-
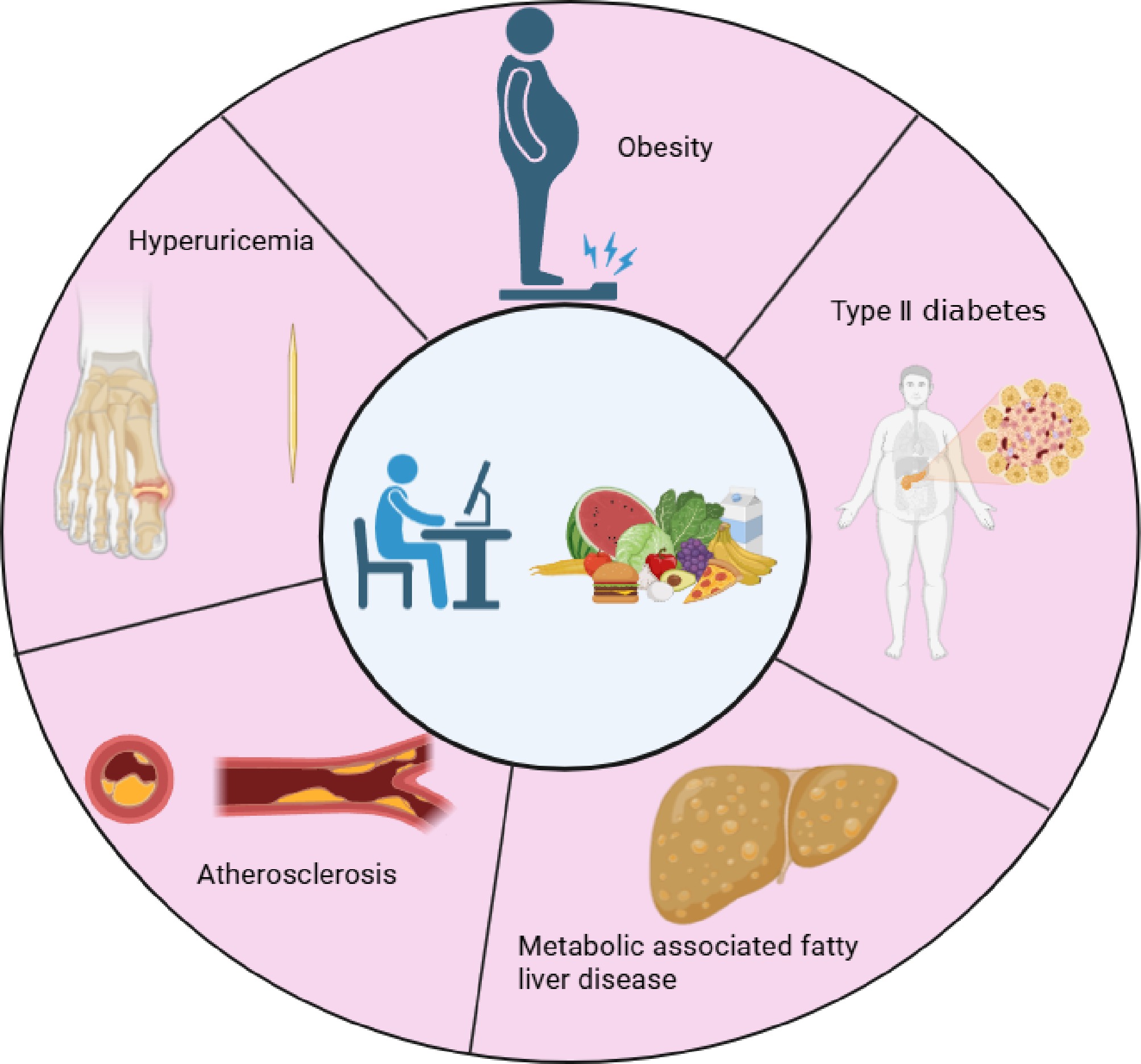
The most prevalent chronic metabolic diseases (CMDs), including Type 2 diabetes mellitus (T2DM), obesity, metabolic dysfunction-associated steatotic liver disease (MASLD), hyperuricemia, and cardiovascular diseases, are nutrition- and exercise-related disorders that remain clinically challenging to cure effectively (Fig. 1)[1]. As the pathogenesis of various CMDs shares common etiological factors related to dietary patterns and physical inactivity, significant interconnections exist among these conditions. T2DM primarily arises from insulin resistance caused by dysregulation of glucose and lipid metabolism, accompanied by characteristic manifestations such as hyperinsulinemia, dyslipidemia, hyperuricemia, and MASLD[2]. Similarly, obesity develops through surplus energy and excessive lipid accumulation resulting from imbalanced energy intake and expenditure[3]. Epidemiological data indicate that since the 21st century, mortality and disability rates associated with common CMDs have surged dramatically, with disease burdens showing continuous escalation[4]. These findings underscore the critical need for coordinated global health initiatives to counteract the rising trends of these debilitating disorders and improve population health outcomes worldwide.
As one of the most widely consumed beverages globally, tea, made from young shoots and leaves of Camellia sinensis, is classified into six major categories depending on the degree of fermentation and processing techniques: green tea, white tea, yellow tea, oolong tea, black tea, and dark tea[5]. Tea leaves contain diverse bioactive compounds, including polyphenols, alkaloids, and amino acids, with distinct compositional profiles across tea varieties resulting from different manufacturing processes and fermentation levels[6]. Both whole tea preparations and their bioactive constituents demonstrate significant potential in modulating the development and progression of T2DM[7]. Accumulating evidence indicates that green tea extracts effectively regulate blood glucose levels and ameliorate insulin resistance in T2DM patients[8]. Beyond nonfermented teas, fermented varieties such as black tea and dark tea also exhibit notable hypoglycemic effects[9]. Among tea's components, catechins and their oxidative polymers are recognized as principal bioactive mediators[10]. This review systematically examines the current research on the therapeutic mechanisms of tea catechins and their oxidative polymers against CMDs, summarizes their demonstrated efficacy, and discusses potential clinical applications.
