-
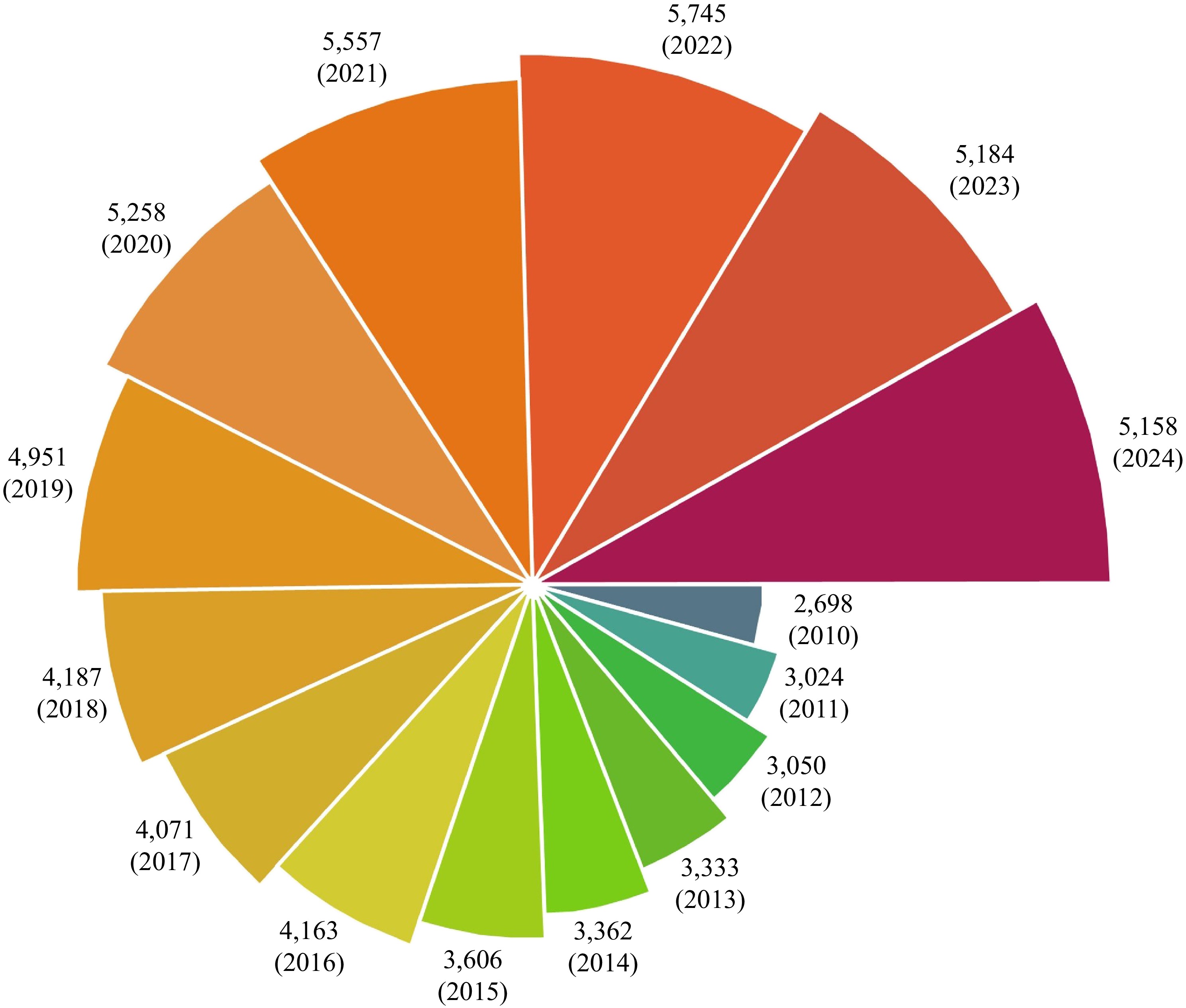
The demand for fish products has propelled the industry into a phase of unprecedented growth and transformation. Fish could be a sustainable and nutritious component of the human diet for obtaining high-quality proteins. Fish proteins are renowned for their rich content of essential amino acids, such as lysine, methionine, phenylalanine, with levels higher than those from terrestrial meat proteins. As shown in Fig. 1, the publications on fish proteins have steadily increased over the past two decades, driven by their growing recognition of potential applications. Edible fish products account for merely 40% of the total fish yield. The remaining 60%, including fish heads, bones, entrails, and skins, are commonly regarded as waste[1]. However, waste from fish still contains a considerable amount of protein, which can be applied in the food industry.
Generally speaking, natural fish proteins rarely exhibit ideal functional properties such as solubility, emulsifying capacity, and gel formation, which limits their direct use in various applications[2]. These limitations are largely determined by the molecular structure and spatial conformation of proteins[3]. For this reason, modification strategies can be beneficial to creating innovative food products, with ideal functional properties to satisfy consumer needs. Although several individual studies have explored these modification methods, there is currently a lack of comprehensive reviews that integrate all mainstream and emerging technologies in fish protein modification. In response, emerging interdisciplinary tools such as artificial intelligence (AI), big data analytics, and molecular simulations have opened new opportunities to design precise, efficient, and eco-friendly modification strategies.
In this review, the relationship between functional properties and modification strategies of fish proteins is discussed, along with their future trends and challenges. With the aim to promote the connection between academic research and practical applications, this review can provide a reference for both researchers and practitioners to promote the development and innovation of fish proteins.
-
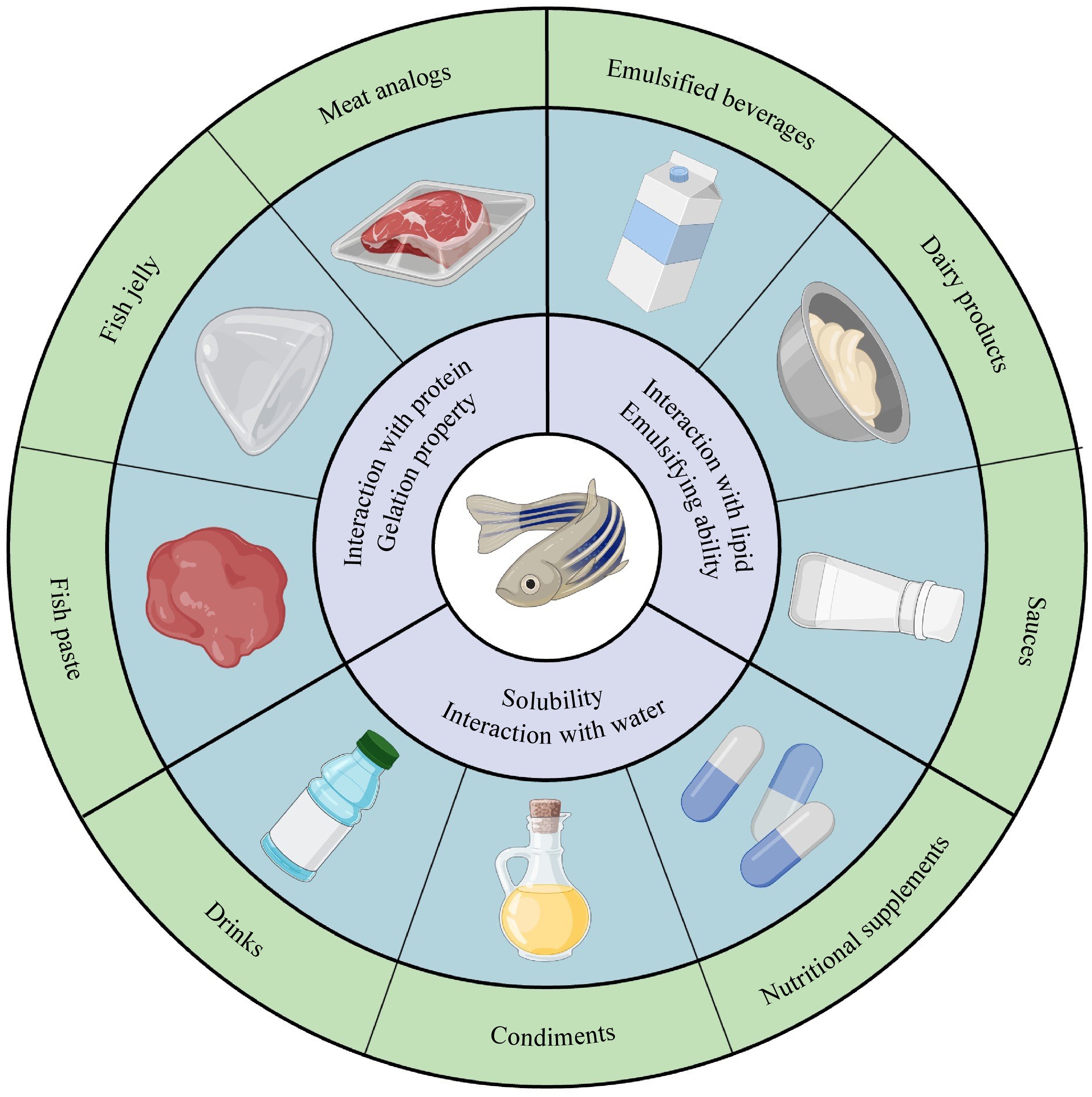
Protein functional properties could influence the behavior of fish proteins during processing, storage, and consumption. As shown in Fig. 2, the primary functional properties of fish proteins can be generally grouped into three classifications[4]. Specifically, protein-water, protein-fat, and protein-protein interactions govern solubility, emulsification, and gel formation, respectively, further determining food structure and texture. All of these functional properties are determined by the molecular characteristics of fish proteins, including factors like hydrophobicity, surface charge, molecular weight, secondary structures, and intermolecular forces. Furthermore, environmental conditions including pH, temperature, and the existence of other ingredients can induce molecular changes to affect protein functionality, thereby impacting the overall food quality[5].
