-
Among all the novel tobacco products, heated tobacco products have drawn particular attention. Unlike the high-temperature combustion process (including chemical reactions such as distillation, cracking, and combustion) of traditional tobacco products, aromatic tobacco substances are generated by distillation and low-temperature pyrolysis of reconstituted tobacco sheets in a heating device for heated tobacco products[1]. In addition, the smoke release process is quite simple without any requirement for burning[2,3]. Moreover, the heating temperature can be tuned to as low as 300°C, which significantly reduces the harmful ingredients released during combustion that pose a threat to human health[4]. However, it is difficult for reconstituted tobacco sheets to produce enough aromatic substances through low-temperature pyrolysis. The concentration of pyrolysis gas, the aroma's intensity, and the sensory quality of heated tobacco products are all far inferior to traditional tobacco products[5−7]. To provide an effective solution to this issue, it would be rational to develop low-temperature pyrolysis technology for smoking materials, such as reducing the pyrolysis temperature of the active ingredients in reconstituted tobacco sheets and ensure the efficient release of aromatic substances at low temperatures. A catalyst is defined as "a substance that increases the rate of a reaction without modifying the overall standard Gibbs energy change in the reaction", according to the International Union of Pure and Applied Chemistry (IUPAC)[8]. As such, the addition of appropriate catalysts not only dramatically reduces the energy barrier for the pyrolysis reaction but also ensures the efficiency of the reaction under relatively mild conditions[9]. Since the reconstituted tobacco sheets used for heated tobacco product are a kind of porous carbohydrate-based biomass material, with the end use in mind, various catalysts for catalytic conversion of biomass could show considerable activity in the pyrolysis process at low temperatures, such as molecular sieves[10−13], mesoporous materials[14−18], metallic oxides[19−22], alkali metal/alkaline earth metal salts[23−27], and so on. Among these, alkali metal salts exert their catalytic effects through coordination with the hydroxyl groups adjacent to the glucose ring, thereby effectively reducing the cellulose's crystallinity and destabilizing the ring structure, ultimately promoting the formation of low-molecular-weight oxygenates and enhancing the conversion of anhydro sugars to ketones, which is important for the regulating the formation of aromatic compounds from tobacco. In addition, trial-and-error experiments based on our previous work indicated that alkali metal salts, particularly acetate, lower the pyrolysis temperature of reconstituted tobacco sheets and increase the total weight loss at 100–300 °C, accompanied by higher release of flavor substances[28,29]. Therefore, they have the potential to be used to assist the low-temperature pyrolysis of reconstituted tobacco sheets. Moreover, alkali metal salts are intrinsic components of tobacco leaves, which means they do not introduce completely foreign elements into the tobacco products. Because of the relative thermal stability of alkali metal salts up to 300 °C, they are unlikely to generate new harmful substances in the smoke of heated tobacco products, thereby exhibiting low toxicity and sufficient safety[29−31]. In addition, these salts are generally stable under ambient conditions when stored in sealed packages. In these cases, clearly, to meet the ever-increasing demands for safety and sensory quality in heated tobacco products, alkali metal salts are highly desirable to assist with this but remain a formidable challenge in practical application.
In our previous study, building on the experience of reducing the pyrolysis temperature of reconstituted tobacco sheets by adding alkali metal/alkaline earth metal salts, e.g., acetates of sodium (Na), potassium (K), magnesium (Mg) and calcium (Ca), thermogravimetry–Fourier transform infrared (TG-FTIR) spectra and pyrolysis–gas chromatography–mass spectrometry (Py-GC-MS) were used to investigate the thermogravimetric properties and the distribution of pyrolysis gas at low temperatures[29]. In that work, thanks to the design, after the addition of Na or K salt, the pyrolysis temperature of reconstituted tobacco sheets was decreased; in addition, the total weight loss and the concentration of pyrolysis gas were significantly increased at 100–300 °C. Notably, the concentration of carbonyl compounds and compounds containing C-O bonds was also increased. These findings indicate that Na or K salts play an important role in promoting low-temperature catalytic pyrolysis and in enhancing the flavor of heated tobacco products. Nevertheless, despite the extensive exploration of low-temperature pyrolysis promoted by alkali metal salts, their practical application still faces challenges because of the lack of tools to impart in situ catalytic ability to reconstituted tobacco sheets, such as the lack of systematic guidance on the control of the dosage and the absence of the kinetic parameters required for designing suitable heating profiles. To address these issues, it is essential to develop a quantitative framework that links the salt dosage with pyrolysis gas and kinetics, while adopting impregnation as the salt addition method to ensure homogeneous distribution within the reconstituted tobacco sheets and to mitigate potential problems of uneven catalysis in practical applications.
This study establishes a clear quantitative relationship between the addition of salt and the pyrolysis behavior of reconstituted tobacco sheets, providing a responsible approach for modulating thermal reactions through the integrated analyses using TG-DTG and Py-GC-MS. The results demonstrate that the pyrolysis pathways, apparent activation energy, and the release of aromatic substances were highly dependent on the addition of alkali metal salts, thus providing opportunities for controlled pyrolysis at relatively low temperatures and for refining the heating profiles. Furthermore, the quantitative changes in the kinetic parameters induced by alkali metal salts were also clarified, providing mechanistic insights into their catalytic roles. The reliability of these findings was further reinforced by cross-validation of the kinetic parameters using the Flynn–Wall–Ozawa (FWO) and Kissinger methods. Ultimately, these results revealed not only the catalytic efficiency of alkali metal salts in promoting low-temperature pyrolysis but also their ability to enhance the generation of aromatic substances, thereby improving the overall smoking quality of heated tobacco products. This study is expected to pave a new way toward modulating the pyrolysis behavior of reconstituted tobacco sheets, while providing valuable insights into a mechanistic basis of alkali metal salts' catalysis.
-
Information on the materials and instruments used is provided in the Supplementary File 1.
Preparation of reconstituted tobacco sheets with different alkali metal salt additives
-
Reconstituted tobacco sheets were first ground into powder with an appropriate particle size, followed by complete impregnation with an aqueous solution of the alkali metal salts. Optimized impregnation conditions were achieved through tests based on our previous work[28,29]. For preparing reconstituted tobacco sheets with the addition of 5 wt.% K element (where addition refers to the mass percentage of the metal element [K] and the quality of the K element was converted from the added amount of K salt) 0.377 g C2H3KO2 was dissolved in 3.0 g of deionized water, then transferred into a glass beaker containing 3.0 g of powder obtained from reconstituted tobacco sheets and stirred well for 3 h, then heated to 100 °C to remove water. Next, the sample was sealed at 25°C and left to stand for 12 h to produce the final 5 wt.% K addition sample, named K5. Reconstituted tobacco sheets with added K or Na (i.e., Kn or Nn) were achieved after complete impregnation with aqueous solutions of C2H3KO2 or C2H3NaO2·3H2O with the designed concentration, where, n signifies the amount of the metal element (Na or K) added in the experiment. Nevertheless, K or Na is an essential macronutrient involved in tobacco plants, so the actual contents of alkali metal ions in the reconstituted tobacco sheets were not equivalent to the amount added. As a blank control, a sample without additional alkali metal salt, named KB, was prepared by the same procedure, in which the aqueous solution of alkali metal salts was replaced by deionized water.
