-
With the escalating global climate crisis and the urgent call for carbon neutrality, the transition from fossil fuels to renewable energy systems has become imperative to mitigate atmospheric CO2 levels and achieve the United Nations Sustainable Development Goals (SDGs)[1]. Among renewable energy sources, biomass has garnered significant attention due to its potential to be converted into diverse energy products (e.g., syngas, bio-oil, biochar) and functional chemicals, as well as its potential for carbon circularity and negative emissions[2,3]. According to the 2023 statistical data release by the International Energy Agency (IEA), modern bioenergy is the largest source of renewable energy globally, accounting for 55% of the end-use consumption market for renewable energy—surpassing the combined total of wind, solar, hydro, and geothermal energy[4].
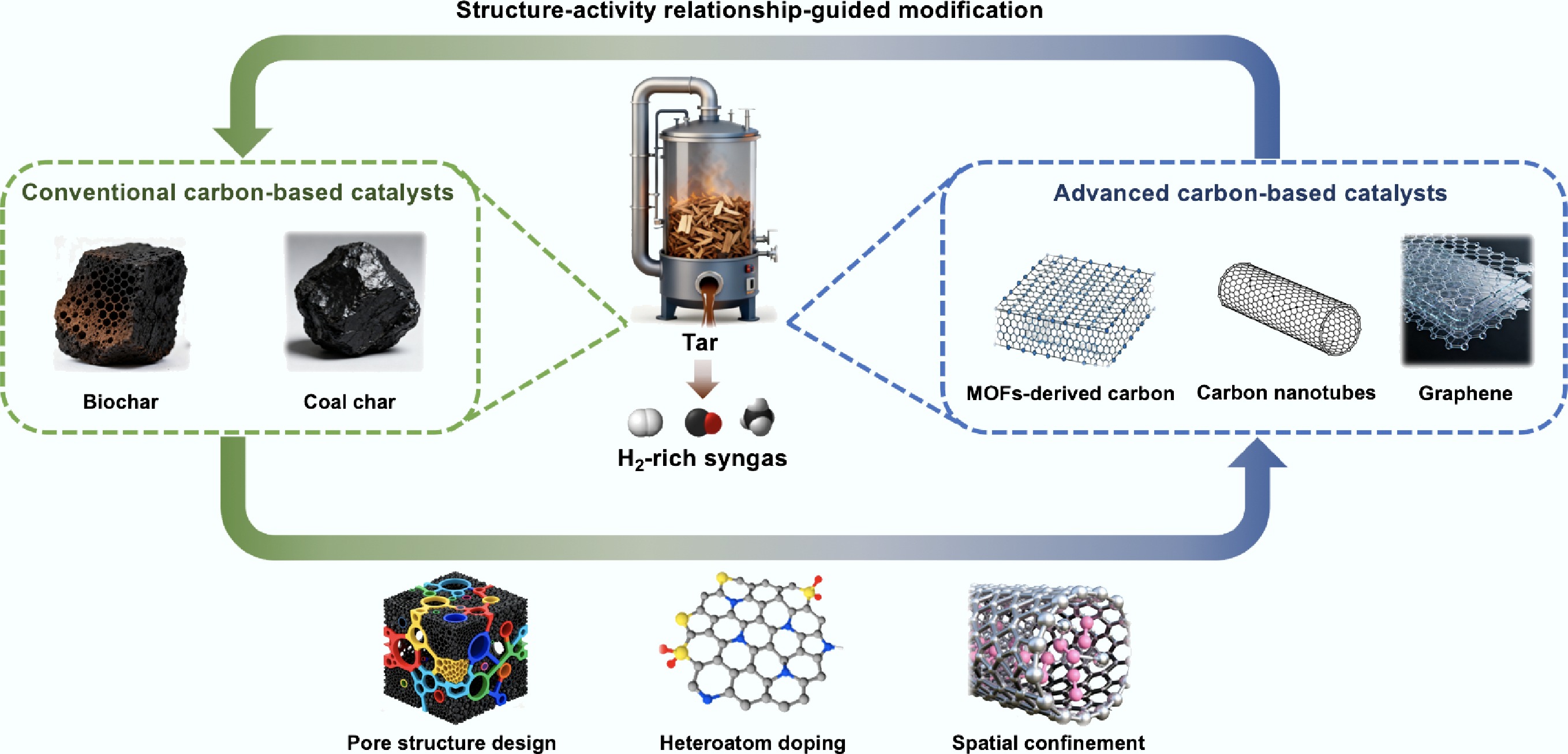
Currently, biomass gasification for syngas production is regarded as one of the most viable approaches for biomass resource utilization[5]. Gasification is a thermochemical process that converts biomass into renewable syngas (primarily CO, H2, and CH4) under oxygen-limited conditions, serving as feedstock for downstream applications, including heat/power generation and green methanol/biodiesel production[6,7]. However, the inevitable formation of tar remains a major obstacle in syngas production. Tar contains hundreds of harmful aromatic compounds that readily cause equipment blockage, corrosion, and affect syngas quality[8,9]. Simply removing tar physically from the syngas stream not only reduces biomass energy conversion efficiency but also poses risks of secondary pollution by generating coking wastewater. Therefore, efficient in-process tar conversion is essential for sustainable biomass gas production. Catalytic reforming of biomass tar (Fig. 1) is a key link in biomass gasification technology, which can effectively remove complex tar compounds and generate valuable syngas simultaneously[10]. During this process, the catalyst effectively lowers the reaction activation energy, facilitating the conversion of tar into small-molecule gaseous products, thereby enabling real-time tar removal[11]. This process can not only maintain equipment cleanliness, but also ensure seamless integration with downstream syngas utilization processes, thereby reducing energy waste[12].
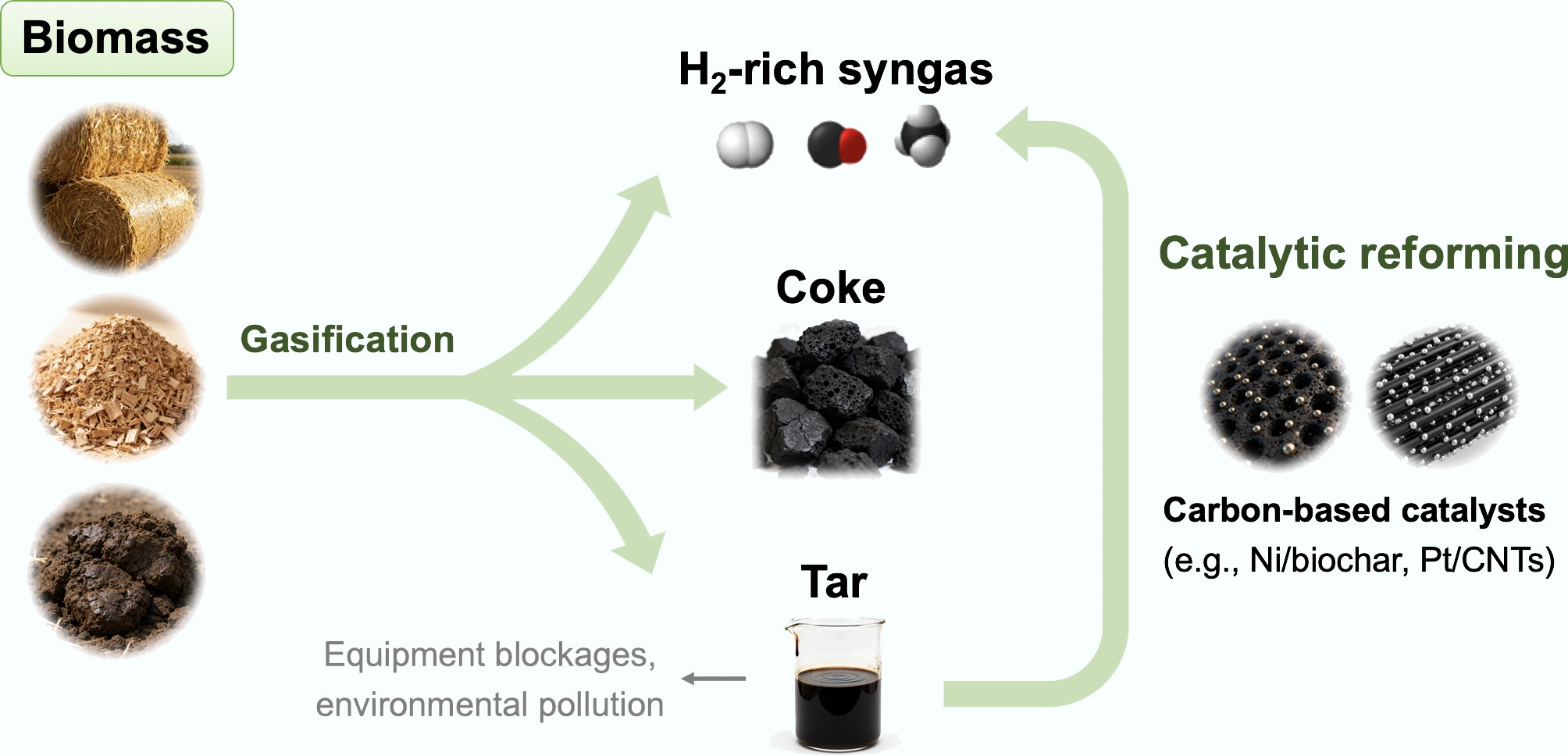
Figure 1.
Schematic illustrating the use of catalytic reforming technology to address the tar issue and enhance the yield of valuable syngas in the biomass gasification process.
The advancement of biomass tar catalytic reforming technology is fundamentally contingent upon innovations in catalyst development. Catalysts can be systematically classified into four principal categories based on the support functional properties: natural mineral-based catalysts, metallic oxide-based catalysts, molecular sieve-based catalysts, and carbon-based catalysts[13,14]. Among them, carbon-based catalysts have attracted extensive attention owing to their distinct properties. Table 1 compares the primary advantages and limitations of carbon-based catalysts and non-carbon-based catalysts in biomass tar catalytic reforming. Compared with conventional non-carbon-based materials, carbon-based catalysts exhibit well-developed porous structures and excellent metal dispersion capability, which effectively facilitate the adsorption of reactants, mass transfer processes, and the exposure of active sites[15,16]. In terms of reaction efficiency, carbon-based catalysts demonstrate outstanding low-temperature catalytic activity and energy-saving characteristics[17,18]. For instance, Ni-Co/carbon catalysts can achieve nearly complete tar conversion at 700 °C, whereas non-carbon-based materials typically require higher reaction temperatures (> 750 °C)[15]. Furthermore, the synergistic effect between carbon-based supports and metal components—such as enhanced reactant adsorption driven by electron transfer and the regulation of reaction pathways mediated by bimetallic interfaces—significantly enhances the selectivity towards target products (e.g., H2)[19−21]. In contrast, conventional non-carbon materials, constrained by micropore diffusion limitations or inert surfaces, often result in the formation of C1−C2 hydrocarbons (products of excessive tar cracking) and low H2 selectivity[22]. In addition, carbon-based supports can promote the precipitation of carbon atoms deposited during the reaction through the vacancies of metal particles, thereby facilitating the formation of graphitized carbon nanofibers (CNFs)[23,24]. This 'using carbon to manage carbon' approach can effectively address the long-standing issue of rapid deactivation of conventional catalysts caused by coke deposition on active sites, which endow carbon-based catalysts with unique advantages throughout the reaction process.
Catalyst categories Typical representatives Key advantages Critical limitations Optimal application scenarios Non-carbon-based catalysts Natural minerals (dolomite, olivine)
Metal oxides (Al2O3, MgO)
Molecular sieves (ZSM-5, HZSM-5)High intrinsic reforming activity
Strong C–C and C–H bond cleavage ability
Well-established industrial experienceSevere coke deposition and metal sintering
High operating temperature
Diffusion limitations for bulky tar molecules
Frequent regeneration requiredHigh-temperature secondary tar reforming
Centralized and industrial-scale gasification systemsCarbon-based catalysts Biochar, coal char, activated carbon
CNTs, graphene, MOFs-derived carbonsDeveloped and tunable pore structures
Excellent metal dispersion
Low-temperature activity
Strong metal-support interaction
Unique coke management via directional carbon transformationLower mechanical strength (especially conventional chars)
Surface heterogeneity
Higher cost and scalability issues for advanced carbonsLow-temperature tar reforming
Distributed biomass gasification
Long-term stable operation and advanced catalytic systemsIn recent years, researchers have developed a wide range of carbon-based catalysts, which exhibit significant variations in raw material sources, preparation methodologies, and structural properties. To better facilitate the research and development of carbon-based catalysts, this review classifies them into two categories: conventional carbon-based catalysts (char-based catalysts), and advanced carbon-based catalysts (MOFs derivatives, carbon nanotube-based catalysts, graphene-based catalysts, etc.)[30,31]. Conventional carbon-based catalysts hold promising application prospects in the catalytic reforming of biomass tar, attributed to their abundant raw materials, straightforward preparation processes, and intrinsic porous structures[32]. However, defects including inert surface chemical properties and poor mechanical strength restrict the catalytic efficiency and stability. More importantly, the inherent microscopic morphological heterogeneity of conventional carbon-based catalysts renders them difficult to establish stable structure-activity relationships, posing challenges in exploring reaction mechanisms and directions for catalyst modification. Utilizing advanced carbon materials with tunable and controllable microstructures as a platform enables precise regulation of pore architecture, surface chemistry, and metal-support interactions, offering a viable approach to address the limitations of conventional carbon materials[33,34]. This strategy not only provides optimization insights for enhancing the directional conversion of coke in carbon-based catalysts, but also establishes a robust framework for elucidating structure-activity relationships in carbon-based catalysts under external field-enhanced conditions, such as microwave-assisted processes, thereby promoting their practical application in the efficient conversion of biomass tar[35].
This review systematically explores the research progress of carbon-based catalysts in biomass tar conversion, with a focus on conventional carbon materials and advanced carbon materials, examining their structural properties, catalytic performance, mechanisms of action, and coke management strategies. It also addresses current challenges, modification strategies, and future directions for the development of efficient, stable, and sustainable carbon-based catalysts for biomass tar reforming. By integrating insights from structure-activity relationships and advanced material design, this work aims to provide a comprehensive framework for the synergistic development of carbon-based catalytic systems.
-
Biomass tar is defined as a complex mixture of condensable hydrocarbons with a molecular weight higher than that of benzene, primarily composed of aromatic compounds, oxygen-containing compounds, and polycyclic aromatic hydrocarbons (PAHs), which is formed during gasification or pyrolysis[36]. Tar can be further characterized as the substance in syngas that condenses inside gasifiers or downstream equipment[37], posing significant challenges to the utilization of syngas. The classification of biomass tar is typically conducted based on its formation stages and chemical composition: primary tar includes cellulose-derived products (e.g., levoglucosan), hemicellulose-derived compounds, and lignin-derived products; secondary tar comprises phenolics and olefins; tertiary tar consists of alkyl derivatives of aromatics and condensed polycyclic compounds such as naphthalene, anthracene, and pyrene[38,39]. The distribution of these tar fractions depends on gasifier design, with updraft gasifiers producing primarily primary tar and downdraft gasifiers generating more tertiary tar due to higher temperature-induced cracking[40,41].
