-
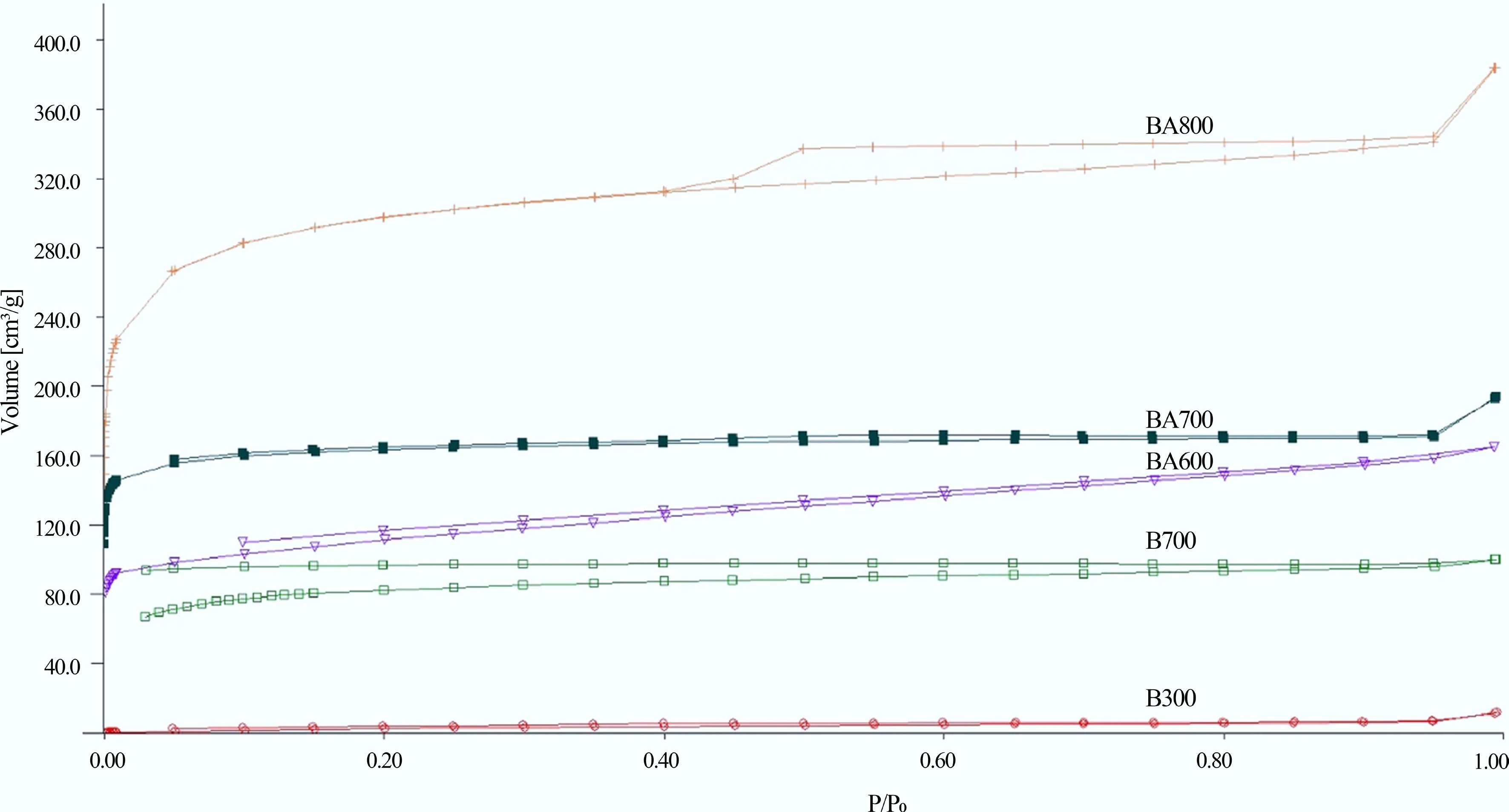
Figure 1.
