-
Fresh-cut vegetables are in high demand worldwide for their nutritional benefits and convenience. Among these, fresh-cut lotus (Nelumbo nucifera Gaertn.) root has gained popularity as a novel, minimally processed product in China[1]. However, microbial contamination and enzymatic browning pose major challenges for the industry. Mechanical injury during processing disrupts cellular compartmentalization, releasing vacuolar phenolics and oxidative enzymes into the cytosol. As observed in lettuce stems, this triggers rapid metabolic shifts and oxidative cross-talk, culminating in discoloration and economic losses[2,3]. This browning cascade is equally severe in white-fleshed vegetables such as lotus root, where high phenolic content and intrinsic enzymatic activity accelerate quality deterioration[4].
Polyphenol oxidase (PPO) is established as the principal enzyme responsible for enzymatic browning in fresh-cut fruits and vegetables[3]. This copper-containing metalloenzyme catalyzes the oxidation of phenolic compounds to highly reactive o-quinones, which subsequently polymerize into brown melanins, compromising product quality[5]. The overall browning process can be modulated by other enzymes. Phenylalanine ammonia-lyase (PAL) drives the synthesis of phenolic substrates, while peroxidase (POD) can further oxidize phenolics in the presence of H2O2, potentially acting in synergy with PPO[6]. Despite this foundational knowledge, the precise regulatory mechanisms controlling phenolic metabolism-related enzyme activities in complex postharvest systems, such as fresh-cut lotus root, remain insufficiently resolved.
Conventional anti-browning strategies (e.g., chemical inhibitors, thermal processing) often compromise sensory attributes or lack eco-compatibility. Irradiation has emerged as a promising non-thermal alternative, capable of eliminating pathogens while extending shelf life[7,8]. Low–to–medium doses (< 5 kGy) may further inhibit browning, as evidenced in shredded cabbage, where γ-irradiation (2 kGy) suppressed PAL activity and gene expression[9]. While γ-irradiation has been demonstrated to effectively delay browning and decay in various fruits and vegetables[9,10], its efficacy and underlying mechanism for highly browning-sensitive root vegetables like lotus root are less comprehensively studied. Existing research on lotus root has primarily focused on the changes in sensory and physicochemical properties during storage post-irradiation. The current mechanistic understanding of irradiation-induced PPO inhibition is often attributed to indirect causes such as gene expression, downregulation, or oxidative stress, while direct experimental evidence on whether and how irradiative energy disrupts the secondary and tertiary structure of PPO, leading to the loss of catalytic function, is still lacking. Therefore, this study evaluated the effects of 60Co-γ irradiation (1–3 kGy) on microbial safety, color quality, and browning-related metabolism in fresh-cut lotus root during storage. It is hypothesized here that, irradiation inhibits browning in fresh-cut lotus root by inducing conformational changes in PPO, disrupting its catalytic function through structural alterations. This multi-scale mechanistic investigation will not only provide a solid theoretical basis for applying γ-irradiation to preserve lotus root, but also offers new insights into the universal molecular mechanisms by which irradiation technology controls enzymatic browning in fresh produce.
-
Lotus roots (Nelumbo nucifera Gaertn. cv. Qian Shui Ou) were harvested from a commercial field located in Guangzhou, Guangdong, China in October 2024, and transported to the laboratory within 3 h under an ice bath. Uniform roots without mechanical damage were washed with tap water and sliced into pieces with the thickness of 0.5 cm using a sharp knife. The slices were divided into four groups packed separately in polypropylene plastic bags and treated with 60Co-γ irradiation at 1, 2, and 3 kGy. Irradiation was performed at a dose rate of 1.0 kGy/h at ambient temperature (approx. 25 °C). Slices without radiation treatment were used as a control. The treated samples were stored at 25 °C with a relative humidity of 65% ± 5% for 9 d. Each bag contained approximately 500 g of samples. Three replicates were applied for each treatment.
Microbial counts
-
Fresh-cut lotus root samples (25 g) were homogenized with 225 mL of sterile physiological saline (0.85% NaCl). Following serial decimal dilution, aliquots were spread-plated onto plate count agar (PCA). After incubation at 37 °C for 48 h, visible colonies were enumerated. Total bacterial counts were calculated as log10 CFU/g. Data is presented as mean values ± standard deviation from three biological replicates (n = 3).
Color measurement and browning degree analysis
-
Surface color of fresh-cut lotus root was quantified using a chromameter (CR-400, Konica Minolta, China). The instrument was preheated for 15 min and calibrated prior to measurements. For each sample, the measurement head was placed in direct contact with three randomly selected positions on the cut surface. Color parameters (L, a*, b*) were recorded, with total color difference calculated as ΔE, where higher ΔE values indicate greater visual discoloration.
Five grams of tissue was homogenized in 20 mL ice-cold distilled water using a polytron homogenizer. The slurry was centrifuged (5,000 g, 20 min, 4 °C) in a refrigerated centrifuge (Eppendorf 5430R, Germany). The resulting supernatant was equilibrated to 25 °C for 5 min before measuring absorbance at 410 nm (A410). Browning index (BI) was calculated as A410 × 10.
Total phenolic compound content
-
Total phenolic content was assessed using the Folin-Ciocalteu assay with minor modifications. Briefly, 3.0 g of sample material was homogenized in 30 mL of 60% (v/v) aqueous ethanol. The homogenate was centrifuged at 10,000 g for 5 min at 4 °C. The supernatant was adjusted to a final volume of 40 mL with extraction solvent (60% ethanol). 0.125 mL of diluted extract was combined with 0.625 mL deionized water. After adding 0.125 mL Folin-Ciocalteu reagent, the mixture was vortexed for 10 s and incubated in darkness (25 °C, 3 min). Subsequently, 1.25 mL of 7% (w/v) sodium carbonate (Na2CO3) and 1.0 mL deionized water were introduced. Following thorough mixing, reaction mixtures were developed in a temperature-controlled water bath at 25 °C for 90 min. Absorbance was measured at 760 nm against a reagent blank using a UV-1800 spectrophotometer (Shimadzu, Japan). Total phenolic content was calculated based on a gallic acid standard curve and expressed as milligrams of gallic acid equivalents per kilogram of fresh weight (mg/kg FW).
Soluble quinone content
-
Soluble quinone content was quantified following Wang et al. with modifications[11]. Fresh-cut lotus root tissue (5.0 g) was homogenized in 20 mL of absolute ethanol. The homogenate was centrifuged at 10,000 g for 10 min (4 °C). The supernatant was collected and immediately subjected to spectrophotometric analysis. Absorbance measurements were recorded at 437 nm using a UV-1800 spectrophotometer (Shimadzu, Japan) with anhydrous ethanol as a blank. Soluble quinone levels are expressed as absorbance units at 437 nm.
