-
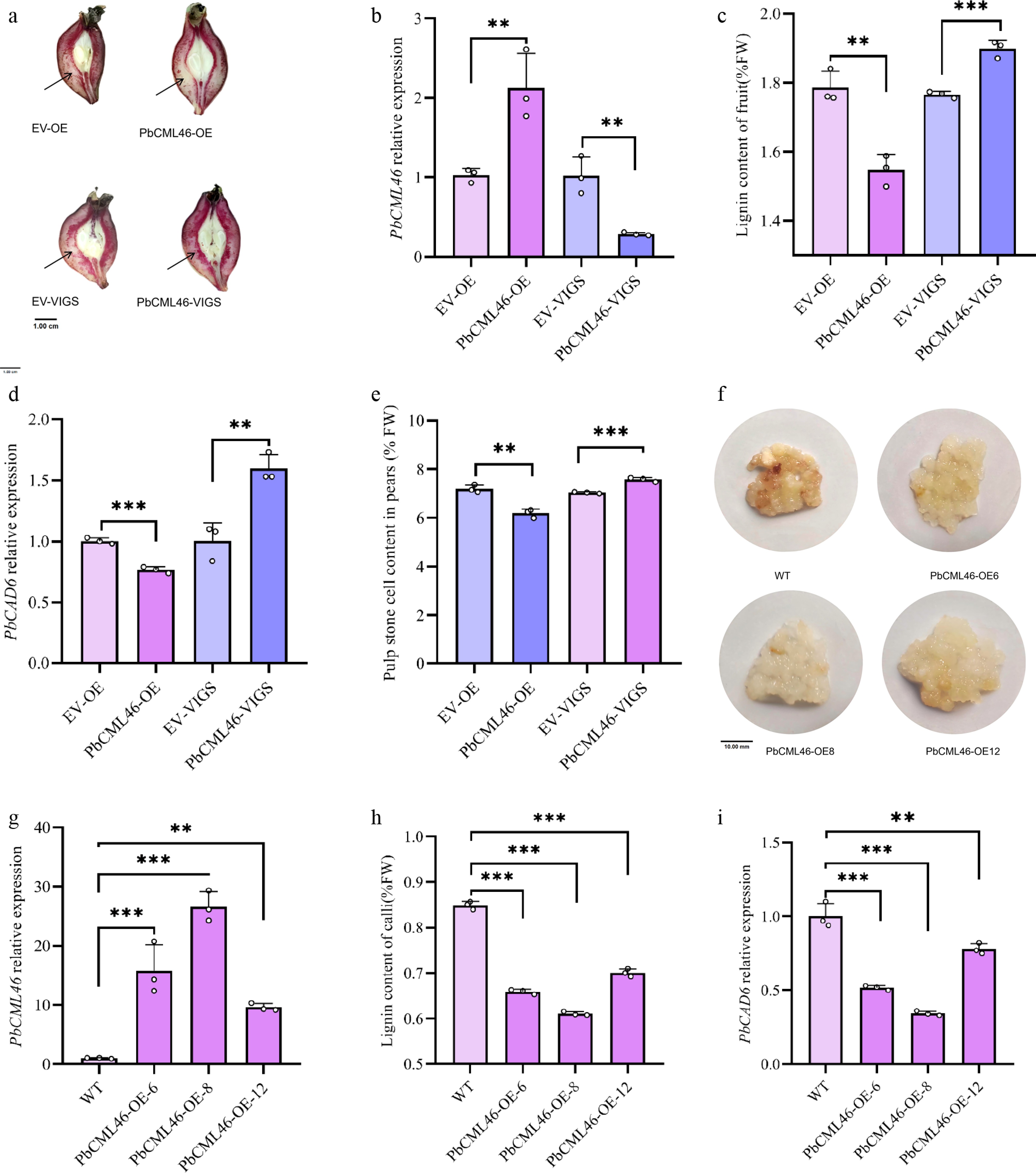
Figure 1.