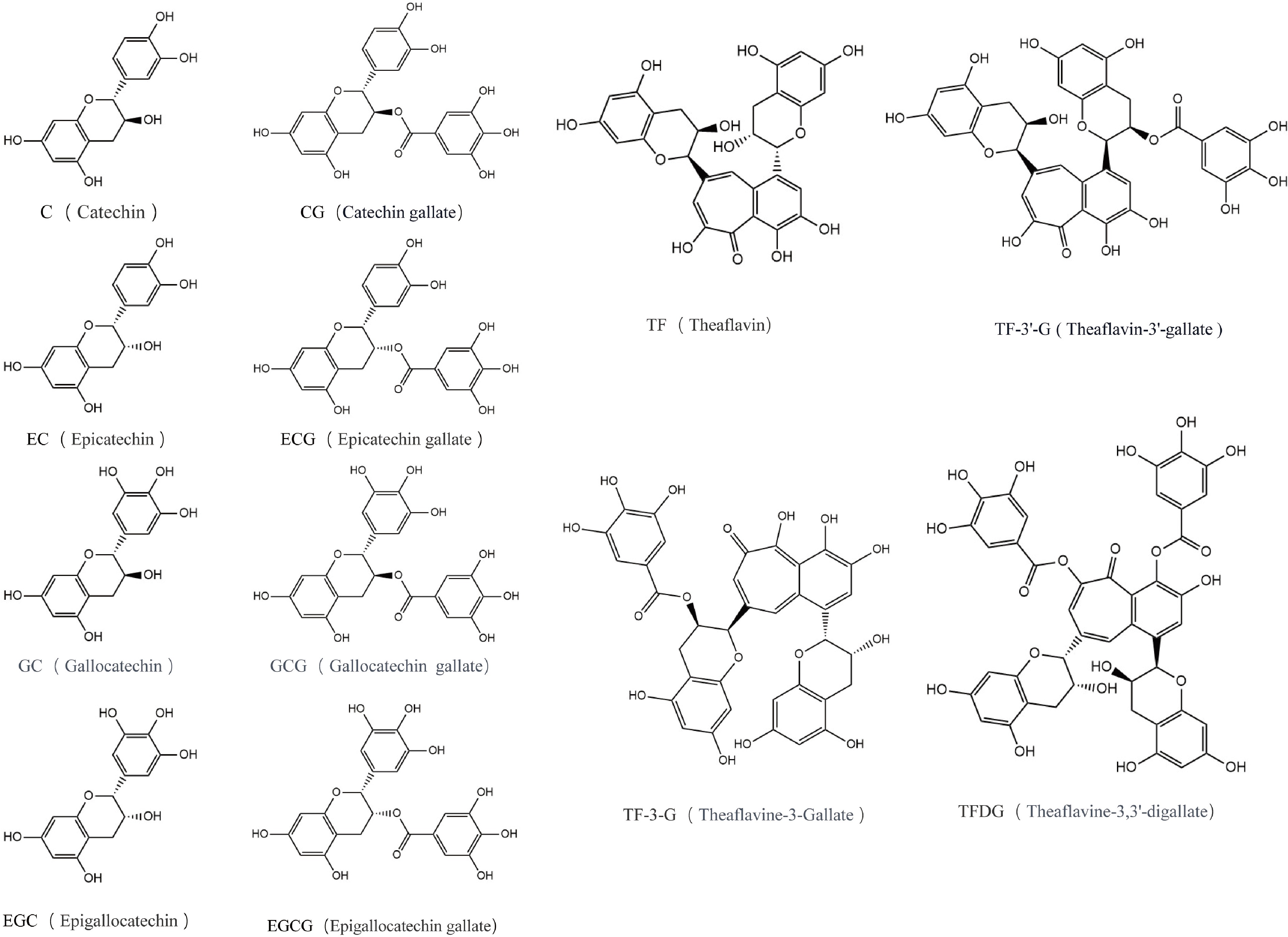
The characteristic flavor profile and therapeutic properties of tea originate from its diverse phytochemical composition. Polyphenolic compounds, particularly epigallocatechin gallate (EGCG), epigallocatechin (EGC), epicatechin gallate (ECG), and epicatechin (EC), constitute the predominant bioactive constituents, demonstrating multifunctional physiological activities, including antioxidant and anti-inflammatory effects[11]. Through enzymatic catalysis by polyphenol oxidase, these catechins undergo oxidative polymerization during tea processing, generating complex derivatives such as theaflavins, thearubigins, and theabrownins (Fig. 2)[12]. These oxidation-derived polymers not only determine the organoleptic characteristics of tea products (color and taste) but also exhibit diverse bioactivities. It is noteworthy that "tea polyphenols" is a broad term encompassing a class of phenolic compounds in tea, including flavonols, phenolic acids, and anthocyanins, in addition to the most abundant flavan-3-ols (catechins). However, this review will primarily focus on the catechins (e.g., EGCG, EGC, ECG, EC) and their enzymatic oxidation-derived polymers (e.g., theaflavins, thearubigins, theabrownins), as they represent the most predominant and well-characterized fraction responsible for the observed metabolic benefits discussed herein. This focused scope allows for a detailed and coherent examination of their specific pharmacology and mechanisms. Mechanistic studies have revealed that both catechins and their oxidative metabolites possess unique structural configurations enabling therapeutic effects against various chronic metabolic disorders through multiple pharmacological pathways.
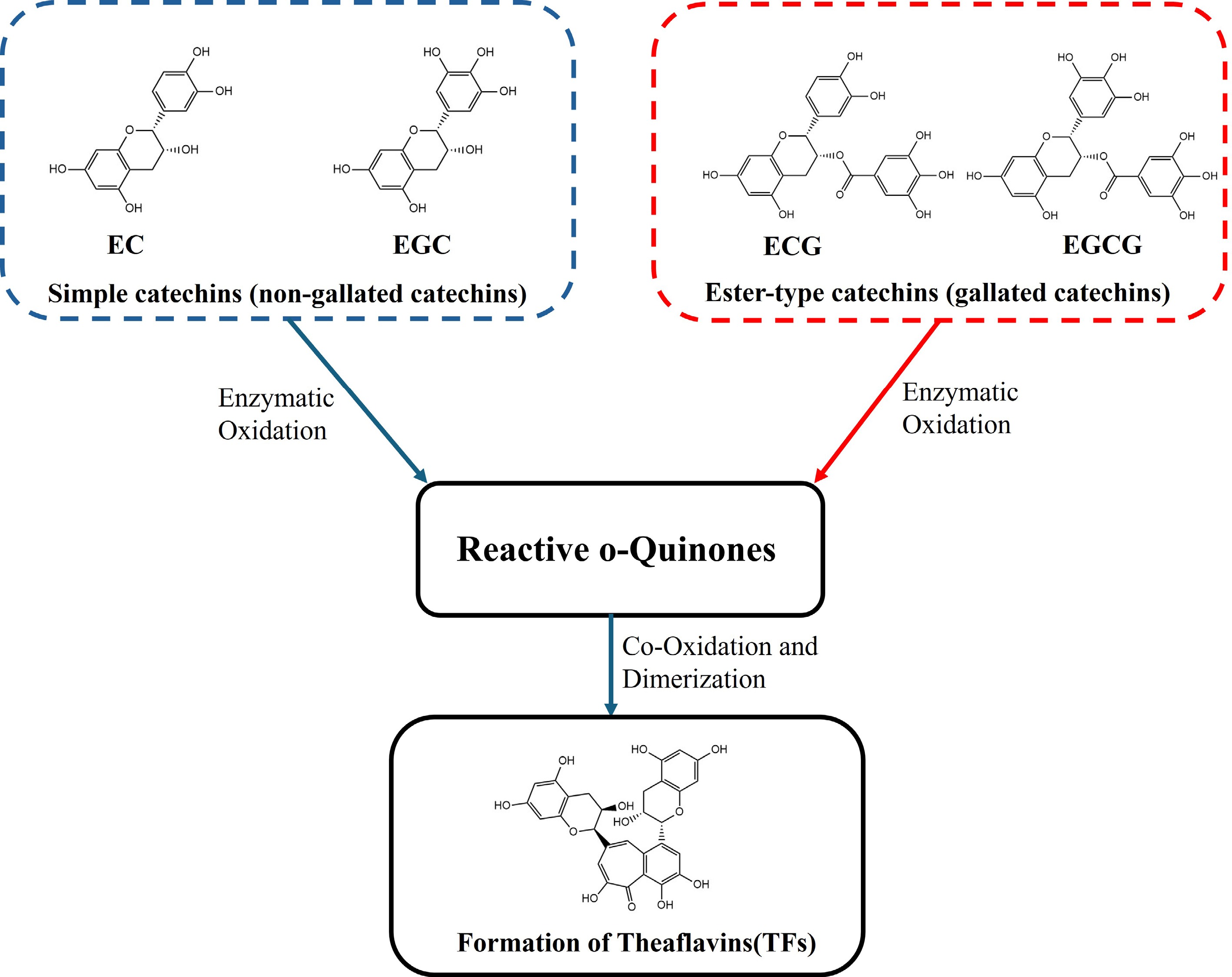
The characteristic flavor and bioactivity of different tea types are largely determined by the transformation of these native catechins during processing. As illustrated in Fig. 3, the enzymatic oxidation of catechins by polyphenol oxidase initiates a cascade of polymerization reactions. This process generates a spectrum of oxidized tea polyphenols (OTPs), starting with the formation of theaflavins from specific pairwise combinations of catechins, followed by further oxidation to thearubigins and eventually theabrownins. This transformation pathway is fundamental to understanding the distinct compositional profiles and associated health effects of semifermented (e.g., oolong tea) and fully fermented teas (e.g., black tea and dark tea) compared with nonfermented green tea.
-
T2DM serves as a prime example of the shared pharmacological framework, with tea catechins and their polymers addressing its core pathophysiologies of insulin resistance, inflammation, and gut dysbiosis. As a global chronic metabolic disorder[13], T2DM demonstrates escalating prevalence worldwide. Current preventive and therapeutic strategies primarily target the fundamental pathophysiological feature of T2DM: dysregulation of glucose and lipid metabolism. Lifestyle interventions involving dietary modification and physical exercise remain cornerstone approaches for the prevention and management of T2DM[14]. For patients progressing to intermediate or advanced stages, pharmacological