-
Tea (Camellia sinensis), one of the world's most widely consumed non-alcoholic beverages, is well known for its complex flavor and health benefits[1−3]. Its functional and sensory characteristics are due to the presence of bioactive constituents, such as theanine, caffeine, flavonoids, sugars, and aromatic compounds[4]. Anji Baicha is a premium green tea derived from the albino cultivar 'Baiye1' and is extensively cultivated in the Anji County of Zhejiang Province (China). It is valued for its signature 'umami' taste and high market value. Theanine (γ-glutamyl-L-acetamide), the predominant non-protein amino acid, largely determines the unique characteristic of the albino tea cultivars[4,5]. It is synthesized in roots during winter and mobilized to shoots in spring. However, 'Baiye1' exhibits temperature sensitivity and low yield, likely due to chloroplast deficiency[6,7]. Therefore, it is essential to develop and implement innovative and effective soil fertilization strategies to improve the quality and yield of Anji Baicha.
Nitrogen (N) is a macronutrient essential for the growth and development of all plants, including tea. In traditional tea cultivation, N fertilizer is combined with phosphorus (P) and potassium (K) fertilizers to achieve a balanced NPK status[8]. While balanced N-P-K application enhances soil quality and boosts tea yield and quality, long-term chemical N fertilization causes environmental issues like soil acidification[9]. Excessive N application reduces tea quality, while P overuse impairs tea aroma[10]. Studies have demonstrated that supra-optimal levels of P and K decrease chlorophyll content in above-ground tissues and negatively influence amino acid (theanine, glutamic acid) concentrations in new shoots of tea plants[11], compromising quality. On the other hand, a deficiency in soil nutrients negatively influences the quality and yield of tea[12]. Collectively, these findings indicate that a balanced application of N, P, K, and micronutrients is essential for optimizing tea yield and theanine content[13]. Therefore, targeted fertilization strategies should be developed specifically for albino tea cultivars.
In tea plantations, combining chemical and organic fertilizers with soil conditioners effectively mitigates soil acidification and modifies the soil microbiota[14,15]. For instance, the addition of humic acid enhances soil structure and fertility, leading to increased crop yields[16]. Besides, magnesium (Mg) availability plays a crucial role in regulating theanine biosynthesis in root tissues and facilitating its transport to developing shoots, a process strongly associated with enhanced tea quality[17,18]. A recent study has indicated that synthetic microorganisms enhance nitrogen-use efficiency (NUE) and facilitate theanine accumulation in tea plants[19]. However, failing to implement proper field management measures to maintain leaf albinism often results in insufficient soil N, P, and K in Anji Baicha plantations[20], inadvertently limiting growth and compromising yield and quality. While N and Mg regulate chlorophyll synthesis, the specific impact of these nutrients on greening in 'Baiye1' remains uncharacterized. Therefore, a balanced and effective nutrient management strategy is needed to improve soil quality, enhance yield, and ensure quality in Anji Baicha plantations.
The present study evaluated the impact of rational fertilization, involving optimized N-P-K ratios with Mg supplementation, on tea performance, soil nutrients, and microbial communities. Two novel treatments, namely optimized fertilization treatment (OPT) and OPT with Mg supply (OPT+Mg), were implemented in the tea plantation, using farmers' practice (FFP) as the control. The study's objectives were to: (1) quantify the effects of N and Mg on 'Baiye1' leaf greening, and (2) identify the key drivers of yield and quality in the tea. The findings will provide a theoretical foundation for developing fertilization strategies to improve soil health and productivity of Anji Baicha plantations.
-
A field experiment was carried out at a site in Anji County (30°45' N, 119°46' E; 50 m a.s.l.), Zhejiang Province, China, which is the native of the albino tea cultivar 'Baiye1'. This area experiences a humid subtropical climate, with a mean annual temperature of 15.43 °C and an average annual rainfall of 1,500 mm. The soil is classified as yellow loam, and the topsoil (0–20 cm) possesses the following characteristics: pH 3.8, alkaline-hydrolyzable N 155 mg·kg−1, total N (TN) 2.3 g·kg−1, soil organic carbon (SOC) 25.35 g·kg−1, available P 315 mg·kg−1, and available K 199 mg·kg−1. The clonally propagated seedlings of 'Baiye1' were established in the field in 2006, at a spacing of 1.5 m between rows and 0.33 m within rows.
Experimental design
-
In 2021, the study was initiated implementing three fertilization treatments and six replicate plots per treatment in the tea plantation: (1) FFP: 3,000 kg·ha−1 organic fertilizer (NPK ≈ 2:1.5:1.5) + 1,200 kg·ha−1 compound fertilizer (15:15:15); (2) OPT: 4,500 kg·ha−1 humic acid-enriched organic fertilizer + 1,200 kg·ha−1 compound fertilizer (21:6:13) + 100 kg·ha−1 CaO soil conditioner (pH = 8.0−10.0); (3) OPT+Mg: OPT components + 60 kg·ha−1 MgSO4 (Supplementary Table S1). The organic fertilizer, conditioner, and MgSO4 were applied once a year in October. Meanwhile, the compound fertilizer was applied in three stages: 25% in February, 25% in May, and 50% in October. All fertilizers were applied in bands prepared at a depth of 15−20 cm between the plant rows.
Every year, in May, the plants were pruned according to local farming practices. The canopy, including the leaves and branches (30–40 cm above the ground), was removed from each plant during the process and returned to the tea plantations. Additionally, manual weeding was carried out twice a year, once in June and July, and again in September.
Sampling and analysis of tea shoots and roots
-
In April 2022, one bud and two leaves were harvested from each plant to assess the yield. Some of the new shoots were immediately microwave-fixed and dried for quality and nutrition determination; the other part of the new shoots was immediately frozen with liquid nitrogen and then dried for the determination of chlorophyll content. Similarly, roots were excavated, rinsed, and oven-dried to constant weight. Both the shoot and root tissues were finely ground and stored for subsequent chemical analysis.
The N content was determined with a Vario Max CN analyzer (Elementar, Germany), while the powdered sample was digested at 550 °C and dissolved in dilute HNO3 to analyze the content of P, K, and other mineral elements by inductively coupled plasma-optical emission spectrometry (ICP-OES; iCAP 7400; Thermo Fisher Scientific, USA).
The modified ninhydrin method was used to determine amino acids (AA) in shoots and roots, and Fe-tartrate colorimetry was used to determine tea polyphenols (TP)[21]. Here, 100 mg of the powdered sample was initially incubated in 5 mL of boiling ddH2O (5 min water bath, 2.5 min vortex). After the reaction, the AA and TP were detected using a UV-Vis spectrophotometer (Shimadzu, UV-2550, Japan) at wavelengths of 570 and 540 nm, respectively.
Further, to determine the composition of free AAs in shoots and roots, high-performance liquid chromatography (HPLC) was performed after derivatization with a Waters AccQ•Tag™ Kit (Waters Corporation, Milford, MA, USA). Similarly, the catechin content was evaluated using an HPLC system (Waters Corporation, Milford, MA, USA) equipped with a C18 reversed-phase column (250 mm length × 4.6 mm internal diameter) under conditions described previously[22]. Both groups of compounds were quantified by comparing the chromatographic peak areas with those of the authentic standards.
Frozen-dried tea leaf samples (100 mg) were homogenized in 10 mL of 80% acetone containing approximately 1 g of CaCO3. The ground material was filtered until the residue became colorless. Absorbance at 663 and 645 nm was measured using a UV-Vis spectrophotometer (Shimadzu UV-2550, Japan). The concentrations of chlorophyll a and chlorophyll b were calculated according to the following equations:
$ \rm{Chl\; a\; (mg/g)=(12.7\times OD_{663}-2.69\times OD_{645})/M} $ $ \rm{Chl\; b\; (mg/g)=(22.9\times OD_{663}-4.68\times OD_{645})/M} $ Total chlorophyll content was obtained as the sum of Chl a and Chl b. 'M' represents the mass (g) of the leaf sample used[12].
