-
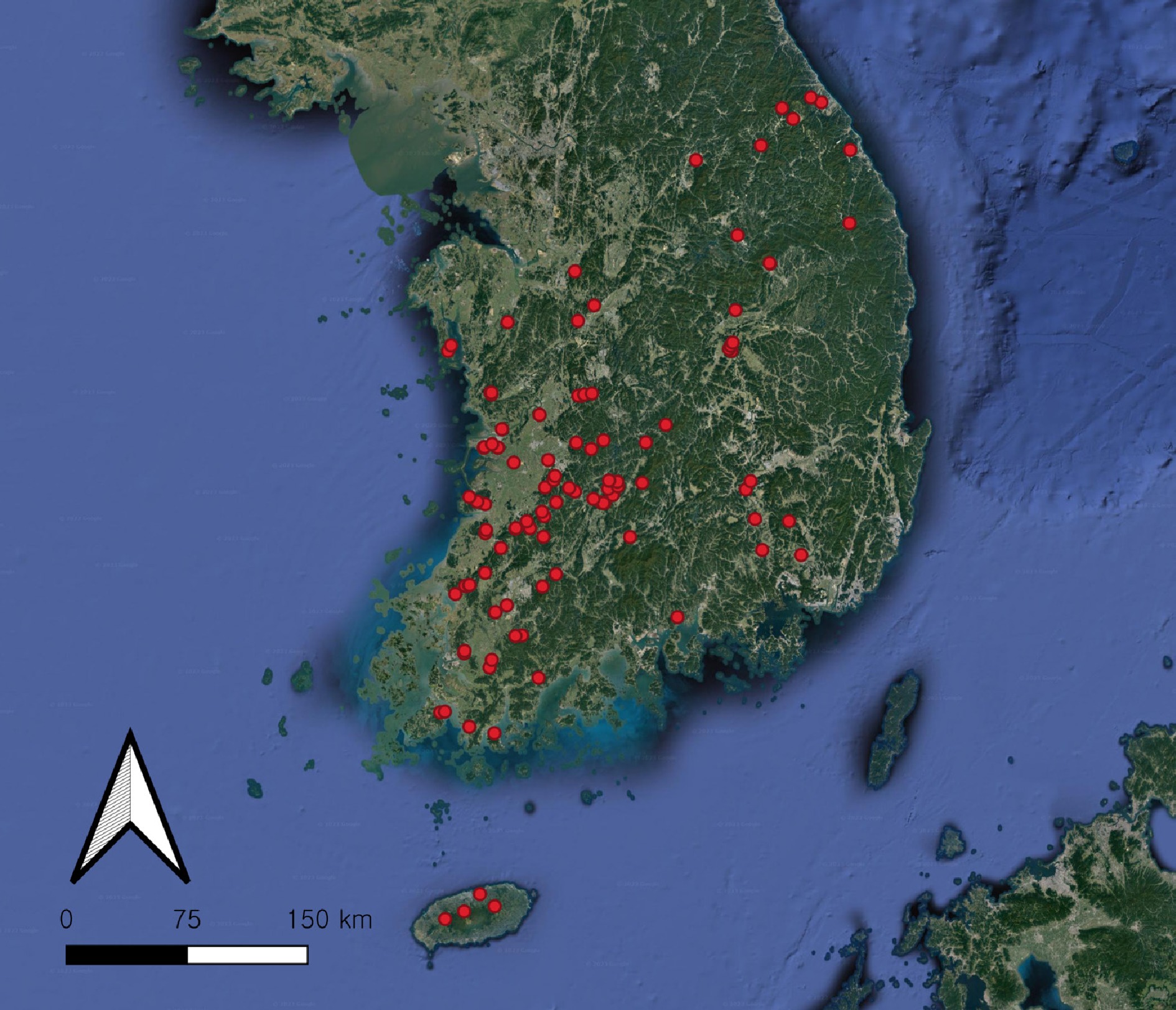
Figure 1.
Location of the 121 sampling sites (red dots) in South Korea. The map was created using QGIS v.3.24.
-
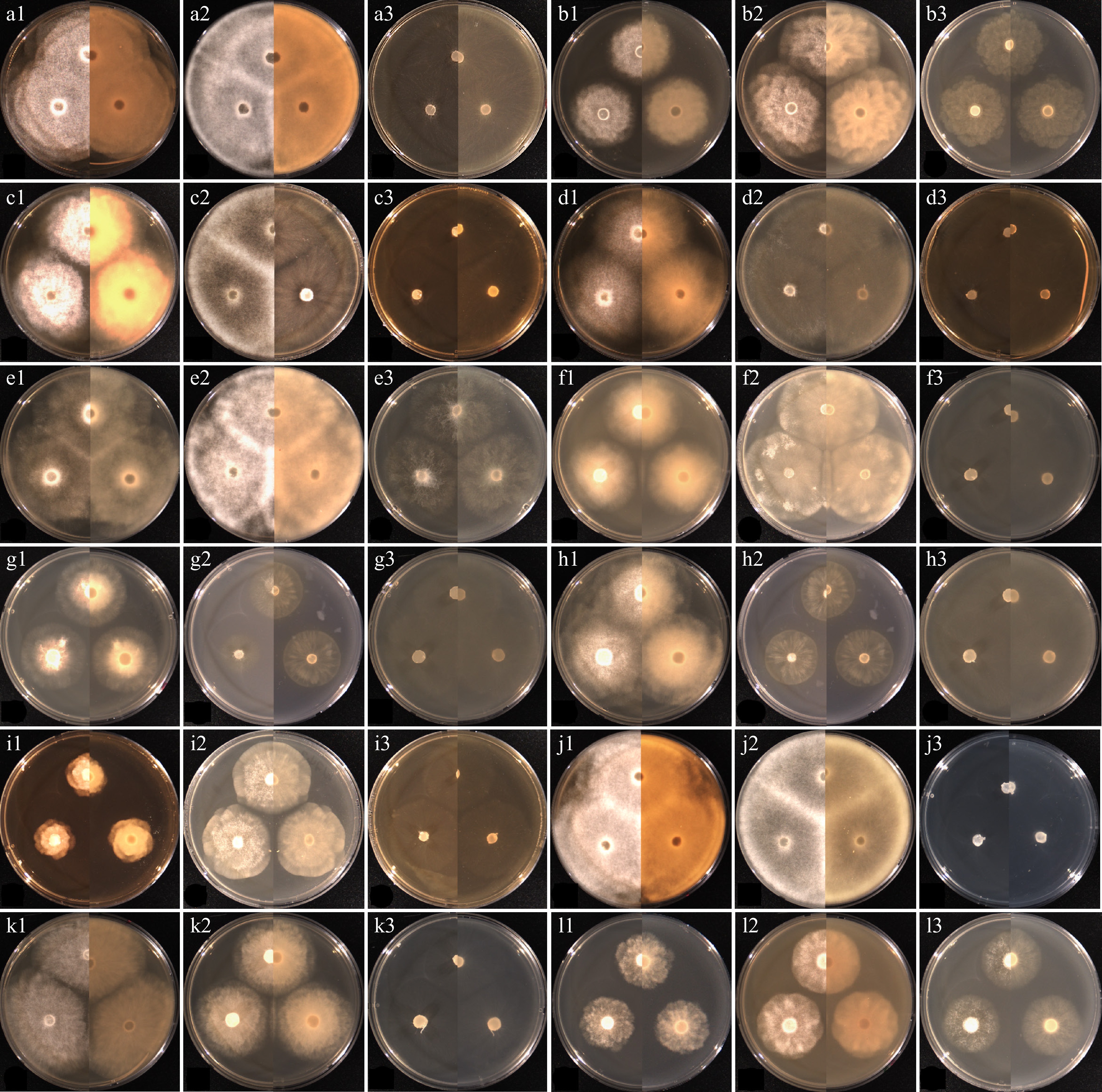
Figure 2.
Cultural characteristics of the isolates of Elongisporangium, Globisporangium, and Phytophthora species, which have previously been unrecorded in Korea, obtained from this study. (a) Elongisporangium undulatum W923, (b) Globisporangium alternatum W1167, (c) G. attrantheridium W786, (d) G. macrosporum W716, (e) G. marsipium W1296, (f) G. nagaii W1534, (g) G. paddicum W1603, (h) G. rostratifingens W1608, (i) G. selbyi W743, (j) G. sylvaticum W769, (k) G. terrestre W2010, and (l) Phytophthora inundata W1757 on (1) PDA, (2) V8A, (3) CMA, after 72 h at 25 °C (left: observed view and right: reverse view).
-
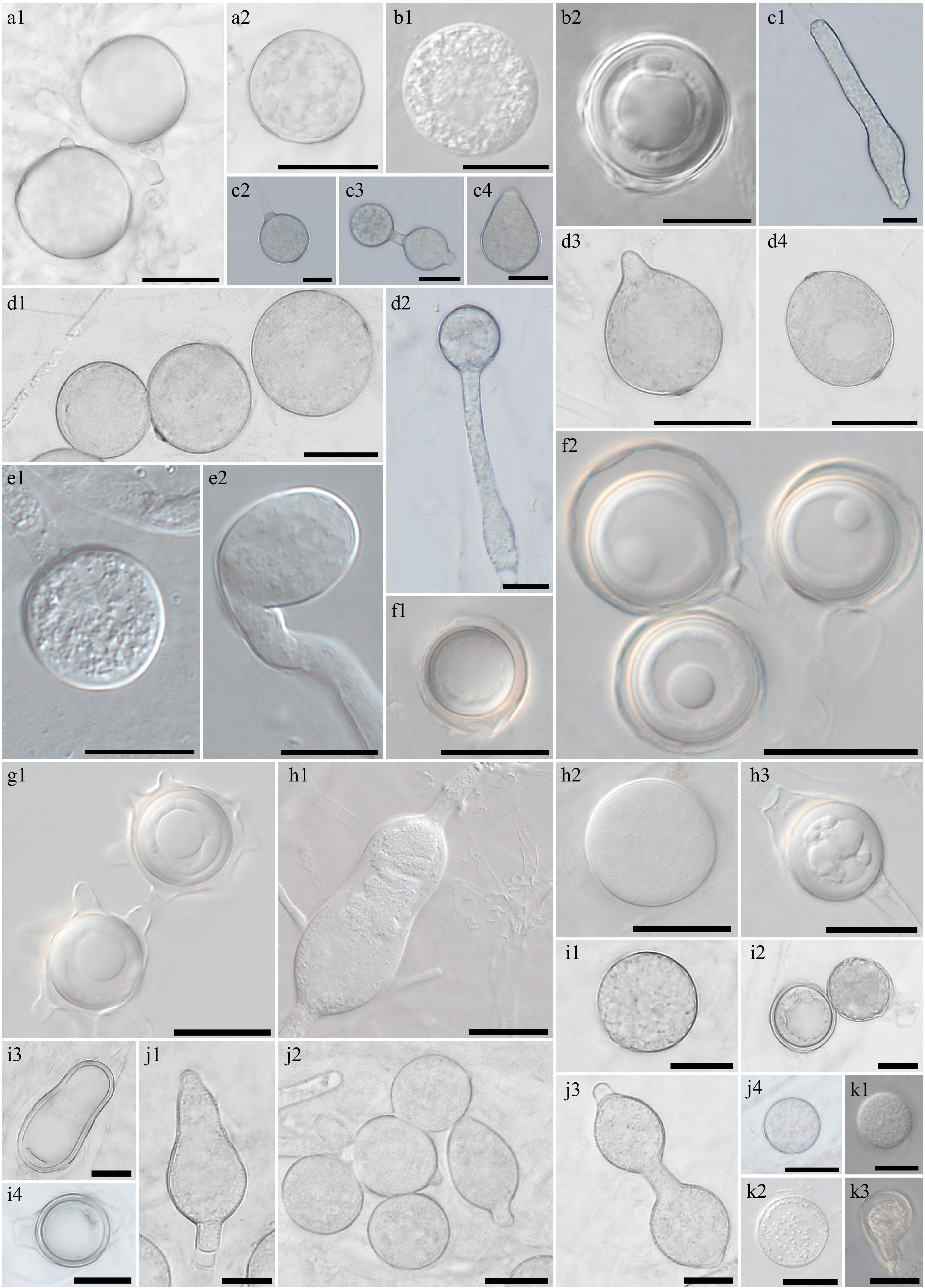
Figure 3.
