-
The Megalurothrips usitatus, belonging to the family Thripidae (Thysanoptera), is a devastating pest of leguminous crops in tropical and subtropical regions. Its host range includes economically important legumes such as cowpea (Vigna unguiculata) and common bean (Phaseolus vulgaris). This pest is characterized by its broad host range adaptability, diverse modes of damage, and rapid evolution of pesticide resistance, making it a critical factor that limits the production of high-quality, high-yield legume crops[1−3]. Both adult and nymphal stages of M. usitatus feed on host flowers, tender leaves, and young fruits, utilizing their scraping-sucking mouthparts. Through abrading the plant epidermis and extracting cellular fluid, they directly undermine the structural integrity and physiological functionality of photosynthetic tissues, thereby inducing silvery-white scarring on leaves, chlorophyll degradation, and substantial reduction of photosynthetic capacity[4,5]. Damage to floral structures frequently precipitates petal deformation and pollination failure, whereas feeding on young fruitlets results in persistent pitting or fruit deformation, including pod curvature and surface roughening in cowpea. Consequently, severe damage is inflicted on cowpea crops, resulting in a significant decline in quality[6−8]. Furthermore, this insect exhibits diminutive body dimensions, with adults measuring approximately 1.5–2.0 mm and nymphs merely 0.8–1.2 mm in length. The species demonstrates pronounced thigmotaxis and frequently conceals itself within the floral structures of leguminous plants, encompassing pistils and petal folds, as well as beneath the calyx of young fruitlets[9]. This ecological behavior considerably exacerbates the difficulty of field control. Cowpea, a leguminous vegetable esteemed for its palatability and nutritional value, occupies considerable importance in human dietary patterns. Hainan Province, situated in tropical regions and characterized by the absence of winter, abundant solar radiation, and copious precipitation, provides environmental conditions exceptionally favorable for cowpea cultivation. Consequently, cowpea has become an essential cash crop vital to the economic livelihoods of local farming communities[10,11]. The intensification of large-scale cowpea production in Hainan has, however, provided M. usitatus with a stable and conducive habitat for population proliferation. This development has correspondingly precipitated a substantial escalation in chemical insecticide dependence. Historically, pyrethroid insecticides—specifically permethrin and deltamethrin—have been extensively deployed against M. usitatus owing to their superior efficacy and relatively benign mammalian toxicity profiles. However, the rate of resistance development in this pest population has considerably surpassed initial projections[12−15].
Voltage-gated sodium channels (VGSCs) are extensively distributed throughout the nervous systems of both vertebrate and invertebrate organisms, serving as fundamental molecular substrates for the transduction of electrical signals within nerve cells. The functional integrity of these channels directly determines the fidelity and efficiency of neuronal signal propagation[16]. As specialized membrane protein channels possessing critical physiological roles, VGSCs additionally constitute the principal target site for various animal-derived neurotoxins, encompassing tetrodotoxin, scorpion toxins, and batrachotoxin[17]. Moreover, they function as the primary molecular targets of classical insecticides, including pyrethroids and dichlorodiphenyltrichloroethane (DDT)[18]. The α-subunit comprises four homologous domains (Domains I–IV), each containing six transmembrane segments (S1–S6). Among these transmembrane segments, the S4 segment functions as the voltage sensor, whereas the S5–S6 segments collectively constitute the ion-conducting pore of the channel[19−21]. Pyrethroid insecticides exert their insecticidal activity by binding to two distinct regions within the VGSC[22,23]. This binding interaction destabilizes the channel's open conformation, suppresses channel inactivation, and consequently permits sustained sodium ion influx. The resultant perturbation of neuronal electrical signaling ultimately produces paralysis and lethality in insects[24,25]. Amino acid substitutions within the sodium channel can alter its three-dimensional architecture, thereby reducing the binding affinity of pyrethroids to their target site. This mechanistic basis of resistance is designated as knockdown resistance (kdr). Presently, numerous kdr mutations conferring elevated resistance levels have been identified across diverse insect species, with these mutations predominantly concentrated within Domains II and IV of the VGSC[26,27].
Pyrethroid insecticides represent synthetic derivatives produced through chemical modification of bioactive constituents derived from natural pyrethrins. According to the presence or absence of an α-cyano group within their molecular structure, they are categorized into two distinct classes: Type I pyrethroids, lacking an α-cyano group, exemplified by permethrin, bifenthrin, etc.; and Type II pyrethroids, containing an α-cyano group, represented by deltamethrin and λ-cyhalothrin, etc[28,29]. Empirical evidence demonstrates that Type I pyrethroids exert their toxic effects by binding to specific sites on channel proteins, disrupting normal transmembrane sodium ion transport, and eliciting repetitive electrical discharge (i.e., repetitive firing) in nerve axons. This aberrant electrophysiological activity precipitates sustained hyperexcitation of presynaptic nerve terminals, ultimately disrupting synaptic signal transmission[30]. Although both types of agents target VGSCs, the toxicological mechanisms of type II pyrethroids differ significantly from those of type I. Type II pyrethroids do not induce repetitive firing in neural tissues; instead, they prolong the duration of sodium currents. This prolongation results in sustained depolarization of the cell membrane, which hinders the generation and conduction of normal action potentials, thereby disrupting synaptic physiological function[20]. Mutations in VGSC conferring knockdown resistance (kdr) represent a principal focus of contemporary research in the field of insecticide resistance mechanisms[31]. Grounded in this mechanistic foundation, detection of kdr mutation frequencies at specific loci within insect VGSCs enables inference of the proportion of resistant individuals within a population, thereby facilitating indirect assessment of population-level resistance status and providing a molecular technical framework for early detection of insecticide resistance development[32].
Based on the above research background, this investigation seeks to elucidate the resistance evolution dynamics of M. usitatus in Hainan Province toward pyrethroid insecticides through systematic mutation site detection and resistance level assessment conducted over three consecutive years. Furthermore, by characterizing the complete-length VGSC gene sequence of this species and identifying resistance-associated mutation sites, the molecular mechanism underlying resistance formation is further clarified. By delineating the resistance evolution trajectory of M. usitatus to pyrethroids, this research endeavors to provide a theoretical framework and practical guidance for the scientifically-informed formulation of insecticide rotation strategies, judicious insecticide application, and retardation of resistance progression in field populations.
