-
Water is the cornerstone of sustaining life on Earth. However, up to 6 billion people are globally projected to face severe drinking water scarcity by 2050[1] according to the United Nations World Water Development Report. Concurrently, approximately 380 billion cubic meters of wastewater are generated globally each year, and this number is expected to rise continuously. The discharge of untreated wastewater introduces a wide variety of pollutants into water bodies, ranging from traditional heavy metals, dyes, and nutrients to emerging contaminants known for high persistence and ecotoxicity, such as antibiotics, perfluorinated compounds, and microplastics (MPs). To this end, wastewater treatment is of vital importance.
Various water treatment technologies, such as chemical precipitation, membrane separation, and advanced oxidation, have been widely studied and applied to face this challenge. However, these technologies often have inherent limitations, including high costs, high energy consumption, potential for secondary pollution, or limited removal efficiency for low-concentration emerging contaminants[1]. Adsorption is favored in this paper due to its simplicity, high efficiency, and relatively low cost. Biochar is an emerging adsorbent prepared via the thermochemical conversion of biomass waste, such as agricultural/forestry waste and municipal sludge. This process aligns well with the principles of the circular economy. Its porous structure, high specific surface area, and rich surface chemistry give it significant potential for removing various pollutants from water.
However, the practical application of pristine biochar faces several bottlenecks. First, its adsorption capacity and selectivity are often insufficient for highly toxic or emerging contaminants in complex water matrices. Second, its powder form complicates separation and recovery from treated water, posing a risk of secondary release. To overcome these bottlenecks, engineering and compositing have become inevitable trends. Compositing biochar with nano-metal oxides, magnetic materials, or functional polymers is an effective strategy. This approach precisely endows the material with enhanced adsorption/catalytic activity, specific targeting functions, and properties facilitating magnetic separation[2,3]. This strategy enables biochar-based composites to demonstrate efficacy far superior to that of pristine biochar in treating complex wastewater, particularly for emerging contaminants.
Currently, most research focuses on the synthesis and characterization of new materials. However, integrated discussions on key issues remain insufficient, including the systematic summarization of composites for addressing emerging contaminants, in-depth analysis of life-cycle environmental risks, and the maximization of 'net environmental benefits' through green design. Consequently, this paper aims to present a forward-looking review that systematically outlines the construction strategies of biochar composites and the removal mechanisms for both traditional and emerging contaminants. Special emphasis is placed on examining the trade-off between performance and risk in environmental applications. The ultimate goal is to provide a systematic knowledge foundation and design framework to drive the field toward a more sustainable future.
-
Biochar is a carbon-rich solid material prepared via pyrolysis carbonization at relatively low temperatures (≤ 700 °C) under completely or partially oxygen-limited conditions. As the initial step and key foundation of the preparation process, feedstock selection not only directly affects biochar yield, but also determines its physical structure, chemical composition, and subsequent application performance. Traditional feedstock sources for biochar are diverse, mainly including agricultural wastes (e.g., rice husks, straw), forestry residues (e.g., sawdust, branches), livestock manure, and municipal sludge[4]. These raw materials have characteristics such as abundant reserves, high renewability, and low cost. However, they differ significantly in elemental composition, ash content, and structural components. Consequently, the resulting biochars exhibit marked differences in specific surface area, pore structure, surface functional groups, and nutrient content.
In recent years, the scope of feedstock selection has continuously expanded and innovated due to the increasing demand for functional diversity in biochar. Tiwari et al. systematically explored the potential of using invasive plant biomass for biochar preparation[5]. They pointed out that this method not only helps control the spread of invasive species and alleviate ecological pressure, but also converts harmful biomass into high-value functional materials. Biochar derived from invasive plants (e.g., water hyacinth, ragweed) typically possesses superior pollutant adsorption performance and stable carbon sequestration capabilities due to its distinct cellular structures and chemical compositions. Thus, it shows broad prospects in restoring degraded environments, such as coal mine spoil heaps and heavy metal-contaminated soils.
Furthermore, co-pyrolysis of wastes with different physicochemical properties has become an effective strategy for precisely regulating biochar performance. For instance, a study co-pyrolyzed cellulose-rich aquatic plants with lignin-rich sawdust[6]. Biochar with a hierarchical pore structure was successfully prepared by leveraging the complementarity of the decomposition behaviors during pyrolysis. This material significantly outperformed those prepared from single feedstocks in terms of specific surface area and pore volume. It not only enhanced its adsorption capacity for organic pollutants and heavy metals, but also provided new insights for designing high-performance environmental adsorbents.
Recently, the co-pyrolysis of industrial by-products with biomass has emerged as a research hotspot, reflecting the green concept of resource recycling. For example, research has utilized the one-step co-pyrolysis of iron-rich red mud and agricultural pomelo peel to prepare magnetic biochar (RMBC). This material achieved synergistic resource utilization of two wastes. Moreover, the iron species loaded on its surface served as efficient catalytic active sites to activate peroxymonosulfate (PMS) for degrading dye pollutants in water, demonstrating good potential in advanced oxidation processes[7]. Another study explored converting fruit wastes (e.g., orange peel and walnut shell) into biochar via pyrolysis at 500 °C[8]. It systematically evaluated the effect of biochar in adsorbing and removing arsenic (As) from soil and water. The result showed that walnut shell biochar (WaSB500) achieved an arsenic removal efficiency of 88.8%, significantly outperforming similar materials. This was attributed to its higher degree of pore development and richer surface oxygen-containing functional groups.
In summary, feedstock selection is a critical step in biochar preparation. It directly regulates the final elemental composition, ash content, pore structure, and surface chemistry (as summarized in Table 1). Consequently, it influences the practical performance of biochar in environmental remediation, agricultural improvement, and energy catalysis. In the future, biochar feedstock strategies will become more diversified, precise, and functionalized with a deeper understanding of the feedstock-process-performance correlation mechanism.
Table 1. Preparation and properties of biochar
Category Method/material/
parameterEffect on biochar characteristics Mechanism/principle Ref. Feedstock selection Agricultural/forestry wastes Renewable, low-cost; elemental/ash variations significantly affect SSA, porosity, and functional groups. Intrinsic components (lignin, cellulose, ash) determine physicochemical structure. Hayder et al.[4] Invasive plants (water hyacinth, ragweed) Superior pollutant adsorption and stable carbon sequestration. Utilization of specific cellular structures and chemical compositions. Tiwari et al.[5] Co-pyrolysis (aquatic plants and sawdust) Hierarchical pore formation; SSA and pore volume superior to single feedstocks. Complementary pyrolysis behaviors of different components. Loc et al.[6] Co-pyrolysis (red mud and pomelo peel) Magnetic biochar; surface iron sites activate PMS for dye degradation. Synergistic waste utilization; introduction of magnetic/catalytic functions. Gai et al.[7] Fruit waste (walnut shell) 88.8% As removal efficiency. Developed porosity and abundant surface oxygen groups. Kumar et al.[8] Method Pyrolysis Produces biochar, bio-oil, syngas. Slow pyrolysis: high char yield; fast: high bio-oil. Thermochemical decomposition under oxygen-limited conditions. Cha et al.[9] Hydrothermal carbonization (HTC) Suitable for high-moisture feedstocks; hydrochar has higher carbon content. Hydrolysis, dehydration, decarboxylation, and aromatization. Funke et al.[10] Gasification Low char yield; rich in alkali salts/minerals, suitable for soil remediation. Partial oxidation targeting gaseous products. You et al.[11] Parameter Temperature Ash, SSA, pH increase; H/C and (O+N)/C decrease; aromaticity increases. Volatile release, micropore development, and unstable component removal. Tomczyk et al.[12] Atmosphere (CO2 vs N2) BET area in CO2 (303.73 m2/g) >> N2
(24.44 m2/g).CO2 selectively etches carbon skeleton, promoting pore expansion. Banik et al.[13] Property Elemental composition Lignocellulosic: high C, low ash; manure/sludge: high ash, nutrient retention. Temperature-dependent decomposition of hemicellulose, cellulose, and lignin. Ippolito et al.[14] BET surface area Increases then decreases with temperature; CO2 enhances SSA. Pore creation via volatile release vs collapse at high temps; CO2 etching. Zhao et al.[15] Functional groups Low-temp: rich in O-groups (–OH, –COOH); High-temp: group loss, hydrophobicity. Thermal decomposition of unstable oxygen-containing groups. Gomez-Eyles et al.[16] CEC Peaks at 250–300 °C; oxidative modification boosts CEC. Correlates with acidic O-groups; modification adds new groups. Kharel et al.[17] Note: SSA, specific surface area; CEC, cation exchange capacity. Different biochar preparation methods
-
Biochar preparation involves a series of carbonization processes, primarily including pyrolysis, gasification, and hydrothermal carbonization (HTC). These methods convert biomass feedstocks into carbon-rich solid materials via distinct reaction conditions and mechanisms.
Preparation via pyrolysis
-
Pyrolysis occurs under oxygen-free conditions, typically within a temperature range of 300–900 °C[9]. This process generates three types of products: solids, liquids, and gases. The solid product is commonly termed biochar, the liquid product is bio-oil, and the gaseous product is syngas, which mainly contains carbon dioxide, hydrogen, and carbon monoxide.
The heating rate determines the pyrolysis speed, influencing biochar characteristics as well as the yields of bio-oil and biogas[18,19]. Prolonged residence time allows for more complete biomass decomposition, but reduces biochar yield. Pyrolysis is categorized into fast and slow pyrolysis. In fast pyrolysis, feedstock is generally added to the reactor after the target temperature is reached, with a residence time of only a few seconds. Conversely, in slow pyrolysis, feedstock is introduced at the onset of heating, with residence times ranging from half an hour to several hours.
HTC method
-
HTC is a wet carbonization technology conducted in an aqueous medium. Biomass feedstock is crushed, mixed with deionized water, and placed in a sealed reactor to react at 150–300 °C and autogenous pressure. The process involves steps such as hydrolysis, dehydration, decarboxylation, and aromatization, ultimately producing a solid product known as hydrochar. Unlike pyrolysis, HTC is particularly suitable for directly treating feedstocks with high moisture content, thereby avoiding the high energy consumption associated with drying.
HTC is performed in the aqueous phase, where feedstock and water are mixed and reacted under elevated temperature and pressure, typically not exceeding 250 °C. Compared to pyrolysis and gasification, biochar prepared via HTC generally possesses a higher carbon content[10]. As a representative wet carbonization method, this process uses sludge as feedstock and water as the thermal medium in a closed system. Operating at specific temperatures (150–250 °C) and pressures, it undergoes hydrolysis, dehydration, decarboxylation, condensation, and aromatization reactions. These reactions increase carbon content while decreasing hydrogen and oxygen levels, generating high-energy sludge-based biochar. The process features mild reaction conditions and is not limited by moisture content[20,21].
Preparation via gasification
-
Gasification utilizes gasifying agents (such as air, oxygen, or steam) to partially oxidize the feedstock. Its operating temperature is typically above 800 °C[11], requiring the introduction of small amounts of oxygen or steam. Similar to pyrolysis, gasification generates solid, liquid, and gaseous products. However, as the gaseous product is the primary target, the biochar yield is usually lower than that of pyrolysis.
Biochar produced during gasification typically contains high levels of alkali salts and alkaline earth minerals. These components can immobilize many heavy metal pollutants, making the biochar suitable for direct use as a remediation agent for problematic soils[22]. Deal et al.[23] reported that problematic soils amended with gasifier-derived biochar resulted in higher corn yields, with the soluble ash content of the biochar exhibiting the most significant impact on soil productivity.
Impact of process parameters on biochar characteristics
-
Biomass type and pyrolysis parameters (e.g., temperature, heating rate, and residence time) collectively influence the composition and properties of the pyrolysis products[24]. Generally, higher pyrolysis temperatures reduce biochar yield due to the intensified primary or secondary decomposition of carbonaceous matter[25].
Research indicates that as pyrolysis temperature rises, the ash content, specific surface area, and pH of biochar typically increase, whereas the H/C ratio (reflecting aromaticity) and (O + N)/C ratio (reflecting polarity) decrease. The increase in pH may be associated with the release of volatiles and the precipitation of alkali salts during pyrolysis[26]; the increases in ash content and specific surface area are attributed to the loss of carbon components and the development of microporous structures at high temperatures[27]. Additionally, higher temperatures facilitate the removal of unstable components and the formation of finer pores, thereby increasing the total pore volume of the biochar[28,29]. These trends in porosity development are clearly illustrated in Fig. 1.
