-
Root and tuber crops (RTCs), including potato (Solanum tuberosum), cassava (Manihot esculenta), sweet potato (Ipomoea batatas), yam (Dioscorea spp.), and taro (Colocasia esculenta), are globally significant staple foods, occupying an indispensable position, particularly in developing countries of the tropics, and sub-tropics[1]. RTCs demonstrated multiple advantages in playing a critical role in global food security. Firstly, these crops exhibit remarkable adaptability, thriving across diverse climate zones, from productive lands in tropical regions to marginal lands subject to drought, salinity, and high altitude[2]. Secondly, RTCs are characterized by high and stable yields, which effectively meet food and nutritional demands, containing significant levels of Vitamin A, Vitamin C, dietary fiber, and protein, thus playing an important role in diversifying diets and mitigating micronutrient deficiencies, especially within resource-constrained communities[3]. Thus, RTCs play a pivotal role in global food security. They provide essential energy and nutrition for human consumption, and offer significant economic and environmental benefits within diverse agricultural ecosystems[4]. For instance, the acreage and production of cassava and sweet potato are continually increasing globally, particularly in developing countries, where these crops have become primary food sources for many communities[4]. Additionally, crop residues from RTCs (such as peels, leaves, and stems) can be utilized as animal feed, further enhancing the efficiency of agricultural resource use[5].
Potato and cassava respectively, represent RTCs produced from temperate and tropical regions, which are especially crucial to global food security. Potato is the world's fourth-largest staple crop, and its high yield and cold tolerance enable cultivation across a wide range of climatic conditions, including the tropics[6]. Cassava, conversely, is renowned for its tolerance to low soil fertility and high resilience to pests and diseases[7], leading to its widespread cultivation in the lowland tropics in Southeast Asia, Africa, and South America[8]. These two crops not only serve as primary sources of carbohydrates for billions of people, but also play a critical role in addressing climate change and food crises.
Tuber crops are not only key crops in the global food security system, but also play a significant role in agricultural carbon cycling and climate change mitigation. Agricultural ecosystems incorporating tuber crops can significantly enhance the soil organic carbon pool through the adoption of management practices, such as conservation tillage and cover cropping[9]. By effectively transporting and storing photosynthetic products in underground tubers or roots, these crops achieve considerable carbon accumulation and possess long-term carbon storage potential[10]. High-yielding root and tuber crops like cassava have a strong capacity for carbon assimilation and allocation, enabling the formation of relatively stable carbon pools in root tissues, thereby enhancing the carbon sink function of farmland ecosystems[11]. Potato exhibits a significant increase in biomass and tuber yield under elevated CO2 conditions, suggesting its strong carbon sequestration potential in high-carbon environments[12]. These practices can increase crop yield and enhance soil resilience to extreme climate events. By leveraging the multifunctional roles of tuber crops within ecological agriculture—for instance, supporting cropping system diversification, reducing reliance on chemical fertilizers, and unlocking their potential for soil remediation[13]—it is possible to further advance the circular economy across the agricultural sector. This contributes substantially to building climate-resilient agriculture and achieving carbon neutrality goals[14].
Although intensive RTC production systems can increase yield, the high energy consumption and fertilizer inputs often lead to significant greenhouse gas emissions and carbon footprints. Thus, future efforts should combine genetic improvement—such as optimizing carbon assimilation and transport pathways—with systemic management strategies, such as precision fertilization and biochar application, to fully realize the potential of RTCs in global carbon reduction and sustainable agriculture.
-
RRCs serve as vital global food sources and contribute significantly to terrestrial carbon sequestration. In 2025, their worldwide production maintains an upward trend, driven by yield improvements in major producing countries. These crops exhibit high energy density (297–525 kcal per 100 g) and substantial carbon storage potential, primarily due to elevated starch content (11.8–31.4 g per 100 g)[15]. Moreover, crop residues enhance soil organic carbon levels, thereby reducing CO2 emissions. Reduced- and no-tillage systems for potatoes, for instance, enable additional annual soil carbon sequestration on a global scale, mitigating CO2 release[16]. Evidence indicates that approximately 20% of root-derived carbon inputs enter non-rhizosphere soil and engage in long-term cycling, underscoring the pivotal role of roots in carbon sequestration[17]. From the source–sink–flow framework, the carbon sequestration advantages of root and tuber crops manifest as follows. Source strength: leaves display high photosynthetic efficiency, effectively fixing atmospheric CO2. Sink capacity and location: the underground sinks (tubers or roots) feature a high harvest index (e.g., 0.8–0.9 for potatoes), exceeding that of many cereals (e.g., ~0.5 for wheat)[18]. This configuration directs photosynthates straight to the soil, forming stable carbon reservoirs. Flow efficiency: transport pathways from source to sink are shorter, with sinks providing positive feedback to sustain leaf photosynthetic capacity[18]. Carbon persistence: deep roots deliver carbon to deeper soil layers (0–2 m), where physical protection promotes stability[17]; in contrast, cereal carbon inputs concentrate in surface layers and are readily removed during harvest[18].