-
The susceptible strain (HK-S) was collected from Yunnan and reared on fresh cowpea without pesticide residues. Four field populations, designated as CM-R (CM2024, CM2025, CM2026), LD-R (LD2024, LD2025, LD2026), SY-R (SY2024, SY2025, SY2026), and LS-R (LS2024, LS2025, LS2026), were collected from Chengmai County, Ledong Li Autonomous County, Sanya City, and Lingshui Li Autonomous County, Hainan Province, China, between 2024 and 2026, respectively; detailed geographic coordinates are provided in Table 1. The susceptible strain (HK-S) was collected from Yunnan and reared on fresh, pesticide-free cowpea. All tested insect sources were reared under artificial climate conditions set at a temperature of 26 ± 1 °C, a relative humidity of 65% ± 5%, and a photoperiod of 16:8 h (light : dark); these rearing conditions were maintained from 2019 to 2025. The tested insecticides were as follows: deltamethrin (purity 100%), bifenthrin (purity 100%), and cypermethrin (purity 100%), all purchased from Beijing J&K Scientific Ltd (Beijing, China).
Table 1. Field collection information table of M. usitatus.
Strains Collection site Year Longitude and latitude Host CM-R Chengmai County, Hainan Province 2024−2026 N 20°20'31", E 110°30'11 Cowpea LD-R Ledong Li Autonomous County, Hainan Province 2024−2026 N 18°40'03", E 109°30'25" Cowpea SY-R Yazhou District, Sanya City, Hainan Province 2024−2026 N 18°23'05", E 109°9'44" Cowpea LS-R Lingshui Li Autonomous County, Hainan Province 2024−2026 N 18°31'42", E 110°1'35" Cowpea Bioassay
-
Technical-grade insecticides were initially dissolved in acetone to prepare stock solutions, and subsequently diluted with 0.1% Triton X-100 solution (Sigma, USA). Six to eight concentration gradients were established for each insecticide, and each concentration was tested with three mechanical replicates. In this study, bioassays against M. usitatus were conducted using the residual film method, with the detailed procedure described as follows: Pre-treatment of experimental materials: Fresh, pesticide-free cowpea pods were selected; only those with a diameter suitable for insertion into 1.5 mL centrifuge tubes were chosen. The selected pods were rinsed, air-dried, and cut into 0.6–0.8 cm segments. A 20-mesh insect-proof screen was cut into rectangles of approximately 2.5 cm per side and set aside. Chemical treatment: The pre-treated cowpea segments and insect-proof net pieces were immersed together in the corresponding insecticide solution for 5 min. They were then removed with disposable forceps, placed on absorbent paper, and air-dried in a fume hood. Additional 1.5 mL centrifuge tubes were prepared; a hole was drilled through the center of each tube cap with a high-temperature electric soldering iron to ensure air exchange. For each concentration, 1.5 mL of the corresponding insecticide solution was added to the tubes. After 1 h of soaking, the solution was discarded, and the tubes were drained of residual liquid and air-dried. Insect inoculation: Using a laboratory-patented aspirator, adults of the laboratory-reared susceptible strain and field-collected adults were separately transferred into the insecticide-treated centrifuge tubes. Each tube received 25 adults. Subsequently, the insecticide-treated cowpea segments were added, and the 20-mesh netting was finally secured with the tube cap to prevent the escape of test insects. The inoculated centrifuge tubes were placed horizontally in a controlled climate chamber set at 25 °C, 70% relative humidity, and a photoperiod of 16 h light : 8 h dark (L : D). Data recording and analysis: Adult mortality in each treatment group was scored 48 h after insecticide exposure. Statistical analysis of the experimental data was performed using PoloPlus 2.0 software.
Cloning of the VGSC gene from M. usitatus
-
Total RNA was extracted from adult M. usitatus using Trizol® Reagent (Ambion, USA) and reverse transcribed into cDNA with the PrimeScript™ II 1st Strand cDNA Synthesis Kit (TaKaRa, Japan). Specific primers MuNav-5(F)/3(R) (synthesized by Tsingke, Beijing, China; sequences are listed in Table 2) were designed, and the full-length VGSC gene was amplified using cDNA as the template. PCR products were examined by 1% agarose gel electrophoresis, purified and recovered using an Omega Bio-Tek kit (USA), ligated into the PGH19 vector, and then transformed into XL-10-gold competent cells (Biomed, Beijing, China). Following 90 min of shaker incubation, the cells were spread onto LA plates containing 10 mg/mL ampicillin and incubated inverted for 24 h. Single colonies were picked and subjected to colony PCR to eliminate false positives. Positive clones were then cultured for amplification, and plasmids were extracted using an Omega kit (USA). The extracted plasmids were verified by restriction digestion with Hind III-HF enzyme (NEB, Beijing, China) and subsequently sent to Tsingke Biotechnology Co., Ltd for sequencing.
Table 2. Primers for the full-length application and mutation frequency detection of VGSC in M. usitatus.
Primer name Primer sequence Usage MuNav-5(F) TTGGCAGTCAATTCCCCGGGGCCACCATGCCGAGTGTCCGGGAGTCG Full-length cloning of VGSC MuNav-3(R) AGTCGTAACCAGATCAAGCTTTCAGACATCCGCAAGGGCCGGAG Domain II-5(F) ACATTTTCTGCGTGTGGGACTGC Detection of mutation sites in Domain II Domain II-5(R) CCAGTTGATAAATCGTGATATTC Genotyping of pyrethroid resistance-associated mutations
-
Thirty adults of M. usitatus were selected from each of the five populations (HK-S, CM-R, LD-R, SY-R, and LS-R), and genomic DNA was individually extracted from each adult using a DNA extraction kit (Beijing Biotech). Specific primers, Domain II-5(F) and Domain II-3(R) (synthesized by Tsingke, Beijing, China), were designed to target the Domain II region of the VGSC gene. PCR amplification was performed using Vazyme Rapid Taq Master Mix. The PCR products were purified and recovered using an Omega kit (USA), ligated into the Vazyme T-vector, and transformed into StBl2 competent cells. After 90 min of incubation with shaking, the cells were spread onto LA solid medium plates containing 10 mg/mL ampicillin and incubated inverted for 24 h. Single colonies were picked and subjected to colony PCR for verification. Positive clones were cultured for amplification and then sent to Tsingke Biotechnology Co., Ltd for sequencing. The obtained sequencing data were analyzed using DNASTAR Lasergene V7.1 Editseq software, and the frequency of the target mutation sites was calculated.
