-
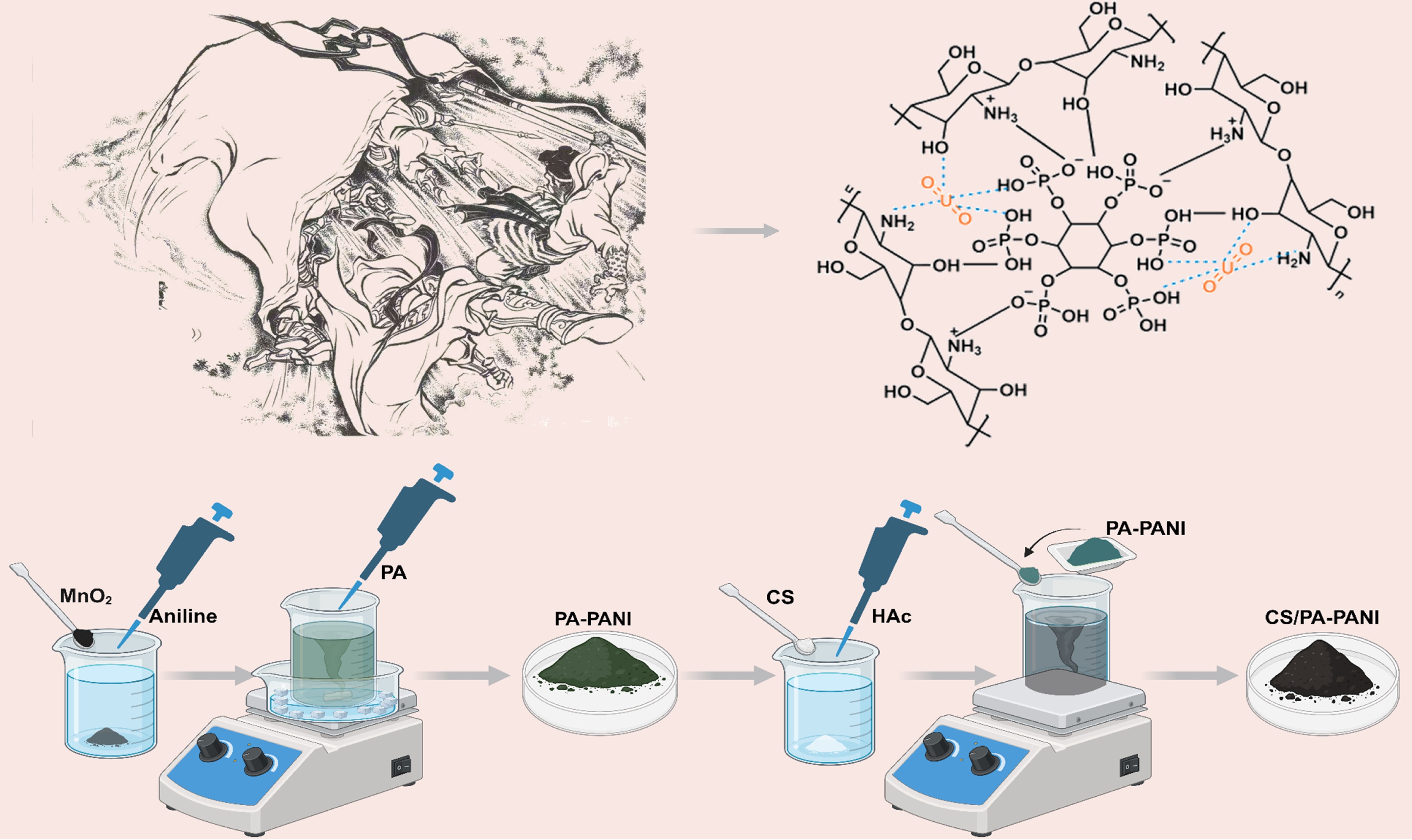
Figure 1.
The preparation process of CS/PA-PANI.