PbCML46 functions in lignin biosynthesis of stone cells in pear fruit. (a) Histochemical localization of stone cell tissue in pear fruits with empty pCAMBIA, empty pTRV, PbCML46 overexpression, and PbCML46 silencing (pear young fruits 35 d after full bloom). (b) qRT-PCR analysis of the relative expression levels of PbCML46 in the infiltration zones of pear fruits with empty pCAMBIA, empty pTRV, PbCML46 overexpression, and PbCML46 silencing. (c) Measurement of lignin content in the infiltration zones of pear fruits with empty pCAMBIA, empty pTRV, PbCML46 overexpression, and PbCML46 silencing. (d) qRT-PCR analysis of the relative expression levels of PbCAD6 in the infiltration zones of pear fruits with empty pCAMBIA, empty pTRV, PbCML46 overexpression, and PbCML46 silencing. (e) Measurement of stone cell content in the osmotic sites of pear fruits with empty pCAMBIA, empty pTRV, PbCML46 overexpression, and PbCML46 silencing. (f) Lignin staining phenotype of wild-type and PbCML46-overexpressing callus. (g) qRT-PCR validation of the relative expression level of PbCML46 in stably overexpressing callus. (h) Measurement of lignin content in stably overexpressing callus. (i) qRT-PCR analysis of the relative expression level of PbCAD6 in stably overexpressing callus. Values represent the mean ± SD of three independent biological replicates. ANOVA analysis and Tukey's test were used (** p < 0.01; *** p < 0.001).
-
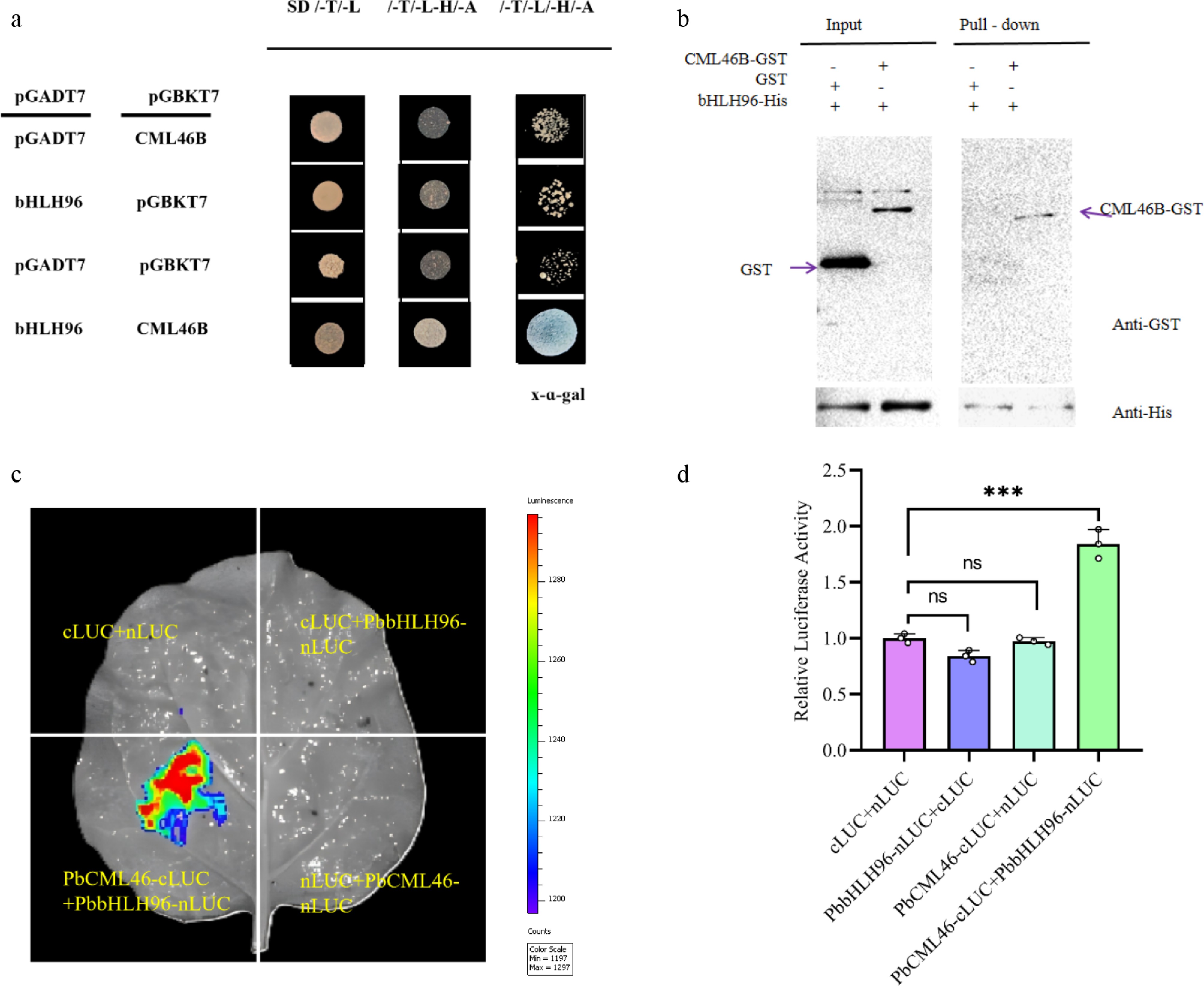
Figure 2.