Characterizations
Calculation of actual alkali metal salts
-
To quantitatively characterize the amount of the metal elements in reconstituted tobacco sheets, inductively coupled plasma–optical emission spectrometric analysis (ICP‒OES) was conducted. First, 0.10 g of the Kn or Nn sample was dissolved in an aqueous solution of hydrochloric acid (0.05 M, 100.00 mL), shaken for 30 s, then allowed to stand for stratification. Subsequently, 10 mL of the supernatant was diluted in 100 mL ultra-pure water. After being filtered through an aqueous phase filtration membrane, the samples were tested on an ICP‒OES analyzer. According to the standard curve of different elements, the ion concentration in the solution was determined, and then the actual alkali metal salt content in the reconstituted tobacco sheets was determined.
Thermogravimetric properties
-
For this analysis, 3.0 mg of reconstituted tobacco sheets containing alkali metal salt was evaluated on a thermogravimetric analyzer at a ramp rate of 10 °C/min from 30 to 500 °C, under a nitrogen atmosphere with a flow rate of 100 mL/min. Each TGA experiment was performed in triplicate, and the results showed good reproducibility and stability.
Identification of pyrolysis gas components
-
The gas compounds generated during low-temperature pyrolysis of the reconstituted tobacco sheets (about 5.0 mg) were introduced into the GC-MS equipment. The reconstituted tobacco sheets were firstly placed into a preheated pyrolysis tube and subsquently dried at 100 °C for 60 s, then heated from 100 to 300 °C at a ramp rate of 20 °C/s and held for 20 s at 300 °C, under an inert atmosphere (helium, 99.99%) after sample injection. The pyrolysis gas was split in the GC inlet (200 °C) at a split ratio of 10:1, with a helium flow rate of 1 mL/min. Then the pyrolysis products were separated using the GC oven program as follows: (1) 3 minutes at 40 °C, (2) heating at a rate of 5 °C/min to 240 °C, (3) holding for 3 minutes at 240 °C, (4) heating at a rate of 20 °C/min to 280 °C and (5 holding for 32 min at 280 °C. The DB-5MS separation column was used for GC (30 m × 250 µm × 0.25 µm film thickness, Agilent Technologies). The temperature of the GC-MS transfer line was 280 °C and the MS source temperature was 230 °C. The mass spectrometer was operated in electron ionization mode at 70 eV at a scanning range of 30–350 m/z. Specifically, the solvent delay time was set to 2.5 min. The separate compounds were identified by comparing their mass spectra to those in the NIST MS library (2014), within a m/z range of 35–300 Da.
Analysis of reaction kinetics
-
According to the thermodynamics of irreversible processes, the pyrolysis of reconstituted tobacco sheets of heated tobacco products was always classified as a solid-state pyrolysis reaction, given that the FWO method can be widely used in kinetic analyses of tobacco pyrolysis[32,33]. Considering that the apparent activation energy (Eα) values of the pyrolysis reaction will be affected by the heating rate, the thermodynamics of the samples with or without Na/K salt at four heating rates were analyzed (i.e., heating rates of 5, 10, 15, and 20 °C/min). Finally, the Eα values were calculated by FWO method.
-
Considering that the addition of a catalyst always has a significant influence on the catalytic effect, reconstituted tobacco sheets with K or Na doped at a range of concentrations were prepared. As shown in Fig. 1 and Supplementary Table S1, the real content of Na increased from 0.05 wt.% to 4.27 wt.% along with the increase in the impregnation concentration, reaching up to 85 times that of the KB sample. By contrast, the actual content of K changed from 1.59 wt.% to 6.56 wt.%, increasing fourfold. This difference was mainly caused by the difference between the Na and K content in the KB samples, which is caused by physiological changes and the composition of fertilizer applied to tobacco plants during growth[34].
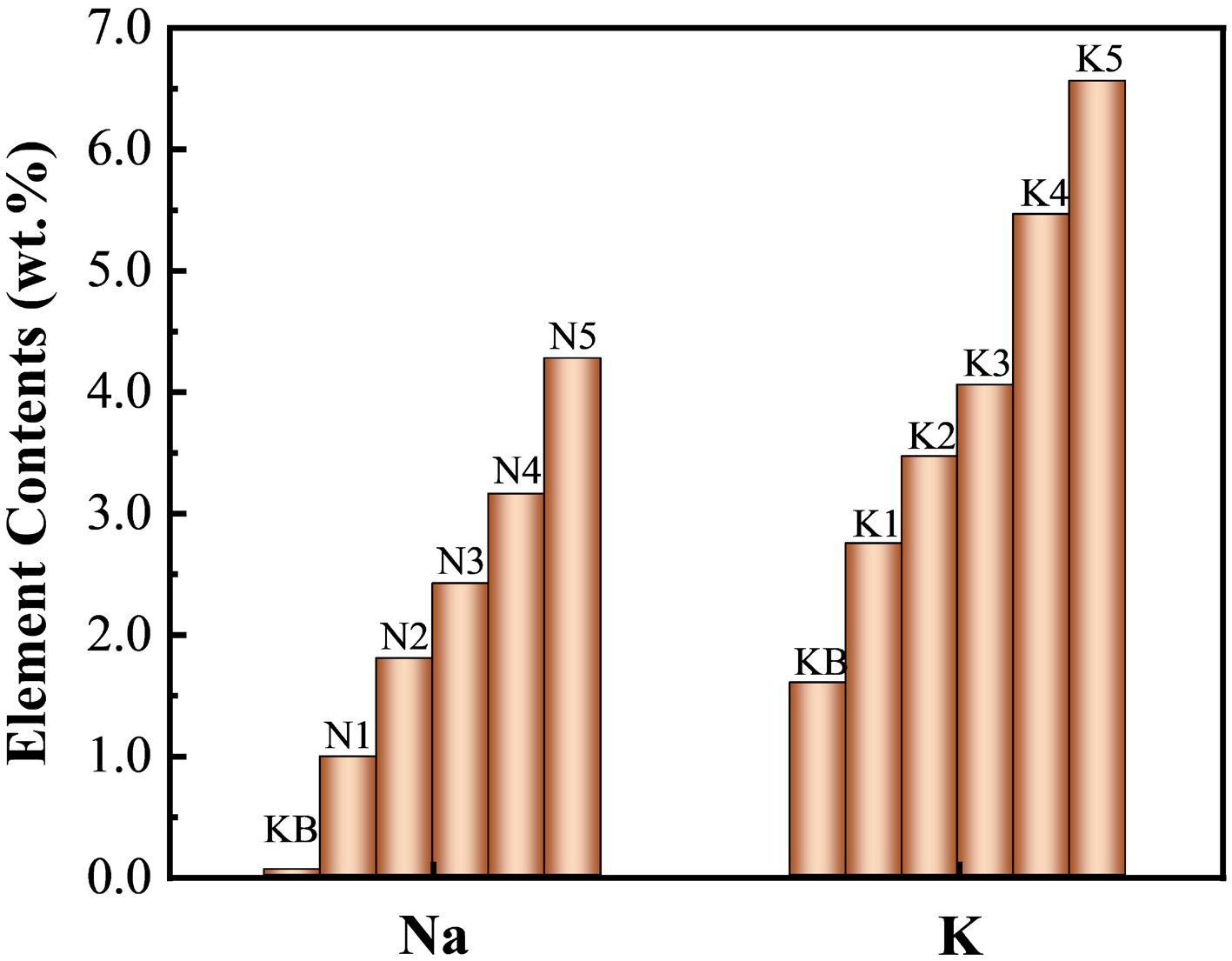
Figure 1.
Contents of Na or K elements in reconstituted tobacco sheets after adding alkali metal salts (measured by ICP‒OES).
