-
Fruits are indispensable components of human diets due to their richness in vitamins, minerals, and dietary fibers[1]. The ripening process, which is the final stage of fruit development, is a dynamic process marked by biochemical and physiological transformations in texture, pigmentation, flavor, and aroma[2]. These changes not only determine sensory quality but also directly influence storability, marketability, and ultimately, consumer acceptance. Therefore, elucidating the molecular and hormonal mechanisms underlying fruit ripening is crucial for developing targeted strategies to regulate ripening-related traits. Such knowledge enables the optimization of fruit quality for consumption, extension of shelf life for storage, and reduction of postharvest losses, thereby enhancing economic returns throughout the supply chain.
Plant hormones, as a class of signal molecules produced by plants themselves and exerting effects at low concentrations, play crucial roles in regulating fruit ripening[3]. Among them, ethylene and abscisic acid (ABA) are two well-known plant hormones related to fruit ripening, which can affect the texture, color, and flavor of most fruits[4]. Fleshy fruits are generally divided into climacteric and non-climacteric types according to whether there is a characteristic burst of respiration during ripening[5]. During the ripening of climacteric fruits such as tomato and kiwifruit, a significant surge in ethylene production typically occurs, which is a key factor in the ripening process and underscores the importance of ethylene in fruit maturation. In contrast, non-climacteric fruit (e.g. strawberry and grape) display relatively low ethylene release, with greater emphasis placed on ABA in regulating ripening processes, albeit ethylene still plays a role[2].
Decades of research have elucidated the individual contributions of ethylene and ABA to ripening. However, recent studies highlight their interdependence, challenging the traditional view of hormonal autonomy[6,7]. For instance, ABA accumulation often precedes ethylene production in climacteric fruits, suggesting a priming role for ABA in ethylene biosynthesis. Conversely, ethylene may fine-tune ABA levels through transcriptional regulation. This bidirectional interaction forms a complex regulatory network that coordinates ripening progression. The initiation of numerous ripening-related traits, including fruit softening and peel degreening, is co-regulated by ethylene and ABA in a coordinated manner[8]. While previous reviews have predominantly focused on the individual roles of either ABA or ethylene in fruit ripening, with any discussion of their interplay often remaining cursory, this review specifically delves into the interplay mechanisms between ethylene and ABA during ripening, offering a more in-depth and targeted examination. Here, we consolidate current knowledge on ethylene-ABA crosstalk, addressing questions: How do these hormones reciprocally influence biosynthesis and signaling? Are their interactions conserved across diverse fruit species? These latest findings provide new insights into the regulation mechanisms of plant hormones on fruit ripening and the development of more accurate and efficient fruit quality regulation technologies.
-
Ethylene is recognized as a key phytohormone in the ripening of fruits, with extensive research focusing on its regulatory mechanisms[5,9]. However, the trigger of ethylene biosynthesis at the beginning of fruit ripening remains to be discovered. Ethylene biosynthesis is initiated by the conversion of S-adenosylmethionine (SAM) to 1-aminocyclopropane-1-carboxylic acid (ACC) via ACC synthase (ACS), followed by ACC oxidase (ACO)-mediated ethylene release[10]. Studies have shown that in various fruits, including peach and persimmon, the peak of ABA content occurs before that of ethylene during the ripening process, suggesting that ABA plays a regulatory role by inducing ethylene release[11]. Numerous studies have demonstrated that ABA orchestrates ripening in both climacteric and non-climacteric fruits, and that ABA can regulate ethylene biosynthesis in numerous fruit species[12]. For instance, in non-climacteric fruits such as strawberry and grape, abscisic acid (ABA) and ethylene form a coordinately regulated positive feedback loop. ABA drives ethylene biosynthesis by upregulating the expression of key ethylene biosynthesis genes (e.g., FaACO1 in strawberry and VvACO1 in grapevine). In turn, ethylene enhances the expression of ABA biosynthesis genes (e.g., FaNCEDs in strawberry and VvNCED1 in grapevine), thereby amplifying the ABA signal[13]. The application of ABA to climacteric fruits such as mango, peach, and banana stimulates ethylene biosynthesis, thereby accelerating the fruit ripening process[14]. The suppression of ZDS, a gene pivotal in the biosynthesis of ABA, led to a reduction in ABA levels and an extended period of ethylene production in tomato fruit[15]. In contrast, the elevation of ABA levels due to the suppression of SlUGT75C1, which encodes an enzyme involved in ABA metabolism, also augmented ethylene release[16]. Analogously, over-expression of DkBG1, an enzyme that hydrolyzes ABA-glucose ester to liberate ABA, accelerated the release of ethylene by enhancing ABA levels[4]. Based on the aforementioned evidence, the regulatory role of ABA in ethylene biosynthesis exhibits remarkable context-dependence and bidirectional characteristics. The key focus for future research lies in elucidating the molecular switch that determines its positive or negative regulation, as well as in deciphering the precise spatiotemporal regulatory mechanisms underlying its action during the initiation of fruit ripening.
