-
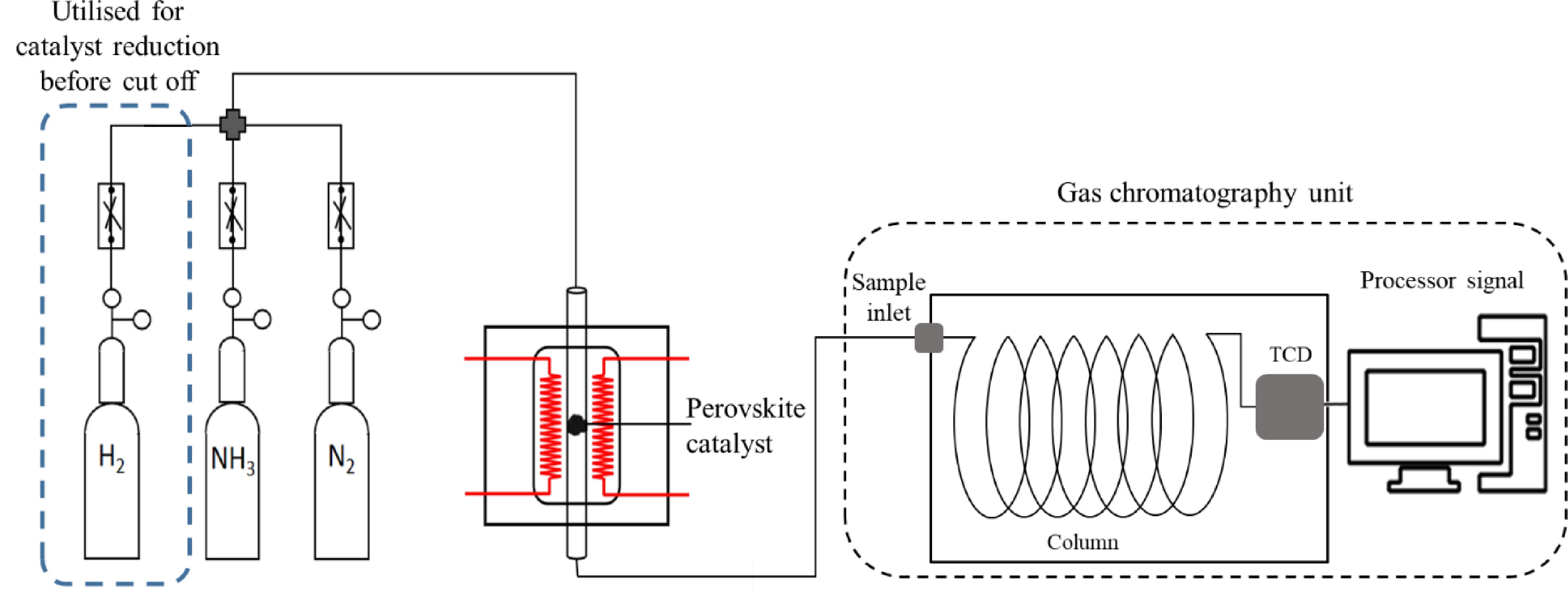
Figure 1.
Schematic of the experimental device for ammonia decomposition.
