-
Plastic pollution has emerged as one of the most pressing environmental challenges of the Anthropocene. It is estimated that approximately 12,000 million metric tons of plastic waste will be accumulated globally by 2050, with soils and sediments serving as major long-term reservoirs for these persistent materials[1]. As plastics fragment, they generate microplastics (MPs), i.e., particles smaller than 5 mm, which have become widespread contaminants in terrestrial ecosystems[2]. Increasing evidence shows that soils and sediments serve as major reservoirs of these particles[3]. Key pathways introducing MPs into soils include agricultural practices such as plastic mulching, application of sewage sludge and compost, irrigation with reclaimed water, and atmospheric deposition[4]. MPs pose growing eco-toxicological risks by being ingested through the food chain and acting as vectors for the adsorption and transfer of other pollutants across food webs[5,6]. Recently, growing evidence suggests that their persistent presence in soils can influence soil ecosystem functions by altering soil microbial communities and associated functional genes. However, a comprehensive understanding of how microplastics regulate soil microbial processes remains limited, thereby constraining our predication of the impacts of MPs on soil ecosystem functions.
MPs provide novel surfaces for microbial colonization and directly interact with soil microbiomes, thereby altering the abundance and activity of functional genes related to biogeochemical cycles[7,8]. For example, long-term exposure to residual plastic mulch in farmland soils has been associated with reduced abundance of carbon-cycling genes, including β-glu and chiA, and shifts in nitrogen-related genes such as nifH, nirS, and nirK[9,10]. In addition, as MPs act as carriers that facilitate the co-transport of heavy metals and antibiotics, they promote the dissemination of antibiotic resistance genes (ARGs)[11]. This is exemplified by a recent study showing that bio-based polylactic acid (PLA) microplastics significantly enriched and disseminated ARGs under the combined pressure of zinc and sulfadiazine in marine environments, serving as a more potent vector than traditional polyethylene (PE)[12]. Similarly, MPs have been shown to enrich antibiotic resistance genes in soils, particularly when combined with co-contaminants that exert selective pressure[9]. Importantly, microplastic effects vary with polymer type, particle size, aging state, and concentration, resulting in variable responses in functional gene profiles[4]. Even biodegradable plastics, such as polylactide and polybutylene succinate, can disrupt carbon and nitrogen cycling by altering microbial gene expression through changes in carbon availability or nutrient stoichiometry[13,14]. Against this background, a functional gene-centered perspective has emerged as a useful approach for synthesizing microplastic impacts on soil ecosystems.
Emerging evidence suggests that MPs create distinct microhabitats, such as the plastisphere, i.e., the microbial biofilm formed on microplastic surfaces[4,15], and soil fauna gut microbiomes, which can act as hotspots for functional gene enrichment and horizontal gene transfer[16]. Soil fauna, including earthworms and collembolans, can ingest MPs, potentially altering gut microbial functions and facilitating the transport of resistance genes through food webs[15,17]. In parallel, global environmental changes, including climate warming, drought, and altered precipitation patterns, are likely to interact with MP pollution and modulate microbial functional gene responses[18,19]. However, most studies to date rely on short-term laboratory experiments under controlled conditions, limiting our ability to capture the complexity of real soil systems and multi-stressor interactions[4].