ABA signaling encompasses four pivotal components: ABA receptors PYR/PYL/PCAR, type 2C protein phosphatases PP2Cs, the subfamily 2 of the SNF1-related kinases SnRK2s, and ABA-responsive element binding proteins AREB/ABFs[17]. These elements directly interface with ethylene pathways (Fig. 1). For example, studies have demonstrated that both over-expression and repression of ABA receptor SlPYL9 modulate the expression of genes involved in the ethylene biosynthesis pathway, including SlACS2/4, SlACO1[18]. Furthermore, SlAREB1 is capable of directly binding to and trans-activating the promoter of NOR, leading to accelerated ethylene release and tomato fruit ripening[19]. Studies have demonstrated that lipocalins (e.g., SlTIL1/2) function as a critical hub integrating ABA and ethylene signaling pathways in tomato. Their expression is directly induced by ABA and further promotes ethylene biosynthesis and signaling via upregulating key ethylene biosynthetic genes (e.g., SlACS2 and SlACO1) and enhancing reactive oxygen species (ROS) scavenging[20]. Studies demonstrate that SlAREB1, a central transcription factor in the ABA signaling pathway, orchestrates ABA-promoted tomato fruit ripening through direct transcriptional activation of ACO5 (a key ethylene biosynthesis gene) and ethylene response factor (AP2/ERFs)[21]. SlABF4 functions as a pivotal mediator of ABA signaling and serves as a central repressor of tomato fruit ripening. It operates through a dual mechanism: directly suppressing the transcription of ethylene biosynthesis genes (SlACS2 and SlACS12), and physically interacting with ripening-related transcription factors—specifically antagonizing the transactivation activity of SlFUL1 while cooperatively enhancing the transcriptional repression exerted by SlMADS1[22]. It has been speculated that MdABF2-like facilitates ethylene release by regulating the expression of MdACS1/3 and MdACO1 in apple[23]. Additionally, protein kinase MdSnRK2‐I can directly phosphorylate MdACS1 to enhance its stability, or phosphorylate and stabilize the transcription factors (TFs) MdHB1/2, thereby augmenting their transcriptional activation of MdACO1. Both mechanisms result in increased ethylene production in apple fruit[24].