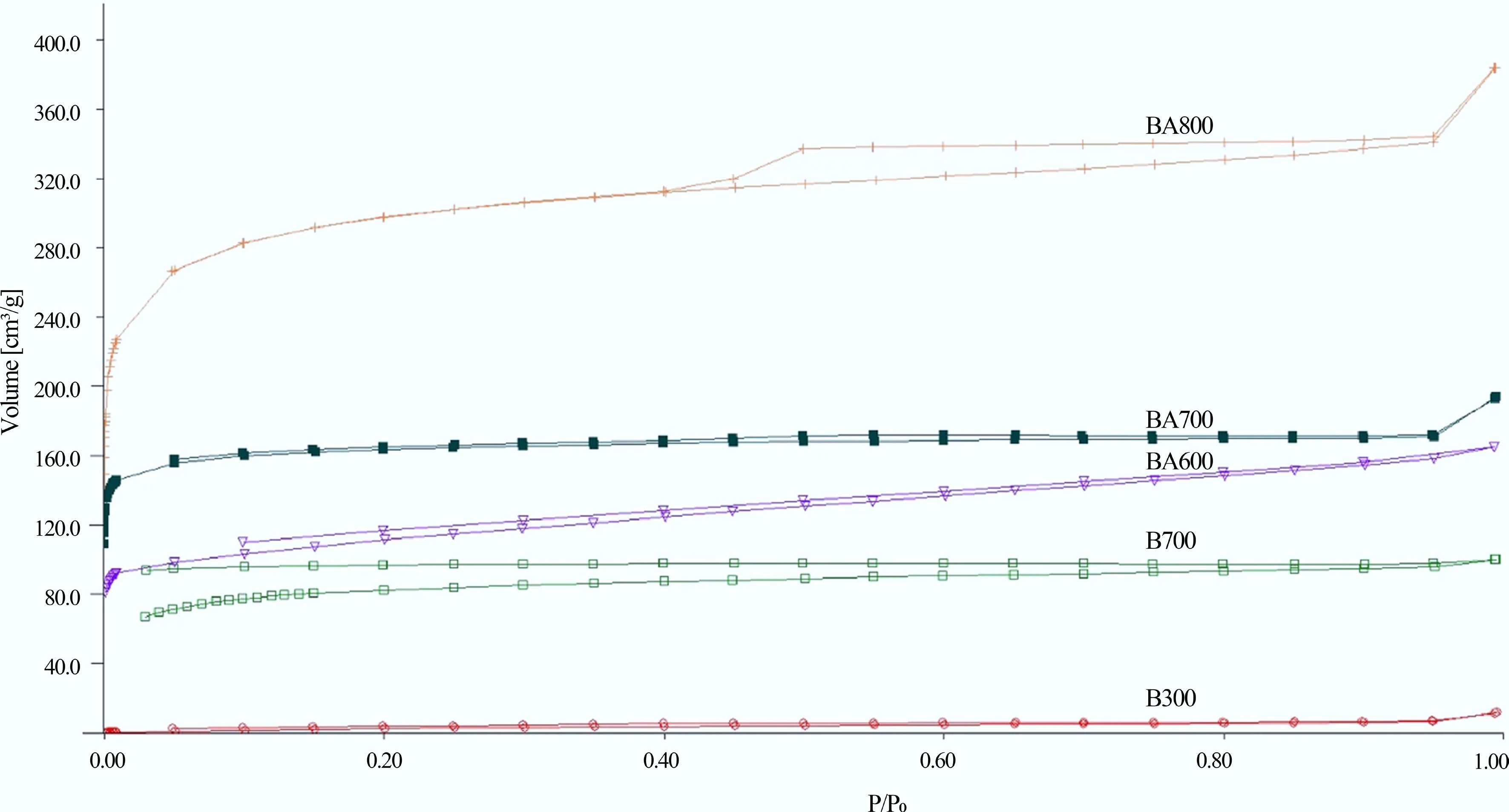
Figure 1.
N2 adsorption isotherms at 77 K for pure (B300, B700) and steam-activated (BA600, BA700, and BA800) biochar[30].
Other preparation processes
-
Beyond the mainstream methods mentioned above, biochar can also be prepared via processes such as flash carbonization and torrefaction[31,32]. Flash carbonization is typically conducted under a pressure of 1–2 MPa and temperatures of 300–600 °C in an inert atmosphere. With a feedstock residence time of approximately 30 min, it converts biomass into a predominantly solid product. Torrefaction is implemented at lower temperatures (200–300 °C). Through partial oxidation, moisture and volatile components are removed from the feedstock, generating a hydrophobic solid product. Biochar obtained from this process typically exhibits a low oxygen-to-carbon ratio[9]. However, due to partial oxidation, the ordering of its carbon matrix structure is relatively low, which may result in reduced adsorption performance.
Preparation method selection, optimization, and future trends
-
As indicated above, biochar preparation methods are diverse. The selection and process optimization do not follow a single standard, but rely heavily on the target application scenario (e.g., adsorption, catalysis, soil improvement) and varying definitions of efficiency (e.g., maximum material performance, minimum energy consumption, shortest duration, or highest yield). Table 2 summarizes the characteristics and applicability of the primary preparation methods.
Table 2. Comprehensive comparison and selection guide for major biochar preparation methods
Method Core features and efficiency advantages Major limitations Typical temperature Recommended applications Ref. Slow pyrolysis High biochar yield; stable structure; high degree of aromaticity. Time-consuming; relatively low heating efficiency. 400–600 °C Prioritizing char yield and stable carbon sequestration (e.g., soil amendment). Do et al.[33] Fast pyrolysis High bio-oil yield; rapid process. Low biochar yield; difficult to tune physicochemical properties. 500–700 °C Primarily targeting bio-energy (bio-oil) co-production. Cha et al.[9] Hydrothermal carbonization (HTC) Directly treats high-moisture feedstocks (avoids drying energy); rich surface functional groups. Product (hydrochar) typically has a lower degree of carbonization 180–250 °C Immediate valorization of high-moisture wastes (e.g., sludge, food residues). Xu et al.[21] Co-pyrolysis/
modified pyrolysisSynergistic effects; enables one-step doping, magnetization, or pore optimization; simplifies processing. Precursor formulation and reaction mechanisms can be complex. Target-dependent (e.g., magnetic: 250–600 °C) Targeted preparation of functional composites (e.g., magnetic adsorbents, catalysts). Zheng et al.[34] Gasification Primary target is Syngas (H2, CO); mature technology for large-scale continuous production; high energy efficiency. Very low biochar yield (5%–15%); strict process control required; tar and ash issues; complex equipment/operation. 600–1,200 °C Large-scale bio-energy utilization (power/hydrogen generation); biochar used for sequestration or soil amendment. You et al.[11] In summary, biochar preparation is evolving from a singular pursuit of yield or performance toward multidimensional efficiency optimization characterized by target-oriented design, energy intensiveness, and process intelligence. For instance, microwave-assisted pyrolysis (MAP) has emerged as a highly efficient alternative to conventional methods, offering volumetric heating, significant reductions in activation energy, and rapid structural control. Future research should prioritize reverse design based on application demands, combining such novel heating methods with AI-driven intelligent optimization algorithms to achieve efficient, controllable, and sustainable conversion from feedstock to functional products[35].
Physicochemical properties of biochar
Elemental composition of biochar
-
The elemental composition of biochar is a critical characteristic, significantly influenced by feedstock type and pyrolysis temperature[14]. As the foundational input for pyrolysis, feedstocks are diverse and broadly categorized into woody biomass (e.g., wood, bark), crop residues (e.g., straw, stalks), and manures/biosolids (e.g., livestock manure, sludge). Woody and agricultural biomass typically contain 15%–25% lignin, 35%–50% cellulose, and 15%–40% hemicellulose[36]. Variations in these components directly determine the elemental profile of the final biochar.
Regarding feedstock, woody materials usually contain negligible amounts of inorganic components like nitrogen (N), potassium (K), calcium (Ca), and sulfur (S). Consequently, biochar derived from them exhibits high carbon content and low ash, limiting its nutrient retention capacity. Conversely, manure or biosolid feedstocks are rich in minerals and nutrients. Biochar produced from these sources not only possesses high ash content and strong nutrient retention, but also often exhibits a higher calcium carbonate equivalent (CCE), indicating excellent potential for neutralizing acidity[37,38]. Furthermore, feedstocks rich in mineral salts (e.g., chlorides) may promote volatile release during pyrolysis, thereby reducing biochar yield[39]. For instance, Choudhary et al.[40] systematically compared nutrient-rich sugarcane press mud and farm manure with low-nutrient rice straw, further validating the decisive role of feedstock source on elemental composition.
Beyond feedstock type, pyrolysis temperature is a key parameter regulating elemental composition. The three major biomass components—hemicellulose, cellulose, and lignin—decompose in distinct temperature ranges: hemicellulose at 150–350 °C, cellulose at 275–350 °C, and the more structurally stable lignin gradually between 250 and 500 °C. The complex aromatic structure of lignin leads to slow degradation during pyrolysis, contributing to higher biochar yield[41,42]. As pyrolysis temperature rises, both carbon and ash contents in the biochar tend to increase. The rise in carbon content stems from enhanced polymerization and aromatization, forming denser, more stable condensed aromatic structures. The increase in ash content results from the concentration and enrichment of mineral elements such as Na, Ca, Mg, K, and Si within the char matrix at high temperatures[12]. Therefore, pyrolysis temperature regulates not only carbon stability, but also ash composition and the potential environmental functions of the biochar.
Specific surface area of biochar
-
The specific surface area is a core parameter for evaluating the physical adsorption performance of biochar, directly determining its capacity and efficiency in accommodating gas molecules or pollutants in solution. The formation and evolution of this property are primarily governed by pyrolysis conditions and feedstock properties.
During pyrolysis, pore structure generation essentially results from the thermochemical conversion of organic components in the precursor. As temperature rises, volatile components like esters and aliphatic alkyls undergo bond-breaking and escape. Simultaneously, the originally encapsulated aromatic lignin skeleton is exposed, constructing abundant micropores (< 2 nm) and mesopores (2–50 nm) within the carbon matrix[43]. The final pyrolysis temperature is a critical factor. Typically, specific surface area initially increases and then decreases with rising temperature. In the low-to-medium range (e.g., 300–600°C), continuous volatile release expands existing channels and creates new pores, significantly increasing surface area. However, at excessive temperatures (e.g., > 700 °C), pore walls may collapse or merge due to over-graphitization or high-temperature sintering, causing surface area reduction. Concurrently, the pore structure evolves: high temperatures widen some micropores into meso- or macropores, reducing the micropore fraction, but increasing total pore volume[44].
In addition to temperature, pyrolysis atmosphere significantly regulates pore development. Banik et al.[13] clearly demonstrated it by comparing rice straw biochar prepared under CO2 vs N2 atmospheres. In CO2, the surface area increased from 2.1 m2/g at 300 °C to 303.73 m2/g at 800 °C, whereas in N2, it only rose from 1.8 m2/g to 24.44 m2/g. This vast difference is attributed to CO2 acting as a weak oxidizing agent, selectively etching the carbon skeleton to promote pore expansion and connectivity. Zhao et al.[15] provided microstructural evidence, noting that high temperatures drive the phase transition of ordered cellulosic carbon structures into disordered amorphous carbon. This process, accompanied by volume shrinkage and the generation of new defects and pores, collectively boosts surface area. Furthermore, advanced activation strategies can maximize this property; for example, employing a microwave-assisted two-step activation strategy (e.g., preliminary H3PO4 activation followed by KOH etching) can construct hierarchically porous biochar with ultra-high specific surface areas exceeding 3,000 m2/g. Precisely tuning the proportion of mesopores to micropores in such structures is critical for simultaneously optimizing adsorption capacity and mass transfer kinetics[45].
However, regardless of condition optimization, intrinsic feedstock characteristics remain the fundamental determinant of pore structure development. Janu et al.[46] provided a typical case: using sewage sludge with high ash and low cellulose content, the specific surface area did not change significantly with pyrolysis atmosphere (N2 vs CO2). The primary reason was that abundant ash and inorganic matter blocked pores and inhibited the ordered evolution of the carbon skeleton, indicating that feedstock chemistry largely dictates the final pore formation pathway and performance ceiling.
Surface functional groups of biochar
-
The surface chemical properties of biochar and its interaction with environmental pollutants are largely determined by the type and abundance of surface functional groups, including oxygen-containing groups such as hydroxyl (–OH), carboxyl (–COOH), phenolic, and carbonyl (C=O). These groups can interact with pollutant molecules through electrostatic interactions, hydrogen bonding, or π–π electron coupling. These groups significantly influence adsorption affinity and selectivity. However, the composition and distribution are dynamic, regulated by factors including pyrolysis temperature, feedstock composition, and subsequent modification methods.
