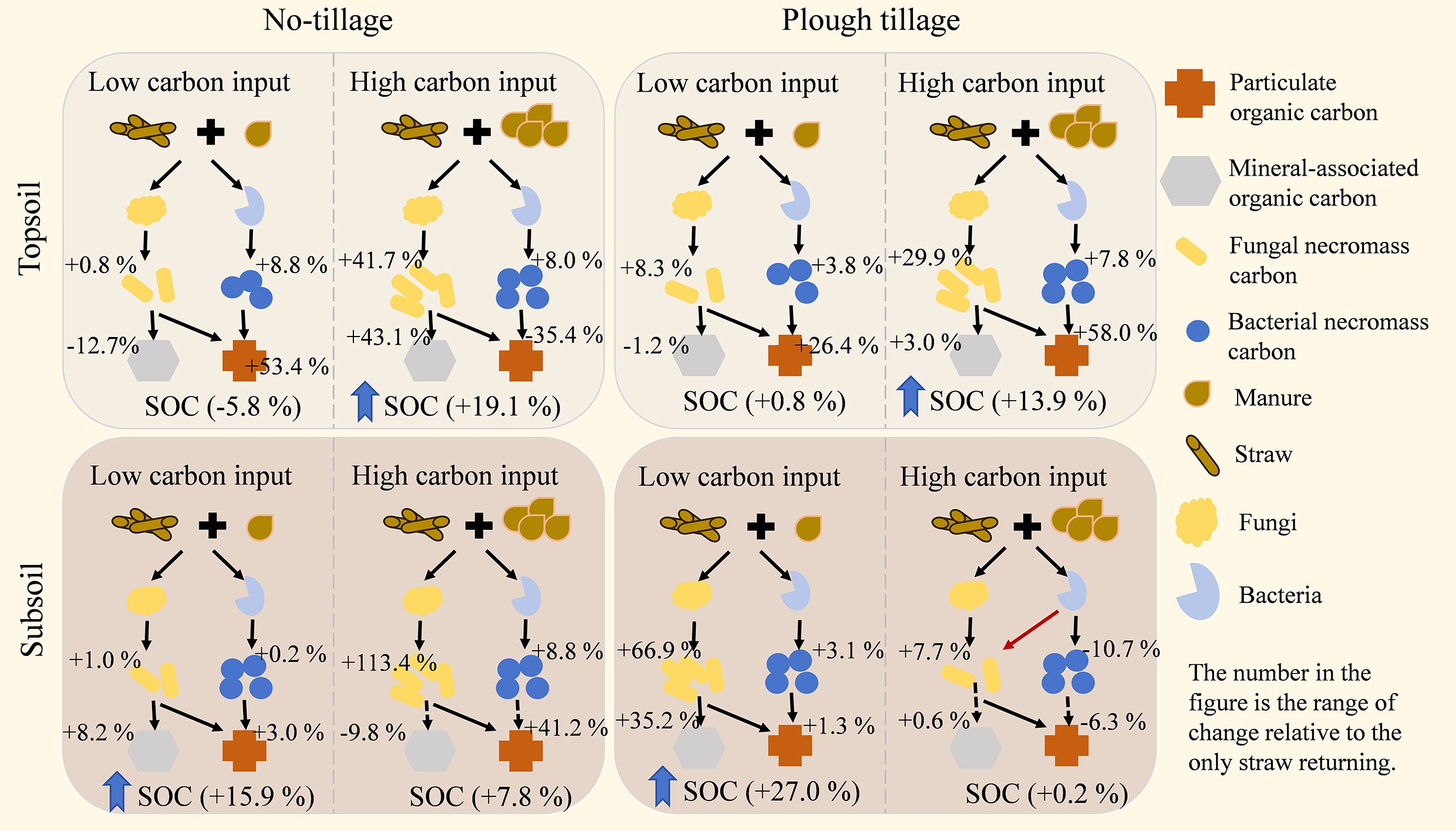
-
The global community is currently grappling with the dual challenges of climate change and escalating food demand. Enhancing soil carbon sequestration through agricultural management has emerged as a key priority within the international scientific agenda[1,2]. Soil organic carbon (SOC) constitutes a fundamental component of terrestrial ecosystems, playing an indispensable role in maintaining soil fertility, supporting microbial life, and influencing the global carbon cycle[3,4].
The primary avenue for carbon input into agricultural soils is through the addition of exogenous organic materials, notably crop residues and animal manures[5]. The introduction of exogenous organic matter leads to its transformation into stable mineral-associated organic carbon (MAOC) and unstable particulate organic carbon (POC) through complex biological and abiotic processes before being stored in the soil[6]. These inputs undergo complex biotic and abiotic transformations, ultimately contributing to the formation of relatively stable MAOC and more labile POC, which are then sequestered in the soil matrix[7], whereas small-molecule soluble organic carbon in livestock and poultry manure is more likely to combine with the mineral surface to form MAOC[8]. More and more researchers have aimed to combine the advantages of straw and manure, and have begun to apply livestock and poultry manure as a field management practice after returning straw[9,10]. Studies have shown that the SOC content achieved with straw and livestock manure application is 12.6% to 26.9% higher than that achieved with returning straw returning[11], and the mixed application of manure and straw significantly increases the content of POC and MAOC, and increases the proportion of MAOC[12]. Microorganisms are the main decomposers of straw and manure after their entry into the soil[13]. Their catabolism and anabolism convert the input carbon into their own biomass and metabolites[14], as well as cell debris and extracellular polymers and other derivatives produced after microbial death, which collectively form microbial necromass carbon (MNC)[15,16]. Microbial biomass carbon (MBC) and MNC, as secondary products from the microbial transformation of input organic matter, have a strong correlation with SOC components[8,17]. MBC is a direct manifestation of vigorous microbial metabolism, and its content is negatively correlated with POC, which is usually associated with rapid turnover. Some studies have also suggested that the utilization preference of microorganisms for MNC exceeds that for POC[18,19], and MNC is an important precursor for the formation of MAOC[20]. Thus, MBC and MNC are intricately involved in the dynamic equilibrium between carbon input pools and both the fast-cycling and stable carbon fractions. However, this source–sink relationship is markedly influenced by the tillage practices applied in farmland systems.
The tillage method determines the initial distribution of exogenous carbon in the soil, Under no-tillage conditions, carbon is mainly concentrated in the topsoil (0–20 cm), whereas plough tillage results in a more even distribution of the input carbon in the subsoil (20–40 cm). The carbon enrichment in specific soil layers promotes soil microorganisms to secrete more carbon-decomposing enzymes, enhances microbial metabolic activity, leads to changes in MBC and MNC[21], and increases the stratification rate of SOC[22]. However, there are significant differences in the soil aeration and nutrient content between the topsoil and subsoil under natural factors and human management. Changes in the soil structure and aeration caused by tillage will first increase the activity of aerobic microorganisms in the soil and improve the availability of nutrients[23]. However, tillage also aggravates the mineralization process of soil carbon and weakens the microbial availability of soil carbon[24]. Therefore, the strategy of combining tillage with exogenous carbon supplementation can not only maintain microbial life activities but also enhance the sequestration capacity of farmland soil carbon. Microbial activity and community structure have significant effects on the dynamics of organic carbon components[25]. However, current research has mainly focused on the composition of surface microbial communities or the independent role of MNC, resulting in a limited understanding of the interactions among these factors in the dynamics and stability mechanism of SOC components in different soil layers. A systematic understanding of the layered differences in microbial carbon sequestration mechanisms under the combined effects of tillage and carbon input is essential for further understanding and improving SOC sequestration.
Therefore, on the basis of the the multi-site experimental platform of organic fertilizer application in Jilin Province, we used farmland soil under straw combined with organic fertilizer as the research material to study the community composition, diversity, MBC, MNC and enzyme activity related to the carbon cycle. The effects of carbon input and tillage on organic carbon sequestration in the topsoil and subsoil were evaluated by combining a partial least squares structural equation model (PLS-PM) with SOC and its fractions. Through this study, we aimed to clarify (1) the overall and layer-specific differences imposed by tillage and carbon inputs on SOC and its components; (2) the effects of tillage and carbon input on the microbial community and its activity, and the layer-specific differences in microbial-derived carbon; and (3) the differences in organic carbon components and organic carbon accumulation caused by changes in the microorganisms and microbial-derived carbon in different soil layers. We speculate that the content of SOC and its components in different soil layers varies with carbon input as a result of the different effects of carbon inputs and tillage on the accumulation of microorganisms and MNC in different soil layers. This systematic analysis broadens the understanding of microbial mediation in SOC sequestration and provides targeted management strategies for farmland carbon sequestration.
-
In 2023, a field experiment platform was established in four representative agricultural counties (cities) in Jilin Province, China. The experiment sites include Gongzuling City, Nongan County, Shuangliao City and Shulan City (Fig. 1), all of which belong to the temperate monsoon continental climate zone. The four seasons are distinct, and rainfall and heat are in the same period. The main planting mode is continuous cropping of maize. Before initiating the experiment, consistent field management practices had been implemented for two years in each experimental plot to ensure that the initial physical and chemical properties of the soil in the experiment plot were consistent. The information of the experiment plots is described in detail in Table 1. The same treatments were arranged in each experimental plot, comprising the tillage factors no-tillage (NT) and plough tillage (PT) with a disturbance depth of 30 cm. At the same time, a split-plot design was established with the following treatments: maize straw only (S), maize straw + low exogenous carbon at 600 kg C ha−1 yr−1 (SLC), maize straw + medium exogenous carbon at 1,200 kg C ha−1 yr−1 (SMC), and maize straw + high exogenous carbon as 2,400 kg C ha−1 yr−1 (SHC). The organic fertilizer used as the exogenous carbon source was uniform particles made of livestock and poultry manure, and the nutrient content was 423.0 g kg−1 C, 38.3 g kg−1 N, 3.1 g kg−1 P2O5, and 8.5 g kg−1 K2O. In each experimental year, the amount of chemical fertilizer input was consistent, at 156 kg ha−1 N, 75 kg ha−1 P2O5, and 72 kg ha−1 K2O. Each experimental plot was six ridges every 12 m, with a ridge spacing of 0.65 m. Each treatment was repeated three times.
