-
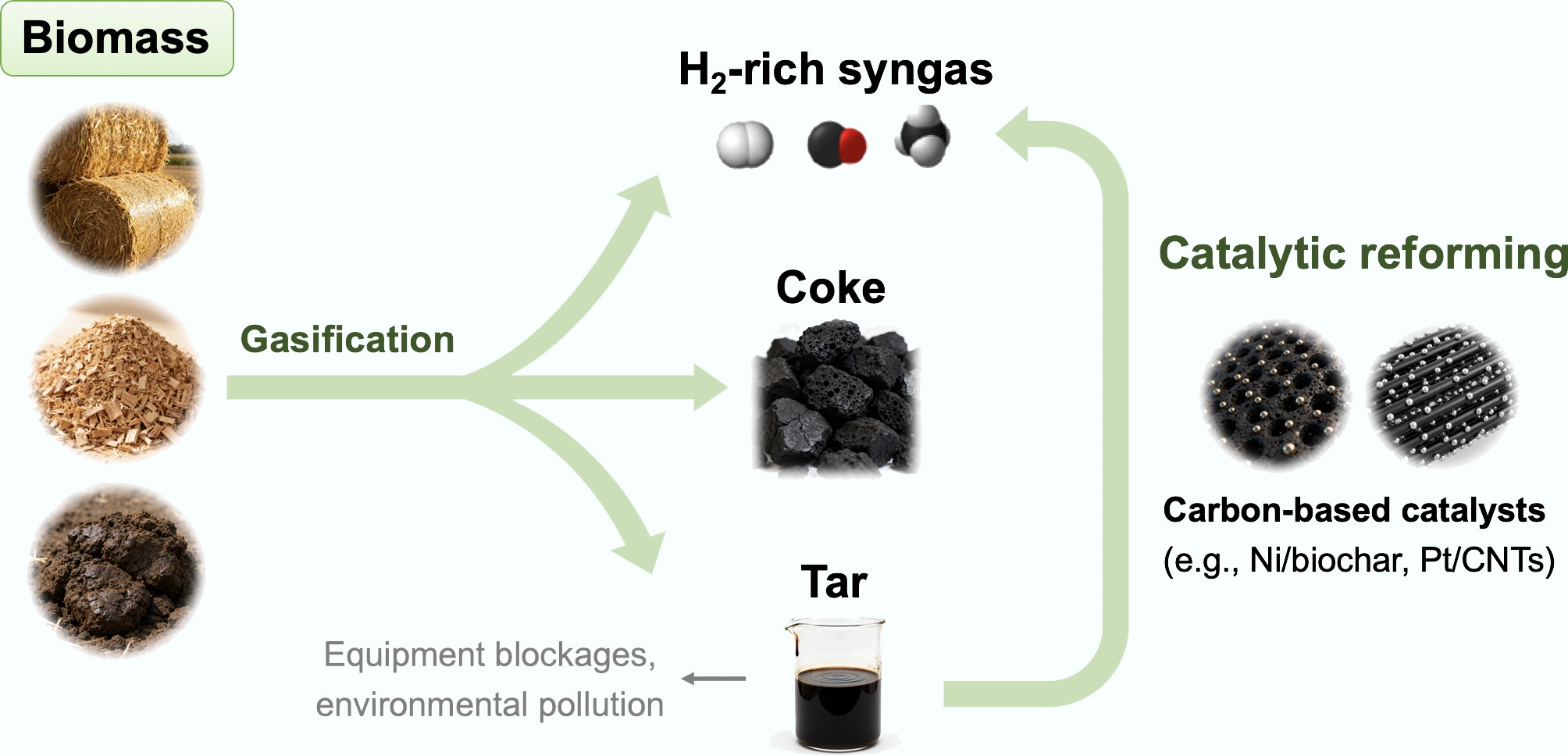
Figure 1.
Schematic illustrating the use of catalytic reforming technology to address the tar issue and enhance the yield of valuable syngas in the biomass gasification process.