The solubility of fish proteins refers to their ability to disperse uniformly in a specific solvent. Generally, the high solubility is beneficial for the protein's other functional properties[6]. Therefore, improving solubility is a crucial step in enhancing fish protein utilization. However, in many cases, fish proteins contain a high proportion of hydrophobic amino acids, causing the proteins to be insufficiently soluble for specific applications. Moreover, fish proteins can function as emulsifiers, which is primarily due to the amphiphilic nature of fish proteins to stabilize the oil-water interface. However, emulsions stabilized by fish proteins are generally susceptible to aggregation over time, undermining the physical stability[7]. Furthermore, fish protein gelation involves protein denaturation and aggregation, leading to the formation of a cross-linked network[8]. However, fish protein gels tend to exhibit relatively weak gelation and rheological properties owing to the limited amount of amino acids like proline and hydroxyproline, which are known to promote the formation of robust gel networks[9]. Therefore, given these structural challenges associated with functional properties of fish proteins, it is particularly important to conduct fish protein modification strategies, expanding their functionalities and applications.
-
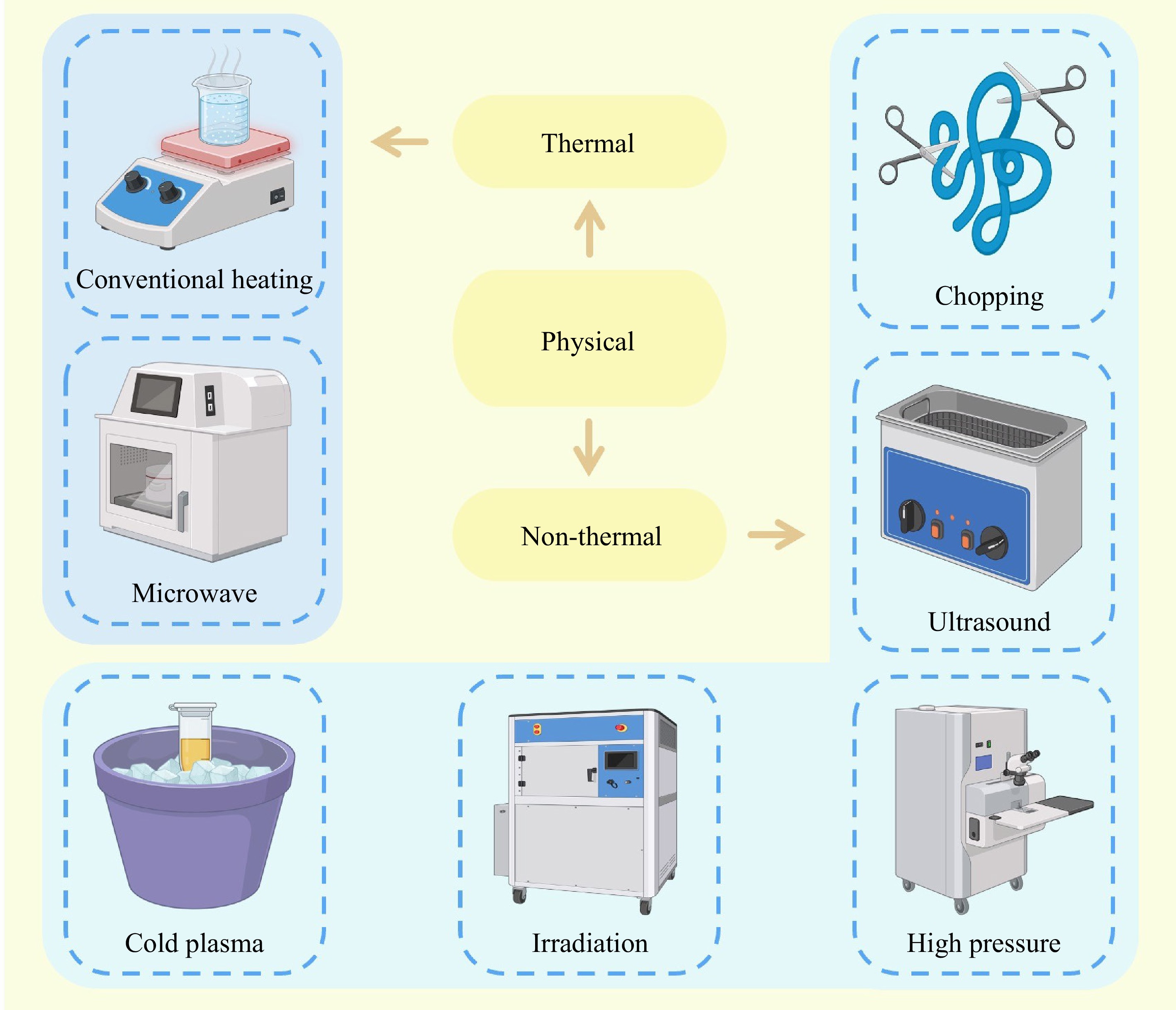
Physical modification of fish proteins involves various thermal or non-thermal techniques without altering their chemical structures (Fig. 3). In the physical modification, it is not required to apply significant levels of organic or acid treatments, as well as additional separation and purification steps, offering a straightforward and environmentally-friendly method (Table 1).
Table 1. Physical modification methods for fish proteins.
Modification
methodsFish protein Treatment conditions Important findings Ref. Ultrasound Atlantic mackerel (Scomber scombrus) side streams 300, 450, and 600 W at frequency of 20 kHz for 10 min Protein solubility increased linearly with an increase in ultrasound power. [7] Myosin from silver carp Varying power output (100, 150, 200, and 250 W) for 3, 6, 9, and 12 min Myosin aggregates were converted to smaller ones with a more uniform distribution and an obvious enhancement in solubility. [11] MP from
Coregonus peled100, 150, 200, and 250 W with different treatment time (0, 3, 6, 9, and 12 min) at a frequency of 20 kHz Solubility, emulsification capacity, and emulsion stability were improved significantly, and each decreased with an increase in treatment time (up to 12 min). [13] MP from Nemipterus virgatus surimi At different levels of power output (0, 150, 300, 450, 600 W) at 20 kHz for
12 minModerate ultrasound can improve the emulsion stability and emulsion treated at 300 W showed the best stability. [92] MP from golden pompano (Trachinotus ovatus) (i) Ultrasonic time (60, 120, and 180 s) at the ultrasonic power of 150 W; (ii) ultrasonic power (100, 150, and 200 W) at the ultrasonic time of 120 s, respectively Both EAI and ESI significantly increased and then decreased with increasing ultrasonic time and power. The maximum values of EAI and ESI were obtained at the ultrasonic time of 120 s and ultrasonic power of 150 W. [14] MP from Asian sea bass Varying amplitudes (40% and 60%) for different time (5, 10, and 15 min) at a frequency of 20 kHz ± 50 Hz and high-intensity power of 750 W Solubility and emulsifying properties were augmented when subjected to ultrasonication at 40% amplitude for 15 min. [15] Common carp (Cyprinus carpio) MP different power (0, 100, 300, and 500 W)
for different time (59.05, 49.67, 45.16, and 35.5 min)Appropriate ultrasonic intensity (300 W) and time (45.16 min) could improve gel strength and WHC. [16] High pressure Muscle protein from smoked rainbow trout 0, 200, 400, or 600 MPa for 3 min at 4 °C Protein solubility and WHC were reduced with increasing pressure. [18] Natural actomyosin from threadfin bream (Nemipterus
spp.)Different pressures (200, 400, 600 MPa) at room temperature for 10, 30, 50 min Protein solubility decreased with increasing pressure and time. [19] Sarcoplasmic proteins from sea bass fillets pressure levels of 100, 250, and 400 MPa; pressure holding times of 0, 5, 15, and
30 min; and pressurization rates of 8 and 14 MPa/sProtein solubility reduced with increase in pressure level and holding time. Pressurization rate did not influence sarcoplasmic protein solubility. [93] Sarcoplasmic proteins from hake (Merluccius merluccius) 200, 400, and 600 MPa for 6 min at 20 °C In contrast to high-pressure levels (400 and 600 MPa), treatment at 200 MPa produced emulsions with enhanced stability, greater consistency index, and shear-thinning behavior. [22] Myosin from golden threadfin bream (Nemipterus virgatus) 100, 200, 300, 400, and 500 MPa for
3 minPressure at 100−300 MPa could markedly improve the gelation properties but gradually decreased as pressure further increased. [17] Golden threadfin bream (Nemipterus virgatus) myosin 150, 300, 450, and 600 MPa (± 15 MPa)
for 5 minmoderate treatment (≤ 300 MPa) strengthened the water retention and structure properties of myosin gel, while stronger treatment (≥ 450 MPa) weakened them. [20] Microwave Sarcoplasmic and MP from grass carp Different microwave power levels (300, 600, and 900 W) and heating times (0−60 s) A negative correlation was observed between protein solubility and microwave power and time. [27] Fish protein hydrolysates from farmed rainbow trout Oncorhynchus mykiss 3, 5, and 15 min (1,200 W, 20% power with 50% duty cycle at 50–55 °C), compared with conventional heating (water bath at 50 °C) Microwave treatment demonstrated higher protein solubility, emulsifying activity and emulsion stability than conventional heating with the same treatment. [94] Tilapia skin fish protein Various microwave power of 240, 400, 640, and 800 W for 1 and 4 min At the same microwave time or power, EAI and ESI increased with increased power or time. Microwave could significantly increase gel strength and hardness. [28] Protein from Alaska Pollock (Theragra chalcogramma) Various powers (300, 400, or 500 W) and time (10, 20, or 30 min) The gelling ability of microwave-heated protein was higher than that produced by water bath heating. Increasing microwave power and time resulted in decreases in both gel strength and WHC. [26] MP from silver carp Microwave (single-mode 2,450 MHz synthesizer) and water bath heating at