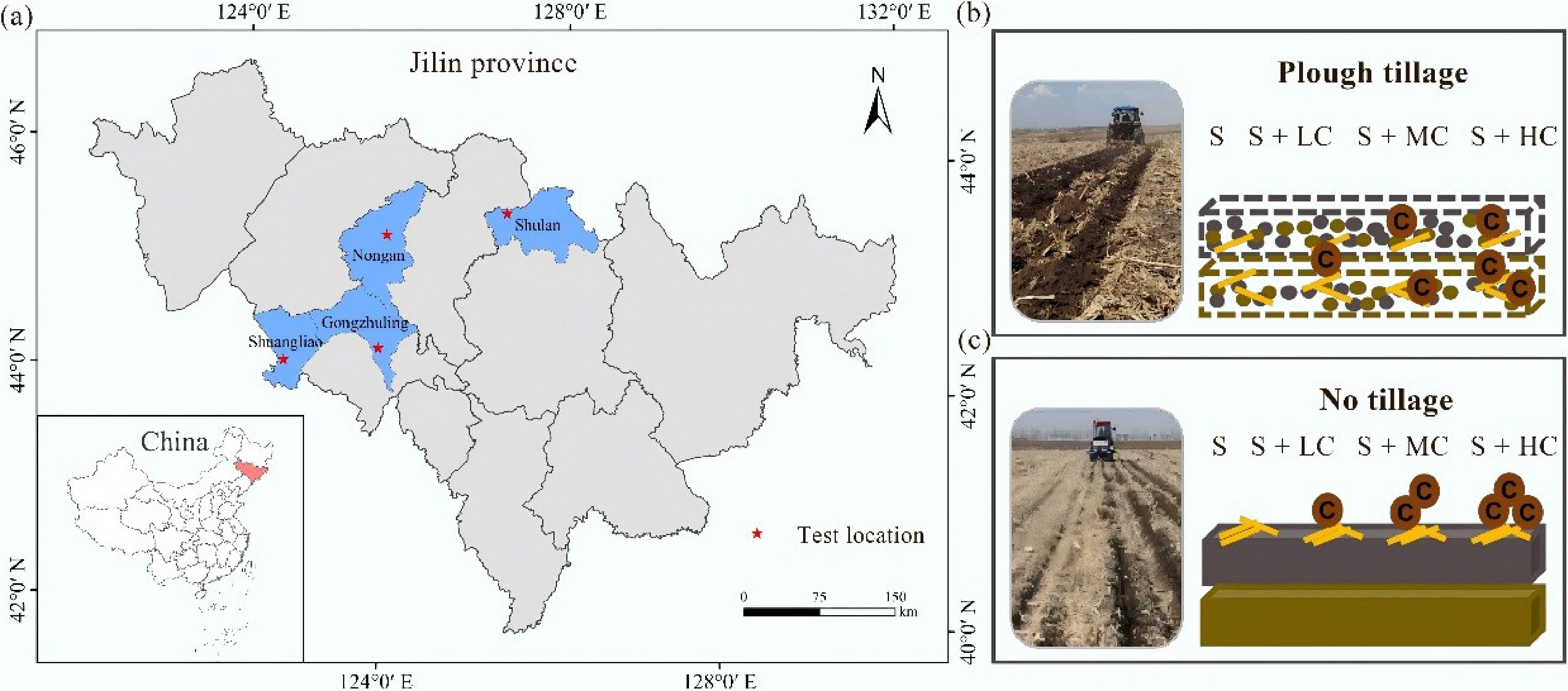
Figure 1.
(a) The experimental area's location. Chart of carbon inputs in different soil layers under (b) plough tillage and (c) no tillage.
Table 1. Field experiment site description and properties in the initial 0–40 cm
Soil layer Site Soil bulk density
(g cm−3)pH
(1:2.5)Soil organic carbon
(g kg−1)Total nitrogen
(g kg−1)Total phosphorus
(g kg−1)Total potassium
(g kg−1)Sand
(%)Silt
(%)Clay
(%)Longitude and latitude Soil type 0–20 cm Gongzhuling 1.3 5.9 9.0 0.59 0.64 10.2 35.4 40.9 23.7 43°58' N, 124°76' E Loamy clay Nongan 1.4 8.1 15.7 0.82 0.91 12.8 34.3 41.1 24.6 44°05' N, 124°51' E Clay loam Shuangliao 1.5 8.3 10.7 0.73 0.97 10.7 34.3 42.2 23.5 43°64' N, 123°58' E Sandy loam Shulan 1.4 5.4 11.1 0.83 1.04 12.5 37.3 44.2 18.5 44°51' N, 126°71' E Sandy clay loam 20–40 cm Gongzhuling 1.6 6.4 8.6 0.58 0.71 10.9 34.4 41.7 23.8 Nongan 1.5 8.1 16.3 0.69 0.63 10.4 33.3 41.7 25.1 Shuangliao 1.5 8.4 10.7 0.57 0.57 11.8 35.5 39.7 24.7 Shulan 1.6 5.8 4.9 0.86 0.70 12.9 37.1 40.9 21.9 Soil sampling and analysis
-
Soil samples of topsoil (0–20 cm) and subsoil (20–40 cm) were collected after teh maize harvest in October 2024. Visible rocks and plant residues were removed during the collection process. One part of the collected samples was passed through an 8-mm sieve and stored, and the other part was stored in a refrigerator at −80 °C in a 50-mL sterile centrifuge tube. During the collection process, the soil was collected with a steel ring knife (volume: 100 cm3) for determination of the soil's bulk density (BD).
The soil samples were ground in the laboratory, and the following measurements were performed. SOC and total nitrogen (TN) were measured using an elemental analyzer, the total phosphorus (TP) in the soil was determined by the NaOH–soil co-melting method and an ultraviolet–visible spectrophotometer, and the total potassium (TK) was determined by flame photometry. The pH value of the soil was measured using a pH meter at a water to soil ratio of 1:2.5. Soil electrical conductivity (EC) values were measured using an EC meter. Soil texture was analyzed via the sodium hexametaphosphate shock separation method. Soil enzyme activities, including those of α-glucosidase (AG), β-glucosidase (BG), cellobiohydrolase (CBH), and xylanase (XY), were assayed using a Solarbio activity assay kit and quantified by a microplate reader (BioTek Synergy LX, Agilent, USA).
Soil MBC and MNC
-
The soil moisture content was adjusted to about 0.5 g g−1 (mass moisture content) and preincubated in a dark environment at 25 °C for one week. Two samples of 20 g of fresh soil (equivalent to the weight of dry soil) were placed into beakers. One beaker was placed in a vacuum dryer for chloroform fumigation, and the other was used as a nonfumigated control. Next 50 mL of an ethanol-free chloroform and 1 mol L−1 of a NaOH solution were added to the fumigation dryer. In addition, 10 small glass beads were added to prevent boiling. The dryer was sealed with Vaseline and vacuumed until the chloroform boiled for 2 min. Then it was placed in a dark environment for 24 h. The soil samples from the control group and the fumigation group were placed in separate triangular flasks, then 100 mL of a 0.5 mol L−1 K2SO4 solution was added and shaken at 200 rpm for 30 min at 25 °C on a shaker. The filtrate was transferred to a plastic bottle after filtration. The MBC value was calculated from the difference in the total extractable carbon and nitrogen between the fumigated soil and the nonfumigated soil, and was divided by a correction factor of 0.45 for MBC. The microbial entropy was calculated as
$ \rm Microbial\;entropy = MBC/SOC $ (1) As a biomarker of microbial necromass, soil amino sugars were quantified according to established protocols. Specifically, 0.5 g of the air-dried soil samples was accurately weighed and hydrolyzed with 6 mol L−1 HCl at 105 °C for 8 h, maintaining constant temperature conditions. After cooling to room temperature, 100 μL of inositol was added to the hydrolysate as an internal standard for vortex mixing and filtration. The clarified solution was concentrated by rotary evaporation (65 °C), neutralized with KOH (pH 6.6–6.8), and then centrifuged (4,000 rpm for 10 min) for phase separation. The colloidal part was dissolved in methanol and centrifuged repeatedly to remove salt. The purified extract was transferred to a derivatization container for nitrogen-assisted solvent evaporation (45 °C), redissolved in ultrapure water, and freeze-dried. The derivatization process included the gradual addition of 300 μL of the reaction reagent and incubation at 80 °C for 30 min, followed by acetylation with 1 mL of acetic anhydride for 20 min at the same temperature. The post-derivatization treatment included phase separation with 1 mL of dichloromethane and 1 M hydrochloric acid. The organic layer was washed with deionized water, dried under nitrogen purging (45 °C), and then redissolved in 300 μL of n-hexane/ethyl acetate (1:1 v/v) for gas chromatography analysis. Chromatographic separation enables the quantification of glucosamine (GluN), mannose amine (ManN), galactosamine (GalN), and muramic acid (MurA). Fungal necromass carbon (FNC, g kg−1) and bacterial necromass carbon (BNC, g kg−1) were calculated according to the following formulas:
$\rm FNC = \left(\dfrac{GluN}{179.17}-2\times \dfrac{MurA}{251.23}\right)\times 179.17 \times 9 $ (2) $ \rm BNC = MurA \times 45 $ (3) These formulas include the molecular weights of GluN (179.17 g mol−1) and MurA (251.23 g mol−1), where the coefficients 9 and 45 are applied for carbon conversion of fungal and bacterial necromass, respectively[26].
