-
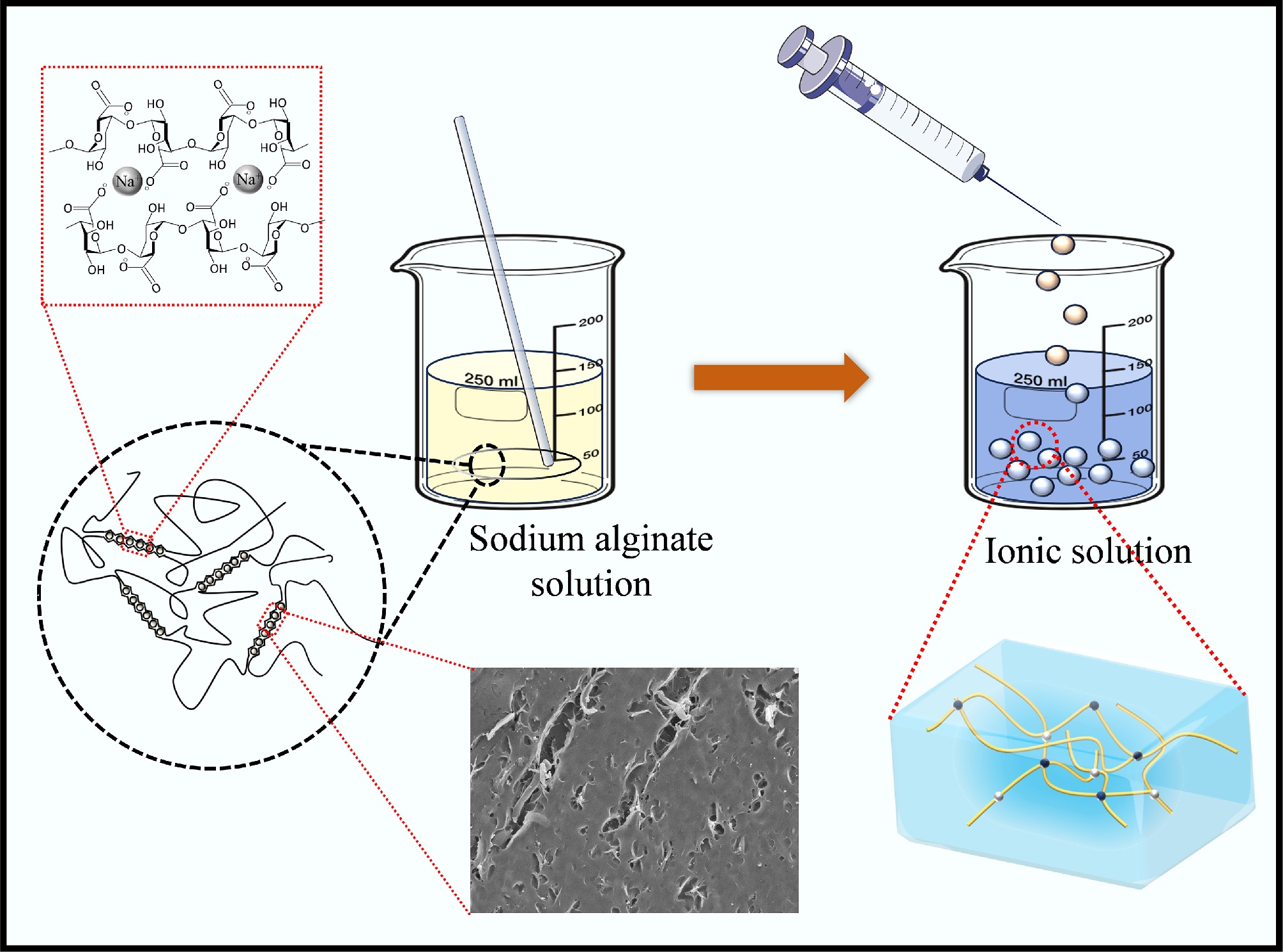
Figure 1.
The production process of sodium alginate composite gel microspheres.