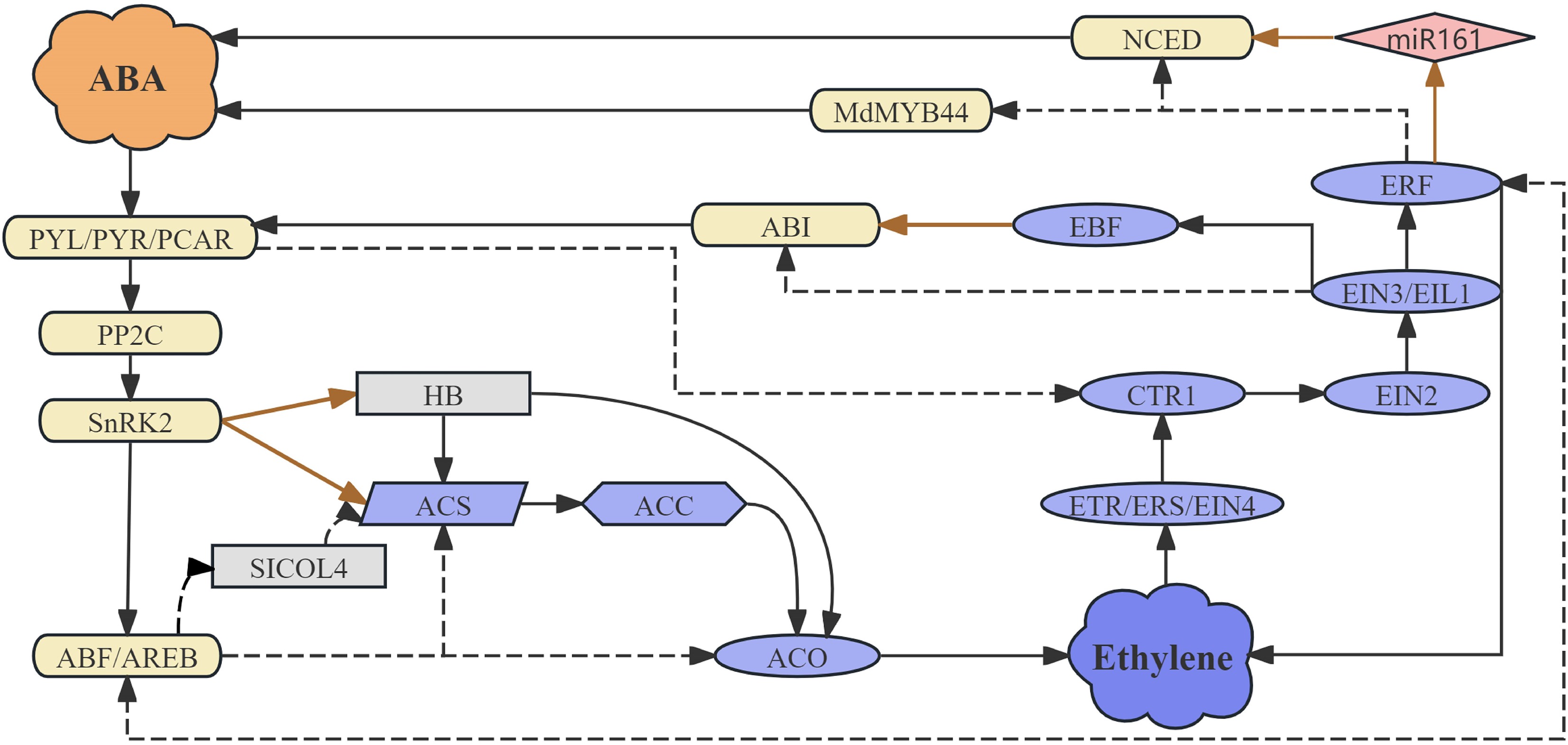
Figure 1.
The interactions between abscisic acid (ABA) and ethylene in fruit ripening. ABI: ABA insensitive; ACO: 1-aminocyclopropyl-carboxylic acid oxidase; ACC: 1-aminocyclopropane-1-carboxylic acid; ACS: 1-aminocyclopropane-1-carboxylic acid synthase; ABF/AREB: ABA responsive element binding factor; CTR1: Constitutive triple response 1; EBF: EIN3-binding F-box protein; EIN2: Ethylene insensitive 2; EIN3: Ethylene insensitive 3; EIL1: Ethylene Insensitive 3-like 1; ERF: Ethylene response factor; Ethylene response (ETR)/Ethylene response sensor (ERS)/EIN4: Ethylene receptors; HB: Homeobox transcription factor; MdMYB44: MYB family transcription factor; miR161: microRNA 161; NCED: 9-cis-cyclocarotenoid dioxygenase; PP2C: Protein phosphatase 2C; PYR/PYL/PCAR: ABA receptors; SlCOL4: ABA signaling-regulated transcriptional repressor; SnRK2: SNF1-related kinases. In the figure, solid lines represent direct regulation, dashed lines represent indirect regulation, and the brown straight lines denote post-transcriptional regulation.
