-
Medicinal mushrooms have become an important research topic in the fields of pharmaceutical, nutraceutical, and functional food in recent years due to their richness in natural bioactive components[1]. The main secondary metabolites they contain include polysaccharides, phenolic compounds, sterols, and terpenoids, and these components have versatile biological effects in protecting human health and treating various diseases[2]. Especially complex polysaccharides such as beta-glucan play a critical role in the modulation of the immune system, increasing the body's defense capacity against infections and tumoral cells. These mushrooms can also suppress excessive responses in autoimmune diseases or strengthen weakened immune functions by showing balancing (immunomodulatory) effects on the immune system[3]. Their antiviral and antibacterial activities enable them to be used as alternative natural agents, especially in the fight against resistant pathogens. Owing to their substantial antioxidant capacity, they contribute to the mitigation of free radical induced oxidative stress and thereby play a critical role in maintaining cellular integrity[4,5]. In addition, various in vitro and in vivo studies have shown that extracts obtained from some medicinal mushroom species suppress the proliferation of cancer cells, stimulate apoptosis mechanisms, and inhibit tumor growth[6]. At the same time, thanks to their anti-inflammatory properties, they provide therapeutic support by positively affecting the course of diseases associated with chronic inflammation[7]. With all these properties, medicinal mushrooms are one of the valuable biological resources containing natural and versatile bioactive compounds that are evaluated as complementary treatment agents in modern medicine. Therefore, this study aimed to comprehensively evaluate the antioxidant, antimicrobial, antiproliferative, and anticholinesterase activities of the mushroom Lactarius salmonicolor R. Heim & Leclair.
Lactarius salmonicolor is an edible mushroom species with high commercial value, naturally distributed especially in Europe, Asia, and North America. It attracts attention for both its nutritional value and its bioactive compounds[8]. It has a nutritional profile with high moisture (86.8%–91.1%), protein (9.4%–19.0%), carbohydrate (71.8%–83.9%), and low-fat content (0.6%–1.1%) on a dry weight basis. The energy value varies between 372.1–382.6 kcal per 100 g of dry matter[9]. When evaluated in terms of fatty acid profile, stearic, oleic, linoleic, and palmitic acids are the main components, and the high rate of unsaturated fatty acids is a positive feature in terms of nutrition[10]. In addition, it is rich in essential minerals such as calcium, magnesium, selenium, and zinc; however, the level of toxic heavy metals such as arsenic (As), cadmium (Cd), and mercury (Hg) is at a level that does not pose a risk to human health[11]. Lactarius salmonicolor shows high antioxidant capacity with its phenolic compounds and tocopherols (especially β-tocopherol) content[12]. In terms of antimicrobials, it has been reported to be effective against some Gram-positive and Gram-negative bacteria, but it has not been reported to have a significant effect against yeast species[11]. Lactarius salmonicolor, which is also an important species in terms of ecological and economic aspects, is being evaluated by creating distribution maps within the scope of sustainable forest management, and developing strategies for the protection of the species[8].
Although several studies have investigated the biological activities of wild mushrooms, comprehensive data on the ethanol extract of L. salmonicolor remain limited. Existing reports are few, and often focus on a single activity. Therefore, this study aims to fill this gap by evaluating the antioxidant, anticholinesterase, antiproliferative, and antimicrobial properties of the ethanol extract of L. salmonicolor, in an integrated manner.
-
The samples of L. salmonicolor species evaluated in this study (Fig. 1) were collected during field studies conducted in the Kastamonu province of Türkiye. The samples obtained as a result of the field studies were transferred to the laboratory, and dried under controlled temperature conditions (45 °C for 48 h) to obtain a suitable form for analysis. For the extraction process, 10 g of dry mushroom sample was extracted with 250 mL of ethanol in a Soxhlet apparatus at 50 °C for approximately 6 h. The obtained ethanolic extract was concentrated with a Buchi R-100 model rotary evaporator operating at 40 °C to remove solvent residues. The final extract was stored at +4 °C to maintain its chemical stability before analysis.
Antioxidant activity tests
-
The total antioxidant status (TAS) and total oxidant status (TOS) of the ethanolic L. salmonicolor extract used in this study were quantitatively analyzed using commercial kits from Rel Assay Diagnostics (Rel Assay Diagnostics, Mega Tıp, Gaziantep, Turkey). All measurements were performed in full compliance with the protocol instructions provided by the manufacturer. Trolox was used as a standard reference compound in the determination of TAS, and hydrogen peroxide was used for calibration in the TOS analysis. The results obtained were reported in mmol/L for TAS, and µmol/L for TOS[13,14]. The Oxidative Stress Index (OSI) value was calculated by converting both parameters to the same unit and then dividing TOS by TAS[15].