Microbial community composition
-
DNA was extracted and amplified via the cetyltrimethylammonium bromide (CTAB) method. The washed DNA sample was diluted to a concentration of 50 ng μL−1, and then the purity and concentration of the DNA were confirmed by 1% agarose gel electrophoresis. Universal primers (338F, 5'-CAAGCTGCAGGACGAGACTC-3'; 806 R, 5'-TTACTTGCCTGTCCGACAG-3') were used to amplify the bacterial 16S rRNA gene region (about 420 bp), and the target was the V3–V4 hypervariable region. For the fungal genes, ITS1 primers (5'-TGGAGCTGTTGGTGTCGCTG-3') and ITS2 primers (5'-AGCTGCGTGTCATCGCGTG-3') were used to amplify the internal transcribed spacer 1 (ITS1) region. The bacterial 16S rRNA and fungal ITS gene sequences were analyzed using the high-throughput sequencing platform of Illumina Miseq (Biomarker Technologies, Rohnert Park, CA, USA). QIIME2 Softer was used for β diversity analysis to evaluate the similarity of species diversity among different samples.
Statistical analysis
-
Two-way analysis of variance (ANOVA) (least significant difference [LSD], Duncan's test, p < 0.05) was performed using SPSS 24.0 software to analyze the differences in organic carbon fractions, the soil's chemical and physical properties, microbial necromass, and enzyme activity under different treatments. Pearson's correlation was used to test the relationship between organic carbon and microbial necromass, using R 4.3.2. In addition, the random forest prediction model and the least path square structural equation model were constructed using the R packages randomForest and plspm, respectively, to evaluate the effects of MNC and MBC on the organic carbon fractions. In the random forest prediction model, the importance of each predictive variable is quantified by the increase in the percentage of the mean square error (MSE). A higher MSE% value indicates greater importance. The R2 index evaluates the goodness of fit of the model. The closer the value is to 1, the higher the prediction's accuracy is. The structural equation model uses goodness of fit (GOF) to evaluate the fitting quality of the model. Multiple linear regression was used to summarize the influence of all factors on SOC, and the optimal model was selected according to the Akaike information criterion (AIC) and the Bayesian information criterion (BIC). Graphics were generated using Origin 2024 software.
-
Tillage and carbon input had a significant effect on SOC (Fig. 2). Overall, compared with returning straw alone, increasing the C input rates could increase the SOC content, with an average increase of 7.7% in no-tillage SOC and an average increase of 10.1% in plough tillage SOC. However, the changes in SOC showed different trends between the topsoil and subsoil (Supplementary Fig. S1). Specifically, SOC showed an increasing trend with an increase in the C input rates in the topsoil. The SOC content of the SHC treatment under no-tillage and plough tillage increased by 19.1% and 13.9% on average compared with the S treatment. In the subsoil, the SOC content under the plough tillage treatment decreased with an increase in the C input rates. Compared with the S treatment, SOC content under the SLC treatment increased by 27.0%, whereas the SOC content under the SHC treatment increased by only 0.2%.
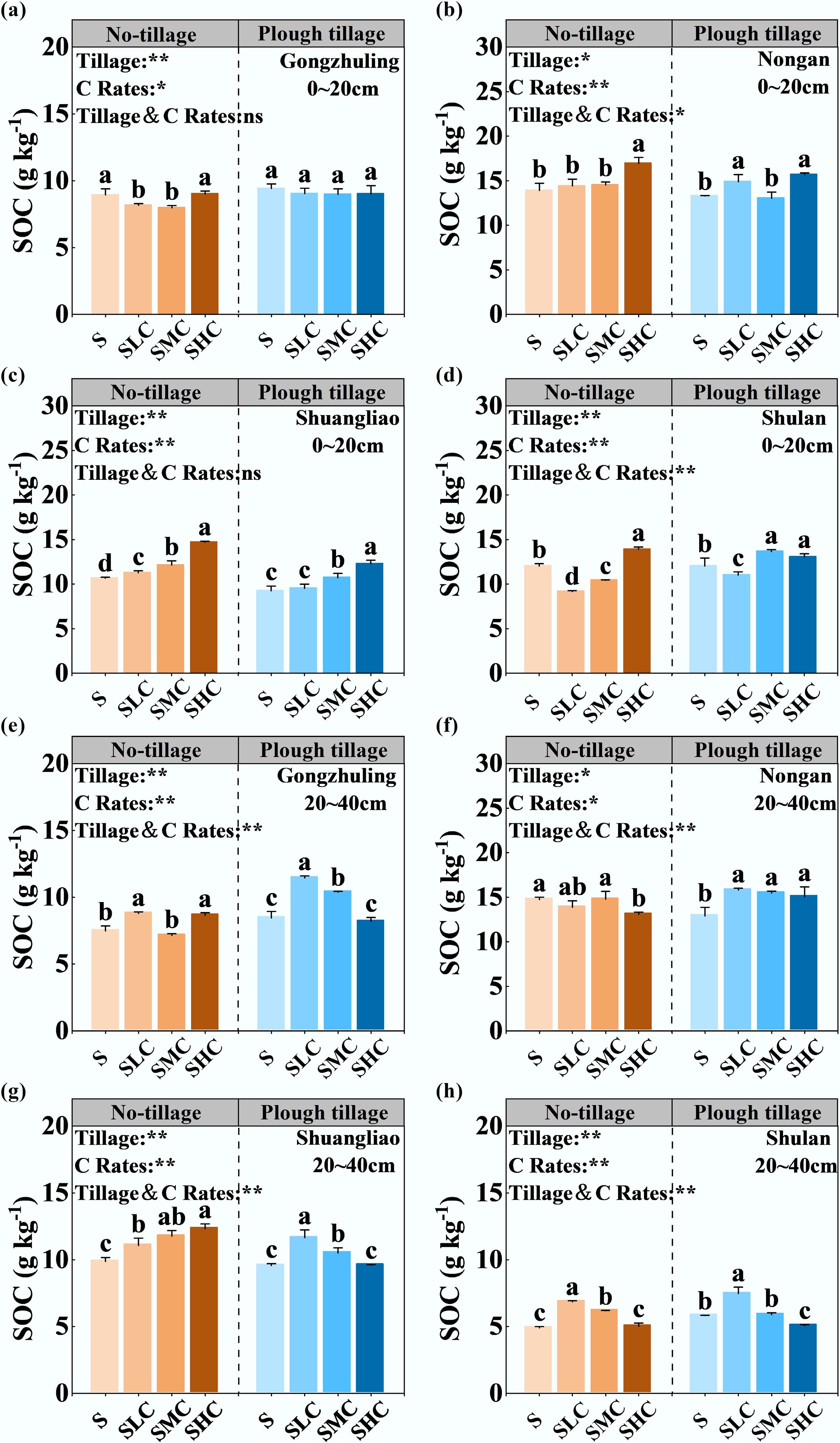
Figure 2.
SOC content as affected by tillage and carbon input. SOC content of topsoil in (a) Gongzhuling, (b) Nongan, (c) Shuangliao, and (d) Shulan. SOC content of subsoil in (e) Gongzhuling, (f) Nongan, (g) Shuangliao, and (h) Shulan. Bars (mean ± standard deviation [SD], n = 3). Bars with different lowercase letters are significantly different among different treatments (p < 0.05). The main and the interactive effects of tillage (Tillage) and carbon input rate (C Rates) are shown: **, p < 0.01; *, p < 0.05; ns, not significant at p > 0.05.
Increasing the C input rates increased the MAOC content in the topsoil, which increased by 7.5% under no-tillage and 3.9% under plough tillage (Fig. 3). Under the no-tillage treatment, SHC treatment significantly increased MAOC compared with the S treatment, with an average increase of 43.1%. Under plough tillage, the POC content increased by 15.9% compared with the S treatment after increasing the C input rate. In the subsoil, compared with the S treatment, the MAOC content under no-tillage decreased by 5.4%, whereas the MAOC content under plough tillage increased by 16.6%. However, we found that with the increase in the C input rate under plough tillage, MAOC content decreased to a level similar to or lower than that of the S treatment. Except for Nongan, the POC content of the SHC treatment in the other three experimental sites was significantly lower than that of the S treatment, with an average decrease of 17.0%.