-
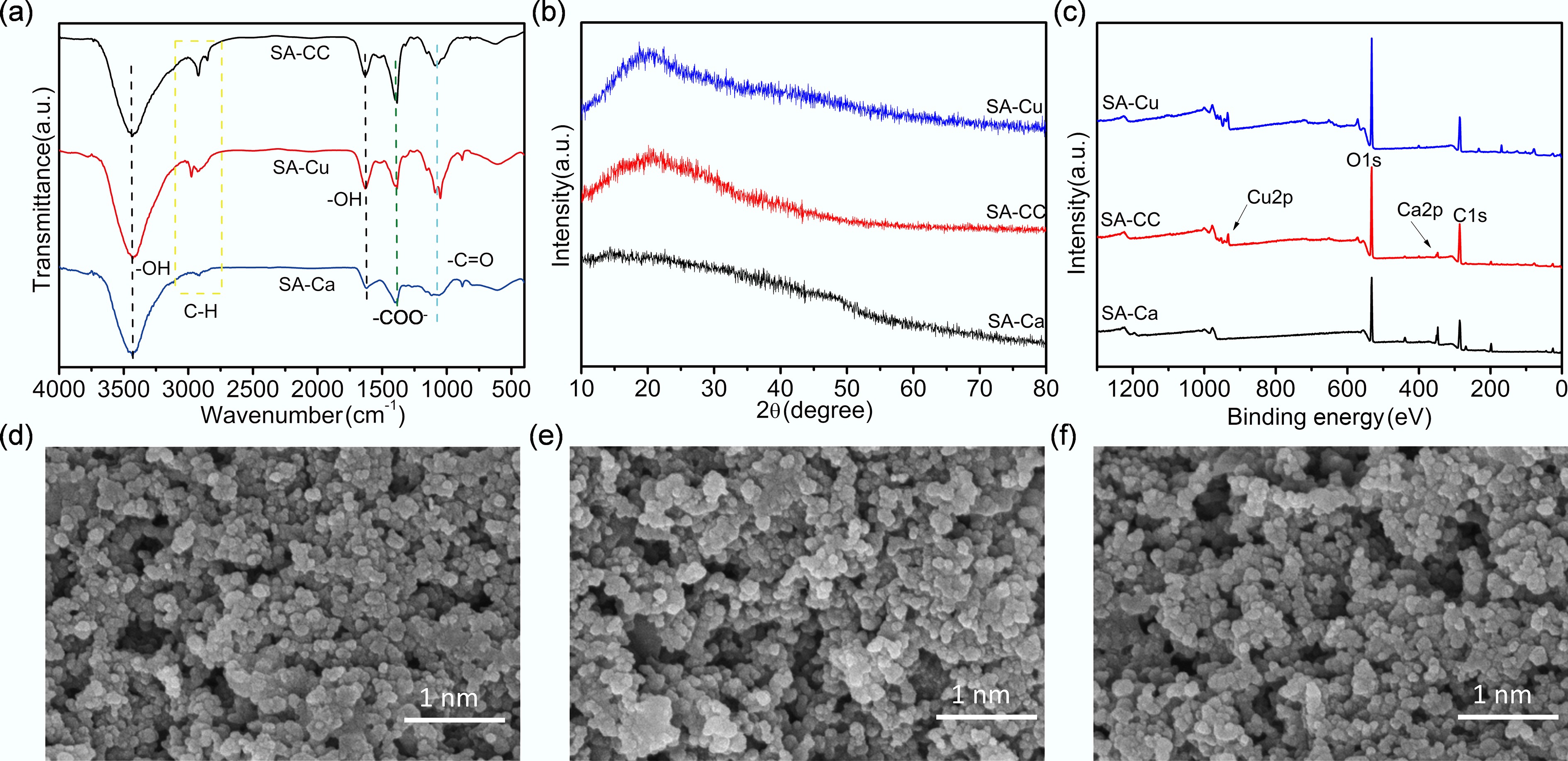
Figure 2.
(a) FTIR spectra, (b) XRD patterns, and (c) XPS spectra of SA-Ca, SA-CC, and SA-Cu, SEM images of (d) SA-Ca, (e) SA-Cu, and (f) SA-CC.