Pyrolysis temperature is critical for functional group stability and evolution. The results of Janu et al.[46] showed that biochar from Picea abies sawdust pyrolyzed at relatively low temperatures (300–600 °C) is rich in oxygenated groups like phenolic, alcohol, and hydrogen-bonded hydroxyls. However, these groups are more thermally labile than the stable aromatic structures formed at high temperatures. Generally, key oxygenated groups like hydroxyl, carboxyl, and phenolic show the most distinct response between 350–400 °C. At higher temperatures, intense pyrolysis causes the loss of these groups, shifting the surface chemistry from polar and hydrophilic to non-polar and hydrophobic[16,47].
Oxygen-containing functional groups decisively influence surface charge and ion exchange behavior. Acidic groups like carboxyl, phenolic, and hydroxyl undergo proton dissociation in solution, imparting persistent negative charges to the biochar surface, thereby enabling efficient proton and cation exchange[48]. Consequently, the cation exchange capacity (CEC) is closely related to the quantity of surface functional groups. Generally, biochar CEC peaks at 250–300 °C and then declines as oxygenated groups decompose at higher temperatures[12]. Additionally, feedstock composition and surface modification also regulate CEC. For example, Ambaye et al.[49] noted that using iron-rich feedstocks enhances surface charge effects via metal-oxygen coordination, boosting CEC. Kharel et al.[17] found that ozone-modified biochar, enriched with carboxyl groups, showed a roughly 10-fold increase in CEC.
Although negatively charged biochar typically adsorbs cationic pollutants more easily, precise surface modification can effectively enhance anion removal. For instance, Liu et al.[50] reported a biochar-based nano-zero-valent iron composite achieving 89%–97% nitrate removal. The mechanism involved zero-valent iron reducing NO3− to harmless products like NO2−, NH4+, and N2, with the biochar carrier providing a dispersion medium and electron transfer channels.
Furthermore, functional group composition can be tailored via chemical modification. Yakout[51] compared rice straw biochar treated with different oxidants. H2SO4 and HNO3 treatments weakened aliphatic C–H stretching and enhanced aromatic characteristics, introducing new C=O stretching peaks attributed to carbonyl, lactone, and quinone structures. Conversely, KMnO4 treatment broadened the –OH absorption peak, indicating strong hydrogen bond synergy between hydroxyls and adjacent carbonyls, thereby optimizing surface reactivity and catalytic potential.
pH and redox properties
-
The pH and redox properties of biochar are macroscopic manifestations of its surface chemistry at the water-solid interface, directly regulating the environmental fate and remediation efficiency of pollutants with variable oxidation states (e.g., heavy metals, metalloids). These properties are not static, but are synergistically shaped by feedstock chemical composition and the thermal history of pyrolysis.
Biochar pH generally increases with rising pyrolysis temperature. This is driven primarily by two mechanisms. First, high-temperature pyrolysis decomposes biomass components like cellulose and hemicellulose, eliminating acidic oxygenated groups such as carboxyl (–COOH) and phenolic hydroxyl groups. Second, alkali and alkaline earth metals (e.g., K, Ca, Mg) in the feedstock enrich in the ash as stable carbonates or oxides. Upon hydrolysis in water, these generate OH–, contributing to alkalinity[12,52]. An et al.[52] found that pyrolyzing three types of industrial hemp stalks at varying temperatures increased biochar pH and conductivity, while FTIR data confirmed a reduction in acidic surface oxygen groups. Zhang et al.[53] further confirmed that feedstock and pyrolysis temperature are key determinants of pH, capable of raising soil pH by an average of 5.59 units. For example, Li et al.[54] produced biochar from mineral-rich manure or straw at high temperatures (> 500 °C) with pH values reaching 9–11. This intrinsic alkalinity effectively neutralizes acidic environments and, crucially, promotes immobilization of cationic heavy metals (e.g., Cu2+, Cd2+, Pb2+) via hydroxide precipitation or surface complexation by raising ambient pH[55]. Research indicates that oxidative modification to increase surface oxygen groups significantly enhances Pb2+ adsorption and passivation under neutral conditions, highlighting the synergistic regulation of pH and surface chemistry[56].
Biochar redox activity is heterogeneous; its intensity and active site types are precisely regulated by pyrolysis temperature and feedstock attributes, directly determining its performance as an environmental electron mediator[57]. Pyrolysis temperature regulates redox activity by reshaping the carbon skeleton. Typically, biochar prepared in the medium-to-high range (400–700 °C) exhibits the highest electron accepting and donating capacities[58]. This is because rising temperatures increase aromatization, forming more extended conjugated π-electron systems that underpin electron storage and shuttling[59]. Simultaneously, temperature governs the transformation of key surface groups: at medium temperatures (approx. 400–600 °C), lignin pyrolysis favors the formation of quinone groups (C=O), which serve as primary electron acceptors. Higher temperatures (> 700 °C) may cause functional group loss, and highly graphitized carbon structures may contribute new electron transfer pathways[57]. For instance, Wu et al.[59] prepared nano-biochar from wheat straw at 400 °C and 700 °C via grinding, sonication, and separation. The study showed that nano-biochar prepared at 700 °C possessed significantly higher electron transfer capacity than the 400 °C sample.
Feedstock chemical composition also influences biochar redox properties. Feedstocks rich in variable-valence metals (e.g., manures, certain sludges) produce biochar where metals exist as oxides or are bound to the carbon matrix, serving as potent redox-active centers[57]. Additionally, variations in organic components (e.g., lignin/cellulose ratio) affect the generation and stability of persistent free radicals and quinone-phenol groups during pyrolysis[60]. Consequently, biochars derived from different feedstocks may exhibit significantly different redox capacities even when pyrolyzed at the same temperature.
-
To address the limitations of pristine biochar in environmental remediation, biochar composites are constructed using biochar as the matrix. These materials maximize the inherent physicochemical properties of biochar while integrating the performance advantages of constituent components. Surface modification with inorganic or organic agents via physical or chemical means is a key strategy to enhance utilization efficiency. Currently, common biochar-based composites include magnetic biochar, nano-metal oxide/hydroxide biochar, and functional nanoparticle-coated biochar composites[61]. Table 3 summarizes the synthesis methods for different biochar composites.
Table 3. Synthesis methods of different biochar composites
Biomass/feedstock Synthesis method Active species Target pollutant Ref. Pine sawdust and sodium alginate Cross-linking with FeCl3 and sodium alginate for simultaneous molding and magnetization, followed by low-temperature pyrolysis. Fe3O4 Pb(II) in soil Zeng et al.[62] Elephant grass Grinding biomass with K2FeO4, followed by pyrolysis at
700 °C for 1 h under nitrogen atmosphere.nZVI (nano zero-valent iron) Sulfamethoxazole He et al.[63] Shiitake mushroom Precursor formation via iron salt adsorption by shiitake mushrooms, followed by high-temperature calcination. Fe/Fe4N Microwave absorption (functional materials) Qiang et al.[64] Wheat straw Impregnation with iron and tin salts, followed by pyrolysis. Fe3O4 Tetracycline Zhang et al.[65] Tobacco stalk Co-pyrolysis with biomass using FeCl2·4H2O as the magnetizing agent. Magnetic metal oxides Phosphorus Yu et al.[66] Preparation of magnetic biochar composites (MBCs)
-
Biochar-based materials typically exhibit excellent adsorption potential due to their large pore volumes and active sites (Fig. 2). Nevertheless, pristine biochar is difficult to separate and recover from water bodies due to its low density and fine particle size, posing a risk of secondary pollution. MBCs address this by coating biochar surfaces with magnetic iron oxide nanoparticles, such as K2FeO4[34], γ-Fe2O3, and CoFe2O4. This introduces magnetism and active iron oxide sites for pollutant removal. MBCs facilitate efficient separation and recovery via external magnetic fields, and the adsorption capacity can be further enhanced through magnetic iron oxide impregnation.
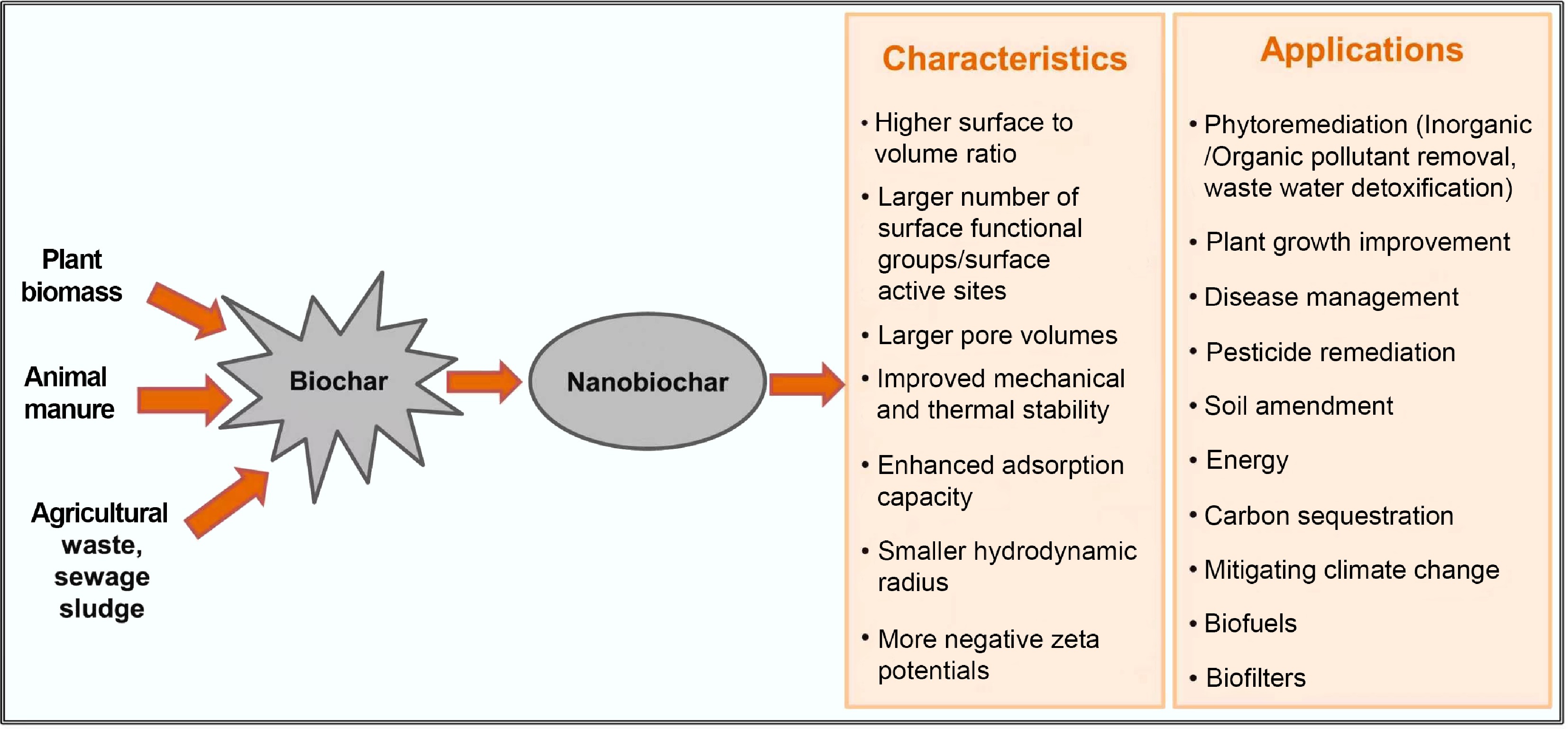
Figure 2.
Nanobiochar and biochar nanocomposites: characteristics and applications[67].
Pre-pyrolysis pretreatment method
-
This method involves loading Fe3+ or Fe2+ onto the biomass surface via impregnation or chemical co-precipitation prior to pyrolysis. Subsequent pyrolysis or microwave heating simultaneously achieves biomass carbonization and magnetic fixation of iron components, yielding MBCs. The primary advantage of this approach is its high process integration and one-step completion. Additionally, magnetic species (e.g., Fe3O4) and the nascent carbon skeleton form strong chemical bonds (e.g., Fe–O–C) in situ during pyrolysis, resulting in high material stability. This is a classic method widely adopted in both laboratory and engineering contexts[68−70]. For instance, Zhang et al.[71] successfully prepared a novel magnetic biochar adsorbent by pyrolyzing ferric chloride-treated biomass. This composite exhibited excellent ferromagnetism (saturation magnetization of 69.2 emu/g) and strong adsorption for arsenic in water. Similarly, Zeng et al.[62] prepared magnetic biochar spheres using pine sawdust and sodium alginate as carbon sources, with FeCl3 serving as both a cross-linker and magnetizing agent. The material possessed a large specific surface area and favorable pore structure, achieving an optimal removal rate of 67.53% and an adsorption capacity of 3.11 mg/g. Chen et al.[72] loaded Fe3+/Fe2+ onto orange peel powder via chemical co-precipitation before pyrolysis at three different temperatures, producing three novel MBCs. SEM and XRD analyses confirmed the presence of magnetic iron oxides. However, this method has several limitations. First, converting iron salts into magnetic species typically requires high temperatures (> 500 °C), which can cause the extensive decomposition of oxygen-containing functional groups and the agglomeration of iron species, thereby limiting adsorption capacity and active site density[34]. Second, precursor salts may block natural pores in the biomass, adversely affecting the specific surface area of the composite[69].