Anticholinesterase activity test
-
In this study, the anticholinesterase activity of the ethanolic extract obtained from L. salmonicolor was evaluated based on the colorimetric method developed by Ellman et al[16]. Inhibitory effects on acetylcholinesterase (AChE), and butyrylcholinesterase (BChE) enzymes were determined, and the standard inhibitor called galantamine was used as a comparative reference. For the analyses, different extract concentrations in the range of 200–3.125 μg/mL were prepared, and test solutions were created. In each experiment, 130 μL of 0.1 M phosphate buffer, prepared at pH 8.0, 10 μL of extract solution, and 20 μL of enzyme solution (AChE or BChE) were added to each of the 96-well microplates, respectively. The prepared reaction mixtures were incubated at 25 °C, and in the dark for 10 min. After incubation, 20 μL of DTNB [5,5′-dithiobis-(2-nitrobenzoic acid)] solution, and 20 μL of acetylcholine iodide (AChI) or butyrylcholine iodide (BChI) as substrate were added to each well to initiate the enzymatic reaction. Enzyme activities were measured spectrophotometrically at 412 nm wavelength, and the inhibitory effect of the extract was determined by calculating IC50 (half maximal inhibitory concentration) values. All experiments were performed in triplicate, and the reliability of the results was confirmed statistically.
Antiproliferative activity test
-
In this study, the cytotoxic effects of ethanolic extract obtained from L. salmonicolor on A549 (lung adenocarcinoma; ATCC CCL-185) cell line were evaluated by MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] based cell viability test. The extract was prepared at 25, 50, 100, and 200 μg/mL concentrations to be used in experimental applications. When the cell cultures reached 70%–80% confluency, detachment from the surface was performed using 3.0 mL Trypsin-EDTA solution (Sigma-Aldrich, MO, USA), and the suspended cells were seeded in 96-well culture plates at appropriate density (1 × 104/well). A 24-h incubation period was applied for adaptation of the cells to the ambient conditions. After this period, the prepared extract solutions were added to the cells, and a second 24-h incubation was performed. At the end of the application period, the culture medium was removed, and MTT solution, prepared at a concentration of 1 mg/mL, was added to each well (The cells were incubated for 4 h at 37 °C). The purple formazan crystals formed by MTT reduced by the metabolically active mitochondria of the cells were dissolved with the help of dimethyl sulfoxide (DMSO) (Sigma-Aldrich, MO, USA) after incubation. The optical density (OD) values related to cell viability were measured with an Epoch model microplate reader (BioTek Instruments, Winooski, VT, USA) operating at a wavelength of 570 nm, and analyzed based on the obtained data[17].
Antimicrobial activity test
-
The minimum inhibitory concentrations (MICs) of the extract were determined against a panel of reference bacterial and fungal strains. The bacterial strains tested were Staphylococcus aureus ATCC 29213, methicillin-resistant S. aureus (MRSA) ATCC 43300, Enterococcus faecalis ATCC 29212, Escherichia coli ATCC 25922, Pseudomonas aeruginosa ATCC 27853, and Acinetobacter baumannii ATCC 19606. For antifungal evaluation, Candida albicans ATCC 10231, C. krusei ATCC 34135, and C. glabrata ATCC 90030 were used. All reference strains were obtained from the American Type Culture Collection (ATCC). Bacterial strains were subcultured in Mueller-Hinton broth (MHB), whereas yeast strains were grown in RPMI 1640 broth. The turbidity of each suspension was adjusted to the 0.5 McFarland standard to obtain a standardized inoculum, which was then further diluted to approximately 106 CFU/mL. The ethanolic extract was tested at concentrations ranging from 800 to 12.5 µg/mL, by incorporating the appropriate amount of extract into molten Mueller-Hinton agar, which had been dispensed into sterile tubes (9 mL per tube), mixed thoroughly, and poured into Petri dishes. Solvents used for extract preparation were also tested alone as negative controls to exclude any intrinsic antimicrobial effect. For each microorganism, 0.01 mL of the standardized inoculum (104 CFU/spot) was applied to the surface of the agar plates containing the different extract concentrations. All plates were incubated at 35 °C for 16–20 h for bacterial strains, and 48 h for yeast strains. After incubation, plates were examined visually. The presence of colonies was interpreted as microbial growth, whereas the absence of visible colonies was considered complete inhibition. MIC values were defined as the lowest extract concentration that prevented visible growth and were expressed in µg/mL[18].