interventions become essential, with mainstream therapeutics including insulin analogs, biguanides, statins, SGLT2 inhibitors, DPP-4 inhibitors, and GLP-1 receptor agonists. These agents ameliorate hyperglycemia through three principal mechanisms: enhancing insulin secretion, improving insulin sensitivity, and suppressing postprandial glucose elevation. However, long-term administration of these medications may induce adverse effects such as hypoglycemia, lactic acidosis, urinary tract infections, and allergic reactions[15]. In contrast, bioactive components derived from traditional herbal medicine and functional foods have gained significant research attention because of their multitarget mechanisms and favorable safety profiles. Emerging evidence indicates that puerarin from Pueraria lobata regulates mitochondrial function through phosphorylation of adenosine monophosphate-activated protein kinase (AMPK) to exert hypoglycemic effects[16]. These findings exemplify the growing exploration of natural compounds as complementary therapeutic agents against T2DM pathogenesis.
Tea catechins and their oxidative polymers demonstrate multi-target therapeutic potential against T2DM through diverse bioactive mechanisms (Table 1)[17]. Experimental studies have revealed that aqueous extracts of green tea that are rich in catechins significantly enhance insulin sensitivity in insulin-resistant murine models, with catechins identified as the principal bioactive mediators[18]. The therapeutic efficacy of α-glucosidase inhibitors in managing diabetes has been established as an effective strategy for controlling postprandial hyperglycemia and its pathological complications[19]. In vitro investigations demonstrate that various green tea catechins act as potent α-glucosidase inhibitors, effectively suppressing enzymatic activity to delay intestinal glucose absorption and mitigate postprandial glycemic spikes[20]. Beyond total catechin complexes, individual catechin monomers, including EGCG, EGC, and ECG, exhibit distinct hypoglycemic properties. Tiantian Zhu et al. reported that administering EGCG ameliorates β-cell dysfunction in T2DM models, thereby improving systemic insulin resistance[21]. Chronic hyperglycemia-induced mitochondrial dysfunction in pancreatic β-cells was shown to be counteracted by EGCG through dose-dependent modulation of the expression of Dynamin-related protein 1 (DRP1), facilitating the restoration of mitochondrial membrane potential and attenuation of apoptosis[22]. Furthermore, EGCG exhibits NLRP3 inflammasome inhibitory activity, suppressing inflammatory pathways and enhancing glucose tolerance[23]. The gut microbiota's pivotal role in modulating glucolipid metabolism has been mechanistically linked to (-)-epicatechin (EC) bioactivity. Experimental evidence confirms that administration of EC remodels the gut microbiota composition in Goto-Kakizaki (GK) rats, suppresses lipopolysaccharide (LPS) production, and enhances hepatic insulin signaling transduction, collectively contributing to mitigating T2DM[24]. These cumulative findings substantiate the multimechanistic hypoglycemic effects of tea catechins through insulin resistance amelioration pathways.
Table 1. Pharmacology of tea catechins and their polymers.