PbCML46 interacts with PbbHLH96 to regulate lignin biosynthesis in stone cells of pear fruit. (a) Yeast two-hybrid assay showing the interaction between PbCML46 and PbbHLH96. (b) In vitro pull-down assay validating the interaction between PbCML46 and PbbHLH96. '+' and '−' indicate the presence and absence of the specified protein, respectively. (c), (d) Luciferase complementation imaging assays demonstrating the interaction between PbCML46 and PbbHLH96 in planta. Values represent the mean ± SD of three independent biological replicates. One-way ANOVA was performed (*** p < 0.001).
-
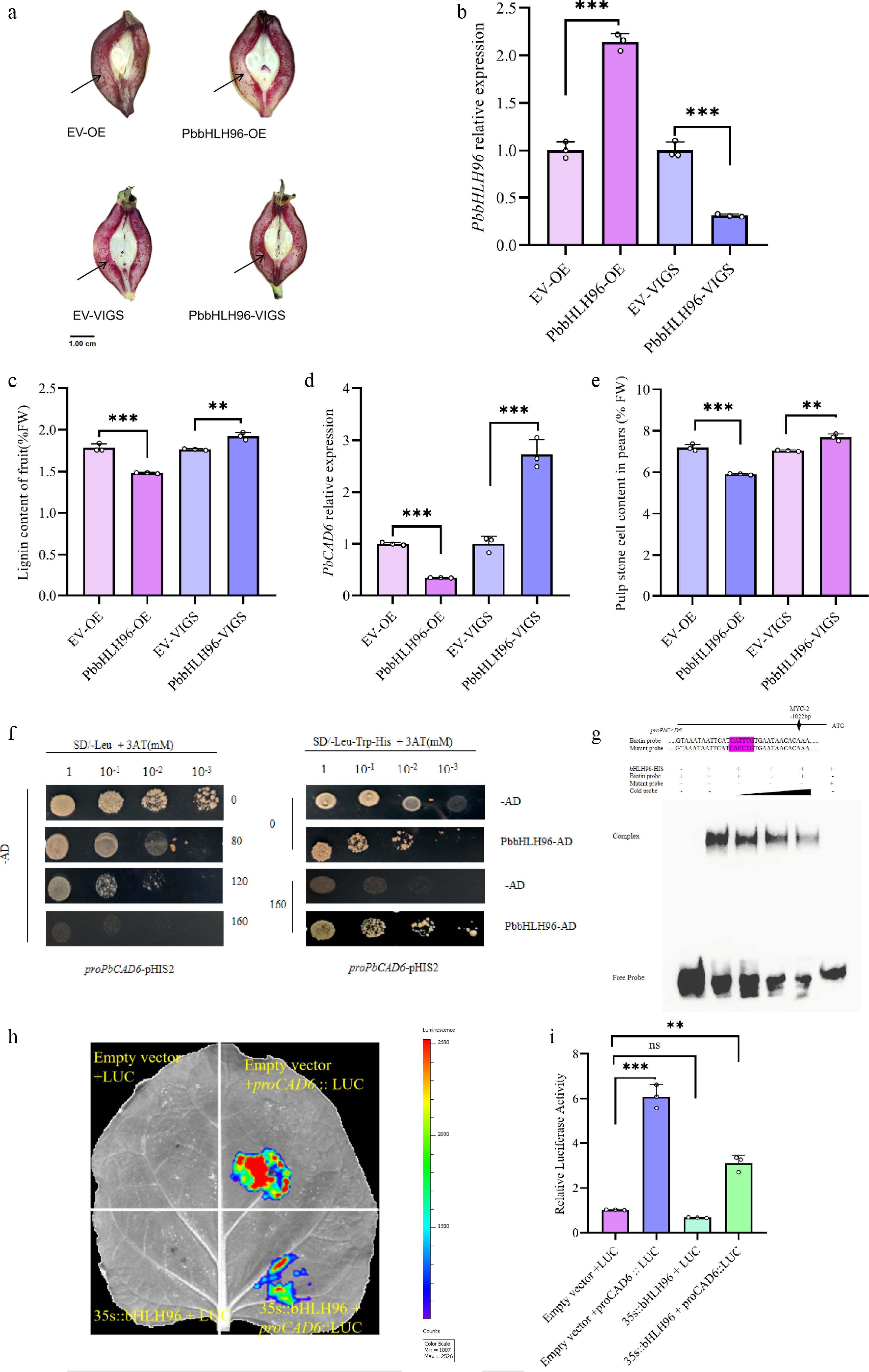
Figure 3.
PbbHLH96 regulates lignin biosynthesis in pear stone cells by binding to and repressing the PbCAD6 promoter. (a) Transient transformation of 'Shannongsu' pear fruits at 35 d after full bloom (DAFB) with PbbHLH96 overexpression and silencing constructs. Longitudinal fruit sections were stained with