90 °C for 20 minMicrowave treatment generated a more stable, denser, and well-structured network compared with conventional heating. [30] Ultrasound
-
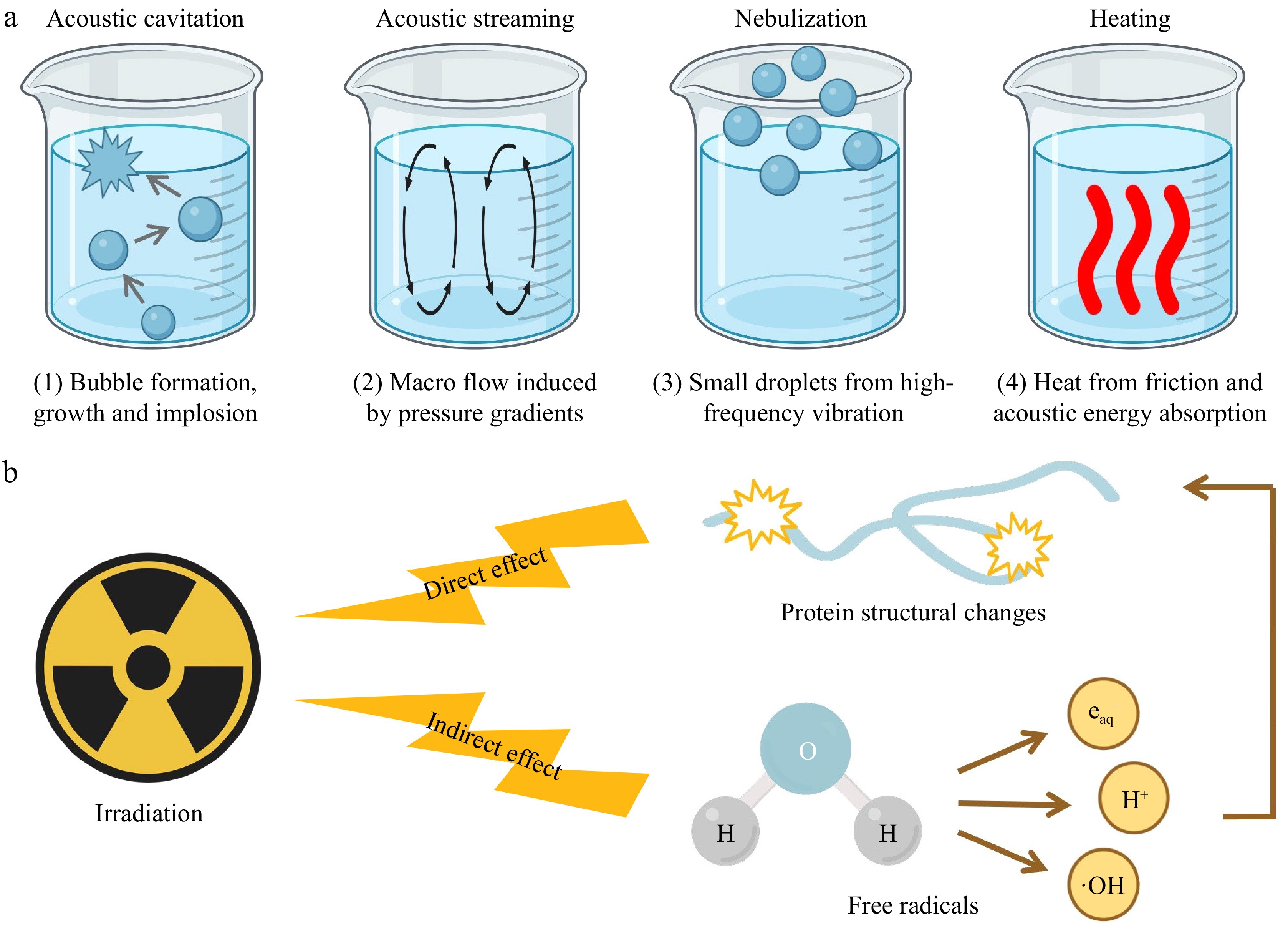
Ultrasound can be classified into low-intensity ultrasound used mainly for non-destructive food analysis, and high-intensity ultrasound that modifies food functional properties through four primary mechanisms (Fig. 4a). These features can disrupt the non-covalent interactions in fish proteins, resulting in alteration in molecular conformation and exposure of internal groups[10]. Furthermore, high-intensity ultrasound can also affect the aggregation state between fish proteins, facilitating either depolymerization or re-aggregation. Previous studies have elucidated the correlation between ultrasound and fish protein solubility. Cropotova et al.[7] have found a linear increase in the solubility of protein from Atlantic mackerel side streams with strengthened ultrasound power, and the ultrasound-treated samples exhibit significant differences compared with the untreated one as the study of Liu et al.[11]. Furthermore, ultrasound treatment can cause denaturation of protein with reduced secondary structure and increased flexibility. The reduction in their molecular size broadens the contact surface of fish proteins with water molecules, thus improving their solubility[12]. However, an extended processing time could weaken the solubility of myofibrillar proteins (MP) from Coregonus peled to a small extent with increased ultrasound power[13]. This phenomenon may result from disulfide linkages and hydrophobic interactions, leading to the recombination of macromolecular aggregates to cause the loss of protein solubility.
Besides, the ultrasound technique can also influence the emulsification and gelatinization of fish proteins. Pan et al.[14] subjected MP from golden pompano to ultrasound with different time and intensity, showing that both emulsifying activity index (EAI) and emulsifying stability index (ESI) increased and subsequently decreased with the escalation of ultrasonic time and power, as shown in previous research[15]. Ultrasound treatment can partially unfold fish proteins, exposing functional groups that enhance their adsorption and interaction at the oil-water interface[13]. Furthermore, ultrasound at an appropriate power can optimize the gelation performance of fish protein with improved storage modulus and gel strength[16].
High pressure
-
High-pressure treatment applies pressures above 100 MPa via fluid media, ensuring uniform pressure distribution based on the isostatic principle, which can induce the penetration of water within the protein to result in the hydration of protein chains[17]. This effect can impact the behavior of fish proteins, such as denaturation, unfolding, and aggregation. Since high pressure does not alter the covalent bonds, it preserves protein primary structure but disrupts secondary and tertiary structures by breaking ionic bonds and hydrophobic interactions.
The solubility of fish proteins exhibits a negative correlation with both the pressure level and holding time during high pressure[18,19]. Generally, the decreased solubility of fish proteins is linked to the pressure-induced reduction in protein volume, resulting in the conformational alteration of proteins. Protein molecules unfold to expose the hydrophobic residues under high pressure for weakening the hydration ability, and the pressure-induced denaturation can cause the generation of insoluble aggregates via intermolecular disulfide bridges. While solubility generally decreases with increasing pressure, other functional properties such as gelation can show opposite trends under moderately high-pressure conditions. moderate high-pressure treatment (≤ 300 MPa) is confirmed to enhance the gelling properties of myosin from golden threadfin bream[17,20]. However, with pressures higher than 400 MPa, this functionality can be weakened to some extent. Moderate high-pressure treatment produces a homogeneous reticular gel by exposing hydrophobic residues and sulfhydryl groups, thereby strengthening protein-protein interactions and gel properties[21]. Previous research indicates that the development of gel structure is largely contingent on the relative rate of protein unfolding and aggregation[21]. Fish proteins modified by ultra-high pressure undergo severe denaturation and aggregation, to make it difficult to dissociate and expand. Consequently, the unfolding speed of protein molecules becomes slow to prompt the formation of irregular networks. The emulsification of sarcoplasmic proteins from hake treated with high pressure presents a similar tendency[22]. High-pressure treatment at 200 MPa improves the emulsifying stability of hake sarcoplasmic proteins by reducing protein size and exposing hydrophobic groups, whereas higher pressures induce denaturation that promotes droplet flocculation and reduces emulsion stability.
Irradiation
-
Irradiation is an effective non-thermal modification technique, modifying fish protein directly or indirectly (Fig. 4b). Free radicals can induce the crosslinking and polymerization of fish proteins to alter their molecular configuration and composition. In addition to crosslinking, irradiation-generated radicals can also trigger oxidative modifications such as carbonylation, fragmentation, and aggregation, further impacting the structural and functional properties[23]. Shi et al.[24] have indicated that irradiation has a detrimental influence on the emulsification characteristics of MP from grass carps. The phenomenon occurs because hydroxyl and superoxide anion radicals formed by water radiolysis can alter the primary structure of fish proteins and result in cross-linking to affect the proteins' interaction with oil. A 7 kGy irradiation dose is considered optimal to enhance the gel properties of hairtail proteins[25]. This moderate level of irradiation may induce a balanced degree of protein unfolding and cross-linking, which facilitates the formation of an ordered and compact gel network.