-
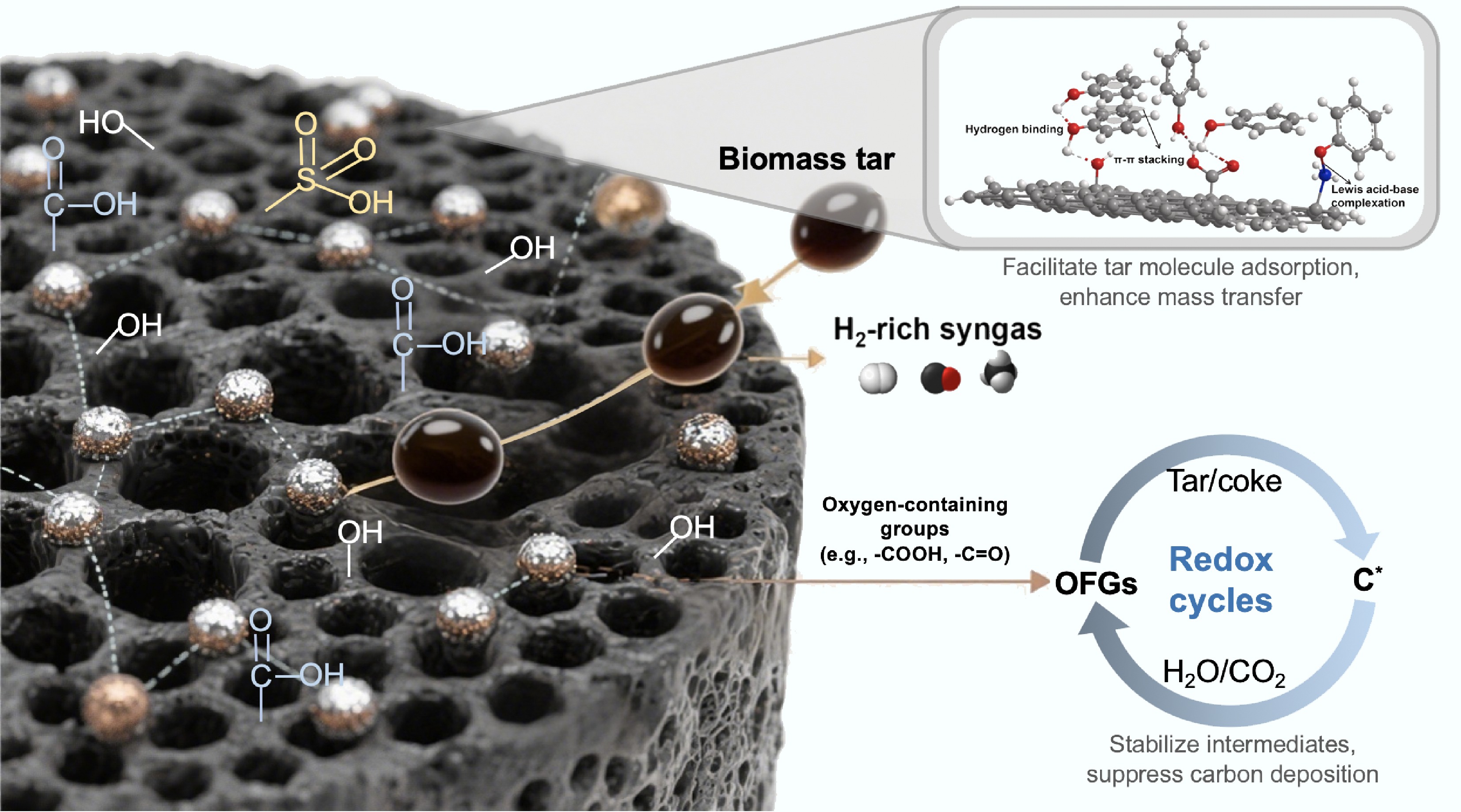
Figure 2.
Schematic diagram of the catalytic mechanism of carbon-based catalysts in biomass tar conversion.