-
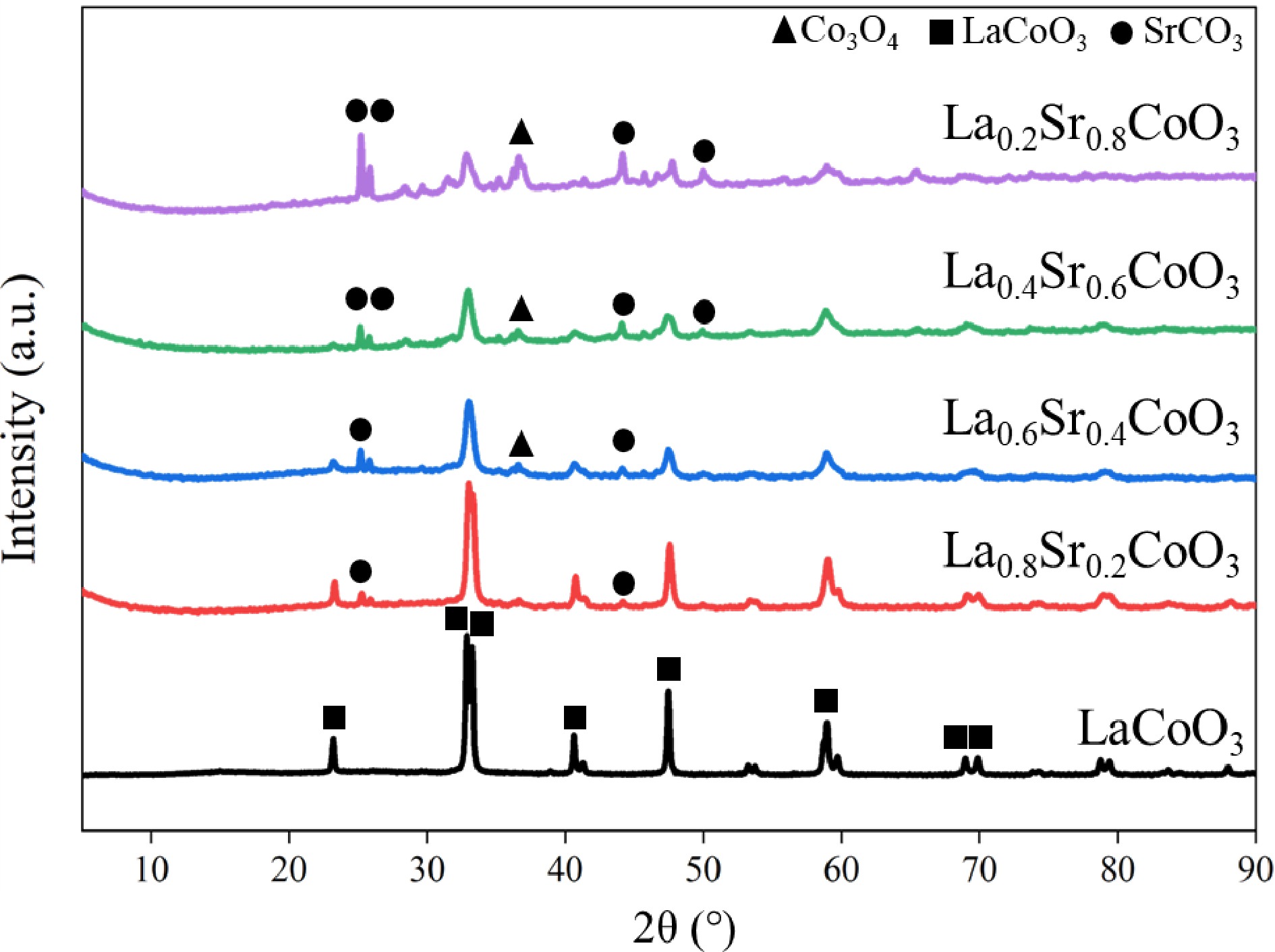
Figure 2.
X-ray diffraction pattern of La(1−x)SrxCoO3 perovskite.
-
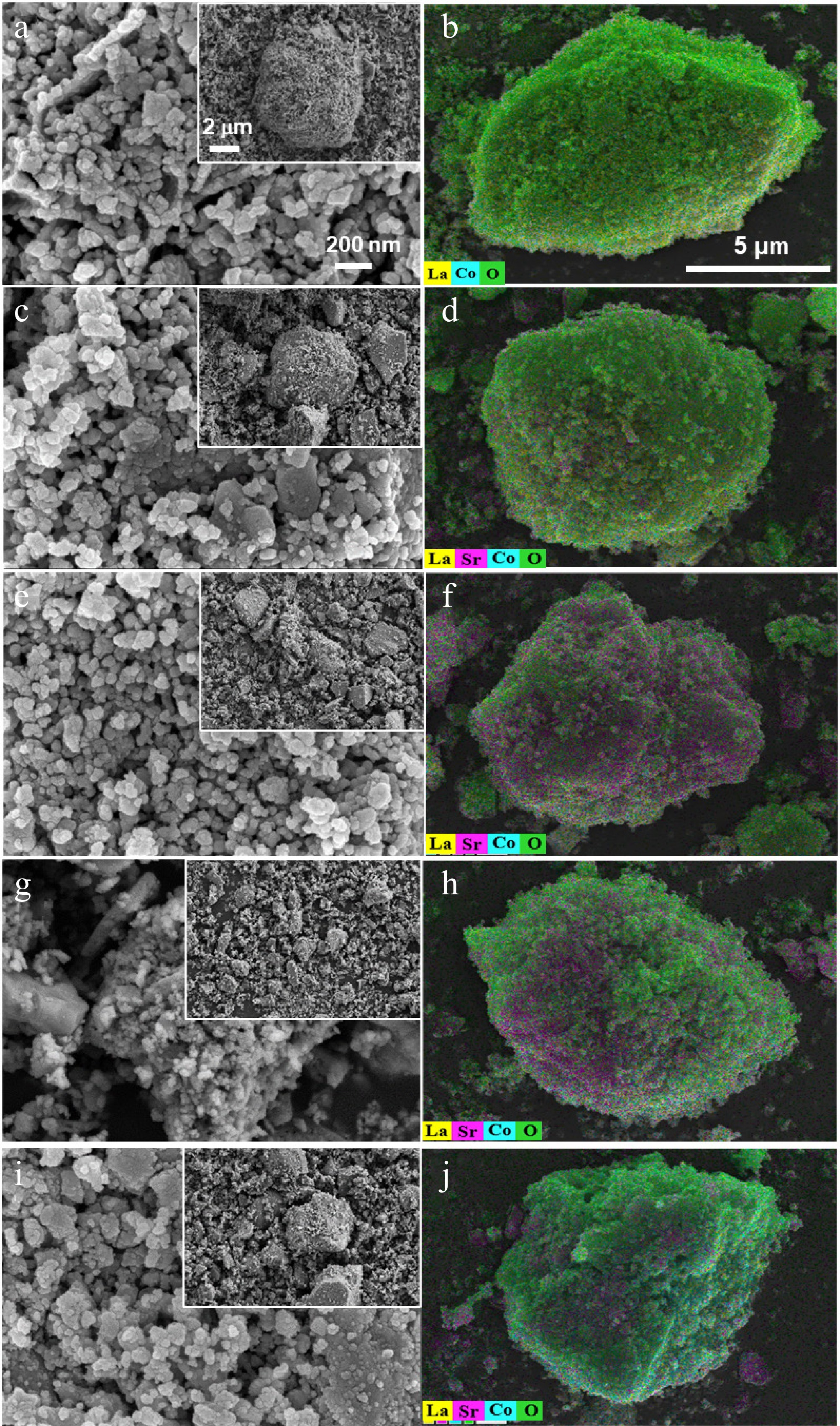
Figure 3.
SEM (first column) and EDS images (second