Morphological characteristics of Elongisporangium, Globisporangium, and Phytophthora species, which have previously been unrecorded in Korea, obtained from this study. Microscopic structures observed under a microscope: (a1), (a2) Chlamydospores of Elongisporangium undulatum W923, (b1) sporangium, (b2) oogonium and oospore of Globisporangium alternatum W1167, (c1), (c3), (c4) hyphal swellings, and (c2) sporangium of (d1)–(d4) G. attrantheridium W786, sporangia of G. macrosporum W716, (e1) sporangium, (e2) hyphal swelling of G. marsipium W1296, (f1), (f2) oogonia and oospores of G. nagaii W1534, (g1) oogonia with ornamentation of G. paddicum W1603, (h1), (h2) sporangia, oogonium and (h3) oospore of G. rostratifingens W1608, (i1) sporangia and (i2)–(i4) oogonia of G. selbyi W743, (j1)–(j4) hyphal swellings of G. sylvaticum W769, (k1), (k2) sporangium, and (k3) oogonium of G. terrestre W2010 (scale bars: a1, a2, d1–k3 = 20 μm, b1–c4 = 10 μm).
-
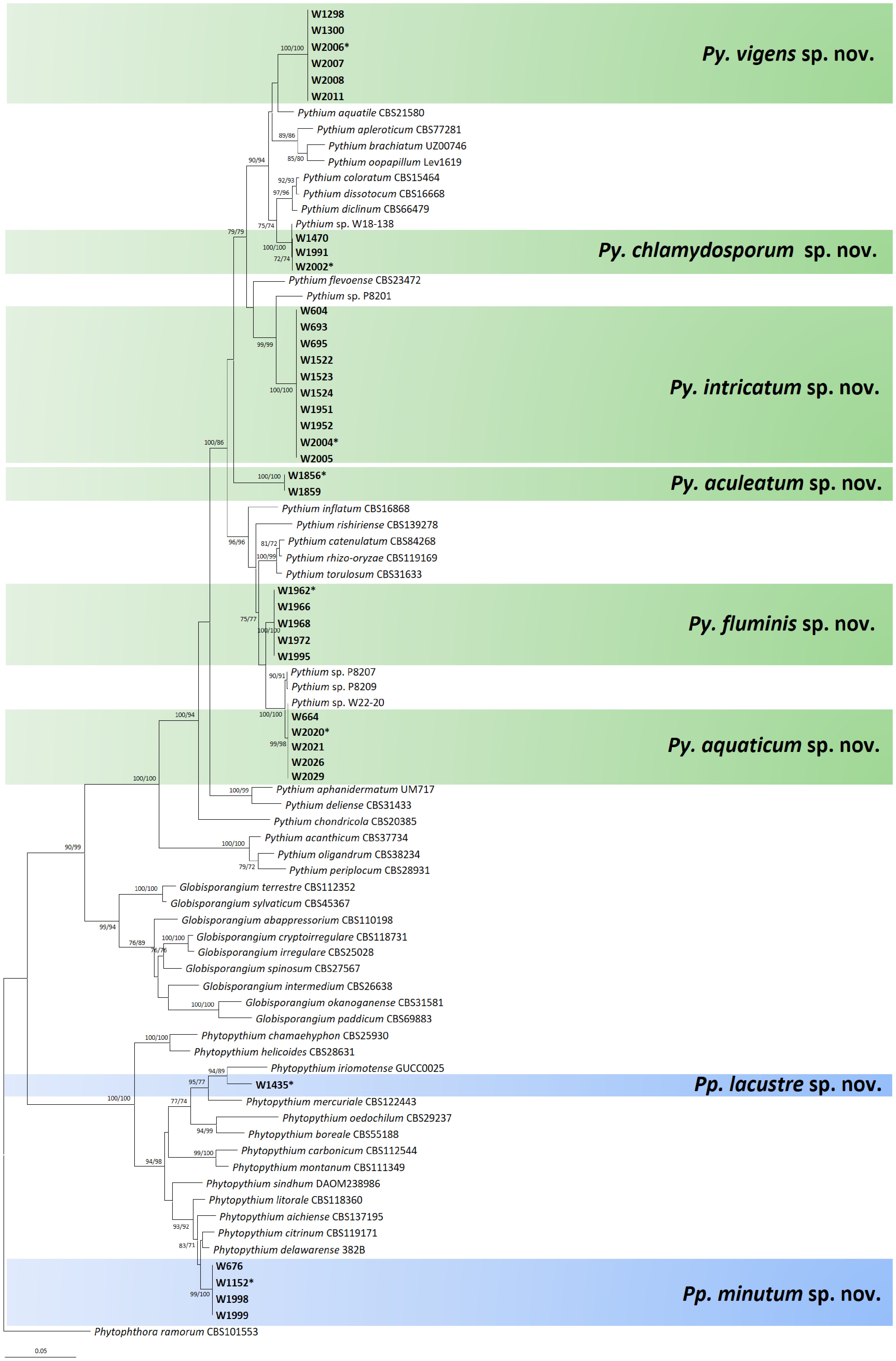
Figure 4.
Multigene phylogenetic tree of Phytopythium and Pythium species from the minimum evolution analysis based on internal transcribed spacer (ITS), cytochrome c oxidase subunit I (cox1), and II (cox2) mtDNA sequences. Bootstrapping values (minimum evolution BP/maximum likelihood BP) higher than 70% were given above or below the branches (1,000 replicates). The strains of new species obtained from this study are shown in bold (ex-type culture with asterisk). Phytophthora ramorum was used as an outgroup. The scale bar equals the number of nucleotide substitutions per site.
-
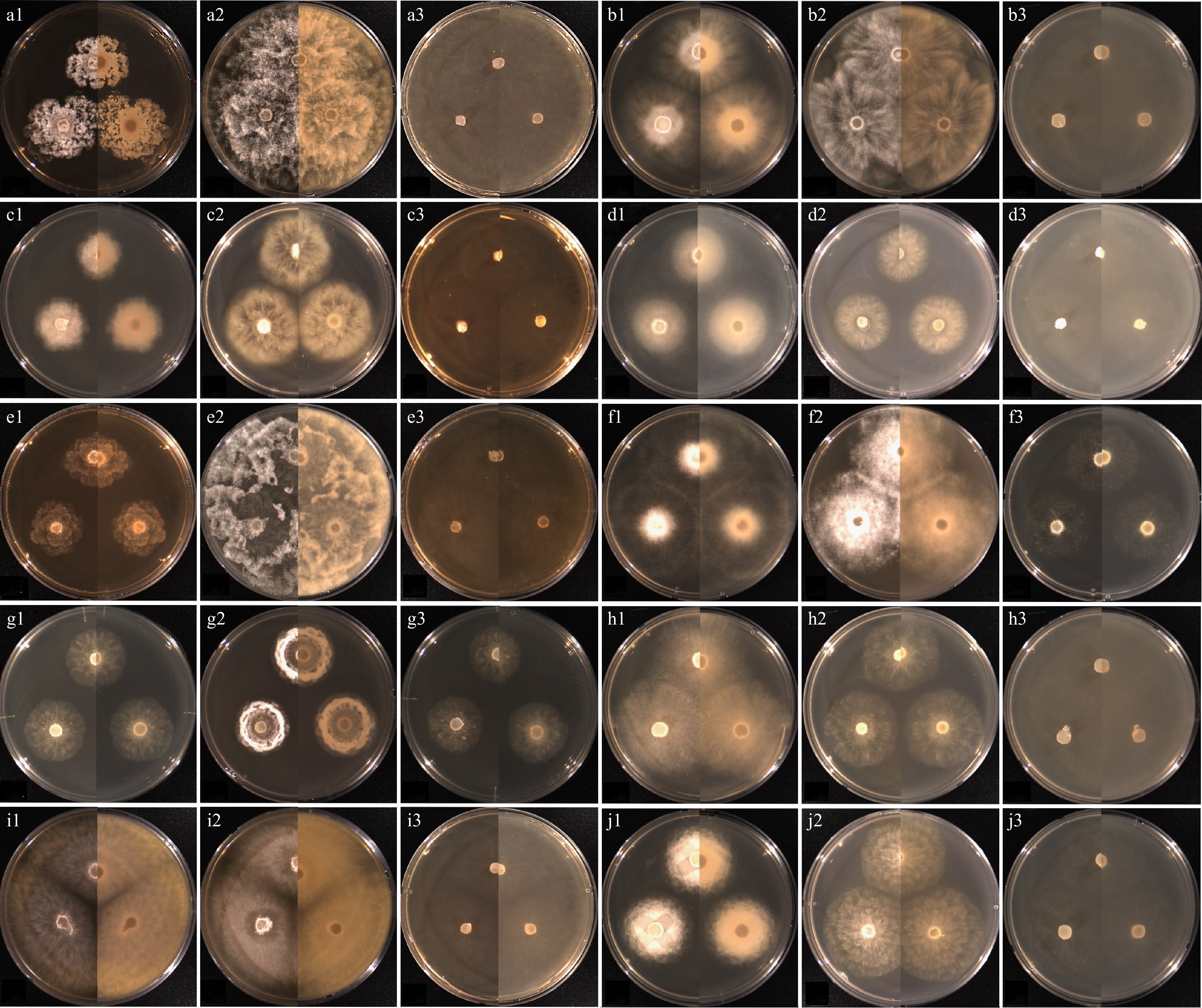
Figure 5.
Cultural characteristics of the isolates of Pythium species, which have previously been unrecorded in Korea, obtained from this study. (a) Pythium acanthicum W927, (b) Py. apleroticum W2032, (c) Py. biforme W813, (d) Py. brachiatum W1541, (e) Py. cedri W754, (f) Py. insidiosum W1285, (g) Py. oryzicollum W1211, (h) Py. pachycaule W1650, (i) Py. subutonaiense W929, and (j) Py. utonaiense W1684 on (1) PDA, (2) V8A, (3) CMA, after 72 h at 25 °C (left: observed view and right: reverse view).
-
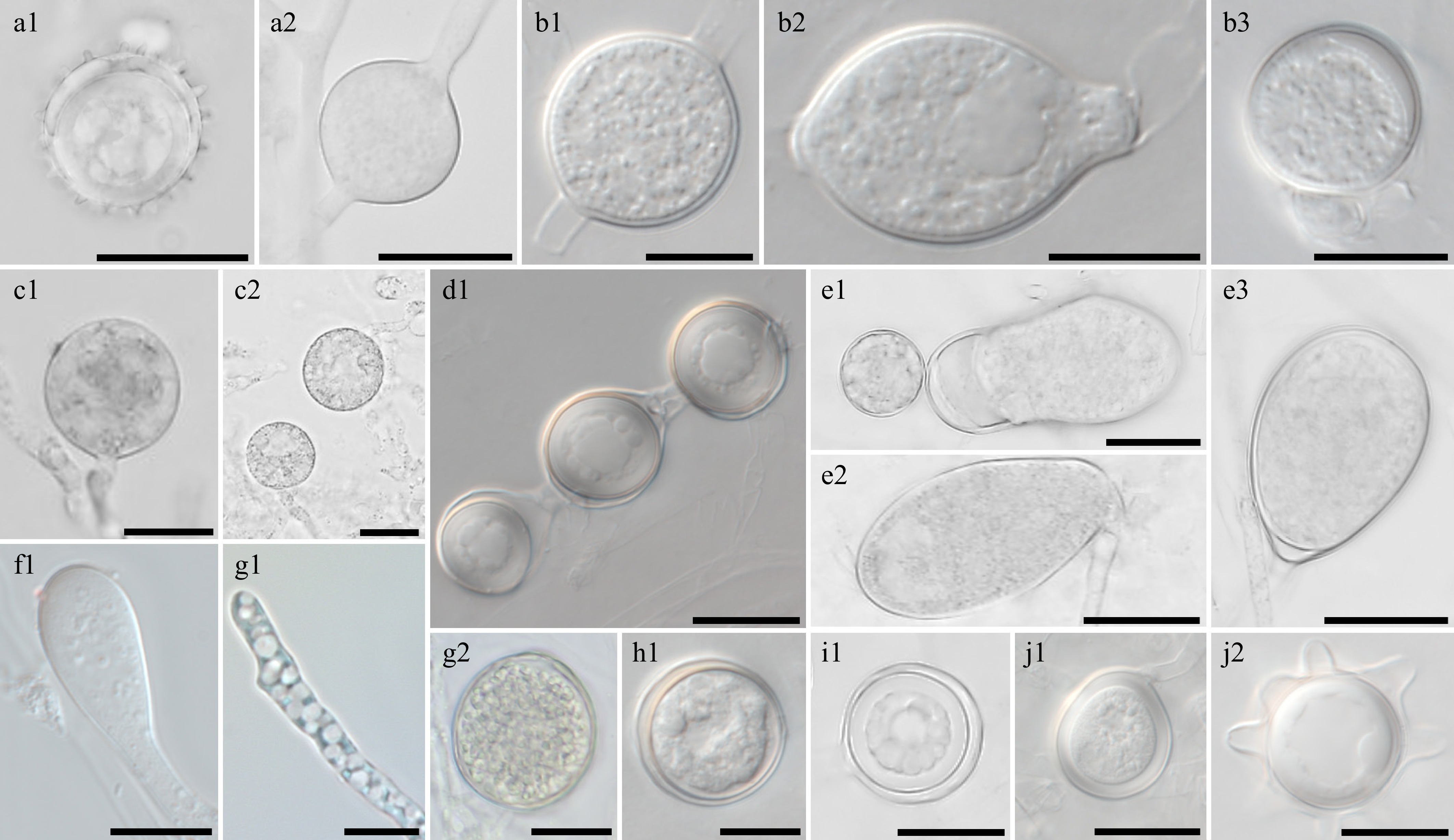
Figure 6.