-
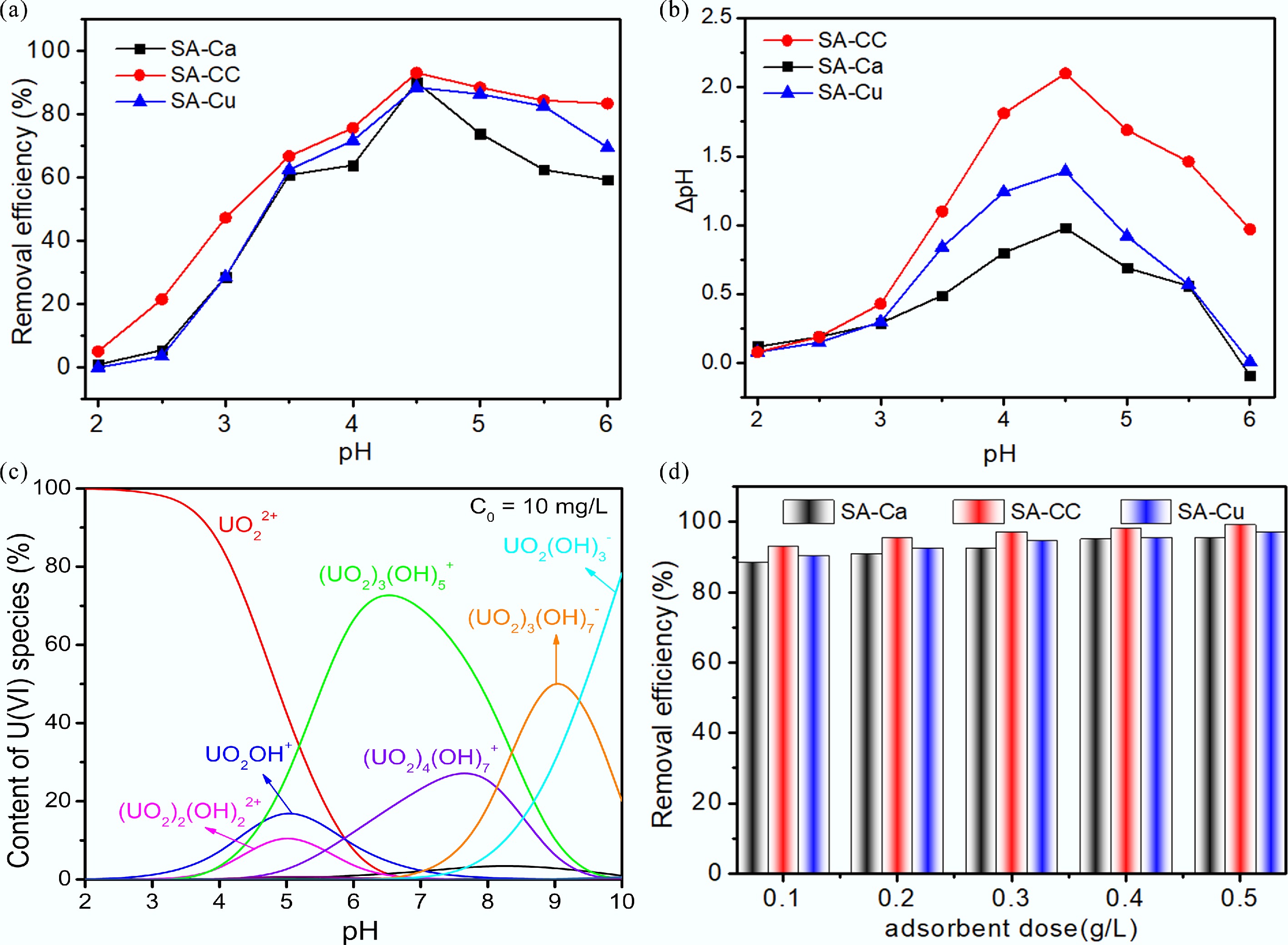
Figure 3.
(a) Effect of pH on U(VI) removal; (b) Zero charge of samples at various pH values (ΔpH = pHfinal – pHinitial); (c) Speciation of U(VI) at pH (2–10); (d) Effect of adsorbent dose on U(VI) removal.