Once the target sample was ready, the thermogravimetric properties were evaluated by TG-DTG to lay the foundation for studying its pyrolysis performance. The TG-DTG results show that whether alkali metal salt was added or not, the whole pyrolysis process of reconstituted tobacco sheets from 35 to 500 °C can be divided into three stages. Notably, sodium acetate trihydrate dehydrates below 150 °C and remains stable up to 450 °C, whereas potassium acetate shows no significant decomposition below 300 °C, indicating that both salts maintain sufficient thermal stability and exert a negligible influence on the heating processes[28]. As can be observed in Fig. 2, the water molecules adsorbed in the reconstituted tobacco sheets mostly evaporated below 105 °C, which was defined as the first weight loss stage. In this stage, the weight loss was about 1.44–5.46 wt.% (Supplementary Table S2). The second weight loss stage occurred at 105–235 °C, and was mainly attributed to the volatilization of functional components (e.g., glycerin, propylene glycol, and nicotine)[28,29]. This was closely followed by the third weight loss stage, which started at 235 °C and ended at 375 °C. The weight loss in this stage was caused by the volatilization of nicotine and aromatic substances, as well as the decomposition of cellulose, etc.[28,29].
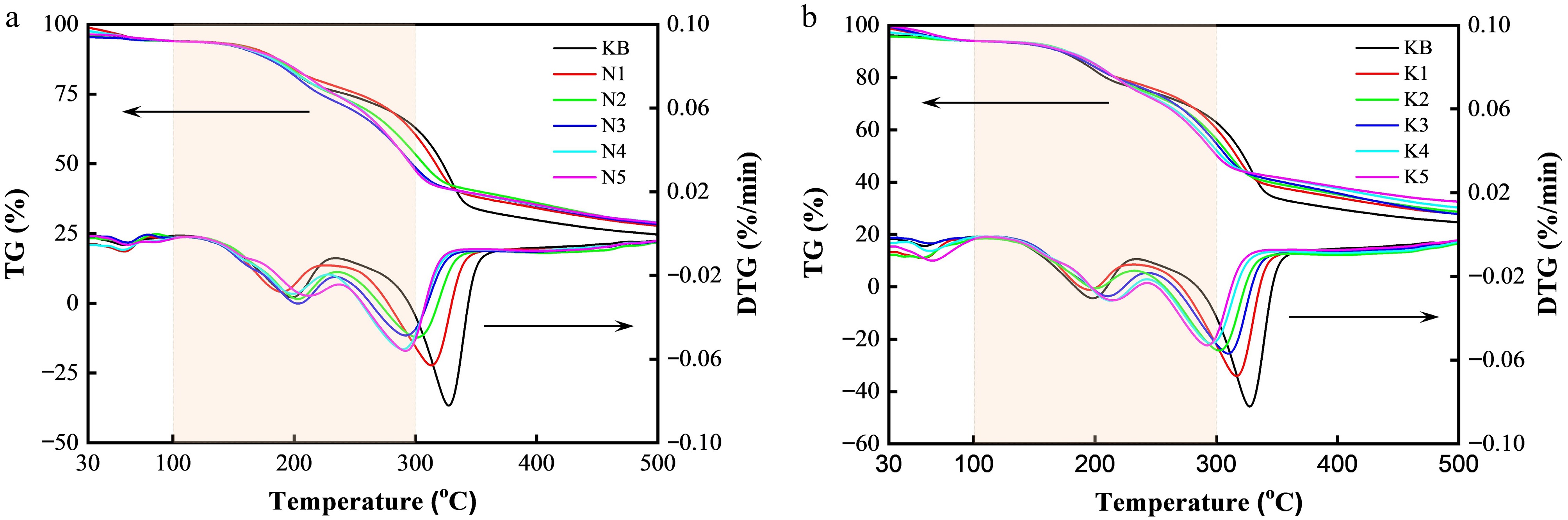
Figure 2.
TG and DTG curves of reconstituted tobacco sheets with varying additions of (a) Na salt and (b) K salt.
Compared with the KB sample, the temperature corresponding to the highest thermal decomposition rate and the total weight loss in the second weight loss stage showed irregular changes with the increase in Na content, indicating that the external Na salt addition had no obvious effect on this stage (Fig. 3a, b). However, as shown in Fig. 3c and Supplementary Table S2, as the Na contents increased from 0.05 wt.% to 3.15 wt.%, the temperature corresponding to the highest thermal decomposition rate decreased from 328 to 292 °C in the third weight loss stage, but only marginal change was seen with further increases in the Na content.
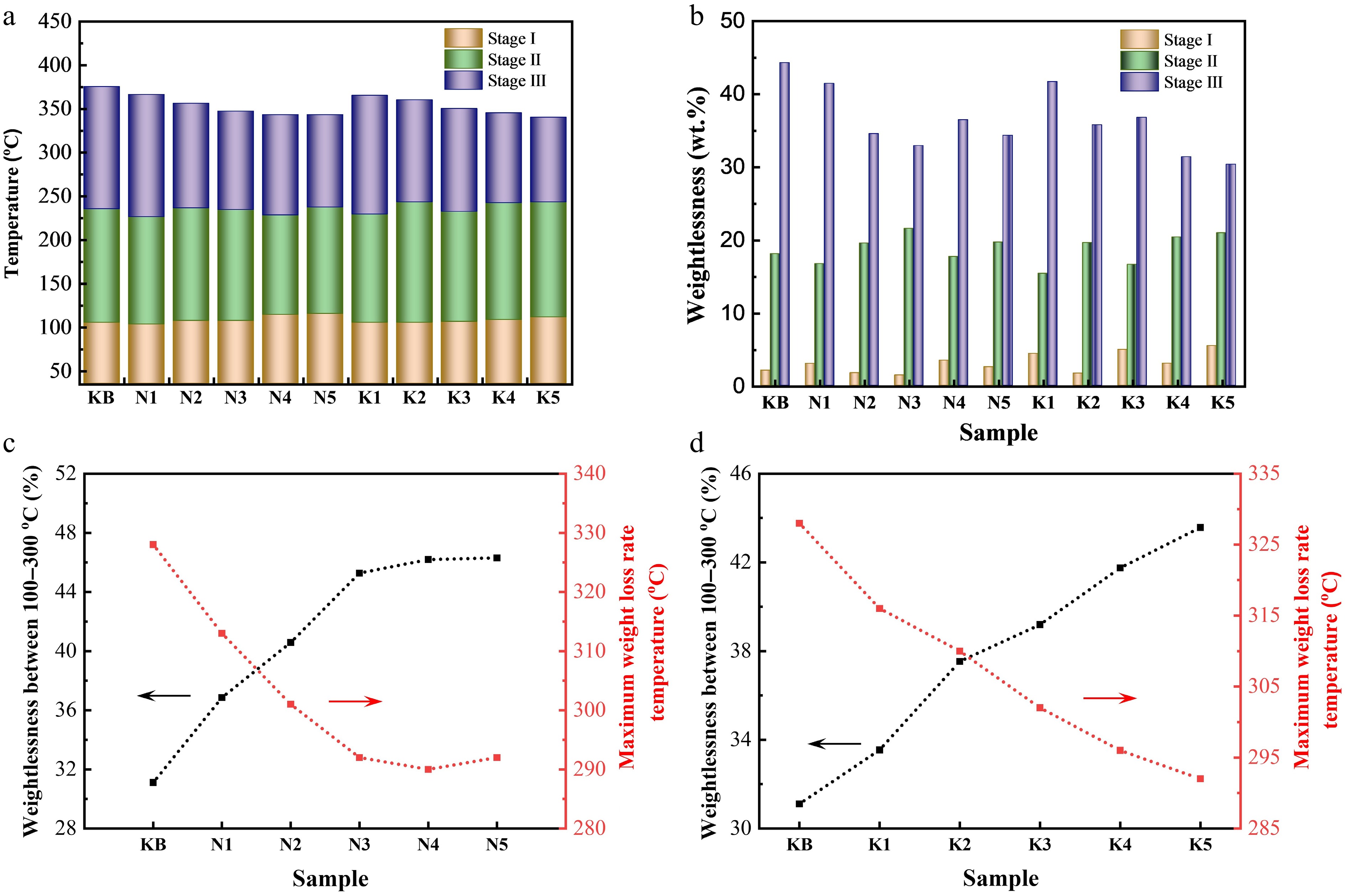
Figure 3.