PAL, POD, and PPO activity assay
-
PAL activity was determined using the method described by Zhang et al. with minor modifications[1]. Approximately 2 g of sample tissue was homogenized in 5 mL of 0.05 mmol/L sodium borate buffer (pH 8.8) containing 0.5 g polyvinylpyrrolidone (PVP), 5 mmol/L 2-mercaptoethanol, and 2 mmol/L EDTA, using a pre-chilled mortar. The homogenate was centrifuged at 12,000 g for 20 min at 4 °C, and the resulting supernatant was used as the crude enzyme extract. The reaction mixture for the PAL assay comprised 3.5 mL of 0.1 mol/L sodium borate buffer (pH 8.7), 1 mL of 10 mmol/L L-phenylalanine, and 0.5 mL of the enzyme extract. The change in absorbance at 290 nm was recorded, and one enzyme unit (U) was defined as a 0.01 absorbance change per min per gram of fresh weight.
POD and PPO activities were assayed following the method of Li et al.[12]. An aliquot of 3.0 g samples was homogenized with 50 mL of ice-cold 0.05 M phosphate-buffered saline (PBS, pH 7.0) in a pre-chilled mortar. To obtain the crude enzyme extract for POD and PPO assays, the homogenate was centrifuged (10,000 g, 10 min, 4 °C) and the supernatant was collected. For POD activity determination, the reaction mixture was prepared by combining 2 mL of acetic acid buffer (with 5 mmol/L benzidine) and 1 mL of the enzyme extract. For PPO activity, the reaction system was prepared by combining 1.0 mL of supernatant, 1.0 mL of 0.1 mol/L catechol, and 1.5 mL of phosphate-buffered saline (PBS). Following a 5 min incubation at 37 °C, the POD reaction was initiated by adding 1 mL of 0.3% H2O2. POD activity was then quantified by monitoring the increase in absorbance at 470 nm for 2 min. PPO activity was determined by measuring the absorbance change at 420 nm. Similar to PAL, one unit (U) of PPO/POD activity was defined as the amount of enzyme that caused an increase in absorbance of 0.01 per min under the assay conditions. The activity was expressed per gram of fresh weight (U/g FW).
PPO extraction and partial purification
-
Following irradiation, PPO was then individually extracted and purified from the tissues of the treatment group. Lotus root slices were homogenized with a Bamix mixer to make a puree. The tissue homogenate was washed twice with pre-chilled PBS (pH 7.4) to remove impurities. To obtain the crude protein extract, the sample was treated with PBS (pH 7.4), supplemented with protease inhibitors (1 mM PMSF and 5 mM EDTA) for lysis, and incubated on ice for 30 min. The mixture was then centrifuged at 12,000 g for 30 min at 4 °C, and the supernatant was collected. Following the gradual addition of solid ammonium sulfate to 40% saturation, the crude protein solution was stirred on ice until fully dissolved and then stored at 4 °C for 8 h. The resulting precipitate was collected by centrifugation, resuspended in PBS buffer, and the supernatant was discarded. The samples were further purified by size-exclusion chromatography using an ÄKTAprime plus system equipped with a Superdex 200 column. The analysis was carried out under the following conditions: a mobile phase of 50 mM PBS buffer, a flow rate of 1 mL/min, and an injection of 5 mL of dialyzed sample. Protein concentration was determined by the Bradford method, using absorbance at 595 nm and a bovine serum albumin (BSA) standard curve. The final partially purified PPO had a concentration of approximately 3 mg/mL. For spectroscopic analyses, this stock was diluted with 50 mM PBS to the required working concentrations (0.5 mg/mL for CD, and 0.1 mg/mL for fluorescence). Simultaneously, the purity of the protein extract was evaluated using sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) (Supplementary Fig. S1).
PPO enzyme gene expression (qRT-PCR)
-
Following total RNA extraction (Trizol method) and quality assessment (purity and concentration), cDNA was synthesized using a reverse transcription kit (Takara, Dalian, China) as per the manufacturer's instructions. Gene expression was subsequently analyzed by real-time PCR (RT-PCR). The qRT-PCR assay was conducted using a 7500 Real-Time PCR System (Applied Biosystems, USA). The program is comprised of the following steps: an initial incubation at 37 °C for 2 min, followed by a reverse transcription step at 55 °C for 15 min and enzyme inactivation at 85 °C for 5 min. Amplification proceeded for 40 cycles with denaturation at 95 °C for 10 s, annealing at 56 °C for 10 s, and extension at 72 °C for 10 s. For each sample, reactions were set up in triplicate to ensure the reproducibility of the results. The mRNA expression level was normalized to the expression level of the housekeeping gene GAPDH (forward, GTCACCACTGACACGTTGGC, and reverse, AGTGGTCGTTGAGGGCAATG). The primer was used for qRT-PCR: N1PPO (Nelumbo nucifera polyphenol oxidase) forward, TTGGAAGCCGAAGATGA and reverse, CACGAAGCGAACACTATC.
Fluorescence emission spectra analysis
-
The protein solution was diluted to 0.1 mg/mL with 50 mM PBS (pH 7.4) for analysis. Fluorescence emission spectra (280–400 nm) was recorded at 25 °C on an RF-600 spectrophotometer (Shimadzu, Tokyo, Japan) using an excitation wavelength of 265 nm. The spectra was acquired with both excitation and emission slits set to 1 nm and a scanning speed of 6,000 nm/min, and were baseline-corrected against a PPO-free control.
Circular dichroism spectrum (CD)
-
CD spectra of the protein solution (0.5 mg/mL in 50 mM PBS, pH 7.4) were acquired using a CHIRASCAN spectropolarimeter (Applied Photophysics, UK), scanning from 190 to 250 nm at a speed of 50 nm/min.
Data analysis
-
The results from at least three independent experiments are reported as mean ± standard deviation. Statistical comparisons were performed by one-way ANOVA with Duncan's multiple range test in SPSS Statistics 17, considering differences significant at p < 0.05.
