-
Understanding floral biology—particularly the timing of anthesis, pollen maturation, and viability—is indispensable for hybridization, germplasm enhancement, and yield improvement. Despite okra's capacity for self-pollination, artificial cross-pollination can enhance genetic diversity and trait introgression which is particularly important in modern breeding strategies[1]. Flower development and pollen viability are critical determinants of reproductive success in crop plants, particularly in species like okra (Abelmoschus esculentus), where controlled pollination is essential for hybrid breeding. Pollen, the male gametophyte, has evolved to transfer sperm cells to the female gametophyte for fertilization, producing fruits and seeds—the staples of the human diet. In order to transmit genetic information between individuals, pollen must survive in the environment for a duration that varies significantly between species[2]. The ability of a pollen grain to endure in the environment while retaining its capability to germinate and form a tip-growing pollen tube on a receptive stigma or pistil is known as pollen viability[3]. The quality of intact pollen is assessed using a number of techniques: (i) measurement of pollen size; (ii) vital staining for the presence of cytoplasm or enzymatic activity in pollen grains; (iii) analysis of the germinability of pollen in vitro or in vivo; (iv) estimation of the 393 Burešetal: pollen viability and hybridization in Cirsium proline content in pollen, and (v) assessment of the ability of pollen to affect fertilization and seed set[4−6]. Additionally, pollen viability can be used to determine a hybrid's reproductive fitness, or its capacity to donate pollen in natural populations[7−9]. Seed production of crops is usually closely related to pollen viability[10−12]. In crop crosses, the likelihood of pollination failure will rise if low-viability pollen grains are pollinated onto the stigmas of female parents. Consequently, there will be a low seed set and decreased breeding efficiency[13]. Research on pollen viability in hermaphrodite species offers crucial information for comprehending the species' field reproduction, as the observed viability percentage may explain the abortion, or even the reproductive success of a species. Low pollen viability, less than 70%, can lead to issues for the species, including infertility[14]. For plant breeding, conservation, and cultivation, the pollen-viability rate is crucial because, in allogamous species like the one being studied, gene flow through pollen increases the likelihood of the formation of various allele combinations and, consequently, genetic variability[15].
Plants flower 35 to 65 DAS in okra
-
The 3rd to 19th nodes, and typically the 5th to 7th nodes, are where hermaphrodite golden yellow blooms occur[16]. Flowers open at dawn, and remain open until evening. Anthers are placed in concentric stacked circles around a stamen column, which surrounds the style in the center of the flower (5−6 anthers per circle). Stigmas are found far away from the anthers and are essentially separated. At anthesis, the sticky pollen grains (about 100 per anther) are not always in touch with the stigmas. The timing is crucial; the desired female parent's buds are chosen when they are ripe, but not yet open (usually distinguished by a small amount of color and size), then emasculation is carried out. Okra has a tendency for protogyny, meaning that the stigma becomes receptive just before the anthers release pollen. The anthers usually dehisce at or just before anthesis. Research indicates that pollen viability peaks during flower opening and that pollen release in okra is mostly synchronized with anthesis[17]. The stigmatic surface is sensitive throughout the day of anthesis and can be receptive as early as a few hours before flower opening[18]. Therefore, the best time for pollination is in the early morning.