Diseases Specific compound/
extract usedActivity/mechanism(s) of action Cell lines/model Dose Application (route of amelioration) Reference T2DM Green tea polyphenols and different tea types Inhibiting α-glucosidase activity 2.33 µg/mL; 0.25–0.016 mg/mL [20] EGCG Reduces blood glucose and improves insulin resistance and islet β-cell disorder High-sucrose high-fat diet (HFD) with streptozotocin-induced diabetic rats 25, 50,
100 mg/kg/dIn vivo (oral gavage) [21] EGCG Alleviates high glucose-induced pancreatic β-cell dysfunction by targeting the DRP1-related mitochondrial apoptosis pathway MIN6 cells 10, 20, 40 μM In vitro [22] EGCG Inhibitor of NLRP3 inflammasome activation; improves glucose tolerance Mouse bone marrow-derived macrophages; HFD mice 20, 30, 40 μM;
50 mg/kgIn vitro; in vivo (oral gavage) [23] (-)-Epicatechin Modulates the gut microbiota and liver insulin signaling pathways HFD rats 40, 80 mg/kg In vivo (oral gavage) [24] EGCG-derived oxidation products Activation of the beneficial axis of RAS and suppression of the deleterious axis of RAS, SELENOP, TXNIP, or renal PEPCK/G6Pase-α db/db mice 5, 10, 40 mg/kg In vivo (intraperitoneal injection) [25] Theaflavins (TFs) Promoting mitochondrial biogenesis and improving the hepatocellular insulin resistance induced by free fatty acids HepG2 cells 2.5, 5, 10 μg/mL In vitro [26] Theabrownin (TB) from Fu Brick tea Altering the gut microbiota and improving intestinal tight junction integrity HFD mice 100, 200,
400 mg/kgIn vivo (oral gavage) [28] Obesity Green tea polyphenols Changing the composition and diversity of colonic microbiota HFD mice Diet supplemented with 0.05, 0.2, 0.8% In vivo [42] Green tea polyphenol extract Promoting the induction of thermogenic cells by reprogramming the initial steps of adipocyte commitment Cafeteria diet mice 500 mg/kg In vivo (oral gavage) [43] Green tea polyphenol extract Inducing adaptive thermogenesis and browning in scWAT HFD mice 500 mg/kg In vivo (oral gavage) [44] Green tea polyphenols Inhibition of erk1/2 activation, alleviation of PPARγ phosphorylation, and increased PPARγ expression HFD rats Drinking water supplemented with 0.8, 1.6,
3.2 g/LIn vivo [45] Polyphenol-enriched oolong tea Increases lipid excretion into feces Healthy adult subjects 19.3 ± 12.9 g/3 d In vivo [46] EGCG Increases the expression of hepatic TGR5 and decreases the expression of intestinal FXR-FGF15 HFD rats 40, 160 mg/kg In vivo (oral gavage) [47] EGCG Induces fat deposition by targeting HSP70 through the activation of AMPK–SIRT1–PGC-1α in porcine subcutaneous preadipocytes Porcine subcutaneous preadipocytes 100 μM In vitro [48] EGCG Downregulated MAPK7 mRNA and protein levels time- and dose-dependently 3T3-L1 cells 10, 20, 50 μM In vitro [49] EGCG Upregulation of Beclin1-dependent autophagy and lipid catabolism in WAT HFD mice 20 mg/kg In vivo (oral gavage) [50] EGCG Promotes fat oxidation HFD mice Diet supplemented with 0.5, 1% In vivo [51] Oxidized tea polyphenols (OTPs) Alleviates the accumulation of lipids in liver and visceral white adipose tissue and promotes lipid excretion HFD rats Diet supplemented with 2% In vivo [53] MASLD Green tea extract (GTE) Prevents dietary-induced liver steatosis HFD rats Diet supplemented with 1.1, 2% In vivo [65] EGCG Possesses a Bmal1-dependent efficacy against insulin resistance conditions by strengthening insulin signaling and eliminating oxidative stress HepG2 cells/primary hepatocytes 0, 25, 50, 100 μM In vitro [66] Tea polyphenols and EGCG Promoting acid-producing bacteria HFD rats 200, 400,
800 mg/kgIn vivo (oral gavage) [67] EGCG Decreases bile acid reabsorption HFD mice Diet supplemented with 0.32% In vivo [68] Green tea extract rich in EGCG Activation of AMPK via LKB1 in the liver HFD mice 50 mg/kg In vivo (oral gavage) [70] AS EGCG Mediated by AP-1 inactivation through ERK and JNK Human primary T cells 0.1, 1, 5, 10, 20 μM In vitro [81] (-)-Epicatechin gallate (ECG) Inhibition of the phosphorylation of p65 in the NF-κB pathway in the aorta VSMCs;
HFD mice10, 20, 50 μM;
5, 25, 50 mg/kgIn vitro; in vivo (intraperitoneal Injection) [82] (-)-Epicatechin gallate (ECG) Inhibition of intracellular NF-κB signaling pathway proteins and activation of the HO-1/Nrf2 signaling pathway [83] Tea polyphenols Promoting the proliferation of the intestinal Bifidobacteria HFD mice Drinking water supplemented with 0.4, 0.8,
1.6 g/LIn vivo [84] Green tea, black tea Antioxidant properties of the intervention HFD rabbits 200 mg/kg In vivo [85] Green tea polyphenols Increasing the mRNA and protein expression levels of hepatic PPARα and autophagy markers HFD-fed male ApoE-knockout mice Drinking water supplemented with 3.2, 6.4 g/L In vivo [86] Hyperuricemia Green tea polyphenols Decreasing uric acid production and increasing uric acid excretion PO-induced hyperuricemic mice 300, 600 mg/kg In vivo (oral gavage) [93] EGCG Inhibition of XOD activity and GLUT9 expression and the promotion of OAT1 expression BRL 3A rat liver cells;
PO-induced hyperuricemic mice10, 20, 40 µM;
25, 50, 100 mg/kgIn vitro; in vivo (oral gavage) [95] Fermented tea extracts Inhibition of xanthine oxidase activities LO-2 cells 0.12, 2 mg/mL In vitro [97] Pu-erh tea polyphenols Reshaping the gut microbiota CRD-induced hyperuricemic mice In vivo [98] The enzymatic oxidation of tea catechins by polyphenol oxidase generates OTPs, primarily through oxidative transformation of various catechin derivatives. Accumulating evidence substantiates the glucose-regulatory efficacy of these oxidative polymers in diabetes management. Notably, EGCG, a key catechin component, undergoes auto-oxidation to form EGCG-derived oxidation products. These derivatives alleviate diabetic symptoms by differentially modulating the renin–angiotensin system (RAS) axes, suppressing the detrimental renal RAS axis while activating the beneficial hepatic RAS axis, thereby downregulating the expression of the hepatic and renal selenoprotein P (SELENOP) gene and thioredoxin interacting protein (TXNIP) to enhance insulin sensitivity in db/db diabetic mice[25].
Theaflavins, formed through oxidative polymerization of catechins during tea processing, demonstrate insulin-sensitizing effects via multiple pathways. Experimental studies have revealed their capacity to ameliorate palmitate-induced insulin resistance in HepG2 cells through a mechanistic triad: upregulating the expression of glucose transporter 4 (GLUT4) protein, elevating Akt phosphorylation at Ser473 levels, and reducing IRS-1 phosphorylation at Ser307[26]. Furthermore, theaflavins regulate hepatic glucose metabolism by modulating the gut microbiota–liver axis, enhancing glycogen synthesis while attenuating hepatic lipid accumulation, thereby improving systemic insulin resistance[27]. Theabrownins, representing advanced oxidation products with higher molecular complexity than theaflavins, constitute the major pigmented components in dark tea. Zhongting Lu et al. demonstrated that administration of theabrownins restores the gut barrier;s integrity by remodeling its microbial composition and associated metabolites (L-ornithine, α-ketoglutarate, and glutamine), effectively mitigating high-fat diet-induced insulin resistance and hepatic inflammation[28].