-
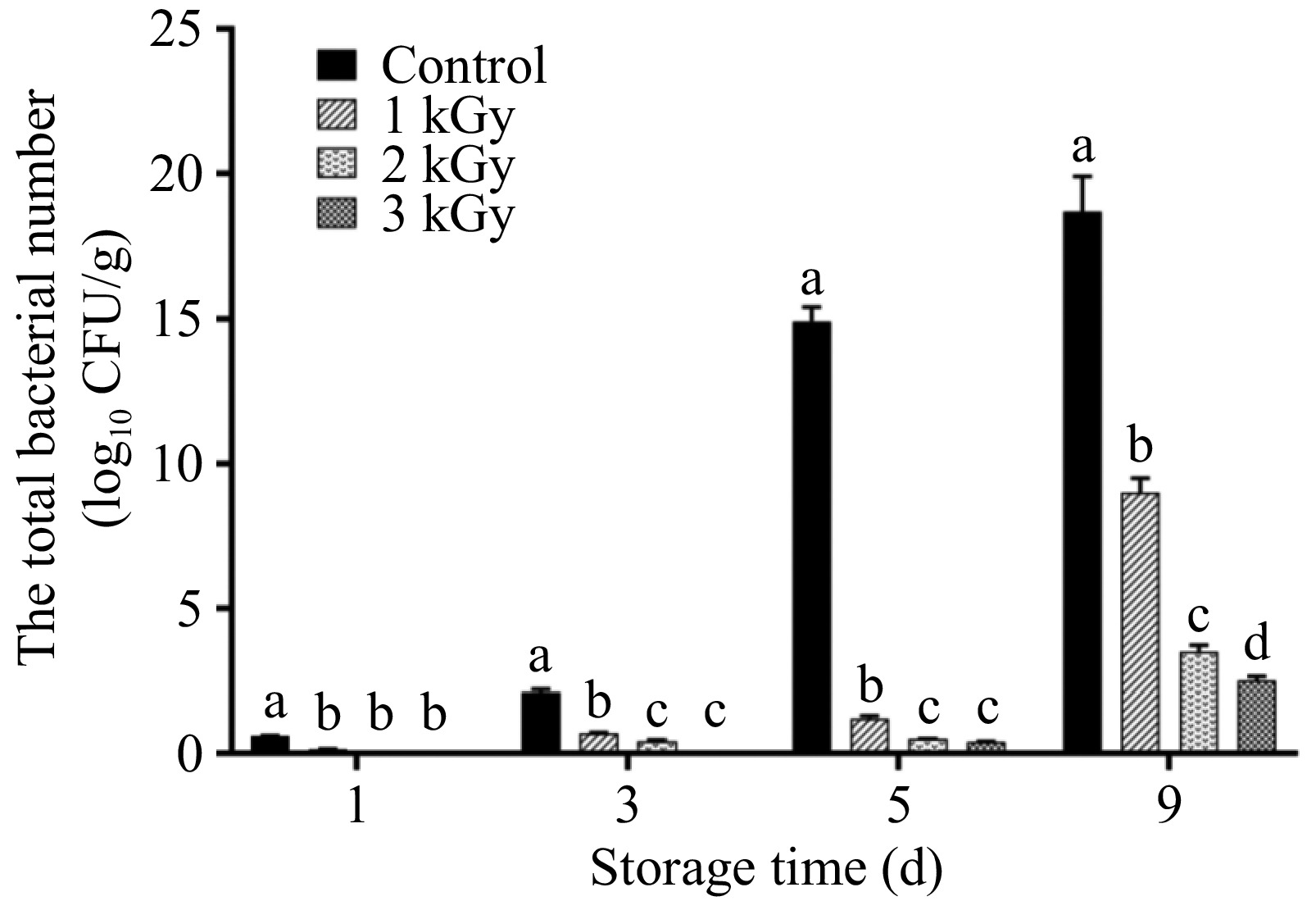
Microbial contamination is a significant challenge in the storage of fresh-cut vegetables. Minimal processing operations such as cutting and dicing, critically determine initial microbial load and the subsequent pattern of microbial growth[13]. Contamination by human, animal, or environmental pathogens can occur during primary production and processing[14], with fresh-cut produce known to harbor pathogens such as Escherichia coli O157:H7, Salmonella enterica, and Listeria monocytogenes[13,15]. Different produce items vary in their ability to support bacterial growth. For example, in fresh-cut lettuce, storage for just 3 d led to a substantial increase in total colony count (exceeding 5 log10 CFU/g), notably from Pseudomonas, resulting in significant sensory quality deterioration[16]. Similarly, in fresh-cut lotus root, the cutting process disrupts the tissue, creating an environment conducive to microbial proliferation. As shown in Fig. 1, total viable counts in untreated (control) lotus root increased rapidly, reaching 15.2 log10 CFU/g by day 5. In sharp contrast, irradiation significantly (p < 0.05) suppressed growth at this critical stage, with the values of 2.6, 1.1, and 0.8 log10 CFU/g for the 1, 2, and 3 kGy treatments, respectively (Fig. 1). This dose-dependent inhibition persisted throughout storage. On storage day 9, lotus root treated with 1 kGy irradiation reduced the total bacterial number to only half the level of the control group. The total viable counts in the 2, and 3 kGy treatments remained below 5.0 log10 CFU/g, with the 3 kGy treatment demonstrating the strongest antimicrobial efficacy. These results clearly indicate that irradiation is a highly effective bactericidal treatment for controlling microbial populations and enhancing the shelf-life of lotus root slices.
Effects of irradiation on color and browning index of fresh-cut lotus root
-
The development of browning was monitored in the lotus root slices throughout storage (Fig. 2). Irradiation from different sources i.e., γ-ray, ultraviolet light, and diode laser has been proven to inhibit browning in fresh-cut vegetables such as celtuce, cabbage, and bamboo shoot[9,17,18]. In this study, browning occurred in the control treatment after 3 d of storage, whereas slices treated with 2, and 3 kGy irradiation exhibited slight browning. This visual improvement was quantitatively confirmed by color measurements, as shown in Table 1. The L* value (lightness) decreased over time in all groups, but the decline was significantly mitigated by irradiation. At the end of storage, the L* values of samples treated with 1, 2, and 3 kGy were 1.4%, 12.4%, and 22.9% higher than the control, respectively. Consequently, the 3 kGy treatment showed the smallest overall color change (ΔE). Consistent with color parameter trends, the browning index (BI) increased with prolonged storage, rising from 4.87 to 15.57 over the 9 d storage period. Starting from the third day of storage, irradiation exerted a significant inhibitory effect on browning. Specifically, BI of samples treated with irradiation doses of 1, 2, and 3 kGy was respectively lower by 19.6%, 25.3%, and 28.6% compared to the control group. Notably, the inhibitory effect of irradiation on browning was dose-dependent during the late stage of storage. The 3 kGy dose demonstrated the most pronounced effect, with its final BI being approximately half of that in the control group. The observed browning degree showed a strong positive correlation with PAL and POD activities, but a significant negative correlation with total phenol content, suggesting that phenol consumption by these enzymes may accelerate browning[19]. Therefore, we further examined the activities of the relevant enzymes.