phloroglucinol-HCl and photographed 12 d after infiltration. qRT-PCR analysis of (b) PbbHLH96 and (c) PbCAD6 expression in transiently transformed fruits. (d) Lignin content measurement at the infiltration sites. (e) Determination of stone cell content in the infiltration site regions of pear fruits with empty pCAMBIA, empty pTRV, PbbHLH96 overexpression, and PbbHLH946 silencing. (f) Yeast one-hybrid assay showing the interaction between PbbHLH96 and the PbCAD6 promoter. (g) Electrophoretic mobility shift assay (EMSA) confirming that PbbHLH96 binds in vitro to the MYC cis-element within the PbCAD6 promoter. (h), (i) Luciferase reporter assay demonstrating that PbbHLH96 represses PbCAD6 promoter activity. Values represent the mean ± SD of three independent biological replicates. Statistical analysis was performed using ANOVA and Tukey's test (** p < 0.01; *** p < 0.001).
-
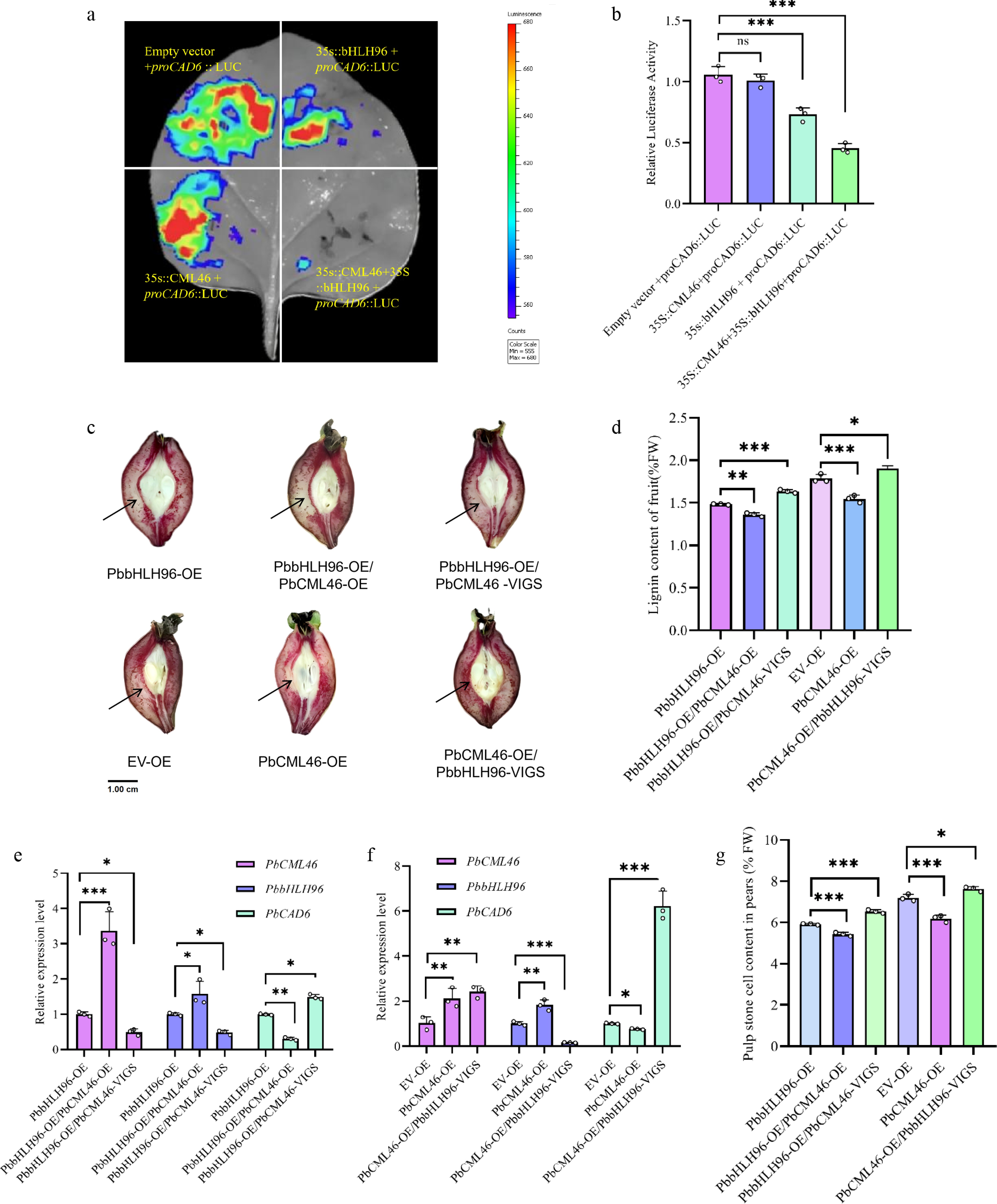
Figure 4.
PbCML46 interacts with PbbHLH96 to enhance its regulatory effect on PbCAD6 and inhibit lignin biosynthesis in pear stone cells. (a) Dual-luciferase reporter (DLR) assay showing that the interaction between PbCML46, and PbbHLH96 enhances the repression of PbCAD6 promoter activity by PbbHLH96. (b) Quantitative analysis of the DLR assay shown in (a). (c) Transient co-transformation in pear fruit via co-injection of overexpression and silencing constructs. Longitudinal fruit sections were stained with phloroglucinol-HCl and photographed 12 d after infiltration. (d) Lignin content measurement (e), (f) qRT-PCR analysis of PbCAD6, PbCML46 and PbbHLH96 expression in the infiltrated areas. (g) Determination of stone cell content in the infiltration regions of pear fruits after instantaneous injection. Values represent the mean ± SD of three independent biological replicates. Statistical analysis was performed using ANOVA (* p < 0.05; ** p < 0.01; *** p < 0.001).