-
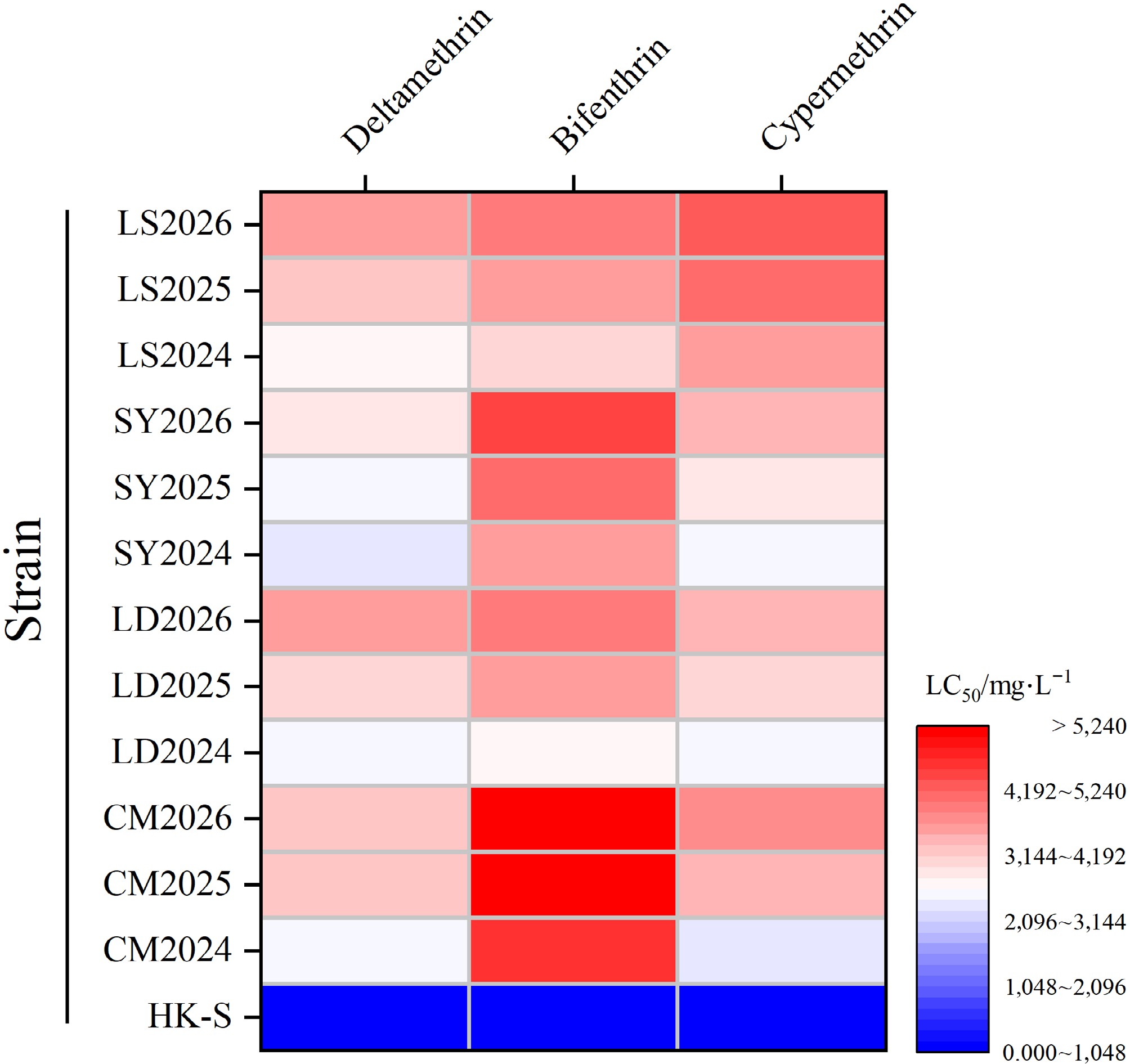
Using the residual film method, the toxicity of three pyrethroids against the susceptible strain (HK-S) and four field populations (CM-R, LD-R, SY-R, LS-R) of M. usitatus was consecutively determined over three years; the results are presented in Tables 3−5. The bioassay results from 2024 indicated that the CM-R strain exhibited the highest resistance, with maximum LC50 values of 4,642.120, 2,489.687, and 2,380.572 mg/L for the three insecticides, respectively. The SY-R and LS-R strains followed, with maximum LC50 values of 3,602.825, 2,049.838 mg/L, and 2,459.628 mg/L and 2,999.842, 2,699.051 mg/L, and 3,652.185 mg/L, respectively. The LD-R strain showed comparatively weaker resistance, yet its levels remained higher than those of HK-S, with maximum LC50 values of 2,710.853, 2,579.637, and 2,461.168 mg/L.
Table 3. Resistance levels of M. usitatus to Deltamethrin.
Treatment Strain Sample No. Slope ± standard error Degree of freedom Chi-square LC50 (95% confidence interval) Resistance ratio Deltamethrin HK-S 444 1.975 ± 0.239 4 0.279 8.148 (5.853−10.311) 1 CM2024 461 2.545 ± 0.016 4 4.864 2,489.687 (1,473.860−4,688.216) 305.6 CM2025 450 5.197 ± 0.571 4 0.173 3,181.355 (2,890.989−3,514.066) 390.4 CM2026 449 3.920 ± 0.433 4 0.210 3,302.052 (2,945.182−3,755.924) 405.3 LD2024 445 1.162 ± 0.133 4 7.676 2,579.637 (2,469.245−3,151.628) 316.6 LD2025 441 1.096 ± 0.217 4 3.294 3,102.792 (1,902.736−5,426.599) 380.8 LD2026 450 2.947 ± 0.329 4 0.091 3,573.267 (3,090.145−4,270.751) 438.6 SY2024 438 0.905 ± 0.127 4 0.890 2,409.838 (1,472.794−3,195.164) 295.8 SY2025 446 1.477 ± 0.073 4 1.011 2,583.096 (1,401.633−5,023.782) 317.0 SY2026 447 3.102 ± 0.308 4 1.268 2,962.999 (2,131.597−4,757.661) 363.6 LS2024 443 2.190 ± 0.169 4 3.672 2,699.051 (2,730.686−2,857.614) 331.3 LS2025 440 2.174 ± 0.228 4 1.806 3,276.117 (1,396.584−6,403.851) 402.1 LS2026 448 2.952 ± 0.316 4 1.281 3,553.753 (2,280.381−4,217.518) 436.2 Resistance fold = Field population LC50/Sensitive strain (HK-S) LC50. HK-S is an indoor strain that has not been exposed to insecticides for an extended period, specifically from 2019 to 2025, and its LC50 serves as the fixed benchmark for all comparisons. Table 4. Resistance levels of M. usitatus to Bifenthrin.
