-

Figure 1.
Worldwide anthropogenic and natural sources of heavy metal contamination.
-
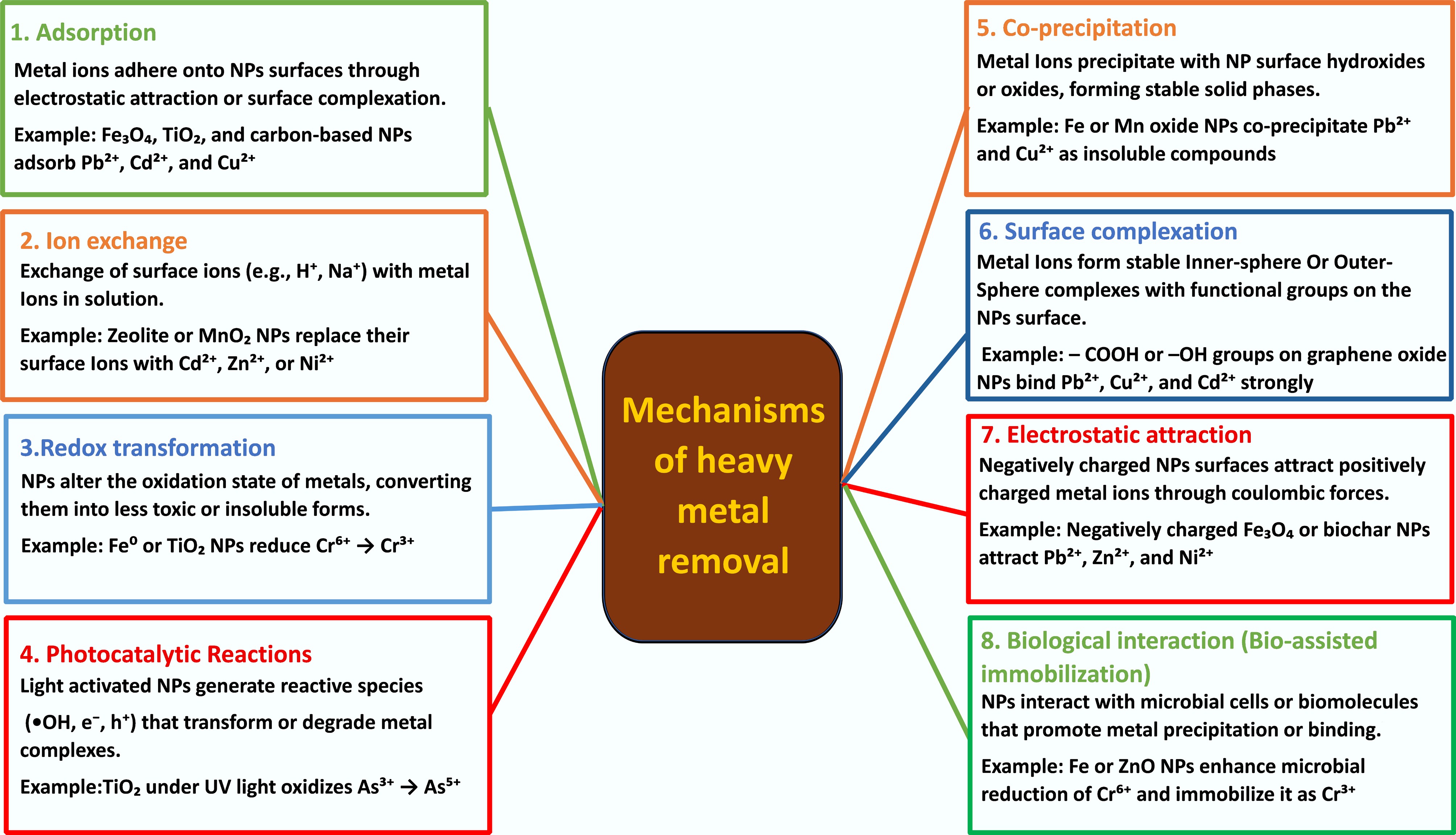
Figure 2.
Schematic representation of major mechanisms by which nanoparticles remove or immobilize heavy metals.
-
Figure 3.
Integration of nanoremediation into sustainable and hybrid systems.