-
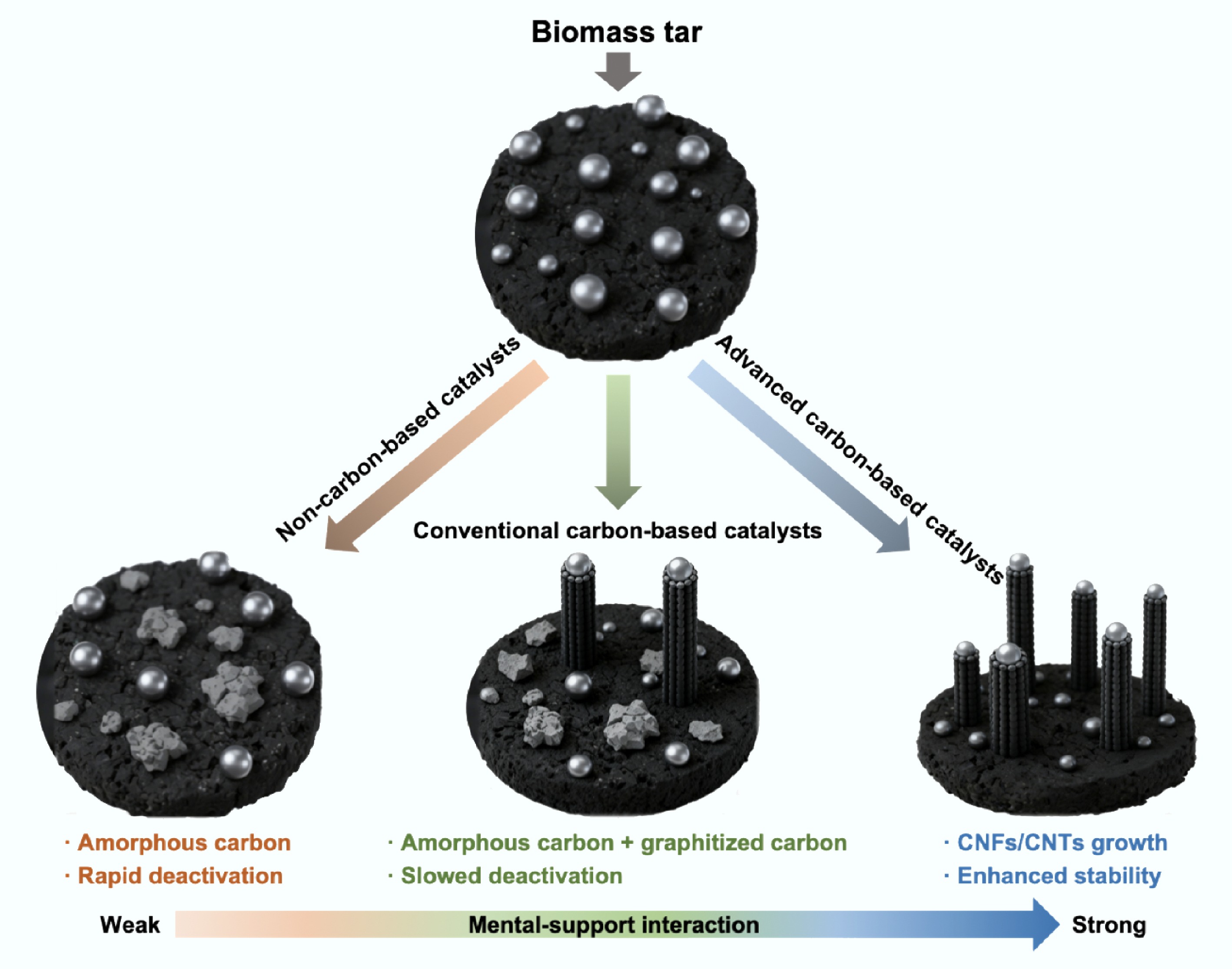
Figure 3.
Comparative schematic of coke conversion pathways over different catalyst systems.
-
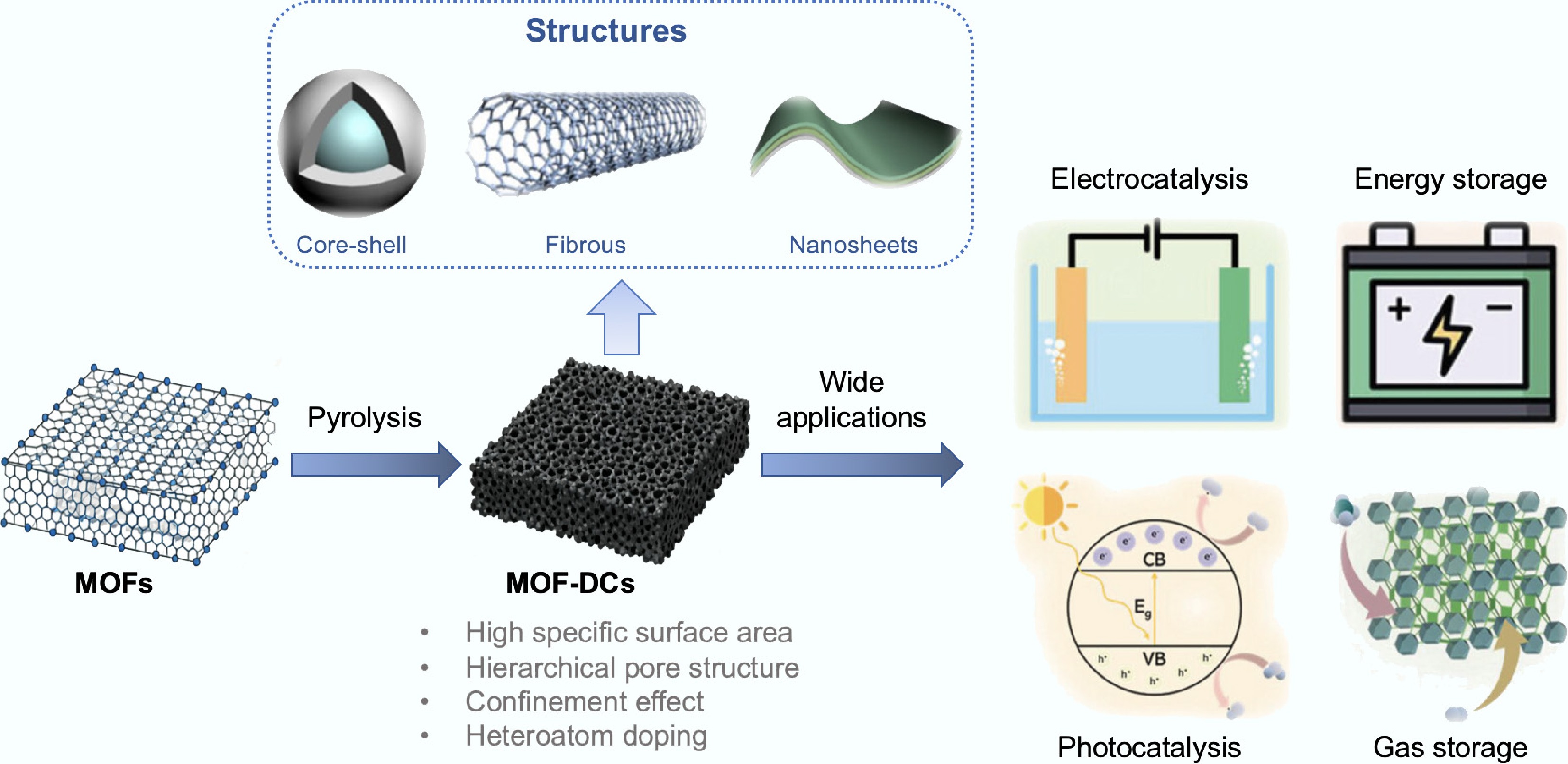
Figure 4.
