-
The contamination of terrestrial and aquatic environments by potentially toxic elements (PTEs), notably lead (Pb), cadmium (Cd), arsenic (As), chromium (Cr), mercury (Hg), and nickel (Ni), is one of the most persistent global threats to ecosystem integrity and human health (Fig. 1)[1]. Anthropogenic activities such as mining, smelting, fossil fuel combustion, electroplating, and the extensive use of agrochemicals have dramatically elevated heavy metal concentrations in soils, sediments, and water bodies[2]. PTEs are non-biodegradable and prone to accumulate or undergo biomagnification at different trophic levels of food chains, leading to long-term ecological degradation and public health risks, including carcinogenesis, oxidative stress, neurotoxicity, and organ dysfunction[3]. Globally, more than 20 million hectares of arable land have been degraded by PTE contamination, particularly in industrializing regions of Asia, Africa, and Eastern Europe[4].
PTEs originate from both natural (geogenic) and anthropogenic sources[5]. Natural sources include weathering of parent materials, volcanic emissions, geothermal activity, and aeolian dust transport, which together determine background concentrations of elements such as Ni, Cr, cobalt (Co), and Cd[6]. Piwowarska et al.[7] reported that quantitative source-apportionment studies show strong inter-element and regional differences; for example, Cd is dominated by anthropogenic emissions in many soil-water settings, with an estimated global anthropogenic: natural emission ratio on the order of 7:1, while Ni is frequently dominated by geogenic (lithogenic) inputs in non-industrial areas[8]. Chromium and Co commonly show large industrial/traffic contributions in urban and mining-affected regions, although their relative shares vary substantially by site (typical receptor-model results report combined natural sources ≈30%–40%, and anthropogenic sources [industry, traffic, coal/biomass combustion, atmospheric deposition] ≈60%–70% for mixed urban/industrial soils and sediments)[9]. However, modern contamination is overwhelmingly caused by anthropogenic sources, as explained in Table 1.
Table 1. Summary of key heavy metals, their predominant anthropogenic and natural sources, and associated environmental and health risks
Heavy metal Natural sources Anthropogenic sources Systemic impacts and human-health risks Ref. Arsenic (As) Natural groundwater geochemistry, volcanic emissions, and weathering of rocks containing arsenic. Ore mining and smelting; burning coal; use in wood preservatives and insecticides; contaminated groundwater from irrigation; industrial discharge Exposure in the long-term causes skin lesions and keratosis, peripheral vascular disease, lungs, bladder, and skin malignancies, crop and water contamination, and ecological bioaccumulation. [10,11] Antimony (Sb) Weathering occurs naturally in ore minerals such as stibnite (Sb2S3). Lead-acid batteries, electronics, flame retardants, and antimony mining and refining Pollution of the soil and water; irritation of the skin, eyes, and respiratory system; aquatic toxicity; and possible carcinogenicity. [10] Cadmium (Cd) Mineral soils, volcanic emissions, and natural weathering of parent rocks. Phosphate fertilizers, battery manufacturing, electroplating, mining and smelting, and municipal and industrial trash Food chain buildup, respiratory toxicity via inhibition, kidney failure (proteinuria), bone demineralization/osteomalacia, and carcinogenic risk. [12] Chromium (Cr, especially Cr VI) Leaching from ultramafic rocks in natural soils and rocks. Chromate pigments, plating, leather tanning, stainless steel manufacturing, and industrial wastes Soil and water contamination; oxidative stress; carcinogenic and respiratory hazards; inhibition of plant growth. [13,14] Copper (Cu) Volcanic eruptions, natural rocks and soils, and copper-mineral deposits. Cu mining and refining, electronics and electrical wiring, antifouling coatings, and agriculture (Cu fungicides) Excess copper can cause oxidative stress, gastrointestinal problems, and liver damage (such as Wilson's-type toxicity); excessive copper also damages aquatic life and stunts the growth of plant roots. [15] Cobalt (Co) Weathering of soils, ultramafic rocks, and minerals containing cobalt. Hard metals, pigments, rechargeable batteries, and cobalt mining and refining Excess cobalt (Co) can cause cardiomyopathy, thyroid dysfunction, and sensory (vision/hearing) effects; environmentally, it contributes to aquatic toxicity and bioaccumulation. [16] Iron (Fe) Large natural reservoir: soils, volcanic emissions, and igneous and metamorphic rocks. Iron/steel industry; mining; metallurgical slag; acid mine drainage mobilizing Fe and other metals Although necessary, too much soluble iron can lead to oxidative stress and organ damage; in the environment, too much iron can precipitate, change habitats, and lower biodiversity (particularly in aquatic systems). [17] Lead (Pb) Lead-containing mineral weathering, natural soils, and aerosols from crustal sources. Lead gasoline uses in the past, battery recycling, smelting, paint, e-waste, mining, and industrial pollutants Effects on wildlife, chronic organ toxicity, soil and dust pollution, and neurotoxic consequences, particularly in youngsters. [18,19] Molybdenum (Mo) Naturally occurring in volcanic emissions, rocks, and soils. Alloying, mining, steel production, and fertilizers (Mo salts) Impacts on wildlife, long-term organ poisoning, dust and soil contamination, and neurotoxic effects, especially in children. [20] Mercury (Hg) Weathering, volcanic emissions, and methylation of organic materials in aquatic environments. Combustion of coal; artisanal gold mining; industrial chemical applications (batteries, chlor-alkali); and methylation-contaminated fish and seafood Fish biomagnification, neurodevelopmental toxicity, reproductive and cardiovascular consequences, and contamination of aquatic ecosystems. [21] Nickel (Ni) Ultramafic rock weathering and natural mineralization. Coal burning, garbage incineration, stainless steel, nickel mining and refining, and battery manufacturing Exposure to Ni, particularly soluble Ni (II) compounds, can cause skin allergies and dermatitis, respiratory issues (such as occupational asthma), and even cancer; environmental effects include phytotoxicity and soil microbial disturbance. [22] Vanadium (V) Minerals containing V (basaltic rocks, volcanic ash, and oil shales). Vanadium alloys, battery technologies, and oil/coal combustion (fly ash) Exposure to vanadium, particularly V+5, can induce respiratory irritation, renal damage, and gastrointestinal distress; environmental effects include leaching from ash, plant absorption, and microbial toxicity. [23] Mining and ore processing generate metal-rich tailings and acid mine drainage, mobilizing PTEs into surrounding ecosystems[24]. Smelting and metallurgical industries release metals via atmospheric fallout, while fossil fuel combustion, particularly coal burning—emits PTEs via fly ash and flue gases[25]. Agriculture contributes additional inputs from phosphate fertilizers, sewage sludge, and pesticide residues, while urbanization introduces diffuse emissions from traffic, building materials, and waste disposal[26]. These processes generate both point-source and diffuse pollution, resulting in complex contamination patterns across soil and water matrices[27].
Once released, PTEs are subjected to complex flow, and can be biomagnified at various food chain levels, and their behavior is regulated by speciation processes, pH, redox potential (Eh), and interactions with mineral and organic materials[4]. They are recycled into environmental compartments (biological and hydrological), adsorbed, and their persistence and bioavailability are determined by precipitation, complexation, and adsorption[28]. Historical industrial effluents have deposited heavy metal pollution in soils; consequently, during decontamination efforts, metals not only remain in soil and accumulate in sediments, but also leach into water bodies under varying geoenvironmental conditions[29]. For instance, Rodriguez-Freire et al.[30] reported that the 2015 Gold King Mine waste release in the Animas River (USA) discharged millions of gallons of metal-laden mine water containing Cd, Pb, As, and other PTEs. Most of the metals were reported to have settled into the riverbed sediments, creating a legacy contamination source.
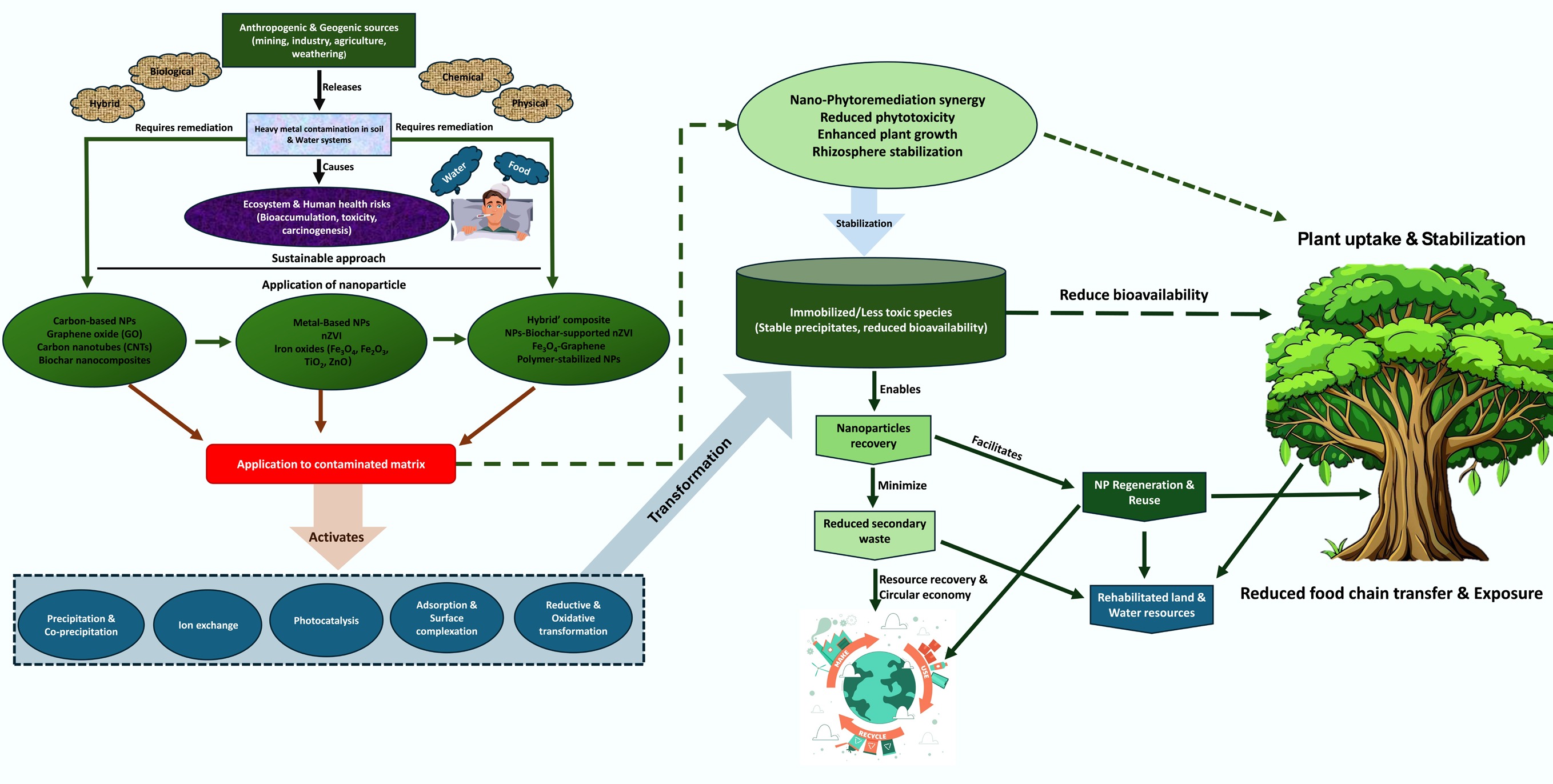
To mitigate PTE pollution, various approaches have been employed; however, traditional physical treatment procedures— including excavation, soil washing, landfill, and chemical stabilization—have not been very successful due to their laborious nature and the additional processing costs incurred from the use of heavy machinery, which is later responsible for soil compaction. These methods are generally expensive, time-intensive, and may generate secondary pollution, or merely transfer contaminants elsewhere[21]. More sustainable alternatives include biological methods, such as microbial and nano-phytoremediation, which tend to be slow but highly location-specific, and are dependent on soil pH, salinity, and redox conditions[31]. This creates an urgent need to identify cost-effective, flexible, sustainable, and environmentally friendly remediation strategies that might respond to the chemical complexity and heterogeneity of contaminated matrices (Fig. 2).
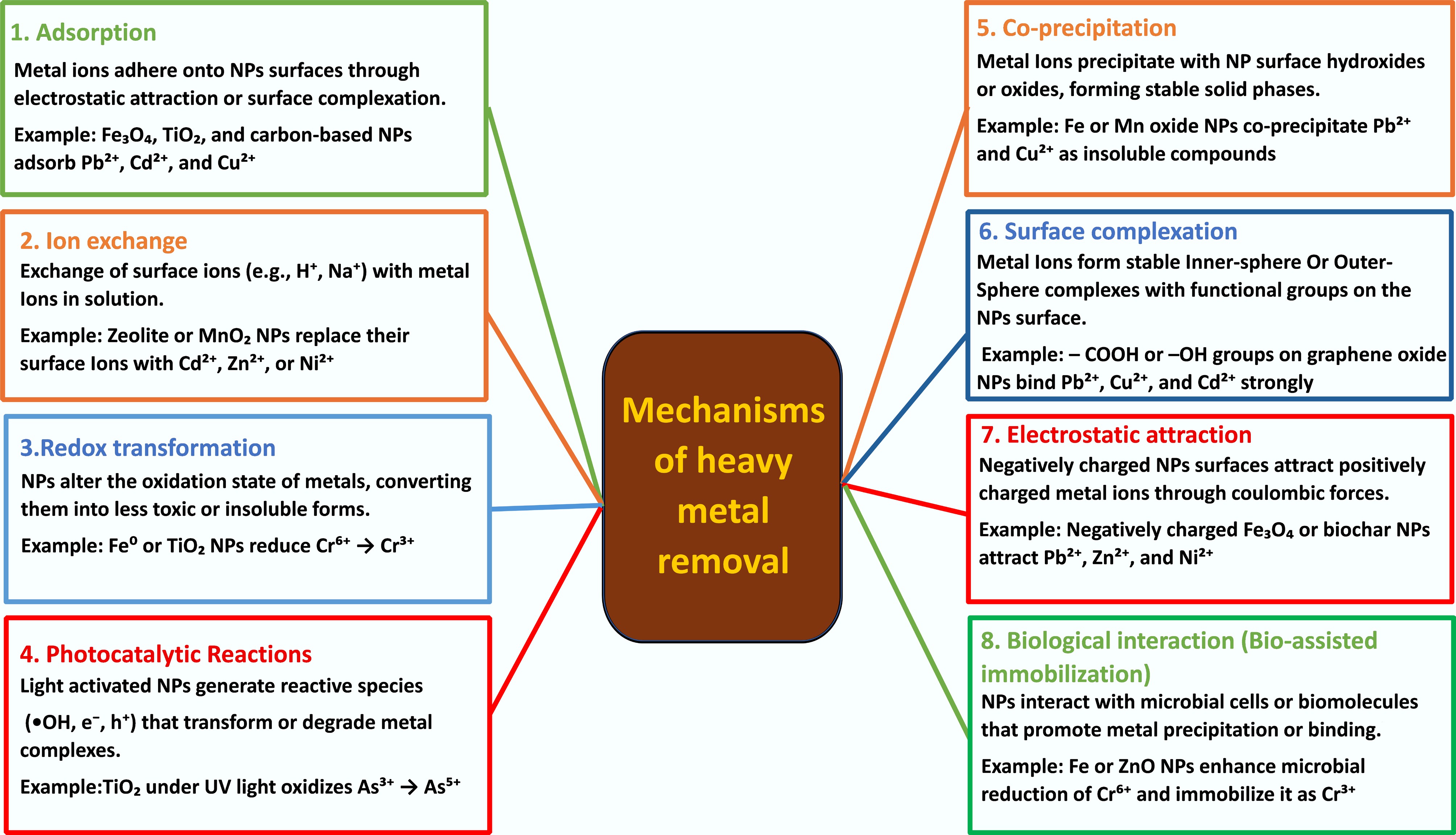
Figure 2.
