-
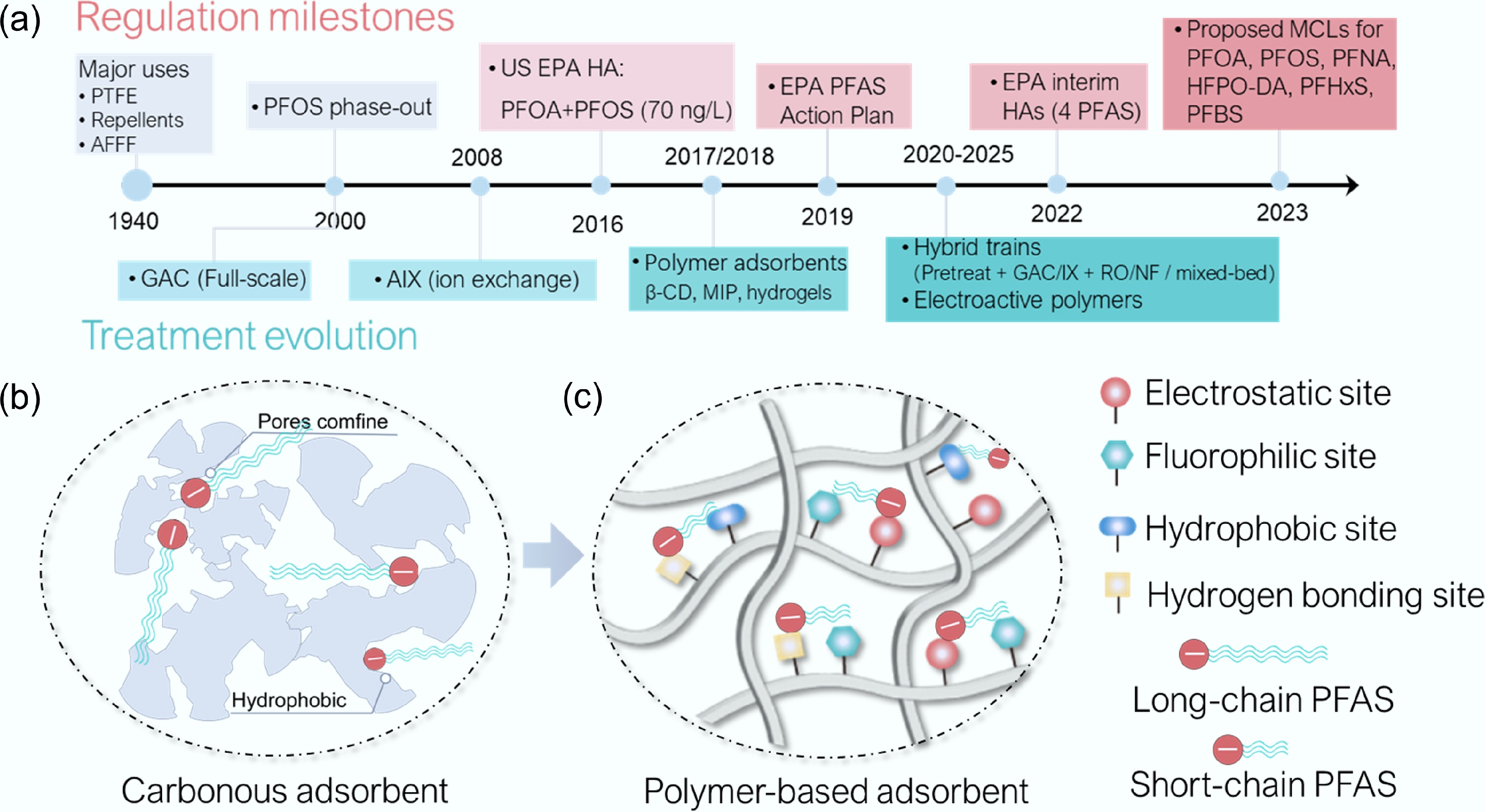
Figure 1.
Novel polymer-based adsorbents for PFAS remediation: history and concept. (a) Timeline highlighting key milestones in the development and applications of polymer-based adsorbents for PFAS removal, aligned with major US Safe Drinking Water Act (SDWA) regulatory actions and advisories that are progressively tightening PFAS limits (Supplementary Table S1). (b) Carbonaceous, and (c) polymer-based PFAS adsorption processes.