The regulation of ethylene signaling by ABA
-
The ethylene signaling pathway in plants is a well-characterized transduction cascade that regulates diverse developmental and stress responses, including fruit ripening, senescence, and abiotic stress adaptation. The core components of this pathway include: Ethylene receptors (e.g., ETR1, ERS1, EIN4), CTR1 (Constitutive triple response 1), EIN2 (Ethylene insensitive 2), EIN3/EIL1, and ERFs[25,26]. Extensive research has demonstrated that the expression of genes that are closely linked to ethylene signaling, such as ZjERS2, ZjEBF2, and EIL3 in the case of Chinese jujube and CTR1 in strawberry, is positively influenced by the presence of ABA[27,28]. Furthermore, the manipulation of the ABA receptor SlPYL9, through both over-expression and repression, has been observed to modify the expression levels of genes pertinent to ethylene signaling, including SlCTR1, SlETR3, and SlERF2. In tomato, ABA promotes fruit ripening by transcriptionally repressing the negative regulator SlCOL4, thereby relieving its suppression of key ethylene biosynthesis genes (e.g., SlACS2, SlACS4, SlACO1) and signaling components[29]. These empirical findings strongly indicate that ABA has the capacity to influence the signal transduction pathway of ethylene (Fig. 1).
Current evidence indicates that in both climacteric and non-climacteric fruits, ABA coordinates the ripening process by modulating the expression and activity of critical components involved in ethylene biosynthesis or signaling. However, in non-climacteric fruits, the ABA content is generally higher, and its regulatory effect on ethylene is more pronounced. In contrast, research on the influence of ABA on ethylene in climacteric fruits remains relatively limited, and most existing studies have focused on exogenous ABA treatments, while the endogenous ABA content in these fruits is comparatively low.
In addition, a key mechanistic question remains unresolved: whether ABA acts directly on ethylene signaling components or indirectly affects downstream signaling by regulating ethylene biosynthesis. Clarifying this distinction is critical for a comprehensive understanding of the hormonal interaction network governing fruit ripening.
-
There is no doubt that ABA plays a significant role in the regulation of ethylene during the process of fruit ripening. However, the extent to which ethylene controls the biosynthesis of ABA in fruit has been less thoroughly investigated and reported. During the ripening process of hot peppers, a notable peak in ethylene emission was observed to precede a sharp increase in ABA levels, suggesting a potential regulatory role of ethylene on ABA biosynthesis[30]. In peach fruit, studies have demonstrated that treatment with 1-methylcycopentene (1-MCP, an ethylene inhibitor) can lead to a reduction in ABA content, along with changes in the transcription levels of PpNCED2/3, which are pivotal genes responsible for ABA biosynthesis. In blueberry fruits, it was found that ethylene accelerates carotenoid degradation and promotes ABA accumulation by down-regulating carotenoid biosynthetic genes while concurrently up-regulating the expression of key genes in ABA biosynthesis, such as VcNCED1 and VcNCED3[31]. Ethylene downregulates the expression of miR161, thereby relieving its suppression of the key ABA biosynthetic gene FaNCED1, which ultimately promotes ABA accumulation and collectively drives fruit ripening[32]. Furthermore, the TF PpERF3 has been identified to directly activate PpNCED2/3, thereby indicating that ethylene can indeed impact the biosynthesis of ABA within the fruit[33]. Another member of the ERF family protein, PpeERF2, has also been implicated in the accumulation of ABA in fruit, where it functions by suppressing the expression of NCEDs. The over-expression of FveERF (FvH4_5g04470.1) in strawberry has also been shown to affect ABA accumulation, demonstrating that this regulatory effect is not confined to a single type of fruit[34]. Evidence indicates that during the late stages of apple development, the increase in ABA levels occurs concomitantly with the activation of ethylene metabolism, suggesting that ethylene may promote ABA biosynthesis by regulating genes such as NCEDs. Meanwhile, ABA catabolism is also modulated by ethylene, and ethylene-dependent ABA breakdown pathways lead to significant varietal differences in the accumulation of ABA derivatives, such as ABA-GE[35]. Collectively, current evidence confirms that ethylene can regulate ABA biosynthesis in fruits through components such as ERFs. However, whether and how other critical elements within the ethylene signaling pathway—such as receptors and EIN2/EIL—participate in modulating ABA synthesis remains to be systematically elucidated.