Microwave
-
Microwaves modify fish proteins by inducing carbon-centered free radicals through absorption by polar groups, thereby altering protein secondary and tertiary structures[26]. Besides, the thermal effect of microwave is another important consideration. Microwave heating arises from energy absorption that drives the rotation and collision of dipolar water molecules and the motion of ions, converting electromagnetic energy into heat and accelerating protein structural and functional changes. Cai et al.[27] have discovered that microwave treatment reduces the solubility of grass carp sarcoplasmic and myofibrillar proteins, with moderate power disrupting non-covalent bonds to alter protein-water and protein-protein interactions, while high power induces irreversible denaturation and aggregation via disulfide bonding and hydrophobic interactions.
As the research of Kang et al.[28], high-power and prolonged microwave treatment increases the EAI and ESI of tilapia skin proteins by enhancing molecular flexibility and interfacial adsorption. Meanwhile, Li et al.[29] have discovered that fish proteins treated with appropriate microwave exhibit improved gel strength and water-holding capacity (WHC), especially at low power levels and short exposure times. Interestingly, Jiao et al.[30] employed an animal nesting model to show that microwave-treated fish proteins form ordered, compact bee-nest-like aggregates, whereas water-bath treatment results in loose, unstable ant-nest-like structures.
Chemical modification
-
Chemical modification of fish proteins encompasses a broad spectrum of techniques aimed at enhancing or altering protein structure and functionality, thereby increasing their suitability and versatility in various food and nutraceutical applications. These techniques typically involve applying chemical agents and treatments, such as pH-shift methods, phosphorylation, enzymatic cross-linking, and other chemical processes (Fig. 5). Based on their modification mechanisms, the ways in which these chemical methods influence fish proteins, and key findings are summarized in Table 2.
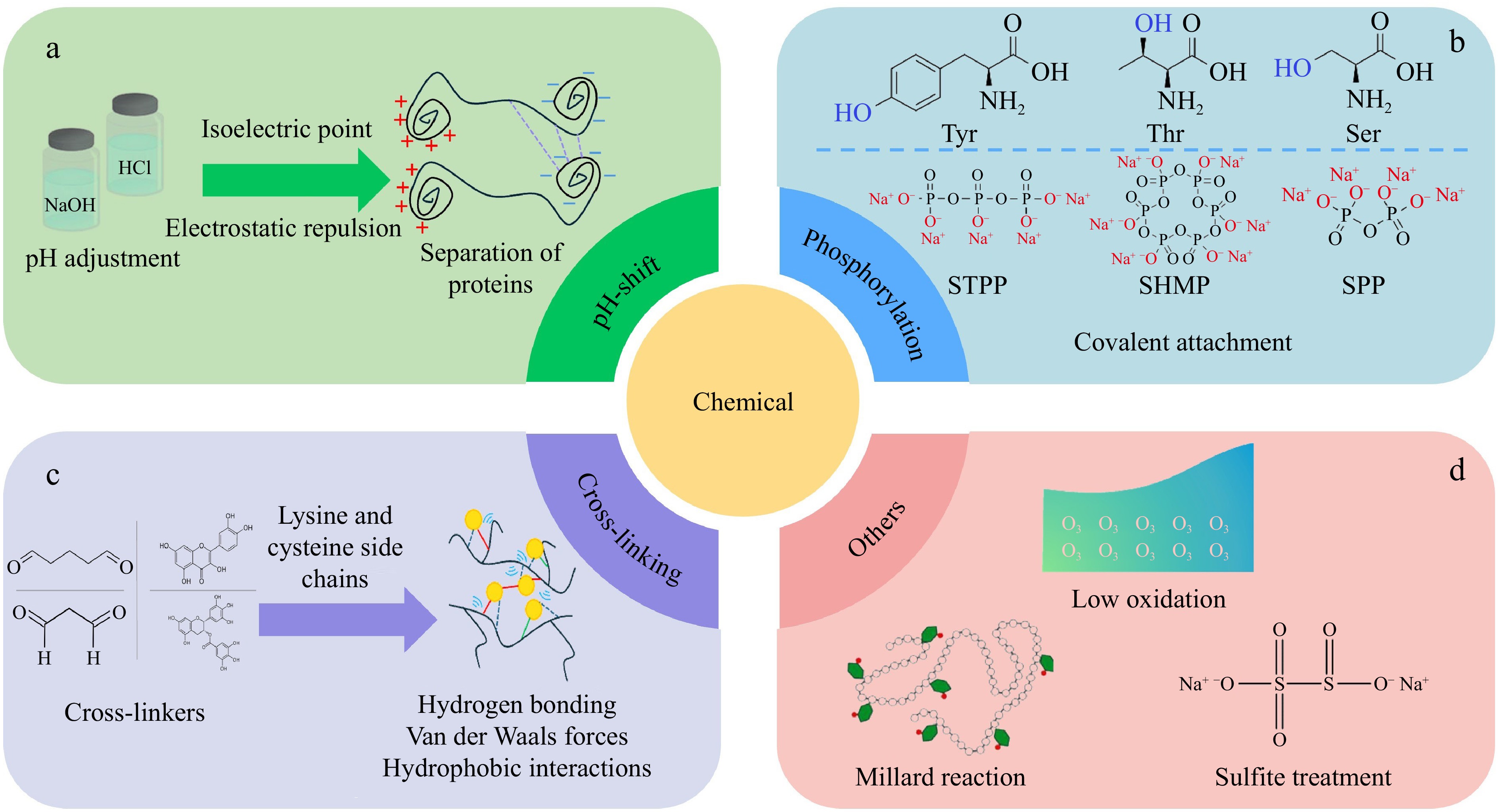
Figure 5.
Chemical modification methods of fish proteins: (a) pH-shift, (b) phosphorylation, (c) cross-linking, and (d) others.
Table 2. Chemical modification methods for fish proteins.
Modification
methodsFish protein Treatment conditions Important findings Ref. pH shift Myosin from blue round scad (Decapterus maruadsi) Acidic or alkaline solubilization followed by neutralization; heating from 20–90 °C Acidic pH-shift irreversibly damaged myosin, preventing gelation; alkaline treatment partially restored structure and gel-forming ability compared to native protein [34] Common carp (Cyprinus carpio) protein isolates Solubilization at pH 2.5 (acidic) or 12.5 (alkaline), followed by precipitation at pH 5.5 Acidic and alkaline isolates showed high protein yield (~76% to 88%), reduced fat/ash, improved digestibility (7–8 ×). [31] Protein from salmon, cod, and herring by-products Acidic or alkaline solubilization at pH 11.5–12.5, isoelectric precipitation at pH 5.5 Alkaline process maximized yield (salmon > herring > cod); solubilization pH effects on gel strength, WHC, and color was species-specific. [32] Baltic herring (Clupea harengus membras) muscle protein isolates Alkaline pH-shift extraction (solubilization pH ~11.2, precipitation pH ~5.4), tested in surimi-type gels and fish balls Reduced fishy odor; gels matched surimi hardness/cohesiveness but were darker; altering pH softened gels without improving whiteness. [35] Bighead carp (Aristichthys nobilis) myosin particles Myosin particles (0.6 M NaCl) used for Pickering emulsions; stability tested over pH 3–11 Emulsions showed high stability at neutral–alkaline pH; acidic pH (≤ 5) caused droplet aggregation due to myosin's proximity to pI. [3] Phosphorylation Grass carp (Ctenopharyngodon idellus) MP Addition of sodium tripolyphosphate, tetrasodium pyrophosphate, sodium hexametaphosphate (0.1%–0.5%, w/w) Sodium tripolyphosphate and tetrasodium pyrophosphate improved gel strength, WHC, and reduced water mobility [36] Fish gelation Phosphorylation with sodium pyrophosphate (FG : TSPP ratio 40:0 to 40:6) at pH 7.0 Increased gel strength, viscosity, emulsifying properties; reduced water mobility; excessive TSPP shortened gelation time. [38] Fish scale gelatins from Coregonus peled, Esox lucius, and Ctenopharyngodon idella Phosphorylation via sodium trimetaphosphate (STMP), phosphorylating for 0–3 h Increased surface hydrophobicity and emulsification activity/stability; structural changes supported improved emulsions. [40] Mantis shrimp (Oratosquilla oratoria) MP Phosphorylation via sodium tripolyphosphate at low ionic strength (0.1 mol/L KCl) Phosphorylation improved solubility, gel strength, and emulsifying properties; enhanced WHC; modified protein structure for better functionality. [41] Chemical
cross-linkingFish skin gelatin (salmon gelatin, SG; tilapia gelatin, TG) Crosslinked with glutaraldehyde (GA) at 0.05%–0.15% to form hydrogel At 0.05% GA, hydrogels had optimal swelling, porosity, and α-glucosidase binding with best antidiabetic potential. [95] Antarctic krill protein (AKP) combined with cellulose Glutaraldehyde (GA) cross-linking to form multiple network with cellulose Multiple cross-linking enhanced crystallinity, orientation, mechanical and moisture-retention properties at 0.2 wt% GA. [96] Grass carp (Ctenopharyngodon idella) myosin Binding with chlorogenic acid (CGA) and rosmarinic acid (RA); spectroscopic analyses (fluorescence, UV–Vis, CD) CGA and RA bind myosin via H-bonding/van der Waals and hydrophobic; increase α-helix, protect sulfhydryls, complexes show antioxidant effects and promote aggregation. [97] Silver carp (Hypophthalmichthys molitrix) MP Crosslinked with oxidized polyphenols with layered double hydroxide nanosheets Dual modification enhanced tensile strength, barrier, and antioxidant activity; improved film stability for food packaging applications. [48] pH shift
-
The pH-shift method is essential for enhancing the functional properties of fish proteins in food processing. One of the primary effects of pH modification on fish proteins is the structural conformation alteration. Extreme pH levels increase electrostatic repulsion, causing protein unfolding and hydrophobic exposure. This improves solubility and facilitates the removal of lipids, pigments, and connective tissue. It has been reported that fish proteins treated through pH-shift can exhibit a high nutritional value, retaining essential amino acids necessary for human health[31]. In addition to solubility, employing a pH-shift strategy significantly improves the gelation property of fish proteins[32]. By altering the pH, it is possible to form strong, stable gel networks for improving WHC and gel matrix formation, which is vital for creating high-quality fish-based products[33]. It's worth noting that alkaline-treated proteins tend to refold close to their native structure, preserving their ability to form strong gels compared with acid treatment[34]. One previous study explored the production of protein isolate from Baltic herring using an alkaline pH-shift process, demonstrating a high yield (~82%), reduced fishy odor due to lipid removal, and good textural qualities in surimi-type gels and fish balls[35]. Moreover, it has been demonstrated that fish proteins obtained via pH-shifting often exhibit high emulsification activity compared to traditional extraction[3]. This is attributed to the exposure of hydrophobic groups during the extreme pH conditions to promote the interactions in emulsions.