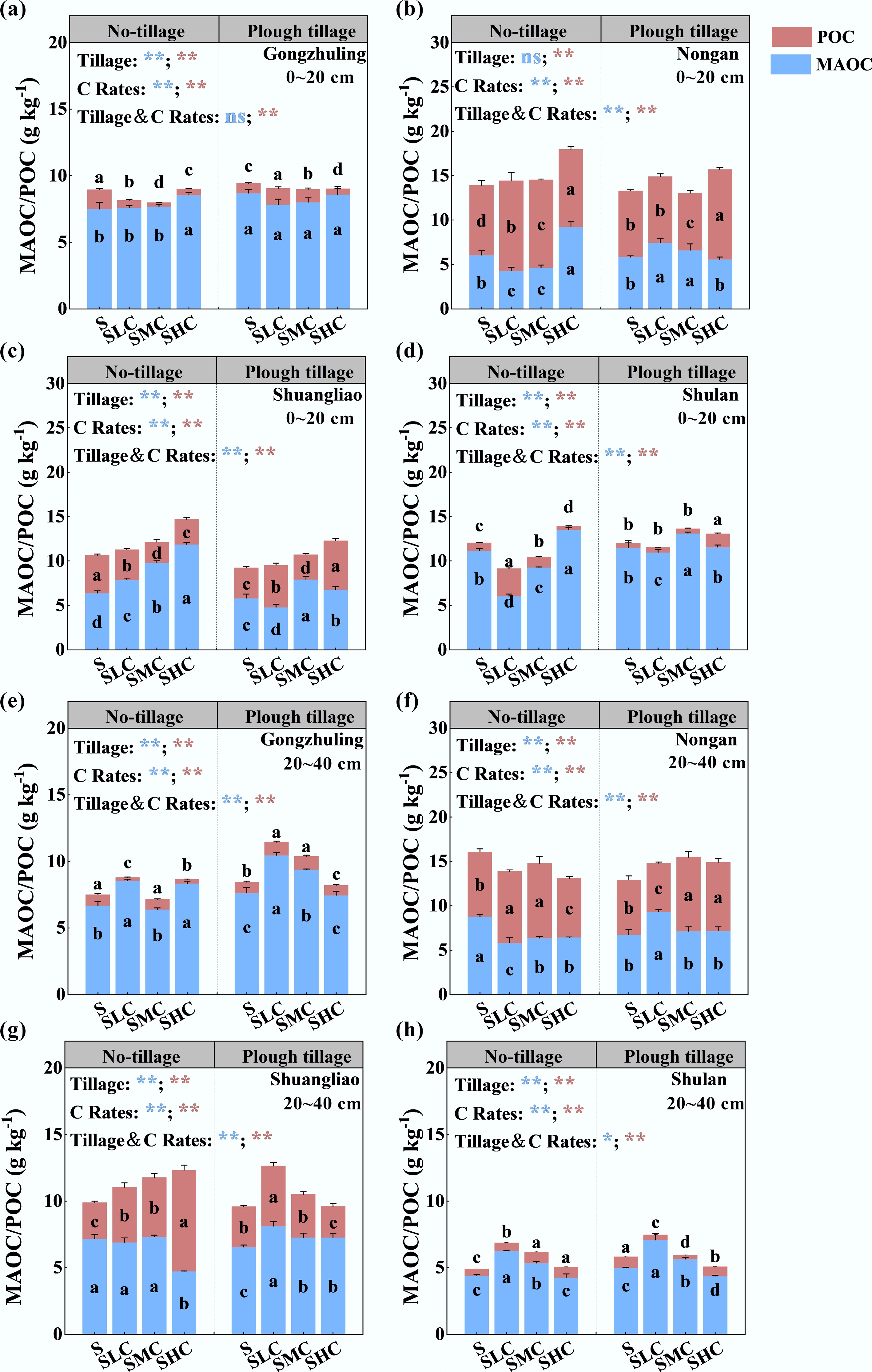
Figure 3.
Effects of different tillage and carbon inputs on the distribution of POC and MAOC in the soil. The content of POC and MAOC in the topsoil of (a) Gongzhuling, (b) Nongan, (c) Shuangliao, and (d) Shulan. The content of POC and MAOC in the subsoil of (e) Gongzhuling, (f) Nongan, (g) Shuangliao, and (h) Shulan. The lowercase letters in the blue and red columns represent the differences between treatments (p < 0.05, n = 3), and the line segments on the column represent the standard deviations of POC and MAOC. The main and interactive effects of tillage (Tillage) and the carbon input rate (C Rates) are shown separately for MAOC (blue) and POC (red): **, p < 0.01; *, p < 0.05; ns, not significant at p > 0.05.
Changes in microbe-derived carbon in topsoil and subsoil
-
Tillage and C input rates and their interactions had significant effects on the MBC content (Fig. 4). Overall, the MBC trends in the topsoil and subsoil were similar, and increasing the C input rates significantly increased the soil MBC content. Compared with the S treatment, the MBC content in the topsoil and subsoil increased by 35.0% and 30.7%, respectively, after the application of exogenous C. However, tillage produced different characteristics in different soil layers. In the topsoil, the MBC content under no-tillage and plough tillage increased by 36.9% and 33.0%, respectively, whereas in the subsoil, the MBC content under no-tillage and plough tillage increased by 25.6% and 35.9%, respectively.
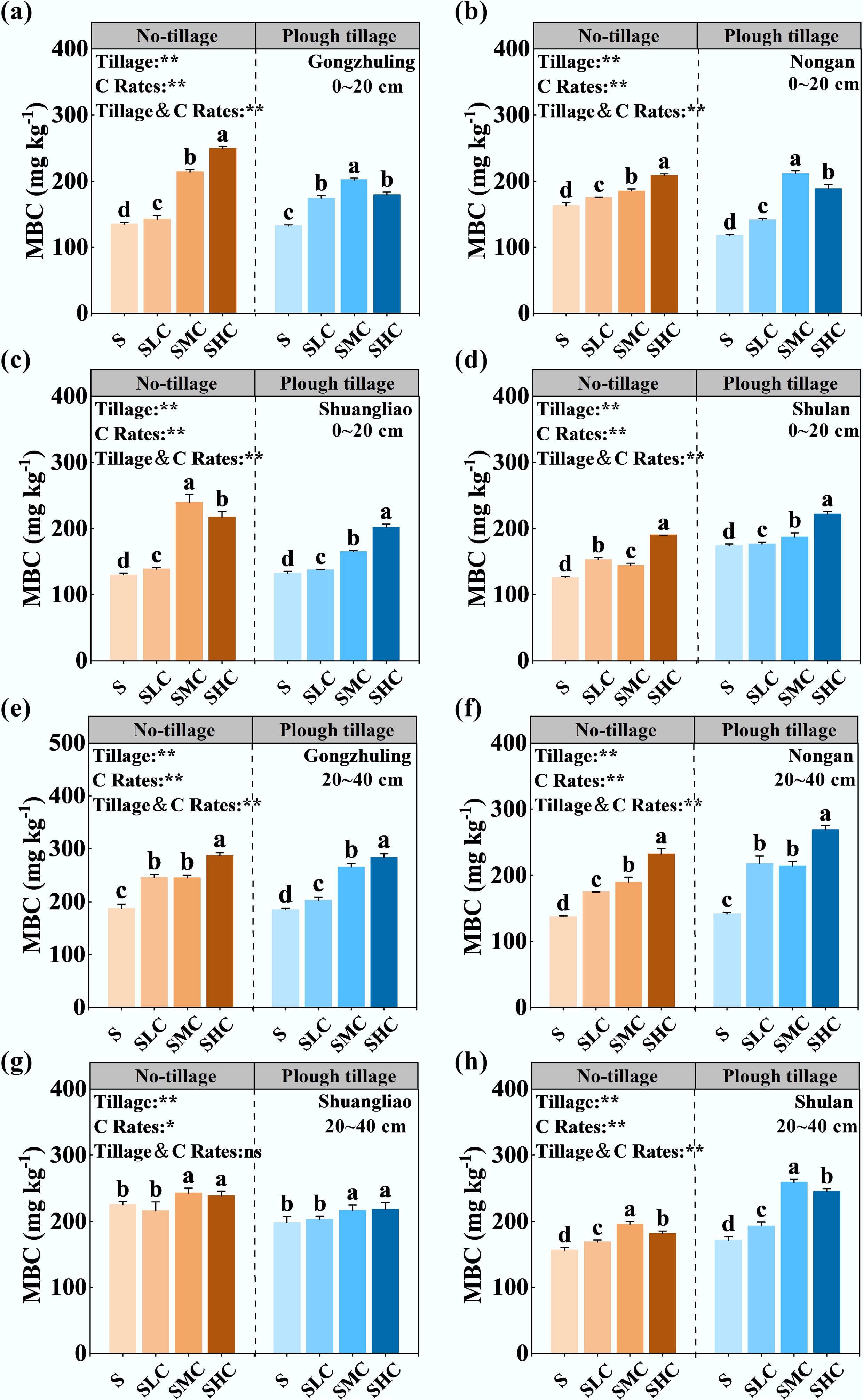
Figure 4.
Effects of tillage and carbon input on MBC. The MBC content of topsoil in (a) Gongzhuling, (b) Nongan, (c) Shuangliao, and (d) Shulan. MBC content of subsoil in (e) Gongzhuling, (f) Nongan, (g) Shuangliao, and (h) Shulan. Bars (mean ± SD, n = 3) with different lowercase letters are significantly differentamong treatments (p < 0.05). The main and interactive effects of tillage (Tillage) and carbon input rate (C Rates) are shown: **, p < 0.01; *, p < 0.05; ns, not significant at p > 0.05.
In the topsoil, the MNC content under increased C input rate treatments was increased by an average of 17.6% (under no-tillage) and 13.6% (under plough tillage) compared with the S treatment (Fig. 5). The proportion of FNC in MNC was higher than that of BNC. Compared with the S treatment, the content of FNC under the increased C input rate treatments increased by 20.2% and 16.8% under no-tillage and plough tillage, respectively. The content of FNC in the topsoil increased with an increase in the C input rate. Compared with the S treatment, the BNC content in the topsoil increased by 9.6% under no-tillage and 3.8% under plough tillage. In the subsoil, the MNC content under the no-tillage treatment increased with an increase in the C input rate. Under the plough tillage treatment, the MNC content reached the highest under the SLC treatment, and then decreased with an increase in the C input rate. The FNC and BNC contents of the SHC treatment in the subsoil under the plough tillage treatment decreased by 3.6% and 8.6% on average compared with SLC.