-
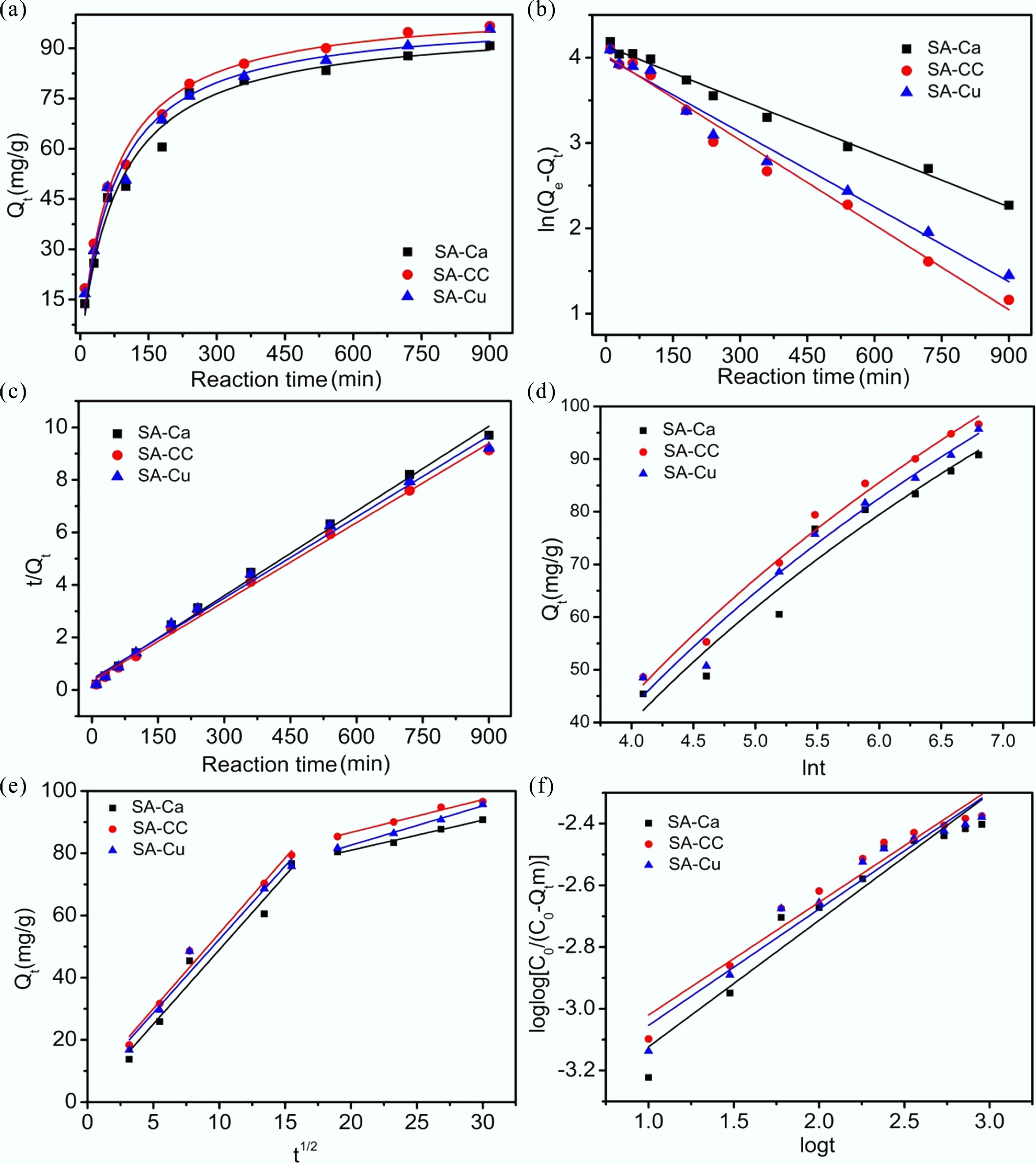
Figure 4.
(a) Effect of contact time on U(VI) removal; (b) Pseudo-first-order kinetic models; (c) Pseudo-second-order kinetic model; (d) Elovich kinetic model; (e) Intraparticle diffusion kinetic model; (f) Bangham kinetic model of U(VI) removal on SA-based materials.