-
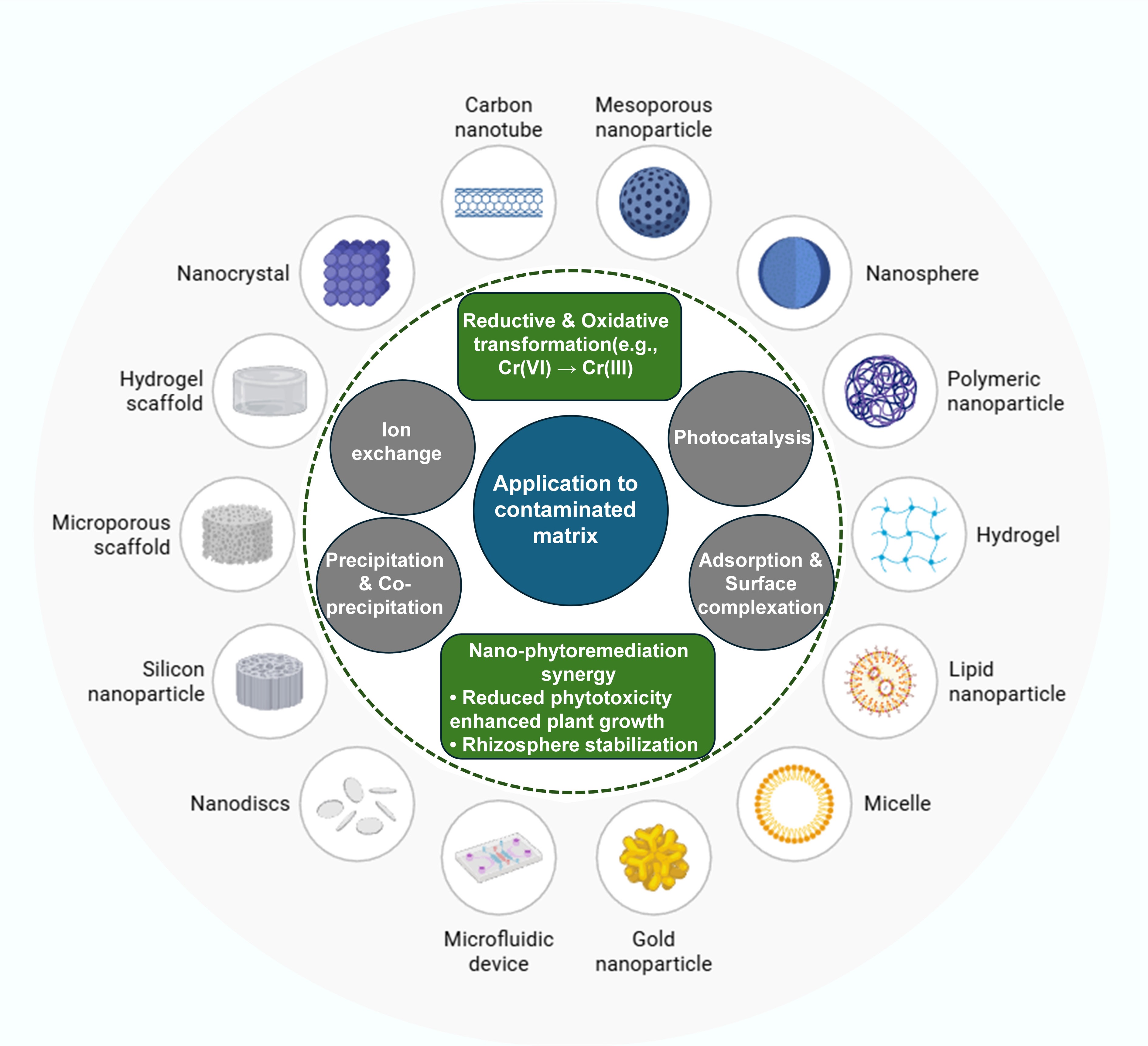
Figure 4.
Mechanisms associated with different nanomaterials for the remediation of inorganic pollutants.