Post-pyrolysis loading method
-
Another approach involves first pyrolyzing biomass into biochar and subsequently loading magnetic materials onto its surface via liquid-phase precipitation, liquid-phase reduction, or mechanical ball milling. The core advantage of this method lies in its high design flexibility and controllability[68,69]. On one hand, biochar with high specific surface area and developed pore structure can be pre-screened or prepared as an ideal carrier. On the other hand, loading parameters (e.g., pH, concentration, time) can be precisely regulated independently of the pyrolysis process, facilitating control over magnetic particle size, morphology, and dispersion. Jin et al.[73] prepared iron/sulfur-modified magnetic biochar via a hydrothermal-calcination method (optimally at 750 °C). Through synergistic adsorption-oxidation, this material achieved a sulfamethoxazole removal rate of 95.5% within 30 min, significantly outperforming pristine biochar. This enhancement was attributed to the enrichment of surface functional groups and promoted electrochemical performance, which collectively promoted PAA activation. Quan et al.[74] prepared biochar-supported nano zero-valent iron (B-nZVI) via liquid-phase reduction. Characterization using TEM, SEM, XRD, XPS, and BET analysis showed that nZVI was uniformly dispersed on the biochar surface without significant agglomeration, exhibiting a high removal rate for Acid Orange 7 in water. Additionally, mechanical ball milling is perhaps the simplest method for preparing MBCs and is widely used for large-scale production. Ball-milled MBCs possess high external and internal specific surface areas and acidic surface functional groups, demonstrating excellent adsorption capacity for Ni(II) in aqueous solutions[75]. However, limitations include the relatively weak binding force (mainly physical adsorption or surface deposition) between magnetic particles and biochar, posing a risk of detachment in harsh environments. Furthermore, a secondary calcination step is often required to stabilize the magnetic phase, increasing process complexity and energy consumption[68,69].
In summary, pre-pyrolysis pretreatment and post-pyrolysis loading form complementary strategies for MBC preparation. The former excels in process simplicity and material stability, making it suitable for scenarios requiring high durability and large-scale application. The latter offers greater potential for precise design and functionality, applicable for developing high-performance composites with specialized structures[76]. Future research could explore synergistic strategies, such as using mild pre-pyrolysis pretreatment (e.g., low-temperature oxidative synergy) to build a stable magnetic matrix—avoiding the defects of functional group decomposition and iron agglomeration caused by traditional high temperatures (> 500 °C)[34,77]—followed by post-loading to precisely introduce secondary functional components (e.g., catalytic sites). This approach aims to create next-generation environmental remediation materials combining strong magnetism, high stability, and specific catalytic degradation capabilities[65]. Such stepwise synergistic strategies have been validated as feasible pathways in frontier research.
Preparation of nano-metal oxide/hydroxide biochar composites
-
Novel composites constructed from biochar and nanomaterials (e.g., MgO, TiO2) significantly increase surface functional groups and active sites, enhancing catalytic degradation capabilities[78]. Consequently, the synthesis of nano-biochar composites is a promising approach for environmental remediation. Preparation methods include bio-accumulation (bio-enrichment), metal salt pretreatment, and post-pyrolysis insertion of metal oxide nanoparticles. As detailed in Table 2, the first two methods primarily involve impregnating target metals into biomass prior to pyrolysis, while the latter introduces nano-metal oxides/hydroxides directly into the pyrolyzed biochar.
Bio-accumulation method
-
Bio-accumulation involves growing biomass to enrich specific elements from soil and fertilizers, which are subsequently retained and concentrated in the biochar during pyrolysis. The core advantage of this method is its green and economical nature, combining phytoremediation with waste valorization to realize a sustainable 'soil-to-adsorbent' synthesis pathway[79]. For example, Yao et al.[80] prepared innovative engineered biochar via the slow pyrolysis of magnesium-enriched tomato tissues. To achieve Mg enrichment, tomato plants in peat substrate were irrigated twice weekly with Hoagland's solution containing 25 mM magnesium. The resulting Mg-biochar composite exhibited superior phosphate adsorption performance. Statistical analysis revealed a strongly significant correlation between phosphate removal and Mg content in the biochar, confirming that enriched Mg was the dominant factor controlling phosphorus removal. Li et al.[81] prepared nano-ZnO/ZnS modified biochar from zinc-contaminated corn stover via slow pyrolysis. This zinc-mineral modified biochar possessed a superior pore structure compared to ordinary biochar and demonstrated strong adsorption capacities for Pb(II), Cu(II), and Cr(VI) (maximum capacities of 135.8, 91.2, and 24.5 mg/g, respectively), significantly outperforming ordinary biochar. This indicates that biochar composites obtained via bio-accumulation offer an effective, economical method beneficial for waste utilization and environmental management. However, the lengthy preparation cycle (dependent on plant growing seasons) and the variability of target metal enrichment due to soil properties, climate, and agricultural management lead to poor controllability and reproducibility. Precisely regulating nanoparticle size and distribution remains difficult[79]. Therefore, further research is needed to obtain biochar with optimized physicochemical properties via this route.
Metal salt pretreatment method
-
The principle of metal salt pretreatment is similar to the impregnation method used for magnetic biochar. Biomass impregnated with a target metal salt solution allows metal ions to attach to the surface and penetrate the interior. Upon pyrolysis, these particles convert into nano-metal oxides/hydroxides, transforming the metal-loaded biomass into a biochar-based nanocomposite[80]. This imparts new functional properties and expands its application potential. Specifically, pyrolyzing iron-treated biomass can yield magnetic biochar adsorbents[71]. The main advantage is the relatively simple process; metal precursors achieve initial molecular-level dispersion within the 3D structure of biomass, facilitating the formation of well-dispersed nanoparticles after pyrolysis. Additionally, metal loading can be conveniently adjusted via impregnation solution concentration[82]. However, the simultaneous decomposition of metal salts and biomass carbonization during pyrolysis may cause some nanoparticle agglomeration. Furthermore, salts at high temperatures may catalyze graphitization or etch the carbon skeleton, affecting the final development of the pore structure[79,82].
Common pretreatment reagents include AlCl3[71], CaCl2[71], MgCl2[71,83], KMnO4[84], and MnCl2[85], which ultimately form nanoparticles such as Al2O3, AlOOH, CaO, MgO, and MnOx on the biochar surface. For instance, Lawrinenko et al.[86] pretreated biomass with ferric and aluminum chloride, followed by slow pyrolysis. The formation of Al–O–C and Fe–O–C structures promoted a wider distribution of metal atoms, enhancing anion exchange capacity through the formation of metal oxyhydroxide surface coatings with high points of zero charge. Zhang et al.[87] synthesized highly porous MgO-biochar nanocomposites by slowly pyrolyzing MgCl2-pretreated biomass. Results showed that MgO nanoflakes were uniform in morphology and evenly dispersed on the biochar matrix, exhibiting excellent removal efficiencies for phosphate and nitrate in water.
Post-pyrolysis insertion of metal oxide nanoparticles
-
Biochar composites with metal oxide nanoparticles attached can also be synthesized after biomass pyrolysis. Common methods include evaporation[88], heat treatment[89], conventional wet impregnation[90], and direct hydrolysis[91], all involving biochar treatment in the presence of metal salts. The greatest advantage of this method is high flexibility in design and regulation. Since the biochar carrier already possesses a stable pore structure, researchers can select optimal nano-active components for specific pollutants and independently optimize loading conditions (e.g., pH, temperature, concentration) to control nanoparticle size, crystallinity, and distribution within pores[92]. However, the main challenge lies in achieving robust and uniform loading. Simple physical mixing or impregnation can lead to agglomeration or pore blockage, reducing the effective specific surface area. Weakly bound loads also pose a risk of leaching during dynamic water treatment, potentially causing secondary pollution[79,92]. Consequently, Cope et al.[88] modified biochar with iron oxide using dissolved ferric nitrate nonahydrate to construct surface chemistry favorable for arsenic adsorption. Results showed that the modified specific surface area and arsenate adsorption increased by 2.5 and 2 orders of magnitude, respectively, compared to pristine biochar. Liang et al.[93] successfully synthesized MnO2 biochar composites by modifying biochar derived from aerobically composted swine manure. The material featured a rougher surface morphology, larger specific surface area, and pore volume, with adsorption capacities for Pb(II) and Cd(II) (268.0 and 45.8 mg/g, respectively) significantly superior to pristine biochar (127.75 and 14.41 mg/g).
In summary, the core objective of compositing nano-metal oxides/hydroxides with biochar is to introduce highly active reaction centers into the relatively inert carbon matrix. Whether through the green pathway of bio-accumulation or the engineering pathways of pretreatment or post-loading, success hinges on achieving high dispersion, strong binding, and stability of the active components. Future research should transcend simple physical mixing or impregnation, aiming to construct active sites with distinct chemical structures and electronic interactions at the interface. Examples include the precise regulation of pyrolysis atmosphere and temperature to grow metal oxides with specific crystal facets in situ on the biochar surface, or designing core-shell structures to synergize adsorption and catalysis. This will not only greatly enhance removal efficiency and selectivity for pollutants (especially refractory organics), but also drive the evolution of such materials from broad-spectrum adsorbents to efficient, specific environmental catalysts[65,94].
Preparation of functional nanoparticle-coated biochar composites
-
Biochar nanocomposites represent an emerging class of advanced materials synthesized by immobilizing functional nanoparticles—such as chitosan, graphene, carbon nanotubes, and nano zero-valent iron—onto the biochar surface. These composites not only fully integrate the respective advantages of the biochar matrix and nanomaterials, but also improve their physicochemical properties, achieving efficient pollutant removal (especially of organics) and even enabling synergistic adsorption-degradation processes.
Pretreating biomass with functional nanoparticles prior to pyrolysis, typically via dip-coating, can convert biomass into biochar-based functional composites. This method offers process integration: during pyrolysis, volatiles and reducing gases from biomass decomposition may facilitate the chemical reduction or modification of functional materials (e.g., reducing graphene oxide) and potentially form strong chemical bonds between nanoparticles and the carbon matrix. However, high-temperature pyrolysis may damage the delicate structures of certain functional nanoparticles (e.g., carbon nanotube integrity) or specific functional groups. Furthermore, nanoparticle addition may interfere with the normal biomass pyrolysis process, affecting intrinsic pore formation[79,95]. Tang et al.[96] synthesized graphene/biochar composites by pretreating wheat straw with graphene followed by slow pyrolysis. Structural analysis indicated that graphene coated the biochar surface mainly via π–π interactions, resulting in larger specific surface area, more functional groups, higher thermal stability, and stronger phenanthrene and mercury removal capabilities compared to pristine biochar. This demonstrates that graphene/biochar composites are efficient, economical, and eco-friendly multifunctional adsorbents. Pi et al.[97] developed a low-cost composite for synergistic adsorption and photocatalytic treatment by pyrolyzing a mixture of melamine and biochar at 520 °C. The material showed good removal of the cationic dye methylene blue. Unlike traditional adsorbents that reach saturation, this composite maintained continuous decontamination ability via photocatalytic degradation of adsorbed organics under light.
Alternatively, loading functional nanoparticles (e.g., zero-valent iron[98,99], chitosan[100], Mg-Al layered double hydroxides[101]) onto the surface of pre-pyrolyzed biochar can also achieve an organic combination of advantages. This method retains the inherent benefits of biochar while successfully introducing nanoparticle characteristics. These immobilized nanoparticles act as highly efficient active sites, significantly enhancing pollutant adsorption. Notably, however, nanoparticle loading may partially block biochar pores, though their superior performance often compensates for this structural cost. For instance, the chitosan surface modification strategy employed by Zhou et al.[100] combined chitosan's high chemical affinity with biochar's porous structure. Yet, the study revealed a negative effect: chitosan loading blocked some pores, significantly reducing specific surface area. This suggests that when introducing functional nanomaterials, the trade-off between performance gains and structural costs must be carefully weighed and minimized through targeted optimization.