column) of (a), (b) LaCoO3, (c), (d) La0.8Sr0.2CoO3, (e), (f) La0.6Sr0.4CoO3, (g), (h) La0.4Sr0.6CoO3 and (i), (j) La0.2Sr0.8CoO3.
-
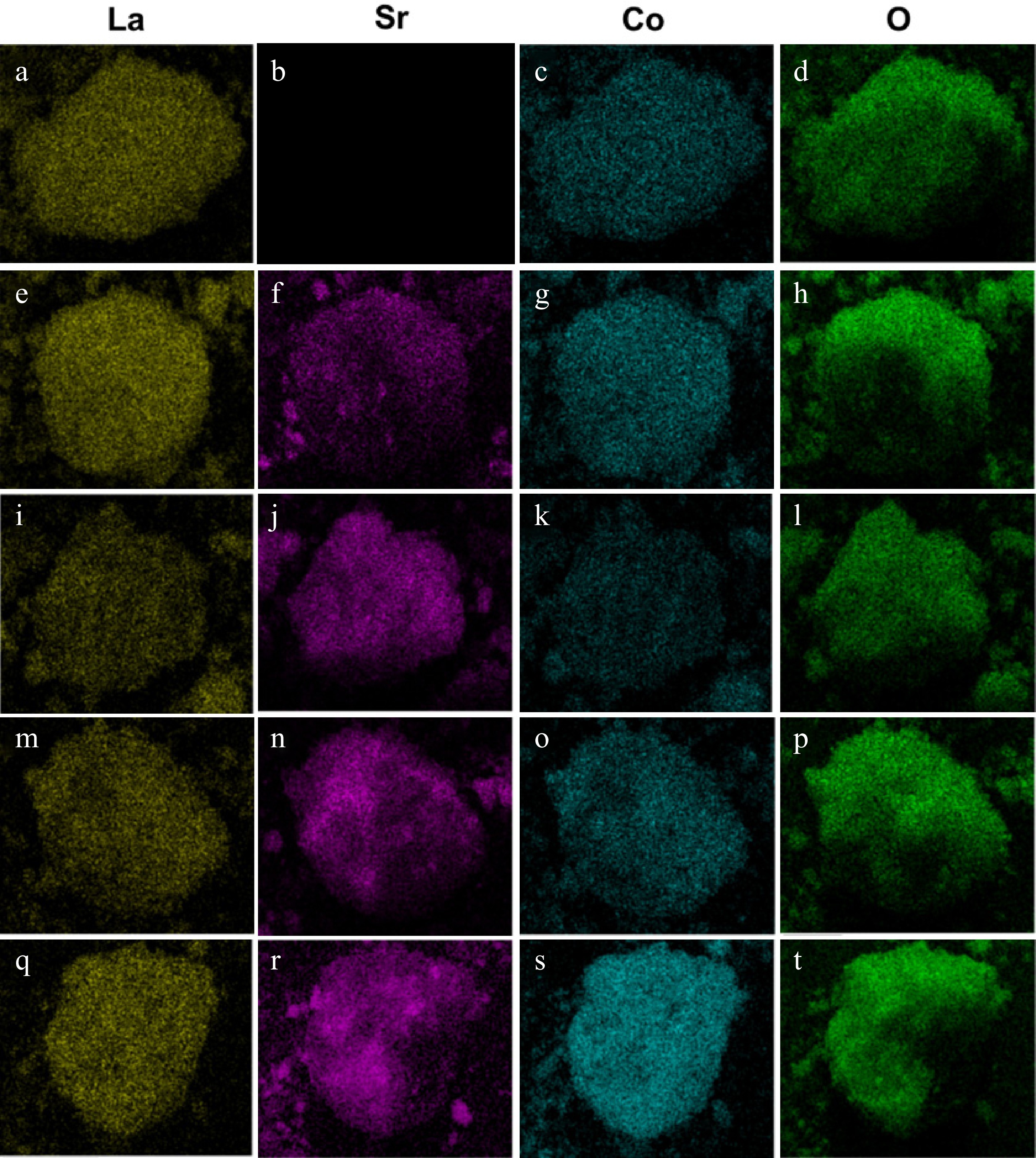
Figure 4.
