-
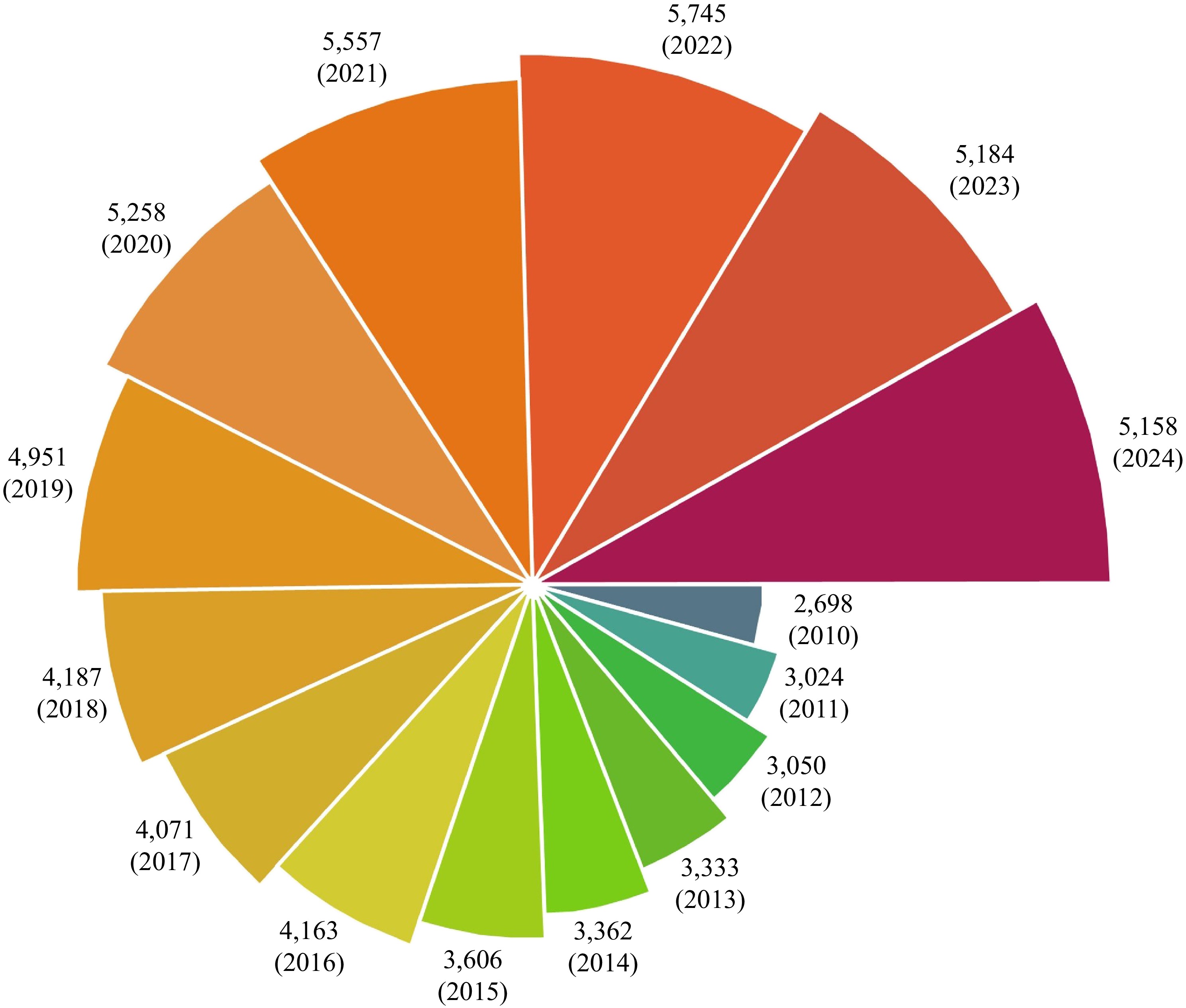
Figure 1.
Annual publication trends on fish protein research.