Thus, RTCs achieve efficient photosynthate allocation through coordinated source-sink-flow dynamics. Their underground sinks facilitate direct carbon transfer to the soil, enhancing sequestration durability and stability. Growth monitoring of poplars showed that during the 1 to 3 year observation period, the conversion efficiency of belowground carbon inputs (e.g., fine roots, root exudates) to soil organic carbon (SOC) reached 76%, while that of aboveground carbon inputs was only 9%[19]. Storage organs such as tubers and taproots exhibit low metabolic activity, and slow decomposition rates of nutrients and carbon substances. Unharvested tuber residues and deep roots, as underground carbon storage pools of plants, contain carbon substances that are not easily mineralized rapidly, and can serve as stable carbon pools[19,20].These attributes showed a unique value in agricultural ecosystem carbon sinks in the following areas. Soil carbon storage: RTCs possess extensive root systems that increase soil organic carbon content. For example, reduced- and no-tillage systems, widely adopted in root and tuber cultivation, markedly enhance soil carbon sequestration. Low carbon emissions: carbon emissions during growth and processing remain relatively low. For instance, potatoes exhibit a carbon emission factor of 0.17–0.23 kg CO2 equivalent per kg of food, lower than that of many cereal crops[21]. Strong adaptability: RTCs adapt well to diverse environmental and soil conditions. They grow across various ecosystems and thus contribute to carbon sequestration over broader regions.
Underground carbon allocation and stability are considered as a unique trait of RTCs; they efficiently partition a substantial portion of photo-assimilated carbon to underground organs (sink), creating a significant carbon sink that contributes to soil organic carbon (SOC) pools. This allocation is driven by strong sink strength in tubers and roots. For instance, a key enzyme, H+-pyrophosphatase (IbVP1) in sweet potato, enhances carbon flux from leaves to storage roots by promoting phloem loading and long-distance sucrose transport[22]. In potatoes, elevated CO2 concentrations stimulate net photosynthesis, leading to increased aboveground and underground biomass accumulation, which improves harvest indices and irrigation water use efficiency[23]. A considerable amount of photosynthetically fixed carbon is released into the soil through root exudates, shaping microbial communities and further stabilizing carbon in the rhizosphere[24]. The stability of this underground carbon is a crucial aspect. Carbon incorporated into tubers and roots, especially as starch, and released via exudates, can contribute to more stable soil carbon fractions compared to aboveground biomass, which decomposes rapidly[19]. Furthermore, genetic studies on potatoes using modified and wild-type lines show that carbon allocation patterns from source leaves to tubers are a fundamental and regulated process, ensuring carbon is directed to underground sinks[20]. This effective underground partitioning and subsequent stabilization underscore the potential of RTCs in climate-smart agriculture by enhancing soil carbon sequestration.
Aboveground photosynthetic efficiency and carbon dioxide fixation in RTCs
-
RTCs are emerging as critical players in agricultural carbon sequestration. Their unique physiological characteristic is the high partitioning efficiency of photosynthetically fixed carbon towards dense, starch-rich, and recalcitrant underground storage organs. This underground allocation strategy results in substantial carbon loading into the soil system, where the carbon is more chemically and physically stable than ephemeral aboveground biomass. Consequently, RTCs offer a vital pathway for medium/long-term carbon storage, contributing significantly to climate change mitigation efforts.