Despite growing interest in microplastic effects on soil ecosystems, current understanding remains fragmented. Microbial functional genes provide a powerful lens for examining how microplastics influence soil biogeochemical processes. They serve as sensitive and valuable indicators for assessing soil health and its responses to management measures[20]. Existing studies have largely emphasized changes in soil physicochemical properties, microbial community composition, or bulk biogeochemical processes, while microbial functional gene responses have not been systematically synthesized. In addition, plastisphere-associated communities often represent a small fraction of total soil biomass, leading to signal dilution in bulk-soil analyses; moreover, functional redundancy among soil microbes and co-occurring environmental stressors further complicate attribution of gene-level responses specifically to microplastic exposure.
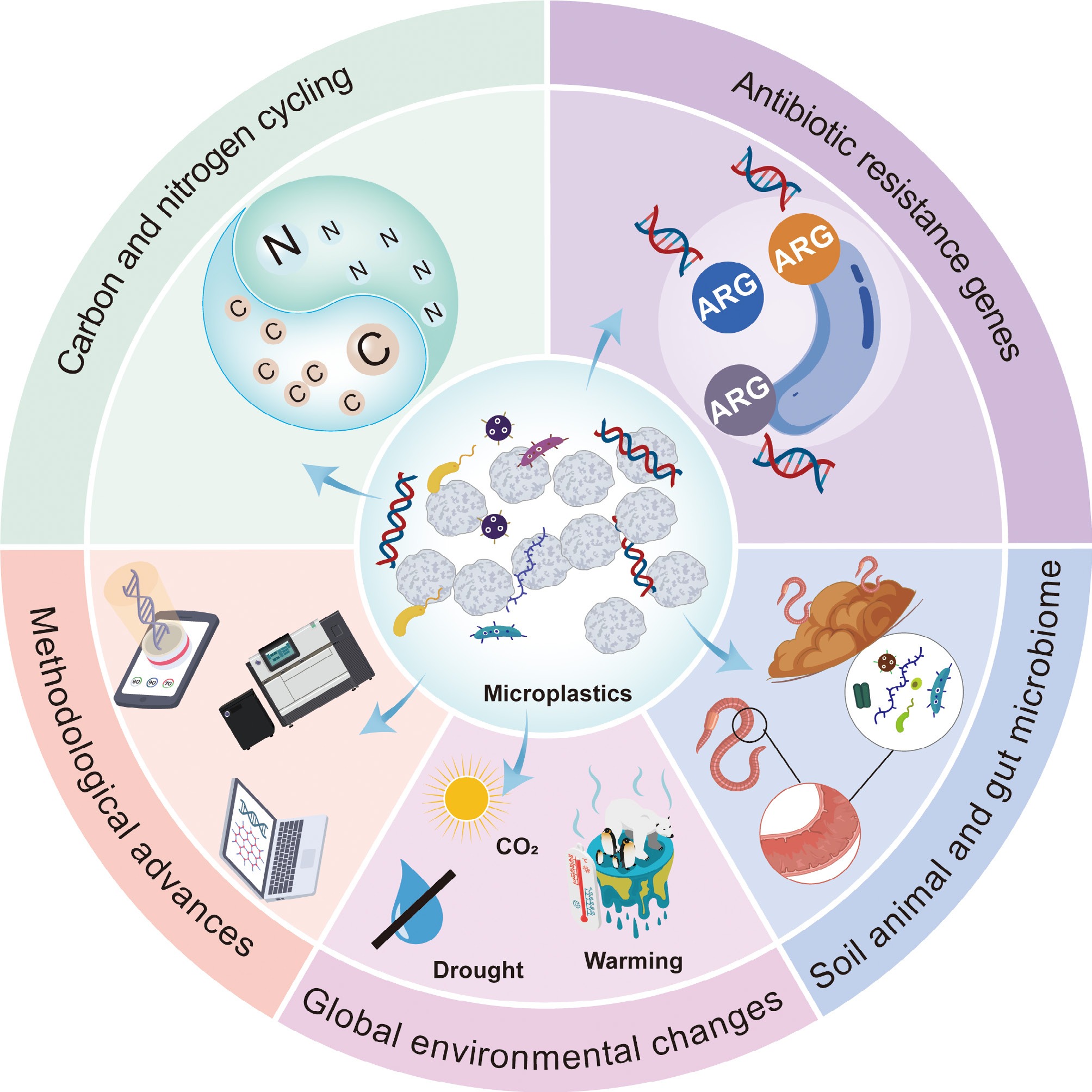
To address these limitations, this review adopts a functional gene-centric perspective and focuses on three major gene categories most relevant to microplastic pollution research: carbon cycling genes, nitrogen cycling genes, and ARGs. These functional gene groups underpin key ecological functions and risks, including greenhouse gas emissions (e.g., CO2, N2O, and CH4), nitrogen transformation efficiency, and the dissemination of ARGs. Together, they offer an integrative framework for organizing current evidence on microplastic–microbe interactions in soil systems. By synthesizing functional gene-based evidence across diverse soil contexts, this review identifies key knowledge gaps and outlines priorities for long-term field studies and mechanistic investigations under global change scenarios.
-
MPs exert profound and complex effects on soil carbon and nitrogen cycling by altering key biochemical processes and microbial functions[21−24]. These effects are mediated through multiple interconnected pathways, including exogenous carbon input, surface-mediated interactions, and the selective shaping of microbial communities, which collectively reprogram the transformations of soil carbon and nitrogen, and the expression of related functional genes (Fig. 1). Regarding the carbon cycle, MPs can alter the content of dissolved organic carbon (DOC)[25−27]. These alterations occur through multiple pathways, including the sorption of DOC and other soil metabolites onto MPs surfaces to form an eco-corona, which can reduce the bioavailability of sorbed DOC, and compete for surface adsorption sites[28]. Concurrently, MPs can serve as a source of labile DOC through the leaching of plastic additives and polymer degradation products, which directly stimulate microbial activity upon release into the environment[29]. Collectively, these processes influence soil and aquatic carbon cycling. MPs can also accelerate soil organic matter (SOM) mineralization[30], and stimulate soil respiration[31], leading to increased CO2 emissions. For instance, PE MPs at 5% (w/w) significantly increased CO2 emissions by 24%–28.67%, primarily attributed to the increased relative abundance of Mycobacterium, Aeromicrobium, Amycolatopsis, and Mortierella species[32]. A global meta-analysis further confirms that MP exposure significantly enhances the abundance of functional genes governing soil carbon decomposition (e.g., abfA, manB, sga), thereby accelerating the mineralization of soil organic carbon and leading to a substantial increase in soil CO2 emissions (by 54.3% on average)[33]. In addition to promoting CO2 emissions, MPs can also profoundly influence methane (CH4) dynamics, a potent greenhouse gas. A study revealed that the co-occurrence of PE (0.5%, w/w), and hydrochar (1%, w/w) significantly enhanced the abundance of the methanogen gene mcrA and increased soil CH4 emissions and greenhouse gas intensity by 83.5% and 36.5%, respectively, compared to the application of hydrochar alone[34]. Conversely, other research indicates that MPs, especially after aging, can suppress CH4 emissions. For example, in paddy soils, aged PLA MPs were found to act as an electron shuttle, diverting electrons to nitrate and iron reduction instead of methanogenesis. This redirection was corroborated by a significant down-regulation of the key methanogenic gene mcrA alongside an up-regulation of genes involved in anaerobic methane oxidation (e.g., fdh, frmB), thereby inhibiting methanogenesis and reducing overall CH4 flux[35].