Treatment Strain Sample No. Slope ± standard error Degree of freedom Chi-square LC50 (95% confidence interval) Resistance ratio Bifenthrin HK-S 444 2.248 ± 0.293 4 0.461 7.610 (5.501−9.515) 1 CM2024 446 0.810 ± 0.120 4 2.060 4,642.120 (2,744.790−5,345.450) 610.0 CM2025 445 2.746 ± 0.386 4 0.704 5,154.927 (4,253.658−6,853.767) 677.4 CM2026 450 2.233 ± 0.291 4 0.461 5,222.599 (4,167.747−7,257.130) 686.3 LD2024 441 0.713 ± 0.124 4 0.154 2,710.853 (1,747.671−5,465.877) 356.2 LD2025 429 1.541 ± 0.155 4 1.045 3,508.291 (2,491.721−7,786.022) 461.0 LD2026 449 3.409 ± 0.416 4 0.428 3,964.617 (3,461.532−4,705.489) 521.0 SY2024 438 0.801 ± 0.129 4 0.025 3,602.825 (2,246.790−7,576.970) 473.4 SY2025 430 1.206 ± 0.022 4 2.964 4,127.643 (2,578.660−7,906.107) 542.4 SY2026 448 3.059 ± 0.397 4 0.495 4,440.504 (3,790.218−5,507.876) 583.5 LS2024 446 2.902 ± 0.222 4 7.844 2,999.842 (2,771.627−3,289.249) 394.2 LS2025 450 1.794 ± 0.067 4 2.454 3,601.884 (2,391.087−6,201.144) 473.3 LS2026 449 4.741 ± 0.580 4 4.453 3,946.913 (3,371.774−4,824.179) 518.7 Resistance fold = Field population LC50/Sensitive strain (HK-S) LC50. HK-S is an indoor strain that has not been exposed to insecticides for an extended period, specifically from 2019 to 2025, and its LC50 serves as the fixed benchmark for all comparisons. Table 5. Resistance levels of M. usitatus to Cypermethrin.
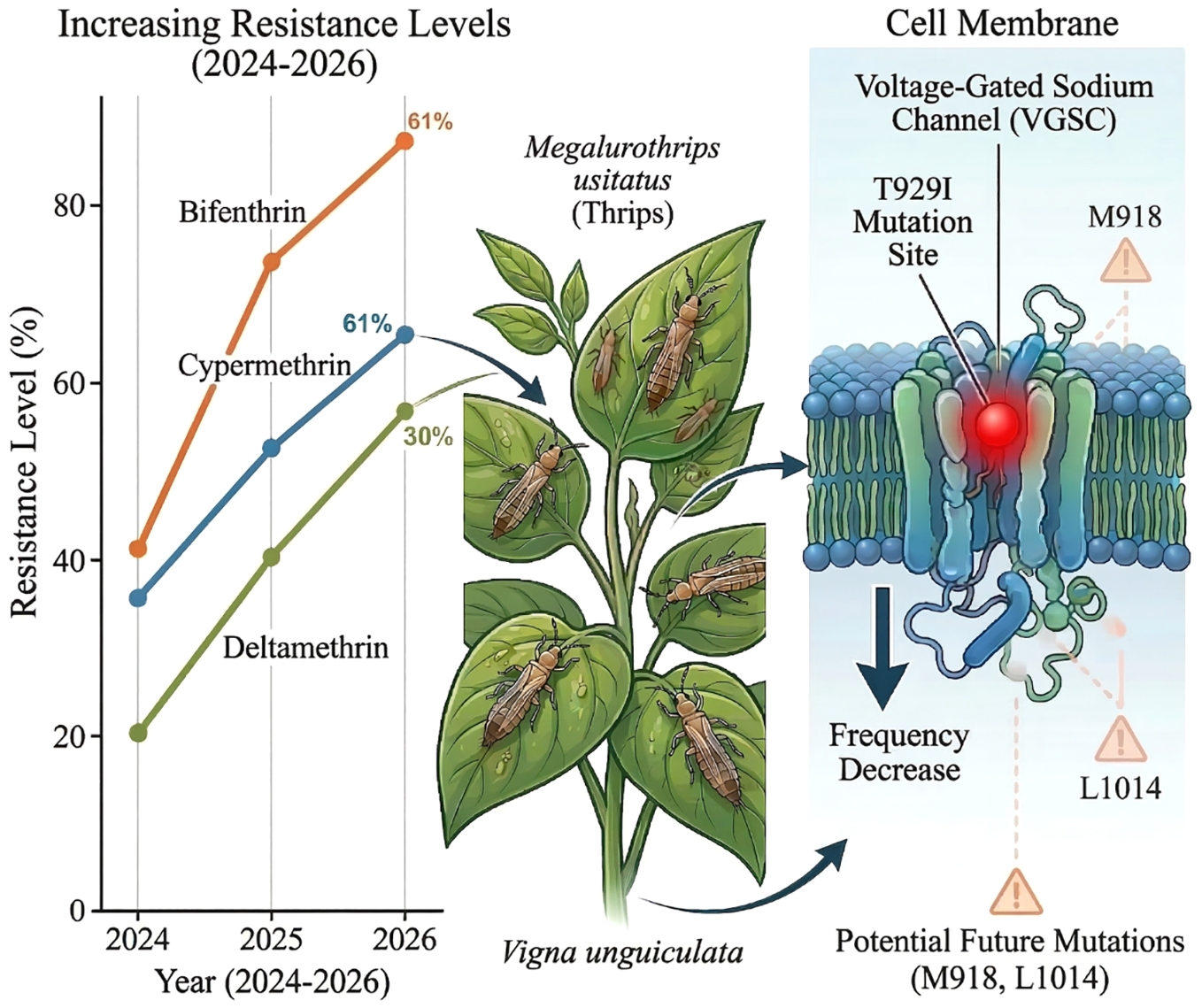
Treatment Strain Sample No. Slope ± standard error Degree of freedom Chi-square LC50 (95% confidence interval) Resistance ratio Cypermethrin HK-S 447 1.730 ± 0.222 4 0.566 6.756 (4.387−9.029) 1 CM2024 425 2.871 ± 0.062 4 2.524 2,380.572 (1,209.860−4,688.216) 352.4 CM2025 450 2.102 ± 0.219 4 0.425 3,342.027 (2,773.125−4,222.895) 494.7 CM2026 450 1.651 ± 0.179 4 0.081 3,738.598 (2,939.492−5,142.503) 553.4 LD2024 445 2.203 ± 0.187 4 1.724 2,461.168 (2,390.935−2,937.372) 364.3 LD2025 445 2.017 ± 0.884 4 1.027 3,055.231 (1,491.087−4,923.077) 452.2 LD2026 450 2.250 ± 0.262 4 2.250 3,319.287 (2,828.367−4,040.714) 491.3 SY2024 437 0.866 ± 0.125 4 0.816 2,459.628 (1,644.078−4,515.853) 364.1 SY2025 429 2.151 ± 0.017 4 1.739 2,963.782 (1,708.851−4,996.745) 438.7 SY2026 448 2.970 ± 0.319 4 1.205 3,359.198 (2,913.821−3,984.137) 497.2 LS2024 441 0.891 ± 0.125 4 0.939 3,652.185 (2,219.346−8,724.188) 540.6 LS2025 448 2.966 ± 0.693 4 1.831 4,102.857 (3,201.822−7,012.696) 607.3 LS2026 449 3.874 ± 0.509 4 0.535 4,314.532 (3,795.844−5,094.333) 638.6 Resistance fold = Field population LC50/Sensitive strain (HK-S) LC50. HK-S is an indoor strain that has not been exposed to insecticides for an extended period, specifically from 2019 to 2025, and its LC50 serves as the fixed benchmark for all