Schematic representation of major mechanisms by which nanoparticles remove or immobilize heavy metals.
In this regard, nanotechnology has emerged as a promising tool in environmental remediation. Nanoparticles are physicochemical systems with dimensions of 1–100 nm, that possess distinctive characteristics, such as a large surface-area-to-volume ratio, controlled surface chemistry, and redox activity[32], which allow toxic metals to be adsorbed, reduced, and immobilized in a highly efficient manner[33]. Nanomaterials used to remediate pollution, collectively referred to as nanoremediation, have become internationally recognized owing to their capacity to induce rapid and in situ detoxification with limited ecosystem impact[34]. Nanomaterials that have been found to have an excellent ability to remove or transform contaminants through different mechanisms, including adsorption, ion exchange, redox transformation, and co-precipitation, include nZVI, oxides of iron, titanium dioxide, and carbon-based materials (graphene oxide, biochar-supported materials)[35]. Despite the extensive efforts undertaken, there are still challenges in scaling nanoremediation technologies for field application. The passivation and transformation of nanoparticle surfaces in the natural environment can reduce reactivity, and uncertainties concerning their environmental fate, ecotoxicity, and their long-term stability remain largely unknown[36]. Although many prior reviews on nanoremediation have widely reported laboratory-level performance and categorized nanomaterials by their composition, they fail to effectively combine the mechanistic understanding with environmental authenticity and scalability[37−40]. The available literature tends to fragment the discussion, either focusing on the adsorption properties of nanoparticles, redox processes in idealized aqueous systems, or individual field trials, without addressing the fundamental science-field-scale interface issues in a systematic way. This review is the first to provide synthesize mechanistic pathways (adsorption, ion exchange, redox, photocatalysis, and co-precipitation) in connection with their sensitivity to dynamic environmental parameters (pH, Eh, natural organic matter [NOM], ionic strength, and aging) in soil, water, and plant systems. Unlike previous literature, performance differences are highlighted across scales—that is, between laboratory, mesocosm, and field level performance—and address emerging regulatory and ecotoxicological concerns. This study offers a translational framework that can be used to transform nanoremediation beyond bench-scale potential into informed and sustainable field application by connecting nanomaterial design with site-specific challenges in geochemistry and delivery. Future research must address the existing gaps to make nanoparticles more stable, standardize evaluation, and integrate nanoremediation with the sustainable management of soil and water.
Based on the above knowledge, this review aims to provide a mechanistic and practically oriented synthesis of the current situation in the nano-remediation of heavy-metal-polluted environments. It addresses: (1) the physicochemical principles of the interactions between nanoparticles and metals; (2) the potential for the biodegradation and remediation of heavy metals using nanomaterials in the soil, water, and plant system, and (3) the most important aspects of nano-remediation efficiency and field implementation. This review highlights opportunities and pathways to advance nano-remediation as a safe, effective, and sustainable process for decreasing PTE contamination through synthesizing existing knowledge, and addressing essential research gaps. Unlike previous reviews that primarily focus on laboratory-scale performance or specific nanomaterial classes, the present review integrates mechanistic pathways with environmental parameter sensitivity, scale-dependent performance discrepancies, and regulatory considerations, thereby providing a comprehensive framework for translating nanoremediation technologies toward real-world implementation.
-
Nanoparticles (NPs) have emerged as a useful tool for environmental remediation due to their high reactivity and surface chemistry, which can be tailored to enhance performance. The use of NPs in the removal of PTEs is directly associated with their structural classification and physicochemical properties[41]. Generally, NPs employed in remediation can be classified into three broad categories, including metal-based nanoparticles, carbon-based nanoparticles, and hybrid/composite nanomaterials. Each category has its own unique features and mechanisms for PTE removal, including adsorption, ion exchange, reduction, and co-precipitation in contaminated environments.
Metal-based nanoparticles
-
One of the most widely studied categories of nanomaterials for remediation includes metal-based nanoparticles (MNPs) and metal oxide nanoparticles. Examples include nanoscale zero-valent iron (nZVI), nanoparticulate iron oxides minerals (Fe3O4, Fe2O3), titanium dioxide (TiO2), zinc oxide (ZnO), manganese dioxide (MnO2), and copper oxide (CuO). These nanoparticles exhibit enhanced physicochemical reactivity because of their high surface-to-volume ratio, redox-active cores, and high densities of surface hydroxyl groups[42].
The surface chemistry and redox potential are the main factors contributing to the unique reactivity of NPs, which can be further enhanced via surface modification and functionalization, resulting in engineered nanoparticles (ENPs). Owing to functionalization, nZVI exhibits a core-shell structure that includes an Fe0 core and an oxidized Fe(II)/Fe(III) shell, which enables the adsorption and reductive precipitation of metal ions[43]. The Fe0 core donates electrons to reduce PTE ions to less bioavailable or reductively transformed forms, and the oxidized shell provides surface sites through which metals can be adsorbed and co-precipitated. Redox-based mechanisms have also been reported by Yang & Shen[42], who demonstrated that TiO2 and ZnO nanoparticles possess high oxidative and photocatalytic activity under UV or visible light irradiation.
Morphology and crystallinity of metal-based nanoparticles are also known to exert significant influence on their reactivity. Magnetic nanoparticles (MNPs) occur in diverse morphologies, including spherical, rod-shaped, and plate-like forms, which directly affect the density of reactive surface sites, and the accessibility of adsorbates. Their particle size typically ranges from 10 to 100 nm, a scale at which surface-dominated and quantum effects govern reactivity and adsorption performance. For example, nZVI particles with diameters of approximately 50 nm exhibit a specific surface area of ~20–25 m2/g[44], and Fe3O4 nanoparticles of similar size exhibit ~60 m2/g[45], which might enhance adsorption potential towards PTE remediation via sorption, surface complexation, and redox-mediated processes. However, environmentally exposed ENPs might undergo different processes such as particle aggregation, surface oxidation, and metal leaching, commonly leading to reduced surface activity, loss of magnetic responsiveness, and diminished adsorption efficiency. These limitations significantly constrain the long-term stability and field-scale applicability of MNPs. To overcome these challenges, various stabilization strategies have been developed, including polymer coating, surface functionalization, and immobilization onto solid support matrices, which improve dispersibility, inhibit surface passivation, and enhance environmental durability[46].
Metal-based nanoparticles have promising potential to remediate organic and inorganic contaminants through several dominant and often intricate mechanisms. Surface adsorption and complexation involve the interaction of dissolved metal ions with reactive surface functional groups—such as hydroxyl, oxide, or other oxygen-containing moieties—resulting in the formation of inner- or outer-sphere coordination complexes[47]. Reductive transformation occurs when nanoparticles act as electron donors, transferring electrons to redox-active metals, and converting them into less toxic or less mobile forms, such as Cr redox-mediated conversion to Cr(III)[14]. Precipitation and co-precipitation subsequently take place when reduced or strongly adsorbed metals form insoluble hydroxide or oxide phases that nucleate and grow on the nanoparticle surface, further immobilizing the contaminants. Together, these coupled processes govern the high removal efficiency of metal-based nanoparticles in aqueous and soil environments[43].
Carbon-based nanoparticles
-
Carbon-based nanomaterials (CNMs) have attracted considerable interest due to their structural diversity, stability, and tunable surface chemistry. The most widely studied CNMs include carbon nanotubes (CNTs), graphene, and graphene oxide (GO/rGO), carbon quantum dots, and biochar-based nanoparticles[48]. They are characterized by high specific surface area (up to 1,000 m2/g, abundant surface functional groups (e.g., hydroxyl, carboxyl, epoxy), and excellent electrical conductivity. Carbon nanomaterials react with PTEs mainly through absorption and surface complexation processes. The electrostatic attraction of metal ions between negatively charged functional groups (–COOH, –OH, C=O), and positively charged metal ions allows the rapid sorption of PTEs[49]. The degree of surface oxidation or functionalization directly influences sorption capacity, as an increased density of oxygen- or nitrogen-containing functional groups enhances metal–ligand coordination and surface complexation. Compared with pristine carbon nanotubes (CNTs), functionalized CNTs bearing carboxyl (–COOH) or amino (–NH2) groups exhibit significantly higher adsorption performance toward divalent metal ions. For instance, surface functionalization has been reported to increase Pb(II) and Cu(II) adsorption capacities by approximately 2–5-fold, with removal efficiencies improving from <40% for pristine CNTs to > 80%–90% for functionalized CNTs under comparable conditions. This enhancement is attributed to the formation of strong inner-sphere complexes between metal ions and deprotonated carboxyl or amine groups, as well as improved surface hydrophilicity and dispersibility of the modified CNTs[50].
Another key process associated with CNMs is ion exchange, in which metal ions in solution substitute weakly bound ions (e.g., H+, Na+) on the carbon surface. The ion-exchange capacity of graphene oxide and biochar nanoparticles is determined by the heterogeneous distribution of acidic and basic sites on their surfaces[51]. Moreover, the adsorption of organometallic and mixed contaminants is also facilitated by π−π and van der Waals forces. In addition to adsorption, CNMs have several practical benefits: (1) they have high sorption potential and are chemically stable; (2) they can be regenerated by a simple chemical or thermal treatment; and (3) they may be used in composite systems with metal oxides or polymers to increase their functionality. Nevertheless, they also have certain shortcomings, such as aggregation of CNTs in aqueous solutions, reduced selectivity in multifaceted mixtures, and potential environmental toxicity when released[52]. These problems can be mitigated by functionalizing or immobilizing CNMs on larger, inert supports.
The high remediation efficiency of carbon-based nanomaterials arises from the strong interplay between their surface chemistry and metal-binding mechanisms. Oxygen-, nitrogen-, and sulfur-containing functional groups on graphene oxide, carbon nanotubes, and biochar-derived nanocomposites facilitate inner-sphere complexation with soft and borderline metal ions, while π–metal interactions between delocalized aromatic domains and metal ions further enhance adsorption affinity. In addition to surface complexation, partially reduced graphene structures can participate in electron transfer processes, promoting the reduction of redox-sensitive contaminants. Recent mechanistic studies have demonstrated that heterogeneity in functional group distribution and defect density governs both adsorption selectivity and redox reactivity, emphasizing the need to tailor carbon nanomaterials at the molecular scale for site-specific heavy-metal remediation[49].
Hybrid and composite nanoparticles
-
Hybrid nanocomposites are nanomaterials that combine two or more classes of nanomaterials to achieve synergistic behavior beyond that of the individual constituents. More often, the hybrids consist of metal or metal oxide nanoparticles and carbonaceous materials such as functionalized biochar, graphene–metal complexes, or CNTs. These combinations take advantage of the redox reactivity of metals and the high surface area and stability of carbon structures. For instance, carbon-supported nano zero-valent iron (C–nZVI), and Fe3O4-graphene composites have been reported to eliminate several PTEs simultaneously, with improved kinetics and adsorption capacities[53]. The carbon matrix acts as a dispersive and protective coating that prevents aggregation of metal particles, and makes them more accessible for electron transfer and adsorption of metal ions. Concurrently, the metallic component provides the redox potential for transformation and immobilization of contaminants. The hybrid composites therefore operate via multi-mechanistic routes, which include surface adsorption to the carbon carrier, reduction utilizing metallic centers, and consequent precipitation or co-precipitation.
One of the advantages of hybrid materials is their increased stability and reusability[54]. Qureashi et al.[43] reported that magnetic hybrids, e.g., Fe3O4–biochar and Fe3O4–graphene, can be readily separated and recovered with the help of external magnetic fields. Furthermore, the selective adsorption of hybrids can be fine-tuned according to the desired specific binding properties, either through functional groups introduced via surface modification or intermediate binding sites doped with heteroatoms[51].
The superior performance of hybrid nanomaterials is primarily attributed to interfacial charge-transfer mechanisms arising from heterojunction formation between carbonaceous supports and metal or metal oxide phases. In rGO–metal oxide composites, graphene derivatives function as electron sinks, facilitating rapid separation and transport of photogenerated charge carriers, thereby suppressing electron–hole recombination and enhancing photocatalytic and redox-driven metal transformation. For example, rGO–ZnO nanocomposites have demonstrated significantly improved Cr(VI) adsorption capacity and faster reduction kinetics compared with their individual components, which has been attributed to enhanced interfacial electron transfer and increased density of reactive surface sites[55]. Similarly, TiO2-graphene heterostructures exhibit superior photocatalytic reduction efficiency toward redox-sensitive contaminants due to improved charge separation, and extended electron lifetime at the nanoscale interface[56,57]. These synergistic mechanisms highlight the importance of nanoscale interface engineering in the rational design of hybrid nanomaterials with optimized adsorption, redox, and photocatalytic functionalities for heavy-metal remediation. These characteristics render hybrid nanocomposites highly compatible with sustainable, field-implementable remediation technologies. However, hybrid materials also face challenges related to the complexity of synthesis and increased production costs compared with single-component systems. Optimizing composition, particle dispersion, and surface modification is essential to translate laboratory-scale success into real-world environment applications.
-
Nanoparticles have unique physicochemical properties that set them apart from their bulk counterparts, and explain their exceptional effectiveness in the remediation of heavy metals. The most significant of these is their very high surface-area-to-volume ratio due to the dominance of the surface atoms[41], which results in a highly reactive surface. This increases the number of adsorption sites, and the speed of interfacial reaction kinetics, facilitating the quick sorption, reduction, or transformation of PTEs in aqueous systems and soil[58]. The increased catalytic activity is also attributed to the high surface area, which facilitates the nucleation of insoluble metal precipitates, thereby enhancing immobilization and stability of contaminants in environmental matrices.