Structural advantages and applications of MOFs-derived carbon materials.
-
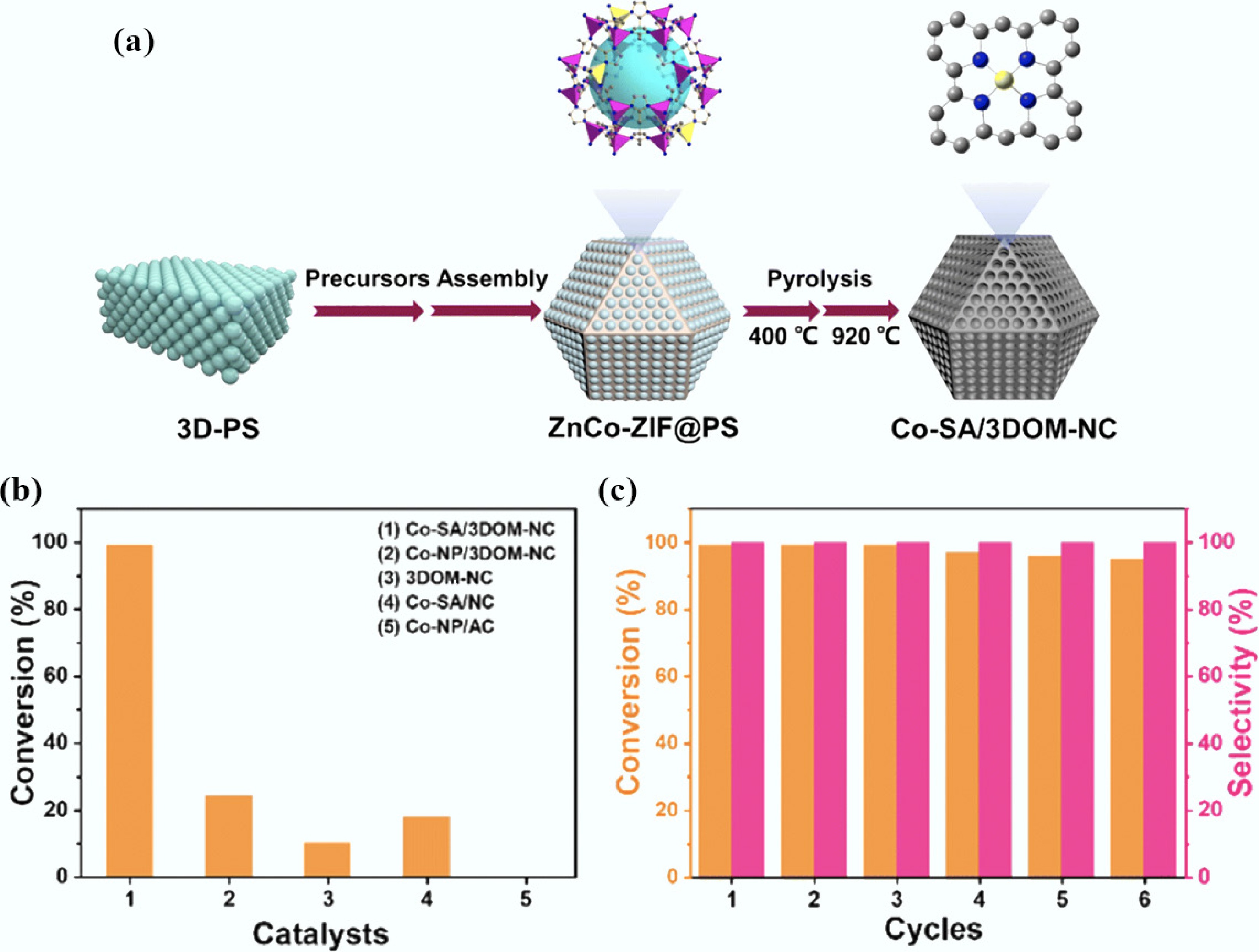
Figure 5.
(a) Schematic illustration of the fabrication of Co-SA/3DOM-NC; (b) The comparison of