The regulation of ABA signaling by ethylene
-
The signaling of ABA is also subject to regulation by ethylene (Fig. 1). When subjected to 1-MCP treatment, the ethylene signaling pathway was found to be inhibited, and this resulted in changes to the expression of PpPYR, PpPP2C, and PpSnRK2, thereby influencing ABA signal transduction in peach fruit[36]. The ethylene F-box protein EBF1 has been shown to physically interact with an ABA-insensitive protein ABI5-like, modulating its transcriptional activity of key genes involved in starch and cell wall degradation[37]. In apple, it was found that MdERF023, acting as a downstream component of the ethylene signaling pathway, mediates the suppression of ABA signaling by inhibiting its key positive regulator, MdMYB44-like[38]. In blueberry, ethylene amplifies ABA signaling by upregulating key ABA biosynthetic genes and the ABA receptor PYR1-like, implicating a synergistic interaction between these hormones during signal transduction[39]. Additionally, EIN3 has been found to inhibit ABI4, influencing its regulation of ABA biosynthesis[40].
Based on the aforementioned literature analysis, ethylene can regulate ABA biosynthesis in both climacteric and non-climacteric fruits by modulating key genes such as NCEDs. However, the regulatory pathways and interaction patterns may exhibit species-specific variations. This indicates that the ethylene-mediated regulation of ABA shares conserved molecular nodes but also involves distinct signaling routes across different fruit types. Moreover, existing evidence suggests that ethylene fine-tunes ABA responses through multi-layered modulation of key components within the ABA signaling pathway. The extensive interactive network between ethylene and ABA signaling further underscores their bidirectional, synergistic, and reciprocal relationship in coordinating fruit ripening.
Understanding these interactions is crucial for developing strategies to manipulate fruit ripening and improve postharvest quality. However, the current regulatory network is likely incomplete. Therefore, future research should focus on identifying additional players in this regulatory network and elucidating the mechanisms underlying their interactions.
-
It is well-established that ethylene and ABA have a mutual regulatory relationship in the context of fruit ripening. However, recent research has unveiled a more complex network of factors that can simultaneously affect the levels of both ethylene and ABA (Fig. 2), thus playing a pivotal role in modulating the ripening process of fruits.