comparisons. Compared with 2024, the resistance levels of the four geographical populations to deltamethrin, bifenthrin, and cypermethrin all increased in 2025. The LC50 values of the four populations against deltamethrin were 3,181.355, 3,102.792, 2,583.096, and 3,276.117 mg/L, respectively. Against bifenthrin, the LC50 values were 5,154.927, 3,508.291, 4,127.643, and 3,601.884 mg/L, respectively. For cypermethrin, the LC50 values were 3,342.027, 3,055.231, 2,963.782, and 4,102.857 mg/L, respectively. In 2026, resistance to the three pyrethroids continued to increase in the four field populations, albeit at a decelerated rate, and differential resistance patterns were observed. For deltamethrin: LD-R and LS-R exhibited the highest resistance (LC50 = 3,573.267 and 3,553.753 mg/L, respectively), followed by CM-R and SY-R (LC50 = 3,302.052 and 2,962.999 mg/L, respectively). For cypermethrin: SY-R and LD-R displayed comparable resistance levels (LC50 = 3,359.198 and 3,319.287 mg/L, respectively), while LS-R exhibited higher resistance than CM-R (LC50 = 4,314.532 and 3,738.598 mg/L, respectively). For bifenthrin: LD-R and LS-R exhibited comparable resistance (LC50 = 3,964.617 and 3,946.913 mg/L, respectively); CM-R possessed the highest resistance (LC50 = 5,222.599 mg/L), followed by SY-R (LC50 = 4,440.504 mg/L). Based on three consecutive years of resistance monitoring, significant selectivity differences were observed among geographical populations in response to distinct insecticides. The relative resistance ranking of the four field populations to the three pyrethroid insecticides was bifenthrin > cypermethrin > deltamethrin. The overall resistance levels of the four geographical populations to the three tested pyrethroid insecticides were ranked as follows: CM-R > LS-R > SY-R > LD-R (Fig. 1).
SNP/INDEL analysis of pyrethroid resistance-associated mutations
-
To assess potential sequence variations among different geographical populations of M. usitatus and the selected strains for pesticide screening, this study cloned and validated the integrity and correctness of the VGSC gene MuNav1-1 of M. usitatus from the experimental population. The sequence exhibited complete alignment with the reference sequence (accession number MZ043856)[5], suggesting that this gene is highly conserved within the experimental population. Consequently, it can be employed for subsequent detection of resistance mutations and functional verification. After obtaining the full-length gene of the VGSC from the M. usitatus, a 3D simulation of the VGSC was constructed based on its basic structure and amino acid sequence information (Fig. 2).
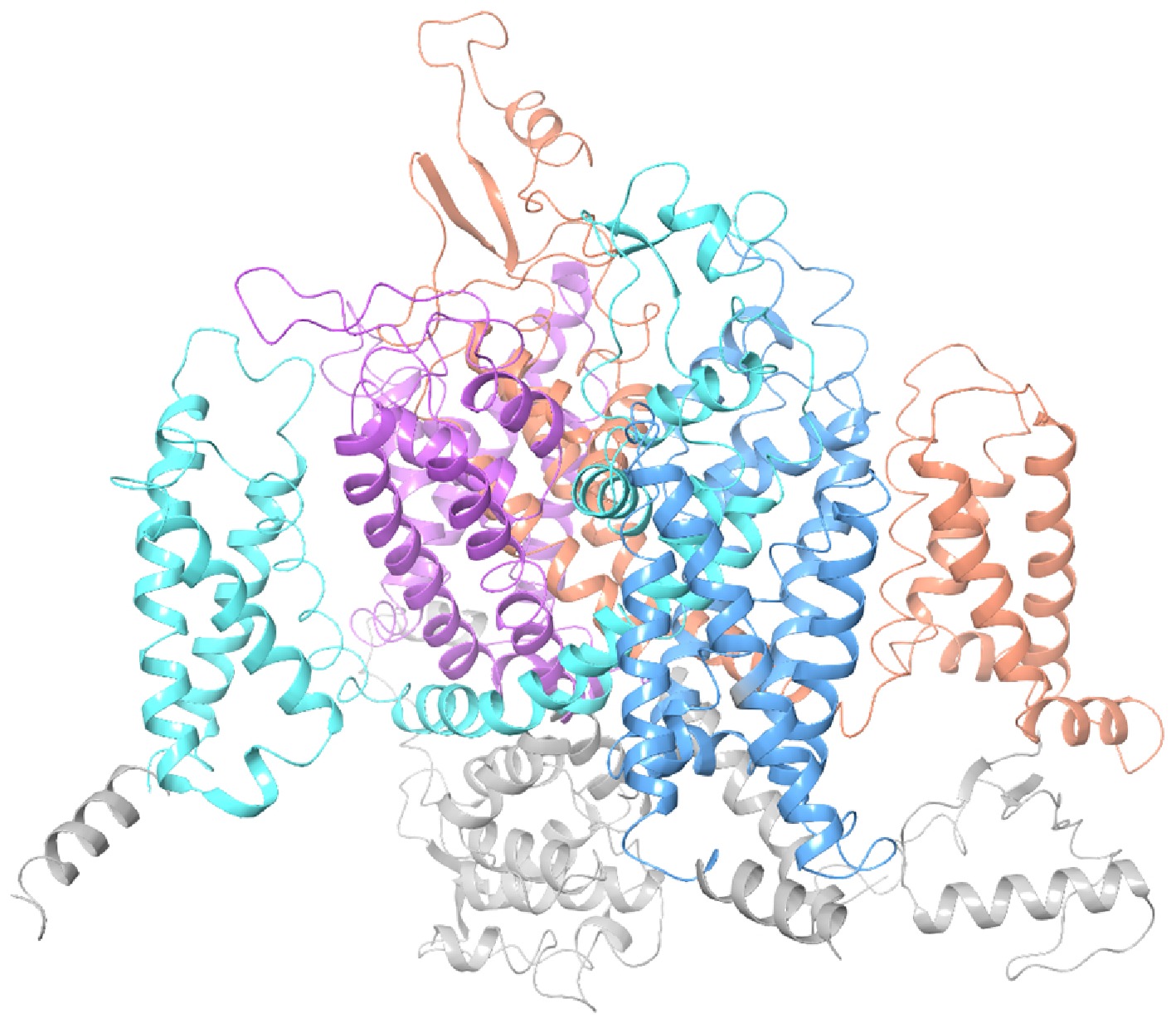
Figure 2.