-
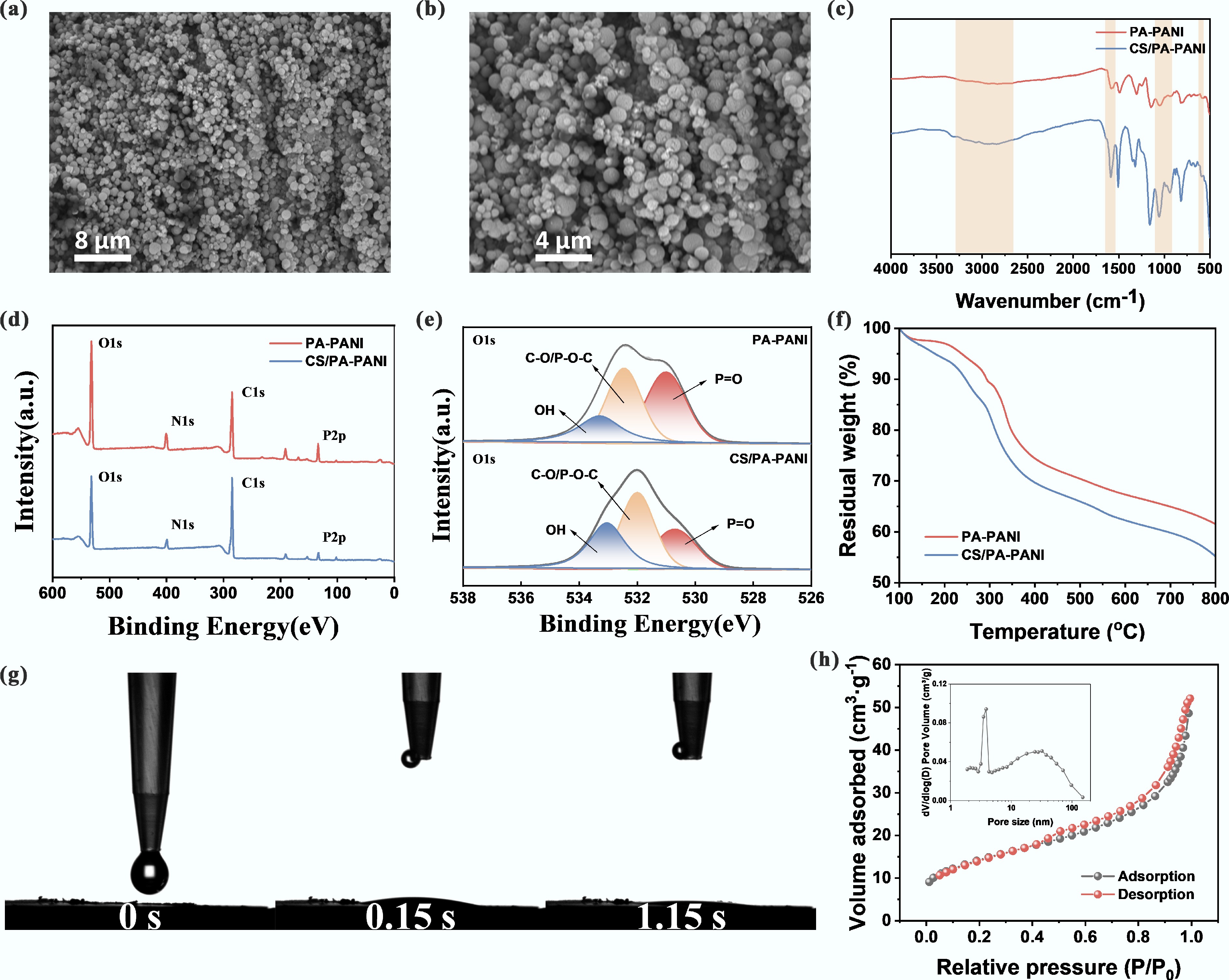
Figure 2.
Synthesis and characterization of CS/PA-PANI. (a), (b) SEM of CS/PA-PANI. (c) FTIR of PA-PANI and CS/PA-PANI; (d) XPS survey spectroscopy of PA-PANI and CS/PA-PANI; (e) XPS O 1s spectra deconvoluted to components at high resolution; (f) Thermogravimetric analysis (TGA) profiles in N2 atmosphere; (g) Time dependent evolution of water contact angle and hysteresis of CS/PA-PANI; (h) N2 adsorption-desorption curve and corresponding pore size distribution (inset) of CS/PA-PANI.