(a) Weight loss transition temperatures of reconstituted tobacco sheets during pyrolysis at 35–500 °C. (b) Weight loss of reconstituted tobacco sheets in weight loss Stages I–III. Weight loss (left) at 100–300 °C and maximum weight loss rate at temperatures below 500 °C (right) of reconstituted tobacco sheets with added (c) Na salt or (d) K salt.
It is worth noting that the standard operative temperature of heated tobacco products is generally about 300 °C; therefore, the thermal weight loss between 100 and 300°C was our main focus, as this indirectly represents the release efficiency of key substances. As shown in Fig. 3c and Supplementary Table S2, the total weight loss between 100 and 300 °C increased to 45.27 wt.% for the N3 sample, which was 14.16 wt.% more than that of the KB sample. In addition, the total weight loss from 100 to 300 °C was proportional to the amount of Na added when less than 2.41 wt.% was added. However, along with a further increase in added Na, the total weight loss at 100–300 °C did not change significantly. In conclusion, when the actual content of Na in reconstituted tobacco sheets is 2.41 wt.%, an optimal catalytic effect can be achieved. Taking the catalyst's cost into consideration, the optimal addition of Na elements was about 3 wt.%.
The thermogravimetric curves and characteristic temperatures of reconstituted tobacco sheets with added K are shown in Figs. 2b and 3d and Supplementary Table S2. The introduction of K had a similar level of catalytic activity to Na, resulting in the maximum weight loss rate appearing at lower temperatures as the added amount increased. This characteristic temperature gradually decreased from 328 °C for the KB sample to 292 °C for the K5 sample. At the same time, with an increase in the addition of K salt, the total weight loss at 100–300 °C increased from 31.11% to 43.57%. Therefore, the optimal catalytic effect can be achieved when the amount of K added is 5 wt.%.
The results indicated that, considering both the reduced energy consumption of the smoking device and the increased weight loss in the range of 100–300 °C, the best catalytic effect can be achieved when 3 wt.% Na or 5 wt.% K were added. The weight loss of the Na- or K-doped reconstituted tobacco sheets between 100 and 300 °C increased by 45.5% and 40.1% compared with the blank sample. In comparison, the catalytic effect of Na salt was slightly better than that of K salt.
Pyrolysis gas components affected by the amount of alkali metal salt in reconstituted tobacco sheets
-
Representative pyrograms of the gas generated from reconstituted tobacco sheets at 300 °C are presented in Figs. 4 and 5. It is apparent that the yields of detectable pyrolysis products were significantly impacted by the pyrolysis catalysts, notably by changing the intensity of detectable compound peaks. The main compounds detected by Py-GC–MS analysis are summarized in Supplementary Tables S3 and S4. These substances corresponding to the peaks in the pyrograms were qualitatively indicated by searching against the mass spectral database with matching degrees of more than 80, and a semi-quantitative analysis was also carried out according to the peak area at the same time. It was also clear that the contents of acetic acid and glycerol acetate in pyrolysis gas increased gradually with the increase in added Na or K salts, and the contents of glycerol and nicotine decreased under the same conditions. This trend can be mechanistically explained by the decomposition of the added acetate in the heating process, accompanied by the formation of acetic acid combined with glycerol through the esterification reaction to form (di-, tri-) acetin. The generated acetic acid also neutralized nicotine in the pyrolysis gas, promoting the transfer of nicotine from the dissociative state in gas to total particle matter (TPM), thereby leading to a gradual decrease in the nicotine content of the pyrolysis gas. In addition, when the addition of Na increased to 1 wt.%, the contents of 1-hydroxy-2-propanone and 2-furan methanol in the pyrolysis gas showed a tendency to increase, indicating the depolymerization of cellulose, since these compounds are known intermediates derived from sugars such as cellulose during pyrolysis. But when the addition of Na increased further to 2–5 wt.%, the contents of 1-hydroxy-2-propanone and 2-furan methanol in the pyrolysis gas remained constant, suggesting further decomposition of cellulose via secondary reactions. In the case of lignin-derived products, the concentration of phenol in the pyrolysis gas rose proportionally with the amount of salt, consistent with the facilitated cleavage of ether bonds. Moreover, as a key component of tobacco aroma, the content of megastigmatrienone in the pyrolysis gas gradually increased with the increase in added alkali metal salts. This may be caused by the intensified pyrolysis reaction, which makes the skeleton more easily damaged. Overall, both Na salt and K salt exhibited similar catalytic effects, mainly by lowering the activation barrier of primary pyrolysis reactions and promoting secondary transformations. Therefore, the optimization of alkali metal salts could enrich the pyrolysis gas components and enhance the aromas' concentration and intensity at relatively low temperatures.
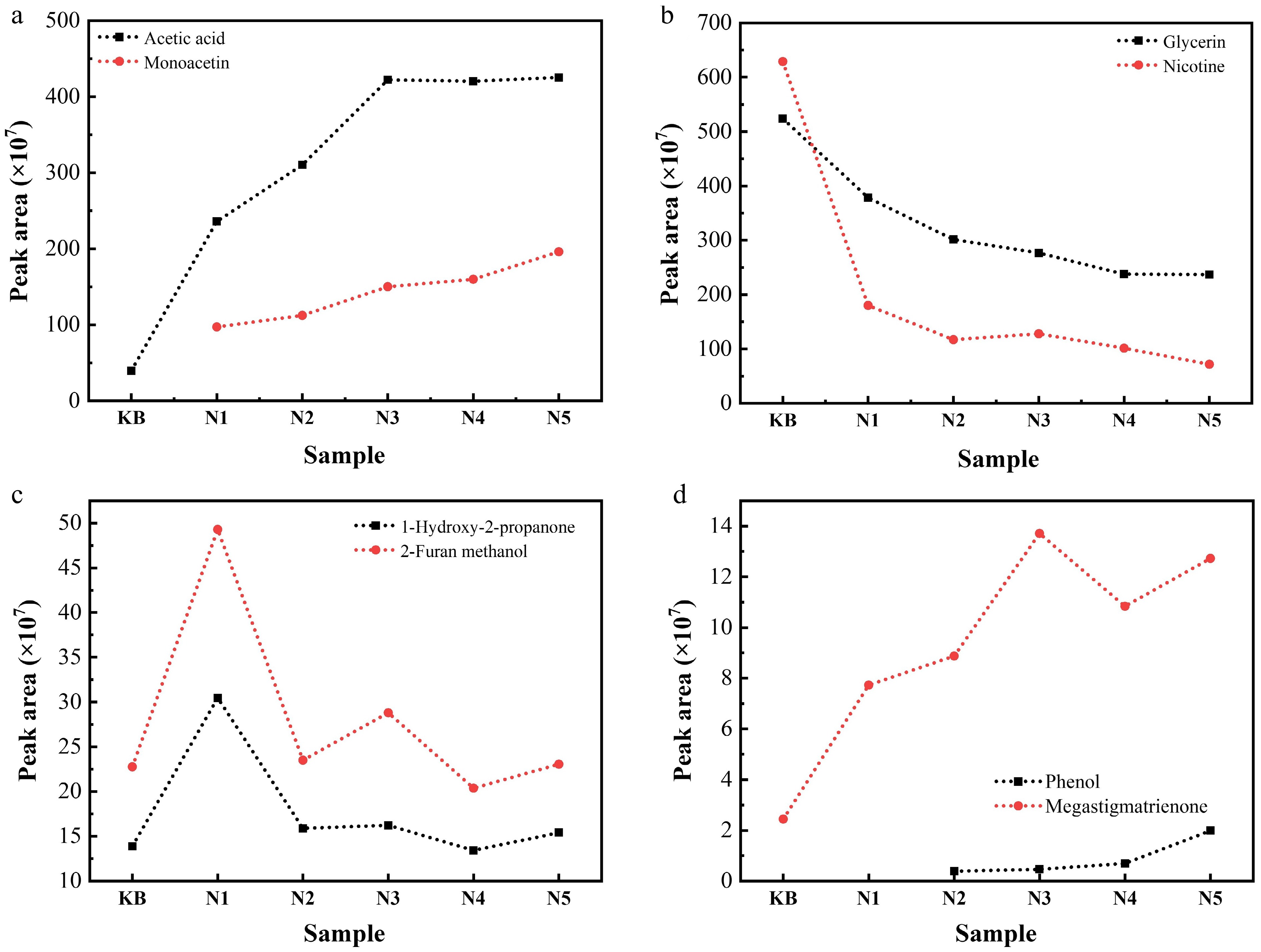
Figure 4.