In terms of physicochemical properties, biomass tar is characterized by high viscosity, high condensability, high carbon content (54%–63%), high calorific value (24–36 MJ kg−1), and low sulfur and nitrogen contents[42−44]. Its light fractions (< 200 °C) have lower viscosity and higher hydrogen content (6.3%–6.8%), making them suitable for use as liquid fuels; while its heavy fractions (> 200 °C) have higher viscosity and are appropriate for application as chemical raw materials (e.g., antipolymerization agents, plasticizers)[45,46]. Furthermore, the properties of tar are significantly influenced by raw material types (for instance, the aromaticity of wood chip tar is higher than that of straw tar), and pyrolysis conditions (temperature, catalysts); after catalytic cracking, the proportion of aromatic hydrocarbons can be further increased (reaching over 50%)[47−49]. Biomass tar is both a pollutant and a potential energy and chemical resource, and its efficient utilization necessitates the integration of technologies like catalytic reforming to minimize environmental impacts[50].
Catalytic reforming of biomass tar refers to a thermochemical process that, under the action of a catalyst, uses reaction media such as steam (H2O), carbon dioxide (CO2), or oxygen (O2), to convert the tar generated during the biomass gasification process into high-value syngas (mainly composed of H2, CO, CO2, and CH4)[51−53]. By reducing tar content and increasing syngas yield, this technology can address issues caused by tar, including equipment corrosion, environmental pollution, and low energy efficiency. Compared with thermal cracking, which typically requires temperatures above 1,000 °C, catalytic reforming reactions can occur at relatively low temperatures, generally in the range of 500–900 °C[32,54,55]. This significant reduction in reaction temperature leads to lower energy consumption and potentially lower equipment costs, making catalytic reforming a more cost-effective option.
By employing steam as a reaction medium, catalytic steam reforming has emerged as a highly attractive approach for efficiently removing tar while simultaneously producing valuable syngas (primarily a mixture of H2 and CO)[56]. During the catalytic steam reforming of tar, multiple reactions occur concurrently, as outlined in the following Eqs (1) to (8), with their prevalence varying depending on the reaction conditions[57]:
Steam reforming:
$ {\rm{C}}_{\rm{n}}{\rm{H}}_{\rm{m}}({\rm{tar}}) + {\rm{n}}{\rm{H}}_2 {\rm{O}} \rightarrow ({\rm{n}} +{\rm{m}}/2) {\rm{H}}_2+ {\rm{n}}{\rm{CO}} $ (1) Dry reforming:
$ {\rm{C}}_{\rm{n}}{\rm{H}}_{\rm{m}}({\rm{tar}}) + {\rm{n}}{\rm{CO}}_2\rightarrow({\rm{m}}/2) {\rm{H}}_2+ 2{\rm{n}}{\rm{CO}} $ (2) Partial oxidation:
$ {\rm{C}}_{\rm{n}}{\rm{H}}_{\rm{m}}({\rm{tar}}) + ({\rm{n}}/2) {\rm{O}}_2\rightarrow({\rm{m}}/2) {\rm{H}}_2+ {\rm{nCO}} $ (3) Thermal cracking:
$ {\rm{C}}_{\rm{n}}{\rm{H}}_{\rm{m}}({\rm{tar}}) \rightarrow {\rm{C}}^*+ {\rm{C}}_{\rm{x}}{\rm{H}}_{\rm{y}}({\rm{smaller}}\; {\rm{tar}}) + {\rm{gas}}$ (4) Carbon formation:
$ {\rm{C}}_{\rm{n}}{\rm{H}}_{\rm{m}}({\rm{tar}})\rightarrow {\rm{nC}}+ ({\rm{m}}/2) {\rm{H}}_2 $ (5) Hydrocracking/hydroreforming:
$ {\rm{C}}_{\rm{n}}{\rm{H}}_{\rm{m}}({\rm{tar}}) + {\rm{H}}_2\rightarrow {\rm{CO}} + {\rm{H}}_2+ {\rm{CH}}_4+ \cdots + {\rm{coke}} $ (6) Water-gas shift (WGS) reaction:
${\rm{CO}} + {\rm{H}}_2{\rm{O}} \rightarrow {\rm{H}}_2+ {\rm{CO}}_2 $ (7) Methanation:
$ {\rm{CO}} + 3{\rm{H}}_2\rightarrow {\rm{CH}}_4+ {\rm{H}}_2{\rm{O}} $ (8) To further enhance tar catalytic reforming activity, active metal components and supports can be designed during the development of carbon-based catalysts to facilitate different reaction steps.
Insights into tar reforming catalysts
-
In carbon-based catalyst systems, the effectiveness of biomass tar reforming is governed by the synergistic interaction between carbon supports and metal active sites. Unlike conventional oxide supports, carbon materials are not inert carriers but actively participate in tar conversion through their porous structure, surface chemistry, and electrical conductivity[58]. These intrinsic properties enable enhanced adsorption of bulky tar molecules, improved mass and electron transfer, and unique regulation of coke evolution, which collectively determine catalytic activity, stability, and long-term performance[59]. Therefore, this section focuses specifically on carbon supports and metal active sites in carbon-based catalysts, emphasizing their distinct roles in tar reforming.
Supports
-
Carbon supports play a central role in determining the performance of carbon-based catalysts for biomass tar reforming. Unlike non-carbon supports, carbon materials provide tunable pore structures, rich surface functional groups, and intrinsic electrical conductivity, which collectively enhance tar adsorption, mass transfer, and reaction kinetics[60]. More importantly, carbon supports actively regulate coke formation and transformation, enabling the conversion of amorphous carbon into ordered graphitic structures and thereby mitigating catalyst deactivation[61].
Biochar, a by-product of biomass gasification, has been widely used as a catalyst support in reforming reactions due to its tunable pore structure, high specific surface area (SSA), and good thermal stability[60,62,63]. However, comparative studies indicate that the catalytic performance of biochar-supported systems is governed not only by surface area, but also by surface chemistry and inherent inorganic species. For example, Wang et al.[64] demonstrated that activating peat char via KOH or CO2 significantly enhanced its interaction with Ni, leading to a high toluene conversion of 95.3%. This improvement highlights the role of pore development in facilitating metal dispersion and reactant accessibility. In contrast, Xu et al.[65] showed that biochar with abundant inherent K species could markedly enhance Ni-based catalytic activity even at relatively low temperatures (450–500 °C), primarily by increasing local water adsorption near Ni sites rather than solely relying on textural properties. Compared with alkali-modified biochar systems, Li et al.[66] reported that Fe-doped corn stalk char achieved similarly high toluene conversion (94.3%) in CO2 reforming, which was attributed to its large surface area and interconnected pore channels that promoted efficient mass transfer and active site utilization.
Activated carbon (AC) materials with high specific surface area (SSA) and porosity can provide abundant contact areas between catalysts and reactants[67,68]. The active phases and promoters are typically loaded on the support surface to ensure catalyst stability. Xu et al.[69] synthesized a nickel-silica nanocomposite coated with carbon (Ni-SiO2@C) on monolithic wood-derived carbon featuring abundant channels. The Ni-SiO2 was introduced into the channels of biomass-derived carbon via an evaporation-induced self-assembly method, followed by heat treatment to fabricate Ni-SiO2@C/WC. The Ni-SiO2@C formed a thin layer covering the carbon channels through the formation of Ni−O−Si or C−C/C−O−Si bonds. Reforming reactions were conducted in a fixed-bed reactor using toluene as the reactant. The results indicated that reaction temperature and time were found to significantly affected toluene conversion, with conversion increasing as temperature rose. Among various catalysts, Ni-15SiO2@C/WC exhibited the highest performance, achieving 97% toluene conversion, attributed to its unique morphology and structure. The uniformly coated thin-layer Ni-SiO2@C protected particles from aggregation, while the multi-level channels ensured efficient mass transfer and gas diffusion.
Carbon nanotubes (CNTs) have emerged as a promising support material in catalysis, garnering significant attention due to their large surface area, excellent electrical conductivity, and high thermal stability[70,71]. Shahsavar et al.[72] developed Ce- or Zr-doped CuZn/CNTs catalysts, demonstrating that the preparation method profoundly influences their morphology, structure, and performance. Different synthesis approaches for CeCuZn/CNTs resulted in varied methanol conversion efficiencies and selectivities toward H2, CO2, and CO. Notably, the CeO2-promoted CuZn/CNTs catalyst prepared via the impregnation method achieved an 85.2% methanol conversion, while the microwave-assisted polyol method yielded CeCuZn/CNTs with the highest activity at 300 °C, highlighting the critical role of synthesis techniques in optimizing catalytic performance.
Metal active sites
-
In carbon-based catalysts, metals function as metal active sites rather than independent catalytic phases. Their catalytic performance is strongly influenced by the physicochemical properties of the carbon support, such as surface functional groups, defect sites, and electrical conductivity[73,74]. Carbon supports regulate the dispersion, electronic structure, and stability of metal active sites, thereby controlling C–C and C–H bond activation, syngas selectivity, and coke formation pathways during tar reforming[75,76]. Metals commonly used in biomass tar conversion can be classified into noble metals (including Pt, Pd, etc.) and transition metals (including Ni, Co, Fe, etc.).
Noble metal catalysts exhibit exceptional catalytic activity, robust sulfur resistance, and long-term stability in tar steam reforming[77,78]. Tomoshige et al.[79] systematically evaluated noble metal catalysts supported on CeO2/SiO2 for tar steam reforming and compared their performance with commercial Ni-based catalysts. The study revealed that the Rh/CeO2/SiO2 catalyst outperformed Ni-based counterparts, with Rh demonstrating significantly higher activity among noble metals. The selectivity order was determined to be Rh > Pt > Pd > Ru = Ni. Recent research has focused on Ru supported on 12SrO–7Al2O3 nanocages, which effectively incorporates active oxygen ions into the framework. This catalyst showed superior performance in steam reforming of dodecane and toluene, featuring high coking resistance[80,81]. To enhance economic viability, low-loadings (< 1 wt%) of noble metals on specialized supports have been developed, achieving high catalytic activity at low temperatures and emerging as promising candidates for industrial tar reforming[82,83]. Despite these advancements, the high cost of noble metals remains a critical challenge compared to non-noble alternatives.
Ni-based catalysts are widely favored in the catalytic reforming of tar model compounds and real biomass tar due to their low cost, earth-abundant reserves, and exceptional capability to cleave C–C/C–H bonds in aromatic molecules[84]. As summarized in Table 2, Ni-based systems generally exhibit high initial tar conversion. However, their long-term performance varies significantly depending on catalyst composition and structural stabilization strategies. A direct comparison between monometallic and bimetallic systems reveals that rapid deactivation of conventional Ni-based catalysts is primarily associated with metal sintering and amorphous carbon accumulation under reforming conditions. For instance, Claude et al.[85] demonstrated that while 10Ni/Al2O3 suffered severe activity decay after only 5 h due to coke-induced site blockage, the introduction of Mn and Mo effectively stabilized Ni particles and suppressed disordered carbon formation, enabling the bimetallic 10Ni-2Mn-2Mo/Al2O3 catalyst to maintain high conversion over 30 h of continuous operation. Similarly, Zou et al.[86] showed that Ni–Fe/palygorskite outperformed its monometallic counterparts, which was attributed to reduced Ni particle size and enhanced metal dispersion, highlighting the critical role of synergistic bimetallic interactions in improving both activity and durability. Collectively, these studies indicate that secondary metal doping does not merely enhance initial activity, but fundamentally alters deactivation pathways by regulating metal–support interactions and coke evolution behavior.
Table 2. Comparison of catalytic activities of different Ni-based catalysts for the conversion of tar model compounds and biomass tar
Catalyst Feedstock Reaction condition Conversion (%) Ref. Ni/liginte char Toluene 650 °C, S/C = 3.4 83.0 [87] Ni/ZSM-5 Toluene 600 °C, S/C = 2 83.1 [88] Fe-Ni/AC Toluene 600 °C, S/C = 2 93.8 [89] Ni-Pt/Ce0.8Zr0.2O2 Toluene 600 °C, S/C = 3 96.0 [90] Ni/biochar Naphthalene 900 °C, S/C = 2 92.0 [91] Ni/biochar Wheat straw 800 °C, S/B = 4 90.0 [92] Ni/Al2O3 Wood residue 550 °C, S/B = 0.6 85.2 [93] Ni/CeO2/Al2O3 Wood residue 550 °C, S/B = 0.6 92.4 [93] Ni/La2O3/Al2O3 Pine wood 600 °C, S/B = 4 96.4 [94] Ni-Co/Al2O3 Pine wood 600 °C, S/B = 4 99.0 [95] a S/C: Steam-to-carbon molar ratio; b S/B: Steam-to-biomass mass ratio. Therefore, developing efficient and low-cost active metals or metal combinations, as well as exploring methods to enhance the stability of Ni-based catalysts represents an important research direction in this field.
Tar reforming mechanisms
-
The conversion of biomass tar on carbon-based catalysts involves two critical and interrelated processes: active reforming, and coke transformation. Active reforming primarily refers to the catalytic conversion of gaseous or adsorbed tar molecules into small-molecule products through metal-catalyzed C–C and C–H bond activation[96]. This process is dominated by the intrinsic activity of metal sites and directly determines tar conversion efficiency and syngas yield. In contrast, coke transformation describes the evolution behavior of solid carbon species formed during tar cracking, including their nucleation, migration, precipitation, and structural rearrangement on the catalyst surface[97]. Rather than contributing directly to tar conversion, coke transformation governs the deactivation pathway and long-term stability of the catalyst by determining whether deposited carbon passivates active sites or is redirected into ordered graphitic structures. The unique advantage of carbon-based catalysts lies in their ability to decouple these two processes: while metal sites drive active reforming, the conductive and structurally tunable carbon matrix redirects deposited carbon toward ordered graphitic structures instead of inactive amorphous coke. This dual-function mechanism forms the basis of the so-called 'coke management' concept that distinguishes carbon-based catalysts from conventional oxide or mineral systems.