Phosphorylation
-
Phosphorylation is another key method for enhancing fish protein functionality. Phosphates such as sodium tripolyphosphate, sodium hexametaphosphate, and sodium pyrophosphate are commonly employed to modify both the structural and functional properties of these proteins[36,37]. The mechanism of phosphorylation involves the covalent attachment of phosphate groups to the hydroxyl groups of amino acids such as serine, threonine, and tyrosine. The introduction of negative charges into the protein via this modification breaks intermolecular links, enhancing its solubility and WHC[38]. Phosphate groups also promote protein unfolding, increasing their accessibility to water and allowing the interactions with other food components. This property is particularly beneficial in fish proteins, given their tendency to denature and form aggregates when processed[39].
One of the major applications of phosphorylation is to improve the gelation property of fish proteins. Some studies have shown that phosphorylation improves the elasticity and firmness of fish gelatin. For example, the use of sodium tripolyphosphate could enhance gel strength, increase surface hydrophobicity, and alter the structural properties of fish gelatin by increasing β-sheet/β-turn contents[38]. Phosphorylation also plays a significant role in improving the emulsifying properties. The introduction of phosphate groups increases the surface activity of fish proteins, allowing them to adsorb effectively at the oil-water interface[40]. Another important application of phosphorylation is in improving the cryoprotective properties of fish proteins. Freezing and thawing can cause significant damage to fish proteins, leading to moisture loss, texture degradation, and protein denaturation. Phosphorylation helps mitigate these effects by increasing the water-binding capacity of proteins to reduce ice crystal formation during freezing and maintain the structural integrity of fish products[41]. This property is particularly valuable in frozen fish products for maintaining the texture and moisture content of fish products.
Chemical cross-linking
-
Chemical cross-linking is another common technique used in fish proteins to create cohesive and stable protein matrices. The mechanism of chemical cross-linking involves covalent bonds and intermolecular forces between protein molecules[42]. Cross-linking agents such as aldehydes and phenolic compounds facilitate chemical reactions by interacting with specific amino acid residues in proteins. These interactions strengthen the fish protein structure, improving its gel-forming capacity and resistance to physical stress.
Aldehydes, such as glutaraldehyde and malondialdehyde, are well-known for their ability to induce protein cross-linking through the formation of covalent bonds between lysine residues on protein molecules[43]. These aldehydes react with the amino groups of lysine side chains, creating stable cross-links to strengthen the protein matrix to form gelation for improving overall texture. Glutaraldehyde is effective at forming stable protein networks, resulting in firm and elastic gels with highly resistant to mechanical stress[44]. Besides, malondialdehyde-induced cross-linking enhances the WHC and gel strength of MP in silver carp, nearly doubling compared to the control, which is critical for reducing water loss during cooking[45].
Phenolic compounds have emerged as a promising alternative to traditional chemical cross-linkers due to their antioxidant properties and lower toxicity compared to synthetic agents. For example, epigallocatechin gallate (EGCG), quercetin, catechins, and other plant-derived polyphenols can induce protein cross-linking through their ability to form electrostatic interactions, Van der Waals forces, and hydrogen bonding with protein molecules, particularly through interactions with amino acid residues like lysine and cysteine[46]. The interactions between fish proteins and phytophenols, whether covalent or non-covalent, result in the improvement of protein functionality, making phytophenols attractive to improve the fish quality in the food industry due to their natural origin and potential health benefits[47]. Additionally, phenolic-induced cross-linking has been shown to improve the oxidative stability of fish proteins, protecting them from degradation during processing and storage. For example, one study has developed mechanically robust and antioxidative films using silver carp MP with oxidized polyphenols and layered double hydroxide[48].
Others
-
In addition, other chemical modification methods, such as oxidation, Maillard reaction, and sulfite treatments, also play important roles in fish protein modifications. Oxidation reactions are commonly encountered in fish protein processing, which are rich in polyunsaturated fatty acids and prone to oxidative degradation. However, excessive oxidation can lead to protein denaturation, off-flavors, and compromised nutritional value. Controlling oxidation with mild oxidizing agents can improve fish protein functionality by inducing favorable structural changes to minimize negative impacts. For instance, ozone water rinsing could oxidize MP of grass carp, improving their color and aroma characteristics without degrading protein quality[49]. Maillard reaction occurs between reducing sugars and amino acids in proteins, which not only enhances flavor development through browning reactions but also improves protein functionality by inducing glycation for fish proteins. Glycation involves the attachment of sugar molecules to protein amino groups, improving emulsifying properties in fish protein-based products. Li et al.[50] have found that glycation of chum salmon MP with uronic acid-type reducing sugars via the Maillard reaction enhances the anti-inflammatory activity of the proteins and improves their emulsifying properties. Sulfite treatment is also used to modify the properties of fish proteins, particularly in preventing oxidation and enhancing protein solubility. Du et al.[51] have demonstrated that sulfite-mediated self-assembly improves the quality of silver carp MP, leading to the enhancement of gel formation and texture. The reduction of disulfide bonds by sulfites could prevent protein aggregation and improve the solubility and functionality of fish proteins, making them suitable for use in processed foods. However, sulfites must be carefully controlled due to potential allergenic responses in sensitive individuals and regulatory limitations on sulfite levels in food products.
Biological modification
-
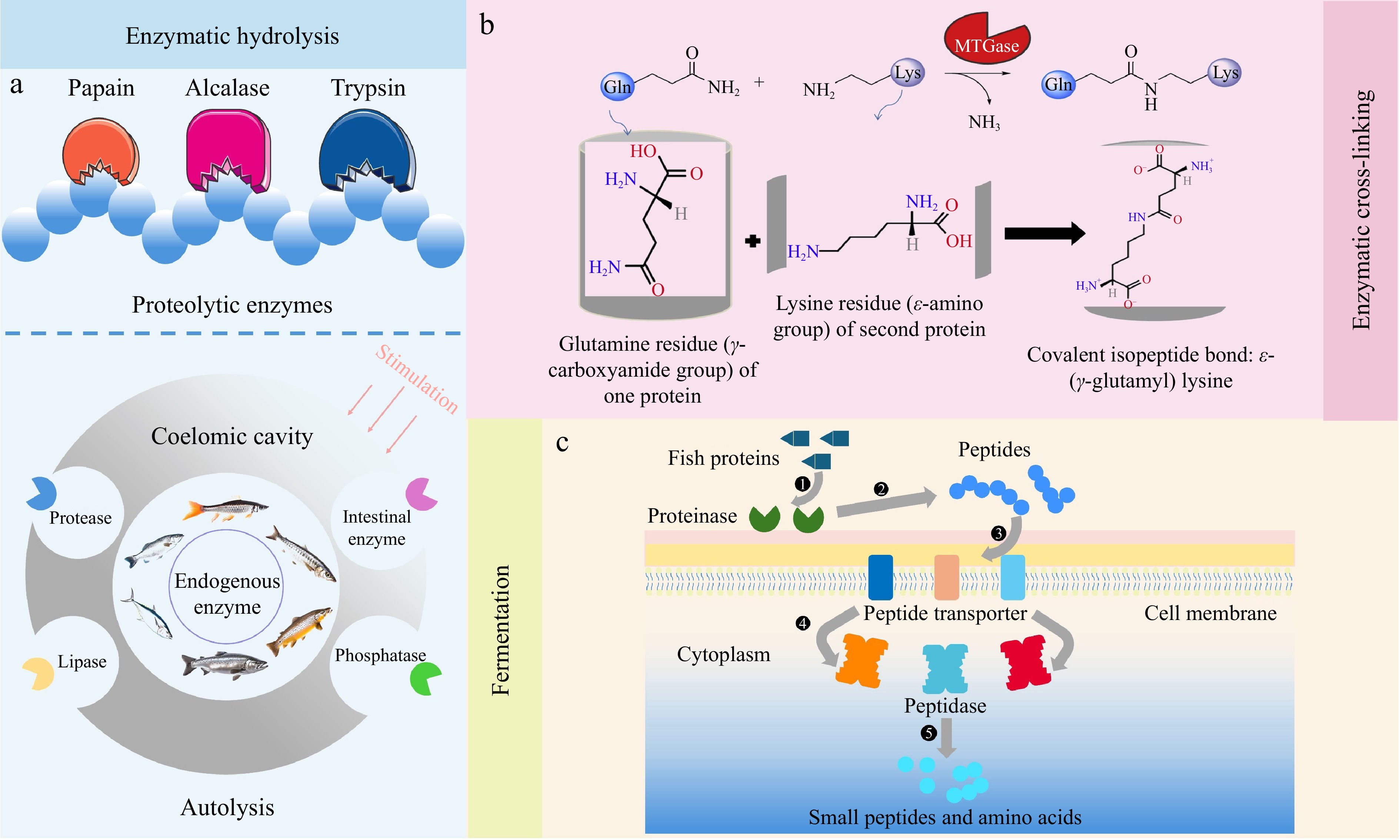
Biological modification methods have been extensively studied for their ability to improve the nutritional and functional properties of fish proteins. These techniques employ natural processes such as enzymatic reactions and microbial activity to enhance protein solubility, bioavailability, and stability while producing valuable bioactive compounds. This section explores the main biological modification approaches, including enzymatic hydrolysis, enzymatic cross-linking, and fermentation (Table 3). The modification mechanisms in fish protein functionality are presented, offering broader possibilities for its application (Fig. 6).