Elemental distribution of La, Sr, Co and O for (a)–(d) LaCoO3, (e)–(h) La0.8Sr0.2CoO3, (i)–(l) La0.6Sr0.4CoO3, (m)–(p) La0.4Sr0.6CoO3 and (q)–(t) La0.2Sr0.8CoO3.
-
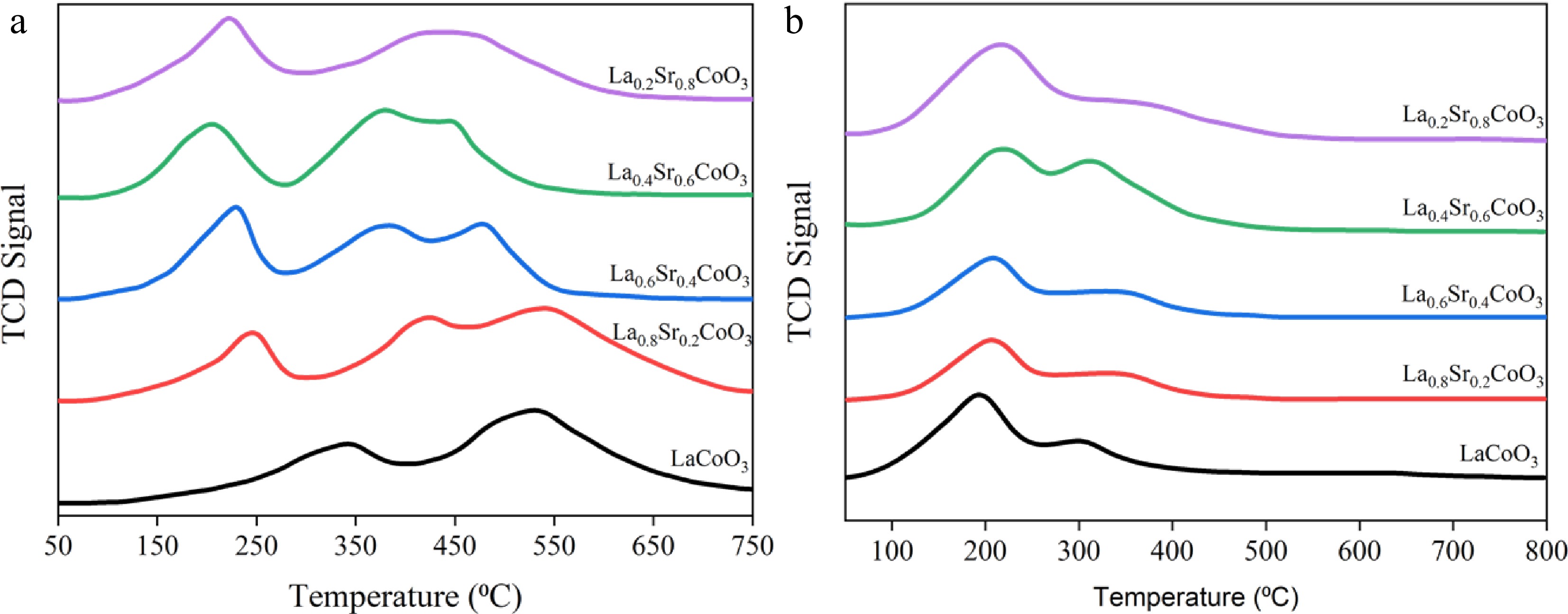
Figure 5.