Morphological characteristics of Pythium species, which have previously been unrecorded in Korea, obtained from this study. Microscopic structures observed under a microscope: (a1) oogonium and (a2) sporangium of Pythium acanthicum W927, (b1), (b3) oogonium and (b2) hyphal swellings of Py. apleroticum W2032, (c1), (c2) hyphal bodies of Py. biforme W813, (d1) oogonia in a chain of Py. brachiatum W1541, (e1)–(e3) sporangia of Py. cedri W754, (f1) Py. insidiosum W1285, (g1) Py. oryzicollum W1211, (g2) oogonium and oospore of Py. oryzicollum W1211, (h1) Py. pachycaule W1650, (i1) Py. subutonaiense W929, and (j1), ( j2) Py. utonaiense W1684 (scale bars: a1, a2, c1–e3, g2, i1–j2 = 20 μm, b1–b3, f1, g1, h1 = 10 μm).
-
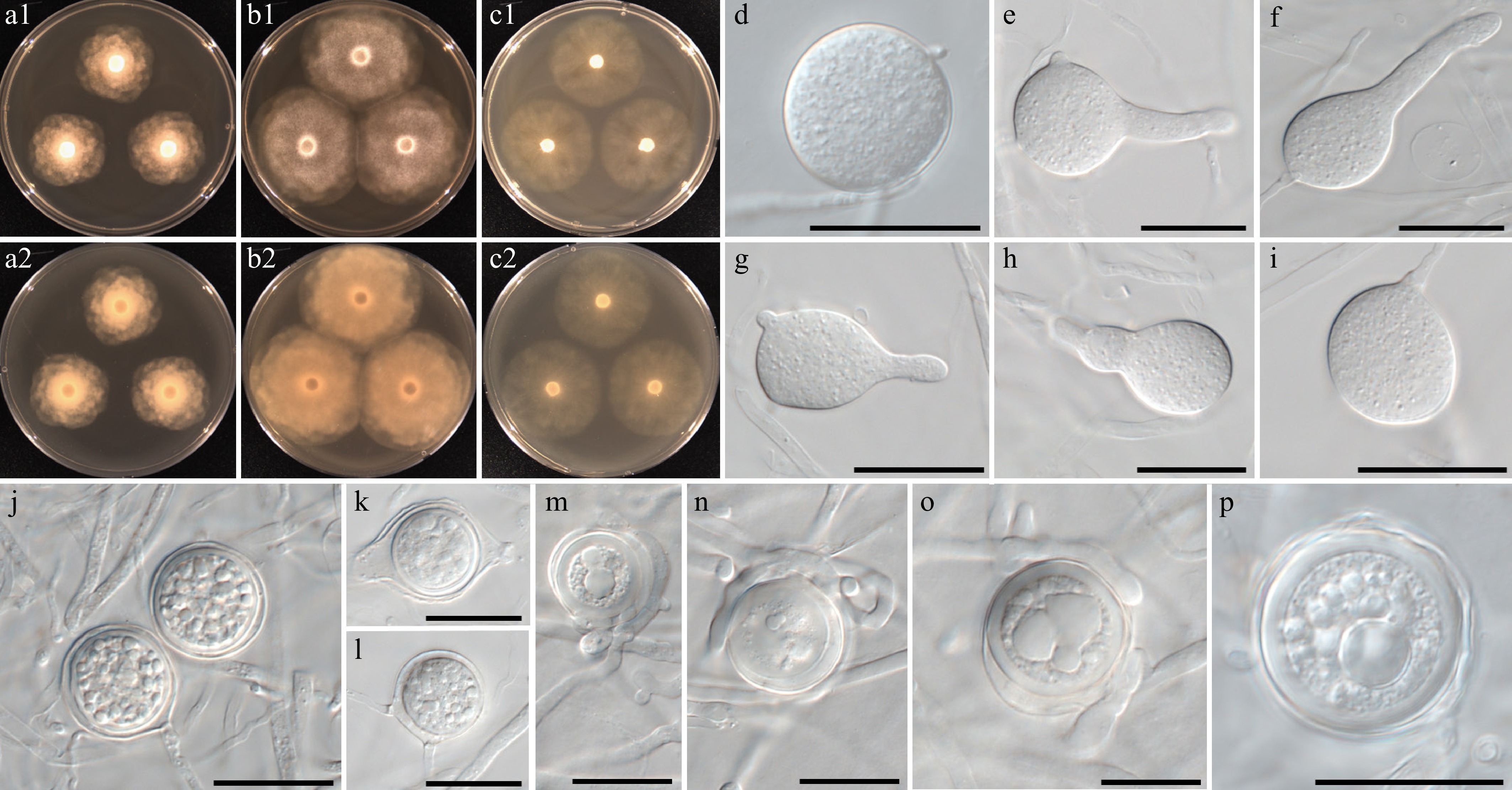
Figure 7.
Colony patterns and morphology of Phytopythium minutum W1152. Cultural characteristics of (a) PDA, (b) V8A, (c) CMA, after 72 h at 25 °C (1: observed view and 2: reverse view). (d)–(p) Microscopic structures observed under a microscope: (d)–(i) (sub-)globose to (ob-)pyriform sporangia with or without discharge tube, (j)–(p) globose oogonia with one or multiple antheridia per oogonium, sometimes encircling the oogonium (scale bars: 20 μm).
-
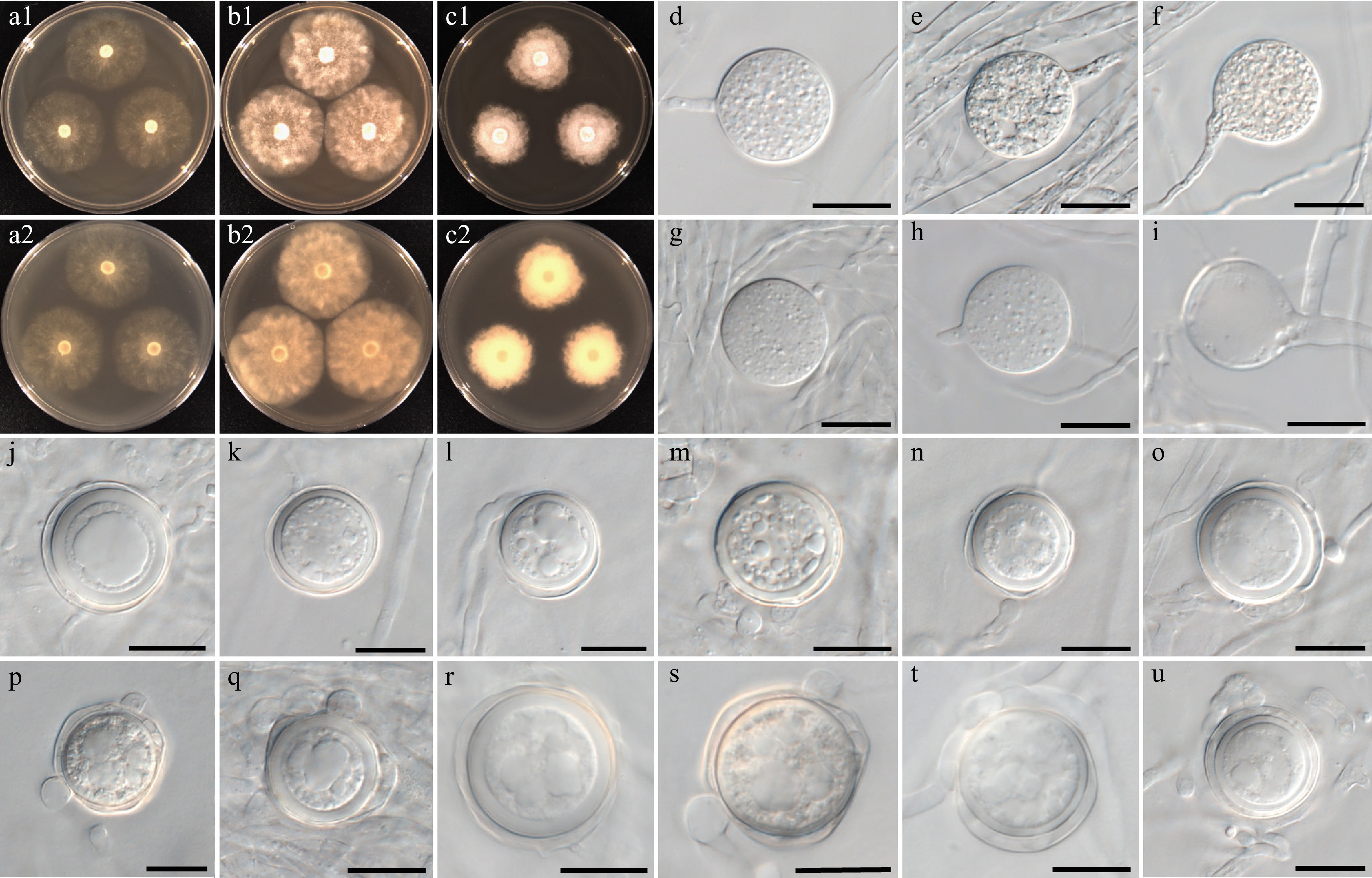
Figure 8.
Colony patterns and morphology of Phytopythium lacustre W1435. Cultural characteristics of (a) PDA, (b) V8A, and (c) CMA, after 72 h at 25 °C (1: observed view and 2: reverse view). (d)–(u) Microscopic structures observed under a microscope: (d)–(i) (sub-)globose sporangia with (h) growing release tube, and (i) empty sporangium, (j)–(u) (sub-)globose oogonia with one to two club-shaped or filamentous inflated antheridia per oogonium (scale bars: 20 μm).