A defining advantage of nanoparticles is their modifiable surface chemistry, which provides the primary control over interfacial interactions with contaminants. Through controlled synthesis or post-functionalization, nanoparticle surfaces can be engineered with hydroxyl, thiol, carboxyl, or amine moieties, thereby regulating surface charge, hydrophilicity, and binding affinity toward specific metal ions[59]. Such functional group modification enables selective complexation or electrostatic attraction of priority contaminants, including As, Cr, Pb, and Cd. In parallel, tailoring surface functionalization allows adjustment of the point of zero charge and surface potential, which enhances colloidal stability under complex environmental conditions, suppresses aggregation, and preserves reactive surface area and long-term reactivity[58]. Beyond surface chemistry, size- and shape-dependent physicochemical properties further dictate nanoparticle performance. At the nanoscale, quantum size effects alter band-gap energies and electron mobility, imparting enhanced redox activity to materials such as nanoscale zero-valent iron (nZVI), TiO2, and ZnO. These electronic properties facilitate rapid electron-transfer reactions, including the reduction of toxic Cr(VI) to the less mobile Cr(III), as well as photo-catalytically assisted degradation of co-occurring organic pollutants[60]. Multi-mechanistic cooperation occurs, where adsorption, redox transformation, and precipitation operate simultaneously: adsorption onto graphene oxide or biochar surfaces captures metals, redox-active sites on nZVI reduce Cr(VI) to Cr(III), and secondary precipitation stabilizes the immobilized metals, with adsorption generally being the fastest initial process, followed by reduction and precipitation[40]. Concurrently, morphological control—through the fabrication of spheres, rods, or sheet-like structures—modulates surface energy and the exposure of reactive crystallographic facets, thereby influencing adsorption capacity and catalytic turnover rates. In practical remediation systems, magnetic functionality provides an additional design dimension that addresses post-treatment recovery and reuse. Superparamagnetic iron oxide nanoparticles (SPIONs) exhibit high magnetization under an external magnetic field while losing residual magnetism upon field removal, enabling efficient separation after remediation and preventing irreversible aggregation[61]. Moreover, magnetic cores can serve as recoverable scaffolds for catalytic or sorptive shells, yielding multifunctional nanocomposites with enhanced operational control. Finally, the construction of hierarchical nanostructures with interconnected mesopores and high surface areas promotes superior mass transfer and contaminant diffusion to active sites, enabling simultaneous adsorption, ion exchange, precipitation, and redox reactions while minimizing diffusion limitations typical of conventional bulk sorbents[62]. In this context, 'ideal behavior' refers to the theoretical assumption of uniform surface adsorption sites and monolayer coverage, as represented in the Langmuir model, whereas 'non-ideal behavior' accounts for heterogeneous surface energies, multilayer adsorption, and site interactions, which are often better described by the Freundlich model. This distinction is important in environmental applications, as natural soils and water matrices rarely exhibit uniform adsorption properties, and both models provide complementary insights into nanoparticle–metal interactions.
The primary significance lies in colloidal stability and transformation in the natural environment because they dictate the behavior and movement of nanoparticles within the environmental matrix. pH, ionic strength, and natural organic matter are key parameters that control aggregation, dissolution, and surface charge characteristics of nanoparticles[36]. Improved dispersibility enhances contaminant contact; however, excessive mobility may increase ecological risk, and stabilizers, or immobilization matrices are therefore commonly used to balance reactivity and environmental safety. In particular, oxidation, sulfidation, or coating of reactive nanoparticles can occur under environmental conditions, and during these transformations, nanoparticle surface properties, and remediation performance, may vary significantly[58].
-
ENPs eliminate PTEs in aqueous and soil environments through a limited number of coupled physicochemical processes—adsorption (surface complexation and surface precipitation), ion exchange, redox and photocatalytic reactions, and co-precipitation (Fig. 3). The use of magnetic recovery for composite materials also enhances their operational sustainability. These mechanisms commonly occur in concert but can be discussed independently; a single nanocomposite can adsorb, reduce, and immobilize metals through secondary mineral formation simultaneously. Understanding the prevailing pathways under specific environmental conditions is essential for enhancing removal efficiency, predicting by-product formation, and preserving the long-term stability of immobilization[35,63,64].
Adsorption and surface complexation
-
Adsorption is the most widespread and general mechanism for metal removal by ENMs, primarily driven by their high surface area, reactive functional groups, and variable surface charge. It consists of electrostatic attractions, inner- and outer-sphere complexation, as well as surface precipitation[65,66]. Oxygen-, nitrogen-, or sulfur-containing surface groups (e.g., hydroxyl, carboxyl, amine, and thiol) firmly bind metal cations (Pb(II), Cd(II), and Cu(II)) to create coordination complexes, which may be either reversible (outer-sphere), or tightly chemisorbed (inner-sphere)[67].
Carbon-based nanomaterials such as graphene oxide, carbon nanotubes, and biochar composites, as well as nanoparticulate metal oxides (Fe3O4, MnO2, TiO2), have a high adsorption capacity, and can be functionalized with a specific ligands (thiol, amine, and phosphates) to enhance selectivity toward soft metals (Hg[II]) and biologically relevant anions (As[V])[64]. Non-ideal adsorption behavior indicates the presence of heterogeneous surface sites and multilayer adsorption, and this behavior is usually modeled using Langmuir or Freundlich isotherms. However, environmental factors influencing adsorption performance include pH, ionic strength, and natural organic matter (NOM). Acidic conditions diminish cation uptake by protonating surface functional groups, whereas alkaline conditions enhance uptake through surface deprotonation and increased metal complexation. Conversely, oxyanionic absorbates, such as arsenate, are preferentially adsorbed at low pH via ligand exchange with protonated surface hydroxyls[63]. Competitive ions (Ca[II], Mg[II]) can block active sites, and NOM can form soluble metal-organic complexes, thereby lowering capacity[36]. Thus, adsorption efficiency is directly governed by nanomaterial surface properties, including functional group density, surface charge, and surface heterogeneity, which collectively determine metal selectivity, binding strength, and resistance to competitive ions under complex environmental conditions.
Ion exchange
-
Ion exchange is a stoichiometric exchange of structural or surface-bound ions (e.g., H+, Na+, Ca+, etc.) with target metal ions at exchangeable positions on layered or functionalized nanomaterials[63]. Layered double hydroxides (LDHs), sulfonated or carboxylate carbonaceous clays, and sulfonated or carboxylate carbonaceous composites demonstrate strong cation-binding abilities and sorbent reversibility and can be regenerated using acid or salt elution[64]. Selectivity is governed by hydrated ionic radius, charge density, and the presence of competing ions in the solution. The presence of Ca(II) and Mg(II) can cause a significant inhibition of target metal uptake in complex aqueous matrices. Thus, adsorption or reduction processes are often combined with ion-exchange nanomaterials to improve overall removal efficiency and selectivity. Consequently, ion-exchange performance is controlled by structural properties such as layer spacing, exchangeable site density, and hydrated ionic radius, which dictate the selectivity and reversibility of metal uptake in complex aqueous and soil systems.
Reductive and oxidative transformations
-
Reduction-based mechanisms are particularly useful for redox-sensitive contaminants such as Cr, As, and Hg where nanozero-valent iron (nZVI) is the most widely studied reduction-based nanomaterial that releases electrons to oxidized metal ions and reduces them to less toxic and less soluble compounds like Cr(III)[35,68]. This process is achieved through electron transfer from the Fe0 core, followed by the oxidation of Fe(0) to Fe(II)/Fe(III) oxides or hydroxides, which in turn provide reactive surfaces for metal adsorption and reduction[69]. The reactivity of the nanoparticles (size, surface passivation) and solution pH are the key factors regulating reaction rates; additionally, the absence or presence of competing electron acceptors (O2, NO3−) strongly influences surface efficiency. Surface modification, bimetallic doping (e.g. Pd, Fe) or supporting nZVI on biochar or silica matrices enhances dispersion, reduces passivation, and improves stability[70]. Alternative electron-donor systems for multi-metal reduction include iron-to-sulfide systems, such as sulfide-bearing nanoparticles, as well as doped semiconductors[71]. The effectiveness of redox-driven remediation is therefore governed by intrinsic material properties such as electron-donating capacity, surface passivation behavior, and corrosion kinetics, which collectively control contaminant transformation rates and long-term stability of reduced metal species.
In photocatalytic nanomaterials, where electrons and holes drive reduction or oxidation reactions, photoactivation is induced using photocatalyst nanomaterials (TiO2, ZnO, Ag/TiO2). Incident light promotes excitation of electrons from the valence band to the conduction band, enabling the reduction of metal ions such as the Cr(VI), while photogenerated holes and reactive oxygen species (ROS) oxidize organic ligands or co-contaminants associated with metals[57,72]. Improving quantum efficiency is achievable through visible-light-performing modifications (e.g., silver [Ag] or nitrogen [N] doping), and by suppressing charge recombination via heterojunction design and surface doping[73]. The ability represents a major sustainability advantage of photocatalysis; however, limitations include light attenuation in turbid matrices, catalyst specificity, and the potential ecotoxic effects of reactive oxygen species (ROS). Sunlight can facilitate catalytic reactions and activate magnetic-core composites promoting contaminant immobilization as well as catalyst recovery and reuse. Photocatalytic remediation outcomes are thus determined by band-gap energy, charge-carrier lifetime, and interfacial charge-transfer efficiency, which together regulate reactive species generation, and the extent of metal reduction or immobilization under environmental light conditions.
At the mechanistic level, redox-driven nanoremediation is governed by interfacial electron-transfer processes between reactive nanomaterial surfaces and metal species. In the case of nanoscale zero-valent iron, electrons are transferred from the Fe0 core to adsorbed contaminants through surface oxide layers, enabling the reduction of Cr(VI) to Cr(III), and As(III) to less mobile forms As(V), while concurrent corrosion reactions generate Fe(II)/Fe(III) oxides that further immobilize metals via co-precipitation and surface complexation[74,75]. During this process, surface reconstruction and passivation gradually occur as iron oxides and hydroxides accumulate, leading to a decline in reactivity over time. In photocatalytic systems, such as TiO2- and ZnO-based nanomaterials, photoexcitation generates electron–hole pairs that participate in redox reactions at the catalyst surface, with photogenerated electrons driving metal reduction and photogenerated holes contributing to the formation of reactive oxygen species from water and dissolved oxygen. The efficiency of these pathways is strongly influenced by charge-carrier lifetime, surface defect density, and the availability of co-reactants, highlighting the importance of both material design and environmental conditions in sustaining long-term redox and photocatalytic activity[56,74].
Precipitation and co-precipitation
-
In precipitation and co-precipitation, new solid phases, including hydroxides, oxides, carbonates, or sulfides may form and surround or entrap metal ions, although not all phases necessarily form simultaneously. Most redox or adsorption reactions are closely coupled with these processes, including the reduction of Cr(VI) by nZVI to produce Cr(III) hydroxides, which co-precipitate with Fe oxyhydroxides. Similarly, sulfonated iron nanoparticles can form FeS phases, which entrap Pb(II), Cd(II), and Zn(II) through the formation of insoluble metal sulfides[70]. The thermodynamic stability and long-term effectiveness of co-precipitation for metal immobilization remain uncertain, except in cases where stable crystalline or mixed-metal phases are formed. However, under changing environmental conditions such as acidification, redox change, or organic complexation, poorly crystallized precipitates may become unstable and remobilize[76]. Therefore, achieving stable field-scale remediation requires the formation of long-lasting mineral phases or the entrapment of metals within porous supports. Across all mechanisms, the remediation outcome is ultimately determined by the alignment between NP physicochemical properties and site-specific environmental conditions, highlighting the importance of property-guided material selection rather than mechanism-agnostic deployment.
Magnetic recovery and recyclable nanomaterials
-
The introduction of magnetic nanocomposites (Fe3O4, γ-Fe2O3) enables the rapid recovery of nanoparticles after the remediation process using low-gradient magnetic separation[61,77]. Silica, polymers, or carbonaceous shells can also stabilize magnetic cores, further enhancing stability, and allows functionalization with chelating ligands for selective adsorption. Superparamagnetic behavior, characterized by negligible remanence, enables dispersion and recovery. These materials can be regenerated via acid or salt desorption, maintaining performance over multiple cycles[66]. A highly effective and low-waste approach for the scalable application of magnetic composites is one in which adsorption or redox activity is combined with a magnetic core.
Mechanistic synergies and environmental controls
-
In real environmental systems, mechanisms do not operate independently. For example, graphene oxide and magnetite can be combined with biochar to simultaneously achieve adsorption, redox transformation, and recoverability: biochar reduces Cr(VI) while sorbing Cr(III)[66] in the form of secondary Fe-Cr hydroxides[70], which might precipitate concurrently (Fig. 4). Environmental parameters, including pH, redox potential, ionic strength, NOM concentration, temperature, and particle aging, govern the processes that occur and the duration of metal immobilization[36]. Reductions in reactivity or the maintenance of stability through processes such as sulfidation or hybridization are advantageous in controlling nanoparticle behavior. Subsequent field application requires leveraging mechanistic knowledge, including site-specific geochemical characterization and life cycle assessment, to optimize nanoremediation performance. Although individual environmental factors are often discussed in isolation, their relative influence on nanomaterial performance can vary substantially depending on site-specific conditions. As summarized in Table 2, pH and ionic strength primarily govern electrostatic interactions and surface complexation behavior, whereas natural organic matter and particle aging exert longer-term control by modifying surface reactivity, aggregation state, and redox capacity. Comparative evaluation of these parameters demonstrates that nanomaterial effectiveness observed under controlled laboratory conditions may not directly translate to complex natural systems without accounting for coupled environmental effects.