Soil sampling and analysis
-
In April 2022, coinciding with the spring tea harvest, soil was sampled from five randomly selected points along a row in each experimental plot, avoiding the fertilization furrows. The surface debris, including leaves and pruning litter, was removed, and soil was sampled from a depth of 0–20 cm using a stainless-steel corer (5 cm diameter). The visible stones, plant residues, and roots were removed by hand, and the soil was sieved (< 2 mm) and split into two portions: one stored at –80 °C for molecular analysis, and the other air-dried for chemical analysis.
Soil pH was assessed in a 1:2.5 (w/v) soil-deionized water suspension with an ORION 3 STAR pH meter (Thermo Fisher Scientific, USA). Ammonium (NH4+) and nitrate (NO3−) were extracted by shaking soil with 2 M KCl (1:10 w/v) for 2 h at 240 rpm and quantified using a SAN++ continuous-flow analyzer (Skalar, Netherlands). Total N (TN) and soil organic carbon (SOC) were measured with a Vario Max CN elemental analyzer (Elementar, Germany). Available nutrients (P, K, Ca, Mg, Al, S, Fe, Mn, Cu, and Zn) were extracted from the soil with the Mehlich-3 solution (2.5 g soil in 25 mL extractant, 5 min shaking)[23] and analyzed by inductively coupled plasma atomic emission spectrometry (ICP-AES; Thermo Jarrell Ash, USA).
Further, the soil quality index (SQI) was computed using the Soil Management Assessment Framework (SMAF) to holistically assess soil nutrient status[24]. Initially, a total dataset (TDS) was constructed from the measured soil parameters. Principal component analysis (PCA) was employed to assign weight coefficients to each parameter. Subsequently, all parameters were standardized to a 0.1–1.0 scale using appropriate standard scoring functions (SSF). The SQI was then derived from the weighted sum of scored parameters using the following equations[25]:
$ \mathrm{Soil}~\mathrm{quality}~\mathrm{index}~=\sum \limits_{i=1}^{n}(Wi\times \mathrm{Si}) $ where, Wi represents the weight of each parameter, Si represents its normalized score, and n represents the total number of parameters. Generally, higher SQI corresponds to improved soil quality.
DNA extraction, amplification, and sequencing
-
Genomic DNA was isolated from 0.5 g of fresh soil using a FastDNA® SPIN Kit (MP Biomedicals, Irvine, CA, USA). The purity and concentration of the extracted DNA were verified with a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, USA). DNA samples with acceptable purity and concentration were stored at −20 °C.
Polymerase chain reaction (PCR) was performed with the DNA to amplify the target genes. For bacterial communities, the V3−V4 region of the 16S rRNA gene was amplified with the primers 338F (5'-ACTCCTACGGGAGGCAGCAG-3') and 806R (5'-GGACTAACHVGGGTWTCTAAT-3'). For fungal communities, the ITS1 region was amplified with the primers ITS1F (5'-CTTGGTCATTTAGAGGAAGTAA-3') and ITS2R (5'-GCTGCGTTCTTCATCGATGC-3')[26]. The PCR mixture (20 µL) contained 4 µL of 5× Fast Pfu buffer, 2 µL of 2.5 mM dNTPs, 0.8 µL of each primer (5 µM), 0.4 µL of Fast Pfu DNA polymerase, and 10 ng of template DNA. The thermocycling process was carried out as follows: initial denaturation at 95 °C for 3 min; 27 cycles of amplification at 95 °C/30 s → 55 °C/30 s → 72 °C/45 s; final extension at 72 °C for 10 min. The amplicons were gel-purified (2% agarose; YuHua PCR Clean-Up Kit; Shanghai, China), quantified (Qubit 4.0; Thermo Fisher Scientific, USA), pooled (equimolar concentrations), and subjected to paired-end sequencing (Illumina Nextseq2000; Majorbio Bio-Pharm, Shanghai, China).
Bioinformatic processing
-
The raw sequences were quality-filtered (QIIME v1.9.1) by removing reads < 150 bp or those with homopolymer repeats[27]. The filtered reads were then assembled (FLASH v1.2.11). Operational taxonomic units (OTUs) were clustered at a 97% similarity threshold. The bacterial and fungal OTUs were taxonomically classified using SILVA (Release 128) and UNITE (Release 6.0) databases[28], respectively. The raw sequencing data have been deposited in the Genome Sequence Archive under the accession number PRJCA039142.
Construction of microbial co-occurrence networks
-
To investigate the microbial interactions in the soil under optimized fertilization, co-occurrence networks were constructed. Initially, the microbial OTUs with an average relative abundance below 0.01% or occurring in fewer than 20% of all samples were excluded. Then, the network inference was performed as follows: (1) A correlation matrix was generated based on the Spearman ranks of the filtered OTUs using the 'psych' package in R (version 4.5.0); (2) Correlations with FDR-adjusted p > 0.001 or absolute Spearman's |ρ| < 0.6 were filtered out; (3) The significant correlations were used to build the co-occurrence networks and compute the topological metrics via the 'igraph' R package[29]; (4) Finally, the networks were visualized in Gephi (version 0.9.2) using the Fruchterman-Reingold layout algorithm. In the network, the node size corresponds to OTU relative abundance; node color represents taxonomic phylum; edges denote the significant (red; p < 0.05) or non-significant (blue) correlations.
Statistical analysis
-
All data were analyzed in R (version 4.5.0). One-way ANOVA followed by Duncan's post-hoc test was used to compare tea yield, chlorophyll content, tea quality features, leaf nutrient content, soil properties, soil microbial α-diversity (OTU level), and soil microbial abundance among the three treatment groups.
The similarities in microbial community composition in the three treatments were assessed via PCoA (Bray-Curtis dissimilarity). Then, PERMANOVA was applied to test the influence of fertilization on overall microbial community structure. A Mantel test was conducted using the 'linkET' package to further evaluate the relationship between the microbial communities and the soil properties.
Random forest (RF) analysis was implemented with the 'randomForest' package to select the top ten bacterial/fungal genera (biomarkers) across the fertilization treatments. Additionally, RF regression was used to identify the predominant factors (soil available nutrients, microbial taxa, and community features) associated with the variations in tea shoot yield and AA content.
-
The present study found significant variations in the yield and quality of tea shoots among the three fertilization regimens (Fig. 1; Supplementary Fig. S1). The optimized fertilization regimens OPT and OPT+Mg increased tea yield by 22% and 29%, respectively, compared to FFP (p < 0.05). These two regimens increased the growth indicators of tea, such as height and crown width, also (Supplementary Fig. S1).
