-
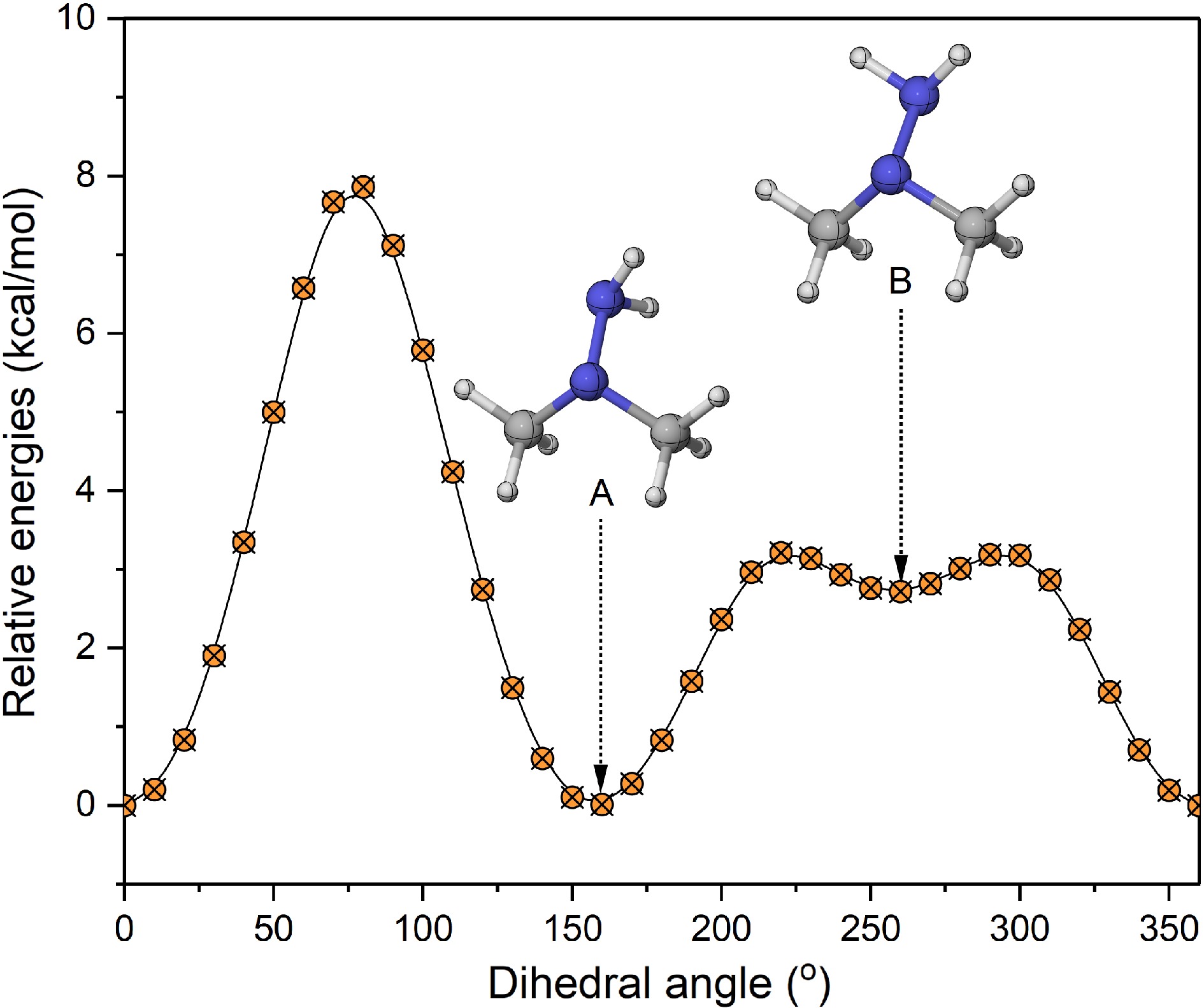
Figure 1.
Relative energy of the amine group rotation in the UDMH molecule (unit: kcal/mol).
-
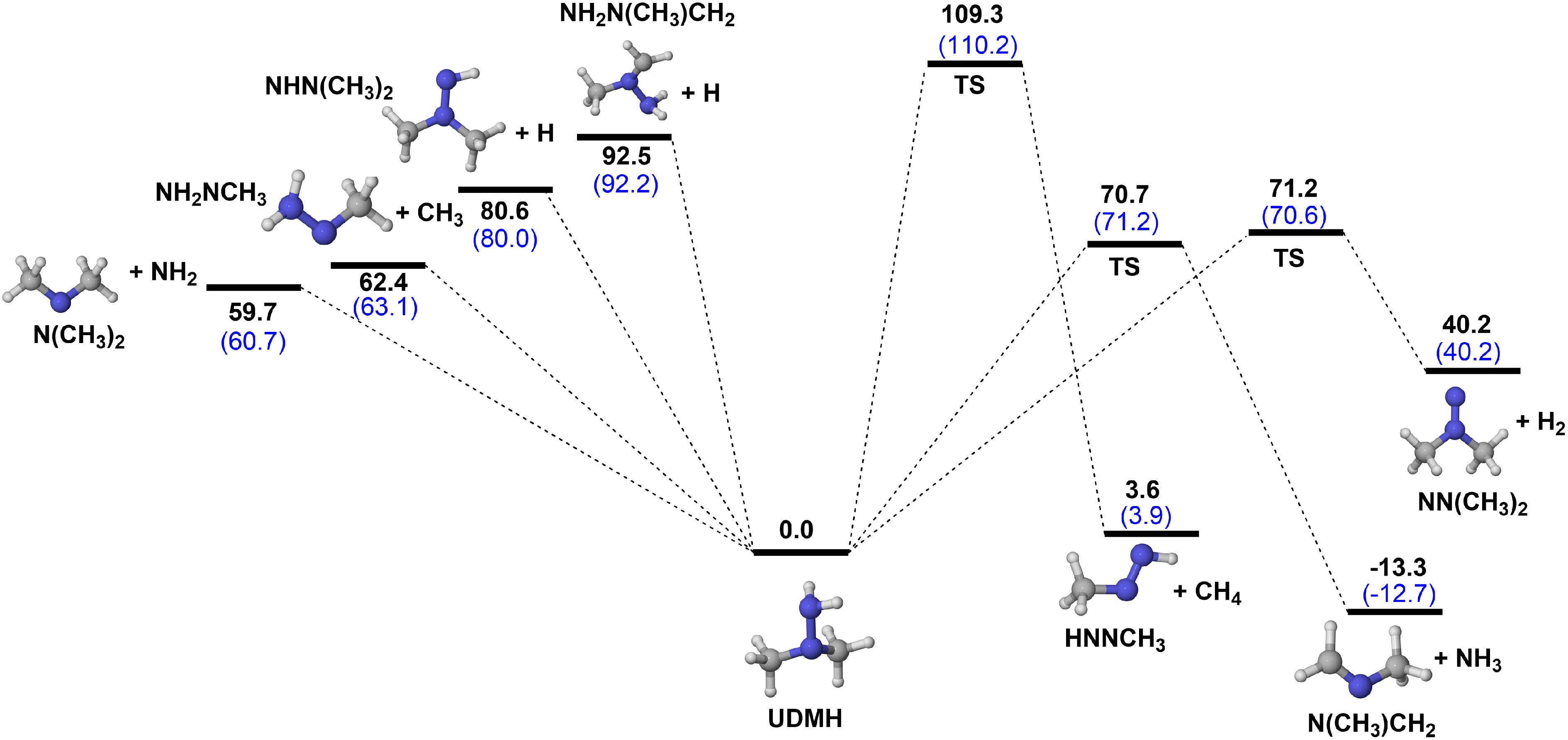
Figure 2.