Statistical analysis
-
All experimental procedures were conducted in triplicate (n = 3), and the results are expressed as mean ± standard deviation (SD). Descriptive statistical analyses were carried out using IBM SPSS Statistics version 25 (IBM Corp., Armonk, NY, USA).
-
Mushrooms, especially edible and medicinal species, have significant antioxidant potential thanks to natural components such as phenolic compounds, flavonoids, ascorbic acid, ergothionein, and carotenoids[19,20]. These components contribute to the protection of cellular structure by playing a role in reducing oxidative stress caused by reactive oxygen species and free radicals[21]. Antioxidant capacities of mushrooms may vary depending on species characteristics, growth environment, extraction method, and solvent used[22]. These activities, evaluated by methods such as DPPH, ABTS, FRAP, and CUPRAC, reveal that mushrooms are not only organisms with nutritional value, but also natural antioxidant sources that can be evaluated pharmacologically[23]. In this study, the antioxidant potential of L. salmonicolor was evaluated. The obtained data are presented in Table 1.
Table 1. Total antioxidant and oxidant status and oxidative stress index of Lactarius salmonicolor.
Sample TAS (mmol/L) TOS (µmol/L) OSI (TOS/[TAS x 10]) Lactarius salmonicolor 3.844 ± 0.049 10.452 ± 0.126 0.272 ± 0.001 * Values were analysed with three repetitions and standard deviation values are given; TAS: Total Antioxidant Status (mmol/L), TOS: Total Oxidant Status (μmol/L); OSI: Oxidative Stress Index (calculated as (TOS/[TASx10]). Total antioxidant capacity (TAS), total oxidant level (TOS), and oxidative stress index (OSI) data of L. salmonicolor species have not been previously reported in the literature. However, it has been reported that this species has antioxidant activity using different methods[11,24]. In this study, the antioxidant capacity of L. salmonicolor was determined quantitatively for the first time using Rel Assay commercial kits. This method is a reliable and standardized analysis method that allows the precise evaluation of total antioxidant and oxidant load in biological samples. When compared with different mushroom species, the TAS value of L. salmonicolor (3.844 mmol/L) is lower than that of many species reported in the literature. For example, species such as Otidea onotica (8.866 mmol/L), Lactarius deliciosus (7.468 mmol/L), Hericium erinaceus (5.426 mmol/L), Cantharellus cibarius (5.511 mmol/L), and Phellinus hartigii (4.98 mmol/L) are observed to have higher TAS values[25−29]. Since the TAS value reflects the total effect of all antioxidant components (phenols, flavonoids, ascorbic acid, etc.) in the body of the organism, this difference indicates phytochemical diversity among species[30]. When evaluated in terms of TOS, the value of L. salmonicolor (10.452 µmol/L) was lower than that of Otidea onotica (14.724 µmol/L) and Lactarius deliciosus (13.161 µmol/L), but higher than that of Phellinus hartigii (9.27 µmol/L), Hericium erinaceus (6.621 µmol/L), and Cantharellus cibarius (7.289 µmol/L). The TOS value reflects the amount of reactive oxidant compounds found in the biological system, and a high TOS level indicates an increased risk of oxidative damage in the organism[30]. The oxidative stress index (OSI) is calculated as the ratio of TOS to TAS and reveals the oxidant-antioxidant balance in the system more comprehensively[30]. In this study, the OSI value of L. salmonicolor (0.272) is significantly higher than other species reported in the literature. Comparatively, OSI values are lower in Otidea onotica (0.166), Phellinus hartigii (0.19), Lactarius deliciosus (0.176), Hericium erinaceus (0.122), and Cantharellus cibarius (0.132). This situation shows that the oxidant compounds in L. salmonicolor are higher than its antioxidant defense capacity, and therefore, oxidative stress is more dominant in this species. When evaluated in general, although the antioxidant capacity of L. salmonicolor is limited, its oxidative load was found to be relatively high. This situation may be due to various environmental and biological factors such as the metabolic structure of the species, the ecosystem conditions in which it was collected, the developmental stage, and the extraction method. In future studies, detailed profiling of the phenolic and flavonoid components of this species, and the investigation of the effects of different extraction and culture conditions will allow a more comprehensive evaluation of the biological activity potential.