The beneficial effects and underlying mechanisms of tea catechins and their polymers on obesity
-
Obesity, a chronic metabolic disorder stemming from dysregulated energy metabolism, shares global prevalence with T2DM as predominant metabolic diseases. Current antiobesity strategies primarily focus on energy intake restriction and expenditure enhancement, with growing emphasis on pharmacological adjuvants to optimize the therapeutic outcomes[29−31]. Multiple natural bioactive molecules demonstrate significant antiadipogenic effects through the dual mechanisms of inhibiting lipogenesis and stimulating lipolysis. Experimental evidence reveals that D-limonene from citrus essential oils markedly suppresses lipid accumulation in adipocytes while activating the AMPK signaling pathway to enhance fat breakdown[32]. Similarly, the antiobesity activity of ginsenoside Rh2 is mediated through AMPK-dependent pathways[33]. Given obesity's etiology as an overnutrition-related metabolic disorder, modulation of the gut microbiota emerges as a critical therapeutic target[34]. Laminaria japonica-derived fucoidan achieves antiobesity effects by remodeling the gut microbial architecture[35], exemplifying how diverse natural molecules exert beneficial metabolic actions through multitarget mechanisms. Tea contains a diverse array of bioactive compounds, with tea catechins and their polymers constituting the most abundant and pharmacologically active fraction. Although other components, such as caffeine[36], tea polysaccharides, and L-theanine, also exhibit health-promoting properties, the antiobesity effects of tea are predominantly attributed to its catechin-rich constituents. This section will therefore focus on the mechanisms of tea catechins and their oxidative derivatives.
In addition to their beneficial effects on T2DM, tea catechins and their oxidative derivatives demonstrate comparable efficacy in ameliorating obesity and modulating its associated metabolic dysfunctions (Table 1). First, as green tea has the highest content of catechins, it has significant antiobesity effects overall[37]. These effects include alleviating intestinal microbial imbalance and regulating intestinal metabolites[38,39], promoting fat breakdown[40], and improving metabolic dysfunction[41], etc. Mechanistic studies confirm catechins' central role in these processes. Li Wang et al. revealed that green tea catechins ameliorate adiposity by reversing high fat diet (HFD)-induced elevation of the Firmicutes/Bacteroidetes (F/B) ratio in the gut microbiota[42]. At the molecular level, green tea catechins activate the PPARγ/FGF21/AMPK/UCP1 signaling axis to promote white-to-beige adipose tissue conversion, a critical mechanism for augmented energy expenditure[43]. Upregulation of adiponectin mediates their dual antiobesity effects: stimulating white adipose thermogenesis[44] and suppressing lipid accumulation[45]. Concurrently, these compounds inhibit intestinal lipid absorption through a physicochemical interaction with dietary fats[46]. Individual catechins demonstrate comparable antiadipogenic properties. EGCG replicates the multimechanistic effects observed with whole catechin extracts, including modulation of the gut microbiota[47], energy metabolism activation[48], inhibited adipocyte proliferation[49], and induction of white adipose autophagy[50]. Notably, EGCG exhibits a unique capacity to enhance fatty acid oxidation pathways[51]. OTPs, which are particularly abundant in black and pu-erh teas as a result of specific processing techniques, exert antiobesity effects through distinct mechanisms: suppressing lipid absorption/ingestion to reduce caloric intake, activating AMPK-mediated lipolysis[52], and inhibiting the accumulation of adipocyte lipids[53]. Theaflavins, characteristic oxidative polymers that are abundant in black tea, demonstrate multimechanistic antiobesity efficacy[54]. Experimental evidence confirms that administration of theaflavins ameliorates HFD-induced obesity in murine models through the SIRT6/AMPK/SREBP-1/FASN signaling axis, effectively suppressing hepatic lipogenesis[55]. In oolong tea extracts, theaflavins and complementary catechins constitute the key mediators of lipid metabolism regulation and gut microbiota modulation. Their antiadipogenic effects correlate significantly with enriched abundance of specific microbiota, particularly Candidatus Arthromitus and Hydrogenoanaerobacterium species[56].
The beneficial effects and underlying mechanisms of tea catechins and their polymers on MASLD
-
MASLD, a metabolic disorder stemming from dysregulated glucose and lipid metabolism, manifests primarily through ectopic lipid accumulation in the hepatocytes. This condition frequently co-occurs with obesity and T2DM as a comorbidity[57], and was formerly known as nonalcoholic fatty liver disease (NAFLD)[58]. Consequently, therapeutic strategies including novel hypoglycemic agents, antihyperlipidemic drugs, lifestyle modifications, nutritional interventions, and exercise regimens have been extensively investigated for managing MASLD[59]. Current NAFLD control strategies target the core pathological features, namely hepatic lipid deposition, oxidative stress, and inflammation, with accumulating evidence supporting the efficacy of natural compounds in disease modulation[60]. Exercise training constitutes an effective nonpharmacological intervention, attenuating NAFLD's progression through amelioration of hepatic oxidative stress and inflammatory responses[61]. In adjunctive therapy, natural products demonstrate multitarget therapeutic advantages because of their low toxicity profiles. For instance, Astragalus polysaccharides mitigate HFD-induced NAFLD by inhibiting hepatic lipid translocation[62]. Catechin supplementation similarly suppresses hepatic steatosis and enhances antioxidant capacity, collectively improving NAFLD's pathology[63]. Mechanistic studies reveal that caffeine—a primary tea alkaloid—attenuates NAFLD's progression through myokine-mediated regulation by stimulating skeletal muscle secretion of interleukin-6 (IL-6), which subsequently modulates hepatic metabolic pathways[64]. The catechins and their polymers in tea have significant antiobesity and anti-T2DM effects. Therefore, studies on related complications have also demonstrated that these compounds exert remarkable beneficial effects on NAFLD. Experimental studies have highlighted green tea extract (GTE), enriched with native catechins, as a potent inhibitor of NAFLD's progression[65]. Mechanistically, tea catechins activate AMPK (a master regulator of energy homeostasis) to suppress hepatic lipogenesis while ameliorating mitochondrial dysfunction in energy metabolism-impaired hepatocytes[66]. Gut microbiota remodeling emerges as another critical pathway, where catechin administration increases the abundance of beneficial bacteria (Bacteroidetes, Faecalibacterium, Parabacteroides, Akkermansia) and enhances acid-producing genera (Butyricimonas, Desulfovibrio), thereby attenuating diet-induced dyslipidemia and hepatic steatosis[67]. As a monomeric catechin, EGCG reduces intestinal bile acid reabsorption, lowering luminal bile acid levels to inhibit lipid absorption, ultimately mitigating HFD-induced metabolic dysregulation and fatty liver disease[68]. Dipeptidyl peptidase-4 (DPP-4), the primary enzyme degrading incretins like glucagon-like peptide-1 (GLP-1), represents a therapeutic target for T2DM and its complications. EGCG suppresses both DPP-4's expression and enzymatic activity, reducing hepatic lipid accumulation and preventing liver injury to prevent and ameliorate NAFLD[69]. Furthermore, EGCG, the predominant constituent of green tea extract, targets AMPK as a key regulatory node to inhibit HFD-induced hepatic steatosis in murine models[70]. In a clinical validation, a double-blind placebo-controlled study demonstrated that catechin-rich green tea extract significantly reduces hepatic fat content and inflammation markers in NAFLD patients[71]. Oxidized catechins similarly attenuate hepatic lipid accumulation through modulation of lipid metabolism regulators, though their mechanisms diverge from native compounds[53]. Given NAFLD's frequent comorbidity with obesity and T2DM, the therapeutic targets and pathways underlying catechin-mediated improvement in NAFLD substantially overlap with those observed in obesity and diabetes intervention studies. In an acute hepatic ischemia–reperfusion study, theaflavins counterintuitively demonstrated hepatoprotective effects in steatotic livers by attenuating hepatic steatosis, oxidative stress, and inflammation while reducing apoptosis of hepatocytes[72]. Separately, theaflavin-3,3'-digallate (TF3)—a oxidized polymer isolated from black tea—exerts antisteatotic properties by suppressing hepatic lipid accumulation through dual regulatory mechanisms: modulating the Fads1/PPARδ/Fabp4 signaling axis and orchestrating gut microbiota-mediated lipid metabolism[73].