Figure 2.
Pictures of fresh-cut lotus root subjected to 60Co-γ irradiation at 1, 2, and 3 kGy during storage.
Table 1. Changes in color and browning index of fresh-cut lotus root during storage.
Parameters Storage time (d) Treatments Control 1 kGy 2 kGy 3 kGy Color
(L*)0 50.91 ± 1.99b 50.21 ± 0.85a 50.98 ± 0.77b 50.13 ± 1.58ab 3 41.81 ± 1.32a 40.48 ± 1.07b 40.34 ± 1.41b 40.68 ± 0.78b 5 40.02 ± 1.94a 38.98 ± 2.00a 39.70 ± 2.08a 40.23 ± 2.00a 9 31.70 ± 1.85c 32.14 ± 1.60c 35.62 ± 2.66b 38.98 ± 2.28a Color
(ΔE)0 45.51 ± 1.61a 44.08 ± 1.80a 45.46 ± 1.73a 44.48 ± 1.89a 3 47.74 ± 1.33a 46.67 ± 0.89b 46.6 ± 1.11b 46.33 ± 0.63b 5 54.67 ± 1.86c 56.96 ± 0.84a 54.98 ± 0.68bc 56.1 ± 1.49ab 9 44.28 ± 2.07a 36.64 ± 1.94c 40.19 ± 2.73b 35.88 ± 2.07c Browning Index (BI) 0 4.87 ± 0.80a 4.87 ± 1.28a 4.80 ± 1.12a 4.83 ± 1.15a 3 8.94 ± 2.59c 7.19 ± 0.73b 6.68 ± 0.92ab 6.38 ± 1.62a 5 14.07 ± 2.07c 7.37 ± 0.31a 7.13 ± 0.82ab 6.91 ± 2.06b 9 15.57 ± 2.73a 10.88 ± 0.48c 9.84 ± 0.52c 7.98 ± 0.67b Different letters in the same row indicate significant difference (p < 0.05). Phenolics and soluble quinone content
-
Phenolic substances are the substrates for browning. The total phenol content in differently treated samples gradually decreased during storage (Fig. 3). This is contrary to previous studies where total phenol content documented a rise in these compounds over time in wounded fresh-cut lotus roots[1,19]. In the present study, this degradation was significantly slower in irradiated samples. The 3 kGy treatment was most effective, best preserving phenolic content during the critical first 3 d of storage.
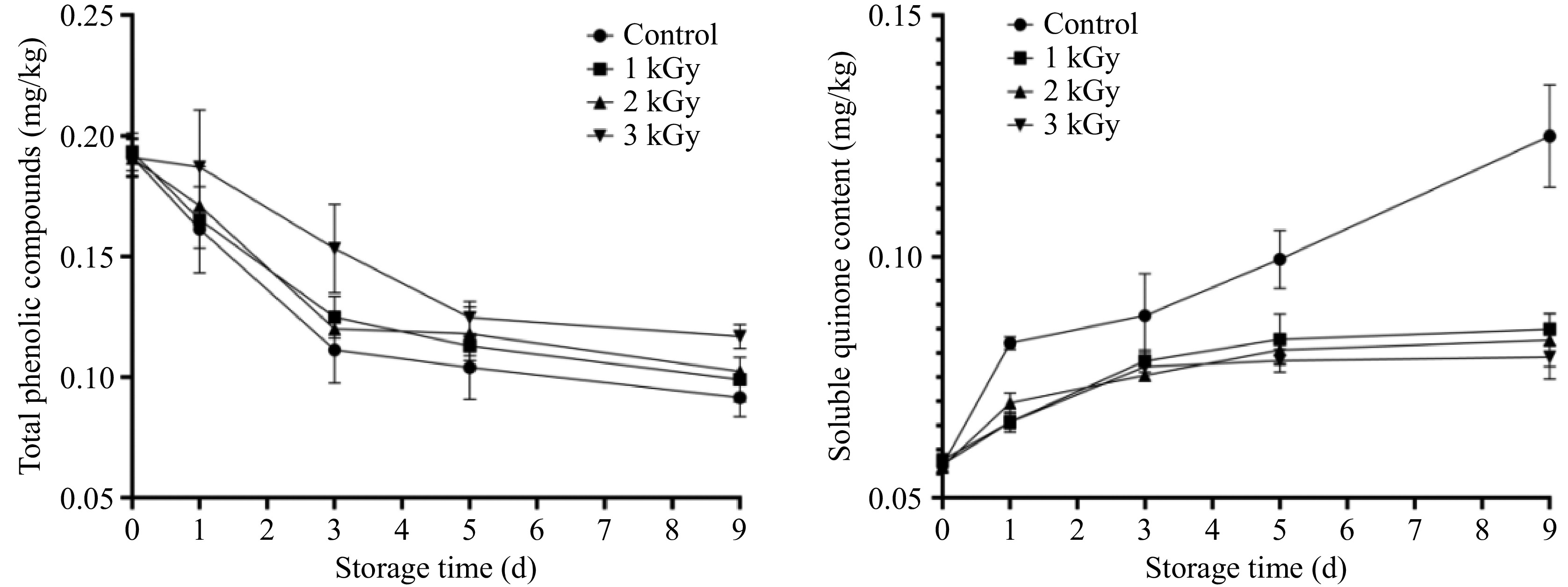
Figure 3.
The dynamic changes of total phenolic compounds, and soluble quinone content of lotus root slices subjected to irradiation during storage.
Quinones, formed via phenolic oxidation, are direct browning precursors. Soluble o-quinone accumulation strongly correlates with visual deterioration, exhibiting an inverse relationship with color brightness (L* value) in horticultural products[16]. Concurrently, the concentration of soluble o-quinones, the direct precursors to brown pigments, increased rapidly, peaking on day 5 (Fig. 3). The accumulation was drastically higher in the control group, which reached quinone levels 2.8-fold greater than the irradiated groups at the peak. By day 9, quinone concentrations in controls doubled those in irradiated samples, confirming that irradiation persistently suppressed oxidation kinetics. The marked reduction in quinones led to a corresponding decrease in browning intensity. This result was perfectly aligned with the measurements of color difference and browning index.