Table 3. Biological modification methods for fish proteins.
Modification methods Fish protein Treatment conditions Important findings Ref. Enzymatic hydrolysis Bighead carp (Hypophthalmichthys nobilis) protein hydrolysate (ficin-mediated) Ficin hydrolysis at pH 6, 40 °C, 3% enzyme/substrate for 1–6 h At DH 20.15%, highest yield, protein content, amino acid content, and solubility; OHC (oil-holding capacity) highest at lowest DH. [54] Chinese sturgeon (Acipenser sinensis) protein hydrolysate Papain hydrolysis (pH 6, 70 °C, 3% E/S, 6 h) DH reached 24.9%; high yield (17.5%), solubility (87%–99%), emulsifying/stability indices, WHC, OHC, and foam capacity. [55] Seabream (Sparus aurata) and seabass (Dicentrarchus labrax) fish protein hydrolysates Enzymatic hydrolysis of by-products (frames & trimmings, heads, viscera) using Alcalase; optimized via response surface methodology Frames & trimmings produced hydrolysates with highest antioxidant and antihypertensive activity, MW ~1.4–2.0 kDa, > 86% digestibility, and excellent essential amino acid profile. [57] Rainbow trout (O. mykiss) by-product autolysate Autolysis at 40 °C for 1 h, followed by heat inactivation Autolysis enriched small peptides but increased lipid and protein oxidation; hydrolysate still displayed antioxidant activity. [62] Enzymatic cross-linking Surimi (silver carp MP gel) MTGase-crosslinked at 29.7%, 45.8%, 63.0%; frozen via liquid nitrogen immersion (LNF), –35 °C, –18 °C Higher cross-linking (45%–63%) improved texture retention, reduced oxidation, and preserved aroma—LNF was superior to air freezing. [64] Surimi MTGase-induced cross-linking at increasing degrees < 30% cross-linking: mushy; ~40% to 55%: elastic/tough; ≥ 64%: brittle; WHC fell; pore structure and hydrophobicity altered. [65] Rainbow trout (Oncorhynchus mykiss) mince Myofibrillar restructuring with 0%–1.0% microbial transglutaminase (MTGase) during refrigerated storage MTGase reduced spoilage indicators (TVB-N, TMA-N), suppressed free amino acids increase, and inhibited psychrophilic and coliform growth—best at 1.0%. [66] Tambaqui (Colossoma macropomum) skin gelatin Transglutaminase (TGase) crosslinking at 1%–3% (w/w) 3% TGase increased Bloom strength; film thickness and mechanics unchanged; solubility and water vapor permeability significantly reduced. [67] Fermentation Enrichment of fermented fish sausages Selected autochthonous Lactobacillus strains (from fish gut microbiota); applied as starter cultures Three strains (IDs 11, 68, 69) reduced fermentation time (~2 d) and ensured microbiological safety, showing probiotic potential. [70] Grass carp (Ctenopharyngodon idella) fish protein (semi-dried meat) Fermented with Monascus purpureus Went M3.439; then subjected to in vitro digestion and fermentation assays Fermentation enhanced free amino acids and glutamate, reduced ammonia/indole, increased propionic acid and Parabacteroides during in vitro fermentation, suggesting improved digestibility and gut health. [94] Atlantic cod (Gadus morhua) muscle protein Fermentation with Lactobacillus helveticus strain Lh191404 Fermentation disrupted allergen structure and linear epitopes, reducing immunoreactivity [72] Crucian carp (Carassius auratus) suanyu (fermented fish) Mixed starter culture: Enterococcus rivorum + E. lactis, compared to natural fermentation over 13 d Mixed culture boosted protease activity, TCA-soluble proteins, softness and flavor compounds (aldehydes, glutamate); protease activity correlated with hardness, chewiness, flavor. [73] Enzymatic hydrolysis
-
Enzymatic hydrolysis has gained significant attention for modifying fish proteins due to its ability to produce bioactive peptides and enhance functional properties. This process uses proteolytic enzymes to break down proteins into small hydrolysates, possessing diverse applications in food, nutraceuticals, and aquaculture[52]. The effectiveness of enzymatic hydrolysis is influenced by many factors, including the type of enzyme, the degree of hydrolysis (DH), and the conditions under which hydrolysis occurs. Common proteolytic enzymes employed for fish protein hydrolysis include papain, Alcalase, trypsin, and pepsin, each exhibiting distinct specificities for cleaving peptide bonds[53]. For example, Alahmad et al.[54] have demonstrated that alcalase is particularly effective in producing hydrolysates from bighead carp with high nutritional and functional qualities. Moreover, the DH is a critical parameter to influence the functional attributes of the hydrolysates. Noman et al.[55] have observed that adjusting the DH during hydrolysis can tailor the functionality of protein hydrolysate from Chinese sturgeon for specific applications in food systems. A high DH generally leads to small peptides to enhance solubility and bioavailability but may reduce gelation and emulsification properties[56]. Furthermore, parameters including temperature, pH, enzyme concentration, and substrate-to-enzyme ratio can be adjusted to influence the DH and the characteristics of the resulting peptides. Valcarcel et al.[57] have demonstrated that optimizing these parameters could enhance the functional properties of protein hydrolysates from seabream and seabass byproducts. This highlights the importance of process control in maximizing the efficiency and functionality of enzymatic hydrolysis for fish protein modification.
One of the major advantages of enzymatic hydrolysis is its capacity to produce bioactive peptides with a range of health benefits. Yathisha et al.[58] have found that fish protein hydrolysates exhibit potent antihypertensive activity due to the presence of bioactive peptides that inhibit angiotensin-converting enzyme, making enzymatic hydrolysates valuable in the development of functional foods and nutraceuticals targeting cardiovascular health. Additionally, the antioxidant properties of fish protein hydrolysates further highlight their potential in mitigating oxidative stress in biological systems, thus contributing to the prevention of chronic diseases, as noted by Wijethunga et al.[59]. Meanwhile, when conducted under optimized conditions, the hydrolysis of fish proteins can reduce bitterness and fishy odors by breaking down undesirable compounds, and facilitating downstream flavor enhancement strategies, thereby improving palatability and suitability for food applications[60].
In addition to conventional enzymatic hydrolysis, extensive research has been done on autolysis and self-digestion with fish proteins. It is an enzymatic process, as shown in Fig. 6a, where endogenous enzymes like cathepsin within fish tissues naturally cleave proteins into peptides and amino acids[61]. This method is particularly beneficial for utilizing fish byproducts, as it requires no external enzyme addition and results in high-value protein hydrolysates rich in bioactive compounds. Nikoo et al.[62] have demonstrated that the autolysis of rainbow trout byproducts enhances lipid and protein breakdown, producing hydrolysates with improved nutritional and functional qualities. Moreover, autolytic hydrolysis has been shown to yield peptides with antioxidant activities, improve digestibility and bioavailability in the fish, offering a sustainable potential for food and nutraceutical application[52].
Enzymatic cross-linking
-
Enzymatic cross-linking has gained significant attention in the fish protein modification because of its capacity to enhance the functional properties of proteins, particularly in enhancing gelation, texture, and WHC. Among various enzymes, microbial transglutaminase (MTGase) is the most widely employed in the food industry. The mechanism of action of MTGase, as shown in Fig. 6b, involves catalyzing acyl transfer reactions between the γ-carboxamide group of glutamine residues and the ε-amino group of lysine residues, forming covalent bonds to stabilize the protein network[63]. This reaction results in a cohesive protein matrix for improving the textural properties of fish-based products. The application of MTGase in fish protein modification offers an ideal alternative to chemical additives, aligning with the surging demand for clean labels in the food industry.
MTGase plays a crucial role in improving gel strength and elasticity in surimi production. Surimi is rich in fish proteins used in products like fish ball and imitation crab meat. The enzymatic cross-linking by MTGase enhances the WHC and resilience of surimi, especially after freezing and thawing for avoiding weaken protein structure[64]. It is noted that MTGase cross-linking could improve the physiochemical properties of surimi gel to enhance consumer acceptability at a moderate cross-linking degree[65]. Meanwhile, this modification is particularly useful in producing restructured fish products from byproducts, allowing for broadening of the utilization of fish resources to enhance the product quality. For example, the quality of rainbow trout can be improved by adding exogenous MTGase, which extends shelf life and eliminates unpleasant odors[66]. Moreover, enzymatic cross-linking has also been applied to enhance the mechanical properties of fish gelatin films in packaging and edible coatings. de Oliveira et al.[67] have reported that MTGase cross-linking improves the tensile strength and barrier properties of tambaqui (Colossoma macropomum) skin gelatin films, making them suitable for food packaging. This highlights the versatility of enzymatic cross-linking in extending the fish protein applications in sustainable packaging beyond the traditional food product modification.