-
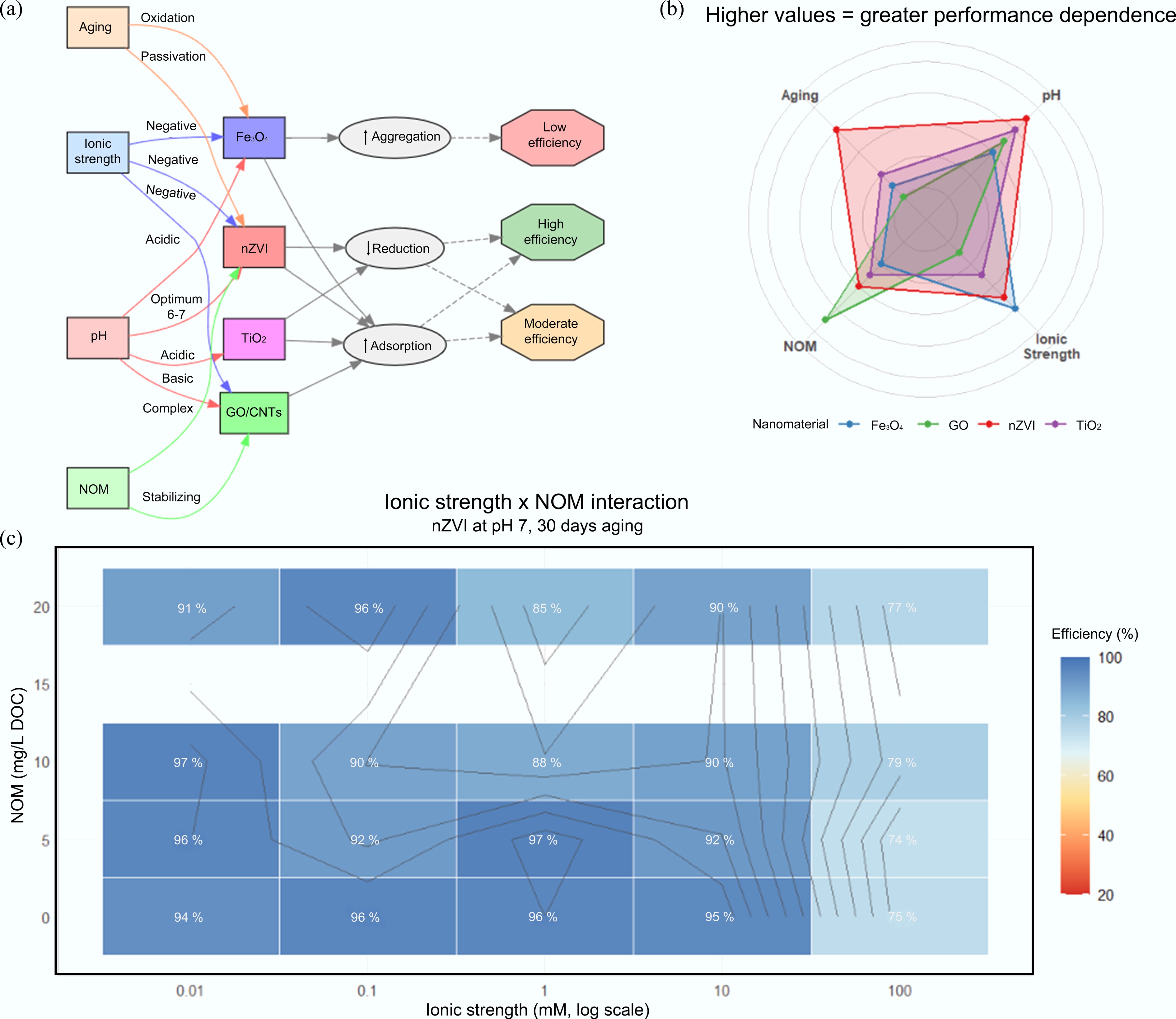
Figure 5.
Environmental parameter controls on nanomaterial remediation efficiency. (a) Schematic overview of key parameter-nanomaterial-performance relationships: pH critically governs nZVI reactivity (optimum ~6 to 7), ionic strength generally suppresses performance via enhanced aggregation, natural organic matter (NOM) exerts complex (stabilizing or competitive) effects, and aging drives progressive surface passivation. (b) Radar chart comparing relative sensitivity of four common nanomaterials to each parameter; radial distance indicates degree of performance dependence (nZVI shows highest sensitivity to pH and aging, while graphene oxide/carbon nanotubes [GO/CNTs] are most affected by NOM). (c) Interaction heatmap for nZVI at pH 7 after 30-d aging, showing how elevated ionic strength and NOM jointly depress removal efficiency, with strongest inhibition occurring under high ionic strength (> 10 mM) coupled with high NOM (> 15 mg/L DOC).