-
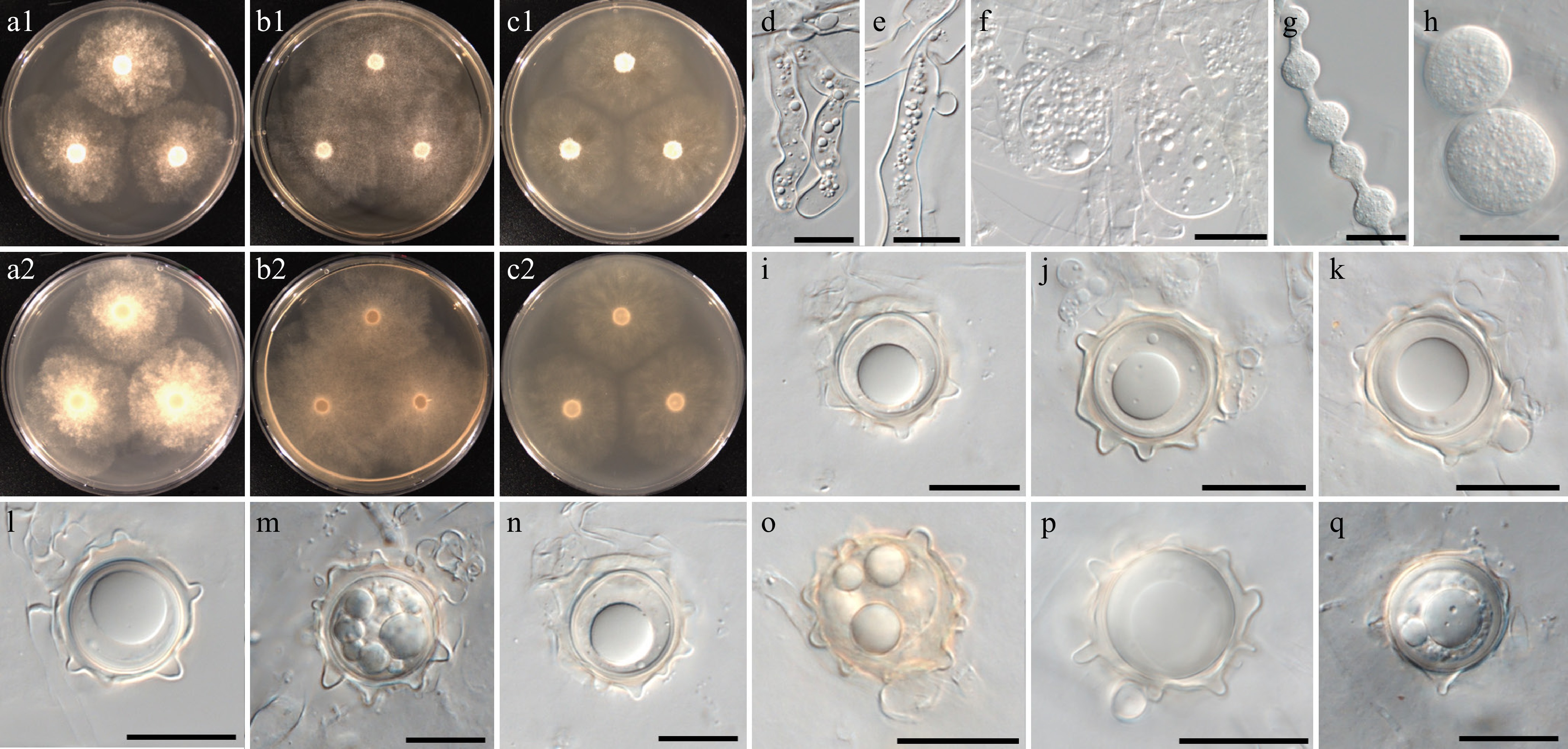
Figure 9.
Colony patterns and morphology of Pythium aculeatum W1856. Cultural characteristics of (a) PDA, (b) V8A, (c) CMA, after 72 h at 25 °C (1: observed view and 2: reverse view). (d)–(q) Microscopic structures observed under a microscope, (d)–(f) inflated filamentous sporangia, (g), (h) hyphal swellings, (i)–(q) globose oogonia with ornamented wall and globose or clavate antheridia (scale bars: d–h = 10 μm, i–q = 20 μm).
-
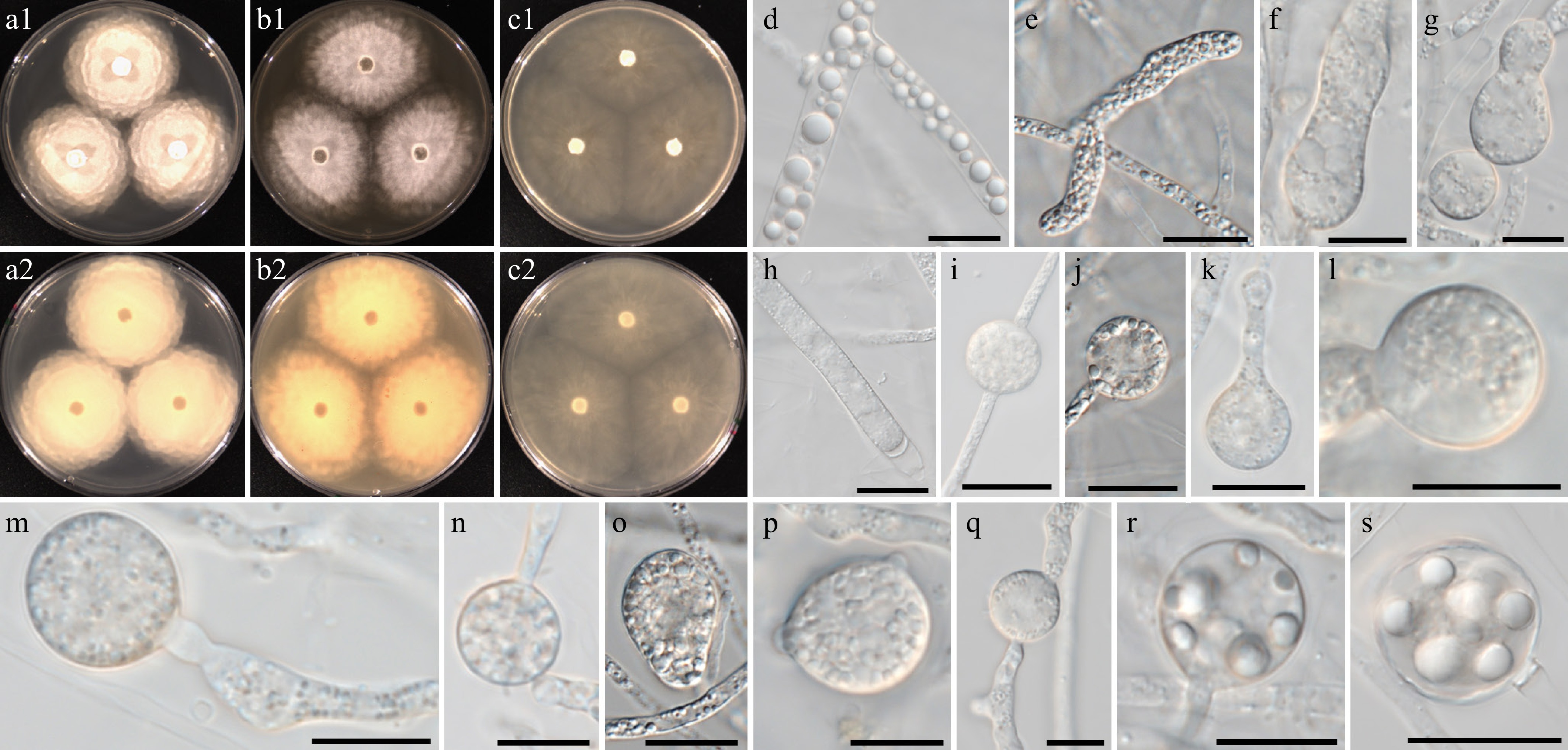
Figure 10.
Colony patterns and morphology of Pythium aquaticum W2020. Cultural characteristics on (a) PDA, (b) V8A, (c) CMA, after 72 h at 25 °C (1: observed view and 2: reverse view). (d)–(s) Microscopic structures observed under a microscope, (d), (r), (s) lipid globules inside the hyphae, (e) filamentous, inflated sporangia, (f)–(q) cylindrical, clavate, (sub-)globose, ovoid or peanut-shaped hyphal swellings (scale bars: 10 μm).
-
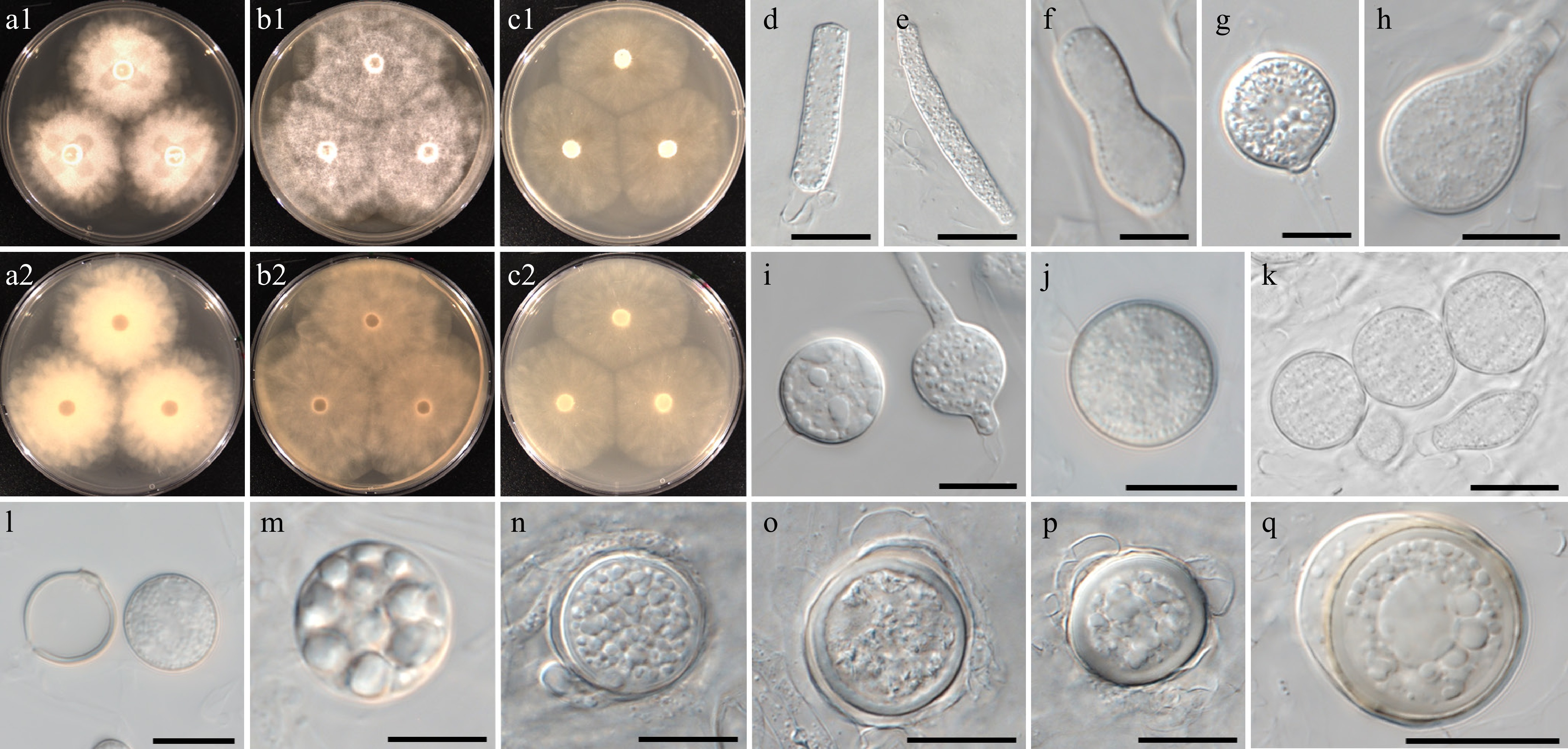
Figure 11.
Colony patterns and morphology of Pythium chlamydosporum W2002. Cultural characteristics of (a) PDA, (b) V8A, (c) CMA, after 72 h at 25 °C (1: observed view and 2: reverse view). (d)–(q) Microscopic structures observed under a microscope: (d), (e) filamentous slightly inflated structures, (f)–(i) cylindrical, clavate, (sub-)globose, ovoid or peanut-shaped hyphal swellings, (j), (k) chlamydospores, (l) empty and mature chlamydospore, (m)–(q) globose oogonia with antheridia (scale bars: 10 μm).