N2 adsorption isotherms at 77 K for pure (B300, B700) and steam-activated (BA600, BA700, and BA800) biochar[30].
-
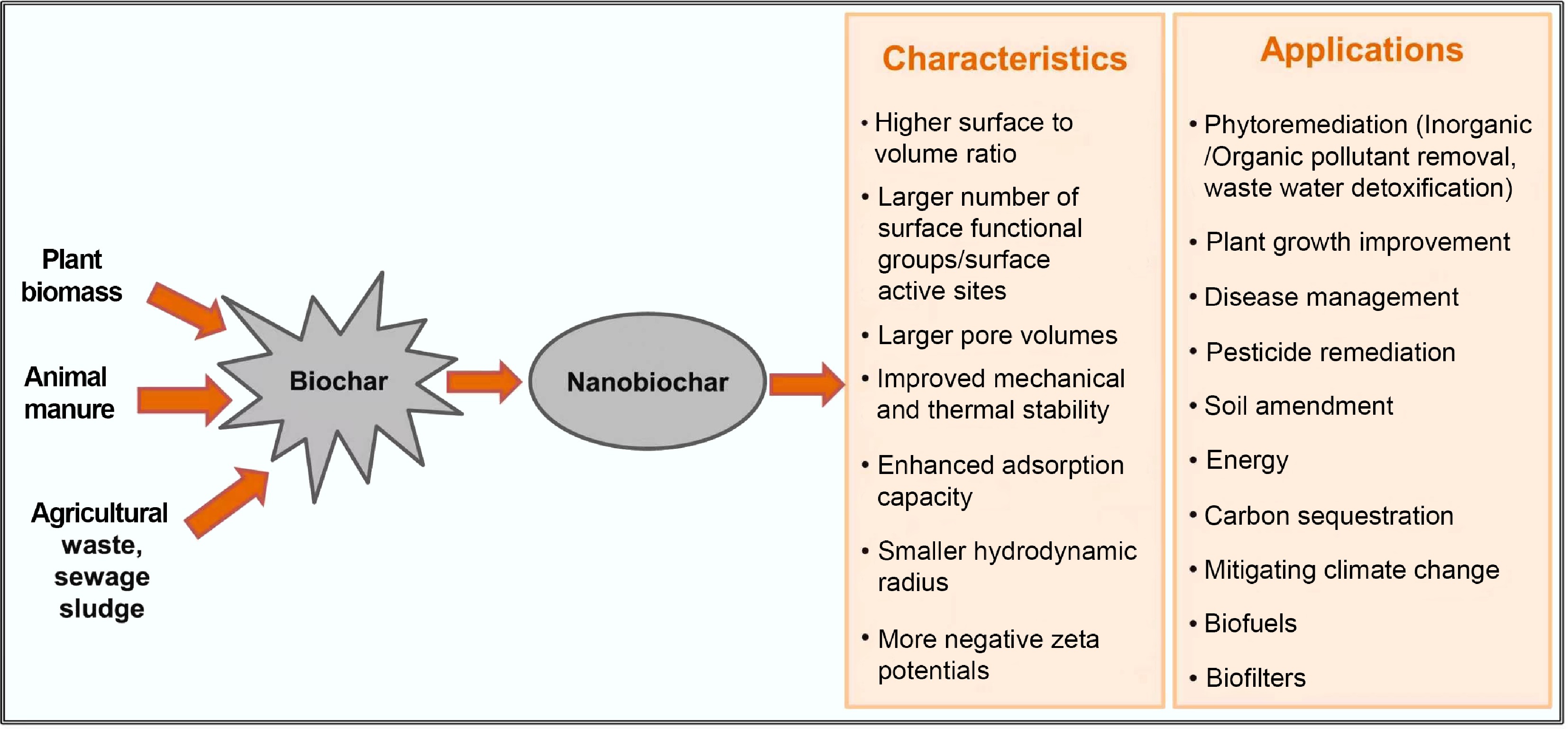
Figure 2.