Fermentation
-
Fermentation is widely used in food modification that leverages the metabolic activities of microorganisms. In fish protein modification, fermentation serves as a preservation method and impacts protein breakdown, bioactive compound production, and flavor development[68]. The application of fermentation in fish processing has demonstrated its ability to improve digestibility, generate bioactive peptides, and produce a variety of flavor-enhancing compounds.
The breakdown of fish proteins during fermentation is driven by the proteolytic activity of microorganisms such as lactic acid bacteria (LAB), yeasts, and fungi[69]. LAB are the most used microorganisms in fish fermentation due to their ability to low the pH, inhibit spoilage organisms, and produce proteolytic enzymes to degrade fish proteins into functional peptides[70]. Fungi, such as Monascus, have also been applied in fish fermentation to enhance the digestibility of fish proteins and produce peptides with antioxidant activities, as seen in Liu et al.[71]. This highlights the significance of selecting suitable microorganisms capable of releasing enzymes, as seen in Fig. 6c, such as certain proteinases and peptidases, to facilitate the hydrolysis of fish proteins into smaller peptides and amino acids, thereby improving both their digestibility and bioavailability. Moreover, Zou et al.[72] have reported that fermentation of Atlantic cod with Lactobacillus helveticus, particularly strain Lh191404, effectively degraded allergenic proteins and disrupted IgE-binding epitopes, reducing immunoreactivity and indicating potential for hypoallergenic fish products. This property is particularly important in expanding the consumer base for fish-derived products to include individuals with seafood allergies.
Fermentation also plays a vital role in improving the functional properties of fish proteins, such as emulsification and foaming capacity, which are essential for their application in processed foods. The enzymatic activity of microorganisms during fermentation enhances the surface-active properties of fish proteins to stabilize emulsions and foams effectively. Xu et al.[68] have highlighted the technological roles of microorganisms in fish fermentation, showing that the metabolic byproducts produced by LAB and yeasts contribute to the formation of stable protein-lipid interfaces, which are crucial for the development of fish-based emulsions and spreads. Yuan et al.[73] have conducted a study on the dynamic changes in texture and flavor compounds during the fermentation of crucian carps, showing that the breakdown of proteins and lipids leads to an improved mouthfeel and enhanced sensory attributes. This makes fermentation a valuable tool for developing processed fish products with good texture and stability, such as fermented fish sausages and surimi-based products.
The fermentation process is crucial for improving the nutritional and functional properties of fish proteins, and enhancing flavor profiles, which is essential in traditional fermented fish products like fish sauce and other fish-based condiments. Feng et al.[74] have highlighted the complex relationship between lipid metabolism and microbial activity during fish fermentation, showing that microbial action influences the formation of volatile flavor compounds. The interaction between lipid degradation and the activity of LAB and other microorganisms leads to the production of distinct flavors in the fermented fish products. Yang et al.[75] have further supported this by analyzing microbial succession in the fermentation of traditional Chinese fish products, Chouguiyu, where the dynamic changes in microbial communities are linked to shifts in flavors and aroma compounds. Overall, this microbial activity is crucial in developing desirable sensory attributes, such as umami and savoury flavors in fish-based fermented products.
Other modifications
-
Aside from physical, chemical, and biological methods, the modification of fish proteins through other methods has been widely studied to enhance their functional properties. These modifications, ranging from the addition of specific substances to deep eutectic solvents and emerging AI-assisted techniques, are designed to improve the structural integrity, solubility, emulsifying ability, and gel strength of fish proteins, offering a versatile approach to meet diverse application demands in the food industry.
Additives
-
Various additives, such as polysaccharides, fat, and salt ions, have been developed and applied for fish protein modification. Wu[76] showed that Opuntia dillenii polysaccharide improves the WHC and textural properties of Trichiurus lepturus MP by forming a stable protein-polysaccharide gel network. Ren et al.[77] pointed out that chitosan can enhance the hydrophilicity, thermal stability, and structural compactness of MP from golden threadfin bream, enabling the formation of stable high-internal-phase emulsions for advanced food applications.
He et al.[78] demonstrated that fish oil exerts protein-dependent effects on fish MP gels and emulsions: appropriate amounts enhance gel strength and emulsion stability by filling network gaps, restricting protein mobility, and forming physical bridges between oil droplets and proteins. However, when the oil content exceeds the capacity of the protein network, oil leaking occurs because proteins are insufficient to cover the surface of extra oil, resulting in an increased proportion of free liquid[78]. Besides, Na+ addition increases the gel strength of grass carp MP by enhancing protein hydration and stabilizing protein interactions.[79]. Additionally, the low gel strength observed in the Mg2+ and Ca2+ treatments indicates the negative impact on the gel integrity.
Deep eutectic solvents
-
Deep eutectic solvents (DES) have emerged as a promising green processing medium for the extraction and modification of fish proteins and bioactive peptides from fishery by-products. DES are formed by combining a hydrogen bond donor and a hydrogen bond acceptor, often from biodegradable, food-grade components such as choline chloride, organic acids, urea, or polyols, to create a eutectic mixture with a melting point lower than the individual components[80]. These solvents possess tunable physicochemical properties, such as polarity, viscosity, and hydrogen bonding capacity, that can be optimized to selectively solubilize and modify proteinaceous materials without the need for hazardous organic solvents[81].
Recent studies have demonstrated that DES can serve as a sustainable and efficient medium for extracting proteins and bioactive derivatives from fish processing residues, offering comparable or superior yields to conventional solvents while enhancing functional properties. For example, betaine/polyol-based DES applied to sardine processing waste have achieved protein recoveries similar to water extraction but have produced extracts with up to threefold higher antioxidant and over 250-fold greater antimicrobial activities, likely due to selective solubilization of bioactive amino acids and peptides[82]. Similarly, betaine–propylene glycol DES, when coupled with ultrasound pretreatment and an aqueous two-phase system purification, have effectively extracted proteins from tilapia viscera, which are subsequently hydrolyzed by alcalase to yield low-molecular-weight peptides with potent DPP-IV inhibitory activity[83]. The combined DES–ultrasound approach enhances protein structural disruption and mass transfer, facilitating peptide release and improving extraction efficiency. These findings underscore the potential of DES, particularly when integrated with physical intensification techniques, as green alternatives for valorizing fish byproducts into high-value functional ingredients.
AI-assisted
-
In recent years, AI has emerged as a promising tool for accelerating fish protein modification by enabling rapid prediction, design, and optimization of processing strategies and functional properties. AI techniques can model complex, non-linear relationships between protein sequences, structural features, and bioactivities, reducing reliance on labor-intensive experimental screening. Zhao et al.[84] have reviewed how methods such as random forests, convolutional neural networks, and other deep learning architectures have accelerated bioactive peptide discovery, functional characterization, and structure–activity relationship modeling. For example, Zhang et al.[85] have demonstrated a generative modeling pipeline, where a long short-term memory (LSTM) model iteratively creates de novo peptide sequences and a DeepPep screening model evaluates binding potential, effectively enabling AI-driven generation and prioritization of novel bioactive peptides.
The integration of bioinformatics with machine learning (ML) has also been successfully applied to fish-derived peptides. Jafarpour et al.[86] have combined proteomic analysis of codfish (Gadus morhua) frame hydrolysates with ML-based bioactivity prediction to identify specific peptides contributing to emulsifying and antioxidative properties. Similarly, Phetchthumrongchai et al.[87] have hydrolyzed skipjack tuna (Katsuwonus pelamis) roe proteins, and applied k-nearest neighbors (k-NN) and random forest (RF) models to predict multiple bioactivities, including antihypertensive, antiviral, and antiparasitic functions. These predictive approaches facilitated the rapid selection of multifunctional peptide candidates for further validation, underscoring the role of AI/ML as a complementary tool to conventional physical, chemical, and biological modification techniques in the valorization of fishery by-products.