(a) H2-TPR curve and (b) CO2-TPD image of La(1−x)SrxCoO3 perovskites.
-
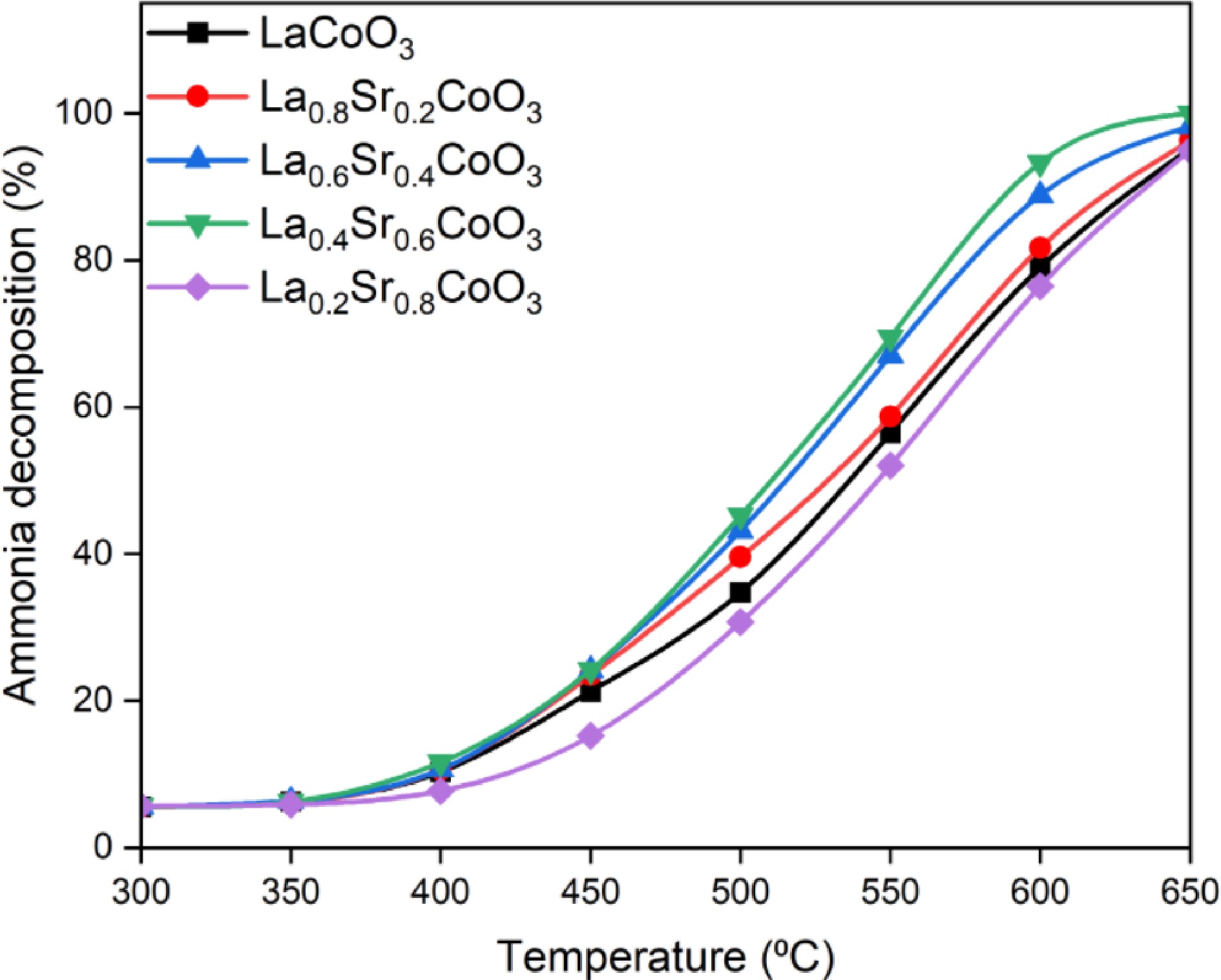
Figure 6.