The detailed mechanism during 'coke management' can be illustrated as below: First, large tar molecules are physically adsorbed onto the abundant porous structure and extensive surface area of the carbon support. Wang et al.[98] demonstrated that bamboo-derived microporous activated carbon with high surface area exhibited strong adsorption toward toluene, with adsorption characteristics governed by pore diffusion and surface interaction mechanisms. Subsequently, the oxygen-containing functional groups on the carbon support surface interact with the aromatic rings or methyl groups in tar molecules via intermolecular forces such as n-π bonds, π-π bonds, and hydrogen bonds[99]. This interaction results in stronger chemical adsorption, enabling the initial activation and enrichment of tar molecules. Tar molecules adsorbed on the carbon support then migrate to the metal-support interface, or directly contact metal particles. Acting as active centers, metal nanoparticles efficiently catalyze the cracking of large tar molecules—for instance, breaking the C–H bonds and C–C bonds of toluene—producing small-molecule gases and active carbon species (e.g., C*, CHx fragments)[100]. This behavior was observed in Ni nanoparticles embedded within carbon nanofibers, which showed high tar conversion and effective catalytic cracking performance[101]. The active carbon atoms generated at the cracking interface dissolve into the bulk phase of the metal nanoparticles. Owing to the presence of concentration gradients or temperature gradients across the metal particles, the carbon atoms diffuse from the contact surface of the particles (the side where the cracking reaction occurs), to the opposite side[102]. When the carbon atoms become supersaturated on the opposite side, they precipitate out of the metal nanoparticles. The precipitated carbon atoms undergo epitaxial growth on specific low-energy crystal planes of the metal crystal (e.g., the [111] plane of Ni), forming a graphene sheet structure[103]. Recent experimental work investigating carbon deposition mechanisms during tar catalytic reforming on Ni-based catalysts confirmed that filamentous carbon growth occurs via carbon dissolution and subsequent reprecipitation at the metal surface[104]. Here, the crystal structure of the metal particles acts as a template for graphitization. The final morphology of the carbon nanofibers (e.g., tubular, lamellar) is controlled by the size and morphology of the metal particles, as well as their interaction with the support. Mazo et al.[105] reported that strong interactions between Ni and a porous glass support favored a base-growth mechanism of carbon nanofibers, with the support anchoring Ni particles and influencing fiber morphology. Generally, the size of the metal particles determines the diameter of the carbon nanofibers; small and uniform metal particles facilitate the formation of carbon nanofibers with consistent diameters[106]. When the interaction between the metal particles and the support is weak, the metal particles may be 'lifted' by the newly formed graphite layers and remain at the top of the fibers to continue catalyzing growth[107]. In contrast, when the interaction is strong, the metal particles are firmly anchored to the support surface. Carbon fibers then 'grow upward' from above the metal particles, while the metal particles stay at the bottom of the fibers[108].
The catalytic mechanisms of carbon-based catalysts in biomass tar conversion primarily involve metal-support interactions (MSI) between the carbon matrix and active metal species. As shown in Fig. 2, the hierarchical porosity (micro/mesopores) and abundant surface functional groups (e.g., -OH, -COOH, -SO3H) of carbon supports facilitate tar molecule adsorption via hydrogen bonding and π−π interactions, while enhancing mass transfer to metal active sites[109−111]. Dispersed metal nanoparticles weaken C–C/C–H bonds through electron transfer, promoting tar cracking and steam reforming into syngas (H2 + CO)[23,111,112]. Moreover, surface oxygen-containing groups (e.g., carbonyls, quinones) can participate in redox cycles, stabilizing intermediates and suppressing carbon deposition via dynamic oxidation of CHx precursors[113,114]. Additionally, some unique nanostructured carbons (e.g., MOFs-derivatives) spatially confine metal particles to mitigate sintering, while conductive frameworks accelerate electron transfer[115]. Acid-base interactions, such as pyridinic-N sites stabilizing phenolic intermediates, further optimize reaction pathways[88,116]. Advanced in situ characterization (e.g., XAS, ETEM) reveals dynamic structural evolution, guiding the design of robust catalysts with tailored active sites and defect engineering[88,114].
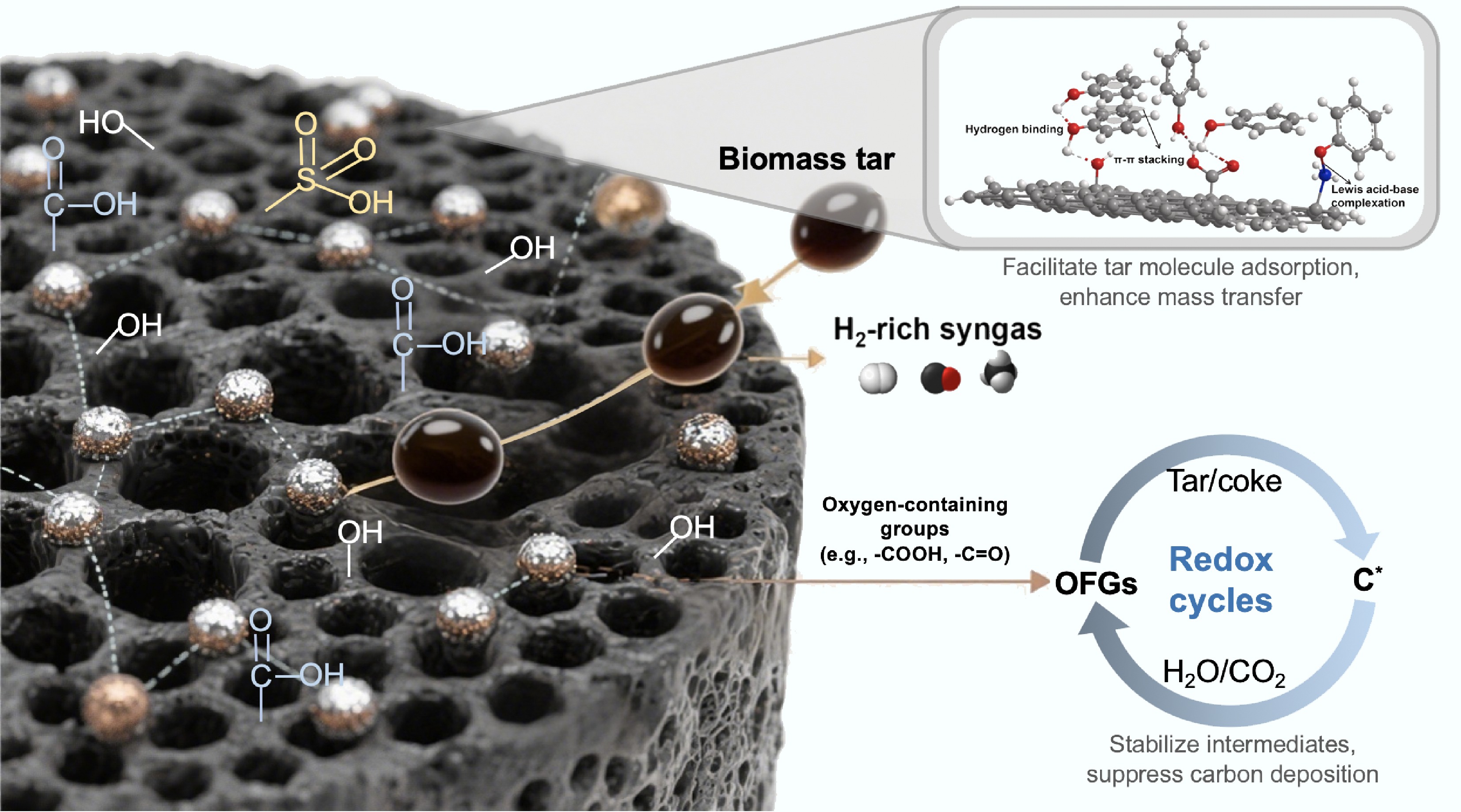
Figure 2.
Schematic diagram of the catalytic mechanism of carbon-based catalysts in biomass tar conversion.
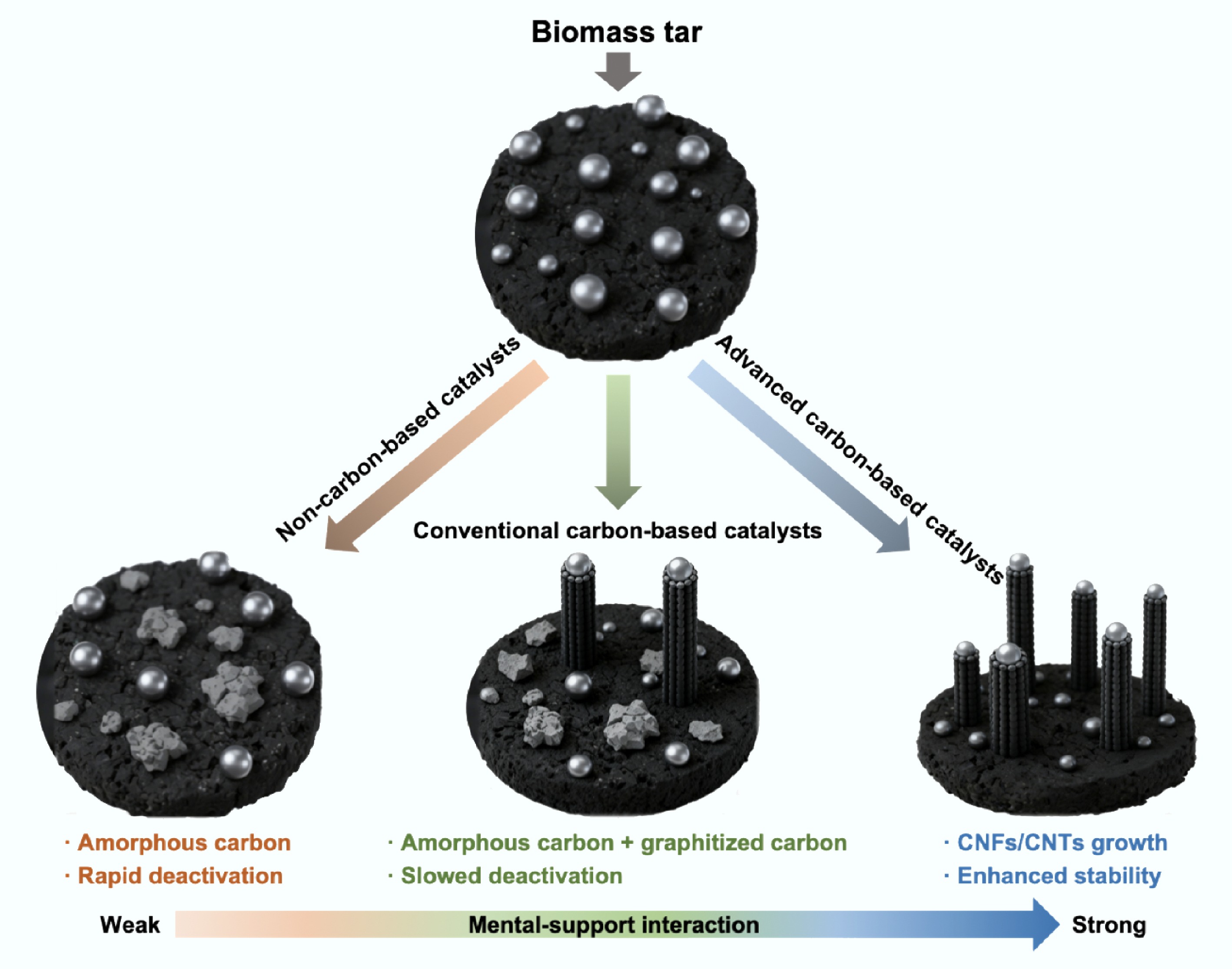
As is known, coke deposition is a bottleneck issue in the catalytic reforming of tar. Its deposition on the catalyst surface covers the active sites, thereby leading to catalyst deactivation[117]. As illustrated in Fig. 3, the evolution pathways of coke during biomass tar reforming are strongly dependent on the nature of the catalyst support and the strength of metal-support interactions. In non-carbon-based catalysts with weak metal-support interactions, reactive carbon species generated from tar cracking tend to deposit randomly as amorphous carbon, rapidly encapsulating metal particles and blocking active sites[118]. For conventional carbon-based catalysts, moderate metal-support interactions partially regulate carbon deposition, leading to the coexistence of amorphous carbon and short carbon filaments, which slows, but does not completely prevent catalyst deactivation[119]. In contrast, advanced carbon-based catalysts with strong metal-support interactions enable a carbon dissolution-precipitation mechanism, in which reactive carbon species dissolve into metal nanoparticles and subsequently precipitate in an ordered manner as carbon nanotubes or graphitic structures[120]. This directed conversion of amorphous coke into structured carbon effectively transforms coke from a deactivation factor into a dynamically regulated product, highlighting the critical role of metal-support interactions in achieving efficient coke management and long-term catalyst stability.
In summary, carbon-based catalysts not only significantly enhance tar conversion efficiency and syngas yield in the process of biomass tar catalytic reforming, but also achieve targeted regulation of coke deposition through their unique coke management mechanism, effectively mitigating the rapid deactivation issue caused by coke deposition in traditional catalysts. Therefore, systematic research on the structural design, performance optimization, and reaction mechanisms of carbon-based catalysts holds significant theoretical value and practical importance for advancing the efficient and clean conversion of biomass tar.
-
Conventional carbon-based catalysts, predominantly derived from biomass or coal carbonization processes, represent the most practically accessible class of carbon materials for biomass tar reforming[121]. Table 3 lists the general advantages and challenges of commonly used conventional carbon-based catalysts (e.g., biochar, coal char) in the catalytic reforming of biomass tar. Although conventional carbon-based catalysts exhibit advantages such as low-temperature activity and economic feasibility, their poor mechanical strength and insufficient stability have restricted their practical applications[122−124]. Therefore, there is an urgent need to design catalyst modification strategies to enhance the application potential of conventional carbon-based catalysts.
Table 3. General advantages and challenges of catalysts with biochar or coal char as the support[125−130]
Support Advantages Challenges Biochar Low-temperature high efficiency
Excellent pore structure and surface functional groups
Unique coke conversion mechanism
Sustainability and low costPoor mechanical strength
Poor surface chemical stability
Interference from alkali metal ash
Difficulty in coke controlCoal char Excellent pore structure
Good metal dispersibility
Directional coke conversion
Low cost and recyclableMetals are prone to sintering
Insufficient mechanical strength
Prominent coke issue
High variability in raw materialsBiochar
-
Biochar is a sustainable and low-cost carbon-based material, defined as a carbon-rich, porous solid, produced through the thermal decomposition of biomass under oxygen-limited conditions at moderate temperatures (350–700 °C)[131−133]. Biochar-supported catalysts exhibit highly variable performance in biomass tar reforming, primarily due to differences in feedstock origin, pyrolysis conditions, and inherent inorganic species[27,134]. Unlike engineered carbon materials, biochar possesses a non-uniform pore structure and chemically heterogeneous surface, which complicates the establishment of reproducible structure-activity relationships[113].