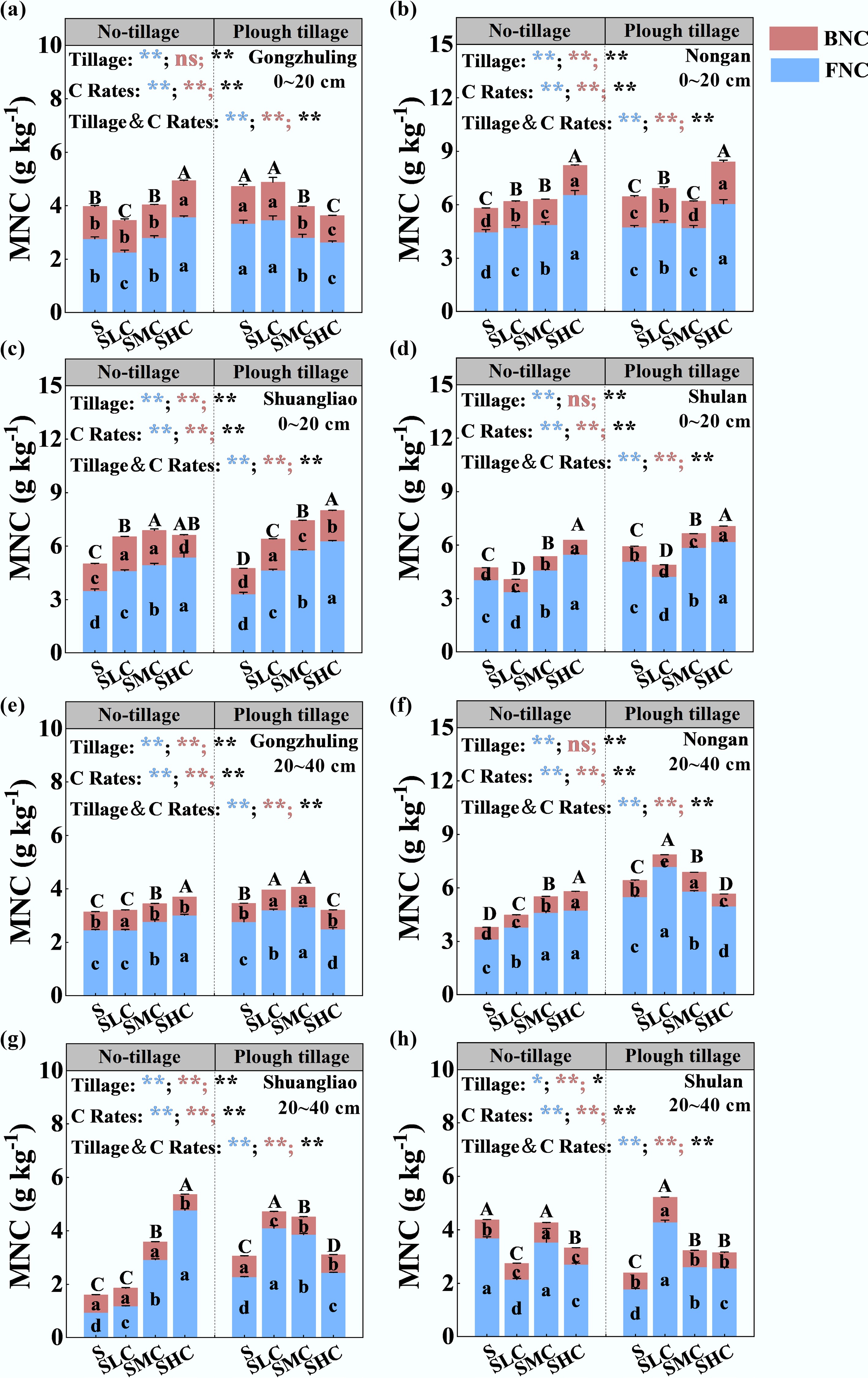
Figure 5.
Contents of BNC, FNC, and MNC under different tillage treatments and C rates. The MNC content of topsoil in (a) Gongzhuling, (b) Nongan, (c) Shuangliao, and (d) Shulan. MNC content of subsoil in (e) Gongzhuling, (f) Nongan, (g) Shuangliao, and (h) Shulan. For each necromass carbon type, bars (mean ± SD, n = 3) with different lowercase letters are significantly different among treatments (p < 0.05). Letters for FNC, BNC, and MNC are colored blue, red, and black, respectively. The main and interactive effects of tillage (Tillage) and carbon input rate (C Rates) are shown separately for FNC, BNC, and MNC: **, p < 0.01; *, p < 0.05; ns, not significant at p > 0.05.
Effects of tillage and carbon inputs on soil physicochemical properties and extracellular enzyme activities
-
Plough tillage significantly decreased bulk density, but there was no significant difference in BD between different C input rates (Supplementary Table S1). Additional C inputs significantly increased the soil TN content. Compared with the S treatment, TN in the topsoil and subsoil increased by 19.3% and 15.9%, respectively. Plough tillage significantly affected the contents of TP and TK in the soil. Compared with the no-tillage treatment, the TP content under plough tillage increased by 12.1%, whereas the TK content under plough tillage decreased by 3.0%. TK was affected by the interaction of tillage, carbon input, and soil layer. The TK content of the subsoil under the ploughing + SHC treatment was 7.2% higher than that under the ploughing + S treatment. The effects of C input rates and tillage on soil enzyme activities were significantly different between the different soil layers. The enzyme activities in the subsoil were higher than those in the topsoil. On average, the activities of AG, CBH, and XY increased by 3.8%, 10.8% and 7.5%, respectively. The activity of CBH and BG increased with an increase in carbon inputs, but this trend was not reflected in the subsoil. The activities of AG and BG under the SHC treatment were significantly lower than those under the SLC treatment with additional carbon input, which was more obvious under plough tillage.
Differences in microbial community and diversity between the topsoil and subsoil
-
The taxonomic distribution of bacterial and fungal communities was evaluated at the phylum level, and the sequences in each operational taxonomic unit were analyzed with 97% similarity (Figs 6 and 7). In each treatment, Acidobacteriota and Proteobacteria were the dominant phyla of soil bacteria, with relative abundances of 14.2%–27.9% and 21.0%–36.8%, respectively. Basidiomycota and Ascomycota were the dominant phyla of soil fungi, with relative abundances of 12.6%–56.5% and 38.8%–69.0%, respectively. Compared with the topsoil, the relative abundance of Proteobacteria in the subsoil decreased by 7.8% on average, and it always showed that the level under plough tillage was higher than that under no-tillage. The relative abundance of Basidiomycota decreased by 6.9% and Ascomycota increased by 5.3% in the topsoil compared with that under no-tillage. In the subsoil, the relative abundance of Basidiomycota in under plough tillage increased by 4.8% compared with no-tillage, whereas the relative abundance of Ascomycota increased by 4.3%. Soil microbial diversity showed different degrees of difference between two different soil layers, especially under plough tillage (Fig. 8). The diversity of bacteria and fungi (Shannon, Simpson) in the topsoil showed an increasing trend with an increase in the C input rates, whereas the diversity of bacteria and fungi in the subsoil showed a decreasing trend with an increase in the C input rates.
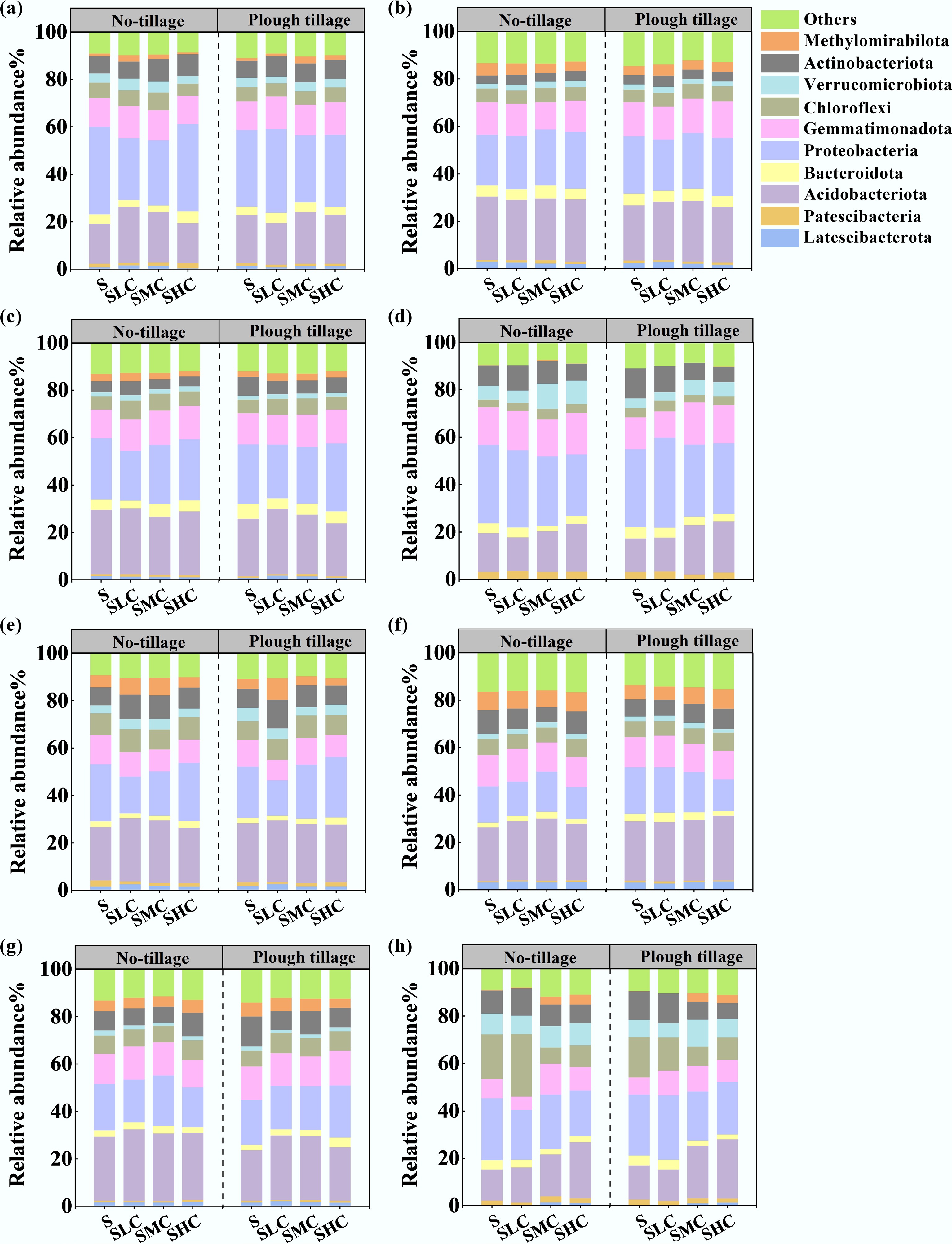
Figure 6.
Relative abundance of bacterial community composition in (a)–(d) the topsoil and (e)–(h) subsoil across the four experimental sites: (a), (e) Gongzuling, (b), (f) Nonggang, (c), (g) Shuangliao, and (d), (h) Shulan.