-
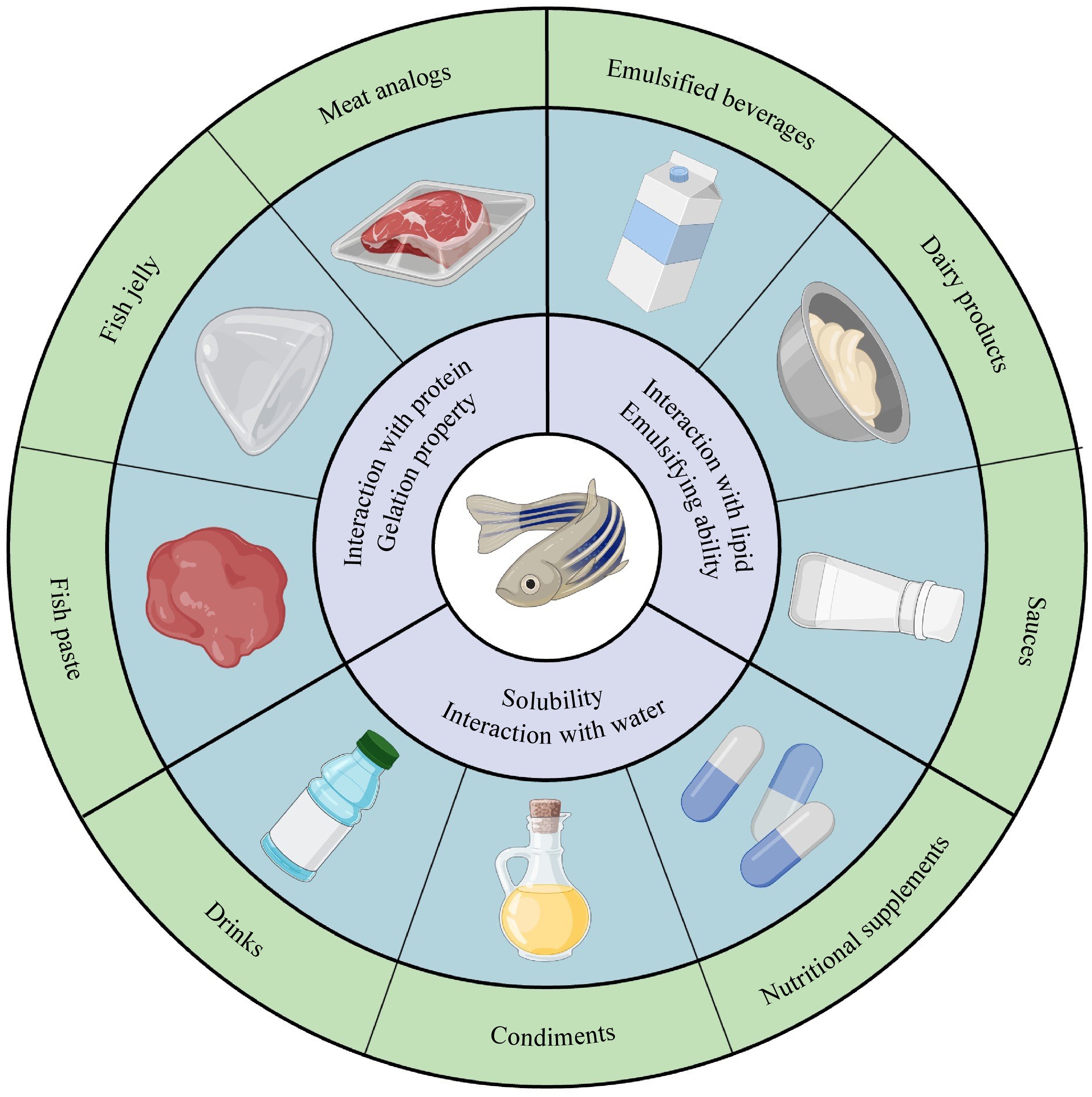
Figure 2.
Relationship between fish protein properties and applications.
-