-
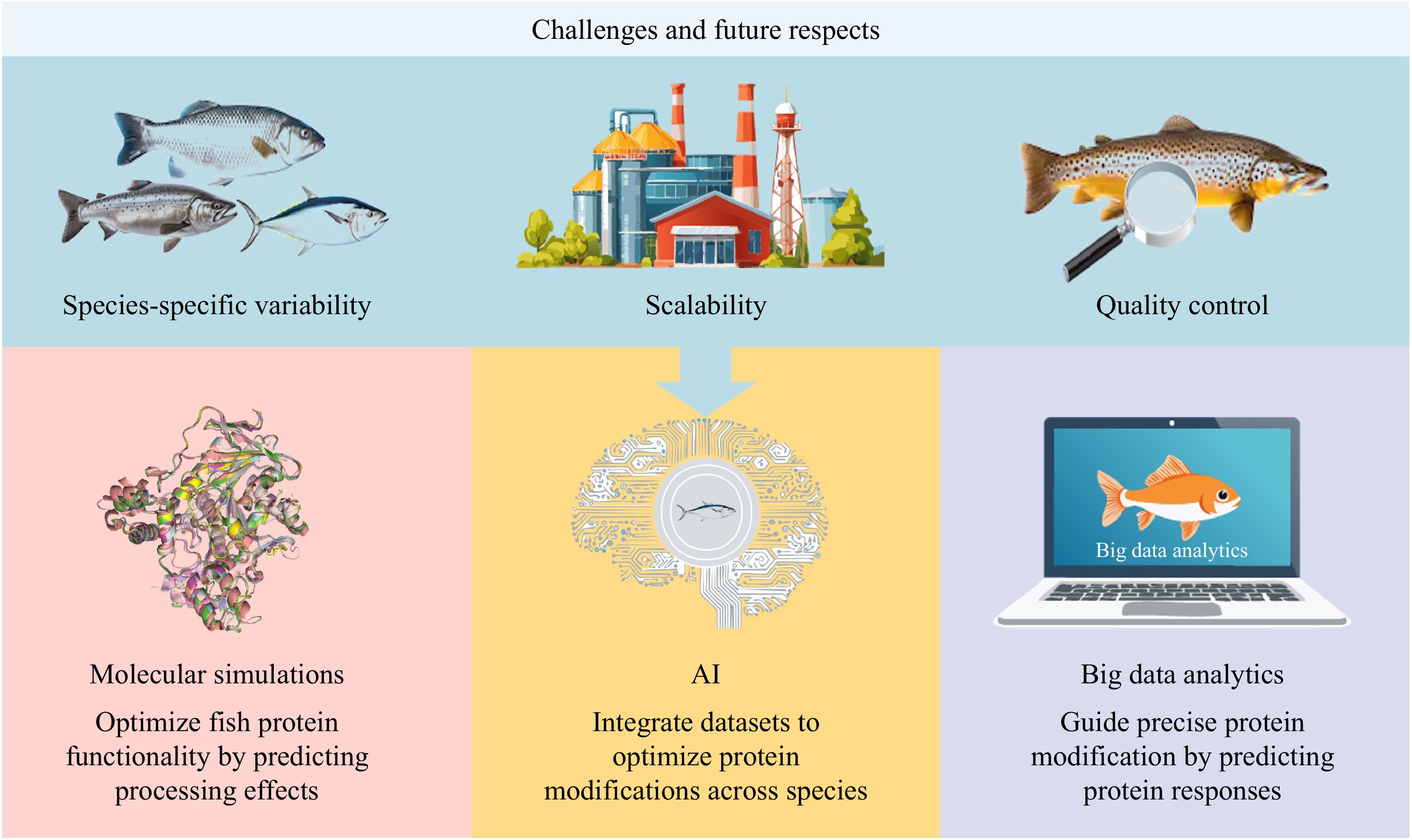
The modification of fish proteins has advanced through physical, chemical, and biological methods. However, challenges such as species-specific variability, scalability, and quality control necessitate innovative solutions (Fig. 7). The intersection of fish protein modification with advanced technologies such as molecular simulations, AI, and big data analytics presents a transformative opportunity to optimize processes, enhance sustainability, and address industry challenges. By incorporating these advanced tools, researchers and industries can enhance the precision and efficiency of fish protein modification methods, unlocking new potential in the field. Molecular simulations provide powerful insights into the structural and functional behavior of fish proteins under various modification conditions. By using techniques such as molecular dynamics and quantum mechanics/molecular mechanics simulations, researchers can predict how proteins respond to enzymatic hydrolysis, pH-shifting, or high-pressure treatments at the atomic level[88]. These simulations can reveal the effects of specific treatments on protein folding, stability, and binding affinities, guiding the design of precise and effective modification strategies. For example, molecular dynamics simulations can model the impact of enzymatic cleavage on protein solubility and identify optimal cleavage sites, while quantum mechanics/molecular mechanics simulations can predict how phosphorylation affects emulsification properties.
AI-driven optimization offers a powerful tool to improve fish protein modification processes. ML models can predict the effects of processing parameters, such as pH and temperature, on the functional properties of fish proteins[89]. This capability enables researchers to fine-tune conditions for achieving specific goals without extensive experimental trials. Furthermore, AI can be employed in real-time monitoring systems to adjust parameters dynamically, ensuring consistent quality and reducing waste. These smart systems are especially valuable in scaling laboratory-level methods to industrial production, where variability in raw materials and environmental conditions often complicates process standardization. Big data analytics further enhances the integration of these tools by enabling large-scale data aggregation and analysis across fish protein studies. For example, big data can merge proteomic and metabolomic information to chart the molecular compositions and functional traits of fish proteins from various species[90]. This information can guide the selection of optimal modification strategies tailored to specific species and applications. For example, enzymatic hydrolysis can be optimized by predicting the most efficient combinations of enzymes and conditions based on the characteristics of raw material through big data analytics[91]. Additionally, combining molecular simulations with big data tools can enable multiscale modeling, linking atomic-level protein behavior with larger-scale functional outcomes.
However, high computational costs, data security concerns, and the need for interdisciplinary collaboration often limit implementation. In addition, a primary bottleneck for AI implementation is the scarcity of standardized, high-quality datasets. Current data on fish protein modifications are often fragmented across isolated studies with varying experimental protocols and species sources. This lack of data uniformity makes it difficult to train robust machine learning models capable of accurately predicting structure-function relationships across diverse species. Furthermore, effective deployment requires expertise in both biotechnology and data science, emphasizing the importance of collaborative efforts between academia, industry, and technology providers. Overall, integrating molecular simulations with AI and big data in fish protein modification represents a promising frontier. These tools provide great insights into protein behavior, enabling precise, efficient, and sustainable modification strategies. By addressing current challenges and fostering interdisciplinary collaboration, the fish protein industry can leverage technological advancements to meet the growing demand for functional, sustainable, and high-quality food products.
-
Fish proteins, rich in essential amino acids and possessing notable functional properties such as gelation, emulsification, and water retention, have immense potential in the food industry. However, these proteins are susceptible to structural degradation during processing, which can limit their applicability. To enhance the functional qualities of fish proteins, a variety of modification techniques have been developed, spanning physical, chemical, biological, and other novel approaches, several of which have proven effective in transforming low-value byproducts into high-value and bioactive isolates suitable for human consumption. These techniques not only enhance the performance of fish proteins in food applications but also promote the sustainable and economical utilization of fish processing byproducts. Despite this significant progress, challenges such as balancing functionality and sensory quality, cost constraints, scalability, and regulatory compliance remain critical. Future research should focus on integrating modification strategies and leveraging emerging technologies, such as molecular simulations, AI and big data analytics, to optimize outcomes. By prioritizing sustainability, safety, and efficiency, the fish protein industry can continue to innovate and meet the growing global demand for health-conscious and functional food ingredients.
-
The authors confirm their contributions to the paper as follows: investigation, writing − original draft: Li H; formal analysis, writing − original draft: Zheng Z; writing − original draft: Wu D; project administration, visualization: Shi L; formal analysis: Li Z; project administration: Cai S; writing − review and editing, methodology, and supervision: Fu C; writing − original draft, supervision: Ren Z. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets generated during and/or analyzed in the current study are available from the corresponding author on reasonable request.
-
This study was funded by the Key Research and Development Project of Hainan Province (Grant Nos ZDYF2022XDNY335, ZDYF2024XDNY204), Natural Science Foundation of Fujian Province (Grant No. 2025J01864, 2024J09042) and Science and Technology Project of Jiangsu Province (Grant No. BZ2022056). We also thank the Biomedical and Health Technology Platform at the National University of Singapore (Suzhou) Research Institute, and the 5th High-Level Entrepreneur-ship and Innovation Team Project of Putian City (Added-value Processing of Predominant Aquatic Products in Putian City).
-
The authors declare that they have no conflict of interest.
-
Authors contributed equally: Haoquan Li, Zihan Zheng, Di Wu
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press on behalf of China Agricultural University, Zhejiang University and Shenyang Agricultural University. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Li H, Zheng Z, Wu D, Shi L, Li Z, et al. 2026. Modification of fish proteins in the food industry: a comprehensive review of technologies, challenges and future trends. Food Innovation and Advances 5(1): 87−101 doi: 10.48130/fia-0026-0003
Modification of fish proteins in the food industry: a comprehensive review of technologies, challenges and future trends
- Received: 02 November 2025
- Revised: 12 January 2026
- Accepted: 12 January 2026
- Published online: 13 March 2026
Abstract: The fish production industry is experiencing rapid growth due to the high nutritional value and rich protein content of fish. However, the functions of fish proteins require improvement to meet industry demands, especially when derived from low-value fish or processing side streams. To enhance sustainability and utilization, modification strategies have been developed, including physical techniques involving ultrasound, high pressure, and microwave without altering chemical composition. Chemical modifications, including pH-shift, phosphorylation, and cross-linking, enable precise manipulation of protein behavior. Biological approaches, such as enzymatic hydrolysis, enzymatic cross-linking, and fermentation, have gained attention due to their ability to produce bioactive peptides and improve protein digestibility. These techniques can alter the molecular structure and interactions of fish proteins, thereby improving their functional attributes and expanding their applications in food processing. Despite these advancements, challenges for fish protein modifications still exist, such as species variability, scalability, and quality control. To address these challenges, the integration of artificial intelligence, molecular simulations, and big data analytics is highlighted, offering data-driven predictive approaches to optimizing modification conditions and accelerating industrial translation. This review comprehensively discusses the modification techniques, challenges, and prospects for fish proteins, emphasizing their potential to drive innovative, high-quality aquatic products with diverse applications.