Numerous experimental studies have confirmed the effectiveness of biochar-supported catalysts in biomass tar conversion. However, their catalytic behaviors vary considerably depending on char origin, metal-support interaction, and reaction conditions. For instance, Wang et al.[135] reported that Ni supported on coal char outperformed its wood-char counterpart in terms of tar removal efficiency and short-term stability, which can be attributed to the higher aromaticity and more ordered carbon structure of coal char that favor stronger metal anchoring and directed coke evolution. In contrast, Ni supported on red cedar char exhibited pronounced sensitivity to operating parameters, where elevated temperature and pressure enhanced tar conversion, whereas excessive steam led to activity suppression due to competitive adsorption and possible metal site blockage[136]. Compared with Ni-based systems, Fe-supported biochar catalysts display a distinct selectivity pattern: they preferentially promote the conversion of oxygenated, non-aromatic species at lower temperatures (< 700 °C), while facilitating secondary aromatization reactions under high-temperature steam reforming conditions (> 700 °C)[137]. These comparative results indicate that biochar-supported catalysts are not universally interchangeable; rather, their performance is governed by the interplay between char structure, metal identity, and reaction environment, underscoring the necessity of tailoring both support properties and active metal selection for targeted tar reforming applications.
In the process of biomass tar conversion, the biochar-supported catalysts exhibit excellent performance in coke management. Chen et al.[138] found in their study on the Ni-Ca/BC catalyst that the addition of calcium reduced the nickel grain size, effectively directing the conversion of amorphous carbon deposits to CNTs, which significantly reduced the carbon deposition amount to 3.6 mmol g−1, much lower than that of the conventional Ni/Al2O3 catalyst (> 15 mmol g−1). In terms of catalytic performance, biochar supports exhibit remarkable low-temperature high efficiency. Sun et al.[109] synthesized the Ni-Ca/BC catalyst, which can achieve a tar conversion rate of 91.8% and an ideal H2/CO ratio of 1.05 at 700 °C, whereas conventional zeolite-based catalysts require temperatures above 800 °C to achieve similar results. This high efficiency stems from the high specific surface area and mesopore-dominated hierarchical pore structure of biochar, which not only facilitates the adsorption and mass transfer of tar molecules but also enables the high dispersion of metal nanoparticles[139]. Furthermore, the addition of calcium in Ni-Ca/BC further optimizes metal dispersion and inhibits high-temperature sintering[109].
In terms of economy and sustainability, biochar supports have prominent advantages. Derived from agricultural or forestry waste (e.g., straw, bamboo residues), biochar is produced at 500–800 °C, significantly lower than molecular sieves (> 1,000 °C), reducing costs by over 60%[140,141]. Shi et al.[142] loaded Zr on pyrolytic char of bamboo waste as a support; its raw material cost is only one-fifth of that of conventional alumina catalysts, and it realizes the closed-loop utilization of waste biomass.
However, biochar supports still face numerous challenges in practical applications. They suffer from insufficient mechanical strength and wear resistance (Mohs hardness less than 3), making them prone to abrasion in fluidized bed environments with high shear force[143]. Go et al.[144] reported that the particle breakage rate of BC-supported catalysts exceeds 20% after 50 h of continuous operation, whereas Al2O3 supports can operate stably for over 100 h under the same conditions. The surface chemical stability of biochar supports is another issue. Acidic functional groups (e.g., sulfonic groups) are liable to hydrolyze and leach in high-temperature steam environments, resulting in a drop in the catalytic efficiency of functionalized BC catalysts from 95% to 77.6% after five reuse cycles[142]. Meanwhile, inherent alkali metal ash (e.g., K+, Ca2+) in raw materials may cover active sites, weakening the metal-support interaction[145]. Regulating the type of carbon deposition also poses difficulties. When metal dispersion is uneven, random carbon deposits will quickly cover active sites, accelerating catalyst deactivation[146]. Additionally, the number and strength of surface acidic sites on biochar are generally lower than those of zeolite molecular sieves (e.g., ZSM-5), which limits its ability to deeply crack heavy tar components. Comparative studies have shown that biochar supports achieve a selectivity of 51.98% for polycyclic aromatic hydrocarbons (PAHs), while ZSM-5 can enhance the selectivity for monocyclic aromatic hydrocarbons (MAHs) to 21.82%[147,148].
To address the aforementioned limitations, several modification strategies have been developed to enhance the performance of biochar-supported catalysts. Composite material design involves combining biochar with clay minerals (e.g., bentonite), ceramic oxides (e.g., SiO2, Al2O3), or carbon nanomaterials to form hybrid supports, significantly improving mechanical strength and thermal stability[149,150]. Surface modification via acid/alkali treatment, oxidation, or heteroatom doping can tailor surface chemistry, strengthen metal anchoring, and regulate hydrophilicity/hydrophobicity, thereby enhancing catalytic activity and steam resistance[151]. Structural optimization through physical/chemical activation or template methods can create more ordered hierarchical pore networks, promoting mass transfer of large tar molecules[152]. Metal site engineering, such as introducing a second metal (e.g., Ca, Fe, Co) or employing alternative loading methods (e.g., ion exchange) can improve metal dispersion, inhibit sintering, and optimize coke conversion pathways[153]. Despite these improvements, the enhancement effects are often limited, and the underlying mechanisms—such as the precise role of heteroatoms in metal-support interactions, or the long-term stability of composite interfaces under reactive conditions—remain insufficiently elucidated, hindering the rational design of next-generation biochar catalysts.
Coal char
-
Coal char is another widely used char-based catalyst support. In fundamental laboratory investigations, chars generated from coal pyrolysis and gasification have emerged as promising supports for tar reforming catalysts[154]. Coal char supports exhibit outstanding performance in optimizing pore structure and metal dispersibility. For instance, deashed fat coal exhibits a mesopore-dominated structure (> 60% mesopores, 2–5 nm), which facilitates the diffusion of large tar molecules and improves Ni dispersion[155]. The NiAWBC catalyst achieved a 40% higher metal dispersion than conventional supports, with Ni particles controlled below 20 nm, thereby reducing sintering risk[156]. In addition, metal loading via methods such as ion exchange can significantly improve metal dispersibility. By optimizing ion exchange conditions (e.g., pH) and carbonization temperature, Wang et al.[157] successfully synthesized a Ni/lignite char catalyst with a Ni loading of 10.6 wt%. The size of metallic Ni particles is only 5–10 nm, and its performance at 650 °C is superior to that of commercial Ni/Al2O3 catalysts (Ni loading of 20 wt% ± 2 wt%). The research group further reduced the Ni particle size through lignite pretreatment (e.g., acid washing, oxidation, or their combination), enhancing catalytic activity and stability at 650 °C[87,158]. Tang et al.[159] developed a highly dispersed cobalt catalyst on lignite char via the ion exchange method for catalytic cracking of tar during biomass gasification. This catalyst achieved nearly 100% toluene conversion at 400 °C and maintained excellent stability in a 100-h continuous test.
Coal char supports also exhibit a unique directional conversion mechanism of coke. The synergistic effect between metals and coal char can regulate the carbon deposition pathway. For example, in the Ni-Ca/coal char catalyst, the addition of Ca2+ helps reduce the size of Ni crystal grains and guides the conversion of amorphous carbon deposits to CNTs, thereby significantly reducing the carbon deposition amount to 3.6 mmol g−1, which is much lower than that of conventional Ni/Al2O3 catalysts (> 15 mmol g−1)[104,160]. Yuan et al.[161] modified coal char supports with Fe, which effectively inhibited the coverage of random carbon deposits by enhancing the gasification reaction (C + H2O → CO + H2). The results showed that the carbon deposition rate of the Ni-Fe/coal char catalyst was 50% lower than that of the monometallic Ni catalyst.
Coal char supports also have significant advantages of low cost and ready availability of raw materials. After deashed fat coal is subjected to acid treatment to remove ash (e.g., SiO2, Al2O3), its specific surface area can be significantly increased to 500–800 m2 g−1, with a cost only 1/3–1/5 that of molecular sieve supports[155]. The Ni/deashed fat coal catalyst (NiAWBC) developed by Tong et al.[156] reduces raw material costs by more than 60% compared to commercial Al2O3 supports, and effectively utilizes the inherent aromatic structure of coal to enhance thermal stability. Zhang et al.[162] systematically compared the performance of chars derived from biomass, lignite, bituminous coal, anthracite, and graphite as tar reforming catalysts. The results showed that chars derived from biomass and low-rank coal exhibited significantly higher catalytic activity than those derived from high-rank fuels. It is worth noting that spent coal char catalysts can be recycled to the gasification system for subsequent utilization, thereby reducing environmental impacts[163].
However, coal char supports still face several challenges in practical applications. The issue of carbon deposition-induced deactivation is particularly prominent. Excessively strong surface acidity of coal supports or uneven metal dispersion can easily trigger deep cracking of biomass tar, generating refractory graphitic carbon. For example, He et al.[97] found in their study on biomass tar cracking using Ni/coal char catalysts that, after 60 min of reaction, graphitic carbon accounted for 45% of the total carbon deposition. As the reaction time progressed, the proportion of graphitic carbon further increased to over 60%. Studies by Klinghoffer et al.[164] showed that for coal char catalysts with uneven metal dispersion, the conversion rate dropped from the initial 85% to 40% after 10 h of tar cracking reaction, whereas the conversion rate of catalysts with well-dispersed metals remained above 70%. Metal sintering is another key issue. In high-temperature environments, metal particles tend to agglomerate, leading to catalyst deactivation. For instance, in monometallic Ni/coal char catalysts, Ni particles increased from 5 to 20 nm at 800 °C, resulting in a 30% decrease in catalytic activity[165]. Additionally, the insufficient mechanical strength of coal supports restricts their long-term operation. Their mechanical strength is generally lower than that of ceramic supports (e.g., MgO, Al2O3), making them prone to pulverization during long-term operation[166].
To mitigate the challenges of coal char supports, recent research has focused on advanced modification approaches. Composite reinforcement by incorporating ceramic binders (e.g., silica sol, alumina) or carbon nanomaterials can enhance mechanical integrity and attrition resistance, extending catalyst lifespan in fluidized beds[167]. Surface functionalization through oxidation, sulfonation, or nitrogen doping, modifies surface acidity/basicity and electronic properties, improving metal dispersion and coke resistance[73,168]. Pore structure engineering using dual-activation (e.g., KOH followed by CO2), or soft/hard templates, can construct interconnected macro-mesoporous networks, facilitating tar molecular diffusion and reducing pore blockage[169,170]. Metal site optimization via bimetallic systems (e.g., Ni-Fe, Ni-Co) or confined loading techniques (e.g., encapsulation) helps maintain small metal particle sizes under high temperatures, suppressing sintering and directing coke toward graphitic forms[171,172]. However, these strategies often face practical constraints, such as increased complexity and cost, and the fundamental understanding of how modified surface sites dynamically interact with tar intermediates during long-term operation remains incomplete. This knowledge gap limits the systematic optimization of coal char catalysts for industrial-scale applications.
Overall, conventional carbon-based catalysts exhibit remarkable advantages in low cost, high efficiency at low temperatures, and sustainability in the catalytic reforming of biomass tar[27]. However, their practical application still faces numerous challenges, including insufficient mechanical strength, poor surface chemical stability, difficulties in regulating carbon deposition, and limited ability to crack heavy tar components[173,174]. Furthermore, since conventional carbon-based catalysts are mostly derived from carbonization products, the uncontrollable carbonization process can result in difficulty in achieving directional control over their micro-morphology, pore structure, and surface chemical sites[175]. As a result, it is difficult to establish a stable 'structure-activity' relationship, which limits the in-depth analysis of reaction mechanisms and the directed optimization of catalytic performance. Therefore, there is an urgent need to develop advanced carbon-based materials with finely tunable structures. Through the precise design of their microstructure, surface chemistry, and metal-support interactions, the intrinsic mechanism of catalytic reactions can be revealed, and theoretical guidance and practical pathways can be provided for the modification of conventional carbon materials[62,176]. This will thereby promote the efficient, stable, and sustainable application of carbon-based catalysts in the clean conversion of biomass tar.
-
In recent years, the development of advanced carbon materials with nanoscale controllability has advanced rapidly. These materials, which feature regular morphologies and precisely constructed active sites, can be fabricated via methods such as the soft template method and hard template method[177,178]. They exhibit significant potential in structural regulation, active site distribution, and reaction pathway optimization, providing a novel catalyst platform for the efficient conversion of biomass tar. Table 4 summarizes how representative structural features of advanced carbon materials highlight the intrinsic limitations of conventional carbons, and provide transferable guidelines for their targeted modification. Based on their material properties, this paper systematically reviews advanced carbon-based catalysts by classifying them into the following categories.