-
Heavy metal Natural sources Anthropogenic sources Systemic impacts and human-health risks Ref. Arsenic (As) Natural groundwater geochemistry, volcanic emissions, and weathering of rocks containing arsenic. Ore mining and smelting; burning coal; use in wood preservatives and insecticides; contaminated groundwater from irrigation; industrial discharge Exposure in the long-term causes skin lesions and keratosis, peripheral vascular disease, lungs, bladder, and skin malignancies, crop and water contamination, and ecological bioaccumulation. [10,11] Antimony (Sb) Weathering occurs naturally in ore minerals such as stibnite (Sb2S3). Lead-acid batteries, electronics, flame retardants, and antimony mining and refining Pollution of the soil and water; irritation of the skin, eyes, and respiratory system; aquatic toxicity; and possible carcinogenicity. [10] Cadmium (Cd) Mineral soils, volcanic emissions, and natural weathering of parent rocks. Phosphate fertilizers, battery manufacturing, electroplating, mining and smelting, and municipal and industrial trash Food chain buildup, respiratory toxicity via inhibition, kidney failure (proteinuria), bone demineralization/osteomalacia, and carcinogenic risk. [12] Chromium (Cr, especially Cr VI) Leaching from ultramafic rocks in natural soils and rocks. Chromate pigments, plating, leather tanning, stainless steel manufacturing, and industrial wastes Soil and water contamination; oxidative stress; carcinogenic and respiratory hazards; inhibition of plant growth. [13,14] Copper (Cu) Volcanic eruptions, natural rocks and soils, and copper-mineral deposits. Cu mining and refining, electronics and electrical wiring, antifouling coatings, and agriculture (Cu fungicides) Excess copper can cause oxidative stress, gastrointestinal problems, and liver damage (such as Wilson's-type toxicity); excessive copper also damages aquatic life and stunts the growth of plant roots. [15] Cobalt (Co) Weathering of soils, ultramafic rocks, and minerals containing cobalt. Hard metals, pigments, rechargeable batteries, and cobalt mining and refining Excess cobalt (Co) can cause cardiomyopathy, thyroid dysfunction, and sensory (vision/hearing) effects; environmentally, it contributes to aquatic toxicity and bioaccumulation. [16] Iron (Fe) Large natural reservoir: soils, volcanic emissions, and igneous and metamorphic rocks. Iron/steel industry; mining; metallurgical slag; acid mine drainage mobilizing Fe and other metals Although necessary, too much soluble iron can lead to oxidative stress and organ damage; in the environment, too much iron can precipitate, change habitats, and lower biodiversity (particularly in aquatic systems). [17] Lead (Pb) Lead-containing mineral weathering, natural soils, and aerosols from crustal sources. Lead gasoline uses in the past, battery recycling, smelting, paint, e-waste, mining, and industrial pollutants Effects on wildlife, chronic organ toxicity, soil and dust pollution, and neurotoxic consequences, particularly in youngsters. [18,19] Molybdenum (Mo) Naturally occurring in volcanic emissions, rocks, and soils. Alloying, mining, steel production, and fertilizers (Mo salts) Impacts on wildlife, long-term organ poisoning, dust and soil contamination, and neurotoxic effects, especially in children. [20] Mercury (Hg) Weathering, volcanic emissions, and methylation of organic materials in aquatic environments. Combustion of coal; artisanal gold mining; industrial chemical applications (batteries, chlor-alkali); and methylation-contaminated fish and seafood Fish biomagnification, neurodevelopmental toxicity, reproductive and cardiovascular consequences, and contamination of aquatic ecosystems. [21] Nickel (Ni) Ultramafic rock weathering and natural mineralization. Coal burning, garbage incineration, stainless steel, nickel mining and refining, and battery manufacturing Exposure to Ni, particularly soluble Ni (II) compounds, can cause skin allergies and dermatitis, respiratory issues (such as occupational asthma), and even cancer; environmental effects include phytotoxicity and soil microbial disturbance. [22] Vanadium (V) Minerals containing V (basaltic rocks, volcanic ash, and oil shales). Vanadium alloys, battery technologies, and oil/coal combustion (fly ash) Exposure to vanadium, particularly V+5, can induce respiratory irritation, renal damage, and gastrointestinal distress; environmental effects include leaching from ash, plant absorption, and microbial toxicity. [23] Table 1.