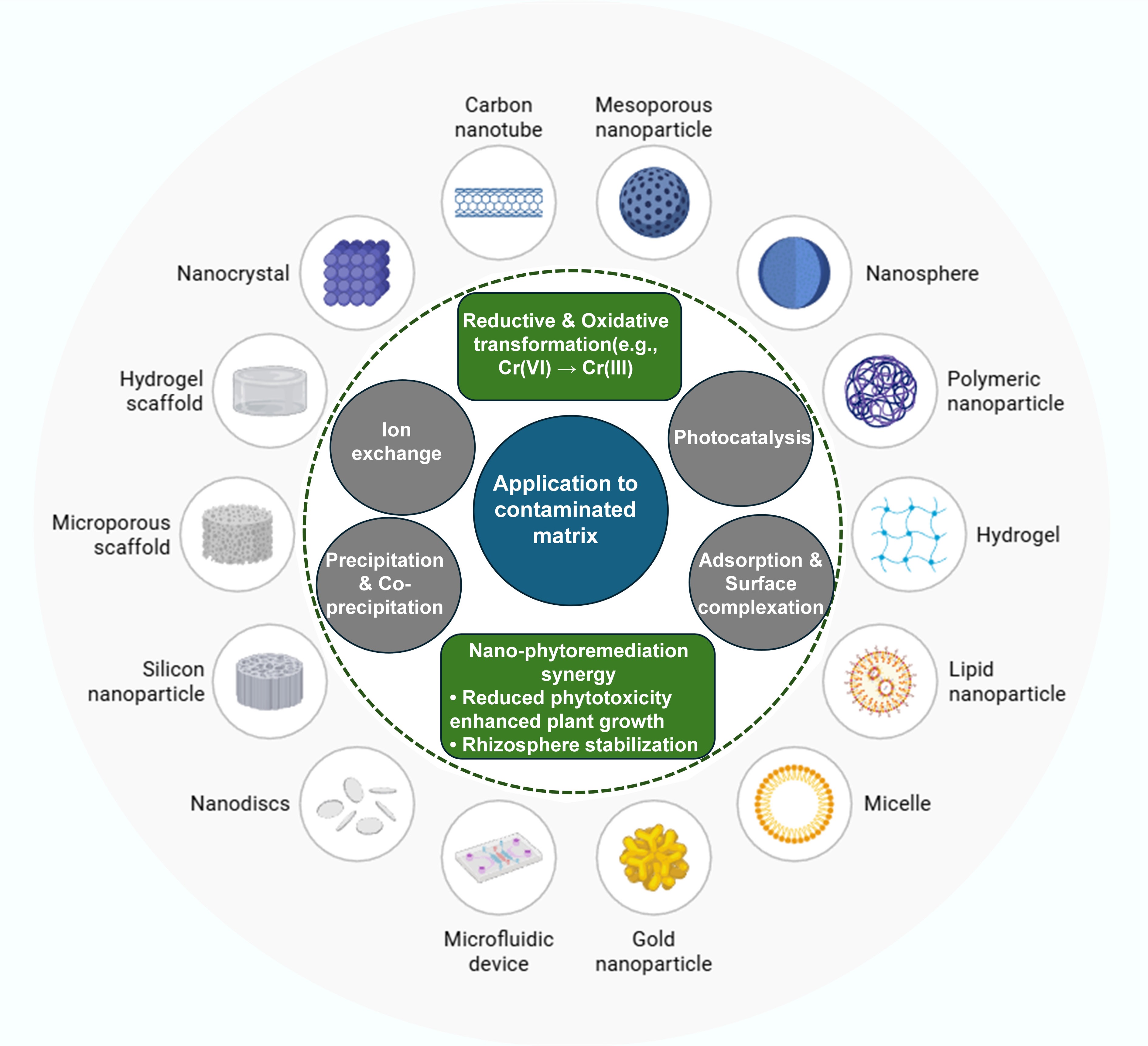
Figure 4.
Mechanisms associated with different nanomaterials for the remediation of inorganic pollutants.
Table 2. Influence of environmental parameters on nanomaterial-based heavy metal remediation
Environmental parameter Typical range studied Observed effect on removal efficiency Dominant mechanism affected Representative references pH 2–10 Decreased adsorption at low pH due to surface protonation; enhanced uptake at near-neutral pH Surface complexation, electrostatic interaction [78,79] Ionic strength 1–100 mM Reduced adsorption efficiency due to charge screening and competition Electrostatic attraction, ion exchange [80] Natural organic matter (NOM) 1–20 mg/L DOC Variable effects; often inhibits adsorption via site blocking and complexation Surface site competition, aggregation [81] Particle aging Weeks–months Decline in reactivity due to oxidation, aggregation, or surface passivation Redox activity, surface availability [82,83] -
Nanoremediation of soils is a process that uses engineered nanomaterials (ENMs) to immobilize, transform, or extract pollutants, primarily heavy metals and metalloids, from contaminated soils and sediments (Table 3). Nanoparticle-based soil remediation is usually employed to stabilize contaminants (immobilization) or to transform (transformation) them into less toxic and less mobile forms, followed by removal or long-term sequestration. Nanomaterials such as nZVI, nano-iron oxides, sulfide-based nanoparticles, carbonaceous nanomaterials (e.g., graphene oxide, carbon nanotubes), and hybrid composites—particularly biochar-supported nZVI and magnetic functionalized sorbents—have been engineered to accomplish these purposes. They are effective because of nanoscale properties, including high specific surface area, tunable surface chemistry, and enhanced reactivity, which allows them to adsorb contaminants effectively, undergo redox transformation, and nucleate secondary mineral phases within soil matrices[34,70].
Table 3. Representative engineered nanomaterials, their dominant removal mechanisms, target metals, and key performance features reported in recent studies
Engineered nanomaterials Predominant mechanistic pathways Target metal ions Key features and performance Key limitations Examples Ref. Carbon-based materials Adsorption via-surface functional-group binding, electrostatic attraction, and π–π interactions Cd(II), Hg(II), Pb(II), Cu(II), Co(II), and Zn(II) High metal-binding capacities (up to 1989 mg/g for Hg(II))
Rapid adsorption kinetics, short equilibrium times, and strong metal–functional group interactions
Surface locations' chemical and thermal stabilityExpensive; challenging recovery and reuse Multi-walled carbon nanotubes, graphene oxide nanosheets, and nano-biochar composites [49] Metal oxides Surface complexation, adsorption, and micro-precipitation Pb(II), Cd(II), Cr(VI), Zn(II), Cu(II), and Hg(II) High capacity for adsorption (up to
1,047.83 mg/g for Zn(II))
Superior reusability and regeneration potential
Adjustable surface chemistry and redox activity
High chemical stability and thermal resistance
Photocatalytic action under UV and visible lightAggregation in high ionic strength, limited selectivity, possible dissolution under acidic conditions Fe2O3, TiO2, Al2O3, ZnO [64] Nanocomposites/
hybrid nanoparticlesRedox and surface complexation; synergistic adsorption plus magnetic separation As(V), Pb(II), Cd(II), Cu(II), and Cr(VI) Rapid and effective magnetic recovery
Enhanced colloidal stability and dispersibility
Reusability in moderate temperatures
Increased surface area due to hybrid synergy
Multipurpose elimination of metals and organic pollutantsComplexity in synthesis, variable stability under environmental conditions, cost, potential secondary contamination TiO2–graphene composites,ZnO–polymer hybrids, Fe3O4@chitosan [85] Zero-valent metals Adsorption, precipitation, and redox transformation As(V), Cr(VI), Pb(II), Cd(II), and Cu(II) Fast reaction kinetics with little secondary pollution
High surface reactivity and electron transfer rate
Strong reducing capability toward toxic ions
Conversion of hazardous metals to less soluble or inert forms
Suitability for both soil and aquatic remediationRapid oxidation, aggregation, reduced long-term reactivity, possible toxic by-products Fe/Ni bimetallic nanoparticles, nZVI, and Fe/Cu core-shell systems [86] Biosorbents/functionalized polymers Ion exchange, adsorption, and surface complexation via −COOH, −NH2, −SH, and −OH Cu(II), Pb(II), Cd(II), and Hg(II) Strong binding through surface functional groups
Excellent regeneration and reusability
Low cost and sustainable materials
High selectivity toward target ions
Biodegradable and ecologically friendlyLower mechanical stability, potential biodegradation over time, slower kinetics compared to metallic nanoparticles Thiol-modified cellulose nanofibers, magnetic chitosan beads, and amine-functionalized biopolymer nanoparticles [60] Zeolites Micro-precipitation, ion exchange, and adsorption Cu(II), Zn(II), Pb(II), and Cd(II) High selectivity and ion-exchange capacity
Adjustable porosity and surface charge
Little efficiency loss during reuse; stability over a broad pH range
Efficacy in multi-metal systemsLimited adsorption capacity for certain metals, sensitive to pH changes, possible clogging in soil/water applications Clinoptilolite nanosheets, surfactant-modified zeolite composites, and nano-zeolite Y [87] Bio-nanocomposites Biocatalysis, biosorption, redox transformation (enzyme/microbe-assisted) As(V), Pb(II), Hg(II), and Cr(VI) Combines biological selectivity with nanomaterial reactivity
Transforms metals enzymatically or microbiologically;
Low toxicity and biodegradable
Sustainable, renewable, and regenerable
Functions well in complicated environmental matrices.Variable reproducibility, sensitivity to environmental conditions (pH, ionic strength), limited large-scale data Magnetite–microbe composites, laccase-immobilized nanoparticles, enzyme-loaded chitosan nanofibers [64] The most common mechanistic pathways of nanomaterial activity are the following: (i) adsorption and surface complexation of dissolved metal ions on reactive nanoparticle surfaces; (ii) redox conversion, such as the reduction of Cr(VI) to Cr(III) by nZVI, which converts mobile toxic forms to less soluble or less bioavailable forms; (iii) co-precipitation and nucleation, in which metals are integrated into new mineral phases on nanoparticle surfaces, and (iv) immobilization by sorptive supports, such as biochar, clay, graphene, and metal oxide nanomaterials. These nanomaterials have the potential to decrease or, in some cases, increase metal phytoavailability depending on surface functionalization and soil chemistry, demonstrating that nanomaterial-soil interactions are complex and species-dependent[84].
Delivery modes and engineering approaches for in-situ and ex-situ soil treatment
-
In practice, the nanoremediation of soils is implemented using various engineering approaches, depending on the site environment and remediation objectives. Surface amendment and mixing of nanoparticle-containing sorbent mixtures (e.g., biochar/nZVI, magnetic nanocomposites), are typically used to rapidly reduce bioavailability in superficial and nonuniformly contaminated soils. For deeper or stratified contamination, groundwater remediation may involve subsurface injection of nanoparticle slurries, with the transport and distribution of the particles and their reactions constrained by soil heterogeneity and high retention rates, often concentrated near injection points[34]. Another potential application in terms of permeable reactive barriers (PRBs) has demonstrated effective performance in locations adjacent to groundwater through the use of reactive nanoscale media, as well as in near-surface environments involving shallow soil-groundwater interfaces; however, their application in unsaturated soils remains challenging[34,35]. These limitations have motivated the development of engineered nanoparticle-based constructs (e.g., biochar-, silica-, and nZVI-supported nano-sorbents) designed to address engineering constraints, enhance mechanical mixing, reduce aggregation, and extend functional longevity in heterogeneous soil systems[66,70]. Field and laboratory studies have reported that biochar-supported nZVI can reduce extractable Cr (VI) and Pb concentrations by 60%–90% within weeks, while PRBs containing reactive nanoscale media can sustain 70%–85% removal efficiency of target metals over several months under varying soil conditions.
Performance outcomes: immobilization, reduction, and bioavailability reduction
-
Quantitative soil remediation assessments often involve measuring the reduction in: (a) extractable concentrations of metal in water; (b) operationally defined and functionally specific metal pools, such as DTPA-extractable fractions; and (c) plant uptake and/or leachate metal fluxes. Reductions in extractable and plant-available fractions, following iron-based amendments, particularly biochar–nZVI composites have been reported to be substantial in laboratory and greenhouse studies. For example, nZVI deposited on biochar has been shown to adsorb approximately 70%–90% of Cr(VI), and to significantly reduce pore-water concentrations and plant uptake of metals under controlled experimental conditions. These effects are commonly accompanied by a corresponding increase in the proportion of metals associated with residual and oxide-bound fractions, indicating enhanced immobilization and stabilization within the soil matrix[70,88]. Weaker, yet encouraging, outcomes have been reported in field and mesocosm experiments, where magnetite and magnetite-functionalized adsorbents reduced Pb migration and leachate concentrations in long-term cultivation systems, but did not significantly alter metal partitioning between soil solid phases and pore water[89]. These findings support the suitability of nanoscale amendments as rapid, in situ measures to mitigate immediate exposure risks, even though the magnitude and persistence of their effects vary considerably depending on soil chemistry and prevailing natural processes.
Case studies and field-relevant demonstrations
-
Although full-scale field tests of unconfined nanoparticles in soils have yet to be conducted because of regulatory issues, several experimental field tests and realistic mesocosm studies demonstrate their potential and limitations. Comparative studies using biochar-supported nZVI in outdoor lysimeters as well as pilot-scale plots in Cr-contaminated soils consistently showed decreases in water-soluble Cr and reduced crop phytoavailability over seasonal periods; however, performance declined in some cases due to nanoparticle aging and variations in local redox conditions[70]. Studies have reported that As-contaminated soils amended with nZVI and graphene oxide (nGOx), as well as other carbonaceous nanomaterials demonstrated that GO could either mobilize or immobilize As depending on its dosage and the properties of the soil; these contradictory results caution against uncritical extrapolation of laboratory-scale findings to field conditions[84]. Long term cultivation studies based on magnetite nanoparticles added to Pb-amended soils demonstrated reduced Pb concentrations in leachates and alleviated plant stress, but also showed alteration in soil microbial communities and, in some cases, increased plant Pb bioavailability, highlighting the ecology trade-offs[89]. Overall, these case studies indicate that although nanomaterials may provide quantifiable remediation benefits in soils, pilot testing, and monitoring at the site are necessary to confirm both effectiveness and environmental friendliness.
Synergies with phyto-management and combined remediation strategies
-
One promising use of soil nanomaterials is the integration of nano-remediation with phytoremediation or phytostabilization. As a soil amendment, nanomaterials can be applied to immobilize metals and simultaneously improve plant health (e.g., by releasing micronutrients or alleviating metal phytotoxicity), thereby supporting the establishment of cover crops or hyperaccumulators that stabilize the site and reduce the risk of erosion. Biochar-supported nZVI can be incorporated into the root zone to decrease the bioavailability of PTEs, and biochar further enhances the physical characteristics and water retention capacity of soils, which enables plant establishment. Conversely, there are nanoparticles (e.g., some metal oxides) that can enhance metal uptake by plants under certain conditions and, therefore, can be deliberately used to promote phytoextraction—the so-called nano-phytoremediation process, which must be strictly controlled to prevent excessive metal accumulation in food chains, particularly in cropping systems[31,34]. These hybrid approaches provide two strategies: (a) trap-then-plant strategies to achieve long-term sequestration and ecosystem restoration; or (b) induced phytoextraction aimed at metal removal followed by biomass harvesting and safe disposal.