Nanobiochar and biochar nanocomposites: characteristics and applications[67].
-
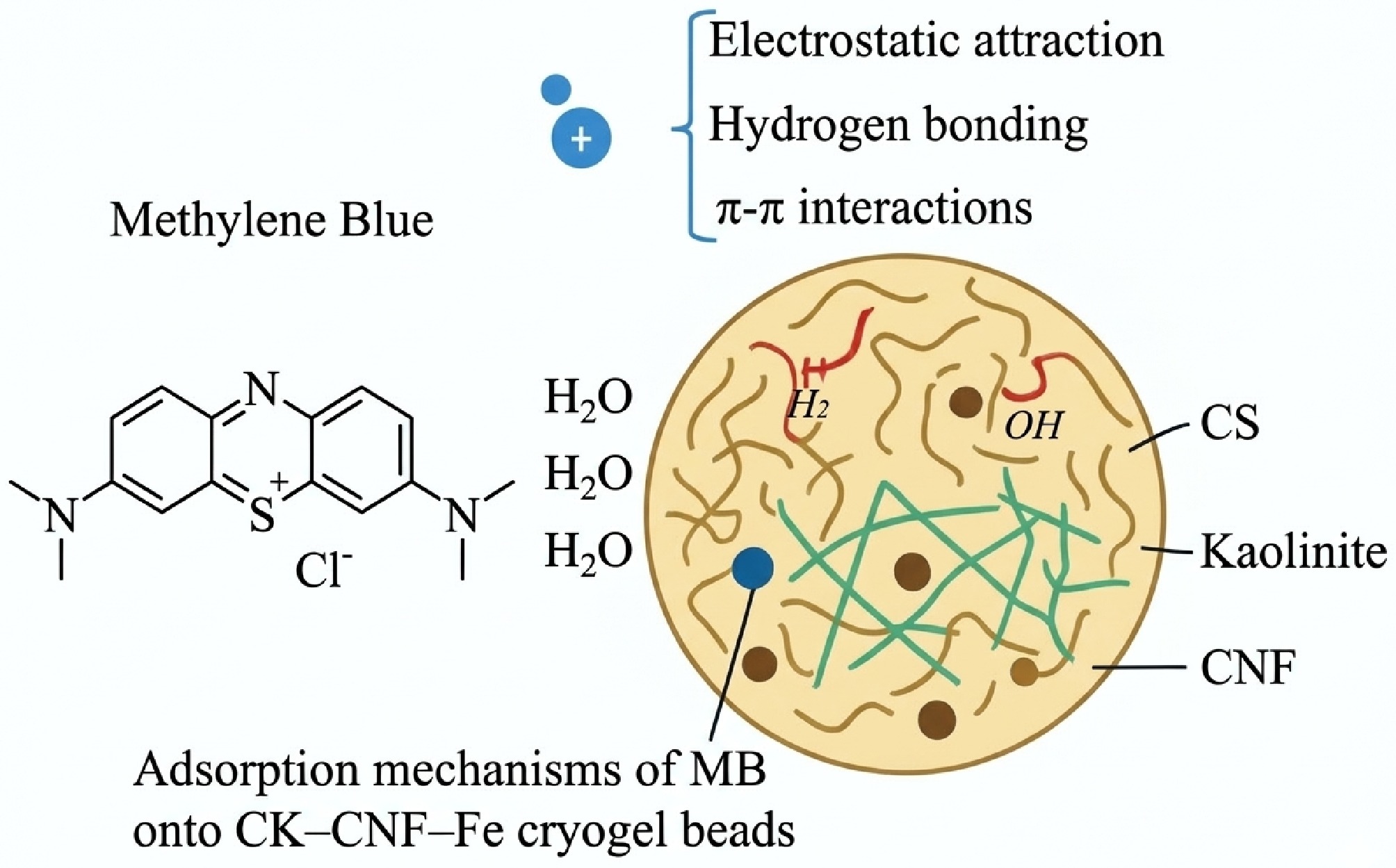
Figure 3.