In summary, functional nanoparticle-coated biochar composites represent a key direction toward high-performance and intelligent environmental materials. The essence of their design lies in sophisticated compositing strategies that combine macroscopic structural advantages of biochar (e.g., hierarchical pores, mechanical strength) with the microscopic properties of functional nanoparticles (e.g., high conductivity, specific affinity, catalytic activity) to generate synergistic effects. Future development is predicted to focus on precise interface engineering: establishing robust, functionalized chemical bridges between nanoparticles and biochar via chemical grafting, in-situ polymerization, or layer-by-layer self-assembly, rather than simple physical attachment. This will effectively prevent active component loss and enable functional programmability, such as developing smart composites responsive to stimuli like pH, light, temperature, or specific molecules for targeted recognition, deep purification, and resource recovery in complex environmental matrices[102].
-
Biochar composites offer a promising strategy for wastewater treatment. These materials possess advantages such as high surface area and porosity[103,104], tailored surface functionality[105,106], and unique physicochemical properties[61,107,108], significantly enhancing their adsorption and catalytic capabilities. Additionally, the synergistic effect between biochar and nanomaterials improves selectivity and specific adsorption efficacy for target pollutants[109]. Through the directional regulation of surface functional groups, biochar composites can achieve efficient removal of diverse pollutants, including dyes[110,111], heavy metals, medical waste, and nitrogen/phosphorus nutrients. A schematic illustration of these interactions is provided in Fig. 3, which details the adsorption mechanisms of Methylene Blue onto composite beads. Table 4 summarizes some applications of biochar composites in wastewater treatment.
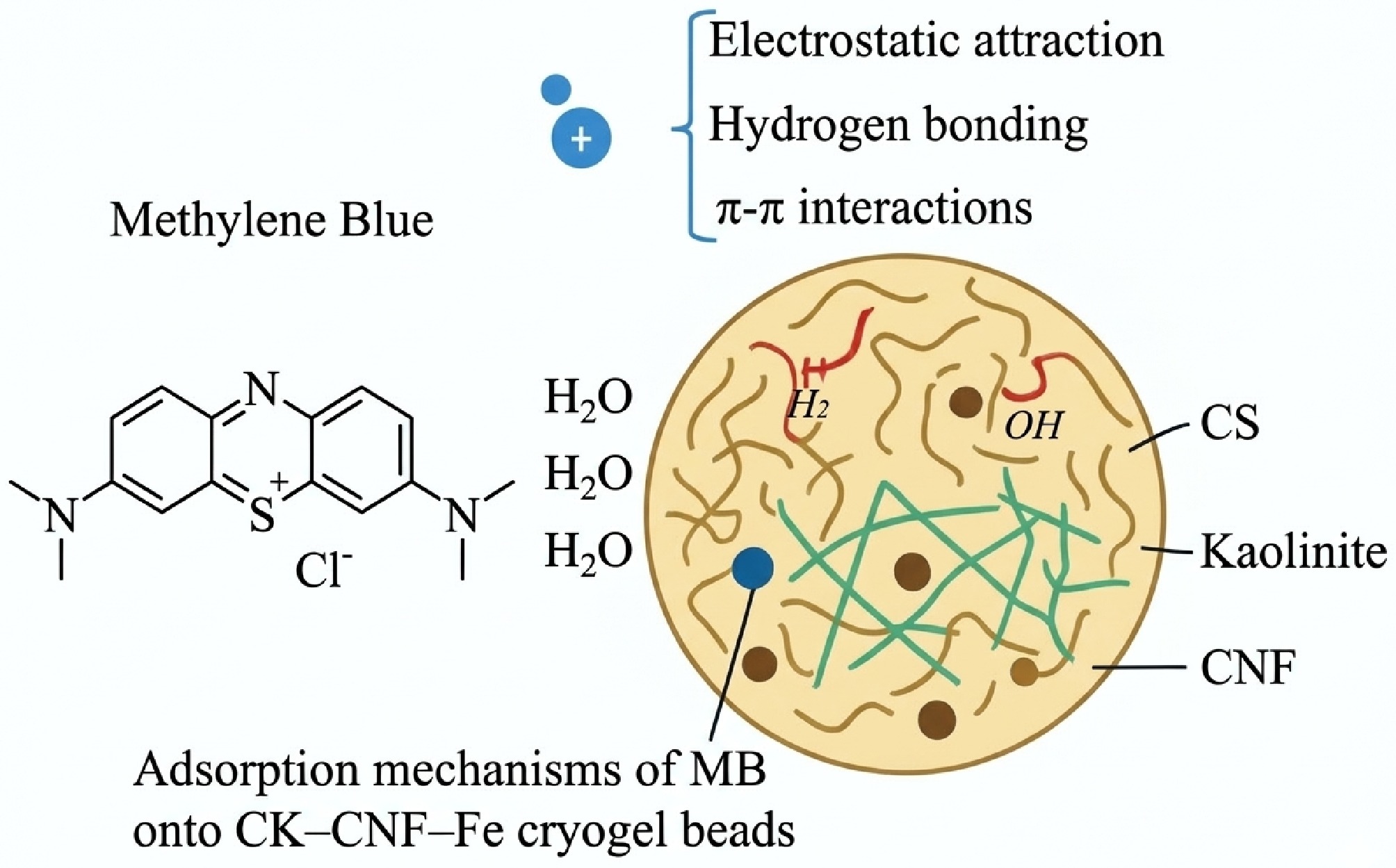
Figure 3.
Schematic illustration of the adsorption mechanisms of MB onto CK–CNF–Fe cryogel beads[112].
Table 4. Application of biochar composites in wastewater treatment
Biomass Synthesis method Composite material Application performance Ref. Pine sawdust One-step synthesis of magnetic biochar via hydrothermal carbonization combined with FeCl3 impregnation. Fe3O4/biochar composite Adsorption capacity of 98.5 mg/g for tetracycline; possesses magnetic separation properties. Ma et al.[113] Rice husk Pyrolysis after co-impregnation with MnCl2 and FeCl3. MnFe2O4/biochar composite Simultaneous removal of As(III) and Cd(II) from water; removal rates > 95%. Khan et al.[114] Peanut shell One-step pyrolysis combining ZnCl2 activation and LaCl3 modification. La-loaded porous biochar composite Deep removal of phosphate from water; adsorption capacity reached 102.3 mg P/g. Yi et al.[115] Orange peel Compositing orange peel biochar with magnetite (Fe3O4). Biochar-magnetite nanocomposite ~90% removal of ciprofloxacin within 60 min; magnetic property enables easy separation/recovery. Sharma et al.[116] Cotton Impregnation with FeCl3 solution followed by sulfidation using H2S gas. FeS/biochar composite High Hg(II) adsorption capacity of 287.4 mg/g in water. Nurlan et al.[117] Reed straw Loading Bacillus subtilis onto UV-radiation modified biochar. UV-modified biochar-Bacillus subtilis composite Saturated adsorption capacity for Cd(II) increased from 23.98 to 49.93 mg/g; particularly effective in weakly alkaline environments. Zhu et al.[118] Sludge Synthesis of manganese-modified composite for activating ferrate(VI). Mn-modified sludge biochar composite Rapid activation of ferrate(VI); 87.39% removal of sulfamethoxazole within 10 min; high removal efficiency for other sulfonamides. Deng et al.[119] Application for the treatment of dye pollutants
-
Dye pollutants in industrial wastewater typically possess complex aromatic structures and strong chemical stability, posing serious threats to aquatic ecosystems. Biochar, as a porous material with developed specific surface area, tunable pore structure, and rich surface functional groups, shows promising application prospects in dye wastewater governance. Its dye removal mechanisms mainly include physical adsorption, chemical adsorption, and catalytic degradation.
In practical applications, biochar feedstocks are diverse, covering agricultural waste, plant biomass, and municipal sludge. For example, a study prepared biochar from green seaweed and systematically evaluated its performance in remediating complex wastewater containing multiple Remazol dyes. Results indicated that under acidic conditions (pH = 2.0), the material exhibited high adsorption capacity for single dyes. When treating actual Remazol wastewater, despite some inhibition from the complex matrix, a decolorization rate of > 95% and total dye removal of > 77.5% were achieved within 240 min, demonstrating biochar's potential for the deep treatment of real wastewater[120].
Dyes and related additives are widely used in the textile, food, paint, paper, printing, and personal care industries. Statistics show that over 10,000 types of natural and synthetic dyes are produced globally each year, with a total output exceeding 7 × 105 t[121]. These pollutants feature complex chemical structures, high environmental stability, and poor biodegradability, becoming major sources of water pollution. Their discharge not only damages aquatic ecology, but also causes human health issues such as dermatitis, rhinitis, allergic reactions, and even cancer, through direct or indirect contact.
Traditional wastewater treatment processes mostly rely on chemical coagulation and biodegradation, making it difficult to achieve complete dye removal. Therefore, developing efficient and environmentally friendly advanced treatment technologies is critical. In this context, biochar is viewed as a material with high potential for dye pollution control due to its renewable feedstocks, flexible preparation, and excellent adsorption/catalytic properties[122]. Multiple studies have utilized various feedstocks to prepare biochar suitable for dye adsorption, such as agricultural residues[123], plant biomass[124], and municipal sludge[125]. Chen et al.[126] evaluated the adsorption performance of macroalgae-derived biochar for malachite green, crystal violet, and Congo red. They found that adsorption capacity for positively charged dyes increased with rising pH, primarily attributed to surface electrostatic attraction. Notably, the maximum adsorption capacity for malachite green reached 5,306 mg/g, benefiting from its highly developed pore structure and unique surface chemistry.
Dyes widely used in the textile, paper, and printing industries are a class of refractory organic pollutants. They possess high solubility and complex aromatic ring structures, remaining highly stable under natural conditions. Dye wastewater not only reduces water light transmittance due to high visibility, leading to hypoxia for aquatic organisms, but its aromatic-rich components also carry biotoxicity, further increasing aquatic ecological risks. Facing such complex pollution, seeking cost-effective and environmentally friendly treatment solutions has become urgent.
Although biochar can effectively adsorb pollutants in water, the inherent limitations of pristine biochar restrict its practical application. Consequently, biochar-based composites have received significant scholarly attention in recent years. Lu et al.[111] used biochar derived from pyrolyzed waste walnut shells as a carrier to successfully prepare a series of TiO2/biochar composite catalysts via hydrolysis for methyl orange degradation. Photocatalytic activity tests showed that the catalytic activity of this material surpassed that of pure TiO2. Its decolorization and mineralization rates for methyl orange reached 96.88% and 83.23%, respectively, attributed to the synergistic effect between biochar and TiO2. Inyang et al.[110] prepared modified biochar by dip-coating hickory or bagasse biomass surfaces with a carbon nanotube suspension, followed by slow pyrolysis at 600 °C in a nitrogen environment. The modified biochar enhanced pollutant adsorption because the surfactant effectively inhibited carbon nanotube agglomeration, promoting the distribution and stabilization of individual carbon nanotube particles on the biochar surface. The removal rates for heavy metal Pb and the dye sulfapyridine (SPY) from wastewater reached 86% and 71%, respectively, far exceeding the effects of pristine biochar.
Application for the removal of heavy metals
-
In recent years, biochar has been widely demonstrated to effectively adsorb heavy metal ions from polluted water bodies due to its large specific surface area, excellent pore structure, and abundant surface functional groups[127,128]. These characteristics enable multiple adsorption mechanisms such as ion exchange, surface complexation, and electrostatic attraction, along with physical adsorption and surface precipitation, as systematically illustrated in Fig. 4.