Environmental fate, aging, and ecotoxicological considerations
-
Uncertainty regarding the environmental behavior of engineered nanoparticles in complex soil matrices is one of the primary concerns in field-scale nanoremediation. When introduced, reactive nanoparticles can be subjected to dynamic changes such as oxidation, sulfidation, coating by humic substances or encasing by secondary precipitates. Although such processes can reduce instantaneous reactivity, they can also have the effect of increasing the stability of particles and limiting mobility. As an example, nanoscale zero-valent iron (nZVI) quickly forms Fe oxyhydroxide shells, which not only sequester reduced metals but also passivate the surface, limiting further electron donation; the surface can also undergo sulfidation, stabilizing immobilized metals, and reducing long-term bioavailability[35,70]. These transformation pathways directly impact the persistence of remedial performance and the potential for unintended ecological effects. Over the long term, the fate of nanoparticles in soils is not well-constrained outside laboratory studies. Field research shows that Fe-based nanoparticles may remain relatively stable for several years, but gradually transform into more stable phases such as magnetite, goethite, or Fe-sulfides. These phases, however, can undergo reductive dissolution under changing redox conditions, which may either release previously immobilized metals or generate reactive intermediates. Long-term aging of ENMs under realistic environmental factors is required to determine the transformation kinetics and identify the durability of metal sequestration. In addition, the interaction of nanoparticles with organic matter may cause an increase in colloidal stability, resulting in off-site transportation, or aggregation, and deposition in soil horizons, forming focal reservoirs of nanoscale materials[36]. The trophic transfer and bioaccumulation are important, but largely unexplored ecological threats. Nanoparticles or products of their transformation may be incorporated by soil microorganisms, plants, and detritivores, and pass into terrestrial food webs. As an illustration, earthworms that have been subjected to nZVI or Fe3O4 nanoparticles have been demonstrated to absorb iron in their tissues, which can then be transferred to higher trophic levels such as birds or small mammals[13]. The nature of transfer, either whole particles, dissolved ions or biomolecule-coated aggregates, determines bioavailability and toxicity. Recent reports indicate that some carbon-based nanomaterials (e.g., graphene oxide) may adsorb to the surface of roots, be absorbed by plants, and be translocated to edible tissues, which is of concern to crop safety and human dietary intake[90]. Quantitative biomagnification factor studies of soil-plant-invertebrate-vertebrate chains are urgently needed for ecological risk assessment. The standard acute toxicity testing does not adequately predict the chronic exposure effects on non-target organisms. Nanomaterials can also cause sublethal oxidative stress, genotoxicity, reproductive impairment, and behavioral alteration in soil fauna during low-dose, long-term exposure. Communities of microbes needed to cycle nutrients and maintain soil health might undergo changes in diversity and activity; some researchers find that nitrifying bacteria or mycorrhizal fungi may be temporarily suppressed after the application of nZVI with recovery periods ranging from weeks to months[13]. Even with a decrease in metal uptake, chronic phytotoxicity can be observed through low rates of germination, distorted root architecture, or reduced photosynthetic rates. Such sublethal endpoints require ecotoxicological testing procedures, which are more extensive, involving multi-generational testing procedures and ecological functions (e.g., decomposition rates, enzyme activities) as opposed to organism survival alone. The interactions between the soil biota and organic matter are dose-dependent and bidirectional. Nanomaterials may change the physicochemical environment of the soil, influence nutrient availability, water retention, and the structure of the habitat. On the other hand, the nanoparticles may be covered by organic matter and microbial exudates in the soil, altering the surface charge, aggregation and reactivity. The context-specific interaction effects can vary: low concentrations of Fe3O4 can facilitate plant growth by correcting an iron deficit, but higher concentrations can cause oxidative stress or physically block root pores[13]. These complex interdependencies must then be captured within a finer graded assessment framework, increasing in complexity, as a single species test, to mesocosm experiments. To fill these knowledge gaps, future studies should focus on the long-term field studies that are accompanied by modern characterization methods (e.g., synchrotron-based spectroscopy, stable isotope tracing) to monitor the transformations of nanoparticles and the process of metabolization of metals in the field. The standardized ecotoxicological procedures need to be established, encompassing chronic exposure conditions, multi trophic level, and delicately sub organismic endpoints. Also, the life cycle and risk-benefit analyses between nanoremediation and conventional methods should consider the fate of residual nanoparticles, possible exposure of the food web, and recovery paths of ecosystems in addition to immediate efficacy. It is only under this extensive consideration that nanoremediation can be promoted as a technology that is environmentally sustainable, and that, in fact, minimizes tradeoffs between environmental integrity and the manipulation of pollution.
Water remediation
-
One of the most pressing environmental issues of the 21st century, is water pollution caused by heavy metals and metalloids. Compared to organic pollutants, heavy metals are non-biodegradable, persist in water bodies, and pose chronic risks to human health, aquatic life, and ecological activities. Traditional water treatment methods, including chemical precipitation, ion exchange, reverse osmosis, and coagulation are effective but limited by high operating costs, sludge generation, and low removal efficiency at low contaminant concentrations (below 1 mg/L). Nanoremediation has emerged as a disruptive technology in recent years, and aiming to overcome these drawbacks by exploiting the physicochemical reactivity, large surface area, and adjustable functionalization of ENMs[34,59,72]. The core principle of nanoremediation in aquatic systems involves multiple interrelated processes such as adsorption, reduction-oxidation (redox) reactions, co-precipitation, photocatalytic oxidation/reduction, and magnetic separation, which may occur individually or synergistically in aqueous matrices. The nanoparticles may be dispersed as a reactive suspension (to treat water in situ), immobilized on supporting matrices (to be used in filtration for ex situ and in columns), or embedded into hybrid systems (e.g., membrane-nanoparticle composite, fluidized beds). The choice of ENMs type and configuration depends on the chemistry of water, the target contaminants, and the environmental context (surface water, industrial effluent, or groundwater)[57,91]. Remediation in water is generally faster than in soil, due to greater accessibility of contaminants and fewer diffusion limitations. However, nanoparticle aggregation, dissolved organic matter (DOM) fouling, and variations in ionic strength can significantly reduce performance. Therefore, recent studies are focused on the development of stabilized, functionalized, or supported nanomaterials, which are capable of preserving colloidal stability and catalytic functionality in realistic water chemistries[61,70]. In aquatic environments, dissolved organic matter (DOM) exerts a 'double-edged sword' effect on the behavior and toxicity of both heavy metals and nanoparticles. On one hand, DOM can significantly mitigate metal bioavailability and promote remediation. This occurs through the formation of stable, soluble complexes with metal ions (e.g., Cd, Cu, Hg), effectively reducing their free ion concentration, which is the most bioavailable form[92]. Furthermore, DOM can also adsorb to nanoparticle surfaces, providing electrostatic, and steric stabilization that enhances colloidal dispersion and prevents aggregation and sedimentation, thereby increasing the potential for transport and dilution[93]. On the other hand, DOM can paradoxically exacerbate metal mobility and toxicity. It can act as a photosensitizer, generating reactive intermediates under solar irradiation that can dissolve metal-bearing minerals, releasing sequestered ions back into the water column. A critical, and often detrimental, mechanism is site competition, where DOM outcompetes toxic metals for binding sites on soil or sediment particles, inhibiting their natural sorption and immobilization[94]. Moreover, in anoxic zones, DOM can serve as a microbial substrate, fueling anaerobic respiration that leads to the reductive dissolution of Fe- and Mn-oxides, concurrently liberating co-precipitated As and other metals. Therefore, the specific chemical composition of DOM (e.g., aromaticity, molecular weight), its concentration, and the prevailing environmental conditions (pH, redox potential) are critical factors that determine whether its protective or facilitative role will dominate. Recent studies demonstrate that engineered nanomaterials, such as Fe3O4, TiO2, and biochar-supported nZVI, can remove 80%–95% of target heavy metals (e.g., Cr, Pb, As) from contaminated water within hours under optimized conditions, with removal efficiency strongly influenced by nanoparticle dose, contact time, and water chemistry[95].
Zero-valent iron nanoparticles (nZVI) and iron oxides
-
Among the various types of nanomaterials, nZVI is one of the most widely used for the removal of heavy metals in water because of its reductive and adsorptive properties. The Fe0 core serves as an effective electron donor, reducing toxic and soluble species, including Cr(VI), As(III), and U(VI), to less mobile oxidation states (Cr[III], As[V], and U[IV]), while the shell of iron (hydr)oxide formed during corrosion provides abundant cationic metal sorption sites[35,69]. The reduction reaction is coupled with the redox reaction, and it can be represented as:
$ \mathrm{Fe^0+Cr(VI)\to Fe^{2+}+Cr(III)} $ followed by the precipitation of Fe–Cr hydroxides. Research studies have documented removal of up to 95% of Cr(VI) and Pb(II) at the neutral pH when freshly prepared nZVI is present[69]. However, nZVI has real problems: it is very reactive and passivates on the surface (forms shells of oxides), aggregates into micro-sized particles, and cannot be easily dispersed in water. Such problems can be mitigated by surface design and composites. For example, starch, carboxymethyl cellulose, and polyacrylic acid layers enhance suspension stability and reduce aggregation via electrostatic and steric repulsion forces[96]. Biochar-, silica-, or activated carbon-supported nZVI increases the accessibility of the reactive sites, extends its life span, and improves transportability. Biochar-supported NZVI in particular has demonstrated stability and capability to immobilize both cationic (Pb, Cd) and oxyanion (As, Cr) pollutants over long periods of time in wastewater systems[70,88]. Another significant category of water remediation material is nanoparticulate iron oxide minerals such as Fe3O4, γ-Fe2O3, and their nanocomposites. Their magnetic properties enable easy separation after treatment, and the abundance of surface hydroxyl groups allows strong affinity for metal cations and oxyanions via complexation and ion exchange. Under optimized pH conditions, magnetite nanoparticles can adsorb up to 200 mg/g of Pb(II), and their magnetic retrieval minimizes the formation of secondary waste[61,77].
Carbon-based nanomaterials
-
Graphene oxide (GO), carbon nanotubes (CNTs), carbon dots, and biochar nanosheets are examples of carbonaceous nanomaterials that have gained enormous interest because of their diversity of structure and versatility of surface chemistry. The functional groups of oxygen (–COOH, –OH, –CO) on GO surfaces, strongly coordinate with heavy metals, and π–π and defect sites can also increase sorption. The sorption capacity of functionalized GO for Cu(II), Cd(II), and Pb(II) can exceed 300 mg/g, with fast kinetics attributed to monolayer complexation and electrostatic attraction[66]. Oxidized or aminated carbon nanotubes can exhibit superior adsorption capacities for Cd, Pb, Zn, and Hg species. Continuous-flow systems have now incorporated CNT-based membranes and composites, which have a high cleaning capacity and high mechanical stability. Nevertheless, high cost, potential cytotoxicity, and regeneration difficulties remain limiting factors[91]. Nano-biochar composites are intermediary materials between natural and engineered materials that provide cost-effective solutions for large-scale applications. Biochar in combination with metal or metal-oxide nanoparticles, not only offers porous support and stability, but also offers active functional groups which can form complexes. Biochar-supported nZVI hybrids are especially efficient in reducing and adsorbing Cr(VI) and Cu(II) in industrial wastewater, enabling simultaneous reduction and adsorption, with easy recovery via magnetic separation[70]. The photocatalytic nanomaterials, especially TiO2, ZnO, and doped forms, use light energy to create electron-hole pairs that can catalyze redox reactions. In the case of heavy metals, the electrons generated by photogeneration react with metal ions, and holes with organic co-contaminants, improving the water quality in general. With pure TiO2 however, solar can only be used in the UV range, which is quite limited[57,72]. The current advances revolve around plasmonic and heterojunction photocatalyst, like Ag/TiO2, Au/ZnO, and TiO2-g-C3 N4 composite, that exhibit better activities in the visible range and increased quantum efficacy. These systems have the ability not only to degrade organic chelating agents (e.g., EDTA) but also to reduce metal ions so that complexation-induced remobilization is prevented. The combined photocatalytic-adsorptive mechanism ensures long-term high removal efficiency, regardless of variations in light intensity and water chemistry[73].
Metal sulfide and bimetallic nanoparticles
-
Metal sulfide nanoparticles (e.g., ZnS, FeS, CuS) are particularly suited for removing soft metal cations like Hg(II), Pb(II), and Cd(II) through precipitation and surface complexation. Their low solubility products yield strong thermodynamic driving forces for sulfide formation, resulting in irreversible immobilization. Additionally, bimetallic nanoparticles (e.g., Fe–Pd, Fe–Cu) exhibit synergistic reactivity, with the noble metal component catalyzing electron transfer from Fe0, thereby accelerating reduction kinetics. Fe–Pd nanoparticles have demonstrated enhanced PTE reduction rates and improved resistance to passivation[69]. The positive aspect of nanoremediation bioreactors in water systems is that nanoparticles can be recovered magnetically, which reduces secondary pollution. Magnetic nanocomposites consist of iron oxide cores with functional materials (silica, polymers, carbon), providing high selectivity and easy recovery. Following adsorption, contaminants may be desorbed using acid or chelating solutions, allowing the nanoparticles to be reused for multiple consecutive cycles, with minimal loss in capacity[61,77]. As an illustration, amino-functionalized Fe3O4@SiO2 nanoparticles maintain more than 80% of their adsorption capacity following five regeneration cycles to eliminate Pb(II) and Cu(II).
Hybrid and coupled systems
-
The rational design of hybrid and coupled systems, involving a set of functional components and remediation mechanisms, is the most promising future direction for nanoremediation in aquatic systems. These artificial systems are able to address the inherent weaknesses of single-mechanism systems by establishing synergistic interfaces in which adsorption, redox transformation, photocatalysis, and magnetic separation can co-exist or occur sequentially, thereby increasing overall efficiency, material stability, and operational sustainability[53,85]. Advanced interfacial processes determine the improved operation of hybrid nanomaterials. The carbonaceous network acts as the electron conduit and a dispersant in composite systems like biochar-supported nanoscale zero-valent iron (BC-nZVI) or reduced graphene oxide-Fe3O4 hybrids. This architecture enables quick transfer of electrons from the reactive metallic core (e.g., Fe0) to contaminants fixed at the surface, which makes the reductive conversion of redox-active metals. At the same time, the carbon support with a high surface area offers a large number of sites to complex and immobilize cationic species (e.g., Pb, Cd), and to simultaneously eliminate oxyanions and cations[70,88]. In addition, the rapid passivation of the metal core is suppressed by the carbon shell that increases the reactive life of the material in complicated aqueous matrices. An example of magnetic-photocatalytic hybrids, including Fe3O4/TiO2 and Fe3O4/g-C3N4, can be considered the combination of the transformation of contaminants and their easy recovery. The photocatalytic element (TiO2, g-C3N4) in these systems absorbs light energy and creates electron-hole pairs. The electrons generated by the photogeneration are used to reduce metal contaminants such as Cr(VI), and the holes can be used to degrade organic pollutants or react with water molecules that co-exist to generate reactive oxygen species, further modifying contaminant speciation[97]. The magnetic core (Fe3O4 or g-Fe2O3) inside the nanocomposites allows the quick separation of the spent nanocomposites from the treated water with an external magnetic field, addressing a severe issue of nanoparticles recovery and preventing secondary contamination[61,77]. This photocatalytic activity, coupled with magnetic recovery, is the key to designing reusable water treatment technology based on catalysts. Another emerging paradigm is the integration of nanoparticles into membrane systems. In this case, nanomaterials (e.g., functionalized CNTs, ZnO) or nZVI are incorporated into or surface-grafted on polymeric membranes. This design combines the size-exclusion capabilities of the membrane with the selective adsorption or catalytic activity of nanoparticles. Such membranes can effectively capture ions of heavy metals while removing organic chelators that may increase metal mobility and thereby providing a barrier to contaminant breakthrough[91]. Recent developments focus on stimuli-responsive or antifouling nanocomposite membranes that are highly permeable and selective under dynamic flow conditions. Finally, hybridization is a strategy to develop multifunctional systems through complementary engineering of properties. These systems are characterized by high reactivity, recyclability, and selectivity, integrating sorption potential of biochar, redox activity regulated by nZVI, photocatalytic activity (of semiconductors), and magnetic retrievability. They are also quite useful in overcoming practical challenges such as nanoparticle aggregation, uncontrolled environmental release, and loss of reactivity; thereby providing a more feasible pathway to translate nanoremediation technologies from a controlled laboratory setting to complex biogeochemical treatment environments.