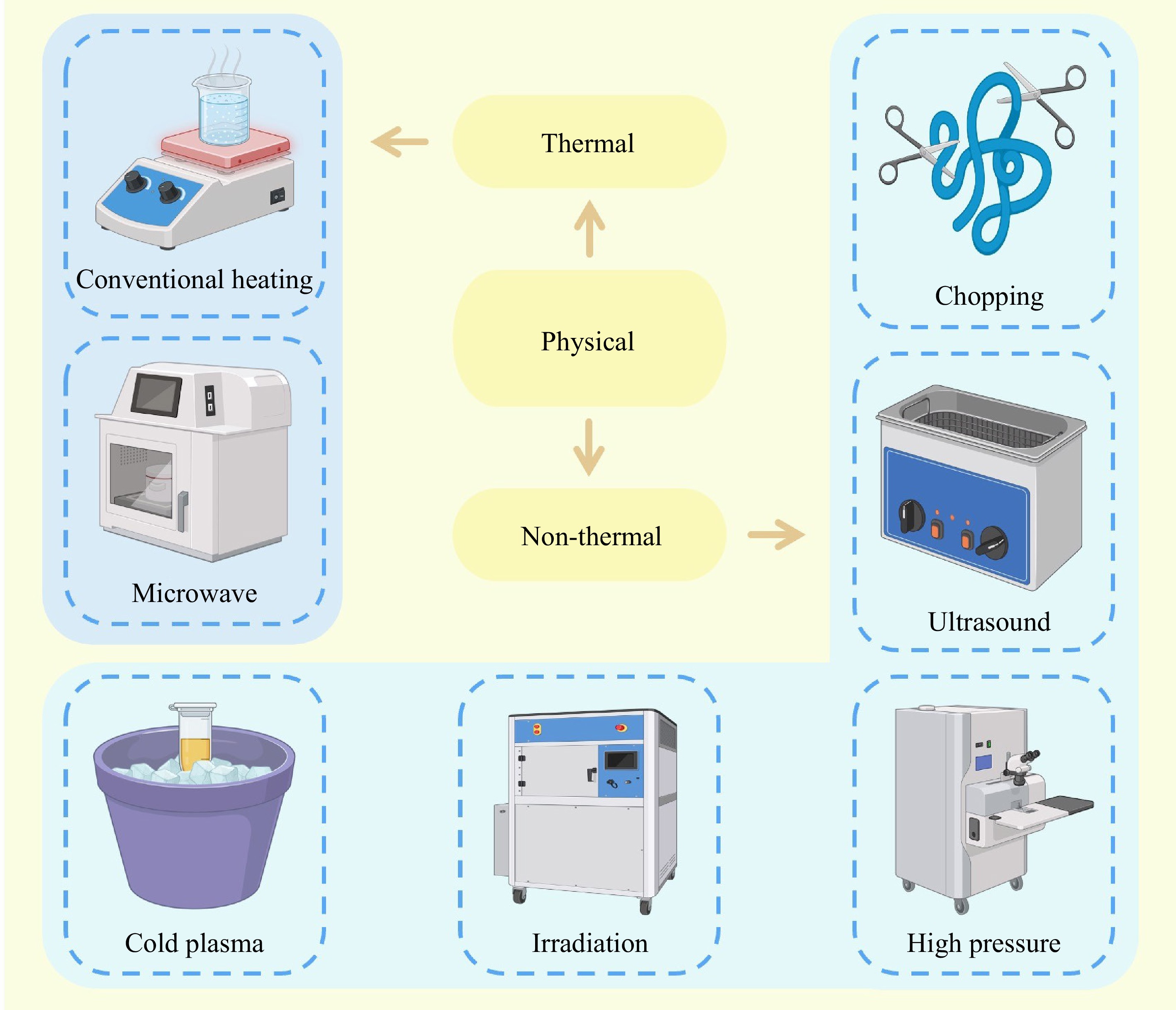
Figure 3.
Physical modification method of fish protein.
-
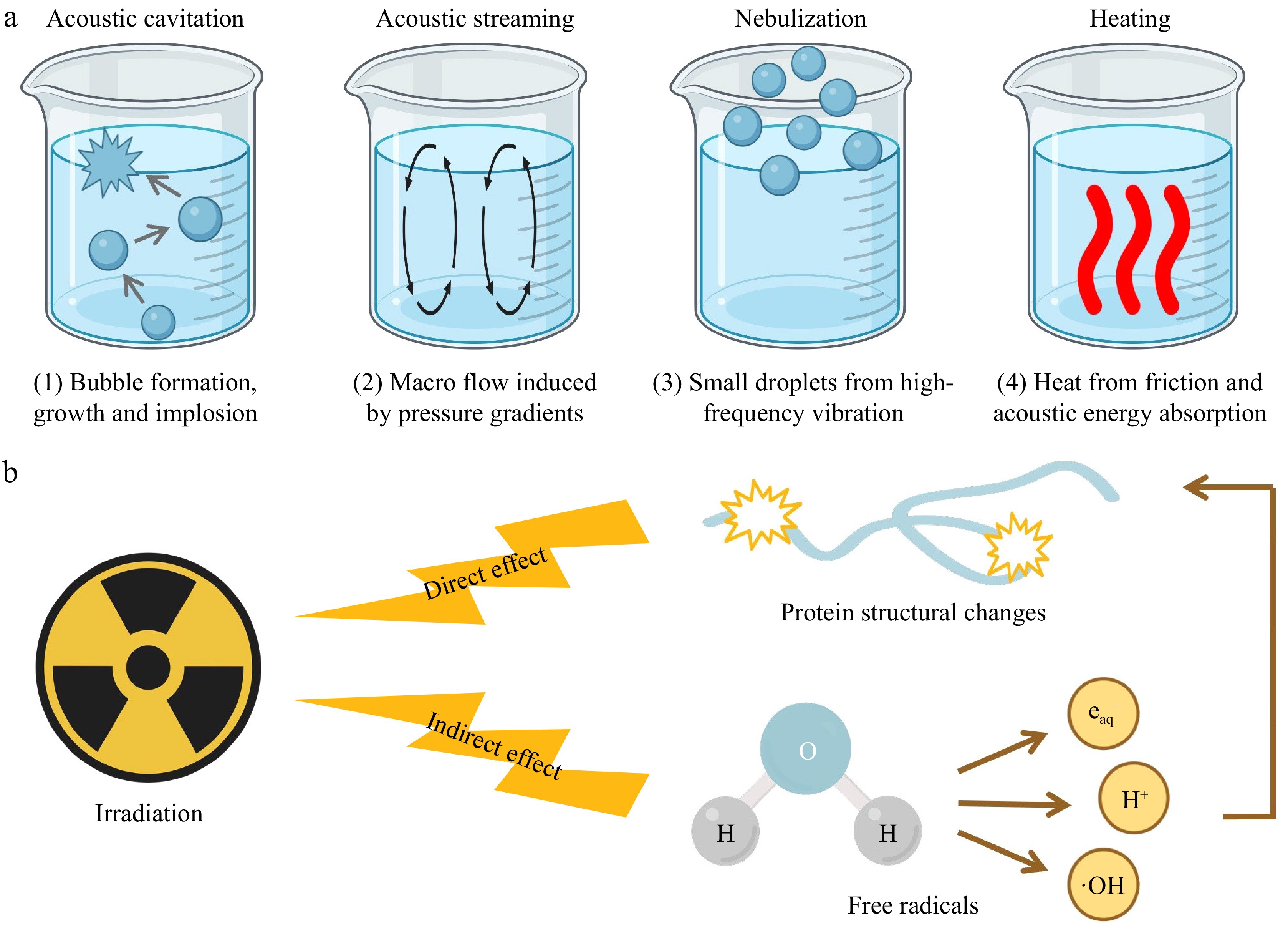
Figure 4.