Table 4. Mapping key features of advanced carbon materials to deficiencies and modification strategies of conventional carbon catalysts[73,179,180]
Features of advanced carbon materials Deficiencies of conventional carbon Mechanistic insights revealed Transferable modification strategies
for conventional carbonOrdered hierarchical pores Random pore networks with high tortuosity and diffusion limitations Directional mass transfer suppresses pore blockage and random coke accumulation Template-assisted activation; pore-channel ordering via soft/hard templates Spatial confinement of metal nanoparticles Metal sintering and uneven dispersion Confined spaces stabilize metal size and inhibit migration Encapsulation strategies; carbon shell or mineral-carbon composites Single-atom or uniformly coordinated metal sites Broad distribution of metal active environments Uniform coordination lowers C–C activation barriers and suppresses coke precursors Heteroatom (N, S) doping to construct stable metal-Nx sites Highly graphitized frameworks Low conductivity and poor control of coke evolution Enhanced electron transfer directs carbon toward graphitic structures Partial graphitization treatments; conductive carbon additives Tunable surface chemistry and hydrophobicity Steam-induced functional group loss Controlled wettability stabilizes active sites under reforming conditions Surface modification to balance hydrophilicity/hydrophobicity MOFs derivatives
-
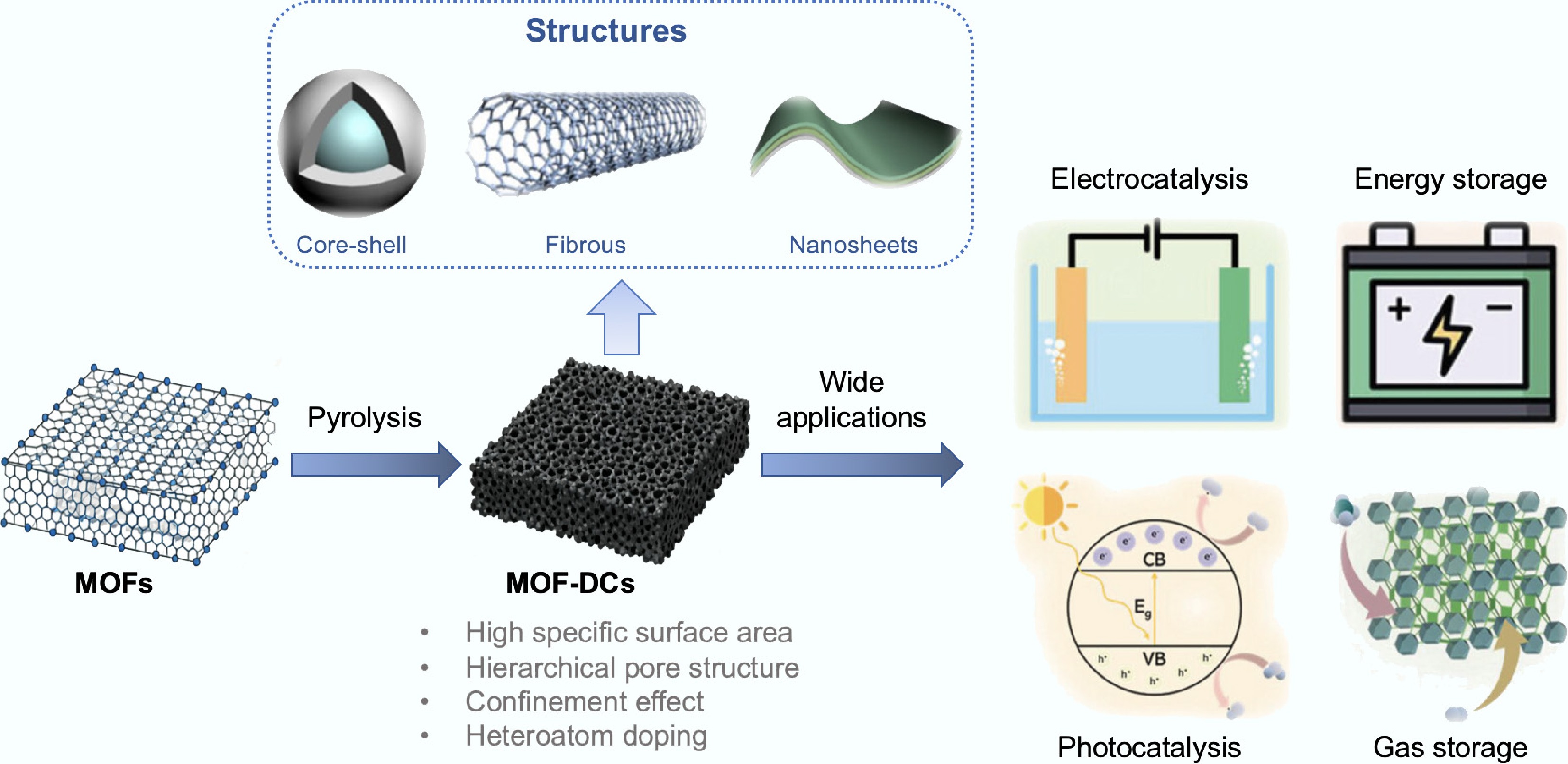
Metal-Organic Frameworks (MOFs) are formed by the connection of metal ions and organic ligands through strong coordination bonds, featuring advantages such as porosity, high specific surface area, and chemical tunability[181]. As shown in Fig. 4, MOFs-derived carbon materials (MOFs-DCs) prepared using MOFs as sacrificial precursors exhibit excellent performance in many fields.
The hierarchical pore structure of MOFs-derived carbons significantly enhances mass transfer efficiency and coke resistance. Zhang et al.[182] fabricated a NiCe-MOFs-derived hierarchical structure catalyst (NiCe-MDC/WC) by in situ growth of layered NiCe-MOF precursors on wood-derived carbon (WC) microchannels, followed by carbonization at 600 °C. In the steam reforming reaction of toluene, the catalyst achieved approximately 99% toluene conversion at 550 °C and maintained high activity for at least 48 h of continuous reaction, demonstrating exceptional durability and stability. The three-dimensional ordered macropore-mesopore hierarchical structure (macropores: 150–300 nm; mesopores: 2–50 nm) constructed via the template method can synergistically accelerate the diffusion of large tar molecules and the exposure of active sites. As shown in Fig. 5, Yao et al.[183] developed the Co-SA/3DOM-NC (three-dimensional ordered macroporous nitrogen-doped carbon-anchored Co single atoms) catalyst. Thanks to its hierarchical pores and high specific surface area (> 1,200 m2 g−1), it achieved a 99% conversion rate in furfural conversion with zero deactivation after six cycles. In contrast, conventional microporous ZIF-derived carbon only reached an 18% conversion rate in the same reaction due to limited mass transfer. Furthermore, the regular pores of MOFs-DCs can act as 'nanoreactors' to guide the directional conversion of amorphous carbon deposits into ordered CNTs through spatial confinement. In the Co@C core-shell structure derived from Co-based MOFs, a carbon layer thickness of 0.334 nm effectively restricts the migration of Co nanoparticles, promoting the directional growth of carbon deposits in the form of graphitized CNTs. This reduces the coking rate to below 10%, significantly alleviating the problem of active site coverage[184].
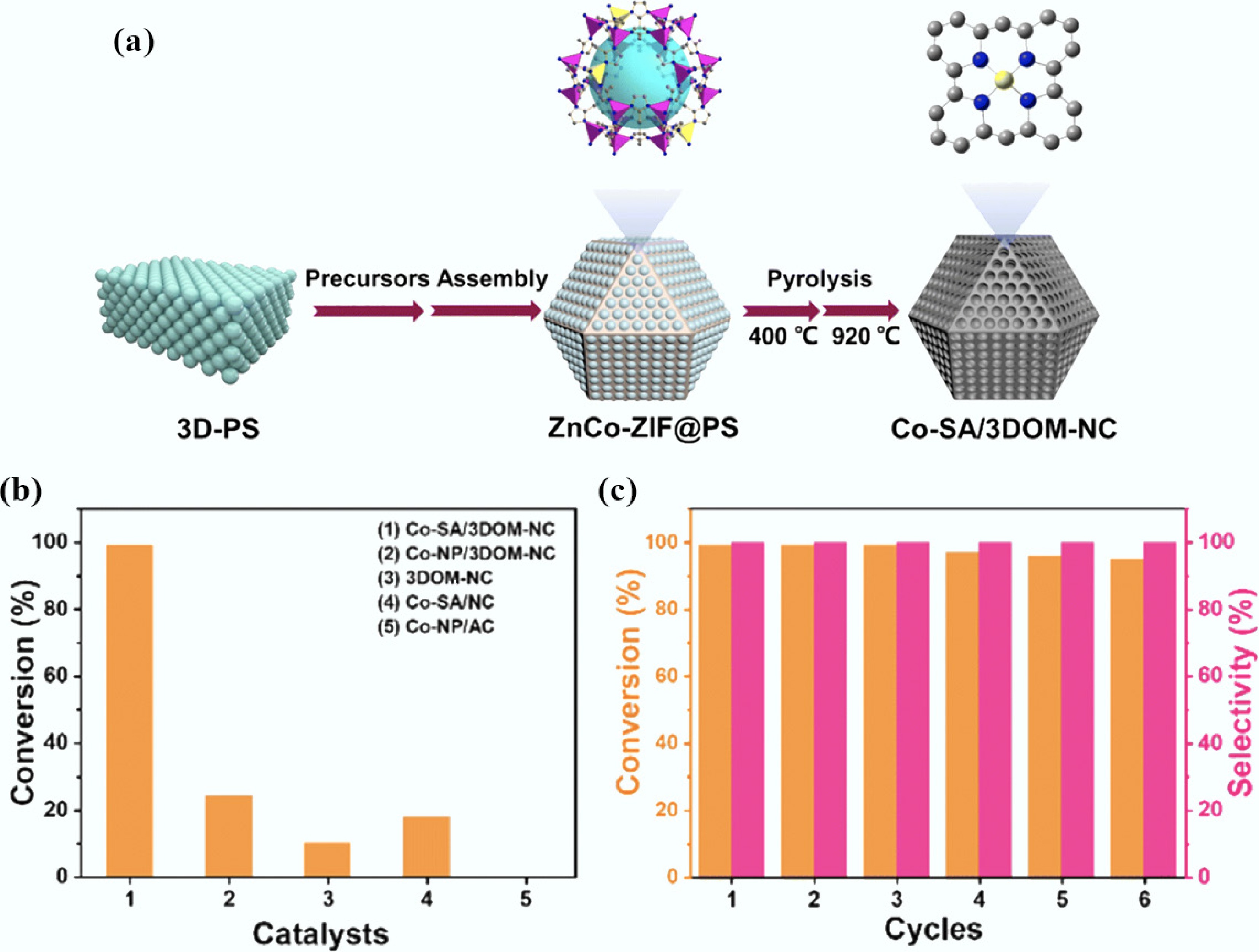
Figure 5.
(a) Schematic illustration of the fabrication of Co-SA/3DOM-NC; (b) The comparison of the FFA conversion between Co-SA/3DOM-NC and other catalysts; (c) Reusability tests of Co-SA/3DOM-NC for the oxidative esterification of FFA[183].
Single-atom site design further enhances the catalytic activity and coke resistance of MOFs-DCs. The periodic coordination environment of MOF precursors enables atomic-level dispersion of metals. For example, CoN4 sites derived from ZnCo-ZIF reduce the cleavage energy barrier of C–C bonds by regulating the d-orbital electronic structure of Co, accelerating aromatic ring opening in tar reforming while reducing the formation of coke precursors[185]. Hydrophobic modification effectively addresses the issue of competitive adsorption with water vapor, thereby enhancing the catalyst's stability in humid environments. Ji et al.[186] synthesized AC@ZIF-8 by growing hydrophobic ZIF-8 on the surface of activated carbon via a PDA binder. Its CO2 adsorption retention rate remained above 90% in high-humidity flue gas, whereas unmodified activated carbon experienced a decay of up to 40%.
The external field response properties of MOF composites have opened up a new avenue for in situ carbon deposition elimination. Fe3O4@MOF composites possess both high saturation magnetization (> 40 emu g−1) and MOF shape selectivity[187]. An external magnetic field enables second-level recovery, and their wave absorption properties induce local hotspots (> 1,000 °C) in a microwave field, promoting the graphitization of amorphous carbon and increasing the carbon deposition conversion rate by 30%. Zhao et al.[188] introduced -OH defects into MOF-Cu through an amino-regulated hydrolysis strategy, forming a hierarchical pore structure. Under full-spectrum irradiation, the 2D defective hierarchical porous MOF-Cu (2D HP MOF-Cu) exhibits broadened light absorption range due to defect energy levels, and new energy levels in the valence band reduce the Gibbs free energy barrier of COOH* → CO*, realizing photocatalytic gasification of carbon deposition. The CO generation rate is 4.4 times that of 3D MOFs.
Compared with conventional catalysts, MOFs-derived catalysts demonstrate remarkable low-temperature efficiency. Chu et al.[189] reported that Ni/MOFs-derived carbon achieved 98% tar conversion at 600 °C, reducing energy consumption by approximately 20% compared to conventional biochar supports (requiring 700 °C). MOFs-derived carbon catalysts exhibit remarkable advantages in the catalytic reforming of biomass tar, with their most prominent features being the directionally designed hierarchical pore structures and precisely constructed active sites[115,182]. Through methods such as template synthesis and ligand regulation, MOFs-derived carbon materials can achieve precise control of pore sizes ranging from micropores to macropores, which significantly facilitates the diffusion of reactant molecules and the exposure of active sites[190]. Meanwhile, their periodic structures provide an ideal platform for the uniform anchoring of metal single atoms or nanoclusters, remarkably enhancing catalytic activity and coking resistance[191−193].
Importantly, the significance of MOFs-derived carbon materials extends beyond their intrinsic catalytic performance, as they provide guidance for overcoming the intrinsic limitations of conventional carbon catalysts. The precisely engineered hierarchical pore networks in MOFs-derived carbons clearly demonstrate that ordered and low-tortuosity mass-transfer pathways, rather than merely high surface area, are essential for suppressing pore blockage and random coke accumulation during tar reforming. This insight directly addresses the diffusion limitations and pore heterogeneity commonly observed in biochar- and coal-char-based catalysts. In addition, the stabilization of single-atom or uniformly coordinated metal sites within well-defined coordination environments highlights the critical role of strong and uniform metal-support interactions in lowering C–C bond activation barriers while suppressing coke precursor formation. These findings suggest that introducing heteroatom-assisted anchoring sites or spatial confinement motifs into conventional carbon matrices represents an effective strategy to improve metal dispersion and long-term stability. Furthermore, the success of core-shell and confined architectures in MOFs-derived systems provides a transferable pathway to mitigate metal sintering and leaching in traditional carbon catalysts through surface coating, partial encapsulation, or composite design, thereby translating fundamental structure-activity relationships into practical modification strategies for low-cost carbon materials.
However, MOFs-derived carbon materials still face challenges in practical applications, including complex synthesis, high costs, and difficulties in scaling up[194,195]. Their synthesis typically relies on solvothermal/hydrothermal methods, which require prolonged reactions under elevated temperatures and pressures, along with significant consumption of organic ligands (e.g., terephthalic acid, 2-methylimidazole) and organic solvents[196]. This leads to substantially higher precursor costs compared to conventional carbon materials. Taking typical ZIF-8 as an example, the cost of its organic ligands account for the majority of the total material cost[197]. Moreover, subsequent high-temperature carbonization (> 800 °C) and acid washing further increase energy consumption and process complexity[198]. During scale-up, issues such as uneven mass and heat transfer, difficulties in controlling crystal growth, and batch-to-batch variations become prominent. Currently, most MOFs-derived carbon materials are still limited to gram-scale laboratory preparation. To advance their industrialization, future efforts should focus on the following cost-reduction and scaling-up pathways: adopting low-cost biomass-derived organic acids (e.g., citric acid, gallic acid) or industrial by-products as ligand alternatives[199]; developing scalable processes such as continuous-flow synthesis and spray drying to improve preparation efficiency and consistency[200,201]; and designing MOF/biochar composite systems or core-shell structures, where a small amount of MOFs-derived carbon serves as a surface modification layer, thereby maintaining performance advantages while significantly reducing overall costs[202]. Through synergistic innovation in materials and processes, MOFs-derived carbon catalysts are expected to achieve widespread application in specific high-value-added scenarios and provide theoretical guidance for the precise modification of conventional carbon materials.