-
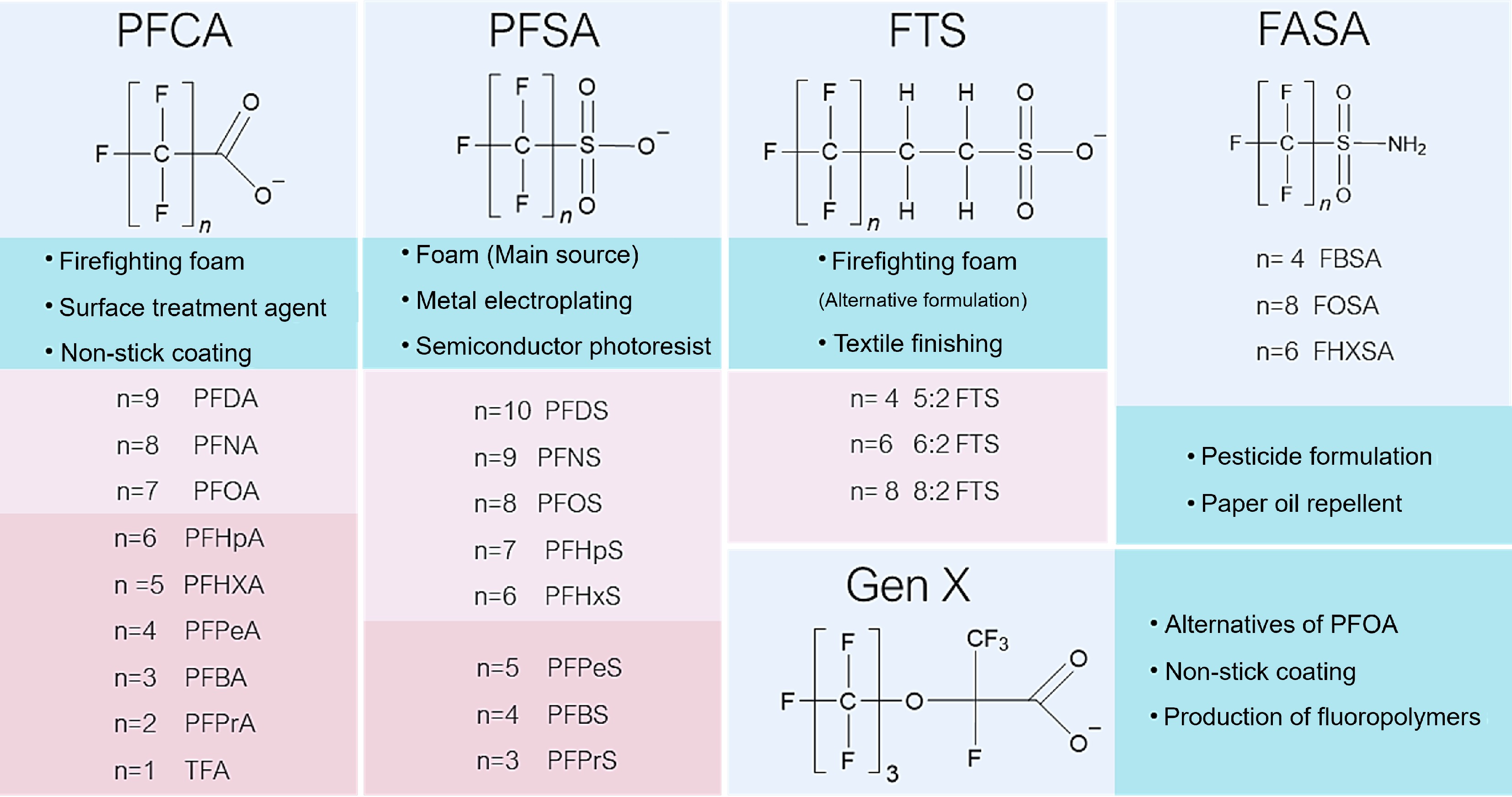
Figure 2.
Chemical structures of widely explored PFAS compounds. n represents individual C−F chain lengths.