PESs for the unimolecular decomposition reactions of UDMH at CCSD(T)/CBS(D+T)//M06-2X/def2-TZVP (black), and CCSD(T)/CBS(T+Q)//M06-2X/def2-TZVP (blue) level (unit: kcal/mol).
-
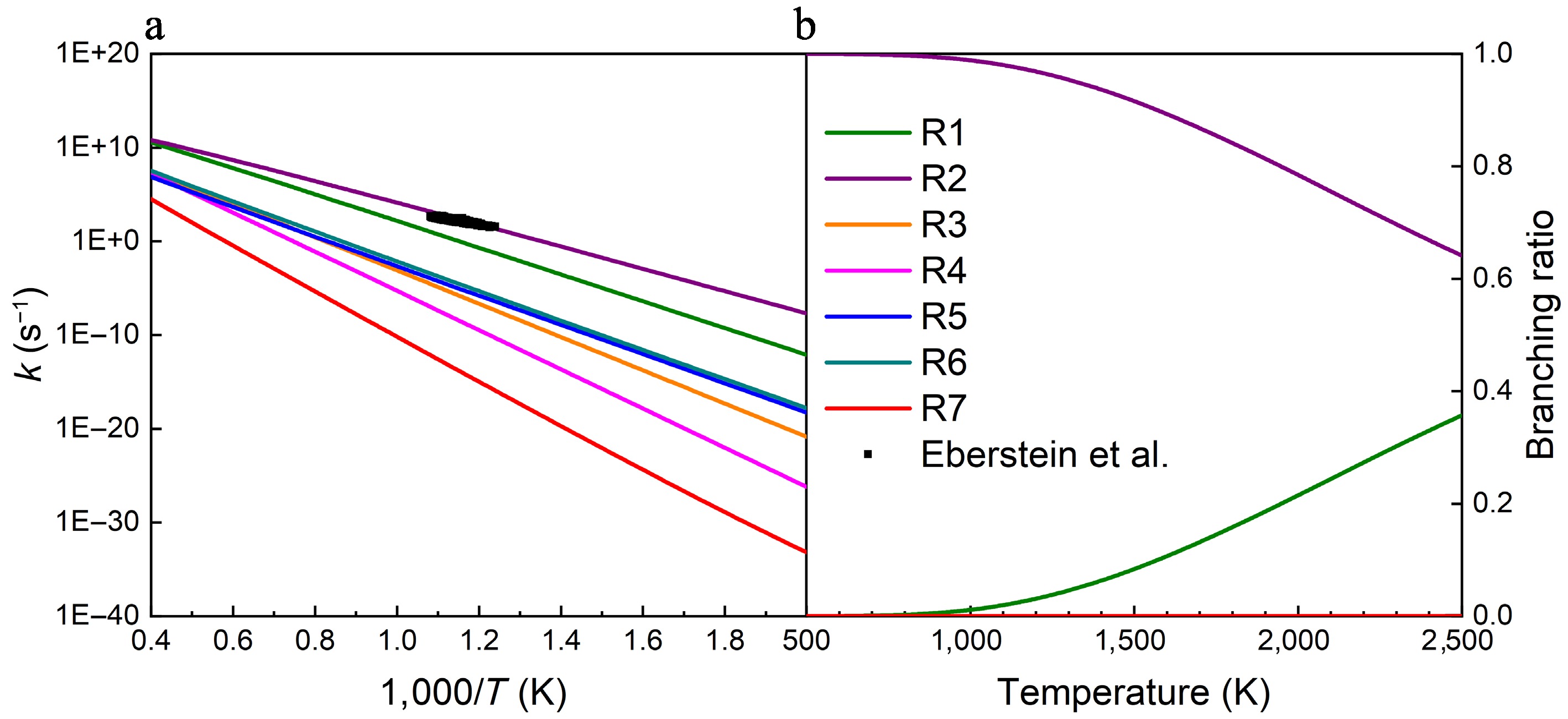
Figure 3.
(a) Rate constants, and (b) branching ratios for the unimolecular decomposition reactions of UDMH at HPL. The black squares represent the total rate constants for the UDMH decomposition reaction measured by Eberstein and Glassman at atmospheric pressure[2].
-
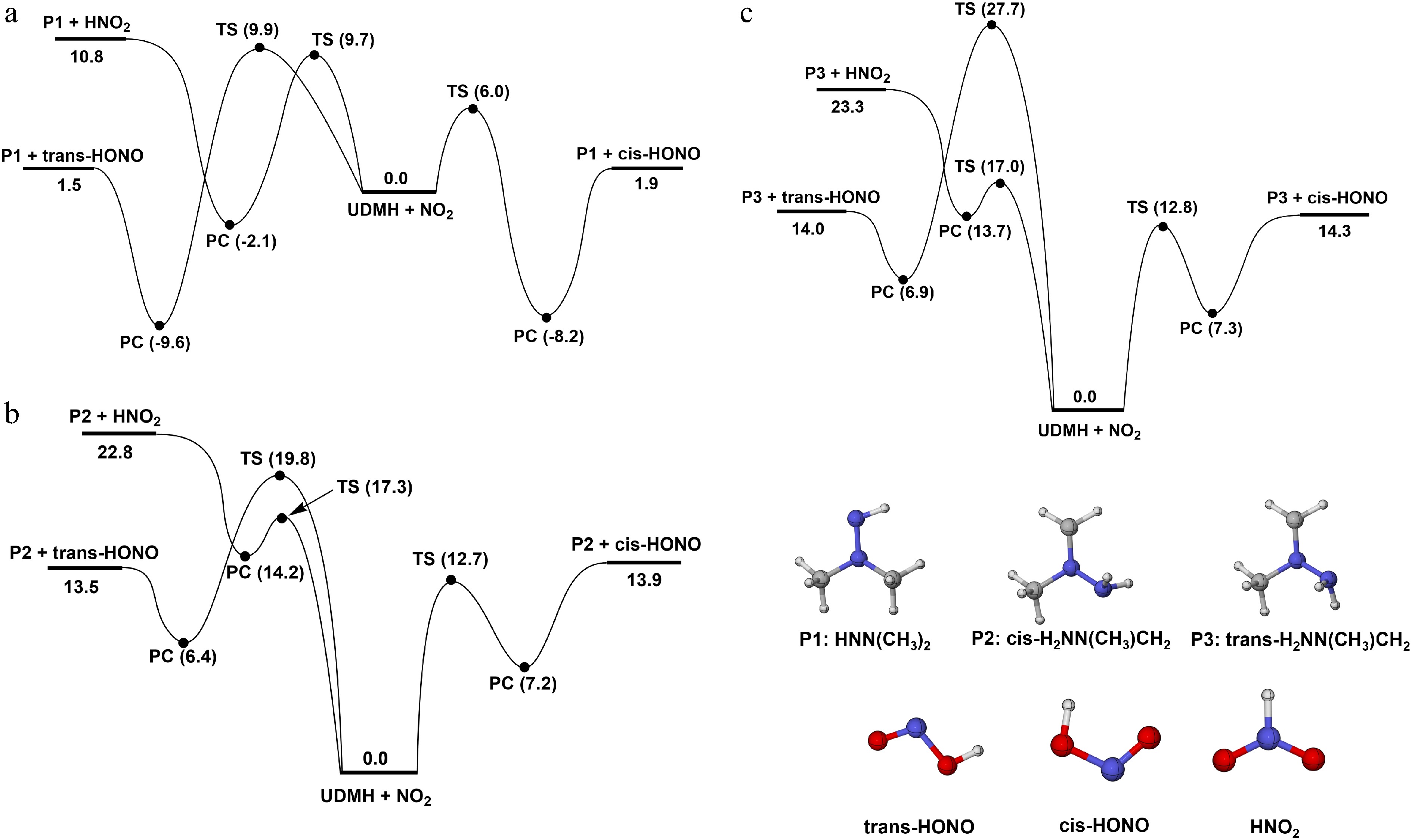
Figure 4.