the FFA conversion between Co-SA/3DOM-NC and other catalysts; (c) Reusability tests of Co-SA/3DOM-NC for the oxidative esterification of FFA[183].
-
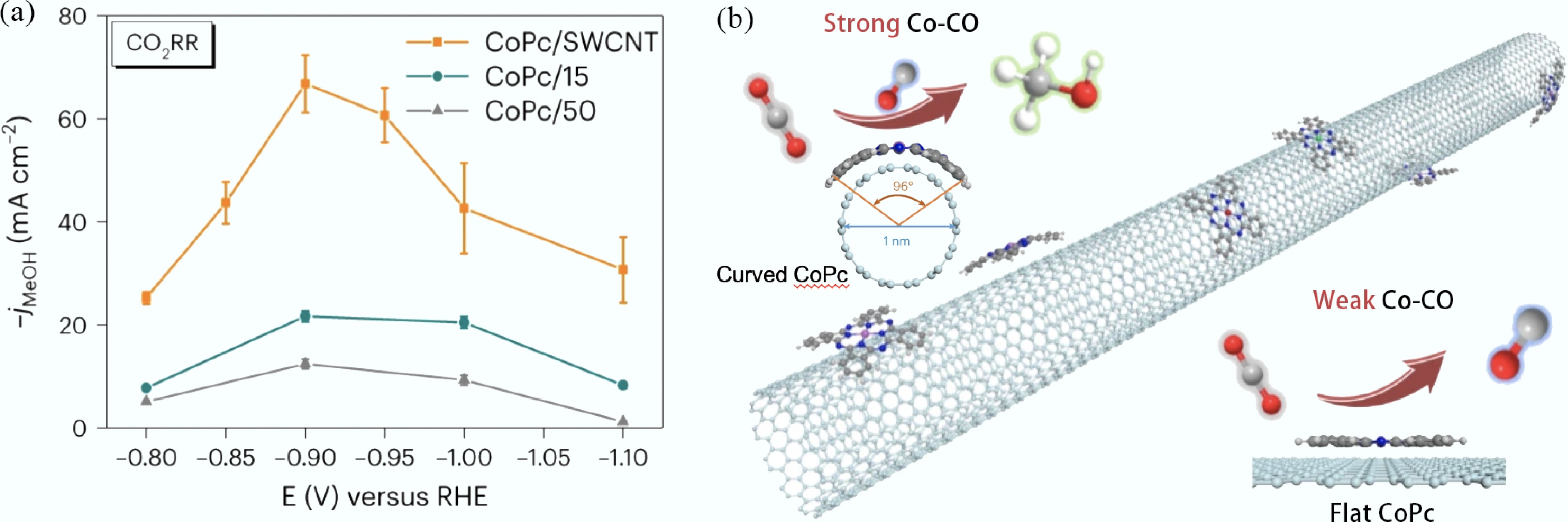
Figure 6.