-
Seven okra varieties namely S-21, SS, M6L3, M6L4, M6L5, M6L6, and M6L7 were grown in separate pots in seven distinct lines during March to June 2022 on the experimental field located on the western side of the Agronomy and Agricultural Extension Department of Rajshahi University. Seeds of mutant lines (M6L3, M6L4, M6L5, M6L6, and M6L7) were collected from the Plant Molecular Biotechnology Laboratory, Department of Biochemistry and Molecular Biology, University of Rajshahi, and the two local varieties (S-21 and SS) were obtained from a local seed vendor named 'Sumi beej vandar' situated in Vodra, Rajshahi City, Rajshahi, Bangladesh. The removal of androecium (stamens) from a bisexual flower is known as emasculation (Fig. 1b). Emasculation was performed in the afternoon from 5 pm to 6 pm by removing the perianth and anthers. Emasculated flowers were immediately covered with polythene bags using paper clips (James clips). The next morning, pollen grains were collected from a healthy (disease-free) male parent and dusted onto the receptive stigmas of emasculated flowers between 7am and 9am. After hand pollination, the stigma of the flower was covered with new polythene bags and tagged with hard papers (laminated) to identify the pollen donor and recipient plant. The tags included the name of the donor plant, the name of the recipient plant, the date of pollination, the date of emasculation, and the name of the breeder. The polythene bags were removed two to three days after artificial pollination to allow the fruits to develop properly. When the capsules reached maturity, they were picked, dried, and the hybrid seeds were stored in a cold place (−20 °C refrigerator) until the F1 generation trial was conducted. The materials required for emasculating the female flowers include forceps, hand gloves, needles, polythene bags, and tags. For pollination, healthy (disease-free) male flowers, a zero-sized brush, polythene bags, and paper clips are needed. Ninety-five percent ethanol can be used to disinfect hands before emasculation and pollination. To tag the flowers, we folded a piece of hard paper to tie around it, indicating that pollination has been performed. Different colored paper can be used to indicate various donor parents or different pollination dates. It should be noted that only disease-free healthy plants were intended to be chosen for the hybridization program.

Figure 1.
(a) Various stages of okra flower development (1−7), at stage 5 emasculation was carried out. (b) An emasculated okra flower before bagging.
A safranin solution was used to test pollen viability[19]. To prepare a 100 ml stock solution, 1 g of safranin was dissolved in 40 ml of 95% alcohol, and then 60 ml of distilled water was added. Twenty milliliters of safranin solution, 40 ml of glycerol, and 20 ml of distilled water were combined to prepare the staining solution. By using a light microscope (XSZ-107 China), counts were taken 1 h after the pollen grains were placed on the staining solution. To calculate the percentage of pollen viability, the number of viable pollen grains in each cultivar was counted under a light microscope using Dino-lite digital microscope (Fig. 2).
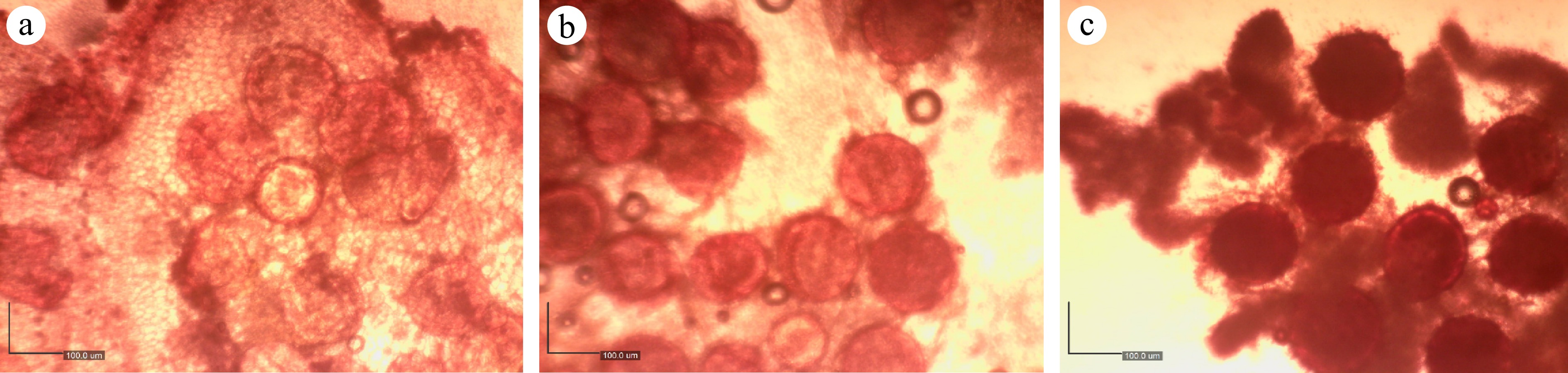
Figure 2.
Stages of pollen development, (a) stage 1, (b) stage 2, and (c) stage 3 (mature pollen under microscope).