Schematic illustration of the adsorption mechanisms of MB onto CK–CNF–Fe cryogel beads[112].
-
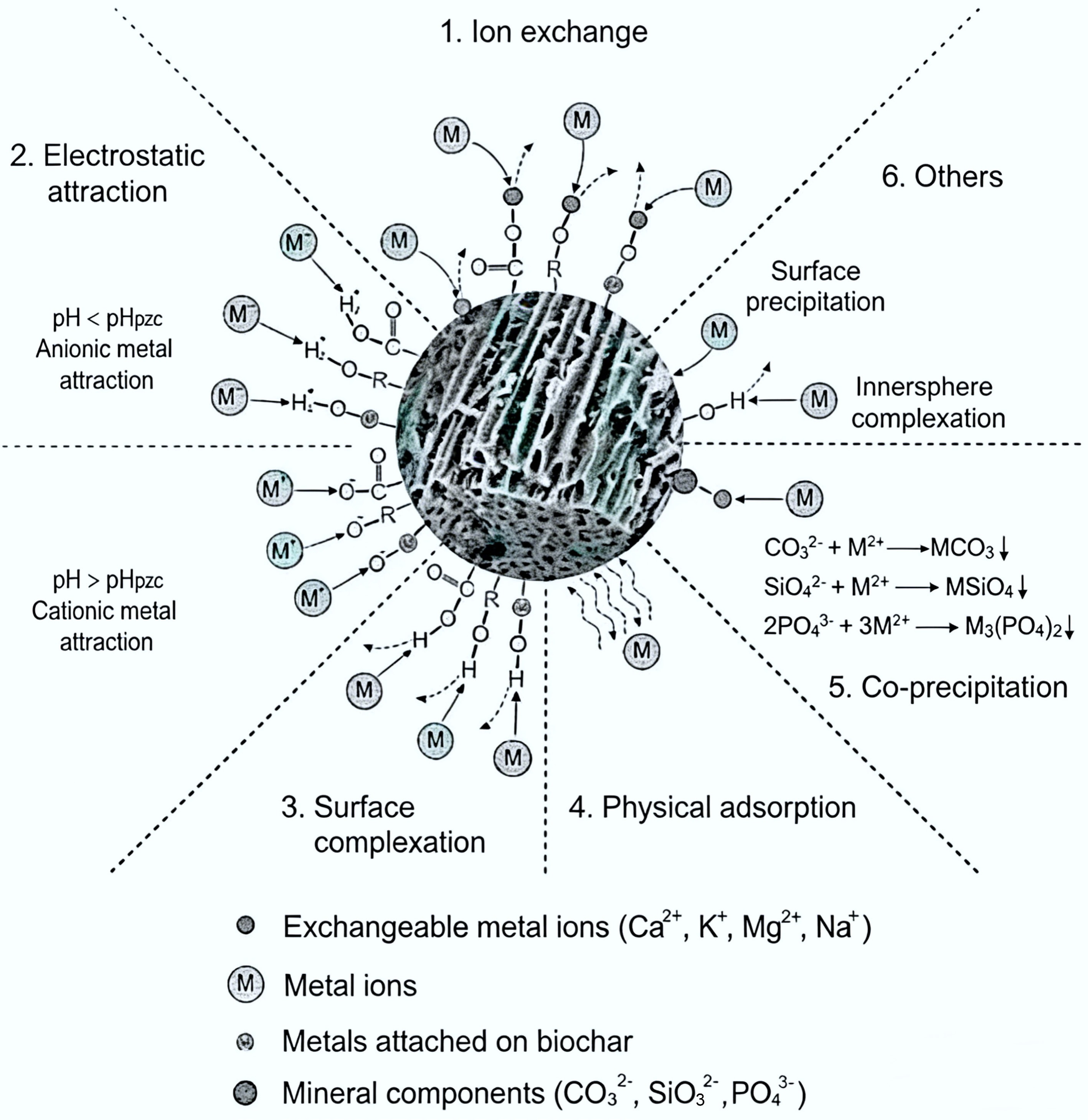
Figure 4.