-
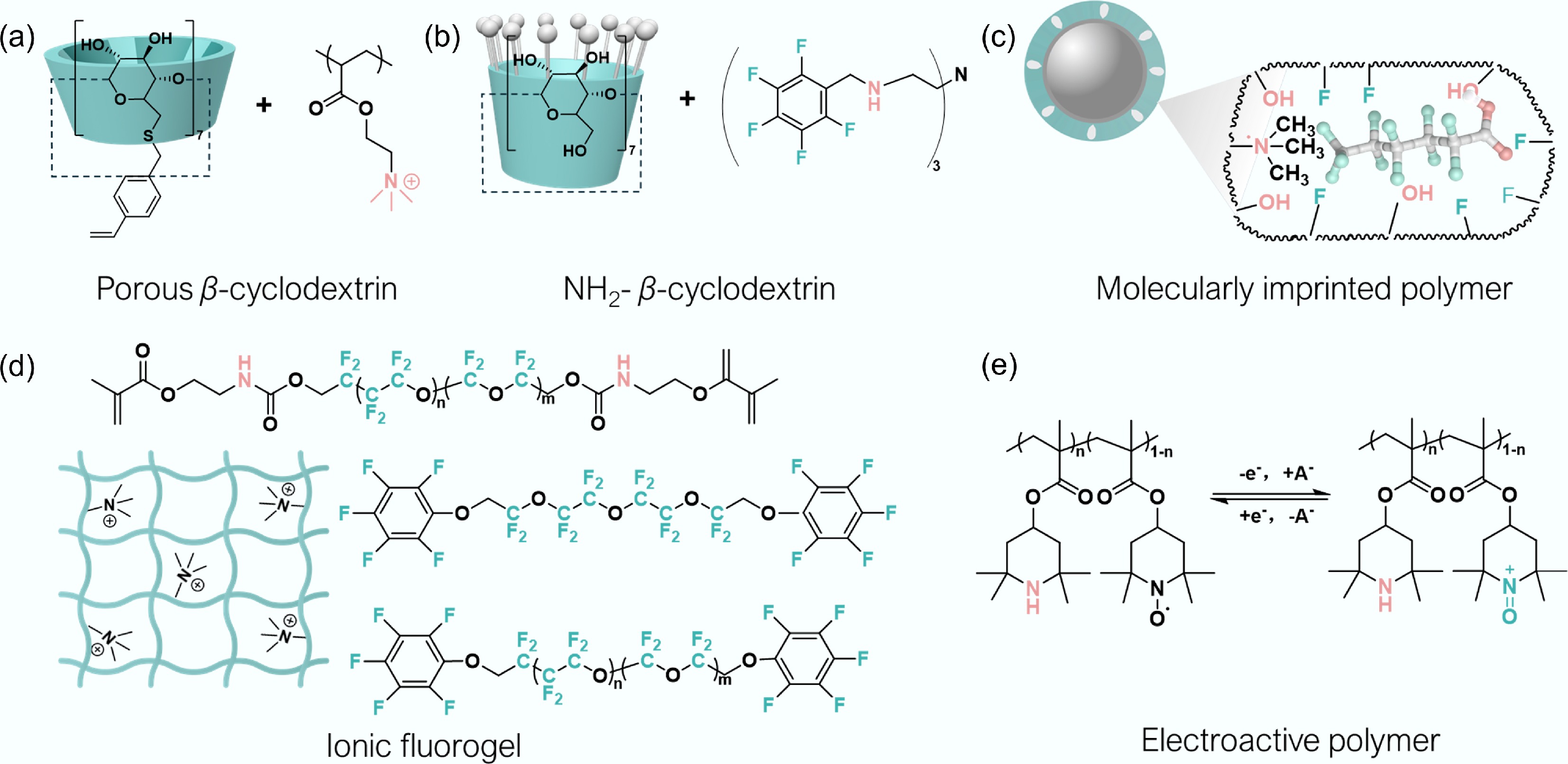
Figure 3.
Summary of chemical structures of the latest representative PFAS polymer-adsorbents. (a) Porous β-cyclodextrin polymers. Adapted from Zhang & Wang[48], with copyright © 2022 American Chemical Society. (b) NH2-β-cyclodextrin polymers. Adapted from Ippolito et al.[49], with copyright © 2024 American Chemical Society. (c) Molecularly imprinted polymer. Adapted from Yang et al.[50], with copyright © 2018, Elsevier. (d) Ionic fluorogel adsorbent. Adapted from Liu et al. and Chang et al. [51,52], with copyright © 2018 and 2022, American Chemical Society, Wiley-VCH. (e) Conducting polymers. Adapted from Qiu et al.[41], with copyright © 2020 and 2024, Wiley-VCH, Elsevier[29].