Anticholinesterase activity
-
Mushrooms are among the valuable biological resources that can exhibit anticholinesterase effects thanks to their rich secondary metabolite profile. In particular, bioactive substances such as phenolic compounds, terpenoids, sterols, and alkaloids contained in some medicinal mushroom species can suppress cholinesterase activity by inhibiting acetylcholinesterase (AChE), and butyrylcholinesterase (BChE) enzymes[31]. This inhibitory effect is of therapeutic importance as it contributes to the preservation of acetylcholine levels in neurodegenerative disorders such as Alzheimer's disease[32]. Mushrooms constitute a remarkable research area in terms of the discovery and development of natural inhibitors with neuroprotective potential[33]. In this study, the anticholinesterase activity of L. salmonicolor was examined, and the results obtained are presented in Table 2.
Table 2. Anticholinesterase activity of Lactarius salmonicolor.
Sample AChE (μg/mL) BChE (μg/mL) Lactarius salmonicolor 58.43 ± 1.58 88.45 ± 1.57 Galantamine 7.61 ± 0.21 15.67 ± 0.18 * Values were studied with three repetitions and standard deviation values are given; AChE: Acetylcholinesterase (μg/mL), BChE: Butyrylcholinesterase (μg/mL). In this study, the inhibition capacity of L. salmonicolor species on acetylcholinesterase (AChE), and butyrylcholinesterase (BChE) enzymes was evaluated. According to the obtained data, the AChE inhibition value of L. salmonicolor was determined as 58.43 ± 1.58 µg/mL, and the BChE inhibition value as 88.45 ± 1.57 µg/mL (Table 2). The positive control substance galantamine used for comparison showed values of 7.61 ± 0.21 µg/mL for AChE and 15.67±0.18 µg/mL for BChE, indicating that the inhibitory effect of L. salmonicolor has a lower potential than galantamine. However, considering that naturally occurring inhibitors are generally multicomponent and work with synergistic effects, the obtained findings indicate the potential pharmacological value of the species. It has been previously reported in the literature that L. salmonicolor has anticholinesterase activity[34]. In addition, it has been reported that the AChE inhibition value of a different Lactarius species, Lactarius deliciosus, is 24.80 µg/mL, and the BChE inhibition value is 51.24 µg/mL[26]. When compared to these values, it is seen that the anticholinesterase effect of L. salmonicolor is weaker. These differences may vary depending on the metabolite diversity between species, collection time, extraction protocol, and polarity of the solvents used. Modulation of the cholinergic system is considered an important target in the treatment of various neurodegenerative diseases, especially Alzheimer's disease[35]. In this context, the inhibitory activity of L. salmonicolor on cholinesterase enzymes indicates that the species is a biological source that can be evaluated in future studies to develop natural anticholinesterase agents. However, advanced phytochemical analyses and biological tests of isolated compounds are needed to reveal the mechanisms of these effects at the molecular level.
Antiproliferative activity
-
Mushrooms are among the important natural resources that show anticancer potential thanks to the various bioactive compounds naturally found in them[36]. In particular, metabolites such as polysaccharides (e.g., beta-glucans), phenolic compounds, terpenoids, sterols, and protein derivatives have been shown in many studies to suppress the proliferation of cancer cells and support programmed cell death (apoptosis)[37]. It has been reported that extracts obtained from some mushroom species have cytotoxic effects on different cancer cell lines, can stop the cell cycle, and inhibit tumor growth[38]. With these properties, mushrooms are considered promising biological materials, especially in studies aimed at the development of natural anticancer agents. In this study, the effects of L. salmonicolor on A549 lung cancer cells were examined, and the results obtained are presented in Fig. 2 as µg/mL.