Adsorption mechanism of biochar[129].
-
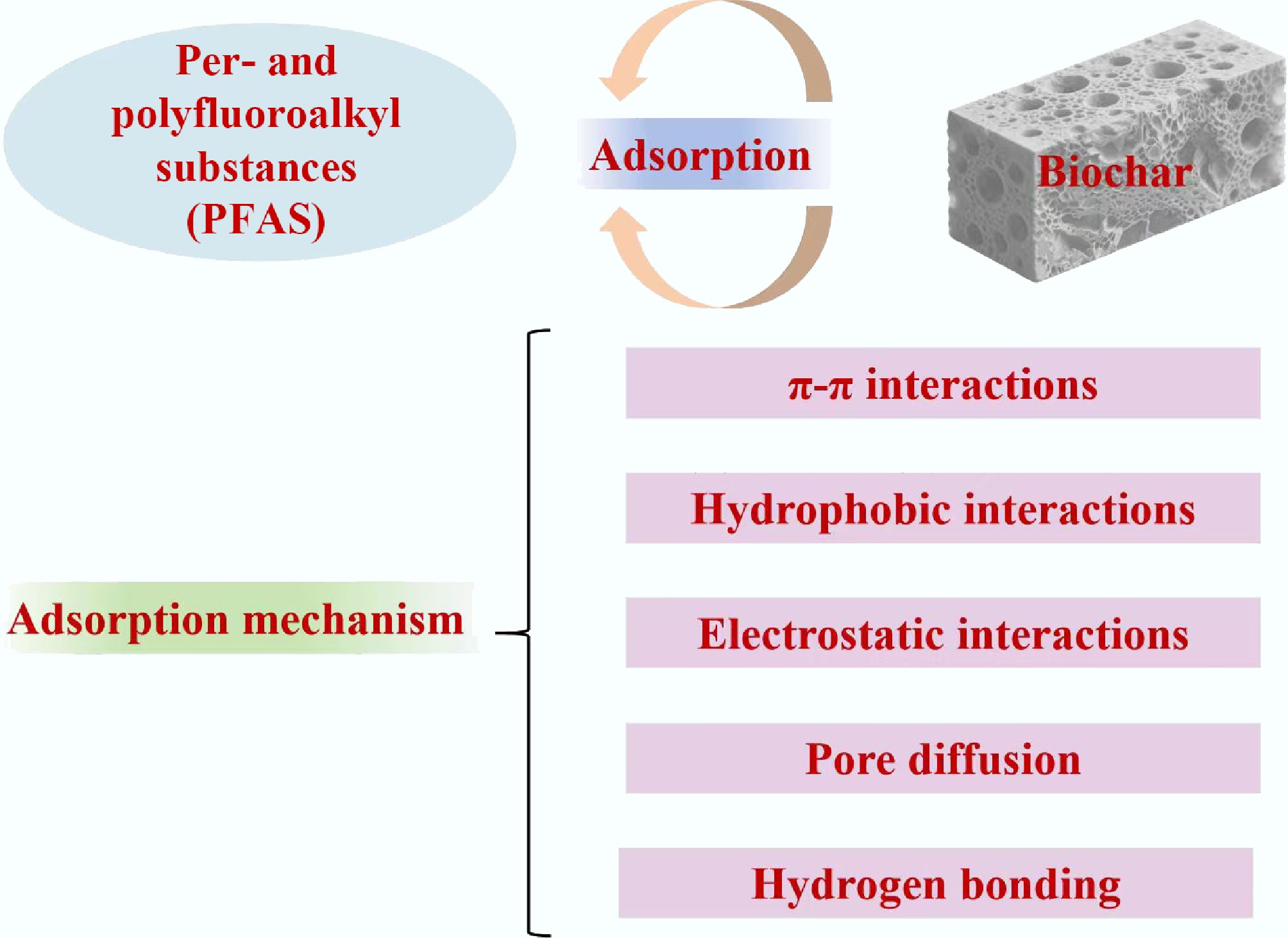
Figure 5.