Summary of key heavy metals, their predominant anthropogenic and natural sources, and associated environmental and health risks
-
Environmental parameter Typical range studied Observed effect on removal efficiency Dominant mechanism affected Representative references pH 2–10 Decreased adsorption at low pH due to surface protonation; enhanced uptake at near-neutral pH Surface complexation, electrostatic interaction [78,79] Ionic strength 1–100 mM Reduced adsorption efficiency due to charge screening and competition Electrostatic attraction, ion exchange [80] Natural organic matter (NOM) 1–20 mg/L DOC Variable effects; often inhibits adsorption via site blocking and complexation Surface site competition, aggregation [81] Particle aging Weeks–months Decline in reactivity due to oxidation, aggregation, or surface passivation Redox activity, surface availability [82,83] Table 2.
Influence of environmental parameters on nanomaterial-based heavy metal remediation
-
Engineered nanomaterials Predominant mechanistic pathways Target metal ions Key features and performance Key limitations Examples Ref. Carbon-based materials Adsorption via-surface functional-group binding, electrostatic attraction, and π–π interactions Cd(II), Hg(II), Pb(II), Cu(II), Co(II), and Zn(II) High metal-binding capacities (up to 1989 mg/g for Hg(II))
Rapid adsorption kinetics, short equilibrium times, and strong metal–functional group interactions
Surface locations' chemical and thermal stabilityExpensive; challenging recovery and reuse Multi-walled carbon nanotubes, graphene oxide nanosheets, and nano-biochar composites [49] Metal oxides Surface complexation, adsorption, and micro-precipitation Pb(II), Cd(II), Cr(VI), Zn(II), Cu(II), and Hg(II) High capacity for adsorption (up to
1,047.83 mg/g for Zn(II))
Superior reusability and regeneration potential
Adjustable surface chemistry and redox activity
High chemical stability and thermal resistance
Photocatalytic action under UV and visible lightAggregation in high ionic strength, limited selectivity, possible dissolution under acidic conditions Fe2O3, TiO2, Al2O3, ZnO [64] Nanocomposites/
hybrid nanoparticlesRedox and surface complexation; synergistic adsorption plus magnetic separation As(V), Pb(II), Cd(II), Cu(II), and Cr(VI) Rapid and effective magnetic recovery
Enhanced colloidal stability and dispersibility
Reusability in moderate temperatures
Increased surface area due to hybrid synergy
Multipurpose elimination of metals and organic pollutantsComplexity in synthesis, variable stability under environmental conditions, cost, potential secondary contamination TiO2–graphene composites,ZnO–polymer hybrids, Fe3O4@chitosan [85] Zero-valent metals Adsorption, precipitation, and redox transformation As(V), Cr(VI), Pb(II), Cd(II), and Cu(II) Fast reaction kinetics with little secondary pollution
High surface reactivity and electron transfer rate
Strong reducing capability toward toxic ions
Conversion of hazardous metals to less soluble or inert forms
Suitability for both soil and aquatic remediationRapid oxidation, aggregation, reduced long-term reactivity, possible toxic by-products Fe/Ni bimetallic nanoparticles, nZVI, and Fe/Cu core-shell systems [86] Biosorbents/functionalized polymers Ion exchange, adsorption, and surface complexation via −COOH, −NH2, −SH, and −OH Cu(II), Pb(II), Cd(II), and Hg(II) Strong binding through surface functional groups
Excellent regeneration and reusability
Low cost and sustainable materials
High selectivity toward target ions
Biodegradable and ecologically friendlyLower mechanical stability, potential biodegradation over time, slower kinetics compared to metallic nanoparticles Thiol-modified cellulose nanofibers, magnetic chitosan beads, and amine-functionalized biopolymer nanoparticles [60] Zeolites Micro-precipitation, ion exchange, and adsorption Cu(II), Zn(II), Pb(II), and Cd(II) High selectivity and ion-exchange capacity
Adjustable porosity and surface charge
Little efficiency loss during reuse; stability over a broad pH range
Efficacy in multi-metal systemsLimited adsorption capacity for certain metals, sensitive to pH changes, possible clogging in soil/water applications Clinoptilolite nanosheets, surfactant-modified zeolite composites, and nano-zeolite Y [87] Bio-nanocomposites Biocatalysis, biosorption, redox transformation (enzyme/microbe-assisted) As(V), Pb(II), Hg(II), and Cr(VI) Combines biological selectivity with nanomaterial reactivity
Transforms metals enzymatically or microbiologically;
Low toxicity and biodegradable
Sustainable, renewable, and regenerable
Functions well in complicated environmental matrices.Variable reproducibility, sensitivity to environmental conditions (pH, ionic strength), limited large-scale data Magnetite–microbe composites, laccase-immobilized nanoparticles, enzyme-loaded chitosan nanofibers [64] Table 3.