PESs for the H-abstraction of UDMH on (a) HNH2, (b) Hcis-CH3, and (c) Htrans-CH3 sites, with NO2 attacking at the CCSD(T)/CBS//M062X/def2-TZVP level (unit: kcal/mol).
-
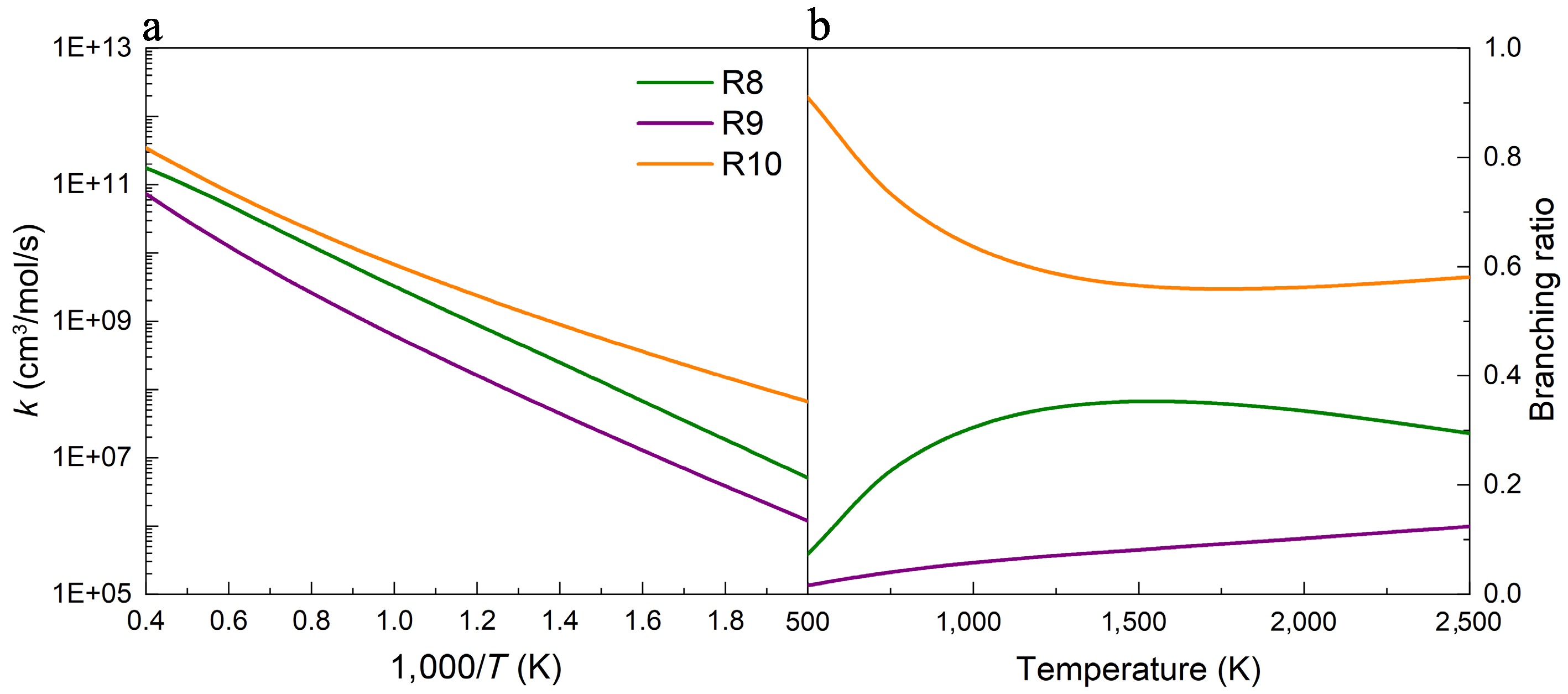
Figure 5.
(a) Rate constants, and (b) branching ratios for the H-abstraction reactions of UDMH on the HNH2 sites, with NO2 attacking at 100 atm.