Possible adsorption mechanism of biochar materials for per-and polyfluoroalkyl substances removal.
-
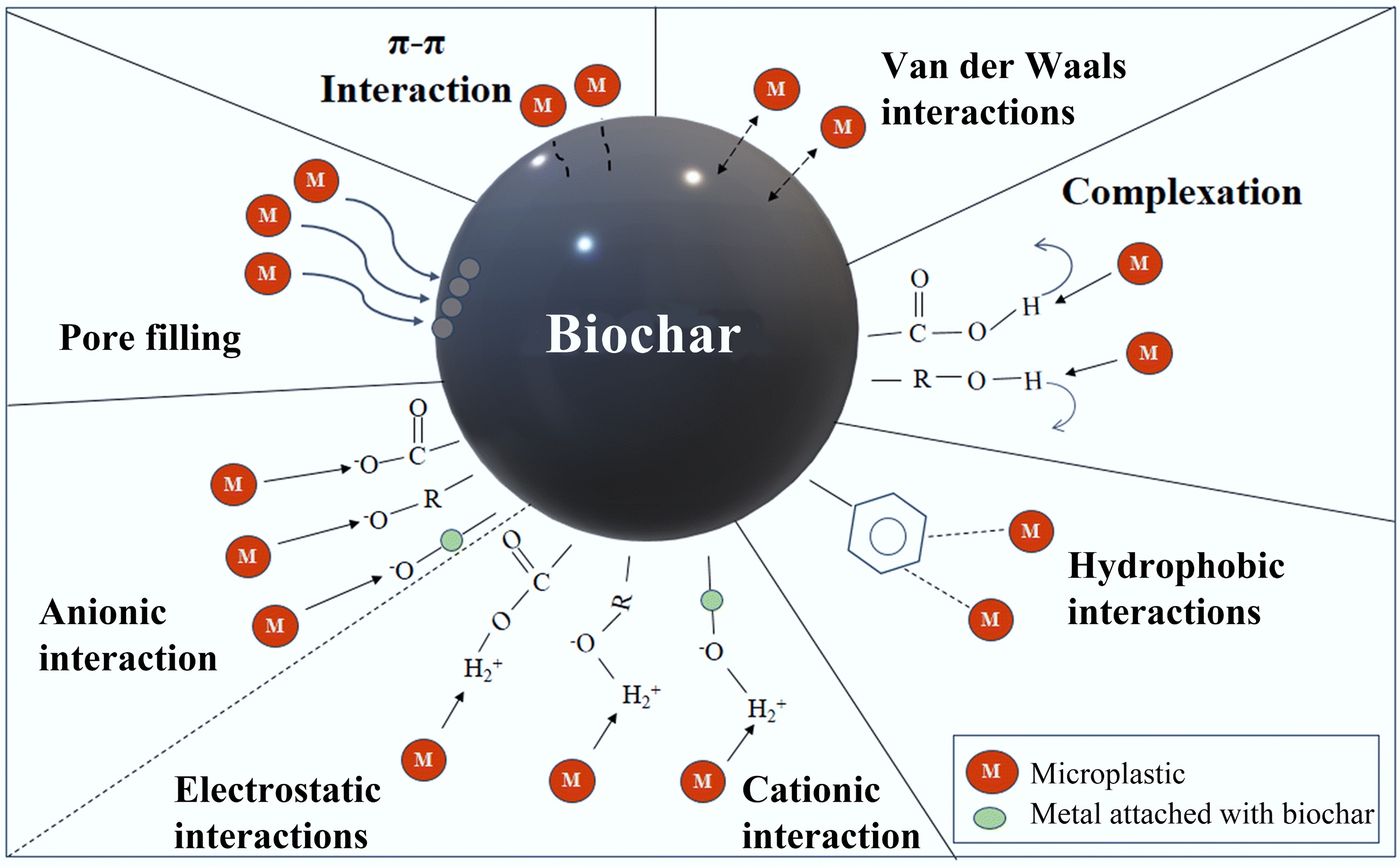
Figure 6.