Representative engineered nanomaterials, their dominant removal mechanisms, target metals, and key performance features reported in recent studies
-
Matrix Typical nanomaterials for remediation Removal efficiency Key influencing factors Matrix-specific challenges and practical caveats Ref. Plant-based and nano-phytoremediation (plant uptake + rhizosphere) TiO2, ZnO, carbon-based nanomaterials (graphene oxide, CNTs), coated magnetic nanoparticles, functionalized Fe nanoparticles (nFe), and nanofertilizers. Plant uptake showed variable increases (10%–300%) over plants alone, while absolute removal efficiency per area remains lower than engineered water or soil treatments; NP-assisted growth can further enhance remediation. Plant factors: Growth rate, stress tolerance, internal antioxidant systems, root architecture, exudates, and plant species (hyperaccumulator vs. high biomass) all influence nanoparticle mobility and bioavailability
Nanomaterial factors: NP type, dose, and coating influence phytotoxicity vs growth stimulation and can enhance contaminant bioavailability or nutrient delivery, improving overall plant-based remediation efficiency.Strengths: low-cost, sustainable, capable of increasing biomass and restoring vegetation.
Caveats: delayed timeline; variability between lab and field; very species- and dose-dependent; risk of NP accumulation in consumable crops and food chains.[98,99] Water remediation (engineered reactors, wastewater, and effluent) nZVI and iron oxides, TiO2 (photocatalysts), ZnO, graphene oxide (GO), carbon nanotubes (CNTs), magnetic Fe3O4 composites, and NPs coated with polymers or chitosan. Heavy metals achieved high removal efficiency (70%–100%), often ≥ 90% for Pb, Cr, and Cu; efficiency can decrease at high contaminant concentrations or upon reuse of nanomaterials, while adsorbents show tens–hundreds mg/g capacity in well-mixed reactors. Environmental factors: pH, temperature, contact time, ionic strength, and competing ions all have an impact on performance; advantages include recoverable magnetic nanoparticles and quick kinetics; disadvantages include fouling, the necessity for NP recovery, and reduced pilot-scale efficiency.
Nanomaterial properties: Size, surface area, charge, stability, functionalization, and dosage influence removal efficiency.Strengths: controlled reactors, recoverable magnetic nanoparticles, and superior contact/kinetics.
Caveats: fouling, NP recovery/regeneration requirements, environmental release risks, and reduced efficiency in pilot-scale or continuous operations compared to batch tests.[86,98,100] Soil remediation (groundwater, ex situ treatment, and in situ amendments) Zeolite-like nanoadsorbents, metal oxide NPs, carbon NMs, biochar-supported NPs, and nZVI (raw and supported/coated). Heavy metals exhibited moderate to high removal efficiency (20%–98%), but performance is highly variable depending on soil properties, species, and nanoparticle application method; long-term stability and NP aggregation can also limit effectiveness. Soil properties:
Contaminant availability and NP behavior are influenced by pH, organic matter, texture, water content, redox potential, and microbial activity.
Nanomaterial properties: Mobility and removal efficiency are influenced by NP stability, dispersion, aggregation, age (oxidation/sulfidation), dose, spatial delivery, and interactions with soil bacteria.Strengths: nZVI can break down some organics and reduce/immobilize metals.
Caveats: limited NP transport (aggregation, filtration), strong influence of soil texture and organic matter, possible long-term ecotoxicity, and lower field efficiencies.[98,100] Table 4.
