-
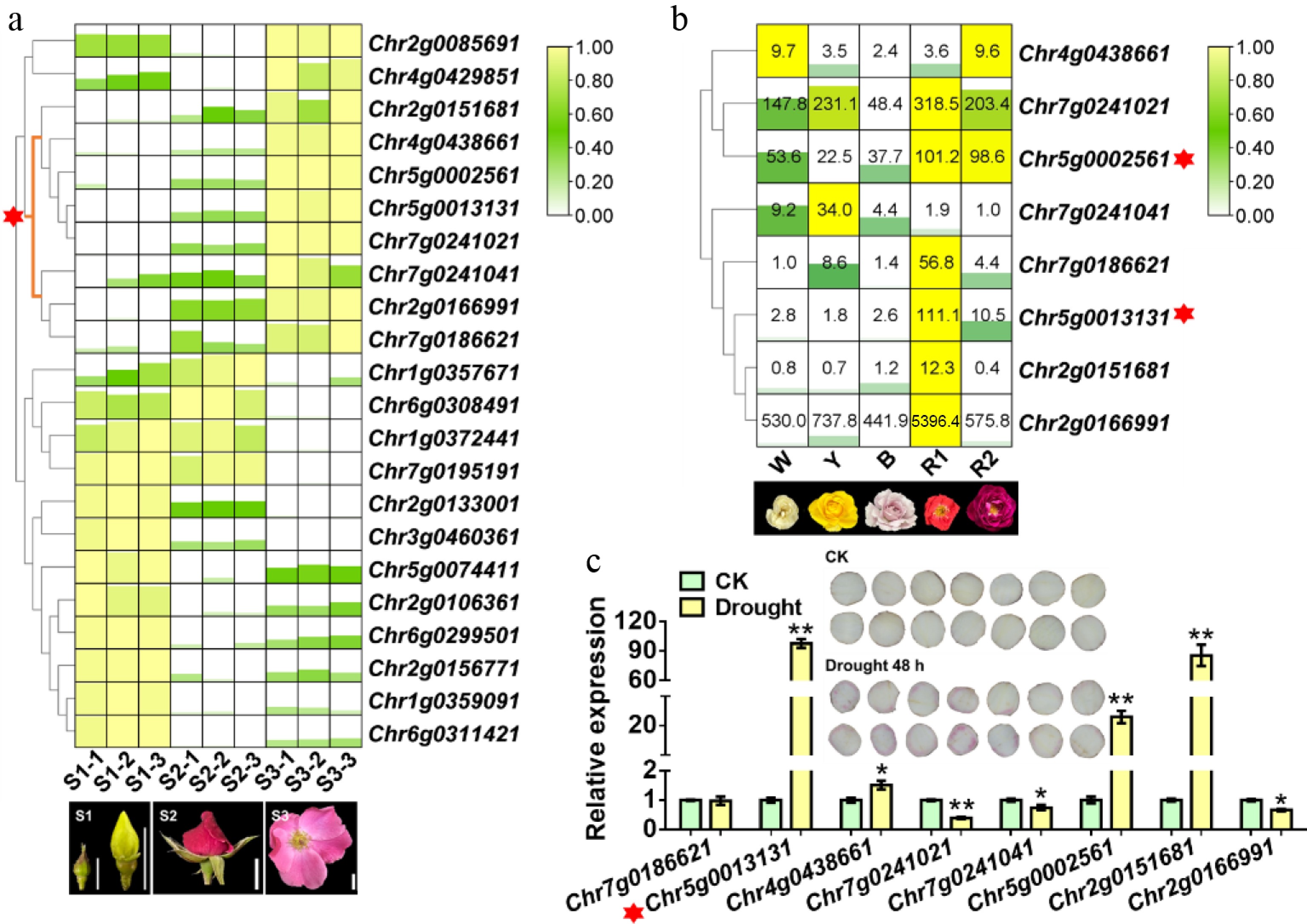
Figure 1.
Identification of a WRKY gene involved in drought-induced anthocyanin accumulation in Rosa chinensis. (a) Heatmap showing differentially expressed WRKY genes from the transcriptome of R. chinensis 'Old Blush' at different flower developmental stages. Representative petal images for each stage are shown. Scale bar: 1 cm. The red asterisk marks the cluster of WRKY genes highly expressed at the S2 and S3 stages, which are critical for anthocyanin accumulation in rose petals. (b) Expression patterns of candidate WRKY genes in rose cultivars with different flower colors. W, white-flowered cultivar (R. hybrida 'Mount Shasta'); Y, yellow-flowered cultivar (R. hybrida 'Yellow Leisure Liness'); B, blue-purple-flowered cultivar (R. hybrida 'Libellula'); R1 and R2, red-flowered cultivars (R. hybrida 'Angela' and R. hybrida 'Yan Li', respectively). Representative petal images are shown. Scale bar, 1 cm. The red asterisk indicates the WRKY genes with relatively high expression in red petals. (c) Relative expression levels of selected WRKY genes in response to drought stress. The phenotype of anthocyanin accumulation in R. hybrida 'Mount Shasta' petal discs with (Drought) or without (CK) 48 hours of drought treatment is shown. The red asterisk marks the stronger response of Chr5g0013131 to drought stress. Data are presented as the mean ± standard deviation (n = 3). Asterisks indicate significant differences (* p < 0.05; ** p < 0.01; Student's t-test).