In terms of carbon fixation capacity, potatoes' core advantage lies in the efficient allocation and long-term retention of carbon to underground sinks. During the tuber bulking stage, 35%–45% of fixed carbon is directionally allocated to tubers and roots. Harvested tubers alone achieve carbon sequestration of 1,500–3,000 kg C ha−1, while unharvested residues (e.g., cull tubers and stolons) contribute an additional 500−1,000 kg C ha−1. These residues, rich in crystalline amylose (accounting for 40%–60% of total starch), decompose at a rate of only 20%–30% per year, with a mean residence time of 2–5 years, significantly enhancing the stability of the soil organic carbon (SOC) pool. Scenario simulations indicate that under optimized management (maximum carbon sequestration + minimum emissions), the carbon flux of potato production can reach 12,830.567 kg CO2eq ha−1·season−1; even under average management conditions, a weak carbon sink effect of −90.703 kg CO2eq ha−1·season−1 is maintained[25]. As key RTCs, potatoes possess a distinct and efficient photosynthetic mechanism while demonstrating substantial carbon fixation potential, serving as a crucial contributor to the carbon sink in agricultural ecosystems. Classified as C3 plants, potatoes rely on the Calvin-Benson-Bassham cycle for CO2 assimilation, with ribulose-1,5-bisphosphate carboxylase/oxygenase (Rubisco) as the core enzyme. Though Rubisco mediates carbon fixation, it exhibits inherent kinetic limitations—specifically, a low specificity factor for CO2 over O2 (Sc/o ≈ 80–100)—leading to photorespiration losses of up to 25%–30% under warm, low-humidity conditions. Nevertheless, at the optimal temperature range of 15–25 °C, potatoes maintain a net photosynthetic rate of 10–20 µmol CO2 m−2·s−1, with peak photosynthetic activity observed during the mid-season vegetative growth stage[12]. Elevated CO2 (eCO2) effectively compensates for Rubisco's limitations: at concentrations of 800 µmol·mol−1, eCO2 not only enhances photosynthetic rate by increasing radiation use efficiency (RUE) and transpiration efficiency (TE), but also suppresses photorespiration. Under heat stress eTeCO2 (24 °C + 800 μmol·mol−1 CO2), eCO2 increased the yield of Favorita by 182% and that of Zhongshu 5 by 370% compared with the pure high-temperature group. Additionally, the tuber number and weight of Zhongshu 5 under eCO2 treatment were both superior to those of the control group[26].
Furthermore, potatoes exhibit strong resilience in carbon fixation against environmental stressors. Under high ozone concentrations, eCO2 sustains carbon fixation by reducing stomatal O3 flux. In response to elevated temperature (eT)-induced inhibition of carbon fixation (e.g., an 83% yield reduction in Favorita), the combined treatment of eCO2 and eT (eTeCO2) alleviates such negative effects by maintaining photosynthetic capacity, and optimizing biomass partitioning to tubers[12,26]. When combined with adaptation measures (e.g., adjusting planting windows and selecting suitable varieties), eCO2 enables a 9%–20% increase in global potato yield. This further confirms potatoes' critical role in agricultural carbon sequestration, highlighting the core value of RTCs in climate-smart agriculture[27].
Cassava also exhibits strong carbon sequestration capacity—it is often classified as a C3–C4 intermediate plant. This classification is based on two key traits: first, its low photorespiratory CO2 compensation point (Gamma, Γ ≈ 25–50 micromoles per mole, μmol mol−1); second, an efficient CO2 recycling mechanism located in the palisade mesophyll tissue[28]. These features enable cassava to exhibit enhanced photosynthetic performance in drought-prone environments. The correlation between temperature and maximum net photosynthetic rate (Amax) in cassava has been well-documented. Citing data from El-Sharkawy et al.[29], research indicates that under warm climatic conditions (mean temperature of 24 °C ranging from 17–30 °C), cassava is capable of achieving maximum net photosynthetic rates (Pnmax) of up to 35 μmol CO2 m−2·s−1. Furthermore, the light saturation point (Isat) necessary to achieve these rates was reported to be high, exceeding 1,800 μmol photons m−2·s−1[30]. In high-yield cassava cultivation systems, the annual carbon fixation capacity reaches 8–12 t of carbon per hectare (tC ha−1), which is equivalent to 29–44 t of CO2 per hectare (t CO2 ha−1). This carbon fixation supports fresh root yields of 15–30 t per hectare (t fresh roots ha−1) over a 9–12 month growing period[10]. Field trials were conducted to assess genetic diversity in photosynthesis (upper canopy net photosynthetic rate), productivity (dry root yield), and their interrelationship, the result suggesting that high photosynthetic capacity can serve as a key selection trait for high-yield cassava[31].
Eight cassava cultivars were used in CO2 enrichment conditions (FACE), to evaluate root sink strength when photosynthesis increases with elevated atmospheric CO2 concentrations—a key consideration for yield improvement as enhanced photosynthetic efficiency requires sufficient sink capacity to avoid carbohydrate accumulation in leaves and photosynthetic suppression—revealing that elevated CO2 increased photosynthesis by 31% without photosynthetic downregulation over the growing season, thereby boosting fresh and dry root biomass by 27% and 37%, respectively[32]. This, all while also improving intrinsic water use efficiency, and leaving leaf protein content as well as leaf and root cyanide concentrations unaffected, suggesting that higher cassava yields can be expected with future increases in atmospheric CO2[32].