Contents of (a) acetic acid and monoacetin, (b) glycerin and nicotine, (c) 1-hydroxy-2-propanone and 2-furan methanol, and (d) phenol and megastigmatrienone in the pyrolysis gas of reconstituted tobacco sheets with different amounts of added Na salt at 300 °C (semiquantitative results represented by the peak area measured via Py-GC/MS).
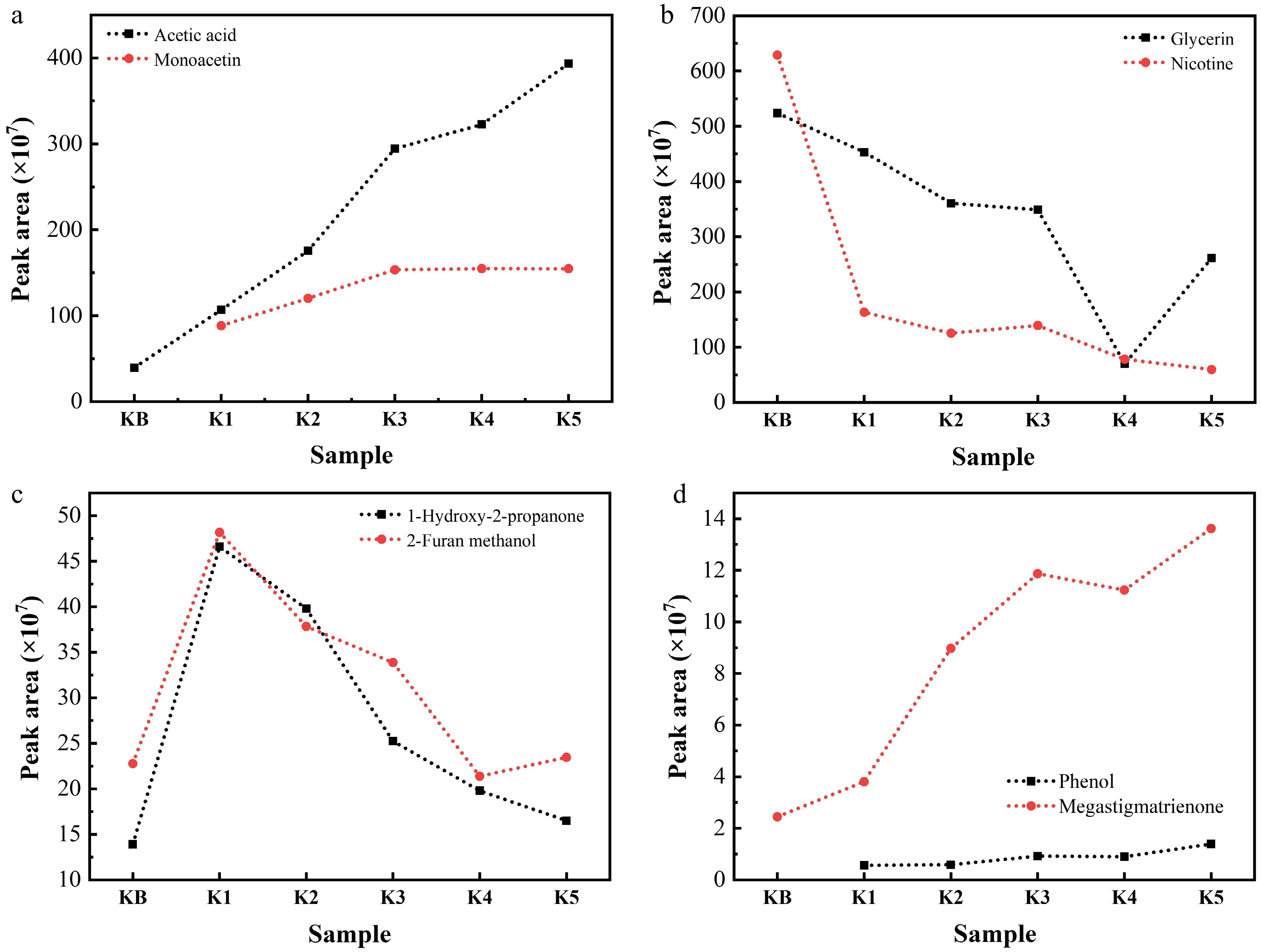
Figure 5.
Contents of (a) acetic acid and monoacetin, (b) glycerin and nicotine, (c) 1-hydroxy-2-propanone and 2-furan methanol, and (d) phenol and megastigmatrienone in the pyrolysis gas of reconstituted tobacco sheets with different amounts of added K salt at 300 °C (semiquantitative restults represented by the peak area measured via Py-GC/MS).
Reaction kinetics of alkali metal salts catalyze the pyrolysis of reconstituted tobacco sheets at low temperatures
-
The reaction kinetics of reconstituted tobacco sheets doped with alkali metal salt were comprehensively examined via the thermogravimetric behavior at different heating rates. As displayed in Fig. 6, the temperature ranges, including the maximum weight loss temperature corresponding to each weight loss stage in the TG-DTG curves, shifted toward higher values at elevated heating rates, such as 15 and 20 °C/min, which can be assigned to the thermal hysteresis phenomenon caused by a larger temperature difference between the solid surface and the interior.