Pollen viability was estimated with the following formula:
$ \mathrm{Pollen\;viability}\text{%} =\frac{\text{Number of viable pollen grains}}{\text{Total number of pollen grains} }\times100 $ -
Flower developmental stages were categorized into seven phases, from small closed buds to fully open flowers (Fig. 1a). Time-lapse observations were made from dawn (as flower opening begins) to dusk (when flowers senesce). Distinct and sequential stages of flower development were clearly observed in okra, providing a practical basis for identifying the optimal stage for emasculation. Seven recognizable developmental stages (stages 1–7) were documented, ranging from immature floral buds to fully opened flowers at anthesis. Early stages (stages 1–3) were characterized by small, tightly closed buds with undeveloped reproductive organs. During intermediate stages (stages 4–5), floral buds increased in size, and internal reproductive structures became distinguishable, although the anthers had not yet dehisced. Stage 5 was identified as the most suitable stage for emasculation, as the buds were mature but unopened, and the anthers were fully developed but had not yet released pollen. At this stage, the stigma was already visible and approaching receptivity, minimizing the risk of self-pollination. In later stages (stages 6–7), flowers showed partial to complete opening, with exposed anthers and visible pollen, making emasculation ineffective due to a high probability of self-fertilization.
Figure 1b illustrates a successfully emasculated flower, where all anthers were carefully removed while preserving the integrity of the central style and stigma. Figure 2 represents various stages of pollen development. Pollen diameter was estimated to be 164 to 170 µm for viable pollen grains at stage 3 (mature pollen) using a Dino-lite digital microscope.
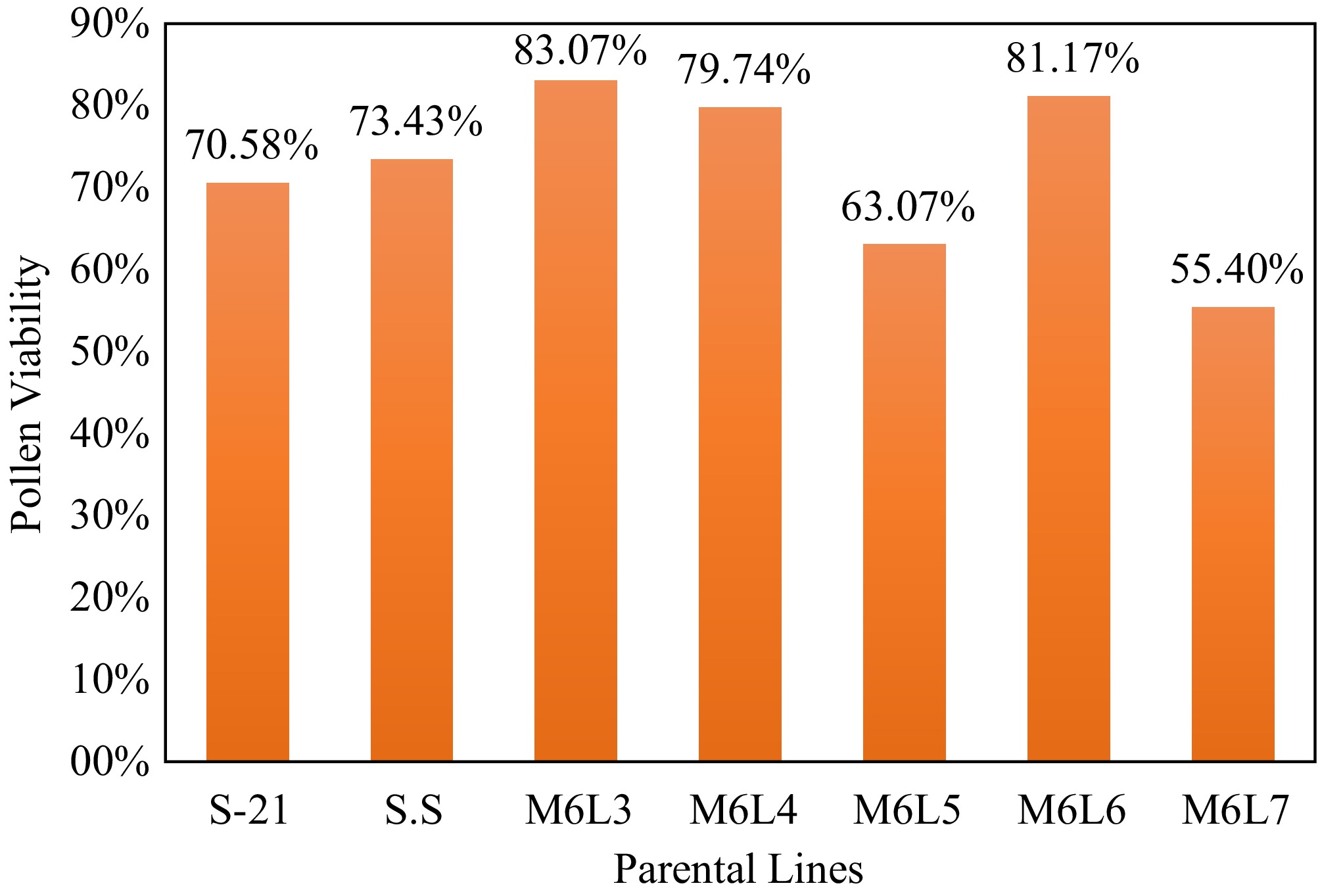
Pollen viability (%) of seven okra lines were estimated, and the results are presented in the graph (Fig. 3). Pollen viability varied markedly among the evaluated okra parental lines, indicating clear differences in male reproductive potential. Viability percentages ranged from 55.40% to 83.07% across the seven parental lines. Among the genotypes, M6L3 exhibited the highest pollen viability (83.07%), followed closely by M6L6 (81.17%) and M6L4 (79.74%), suggesting superior pollen fitness in these lines. Moderate levels of pollen viability were recorded in SS (73.43%) and S-21 (70.58%). In contrast, M6L5 showed comparatively lower viability (63.07%), while M6L7 recorded the lowest pollen viability (55.40%) among all tested lines.
Overall, the observed variation in pollen viability highlights the importance of parental line selection in okra breeding with higher pollen viability percentage. Lines with higher pollen viability, notably M6L3, M6L6, and M6L4, may contribute more effectively to hybrid development and seed production.
-