-
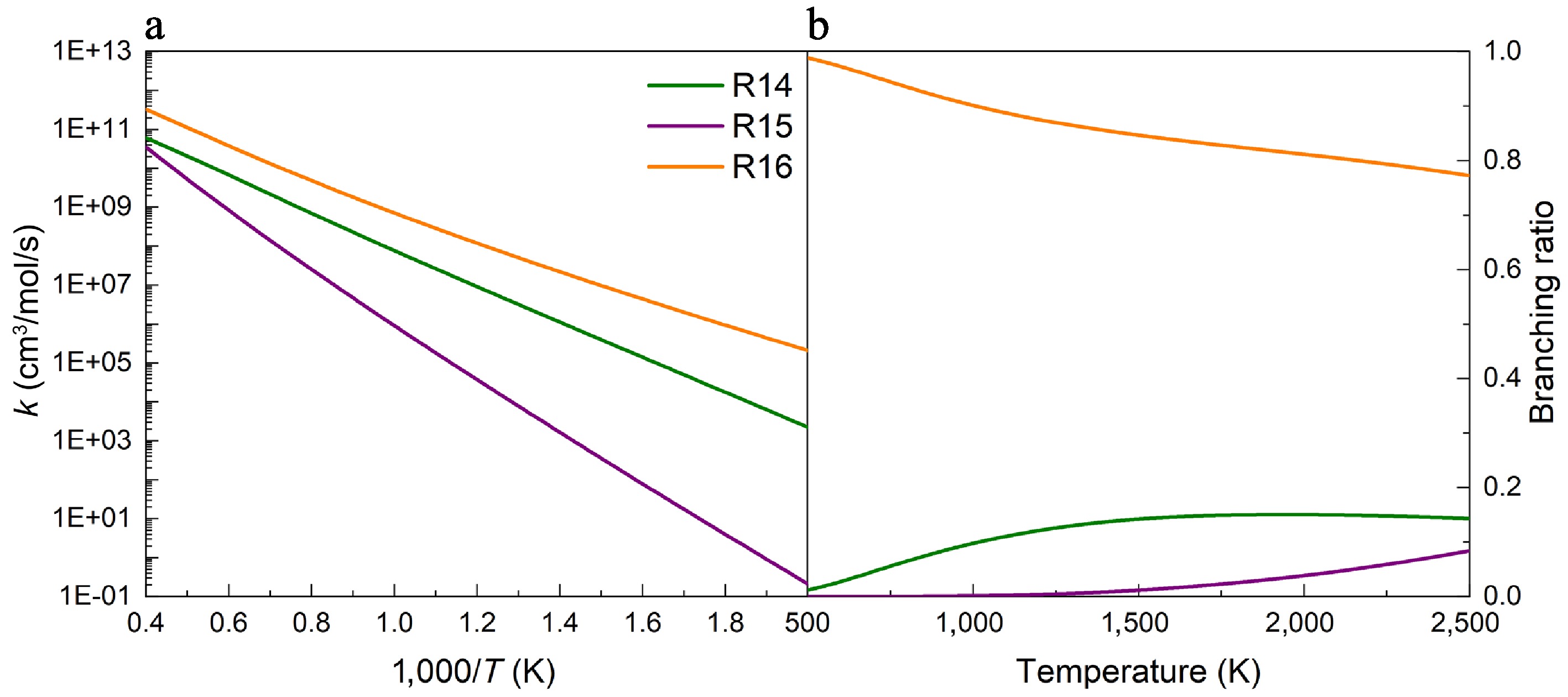
Figure 7.
(a) Rate constants, and (b) branching ratios for the H-abstraction reactions of UDMH on the Htrans-CH3 sites, with NO2 attacking at 100 atm.
-
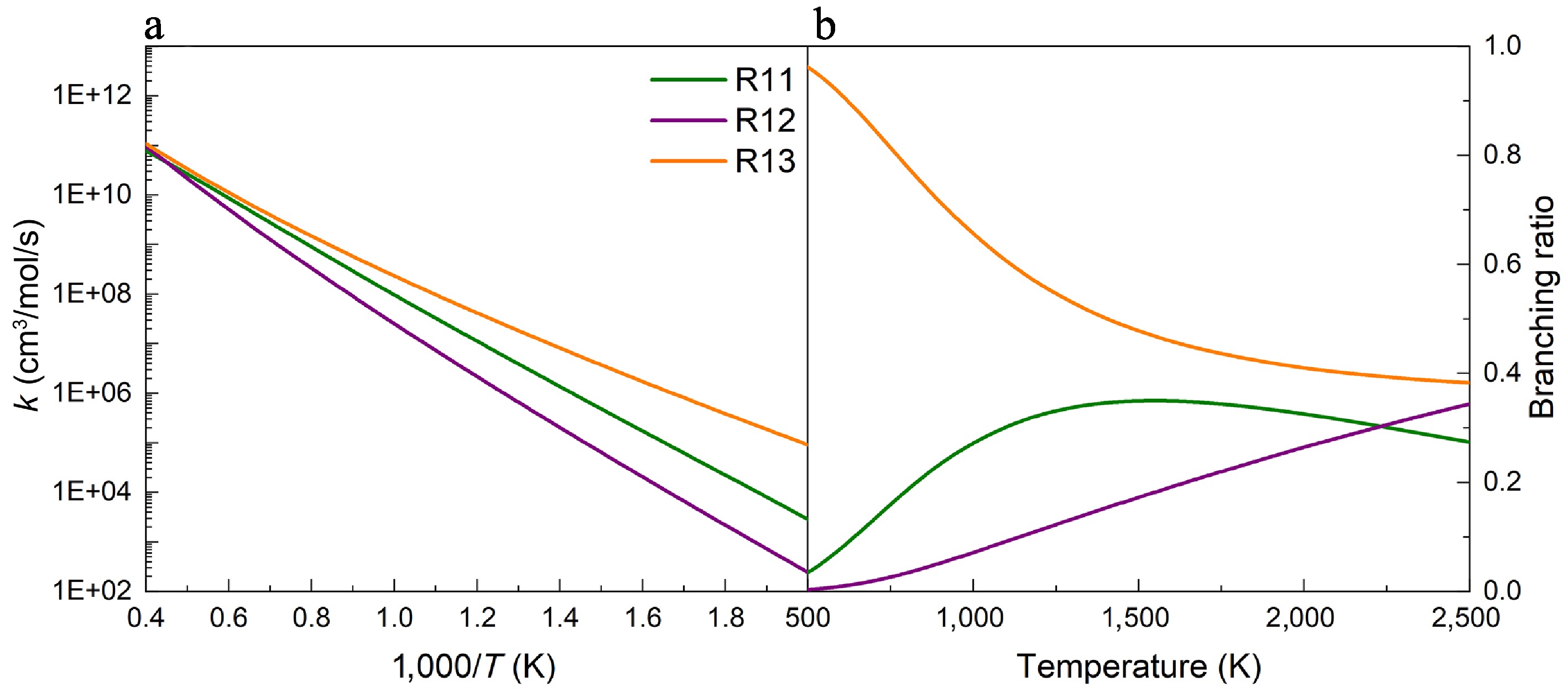
Figure 6.
(a) Rate constants, and (b) branching ratios for the H-abstraction reactions of UDMH on the Hcis-CH3 sites, with NO2 attacking at 100 atm.