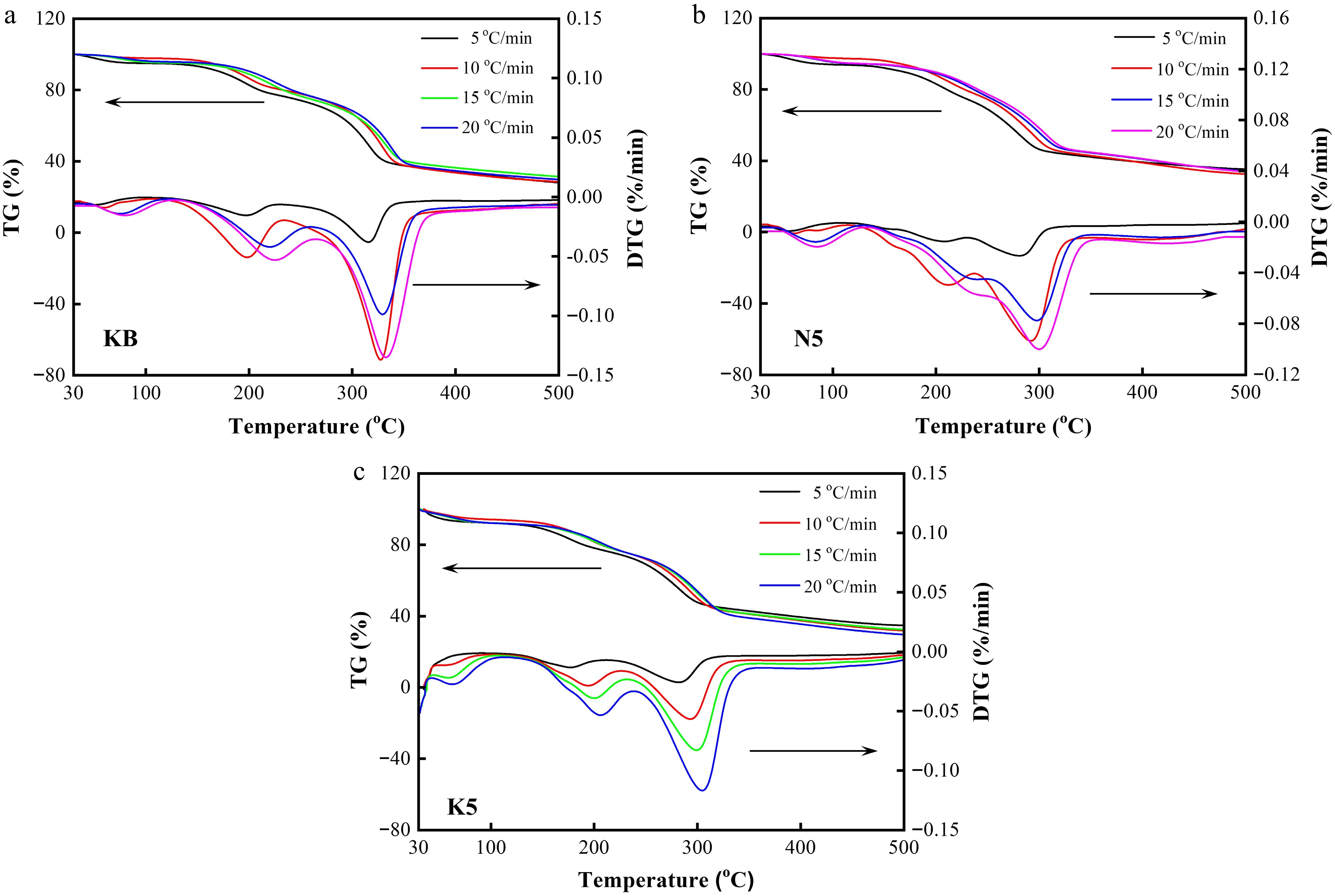
Figure 6.
TG and DTG curves of (a) the KB sample, (b) the N5 sample, and (c) the K5 sample under different heating rates (5, 10, 15, and 20 °C/min, respectively).
The thermogravimetric curves of reconstituted tobacco sheets, both with and without alkali metal salt, were investigated at heating rates of 5–20 °C/min. The apparent activation energy was calculated by linear fitting of the logarithm of the heating rate against 1/T under different conversion ratios (abbreviated as α, α = 0.1~0.8) using the FWO method. For this purpose, the following equation was used:
$ \text{lg}\beta=\text{lg}\dfrac{AE_{\alpha}}{\text{RG(}\alpha\text{)}}-2.315-0.4567\dfrac{E_{\alpha}}{\text{R}T}\mathit{ } $ (1) where,
$ \text{lg}\dfrac{{A}{{E}}_{\alpha}}{\text{RG(}\alpha\text{)}}-2.315 $ $ \ln\left(\dfrac{{\beta}}{{{T}}_{{P}}^{{2}}}\right)=\ln\left(\dfrac{{AR}}{{E}}\right)-\dfrac{{E}}{{R}{{T}}_{{P}}} $ (2) where TP is the peak temperature at the maximum weight loss rate obtained from the DTG curves, and E can be calculated from the slope of the linear fitting line by plotting ln(β/TP2) versus 1/TP for a given value of α.
According to Fig. 6, Eqs. (1) and (2), linear relationships between lgβ and 1/T × 103 and between
$ \text{l}\text{n(}\dfrac{{\beta}}{{{T}}_{{P}}^{{2}}}{)} $ 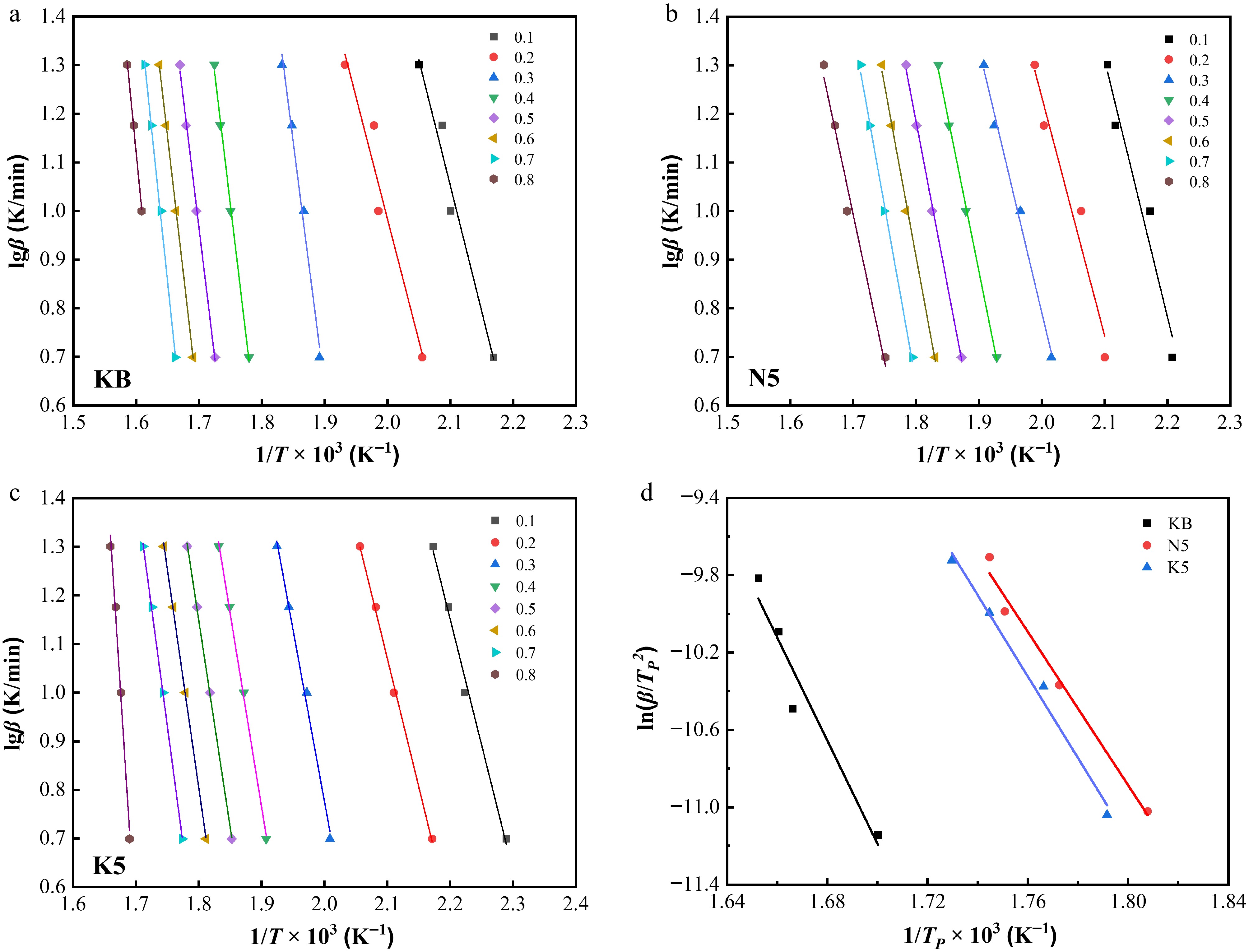
Figure 7.