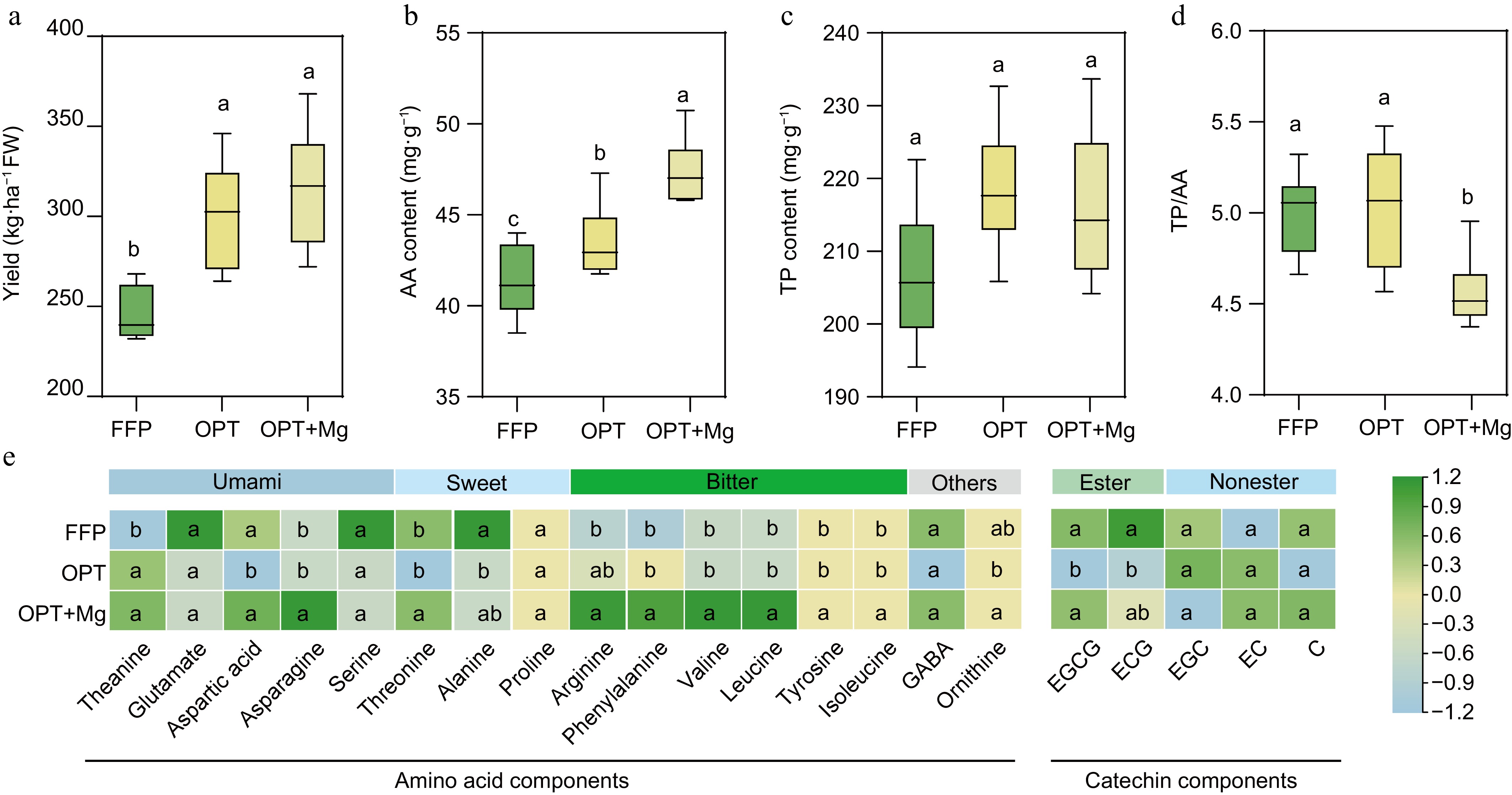
Figure 1.
The yield and quality of Anji Baicha across three soil fertilization regimens. (a) Yield. (b) Total free amino acid (AA) content of the shoots. (c) Total protein (TP) content of the shoots. (d) AA/TP ratio. (e) Heatmap depicting the contents of AA and catechins in the shoots. The heatmap is based on standardized data. ECG: epicatechin gallate, EC: epicatechin, EGC: epicatechin gallate, EGCG: epigallocatechin gallate, and C: catechin.
Then, the AA and TP levels in the tea shoots were analyzed to assess the quality of Anji Baicha under the different fertilization regimens. Here, OPT significantly increased the shoot AA content compared to FFP. The supply of Mg under optimized fertilization further enhanced this positive effect (Fig. 1b). Both OPT and OPT+Mg increased the TP content compared to FFP; however, these differences were not significant (Fig. 1c). Meanwhile, OPT+Mg resulted in a drop in the TP/AA ratio compared with FFP and OPT (Fig. 1d).
Further analysis revealed that the levels of theanine, the key amino acid imparting umami flavor, were significantly elevated in the young shoots under both OPT and OPT+Mg treatments compared to FFP (p < 0.05; Fig. 1e). However, the content of its precursor, glutamate, remained similar across the three fertilization regimens. Additionally, OPT+Mg enhanced the content of arginine, phenylalanine, tyrosine, leucine, ornithine, threonine, asparagine, valine, and isoleucine in the tea shoots compared to FFP or OPT (p < 0.05). Conversely, OPT reduced the levels of alanine and catechins, such as epigallocatechin gallate (EGCG) and epicatechin gallate (ECG), compared to FFP; however, the levels under OPT+Mg were comparable to those under OPT (Fig. 1e).
Changes in chlorophyll content and nutrient levels of tea shoots and roots
-
Typically, a change in leaf color to green would reduce the economic value of Anji Baicha. Therefore, to assess the effects of fertilization on economic value, chlorophyll content of Anji Baicha was measured. Analysis revealed no significant increase in chlorophyll content under OPT or OPT+Mg compared to FFP (Fig. 2a). Likewise, no significant differences were found in the nutrient accumulation patterns in the shoots among the fertilization regimens (Fig. 2b). In contrast, the elemental composition of roots varied substantially among the treatments. Specifically, N and P peaked under OPT; K, Ca, and Mg peaked under OPT+Mg; the microelements Fe, Mn, Cu, and Zn peaked under OPT, followed by FFP and OPT+Mg.
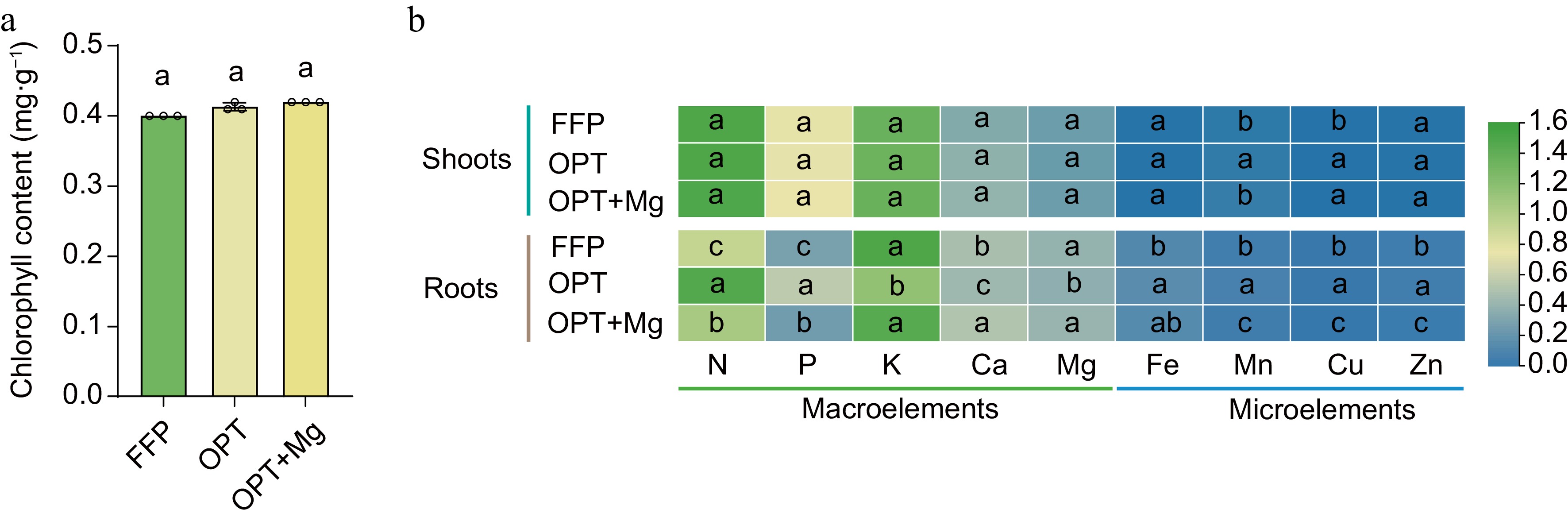
Figure 2.
The content of chlorophyll in the tea shoots and the accumulation patterns of nutrients in the shoots and roots of Anji Baicha across three soil fertilization regimens. (a) Chlorophyll content. (b) Heatmap depicting the content of nutrients in the shoots and roots. The heatmap is based on the log10-transformed data.