3D model of M. usitatus VGSC (orange area: Domain I, blue area: Domain II, light blue area: Domain III, purple area: Domain IV).
Additionally, 30 adult M. usitatus were selected from each of the five populations: HK-S, CM-R, LD-R, SY-R, and LS-R. Their individual DNA was used as a template for cloning the VGSC structure Domain II region using RT-PCR technology. During the analysis, special attention was paid to the super kdr mutation site M918T, which has not been reported in M. usitatus, as well as the kdr mutations L1014F and M918L reported in Thrips tabaci. The sequencing data identification results (Table 6) showed that M918T/L and L1014F were not detected in the tested populations. However, the reported T929I mutation was detected in all populations except for the sensitive strain HK-S, which is located in the VGSC Domain II (Table 7, Fig. 3).
Table 6. Mutation frequency of M. usitatus M918L/T, L1014F, and T929I mutation sites.
Strain Year M918L Mutation frequency M918T Mutation frequency T929I Mutation frequency L1014F Mutation frequency Total samples HK-S 2025 M/L 0 M/T 0 T/I 0 L/F 0 29 CM2024 2024 M/L 0 M/T 0 T/I 100 L/F 0 29 CM2025 2025 M/L 0 M/T 0 T/I 93.3 L/F 0 30 CM2026 2026 M/L 0 M/T 0 T/I 90.0 L/F 0 30 LD2024 2024 M/L 0 M/T 0 T/I 100 L/F 0 30 LD2025 2025 M/L 0 M/T 0 T/I 100 L/F 0 30 LD2026 2026 M/L 0 M/T 0 T/I 96.7 L/F 0 30 SY2024 2024 M/L 0 M/T 0 T/I 100 L/F 0 29 SY2025 2025 M/L 0 M/T 0 T/I 100 L/F 0 28 SY2026 2026 M/L 0 M/T 0 T/I 96.7 L/F 0 30 LS2024 2024 M/L 0 M/T 0 T/I 100.0 L/F 0 29 LS2025 2025 M/L 0 M/T 0 T/I 90.0 L/F 0 30 LS2026 2026 M/L 0 M/T 0 T/I 90.0 L/F 0 30 To ensure the reliability and accuracy of the experimental data, samples that did not meet quality standards were excluded from the final statistical analysis. Table 7. Amino acid sequences of the Domain II-localized T929I mutation and the pyrethroid-binding site (IIL45-IIS5-IIIS6) in M. usitatus.
Channel Segment MuNav1-1 IL45-S5 252VPGLKTIVGA VI ESVKNLRDVI ILTMFSLSVF ALMGLQIY IIL45-S5 934WPTLNLLISI MG RTMGALGNLT FVLCIIIFIF AVMGMQLF IIIL45-S5 1431MQGMRVVVNA LV QAIPSIFNVL LVCLIFWLIF AIMGVQLF IVL45-S5 1749AKGIRTLLFA LA MSLPALFNIC LLLFLVMFIF AIFGMSFF IS6 402PWHMLFFIVI IFLGSFYLVN LILAIVAMSY DE IIS6 1025WSCIPFFLAT VVIGNLVVLN LFLALLLSNF GS IIIS6 1552IYMYLYFVFF IIFGSFFTLN LFIGVIIDNF NE IIIS6 1852TIGITFLLSY LVISFLIVIN MYIAVILENY SQ 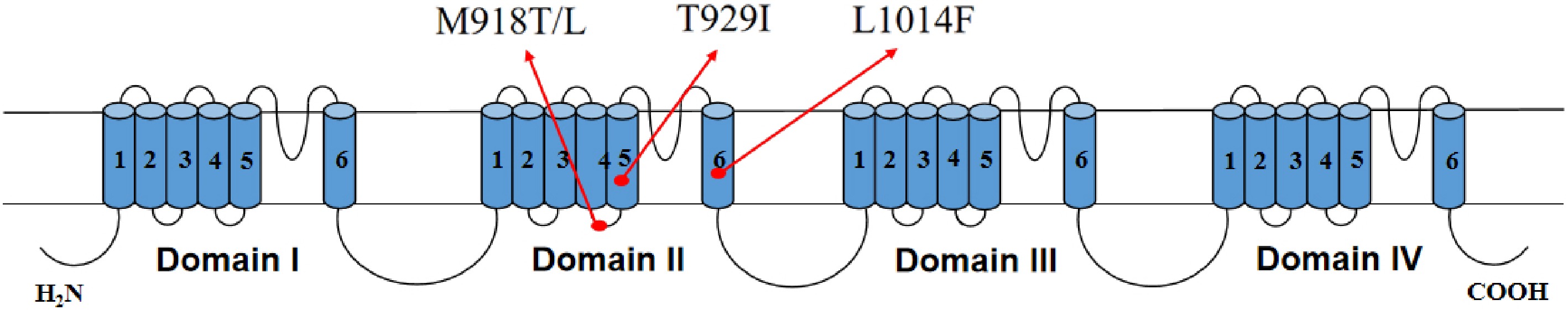
Figure 3.