-
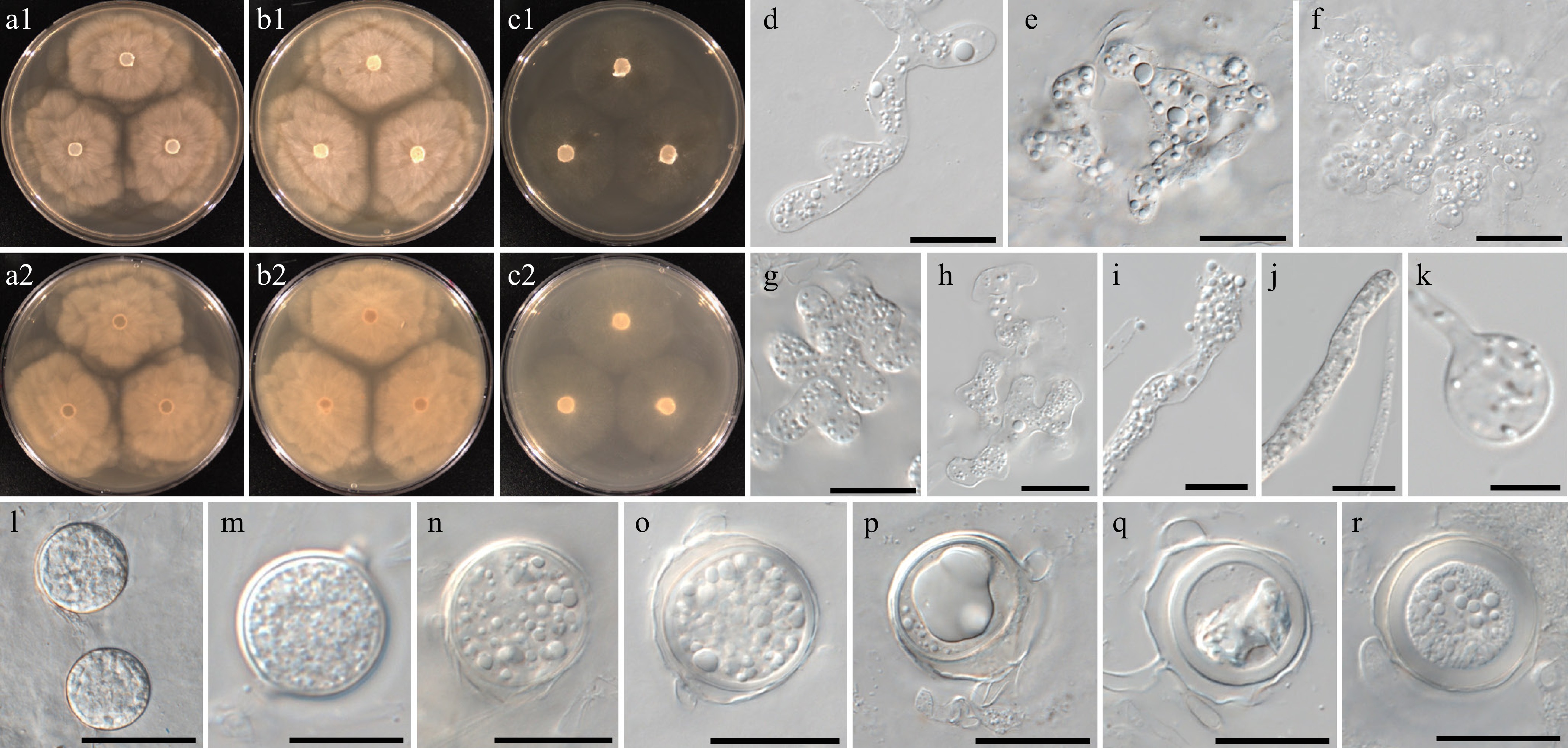
Figure 12.
Colony patterns and morphology of Pythium intricatum W2004. Cultural characteristics of (a) PDA, (b) V8A, (c) CMA, after 72 h at 25 °C (1: observed view and 2: reverse view). (d)–(r) Microscopic structures observed under a microscope: (d) filamentous inflated sporangia, (e)–(h) lobulated sporangia forming toruloid complexes or dendroid structures, (i)–(k) filamentous sporangia and swelling, (l), (m) chlamydospores, (n)–(r) globose oogonia with antheridial cells (scale bars: d–k = 10 μm, l–r = 20 μm).
-
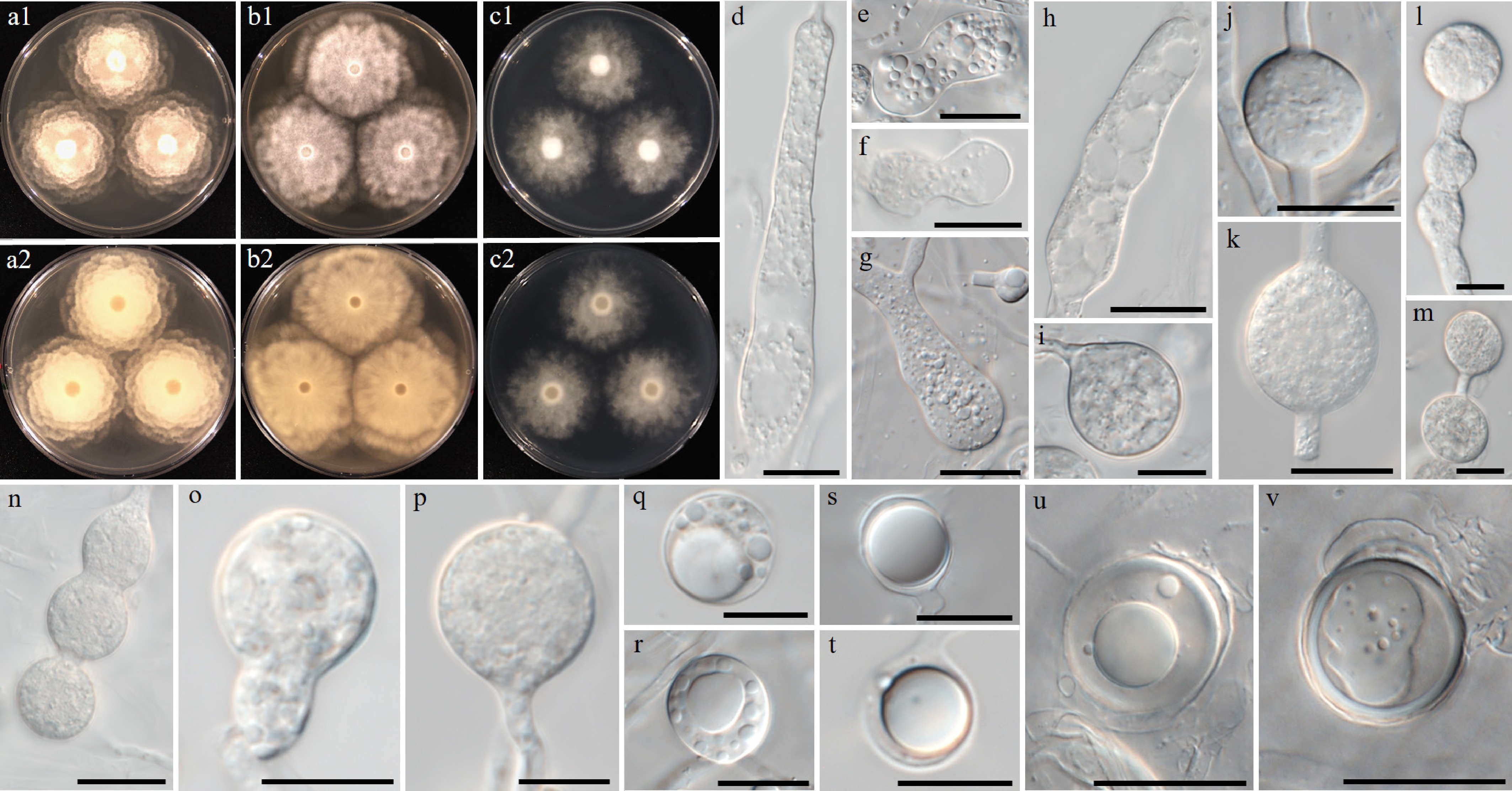
Figure 13.
Colony patterns and morphology of Pythium fluminis W1962. Cultural characteristics of (a) PDA, (b) V8A, (c) CMA, after 72 h at 25 °C (1: observed view and 2: reverse view). (d)–(v) Microscopic structures observed under a microscope: (d) filamentous, inflated sporangium, (e)–(r) clavate, (sub-)globose, ovoid or peanut-shaped, sometimes catenulate hyphal swellings, (s), (t) globose oogonia (u), (v) with antheridia (scale bars: d–p = 10 μm, q–v = 20 μm).
-
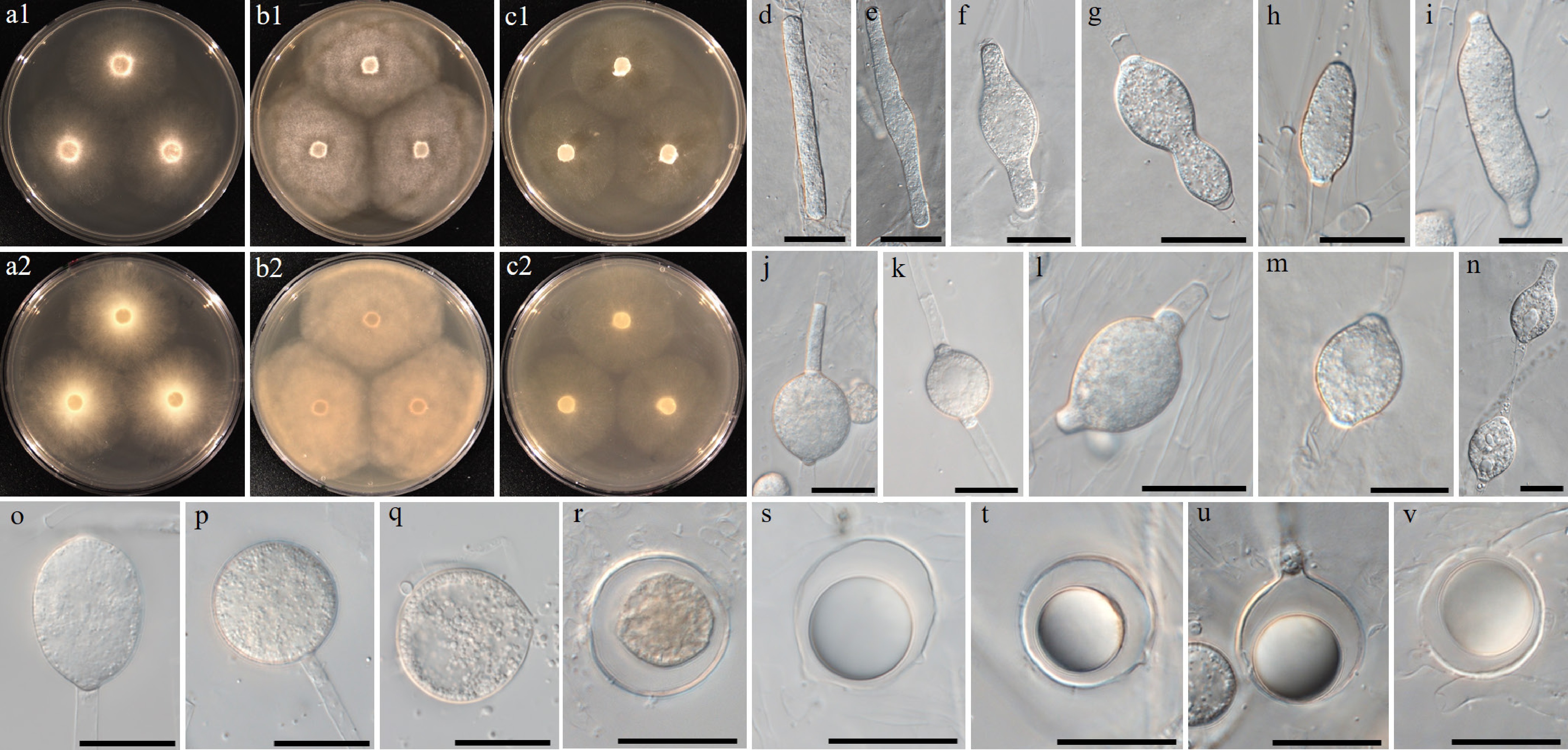
Figure 14.
Colony patterns and morphology of Pythium vigens W2006. Cultural characteristics of (a) PDA, (b) V8A, (c) CMA, after 72 h at 25 °C (1: observed view and 2: reverse view). (d)–(v) Microscopic structures observed under a microscope: (d), (e) filamentous slightly inflated structures, (f)–(q) intercalary, cylindrical, (sub-)globose, ovoid or peanut-shaped hyphal swellings, (n) rarely catenulate, (r)–(v) globose oogonia (scale bars: d–q = 10 μm, r–v = 20 μm).