Adsorption mechanisms underlying the removal of MPs by the biochar[189].
-
Category Method/material/
parameterEffect on biochar characteristics Mechanism/principle Ref. Feedstock selection Agricultural/forestry wastes Renewable, low-cost; elemental/ash variations significantly affect SSA, porosity, and functional groups. Intrinsic components (lignin, cellulose, ash) determine physicochemical structure. Hayder et al.[4] Invasive plants (water hyacinth, ragweed) Superior pollutant adsorption and stable carbon sequestration. Utilization of specific cellular structures and chemical compositions. Tiwari et al.[5] Co-pyrolysis (aquatic plants and sawdust) Hierarchical pore formation; SSA and pore volume superior to single feedstocks. Complementary pyrolysis behaviors of different components. Loc et al.[6] Co-pyrolysis (red mud and pomelo peel) Magnetic biochar; surface iron sites activate PMS for dye degradation. Synergistic waste utilization; introduction of magnetic/catalytic functions. Gai et al.[7] Fruit waste (walnut shell) 88.8% As removal efficiency. Developed porosity and abundant surface oxygen groups. Kumar et al.[8] Method Pyrolysis Produces biochar, bio-oil, syngas. Slow pyrolysis: high char yield; fast: high bio-oil. Thermochemical decomposition under oxygen-limited conditions. Cha et al.[9] Hydrothermal carbonization (HTC) Suitable for high-moisture feedstocks; hydrochar has higher carbon content. Hydrolysis, dehydration, decarboxylation, and aromatization. Funke et al.[10] Gasification Low char yield; rich in alkali salts/minerals, suitable for soil remediation. Partial oxidation targeting gaseous products. You et al.[11] Parameter Temperature Ash, SSA, pH increase; H/C and (O+N)/C decrease; aromaticity increases. Volatile release, micropore development, and unstable component removal. Tomczyk et al.[12] Atmosphere (CO2 vs N2) BET area in CO2 (303.73 m2/g) >> N2
(24.44 m2/g).CO2 selectively etches carbon skeleton, promoting pore expansion. Banik et al.[13] Property Elemental composition Lignocellulosic: high C, low ash; manure/sludge: high ash, nutrient retention. Temperature-dependent decomposition of hemicellulose, cellulose, and lignin. Ippolito et al.[14] BET surface area Increases then decreases with temperature; CO2 enhances SSA. Pore creation via volatile release vs collapse at high temps; CO2 etching. Zhao et al.[15] Functional groups Low-temp: rich in O-groups (–OH, –COOH); High-temp: group loss, hydrophobicity. Thermal decomposition of unstable oxygen-containing groups. Gomez-Eyles et al.[16] CEC Peaks at 250–300 °C; oxidative modification boosts CEC. Correlates with acidic O-groups; modification adds new groups. Kharel et al.[17] Note: SSA, specific surface area; CEC, cation exchange capacity. Table 1.