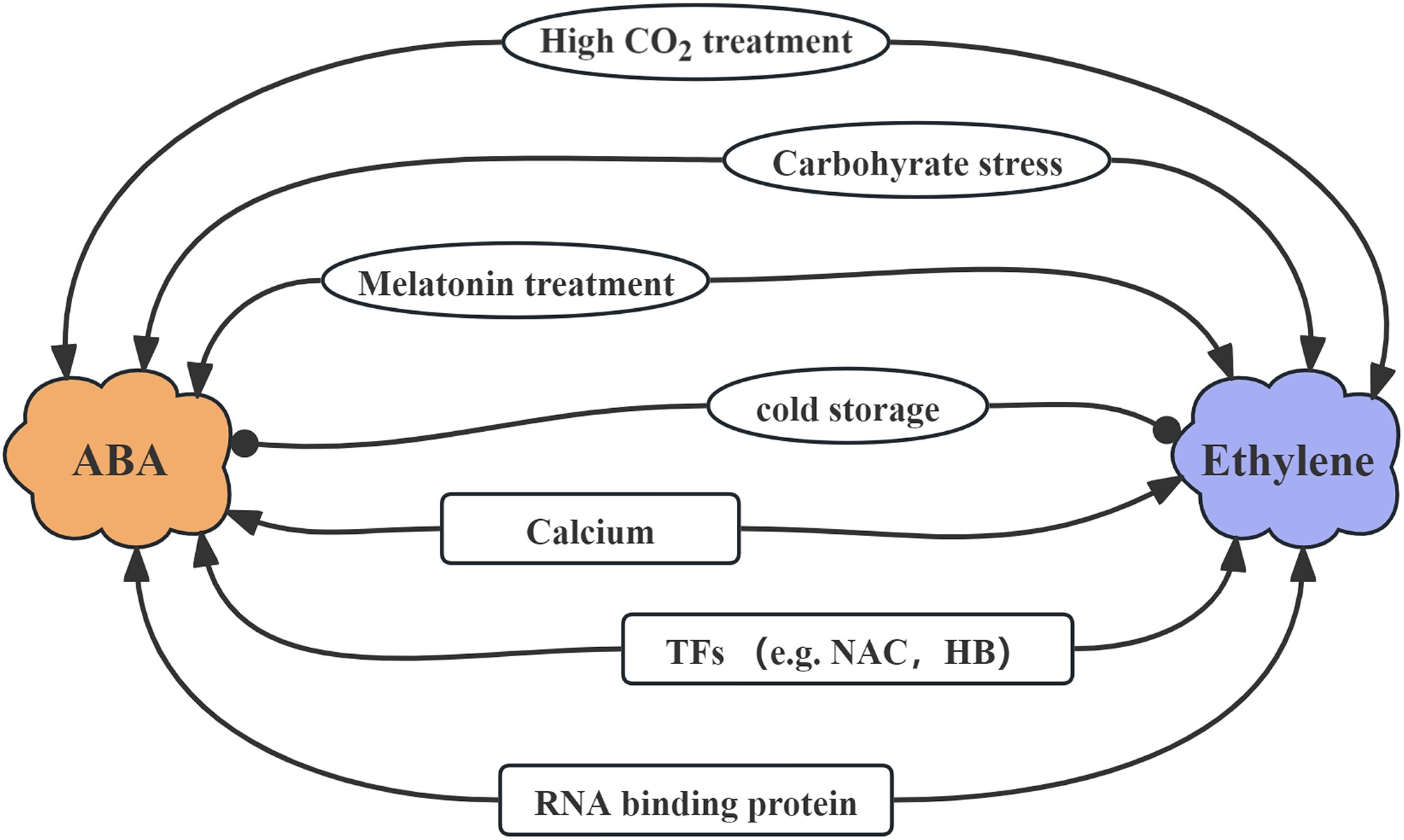
Figure 2.
Some co-regulators of ethylene and abscisic acid (ABA) in fruit. The triangular arrow indicates a promoting effect, while the circular arrow represents an inhibitory effect. TF: transcription factor.