-
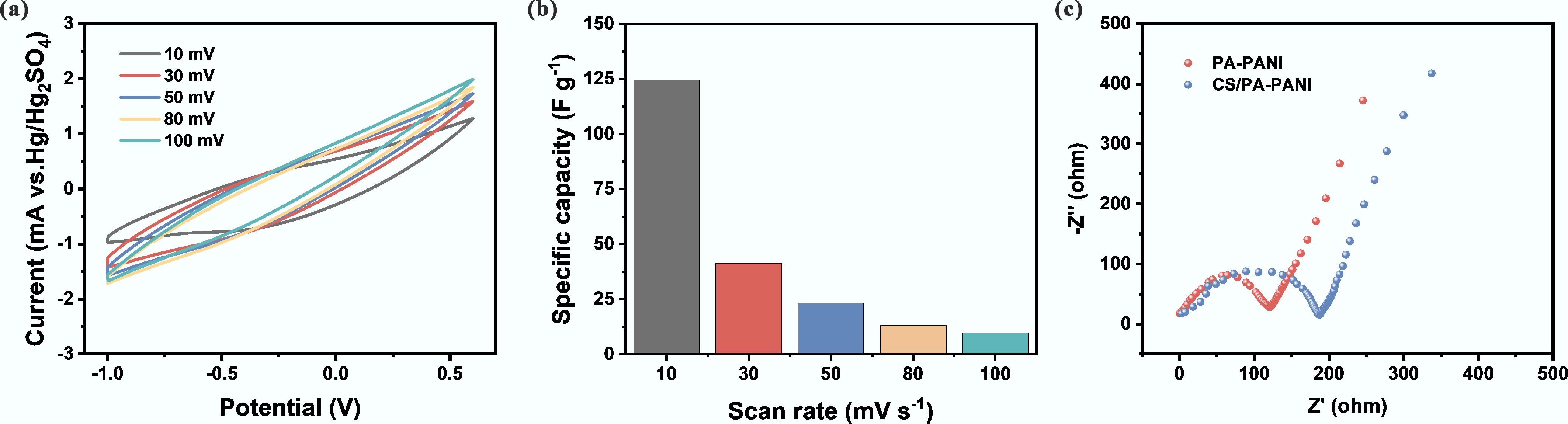
Figure 3.
Electrochemical performance evaluation of CS/PA-PANI. (a) CV curves of CS/PA-PANI at various scanning rates; (b) Specific capacitance of CS/PA-PANI at various scanning rates; (c) EIS spectra of the CS/PA-PANI and PA-PANI.
-
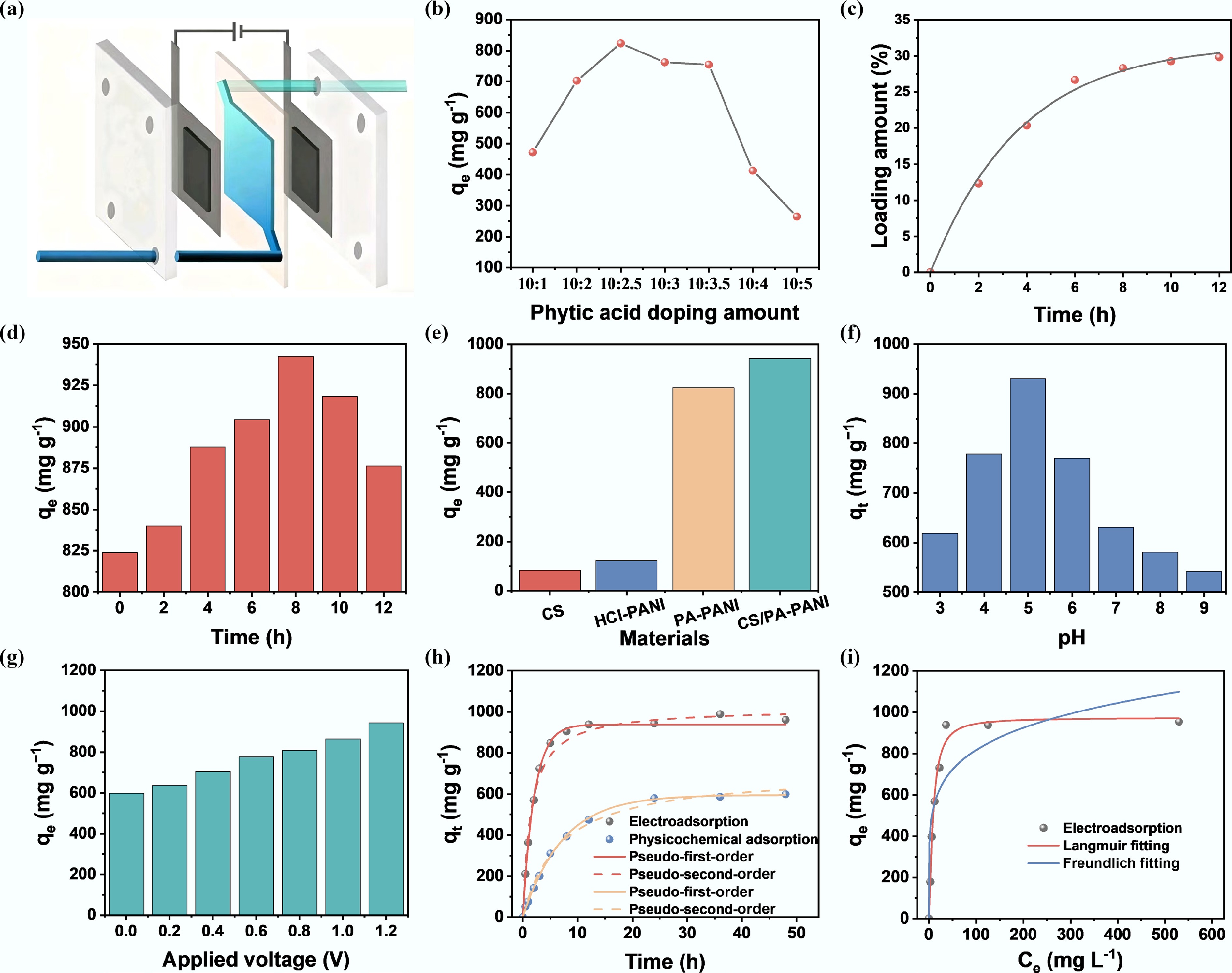
Figure 4.
Uranium electrosorption behavior of CS/PA-PANI electrodes. (a) Schematic illustration of CS/PA-PANI employed as an immobile electrode in the CDI system. (b) Effect of phytic acid doping amount on uranium electrosorption. (c) Change of chitosan loading concentration with time. (d) Effect of chitosan doping time on adsorption performance. (e) U(VI) electrosorbed by various samples. Influence of pH of (f) solution and voltage (g) applied on U(VI) electrosorption. (h) Kinetics of adsorption CS/PA-PANI with the respective fitting equations in the model of pseudo-first-order and pseudo-second-order. (i) Adsorption isotherms at initial U(VI) concentrations of 50, 100, 200, 300, 400, and 500 mg L−1. Experimental conditions: solution volume: 50 mL; pH adjusted using dilute Na2CO3 or HNO3 solution.