-
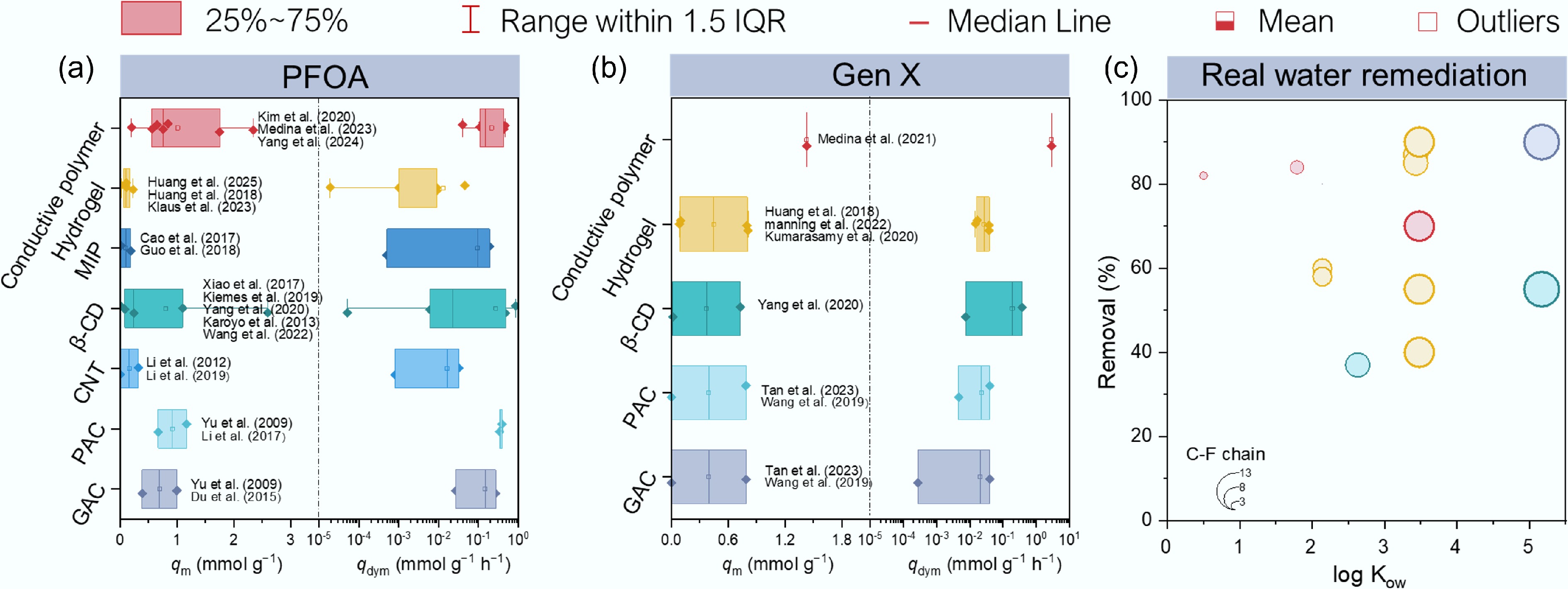
Figure 4.
Comparison of carbon-based and polymer-based adsorbents for PFAS remediation. (a), (b) Comparison of qm and dynamic adsorption capacity (qdyn, mmol g−1 h−1) value for (a) PFOA (n = 18), (b) Gen X (n = 18) in reported literature (listed in Supplementary Tables S2 and S3). The centerline, upper limits and the lower limits of the box, upper whiskers and lower whiskers, and the point represents the median, 25% of the maximum and minimum, the maximum and minimum, and the average of qm or qdyn in different adsorbents. (c) Comparison of the short-chain PFAS removal efficiency of different polymer-adsorbents (listed in Supplementary Tables S4).