Topological structure diagram of the VGSC M918L/T, T929I and L1014F mutation sites in the M. usitatus.
Compared to the sensitive strain HK-S, the T929I mutation was detected in all four field populations (CM-R, LD-R, SY-R, LS-R) across all years, whereas the frequency of this mutation was zero in HK-S, with no detection of this mutation. In 2024, the T929I mutation frequency in all four field populations reached 100%, representing the highest mutation frequency detected at this locus. In 2025, the mutation frequency at this locus began to differentiate among populations, with LD2025 and SY2025 maintaining a 100% mutation frequency, while the mutation frequency in CM2025 decreased to 93.3%, and that in LS2025 decreased to 90.0%. In 2026, the T929I mutation frequency in all four field populations showed varying degrees of decline. The mutation frequency in CM2026 further decreased to 90.0%, while that in LD2026 and SY2026 both decreased to 96.7%. LS2026 maintained the same mutation frequency of 90.0% as in 2025. Overall, from 2024 to 2026, the T929I mutation frequency in the four field populations showed a decreasing trend from the initial 100% year by year, with no increase in mutation frequency observed. Furthermore, the differentiation in mutation frequency among populations began to emerge in 2025 and continued to persist in 2026 (Table 6).
-
In agricultural production practices, diverse pesticides are extensively utilized for managing crop diseases and arthropod pests, thereby providing essential support for sustaining food production. However, with the continuous escalation of pesticide applications, target pests have progressively developed resistance globally, presenting a formidable challenge to the Integrated Pest Management (IPM) system worldwide[33,34]. Pyrethroid insecticides, representing the second most extensively used class of commercial insecticides globally, are deployed to control arthropod pests in agricultural and urban environments[35]. Their application is particularly prevalent in tropical and subtropical regions, where pest outbreaks occur with high frequency.
Hainan Province exhibits a typical subtropical climate characterized by year-round warmth and moisture, conditions that facilitate the proliferation and reproduction of insect pests. This ecological setting leads to severe overlapping generations and consequently accelerates the development of insecticide resistance and complicates pest management efforts. Previous investigations have demonstrated that field populations of M. usitatus collected from Haikou, Chengmai, Ledong, and Sanya in Hainan Province exhibit substantial resistance to β-cypermethrin and bifenthrin[36], a pattern further corroborated by the resistance monitoring data presented in this study. Between 2024 and 2025, resistance of M. usitatus to pyrethroid insecticides escalated rapidly. The 2024 bioassay revealed an LC50 value of 2,489.687 mg/L for the CM2024 population, which increased to 3,181.355 mg/L in 2025, representing a notable elevation. The resistance ratio rose from 305.6-fold to 390.4-fold within a single year. Concurrently, the LC50 for bifenthrin reached 5,154.927 mg/L (CM2025), indicating a 1.1-fold increase relative to 2024 and a 45-fold increase relative to 2015[14]. Furthermore, M. usitatus exhibited differential susceptibility among various pyrethroid compounds, with the highest resistance level observed against bifenthrin, followed by cypermethrin, and the lowest against deltamethrin. We hypothesize that this phenomenon may arise from the varying binding affinities and mechanisms associated with the molecular structures and target sites of different insecticides. Furthermore, disparities in the activity of metabolic detoxifying enzymes among distinct populations may also influence drug selectivity. This hierarchy provides a reference framework for optimizing future field applications of pyrethroid insecticides.
Insects primarily circumvent the toxic effects of insecticides through detoxifying enzyme metabolism, target-site mutations, and cuticular barrier mechanisms[37−39]. Previous research has established that pyrethroid resistance in M. usitatus is associated with mutations in the VGSC[40]. The M918T and L1014F substitutions, designated as 'super-kdr' and 'kdr' respectively, confer exceptionally high resistance levels across a diverse range of insect pests. Their association with resistance was initially identified in the housefly (Musca domestica) through molecular cloning and sequencing, and subsequently validated in an electrophysiological expression system using Drosophila melanogaster[41,42]. The escalating prevalence of these mutations among pest populations poses a considerable obstacle to agricultural pest management due to the extraordinarily elevated resistance level conferred[43]. Notably, thrips pests exhibit rapid reproduction rates and considerable adaptability to environmental changes. Under the selective pressure imposed by pesticides, these pests are likely to accumulate multiple mutations in VGSCs, resulting in substantial variation in mutation spectra and resistance levels across different species[44]. In Thrips palmi Karny, the dual mutation I265T/L1014F was initially documented in field populations in Hainan. Between 2020 and 2022, the mutation frequencies of this dual mutation in the local population (HN) increased dramatically from 53.33% to 96.67%, functional verification demonstrated that the combined mutations I265T and L1014F enhance the resistance of T. palmi to pyrethroid insecticides by 20 to 25 fold compared with susceptible populations[19]. In Frankliniella occidentalis, the mutation frequencies of L1014F vary substantially among different populations, ranging from 11.01% (Fo4) to 37.29% (Fo5)[45]. In T. tabaci, the M918L substitution, which is associated with high resistance in various pest species, exists in VGSCs in the heterozygous M/L form; furthermore, the dual mutation M918T/L1014F has also been documented. Notably, these two types of mutations occur exclusively in female-parthenogenetic populations of T. tabaci, and the M918T/L1014F dual mutation is associated with a higher adaptive cost[46].
VGSC mutations can substantially reduce the sensitivity of thrips pests to pyrethroid insecticides. In the resistant population of M. usitatus in Hainan, the mutation frequencies of M283R were 3.33%, 3.33%, and 10.0% from 2019 to 2021, indicating a notable upward trend[47]. As the population resistance level increased annually, the mutations T929I and K1774N emerged sequentially, with the mutation frequencies of T929I reaching 100%. Electrophysiological experiments further confirmed that these mutations confer substantial resistance to pyrethroid insecticides, including permethrin, deltamethrin, bifenthrin, and lambda-cyhalothrin in M. usitatus[48]. In recent years, the mutation rate of T929I has shown a slight decline in M. usitatus of ordinary giant thistle horses, potentially linked to fitness costs, alterations in population genetic structure, or compensatory mutations, although our study did not detect M918T and L1014F mutations, previous research on T. tabaci has established a strong correlation between these super kdr mutations and parthenogenesis, indicating that they may persist as low-frequency heterozygotes, even if undetected in field populations. Therefore, the distinct reproductive traits of thrips, coupled with the limitations of detection methods, imply that these mutations may still present potential risks in M. usitatus, further investigation is required to ascertain whether different breeding populations of M. usitatus will demonstrate resistance differentiation akin to that observed in whiteflies, or if the low frequency of such mutations, attributed to high fitness costs, complicates their detection[49]. From a resistance evolution perspective, the emergence of these sodium channel mutations is closely correlated with high-intensity pesticide selection pressure in agricultural fields[20]. As the overall resistance level of M. usitatus continues to increase, the potential risk for the emergence of M918T/L1014F dual mutations will further escalate.