-
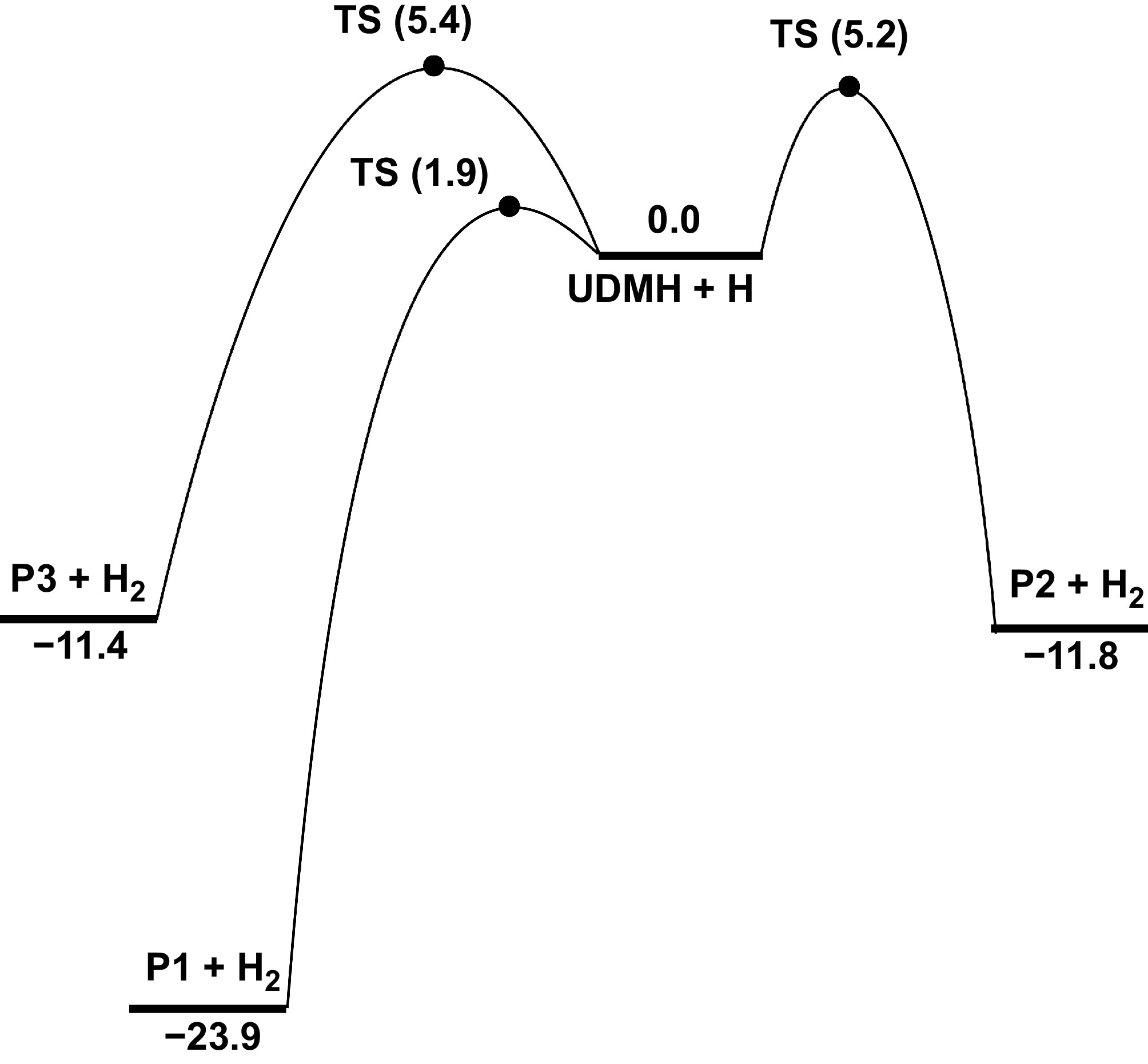
Figure 8.
PESs for the H-abstraction at Hinside atoms of UDMH with H attacking at CCSD(T)/CBS//M062X/def2-TZVP level (unit: kcal/mol).
-
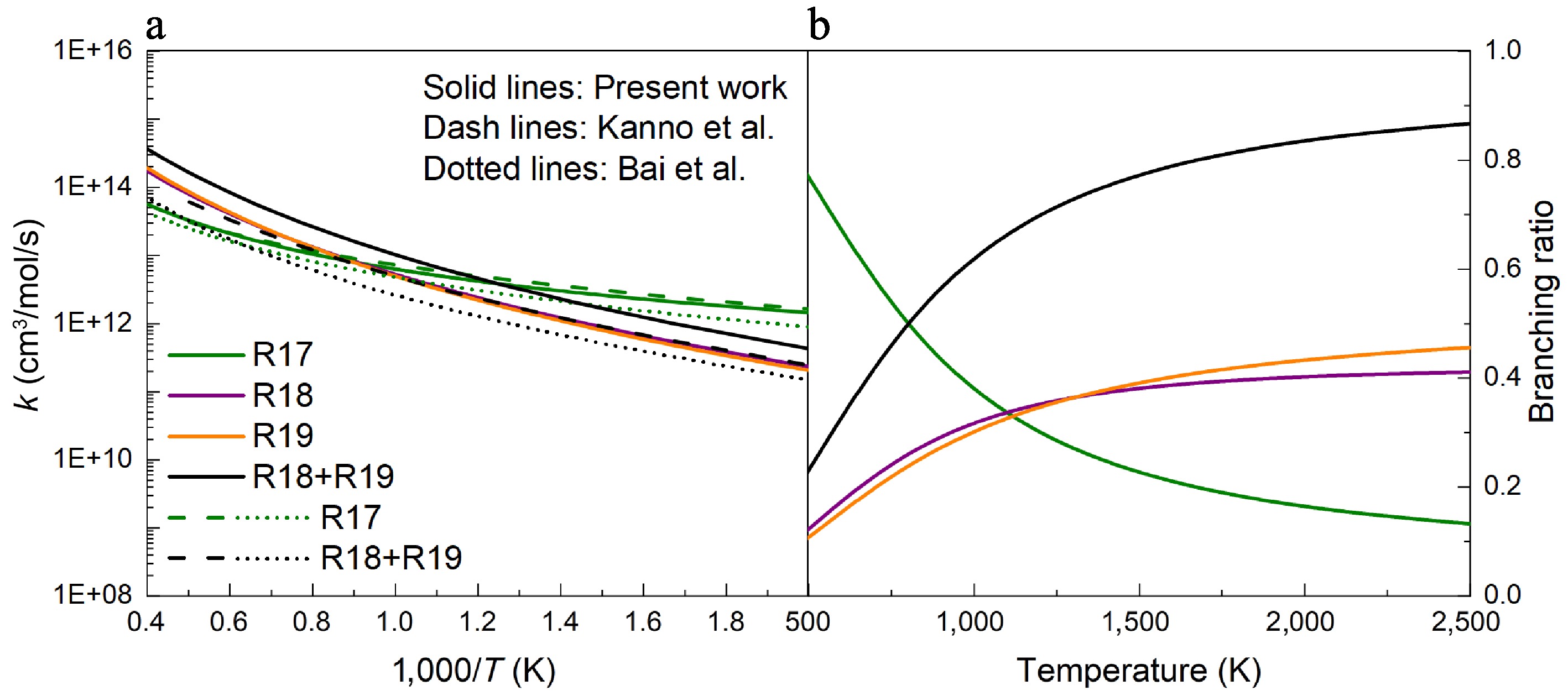
-