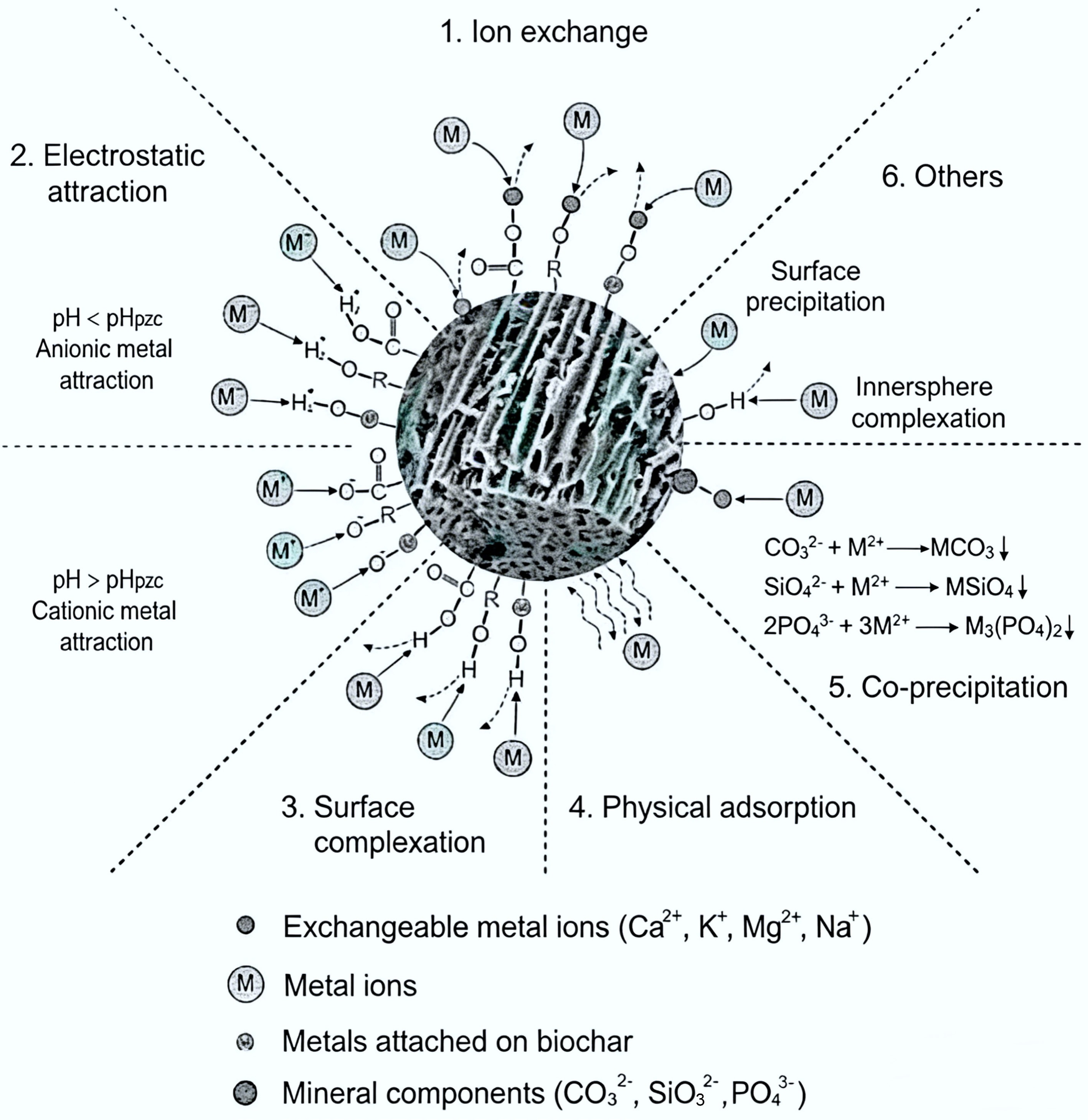
Figure 4.
Adsorption mechanism of biochar[129].
Adsorption capacity of biochar for heavy metals is mainly influenced by feedstock type, pyrolysis conditions, pretreatment, and modification methods. These factors collectively regulate key properties such as pore distribution, specific surface area, pH, surface functional group type/quantity, and cation exchange capacity[130,131]. Liu & Zhang[132] compared the Pb(II) adsorption behavior of biochar derived from rice husk and pine wood, finding that pine biochar's capacity was approximately double that of rice husk biochar, indicating that feedstock type significantly impacts performance. Inyang et al.[133] further pointed out that cationic nutrients (e.g., sodium, potassium, magnesium) enriched in feedstocks like oak and pine contribute to enhancing biochar's cation exchange capacity, thereby improving Pb(II) adsorption via ion exchange mechanisms. Additionally, biochar derived from animal wastes such as poultry litter (including bedding, feed residues, and feathers) and dairy manure typically has high ash content and is rich in inorganic elements[134,135], which can effectively immobilize heavy metals through precipitation and ion exchange[136].
Pyrolysis temperature is another critical regulatory factor, significantly affecting biochar's physical structure and surface chemistry[137]. Generally, as pyrolysis temperature increases, biochar aromatization enhances, pore structure further develops, and specific surface area increases, but the number of surface oxygen-containing functional groups and CEC usually decrease[12,13]. Shi et al.[138] found that increasing pyrolysis temperature could enhance the lead adsorption performance of rice husk biochar. Another study systematically compared the adsorption of Hg2+, Cd2+, and Pb2+ on corn stover biochar prepared at 300 (BC300), 400 (BC400), and 500 °C (BC500), finding that BC400 exhibited optimal heavy metal removal due to having the most abundant oxygen-containing functional groups[139].
Heavy metals are non-biodegradable and accumulate in organisms through the food chain. Due to their toxicity, carcinogenicity, and persistence, which pose serious threats to human health, heavy metal removal is a key focus for biochar composites in wastewater purification. Compared to pristine biochar, introducing nano-metal oxides/hydroxides, magnetic iron oxides, and functional nanoparticles onto the biochar surface allows for direct interaction with heavy metals, significantly enhancing adsorption capacity.
For example, Wang et al.[140] synthesized magnetic biochar by pyrolyzing a mixture of natural hematite and pine biomass. Characterization revealed that compared to unmodified biochar, the hematite-modified biochar not only possessed stronger magnetism but also exhibited superior arsenic removal from aqueous solutions. Moreover, the magnetized biochar could be easily separated by external magnets, making it widely applicable for arsenic removal. Similarly, Zhang et al.[141] prepared chitosan-modified magnetic bamboo charcoal (MBB) for the detoxification of Cr(VI) in sewage. Results showed that chitosan modification provided more active adsorption sites on the MBB surface, enhancing Cr(VI) removal. At 25 °C, the maximum adsorption capacities of MBB and CMBB for hexavalent chromium were 75.8 and 127 mg/g, respectively.
Furthermore, loading different functional nanoparticles simultaneously onto the same biochar can create multifunctional materials with synergistic effects for heavy metal removal. Yan et al.[142] successfully synthesized magnetic biochar/ZnS composites by depositing ZnS nanocrystals on the magnetic biochar surface in a polyol solution. The resulting composite could be easily separated from the solution due to its excellent superparamagnetic properties and exhibited a maximum Pb(II) adsorption capacity of 367.65 mg/g, which is ten times higher than reported capacities for magnetic biochar. Fan et al.[143] prepared a composite embedded with nano zero-valent iron (nZVI) via one-step pyrolysis of a biomass sawdust and Fe2O3 mixture. The surface Fe species could be oxidized to amorphous FeOOH, adsorbing heavy metal As via adsorption or co-precipitation.
Application for the treatment of nitrogen and phosphorus nutrients
-
Eutrophication is an ecological issue triggered by the excessive input of nutrients such as nitrogen and phosphorus into water bodies[144], causing far-reaching and multi-level harm. The surge in nutrients stimulates the proliferation of wild algae (e.g., cyanobacteria, green algae), forming 'algal blooms' that block photosynthesis and cause underwater plant death. Simultaneously, some cyanobacteria produce algal toxins that can accumulate through drinking water sources or aquatic products, directly endangering human health and potentially causing liver and nervous system diseases. Therefore, controlling ammonia nitrogen concentration in water bodies has become an effective means to address eutrophication[145].
Removing nitrogen and phosphorus from wastewater is key to controlling eutrophication. Current technologies include adsorption, chemical precipitation, and biological treatment. Among these, biochar, as an efficient adsorbent, shows unique potential for treating nitrogen and phosphorus nutrients.
Application for the removal of phosphorus
-
Phosphorus removal efficiency of biochar is significantly affected by feedstock type and surface charge. Jung et al.[146] showed that peanut shell biochar pyrolyzed at 700 °C had a significantly higher adsorption capacity for PO4-P (2.0 mg/g) than biochars from oak, soybean, and bamboo at the same temperature. However, pristine biochar generally has limited phosphorus adsorption capacity due to its typically negatively charged surface, which causes electrostatic repulsion with phosphate anions. For example, Reddy et al.[147] reported that wood biochar removed less than 50% of phosphorus from rainwater. This limitation has prompted researchers to develop various modified biochars to specifically enhance phosphate affinity.
Wang et al.[148], aiming for the resource utilization of wetland plants, prepared modified calamus biochar composites via pyrolysis with NaOH pretreatment and FeSO4 as a modifier. The composite showed improved hydrophilicity and polarity, with a significant increase in total specific surface area and micropore volume. Optimal adsorption and phosphorus removal were achieved at a pyrolysis temperature of 673 K and a modifier concentration of 1.0 mol/L, with a phosphorus removal rate and equilibrium adsorption capacity of 98.48% and 24.62 mg/g, respectively.
Application for the removal of nitrogen
-
Nitrogen in water bodies mainly exists as ammonium (NH4+) and nitrate (NO3−), and biochar exhibits different removal mechanisms and effects for each.
Biochar can effectively adsorb NH4+ from water via its microporous structure and cation exchange capacity. Meng et al.[149] used a composite biochar prepared from co-fermentation products to remove up to 95.9% ± 3.4% of NH4+-N, attributed to its larger specific surface area and calcium enrichment. Xu et al.[150] developed an MgO-biochar, which also showed good adsorption performance for ammonium in urine (47.5 mg/g).
The adsorption capacity of pristine biochar for negatively charged NO3− is usually weak. This can be effectively improved by metal-loaded modification. Zhang et al.[87] found that the NO3− adsorption capacity of various biochars modified with magnesium oxide (MgO) was significantly enhanced due to the increased specific surface area provided by MgO nanosheets. Wang et al.[148] further demonstrated that lanthanum-modified oak sawdust biochar showed a significant increase in nitrate adsorption capacity from 2.02 to 22.58 mg/g, mainly attributed to the increase in alkaline functional groups on the modified surface. Yu et al.[151] obtained Prussian blue/biochar composites by combining Prussian blue with modified biochar. The composite could selectively adsorb ammonia nitrogen in wastewater through coordination, achieving > 95% removal and a maximum adsorption capacity of 24.4 mg/g at pH 8, which is 101.3% higher than unmodified biochar. Simultaneously, a Fenton oxidation system constructed using H2O2 could achieve efficient organic matter degradation and ammonia nitrogen adsorption concurrently, showing potential for treating multi-component eutrophic wastewater.
In summary, biochar and its composites have demonstrated significant effectiveness in removing traditional pollutants such as dyes, heavy metals, and nutrients through mechanisms like adsorption, ion exchange, and co-precipitation. However, with changes in industry and lifestyle, large quantities of emerging contaminants are entering the water environment. These possess different molecular structures and environmental behaviors, posing new requirements for purification materials. Therefore, the following section turns to discuss the latest progress, unique mechanisms, and challenges of biochar composites in addressing these novel environmental challenges.
-
As water remediation research deepens in traditional fields like heavy metals[152], organic dyes[153], and pesticides[154], a series of new environmental challenges have emerged. Industrial development and lifestyle changes have led to the continuous entry of emerging contaminants such as antibiotics[155], per- and polyfluoroalkyl substances (PFAS)[156], and MPs[157] into the water cycle. These substances, typically characterized by low environmental concentration, high biotoxicity, stable chemical structure, and resistance to natural degradation[158], pose severe tests for traditional water treatment technologies and have garnered global attention due to their potential ecological and health risks. The 'List of Key Controlled New Pollutants' released by China's Ministry of Ecology and Environment in 2023 is a significant response to such risks.
Therefore, this chapter focuses on biochar and its composites designed for emerging contaminant removal, systematically elaborating on their efficacy, analyzing their microscopic mechanisms, and exploring current challenges and future directions in this field.
Application for the treatment of antibiotics
-
As a widely present class of emerging contaminants, antibiotic residues in the environment pose severe threats to ecosystems and public health. These primarily include sulfonamides, tetracyclines, and fluoroquinolones. Although indispensable in medicine, antibiotics have low metabolic rates in the human body, with approximately 70%–90% excreted unchanged into the environment[159]. These residues not only disrupt the balance of aquatic microbial communities, but also promote the generation and spread of antibiotic-resistant bacteria (ARB) and antibiotic resistance genes (ARGs)[160,161]. Long-term exposure to antibiotic-containing drinking water may directly or indirectly lead to drug resistance in human pathogens[162]. Furthermore, most antibiotic molecules contain stable aromatic or heterocyclic structures, making them difficult to degrade naturally or remove thoroughly via conventional technologies[65]. Therefore, developing efficient treatment technologies to eliminate antibiotics from water is crucial for ensuring ecological safety and human health.
Adsorption, due to its simplicity and mild conditions, is a foundational method for removing organic pollutants. For instance, Pham et al.[163] prepared porous biochar from macadamia nut shells via pyrolysis. Its high specific surface area and rich mesoporous structure effectively adsorbed ciprofloxacin and sulfamethoxazole via a pore-filling mechanism. However, adsorption only achieves phase transfer of pollutants without destroying their stable molecular structures, posing risks of difficult regeneration and secondary pollution. Consequently, research focus has shifted toward catalytic degradation technologies capable of completely mineralizing antibiotics. The core involves transforming biochar from an adsorbent carrier into a catalytic matrix by introducing highly active sites into the porous carbon skeleton to construct heterogeneous catalytic systems.