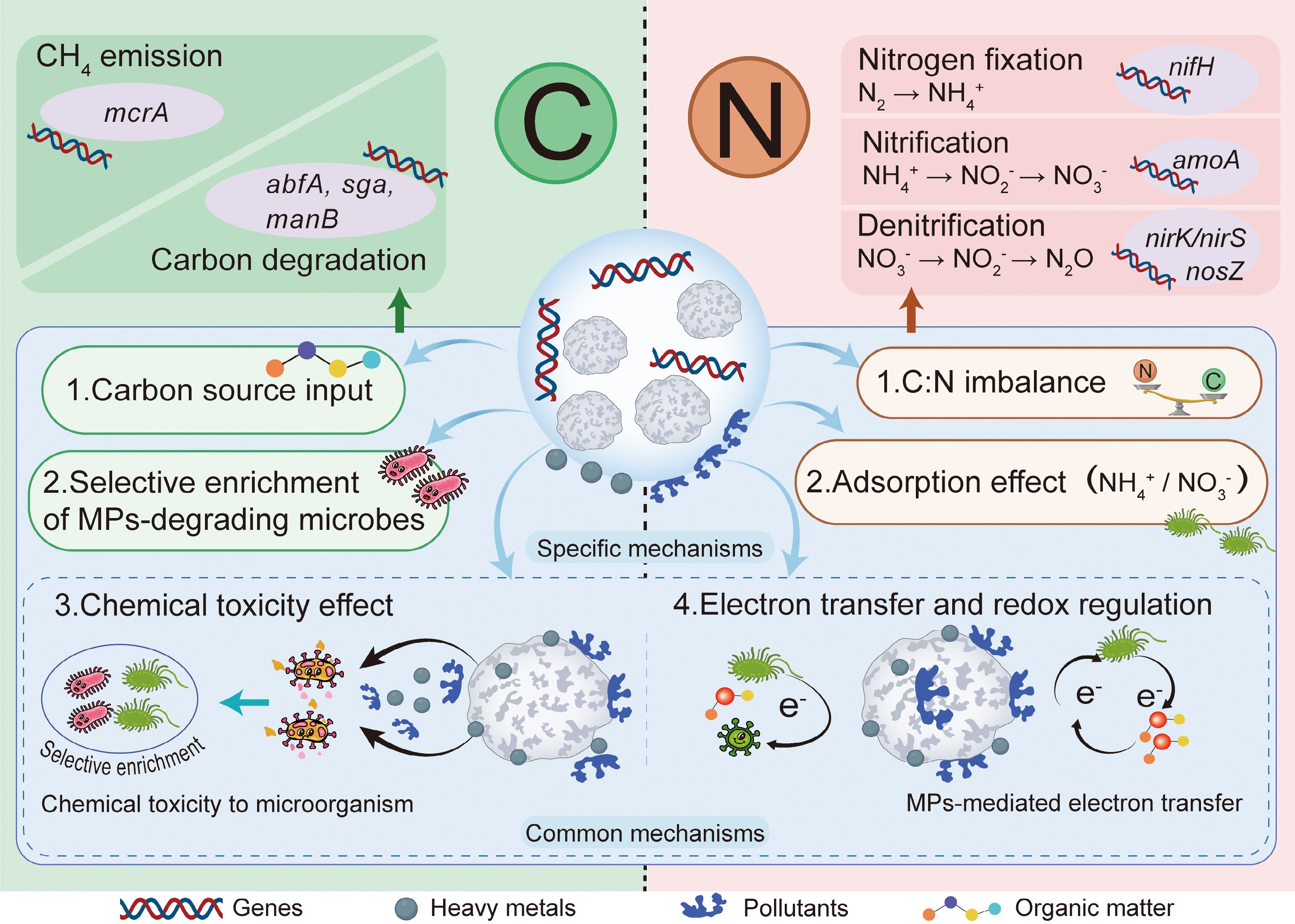
Figure 1.
Microplastics affect soil carbon and nitrogen cycling and related gene expression through multiple pathways. In the carbon cycle, MPs serve as an exogenous carbon input (1) and selectively enrich microbial communities capable of degrading MPs (2). These changes alter the abundance of functional genes involved in carbon metabolism, such as those for carbon degradation (abfA, sga, manB) and methane emission (mcrA), collectively influencing carbon turnover and methane release. In the nitrogen cycle, the carbon input disrupts the soil C:N balance (1), while MP surfaces adsorb ammonium ions (2). These processes jointly alter nitrogen availability and regulate the expression of genes associated with nitrogen fixation (nifH), nitrification (amoA), and denitrification (nirS, nirK, nosZ). Furthermore, MPs selectively shape microbial communities by adsorbing heavy metals and organic pollutants (3), and mediate electron transfer and redox reactions (4). Together, these mechanisms reshape microbial metabolic activity and the expression of genes related to carbon and nitrogen transformations.