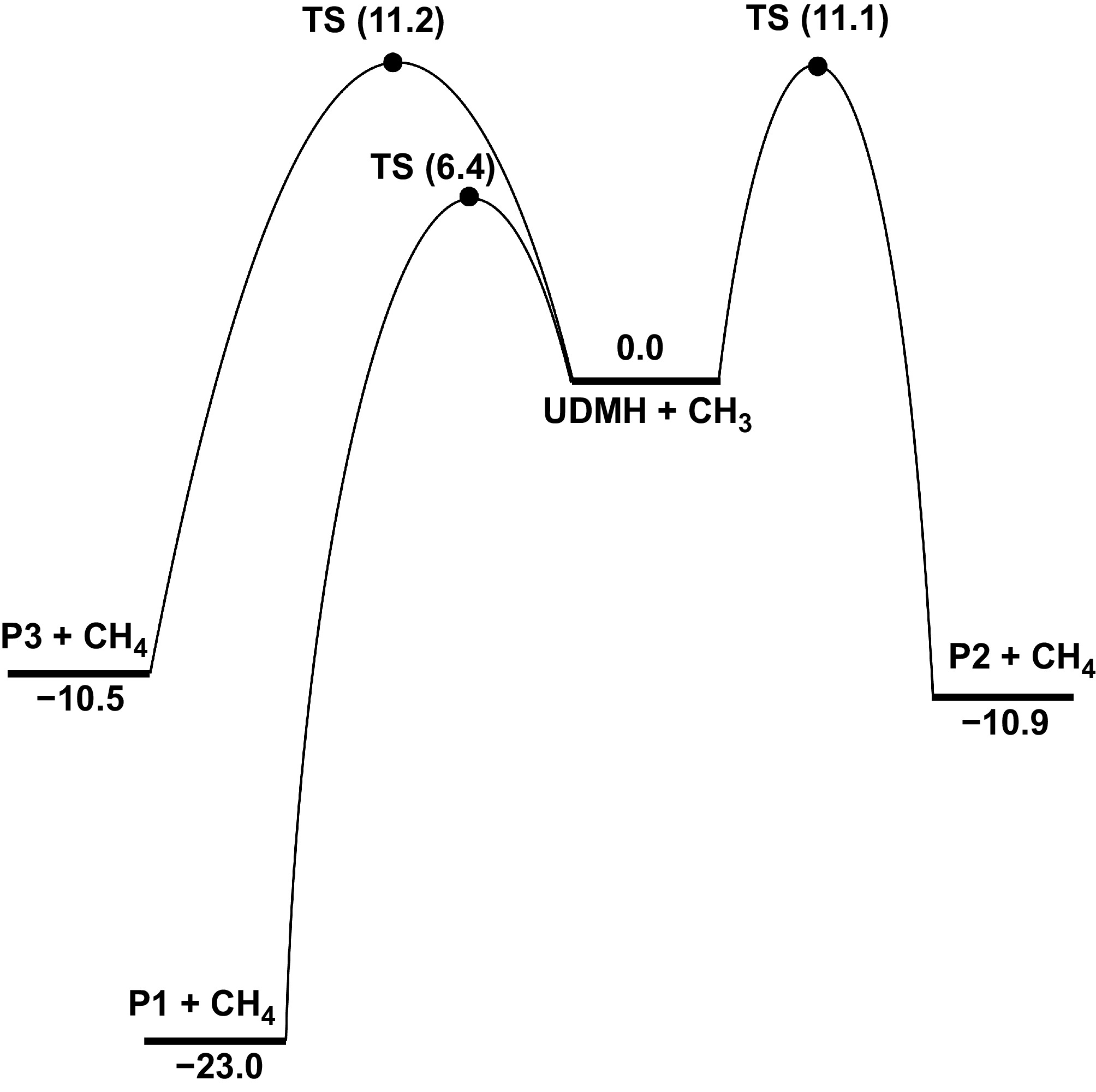
Figure 10.
PES for the H-abstraction at Hinside atoms of UDMH, with CH3 attacking at CCSD(T)/CBS//M062X/def2-TZVP level (unit: kcal/mol).
-
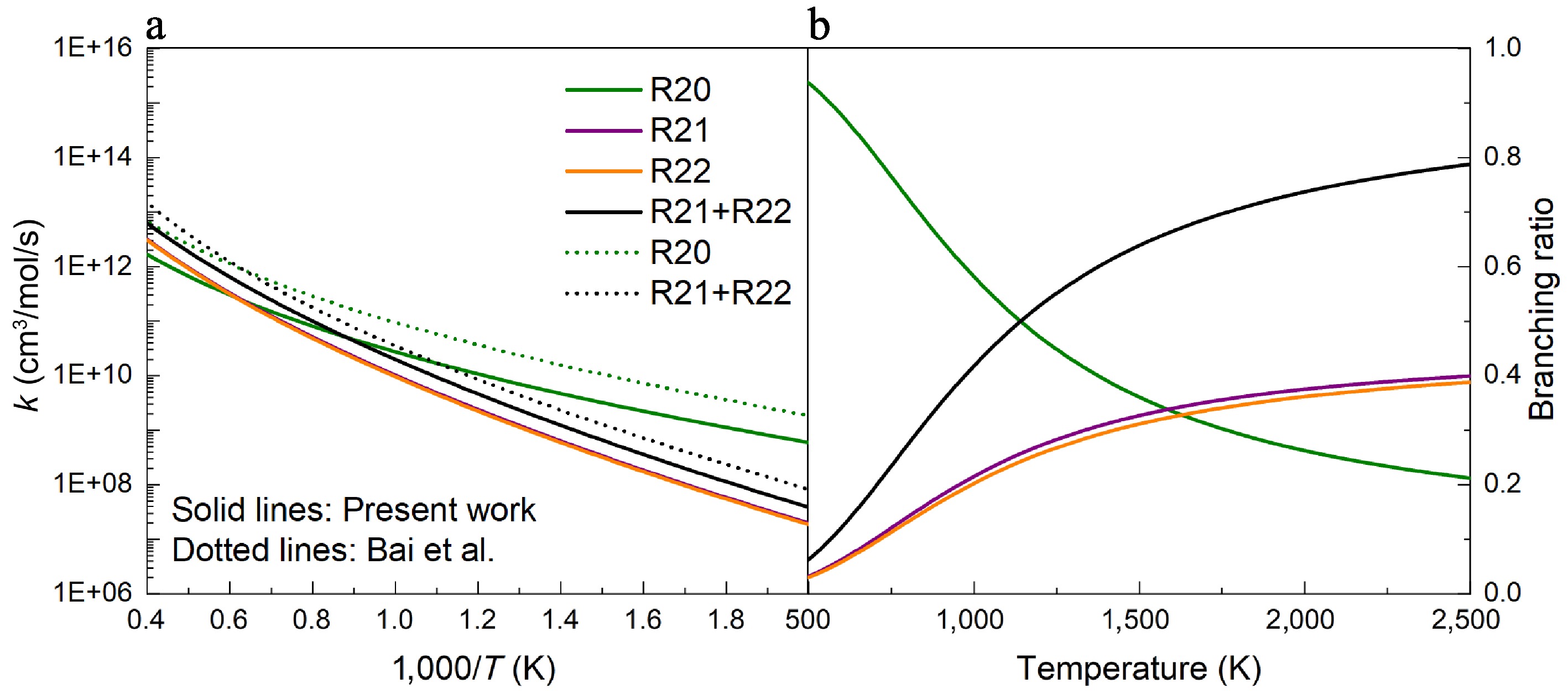
Figure 11.