-
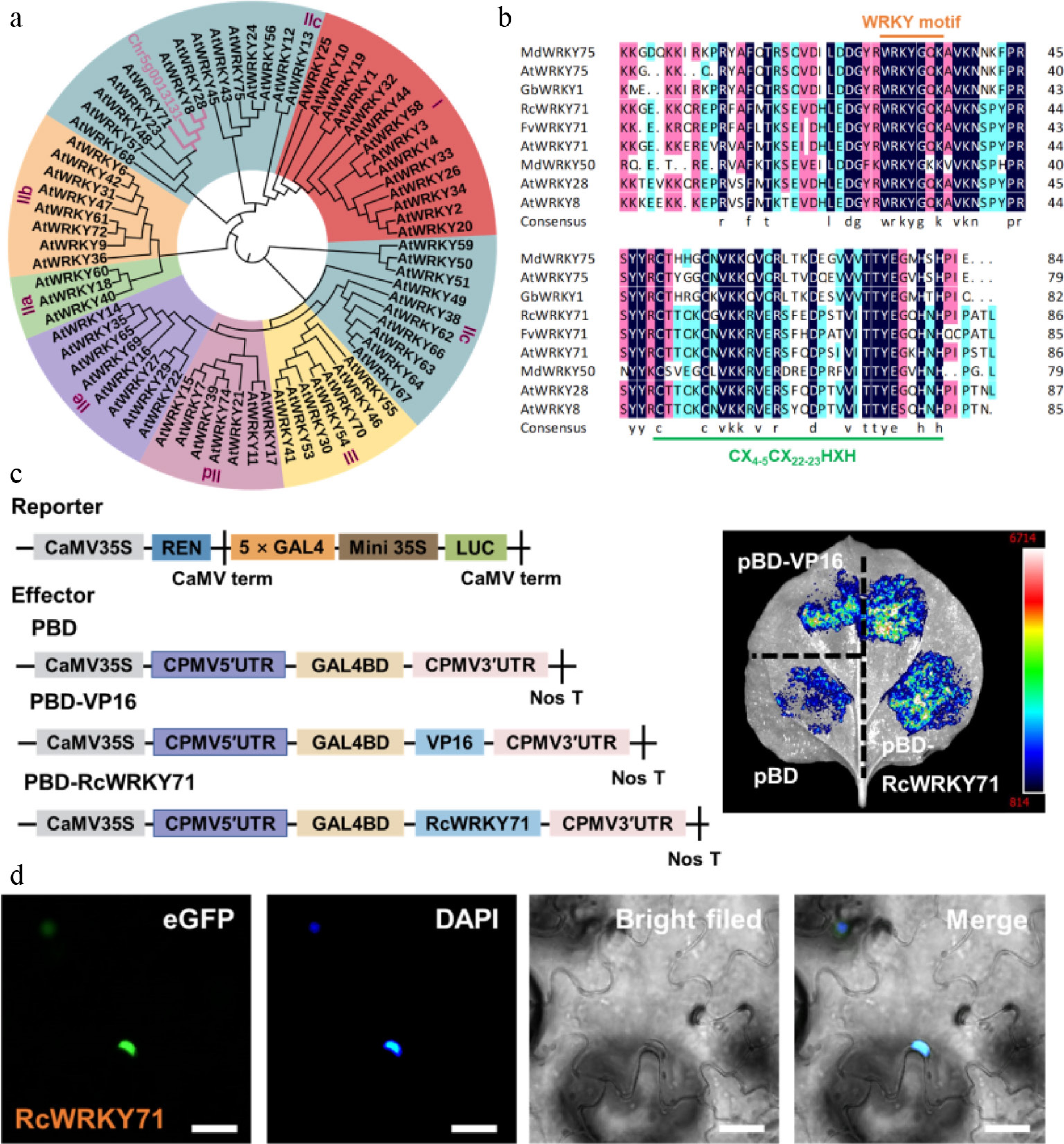
Figure 2.
Phylogenetic analysis, multiple sequence alignment, transcriptional activity assay, and subcellular localization of RcWRKY71. (a) Phylogenetic tree constructed from the full-length amino acid sequences of RcWRKY71 and WRKY family members from Arabidopsis thaliana. (b) Multiple sequence alignment of RcWRKY71 and its homologs from different species. The conserved WRKY domain and zinc finger motif were indicated. (c) Transcriptional activity of RcWRKY71 assessed using the GAL4-based luciferase (LUC) reporter system. The effector and reporter constructs are schematically illustrated. (d) Subcellular localization of RcWRKY71. The RcWRKY71–GFP fusion protein (green) localizes to the nucleus, as shown by co-staining with DAPI (blue). Scale bars, 20 μm.