Alterations in the carbon cycle induced by MPs further regulate nitrogen transformation rates and pathways by altering microbial energy supply and the stoichiometric balance between carbon and nitrogen. As MPs are inherently carbon-rich, they function as exogenous carbon sources that reshape soil carbon and energy dynamics[36], where polybutylene adipate terephthalate (PBAT) and PE stimulate significant DOC accumulation, whereas polyethylene terephthalate (PET) consistently suppresses DOC availability[36]. Microplastic contamination alters carbon cycling by increasing soil organic carbon and dissolved organic carbon, subsequently regulating microbial energy dynamics through enhanced microbial biomass carbon and nitrogen[19]. This process modulates the C/N ratio (e.g., a 19% reduction in ammonium nitrogen under biodegradable MPs) and ultimately affects nitrogen transformation rates via enhanced nirK gene abundance and nitrous oxide emissions[19,37]. In addition to this indirect mechanism, MPs can also directly alter nitrogen availability through the physical adsorption of nitrogenous ions (e.g., NH4+) from the soil solution[38]. Within nitrogen cycling processes, MPs interfere with key processes including nitrogen fixation[39,40], nitrification[41,42], and denitrification[43,44]. Biological nitrogen fixation responds variably to polymer type and concentration. Low concentrations of polyvinyl chloride (PVC) or low-density PE (LDPE) may increase nifH gene abundance and N-fixing bacteria, while higher concentrations often inhibit these groups[45,46]. Additionally, a long-term field study found that more than ten years of residual plastic film exposure increased the abundance of nitrogen fixation (nifH), nitrite reductase (nirS), and N2O reductase (nosZ) genes, but decreased the abundance of nitrite reductase (nirK), indicating a shift in nitrogen transformation potential[10]. Nitrification exhibits distinct sensitivity to MP contamination, with ammonia-oxidizing bacteria (AOB) and archaea (AOA) showing varied responses to different polymers[46,47]. For instance, LDPE at 7% (w/w) increased AOB-amoA gene abundance, whereas PVC-MP reduced it[45,46]. MPs can alter the abundance and composition of denitrification functional genes, thereby affecting the denitrification process and N2O production[48−50]. For example, exposure to polyethylene and polylactide MPs has been reported to significantly increase the relative abundance of key denitrification genes, including nirS, nirK, and nosZ[51], indicating a potential shift in microbial nitrogen transformation capacity. Moreover, a meta-analysis confirmed that MPs increase denitrification rates by 17.8%, and significantly elevate N2O emissions by 140.6% while reducing soil available nitrogen pools (e.g., NO3− and NH4+)[43]. Collectively, MPs alter the structure and function of soil microbial communities, leading to disrupted carbon and nitrogen cycling through altered gene expression and metabolic activity. These perturbations pose a growing threat to agricultural sustainability by potentially reducing soil nutrient use efficiency and amplifying greenhouse gas emissions.
-
The co-occurrence of ARGs and MPs in agricultural soils has been increasingly reported, largely driven by the application of organic fertilizers and sewage sludge[52]. As a result, soil ecosystems have become important reservoirs of both ARGs and MPs, raising global concerns about their combined environmental and health risks[53−55]. MPs alter soil biophysical properties and microbial communities, thereby reshaping the soil resistome and elevating ARG abundance[56]. Long-term plastic film contamination has been shown to significantly increase ARG levels across Chinese farmland, with the highest abundance observed in Xinjiang, where mulching histories are longest[57]. Interestingly, a recent study revealed that the biodegradable PBS particles significantly increased the abundance of ARGs in the soil compared to the control and conventional MPs treatments, owing to the broadened bacterial hosts (i.e., the relative abundance of Proteobacteria as a host increased from 38.5% in the control soils to 58.2%)[58]. These findings highlight the necessity of further exploring the ecological risks and mechanisms of biodegradable MPs.
Beyond their direct effects, MPs act as vectors for co-contaminants such as heavy metals, antibiotics, and pesticides, generating complex co-selection pressures that further promote ARG proliferation[56]. For example, PE increased the bioavailable tetracycline and copper, intensifying ARG enrichment under co-selection by these pollutants in manured soils[59]. Addressing the transmission of ARGs in such combined pollution contexts is therefore vital for sustainable soil management[60].
Once in soil, MPs are rapidly colonized by microorganisms, forming biofilms that create a 'plastisphere', whose physicochemical properties and microbial community structure differ markedly from those of the surrounding soil matrix[61]. This renders it a 'hotspot' for microbial colonization and metabolic activity[62]. Metagenomic analyses have demonstrated that plastispheres selectively enrich ARGs to a greater extent than the surrounding bulk soil, with enrichment patterns varying by polymer type and soil characteristics[63]. For instance, plastispheres of low-density LDPE, polypropylene (PP), and polystyrene (PS) exhibit threefold increases in genes conferring resistance to fluoroquinolones, multidrug efflux pumps, rifamycins, and tetracyclines[64]. Specific bacterial taxa, such as Pseudomonas spp. identified in biofilms on both polyethylene terephthalate and polyhydroxyalkanoate particles were the predominant carriers of glycopeptide and multidrug resistance determinants, whereas Desulfovibrio spp. emerged as the principal hosts of macrolide-lincosamide-streptogramin (MLS) resistance genes on polyethylene terephthalate surfaces[65]. This selective enrichment is tightly coupled to plastisphere-specific shifts in microbial community structure: particular taxa occupy dominant ecological niches on plastic surfaces, serve as the primary ARGs hosts, and mediate horizontal gene transfer (HGT) events[64,66]. The plastisphere also sorbs diverse pollutants, intensifying selective pressure, and accelerating ARG accumulation[67].
Concomitantly, virulence factor genes (VFGs) exhibit a synergistic enrichment pattern within the plastisphere, raising additional ecological risks[60]. Compared with PE-MPs, the PBAT/PLA-MPs show higher abundances of offensive VFGs and co-localization of ARGs and VFGs within shared microbial hosts[60]. This dual enrichment indicates that MPs can simultaneously enhance resistance dissemination and traits of pathogen virulence potential, thereby amplifying their ecological risks to soil health and higher trophic organisms[68].
Overall, evidence is mounting that MP pollution is contributing to the development, transmission, and spread of ARGs. This facilitation operates through several key mechanisms (Fig. 2), including the induction of bacterial membrane permeability, the activation of cellular stress responses, the promotion of biofilm formation, and conjugation events, and the selective enrichment of resistant bacterial hosts. However, most of these studies were carried out in controlled laboratory settings, and further work is needed to better understand the processes of ARG dissemination in more complex soil fields. In addition, a lack of knowledge on potential mechanisms underlying the effects of MPs on ARGs is one of the top challenges in environmental risk assessment.