Plant-based systems: nano-assisted phytoremediation—concepts and mechanisms
-
Plant-based remediation (phytoremediation) involves a bioremediation technique which involves utilizing natural plant capabilities for the recovery, fixation, stabilization or even volatilization of soil, sediment, and water pollutants. Nanotechnology supplements these plant processes in two ways: (1) via nano-enabled amendments to modify contaminant speciation and bioavailability in the rhizosphere (e.g., nano-adsorbents that immobilize labile metal fractions or nano-reducing agents that reduce toxic oxyanions to less mobile forms); (2) via direct nanoparticles-plant interactions, where engineered nanoparticles (ENMs) are used to alter plant physiology, root architecture, nutrient status, and transporter expression, thereby modifying metal uptake (Table 4).
Table 4. Comparison of nanomaterial performance in soil, water, and plant-based remediation systems, including removal efficiencies and influencing factors
Matrix Typical nanomaterials for remediation Removal efficiency Key influencing factors Matrix-specific challenges and practical caveats Ref. Plant-based and nano-phytoremediation (plant uptake + rhizosphere) TiO2, ZnO, carbon-based nanomaterials (graphene oxide, CNTs), coated magnetic nanoparticles, functionalized Fe nanoparticles (nFe), and nanofertilizers. Plant uptake showed variable increases (10%–300%) over plants alone, while absolute removal efficiency per area remains lower than engineered water or soil treatments; NP-assisted growth can further enhance remediation. Plant factors: Growth rate, stress tolerance, internal antioxidant systems, root architecture, exudates, and plant species (hyperaccumulator vs. high biomass) all influence nanoparticle mobility and bioavailability
Nanomaterial factors: NP type, dose, and coating influence phytotoxicity vs growth stimulation and can enhance contaminant bioavailability or nutrient delivery, improving overall plant-based remediation efficiency.Strengths: low-cost, sustainable, capable of increasing biomass and restoring vegetation.
Caveats: delayed timeline; variability between lab and field; very species- and dose-dependent; risk of NP accumulation in consumable crops and food chains.[98,99] Water remediation (engineered reactors, wastewater, and effluent) nZVI and iron oxides, TiO2 (photocatalysts), ZnO, graphene oxide (GO), carbon nanotubes (CNTs), magnetic Fe3O4 composites, and NPs coated with polymers or chitosan. Heavy metals achieved high removal efficiency (70%–100%), often ≥ 90% for Pb, Cr, and Cu; efficiency can decrease at high contaminant concentrations or upon reuse of nanomaterials, while adsorbents show tens–hundreds mg/g capacity in well-mixed reactors. Environmental factors: pH, temperature, contact time, ionic strength, and competing ions all have an impact on performance; advantages include recoverable magnetic nanoparticles and quick kinetics; disadvantages include fouling, the necessity for NP recovery, and reduced pilot-scale efficiency.
Nanomaterial properties: Size, surface area, charge, stability, functionalization, and dosage influence removal efficiency.Strengths: controlled reactors, recoverable magnetic nanoparticles, and superior contact/kinetics.
Caveats: fouling, NP recovery/regeneration requirements, environmental release risks, and reduced efficiency in pilot-scale or continuous operations compared to batch tests.[86,98,100] Soil remediation (groundwater, ex situ treatment, and in situ amendments) Zeolite-like nanoadsorbents, metal oxide NPs, carbon NMs, biochar-supported NPs, and nZVI (raw and supported/coated). Heavy metals exhibited moderate to high removal efficiency (20%–98%), but performance is highly variable depending on soil properties, species, and nanoparticle application method; long-term stability and NP aggregation can also limit effectiveness. Soil properties:
Contaminant availability and NP behavior are influenced by pH, organic matter, texture, water content, redox potential, and microbial activity.
Nanomaterial properties: Mobility and removal efficiency are influenced by NP stability, dispersion, aggregation, age (oxidation/sulfidation), dose, spatial delivery, and interactions with soil bacteria.Strengths: nZVI can break down some organics and reduce/immobilize metals.
Caveats: limited NP transport (aggregation, filtration), strong influence of soil texture and organic matter, possible long-term ecotoxicity, and lower field efficiencies.[98,100] There are a variety of mechanistic ways in which nano-assisted phytoremediation works. Firstly, the nano adsorbent binds to dissolved metal species in soil via Fe oxides, nano biochar-based nZVI, functionalized graphene derivatives, thereby reducing the number of free ions that could be absorbed by the roots to minimize phytotoxicity. This pinning down may be needed in case the remediation goal is phytostabilization, where the focus is on maintaining vegetation cover, while food-chain transfer is of lesser concern[70]. Secondly, oxyanions (Cr[VI], Se[VI]) are carcinogenically catalyzed and converted to less mobile forms Cr(III), Se(IV) or Se(0) with the assistance of redox-active nanoparticles (especially, nZVI or sulfide-bearing nanoparticles), thereby reducing phytoavailability[35,70]. Third, to achieve the dual goals of increasing metal solubility and promoting root establishment, nanoparticles can be applied to enhance phytoextraction. For example, nano-fertilizers or biochar-based nano-amendments (e.g., biochar nanosheets) can enhance root exudation, temporarily acidify the rhizosphere, and facilitate the controlled movement of metal complexes toward roots, thereby improving uptake by hyperaccumulators or specialist cultivars for harvestable biomass[31].
Nanoparticle uptake, translocation, and plant physiological responses
-
Understanding the interactions of ENMs with plant roots and their subsequent uptake into tissues is critical for predicting their biological impacts and environmental risks. The initial uptake of nanoparticles mainly occurs at the root surface, primarily via apoplastic and symplastic pathways. Epidermal cell walls are root barriers that enable passage of small nanoparticles (generally less than 50–100 nm, depending on species and root ultrastructure) across the apoplast; to cross the endodermal Casparian strip and enter the vascular system, the particle or dissolved ionic species must undergo membrane transport processes, as particle size, cellular surface charge, and functionalization have an effect on passage[90]. Endocytosis, via the vesicle trafficking route, and carrier-mediated uptake of dissolved metal ions released during nanoparticle dissolution, are two of the major cellular processes involved in internalization processes. Upon internalization, nanoparticles may either stay in the form of particulate matter after internalization, dissolve to release metal ions, or be covered with plant biomolecules (proteins, organic acids), thereby altering bioavailability and toxicity. Translocation into aerial tissues is primarily dependent on the loading of xylem and the transpiration stream. Metals complexed with low-molecular-weight ligands (e.g., citrate, malate, phytochelatin complex, and others) are easier to transport during the translocation process since it involves small particulate forms, as opposed to the large ones. Phloem movement does not generally produce productive particles, but can help in the redistribution of metals that are complexed with mobile organic ligands[90,101].
The physiological response of plants to nanoparticle exposure is dose- and material-dependent. Low concentrations of nanomaterials may be growth-stimulating (due to the provision of micronutrients and the inhibition of metal stress) or growth-promoting (due to the induction of oxidative stress, affecting membrane integrity, enzyme activity, and hormonal changes)[36]. Therefore, nano-phytoremediation requires careful optimization, balancing beneficial and adverse effects through appropriate selection of nanomaterial type, dosage, and timing of application in relation to plant phenological stage and seasonal timing.
-
Phytoremediation modalities can be broadly classified into classical phytoremediation and nano-assisted phytoremediation. Classical phytoremediation comprises traditional plant-based remediation approaches, while nano-assisted phytoremediation integrates nanomaterials to enhance the efficiency of this classical redundancy. Thus, nano-assisted phytoremediation may be viewed as an advanced extension of classical phytoremediation modalities.
Phytostabilization (immobilize + revegetate)
-
In this method, the nano-amendments that are employed act as primary blockers of the metals, typically the iron oxides, biochar particles, or phosphate-surface-functionalized nanomaterials to block the labile fraction of the metals and allow protective vegetation. The nano-amendment acts as a root zone long-term sorption sink, while plants reduce surface erosion as well as the transportation of dust. Biochar-wrapped nZVI used as a root-zone amendment has been reported to reduce the phytotoxicity of Cr and Pb and to support the naturally growing grass and cover crops in the laboratory, greenhouse, and lysimeter experiments[70,88]. Where long-term stability of immobilized species is required, sulfidation iron-based materials are utilized to promote the formation of less mobile metal sulfides. Such sulfidated iron-based materials provide enhanced long-term stability by converting labile metal species into low-solubility metal sulfides, thereby minimizing remobilization under fluctuating pH and redox conditions.
Phytoextraction (mobilize + harvest)
-
Nanomaterials may be experimentally used to enhance uptake through temporary phytoavailability (via chelation, rhizosphere acidification, or surfactant-like effects) in hyperaccumulator plants. This method can be utilized in situations where the harvested biomass may be disposed of or recycled into metal (phytomining processes). Another type of tool is ENMs preparations and nano-chelators in the development of transient soluble target PTEs and reducing off-site transportation is directed to phytoextraction[31]. Mechanistically, nano-chelators and reactive nanoparticles bind soil-bound metals by the formation of labile metal-ligand complexes or by modifying rhizosphere pH and redox microenvironments, which enhances metal desorption from mineral and organic surfaces. These temporarily mobilized metal species are concentrated at the root zone, thereby increasing membrane transport and xylem loading in hyperaccumulator plants. Also, the short residence time of soluble metal-nano complexes decreases downward leaching and lateral movement, which directs metals toward plant uptake as opposed to off-site dispersion. However, the need to increase the level of the metal absorbed in the aboveground tissues requires strict control to ensure that it does not enter the food chain, especially in croplands.
Phytovolatilization and transformation
-
In a few instances (e.g., certain forms of mercury), where such elements can be volatilized (e.g., some types of mercury), nanomaterials which change the interactions of plant metabolism or the methylation/demethylation processes of microbes can indirectly influence the volatilization rate. Due to the ecological and regulatory complexity of the volatilized contaminants, the pathway is selected with great caution and usually comprises a small part of the nano-phytoremediation plans. The rhizosphere microbiome plays a central role in metal cycling, speciation, and plant health to a much greater extent. Depending on the case, nanomaterials impact microbial communities, killing sensitive groups in some cases and boosting the growth of metal-transforming groups in others. Metal-immobilizing nanoparticles have the potential to reduce microbial toxicity and provide a positive effect on beneficial microbes, and, therefore, certain other ENMs can become an antimicrobial activity that can potentially disrupt symbiotic relationships[13,36]. A new trend that could be considered in the future is the application of nanomaterials in collaboration with plant growth-promoting rhizobacteria (PGPR) and mycorrhizal fungi. This approach aims to regulate bioavailability by using nanomaterials to enhance microbial growth. The results of controlled experiments indicate the possibility of using the positive impact of joint PGPR-biochar-nZVI additions to increase plant biomass and reduce shoot metal concentrations, but show a high degree of dependence on the context[31,90].
Successful field deployment of nano-phytoremediation requires integrative design that addresses the following elements: (1) site characterization including speciation and distribution of metals, pH, redox status, soil texture, and baseline ecological conditions; (2) selection of plant species or cultivars (hyperaccumulators, metal-tolerant grasses, or deep-rooted perennials) matched to climate and remediation objectives; (3) selection and formulation of nanomaterials (supported vs free nanoparticles, stabilized colloids, or pelletized amendments) optimized for target chemistry and delivery; (4) application method and timing (surface incorporation prior to planting, root-zone banding, repeated low-dose applications vs single high-dose application); (5) monitoring framework including labile metal measures (DGT, pore-water sampling), plant tissue analysis, ecotoxicological endpoints, and tracking of nanoparticle fate (transformation, persistence, potential trophic transfer); and (6) stewardship and contingency planning for potential remobilization or unexpected ecological impacts[13,31]. The increasing use of nano-phytoremediation methods and integrated soil–water–plant systems has been demonstrated effectively in pilot-scale and mesocosm studies. Examples include biochar-supported nZVI for chromium-contaminated agricultural soils: outdoor lysimeter and pilot-plot experiments applying biochar–nZVI to Cr(VI)-contaminated soils showed reductions in pore-water Cr concentrations, DTPA-extractable pools, and crop uptake within a single cropping season, allowing cover crops to be used safely. With aging, the development of Fe-oxide shells on nZVI progressively reduced its reductive potential, while sorptive immobilization was maintained[70].
Nano-enabled phytoextraction using hyperaccumulators in field trials combining EDTA-free nano-chelators and segregated harvest of metal-enriched biomass demonstrated increased Zn and Cd uptake into Brassica spp. shoots, enabling more efficient biomass removal with lower chelator dosing and reduced leachate risk compared to conventional chelator-assisted phytoextraction[31]. Integrated groundwater–plant treatments using nZVI and riparian vegetation were also tested: in a pilot study at a Cr-contaminated aquifer, targeted nZVI injections in the hyporheic zone, combined with deep-rooted riparian species, reduced aqueous Cr concentrations and limited downstream dispersion through combined reductive immobilization and vegetative uptake of residual mobile fractions[35]. Similarly, combined nanoparticle amendment and PGPR inoculation for mine tailing revegetation, using iron oxide nanoparticles, composted biosolids, and PGPR, improved seedling survival, increased plant biomass, reduced metal uptake in shoots, and accelerated recovery of soil microbial diversity compared to controls, indicating synergistic benefits for ecosystem restoration[13,31].
Despite these promising results, several caveats remain. Outcomes may vary due to differences in soil chemistry and hydrology, and ecological trade-offs such as microbial community shifts or potential trophic exposure must be carefully monitored and managed over the long term. Although nanomaterials frequently demonstrate high removal efficiencies under controlled laboratory conditions, their performance often declines as systems transition toward mesocosm and field scales. Table 5 compares representative nanoremediation studies across experimental scales, highlighting systematic reductions in removal efficiency, changes in nanoparticle behavior, and emerging transport and stability constraints under increasingly complex environmental conditions.