This study investigated the kdr loci M918T/L, L1014F, and T929I within Domain II of the VGSC binding sites in M. usitatus populations exposed to pyrethroid insecticides. No novel mutations were identified in the tested samples, with the exception of T929I. However, given the annual increase in resistance levels, we anticipate a potential risk for the emergence of M918T/L and L1014F mutations in field populations of M. usitatus. This concern is reinforced by the emergence of populations exhibiting exceptionally high resistance levels in closely related thrips species, including T. tabaci, T. palmi Karny, and F. occidentalis, which have demonstrated high resistance to pyrethroid insecticides[19,43,44]. We performed functional verification of M918T/L and L1014F utilizing the Xenopus oocyte expression system and the two-electrode voltage clamp technique, revealing a substantial correlation with elevated resistance of M. usitatus to pyrethroid insecticides (data not yet published). This finding indirectly suggests the likelihood of mutations arising when field resistance reaches a critical threshold. Such insights provide a theoretical foundation for understanding pest resistance mechanisms and inform proactive scientific prevention and control strategies. This knowledge can facilitate the design of targeted pyrethroid insecticide application strategies, including rational rotation or mixed use of compounds based on the kdr genotypes identified in field populations. Ultimately, the sustained efficacy of pyrethroid insecticides will depend on science-driven pest management approaches that prioritize comprehensive monitoring of mutation frequencies.
-
This study examined the resistance of three pyrethroid insecticides in four strains of M. usitatus in Hainan (CM-R, LS-R, SY-R, LD-R), and one laboratory-sensitive strain (HK-S) from 2024 to 2026. The results indicated a year-on-year increase in resistance among M. usitatus in Hainan Province to pyrethroid insecticides, with interannual variations in the rate of development. Notably, all strains exhibited high levels of resistance to bromoxystrobin, cypermethrin, and chlorpyrifos, with resistance levels rising sharply from 2024 to 2025 and then decelerating in 2026. The overall resistance ranking was CM-R > LS-R > SY-R > LD-R > HK-S, with cypermethrin demonstrating the highest selectivity among the pesticides tested. Additionally, mutation point detection was performed on the target sodium channel related to pyrethroid insecticides over three consecutive years. Molecular testing revealed that T929I was the sole mutation associated with pyrethroid resistance, exhibiting a change in mutation frequency among M. usitatus in Hainan. The mutation frequency reached 100% in 2024 but showed a population-specific decline from 2025 to 2026.
-
The authors confirm contributions to the paper as follows: methodology: Xia G, Yuan D, Wang L, Gao R, Wu S; data curation: Xia G, Yuan D, Wang L, Wang Q, Xie Y, Wayne J; resources: Wu S; draft manuscript preparation: Xia G; writing−review and editing: Wu S; supervision: Wu S; funding acquisition: Wu S. All authors reviewed the results and approved the final version of the manuscript.
-
All data generated or analyzed during this study are included in this published article and its supplementary information files. The core datasets (including bioassay mortality records, LC50 calculation results, and mutation frequency statistics) are presented in the tables and figures of the main text, while detailed raw data and supplementary experimental records are provided in the Electronic Supplementary Information for further reference and reproduction.
-
This study was funded by the National Natural Science Foundation of China (Grant No. 32260666), National Key R&D Program of China (Grant No. 2024YFD1400100), HNARS2023-3-G5, the Science and Technology Commissioner Project of Hainan Province: (Grant No. KJTP202514), "111" Project (Grant No. D20024), Science and Technology special fund of Hainan Province (Grant No. ZDYF2024XDNY250).
-
The authors declare that they have no conflict of interest.
-
Received 21 February 2026; Accepted 16 March 2026; Published online 27 March 2026
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press on behalf of Hainan University. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Xia G, Yuan D, Wang L, Gao R, Wang Q, et al. 2026. Evolutionary dynamics of pyrethroid resistance in the Megalurothrips usitatus of Hainan and detection of voltage-gated sodium channel mutation sites. Tropical Plants 5: e011 doi: 10.48130/tp-0026-0010
Evolutionary dynamics of pyrethroid resistance in the Megalurothrips usitatus of Hainan and detection of voltage-gated sodium channel mutation sites
- Received: 21 February 2026
- Revised: 11 March 2026
- Accepted: 16 March 2026
- Published online: 27 March 2026
Abstract: To elucidate the evolutionary dynamics and molecular mechanisms underlying resistance to pyrethroid insecticides in the Megalurothrips usitatus of Hainan Province, resistance levels to deltamethrin, bifenthrin, and cypermethrin were systematically assessed in four field populations from 2024 to 2026. The voltage-gated sodium channel (VGSC) gene was cloned, and resistance-associated mutation sites were identified. The findings indicated that all four field populations demonstrated elevated levels of resistance to the three pyrethroids. Although resistance continued to escalate from 2024 to 2026, the rate of increase decelerated in 2026. The overall resistance of the populations was ranked as CM-R > LS-R > SY-R > LD-R, with the selectivity of resistance to the insecticides ordered as bifenthrin > cypermethrin > deltamethrin. The full-length VGSC gene of M. usitatus, the T929I mutation, was identified in all four field populations, exhibiting a frequency of 100% in 2024. From 2025 to 2026, this frequency exhibited a declining trend, accompanied by population differentiation. Although no M918L/T or L1014F mutations were detected, these mutations are widely present in other thrips closely related to the M. usitatus, indicating potential risks that may arise in the future. Currently, T929I is the sole mutation associated with pyrethroid resistance identified in the M. usitatus in Hainan, and its decreasing frequency may be linked to novel resistance adaptations within the VGSC of this insect. The findings of this study provide a theoretical foundation for the early monitoring and scientific management of resistance in the M. usitatus in Hainan.