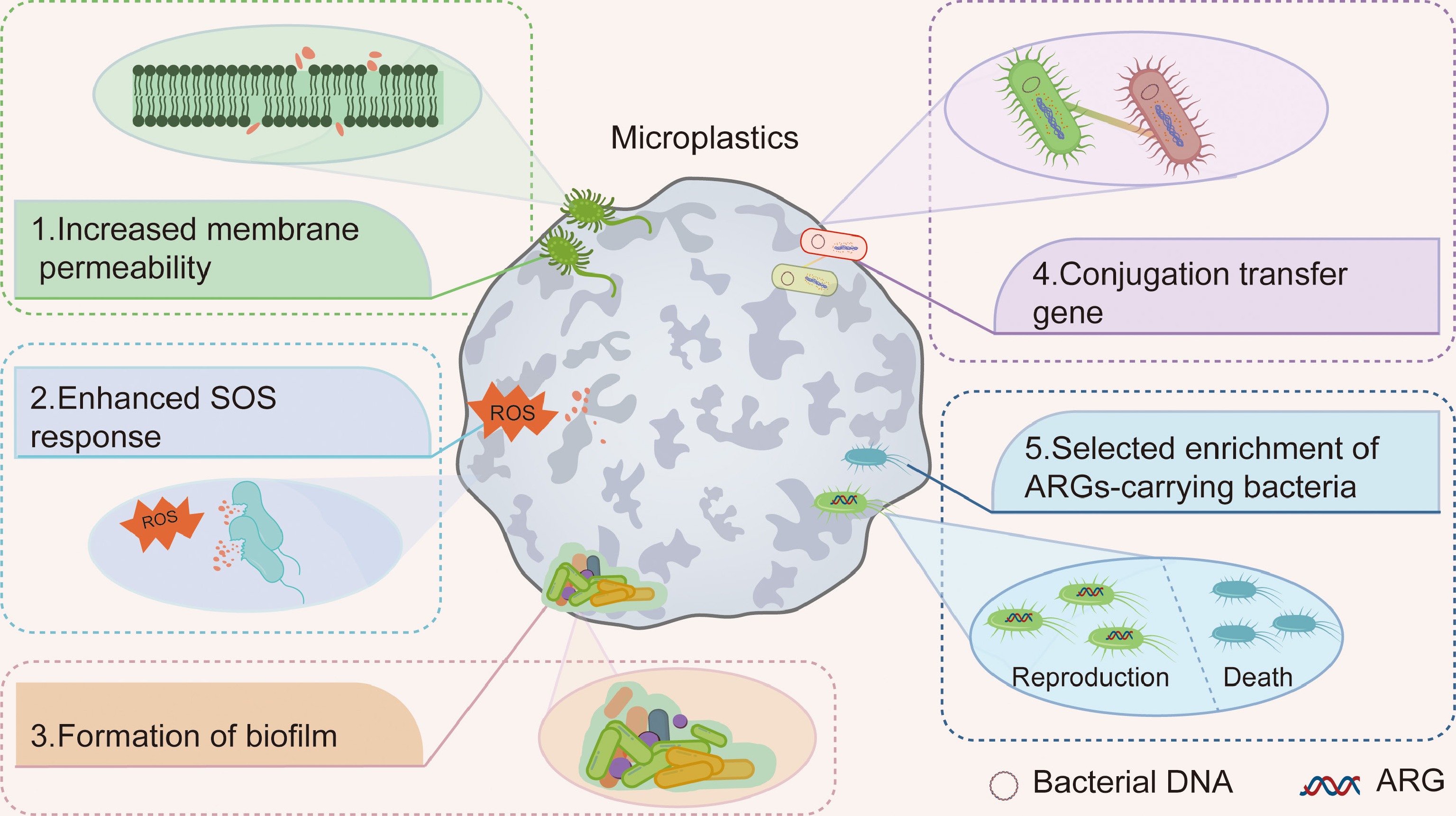
Figure 2.
Mechanisms by which microplastics (MPs) facilitate the dissemination of ARGs. MPs promote ARGs dissemination through five primary mechanisms: (1) increasing bacterial membrane permeability; (2) enhancing the SOS response; (3) facilitating biofilm formation; (4) promoting conjugation-mediated gene transfer; and (5) selectively enriching ARGs-carrying bacteria.
-
Soil animals occupy important positions in terrestrial ecosystems and participate in most below-ground ecological processes. Soil fauna guts, normally colonized by numerous microorganisms, are important reservoirs of biodiversity. It has been confirmed in many faunal groups, such as nematodes, mites, and earthworms, that dominant gut bacterial taxa included Proteobacteria, Firmicutes, Actinobacteria, and Bacteroidetes were identified[62,69]. These gut microbes contribute to host metabolism and health, as they are involved in processes such as the decomposition of organic matter, maintenance of biodiversity, and nutrient cycling of nitrogen and carbon, and degradation of pollutants[70]. Nowadays, the hidden functional genes and microbes in the soil fauna gut have largely been reported. For example, some denitrification genes, such as narG, napA, nirS, and nosZ, which were positively correlated with N2O emissions, are widely observed in the worm gut bacteria[71]. In addition, the fact that soil fauna are sensitive to changes in soils and can be used as valuable bio-indicators of soil quality, in particular in ecological assessment research, is widely recognized[72]. Recently, the gut microbiota of soil fauna has generally been suggested as a biomonitor in ecotoxicological studies[73−75]. For instance, evidence shows that exposure to silver nanoparticles disturbed gut bacterial communities of the collembolan but not its survival or reproduction, suggesting that the gut microbial community of soil fauna could be a sensitive bio-indicator of changes in contaminated soils[76].