Linear relationships between lgβ and 1/T × 103 at different conversion ratios (α) for (a) the KB sample, (b) the N5 sample, and (c) the K5 sample. (d) Linear relationships between ln(β/TP2) and 1/TP × 103 for the KB sample, the N5 sample, and the K5 sample.
Table 1. Kinetic parameters and squared correlation coefficients of the FWO method for the pyrolysis of the blank sample and the Na salt-doped and K salt-doped samples.
α KB N5 K5 Eα/(kJ·mol−1) R2 Eα/(kJ·mol−1) R2 Eα/(kJ·mol−1) R2 0.1 94.17 0.96 96.21 0.93 94.08 0.99 0.2 91.21 0.92 89.41 0.93 95.08 0.99 0.3 183.64 0.99 98.74 0.99 126.00 0.99 0.4 196.93 0.99 116.92 0.99 143.59 0.99 0.5 194.36 0.99 122.95 0.99 153.25 0.99 0.6 203.14 0.99 127.86 0.99 161.73 0.99 0.7 224.98 0.99 131.85 0.99 172.86 0.99 0.8 238.99 0.99 110.38 0.97 239.92 0.99 As displayed in Fig. 8, Eα values of reconstituted tobacco sheets calculated using the FWO method were not constants but changed with the progress of the reaction, indicating that the reaction mechanism controlling the decomposition process changed as the reaction proceeded, and the pyrolysis process of these samples seemed to be quite complex. The first stage of efficiency at a conversion range of 0.1–0.2 was mainly related to the evaporation of micromolecules of volatile components such as water contained in tobacco. The Eα calculated from all these samples showed little difference whether alkali salt was added or not. The efficiency of conversion changed from 0.2 to 0.3 along with the increase in temperature increase, and the Eα derived from all these samples appeared to increase. In the third stage of the reaction, when α was more than 0.3 but less than 0.7, Eα continued to increase in small increments. This may be mainly related to the pyrolysis of macromolecular substances such as cellulose, semi-cellulose, and lignin.
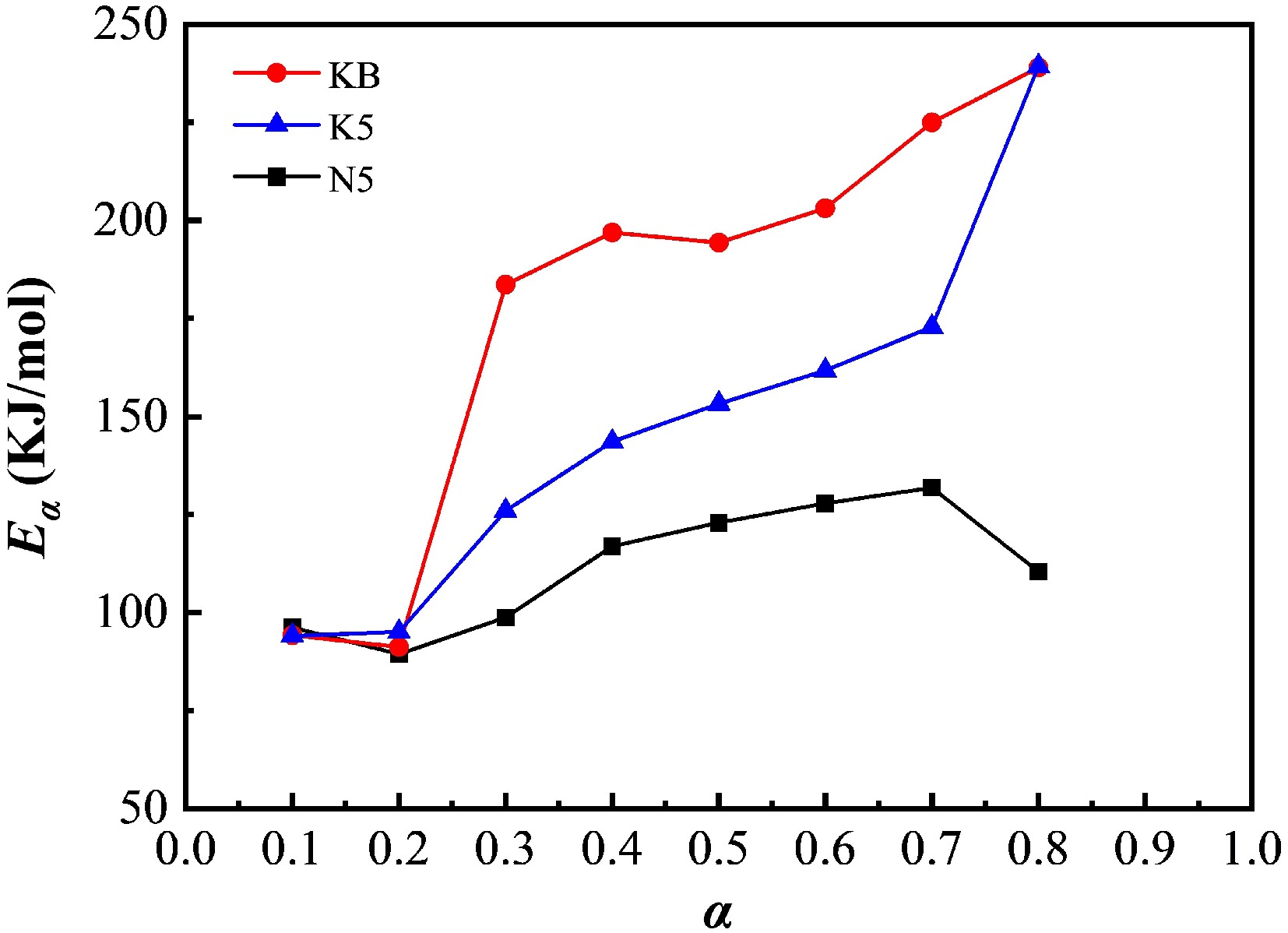
Figure 8.
Eα for the pyrolysis of reconstituted tobacco sheets (KB, K5, N5) at different conversion ratios (α).