-
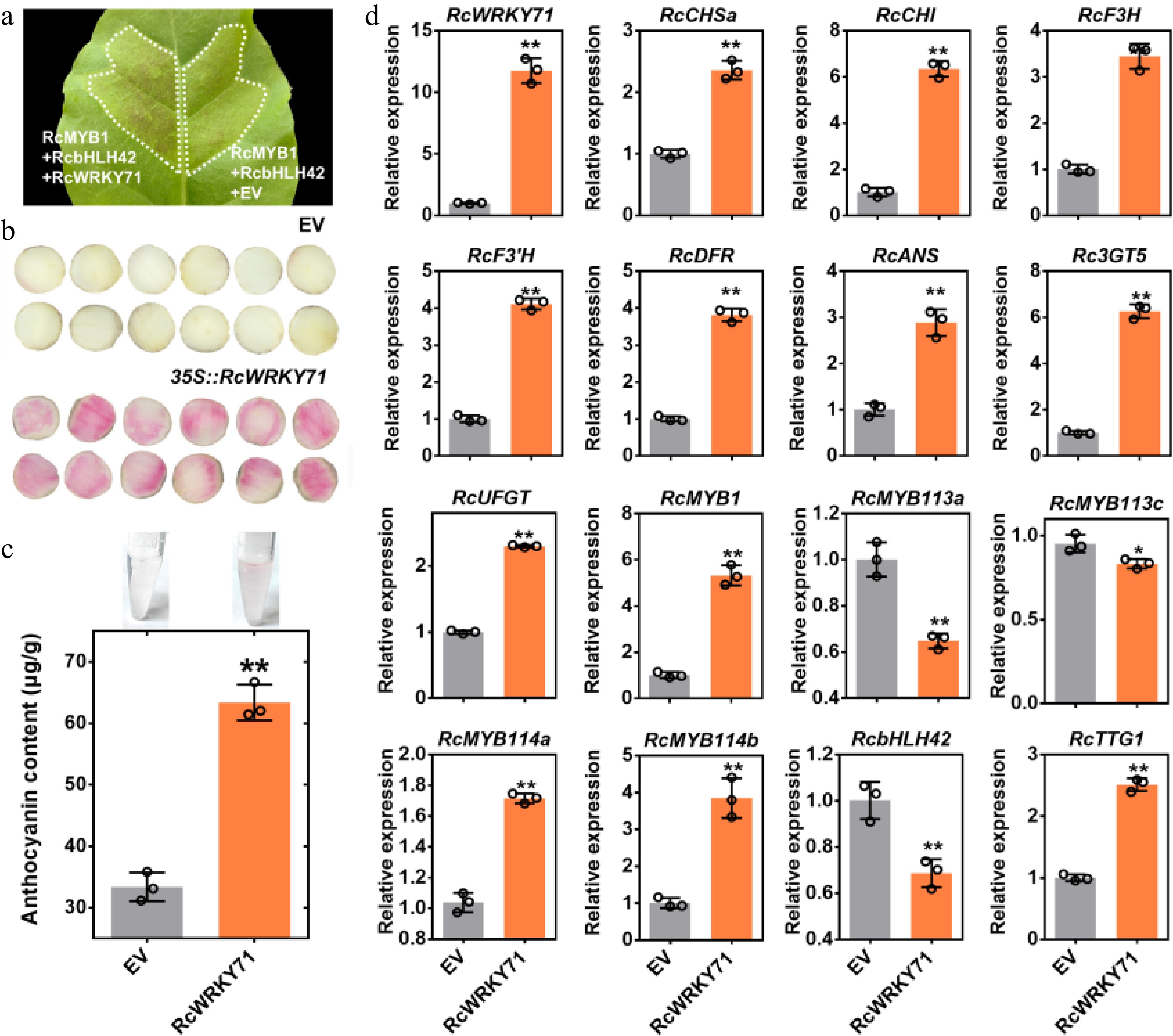
Figure 3.
Phenotypic and molecular analysis of the transient overexpression of RcWRKY71 in tobacco leaves and rose petal discs. (a) Phenotype of tobacco leaves following transient overexpression of RcWRKY71. (b) Phenotype of rose petal discs of the white-flowered rose cultivar 'Mount Shasta' after transient overexpression of RcWRKY71. (c) Anthocyanin content measurement in 'Mount Shasta' petal discs transiently overexpressing RcWRKY71. Data are presented as the mean ± standard deviation (n = 3). Asterisks indicate a significant difference (** p < 0.01; Student's t-test). (d) RT-qPCR analysis of the relative expression levels of RcWRKY71, anthocyanin structural genes, and regulatory genes in 'Mount Shasta' petal discs after transient overexpression of RcWRKY71. Data are presented as the mean ± standard deviation (n = 3). Asterisks indicate a significant difference (* p < 0.05, ** p < 0.01; Student's t-test).