Mechanisms of (a) ultrasound, and (b) irradiation on fish protein modification.
-
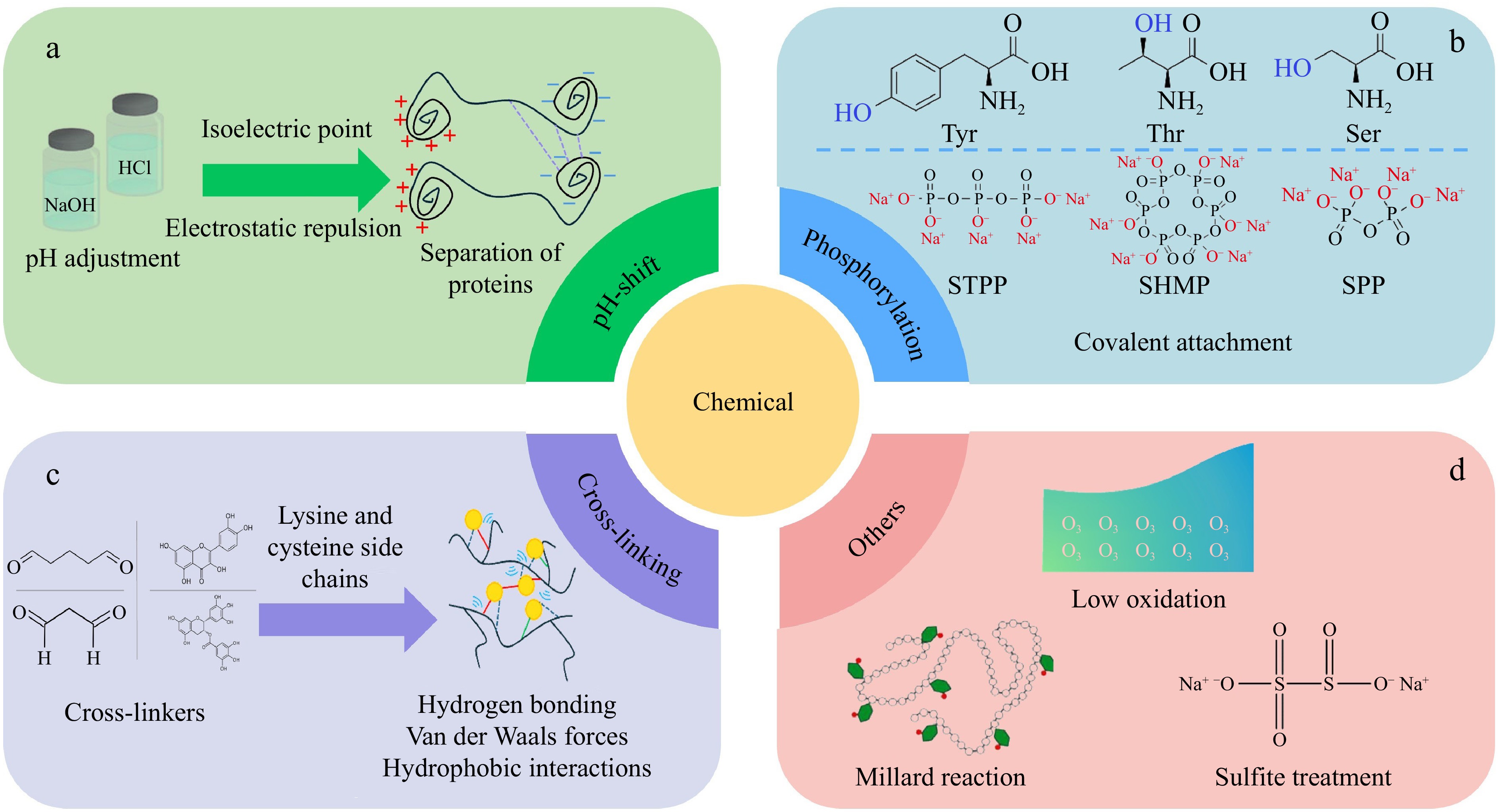
Figure 5.
Chemical modification methods of fish proteins: (a) pH-shift, (b) phosphorylation, (c) cross-linking, and (d) others.