The beneficial effects and underlying mechanisms of tea catechins and their polymers on AS
-
The pathogenesis of atherosclerosis (AS) is profoundly influenced by inflammation and oxidative stress, placing it squarely within the scope of the shared mechanisms outlined above. AS, a cardiovascular-linked chronic metabolic disorder, represents the primary contributor to global cardiovascular mortality. Its pathogenesis initiates with elevated blood lipids, triggering subendothelial lipoprotein accumulation, progressing to plaque rupture, thrombus formation, and consequent narrowing of the arterial lumen[74]. Contemporary therapeutic strategies primarily target inflammatory pathways and cholesterol metabolism, where prolonging the functionality of low-density lipoprotein (LDL) receptors (LDLRs) sustains cholesterol homeostasis, thereby inhibiting atherogenesis[75]. Hyperlipidemia potentiates the progression of AS by inducing the deposition of lipid-laden M1-polarized macrophages within the vascular walls, making the inhibition of macrophage proliferation/activation a viable therapeutic approach[76]. Notably, natural products provide abundant PCSK9 ligands and macrophage activity inhibitors. Experimental studies have demonstrated that naringenin significantly improves plaque's morphology and reduces intraplaque macrophage accumulation[77].
Tea catechins and their oxidative polymers demonstrate multimodal therapeutic potential against AS through diverse bioactivities encompassing anti-inflammatory, antioxidant, and anticholesterolemic effects. Pathophysiological studies have identified inflammation and oxidative stress as the pivotal drivers of atherogenesis (Table 1)[78,79], with tea catechins and their polymers exhibiting significant AS-modulating capacity via their antioxidant properties[80]. Experimental evidence has revealed that EGCG suppresses cytokine secretion (IL-2, IL-4, IFN-γ, tumor necrosis factor-α [TNF-α]) in activated human primary T-cells, suggesting immunomodulatory inhibition of inflammatory atherosclerosis[81]. ECG, another component of catechin, reduces aortic plaque formation in ApoE-/- mice through dual mechanisms: decreasing malondialdehyde (MDA) levels while enhancing superoxide dismutase (SOD) activity to mitigate oxidative stress[82], and inhibiting ox-LDL-induced apoptosis to promote macrophage migration and plaque stabilization via combined antioxidant and anti-inflammatory actions[83].
The gut–vascular axis emerges as a novel therapeutic frontier, with tea catechins attenuating HFD-induced aortic plaque formation in ApoE-/- mice through selective enrichment of Bifidobacterium species[84]. Given the established correlation between the accumulation of LDL cholesterol and atherogenesis, epidemiological data indicate that green tea consumption is associated with a 31% reduction in the formaton of aortic lesions under cholesterol-supplemented dietary conditions[85]. Mechanistic studies also revealed that green tea catechins counteract HFD-induced suppression of vascular autophagy by decreasing serum ox-LDL levels while upregulating autophagy markers (LC3-II, Beclin-1, p62) at both the transcriptional and translational levels in vascular tissues[86]. Theaflavins, prominent oxidative polymers in black tea, exhibit potent antioxidative and anti-inflammatory properties. Mechanistically, they activate the miR-24-mediated Nrf2/HO-1 signaling axis, effectively reducing serum lipid levels and MDA production while suppressing the progression of atherosclerotic plaque and aortic histological alterations[87].
The beneficial effects and underlying mechanisms of tea catechins and their polymers on hyperuricemia
-
Hyperuricemia (gout), a significant contributor to multiple metabolic disorders, is characterized by elevated uric acid (UA) levels. Current management primarily relies on xanthine oxidase (XO) inhibitors and uricosuric agents[88], yet these therapeutics carry the risks of chronic kidney injury[89]. Notably, natural compounds have demonstrated dual benefits in ameliorating hyperuricemia while conferring renal protection. Resveratrol treatment significantly reduces serum UA, creatinine, blood urea nitrogen, and urinary protein levels while attenuating renal fibrosis and inflammation[90]. Purple sweet potato leaf polyphenols suppress hepatic xanthine oxidoreductase (XOD) and adenosine deaminase activities, effectively lowering serum UA while exerting nephroprotective effects[91]. Consequently, dietary intervention represents an effective strategy for hyperuricemia prevention and UA control, with tea catechins and their polymers serving as key bioactive mediators.
It is important to note that the primary pathological consequence of hyperuricemia is the crystallization of urate within the joints and tissues. These crystal deposits activate pattern recognition receptors, including the XO and NLRP3 inflammasome, triggering acute gouty arthritis[92] Gang Chen et al. demonstrated that green tea catechins reduce serum UA through dual mechanisms: inhibiting hepatic XOD expression (decreasing production) and enhancing renal excretion[93]. Crucially, these effects coincide with significant nephroprotection[94]. EGCG monotherapy specifically suppresses hepatic XOD activity while upregulating the expression of renal organic anion transporter 1 (OAT1), confirming its pivotal role in UA homeostasis[95]. Mechanistically, EGCG modulates UA transporters in vivo by inducing UA-secretory transporters (OAT1, OCT1) while suppressing reabsorptive transporters (URAT1, GLUT9). Concurrently, it remodels the gut microbiota by enriching the Lactobacillus, Faecalibacterium, and Bifidobacterium genera. Metabolomic analyses suggest that EGCG-induced microbial metabolite modifications contribute to alleviating hyperuricemia[96]. Fermented tea extracts rich in oxidized catechins exhibit comparable hypouricemic properties[97]. In circadian disruption-induced hyperuricemia models, pu-erh tea catechins restore the gut microbial ecology by elevating Bifidobacterium, Akkermansia, and Faecalibacterium abundance, thereby enhancing UA excretion and reducing serum UA/xanthine levels[98]. Network pharmacology predictions also indicate that theaflavins target multiple genes and proteins in the pathogenesis of gout[99].