-
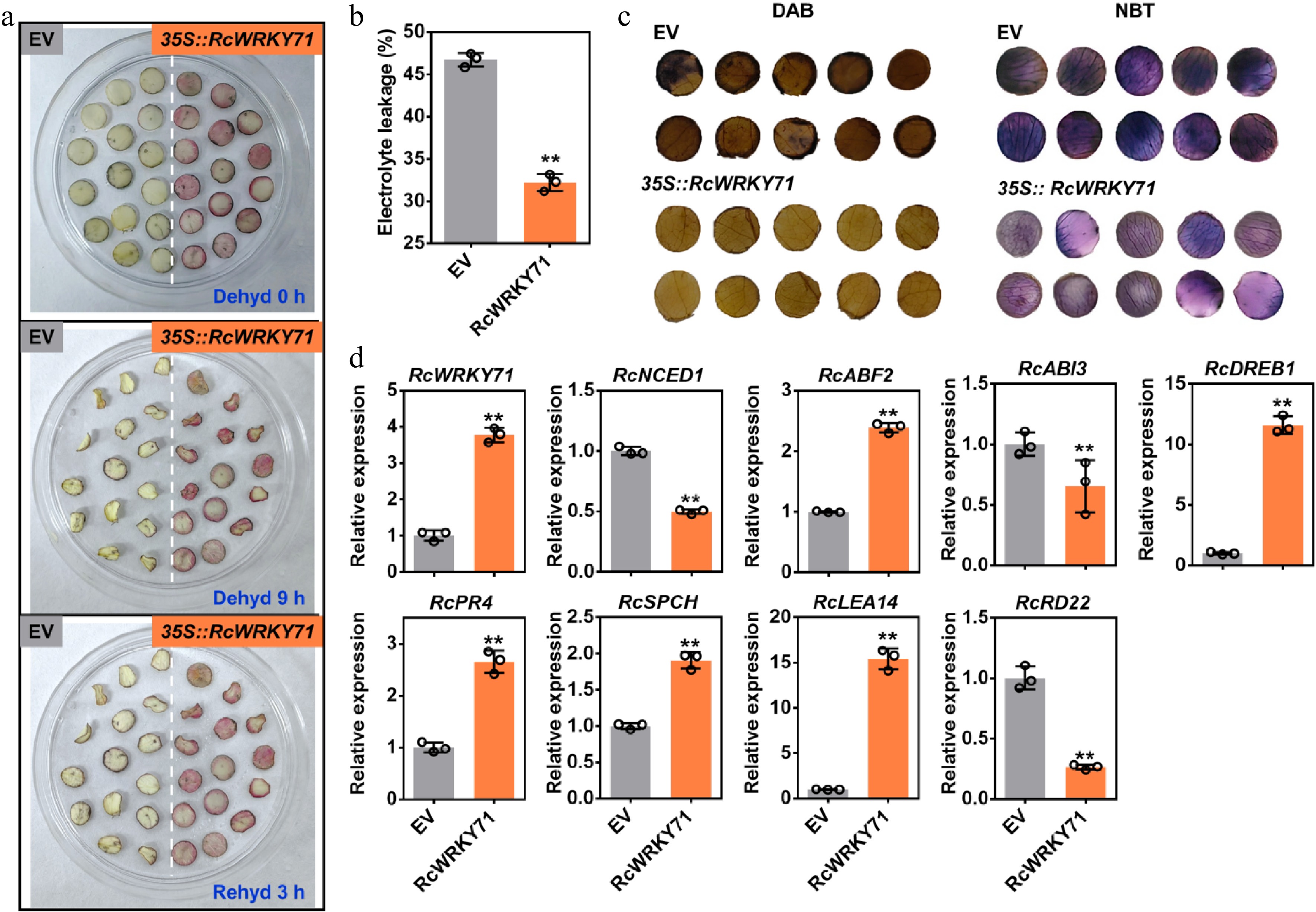
Figure 4.
Physiological and molecular analysis of rose petal discs overexpressing RcWRKY71 under drought stress. (a) Phenotype of petal discs after 5 days of transient overexpression of RcWRKY71 followed by drought treatment. (b) Relative electrolyte leakage measured in petal discs after 5 days of transient overexpression of RcWRKY71 and subsequent drought stress. Data are presented as the mean ± standard deviation (n = 3). Asterisks indicate significant differences (** p < 0.01; Student's t-test). (c) Histochemical detection of hydrogen peroxide (H2O2) and superoxide anion (O2−) using DAB and NBT staining, respectively, in petal discs under the same conditions as in (a). (d) RT-qPCR analysis of the relative expression levels of stress-responsive genes in petal discs after 5 days of transient overexpression of RcWRKY71. Data are presented as the mean ± standard deviation (n = 3). Asterisks indicate significant differences (** p < 0.01; Student's t-test).