-
Taxon Substrate Distribution Ref. Order Species Algae Amphibian Crustaceans Fish Mosquito Nematoda Plant Soil Water Stream Reservoir River Myzocytiopsidales Resticularia marchaliana o o Czeczuga et al.[47] Leptomitales Apodachlya brachynema o o Czeczuga et al.[47] Peronosporales Halophytophthora fluviatilis o o o o Yang & Hong[48] Phytophthora ×alni o o o o o Jung & Blaschke[49], Bjelke et al.[50] Phytophthora taxon aquatilis o o Hong et al.[51] Phytophthora bilorbang o o Corcobado et al.[20], Christova[52] Phytophthora cactorum o o o o Jung et al.[53], Jung et al.[19], Reeser et al.[12] Phytophthora ×cambivora o o o o o o Hwang et al.[54], Remigi et al.[55], Reeser et al.[12], Jung et al.[17], Mullett et al.[56] Phytophthora capensis o o o o o Bezuidenhout et al.[57], Jung et al.[16], Bose et al.[58] Phytophthora chlamydospora* o o o o o o Remigi et al.[55], Reeser et al.[12], Jung et al.[59], Hong et al.[51], Jung et al.[10], Jung et al.[17], Hansen et al.[60], Jung et al.[16], Nam et al.[61], Corcobado et al.[20] Phytophthora cinnamomi o o o o o Hwang et al.[54], Hong et al.[51], Jung et al.[62], Stamler et al.[63], Jung et al.[16] Phytophthora citricola o o o o o Hwang et al.[54], Remigi et al.[55], Hulvey et al.[64] Phytophthora citrophthora o o o o o o Hwang et al.[54], Hulvey et al.[64], Hong et al.[51], Jung et al.[65], Jung et al.[66] Phytophthora cryptogea o o Hong et al.[51] Phytophthora drechsleri o o o o o Jung et al.[65], Jung et al.[16] Phytophthora europaea o o o o o Jung et al.[67], Remigi et al.[55], Hwang et al.[54], Reeser et al.[12] Phytophthora gallica o o Corcobado et al.[20] Phytophthora gibbosa o Jung et al.[59] Phytophthora gonapodyides o o o o o o Hwang et al.[54], Jung[68], Remigi et al.[55], Jung et al.[59], Belisario et al.[69], Corcobado et al.[20] Phytophthora gregata o o o o Jung et al.[59] Phytophthora heveae o o o o Erwin & Ribeiro[8], Hwang et al.[54], Jung et al.[19], Jung et al.[16] Phytophthora honggalleglyana o o Christova[52] Phytophthora hydropathica o o o o o Hulvey et al.[64], Hong et al.[70], Corcobado et al.[20] Phytophthora inundata* o o o o o o Brasier et al.[71], Jung et al.[59], Hüberli et al.[13], Christova[52] Phytophthora irrigata o o o o o Hong et al.[72], Hulvey et al.[64] Phytophthora lacustris o o Corcobado et al.[20], Christova[52] Phytophthora sp. 'lagoariana'* o o o Wallace[73], Nam et al.[61] Phytophthora litoralis o o o o o Hulvey et al.[64], Jung et al.[59] Phytophthora macrochlamydospora o o o o Jung et al.[16] Phytophthora megasperma o o o o o o Erwin & Ribeiro[8], Remigi et al.[55], Jung et al.[59], Reeser et al.[12], Jung et al.[65], Jung et al.[17] Phytophthora mississippiae o o o Hong et al.[51] Phytophthora nemorosa o o o o Reeser et al.[12] Phytophthora nicotianae o o o o Hulvey et al.[64] Phytophthora pini o o o o Reeser et al.[12], Jung et al.[65], Jung et al.[66], Milenković et al.[74] Phytophthora plurivora o o o o o Jung & Burgess[75], Reeser et al.[12], Jung et al.[10], Jung et al.[17], Jung et al.[18], Jung et al.[19], Jung et al.[65], Jung et al.[66] Phytophthora pseudosyringae o o o o o Hwang et al.[54], Jung[68], Reeser et al.[12], Jung et al.[10], Jung et al.[65] Phytophthora ramorum o o o o Werres et al.[76], Reeser et al.[12], Hong et al.[51], Eyre et al.[77], Jung et al.[16] Phytophthora riparia o o o Hong et al.[51], Corcobado et al.[20] Phytophthora siskiyouensis o o o o Reeser er al.[12], Reeser et al.[78] Phytophthora syringae o o o o o o Erwin & Ribeiro[8], Remigi et al.[55], Hulvey et al.[64], Reeser et al.[12], Hong et al.[51], Jung et al.[65], Corcobado et al.[20] Phytophthora thermophila o o o o o Jung et al.[17], Jung et al.[59] Phytophthora vesicula o o Nakagiri[26] Pythiales Elongisporangium anandrum o o Christova[52] Elongisporangium helicandrum o o Czeczuga et al.[47] Elongisporangium undulatum* o o o o o o Khulbe[79], Abdelzaher et al.[80], Czeczuga & Snarka[81] Globisporangium alternatum* o o Rahman et al.[82] Globisporangium attrantheridium* o o o o Nelson & Karp[83] Globisporangium carolinianum o o o Abdelzaher et al.[80], Czeczuga & Snarka[81] Globisporangium debaryanum o Poitras[84] Globisporangium echinulatum o o o o o o Robertson[85], Khulbe[79], Czeczuga[86], Czeczuga & Snarka[81] Globisporangium heterothallicum* o o Nam & Choi[87] Globisporangium intermedium* o o o o o o El-Hissy et al.[88], Czeczuga[86], Czeczuga & Snarka[81], Czeczuga & Muszynska[89], Nam & Choi[87] Globisporangium irregulare* o o o o Poitras[84], Abdelzaher et al.[80], Czeczuga & Snarka[81] Globisporangium lucens o o Czeczuga & Snarka[81] Globisporangium macrosporum* o o In the present study Globisporangium marsipium* o o o o Czeczuga[86], Czeczuga & Muszynska[89], Matsiakh et al.[90] Globisporangium megalacanthum o o o o Czeczuga & Snarka[81], Czeczuga & Muszynska[89] Globisporangium middletonii o o o o Karling[91], Czeczuga & Snarka[81] Globisporangium multisporum o o Poitras[84] Globisporangium nagaii* o o Vetukuri et al.[92] Globisporangium paddicum* o o In the present study Globisporangium pleroticum o o Abdelzaher et al.[80], Czeczuga & Muszynska[89] Globisporangium pulchrum o o o Poitras[84], Khulbe[79] Globisporangium rostratifingens* o o o o In the present study Globisporangium rostratum o o o o o Czeczuga[86], Czeczuga & Snarka[81], Czeczuga & Muszynska[89] Globisporangium selbyi* o o In the present study Globisporangium spinosum* o o o o o o Khulbe[79], Abdelzaher et al.[80], Czeczuga & Snarka[81] Globisporangium splendens o o Poitras[84] Globisporangium sylvaticum* o o o o o o o Abdelzaher et al.[80], Czeczuga[86], Czeczuga & Muszynska[89], Matsiakh et al.[90] Globisporangium terrestre * o o Matsiakh et al.[90] Globisporangium ultimum* o o o o o Poitras[84], El-Hissy et al.[88], Czeczuga[86], Czeczuga & Snarka[81], Czeczuga & Muszynska[89], Nam & Choi[87] Lagenidium giganteum o o Czeczuga et al.[47] Lagenidium microsporum o o Czeczuga et al.[47] Lagenidium oophilum o o Czeczuga et al.[47] Myzocytium megastomum o o Czeczuga et al.[47] Myzocytium rabenhorstii o o Czeczuga et al.[47] Myzocytium zoophthorum o o Czeczuga et al.[47] Phytopythium chamaehyphon* o o Nam & Choi[87] Phytopythium citrinum o o Christova[52] Phytopythium indigoferae o o Czeczuga et al.[47] Phytopythium iriomotense o o Baten et al.[93] Phytopythium litorale* o o o o o Nam & Choi[87], Vetukuri et al.[92], Christova[52] Phytopythium palingenes o o El-Hissy et al.[88], Czeczuga et al.[47] Phytopythium vexans* o o o o Poitras[84], Khulbe[79], Nam & Choi[87] Pythium acanthicum* o o o o Robertson[85], Czeczuga & Snarska[81] Pythium adhaerens o o o Sparrow[94], Abdelzaher & Kageyama[95] Pythium afertile o o o o Robertson[85], Czeczuga & Snarka[81] Pythium amaminum o o o Kikuchi et al.[96] Pythium angustatum o o o Matsiakh et al.[90], Christova[52] Pythium aphanidermatum* o o o o o o Poitras[84], Khulbe[79], Al-Sheikh & Abdelzaher[97], Lodhi et al.[98] Pythium apleroticum* o o In the present study Pythium aquatile* o o o o Czeczuga & Snarka[81], Czeczuga & Muszynska[89], Matsiakh et al.[90], Abdelzaher & Kageyama[95] Pythium aristosporum o o o o o Czeczuga & Snarka[81], Czeczuga[86], Czeczuga & Muszynska[89] Pythium arrhenomanes o o Czeczuga & Muszynska[89] Pythium biforme* o o o Uzuhashi et al.[99] Pythium brachiatum* o o Uzuhashi et al.[99] Pythium cactacearum o o Czeczuga et al.[47] Pythium catenulatum* o o o o Poitras[84], Abdelzaher et al.[80], Czeczuga & Snarka[81], Rahman & Sarowar[100] Pythium cedri* o o In the present study Pythium coloratum o o Robertson[85] Pythium debaryanum o o o o o o Robertson[85], Khulbe[79], Czeczuga et al.[47] Pythium deliense o o Abdelzaher et al.[80] Pythium diclinum* o o o o o Abdelzaher et al.[80], Al-Sheikh & Abdelzaher[97], Nam & Choi[87], Abdelzaher & Kageyama[95] Pythium dissimile o o Czeczuga & Snarka[81] Pythium dissotocum* o o o o o Poitras[84], Abdelzaher et al.[80], Czeczuga & Snarka[81], Al-Sheikh & Abdelzaher[97] Pythium elongatum o o Czeczuga et al.[47] Pythium gibbosum o o Czeczuga et al.[47] Pythium globosum o o Czeczuga et al.[47] Pythium gracile o o o Poitras[84], Czeczuga et al.[47] Pythium graminicola o o Czeczuga et al.[47] Pythium hydnosporum o o o o o Czeczuga et al.[47], Czeczuga & Snarka[81], Czeczuga & Muszynska[89] Pythium imperfectum o o Czeczuga et al.[47] Pythium inflatum o o o o o o o Poitras[84], Robertson[85], Czeczuga[86], Czeczuga & Snarka[81], Nam & Choi[87] Pythium insidiosum* o o Presser & Goss[101] Pythium jirovecii o o Czeczuga et al.[47] Pythium junctum o o Uzuhashi et al.[99] Pythium monospermum* o o o o o Poitras[84], Abdelzaher et al.[80], Czeczuga & Snarka[81], Matsiakh et al.[90] Pythium myriotylum o o o Czeczuga & Snarka[81] Pythium oligandrum o o Czeczuga et al.[47] Pythium oopapillum* o o o Matsiakh et al.[90], Nam & Choi[87] Pythium oryzicollum* o o o o Salmaninezhad & Mostowfizadeh-Ghalamfarsa[102] Pythium pachycaule* o o o o Czeczuga et al.[103], Abdelzaher & Kageyama[95] Pythium papillatum o o Czeczuga et al.[47] Pythium periilum o o Czeczuga et al.[47] Pythium periplocum* o o Czeczuga & Muszynska[89] Pythium phragmitis o o Nechwatal et al.[104] Pythium podbielkowskii o o Czeczuga et al.[47] Pythium polysporum o o Czeczuga et al.[47] Pythium pyrilobum o o Czeczuga & Snarka[81] Pythium rhizo-oryzae* o o o Uzuhashi et al.[99] Pythium rishiriense o o Rahman et al.[82] Pythium subutonaiense* o o Chen & Zheng[105] Pythium sukuiense o o Matsiakh et al.[90] Pythium tardicrescens o o Czeczuga et al.[47] Pythium tenue o o o Poitras[84], Czeczuga et al.[47] Pythium thalassium o o El-Hissy et al.[88] Pythium torulosum* o o o o Poitras[84], Czeczuga & Snarka[81], Czeczuga[86], Abdelzaher & Kageyama[95] Pythium utonaiense* o o Uzuhashi et al.[99] Pythium vanterpoolii o o Czeczuga et al.[47] Saprolegniales Achlya ambisexualis o o o o Czeczuga et al.[47], Robideau et al.[106], Hunjavanit et al.[107],