-
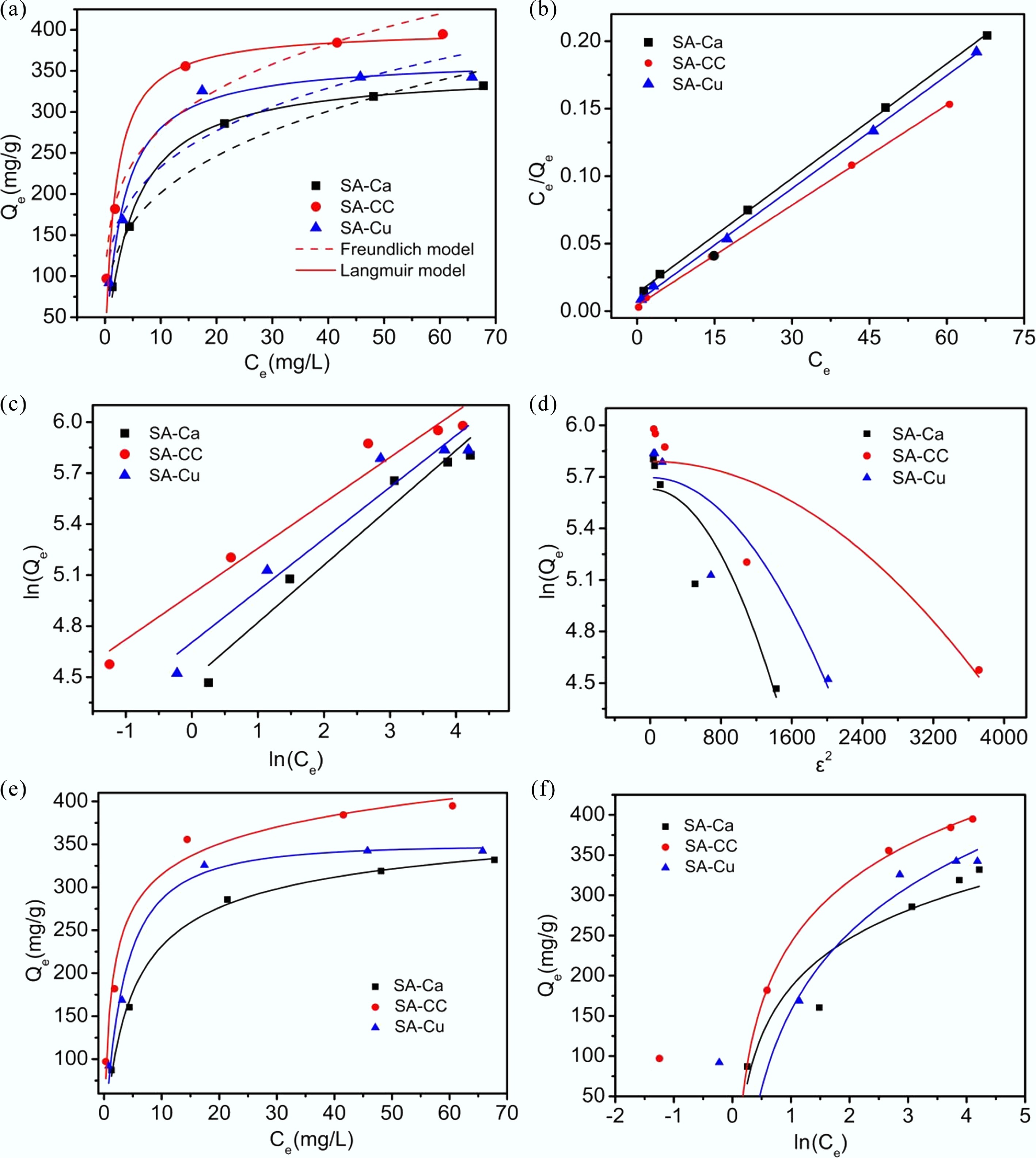
Figure 5.
Adsorption isotherms of U(VI) on SA-based materials at 298.15 K. (a) Nonlinear Freundlich and Langmuir isotherms; (b) Linear Langmuir isotherm; (c) Linear Freundlich isotherm; (d) D-R isotherm; (e) R-P isotherm; (f) Temkin isotherm.