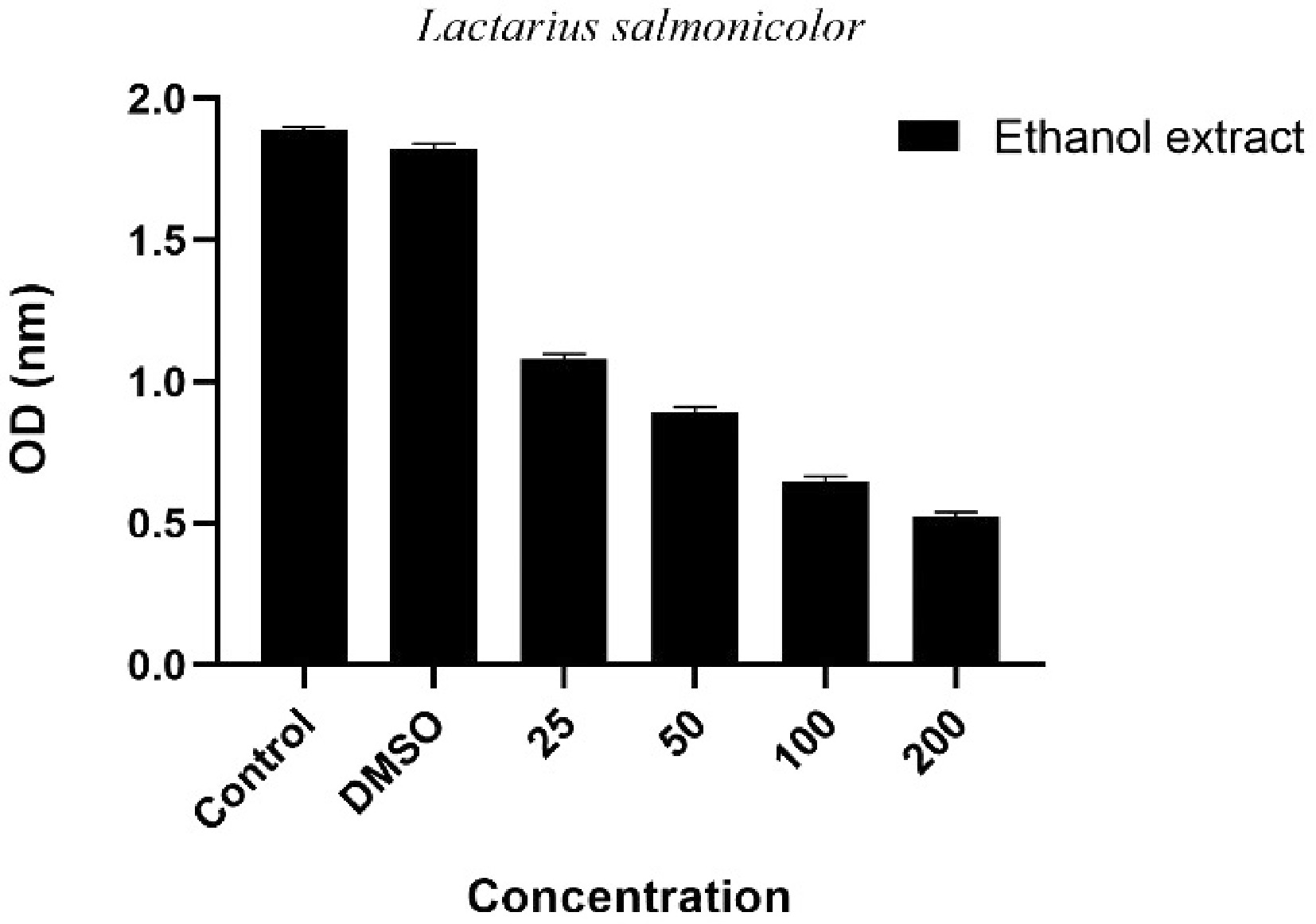
Figure 2.
Dose-dependent antiproliferative effect of Lactarius salmonicolor ethanol extract on A549 lung cancer cells. Control: Group kept only in the medium without any chemical substance applied; DMSO: Control group applied only to DMSO together with the medium; Extract applied groups: Experimental groups given mushroom extract at concentrations of 25, 50, 100, and 200 µg/mL; Values represent mean ± standard deviation (Std).