(a) Rate constants, and (b) branching ratios for the H-abstraction reactions of UDMH with CH3 attacking at HPL calculated in this work and by Bai et al.[25].
-
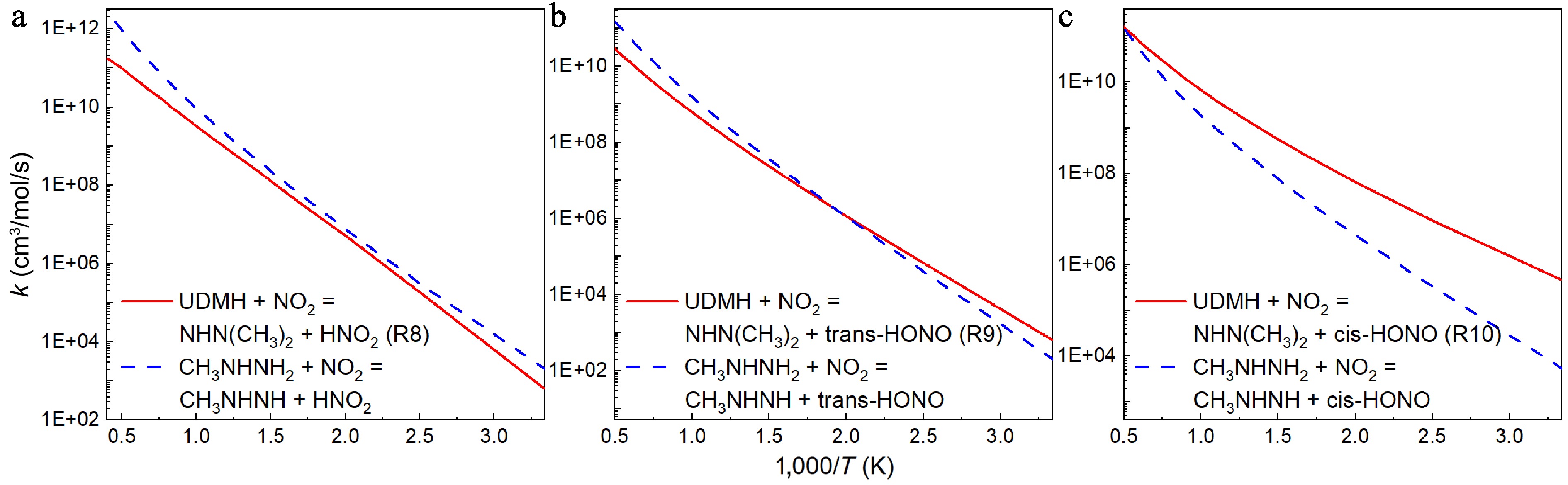
Figure 12.
Comparison between the rate constants for the H abstraction reactions at the NH2 group of UDMH ([a] R8, [b] R9, and [c] R10) and CH3NHNH2[18] with NO2 attacking.
-
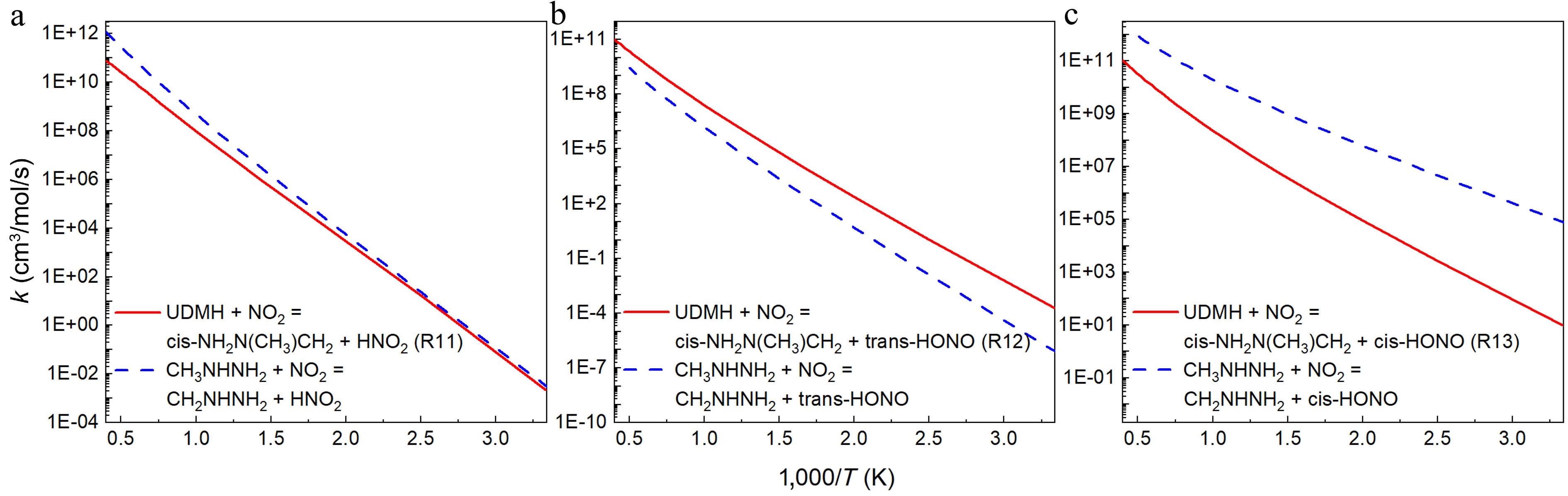
Figure 13.
Comparison between the rate constants for the H abstraction reactions at per CH3 group of UDMH ([a] R11, [b] R12, and [c] R13) and CH3NHNH2[18] with NO2 attacking.