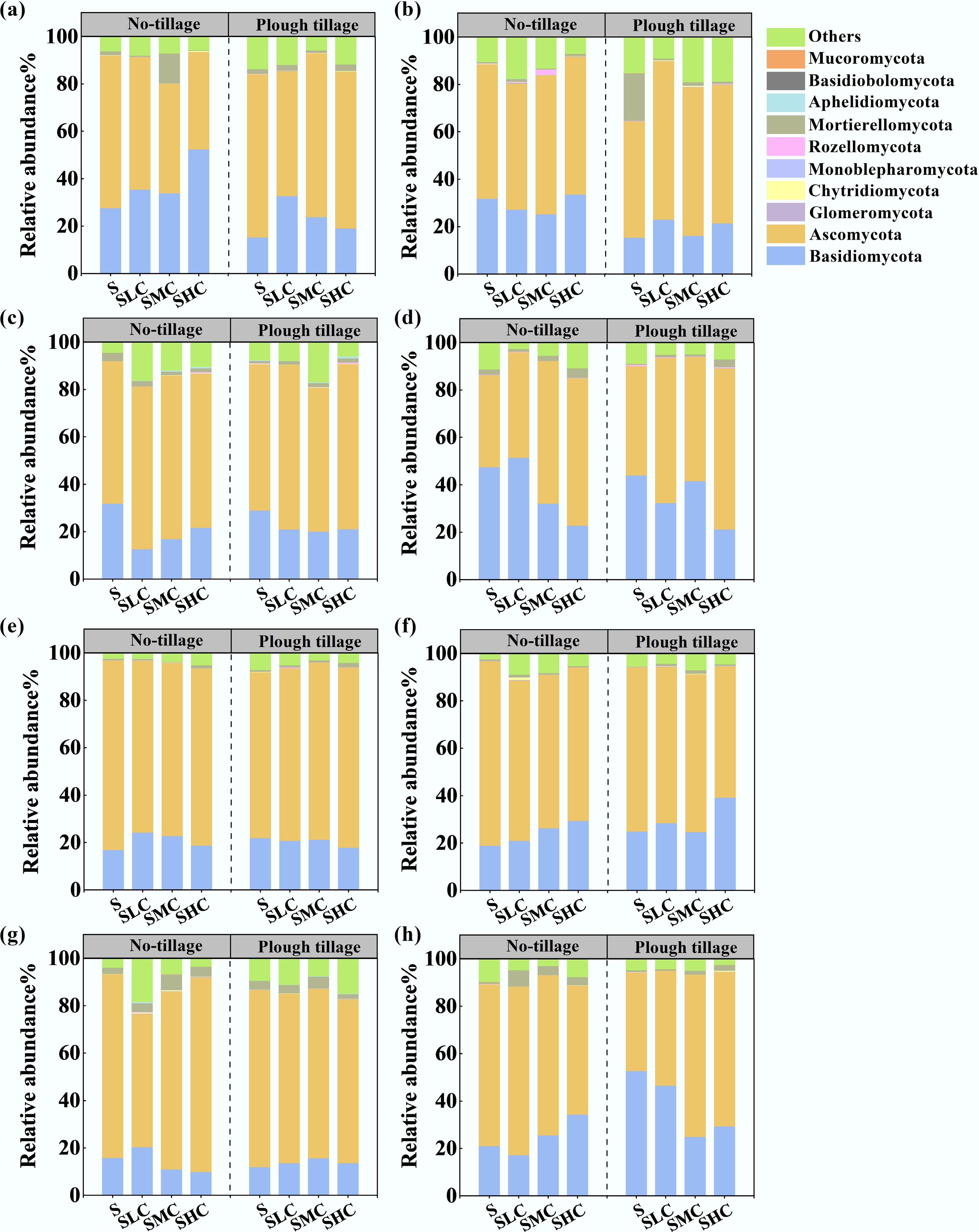
Figure 7.
Relative abundance of the fungal community composition in the (a)–(d) topsoil and (e)–(h) subsoil across the four experimental sites: (a), (e) Gongzuling, (b), (f) Nonggang, (c), (g) Shuangliao, and (d), (h) Shulan.
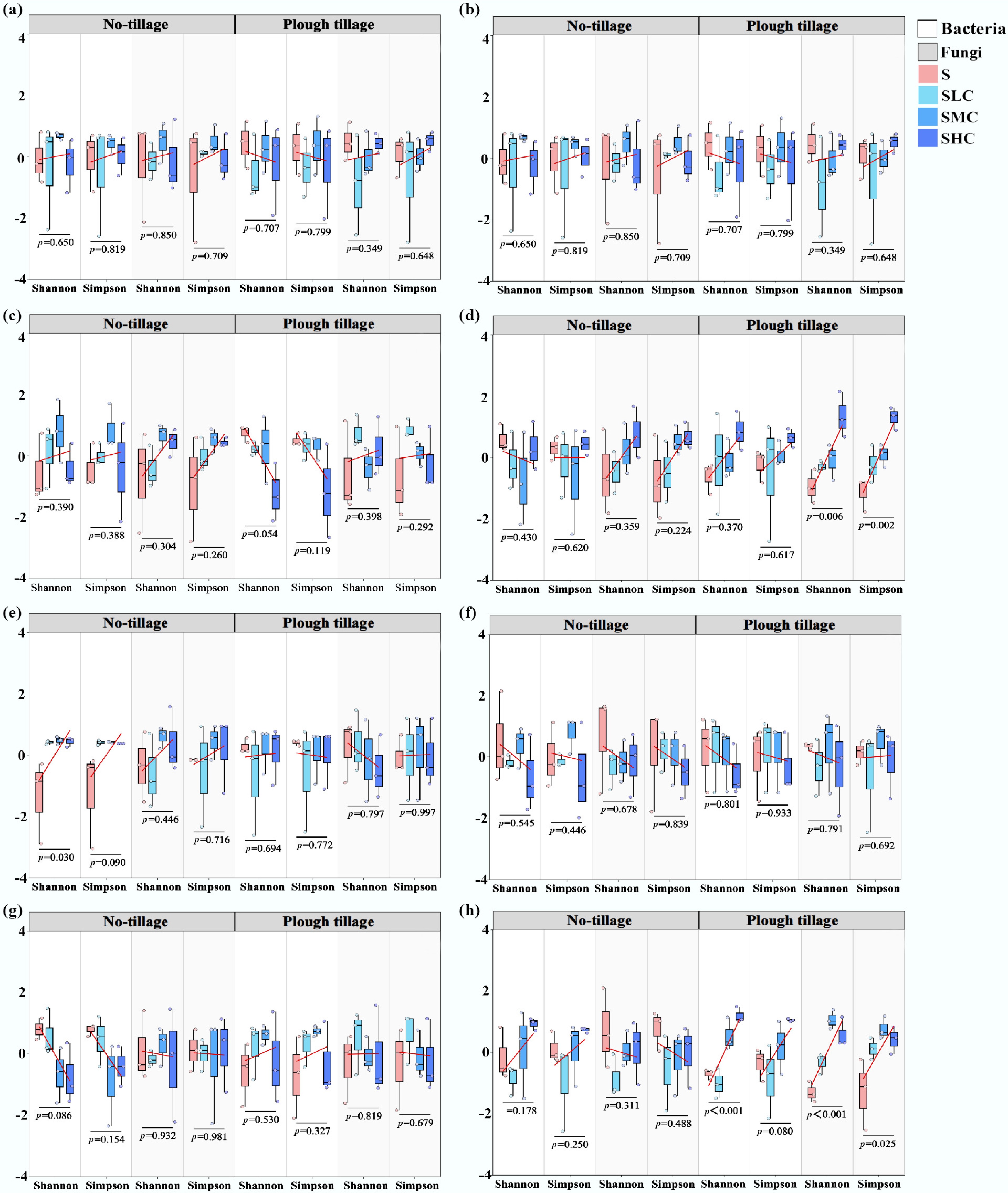
Figure 8.
The α diversity of bacterial and fungal communities in (a)–(d) the topsoil and (e)–(h) the subsoil across the four experimental sites: (a), (e) Gongzhuling, (b), (f) Nongan, (c), (g) Shuangliao, and (d), (h) Shulan. Data are normalized Z-scores. Red lines represent the trend lines obtained by linear fitting, and the associated p-values indicate the significance of the difference in fungal and bacterial α diversity between treatments.
-
The purpose of this study was to increase the amount of manure applied after returning straw to the field to achieve the goal of supplementing carbon inputs and increasing the SOC content. In general, the topsoil's SOC content gradually increased with an increase in carbon input (Fig. 2). The simple carbon-containing substrates (such as proteins and short-chain fats) introduced by fermented manure activate the carbon decomposition behavior of microorganisms[27]. At the same time, the introduction of nitrogen and phosphorus eliminated the nutrient limitations of microbial growth when only straw was returned to the field, and microbial biomass increased rapidly[28]. The rapid turnover of microbial biomass promotes the accumulation of microbial residues[29], which, in turn, promote the enrichment of carbon in the topsoil[30]. However, in the subsoil, the SOC content after carbon inputs showed an opposite trend to that of the surface layer, especially under plough tillage (Supplementary Fig. S1), which contradicted the view that SOC sequestration depended on carbon inputs[31]. Compared with untreated peat, ploughing improves aeration and inputs sufficient carbon into peat, which may enhance microbial respiration. Excessive substrate will be mineralized and lost in the form of carbon dioxide instead of being utilized for biosynthesis[32]. There was also a difference in the MAOC content between soil layers after carbon input (Fig. 3; Supplementary Fig. S2). There were asynchronous characteristics among MBC, MNC, and MAOC accumulation in the subsoil, which may be caused by the difference in the MNC accumulation process and its combination with MAOC[8]. FNC is the main component of MNC (Fig. 5; Supplementary Fig. S3), which is an important precursor for the formation of MAOC. Correlation analysis showed that FNC was more strongly correlated with SOC (Fig. 9a). In general, the turnover time of fungal necromass is longer, which is more conducive to maintaining the stability of SOC[33]. However, after the additional carbon input, the FNC content in the subsoil decreased. Microbial utilization preference studies have shown that bacteria prefer the uptake of simple organic compound substrates, whereas fungi have more advantages in the utilization of complex compound substrates[34]. Therefore, in the competition for simple substrates, bacteria are significantly better than fungi[35]. In the subsoil, the low-molecular-weight organic matter brought by a large amount of carbon input after tillage promotes bacterial reproduction and squeezes the living space of fungi[35], resulting in the accumulation rate of FNC being lower than the decomposition rate. In addition, the mineral activity in the subsoil is low, and the improved aeration caused by tillage results in oxidation of the mineral surface, an increase in the proportion of stable minerals, and loosening of the soil structure, which hinders the binding process of FNC and minerals[12,36,37]. In light of the results of the study, we conclude that there are differences between the topsoil and subsoil under different carbon input and tillage conditions. Under no-tillage conditions, high exogenous carbon inputs are more conducive to carbon sequestration in the topsoil, whereas ploughing combined with low exogenous carbon inputs is more conducive to carbon sequestration in the subsoil.