(a) jMeOH of CoPc/SWCNT, CoPc/15 and CoPc/50 catalysts in a flow cell under CO2 atmosphere; (b) Schematic diagram of the synthetic process of CoPc/SWCNT catalyst and the structure of CoPc on different-diameter CNTs[210].
-
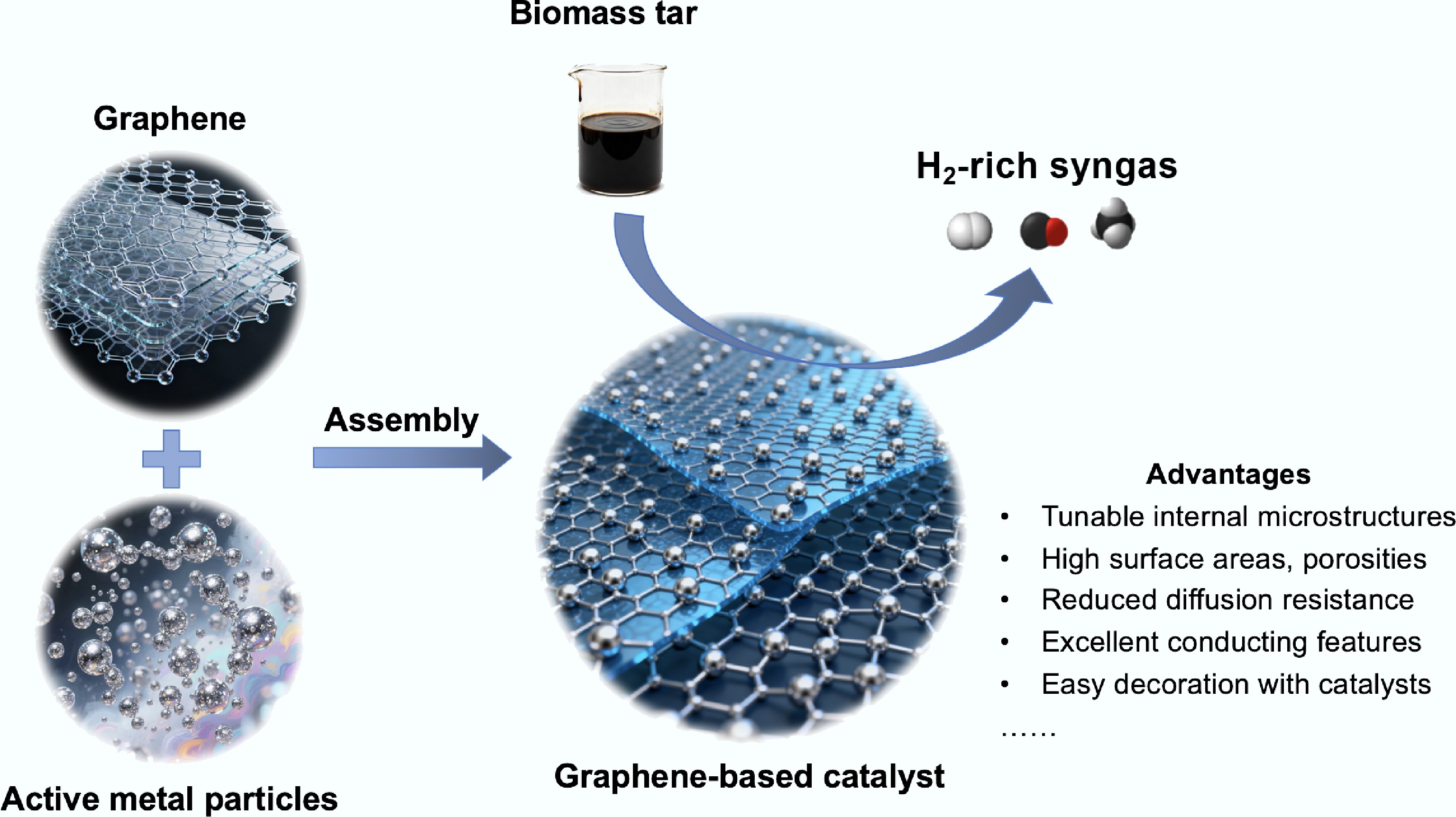
Figure 7.
Synthesis, applications, and prominent structural advantages of graphene-based catalysts.