-
-
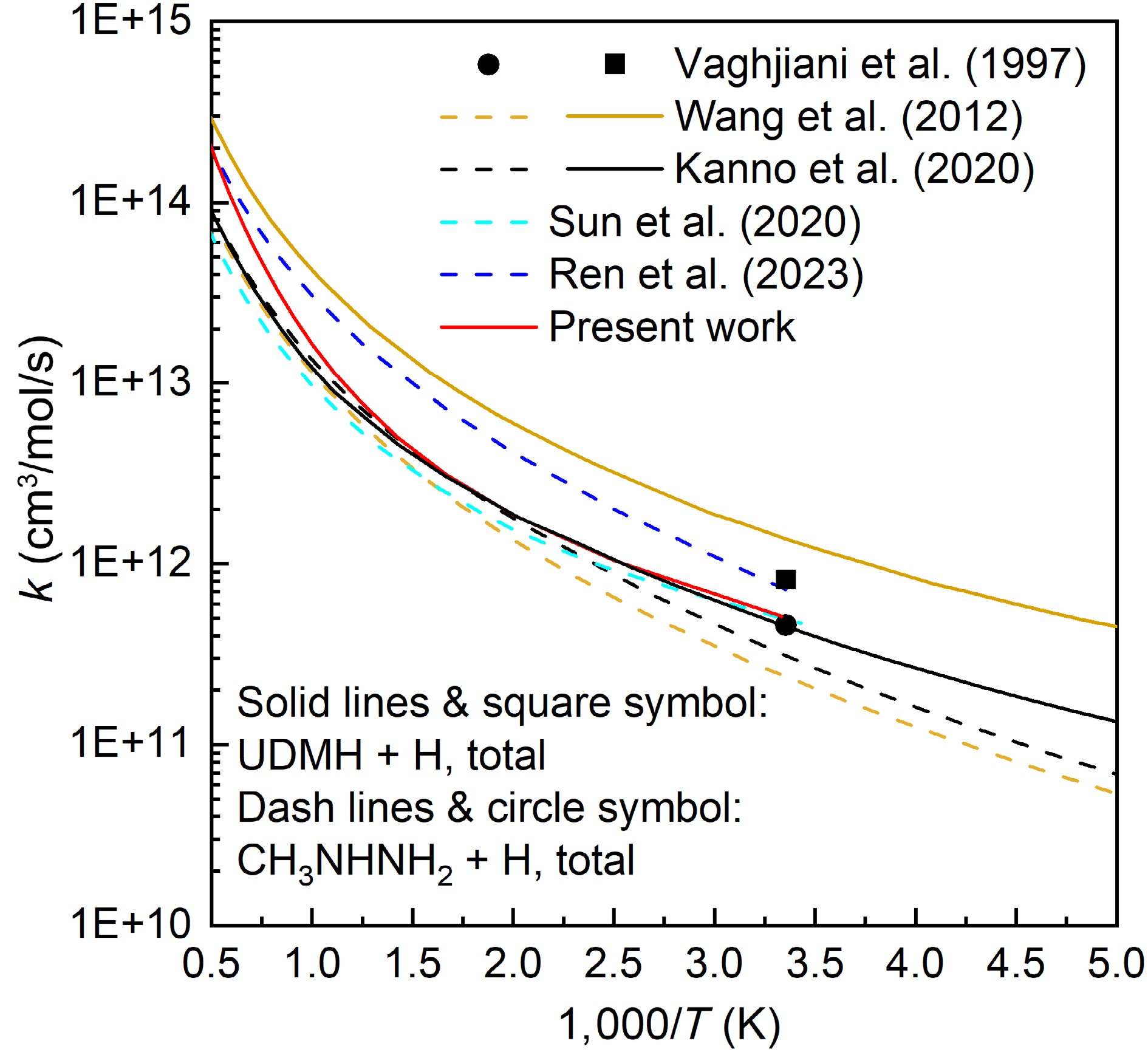
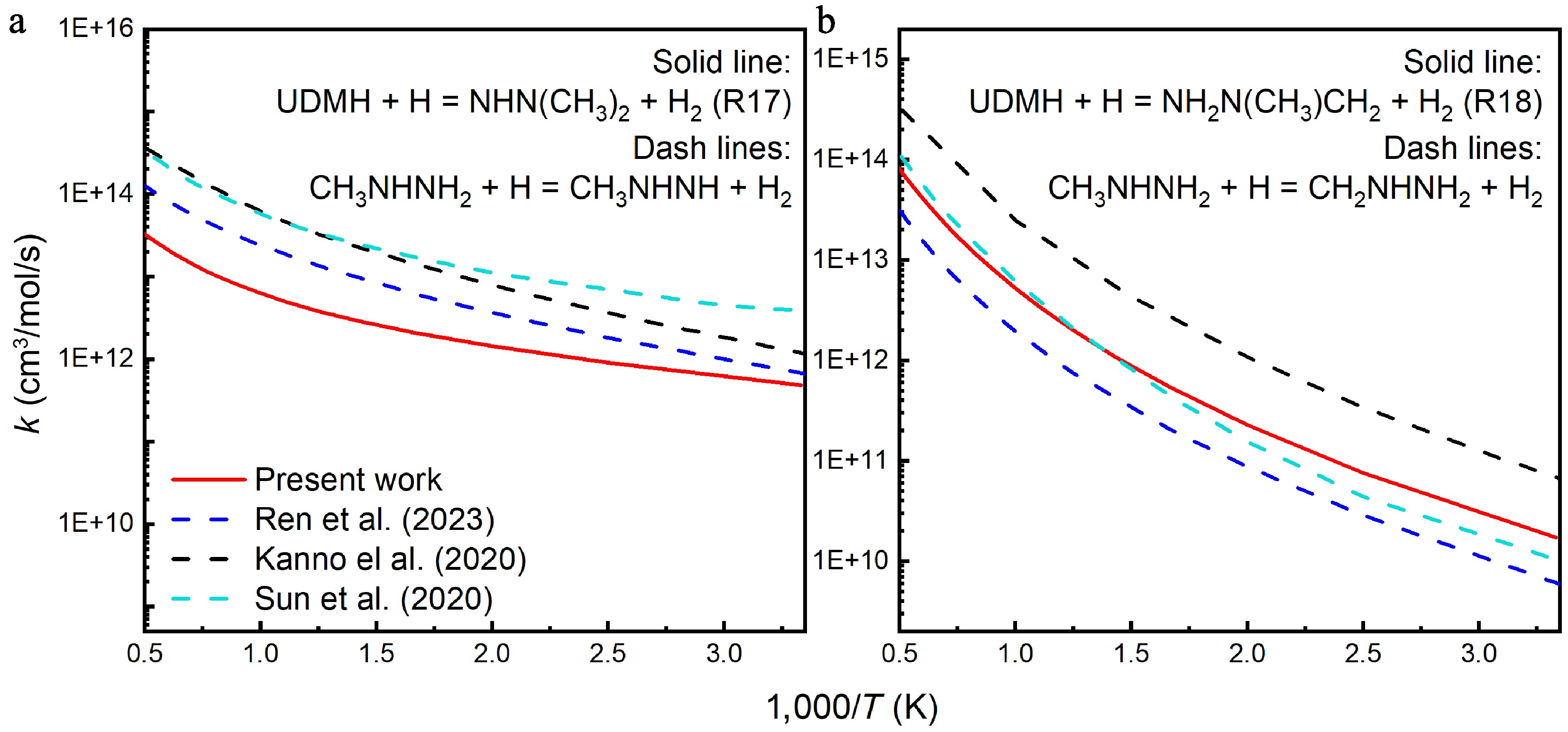
Figures
(15)
Tables
(0)