Changes in the soil's available nutrients and SQI
-
Fertilization significantly changed the soil properties of the Anji Baicha plantations (p < 0.05; Table 1). Optimized fertilization treatments, especially OPT+Mg, significantly increased the levels of most available nutrients in the soil. Among the treatments, OPT+Mg resulted in the highest soil pH, indicating that it alleviated soil acidification. Besides, OPT+Mg resulted in lower soil-available aluminum (Al) content than FFP and OPT. The available soil Cu was also lower under both OPT and OPT+Mg than under FFP (p < 0.05). Conversely, N, K, Ca, Mg, S, Mn, and Zn significantly increased under OPT and OPT+Mg (p < 0.05), whereas SOC, AP, and Fe showed no change.
Table 1. Soil available nutrients and SQI of Anji Baicha plantations under different fertilization regimens.
Parameters Unit Fertilization regimen FFP OPT OPT+Mg pH / 4.31 ± 0.13b 4.26 ± 0.1b 5.66 ± 0.64a SOC g·kg−1 34.06 ± 3.53ns 33.46 ± 0.96 36.25 ± 3.1 N g·kg−1 2.88 ± 0.23b 2.86 ± 0.07b 3.11 ± 0.21a P mg·kg−1 373 ± 43ns 394 ± 28 394 ± 5 K mg·kg−1 173 ± 9c 236 ± 26b 274 ± 5a Ca mg·kg−1 120 ± 16c 26 ± 72b 1852 ± 157a Mg mg·kg−1 28 ± 3c 77 ± 31b 129 ± 4a S mg·kg−1 9.3 ± 0.7c 12.1 ± 1.3b 14.6 ± 0.5a Al mg·kg−1 1746 ± 40a 1633 ± 44b 1418 ± 25c Fe mg·kg−1 536 ± 29ns 551 ± 17 543 ± 3 Mn mg·kg−1 3.17 ± 0.61c 4.2 ± 0.14b 12.39 ± 0.6a Cu mg·kg−1 2.16 ± 0.41a 0.4 ± 0.1c 0.87 ± 0.05b Zn mg·kg−1 3.11 ± 0.42c 2.92 ± 0.55b 5.13 ± 1.81a SQI / 0.36 ± 0.06c 0.43 ± 0.03b 0.63 ± 0.05a The different lowercase letters denote significant differences among treatments, while 'ns' indicates no significant difference. Further, SQI was calculated from these soil parameters to comprehensively assess the soil's response to different fertilization regimens. Compared with FFP, both the optimized fertilization regimens significantly improved the SQI of the tea plantation. SQI increased by 19.4% under OPT and 75% under OPT+Mg compared to FPP (Table 1).
Soil microbial diversity and community composition
-
Furthermore, the Chao1 and Shannon indices were used to assess soil microbial richness and diversity in the tea plantations under different fertilization regimens. Among the various treatments, OPT+Mg exhibited the highest bacterial and fungal Chao1 indices. The bacterial and fungal Chao1 indices under OPT+Mg were higher than those under FFP and OPT (p < 0.05), which showed almost similar values (Fig. 3a, b). Meanwhile, the highest Shannon index for the bacteria was observed under OPT+Mg, followed by FFP and OPT. However, the treatments resulted in no significant change in the fungal Shannon index (Fig. 3d, e).
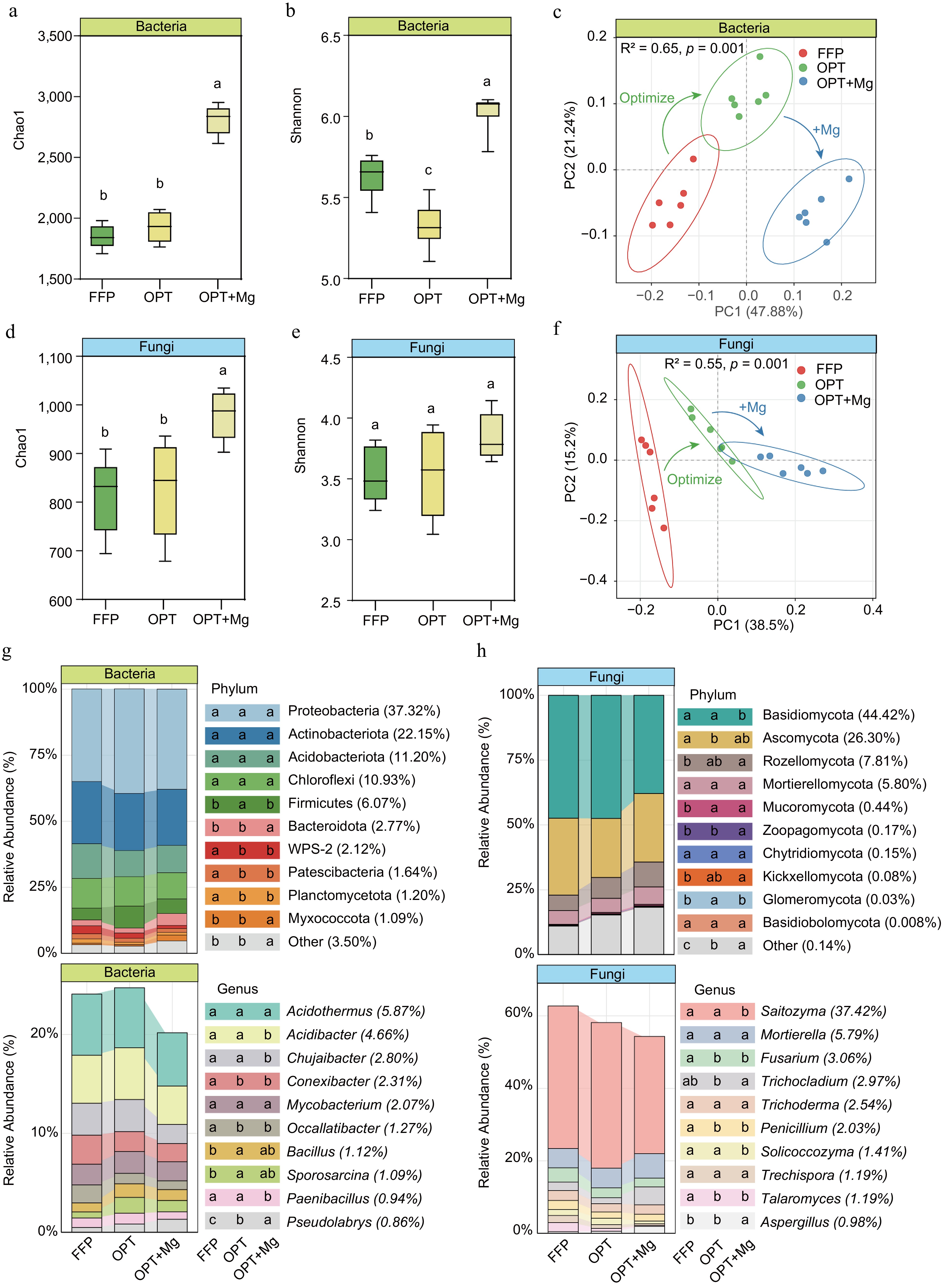
Figure 3.
Soil microbial richness and diversity in Anji Baicha plantations under three fertilization regimens. (a), (d) Chao1 richness estimators. (b), (e) Shannon diversity indices of the bacterial and fungal communities, respectively. (c), (f) PCoA plots (Bray-Curtis distance) showing OTU-level composition of bacterial and fungal communities. (g), (h) The top ten taxonomic groups with the highest relative abundance at the bacterial and fungal phylum and genus levels.
Meanwhile, principal coordinates analysis (PCoA) with Bray-Curtis dissimilarity revealed clear differences in microbial community structure among the fertilization regimes. The first two PCoA axes accounted for 69.12% and 53.7% of the variance in bacterial and fungal assemblages, respectively. PERMANOVA proved that fertilization significantly influenced both bacterial (R2 = 0.65, p < 0.001) and fungal (R2 = 0.55, p < 0.001) community composition. In the ordination plots, the OPT samples were positioned between the FFP and OPT+Mg clusters (Fig. 3c, f), indicating progressive microbial succession under optimized fertilization.