Carbon nanotube-based catalysts
-
Carbon nanotubes (CNTs) have emerged as ideal catalyst supports for biomass tar conversion due to their unique structural characteristics. Their one-dimensional hollow tubular structure (diameter: 5–25 nm) and ultra-high specific surface area (typically > 250 m2 g−1) provide an excellent dispersion platform for metal nanoparticles[203−205]. This regular and ordered pore structure significantly accelerates the diffusion of large tar molecules, and effectively reduces the risk of blockage by amorphous carbon deposits caused by mass transfer limitations[206]. Meanwhile, their spatial confinement effect can inhibit the migration and sintering of metal particles at high temperatures[207]. Liu et al.[208] embedded Ni nanoparticles into the defect sites of nitrogen-doped carbon nanotubes (N-CNTs) via an in situ confinement growth method, controlling the Ni particle size to 4.2 ± 0.8 nm with uniform distribution on the CNT walls. The dispersion degree was 40% higher than that of coal char supports, thereby significantly increasing the active site density and catalytic stability.
Another key advantage of CNTs lies in their high electrical conductivity. Their powder resistivity is as low as 15–22 mΩ·cm, far lower than that of biochar (> 100 mΩ·cm), which greatly enhances the electron transfer efficiency during reactions[209]. Chai et al.[195] loaded Ni-Co bimetals on MOFs-derived CNTs and found that the electron conduction capability of CNTs accelerated electron transfer in tar cracking, reducing the activation energy for C–C bond cleavage by 15%. In addition, Su et al.[210] synthesized the CoPc/SWCNT catalyst by supporting cobalt phthalocyanine (CoPc) on single-walled carbon nanotubes (SWCNTs). As shown in Fig. 6a, in electrocatalytic CO2 reduction, the CoPc/SWCNT catalyst improved electron mobility, achieving a methanol partial current density of > 90 mA cm−2 with a selectivity of > 60% in a series flow electrolyzer.
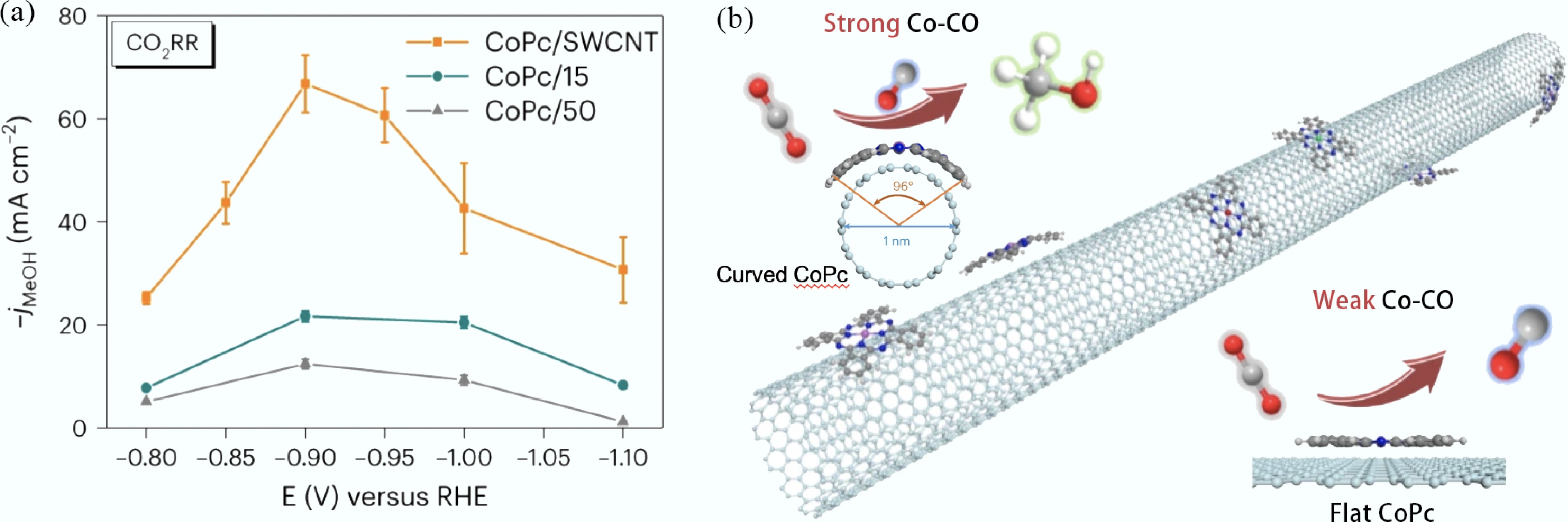
Figure 6.
(a) jMeOH of CoPc/SWCNT, CoPc/15 and CoPc/50 catalysts in a flow cell under CO2 atmosphere; (b) Schematic diagram of the synthetic process of CoPc/SWCNT catalyst and the structure of CoPc on different-diameter CNTs[210].
In addressing the core challenge of carbon deposition-induced deactivation, CNTs exhibit a unique 'active guidance' mechanism. Their curved tube wall surfaces can act as 'nanoscale templates', inducing the ordered growth of carbon deposits into graphitized CNTs along specific directions, rather than allowing them to randomly cover active sites[211]. As shown in Fig. 6b, Su et al.[210] observed, when supporting cobalt phthalocyanine (CoPc) on single-walled carbon nanotubes (SWCNTs, with a diameter of 1.0 ± 0.2 nm), that the phthalocyanine ring undergoes a 96° bending when CoPc is adsorbed on the SWCNT surface. This enhances the binding force with CO intermediates and promotes the conversion of carbon deposits into methanol. Furthermore, heteroatom doping of CNTs can further optimize the electronic structure of metals and regulate the carbon deposition conversion pathway. For example, Williamson et al.[212] found that in N-doped Fe@CNT catalysts, pyridinic nitrogen inhibits carbon deposition by strengthening the metal-support interaction (MSI) and simultaneously promotes the formation of Fe0, reducing the carbon deposition amount to 3.6 mmol g−1 (compared to > 15 mmol g−1 for undoped supports).
In terms of long-term stability, CNTs exhibit significant advantages. The graphitized carbon layers (sp2 hybridization) of CNTs form a dense anti-oxidation barrier due to the planar conjugated structure of carbon atoms (with an interlayer spacing of 0.334 nm), endowing them with stronger anti-oxidation capacity than the amorphous carbon of biochar[211]. Huda et al.[213] formed a uniform Nafion coating (with a thickness of approximately 2 nm) on the surface of Pt/SWCNTs via an in situ spraying method. In a 500-h potentiostatic test (0.9 V), the current density retention rate of Nafion-coated Pt/SWCNTs reached 91.3%, which was significantly higher than the 68.5% of conventional Pt/C. CNTs also possess excellent hydrophobicity (with a contact angle > 120°), which can effectively repel water vapor in the reaction environment and reduce the competitive adsorption of water on active sites[214]. For example, Lu et al.[215] found that the dendritic Fe-N/CNTs cathode, relying on high hydrophobicity, significantly alleviated water flooding in proton exchange membrane fuel cells (PEMFCs) and could operate stably at a voltage of 0.4 V for 100 h, with a voltage decay of only 3.2%. This characteristic is also applicable to biomass tar reforming reactions under high-humidity environments.
Beyond their excellent catalytic performance, CNTs-based catalysts offer critical insights into how well-defined carbon architectures can be leveraged to address the structural deficiencies of conventional carbon materials. The one-dimensional, hollow, and low-tortuosity channels of CNTs demonstrate that directional mass transfer is highly effective in minimizing diffusion resistance and preventing the random accumulation of amorphous coke, a persistent challenge for disordered char-based supports. This suggests that constructing linear or quasi-aligned mesoporous channels in biochar or coal char—via templating, activation, or controlled carbonization—can substantially enhance tar accessibility to active sites. Moreover, the high electrical conductivity of CNTs highlights the importance of electronic transport in facilitating C–C bond cleavage and coke graphitization, indicating that partial graphitization treatments or conductive carbon additives may significantly improve the catalytic efficiency of traditional carbons. In addition, heteroatom doping strategies commonly employed in CNTs to strengthen metal-support interactions and suppress metal sintering provide a directly transferable approach for stabilizing metal nanoparticles on conventional carbon matrices, thereby improving durability under harsh reforming conditions. It is noteworthy that during the catalytic reforming of biomass tar, under the templating guidance of metal nanoparticles and the structural regulation of the support, active carbon species (e.g., C*, CHx fragments) generated from the cracking of tar molecules on metal active sites can be induced to grow into carbon nanotubes[104]. These in situ generated CNTs during the reaction not only do not cover the active sites but may also form new catalytic interfaces, further enhancing the activity and long-term stability of the catalyst[216].
Although carbon nanotube-based catalysts exhibit significant advantages in structure and performance, their industrialization is still hindered by the high cost of synthesis methods and challenges in large-scale production. Current mainstream preparation methods, such as chemical vapor deposition (CVD), require high temperatures (700–1,200 °C) and the use of transition metal catalysts, along with high-purity carbon sources (e.g., ethylene, propylene)[217]. These processes involve complex equipment and high energy consumption. Moreover, post-treatment steps to remove the catalyst support further increase costs[218]. Multiple pricing guides and reports state that high-purity CNTs can cost tens to hundreds of dollars per gram at lab scale—many times more than bulk activated carbon[219]. During scale-up, it is difficult to control the uniformity of the temperature field and gas flow distribution in CVD reactors, leading to inconsistencies in carbon nanotube diameter, length, and defect density, which affects the stability of catalytic performance[220,221]. In addition, metal nanoparticles are prone to migration and sintering during high-temperature reactions, thereby reducing catalyst lifetime[222]. To overcome these bottlenecks, future research may focus on: developing biomass pyrolysis gases or low-cost hydrocarbons as carbon sources, and combining them with floating-catalyst CVD to achieve continuous production[223]; utilizing template or self-assembly techniques to grow carbon nanotube arrays in situ on the surface of conventional carbon supports, constructing biomimetic hierarchical structures that balance performance and cost[224]; and designing spatially confined core-shell structures that encapsulate metal nanoparticles within carbon layers to enhance their high-temperature stability[225]. Through the integration of material design and process optimization, carbon nanotube-based catalysts are expected to enable widespread application in highly efficient, long-lifetime reaction systems and to promote the directional regulation of traditional carbon support structures.
Graphene-based catalysts
-
Graphene, a two-dimensional hexagonal carbon lattice, has shown great promise as a catalyst support for biomass tar conversion, owing to its ultra-high specific surface area (~2,630 m2 g−1), excellent electrical and thermal conductivity, and high mechanical strength[226−228]. Figure 7 demonstrates the prominent structural advantages of graphene-based catalysts and their application in the catalytic reforming of biomass tar. Its two-dimensional planar structure can be engineered to form ordered mesoporous channels through chemical etching or template methods[229−231]. For instance, Chen et al.[232] found that when single-layer graphene grows on a sapphire substrate, its carrier mobility reaches 14,700 cm2 V−1 s−1 and sheet resistance is as low as 587 Ω−1, which can significantly accelerate the transfer of reaction electrons. Moreover, the atomically flat surface of graphene (roughness < 0.5 nm) and its uniformly oriented domain structure (grain boundary density < 0.1 μm−1) can prevent the clogging of disordered pores by large tar molecules, thereby reducing the formation of amorphous carbon deposits. In addition, the structural advantages of graphene not only provide a uniformly dispersed platform for metal nanoparticles, but also, due to its atomic-level thickness and interlayer 'van der Waals' forces, promote the rapid adsorption and diffusion of reactant molecules, greatly improving mass transfer efficiency[233,234].
Heteroatom doping is a key approach to optimizing the performance of graphene-based catalysts. The introduction of graphitic nitrogen (e.g., pyridinic nitrogen) can form metal-like active sites. As shown in Fig. 8a, the nitrogen-doped graphene developed by Lv et al.[235] achieved 99% DFF selectivity in the oxidation of 5-hydroxymethylfurfural (HMF) to 2,5-diformylfuran (DFF) by regulating the electronic structure through nitrogen atoms, with activity superior to that of graphene oxide. Yang et al.[236] found that in phosphorus-doped graphene aerogels (PGAs), phosphorus doping increases the electron density at Zigzag sites on graphene edges. This promotes the adsorption of *CO intermediates in CO2 reduction, enabling carbon deposits to be preferentially converted into graphitized carbon rather than amorphous carbon. The coking rate is only 0.015 mg cm−2 h−1, which is 70% lower than that of undoped graphene. These doping strategies have optimized the carbon deposition conversion pathways by precisely regulating the electronic states of active sites.
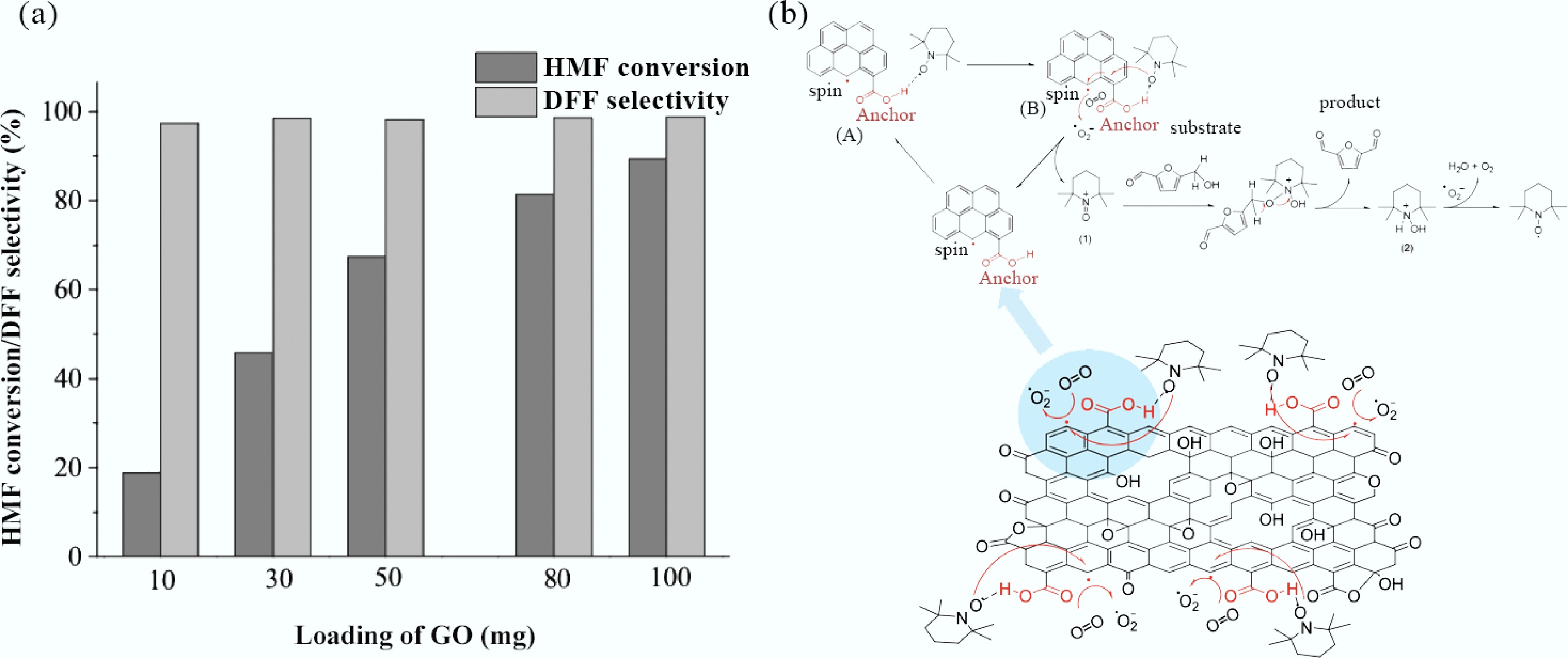
Figure 8.