Environmental and chemical regulators
-
In recent research, many environmental and chemical regulators have been recognized as co-regulators of ethylene and ABA in fruit (Fig. 2). For instance, studies have demonstrated that melatonin treatment stimulates the production of both ABA and ethylene in grape berries, which subsequently accelerates the ripening process of fruit[27,28,41]. In strawberry fruit, exogenous melatonin acts as a negative regulator of ripening, primarily through the following mechanisms: by reducing H2O2 accumulation and down-regulating the expression of NCEDs genes to inhibit both ABA biosynthesis and signaling; and by suppressing the expression of key ethylene biosynthesis genes such as ACO and ACS, thereby reducing ethylene production. Additionally, melatonin further modulates the expression of GAMYB and SnRK2.6, which cooperatively suppress ABA and ethylene pathways, ultimately delaying the ripening process in strawberries[42]. In cherry tomatoes, melatonin delays postharvest ripening and senescence by modulating the endogenous hormonal network, which involves suppressing the biosynthesis of ABA and ethylene while promoting the accumulation of Indole-3-Acetic Acid (IAA)[43]. This finding underscores the significance of melatonin as a potential regulator in controlling fruit ripening. When compared to control (CK) and CO2 + 1-MCP treatments, high CO2 treatment in persimmon fruits has been found to elevate the contents of both ethylene and ABA[11]. This phenomenon is closely associated with the fruit's ripening phenotype, further highlighting the role of environmental factors in influencing fruit maturation. Furthermore, the shortage of carbohydrates resulting from a combination of girdling and defoliation treatments triggers the accumulation of ethylene and ABA in litchi, leading to fruit abscission[44]. In macadamia, carbohydrate deprivation triggers the upregulation of ABA and ethylene signaling while suppressing auxin and other pathways, thereby mediating stress responses such as fruit abscission[45]. On the contrary, cold storage of avocado fruits has been proven to inhibit the accumulation of both ethylene and ABA, thereby delaying the ripening process and extending the shelf life of fruit[46]. These findings indicate that numerous environmental and chemical factors can concurrently regulate the accumulation of ethylene and ABA, further underscoring the intimate relationship that exists between them.
Endogenous genes
-
Multiple genes, particularly TFs, have been identified as critical co-regulators of ethylene and ABA in fruit (Fig. 2). The intricate interplay between these genes and hormones plays a pivotal role in regulating various physiological processes, including fruit ripening. For instance, the over-expression of LYCOPENE β‐CYCLASE (LCYb) has been demonstrated to delay fruit softening and prolong shelf life, accompanied by an augmentation in ABA content and a concurrent reduction in ethylene production[47]. In papaya, calcium signaling, through the activation of its sensor CpCML15, inhibits the negative regulator CpPP2C46/65 in the ABA signaling pathway. This relieves the suppression on downstream transcription factors such as CpABI5 and CpERF003-like, thereby cooperatively promoting fruit ripening[48]. A diverse array of TFs, encompassing MADS-box, MYB, NAC, AP2/ERF, bHLH, and ZIP families, have been reported to play significant roles in the regulation of ABA- and ethylene-related genes during the ripening process of grape berries[49]. These TFs act as molecular switches, orchestrating the expression of target genes. Specifically, in NAC TF RIF-RNAi receptacles, an examination revealed that genes associated with the ethylene signaling pathway were up-regulated, whereas FaNCED3 and a selection of genes implicated in ABA signaling exhibited down-regulation[50]. This finding underscores the notion that the regulation of ethylene and ABA does not necessarily occur in tandem, with one hormone potentially increasing in content while the other decreases, rather than both being concurrently up- or down-regulated.
Notably, both LcHB2 and LcHB3 exhibit the capacity to directly bind to the promoter regions of LcACO2/3, LcACS1/4/7, and LcNCED3 genes, thereby activating their expression to regulate ethylene and ABA accumulation[44]. This direct interaction furnishes unequivocal evidence supporting their co-regulation functions. Research demonstrates that the RNA-binding protein DkRBM24-1 binds specifically to the 3′ UTRs of DkACS1 and DkNCED2, directly enhancing the stability or translation efficiency of their mRNAs and thereby up-regulating these core genes for ethylene and ABA biosynthesis. This regulatory action is strongly induced by high CO2 treatment but suppressed by 1-MCP, indicating its dependence on a functional ethylene signaling pathway[11]. This finding expands our understanding of the regulatory mechanisms governing fruit ripening and hormone biosynthesis.
Overall, in both climacteric and non-climacteric fruits, the interaction network between ethylene and ABA, despite its divergence in different fruit types, is coordinately modulated by common upstream factors, including environmental signals, chemical regulators, and key transcription factors, thus forming a highly integrated co-regulatory module. Based on the mechanisms of these endogenous genes and their crosstalk with hormonal networks, future targeted regulatory strategies can be designed to manipulate the fruit ripening process, opening new avenues for enhancing fruit quality and prolonging postharvest shelf life. This also highlights promising directions for further research in plant biology and horticultural science.