Vega-Ramírez et al.[108]Achlya americana o o o Ogbonna & Alabi[109], Czeczuga et al.[47], Robideau et al.[106], Godlewska et al.[110], Choi et al.[111] Achlya apiculata o o o Ogbonna & Alabi[109], Czeczuga et al.[47], Godlewska et al.[110] Achlya bisexualis* o o o o Leclerc et al.[112], Robideau et al.[106], Choi et al.[111] Achlya caroliniana o o o o o Srivastava & Srivastava[113], Ogbonna & Alabi[109], Czeczuga et al.[47] Achlya colorata o o Czeczuga et al.[47] Achlya crenulata o o Czeczuga et al.[47] Achlya debaryana o o o Czeczuga et al.[47] Achlya diffusa o o Srivastava & Srivastava[113], Ogbonna & Alabi[109], Czeczuga et al.[47] Achlya dubia o o o Shanor & Saslow[114], Ogbonna & Alabi[109], Czeczuga et al.[47] Achlya flagellata o o Hoshina et al.[115], Shah et al.[116], El-Hissy et al.[88], Ogbonna & Alabi[109] Achlya heterosexualis o o Vega-Ramírez et al.[108] Achlya klebsiana o o Ogbonna & Alabi[109], Czeczuga et al.[47], Godlewska et al.[110] Achlya megasperma o o Ogbonna & Alabi[109] Achlya oblongata o o o Ogbonna & Alabi[109], Czeczuga et al.[47], Godlewska et al.[110] Achlya oligocantha o o Czeczuga et al.[47] Achlya orion o o o o Ogbonna & Alabi[109], Czeczuga et al.[47] Achlya polyandra o o Czeczuga et al.[47], Godlewska et al.[110] Achlya prolifera o o o o Ogbonna & Alabi[109], Czeczuga et al.[47], Godlewska et al.[110] Achlya proliferoides o o o Ogbonna & Alabi[109], Czeczuga et al.[47], Godlewska et al.[110] Achlya racemosa o o o Ogbonna & Alabi[109], Czeczuga et al.[47] Achlya rodrigueziana o o Czeczuga et al.[47] Achlya spiralis o o Paul & Steciow[117] Aphanomyces acinetophagu o o Czeczuga et al.[47] Aphanomyces amphigynus o o o El-Hissy et al.[88], Czeczuga et al.[47], Godlewska et al.[110] Aphanomyces astaci o o Unestam & Nylund[118], Czeczuga et al.[47], Godlewska et al.[110], Hsieh et al.[119] Aphanomyces bosminae o o Czeczuga et al.[47] Aphanomyces daphniae o o o Czeczuga et al.[47] Aphanomyces frigidophilus o o o o Ballesteros et al.[120] Aphanomyces helicoides o o o o Srivastava & Srivastava[113], Czeczuga et al.[47] Aphanomyces invadans o o o Iberahim et al.[121] Aphanomyces irregularis o o o o Czeczuga et al.[47] Aphanomyces laevis* o o o o Shanor & Saslow[114], Ogbonna & Alabi[109], Czeczuga et al.[47], Godlewska et al.[110] Aphanomyces ovidestruens o o Czeczuga et al.[47] Aphanomyces patersonii o o Czeczuga et al.[47] Aphanomyces stellatus o o o o Hoshina et al.[115], Shah et al.[116], Ogbonna & Alabi[109] Dictyuchus carpophorus o o El-Hissy et al.[88] Dictyuchus magnusii o o El-Hissy et al.[88] Dictyuchus monosporus o o El-Hissy et al.[88] Dictyuchus sterilis o o El-Hissy et al.[88], Godlewska et al.[110] Geolegnia helicoides* o o o o Steciow et al.[122], Nam et al.[123] Leptolegnia caudata* o o o o o o o o o El-Hissy et al.[88], Czeczuga et al.[47], Nam et al.[123] Newbya androgyna o o o El-Hissy et al.[88], Czeczuga et al.[47], Godlewska et al.[110] Protoachlya papillata o o o Ogbonna & Alabi[109], Czeczuga et al.[47] Pythiopsis cymosa o o El-Hissy et al.[88], Czeczuga et al.[124] Saprolegnia aenigmatica* o o o Sandoval-Sierra et al.[36], Nam et al.[123] Saprolegnia anisospora o o o o o Czeczuga et al.[47], Masigol et al.[125] Saprolegnia asterophora* o o o o o o Czeczuga et al.[47], Godlewska et al.[110], Nam et al.[123] Saprolegnia australis* o o o Vega-Ramírez et al.[108], Rezinciuc et al.[37], Pavić et al.[126], Nam et al.[123] Saprolegnia crustosa o o Czeczuga et al.[47] Saprolegnia delica* o o o o o o Czeczuga et al.[47], Godlewska et al.[110], Pavić et al.[126], Nam et al.[123] Saprolegnia diclina o o o o o o o Czeczuga et al.[47], Fernández-Benéitez et al.[39], Hunjavanit et al.[107], Godlewska et al.[110], Vega-Ramírez et al.[108], Martín-Torrijos et al.[127], Sadinski et al.[128], Nam et al.[123] Saprolegnia eccentrica o o Czeczuga et al.[47] Saprolegnia ferax* o o o o o o Blaustein et al.[129], Czeczuga et al.[47], Romansic et al.[41], Prada-Salcedo et al.[130], Godlewska et al.[110], Vega-Ramírez et al.[108], Groffen et al.[131], Nam et al.[123] Saprolegnia glomerata o o o o o Czeczuga et al.[47], Godlewska et al.[110], Vega-Ramírez et al.[108] Saprolegnia hypogna o o Godlewska et al.[110] Saprolegnia latvica o o Czeczuga et al.[47] Saprolegnia litoralis o o Ogbonna & Alabi[109], Czeczuga et al.[47] Saprolegnia megasperma o o o Nolan[132] Saprolegnia monilifera o o o o Czeczuga et al.[47], Godlewska et al.[110] Saprolegnia monoica o o o o Czeczuga et al.[47], Godlewska et al.[110] Saprolegnia parasitica* o o o o o o Czeczuga et al.[47], Godlewska et al.[110], Vega-Ramírez et al.[108],
Shin et al.[133], Pavić et al.[126], Nam et al.[123]Saprolegnia pseudocrustosa o o Czeczuga et al.[47] Saprolegnia salmonis* o o Hussein et al.[38], Nam et al.[123] Saprolegnia terrestris o o o Vega-Ramírez et al.[108] Saprolegnia torulosa* o o o o o o Godlewska et al.[110], Czeczuga et al.[47], Nam et al.[123] Saprolegnia turfosa o o Godlewska et al.[110] Saprolegnia uliginosa o o o o Czeczuga et al.[47], Vega-Ramírez et al.[108] Saprolegnia unispora o o o o o Czeczuga et al.[47], Godlewska et al.[110], Vega-Ramírez et al.[108] Thraustotheca clavata o o o Ogbonna & Alabi[109], Czeczuga et al.[47], Godlewska et al.[110] Species reported in Korea are shown in bold. Asterisk means oomycetes species isolated in the present study. Table 1.
Oomycetes inhabiting freshwater ecosystems.
-
Character Phytopythium minutum Phytopythium citrinum Phytopythium delawarense Phytopythium aichiense Phytopythium litorale Colony Chrysanthemum pattern on PDA, faintly chrysanthemum on V8A, and faintly striate on CMA Submerged and mixed chrysanthemum-rosette pattern on PCA Radiate pattern on PCA Stellate pattern on V8A Radiate to chrysanthemum pattern on PCA and V8A Growth rate 8 mm per day at 25 °C
on PDA
12 mm per day at 25 °C
on V8A
9.5 mm per day at 25 °C
on CMA9 mm per day at 25 °C on CMA
11 mm per day at 25 °C
on PCA10 mm per day at
25 °C on CMA11.6 mm per day at
25 °C on CMA13.3 mm per day at 25 °C on CMA
12.6 mm per day at 25 °C on PCAProliferation Internal extended Internal extended Internal nested and internal extended Internal nested and internal extended Internal nested and internal extended Hyphal swellings Absent Absent Present Absent Present Sporangia (Sub-)globose, 13–23 (av. 18 ± 3) μm diameter, or ovoid to (ob-)pyriform, 16–20 × 24–27 (av. 18 ± 2 × 26 ± 1) μm, terminal or intercalary (Sub-)globose or pyriform to lemoniform, terminal
or intercalary, 15–35 (av. 24.2) μm diameterGlobose to slightly elongate, limonform or (ob-)void, terminal or intercalary, 25–40 (av. 32.5) μm diameter Sub-globose, ovoid or limonoform, 25–39 × 19–27 (31.4 × 24.9) μm (av. 31.4 μm) diameter (Sub-)globose, broad ovoid or obpyriform, terminal or intercalary, 22.8–32.2 × 20.8–29.1 (av. 28.5 × 26.7) μm Oogonia Smooth, globose to sub-globose or limoniform, 15.5–29.3 (av. 21.5 ± 4) μm diameter Smooth, globose, 20–36 (av. 27.6) μm diameter Smooth, globose, 21–34 (av. 27.2) μm diameter Smooth, globose, 29–40 (av. 34.3) μm diameter Absent Antheridia Monoclinous or diclinous, showing undefined shapes or encircling the oogonium Strictly hypogynous 1–2 per oogonium, diclinous, broad lengthwise contact 1–2 per oogonium, mostly diclinous, and rarely monoclinous, crook-necked or filamentous inflated Absent Oospores Aplerotic or plerotic, globose, 13–26 (av. 18.5 ± 3) μm diameter Plerotic, 18–34 (av. 24.9) μm diameter Aplerotic, 21–26 (av. 24.2) μm diameter Aplerotic, av. 24.2 μm diameter Absent Cardinal growth temperature (minimum/optimum/
maximum temperature)4/30/35 °C n.a. 10/28/34 °C 4/28/34 °C 4/30/35 °C Ref. This study Paul[151] Broders et al.[152] Baten et al.[93] Nechwatal & Mendgen[146] Table 2.
Comparison of Phytopythium minutum with morphologically and phylogenetically related species (n.a. = not available).
-
Character Phytopythium lacustre Phytopythium iriomotense Phytopythium mercuriale Phytopythium oedochilum Phytopythium boreale Colony Radiate to faint chrysanthemum patterns on PDA, V8A and CMA Chrysanthemum pattern on V8A Tightly chrysanthemum on PDA, rosaceous on MEA, slightly stellate on CMA Submerged with some aerial mycelium on CMA, without a special pattern on PCA n.a. Growth rate 12 mm per day at 25 °C
on PDA
14 mm per day at 25 °C
on V8A
10 mm per day at 25 °C
on CMA24.3 mm per day at 25 °C on CMA 9 mm per day at
25 °C on CMA20 mm per day at 25 °C on CMA 20 mm per day at
25 °C on CMAProliferation Internal extended Internal or internal nested Internal or internal nested External, internal and internal nested Internal Hyphal swellings Absent Present Present Absent Present Sporangia Globose to sub-globose, terminal, 24–38
(av. 31 ± 4) μm
diameterGlobose to sub-globose, papillated and terminal
av. 32.7 μmSub-globose to obovoid, papillated and terminal
av. 22 μmSub-globose, lemoniform, obovoid
or ovoid, papillated
av. 32.8 μmAbsent Oogonia Smooth, (sub-)globose, terminal or intercalary, 29–36 (av. 32.5 ± 2) μm diameter Smooth, globose av.
27.3 μm diameter, oogonia with double oospores presentSmooth, globose, av. 29.8 μm diameter Smooth and globose av. 32.8 μm diameter Smooth, globose, av. 32.8 μm diameter Antheridia Monoclinous or diclinous, 1−2 per oogonium, club-shaped or inflated clavate Diclinous, 1−2 per oogonium, crook-necked or occasionally amphigynous antheridial cells are present Mostly diclinous, multiple knotted around the oogonia Diclinous, occasionally monoclinous, 1−2(4) per oogonium, cells were large, curved, and broadly laterally attached to the oogonium Monoclinous, rarely diclinous, 1−2 per oogonium, antheridial cells are amphigynous
or sessilOospores Aplerotic or plerotic, globose, 23–35(av. 29.5 ± 4) μm diameter Aplerotic or plerotic av. 24.4 μm diameter Absent Aplerotic av. 30.3 μm diameter Plerotic av. 22.2 μm diameter Cardinal growth temperature (minimum/optimum/
maximum temperature)10/30/35 °C 10/30/35 °C 5/25–30/43 °C 10/30/35 °C 4/25–30/43 °C Ref. This study Baten et al.[93] Belbahri et al.[153] Van der Plaats-Niterink[150] Duan[154] Table 3.
Comparison of Phytopythium lacustre with morphologically and phylogenetically related species (n.a. = not available).