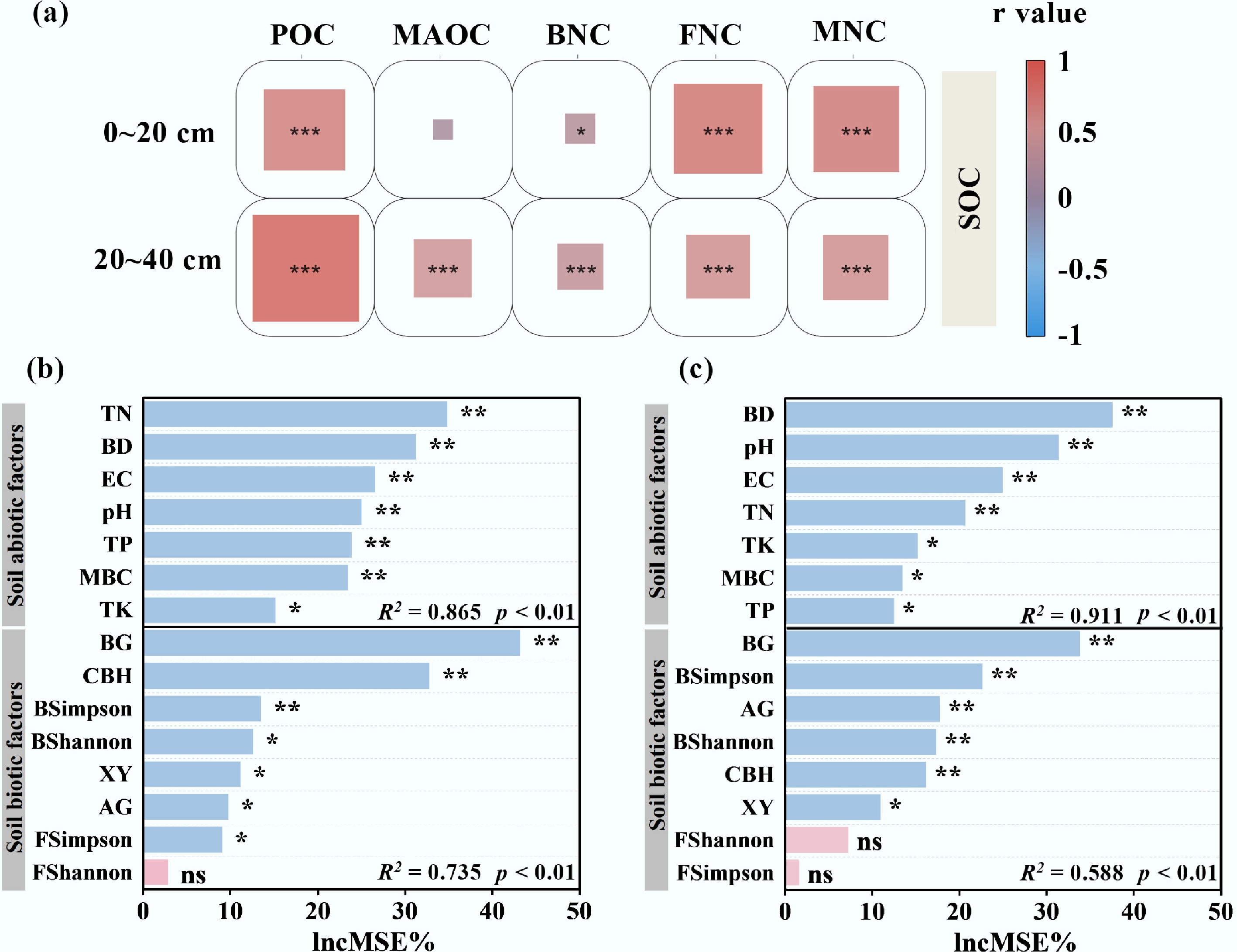
Figure 9.
(a) Relationships between SOC and individual carbon components were analyzed using Pearson's correlation. Random forest models were applied to evaluate the importance of biotic and abiotic factors to SOC in the (b) topsoil and (c) subsoil. Significance levels: *** p < 0.001, ** p < 0.01, * p < 0.05; ns, not significant.
Characteristic differences in SOC accumulation caused by microorganisms
-
Fungi and bacteria in the soil are the main decomposers after carbon input, and they are also the key links in the immobilization of exogenous carbon into SOC[38]. Returning organic matter to the field provides a sufficient carbon source for microorganisms, and the additional input of carbon while balancing the carbon–nitrogen ratio of returned straw can significantly increase the diversity and richness of soil microbial communities[39]. However, this study found that in resource-rich soils, the diversity of fungal and bacterial communities did not change significantly in the short term after tillage and carbon input (Fig. 8). In relatively barren soils (such as the subsoil of Shulan), tillage significantly increased the diversity of soil fungal and bacterial communities as the carbon input increased. Fungi and bacteria occupy the same niche in the soil[35]. The complex competition and cooperation between fungal and bacterial communities in nutrient-rich soils enhances their resistance to soil environmental changes, whereas barren soils increase competition for fresh nutrients[40]. However, in general, microbial richness and the uniformity of species distribution had no significant effect on SOC sequestration (Fig. 9b, c), but specific phyla (Proteobacteria) played a prominent role in the carbon cycle (Supplementary Fig. S5). Proteobacteria is the most abundant phylum in the soil. When the carbon source is sufficient, the microorganisms grow rapidly but they prefer small-molecule carbon sources and promote carbon mineralization[41]. In the subsoil, although the relative abundance of Proteobacteria decreased, the nutrient input after tillage stimulated their activity (Supplementary Fig. S6). After consuming labile organic carbon, these microorganisms may prefer to utilize the relatively easily decomposed FNC through cross-feeding (Supplementary Figs S4 and S8). In addition, the specific properties of microorganisms that obtain carbon and nutrients are reflected by soil enzyme activity[42]. The activities of BG and CBH in the topsoil increased significantly (Supplementary Table S1), indicating that microorganisms in the topsoil preferentially decomposed and utilized the cellulose and hemicellulose in straw[43]. The secondary organic matter formed after the decomposition of organic matter is more conducive to the formation of POC, thereby enhancing the effect of POC on topsoil SOC (Fig. 10a). The activities of AG and BG in the subsoil decreased significantly, indicating that the carbon acquisition strategy of microorganisms in the subsoil turned to the use of simple substrates such as sugars by bacteria[44]. At the same time, combined with the negative correlation between CBH/BG and FNC (Supplementary Figs S7 and S9), it indicates that fungi are more inclined to slowly decompose complex substrates, and carbon may be used for respiration rather than biomass accumulation. The formation of MAOC is mainly driven by the input of microbial metabolic carbon[45]. FNC is decomposed by the dominant bacterial phylum[46], resulting in insufficient precursors for the formation of MAOC (Fig. 10b). Therefore, we conclude that the difference in SOC in response to changes in the carbon inputs between the topsoil and subsoil is attributed to the difference in FNC accumulation caused by changes in the microbial carbon utilization strategies in different soil layers.