Geophagous soil fauna residing in the soil are in direct contact with soil pollutants. Thus, residual MPs in soils can not only cause decreases in soil quality but also constitute a threat to soil animals. The concrete evidence that various soil fauna, such as earthworms[77], nematodes[78], and snails[79], ingest diverse MPs, contributing to accumulation in their body tissues and gut, has been reported in several studies. Crucially, this ingestion directly reshapes the functional gene repertoire within the gut. Studies on the gut microbiome reveal that MPs can act as selective filters for microbial functional genes. For example, exposure to PVC microplastics in a soil-earthworm system was found to significantly increase the abundance and diversity of ARGs in the earthworm gut, with pronounced enrichment of specific genes conferring resistance to aminoglycosides (aadA) and sulfonamides (sul1)[80]. Beyond the microbiome, MPs also directly perturb the host’s own gene expression. In the nematode Caenorhabditis elegans, ingestion of PS-MPs strongly up-regulated oxidative-stress-related genes (e.g., sod-1, ctl-1, gst-4) while down-regulating key genes required for intestinal development and barrier function (e.g., act-5, lin-7), thereby linking MP ingestion to genotoxic and structural damage in the host intestine[81]. In addition, the gut microbiota dysbiosis of soil fauna due to exposure to micro/nano plastics has recently aroused public concern[15]. Micro-polyvinyl chloride and nano-polystyrene exposure disturbed the bacterial diversity and composition in the mesofauna (collembolan and Enchytraeus crypticus) gut[82,83]. Similarly, tire tread particles containing a lot of pollutants led to microbial dysbiosis, and also caused an enrichment of pathogen genera in the gut of E. crypticus[84]. The functional genes within the soil fauna gut are an emerging research focus. Increasing evidence indicates that MPs can disrupt soil fauna metabolism by altering the expression of these gut microbial genes. For instance, a metagenomic study revealed that exposure to polystyrene microplastics significantly altered the abundance and profile of ARGs in the earthworm gut, demonstrating a direct impact of MPs on the functional gene repertoire of the gut microbiota[17]. Moreover, MPs act as vectors, altering the impact of co-existing pollutants on the gut functional genes of soil fauna. For instance, co-exposure to low-density polyethylene microplastics and the fungicide pyraclostrobin significantly increased the relative abundance of antibiotic resistance genes in both soil and earthworm guts by up to 2.15 times, compared to exposure to the fungicide alone[85]. Conversely, the presence of nano-plastics could mitigate the toxicity of arsenic to earthworm gut microbiota by reducing arsenic bioaccumulation and modifying the profiles of arsenic biotransformation genes[86].
In turn, the fate of MPs in soil ecosystems is influenced by soil fauna activity[87]. Several studies clearly showed that earthworms and collembolans could contribute to the migration and redistribution of MPs[88]. Given that soil fauna occupy key positions of soil food webs, MPs ingested and accumulated in soil animals could be transferred to higher trophic levels through the food chain[89], and this deserves special attention in future studies. In addition, plastics could be fragmented or degraded by soil animals[90]. For example, in the mealworm gut, the gut microbiome, especially Citrobacter sp. and Kosakonia sp., has the ability to chemically degrade plastics[91]. The earthworm gut has been reported to be a neglected but important micro-environment for microplastic degradation due to special microbiota and function gene activity in the gut. Recently, the sizes of LDPE plastics were observably decreased after passing through the earthworm gut, and researchers also screened some functional microorganisms, such as Rhodococcus jostii, Mycobacterium vanbaalenii, and Streptomyces fulvissimus, that can degrade MPs from the gut of earthworms[92,93]. However, for these functional degrading microbes that are known to be present in animal guts, much remains unclear about the specific related degradation functional genes, and thus, this limits our ability to predict how gut microbiota and genes respond to the changes induced by plastics.
-
Global environmental change is increasingly recognized as an important driver shaping the environmental fate and ecological impacts of MPs. Due to their persistence, mobility, and widespread distribution, MPs can be transported across terrestrial and aquatic systems through processes such as rainfall runoff and soil erosion, facilitating long-distance migration and redistribution[3,94]. Climate-related factors, including altered precipitation regimes, warming, and drought, have been shown to modify the physical behavior of MPs and their interactions with biota. For instance, Chang et al. used regression random forest models incorporating multiple microplastic characteristics and predicted that a 10 °C increase in global temperature could be associated with an increase in microplastic concentrations, from ~12,500 to ~13,400 particles m−3[95]. Drought conditions can amplify the effects of plastic fragments on plant growth, while environmental changes may also promote the release of additives and sorbed contaminants from MPs, thereby enhancing their ecological risks[96−98].
More importantly, MP-driven shifts in soil microbial carbon and nitrogen cycling may be further reshaped under global change conditions. Microbial communities are highly sensitive to global change drivers, such as warming and drought. Experimental warming has been shown to rapidly alter microbial community structure and enrich functional genes involved in carbon decomposition and nitrogen cycling, potentially accelerating soil carbon loss in diverse ecosystems, including permafrost and temperate grasslands[99,100]. The presence of microplastics may further modify and amplify the effects of global change drivers on microbial communities. For example, under drought conditions, MPs have been shown to shift microbial community composition by favoring specific taxa and reducing bacterial diversity, thereby altering genes and enzyme activities associated with carbon degradation, methane metabolism, and phosphorus cycling, leading to enhanced N2O emissions[101]. MPs altered microbial functional genes involved in carbon and nitrogen cycling (e.g., amoA, amoC, and nirK) primarily under elevated temperature (30 °C), whereas effects at ambient temperatures were not statistically significant, indicating that warming conditions modulate microplastic-driven genomic responses[102]. In some cases, microplastics may even reverse the direction of global change effects on soil microbial processes, as evidenced by contrasting responses of litter decomposition and ecosystem multifunctionality under well-watered conditions[18].