-
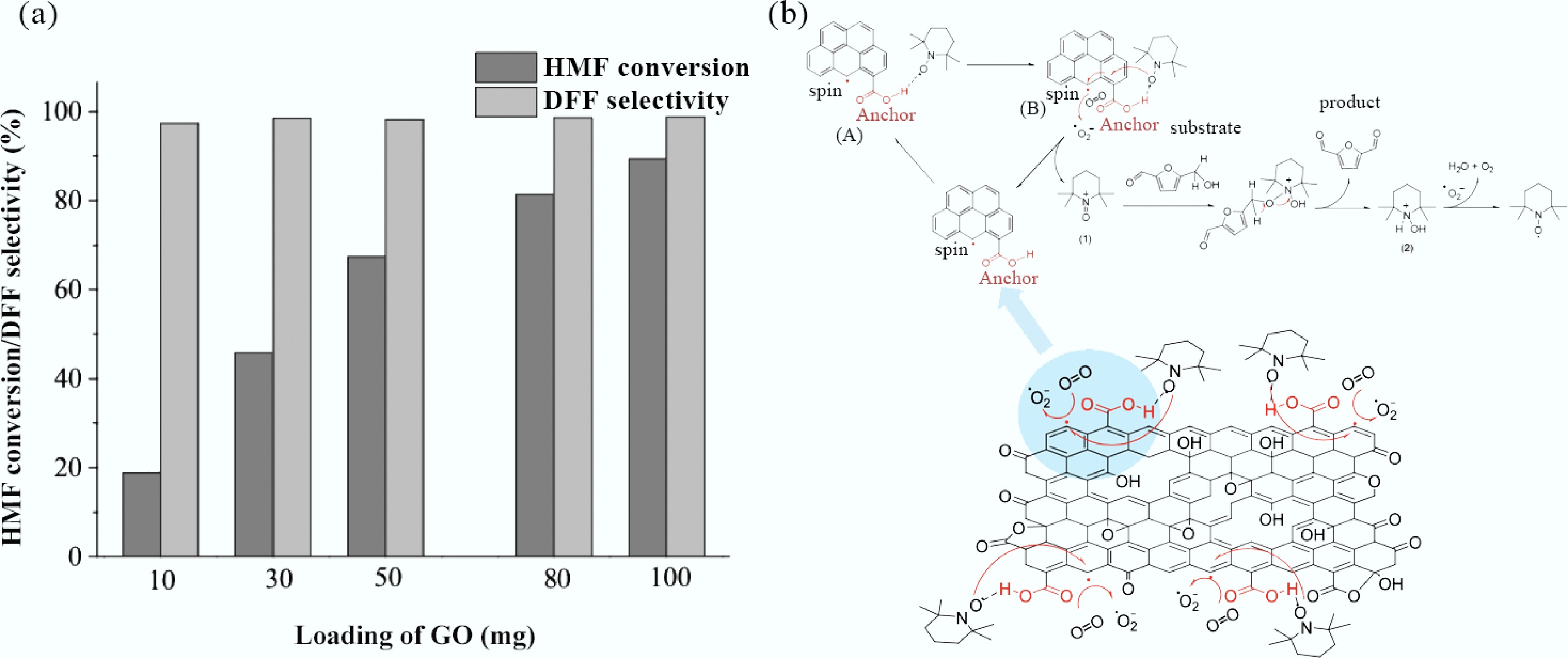
Figure 8.
(a) Effect of the GO loading on aerobic oxidation of HMF into DFF; (b) Proposed reaction pathway for selective oxidation of HMF into DFF with molecular oxygen as the terminal oxidant in GO/TEMPO catalytic system[235].
-

Figure 9.
Modification strategies for conventional carbon-based catalysts based on the structure-activity relationship of advanced carbon materials, and applications of the modified catalysts in various fields.