-
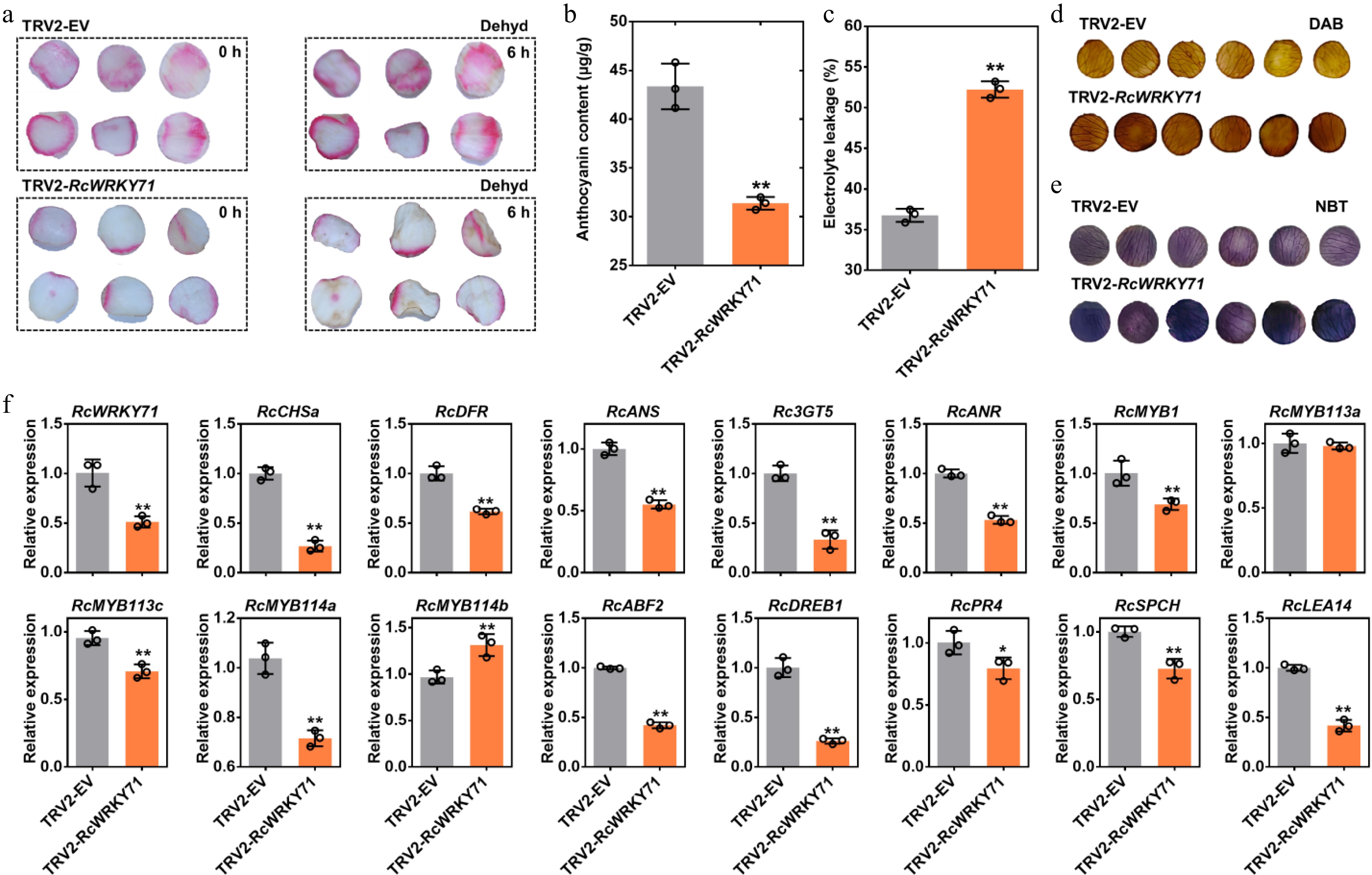
Figure 5.
Phenotypic, physiological, and molecular analysis of RcWRKY71-silenced rose petal discs. (a) Anthocyanin accumulation phenotype and drought stress phenotype in petal discs after RcWRKY71 silencing. (b) Anthocyanin content measurement in RcWRKY71-silenced petal discs. Data are presented as the mean ± standard deviation (n = 3). Asterisks indicate significant differences (** p < 0.01; Student's t-test). (c) Relative electrolyte leakage in RcWRKY71-silenced petal discs under drought stress. Data are presented as the mean ± standard deviation (n = 3). Asterisks indicate significant differences (** p < 0.01; Student's t-test). (d) Detection of hydrogen peroxide using DAB staining in RcWRKY71-silenced petal discs under drought stress. (e) Detection of superoxide anion using NBT staining in RcWRKY71-silenced petal discs under drought stress. (f) RT-qPCR analysis of the relative expression levels of anthocyanin biosynthesis/regulatory genes and stress-responsive genes in RcWRKY71-silenced petal discs under drought stress. Data are presented as the mean ± SD (n = 3). Asterisks indicate significant differences (** p < 0.01; Student's t-test).