60Co-γ irradiation had limited effects on PAL and POD activities
-
Phenolic metabolism alterations are widely recognized to influence enzymatic browning in cut vegetables[3,20]. The activity of PAL, the first enzyme in the phenolic-synthesizing phenylpropanoid pathway, showed a dynamic change in fresh-cut lotus root during storage, initially increased during storage, peaking on day 5 before declining (Table 2). This pattern aligns with studies on cut lettuce and shredded cabbage, where wound-induced stress typically enhances PAL activity[3,9]. However, 60Co-γ irradiation did not significantly alter PAL activity relative to the control at any point during storage. This lack of PAL response to irradiation-induced stress suggested that PAL activity may not be the primary factor regulating enzymatic browning in lotus root slices. The absence of PAL modulation by irradiation in lotus root contrasted sharply with its reported efficacy in cabbage[9], where PAL suppression directly mediated browning inhibition. This discrepancy underscored that irradiation's anti-browning mechanisms were highly commodity-dependent. Similarly, peroxidase (POD) activity was not significantly affected by irradiation treatment (Table 2). In root crops like lotus, which naturally accumulate high levels of preformed phenolics[21], browning control may require targeting oxidation enzymes (e.g. PPO) rather than phenolic biosynthesis pathways.
Table 2. The activities of PAL and POD in lotus roots slices subjected to different doses of 60Co-γ irradiation during storage.
Parameters Storage
time (d)Treatments (U/g FW) Control 1 kGy 2 kGy 3 kGy PAL
activity0 6.05 ± 0.67a 6.65 ± 0.57a 6.15 ± 1.32a 6.45 ± 2.89a 3 10.71 ± 0.06a 10.89 ± 1.30a 9.09 ± 1.22a 9.21 ± 1.65a 5 10.42 ± 0.71a 12.02 ± 0.73b 9.97 ± 0.24a 10.58 ± 0.29a 9 9.46 ± 0.68a 9.23 ± 0.57a 9.75 ± 1.86a 9.98 ± 0.72a POD
activity0 39.77 ± 5.38a 39.91 ± 3.30a 38.23 ± 3.27a 38.76 ± 4.19a 3 56.84 ± 8.71a 54.75 ± 1.86a 56.38 ± 1.95a 53.49 ± 1.13a 5 53.48 ± 1.13a 59.51 ± 9.73b 59.70 ± 8.11b 53.48 ± 4.36a 9 51.47 ± 2.10a 55.62 ± 0.97a 53.16 ± 5.62a 54.89 ± 8.75a Different letters in the same row indicate significant difference (p < 0.05). 60Co-γ irradiation inactivated PPO activity
-
All samples showed a significant storage-dependent increase in PPO activity, while this elevation was notably mitigated by 60Co-γ irradiation (Fig. 4). The control group exhibited a 196% rise in PPO activity, compared to an average increase of only 72% in irradiated groups. On day 5 of storage, PPO activity in the control was 1.7 times higher than in irradiated samples. The inhibitory effect of 60Co-γ irradiation on PPO activity was dose-dependent, with the 3 kGy dose being the most effective. At the molecular level, qRT-PCR analysis revealed that PPO transcript abundance in the 3 kGy irradiated group was only 50.2% of that in the control, indicating 60Co-γ irradiation downregulated PPO transcription to inhibit its activity increase.
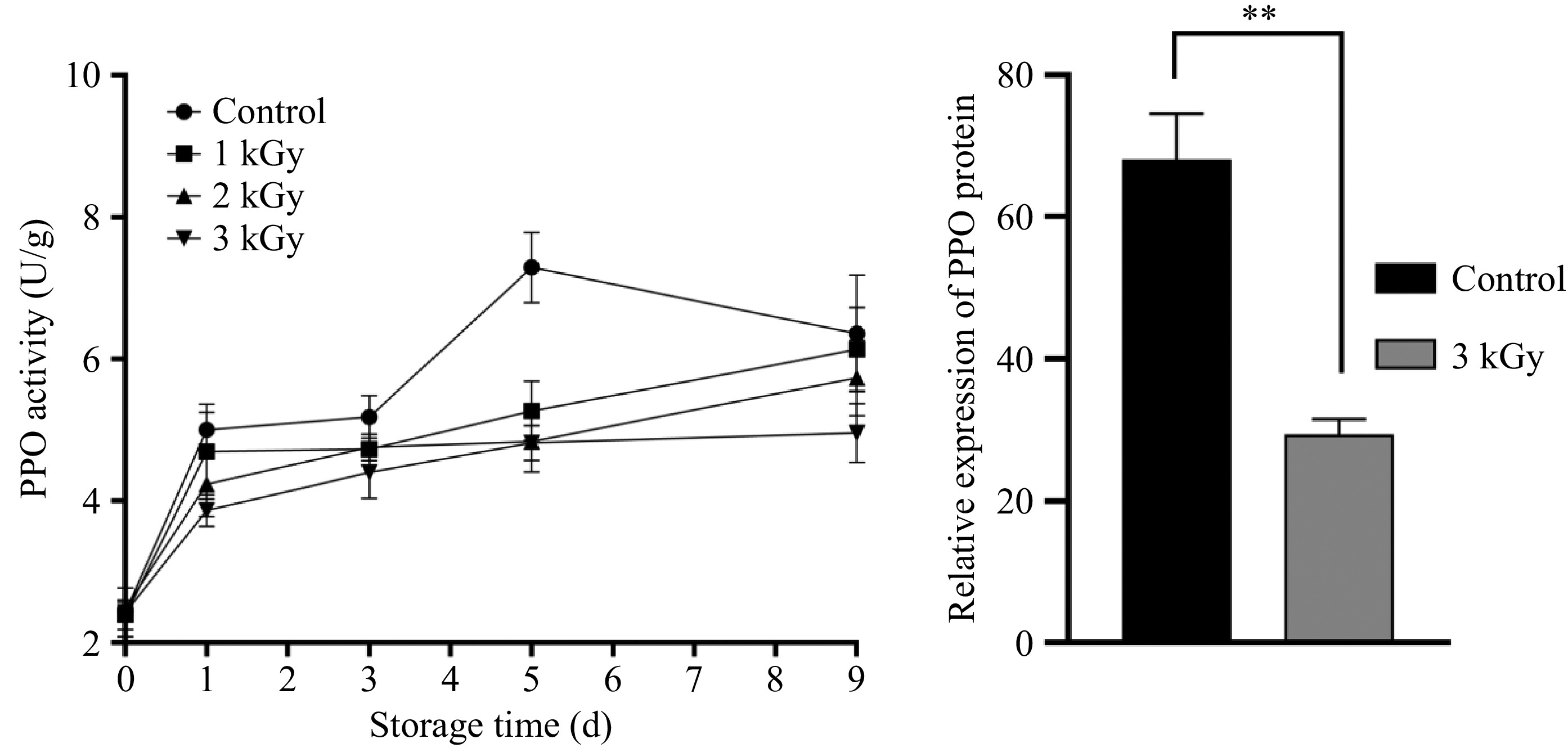
Figure 4.