Subsequently, the microbial community profile of the tea plantation soil was analyzed (Fig. 3g, h). The dominant bacterial phyla (accounting for > 10% of the mean relative abundance) were Proteobacteria (37.32%), Actinobacteriota (22.15%), Acidobacteriota (11.20%), and Chloroflexi (10.93%), which together constituted 82% of the total bacterial community (Fig. 3g). Notably, these dominant bacterial groups exhibited similar relative abundances across the three treatments, while the less abundant bacterial taxa (1%–6%) showed significant shifts. In the fungal community, the predominant phyla were Basidiomycota (44.42%), Ascomycota (26.30%), Rozellomycota (7.81%), and Mortierellomycota (5.80%), collectively accounting for 84% of the sequences. In contrast to the bacterial community, both dominant and rare phyla of the fungal community exhibited significant differences in relative abundance across fertilization regimens (p < 0.05).
Furthermore, to determine the soil microbial biomarkers at the genus level under FFP, OPT, and OPT+Mg regimens, RF analysis was conducted. The analysis identified the top ten bacterial and fungal genera as biomarker taxa and revealed that these taxa were characterized by low abundance. Subsequent comparisons among the fertilization regimens revealed that most microbial biomarkers were significantly enriched in the OPT+Mg-treated soil (p < 0.001). Specifically, 80% of the bacterial biomarkers and 60% of the fungal biomarkers were detected in the soil under OPT+Mg treatment (Fig. 4).
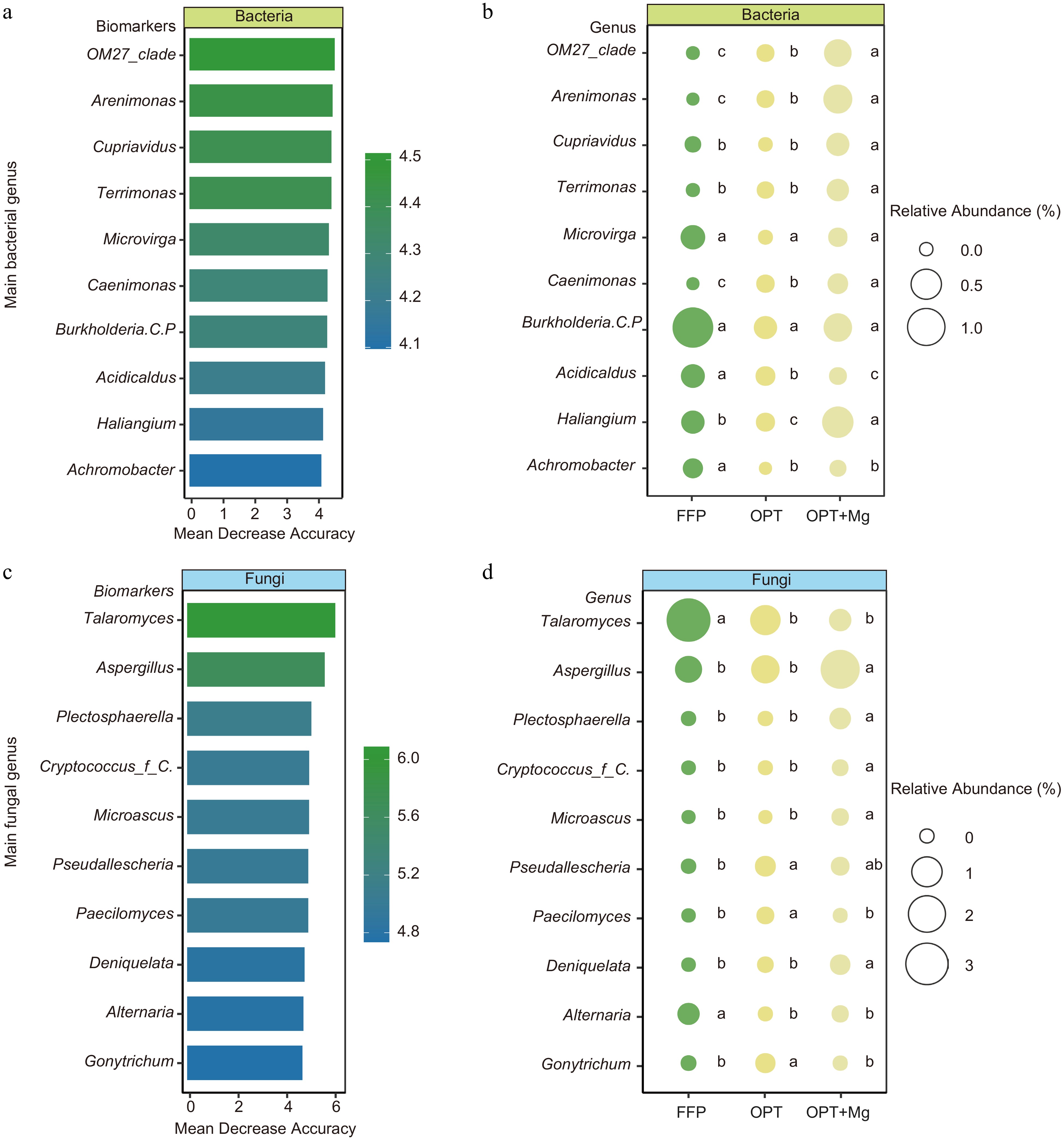
Figure 4.
Identification of microbial biomarkers in Anji Baicha tea plantation soils under different fertilization regimens. The top ten key (a) bacterial, and (c) fungal biomarker genera across the different fertilization regimens. RF regression model based on the relative abundance identified the key microbial genera. The relative abundances of the top ten (b) bacterial, and (d) fungal genera identified as biomarkers in different treatments.
Soil microbial co-occurrence networks
-
The co-occurrence networks based on Spearman correlation ranks revealed the microbial interaction patterns across the fertilization regimens (Fig. 5). Among the bacterial networks, the OPT+Mg network had the most nodes (OTUs) and edges (interactions). The OPT+Mg network had 44.9%−52.7% more nodes than the FFP and OPT networks. Specifically, the bacterial network under OPT+Mg had 18.99%−21.87% more positive (cooperation) links but 19.53%−22.37% fewer negative (competition) links than the networks under FFP and OPT (Fig. 5a−c). Meanwhile, the fungal co-occurrence networks were simpler with fewer nodes and edges under the different fertilization regimens. The fungal networks under the two optimized fertilization treatments had fewer nodes and edges than the network under FFP (Fig. 5d−f). These observations suggest that Mg addition enhanced the positive interactions among the soil bacteria in tea plantations, while having a relatively minor impact on fungi.