Table 5. Comparison of nanoremediation performance across laboratory, mesocosm, and field scales
Scale Typical system and conditions Nanomaterials evaluated Removal efficiency Observed nanoparticle behavior Key limitations Ref. Laboratory Batch reactors; controlled pH, ionic strength; synthetic solutions nZVI, Fe3O4, GO, TiO2 80%–99% metal removal High reactivity; minimal aggregation Overestimation of performance; lack of heterogeneity [49,78] Mesocosm Soil columns, lysimeters, outdoor tanks; semi-natural conditions Biochar–nZVI, Fe oxides, hybrid composites 50%–85% reduction in bioavailable metals Partial aggregation; surface passivation Scale sensitivity; aging effects [70,102] Field In situ soil or aquifer treatment; heterogeneous matrices nZVI slurries,
magnetic composites30%–70% reduction
over monthsStrong aggregation; oxidation; limited transport Delivery constraints; cost; regulatory limits [30,82] -
Geochemical and physicochemical factors that control metal speciation, mobility, and the stability of nanoparticles are the key determinants of the environmental fate and reactivity of heavy metals, as well as the effectiveness of nanomaterial-facilitated remediation (Fig. 5). These parameters dictate how contaminants interact with soil and water matrices, govern the modification pathways, and ultimately define the effectiveness of the remediation interventions[2]. The ecological and toxicological behavior of heavy metals is not a matter of overall concentration, but of chemical speciation of the metals. The process of determining the different forms an element can take, such as free ions, inorganic/organic complexes, adsorbed phases, or precipitated minerals—each with its own solubility, bioavailability, and stability—is known as speciation[103]. These differences underscore the importance of speciation analysis for risk assessment and the design of effective remediation strategies. The major factors controlling metal speciation are pH, Eh, and the presence of ligands, in addition to organic matter. Cationic metals like Cd(II) and Pb(II) are more soluble in acidic conditions and precipitate and adsorb on surfaces in alkaline conditions. On the other hand, low pH values of the oxide surface show a higher sorption of oxyanions like AsO43− or CrO42−[2]. The redox oscillations change the oxidation states and hence the toxicity and solubility of metals, reducing conditions liberate Fe(II) and oxidizing conditions stabilize the Fe(III) oxides that are capable of sequestration of PTEs. Stronger spatial and temporal heterogeneity in metals results from microbial and biogeochemical alterations, including microbial methylation of Hg(II) or As.
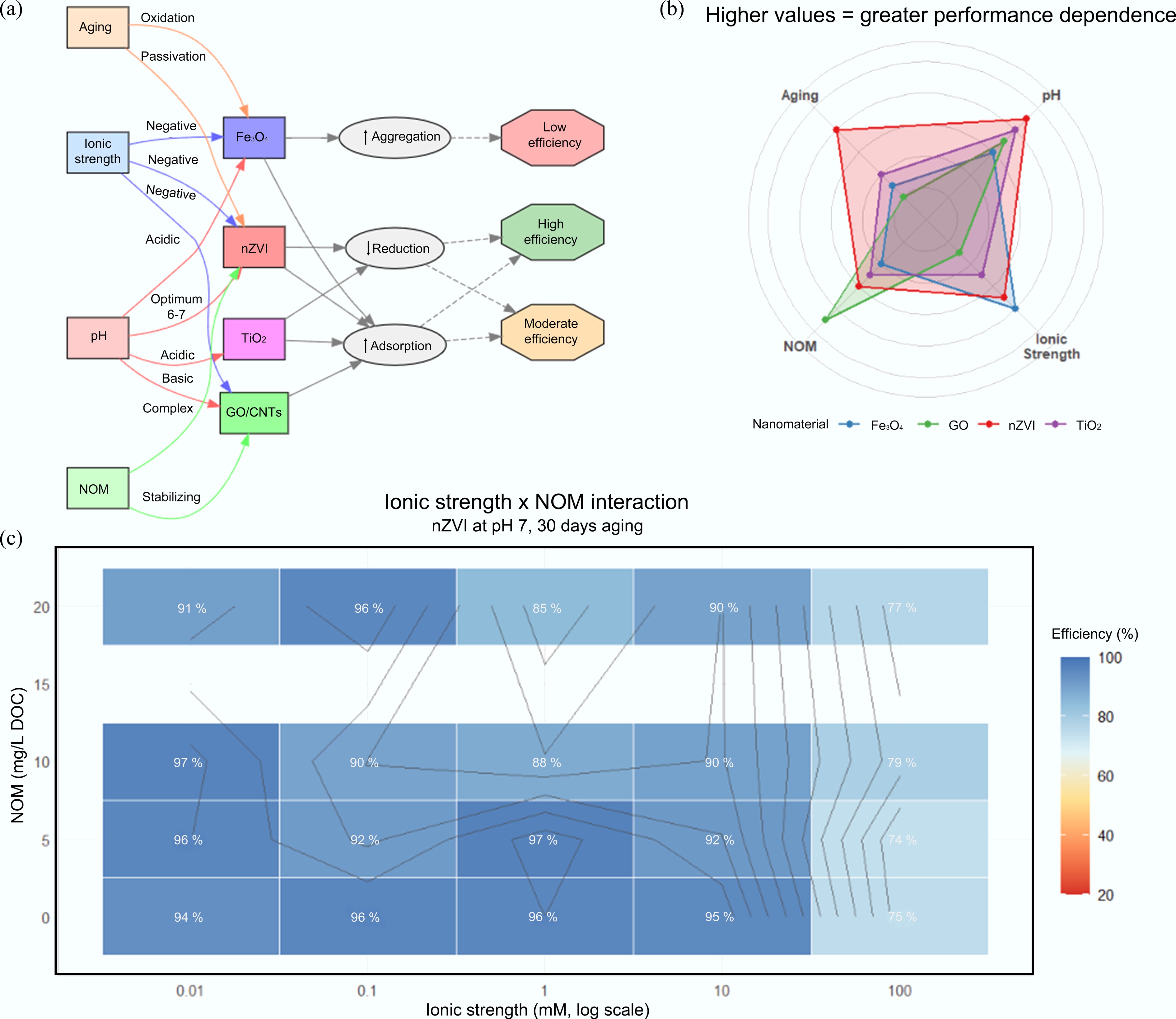
Figure 5.
Environmental parameter controls on nanomaterial remediation efficiency. (a) Schematic overview of key parameter-nanomaterial-performance relationships: pH critically governs nZVI reactivity (optimum ~6 to 7), ionic strength generally suppresses performance via enhanced aggregation, natural organic matter (NOM) exerts complex (stabilizing or competitive) effects, and aging drives progressive surface passivation. (b) Radar chart comparing relative sensitivity of four common nanomaterials to each parameter; radial distance indicates degree of performance dependence (nZVI shows highest sensitivity to pH and aging, while graphene oxide/carbon nanotubes [GO/CNTs] are most affected by NOM). (c) Interaction heatmap for nZVI at pH 7 after 30-d aging, showing how elevated ionic strength and NOM jointly depress removal efficiency, with strongest inhibition occurring under high ionic strength (> 10 mM) coupled with high NOM (> 15 mg/L DOC).
A comparative study of environmental factors reveals that pH plays a key role in regulating solubility and selectivity of adsorption of various metals, whereas Eh is a determining factor for the redox-driven transformation and immobilization ability of nanoparticles. Natural organic matter effects are material-specific, stabilizing certain nanoparticles, e.g., Fe3O4, and inhibiting others, e.g., nZVI. Aggregation is affected by ionic strength, and supported nanomaterials are more effective in high-salt soils than their bare counterparts. The relative significance of each factor thus depends on the nature of nanoparticles and contaminants. As an example, Fe3O4 nanoparticles are most effective in moderately acidic soils with moderate NOM, and nZVI is most effective in reducing conditions with low NOM to optimally remediate Cr (VI). Integrating these insights allows for the tailoring of nanomaterial selection and dosing to site-specific conditions, enhancing both remediation efficiency and ecological safety.
Reactivity and transformation of nanomaterials in remediation systems are also determined by the geochemical drivers that determine the speciation of metals. For example, Fe3O4 nanoparticles achieve optimal adsorption in slightly acidic conditions due to the electrostatic force between protonated surfaces and Cr(III) ions with negative charges[72]. Similarly, the redox reactivity and corrosion of nZVI at acidic to neutral pH conditions enhance the reduction and transformation of contaminants[35,70]. The Eh is also a determinant of the contaminant speciation and controls the transformation and stability of the nanoparticles themselves. Under reducing environmental conditions, Fe0 nanoparticles react to form Fe hydroxides and sulfides that fix metals by co-precipitation and surface sorption. Nevertheless, these reduced species may be re-oxidized under oxidative conditions, which may liberate previously immobilized metals. Thus, the field performance of nZVI is highly dependent on local pH and Eh conditions, and site-specific characterization is necessary prior to application[35]. On the one hand, humic and fulvic substances can stabilize nanoparticle dispersions in water and soil systems through steric or electrostatic repulsion thereby preventing aggregation and enhancing mobility[36,61]. NOM might occupy the adsorption sites of target metals, form soluble metal organic complexes that reduce bioavailability, and occupy reactive surface sites, thereby reducing overall efficacy[66]. Overall, the effect is dispersive and reactive at low concentrations, whereas at high concentrations it inhibits contaminant removal, depending on NOM concentration and system chemistry.
-
The effective transport and delivery of ENMs in soils and sediments remain major challenges for in situ nanoremediation. In heterogeneous porous media, ENMs are prone to aggregation and deposition due to van der Waals attractions, magnetic interactions, and electrostatic screening, which substantially reduce their mobility and reactive surface area. Soil texture, pore-size distribution, mineral composition, and ionic strength strongly influence nanoparticle retention through straining, attachment to grain surfaces, and filtration at pore throats. In addition, natural organic matter may either enhance mobility through steric stabilization or promote aggregation via bridging effects, depending on its composition and concentration. These processes often result in limited subsurface distribution and uneven treatment zones, thereby constraining the effectiveness of field-scale nanoremediation relative to laboratory observations.
Moreover, a significant constraint to nanoremediation at field scale is nanoparticle aggregation. The enhanced speed of agglomeration of particles in question when powered by electrostatic and magnetic forces reduces the surface area, reactivity, and limits subsurface transportation[34]. Aggregation is known to be caused by high ionic strength and divalent cations (Ca[II], Mg[II]), and organic coating or polymeric stabilizers (e.g., carboxymethyl cellulose, polyethylene glycol) can be used to maintain dispersion and enhance particle mobility[69]. However, strong coatings may inhibit the exchange of electrons, thereby creating a trade-off between stability and reactivity[70]. Supported nanomaterials, such as nZVI on biochar or silica supports, are an intermediate that can prevent aggregation, aid in recovery, and remain reactive over time[88].
The concept of environmental safety is receiving increasing attention in nanoremediation research. Nanoparticles may cause toxicity through either reactive oxygen species, releasing metal ions, or by physical interactions between particles and microbial and plant membranes. Nanomaterials with iron are commonly considered environmentally safe, and they will become natural iron oxide and iron hydroxide, yet local concentrations may lead to oxidative stress and alter the structure of the microbial communities[13,36]. The use of less dispersible nanomaterials can be assisted by functionalizion with responsible design, and lower tiers of ecotoxicology of soil and water living organisms.
Beyond short-term toxicity, long-term ecological risks associated with nanoremediation remain insufficiently understood. Engineered nanomaterials introduced into soils and sediments may persist for extended periods, undergo gradual transformation, or accumulate in specific environmental compartments, raising concerns regarding chronic exposure and trophic transfer. Several studies have reported the uptake of metal-based nanoparticles or their transformation products by soil microorganisms, plants, and benthic organisms, with potential propagation through food webs. Although iron-based nanomaterials are generally considered less hazardous due to their eventual transformation into naturally occurring iron oxides, prolonged exposure may still alter microbial community structure, enzyme activity, and nutrient cycling. These findings highlight the need for long-term monitoring, life-cycle assessment, and ecotoxicological evaluation under environmentally realistic conditions before widespread field deployment of nanoremediation technologies. Laboratory studies have shown that field demonstrations with nZVI slurries used in pilot projects have been able to bring contaminated aquifer levels of Cr(VI) and As(V) to acceptable regulatory levels in several months[69]. Similarly, the Fe3O4 and TiO2-g-C3N4 composites have been engineered to be stable and operational in the wastewater treatment systems, having effective recovery and minimal amounts of secondary waste in the application containing biochar supports[57].
-
Despite significant advances in nanoremediation research, large-scale field implementation remains constrained by regulatory uncertainty and policy gaps. Most environmental regulations governing soil and water remediation were originally designed for conventional chemicals or bulk materials and lack explicit provisions for engineered nanomaterials (ENMs). Consequently, nanoremediation technologies are often evaluated under existing chemical risk assessment frameworks that may not adequately capture nanoparticle-specific properties such as size-dependent reactivity, dynamic transformation pathways, environmental mobility, and long-term persistence[104,105]. This regulatory mismatch creates ambiguity in permitting, liability, and monitoring requirements, slowing the translation of promising laboratory results into field-scale applications. The current regulatory landscape across major jurisdictions reveals a patchwork of approaches with limited harmonization. In the European Union, nanomaterials fall under the Registration, Evaluation, Authorization and Restriction of Chemicals (REACH) regulation, which mandates case-by-case risk assessment but lacks standardized testing protocols tailored to environmental remediation scenarios. While recent amendments require enhanced reporting for nanoforms, specific guidance for evaluating in situ remediation—particularly regarding particle mobility, transformation products, and ecosystem-scale effects—remains underdeveloped[106]. In the United States, oversight is primarily managed by the Environmental Protection Agency under the Toxic Substances Control Act (TSCA). Although TSCA's 2016 amendments include provisions for new chemical notifications involving nanomaterials, clear protocols for field deployment, long-term monitoring, and site closure criteria for nanoremediation projects are not yet formalized. Similarly, in Canada, nanomaterials are assessed under the Canadian Environmental Protection Act, but existing guidelines do not differentiate between remedial nanomaterials and industrial nano-additives, creating uncertainty in regulatory classification and data requirements.
Critical policy gaps and data needs must be addressed to enable responsible deployment. Key shortcomings include:
• Standardized characterization protocols: Existing guidelines (e.g., OECD, ISO) focus on material identification, but lack environmental-behavior metrics such as aging-dependent reactivity, colloidal stability in natural matrices, and transformation kinetics under field conditions.
• Tiered risk assessment frameworks: Current chemical-based models inadequately address nanoparticle-specific exposure pathways (e.g., trophic transfer, incidental ingestion by soil fauna) and dose-response relationships that may be non-linear or influenced by particle coating and aggregation state.
• Post-application monitoring requirements: Few regulations stipulate duration, frequency, or endpoints for monitoring nanomaterial fate and residual ecological impacts after remediation, creating uncertainty about long-term stewardship and liability.
• Cross-jurisdictional inconsistencies: Divergent definitions of 'nanomaterial', differing data requirements, and varying acceptable risk thresholds hinder technology transfer and multinational site remediation efforts.