-
Ethylene and ABA operate not as isolated regulators but as interdependent modulators within the intricate processes of fruit ripening. Their interplay encompasses intricate feedback mechanisms, the sharing of endogenous genes, and responsiveness to a variety of environmental cues, collectively constructing a resilient yet highly adaptable regulatory network. Specifically, in climacteric and non-climacteric fruits, this interaction network exhibits distinct characteristics: In climacteric fruits, ABA generally serves as an upstream trigger for ethylene biosynthesis, while ethylene often synchronously regulates ABA synthesis through conserved transcription factors. In contrast, non-climacteric fruits predominantly rely on a positive feedback regulatory loop between the two hormones. Despite these differences, the interplay between ethylene and ABA in both fruit types is coordinately regulated by common upstream factors, including environmental signals, chemical modulators, and key transcription factors, thereby forming a highly integrated co-regulatory module that precisely orchestrates the fruit ripening process.
To move beyond the general understanding of this interaction, future research should focus on achieving breakthroughs at three levels: mechanistic depth, methodological innovation, and practical application. Firstly, systematic comparative biological studies are needed to elucidate the fundamental reasons for the network's specificity across different fruit types (e.g., climacteric vs non-climacteric) and to identify the upstream 'molecular switches' that determine signaling dominance. Secondly, the integration of multi-omics and advanced imaging technologies should be employed to dynamically map how the network responds to and is reconfigured under environmental stresses such as high temperature and drought. Finally, strategies based on key hub genes for precision breeding or the use of temporally controlled delivery technologies to modulate hormone signals can be explored. This will enable the design of green solutions that simultaneously optimize fruit quality and postharvest storage performance, thereby providing a new paradigm for sustainable agricultural development.
-
The authors confirm contribution to the paper as follows: study conception and design: Wu W, Yang ZF; data collection: Cai YJ, Dai H, Zhang YH, Wei ZY, Ni H; analysis and interpretation of results: Wu W, Cai YJ; figure preparation: Cai YJ, Dai H, Zhang YH; draft manuscript preparation: Wu W, Cai YJ; manuscript revision: Shi LY, Yang ZF, Ding HM, Chen W. All authors discussed the content, reviewed the results and approved the final version of the manuscript.
-
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
-
This work was supported by the Zhejiang Provincial Top Discipline of Biological Engineering (Grant No. ZS2025012) and the National Natural Science Foundation of China (Grant No. 32302620).
-
The authors declare that they have no conflict of interest.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press on behalf of Chongqing University. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Cai YJ, Dai H, Zhang YH, Wei ZY, Ni H, et al. 2026. The interactions between ethylene and abscisic acid during fruit ripening. Plant Hormones 2: e008 doi: 10.48130/ph-0026-0006
The interactions between ethylene and abscisic acid during fruit ripening
- Received: 26 December 2025
- Revised: 11 February 2026
- Accepted: 11 March 2026
- Published online: 15 April 2026
Abstract: Fruits serve as vital components of human nutrition, providing essential micronutrients, including vitamins, minerals, and dietary fibers. The terminal phase of fruit development, termed ripening, profoundly impacts quality traits and postharvest economic value. Ethylene and abscisic acid (ABA), two pivotal phytohormones, orchestrate fruit ripening through intricate regulatory networks. While early studies predominantly focused on their independent roles, emerging evidence highlights the dynamic crosstalk between ethylene and ABA as a critical modulator of ripening. This review synthesizes recent advancements in understanding their mutual regulatory mechanisms during ripening, emphasizing ABA's role in modulating ethylene biosynthesis and signaling, and vice versa. Additionally, we discuss external factors and transcription regulators that co-regulate both hormones. These findings not only refine our understanding of hormonal interplay in plant physiology but also present opportunities for targeted manipulation of ripening processes to enhance fruit quality and shelf life.
-
Key words:
- Fruit ripening /
- Hormone crosstalk /
- Ethylene /
- Abscisic acid /
- Postharvest regulation