-
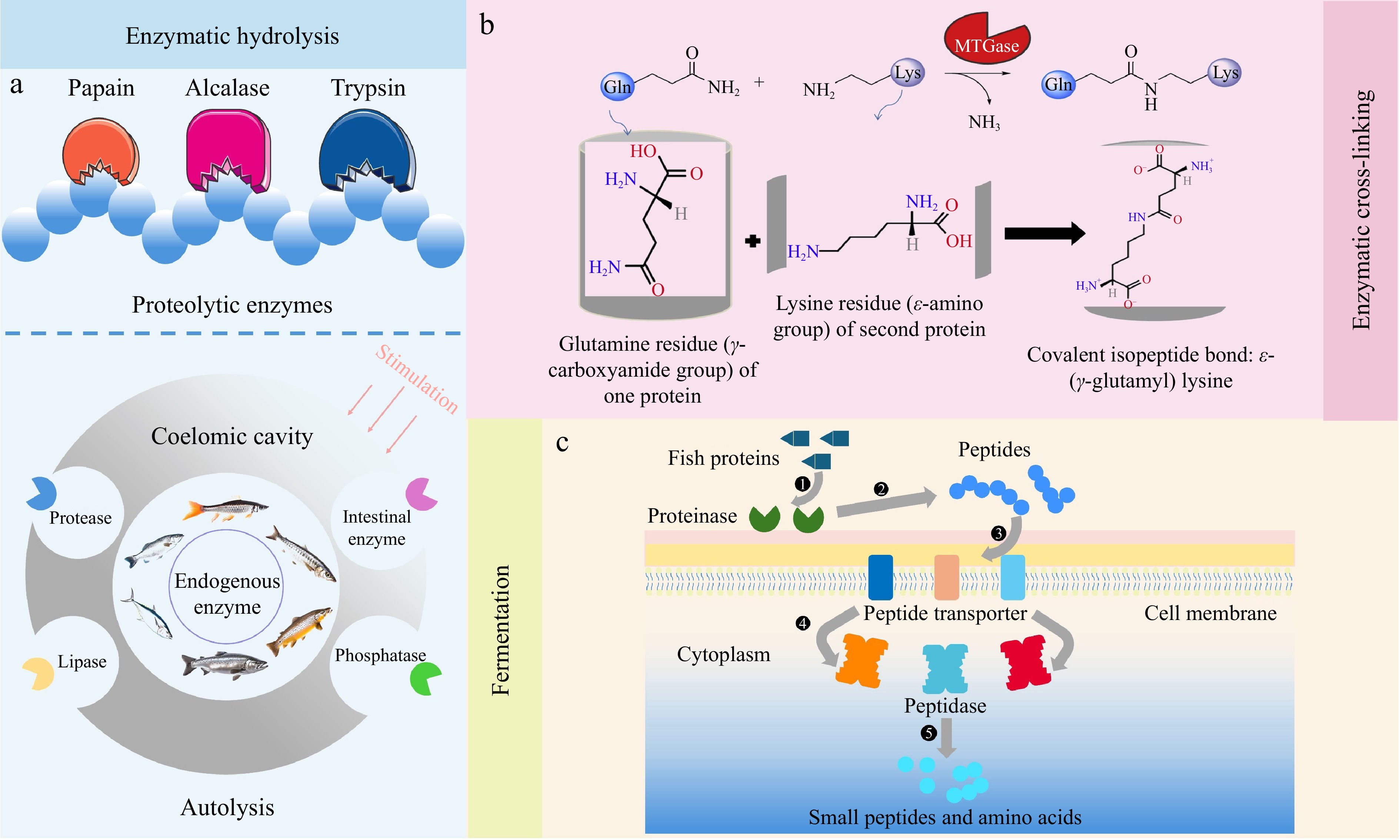
-
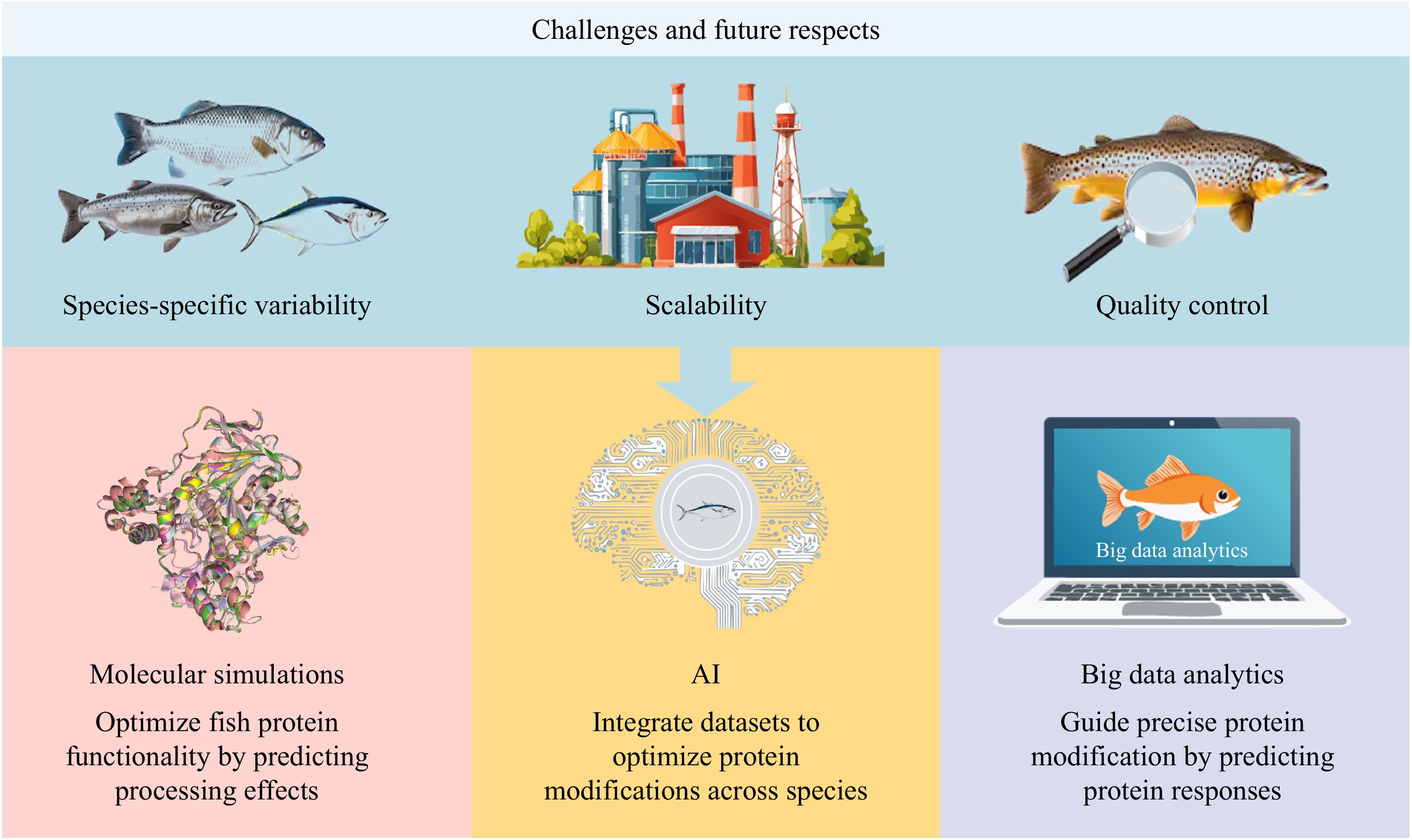
Figure 7.
Challenges and future respects of fish modification methods.
-
Modification
methodsFish protein Treatment conditions Important findings Ref. Ultrasound Atlantic mackerel (Scomber scombrus) side streams 300, 450, and 600 W at frequency of 20 kHz for 10 min Protein solubility increased linearly with an increase in ultrasound power. [7] Myosin from silver carp Varying power output (100, 150, 200, and 250 W) for 3, 6, 9, and 12 min Myosin aggregates were converted to smaller ones with a more uniform distribution and an obvious enhancement in solubility. [11] MP from
Coregonus peled100, 150, 200, and 250 W with different treatment time (0, 3, 6, 9, and 12 min) at a frequency of 20 kHz Solubility, emulsification capacity, and emulsion stability were improved significantly, and each decreased with an increase in treatment time (up to 12 min). [13] MP from Nemipterus virgatus surimi At different levels of power output (0, 150, 300, 450, 600 W) at 20 kHz for
12 minModerate ultrasound can improve the emulsion stability and emulsion treated at 300 W showed the best stability. [92] MP from golden pompano (Trachinotus ovatus) (i) Ultrasonic time (60, 120, and 180 s) at the ultrasonic power of 150 W; (ii) ultrasonic power (100, 150, and 200 W) at the ultrasonic time of 120 s, respectively Both EAI and ESI significantly increased and then decreased with increasing ultrasonic time and power. The maximum values of EAI and ESI were obtained at the ultrasonic time of 120 s and ultrasonic power of 150 W. [14] MP from Asian sea bass Varying amplitudes (40% and 60%) for different time (5, 10, and 15 min) at a frequency of 20 kHz ± 50 Hz and high-intensity power of 750 W Solubility and emulsifying properties were augmented when subjected to ultrasonication at 40% amplitude for 15 min. [15] Common carp (Cyprinus carpio) MP different power (0, 100, 300, and 500 W)
for different time (59.05, 49.67, 45.16, and 35.5 min)Appropriate ultrasonic intensity (300 W) and time (45.16 min) could improve gel strength and WHC. [16] High pressure Muscle protein from smoked rainbow trout 0, 200, 400, or 600 MPa for 3 min at 4 °C Protein solubility and WHC were reduced with increasing pressure. [18] Natural actomyosin from threadfin bream (Nemipterus
spp.)Different pressures (200, 400, 600 MPa) at room temperature for 10, 30, 50 min Protein solubility decreased with increasing pressure and time. [19] Sarcoplasmic proteins from sea bass fillets pressure levels of 100, 250, and 400 MPa; pressure holding times of 0, 5, 15, and
30 min; and pressurization rates of 8 and 14 MPa/sProtein solubility reduced with increase in pressure level and holding time. Pressurization rate did not influence sarcoplasmic protein solubility. [93] Sarcoplasmic proteins from hake (Merluccius merluccius) 200, 400, and 600 MPa for 6 min at 20 °C In contrast to high-pressure levels (400 and 600 MPa), treatment at 200 MPa produced emulsions with enhanced stability, greater consistency index, and shear-thinning behavior. [22] Myosin from golden threadfin bream (Nemipterus virgatus) 100, 200, 300, 400, and 500 MPa for