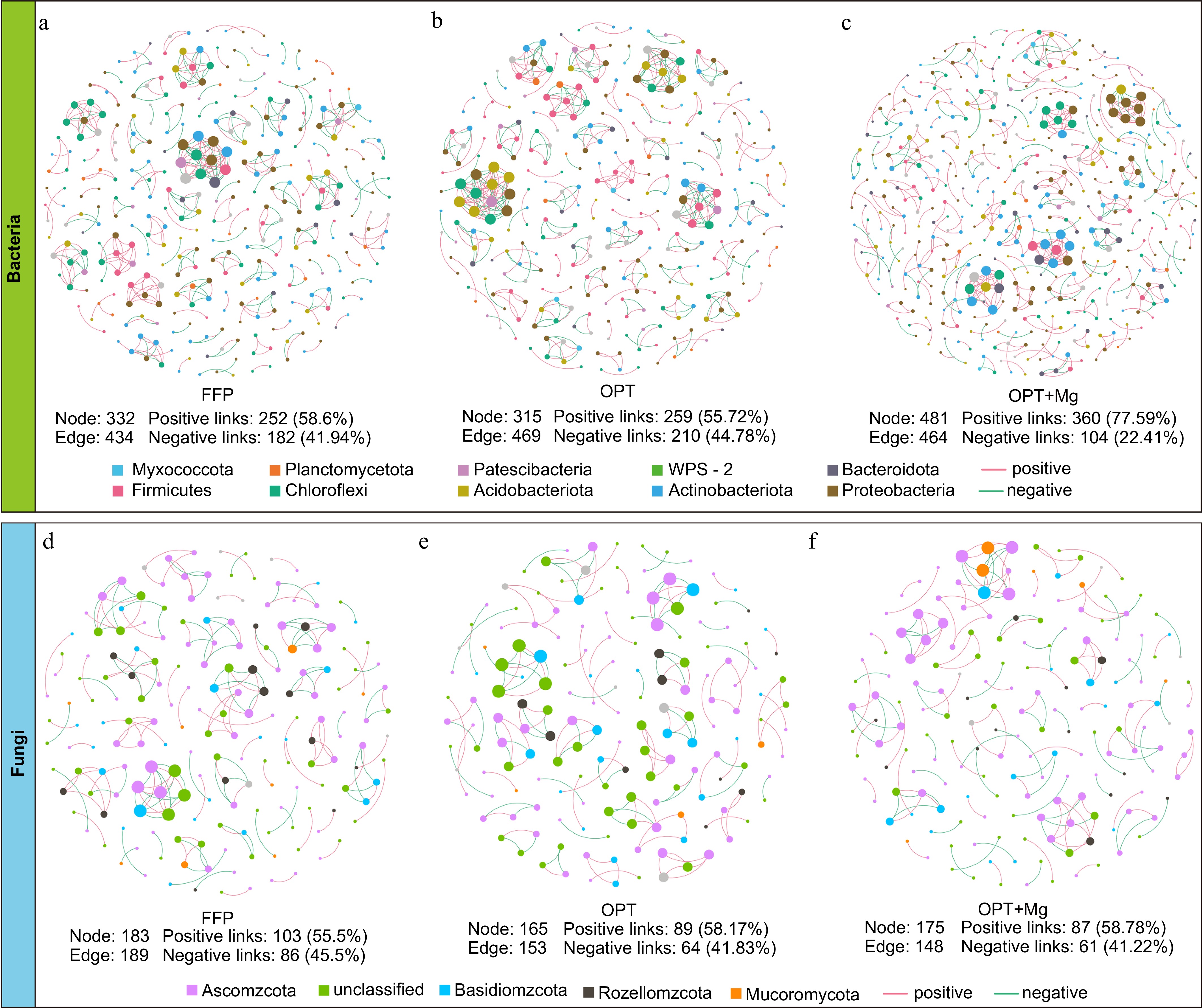
Figure 5.
Soil microbial co-occurrence networks across fertilization regimens in tea plantations. (a)−(c) Bacterial, and (d)−(f) fungal OTU networks. Here, nodes are color-coded based on phylum, and node size is proportional to OTU relative abundance. The edges represent significant correlations (Spearman's |ρ| > 0.6; FDR-adjusted p < 0.05): red indicates the positive correlations, while green indicates the negative correlations. Networks were generated using OTUs with average relative abundance > 0.01% and occurrence frequency > 20%.
Relationships between the microbial community and the variables of the soil
-
Further Pearson's correlation analysis and Mantel test indicated a strong and significant correlation between the soil bacterial community and soil nutrients, suggesting that soil physicochemical properties play a dominant role in shaping bacterial composition. Bacterial diversity was positively correlated with most measured factors, except for SOC, soil N, P, Fe, and Cu contents (Fig. 6a). In contrast, the fungal community was positively associated with most soil physicochemical variables but not with available P. However, fungal diversity was positively correlated with soil pH, K, Ca, Mg, Al, and Mn (Fig. 6b). The heatmap based on correlation coefficients also revealed significant interrelationships among soil physicochemical properties.
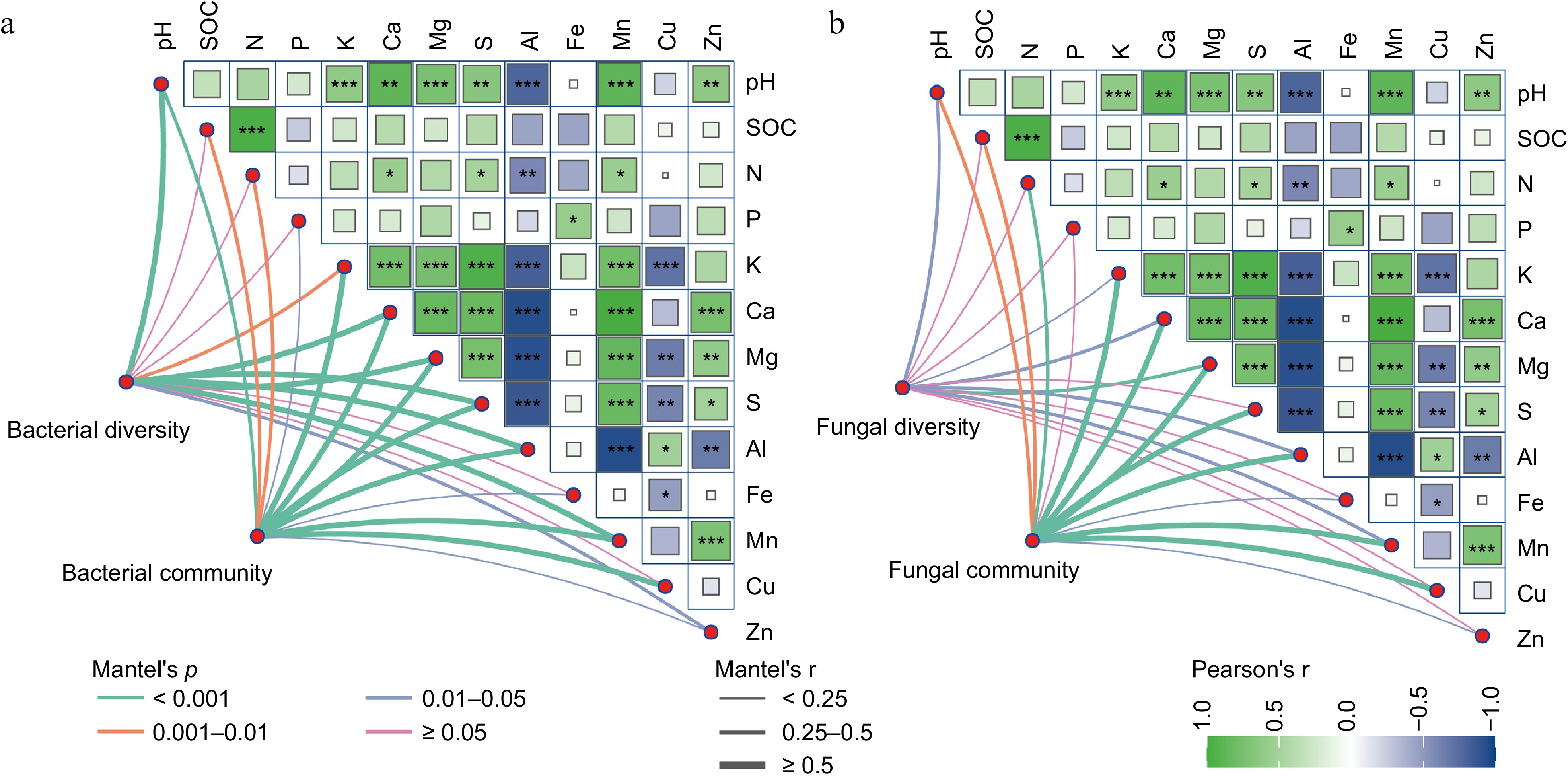
Figure 6.
Pearson correlation heatmap and Mantel test reveal the relationships between soil microbial communities and available nutrients across fertilization treatments. The relations for the (a) bacterial, and (b) fungal communities are shown. In the figure, Pearson correlation coefficients are shown, with green indicating positive correlations and blue indicating negative correlations; * p < 0.05, ** p < 0.01, *** p < 0.001. The Mantel test was used to characterize the correlations; the p-value indicates the strength of the correlation. Green lines represent p < 0.001, orange lines mean 0.001 < p < 0.01, and blue lines mean 0.01 < p < 0.05; the thickness of the lines represents the r values of the Mantel test.