(a) Effect of the GO loading on aerobic oxidation of HMF into DFF; (b) Proposed reaction pathway for selective oxidation of HMF into DFF with molecular oxygen as the terminal oxidant in GO/TEMPO catalytic system[235].
The core breakthrough of graphene-based catalysts lies in their ability to 'actively restructure' carbon deposition conversion pathways. Carboxyl groups (-COOH) and unpaired electrons at the edges of graphene oxide (GO) can synergistically activate tar molecules while promoting the conversion of carbon deposits into ordered carbon structures. As shown in Fig. 8b, when Lv et al.[235] conducted the co-catalyzed oxidation of HMF using GO and TEMPO, they found that the carboxyl groups (-COOH) at GO edges form a dynamic complex with the nitroxyl radicals of TEMPO through a hydrogen-bond network. This significantly reduces the activation energy of HMF oxidation and guides the rearrangement of amorphous carbon deposits into graphite microcrystals, resulting in post-reaction carbon deposits, mainly composed of 3–5 layers of graphene sheets. Teng et al.[237] utilized flash joule heating (FJH) technology, which employs instantaneous ultra-high temperatures (approximately 3,000 K, lasting about 10 s), to directly convert biomass pyrolytic carbon into 2–5 layer graphene (FG). This simultaneously achieves directional graphitization of carbon deposits and volatilization of heavy metals in biomass pyrolytic carbon. In addition, core-shell structure design can further inhibit catalyst deactivation caused by carbon deposition. For instance, Nandakumar et al.[238] designed graphene-encapsulated transition metal@N/C catalysts, the core-shell structure exhibits a confinement effect that can stabilize metal nanoparticles and inhibit carbon deposition, thereby enhancing catalytic stability during tar conversion.
Graphene-based catalysts can also be combined with other functional materials to enhance their comprehensive properties. For example, Li et al.[239] loaded Ni onto biochar-derived graphene via molten salt electrolysis, forming a composite structure of graphitic nanotubes. As anode materials for lithium-ion batteries, this composite exhibited high energy storage performance (specific capacity > 350 mAh g−1), validating its dual potential in energy conversion and storage.
From a broader perspective, the value of graphene-based catalysts lies in their ability to reveal how two-dimensional, electronically conductive carbon interfaces regulate reaction pathways and carbon evolution during tar reforming. The atomically flat surfaces and tunable electronic structures of graphene clearly demonstrate that uniform exposure of active sites and efficient electron transport are key factors in suppressing amorphous carbon formation and promoting directional graphitization. These insights directly address the heterogeneous surface chemistry and poor conductivity of conventional carbon catalysts. In particular, the effectiveness of heteroatom-doped graphene in modulating electron density and metal anchoring strength suggests that controlled heteroatom incorporation into biochar or coal char can be an effective route to tailor surface reactivity and enhance coke resistance. Furthermore, graphene-based composite and coating strategies illustrate a practical pathway for compensating for the mechanical weakness and low conductivity of traditional carbons, by integrating thin graphitic layers or graphene-like domains, without fundamentally altering the low-cost nature of conventional carbon supports.
Although graphene-based catalysts demonstrate excellent performance potential, their industrialization still faces challenges such as high synthesis costs, complex processes, and insufficient structural stability. The preparation of high-quality graphene primarily relies on CVD, which requires single-crystal metal substrates (e.g., copper foil) and ultra-high-vacuum environments, along with high-purity precursors (e.g., methane)[240]. Moreover, the subsequent transfer process tends to introduce defects and contamination, resulting in significantly higher costs compared to traditional carbon materials[241]. Although GO prepared by the redox method allows for batch synthesis, the use of strong oxidizing agents, extensive water-washing steps, and high energy consumption during high-temperature reduction still make its environmental footprint and economic viability less competitive[242,243]. During scale-up, graphene layers are prone to π–π stacking, which reduces the specific surface area and accessibility of active sites[244]. Additionally, metal particles tend to migrate and aggregate on the two-dimensional surface, accelerating sintering, especially at high temperatures[245]. To enhance the feasibility of large-scale applications, future research can explore the following directions: developing biomass flash joule heating technology to directly convert agricultural waste into graphene with controllable defects, significantly reducing raw material and energy costs[233]; creating composite systems of graphene with traditional carbon materials, such as graphene-coated biochar or blends with activated carbon, to improve electrical conductivity and mechanical strength while controlling costs[246]; and utilizing continuous roll-to-roll CVD or liquid-phase self-assembly processes to produce large-area, uniform graphene-based catalytic membranes[247]. With the assistance of artificial intelligence in material design and process optimization, graphene-based catalysts are expected to gradually achieve engineering applications in high-efficiency, long-life catalytic systems.
Other carbon-based catalysts
-
Although the catalysts discussed in this section are also carbon-based, they are not categorized as advanced carbon-based catalysts in this review. The term 'advanced carbon-based catalysts' is herein reserved for model systems with precisely tunable and well-defined structures—such as MOFs-derived carbons, carbon nanotubes, and graphene—that enable systematic investigation of structure-activity-stability relationships. In contrast, the carbon-based catalysts summarized in this section typically possess partially ordered or application-driven architectures, where structural parameters, active site environments, and metal-support interactions are less independently controllable. These materials are therefore discussed separately, not because they lack catalytic relevance, but because their primary value lies in demonstrating practical performance enhancements or engineering-oriented designs, rather than in serving as idealized platforms for mechanistic deconvolution. Their structures often arise from natural templates, macroscopic architectures, or process-integrated synthesis routes, which, while beneficial for mass transfer and stability, do not allow the same level of atomic- or nanoscale precision as advanced carbon materials.
Wood-derived three-dimensional (3D) carbon monoliths represent a class of application-oriented carbon catalysts with aligned macrochannels, offering enhanced mass transfer but limited independent control over nanoscale active site environments[248]. Wang et al.[249] developed a 3D carbon monolithic reactor derived from natural wood, in which the wood-derived carbon support is prepared via nickel nitrate impregnation and high-temperature pyrolysis at 900 °C features low-tortuosity open channels (5–60 μm in diameter) and uniformly loaded Ni nanoparticles (20–60 nm). This unique architecture integrates aligned channels with secondary micropores to enable efficient mass transfer by promoting gas mixing and reactant contact, while the Ni nanoparticles encapsulated by graphitic carbon layers endow the catalyst with remarkable resistance to carbon deposition and metal agglomeration. Experimental results demonstrate that in toluene steam reforming, this catalyst exhibits significantly higher conversion efficiency and operational stability than conventional activated carbon-based counterparts. This structural design not only demonstrates the possibility of converting natural porous structural materials into high-efficiency catalyst supports, but also provides insights for the construction of bionic channels and metal spatial confinement in the design of conventional carbon-based catalysts.
Mesoporous carbon (MC) features a highly ordered mesoporous structure (pore size 2–50 nm) with a specific surface area up to 2,500 m2 g−1, which can effectively promote the diffusion and adsorption of large tar molecules[250,251]. Ordered mesoporous carbons provide improved pore regularity compared with conventional chars; however, their metal coordination environments and electronic properties remain less precisely defined than those of MOFs-derived carbons. Cabrera-Reyes et al.[252] investigated the performance of Ni-supported mesoporous carbon (MC) catalysts in the steam reforming of biomass tar model compounds (e.g., phenol) and regulated the surface chemical properties of mesoporous carbon via phosphorus doping, significantly inhibiting carbon deposition. The results showed that phosphorus-modified mesoporous carbon (P-MC) enhanced the dispersion of Ni nanoparticles by strengthening the strong metal-support interaction (SMSI). Moreover, it guided the directional conversion of carbon deposition precursors to CO2 and H2 through surface acidic sites, ultimately achieving a tar conversion rate approaching 100% and an H2 yield increase of more than 30%. This ordered mesoporous structure and surface chemical modification strategy has provided a viable pathway for addressing the issues of pore disorder and surface inertness in conventional carbon materials, particularly in terms of the efficient diffusion and cracking of heavy tar molecules.
Carbon nanofibers (CNFs) have a unique fibrous structure and good mechanical properties. The high specific surface area (> 300 m2 g−1) and fibrous porous structure (diameter < 100 nm) of CNFs provide uniformly dispersed active sites for metal nanoparticles. Their one-dimensional fiber network significantly promotes the diffusion and adsorption of reactants (such as large tar molecules) while reducing mass transfer resistance[253]. CNFs-supported catalysts can show good catalytic performance in biomass tar conversion. The Ni/CNFs catalyst prepared by Tang et al.[253] via electrospinning combined with high-temperature carbonization exhibited a 92% conversion rate in toluene steam reforming at 700 °C, representing a 20% improvement over conventional activated carbon supports. Studies have found that the inter-fiber gaps (50–200 nm) of CNFs can effectively capture polycyclic aromatic hydrocarbons in tar, thereby reducing carbon deposition. The open pores formed by fiber interweaving provide a new direction for the design of conventional carbon supports, and particularly hold reference value in terms of enhancing the mechanical stability and anti-clogging capability of catalysts. Although CNFs share certain structural similarities with CNTs, they typically exhibit broader diameter distributions and less uniform degree of graphitization, which constrains their use as model catalyst platforms.
Overall, advanced carbon-based catalysts demonstrate significant potential in the catalytic reforming of biomass tar, owing to their tunable structures, excellent electronic properties, and strong coke resistance. The hierarchical pore structures and single-atom site designs of MOFs-derived materials provide an ideal platform for reactant diffusion and intermediate stabilization[254,255]. The high electrical conductivity and spatial confinement effects of carbon nanotubes effectively facilitate electron transfer and inhibit metal sintering[256]. Meanwhile, the two-dimensional planar structure and tunable surface chemistry of graphene creates favorable conditions for the uniform distribution of active sites and the directional conversion of coke deposits[257]. These precise structural modulation strategies not only offer new insights for the rational design of tar conversion catalysts, but also pave the way for the development of carbon-based catalysts in other heterogeneous catalytic processes, such as electrocatalysis, environmental catalysis, and energy conversion. To provide strategic guidance for catalyst selection under different application constraints, a comparative trade-off analysis of representative carbon-based catalysts is summarized in Table 5, focusing on key tensions such as cost vs performance, and scalability vs controllability. In the future, further leveraging the structural advantages of advanced carbon materials, combined with artificial intelligence-assisted design and green synthesis pathways, will promote the efficient, stable, and sustainable application of carbon-based catalysts in a broader range of catalytic fields.
Table 5. Trade-off analysis of different types of carbon-based catalysts for biomass tar reforming[62,258−262]
Catalyst type Key features Cost Performance (activity/stability) Scalability Controllability (structure/sites) Recommended scenarios Biochar-based Abundant, low-temperature active, porous, surface-functionalized Low Moderate activity; limited stability in steam High (abundant biomass feedstocks) Low (heterogeneous structure) Distributed, low-temperature tar reforming; pilot-scale systems Coal char-based Tunable porosity, good metal dispersion, coke-convertible Low to moderate Good low-temperature activity; prone to sintering and coke High (coal resources available) Moderate (pore structure tunable) Industrial gasification with integrated char recycling MOFs-derived carbons Ordered hierarchical pores, single-atom sites, high SSA Fairly high Excellent activity and coke resistance; stability under study Low (complex synthesis, ligand cost) High (precise pore and site design) High-value syngas production; fundamental mechanism studies CNTs-based High conductivity, spatial confinement, oriented coke growth High High activity and long-term stability; good coke management Low to moderate (CVD scale-up challenging) High (diameter, doping, metal confinement tunable) Advanced catalytic systems requiring high conductivity and coke tolerance Graphene-based Ultra-high SSA, excellent conductivity, tunable surface chemistry High Superior activity and coke restructuring; stacking issues Low (CVD/GO production costly) High (surface chemistry and doping controllable) High-efficiency, low- temperature reforming; hybrid catalysts with conventional carbons -
Conventional carbon-based catalysts demonstrate significant cost advantages and low-temperature activity in the catalytic reforming of biomass tar. However, their practical application still faces numerous challenges. Firstly, the inherent heterogeneity of the materials makes it difficult to precisely control metal dispersion, and conventional impregnation methods often lead to the agglomeration of metal nanoparticles, significantly reducing catalytic efficiency[263]. Secondly, during operation, insufficient mechanical strength results in high particle wear rates in fluidized beds, while the hydrolysis and loss of surface functional groups in steam environments further accelerate the decline in catalytic activity[264]. Coke deposition is particularly problematic, as amorphous coke preferentially nucleates at interfacial defects, forming insulating layers that occlude active sites within 10–20 h of operation[265,266]. Furthermore, the limited pore architecture of raw chars further restricts molecular diffusion of bulky tar compounds, creating kinetic bottlenecks that undermine overall process efficiency[164]. To systematically address these challenges, insights from advanced carbon materials provide critical guidance for the rational modification of conventional carbon-based catalysts.
Advanced carbon materials, with their precisely tunable pore structures, excellent electron conductivity, and unique spatial confinement effects, provide critical insights for understanding catalytic mechanisms and guiding the modification of conventional carbon materials. As shown in Fig. 9, methods such as physical activation, heteroatom doping, and metal addition can modify the porous structure and surface chemical properties of conventional carbon-based catalysts, thereby enhancing their catalytic activity, stability, and expanding their application scope. Moreover, inspired by the hierarchical pore design of MOF derivatives, interconnected macropore (150–300 nm) and mesopore (2–50 nm) networks can be constructed in biochar through template methods combined with dual activation using KOH and CO2[267,268]. This significantly optimizes mass transfer efficiency and directs the directional conversion of coke into graphitized CNTs, reducing the coke deposition rate by 30%[269]. Inspired by the high conductivity of carbon nanotubes, nitrogen or phosphorus doping of coal-derived coke can introduce electron-deficient sites, enhancing MSI and lowering the energy barrier for C–C bond cleavage by approximately 15%[270]. Additionally, the two-dimensional planar structure of graphene and heteroatom doping strategies can be applied to optimize the surface chemical properties of conventional carbon supports, improving metal dispersion and resistance to coke deposition. In the future, by simulating the structural characteristics of advanced carbon materials and combining green synthesis pathways with low-cost precursors (e.g., biomass waste), the development of composite carbon carriers with biomimetic channels and spatial confinement effects will be a crucial direction for enhancing the performance and stability of conventional carbon-based catalysts.