-
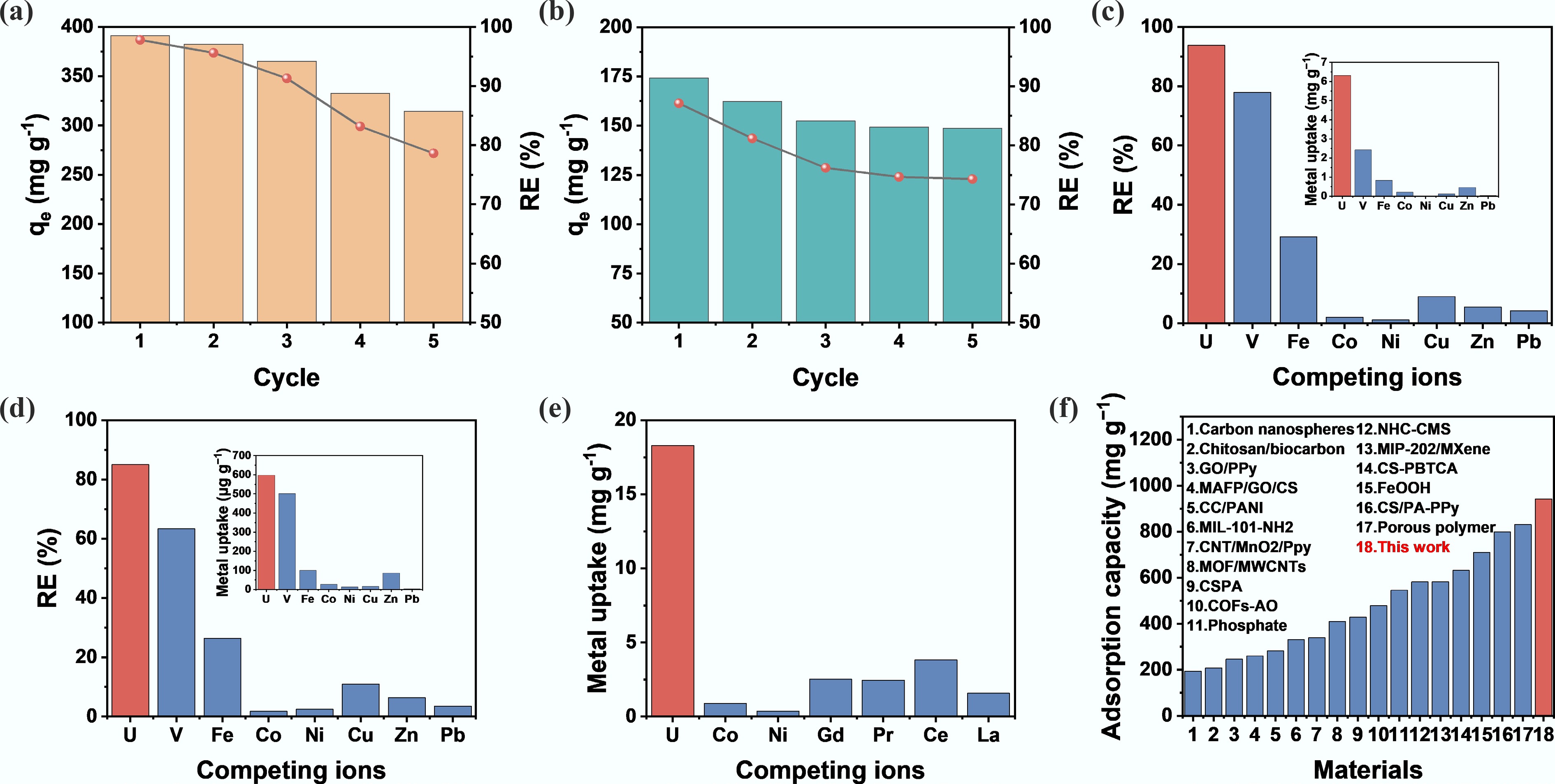
Figure 5.
Cycling stability and selectivity of CS/PA-PANI electrodes in uranium recovery. (a) Cycling reversibility of CS/PA-PANI electrodes eluted with 0.1 mol L−1 HCl under reverse bias for 4 h. The starting concentration, C0 = 200 mg L−1. (b) Five adsorption cycles and one elution of used CS/PA-PANI electrodes with 0.1 mol L−1 HNO3 as the eluent. C0 = 100 mg L−1. (c) Uranium and competing ion uptake by CS/PA-PANI into U-spiked simulated seawater. (d) Comparison of adsorption of uranium in the natural seawater with other competing ions. (e) Co-removal of a mixture of ions on the CS/PA-PANI electrode through simulated nuclear wastewater after 24 h. (f) Comparison of the uranium adsorption capacity of CS/PA-PANI with reported adsorbents[26, 32,33, 50,56−67].