Identification of soil nutrient and microbial variables crucial for predicting tea yield and quality
-
The RF modeling was further carried out to identify the important soil nutrients and microbial properties influencing tea yield and quality (shoot AA content). First, the yield and quality models were generated using the bacterial and fungal biomarker taxa as the independent variables (Fig. 4). The results demonstrated that soil bacterial and fungal biomarkers could predict the tea yield (R2 = 0.44, p = 0.003) and quality (R2 = 0.45, p < 0.001) in the plantations under different fertilization regimens.
Subsequent pie chart analysis indicated that 57% and 43% of the changes in tea yield were due to soil bacterial and fungal biomarkers, respectively. Burkholderia, Caenimonas, Arenimonas, Terrimonas, and Acidicaldus were identified as the bacterial biomarkers crucial for predicting the yield, while Deniquelata and Cryptococcus were identified as the fungal biomarkers. Besides, half of the soil bacterial biomarkers analyzed in this study were identified as crucial (Fig. 7a).
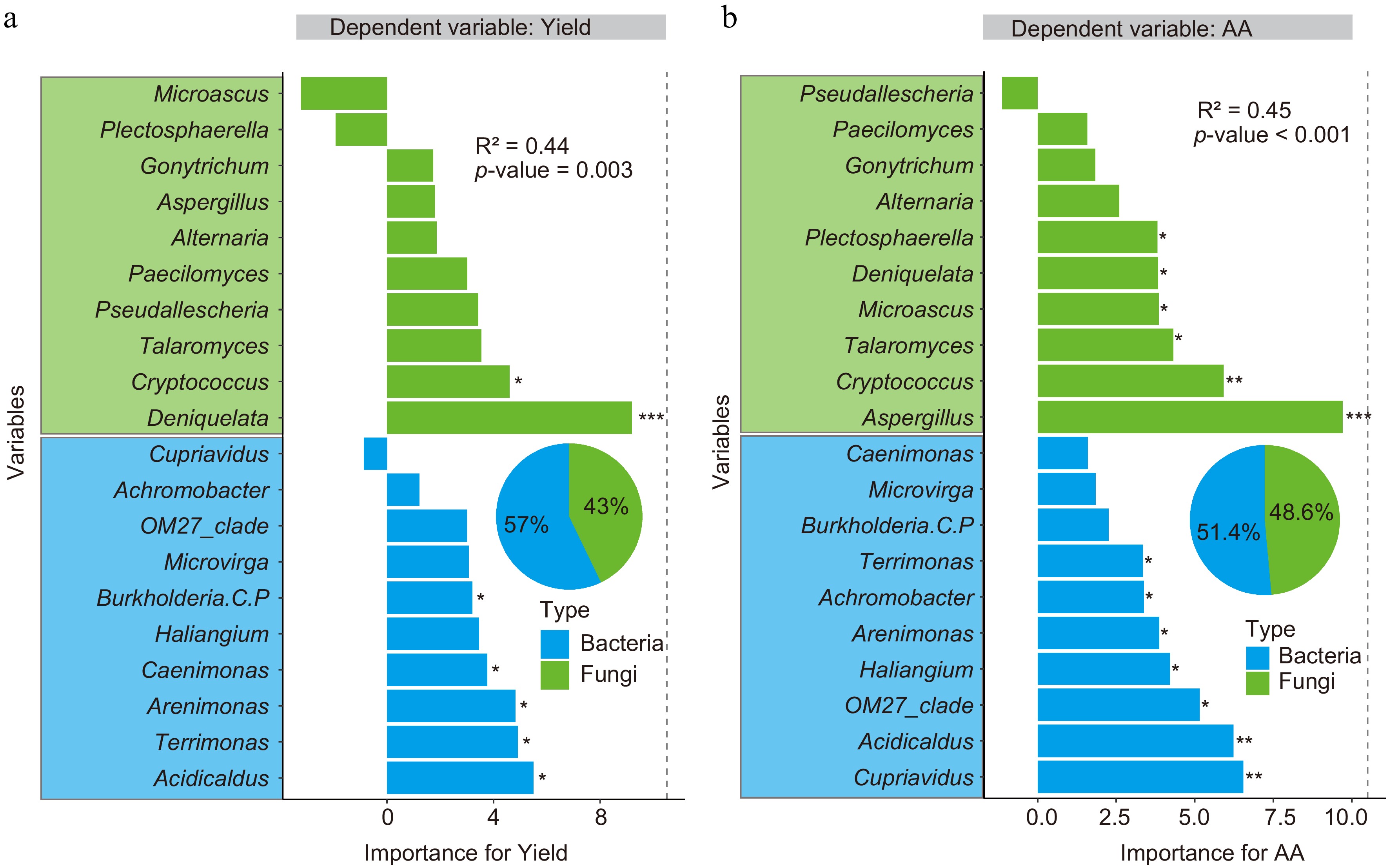
Figure 7.
Random forest analysis identifies the key variables affecting the Anji Baicha (a) yield, and (b) AA content. The genus-level bacteria and fungi are considered biomarkers (Fig. 4). The pie chart shows the relative influence of the bacterial and fungal (genus-level) biomarkers on yield and quality. Here, * p < 0.05, ** p < 0.01, and *** p < 0.001.
The study also indicated that soil bacterial (51.4%) and fungal (48.6%) biomarkers significantly contributed to the variations in tea quality. Cupriavidus, Acidicaldus, OM27-clade, Haliangium, Arenimonas, Achromobacter, and Terrimonas were identified as the critical bacterial biomarkers largely contributing to the changes in tea quality, while Aspergillus, Cryptococcus, Talaromyces, Microascus, Deniquelata, and Plectosphaerella were identified as the crucial fungal biomarkers (Fig. 7b). Overall, the present study identified that four soil bacteria (Acidicaldus, Terrimonas, Arenimonas, and Haliangium) and one soil fungus (Cryptococcus) influenced both tea yield and quality.
-
The rational application of N-P-K fertilizers significantly increased the yield of Anji Baicha tea by 22% compared with farmers' fertilization practice (FFP), highlighting the role of balanced fertilization in improving tea productivity. However, no further significant improvement in yield was observed when Mg was added under optimized N-P-K conditions. Studies have established a positive correlation between soil N levels and tea yield and AA content[21]. In contrast, excessive P and K fertilization has been linked to decreased relative chlorophyll content and reduced levels of free AAs, such as theanine and glutamic acid, in tea leaves[25]. Thus, these findings, combined with earlier reports, suggest that a rational fertilization strategy is essential for enhancing yield while preserving the quality of Anji Baicha.
The optimized N-P-K treatment of the present study significantly increased AA content in new tea shoots. Supplementing Mg under optimized N-P-K fertilization further promoted AA accumulation (Fig. 1b, e). Anji Baicha has high economic value due to its white color characteristic. Notably, N and Mg application did not increase chlorophyll content in Anji Baicha. These observations suggest that the unique characteristics of Anji Baicha, possibly involving genetic constraints in chlorophyll synthesis, allowed the tea to remain white and retain high economic value.
In tea plants, roots mediate N uptake and assimilation. The N is stored and transported primarily in the form of AA, like theanine and arginine[30−32]. Theanine, predominantly synthesized in roots during winter and translocated to new shoots in spring[30], represents a key N reservoir. In the present study, optimized fertilization significantly increased both root N and free theanine content in Anji Baicha (Supplementary Fig. S2). However, Mg supplementation under the optimized N-P-K fertilization regimen reduced theanine accumulation in roots. Research has indicated that the application of optimal levels of Mg to the soil facilitates theanine transport from roots to new shoots[17,18]. Therefore, a divergence was observed in theanine accumulation between roots and new shoots under different fertilization regimens (Fig. 1; Supplementary Fig. S2). It was also found that optimal N-P-K increased AA synthesis, while Mg supply enhanced its transport. These findings prove that rational fertilization boosts yield and enhances tea quality while preserving leaf color.