3 minPressure at 100−300 MPa could markedly improve the gelation properties but gradually decreased as pressure further increased. [17] Golden threadfin bream (Nemipterus virgatus) myosin 150, 300, 450, and 600 MPa (± 15 MPa)
for 5 minmoderate treatment (≤ 300 MPa) strengthened the water retention and structure properties of myosin gel, while stronger treatment (≥ 450 MPa) weakened them. [20] Microwave Sarcoplasmic and MP from grass carp Different microwave power levels (300, 600, and 900 W) and heating times (0−60 s) A negative correlation was observed between protein solubility and microwave power and time. [27] Fish protein hydrolysates from farmed rainbow trout Oncorhynchus mykiss 3, 5, and 15 min (1,200 W, 20% power with 50% duty cycle at 50–55 °C), compared with conventional heating (water bath at 50 °C) Microwave treatment demonstrated higher protein solubility, emulsifying activity and emulsion stability than conventional heating with the same treatment. [94] Tilapia skin fish protein Various microwave power of 240, 400, 640, and 800 W for 1 and 4 min At the same microwave time or power, EAI and ESI increased with increased power or time. Microwave could significantly increase gel strength and hardness. [28] Protein from Alaska Pollock (Theragra chalcogramma) Various powers (300, 400, or 500 W) and time (10, 20, or 30 min) The gelling ability of microwave-heated protein was higher than that produced by water bath heating. Increasing microwave power and time resulted in decreases in both gel strength and WHC. [26] MP from silver carp Microwave (single-mode 2,450 MHz synthesizer) and water bath heating at
90 °C for 20 minMicrowave treatment generated a more stable, denser, and well-structured network compared with conventional heating. [30] Table 1.
Physical modification methods for fish proteins.
-
Modification
methodsFish protein Treatment conditions Important findings Ref. pH shift Myosin from blue round scad (Decapterus maruadsi) Acidic or alkaline solubilization followed by neutralization; heating from 20–90 °C Acidic pH-shift irreversibly damaged myosin, preventing gelation; alkaline treatment partially restored structure and gel-forming ability compared to native protein [34] Common carp (Cyprinus carpio) protein isolates Solubilization at pH 2.5 (acidic) or 12.5 (alkaline), followed by precipitation at pH 5.5 Acidic and alkaline isolates showed high protein yield (~76% to 88%), reduced fat/ash, improved digestibility (7–8 ×). [31] Protein from salmon, cod, and herring by-products Acidic or alkaline solubilization at pH 11.5–12.5, isoelectric precipitation at pH 5.5 Alkaline process