-
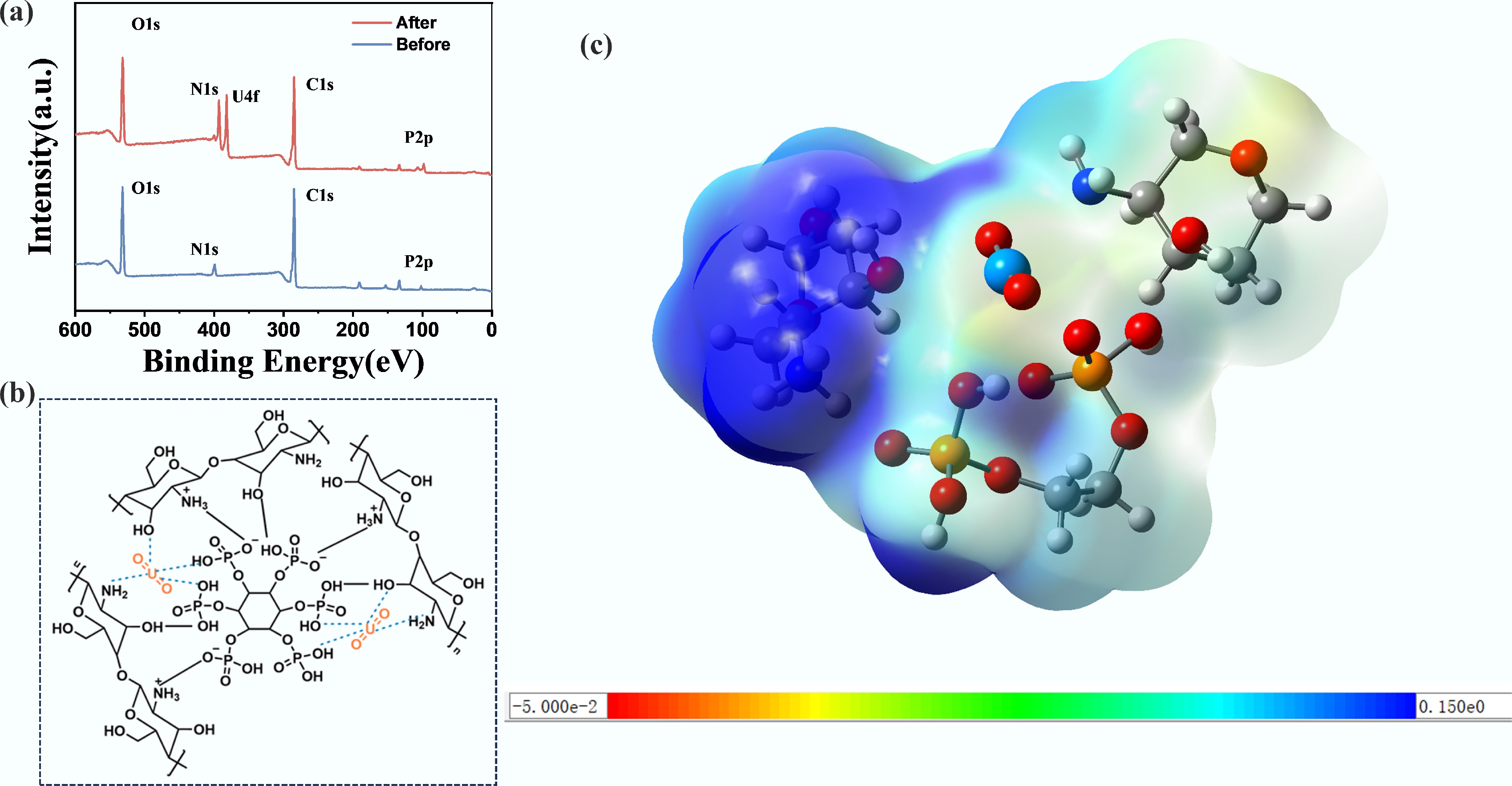
Figure 6.
Mechanistic study of uranium adsorption by CS/PA-PANI composites. (a) XPS spectra of the CS/PA-PANI before and after U adsorption. (b) Structural schematics of functional groups in CS/PA-PANI. (c) Coordination structure diagram of functional groups in CS/PA-PANI with uranyl ion.
Figures
(6)
Tables
(0)