Global change impacts the temperature, precipitation, and nutrient availability in soil ecosystems, causing potential shifts in microbial communities that commonly raise the risk of pathogens, antibiotic resistome, and horizontal gene transfer[103−105]. Many novel pathogenic microbes or ARGs were observed in permafrost thawing due to global warming change[106]. Furthermore, the extreme weather, such as flooding and global warming events, can introduce some pollutants (e.g., metal[loid]s and antibiotics) and resistant microorganisms to the environments worldwide, and this will undoubtedly increase the enrichment and dissemination of ARGs[85,105]. It is noted that MPs can become a hotspot and habitat for microbiota that play an important role in ecosystem functions[107]. Concurrently, microplastics act as selective vectors that concentrate specific ARGs and promote their dissemination across environmental compartments[108]. This synergy ultimately elevates the risk of human exposure, notably through potential transmission along the food chain.
Soil ecosystems are concurrently affected by multiple global environmental change factors, but most studies focus on one or two factors. The interactive effects of multiple, co-occurring stressors therefore, remain largely unexplored. Recent research addressing multiple anthropogenic pressures reveals that combinations of global change factors create substantial uncertainty yet drive consistent directional shifts in soil biodiversity and ecosystem functions[109]. Given the growing prevalence of MPs in terrestrial environments and their documented effects on various functional genes, combined global change factors may modify how MPs influence environmental functional genes. However, information on these interactions in soil ecosystems remains scarce, which limits our understanding of the ecological consequences of MP–climate interactions.
-
Understanding how MPs influence the distribution, expression, and mobility of functional genes in soil microbiomes requires comprehensive and multidisciplinary approaches. Recent methodological advances have significantly enhanced our ability to unravel these mechanisms. Three core methodological directions are emerging:
Recent advancements in multi-omics technologies have significantly enhanced our understanding of how MPs affect soil microbial functional genes. Metagenomic and metatranscriptomic sequencing allow for comprehensive profiling of both the taxonomic structure and the functional potential of soil microbial communities exposed to MPs[110]. Tools such as KEGG, CAZy, and SARG have enabled high-resolution annotation of genes related to metabolism and resistance, revealing MP-induced alterations in nutrient cycling pathways, xenobiotic degradation capacity, and resistome configurations[111]. Additionally, high-throughput quantitative PCR (HT-qPCR), and functional gene arrays such as GeoChip facilitate precise quantification of functional gene families across environmental gradients[110−112], providing deeper insights into how MPs impact the expression of key functional genes. Importantly, microbial carbon use efficiency (CUE) is increasingly recognized as a key indicator for evaluating the effects of MPs on microbial carbon metabolism and soil carbon storage. Changes in CUE reflect shifts in how microbes allocate carbon between growth and respiration, thus offering insights into the broader impact of MPs on soil carbon dynamics[113]. These omics-based approaches have shifted the research focus from taxonomic profiling to functional capacity and activity, offering a more systemic understanding of MP-induced microbial alterations.
Another key direction involves tracing mobile genetic elements (MGEs), and virus-associated functions in the plastisphere. This approach focuses on tracking MGEs and virus-encoded auxiliary metabolic genes (AMGs), which are key mediators of HGT in MP-affected soils. MPs may serve as physical hotspots for genetic exchange. Computational tools such as VIBRANT, DramV, and CheckV have been developed to identify phage genomes, prophages, and their encoded AMGs, which often play roles in nutrient uptake or host stress responses[67,114,115]. These functions provide evidence of virus-mediated microbial adaptation to MP-induced stress. Moreover, tools like MobileOG-db and DefenseFinder allow researchers to detect MGEs and classify host defense systems, including CRISPR-Cas and restriction-modification systems[68,115]. These systems not only regulate HGT but also modulate microbial immunity, potentially influencing the dissemination of ARGs or carbon metabolism-related functions within plastisphere-associated communities. Tracing these mobile elements provides a mechanistic understanding of how MPs facilitate gene flow and microbial co-evolution in soil ecosystems.
In addition, experimental and imaging techniques have advanced the validation of functional shifts in situ. These methods bridge the gap between genomic inference and actual functional verification in microplastic-contaminated soils. Confocal laser scanning microscopy and scanning electron microscopy (SEM) provide direct visual evidence of microbial colonization and biofilm formation on MP surfaces, confirming MPs as microbial niches[116]. Furthermore, advanced isotopic labeling techniques such as Raman-D2O probing and stable isotope probing (SIP) allow identification of metabolically active microbes in MP-amended soils, enabling the direct linkage between gene presence and microbial function[117]. Raman-D2O probing identifies metabolically active microbial cells based on deuterium incorporation, and when integrated with stable isotope probing, single-cell sorting, and downstream genomic analyses, it enables the direct linkage of active microbial populations to their functional gene repertoires and metabolic pathways in microplastic-amended soils[118,119]. Experimental platforms such as soil microcosms incorporating synthetic microbial consortia or gnotobiotic systems are increasingly used to simulate realistic environmental conditions, including drought or nutrient enrichment, under microplastic exposure[112]. These controlled systems provide reproducible settings that are essential for validating predictions derived from multi-omics analyses and for elucidating how MPs shape microbial functional traits and ecological strategies.
Despite substantial progress, several methodological challenges persist. Omics-based approaches largely infer functional potential rather than confirming in situ microbial activity, and the link between functional gene abundance and realized ecosystem processes remains uncertain in MP-contaminated soils. Advanced single-cell strategies combining stable isotope probing with Raman-activated cell sorting have demonstrated the power to directly connect microbial activity, genomes, and metabolic pathways at the single-cell level, yet their application in soils is constrained by technical complexity, limited throughput, and strong spatial heterogeneity. Consequently, integrating high-throughput community-scale omics with targeted single-cell validation remains a key challenge for advancing mechanistic understanding of microplastic effects on soil functional genes.