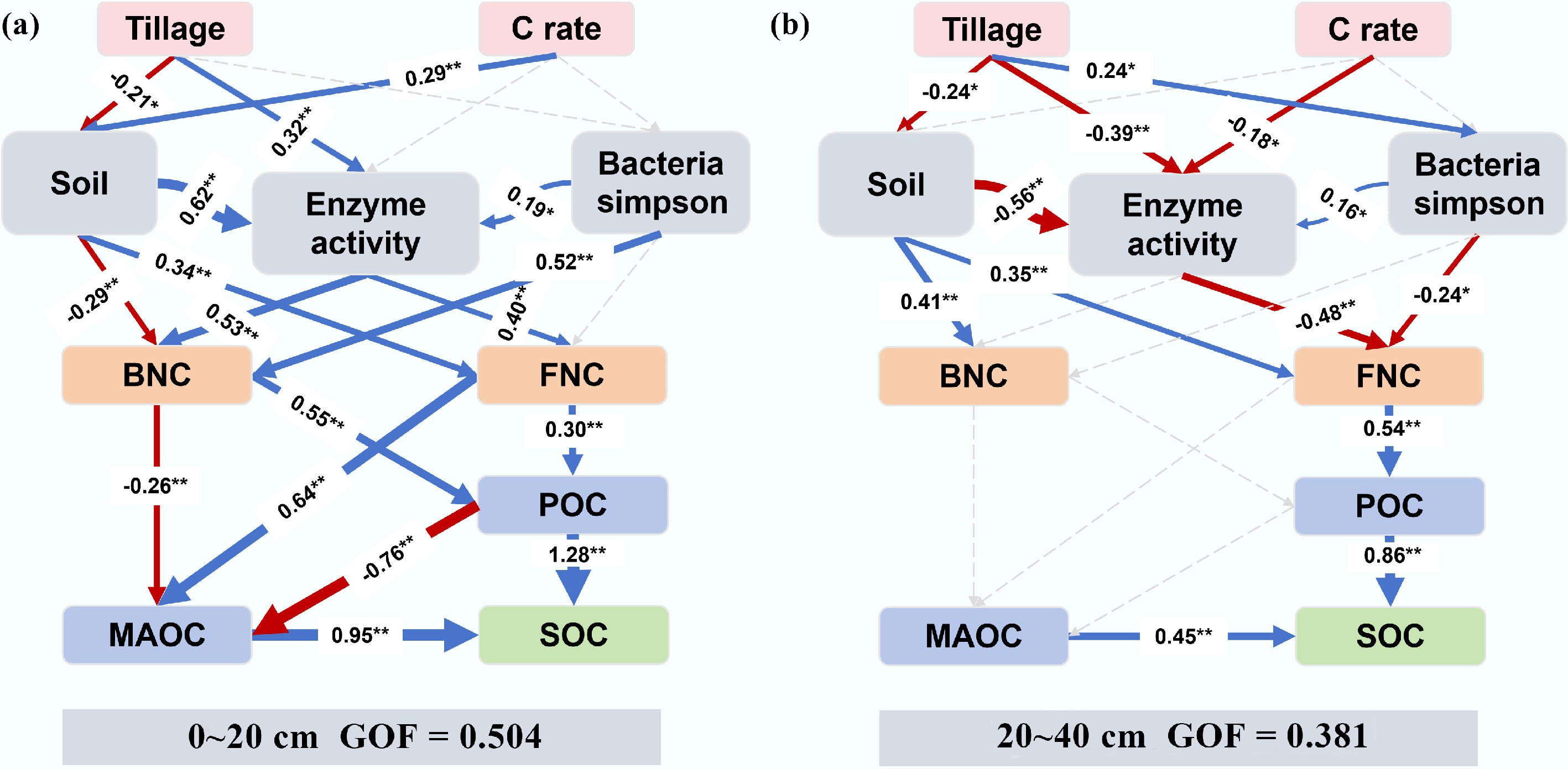
Figure 10.
Relationships between SOC and its components, FNC, and BNC in the (a) topsoil and (b) subsoil, as analyzed by partial least squares path modeling. The arrows' widths correspond to the strength of the causal relationship; numbers are standardized path coefficients. Blue and red lines indicate positive and negative paths, respectively. Solid and dashed lines denote significant (p < 0.05) and nonsignificant paths, respectively: *, p < 0.05, **, p < 0.01. The R2 values indicate the proportion of variance explained for each factor. Models were assessed using the goodness of fit.
The feasibility of strengthening the integrated management of tillage and carbon input
-
Our study showed that the combined application of livestock manure and straw increased SOC in farmland under continuous cropping of maize compared with returning straw alone. However, targeted management measures are needed for different soil types. Through a multiple linear model (Supplementary Fig. S10), it was found that the initial SOC of the soil has a certain negative effect on the SOC sequestration of the topsoil but it has a positive effect on that of the subsoil. In addition, the looser bottom soil is conducive to SOC sequestration. Therefore, for soils similar to those at the Gongzhuling and Shulan experimental sites, where the subsoil BD is higher and the soil is carbon-poor, it is preferable to apply straw and a low amount of exogenous carbon, and promote subsoil carbon sequestration by ploughing. For soils similar to the sandy loam at the Shuangliao experimental site, no-tillage is more suitable to maintain the high soil BD and avoid the loss of SOC[47]. When straw and livestock manure are returned to the field, the addition of carbon-fixing bacteria seems to enhance the carbon sequestration capacity of the topsoil. In addition, long-term no-tillage is more conducive to improving the BD of the subsoil and driving deep SOC sequestration under the synergistic effect of crop roots[48]. For soils similar to the carbon-rich clay loam at the Nongan experimental site, more detailed management is needed to improve the soil carbon sequestration efficiency and maintain the soil carbon balance. Perhaps alternating plough tillage and no-tillage could better maintain SOC sequestration.
-
This study highlights the effects of tillage and carbon input on the differences in carbon sequestration between the topsoil and subsoil. The content of SOC in the topsoil increased with an increase in carbon inputs and was not limited by tillage, whereas the content of SOC in the subsoil reached its highest when straw and low-carbon organic fertilizer were applied, which was more obvious under plough tillage. It is inferred that the difference between topsoil and subsoil is largely caused by the difference in the accumulation of FNC into POC and MAOC. In the subsoil, bacteria such as Proteobacteria and enzymes decompose and reuse FNC, resulting in a decrease in the accumulation of the main organic carbon components (MAOC and POC) in the subsoil. This study confirmed that the application of carbon sources in addition to straw can increase the content of SOC in the topsoil and subsoil, but a large amount of carbon input will weaken the organic carbon sequestration capacity of the subsoil. We suggest adding carbon sources on top of returning straw to strengthen the organic carbon sequestration of farmland soil, and tailoring tillage practices to suit the farmland's SOC, BD, pH, and other characteristics to promote SOC sequestration via comprehensive management measures. There are still some limitations in this study. Because of the short experimental period, the changes in tillage methods and organic matter inputs in the short term may lead to fluctuations in the results. In the future, it may be possible to observe the adaptive changes of the soil and microorganisms, clarify the interaction mechanism between them, and deepen our understanding of farmland soil carbon sequestration over a longer time with additional carbon source inputs.
-
It accompanies this paper at: https://doi.org/10.48130/aee-0026-0006.
-
The authors confirm contribution to the paper as follows: Ximing Liu: conceptualization, methodology, software, visualization, writing; Chang Zhang: writing; Xiaolin Li: resources; Song Cheng: resources; Jinyao Yan: methodology, writing – review & editing; Jingchao Yuan: writing – review & editing; Jianzhao Liu: resources, supervision; Yao Liang: resources, conceptualization, methodology, supervision; Jingkun Lu: resources, supervision; Wei Fan: investigation, conceptualization, methodology, visualization, writing – review & editing, supervision, funding acquisition; Hongguang Cai: investigation, conceptualization, methodology, resources, supervision. All authors reviewed theresults and approved the final version of the manuscript.
-
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
-
This work was supported by the National Key Research and Development Program of China (Grant No. 2023YFD1501100), the Natural Science Foundation of Jilin Province, China (Grant No. 20220301018NY), and the 7th Batch of Jilin Province's Project to Support Young Science and Technology Talents (Grant No. QT202318).
-
The authors declare no conflicts of interest.
-
Farmland soil organic carbon content showed layer-specific differences at different carbon input levels.
Microbial activity and microbial necromass carbon mediated the transformation of input carbon to soil carbon.
The binding ability of microbial source carbon to minerals in the subsoil was weakened.
The appropriate input of carbon from sources other than straw into farmland can enhance soil carbon sinks.
-
Full list of author information is available at the end of the article.
- The supplementary files can be downloaded from here.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Liu X, Zhang C, Li X, Cheng S, Yan J, et al. 2026. Differences in soil organic carbon among soil layers caused by microbial necromass carbon accumulation under different tillage and carbon input regimes. Agricultural Ecology and Environment 2: e009 doi: 10.48130/aee-0026-0006
Differences in soil organic carbon among soil layers caused by microbial necromass carbon accumulation under different tillage and carbon input regimes
- Received: 08 October 2025
- Revised: 15 January 2026
- Accepted: 22 January 2026
- Published online: 05 March 2026
Abstract: Tillage and carbon input are considered to be important management measures for maintaining the sustainable production capacity of farmland soil. However, our understanding of the differences in the response of soil organic carbon (SOC) to tillage and carbon input between different soil layers remains limited. Therefore, we established no-tillage and plough tillage treatments alongside total straw return and exogenous carbon inputs of 600, 1,200, and 2400 kg C ha−1 yr−1 in four maize fields in Jilin Province. After one year, topsoil (0–20 cm) and subsoil (20–40 cm) were collected. Microbial high-throughput sequencing and amino sugar biomarkers combined with a partial least squares structural equation model (PLSPM) were used to analyze the role of microbial necromass carbon (MNC) in the SOC sequestration pathway of topsoil and subsoil, and the differences in SOC between different soil layers. We found that increasing the carbon input in addition to returning straw increased the SOC content of the topsoil as the carbon input increased, whereas the increase in SOC in the subsoil after plough tillage decreased with the increase in C input rates from 27.0% to 0.2%. In the topsoil, additional carbon input increased the soil total nitrogen (TN) content by 19.3%; β-glucosidase (BG) and cellobiohydrolase (CBH) activity by 29.8% and 24.2%, respectively; and microbial biomass carbon (MBC) by 35.0%, which, in turn, promoted fungal necromass carbon (FNC) and bacterial necromass carbon (BNC) by 18.5% and 6.7%, respectively. Through PLSPM analysis, it was found that FNC promoted the accumulation of mineral-associated organic carbon (MAOC) in the topsoil, whereas FNC and BNC together promoted the accumulation of particulate organic carbon (POC). In the subsoil, the changes in enzyme activity and the enzyme ratio showed that carbon input increased the preference of microorganisms for refractory carbon sources compared with returning straw alone, but the negative correlation between CBH/BG and FNC showed that carbon may be utilized more in respiration than in growth after plough tillage improved the aeration of the subsoil. Plough tillage promoted the decomposition of FNC by microorganisms such as Proteobacteria in the subsoil. The reduction in the precursor substance FNC limits the accumulation of MAOC. These findings suggest that carbon sequestration in the tillage layer is not entirely dependent on carbon input, but that higher farmland soil carbon sequestration benefits can be achieved through the comprehensive management of tillage, straw, and organic fertilizer.
-
Key words:
- Carbon input /
- Carbon sequestration /
- Microbial necromass carbon /
- Microbe mediation