Ammonia decomposition performance of La(1−x)SrxCoO3 perovskite between 300−650 °C.
-
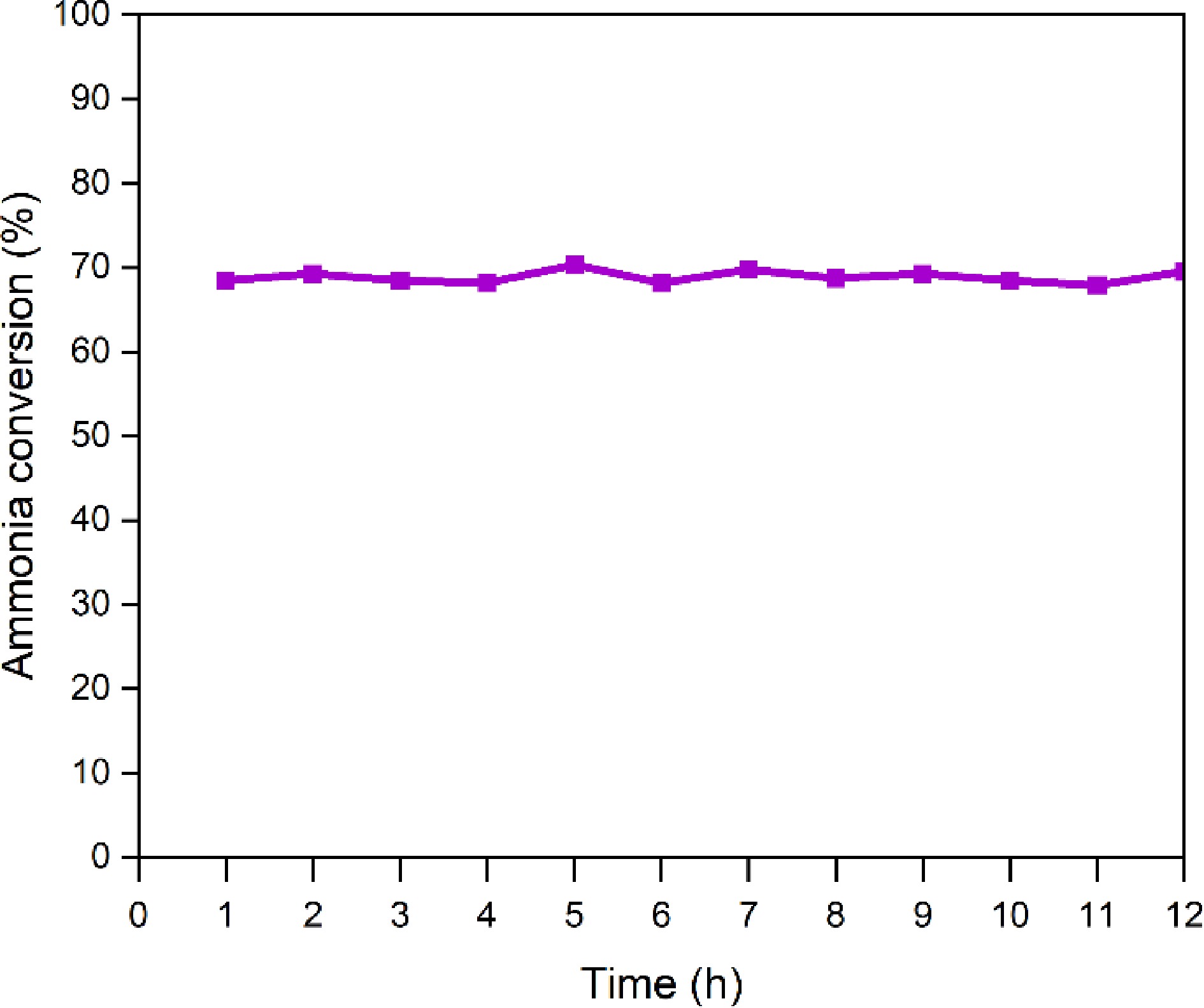
Figure 7.