Preparation and properties of biochar
-
Method Core features and efficiency advantages Major limitations Typical temperature Recommended applications Ref. Slow pyrolysis High biochar yield; stable structure; high degree of aromaticity. Time-consuming; relatively low heating efficiency. 400–600 °C Prioritizing char yield and stable carbon sequestration (e.g., soil amendment). Do et al.[33] Fast pyrolysis High bio-oil yield; rapid process. Low biochar yield; difficult to tune physicochemical properties. 500–700 °C Primarily targeting bio-energy (bio-oil) co-production. Cha et al.[9] Hydrothermal carbonization (HTC) Directly treats high-moisture feedstocks (avoids drying energy); rich surface functional groups. Product (hydrochar) typically has a lower degree of carbonization 180–250 °C Immediate valorization of high-moisture wastes (e.g., sludge, food residues). Xu et al.[21] Co-pyrolysis/
modified pyrolysisSynergistic effects; enables one-step doping, magnetization, or pore optimization; simplifies processing. Precursor formulation and reaction mechanisms can be complex. Target-dependent (e.g., magnetic: 250–600 °C) Targeted preparation of functional composites (e.g., magnetic adsorbents, catalysts). Zheng et al.[34] Gasification Primary target is Syngas (H2, CO); mature technology for large-scale continuous production; high energy efficiency. Very low biochar yield (5%–15%); strict process control required; tar and ash issues; complex equipment/operation. 600–1,200 °C Large-scale bio-energy utilization (power/hydrogen generation); biochar used for sequestration or soil amendment. You et al.[11] Table 2.
Comprehensive comparison and selection guide for major biochar preparation methods
-
Biomass/feedstock Synthesis method Active species Target pollutant Ref. Pine sawdust and sodium alginate Cross-linking with FeCl3 and sodium alginate for simultaneous molding and magnetization, followed by low-temperature pyrolysis. Fe3O4 Pb(II) in soil Zeng et al.[62] Elephant grass Grinding biomass with K2FeO4, followed by pyrolysis at
700 °C for 1 h under nitrogen atmosphere.nZVI (nano zero-valent iron) Sulfamethoxazole He et al.[63] Shiitake mushroom Precursor formation via iron salt adsorption by shiitake mushrooms, followed by high-temperature calcination. Fe/Fe4N Microwave absorption (functional materials) Qiang et al.[64] Wheat straw Impregnation with iron and tin salts, followed by pyrolysis. Fe3O4 Tetracycline Zhang et al.[65] Tobacco stalk Co-pyrolysis with biomass using FeCl2·4H2O as the magnetizing agent. Magnetic metal oxides Phosphorus Yu et al.[66] Table 3.
Synthesis methods of different biochar composites
-
Biomass Synthesis method Composite material Application performance Ref. Pine sawdust One-step synthesis of magnetic biochar via hydrothermal carbonization combined with FeCl3 impregnation. Fe3O4/biochar composite Adsorption capacity of 98.5 mg/g for tetracycline; possesses magnetic separation properties. Ma et al.[113] Rice husk Pyrolysis after co-impregnation with MnCl2 and FeCl3. MnFe2O4/biochar composite Simultaneous removal of As(III) and Cd(II) from water; removal rates > 95%. Khan et al.[114] Peanut shell One-step pyrolysis combining ZnCl2 activation and LaCl3 modification. La-loaded porous biochar composite Deep removal of phosphate from water; adsorption capacity reached 102.3 mg P/g. Yi et al.[115] Orange peel Compositing orange peel biochar with magnetite (Fe3O4). Biochar-magnetite nanocomposite ~90% removal of ciprofloxacin within 60 min; magnetic property enables easy separation/recovery. Sharma et al.[116] Cotton Impregnation with FeCl3 solution followed by sulfidation using H2S gas. FeS/biochar composite High Hg(II) adsorption capacity of 287.4 mg/g in water. Nurlan et al.[117] Reed straw Loading Bacillus subtilis onto UV-radiation modified biochar. UV-modified biochar-Bacillus subtilis composite Saturated adsorption capacity for Cd(II) increased from 23.98 to 49.93 mg/g; particularly effective in weakly alkaline environments. Zhu et al.[118] Sludge Synthesis of manganese-modified composite for activating ferrate(VI). Mn-modified sludge biochar composite Rapid activation of ferrate(VI); 87.39% removal of sulfamethoxazole within 10 min; high removal efficiency for other sulfonamides. Deng et al.[119] Table 4.
Application of biochar composites in wastewater treatment
Figures
(6)
Tables
(4)