Current mainstream modification mechanisms focus on two directions. First, constructing semiconductor heterojunctions for photocatalytic degradation. Compositing semiconductors like graphitic carbon nitride (g-C3N4) and titanium dioxide (TiO2) with biochar leverages biochar's superior electron conductivity and large surface area to promote the separation and migration of photogenerated carriers, thereby enhancing photocatalytic efficiency[164,165]. Xue et al.[164] constructed an S-scheme FeOOH/Bi2MoO6 heterojunction supported on biochar. In the presence of visible light and H2O2, this material coupled photocatalysis with Fenton reactions, achieving a tetracycline degradation rate of 97% within 40 min, maintaining high efficiency across a wide pH range (3–11). The Biochar@ZnFe2O4/BiOBr Z-scheme heterojunction developed by Chen et al.[165] demonstrated that biochar acts as an electron mediator, significantly enhancing interfacial electron migration from BiOBr to ZnFe2O4 and effectively separating photogenerated electrons and holes.
The second direction is introducing transition metal sites to activate persulfate-based advanced oxidation processes (AOPs). By loading nano zero-valent iron, iron oxides, or species like cobalt and manganese onto the biochar surface, active centers are formed to activate PMS or peroxydisulfate (PDS). This generates strong oxidizing species such as sulfate radicals (SO4−) and hydroxyl radicals (•OH), achieving non-selective deep oxidative degradation of antibiotics[166]. Biochar's graphitized structure provides free-flowing electrons, and its large surface area and abundant functional groups (e.g., C=O) can also disrupt the O–O bond of oxidants via electron transfer, synergistically promoting radical generation[167,168]. Shi et al.[169] found that when pyrolysis temperature was raised to 900 °C, the coupling of graphitic nitrogen and graphitic-like structures in nitrogen-doped biochar formed optimal active sites for activating PDS to degrade sulfamethazine. Tran et al.[170] utilized chitosan-crosslinked biochar to effectively remove sulfamethoxazole via multiple mechanisms, including surface positive charge (electrostatic attraction), π–π interactions, hydrogen bonding, and pore filling.
In summary, the application of biochar and its composites in antibiotic removal shows a clear evolutionary path from adsorption to catalytic degradation. The graphitized structure of biochar serves as the material basis for providing catalytic active centers, while elemental doping further introduces functional groups and regulates electron transfer. Its high conductivity also makes it an ideal matrix for separating photogenerated charges. Future research should dedicate efforts to screening more biomass precursors for preparing self-doped biochar, reducing reliance on chemical additives during synthesis, and systematically evaluating long-term stability in practical applications based on green chemistry principles to prevent secondary pollution issues such as metal ion leaching.
Application for the treatment of PFAS
-
PFAS are considered one of the most difficult emerging contaminants to treat due to their extreme chemical stability and potential bioaccumulation. They are frequently detected in aquatic environments, including drinking water, surface water, groundwater, and wastewater[155]. The carbon-fluorine (C–F) bond in PFAS molecules is one of the strongest covalent bonds in organic chemistry, imparting exceptional chemical and thermal stability to these compounds[171]. Due to this persistence, PFAS possess biomagnification potential and can induce oxidative stress, which is linked to heart disease, central nervous system impacts, and cancer risks. Therefore, developing effective technologies to remove PFAS from water environments is crucial.
PFAS are classified into long-chain and short-chain categories based on carbon chain length (primarily defined by the tendency of radicals to break C–C bonds)[172]. Short-chain PFAS possess higher water solubility and are more prevalent in surface water. Their environmental persistence and bioaccumulation may even exceed those of some long-chain counterparts[173]. Adsorption is considered one of the most viable treatment technologies currently because it effectively captures PFAS molecules without generating harmful degradation intermediates. Biochar, as an environmentally friendly adsorbent, demonstrates significant potential in this field[155,174]. Zhang et al.[175] prepared a high-performance acid-modified biochar adsorbent using sludge feedstock. The theoretical maximum adsorption capacities for perfluorooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA) reached 72.17 and 45.88 mg/g, respectively. The study inferred that the adsorption mechanism was primarily attributed to the synergy of three aspects: electrostatic attraction between the positively charged biochar surface and deprotonated PFAS, hydrogen bonding between PFAS anions and surface functional groups, and hydrophobic interactions with long-chain PFAS. Krahn et al.[176] also indicated that sewage sludge-derived biochar exhibited higher PFAS removal efficiency than woody biochar or commercial activated carbon. This was attributed to its higher specific surface area and pore volume, making it more suitable for water and soil remediation. To further enhance performance, Deng et al.[177] developed a novel biochar composite based on sulfobetaine polymers, featuring both quaternary ammonium groups and hydrophobicity. The maximum adsorption capacities for PFOS, PFOA, perfluorobutane sulfonate (PFBS), and perfluorobutanoic acid (PFBA) reached 634, 536, 301, and 264 mg/g, respectively, with excellent regenerability. Such superior performance was attributed to surface sulfonate and amide groups, with possible mechanisms involving electrostatic interaction, hydrophobic interaction, pore diffusion, and complexation. Yu et al.[151] synthesized polypyrrole-functionalized biochar composites via a novel in situ self-sacrificing method. This method utilized Fe3O4 to release Fe3+ in situ as an oxidant, guiding the uniform polymerization of pyrrole monomers on the biochar surface. This overcame polypyrrole agglomeration while imparting a positive charge to the composite surface. The material achieved adsorption capacities of 3.89 and 1.53 mmol/g for short-chain PFBA and PFBS, and 2.55 and 1.22 mmol/g for long-chain PFOA and PFOS, respectively, and could be reused at least five times.
In summary, the adsorption mechanism of biochar for PFAS involves the synergy of hydrophobic interactions, electrostatic attraction, π–π stacking, hydrogen bonding, and pore diffusion (Fig. 5). The dominant mechanism depends on biochar surface properties[151,177]. Although traditionally prepared biochar shows good PFAS adsorption, limitations remain, including limited efficiency for short-chain PFAS and narrow effective pH windows. Based on these mechanisms, targeted modification can significantly enhance performance: heteroatom doping introduces more functional groups, enhancing electrostatic interactions and hydrogen bonding, which favors hydrophilic short-chain PFAS adsorption; acid treatment optimizes surface charge to strengthen electrostatic attraction while regulating hydrophobicity; and steam activation creates abundant mesopores, providing ample adsorption sites and space. Through modification, biochar is expected to achieve high capacity, rapid equilibrium, and efficiency across a wider pH range, making it suitable for complex real-world waters. Future research should prioritize cost control alongside high performance to promote engineering translation.
Figure 5.
Possible adsorption mechanism of biochar materials for per-and polyfluoroalkyl substances removal.
Application for the treatment of MPs
-
MPs refer to plastic particles smaller than 5 mm. They can be classified into primary MPs, which are directly discharged into the environment, and secondary MPs, which form via the fragmentation of larger plastic waste through biotic or abiotic processes, such as UV irradiation and physical abrasion[178]. Statistics indicate that nearly 80% of MPs originate from land-based anthropogenic activities, such as washing synthetic fibers, using personal care products, and the wear of tires and plastic products. These particles subsequently enter water bodies and soil via surface runoff or wastewater, gradually infiltrating the food chain[179]. Wastewater treatment plants, acting as significant sinks for MPs, can also serve as pathways for their diffusion into aquatic ecosystems. It is estimated that approximately 4.8 to 12.7 million tons of plastic enter water bodies globally each year, with nearly 150 million tons of plastic waste accumulated in oceans[180,181]. This poses severe threats to ecosystems and human health, making the development of efficient MP removal technologies urgent.
Since the complete catalytic degradation of MPs involves complex multi-step radical processes requiring stringent catalyst properties, and incomplete mineralization may generate smaller secondary pollutants, adsorption is considered a promising removal technology due to its energy efficiency, environmental friendliness, and lack of harmful degradation byproducts[182]. Among adsorbents, biochar demonstrates unique advantages over sponges and certain metal-based materials in removing aquatic MPs, owing to its porous structure, abundant surface sites, and cost-effectiveness[183]. Its removal mechanisms primarily include hydrophobic interactions, electrostatic attraction, π–π stacking, hydrogen bonding, and pore filling, sharing similarities with the adsorption mechanisms of PFAS[184,185].
Metal modification is a common strategy to enhance biochar's MP adsorption performance. Wang et al.[186] synthesized magnetic and Mg/Zn-modified biochars for removing polystyrene microspheres (1 μm). Experiments showed removal efficiencies of 94.81%, 98.75%, and 99.46% for magnetic biochar, Mg-modified, and Zn-modified magnetic biochars, respectively. This high efficiency was attributed to the combined effects of electrostatic attraction and surface chemical bonding. Babalar et al.[187] loaded Fe3O4 and zeolite onto the biochar surface to prepare a composite (PMACZ). It achieved a maximum adsorption capacity of 736 mg/g for 2 µm MPs and could be reused at least 4 times, with π–π interactions and electrostatic attraction being the primary mechanisms. Zhang et al.[141] further confirmed that iron-modified (FeBC) and iron/zinc co-modified (Fe/ZnBC) biochars achieved removal rates of 96.24% and 84.77% for polystyrene MPs, respectively. Performance remained stable under acidic to neutral conditions (pH 3–7); however, at pH 9–11, removal efficiency dropped significantly due to enhanced surface electrostatic repulsion.
In addition to metal modification, surfactant modification effectively improves biochar performance. Parashar et al.[188] modified rice husk-based magnetic biochar with cetyltrimethylammonium bromide (CTAB). The resulting MBC-CTAB achieved an MP adsorption capacity of 247 mg/g within 3 min. This was mainly attributed to CTAB-introduced amino groups enhancing electrostatic and hydrophobic interactions, hydrogen bonding, and pore filling effects. However, optimal adsorption was only achieved in a slightly acidic environment (pH = 4), indicating the need to broaden its applicable pH range.
In summary, strategies such as metal loading or surfactant modification can effectively regulate biochar surface properties, thereby enhancing adsorption capacity and selectivity for MPs. Future research should explore novel biochars capable of efficiently adsorbing multiple MPs simultaneously across a wider pH range, while considering preparation costs to assess practical feasibility. Furthermore, computational tools such as artificial intelligence, density functional theory (DFT), and molecular dynamics (MD) simulations hold promise for guiding the rational design of more efficient adsorbents at the molecular level. Figure 6 provides a schematic overview of these complex molecular interactions (e.g., Van der Waals forces and complexation) that such computational models aim to optimize.
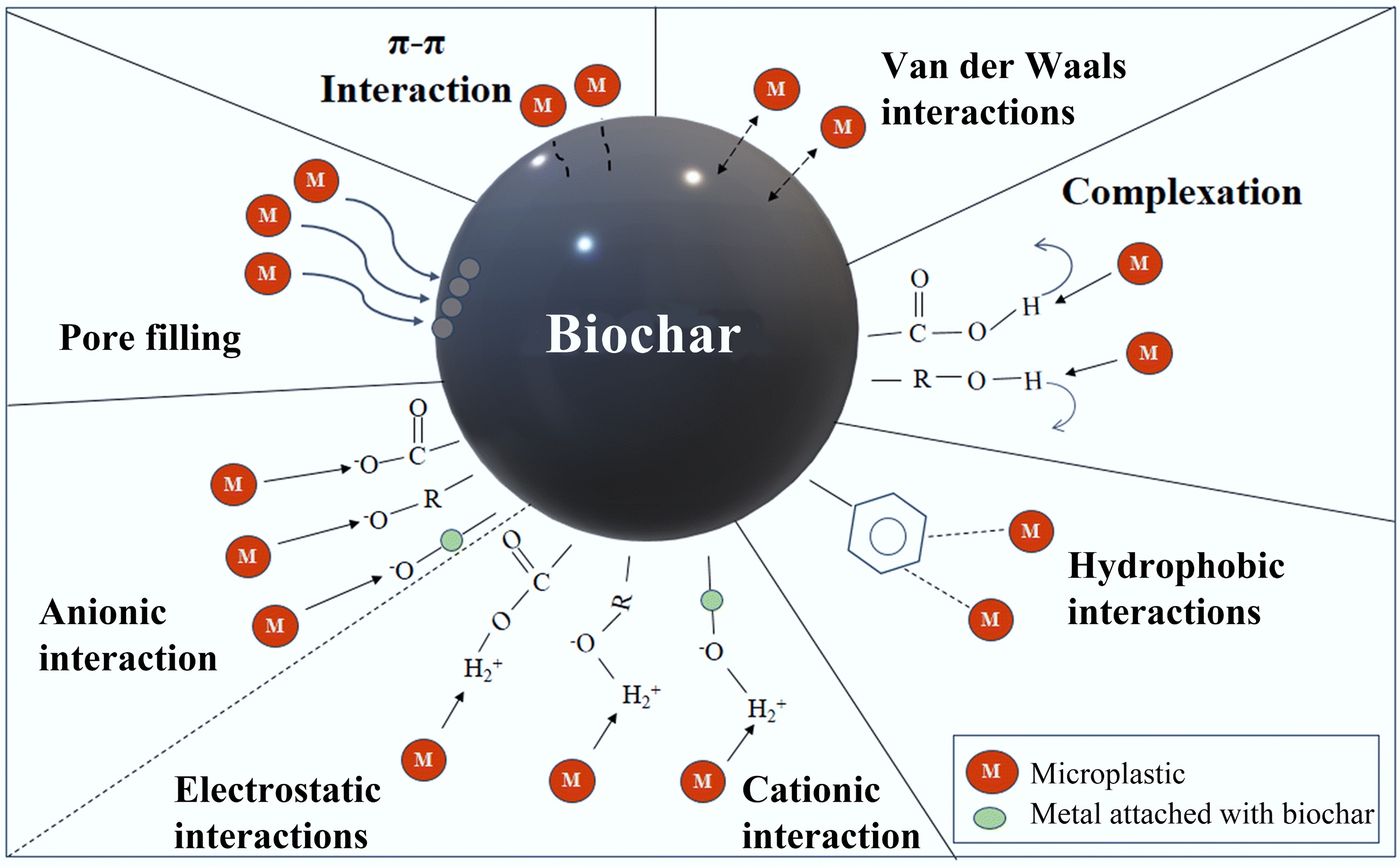
Figure 6.