The PPO activity, and relative gene expression of PPO of lotus root slice samples with and without irradiation. ** Represents significant different between treatments (p < 0.05).
Effect of irradiation on the secondary structure of PPO
-
Conformational changes in PPO induced by gamma irradiation were analyzed by far-UV circular dichroism (CD) spectroscopy. According to CD spectral analysis (Table 3), the native PPO in lotus root was characterized by 35% α-helix and 20% β-sheet content. The present findings on the secondary structure thus confirm the predominant α-helical conformation of PPO[22]. A significant conformational change was observed following the 3 kGy treatment, characterized by a 48.6% reduction in α-helix and a concurrent 40% increase in β-sheet content. Analysis of the CD spectra demonstrated that high-dose irradiation (3 kGy) induced a distinctive structural transition in the protein, marked by the dominance of β-sheet conformations. The CD spectroscopic data were in full agreement with the enzymatic activity results, confirming that the conformational changes in PPO directly underlie the observed alterations in its activity. According to Chutia et al., PPO showed higher vulnerability to DBD plasma inactivation compared to POD in tender coconut water[23]. In this study, gamma irradiation was found to alter protein conformation, leading to a decrease in α-helix and a concomitant increase in β-sheets and random coils. The active site copper of PPO molecules was coordinated by three histidine residues contributed from the four α-helices[24]. Consequently, the disruption of the α-helix structures of PPO might result in the breakdown of active sites and the inactivation of PPO.
Table 3. Effect of 60Co-γ irradiation on the percentage content of lotus PPO secondary structures.
Treatment α-Helix (%) β-Sheet (%) β-Corner (%) Random coil (%) Control 35.4 ± 0.4a 20.6 ± 0.5a 14.0 ± 0.1a 30.1 ± 0.9a 3 kGy 18.4 ± 0.2b 28.5 ± 0.1b 14.2 ± 0.1a 39.0 ± 0.3b The different letters in the same row means significant difference (p < 0.05). Effect of irradiation on the tertiary structure of PPO
-
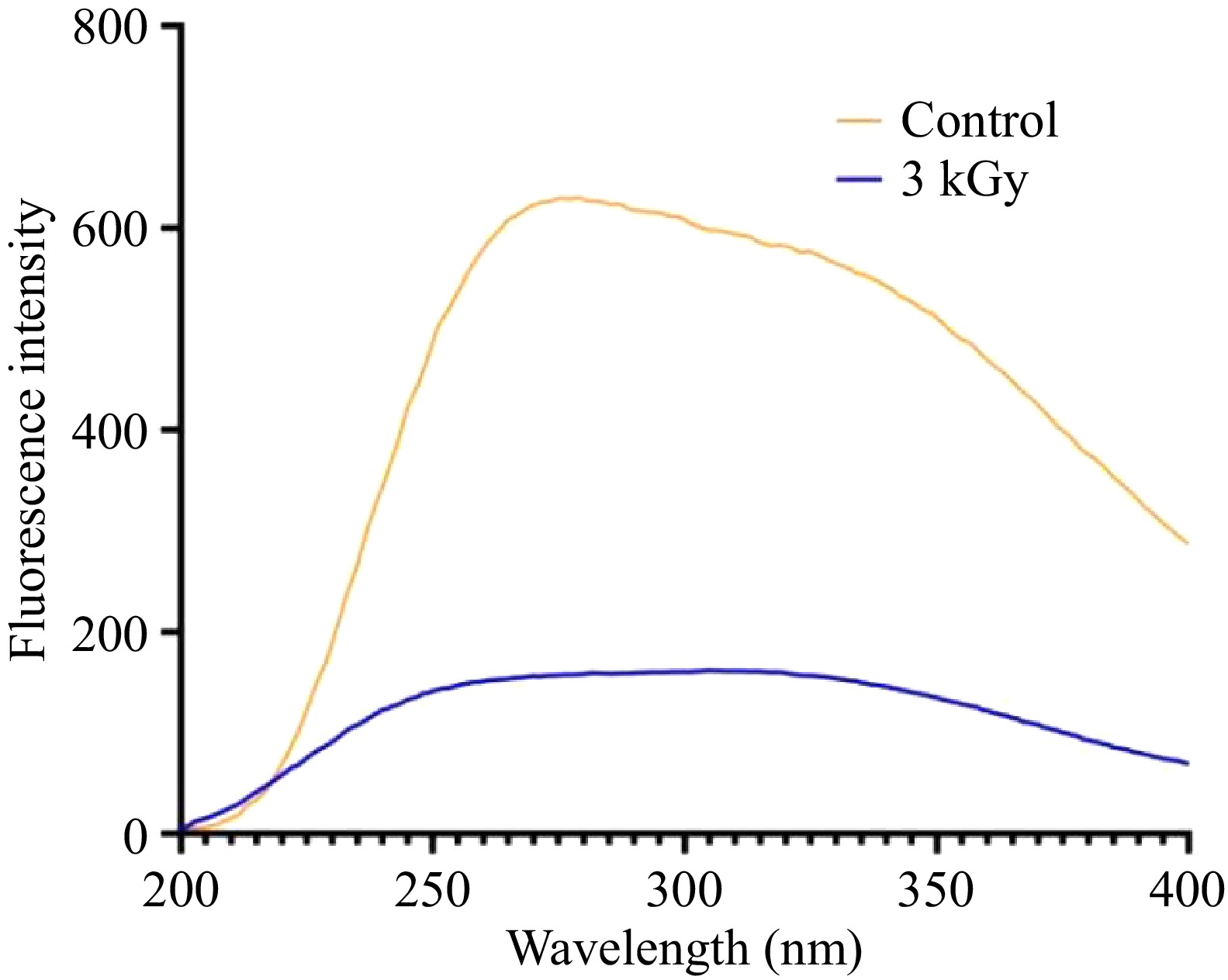
Tertiary structural changes in PPO induced by gamma irradiation were assessed by intrinsic fluorescence spectroscopy (Fig. 5). Compared to the native PPO, irradiation treatment caused a marked decrease in fluorescence intensity. Furthermore, a clear redshift in the maximum emission wavelength was observed. For the 3 kGy treatment group, the emission maximum shifted from 275 to 310 nm (Fig. 5). This spectral change signifies a fundamental alteration in the enzyme's conformation. The concurrent redshift in emission maximum and decrease in fluorescence intensity (Fig. 5) confirmed that gamma irradiation induced microenvironmental alterations around aromatic residues, leading to the disruption of PPO's native tertiary structure.