maximized yield (salmon > herring > cod); solubilization pH effects on gel strength, WHC, and color was species-specific. [32] Baltic herring (Clupea harengus membras) muscle protein isolates Alkaline pH-shift extraction (solubilization pH ~11.2, precipitation pH ~5.4), tested in surimi-type gels and fish balls Reduced fishy odor; gels matched surimi hardness/cohesiveness but were darker; altering pH softened gels without improving whiteness. [35] Bighead carp (Aristichthys nobilis) myosin particles Myosin particles (0.6 M NaCl) used for Pickering emulsions; stability tested over pH 3–11 Emulsions showed high stability at neutral–alkaline pH; acidic pH (≤ 5) caused droplet aggregation due to myosin's proximity to pI. [3] Phosphorylation Grass carp (Ctenopharyngodon idellus) MP Addition of sodium tripolyphosphate, tetrasodium pyrophosphate, sodium hexametaphosphate (0.1%–0.5%, w/w) Sodium tripolyphosphate and tetrasodium pyrophosphate improved gel strength, WHC, and reduced water mobility [36] Fish gelation Phosphorylation with sodium pyrophosphate (FG : TSPP ratio 40:0 to 40:6) at pH 7.0 Increased gel strength, viscosity, emulsifying properties; reduced water mobility; excessive TSPP shortened gelation time. [38] Fish scale gelatins from Coregonus peled, Esox lucius, and Ctenopharyngodon idella Phosphorylation via sodium trimetaphosphate (STMP), phosphorylating for 0–3 h Increased surface hydrophobicity and emulsification activity/stability; structural changes supported improved emulsions. [40] Mantis shrimp (Oratosquilla oratoria) MP Phosphorylation via sodium tripolyphosphate at low ionic strength (0.1 mol/L KCl) Phosphorylation improved solubility, gel strength, and emulsifying properties; enhanced WHC; modified protein structure for better functionality. [41] Chemical
cross-linkingFish skin gelatin (salmon gelatin, SG; tilapia gelatin, TG) Crosslinked with glutaraldehyde (GA) at 0.05%–0.15% to form hydrogel At 0.05% GA, hydrogels had optimal swelling, porosity, and α-glucosidase binding with best antidiabetic potential. [95] Antarctic krill protein (AKP) combined with cellulose Glutaraldehyde (GA) cross-linking to form multiple network with cellulose Multiple cross-linking enhanced crystallinity, orientation, mechanical and moisture-retention properties at 0.2 wt% GA. [96] Grass carp (Ctenopharyngodon idella) myosin Binding with chlorogenic acid (CGA) and rosmarinic acid (RA); spectroscopic analyses (fluorescence, UV–Vis, CD) CGA and RA bind myosin via H-bonding/van der Waals and hydrophobic; increase α-helix, protect sulfhydryls, complexes show antioxidant effects and promote aggregation. [97] Silver carp (Hypophthalmichthys molitrix) MP Crosslinked with oxidized polyphenols with layered double hydroxide nanosheets Dual modification enhanced tensile strength, barrier, and antioxidant activity; improved film stability for food packaging applications. [48] Table 2.