-
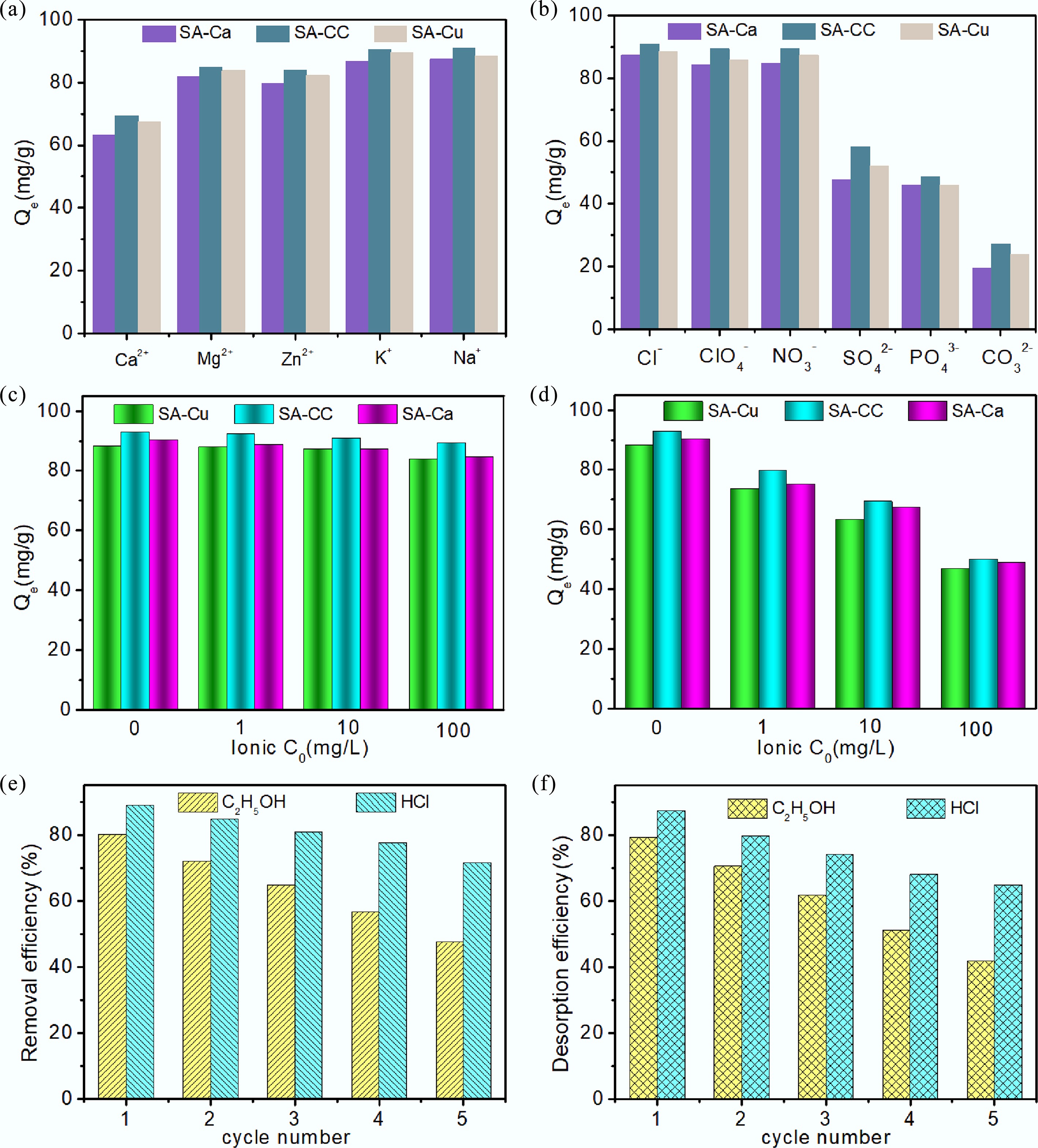
Figure 6.
Influence of (a) cations, and (b) anions on the removal of uranium (VI), influence of ionic strength of (c) NaCl, and (d) CaCl2 on the removal of uranium (VI), (e) removal efficiency, and (f) desorption efficiency of U(VI) onto SA-CC in five cycles with CH3OH and HCl.