-
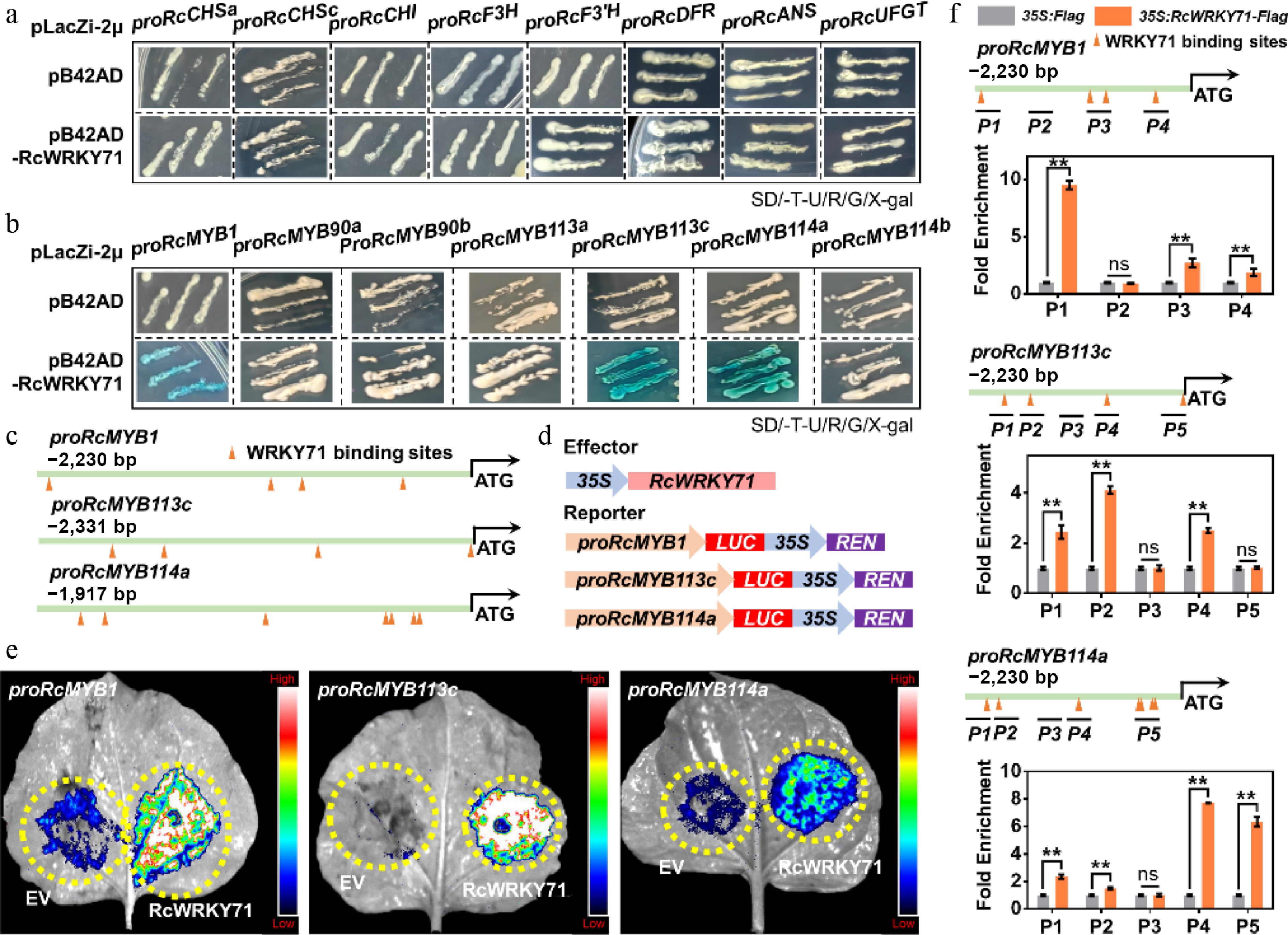
Figure 6.