-
The present findings demonstrate that 60Co-γ irradiation effectively maintains the postharvest quality of fresh-cut lotus root primarily by inhibiting the PPO-mediated enzymatic browning pathway, rather than through a generalized suppression of phenolic metabolism.
The core anti-browning mechanism was the targeted suppression of PPO. It was found that irradiation not only inhibited the storage-induced surge in PPO activity in a dose-dependent manner but also significantly downregulated the expression of the PPO gene itself. This dual action at the transcriptional and enzymatic levels effectively attenuates the entire browning cascade. This is consistent with transcriptomic studies in other irradiated crops, such as Lentinula edodes, where irradiation directly downregulated browning-related enzyme genes[25]. The resulting reduction in PPO activity directly explains the slower oxidation of phenolic substrates and the consequent drastic reduction in the accumulation of soluble o-quinones, which are the direct precursors of brown pigments. The strong correlation between low quinone levels and lower BI and higher lightness (L*) firmly establishes this suppressed oxidative flux as the primary cause for improved visual quality.
Unlike in shredded cabbage, where irradiation inhibits browning by suppressing PAL activity and phenolic synthesis[21], no significant effect of irradiation was observed on PAL activity in lotus root (Table 2). This indicates that the phenolic biosynthesis pathway was not the primary target of irradiation in this tissue. Interestingly, we observed a net decrease in total phenolics during storage (Fig. 3a). This contrasts with some reports where cutting stress alone, without any external physical field intervention, usually stimulates phenolic biosynthesis leading to an accumulation of phenolics in fresh-cut lotus[1,19]. This reversal of trends underscores the profound impact of γ-irradiation. In the typical wound response, the increased synthesis may outweigh oxidation. However, under γ-irradiation, the metabolic balance is shifted. The high constitutive phenolic pool in lotus root[21] is rapidly oxidized by irradiation-generated free radicals. Moreover, irradiation simultaneously and powerfully activated PPO (at least transiently during treatment), and disrupted its structure, leading to sustained high oxidative activity. It is plausible that the rate of phenolic consumption via this oxidation pathway outpaced any potential wound-induced synthesis, leading to the observed net decline. Therefore, in such a system with abundant pre-formed substrates, the critical control point logically shifts from inhibiting synthesis (as in cabbage) to preventing the oxidation of the existing phenolic pool. The results of this study confirm that targeting the oxidation enzyme (PPO) is paramount in this context.
In contrast to PPO, the activities of PAL and POD were not significantly altered by irradiation (Table 2). The lack of a POD response is notable, as a synergistic role for POD in PPO-mediated browning has been proposed in some systems[26]. Its minimal involvement here is consistent with the limited endogenous H2O2 availability typical in many fresh-cut vegetables[27], a condition exacerbated in lotus root. The critical mechanism underlying this H2O2 limitation is exemplified in fresh-cut potatoes, where browning progressed without detectable H2O2 generation, and POD activity only increased significantly upon exogenous H2O2 addition[28]. The main phenolic compound in lotus roots is gallic acid[21], and as a PPO substrate, its ability to induce hydrogen peroxide formation is far weaker than that of catechin[29]. The parallel kinetics of phenolic preservation and quinone suppression under irradiation confirm that browning control in lotus root hinges on intercepting the initial PPO-catalyzed oxidation step. Unlike some leafy vegetables where quinone recycling occurs (e.g., via catechol-POD cycles), lotus root's gallic acid-dominated system lacks efficient quinone reductases[21]. Thus, accumulated o-quinones directly polymerize into melanin. By simultaneously dampening PPO activity and conserving phenolics, γ-irradiation effectively throttles flux through this terminal browning pathway.
The profound inactivation of PPO can be directly explained by the irradiation-induced structural damage observed at both the secondary and tertiary levels. Our CD spectral analysis revealed a fundamental unfolding of PPO, marked by a drastic loss of α-helix and a concomitant gain of β-sheet structure following 3 kGy treatment. This is critically important because the active site of PPO, where the copper ions are coordinated, is primarily constructed from several α-helices[22]. The disruption of this helical framework, as seen in the 48.6% reduction, likely leads to the disintegration of the catalytic center, directly causing loss of function. This structural perturbation is not unique to irradiation. Similar principles of α-helix destabilization have been observed in other physical fields, such as magnetic fields. Studies have demonstrated that magnetic fields can facilitate the ordering of hydrogen bonds and alter the alignment of peptide dipole moments within α-helices, leading to a measurable decrease in helical content[30].
This unfolding at the secondary level was consistent with the perturbation of the tertiary structure, as evidenced by fluorescence spectroscopy. Intrinsic fluorescence originates predominantly from tryptophan (Trp) and tyrosine (Tyr) residues, which are sensitive reporters of local conformational changes. These fluorophores are usually sequestered within the hydrophobic core in the native state. The fluorescence of these residues is highly sensitive to the local environment and is quenched in polar solvents[31]. The observed reduction in fluorescence intensity (Fig. 5) suggested that irradiation causes the burial or quenching of tryptophan residues, reflecting conformational rearrangements and a possible alteration in the surface hydrophobicity of the enzyme. It has been reported that gamma irradiation, which triggers the radical oxidation of protein, could increase the surface hydrophobicity by exposing sulphydryl groups[32], a process indicative of disulfide bond reduction or cleavage. Critically, disulfide bonds are covalent cross-links that provide substantial conformational stability to enzymes; their disruption significantly compromises structural integrity[33]. The observed spectral changes suggests that gamma irradiation induced a partial unfolding of the protein, causing the buried Trp and Tyr residues to become exposed to the surrounding aqueous solvent[34]. Irradiation induced the disruption of the tertiary structure, and the amino acids with high mobility were more exposed to the polar environment, resulting in a red-shifted emission peak[35]. The pronounced redshift and quenching of fluorescence unequivocally demonstrate that the compact, native structure of PPO was loosened, exposing the hydrophobic core and its constituent Trp and Tyr residues to the aqueous milieu[32]. This conformational rearrangement can be attributed to the impact of irradiation-generated free radicals, which attack protein molecules by disrupting stabilizing non-covalent interactions and disulfide bonds[36]. Such attacks lead to a loss of structural integrity, resulting in protein unfolding and denaturation. This structural disorganization could bring the Trp and Tyr residues into close proximity with other amino acid side chains that act as quenchers, and/or promote non-specific aggregation. Both scenarios would lead to fluorescence quenching, consistent with our spectral observations. The resultant conformational change directly impacts enzymatic function. Critically, the direct correlation between such a fluorescence redshift and the loss of PPO activity has been consistently documented in previous studies[33,37]. Therefore, our fluorescence spectroscopic evidence provides a direct structural explanation for the observed inactivation of PPO in irradiated lotus roots.