Okra flowers exhibit protogyny, where the stigma becomes receptive just before anther dehiscence, necessitating careful timing of emasculation and pollination[20]. Most plants require effective pollination to produce fruit and seeds, and knowledge of pollen biology, particularly pollen viability and pollen tube growth is necessary for any logical strategies to increase productivity[21,22]. This experiment also discusses the importance of determining pollen viability in okra plants selected for hybridization purposes. Fitness is significantly influenced by the quantity and quality of pollen that a flower produces. Pollen viability is sometimes linked with pollen quality[23]. Viable pollen grains are microscopic structures enclosed by a complex, multilayered wall that safeguards the male gametophyte. This wall consists of an outer exine, which provides rigidity and surface ornamentation, and an inner intine, which plays a crucial role in pollen germination and in overall pollen viability[24]. In the present study, viable pollen grains were clearly identified under microscopic observation by the presence of intact exine and intine layers with a spherical structure. The visibility and structural integrity of these wall components are considered key indicators of pollen viability in all species. The size of mature pollen also determines the pollen viability of any species. Okra (Abelmoschus esculentus) pollen grains are relatively large, with diameters typically ranging from approximately 125 to 162 µm, which is consistent with observations of spherical pollen morphology in this species[25]. We also observed viable pollen grains with a diameter 164−170 µm in the current experiment. Pollen viability directly influences fertilization efficiency and seed set[26]. High viability in lines M6L3 and M6L6 indicates strong reproductive potential, making them suitable candidates for hybrid breeding pipelines. Conversely, low viability in M6L5 and M6L7 suggests reduced breeding effectiveness, potentially due to environmental stress or pathogen effects on floral development. High pollen viability has been associated with increased seed set and breeding success in other crops, including Brassica and Solanaceae species[27]. Therefore, genotypes with high viability percentages are valuable for breeding programs aiming to enhance genetic recombination.
A deeper understanding of okra's floral biology and effective hybridization techniques is crucial to achieving successful breeding in okra. In this experiment, the developmental stages of the okra flower are shown for a successful hybridization program. For the successful emasculation of flower buds, selecting flowers at the appropriate stage is necessary. A well-developed flower that has not opened yet should be selected for emasculation. To create okra hybrids, breeders typically employ emasculation of the female parent bud followed by controlled pollination. Flowers opened at dawn and remained open for less than a day[28]. We also observed this phenomenon during the hybridization program in the current experiment. Flower development and reproductive phenology are foundational to successful hybridization programs. The protogynous nature of okra flowers requires breeders to timely emasculate flowers to prevent self-pollination and maximize cross success[29]. The identification of the 5th floral developmental stage as optimal for emasculation aligns with the findings in related species, where stigma receptivity precedes pollen release[30].