Effects of optimized soil fertilization on soil quality and microbial characteristics
-
This study found that the OPT+Mg regimen alleviated soil acidification in Anji Baicha plantations (Table 1). Consistent with this observation, the OPT+Mg treatment significantly reduced available Al, indicating a decline in soil acidity[33]. Besides, both OPT and OPT+Mg increased Ca, Mg, and K in the soil, which play critical roles in increasing soil pH[34]. Thus, the increase in soil pH with the OPT+Mg treatment likely reduced the loss of cations (K+, Mg2+, and Ca2+), thereby enhancing soil fertility. The analysis of SQI corroborated these findings (Table 1).
Research has established that fertilization significantly shapes the microbial communities of the soil. Optimizing the N-P-K ratio altered bacterial and fungal diversity in the tea plantation soil, with Mg supplementation amplifying this effect (Fig. 4). These findings are consistent with those of Yang et al., who found that Mg enhances bacterial α-diversity and network complexity but reduces fungal connectivity[35]. Given the established links between microbial diversity and nutrient cycling[36], OPT and OPT+Mg regimens modified soil chemistry and restructured the soil microbial communities[37].
Consistent with prior findings[38], Proteobacteria, Actinobacteria, and Acidobacteria dominated the soil bacterial community in the tea plantation, while Basidiomycota and Ascomycota prevailed among fungi. Though phylum-level composition remained stable, the relative abundance of the microbes shifted significantly across the treatments (Fig. 3g). The RF analysis identified ten low-abundance bacterial genera[39], with differential abundance patterns across the regimens, as biomarkers of these soils (Fig. 4). These biomarkers are believed to positively regulate the yield and quality of Anji Baicha tea.
Key drivers of yield and quality
-
Pearson correlation analysis with the Mantel test revealed a direct association between the soil microbial community (abundance and diversity) and soil physicochemical properties (Fig. 6). The soil microorganisms contribute to organic matter transformation and nutrient cycling and modify the soil habitat through various biochemical and biophysical processes[40]. These activities indirectly influence tea yield and quality. In this study, the application of OPT+Mg increased soil pH, leading to the accumulation of Ca and K. Generally, a soil pH of 5.32 is optimal for soil nutrient availability and good tea quality[41], whereas a slightly lower pH (around 5) favors tea plant growth and nutrient accumulation[42]. In other words, an appropriate soil pH is crucial for improving tea quality. Moreover, Yang et al. reported that Mg fertilization enriches beneficial microbes involved in N fixation and P mineralization[35].
Further, RF analysis identified soil microbial communities as key factors affecting the yield and quality of Anji Baicha tea (Fig. 7a). Among them, bacterial communities played a dominant role. Notably, the genus Burkholderia, known for promoting plant growth, nitrogen fixation, phosphorus solubilization, and stress resistance[43], was identified as an important microbial group. Integrating these findings with results, Mg fertilization likely attracted beneficial microorganisms such as Burkholderia and thereby increased Anji Baicha yield.
Both bacterial and fungal communities were found to influence the amino acid (AA) content of tea shoots (Fig. 7b). Cupriavidus metallidurans has been shown to enhance seed germination and growth of maize, wheat, and tomato[44]. Thus, findings indicate that optimizing the N-P-K ratio in tea plantations not only improved shoot yield and quality but also enhanced soil quality by regulating nutrient cycling. The addition of Mg under optimized conditions increased soil pH and recruited more beneficial microbes, ultimately improving both soil health and tea quality. In summary, research findings indicate that optimizing the N-P-K ratio and supplementing Mg are of great significance for enhancing tea quality and supporting sustainable agricultural practices.
Research limitations
-
The present study demonstrated that Mg supplementation enhances the positive effects of optimized N-P-K fertilization on the yield and quality of the albino tea cultivar. However, the experiment was conducted only in a specific geographic area and over a relatively short timeframe, potentially limiting the strategy's applicability. Future studies should investigate the differences in the effects of these fertilization regimens across diverse locations and plantations. Moreover, the dose-dependent effect of Mg remains unclear. Researchers should also perform experiments with synthetic microbial communities to establish the relationships between microbial communities and tea quality. A detailed investigation considering these aspects will help elucidate the precise mechanism underlying the observed differences in AA accumulation between roots and new shoots in tea plants under different treatments.
-
The present study developed an effective fertilization regimen, involving an optimal N-P-K ratio and Mg supply, for Anji Baicha plantations. This regimen enhanced Anji Baicha tea yield and quality without affecting the leaf albinism trait. Detailed analysis revealed that the changes in the soil nutrient profile under optimized fertilization restructured the soil microbial communities and their interactions. The optimized NPK regimen primarily modulated the low-abundance bacterial taxa, while Mg supplementation synergistically enhanced these effects. These findings prove that integrating customized fertilization with Mg supply is a promising strategy for tea. The study also provides a solid framework for precision fertilization in tea plantations.
This research was supported by the Yunnan Provincial Special Fund for the Construction of a Science and Technology Innovation Base (Grant No. 202402AE090015), the Zhejiang Provincial Natural Science Foundation of China (Grant No. LY23C150008), the National Key Research and Development Program of China (Grant No. 2021YFD1601100), the China Agriculture Research System of MOF and MARA (Grant No. CARS-19), and the National Natural Science Foundation of China (Grant Nos 42407459, 32172634).
-
The authors confirm their contributions to the paper as follows: study conception and design: Yang X, Ma L, Ruan J, Ni K, Shi Y, Long L; experiments performing and data analysis: Yan X, Yang X, Xue J, Huo K; draft manuscript preparation: Yan X. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets generated during and/or analyzed in the current study are available from the corresponding author on reasonable request.
-
The authors declare that they have no conflict of interest.
- Supplementary Table S1 Soil fertilization regimens.
- Supplementary Fig. S1 Tea growth under different treatments.
- Supplementary Fig. S2 Contents of AA and theanine in tea plant roots.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Yan X, Yang X, Ni K, Shi Y, Long L, et al. 2026. Optimizing N-P-K ratios with magnesium supply improves tea yield and quality primarily by modulating soil bacterial communities in an albino tea plantation. Beverage Plant Research 6: e009 doi: 10.48130/bpr-0025-0043
Optimizing N-P-K ratios with magnesium supply improves tea yield and quality primarily by modulating soil bacterial communities in an albino tea plantation
- Received: 10 November 2025
- Revised: 12 December 2025
- Accepted: 16 December 2025
- Published online: 13 March 2026
Abstract: Anji Baicha is produced from 'Baiye 1', which is an albino cultivar of tea (Camellia sinensis). It is highly prized for its unique characteristic of accumulating high levels of free amino acids, especially theanine. Conventional fertilization practices used to maintain leaf albinism often compromise yield and quality. Therefore, the present study used an optimized fertilization strategy, combining adjusted N-P-K ratios with Mg supplementation, to enhance plant nutrition while preserving the desired albinism feature. Field experiments were carried out to compare farmers' practice (FFP), optimized N-P-K (OPT), and OPT+Mg. Compared with FFP, OPT, and OPT+Mg treatment significantly increased tea yield by 22%–29% and the amino acid (including theanine) content in new shoots (p > 0.05), while successfully maintaining leaf albinism. They also improved soil nutrient availability, microbial diversity, and enhanced soil quality by 19.4%–75%. Both OPT and OPT+Mg treatments reshaped soil bacterial and fungal communities, enriching specific beneficial microbial taxa. Random Forest predictive analysis indicated that bacterial biomarkers exerted a greater influence on tea yield and amino acid content than fungal biomarkers, accounting for 57% and 51.4% of the predicted variance for yield and amino acids, respectively (compared to 43% and 48.6% for fungi). Acidicaldus and Deniquelata were identified as key biomarkers for yield, while Cupriavidus and Aspergillus were key biomarkers associated with variation in amino acid content. Thus, findings demonstrate that adequate N fertilization is crucial for optimal growth of this albino tea cultivar, and Mg supplementation under optimal N supply provides synergistic advantages. The study establishes a scientific framework for the sustainable production of Anji Baicha.