In this study, the antiproliferative effect of L. salmonicolor ethanol extract on A549 human lung adenocarcinoma cell line was investigated. The results obtained show that the extract reduces cell viability in a dose-dependent manner (Fig. 2). While the optical density (OD) value was at high levels in the control group, this value decreased significantly at the highest dose of 200 µg/mL. This situation reveals that the extract has a suppressive effect on cell proliferation. The fact that no significant difference was observed between the control group without extract, and the control group with only DMSO application confirms that DMSO has no toxic effect on cells at the tested concentration, and that the obtained inhibition is directly related to the mushroom extract. The gradual decrease in cell viability observed with increasing concentrations of the extract may be due to the cytotoxic effect of the phenolic compounds, terpenoids, and other secondary metabolites in its composition. Although there are limited studies in the literature on the anticancer potential of Lactarius species, it is reported that this effect may vary between species and cell line-based. For example, antiproliferative effects of Lactarius deliciosus have been reported on different cancer cell lines, but these effects generally vary depending on the extraction method, solvent type, and applied concentration[26,39]. The strong inhibitory effect of L. salmonicolor on A549 cells evaluated in this study indicates that this species may be a potential source of natural anticancer compounds. However, advanced molecular studies including apoptotic pathways, cell cycle analyses, and gene expression levels are required to understand the mechanism of this effect in more detail. In addition, it is important to conduct tests on normal cell lines to determine whether this effect shows selective toxicity.
Antimicrobial activity
-
Mushrooms are increasingly gaining attention as natural agents in the fight against microbial infections. The low molecular weight secondary metabolites they contain can suppress the growth of various pathogens, especially by exhibiting some antibacterial and antifungal properties[40]. It is reported that the compounds synthesized by these organisms have different mechanisms of action, such as disrupting cell wall synthesis, inhibiting protein production, or weakening membrane integrity[41]. These suppressive effects of mushroom extracts on various microbial strains allow them to be evaluated as a source of natural antimicrobial compounds. In addition, considering the resistance problems developing against traditional antibiotics, the discovery of new antimicrobial agents from fungi is of great importance[42]. In this study, the effects of L. salmonicolor against different bacterial and fungal strains were investigated, and the results obtained are shown in Table 3 as µg/mL.
Table 3. Minimum inhibitory concentration values of Lactarius salmonicolor.
A B C D E F G H J Lactarius salmonicolor extract 100 100 200 400 400 400 100 50 50 50, 100, 200, 400 µg/mL represents the lowest concentration that stops the growth of microorganisms. A: Staphylococcus aureus ATCC 29213, B: methicillin-resistant S. aureus (MRSA) ATCC 43300, C: Enterococcus faecalis ATCC 29212, D: Escherichia coli ATCC 25922, E: Pseudomonas aeruginosa ATCC 27853, F: Acinetobacter baumannii ATCC 19606, G: Candida albicans ATCC 10231, H: C. krusei ATCC 34135, J: C. glabrata ATCC 90030 In this study, the antimicrobial effects of L. salmonicolor extract against various Gram-positive and Gram-negative bacteria and yeast species were evaluated. The results obtained reveal that this fungus species has a remarkable inhibitory potential, especially against some pathogenic microorganisms. When evaluated in terms of minimum inhibitory concentration (MIC) values, the lowest MIC value of 50 µg/mL against Candida glabrata and C. krusei strains suggests that L. salmonicolor may have a strong antifungal effect. A relatively low MIC value of 100 µg/mL was also observed against the C. albicans strain. This indicates that natural products of fungal origin can be used as alternative treatment agents in fungal infections such as candidiasis. The MIC value was determined as 100 µg/mL against gram-positive bacteria such as Staphylococcus aureus and its methicillin-resistant strain S. aureus MRSA, and these values indicate that the fungal extract may also be effective against resistant bacterial species such as MRSA. This finding is quite important in terms of evaluating natural products as alternative antimicrobial agents in today's world, where antibiotic resistance is rapidly spreading. A higher MIC value of 200 µg/mL was observed against Enterococcus faecalis, suggesting that the spectrum of action may vary depending on the type of microorganism. Higher MIC values were recorded against Gram-negative bacteria. The MIC value was determined as 400 µg/mL for Escherichia coli, Pseudomonas aeruginosa, and Acinetobacter baumannii strains, and this shows that the effect of the extract is limited. It is known that natural compounds are generally less effective against hospital-acquired pathogens that can show multidrug resistance, especially P. aeruginosa and A. baumannii. In this context, the limited effect of L. salmonicolor against these bacteria even at high concentrations suggests that the antimicrobial compounds in the extract may have difficulty in passing the outer membrane structure of Gram-negative bacteria. These findings are generally consistent with the literature reported by Dülger et al.[43] that L. salmonicolor exhibits antimicrobial activity against various microorganisms such as Escherichia coli, Proteus vulgaris, Staphylococcus aureus, Corynebacterium xerosis, Bacillus cereus, Bacillus megaterium, and Mycobacterium smegmatis. However, the spectrum of microorganisms used in this study was kept wider, and the evaluation was made especially against resistant strains. Therefore, this study highlights the broad-spectrum antimicrobial potential of Lactarius salmonicolor, and suggests that future biodirected fractionation studies may contribute to the development of more effective natural antimicrobial agents by isolating active compounds.