-
Pollen viability of seven okra cultivars was determined in this experiment, and this attribute is essential to know the fruit set percentage in the hybridization procedure. Okra breeding has advanced significantly, and this achievement has been largely attributed to the combined application of hybridization techniques and floral biology insights. Breeders have produced okra hybrids that have increased yields and farmers' income by perfecting the art and science of controlled pollination, which involves synchronizing emasculation and pollination to the rhythm of the plant. The future era of okra breeding will essentially be driven by the ongoing fusion of cutting-edge research with traditional expertise (floral biology, hybridization techniques).
Flowers of Abelmoschus esculentus last less than 1 d during their flowering period. The dynamic process of flower opening was divided into seven stages in this experiment to determine the perfect stage at which emasculation should be done. Emasculation was done at the 5th flowering stage. For successful hybridization (artificial hand pollination), it is crucial to know the appropriate stage at which emasculation should be done. Successful hybridization depends on preventing self-pollination, while keeping the female reproductive organs fully functional. Okra flowers are bisexual and protandrous, and anther dehiscence occurs very close to flower opening (anthesis). If emasculation is done too late, pollen may already have been released onto the stigma, resulting in self-fertilization, which defeats the purpose of hybridization.
-
The authors confirmed their contributions to the study as follows: The corresponding author Prof. Hossain MA conceptualized and designed the study on flower development and pollen viability in okra. Field and laboratory experiments were conducted by Piuli ZT, including floral stage assessment and pollen viability analysis. Data collection, statistical analysis, and interpretation were performed by Piuli ZT. Both authors reviewed the results and approved the final version of the manuscript.
-
The datasets generated and analyzed during the current study on flower development and pollen viability in okra are included within the article.
-
The authors express the deepest sense of gratitude to the Agro-technology Laboratory, Department of Agronomy & Agricultural Extension for providing the opportunity of an okra field trial, and the Plant Molecular Biotechnology Laboratory, Department of Biochemistry & Molecular Biology, University of Rajshahi, for providing all the necessary equipment for the experimental work. We, specially thank Saurov MEH for the technical assistance in calibrating the microscope with Dino light 2.0.
-
The authors declare that they have no conflict of interest.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Piuli ZT, Hossain MA. 2026. Assessment of flower development and pollen viability to support okra breeding efforts. Technology in Horticulture 6: e012 doi: 10.48130/tihort-0026-0005
Assessment of flower development and pollen viability to support okra breeding efforts
- Received: 05 January 2026
- Revised: 03 February 2026
- Accepted: 25 February 2026
- Published online: 27 April 2026
Abstract: Pollen viability and longevity are critical determinants of sexual reproduction in plants and underpin key processes in plant physiology, ecology, breeding, and crop-based industries. Effective pollen performance is essential not only for natural plant propagation, but also for breeding programs that involve pollen collection and controlled hybridization to ensure high fruit and seed set. In okra (Abelmoschus esculentus L.), recent advances in breeding strategies increasingly rely on detailed knowledge of floral biology and pollen behavior to enhance yield potential, disease resistance, and cultivar adaptability. The present study evaluated pollen viability percentage to identify suitable parental lines in okra for successful hybridization; flower developmental stages were also studied to determine the appropriate stage of emasculation in okra. Pollen viability (%) was assessed in seven okra cultivars, comprising two local varieties—Selection-21(S-21) and Sobuj-Sathi(SS)—and five mutant lines (M6L3, M6L4, M6L5, M6L6, and M6L7). Notably, among the five mutant lines, M6L3 (83.07%), and M6L6 (81.17%) exhibited significantly higher pollen viability than the local cultivars. High-viability mutant lines identified in this study represent valuable genetic resources for okra breeding programs. Overall, this study highlights the importance of synchronizing floral developmental stages and pollen viability assessment to enhance breeding efficiency and genetic improvement in okra.
-
Key words:
- Abelmoschus esculentus L. /
- Emasculation /
- Floral biology /
- Hybridization /
- Mutants /
- Pollen longevity