Transcriptional regulatory mechanism of RcWRKY71 in anthocyanin accumulation in R. chinensis. (a) Yeast one-hybrid assay testing the binding of RcWRKY71 to the promoters of anthocyanin structural genes. (b) Yeast one-hybrid assay examining the binding of RcWRKY71 to the promoters of SG6 subgroup MYB genes that regulate anthocyanin accumulation. (c) Prediction of WRKY71-binding sites in the promoters of RcMYB1, RcMYB113c, and RcMYB114a using JASPAR. (d) Schematics of the dual-luciferase (LUC) reporter constructs. (e) Transactivation activity of RcWRKY71 on the promoters of RcMYB1, RcMYB113c, and RcMYB114a measured by dual-luciferase assays. (f) ChIP-qPCR analysis of RcWRKY71 binding to the promoters of RcMYB1, RcMYB113c, and RcMYB114a. Data are presented as the mean ± SD (n = 3). Asterisks indicate significant differences (** p < 0.01; Student's t-test).
-
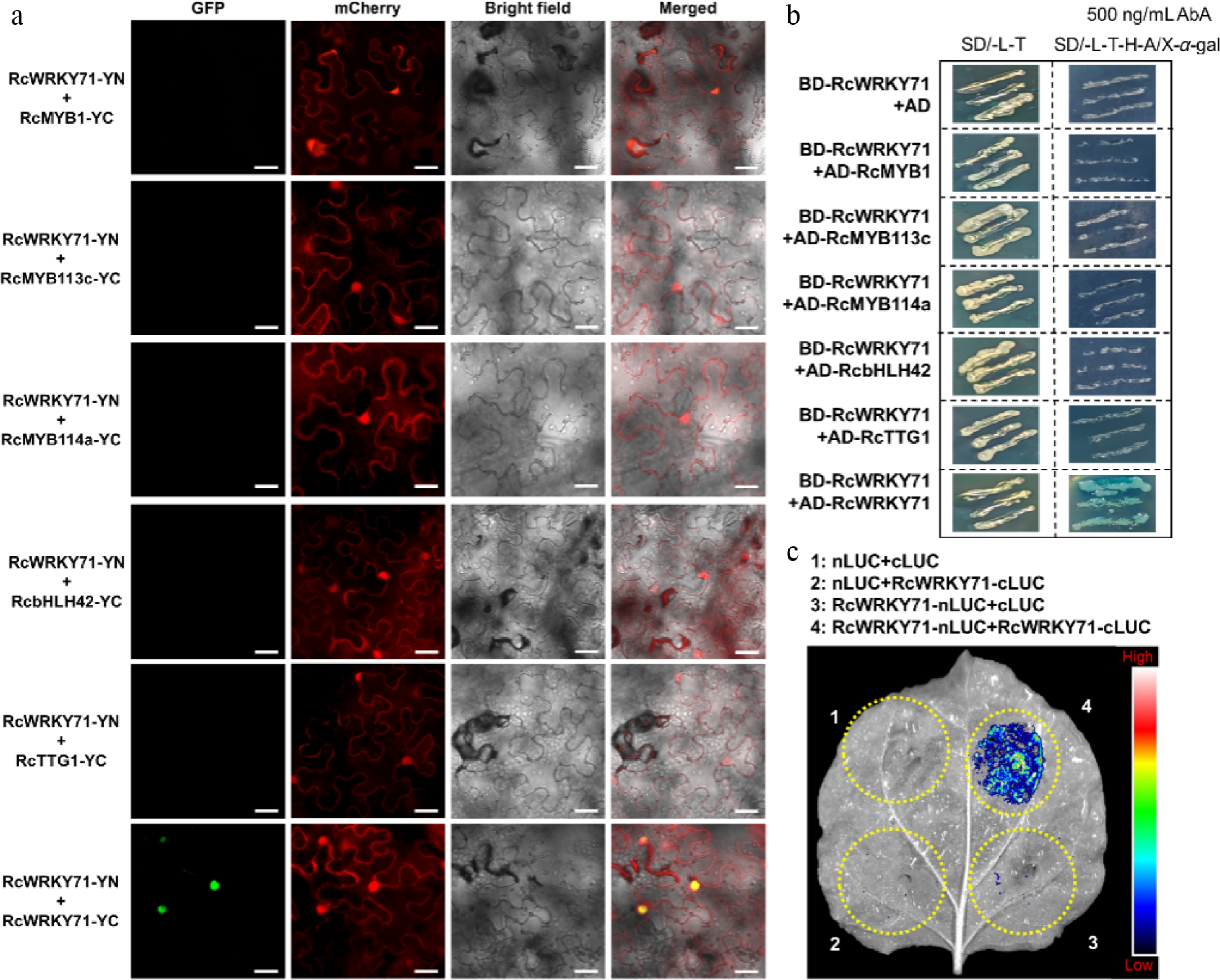
Figure 7.
Post-translational regulatory mechanism of RcWRKY71 in anthocyanin accumulation in R. chinensis. (a) BiFC assay examining the protein interaction between RcWRKY71 and components of the anthocyanin-related MBW complex. Scale bars, 20 μm. (b) Yeast two-hybrid assay testing the protein interaction between RcWRKY71 and MBW complex members. (c) Split-luciferase complementation assay confirming the homomeric interaction of RcWRKY71.