-
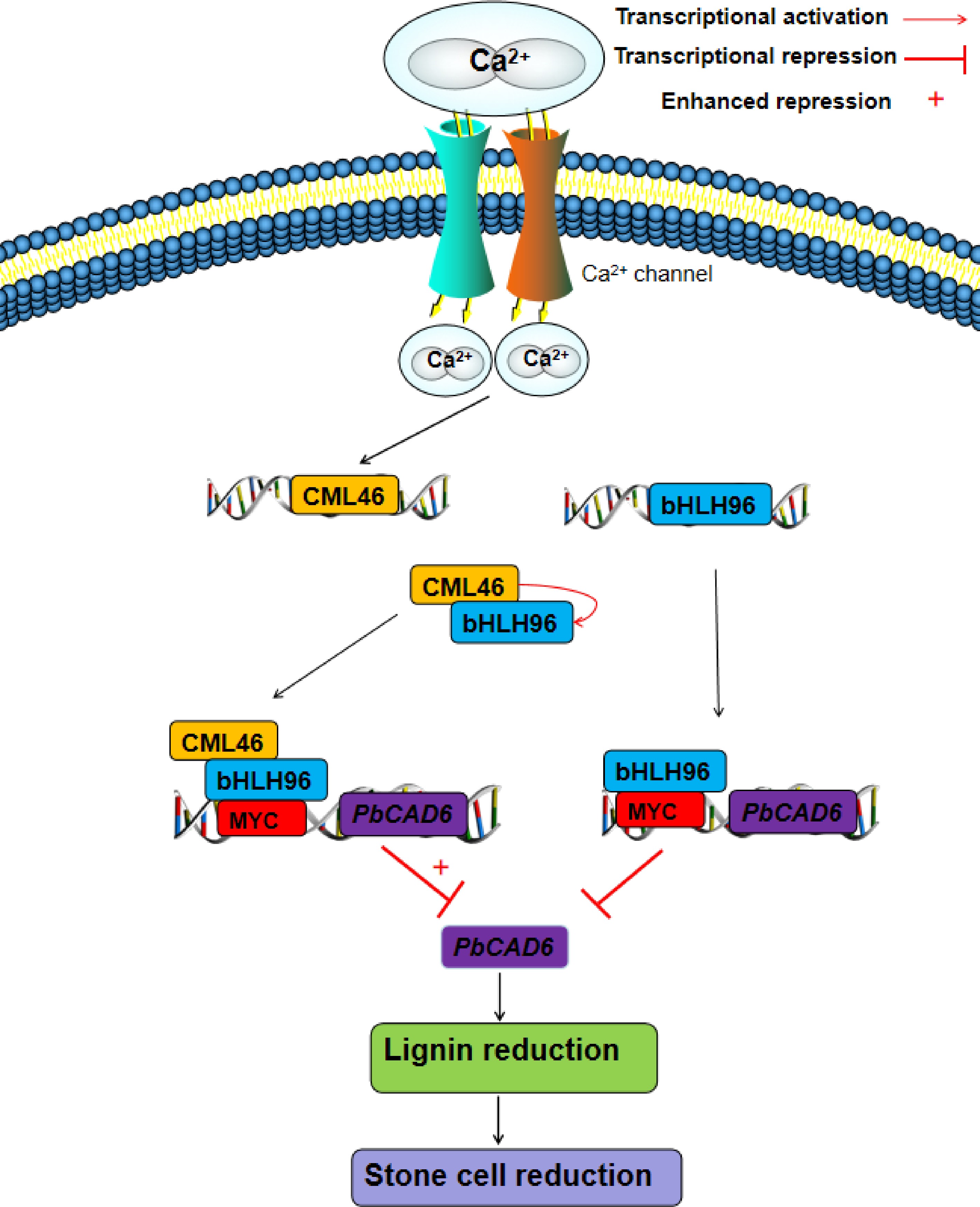
Figure 5.
A proposed regulatory model for the roles of PbCML46 and PbbHLH96 in modulating lignin biosynthesis within stone cells in response to Ca2+ signals in 'Shannongsu' pear. Upon the entry of exogenous calcium ions into the cell, the cytosolic Ca2+ concentration increases. On one hand, this directly induces the expression of PbbHLH96, which in turn binds to the MYC cis-element in the PbCAD6 promoter and represses its expression, thereby inhibiting lignin biosynthesis and stone cell formation. On the other hand, PbCML46 is activated upon Ca2+ binding, undergoing a conformational change that enables its interaction with PbbHLH96, further potentiating the inhibitory effect of PbbHLH96 on downstream PbCAD6 expression, and consequently suppressing lignin and stone cell accumulation. Furthermore, PbCML46 may upregulate PbbHLH96 expression through a feedback mechanism, forming a cascade amplification effect that ultimately leads to a pronounced inhibition of lignin biosynthesis and stone cell formation. This model elucidates the molecular mechanism by which calcium signaling finely regulates lignin metabolism in pear fruit stone cells through the PbCML46-PbbHLH96 module, providing a new theoretical foundation for the improvement of pear fruit quality.
Figures
(5)
Tables
(0)