As an important food crop, sweet potato exhibits considerable carbon fixation capacity (5–9 t C ha−1·yr−1) and plays a significant role in agricultural carbon sinks[33]. This C3 plant exhibits a photosynthetic rate of 15–25 µmol CO2 m−2·s−1 under field conditions, achieving optimal performance at 25–30 °C and moderate irradiance[34]. Its Rubisco kinetics are comparable to those of potato[35]. With a growing cycle of 120–180 d and a fresh root yield of 15–25 t·ha−1, its net annual carbon sequestration capacity ranges from 18–33 t CO2 ha−1[36]. Its carbon fixation capacity is influenced by environmental factors (temperature, light, and CO2 concentration) and genetic characteristics; in the context of global climate change, particularly under elevated CO2 concentrations, sweet potato demonstrates substantial potential for enhanced carbon sink capacity. Studies have shown that sweet potato can allocate 11.2%–56.4% of photosynthates to storage roots during its growth cycle, and the final root yield is significantly correlated with photosynthate partitioning (r = 0.69–0.87)[37]. Similar to other tuber crops, under elevated CO2 (eCO2) at 700 ppm, the photosynthetic rate is significantly enhanced by 30%–50%, primarily mediated by the suppression of photorespiration[38]. This response underscores sweet potato's adaptive potential to eCO2, where eCO2-induced reductions in stomatal conductance (gs) conserve water. Similarly, photosynthetically efficient and climate-smart sweet potato varieties were tested by measuring photosynthesis under ambient (400 ppm), and gradient elevated CO2 concentrations (up to 1,000 ppm), a constant temperature of 30 °C, and the optimal treatment of a photosynthetic photon flux density (PPFD) of 1,500 μmol m−2·s−1[39].
A comparison of photosynthetic and carbon sequestration of RTCs and C4 crops
-
RTCs, encompassing staples such as potato, cassava, sweet potato, taro, and yam, play a critical role in global food security, particularly in tropical regions in developing, or least developed countries in sub-Saharan Africa, where they form dietary foundations and support rural economies[40]. Their photosynthetic and carbon sequestration efficiencies are key to understanding productivity, resilience to climate change, and potential contributions to mitigating greenhouse gas emissions. Mainstream C4 crops, including maize (Zea mays), sorghum (Sorghum bicolor), and sugarcane (Saccharum officinarum), are often benchmarked against RTCs due to their evolved adaptations for higher efficiency in carbon fixation[41]. RTCs, predominantly utilizing the C3 photosynthetic pathway, exhibit lower photosynthetic and carbon sequestration efficiencies compared to C4 crops. In terms of light-use efficiency (LUE), C4 crops (including maize, sorghum, and sugarcane) clearly outperform RTCs (such as cassava, sweet potato, potato, and yam). This aligns with theoretical expectations, theoretical maximum photosynthetic efficiency for C3 plants is approximately 4.6% vs 6.0% for C4 plants, under 30 °C and current atmospheric CO2 levels, with field-realized efficiencies showing C4 crops achieving up to 20%–30% higher carbon fixation due to minimized photorespiration[42,43]. Radiation use efficiency (RUE) reinforces these trends, with C4 crops efficiently converting radiation into dry matter compared to RTCs. Measured net photosynthetic rates confirm this gap: cassava reaches 40–50 μmol CO2 m−2·s−1 under saturating light, sweet potato 21.4–40.3 μmol CO2 m−2·s−1, and yam 20–30 μmol m−2·s−1 (with up to a 68% increase under elevated CO2), while C4 sorghum and maize typically range 32–50 μmol CO2 m−2·s−1[44−46]. For carbon sequestration, C4 plants exhibit higher CO2 fixation fluxes (387.6 vs 201.0 mmol·gDW−1·h−1 for C3), and annual soil carbon inputs (~3.3 Mg C ha−1·yr−1), compared to RTC root-derived inputs of ~2.2 Mg C ha−1·yr−1[47,48]. Although cassava displays intermediate C3–C4 traits that partially mitigate photorespiration[46], RTCs generally remain less efficient, as shown in Table 1.
Table 1. A comparison of photosynthetic and carbon sequestration of RTCs and C4 crops.