-
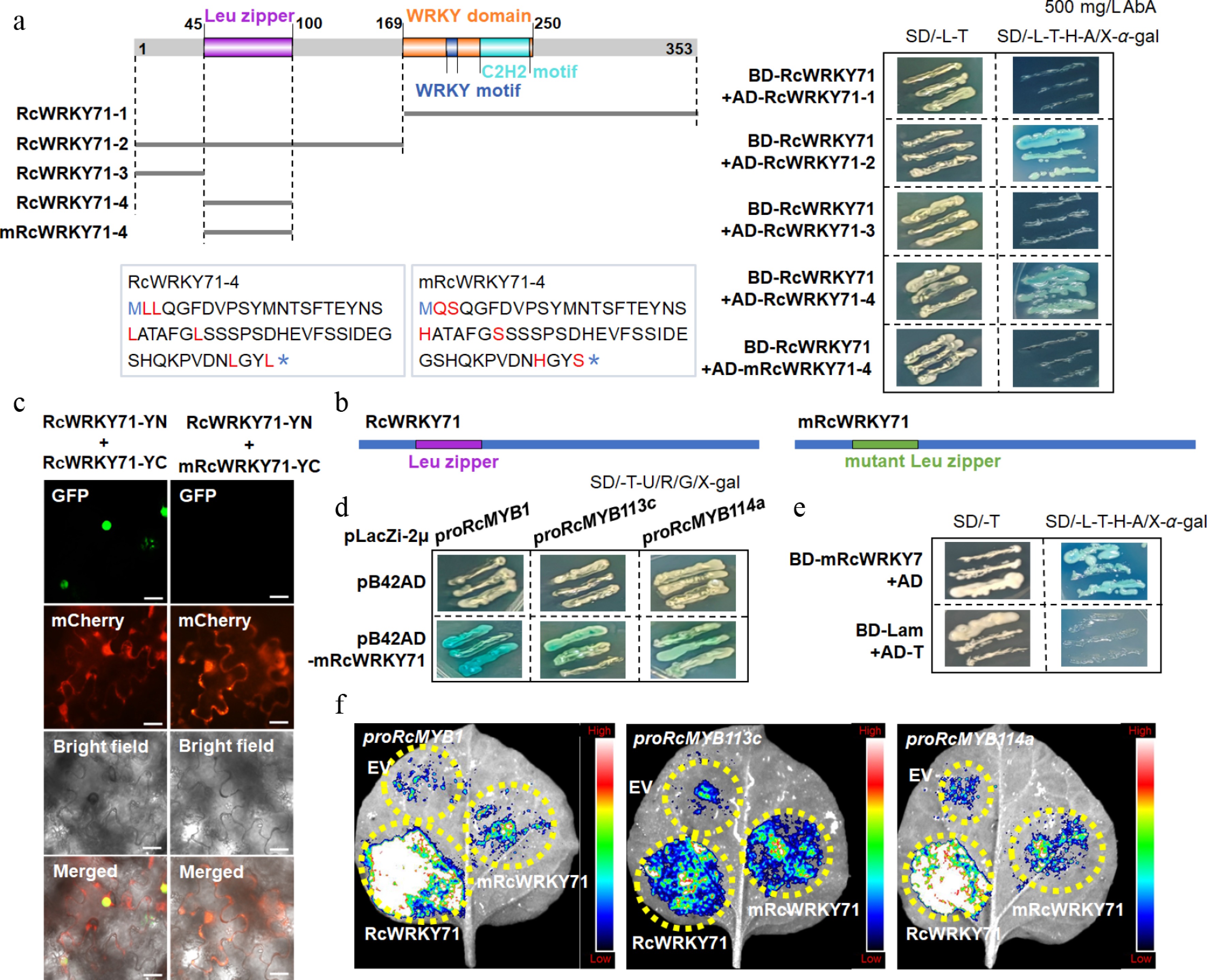
Figure 8.
Effect of Leu-zipper mutation on homodimer formation and function of RcWRKY71. (a) Yeast two-hybrid assay showing the interaction between different truncated versions of RcWRKY71 and the effect of Leu-zipper mutation on protein interaction. (b) Schematic diagram of RcWRKY71 and the mutant mRcWRKY71. (c) BiFC assay confirming that the Leu-zipper mutant mRcWRKY71 fails to interact with wild-type RcWRKY71. Scale bars, 20 μm. (d) Yeast one-hybrid (Y1H) assay demonstrating that the Leu-zipper mutant mRcWRKY71 retains the ability to bind to the promoters of downstream genes. (e) Yeast two-hybrid assay showing that the Leu-zipper mutant mRcWRKY71 maintains its transcriptional activation activity. (f) Dual-luciferase (Luc) assay showing that the transactivation ability of mRcWRKY71 toward downstream gene promoters is significantly attenuated.
Figures
(8)
Tables
(0)