-
Character Pythium aculeatum Pythium flevoense Pythium rishiriense Pythium pyrilobum Pythium inflatum Colony Faint chrysanthemum pattern on PDA and V8, radiate pattern on CMA Chrysanthemum pattern on PDA and CMA Rosette petaloid on PDA, vaguely petaloid on V8A Radiate pattern without a special pattern on PCA Radiate pattern on PCA, slightly radiate pattern on CMA Growth rate 13.5 mm per day at 25 °C on PDA
15.5 mm per day at 25 °C on V8A
13.5 mm per day at 25 °C on CMA10 mm per day at 25 °C
on CMA16 mm per day at
25 °C on PCA22 mm per day at
25 °C on PCA20 mm per day at 25 °C on CMA Hyphal swellings Present. Hyphal bodies, terminal or intercalary, (sub-)globose, limoniform or catenulate Absent Absent n.a. n.a. Sporangia Filamentous, inflated, irregularly swollen Filamentous, not-inflated Filamentous, inflated Filamentous, inflated, irregularly swollen Filamentous, inflated, forming irregular or globose outgrowths Oogonia Ornamented with blunt projections, globose, terminal, 19–27 (av. 21 ± 2) μm diameter Smooth, mostly terminal on short side branches of feather-like hyphae, 17–30 (av. 19) µm diameter Smooth, globose, terminal or intercalary, sometimes in a chain, av. 24.3 μm diameter Smooth, globose or pyriform, terminal or intercalary, 23–33 (av. 27) μm diameter Smooth, globose, terminal or intercalary, 19–27 (av. 22.5) μm diameter Antheridia Monoclinous or diclinous, 1−2 per oogonium, antheridial cells globose or clavate Diclinous, one to several per oogonium, antheridial stalks mostly bifurcate or differently branched near the oogonium 1−2(–5) per oogonium, monoclinous or diclinous, terminal on branched stalk, rarely intercalary, clavate or
crook-necked, hypogynousMonoclinous or diclinous, mostly two or three cells on one long stalk which encircles the oogonium, mostly crook-necked 1−2 per oogonium, diclinous Oospores Aplerotic or plerotic, globose, often yellowish, 16–24 (av. 20 ± 2) µm diameter Aplerotic, occasionally nearly plerotic, 14–24
(av. 17.7) µm diameterMostly plerotic, av. 21.8 μm diameter Aplerotic or plerotic, globose, yellowish, 20–32 (av. 23.5) µm diameter Plerotic, 18–25 (av. 21.5) µm diameter Cardinal growth temperature (minimum/optimum/
maximum temperature)5/20/30 °C 5/25/35 °C 5/35/42 °C 4/25/37 °C 5/30/37 °C Ref. This study Van der Plaats-Niterink[155] Rahman et al.[82] Vaartaja[156] Van der Plaats-Niterink[150] Table 4.
Comparison of Pythium aculeatum with morphologically and phylogenetically related species (n.a. = not available).
-
Character Pythium aquaticum Pythium fluminis Pythium catenulatum Pythium torulosum Pythium rhizo-oryzae Pythium rishiriense Colony Rosaceous pattern on PDA, radiate pattern on V8 and CMA Rosaceous pattern on PDA, chrysanthemum pattern on V8A, irregular pattern on CMA Narrow chrysanthemum pattern on PCA, rosette pattern on CMA Rosette pattern on PCA Narrow chrysanthemum pattern on PCA, rosette pattern on CMA Rosette petaloid on PDA, vaguely petaloid on V8A Growth rate 12.5 mm per day at 25 °C on PDA
9.5 mm per day at
25 °C on V8A
10.5 mm per day at 25 °C on CMA12.5 mm per day at
25 °C on PDA
14.5 mm per day at
25 °C on V8A
10.5 mm per day at
25 °C on CMA14 mm per day at
25 °C on PCA15 mm per day at
25 °C on PCA12 mm per day at 25 °C on PCA 16 mm per day at
25 °C on PCAHyphal swellings Present. Hyphal bodies, terminal or intercalary, cylindrical, clavate, (sub-)globose, ovoid or peanut-shaped Present. Hyphal bodies, terminal or intercalary, clavate, (sub-)globose, catenulate, ovoid or peanut-shaped Present Absent Present. Hyphal bodies, catenulate and terminal Absent Sporangia Filamentous, slightly inflated Filamentous, inflated Filamentous, inflated, irregularly swollen, branched parts of the mycelium Filamentous, inflated Not observed Filamentous, inflated Oogonia Not observed Smooth, globose, terminal or intercalary, 11–22 (av. 15 ± 3) μm diameter Smooth, globose, terminal or intercalary, mostly heterothallic,
av. 22.8 μm diameterSmooth, terminal or intercalary, 11–22
(av. 15) μm diameterSmooth, globose, terminal or intercalary, av.
19.7 μm diameterSmooth, globose, terminal or intercalary, sometimes in a chain, av. 24.3 μm diameter Antheridia Not observed Monoclinous, antheridial cells clavate, bell-shaped,
or with wide contact with oogonium5–(–12) per oogonium, diclinous, occasionally monoclinous in homothallic isolates, clavate or crook-necked, stalk branched 1–2(–3) per oogonium, monoclinous or occasionally diclinous, borne on rarely branched antheridial stalks Wrapping around, making a knot 1–2(–5) per oogonium, monoclinous or diclinous, terminal on branched stalk, rarely intercalary, clavate or
crook-necked, hypogynousOospores Not observed Aplerotic or plerotic, globose, 9.5–20 (av.
14 ± 3) μm diameterMostly plerotic, occasionally aplerotic Plerotic, 10–21 μm diameter Plerotic and aplerotic, 14–
20 μm diameterMostly plerotic, av. 21.8 μm diameter Cardinal growth temperature (minimum/
optimum/
maximum temperature)5/30/40 °C 5/30/40 °C 10/30–35/40 °C 5/25–30/35 °C n.a. 5/35/42 °C Ref. This study This study Van der Plaats-Niterink[150] Van der Plaats-Niterink[150] Bala[157] Rahman et al.[82] Table 5.
Comparison of Pythium aquaticum and Py. fluminis with morphologically and phylogenetically related species (n.a. = not available).
-
Character Pythium chlamydosporum Pythium vigens Pythium aquatile Pythium apleroticum Pythium brachiatum Pythium oopapillum Pythium diclinum Colony Radiate pattern on PDA, V8A, and CMA Faint striate to radiate pattern on PDA, V8A, and CMA Rosette pattern on PCA and CMA n.a. Radiate pattern Vague chrysanthemum pattern on PCA Radiate pattern on CMA and PCA Growth rate 16 mm per day at 25 °C on PDA
18 mm per day at 25 °C on V8A
16 mm per day at 25 °C on CMA22 mm per day at 25 °C
on PDA
28.5 mm per day at 25 °C on V8A
20 mm per day at 25 °C
on CMA17 mm per day at
25 °C on PCA19 mm per day at
25 °C on PCA14 mm per day at 25 °C on PCA 17 mm per day at
25 °C on PCA19 mm per day at
25 °C on PCAHyphal swellings Present. Hyphal bodies, terminal or intercalary, (sub-)globose, rarely ovoid or peanut shaped Present. Hyphal bodies, terminal or intercalary, (sub-)globose, ovoid or peanut-shaped, rarely catenulate Absent Absent Filamentous slightly inflated n.a. n.a. Sporangia Not observed Not observed Filamentous or slightly inflated, forming rectangular, dendroid structures Filamentous, not-inflated Not observed Filamentous, inflated, lobate elements Filamentous, not-inflated Oogonia Smooth, globose, terminal or intercalary, 10–24 (av. 18 ± 4) μm diameter Smooth, globose, terminal or intercalary, 21–26 (av. 23 ± 1) μm diameter Oogonia globose, smooth, terminal or less often intercalary, but frequently clustering in small groups, 19–23 (av. 21 µm) diameter Smooth, globose, terminal or intercalary, 11–20 μm Intercalary or terminal, sometimes in chain; smooth- walled or occasionally with a projection, 13.3–34.4 (av. 22.7) Globose, sub-terminal or intercalary, av.
21.5 μmSmooth, spherical or ovoid, mostly terminal or subterminal, occasionally intercalary, 18–23 μm (av. 20.5) Antheridia Monoclinous or diclinous, 1–2 per oogonium, antheridial cells globose or encircling an oogonium Lacking One, rarely two per oogonium, monoclinous, originating from the oogonial stalk at 2–10 µm or more below the oogonium, or from the parent hypha Diclinous, 1–2 per oogonium. Diclinous and monoclinous; arm-like shaped on unbranched stalks; 1–2 per oogonium Rarely hypogynous, antheridial stalks branched Diclinous, 1–2 per oogonium Oospores Aplerotic or plerotic, globose, 9–22 (av. 16 ± 3) µm diameter Aplerotic, globose, 13–16 (av. 15) µm diameter Aplerotic, 16–19 (av. 17.9) µm diameter Aplerotic, 9–17 µm diameter Plerotic, occasionally aplerotic, 9.4–27.2
(av. 16.4) diameterav. 16.4 μm diameter av. 17.5 μm diameter Cardinal growth temperature (minimum/optimum/
maximum temperature)5/25/30 °C 5/25/35 °C 5/25/35 °C 5/30/40 °C 4/25/30 °C n.a./30/30 °C 5/30/40 °C Ref. This study This study Höhnk[158] Ito & Tokunaga[159] Uzuhashi et al.[99] Bala et al.[160] Ito & Tokunaga[159] Table 6.
Comparison of Pythium chlamydosporum and Py. vigens with morphologically and phylogenetically related species (n.a. = not available)
-
Character Pythium intricatum Pythium flevoense Pythium rishiriense Pythium inflatum Pythium aquatile Colony Chrysanthemum patterns on PDA and V8A, very faint striate pattern on CMA Chrysanthemum pattern on PDA and CMA Rosette petaloid on PDA, vaguely petaloid on V8A Radiate pattern on PCA, slightly radiate pattern on CMA Rosette pattern on PCA and CMA Growth rate 10.5 mm per day at 25 °C on PDA
10.5 mm per day at 25 °C on V8A
8.5 mm per day at 25 °C
on CMA10 mm per day at 25 °C
on CMA16 mm per day at 25 °C on PCA 20 mm per day at 25 °C on CMA 17 mm per day at 25 °C on PCA Hyphal swellings Present. Hyphal bodies, terminal, (sub-)globose Absent Absent n.a. Absent Sporangia Filamentous, strongly inflated, lobulated, forming toruloid complexes or dendroid structures. Filamentous, not-inflated Filamentous, inflated Filamentous, inflated, forming irregular or globose outgrowths Filamentous or slightly inflated, forming rectangular, dendroid structures Oogonia Smooth, globose, terminal or intercalary, 17–27 (av. 22 ± 3) μm diameter Smooth, mostly terminal on short side branches of feather-like hyphae, 17–30 (av. 19) µm diameter Smooth, globose, terminal or intercalary, sometimes in a chain, av. 24.3 μm diameter Smooth, globose, terminal or intercalary, 19–27 (av. 22.5) μm diameter Oogonia globose, smooth, terminal or less often intercalary, but frequently clustering in small groups, 19–23 (av. 21 µm) diameter Antheridia Monoclinous or diclinous, 1–2 per oogonium, antheridial cells globose, clavate or encircling an oogonium Diclinous, one to several per oogonium, antheridial stalks mostly bifurcate or differently branched near the oogonium 1–2(–5) per oogonium, monoclinous or diclinous, terminal on branched stalk, rarely intercalary, clavate or
crook-necked, hypogynous1–2 per oogonium, diclinous One, rarely two per oogonium, monoclinous, originating from the oogonial stalk at 2–10 µm or more below the oogonium, or from the parent hypha Oospores Aplerotic or plerotic, globose, 15–26 (av. 20.5 ± 3) µm diameter Aplerotic, occasionally nearly plerotic, 14–24
(av. 17.7) µm diameterMostly plerotic, av. 21.8 μm diameter Plerotic, 18–25 (av. 21.5) µm diameter Aplerotic, 16–19 (av. 17.9) µm diameter Cardinal growth temperature (minimum/optimum/
maximum temperature)5/25/30 °C 5/25/35 °C 5/35/42 °C 5/30/37 °C 5/25/35 °C Ref. This study Van der Plaats-Niterink[155] Rahman et al.[82] Van der Plaats-Niterink[150] Höhnk[158] Table 7.
Comparison of Py. intricatum with morphologically and phylogenetically related species (n.a. = not available).
Figures
(14)
Tables
(7)