The Eα values calculated using the FWO method are presented in Table 1. Alkali metal salts could significantly promote the decomposition of reconstituted tobacco sheets. For the N5 and K5 samples, the third stage's Eα value shifted from 183.64–224.98 (KB) to 98.74–131.85 or 126.00–172.86 kJ·mol−1, with an average decreased of 80.95 or 49.12 kJ·mol−1, respectively. According to the Kissinger method, the E values for the KB, N5, and K5 samples showed the same general trends, which were calculated to be 221.78, 164.84, and 175.07 kJ·mol−1, respectively. However, the apparent activation energy calculated using the FWO method showed an abnormally steep drop in the N5 sample's results (from 131.85 to 110.38 kJ·mol−1), which can be attributed to the secondary reactions resulting from the oversaturation and aggregation of Na salts added to reconstituted tobacco sheets, indicating the excessive addition of Na salt. It is noteworthy that even though alkali metal salts could not migrate into the crystalline structure of macromolecules, there was a strong tendency for the impregnated salts to break the existing chemical bonds in the main and branch chains of cellulose, etc., which can be attributed to their affinity to polymer organic groups[35]. With this clarification, it should be noted that the absorbed alkali metals are believed to decrease the maximum weight loss temperature and increase the weight loss during pyrolysis. In general, the apparent activation energy required during the pyrolysis of Na assisted was approximately 50% less than that of K-doped reconstituted tobacco sheets, which also indicates that the catalytic effect of the Na salt was superior to that of the K salt. This result is consistent with the conclusion that there is a weaker effect on the maximum weight loss rate temperature in the third weight loss stage and the total weight loss in the range of 100–300 °C of K-doped reconstituted tobacco sheets if the amount of Na and K added is the same.
-
By using TG-DTG and Py-GC/MS, this work pioneered a comprehensive study on the effects of adding alkali metal salts on reconstituted tobacco sheets' pyrolytic properties. The addition of Na salt or K salt provided an effective way to decrease the temperature of the maximum weight loss rate in the third weight loss stage and to increase the total weight loss between 100 and 300 °C. Notably, alkali metal salts play an important role in the catalytic cracking process of cellulose and lignin. therefore, the contents of key components in the pyrolysis gas, such as acetic acid, glycerol acetate, hydroxyacetone, furfuryl alcohol, butyrolactone, phenol, and megastigmatrienone displayed varying degrees of increase, which may suggest immense utility for optimizing fragrance and smoke quality. At the same time, through the combination of the FWO method and the Kissinger method, it was found that the decrease in the apparent activation energy was beneficial for the alkali metal salt-assisted low-temperature pyrolysis process, as the Eα value largely declined by about 80.95 and 49.12 kJ·mol−1 for Na and K-doped reconstituted tobacco sheets, respectively. This result can be attributed to the stronger Lewis acidity of Na+ and the stronger electrostatic interaction between Na+ and oxygen atoms in biomass matrices, which more effectively polarizes oxygen-containing functional groups such as the C–O bonds in cellulose or the ether linkages in lignin, thus facilitating bond cleavage and stabilizing transition states. By contrast, owing to the larger ionic radius and lower electrostatic interaction, K+ exerts a relatively weaker catalytic effect.
In summary, Na salts are more efficient in promoting the quality of pyrolysis gas at relatively low loadings (3 wt.%), whereas K salts act more moderately but require larger added amounts (5 wt.%). Although the low-temperature pyrolysis achieved here shows promising applications for high-quality heated tobacco products, the current research was unable to reveal the in-depth pyrolysis mechanism because of the complexity of tobacco's thermal reactions. In addition, sensory evaluations and biosafety evaluations based on toxicological assessments have not yet been carried out because of experimental constraints. Thus, it is necessary that future work focuses on the impact of adding alkali metal on the actual sensory quality, integrating advanced in situ analytical techniques with kinetic modeling to clarify the multi-step pyrolysis pathways, and incorporating toxicological assessments to ensure safe and reliable application.
-
We systematically elucidated the influence of alkali metal salts on the pyrolysis characteristics of reconstituted tobacco sheets. The introduction of Na+ and K+ salts lowered the temperature of the maximum weight loss rate and enhanced volatile evolution at 100–300 °C. Catalytic promotion of cellulose and lignin degradation led to elevated yields of key aroma precursors, implying potential improvement in smoke quality. Kinetic evaluation using the FWO and Kissinger methods confirmed that adding alkali metal markedly reduced the apparent activation energies, facilitating low-temperature pyrolysis. The stronger Lewis acidity and electrostatic interaction of Na+ accounted for its superior catalytic performance compared with K+. The proposed paradigm here shows great application potential for low-temperature pyrolysis of reconstituted tobacco sheets for heated tobacco products.
We acknowledgment the financial support from the Science and Technology Project of China Tobacco Henan Industrial Co. Ltd. (AW2022022 and BHW202402).
-
The authors confirm their contributions to the paper as follows: study conception and design: Wang Y, Zhao X, Zhang Z; data collection: Du J, Xu H, Ma S; analysis and interpretation of the results: Wang E, Tian H, Han L, Ge Z; draft manuscript preparation: Wang Y, Zhao X, Liu W. All authors reviewed the results and approved the final version of the manuscript.
-
All data generated or analyzed during this study are included in this published article and its electronic supplementary information files.
-
The authors declare that they have no conflict of interest.
- Supplementary Table S1 Contents of alkali metal in reconstituted tobacco sheets for heated tobacco products after soaking in alkali metal salt solutions with different concentration.
- Supplementary Table S2 Weight loss of reconstituted tobacco sheets with varying additions of Na salt (N1-N5) or K salt (K1-K5) in thermo-weightlessness process.
- Supplementary Tables S3 Semiquantitative results of volatile components in the pyrolysis gas of reconstituted tobacco sheets for heated tobacco products with different Na salt additions at 300 °C.
- Supplementary Table S4 Semiquantitative results of volatile components in the pyrolysis gas of reconstituted tobacco sheets for heated tobacco products with different K salt additions at 300 °C.
- Supplementary File 1 Supplementary materials and method to this study.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Wang Y, Du J, Xu H, Wang E, Tian H, et al. 2026. Alkali metal salt-assisted low-temperature pyrolysis of reconstituted tobacco sheets: research into salt addition, and reaction kinetics. Progress in Reaction Kinetics and Mechanism 51: e004 doi: 10.48130/prkm-0025-0029
Alkali metal salt-assisted low-temperature pyrolysis of reconstituted tobacco sheets: research into salt addition, and reaction kinetics
- Received: 06 August 2025
- Revised: 25 September 2025
- Accepted: 28 October 2025
- Published online: 10 February 2026
Abstract: Recent years have witnessed major advances in heated tobacco products. However, optimizing the aroma release and smoke generation of these products remains a formidable challenge. Alkali metal salts have emerged as effective catalysts for pyrolysis, demonstrating the dual benefits of enhanced catalytic efficiency and alleviating coke deposition. To effectively illustrate the effect of adding salt on the low-temperature catalytic pyrolysis performance of reconstituted tobacco sheets, thermogravimetric analysis and pyrolysis gas chromatography–mass spectrometry analyses were carried out, and the Flynn–Wall–Ozawa method and the Kissinger method were also applied to calculate reaction kinetics. The addition of the sodium (Na) salt or potassium (K) salt in reconstituted tobacco sheets was observed to significantly decrease the temperature corresponding to the maximum weight loss rate and increase the total weight loss with the range of 100–300 °C. Meanwhile, the content of key components in the pyrolysis gas gradually increased, indicating that the ability to catalyze cellulose and lignin pyrolysis improved. The results of dynamic analysis show that the apparent activation energy of reconstituted tobacco sheets decreased by an average of 80.95 or 49.12 kJ·mol−1 after adding Na or K salt when the conversion rate was 0.1–0.8. The approach reported herein not only exhibits a fundamental advance in the low-temperature pyrolysis of reconstituted tobacco sheets for heated tobacco products but also increases the concentration of pyrolysis gas and aromatic substances.
-
Key words:
- Alkali metal salt /
- Heated tobacco product /
- Kinetic analysis /
- Pyrolysis /
- Reconstituted tobacco sheet