Chemical modification methods for fish proteins.
-
Modification methods Fish protein Treatment conditions Important findings Ref. Enzymatic hydrolysis Bighead carp (Hypophthalmichthys nobilis) protein hydrolysate (ficin-mediated) Ficin hydrolysis at pH 6, 40 °C, 3% enzyme/substrate for 1–6 h At DH 20.15%, highest yield, protein content, amino acid content, and solubility; OHC (oil-holding capacity) highest at lowest DH. [54] Chinese sturgeon (Acipenser sinensis) protein hydrolysate Papain hydrolysis (pH 6, 70 °C, 3% E/S, 6 h) DH reached 24.9%; high yield (17.5%), solubility (87%–99%), emulsifying/stability indices, WHC, OHC, and foam capacity. [55] Seabream (Sparus aurata) and seabass (Dicentrarchus labrax) fish protein hydrolysates Enzymatic hydrolysis of by-products (frames & trimmings, heads, viscera) using Alcalase; optimized via response surface methodology Frames & trimmings produced hydrolysates with highest antioxidant and antihypertensive activity, MW ~1.4–2.0 kDa, > 86% digestibility, and excellent essential amino acid profile. [57] Rainbow trout (O. mykiss) by-product autolysate Autolysis at 40 °C for 1 h, followed by heat inactivation Autolysis enriched small peptides but increased lipid and protein oxidation; hydrolysate still displayed antioxidant activity. [62] Enzymatic cross-linking Surimi (silver carp MP gel) MTGase-crosslinked at 29.7%, 45.8%, 63.0%; frozen via liquid nitrogen immersion (LNF), –35 °C, –18 °C Higher cross-linking (45%–63%) improved texture retention, reduced oxidation, and preserved aroma—LNF was superior to air freezing. [64] Surimi MTGase-induced cross-linking at increasing degrees < 30% cross-linking: mushy; ~40% to 55%: elastic/tough; ≥ 64%: brittle; WHC fell; pore structure and hydrophobicity altered. [65] Rainbow trout (Oncorhynchus mykiss) mince Myofibrillar restructuring with 0%–1.0% microbial transglutaminase (MTGase) during refrigerated storage MTGase reduced spoilage indicators (TVB-N, TMA-N), suppressed free amino acids increase, and inhibited psychrophilic and coliform growth—best at 1.0%. [66] Tambaqui (Colossoma macropomum) skin gelatin Transglutaminase (TGase) crosslinking at 1%–3% (w/w) 3% TGase increased Bloom strength; film thickness and mechanics unchanged; solubility and water vapor permeability significantly reduced. [67] Fermentation Enrichment of fermented fish sausages Selected autochthonous Lactobacillus strains (from fish gut microbiota); applied as starter cultures Three strains (IDs 11, 68, 69) reduced fermentation time (~2 d) and ensured microbiological safety, showing probiotic potential. [70] Grass carp (Ctenopharyngodon idella) fish protein (semi-dried meat) Fermented with Monascus purpureus Went M3.439; then subjected to in vitro digestion and fermentation assays Fermentation enhanced free amino acids and glutamate, reduced ammonia/indole, increased propionic acid and Parabacteroides during in vitro fermentation, suggesting improved digestibility and gut health. [94] Atlantic cod (Gadus morhua) muscle protein Fermentation with Lactobacillus helveticus strain Lh191404 Fermentation disrupted allergen structure and linear epitopes, reducing immunoreactivity [72] Crucian carp (Carassius auratus) suanyu (fermented fish) Mixed starter culture: Enterococcus rivorum + E. lactis, compared to natural fermentation over 13 d Mixed culture boosted protease activity, TCA-soluble proteins, softness and flavor compounds (aldehydes, glutamate); protease activity correlated with hardness, chewiness, flavor. [73] Table 3.
Biological modification methods for fish proteins.
Figures
(7)
Tables
(3)