Emerging guidelines and voluntary frameworks offer preliminary pathways toward regulatory clarity. Organizations such as the International Organization for Standardization (ISO) and ASTM International have developed technical standards (e.g., ISO/TR 11360:2021 on nanotechnology-environmental risk assessment; ASTM E3247-20 on terrestrial ecotoxicity testing for nanomaterials) that provide methodologies for environmental testing. Similarly, the European Chemicals Agency's (ECHA) 'Guidance on Information Requirements and Chemical Safety Assessment for Nanomaterials' outlines data expectations for persistence, bioaccumulation, and toxicity (PBT) assessment, though its application to remediation-specific scenarios remains interpretive. Voluntary stewardship programs, such as the Nano Risk Framework developed by the Environmental Defense Fund and DuPont, encourage proactive hazard evaluation and lifecycle thinking but lack enforcement mechanisms.
Towards adaptive and risk-proportionate regulation, several policy innovations could accelerate responsible deployment:
• Permitting-by-exception models: For well-characterized, low-mobility nanomaterials (e.g., iron oxides immobilized on biochar), streamlined permitting could be allowed in designated contamination scenarios, contingent on pre-defined monitoring plans.
• Field-trial registries and data-sharing platforms: Centralized databases documenting pilot-scale performance, environmental fate, and ecotoxicological observations—akin to the US EPA's Contaminated Site Clean-Up Information (CLU-IN) database—could build the evidentiary foundation for future regulation.
• Harmonized international criteria: Alignment on core parameters (e.g., particle stability thresholds, ecological screening levels, transformation product identification) would reduce redundant testing and facilitate technology transfer, particularly in transboundary pollution scenarios.
• Stakeholder-engaged governance: Inclusive dialogue among regulators, industry, researchers, and community representatives can co-develop acceptable risk-benefit trade-offs and build public trust—a critical enabler for social license to operate.
Ultimately, regulatory evolution must balance precaution with innovation, recognizing that overly restrictive policies may stall environmental cleanup, while lax oversight risks unintended consequences. Closing the policy-science gap requires concerted efforts to generate robust, field-validated data on nanomaterial fate and effects, translate those findings into practical guidance, and foster regulatory agility through iterative learning from controlled field demonstrations. Only through such collaborative, evidence-informed policymaking can nanoremediation realize its potential as a scalable, safe, and socially accepted solution for heavy-metal-contaminated environments.
-
Although nanotechnology has demonstrated considerable promise for environmental remediation in the laboratory, its transition to sustainable field deployment remains constrained by a suite of unresolved scientific, technical, and governance barriers. The most critical knowledge gaps include long-term stability and transformation of engineered nanomaterials (ENMs) in complex environmental matrices, standardized testing and reporting, life-cycle evaluation, economic feasibility, and a regulatory framework to ensure safe and equitable deployment. Addressing these gaps is essential to realize the full environmental potential of nanoremediation. Nanoparticles degrade after release into the environment, due to aging and processes such as aggregation, oxidation, sulfidation, dissolution, and reaction with natural organic matter, changing their surface chemistry, reactivity, and mobility significantly[47]. Although surface modification can reduce acute toxicity, there is a risk of unintended consequences, including increased acute toxicity or long-term nanoscale additions to sediments[35,70]. Most studies are short-term and lab based, leaving a significant gap in knowledge regarding seasonal or multiyear transformation. To elucidate the transformation processes and the entrapment of the immobilized metals, and the permanence of nano-induced mineral phases, long term field studies with advanced spectroscopic and microscopic tools are needed.
The inconsistency in reporting particle size, surface area, zeta potential, and dissolution behavior and the differences in the expression of exposure measures also prevent interstudy comparability[36]. Additionally, variations in pH, ionic strength, and concentration of organic matter utilized in experiments make it difficult to generalize to field conditions. The application of standardized procedures as per the OECD or ISO requirements for the synthesis of nanomaterials, characterizations, and environmental trials of the same would greatly increase reproducibility and regulatory confidence. It must also be made available in open-access data repositories and interoperable databases, which would facilitate meta-analyses, predictive modeling, and material optimization across research groups[34]. To be truly sustainable, nanoremediation must be viewed as a component of a life cycle that will involve synthesis, deployment, regeneration, and end-of-life management. The energy and chemicals involved in the manufacture of nanoparticles are normally significant,and therefore must be offset by environmental benefits. In addition, transformation products or used nanomaterials contain high concentrations of heavy metals, which are not easy to dispose of or recycle[61].
The retrievable and regenerable systems to emerge as one of the priority areas of inquiry should be systems like magnetic or biochar-supported nanocomposites that can be reused and recovered without any safety issues[31]. The integration of life cycle, techno-economic, and risk analyses will facilitate the evidence-based decision-making regarding the material design and policy use (Table 6). One of the biggest problems with implementation in the field is economic scalability. Many nanomaterials are prepared on a laboratory scale using batch processes, which cannot be directly applied in the large-scale synthesis of nanomaterials. The prohibitive price of functionalized/composite nanoparticles may be too extravagant in heavily polluted regions[34]. Future approaches should be directed toward greener, less costly methods of synthesis, which can involve biosynthesized, waste-derived, or naturally templated nanoparticles that should be aligned with the so-called principle of the circular-economy. In situ delivery systems (i.e., permeable reactive barriers, nano-amended biofilters) can be considered an appropriate step to make the operations more feasible and reduce exposure risks.
Table 6. Summary of major scientific, technical, and regulatory gaps in nanoremediation, and proposed future research directions for field-scale implementation
Category Key challenges and limitations Explanation Proposed research and policy priorities Ref. Scientific gaps Ecotoxicity and long-term impacts Insufficient chronic/trophic-transfer data; possible harm to non-target creatures across trophic levels. Perform extensive, long-term mesocosm experiments and life cycle/ecosystem investigations; measure trophic transfer and bioaccumulation under practical exposure conditions. [107,108] Transformation products and secondary contamination Unknown effects of modified or deteriorated NPs on the environment and living things. Characterize and identify transformation products in practical settings; investigate secondary impacts on geochemistry and biota. [108,109] Environmental fate and transport NP transformations (dissolution, aggregation, aging) and unpredictable mobility in heterogeneous soil–water matrices. Integrate fate-transport models that have been verified in column and field experiments, as well as in-situ characterization techniques (spectroscopy/tracers). [110,111] Bioavailability and mobility under field conditions Lab results may not translate to heterogeneous natural environments. To measure bioavailability, transport, and retention in soils and sediments, conduct field-scale experiments and predictive modeling. [107,112] Interaction mechanisms Limited understanding of NP pollutant and microbe interactions at molecular levels. Utilize controlled mesocosms, synchrotron imaging, and multi-omics to enable mechanism-driven material design and connect mechanisms to results. [112,113] Technical gaps Manufacturing and scalability Lab techniques are costly and difficult to scale for widespread field use. Create low-cost, environmentally friendly, bio- or waste-derived synthesis pathways; conduct techno-economic assessments and pilot-scale production. [113] Material stability under environmental stress Changes in temperature, pH, or ions can cause NPs to break down, lose their reactivity, or congregate. Create robust NPs with surface functionalization, test them in dynamic environments, and take aging studies into consideration. [109] Monitoring and confirmation of performance Absence of field-deployable, real-time techniques to monitor NP activity and fate. Develop standardized KPIs, tracer techniques, and portable sensors for adaptive control and long-term monitoring. [111] Integrating hybrid systems If NPs are not optimized, their integration into chemical or biological processes may result in reduced productivity. To increase productivity while reducing ecological disturbance, study NP–plant–microbe or NP–biochar synergistic systems. [114] Dispersion and delivery In contaminated areas, aggregation lowers NP availability and reactivity. Develop 'safer-by-design' nanoparticles, core-shell, or magnetic structures; maximize in situ distribution using tracers or permeable reactive barriers. [115] Regulatory gaps Gaps in monitoring and liability Aambiguous accountability for unanticipated consequences or failures. Develop legal frameworks for liability, remediation obligations, and post-deployment monitoring. [116] Lack of distinct frames Several countries have environmental laws specifically related to NPs, which are frequently handled like bulk chemicals. Develop global, tiered, risk-based field deployment guidelines that cover permissions and monitoring. [112] Public perception and ethical concerns Skepticism regarding unknown long-term hazards. Conduct socioeconomic impact research, pilot co-design, and open public interaction. [117] Interoperability and data sharing Meta-analysis and cross-study comparison are restricted by the absence of centralized databases. Develop interoperable, open-access databases for performance data, environmental monitoring, and NP characterization. [118] Standardization of risk assessment Inconsistent terminology and test protocols limit comparability. Create standardized reporting guidelines, classification systems, and test procedures for all jurisdictions. [111] The next generation of remediation strategies will be developed using nanotechnology in multi-functional systems that integrate chemical, biological, and physical processes inspired by nature. More successful hybrid solutions can be created by using nano-assisted phytoremediation, a consortium of microbes and nanoparticles, and biochar-nanoparticle composites that can also decrease the quantity of nanoparticles needed and enhance their efficiency[31,66]. These synergistic systems, however, should be well formulated with the ecosystems of interrelations amongst the nanoparticles, plants, and microbes, avoiding undesired ecological disturbances. When nanoremediation is integrated into sustainable land and water management systems, supported by carbon credits, incentives to recover resources, and circular-economy methods, environmental and socioeconomic outcomes are likely to improve in the long run[36].
The uncertainty in the regulations is one of the biggest bottlenecks. Explicit visualizations of the use of engineered nanomaterials in the environment are not provided in most jurisdictions, and even chemical regulations in most cases does not consider the nano-specific applications or modifications. The government and the political movement will demand active and performance-based regulations that would establish a balance between innovation and caution. Open stakeholder engagement will also boost public confidence towards the government and facilitate post-deployment tracking and liability management with minimal effort. Responsible adoption will be further increased with the international harmonization of definitions, testing requirements, and reporting standards.
-
The hyper-reactive, selective, and versatile nature of nanoremediation positions as a promising approach for transforming the remediation of heavy metal-contaminated sites, moving beyond the limitations of conventional approaches. However, its effectiveness is relative to existing technologies under realistic environmental conditions. Engineered nanomaterials exhibit a strong capacity to detoxify and immobilize toxic metals in soil, sediment, and water, and their action involves a combination of mechanisms including adsorption, ion exchange, reduction, photocatalysis, precipitation, as well as magnetic recovery. High efficiency and recoverability are achieved in hybrid nanostructures such as biochar-based zero-valent iron, graphene-magnetite, and functionalized metal oxides, which combine contaminant stabilization with biodegradation synergistic pathways through interaction with biological system,s including microbial consortia and phytoremediation. Nevertheless, uncertainties remain regarding the long-term stability, transformation, and possible ecotoxicological effects of nanomaterials in complex environmental matrices. The development of safe-by-design, recoverable, and biodegradable nanomaterials with minimal environmental impact will enhance the future effectiveness of nanoremediations. In this regard, systematic risk assessment and life-cycle analysis are necessary to guarantee environmental safety. The integration of artificial intelligence, machine learning, and real-time sensing can accelerate the detection and monitoring of materials and the factors governing the remediation process. These data-driven approaches can facilitate site-specific-decision making and enhance the predictability of remediation processes. Nanoremediation is not only an environmental solution, but also an integral part of the economic cycle, linking remediation with resource recovery and ecological restoration, which can increase sustainability and social value. However, its large-scale implementation needs to be developed in a coordinated manner in the fields of science, engineering, and regulation. Finally, the coordinated advancement of science, engineering, and policy will be essential for the future of nanoremediation. Establishing international standards, transparent risk governance, and equitable access to technology is critical to translating laboratory-scale successes into safe, effective, and sustainable solutions for heavy metal pollution. With these concerted efforts, nanotechnology can be used to its full potential as a reliable, environmentally friendly, and sustainable solution to heavy metal contamination.
-
The authors confirm their contributions to the paper as follows: Qasim Ali, Longfei Liu, Qi Li, and Shahzad Ahmed: visualization, writing − original draft preparation; Marwa Yasmeen, Asma Mukhtar, Muhammad Akraam, Muhammad Azeem Sabir, and Adnan Hussain: illustration, proofreading, writing − reviews and editing; Rashid Hussain, Maqshoof Ahmad, and Muhammad Mahroz Hussain: proofreading, writing − reviews and editing; Shengsen Wang: supervision, proofreading, writing − reviews and editing. All authors reviewed the results and approved the final version of the manuscript.
-
Data sharing is not applicable to this article as no data set was generated or analyzed during current study.
-
No funding was received to assist with the preparation of this manuscript.
-
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
-
This review explores the potential of nanomaterials for heavy metal adsorption, reduction, and photocatalysis.
This review summarizes pH, Eh, and NOM critically control nanoremediation effectiveness.
Research highlights nanomaterial ecotoxicity and regulatory gaps as key bottlenecks.
Findings advocate merging nanoremediation with phytoremediation for sustainable gains.
-
Full list of author information is available at the end of the article.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Ali Q, Li Q, Ahmed S, Yasmeen M, Mukhtar A, et al. 2026. Nanoremediation of heavy metal-contaminated environments: mechanisms, advances, and future prospects. Agricultural Ecology and Environment 2: e007 doi: 10.48130/aee-0026-0004
Nanoremediation of heavy metal-contaminated environments: mechanisms, advances, and future prospects
- Received: 13 November 2025
- Revised: 10 January 2026
- Accepted: 17 January 2026
- Published online: 04 March 2026
Abstract: Heavy-metal pollution of soil, sediments, and water remains a major environmental and public health concern worldwide. Conventional remediation methods (excavation, soil washing, chemical stabilization, and pump-and-treat) frequently suffer from high costs, low selectivity, and the generation of secondary wastes. Engineered nanomaterials (ENMs) provide a complementary toolkit that leverages nanoscale properties, such as a large specific surface area, controlled surface chemistry, and redox or photocatalytic functionality to bind, transform, or immobilize toxic metals in situ or ex situ. This review critically synthesizes recent advancements in nanomaterial classes used for heavy-metal mitigation, such as nanoscale zero-valent iron and iron oxides, titanium dioxide, and other metal oxides, carbonaceous materials including graphene derivatives, and biochar-based composites, and hybrid magnetic/functionalized composites. We discussed the detailed mechanistic pathways (adsorption and surface complexation, ion exchange, electron-transfer reduction, photocatalytic transformation, and co-precipitation), and how environmental parameters—pH, redox potential, ionic strength, natural organic matter, and particle aging—influence efficacy and permanence. We compare laboratory, mesocosm, and field studies to highlight practical performance, limitations in transport and delivery, and ecological trade-offs. Remaining barriers to deployment include nanoparticle transformation and mobility in complex matrices, standardized ecotoxicological testing, techno-economic assessment, and regulatory acceptance. The review concludes with recommended research directions to enhance stability, selectivity, recovery, and life cycle sustainability of nanoremediation technologies, emphasizing integrated approaches that combine mechanistic insight with field-scale validation.
-
Key words:
- Nanoremediation /
- Zero-valent iron /
- Graphene oxide /
- Photocatalysis /
- Environmental fate /
- Ecotoxicity