-
The beneficial effects of tea catechins and their oxidative polymers on various CMDs, as detailed in the preceding sections, are underpinned by a common pharmacological framework. Rather than operating through isolated pathways, these compounds exert their pleiotropic effects by simultaneously targeting a network of interconnected biological processes. This section synthesizes these core mechanisms—centering on the amelioration of insulin resistance, the suppression of chronic inflammation, and remodeling of the gut microbiota ecosystem—to provide a unified understanding of how these natural compounds combat metabolic dysregulation across different disease contexts (Table 2). The following discussions on specific diseases should be viewed as specific manifestations and evidence of this multitarget framework.
Table 2. Summary of the key shared pharmacological targets of tea catechins and their polymers.
Shared mechanism Key molecular targets/pathways Representative tea components Insulin resistance AMPK, IRS-1/PI3K/Akt, GLUT4, PPARγ EGCG, theaflavins, green tea catechins Inflammation NLRP3 Inflammasome, NF-κB, TNF-α, IL-6 EGCG, ECG, theaflavins Oxidative stress Nrf2/HO-1, SOD, MDA ECG, EGCG, theaflavins Gut microbiota dysbiosis Microbiota composition, SCFAs, intestinal FXR/FGF19, bile acids EGCG, EC, theabrownins, green tea catechins Amelioration of insulin resistance
-
Insulin resistance is a cornerstone of multiple CMDs. Tea catechins and their polymers enhance insulin sensitivity through multifaceted actions on insulin signaling pathways. A primary mechanism involves the activation of AMPK, a central cellular energy sensor. Upon activation by compounds such as EGCG and theaflavins, AMPK phosphorylates downstream targets to promote glucose uptake and fatty acid oxidation while inhibiting gluconeogenesis and lipogenesis[66,70]. Concurrently, they potentiate the insulin receptor substrate 1/phosphatidylinositol 3-kinase/protein kinase B (IRS-1/PI3K/Akt) signaling axis. For instance, theaflavins have been shown to enhance phosphorylation of Akt and membrane translocation of GLUT4, facilitating cellular glucose uptake[26]. Furthermore, tea catechins and their polymers mitigate factors contributing to insulin resistance, such as oxidative stress and endoplasmic reticulum stress, by upregulating Nrf2-mediated antioxidant defenses[87] and modulating the unfolded protein response. The critical role of this mechanism is exemplified in the improvement of T2DM, the mitigation of obesity-related metabolic dysfunction, and the amelioration of MASLD.
Suppression of chronic inflammation and oxidative stress
-
Sustained inflammation and oxidative stress are critical drivers of CMDs' progression. Tea catechins and their polymers function as potent anti-inflammatory and antioxidant agents, primarily by inhibiting the NLRP3 inflammasome and the NF-κB signaling pathway. EGCG effectively suppresses activation of the NLRP3 inflammasome, thereby reducing the maturation and secretion of proinflammatory cytokines like IL-1β and IL-18[23]. The anti-inflammatory effects are also mediated through the inhibition of NF-κB nuclear translocation, leading to decreased expression of TNF-α, IL-6, and other adhesion molecules[81,83]. In parallel, the antioxidant capacity of these compounds stems from their direct free radical-scavenging properties and their ability to induce endogenous antioxidant enzymes via the Nrf2/HO-1 pathway. Compounds like ECG have been demonstrated to reduce MDA levels and enhance SOD activity, thereby alleviating oxidative damage in vascular tissues[82]. This foundational activity directly contributes to the attenuation of atherosclerosis and is also a key component in alleviating the pathologies of MASLD and hyperuricemia-induced gouty inflammation.
Remodeling of the gut microbiota and the gut–organ axes
-
The gut microbiota plays a pivotal role in regulating the host's metabolism, and its dysbiosis is intricately linked to CMDs. Tea catechins and their polymers exert profound prebiotic-like effects, selectively enriching beneficial microbial populations (e.g., Bacteroidetes, Akkermansia, Bifidobacterium, Faecalibacterium) while suppressing detrimental bacteria[42,67,84]. This microbial remodeling enhances intestinal barrier integrity, reducing the translocation of lipopolysaccarides (LPS) into the circulation and the subsequent metabolic endotoxemia. The biological consequences of catechin-induced microbiota shifts are mediated through the gut–liver and gut–vascular axes. Microbial fermentation of these compounds yields bioactive metabolites, particularly short-chain fatty acids (SCFAs) like butyrate, which exert systemic anti-inflammatory and insulin-sensitizing effects. Moreover, tea catechins, their polymers, and their microbial metabolites modulate bile acid metabolism and the farnesoid X receptor (FXR)–fibroblast growth factor 15/19 (FGF15/19) axis, influencing hepatic glycolipid metabolism[47,68]. Theabrownins from dark tea exemplify this mechanism by restoring the gut barrier's function and altering microbial metabolites, thereby ameliorating systemic insulin resistance and hepatic steatosis[28]. The gut microbiome serves as a key modulator for the effects of tea catechins across CMDs, with its influence being particularly evident in the evidence presented for obesity, T2DM, and hyperuricemia.
The interconnectedness of these pathways underscores the multitarget nature of tea catechins and their polymers. For example, activation of AMPK not only improves insulin sensitivity but also inhibits NF-κB-mediated inflammation. Similarly, gut microbiota remodeling can lead to reduced systemic inflammation and improved insulin signaling. This synergistic targeting of the shared mechanisms provides a robust pharmacological foundation for the efficacy of these compounds against a spectrum of CMDs, as detailed in the previous sections.
-
Tea catechins and their oxidative polymers, a class of ubiquitous phenolic compounds in Camellia sinensis, demonstrate unique therapeutic potential against CMDs such as T2DM and atherosclerosis, primarily through their anti-inflammatory and antioxidant properties (Fig. 4). This review systematically elucidates their multitarget mechanisms involving lipid metabolism regulation, insulin resistance amelioration, and inflammatory pathway suppression. Their natural origin and favorable safety profile provide a scientific rationale for developing dietary interventions aligned with the 'medicine–food homology' concept. Notably, bioactive metabolites generated via gut microbiota-mediated biotransformation may synergize with parental catechins, offering novel perspectives for deciphering their holistic biological effects.