-
This study evaluated the antioxidant, anticholinesterase, antiproliferative, and antimicrobial properties of L. salmonicolor species with a holistic approach. The findings show that this mushroom has a remarkable potential in terms of various biological activities. Antioxidant analyses revealed that the total antioxidant capacity was limited, and the oxidant load was relatively high, suggesting that the species may have a different chemical profile in terms of oxidative stress. Anticholinesterase activity results revealed a certain inhibitory effect on enzymes associated with neurodegenerative diseases such as Alzheimer's, and although this effect was lower than reference substances such as galantamine, it is valuable in terms of naturally derived synergistic compounds. In antiproliferative tests, the mushroom extract showed a dose-dependent cytotoxic effect on the A549 lung cancer cell line, and significantly reduced cell viability. This situation can be explained by the contribution of bioactive metabolites, especially phenolic and terpenoid compounds. In terms of antimicrobial activity, L. salmonicolor was effective especially against Candida species and some Gram-positive bacteria with low minimum inhibitory concentration (MIC) values, while it showed limited activity against Gram-negative bacteria at higher concentrations. These findings reveal that the antimicrobial spectrum of the fungus varies according to the microorganism species and that the outer membrane structures may be determinant in this activity. When evaluated in general, L. salmonicolor has significant potential in terms of natural product research, pharmaceutical biotechnology, and food supplement development with its versatile biological activities. Isolated compound analyses, fractionation studies, molecular mechanism studies, and in vivo bioavailability tests to be conducted on this species in the future will contribute to the acquisition of new findings that will enable the evaluation of the fungus in therapeutic applications.
This research has been funded by the Science Committee of the Ministry of Science and Higher Education of the Republic of Kazakhstan (Grant No. AP19676907).
-
The authors confirm their contributions to the paper as follows: study conception and design, data collection, analysis and interpretation of results, draft manuscript preparation: Khassanov VT, Sevindik M. Both authors reviewed the results and approved the final version of the manuscript.
-
The data that support the findings of this study are available upon request from the corresponding author.
-
The authors declare that they have no conflict of interest.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Khassanov VT, Sevindik M. 2026. Evaluation of antioxidant, anticholinesterase, antiproliferative, and antimicrobial activities of the ethanol extract of Lactarius salmonicolor. Studies in Fungi 11: e005 doi: 10.48130/sif-0026-0003
Evaluation of antioxidant, anticholinesterase, antiproliferative, and antimicrobial activities of the ethanol extract of Lactarius salmonicolor
- Received: 13 October 2025
- Revised: 10 December 2025
- Accepted: 25 December 2025
- Published online: 28 February 2026
Abstract: This study aimed to evaluate the antioxidant, anticholinesterase, antiproliferative, and antimicrobial activities of Lactarius salmonicolor species. Antioxidant capacity was quantitatively determined using Rel Assay commercial kits, and total antioxidant status (TAS) was 3.844 mmol/L, total oxidant status (TOS) was 10.452 µmol/L, and oxidative stress index (OSI) was 0.272. In the anticholinesterase activity assay, the mushroom extract inhibited acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) with IC50 values of 58.43 and 88.45 µg/mL, respectively. In antiproliferative activity evaluations, L. salmonicolor extract reduced cell viability in the A549 human lung cancer cell line in a dose-dependent manner and showed a significant cytotoxic effect at high concentrations. In antimicrobial tests, the mushroom extract showed notable antifungal activity against Candida glabrata and C. krusei strains with a low MIC value of 50 µg/mL. In addition, it exhibited remarkable antibacterial activity against Staphylococcus aureus and methicillin-resistant S. aureus MRSA strains with a MIC value of 100 µg/mL. However, its activity against Gram-negative bacteria was comparatively limited. Overall, the findings indicated that L. salmonicolor is a potential biological source that can be evaluated in research for the development of natural antioxidant, antimicrobial, anticancer, and neuroprotective agents with its multifaceted biological activities.