Adsorption mechanisms underlying the removal of MPs by the biochar[189].
-
LCA, as a systematic method following ISO standards, provides a scientific benchmark for evaluating the environmental sustainability of biochar composites by quantifying energy consumption, resource utilization, and emissions throughout the product's entire life cycle[190]. Its standardized process covers four stages: goal and scope definition, inventory analysis, impact assessment, and interpretation[191]. In the biochar field, LCA is typically conducted using software tools like SimaPro or OpenLCA, integrating background data from databases such as Ecoinvent and Agri-footprint with laboratory-specific foreground data[192].
A typical study must first define clear system boundaries. For instance, Shaheen et al.[193], when comparing palm waste biochar with activated carbon, defined boundaries covering the entire process from feedstock collection and transport, pyrolysis, and modification, to application and final disposal. Case studies indicate that environmental performance varies significantly across materials and process routes. Using the removal of 1 kg of pollutant as the functional unit, ZnCl2-modified biochar exhibited a lower Global Warming Potential (1.58 kg CO2 eq). Comparative studies reveal that biochar derived from waste biomass often possesses a lower comprehensive environmental footprint than traditional activated carbon[194]. However, the environmental cost behind performance enhancement cannot be ignored. Shaheen et al.[190] explicitly noted that complex processes combining metal loading and acid modification for enhanced functionality resulted in significantly higher Global Warming Potential and Cumulative Energy Demand compared to single modification strategies. This highlights the potential surge in carbon costs caused by engineering modification. Furthermore, the use of chemicals during modification introduces potential ecotoxicity risks. Toxicity impact assessments in LCA show that alkaline modifiers generally exhibit higher toxicity to aquatic environments than acidic ones. This necessitates considering safer chemical alternatives during the material design phase, such as replacing sodium hydroxide with potassium hydroxide or implementing process effluent recycling[195].
Current LCA research in this field still faces challenges regarding methodological unification, primarily in functional unit selection, system boundary setting, and the uncertainty involved in scaling laboratory data to industrial simulations. These inconsistencies hinder direct comparisons between studies. Therefore, future development should focus on establishing a standardized LCA framework for biochar composites, enhancing the transparency and consistency of full-chain inventory data, and prioritizing LCA guidance during the green design phase. Selecting low-impact feedstocks and low-carbon modification processes ensures that the environmental benefits of high-performance biochar composites are not offset by excessive resource and environmental costs throughout their life cycle, thereby achieving true synergy between water treatment technology and sustainable development.
-
Biochar and its composites have garnered widespread attention in environmental remediation due to their superior physicochemical properties, such as high specific surface area, rich pore structure, and diverse surface functional groups. They are extensively applied in soil improvement, pollutant adsorption, and wastewater treatment[196]. However, any performance gain from engineering design may be accompanied by potential environmental costs. These risks are closely linked to the material's core functions and must be prudently assessed from a whole-life-cycle perspective[197]. As research deepens and applications expand, these potential secondary environmental risks are becoming increasingly apparent. Systematic recognition of these risks is the cornerstone for promoting truly sustainable material application.
Biochar prepared from feedstocks like sludge and livestock manure may contain heavy metals inherent to the raw material. During pyrolysis, these metals can further enrich in the biochar, reaching concentrations 1.5–2.3 times higher than in the feedstock[198]. When applied to the environment, these heavy metals may be released back into soil or water over time or with changing environmental conditions, causing secondary pollution. Additionally, biochar pyrolysis is accompanied by the generation of polycyclic aromatic hydrocarbons (PAHs). Relevant studies indicate that PAH generation increases exponentially with rising pyrolysis temperature: PAH content is approximately 120 μg/kg at 300 °C, rising sharply to 890 μg/kg at 700 °C[199]. As PAHs are carcinogenic, teratogenic and mutagenic organic pollutants, their presence undoubtedly increases the application risks of biochar. More complexly, the pyrolysis process generates environmentally persistent free radicals (EPFRs) with unique properties. EPFRs have long half-lives (> 30 d), allowing them to persist in the environment and increasing organism exposure risks. Simultaneously, EPFRs can induce the production of reactive oxygen species (ROS), leading to protein and DNA damage, which may potentially induce tumors and cardiovascular diseases.
Beyond the risks of biochar itself, the hazards of modification processes and introduced functional components cannot be ignored. For example, Kookana et al.[200] indicated that the addition of chemicals like potassium hydroxide, sodium hydroxide, hydrochloric acid, and sulfuric acid during composite preparation alters biochar pH, subsequently affecting soil/water environments and microbial growth/reproduction. This suggests that the environmental release and leaching of nano-components or chemical modifiers introduced for high activity is another explicit risk[201]. Wahab et al.[202] synthesized well-crystalline ZnO nanoparticles via soft chemistry and investigated their role in inducing oxidative stress in Cloudman S91 melanoma cells. Results showed that exposure to ZnO nanoparticles caused ROS production and apoptosis in cancer cells. These functional nano-components, characterized by small size and large specific surface area, may migrate over long periods in water bodies once detached from the biochar matrix. They can exert unique nanotoxicity through mechanisms like oxidative stress induction, with environmental behaviors that are complex and difficult to monitor or remediate[197].
After entering the environment, the long-term behavior and ultimate fate of biochar poses greater hidden dangers. On one hand, it interacts with existing environmental pollutants to form composite pollution, the toxicity of which often differs from that of single pollutants. Liu et al.[203] systematically studied the interaction between biochar and Cr or As, finding that redox-induced valence changes of metal ions were key to the combined cytotoxicity of biochar/Cr/As. Specifically, when Cr(VI) was adsorbed on biochar, 86.4% was reduced to the less toxic Cr(III). Conversely, As(III) was partially oxidized (up to 37.2%) to As(V), reaching equilibrium. Since heavy metal toxicity varies greatly with valence state, this change directly affects the complex's toxicity intensity. On the other hand, Azargohar et al.[204] found that steam-activated biochar exhibited toxicity to Daphnia, revealing potential direct biotoxicity of the material itself. More importantly, saturated or deactivated composites, if not planned for safe disposal (e.g., random landfilling), become composite reservoirs for pollutants and functional chemicals. They face the risk of pollutant re-release or soil/groundwater contamination via leachate under changing environmental conditions, effectively transferring and solidifying aqueous pollution into more intractable solid-phase pollution.
Although biochar has achieved excellent results in enhancing pollutant removal, research on its stability and biotoxicity remains weak. Therefore, to avoid secondary contamination and biological damage from toxic materials, systematic evaluation of their stability and biological effects in aqueous/soil environments is required[197]. Future research and application must drive a fundamental shift in material design paradigms, moving from pure performance optimization to synergistic performance-safety design. This requires prioritizing risk prevention: implementing green synthesis and safe substitution at the source, and prioritizing environmentally friendly, low-toxicity, or bio-based modifiers[205]. Structurally, efforts should focus on building robust chemical bonds (e.g., M–O–C covalent bonds) to enhance interfacial stability and fundamentally inhibit active component leaching, while pre-planning safe disposal or resource recovery pathways[206]. Finally, a cradle-to-grave LCA system must be established and adopted. This system should incorporate ecotoxicity testing, long-term leaching experiments, and environmental footprint analysis as essential dimensions for performance evaluation, thereby quantifying the true net environmental benefit[207]. A frontier direction is adopting the Safe and Sustainable by Design (SSbD) framework, systematically assessing and optimizing the balance between function, safety, and sustainability during the early design stages of material development[208].
In summary, true innovation in biochar composites lies in minimizing environmental risk as a core constraint parallel to performance optimization from the very beginning of design. Through systematic strategies of source prevention, process control, and end-of-pipe safety management, deep synergy between environmental remediation efficiency and long-term ecosystem health can be achieved for sustainable development[209].
-
Biochar-based composites have emerged as promising materials for wastewater treatment, showing clear advantages over pristine biochar in terms of adsorption capacity, selectivity, and catalytic activity. These improvements are mainly achieved through rational feedstock selection, controlled thermochemical conversion, and targeted engineering strategies, which enable the regulation of pore structure, surface chemistry, and redox properties. Existing studies demonstrate that biochar-based composites can effectively remove a wide range of conventional pollutants and emerging contaminants from wastewater through adsorption and catalytic mechanisms.
Despite these advances, several challenges still limit their large-scale application. Performance enhancement is often accompanied by increased material complexity, higher energy consumption, and potential environmental risks, such as secondary pollution and long-term stability issues. Moreover, the lack of standardized evaluation methods make it difficult to compare different modification strategies and assess their practical feasibility.
Future research should therefore focus on balancing treatment efficiency with environmental sustainability. Greater attention should be paid to green synthesis routes, low-energy modification strategies, and the long-term stability of biochar-based composites under realistic wastewater conditions. In addition, life-cycle assessment and risk evaluation should be systematically incorporated into material design to ensure net environmental benefits. Application-oriented design, combined with mechanistic understanding, will be essential to promote the transition of biochar-based composites from laboratory studies to practical wastewater treatment systems.
-
The authors confirm their contributions to the paper as follows: all authors contributed to the study conception and design; Qichen Hou and Xinjun Zhang wrote the original draft of the manuscript; Cui Wang and Bo Bai reviewed and edited the manuscript. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets used or analyzed during the current study are available from the corresponding authors upon reasonable requests.
-
This work is supported by the National Natural Science Foundation of China (52406178), China Postdoctoral Science Foundation (2025T180169 and 2024M752736), and the Young Talent Fund of Xi'an Association for Science and Technology (0959202513003).
-
All authors declare that they have no competing interests.
-
Full list of author information is available at the end of the article.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Wang C, Hou Q, Zhang X, Bai B. 2026. Preparation of biochar-based composites and application in removal of conventional and emerging pollutants from wastewater: performance enhancement, mechanisms, sustainability, and risk evaluations. Sustainable Carbon Materials 2: e020 doi: 10.48130/scm-0026-0015
Preparation of biochar-based composites and application in removal of conventional and emerging pollutants from wastewater: performance enhancement, mechanisms, sustainability, and risk evaluations
- Received: 27 November 2025
- Revised: 15 January 2026
- Accepted: 24 March 2026
- Published online: 13 April 2026
Abstract: Biochar derived from biomass waste has been widely investigated for wastewater treatment due to its porous structure and surface functionality. However, pristine biochar often exhibits limited adsorption capacity, low selectivity toward emerging contaminants, and poor separation and recovery performance, which restrict its practical application. To overcome these limitations, biochar-based composites have been extensively developed through controlled synthesis and functionalization strategies. Herein, we critically review and address recent progress in the preparation of biochar and biochar-based composites, with emphasis on feedstock selection, thermochemical conversion processes, and composite engineering approaches, including magnetic biochar, metal oxide- or hydroxide-modified biochar, and functional nanoparticle-loaded biochar. Additionally, the key physicochemical properties governing pollutant removal are discussed, and the adsorption and catalytic mechanisms involved in removing conventional pollutants and emerging contaminants from wastewater are analyzed. Notably, this review highlights the trade-offs between biochar performance enhancement and potential environmental risks, and emphasizes the importance of life cycle assessment and green design for achieving sustainable wastewater treatment applications.