In conclusion, 60Co-γ irradiation generates free radicals that cause conformational changes in PPO (secondary structure destruction and tertiary structure unfolding), which is coupled with the transcriptional downregulation of the PPO gene. In addition to fundamentally altering the browning metabolism, γ-irradiation conferred a decisive practical advantage by ensuring microbial safety throughout storage. The treatment at 2, and 3 kGy effectively maintained total viable counts below 5 log10 CFU/g. This represents a significant extension of the microbiological shelf-life compared to the untreated control. This robust microbial control, achieved concurrently with the profound inhibition of enzymatic browning, underscores the dual and synergistic benefit of γ-irradiation. Gamma irradiation presents a sophisticated postharvest strategy that leverages fundamental structural and transcriptional biology to achieve superior quality preservation in fresh-cut lotus root. The treatments showed no significant negative impact on the primary nutritional marker (total phenolic content) assessed in this study. Their influence on other sensory attributes, such as texture, aroma, and specific flavor compounds, remains to be evaluated. Therefore, future research should include a comprehensive dose-response analysis of these crucial consumer acceptance parameters to fully optimize the irradiation protocol for the commercial production of fresh-cut lotus root.
-
This study demonstrates that 60Co-γ irradiation serves as an effective technology for preserving the quality of fresh-cut lotus root by ensuring microbiological safety and profoundly inhibiting enzymatic browning. The preservation effect is primarily achieved through a significant reduction in microbial load and a multi-targeted intervention in the browning pathway. Specifically, irradiation treatment conserves phenolic substrates, thereby limiting the formation of soluble quinones, the direct precursors of brown pigments. The browning process appears to be dominated by PPO activity and substrate availability, rather than being co-limited by POD. The core mechanism lies in the potent and selective suppression of PPO, which is driven by irradiation-induced conformational alterations in the enzyme's tertiary structure and a concurrent down-regulation of PPO gene expression. Notably, the stability of PAL and POD activities underscore that the plant's inherent stress response can compensate for generalized protein damage, further highlighting the specificity of irradiation towards deactivating the pivotal PPO-mediated browning cascade. Thus, 60Co-γ irradiation presents a scientifically grounded strategy for the fresh-cut industry, effectively decoupling the undesirable browning reaction from the plant's beneficial defense mechanisms to prolong shelf life and maintain visual quality.
-
The authors confirm their contributions to the paper as follows: study conception and design: Ma L, Liu D, Lan B, Wen X; data collection: Zhang S; analysis and interpretation of results: Zhang S, Wang F, Liu D; draft manuscript preparation: Liu D; writing − review and editing: Wang F, Xiao G, Ma L, Liu D; project management: Ma L. All authors reviewed the results and approved the final version of the manuscript.
-
All data generated or analyzed during this study are included in this published article and its supplementary information files.
-
The work was financed by Key Research and Development Project of Guangdong Province (2023B0202050001), Guangdong S&T programme (2023B0202090005), the R&D Projects in Key Areas of Guangdong Province (2023B0202080003), Youth S&T Talent Support Programme of Guangdong Provincial Association for Science and Technology (SKXRC202401), the Guangdong Provincial Key Laboratory of Lingnan Specialty Food Science and Technology.
-
The authors declare that they have no conflict of interest.
-
accompanies this paper online at: https://doi.org/10.48130/fia-0026-0008.
- Supplementary Fig. S1 SDS-PAGE pattern of purified PPO protein (~60 kDa) from irradiated (3 kGy) and non-irradiated (0 kGy) samples.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press on behalf of China Agricultural University, Zhejiang University and Shenyang Agricultural University. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Liu D, Zhang S, Wang F, Lan B, Wen X, et al. 2026. Synergistic action of 60Co-γ irradiation on quality preservation of fresh-cut lotus root: microbial control and targeted polyphenol oxidase suppression. Food Innovation and Advances 5(2): 145−152 doi: 10.48130/fia-0026-0008
Synergistic action of 60Co-γ irradiation on quality preservation of fresh-cut lotus root: microbial control and targeted polyphenol oxidase suppression
- Received: 15 December 2025
- Revised: 06 January 2026
- Accepted: 19 January 2026
- Published online: 08 April 2026
Abstract: Fresh-cut lotus root is highly susceptible to quality deterioration, primarily due to enzymatic browning and microbial growth. This study investigated the efficacy and mechanism of 60Co-γ irradiation in preserving the quality of fresh-cut lotus root. The results demonstrated that irradiation effectively controlled total bacterial numbers within 5 log10 CFU/g throughout the storage period. Irradiation significantly inhibited browning, reducing the browning index by 19.6%, 25.3%, and 28.6% at 1, 2, and 3 kGy, respectively, compared to the control. This inhibitory effect was attributed to the delayed degradation of total phenolics and the suppressed accumulation of soluble quinones, the direct browning precursors. Irradiation induced a dose-dependent inhibition of polyphenol oxidase (PPO) activity, with the 3 kGy treatment exerting the strongest suppressive effect; however, the activities of phenylalanine ammonia-lyase (PAL) and peroxidase (POD) remained unaffected. Circular dichroism (CD) and fluorescence spectroscopy revealed three kGy irradiation induced rearrangements in the secondary structure, and disruption of the tertiary structure of PPO. Specifically, CD analysis definitively characterized conformational changes in PPO, showing a dramatic reduction in the α-helix content from 35.4% to 18.4%. Meanwhile, fluorescence spectra of PPO exhibited a 12 nm redshift accompanied by significant fluorescence quenching. This phenomenon provides direct evidence that the enzyme's tertiary structure was disrupted, which in turn lead to the exposure of its hydrophobic core and ultimately resulted in functional inactivation. 60Co-γ irradiation is a highly effective technology for maintaining the quality of fresh-cut lotus root, primarily through direct microbial inactivation and the suppression of the PPO-mediated browning pathway.