Ammonia conversion rate of the La0.4Sr0.6CoO3 catalyst during the ammonia catalytic decomposition reaction.
-
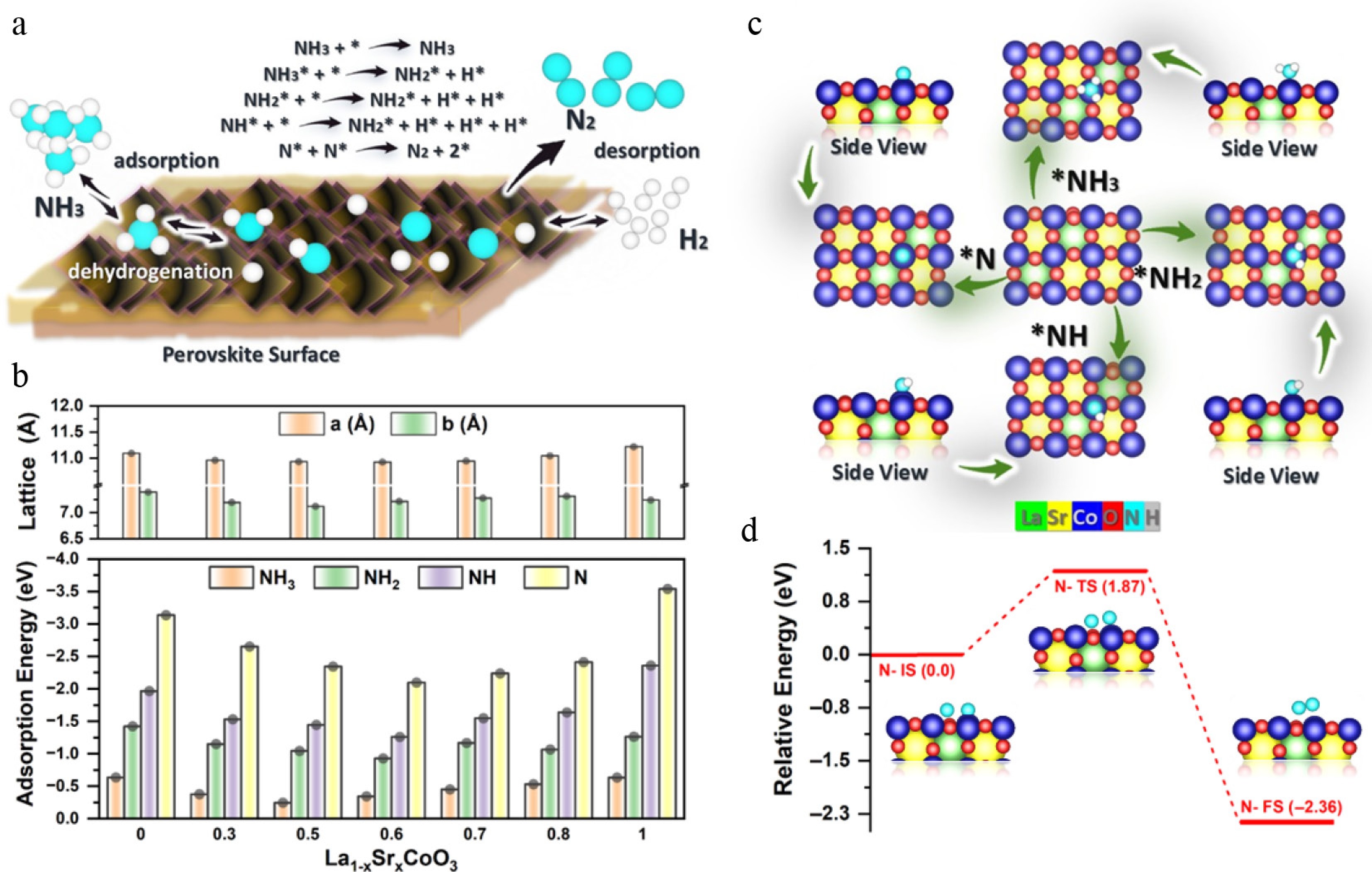
Figure 8.
(a) Pathway of the NH3 decomposition mechanism, with * denoting active sites. (b) Lattice contraction (x ≤ 0.6) to expansion (x > 0.6) and NHx adsorption energies (weakest at x = 0.6). Inset: NH3 on Co (1.82 Å). (c) Optimised NH3/NH2/NH/N structures. (d) N desorption profile: 1.87 eV barrier, exothermic (−2.36 eV) at x = 0.6.