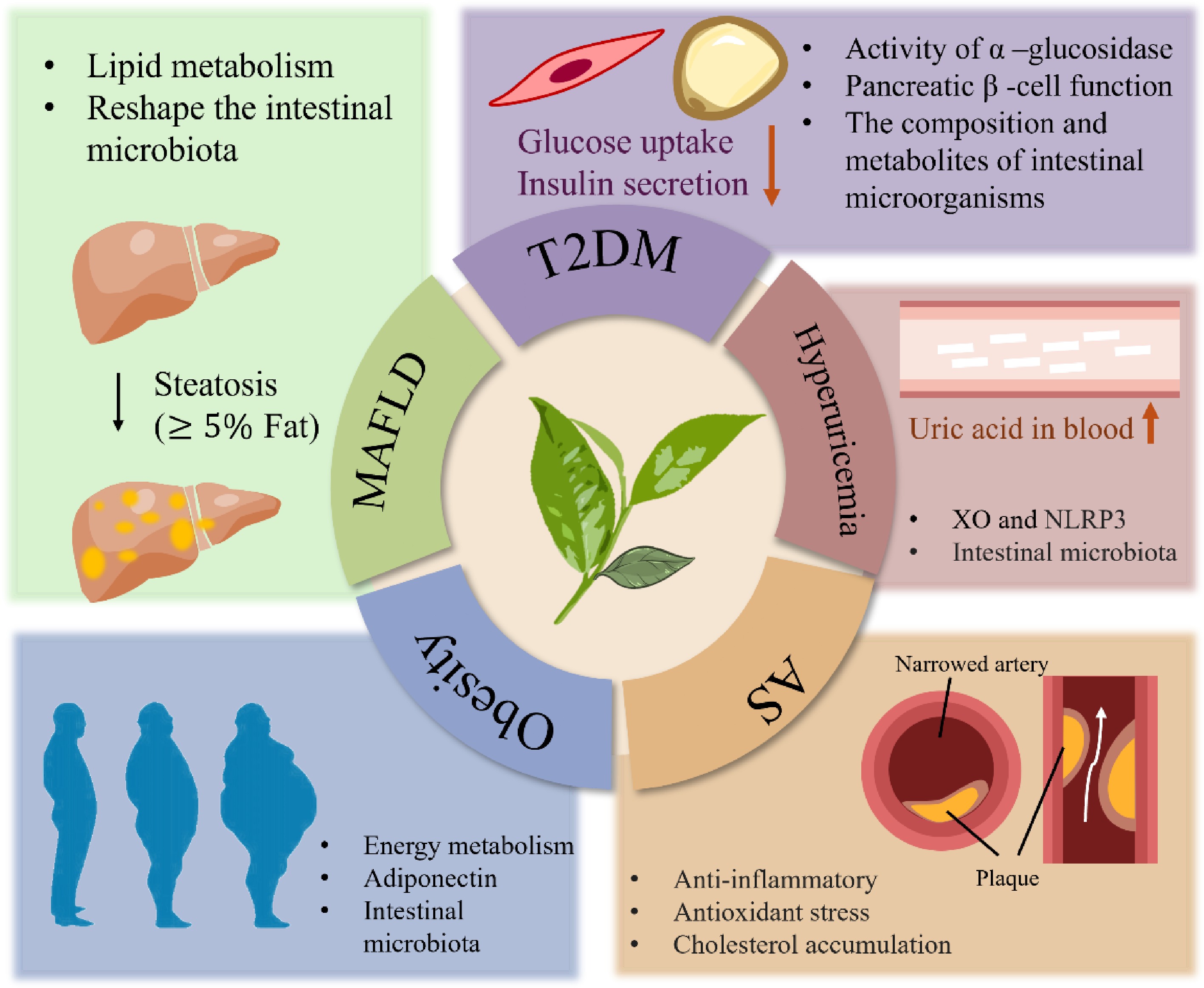
Figure 4.
The improving effect of tea catechins and their polymers on metabolic diseases: pharmacology and mechanism.
Critical limitations impede their clinical translation. Suboptimal bioavailability restricts their therapeutic efficacy, necessitating advanced delivery systems or structural modifications. Catechin–drug interactions and inter-individual metabolic heterogeneity remain insufficiently characterized. Substantial research and clinical trials remain imperative to comprehensively elucidate the efficacy and safety profiles of tea catechins and their polymers in managing CMDs. Concurrently, employing multi-omics technologies to identify the potential therapeutic targets and interaction networks will enable more precise assessment of their pharmacodynamic effects and pharmacokinetic properties. These investigations are crucial for developing advanced therapeutic agents and intervention strategies against CMDs. The integration of precision nutrition with systems biology approaches positions tea catechins and their polymers as pivotal components in metabolic health management, potentially revolutionizing targeted interventions within the diet–microbiota–host triad.
-
The authors confirm contribution to the paper as follows: writing − original draft preparation: Wang Y, Peng W, Wang Q; writing − review and editing: Liu K, Zhou Q; investigation: Wang Y; supervision: Liu K; validation: Zhou Q. All authors reviewed the results and approved the final version of the manuscript.
-
Data availability is not applicable to this article as no new data were created or analyzed in this study.
-
This study was supported by the Scientific and Technological Research Projects of Henan Province (242102110327).
-
The authors declare that they have no conflict of interest.
-
# Authors contributed equally: Yinghao Wang, Wenyuan Peng
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Wang Y, Peng W, Wang Q, Liu K, Zhou Q. 2026. The improvement effect of tea catechins on metabolic diseases: pharmacology and mechanism. Beverage Plant Research 6: e014 doi: 10.48130/bpr-0025-0044
The improvement effect of tea catechins on metabolic diseases: pharmacology and mechanism
- Received: 03 June 2025
- Revised: 19 November 2025
- Accepted: 04 December 2025
- Published online: 15 April 2026
Abstract: Chronic metabolic diseases (CMDs), characterized by Type 2 diabetes mellitus (T2DM), obesity, and atherosclerosis, represent a global health crisis, significantly impairing patients' quality of life and imposing substantial socioeconomic burdens. Although current pharmacological interventions can mitigate disease progression, long-term administration carries the inherent risks of adverse effects. Medicinal food homologous (MFH) substances are emerging as promising interventions for metabolic disorders owing to their multitarget bioactivity, favorable safety profiles, and high patient compliance. Recent research demonstrates that tea catechins and their oxidative polymers exert beneficial effects against metabolic dysregulation through diverse mechanisms, including modulation of glucolipid metabolism, amelioration of insulin resistance, and anti-inflammatory actions. This review systematically synthesizes the molecular mechanisms and key signaling pathways underpinning the beneficial effects of tea catechins and their oxidative polymers in obesity, diabetes, and related syndromes, which include regulating glucolipid metabolism, improving insulin resistance, and exerting anti-inflammatory and antioxidant activities. Furthermore, the article critically discusses the clinical translational potential of these compounds, highlighting future applications as dietary supplements or adjuvant therapeutics. Collectively, these findings establish a theoretical foundation for developing novel natural product-based therapeutics against metabolic disorders.
-
Key words:
- Tea catechins /
- Metabolic diseases /
- Insulin resistance /
- Gut microbiota /
- Oxidative stress /
- Atherosclerosis