-
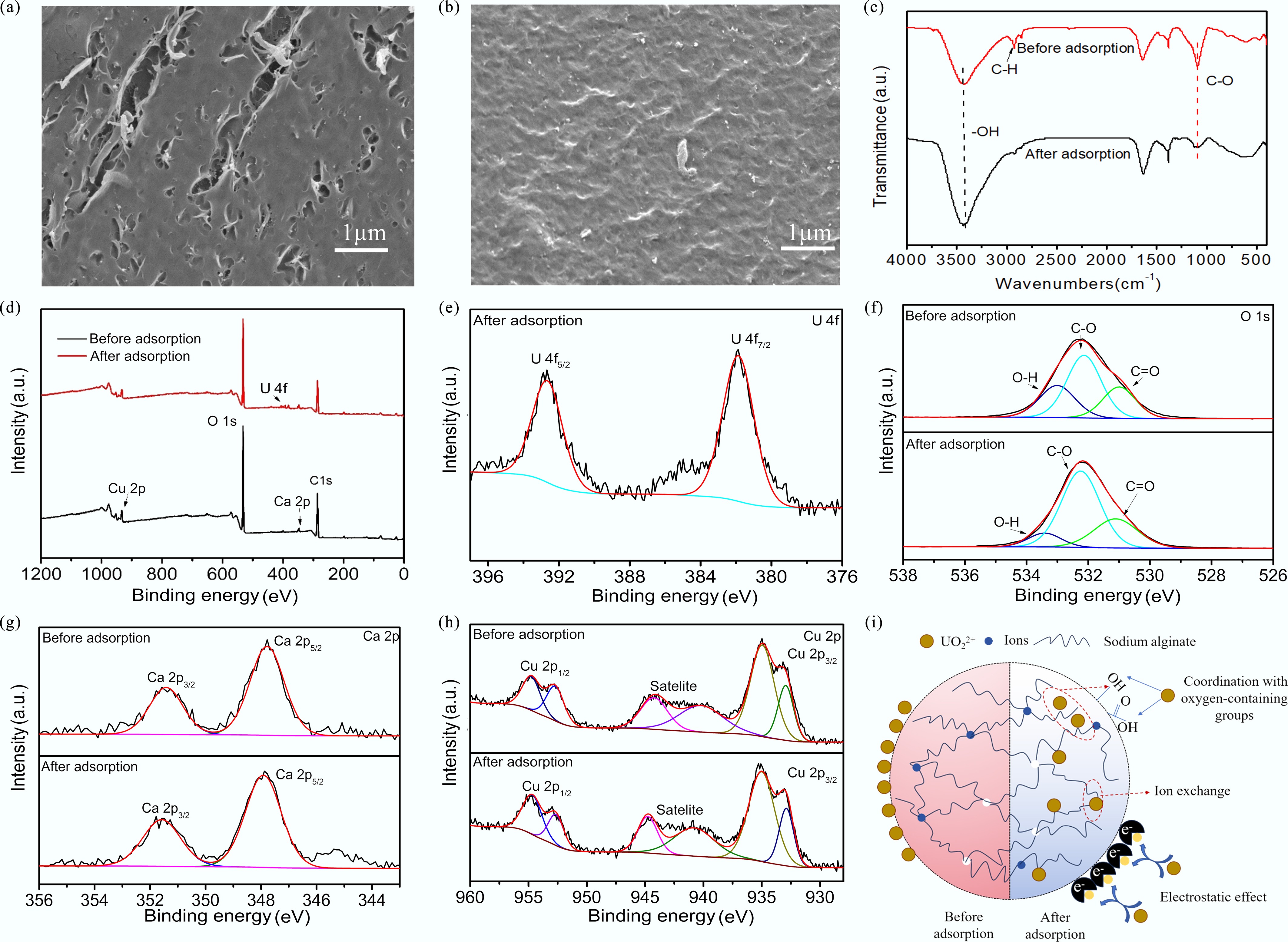
Figure 7.
(a) SEM images of SA-CC before adsorption; (b) SEM images of SA-CC after adsorption; (c) FTIR spectra of SA-CC before and after adsorption; (d) XPS spectra of SA-CC before and after adsorption; High-resolution XPS spectra of (e) U 4f, (f) O 1s, (g) Cu 2p, and (h) Ca 2p of SA-CC before and after U(VI) adsorption; (i) Possible mechanism for the interaction between SA-CC and U(VI).
-
Table 1.
Comparison of the maximum adsorption capacity of uranium (VI) on various adsorbents
Figures
(7)
Tables
(1)