-
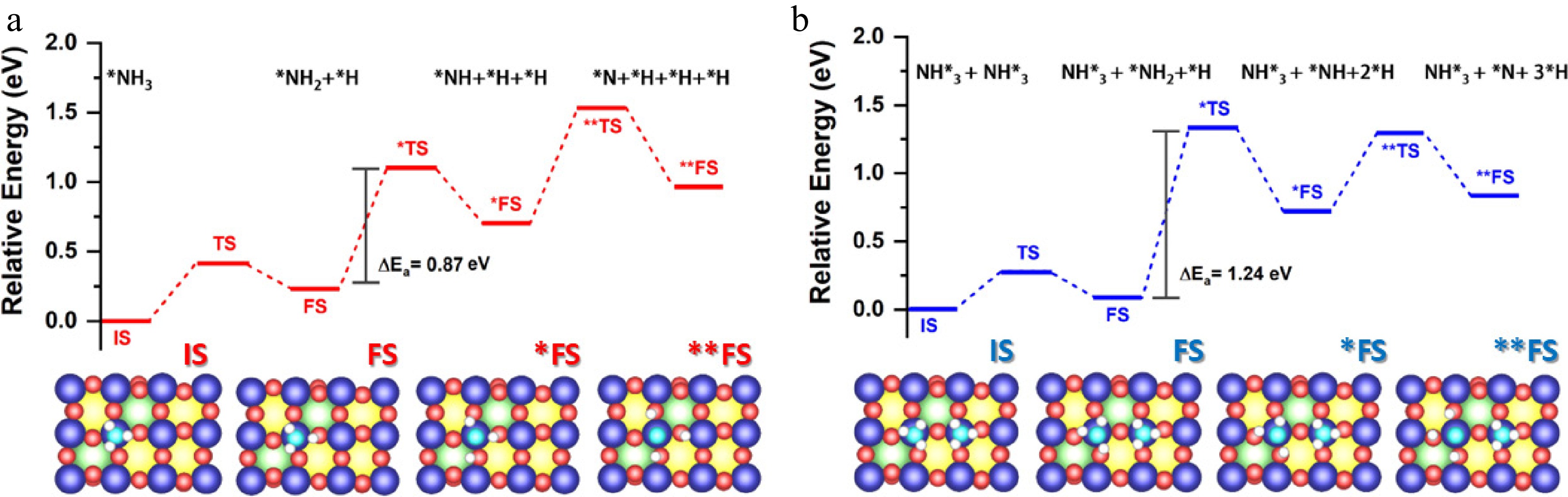
Figure 9.
(a) Mechanism of NH3 decomposition on La0.4Sr0.6CoO3 perovskite catalysts. Reaction pathway showing sequential dehydrogenation steps (NH3* → NH2* → NH* → N*) and N2 desorption, with energy distributions for key intermediates at different Sr doping levels (x = 0–1). (b) Optimised structures (top and side views) illustrating the dimerised decomposition mechanism at the active Co site, with bond lengths of 1.86 Å and relative energies indicated.
-
Catalyst Metallic loading (wt %) GHSV (mL·g−1·h−1) Reaction temperature (°C) Ammonia conversion rate (%) Ref. Ru/N-CNT 7 6,000 400 48.0 [51] Ru/SiO2 10 30,000 500 64.0 [52] Co/γ−Al2O3 5 36,000 500 21.0 [49] Mo/ γ−Al2O3 5 36,000 500 22.4 [49] CoMo/ γ−Al2O3 5 36,000 500 55.0 [49] Fe5Co/CNTs 5 36,000 600 48.0 [55] *HEA-Co45Mo25 8.8 36,000 500 64.5 [50] Co7Mo3/MCM-41 5 36,000 500 51.8 [56] MnN-LiNH2 49.9 13,500 465 75.2 [54] MnN-NaNH2 46.7 13,500 465 32.0 [54] Ni-0.1La/SiO2 − 30,000 500 46.5 [53] Ni-0.1Ce/SiO2 − 30,000 500 40.2 [53] La0.4Sr0.6CoO3 − 37,500 500 43.2 This work *HEA, high-entropy alloy. Table 1.
Performance of different catalysts in thermal catalytic decomposition of NH3.
Figures
(9)
Tables
(1)