Figure 9.
Modification strategies for conventional carbon-based catalysts based on the structure-activity relationship of advanced carbon materials, and applications of the modified catalysts in various fields.
Although carbon-based catalysts exhibit unique coke management advantages in biomass tar reforming, their long-term operational stability and regeneration performance remain major challenges for industrial applications. Unlike conventional non-carbon-based supports, carbon-based catalysts undergo simultaneous structural evolution and chemical environment reconstruction during reaction-regeneration cycles, resulting in a more complex, multi-scale deactivation mechanism. In addition to unavoidable carbon deposition and coverage, the metal active phase may still undergo sintering and migration under prolonged high-temperature and steam conditions, leading to a reduction in the number of effective active sites[271,272]. Meanwhile, some poorly graphitized carbon supports are susceptible to gasification consumption or mechanical wear, causing pore structure collapse and metal loss[273]. Furthermore, repeated oxidation-reduction cycles during regeneration may induce the accumulation of carbon skeleton defects and the rearrangement of surface functional groups, thereby altering the metal-support interaction (MSI) and affecting subsequent reaction pathways[274]. In response to the above issues, recent regeneration strategies have gradually shifted from 'simple coke combustion' to 'structural collaborative repair'. These include using mild atmosphere regulation to achieve selective gasification of non-active carbon, leveraging the metal-carbon interface to induce the reconstruction of coke deposits into ordered carbon nanostructures, and adjusting the distribution of surface defects and functional groups during regeneration to restore the stability of active sites[275−277]. These studies demonstrate that rationally designed regeneration strategies not only help extend the service life of carbon-based catalysts but may also further enhance their inherent capability for targeted carbon regulation. This lays the groundwork for the subsequent design of intelligently optimized catalytic systems, based on fine structural modulation.
Evolution of reaction systems
-
Current research on carbon-based catalysts is predominantly focused on the laboratory scale. Their scale-up to industrial application requires systematic optimization of the reaction system. Firstly, regarding catalyst loss and activity maintenance, completely avoiding support loss is not essential. For conventional carbon materials, although partial gasification of the support during steam reforming leads to mass loss, it can also increase the effective syngas yield. The key lies in maintaining a stable microenvironment for the active sites through structural design (e.g., constructing core-shell configurations, ordered channels), rather than solely pursuing the enhancement of the overall mechanical strength of the support. For instance, introducing metal spatial confinement structures into biochar can protect the active metals from sintering and loss even as the support gradually depletes, achieving a balance between local protection of active sites and controlled overall transformation of the support[278].
Secondly, the introduction of advanced field-assisted technologies is an important approach to enhance reaction efficiency and catalyst anti-deactivation capability. Microwave technology, due to its unique volumetric heating and 'hotspot' effects shows significant advantages in the application of carbon-based catalysts. For example, embedding magnetic Fe3O4 nanocrystals into a carbon support can induce local ultra-high temperatures (> 1,000 °C) under a microwave field, enabling instantaneous gasification of carbon deposition precursors (via the Boudouard reaction) and reducing net energy consumption by approximately 30%[279,280]. Furthermore, external field coupling technologies such as plasma and photoelectrocatalysis also hold promise for activating reactants and modulating reaction pathways at low temperatures. Future work should explore their synergistic mechanisms with carbon-based catalysts and develop intelligent reaction systems based on multi-field coupling.
Regarding reactor design and catalytic bed optimization, reactors suitable for industrialization need to balance mass/heat transfer efficiency with catalyst adaptability. Fluidized-bed reactors offer good heat transfer but demand high catalyst strength, under which conditions carbon supports are prone to abrasion. Fixed-bed reactors have a simple structure but are prone to clogging and exhibit poor heat transfer. Future developments may involve structured reactors, such as 3D-printed wood-derived carbon monolith reactors. Their low-tortuosity open channels can promote gas mixing and reactant contact, while enabling spatial confinement of metal nanoparticles[281]. The particle size, shape, and packing methods of the catalyst also require systematic optimization. For example, adopting gradient pore designs or multi-functional zoned beds adapt to the requirements of different reaction stages, thereby enhancing overall process economics and operational flexibility[282].
Application of AI
-
The research and development of carbon-based catalysts still rely on traditional trial-and-error methods, resulting in slow progress and difficulties in assessing their industrial potential. The introduction of artificial intelligence (AI) technology is reshaping the paradigm of catalyst development. Machine learning (ML) models can analyze vast amounts of experimental data (e.g., biomass feedstock composition, pyrolysis history, and metal loading configurations) to predict optimal catalyst formulations, thereby reducing the number of synthesis iterations by half[283]. For instance, neural networks trained on over 15,000 datasets can recommend Ni-Fe-Co ternary compositions with deviations of less than 5% from the experimentally determined optimal values, demonstrating the power of ML in accelerating multi-component catalyst optimization[284].
Beyond composition prediction, AI is increasingly employed for de novo design and structural optimization of carbon supports. In recent years, researchers have integrated genetic algorithms (GA), Bayesian optimization, and machine learning models with pore-scale mass transfer simulations to systematically regulate the pore size distribution, porosity, and hierarchical pore structure of carbon carriers within high-dimensional design spaces[285−287]. This integration aims to achieve a synergistic enhancement of reactant diffusion efficiency and active site exposure. For instance, data-driven pore-scale modeling frameworks have been employed to establish quantitative relationships between pore structural parameters and mass transfer performance, thereby providing reverse guidance for the rational design of porous carbon carrier structures[287]. Furthermore, GA and Bayesian optimization have been applied to balance key variables—such as micropore volume fraction, mesopore size distribution, and specific surface area—under constrained conditions, enabling the rapid screening of structural solutions with optimal mass transfer-reaction performance[288]. Such AI-assisted design strategies highlight AI's capability to navigate complex design spaces beyond human intuition.
AI also plays a crucial role in predicting catalyst lifespan and optimizing regeneration strategies. In recent years, machine learning models have been utilized to analyze online or offline characterization data—such as reaction rates, conversion decay, carbon deposition levels, and operational parameters—to develop predictive models for the evolution of catalyst activity over time[289]. This enables quantitative assessment of deactivation behavior and remaining service life. For instance, soft sensor models based on random forests (RF), artificial neural networks (ANN), or Gaussian processes can infer the real-time activity state of catalysts from multi-source process data and predict deactivation inflection points in advance[290]. Furthermore, by integrating data-driven models with kinetic or probabilistic frameworks, they provide decision-making support for optimizing regeneration cycles and replacement strategies, shifting regeneration operations from experience-driven to prediction-driven approaches[290]. Such AI-assisted methods not only improve catalyst utilization efficiency but also offer new technological pathways for ensuring stable operation and reducing operational costs.
However, the effectiveness of AI in catalyst design hinges on the availability of high-quality, well-curated datasets. Current applications are often limited to narrow chemical spaces or model reactions, and extrapolation to real biomass tar mixtures remains challenging[291]. Moreover, the 'black-box' nature of some advanced ML models can obscure mechanistic insights, necessitating complementary use of explainable AI (XAI) techniques[292]. Future efforts should focus on building open, standardized catalyst databases, integrating multi-scale simulations with ML, and developing hybrid models that couple data-driven predictions with fundamental catalytic principles.
Techno-economic environmental impact analysis
-
In evaluating the prospects for large-scale application, the integration of life cycle assessment (LCA) and process modeling is crucial. LCA provides a systematic framework to quantify the environmental impacts of carbon-based catalysts across their entire life cycle, encompassing raw material acquisition, synthesis, operational use, regeneration, and final disposal or recycling. This holistic approach is essential for verifying the sustainability claims of carbon-based catalysts and guiding their eco-design.
Carbon-based catalysts, particularly those derived from biomass waste, demonstrate significant potential environmental advantages. Firstly, the use of waste biomass as a feedstock can contribute to carbon negativity by sequestering biogenic carbon in a stable form, potentially offsetting emissions from other process stages. Secondly, their synthesis often requires lower temperatures and less energy-intensive processes compared to conventional catalysts like zeolites or certain metal oxides, leading to a reduced embedded energy footprint[293]. Most notably, their unique 'coke management' capability, which transforms deactivating coke into structured graphitic carbons, represents a form of in situ resource recovery. This not only mitigates catalyst deactivation, but also reduces the generation of solid waste and the need for frequent catalyst replacement, thereby lowering the environmental burden associated with catalyst disposal and production.
When compared to conventional non-carbon-based catalysts, carbon-based catalysts can offer a lower overall carbon footprint and reduced toxicity potential, given their earth-abundant materials and often simpler, less chemical-intensive preparation routes. For example, an Fe3O4@biochar system combined with microwave-assisted coke gasification can achieve net negative carbon emissions (−0.8 kg CO2-eq kg−1 syngas), highlighting a pathway for synergistic environmental benefit[294]. Concerning catalyst regeneration, LCA studies specifically focusing on the regeneration cycles of carbon-based catalysts are still emerging. Preliminary analyses suggest that regeneration strategies leveraging the catalysts' own reactivity (e.g., steam or CO2 gasification of deposited carbon) can be more energy- and material-efficient compared to the high-temperature calcination or chemical treatments often required for oxide-based catalysts[273]. However, the net environmental benefit depends heavily on the number of feasible regeneration cycles and the energy source for regeneration.
Despite these promising aspects, a significant research gap remains: publicly available, comprehensive LCA studies that quantitatively compare the full life-cycle impacts of advanced vs conventional carbon-based catalysts, and against benchmark non-carbon catalysts, are scarce. Most current environmental assessments are limited to single-issue analyses (e.g., carbon emissions during use) or suffer from unclear system boundaries and incomplete inventory data[295]. Future work must prioritize rigorous, standardized LCA to move beyond qualitative claims. By integrating process simulation, techno-economic analysis (TEA), and LCA into a comprehensive digital twin platform, multi-objective optimization algorithms can be employed to simultaneously balance conversion efficiency, long-term stability, economic cost, and environmental footprint[296]. This will provide a robust scientific foundation and decision-support for the sustainable industrialization of carbon-based catalysts, ultimately driving biomass gasification technology toward higher efficiency, lower carbon emissions, and enhanced resource circularity.
-
Carbon-based catalysts have demonstrated remarkable advantages in the efficient conversion of biomass tar and their development provides a robust guarantee for the production of renewable and clean syngas. Addressing the limitations of conventional biochar and coal-based catalysts, such as mechanical instability, proneness to coking, limited catalytic stability, and difficulty in targeted regulation and modification, it is a feasible approach to explore the universal catalytic mechanisms and advantageous characteristics of carbon-based catalysts using advanced carbon-based materials capable of precise regulation of porous/catalytic structures and electronic properties at the microscale, as tools, thereby guiding the targeted modification of conventional catalysts facilitating industrial application. Based on this insight, future research should further deepen mechanism-oriented exploration centered on advanced carbon materials and establish a mechanism library for carbon-based catalysts in the catalytic reforming of tar. Strategies such as heteroatom doping, hierarchical pore structure engineering, and spatial confinement of active metals can be explored for advanced carbon materials. Furthermore, the application of artificial intelligence in catalyst performance retrieval, design, and process optimization can significantly reduce the cost of catalyst development via the trial-and-error method. Moreover, research focus should shift toward economically viable modification technologies that can directionally reproduce the advantageous characteristics of advanced carbon-based catalysts on low-cost and readily available conventional carbon-based catalysts. Meanwhile, in industrial applications, more attention should be paid to improving the mechanical fatigue performance and reproducibility of catalysts, and coupling multi-field domains such as microwave fields, electric fields, and optical fields to further enhance the activity and efficiency of carbon-based catalysts. Rational use of carbon-based catalysts in biomass gasification can substantially contribute to energy transition and carbon neutrality.
We would like to express our sincere gratitude to the Interdisciplinary Innovation Lab. for Energy & Environment at Tianjin University of Commerce for providing the research platform and support.
-
The authors confirm their contributions to the paper as follows: all authors contributed to the study conception and design; literature survey, data collection and analysis were performed by Ziwei Xue and Yuechi Che; Beibei Yan, Xiaoqiang Cui and Jian Li participated in the review and commentary of previous versions of the manuscript; Guanyi Chen provided overall supervision and guided the research direction; the first draft of the manuscript was written by Ziwei Xue and all authors commented on previous versions of the manuscript. All authors reviewed the results and approved the final version of the manuscript.
-
This work was supported by National Natural Science Foundation of China (Grant No. 52306267), and Tianjin Science and Technology Bureau (24ZXTKSN00050).
-
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
-
Full list of author information is available at the end of the article.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Chen G, Xue Z, Che Y, Yan B, Cui X, et al. 2026. Progress of carbon-based catalysts for biomass tar reforming: a review. Sustainable Carbon Materials 2: e012 doi: 10.48130/scm-0026-0007
Progress of carbon-based catalysts for biomass tar reforming: a review
- Received: 14 November 2025
- Revised: 16 January 2026
- Accepted: 25 January 2026
- Published online: 11 March 2026
Abstract: Tar formation remains a major bottleneck for the sustainable utilization of biomass gasification, and catalytic reforming is an effective route for tar conversion. Among various catalyst systems, carbon-based catalysts exhibit unique advantages due to their tunable pore structures, adjustable surface chemistry, and intrinsic electrical conductivity. Unlike non-carbon supports, conductive carbon matrices actively regulate coke evolution by facilitating electron transfer at metal-carbon interfaces, thereby promoting carbon dissolution-precipitation pathways and the transformation of amorphous coke into ordered graphitic structures. This distinctive 'coke management' behavior mitigates active-site encapsulation and directly links electrical conductivity to catalyst stability and long-term reforming performance. Based on structural controllability and functional mechanisms, carbon-based catalysts can be classified into two categories: conventional carbon catalysts (e.g., biochar and coal char), which are low-cost and readily available but exhibit structural heterogeneity, and advanced carbon catalysts (e.g., MOFs-derived carbons, carbon nanotubes, and graphene-based materials), which possess ordered pore networks, well-defined metal-support interactions, and highly graphitized conductive frameworks. While conventional carbons are attractive for large-scale application, their disordered microstructures limit precise control over mass transfer, metal dispersion, and coke evolution. In contrast, advanced carbon materials provide model platforms to elucidate structure-activity-stability relationships and to establish transferable design principles. This review integrates recent progress in both catalyst classes, emphasizing how insights from advanced carbon systems can guide the targeted modification of conventional carbons, and proposes a unified framework linking conductivity, coke behavior, and durability for the rational design of efficient and stable carbon-based catalysts for biomass tar reforming.