-
Catalyst categories Typical representatives Key advantages Critical limitations Optimal application scenarios Non-carbon-based catalysts Natural minerals (dolomite, olivine)
Metal oxides (Al2O3, MgO)
Molecular sieves (ZSM-5, HZSM-5)High intrinsic reforming activity
Strong C–C and C–H bond cleavage ability
Well-established industrial experienceSevere coke deposition and metal sintering
High operating temperature
Diffusion limitations for bulky tar molecules
Frequent regeneration requiredHigh-temperature secondary tar reforming
Centralized and industrial-scale gasification systemsCarbon-based catalysts Biochar, coal char, activated carbon
CNTs, graphene, MOFs-derived carbonsDeveloped and tunable pore structures
Excellent metal dispersion
Low-temperature activity
Strong metal-support interaction
Unique coke management via directional carbon transformationLower mechanical strength (especially conventional chars)
Surface heterogeneity
Higher cost and scalability issues for advanced carbonsLow-temperature tar reforming
Distributed biomass gasification
Long-term stable operation and advanced catalytic systems -
Catalyst Feedstock Reaction condition Conversion (%) Ref. Ni/liginte char Toluene 650 °C, S/C = 3.4 83.0 [87] Ni/ZSM-5 Toluene 600 °C, S/C = 2 83.1 [88] Fe-Ni/AC Toluene 600 °C, S/C = 2 93.8 [89] Ni-Pt/Ce0.8Zr0.2O2 Toluene 600 °C, S/C = 3 96.0 [90] Ni/biochar Naphthalene 900 °C, S/C = 2 92.0 [91] Ni/biochar Wheat straw 800 °C, S/B = 4 90.0 [92] Ni/Al2O3 Wood residue 550 °C, S/B = 0.6 85.2 [93] Ni/CeO2/Al2O3 Wood residue 550 °C, S/B = 0.6 92.4 [93] Ni/La2O3/Al2O3 Pine wood 600 °C, S/B = 4 96.4 [94] Ni-Co/Al2O3 Pine wood 600 °C, S/B = 4 99.0 [95] a S/C: Steam-to-carbon molar ratio; b S/B: Steam-to-biomass mass ratio. Table 2.
Comparison of catalytic activities of different Ni-based catalysts for the conversion of tar model compounds and biomass tar
-
Support Advantages Challenges Biochar Low-temperature high efficiency
Excellent pore structure and surface functional groups
Unique coke conversion mechanism
Sustainability and low costPoor mechanical strength
Poor surface chemical stability
Interference from alkali metal ash
Difficulty in coke controlCoal char Excellent pore structure
Good metal dispersibility
Directional coke conversion
Low cost and recyclableMetals are prone to sintering
Insufficient mechanical strength
Prominent coke issue
High variability in raw materials -
Features of advanced carbon materials Deficiencies of conventional carbon Mechanistic insights revealed Transferable modification strategies
for conventional carbonOrdered hierarchical pores Random pore networks with high tortuosity and diffusion limitations Directional mass transfer suppresses pore blockage and random coke accumulation Template-assisted activation; pore-channel ordering via soft/hard templates Spatial confinement of metal nanoparticles Metal sintering and uneven dispersion Confined spaces stabilize metal size and inhibit migration Encapsulation strategies; carbon shell or mineral-carbon composites Single-atom or uniformly coordinated metal sites Broad distribution of metal active environments Uniform coordination lowers C–C activation barriers and suppresses coke precursors Heteroatom (N, S) doping to construct stable metal-Nx sites Highly graphitized frameworks Low conductivity and poor control of coke evolution Enhanced electron transfer directs carbon toward graphitic structures Partial graphitization treatments; conductive carbon additives Tunable surface chemistry and hydrophobicity Steam-induced functional group loss Controlled wettability stabilizes active sites under reforming conditions Surface modification to balance hydrophilicity/hydrophobicity -
Catalyst type Key features Cost Performance (activity/stability) Scalability Controllability (structure/sites) Recommended scenarios Biochar-based Abundant, low-temperature active, porous, surface-functionalized Low Moderate activity; limited stability in steam High (abundant biomass feedstocks) Low (heterogeneous structure) Distributed, low-temperature tar reforming; pilot-scale systems Coal char-based Tunable porosity, good metal dispersion, coke-convertible Low to moderate Good low-temperature activity; prone to sintering and coke High (coal resources available) Moderate (pore structure tunable) Industrial gasification with integrated char recycling MOFs-derived carbons Ordered hierarchical pores, single-atom sites, high SSA Fairly high Excellent activity and coke resistance; stability under study Low (complex synthesis, ligand cost) High (precise pore and site design) High-value syngas production; fundamental mechanism studies CNTs-based High conductivity, spatial confinement, oriented coke growth High High activity and long-term stability; good coke management Low to moderate (CVD scale-up challenging) High (diameter, doping, metal confinement tunable) Advanced catalytic systems requiring high conductivity and coke tolerance Graphene-based Ultra-high SSA, excellent conductivity, tunable surface chemistry High Superior activity and coke restructuring; stacking issues Low (CVD/GO production costly) High (surface chemistry and doping controllable) High-efficiency, low- temperature reforming; hybrid catalysts with conventional carbons
Figures
(9)
Tables
(5)