-
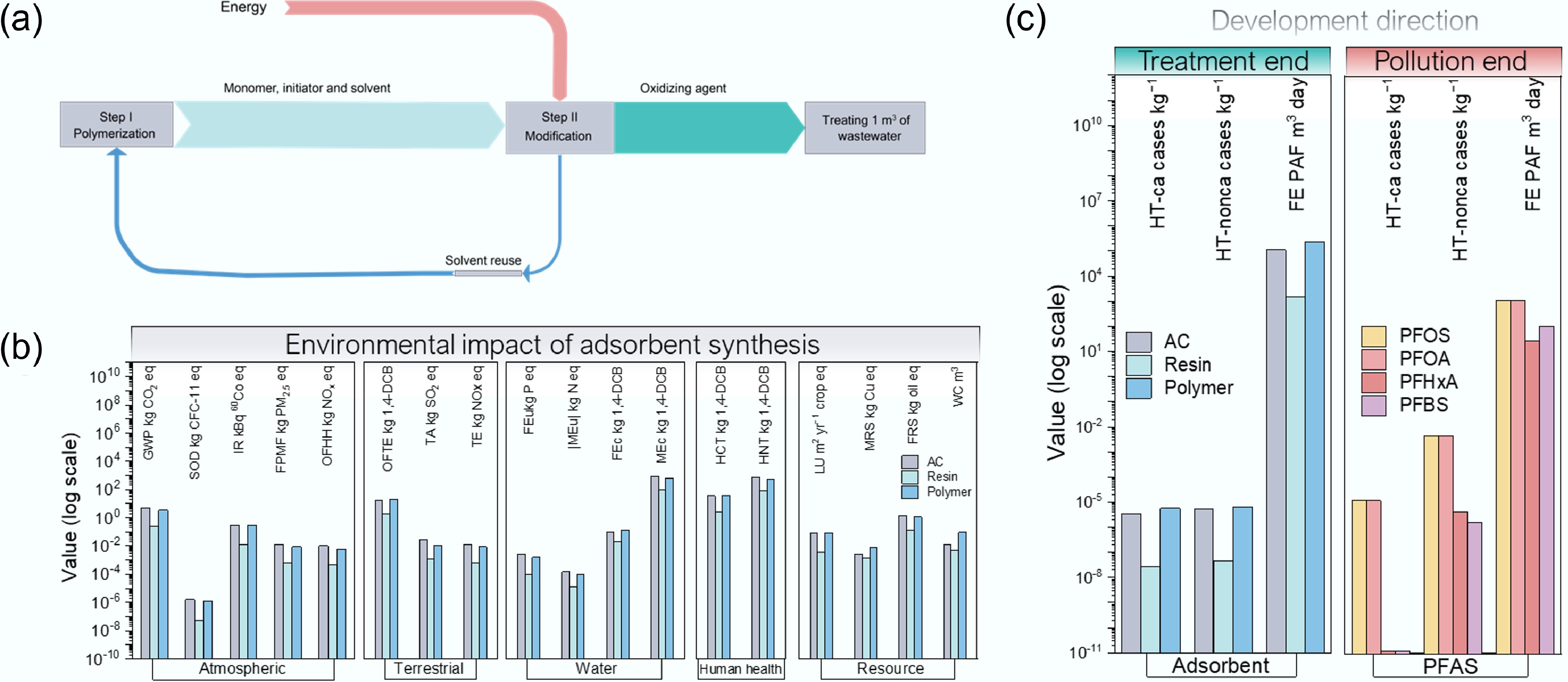
Figure 5.
LCA assessment of the polymer and carbonaceous adsorbents for PFAS remediation. (a) Mass flow of polymer adsorption synthesis, including polymerization of monomer, and modification of the polymer process; (b) Comparison of environmental impacts on MEu, HNT, GWP, TE, and the other 14 categories for polymer and carbonaceous adsorbents (Supplementary Table S7); (c) Environmental damages of polymer and carbonaceous adsorbents and long-chain and short-chain PFAS.
-
Binding type Electrostatic Hydrophobic Fluorophilic Hydrogen bonding Schematic 



Target PFAS moiety Anionic headgroups:
–COO–, –SO3–Fluorinated tail:
–(CF2)n–CF3Fluorinated tail:
–(CF2)n–, –O–CF2–Headgroup O atoms(–COO–/–SO3–) and
ether O (–O–)Sorbent binding site Cationic sites: –NR4+, –NH3+/–NH2+–, imidazolium+/pyridinium+, guanidinium (–C[=NH2+]NH2) Hydrophobic domains: sp2-C (graphitic/aromatic), –(CH2)n–; micropores (geometric confinement) Fluorous domains: –(CF2)n–/–CF3, fluorinated surfaces –C(F)x–, fluorinated pore walls/cavities Donors: –NH–, –OH, –CONH–, –NHCONH–; Acceptors: C=O, –N: (paired with PFAS O atoms) Table 1.
Summary of intermolecular interactions for short-chain PFAS capture
Figures
(5)
Tables
(1)