Comparison of nanomaterial performance in soil, water, and plant-based remediation systems, including removal efficiencies and influencing factors
-
Scale Typical system and conditions Nanomaterials evaluated Removal efficiency Observed nanoparticle behavior Key limitations Ref. Laboratory Batch reactors; controlled pH, ionic strength; synthetic solutions nZVI, Fe3O4, GO, TiO2 80%–99% metal removal High reactivity; minimal aggregation Overestimation of performance; lack of heterogeneity [49,78] Mesocosm Soil columns, lysimeters, outdoor tanks; semi-natural conditions Biochar–nZVI, Fe oxides, hybrid composites 50%–85% reduction in bioavailable metals Partial aggregation; surface passivation Scale sensitivity; aging effects [70,102] Field In situ soil or aquifer treatment; heterogeneous matrices nZVI slurries,
magnetic composites30%–70% reduction
over monthsStrong aggregation; oxidation; limited transport Delivery constraints; cost; regulatory limits [30,82] Table 5.
Comparison of nanoremediation performance across laboratory, mesocosm, and field scales
-
Category Key challenges and limitations Explanation Proposed research and policy priorities Ref. Scientific gaps Ecotoxicity and long-term impacts Insufficient chronic/trophic-transfer data; possible harm to non-target creatures across trophic levels. Perform extensive, long-term mesocosm experiments and life cycle/ecosystem investigations; measure trophic transfer and bioaccumulation under practical exposure conditions. [107,108] Transformation products and secondary contamination Unknown effects of modified or deteriorated NPs on the environment and living things. Characterize and identify transformation products in practical settings; investigate secondary impacts on geochemistry and biota. [108,109] Environmental fate and transport NP transformations (dissolution, aggregation, aging) and unpredictable mobility in heterogeneous soil–water matrices. Integrate fate-transport models that have been verified in column and field experiments, as well as in-situ characterization techniques (spectroscopy/tracers). [110,111] Bioavailability and mobility under field conditions Lab results may not translate to heterogeneous natural environments. To measure bioavailability, transport, and retention in soils and sediments, conduct field-scale experiments and predictive modeling. [107,112] Interaction mechanisms Limited understanding of NP pollutant and microbe interactions at molecular levels. Utilize controlled mesocosms, synchrotron imaging, and multi-omics to enable mechanism-driven material design and connect mechanisms to results. [112,113] Technical gaps Manufacturing and scalability Lab techniques are costly and difficult to scale for widespread field use. Create low-cost, environmentally friendly, bio- or waste-derived synthesis pathways; conduct techno-economic assessments and pilot-scale production. [113] Material stability under environmental stress Changes in temperature, pH, or ions can cause NPs to break down, lose their reactivity, or congregate. Create robust NPs with surface functionalization, test them in dynamic environments, and take aging studies into consideration. [109] Monitoring and confirmation of performance Absence of field-deployable, real-time techniques to monitor NP activity and fate. Develop standardized KPIs, tracer techniques, and portable sensors for adaptive control and long-term monitoring. [111] Integrating hybrid systems If NPs are not optimized, their integration into chemical or biological processes may result in reduced productivity. To increase productivity while reducing ecological disturbance, study NP–plant–microbe or NP–biochar synergistic systems. [114] Dispersion and delivery In contaminated areas, aggregation lowers NP availability and reactivity. Develop 'safer-by-design' nanoparticles, core-shell, or magnetic structures; maximize in situ distribution using tracers or permeable reactive barriers. [115] Regulatory gaps Gaps in monitoring and liability Aambiguous accountability for unanticipated consequences or failures. Develop legal frameworks for liability, remediation obligations, and post-deployment monitoring. [116] Lack of distinct frames Several countries have environmental laws specifically related to NPs, which are frequently handled like bulk chemicals. Develop global, tiered, risk-based field deployment guidelines that cover permissions and monitoring. [112] Public perception and ethical concerns Skepticism regarding unknown long-term hazards. Conduct socioeconomic impact research, pilot co-design, and open public interaction. [117] Interoperability and data sharing Meta-analysis and cross-study comparison are restricted by the absence of centralized databases. Develop interoperable, open-access databases for performance data, environmental monitoring, and NP characterization. [118] Standardization of risk assessment Inconsistent terminology and test protocols limit comparability. Create standardized reporting guidelines, classification systems, and test procedures for all jurisdictions. [111] Table 6.
Summary of major scientific, technical, and regulatory gaps in nanoremediation, and proposed future research directions for field-scale implementation
Figures
(5)
Tables
(6)