-
MPs have emerged as pervasive contaminants in terrestrial ecosystems, where soils act as major reservoirs and long-term sinks. By reshaping microbial communities and altering functional gene repertoires, MPs influence central soil biogeochemistry processes, including carbon decomposition, nitrogen transformation, pollutant detoxification, and resistance to abiotic stress. Evidence now points to their impacts extending beyond bulk soils into specialized niches, such as the plastisphere and soil animal gut microbiomes, which serve as hotspots for functional gene enrichment, horizontal gene transfer, and pollutant co-selection. These findings underscore that MPs, both conventional and biodegradable, not only modify microbial ecology but also mediate broader ecosystem functions under interacting global change pressures. Despite rapid progress, critical knowledge gaps still remain. Current insights are largely derived from short-term laboratory experiments that fail to capture the complexity of field conditions, long-term exposures, and the co-occurrence of multiple stressors such as drought, warming, and heavy metal pollution. Moreover, the functional implications of MPs are still inferred primarily from taxonomic or metagenomic data, with limited mechanistic validation linking gene-level changes to ecosystem-scale processes. The ecological roles of plastisphere-associated viruses are also poorly understood, despite their potential to accelerate gene flow and microbial adaptation.
Future research should pursue several directions. First, long-term and multi-factorial field studies across spatial scales are essential to comprehensively disentangle microplastic effects under realistic environmental scenarios. Second, the distinct impacts of biodegradable and conventional MPs must be systematically elucidated. Emerging evidence reveals a fundamental dichotomy between conventional MPs and their biodegradable counterparts. Conventional MPs can destabilize soil aggregates and redistribute organic carbon, whereas biodegradable MPs often maintain aggregate structure and enhance carbon content across size classes[85]. These contrasting outcomes likely arise from divergent microbial and viral responses. Biodegradable MPs tend to enrich copiotrophic taxa and upregulate genes for glycolysis and amino acid metabolism, while conventional MPs favor oligotrophic communities and enhance genes for complex carbohydrate metabolism[115]. Critically, these microbial shifts may be reinforced by distinct virus–host interactions. Soils amended with conventional MPs exhibit an increased proportion of lytic viruses and a decrease in microbial CUE, whereas biodegradable MP amendments foster lysogenic viruses, AMGs, and stable CUE[115]. Future work should therefore explicitly compare these MP categories by integrating field carbon monitoring with mechanistic studies targeting microbial functional genes, viral dynamics, and key physiological parameters like CUE. Third, advancing active microbiome approaches (e.g., Raman-D2O probing, metatranscriptomics) in combination with metagenomics will allow more precise exploration of the mechanisms by which MPs alter soil microbial functions. The functional gene profiling should be integrated with isotopic tracing and experimental validation to directly link it to real soil carbon and nitrogen fluxes, thereby enabling more quantitative attribution of microplastic impacts to soil biogeochemical processes. Fourth, integration of viral dynamics into microplastic research will be critical for elucidating how viruses, as key regulators of microbial communities, shape functional gene enrichment and metabolic potentials in plastic-associated environments. Future efforts must scale from local experiments to global assessments, embedding microplastic-microbe interactions and their ecological consequences within the broader context of climate change. Such insights are vital for guiding plastic governance, maintaining soil biogeochemical functions and resilience under escalating microplastic pressures in the Anthropocene.
-
The authors confirm their contributions to the paper as follows: Hongtao Wang: funding acquisition, conceptualization, methodology, investigation, writing-original draft. Lijuan Ma: conceptualization, investigation, visualization, writing-original draft. Lihong Xie: investigation, visualization. Ting Xie: investigation. Sha Zhang: investigation, writing–review & editing. Tiangui Cai: investigation. Lu Wang: funding acquisition, conceptualization, supervision, project administration. All authors reviewed the results and approved the final version of the manuscript.
-
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
-
This work was financially supported by the Postgraduate Education Reform and Quality Improvement Project of Henan Province (YJS2026YBGZZ36), Natural Science Foundation of Henan Province (252300421833) and National Natural Science Foundation of China (42207013).
-
The authors declare that they have no conflict of interest.
-
# Authors contributed equally: Hongtao Wang, Lijuan Ma
Full list of author information is available at the end of the article. - Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Wang H, Ma L, Xie L, Xie T, Zhang S, et al. 2026. Effects of microplastic on soil ecosystems: a perspective from functional genes. Environmental and Biogeochemical Processes 2: e008 doi: 10.48130/ebp-0026-0003
Effects of microplastic on soil ecosystems: a perspective from functional genes
- Received: 30 October 2025
- Revised: 29 December 2025
- Accepted: 12 January 2026
- Published online: 12 February 2026
Abstract: The extensive production and persistence of microplastics (MPs) have resulted in their widespread accumulation in terrestrial ecosystems, with soils serving as critical sinks and active interfaces. As emerging pollutants, MPs can alter soil physicochemical properties and microbial habitats, raising concerns about their effects on microbial functional genes that regulate key biogeochemical processes. This review synthesizes recent progress in understanding how MPs influence soil microbial functional genes and their ecological consequences. MPs modify soil structure, enzyme activity, and microbial community composition, leading to shifts in the diversity and expression of genes related to nutrient cycling and antibiotic resistance. Such changes disrupt microbial-mediated carbon and nitrogen transformations and consequently affect soil ecosystem functioning. The plastisphere and soil fauna gut microbiomes have been identified as hotspots for microbial activity and gene exchange, enhancing the redistribution of functional genes within soil environments. Moreover, environmental factors such as warming, drought, and elevated CO2 interact with MPs, further amplifying or modifying these impacts under global change conditions. Finally, this review highlights the importance of integrating multi-omics, computational, and imaging approaches to deepen understanding of microplastic-microbe interactions, and their implications for soil biogeochemical processes and ecosystem functioning.