Crop type Specific crop Light-use efficiency (%) Net photosynthetic rate
(μmol CO2 m−2·s−1)Radiation use efficiency (RUE, g DM MJ−1 PAR) Photosynthetic conditions/
characteristics descriptionCarbon fixation notes Ref. RTCs (C3–C4 intermediate) Cassava 3.6–4.1 17.8–35 (typical field); 40–50 (Saturating light [>1,800 μmol m−2·s−1]) 0.22–1.64 (varied by study, cultivar, season) Low photorespiration;
heat-tolerantHigh carbon allocation to tubers [49,50] RTCs (C3) Sweet potato ≈4.0 15–35 (typical field); 21.4–40.3 (380−1,000 ppm CO2, 1,500 μmol m−2·s−1 PPFD) 0.8–2.5 (varied by
field experiment, cropping system)Closely related to cropping system, planting pattern,
and water contentLow photorespiration; heat tolerant [34,51] RTCs (C3) Potato 3.0–4.0 15–35 (influenced by variety, temperature, moisture); 30–35 (upper limit) 1.6–1.8 (multiple reviews; solar-radiation basis) Pn increases significantly with warming; C3 type, Pn rises with elevated CO2 (indoor tests) Moderate WUE [52−54] RTCs (C3) Yam ≈3.5 20–30 (up to 68% increase at elevated CO2) 0.8–1.8 (varied by experimental conditions) Mostly C3 type, responsive to elevated CO2; some reports
show ~60%–70% Pn increase under high CO2Shade-tolerant, moderate PN [55,56] C4 Maize 4.6–4.9 35–50 (field high-quality/superior cultivar; decreases under moderate/low humidity) 1.6–3.5 (the value is higher under special high-density/ideal conditions) High photosynthetic efficiency; Higher RUE under special stress/emergency conditions High PN and WUE [47,57] C4 Sorghum ≈4.7 32–46 (typical field/
greenhouse)1.4–4.9 (varied by cultivar and water,
3–5 in early stage)High photosynthetic efficiency; maintains photosynthetic and water use efficiency (WUE) under high temperature and drought High drought tolerance [58,59] C4 Sugarcane ≈5.0 30–50 (typical field);
40–50 (midday values)1.6–3.0 (1.6–2.0 in most field studies; cultivar/season led to upper limit near 3) High photosynthetic efficiency; robust under drought; energy sorghum (bioenergy type) has higher RUE than grain sorghum Highest biomass production [60,61] -
In tuber crops, carbon sequestration efficiency remains intrinsically constrained by core physiological and molecular bottlenecks that limits both carbon fixation and subsequent allocation to underground sinks. At the fixation level, the C3 pathway relies on ribulose-1,5-bisphosphate carboxylase/oxygenase (Rubisco), an enzyme with inherently low catalytic turnover (Kcat = 3 s−1) and strong competitive inhibition by O2[62]. These molecular properties result in substantial photorespiratory losses, often removing 20%–50% of previously fixed carbon, and forming a primary biochemical basis for potato's low carbon-sequestration capacity[63]. Compounding this limitation, Rubisco activase (Rca), the chaperone maintaining Rubisco in an active conformation is acutely sensitive to heat stress[64]. Field-level temperatures commonly experienced in potato-growing regions are sufficient to deactivate Rca, causing rapid declines in Rubisco activation status and carbon assimilation. The lack of heat-stable Rca isoforms, coupled with incomplete optimization of Rubisco assembly factors such as Raf1, Raf2, BSD2, Hsp70, and Cpn60 further constrain the amount of functional Rubisco that can be maintained under fluctuating field conditions[65].
Source-sink imbalance and feedback inhibition
-
Beyond fixation, RTCs, such as potato's low-carbon-sink performance reflects a mismatch between photosynthetic 'source' strength and the biochemical and developmental capacity of tubers to absorb incoming carbon flux[66]. When photosynthetic enhancement is not accompanied by proportional increases in sink demand, accumulating sugars in leaf tissue induce sugar-signaling and feedback inhibition pathways that suppress the Calvin cycle and limit overall productivity—an issue widely observed in potato under high light or elevated CO2[67]. This illustrates the core 'source–sink–energy optimization dilemma': without concurrent enhancement of sink strength, improvements in carbon fixation cannot translate into durable increases in carbon storage. Potato therefore suffers from insufficient sink-related genetic tuning, including limited activity of AGPase for starch biosynthesis[68], suboptimal phosphoenolpyruvate carboxylase (PEPC)-mediated CO2 concentration, and restricted phloem unloading capacity. Transport proteins such as SUT, SWEET, and tuber-specific transporters (e.g., StTST2) are often expressed below the levels required for maximal carbon flux into developing tubers, creating another structural barrier to carbon sequestration[69].
Developmental and regulatory constraints
-
Developmental and regulatory constraints further limit low-carbon performance. Tuber initiation, sink differentiation, and carbon-allocation transitions are controlled by tightly interconnected miRNA regulatory networks, including miR156 and miR172[70]. These networks remain largely unoptimized in modern potato breeding, preventing precise manipulation of sink formation and carbon partitioning. Moreover, limited deployment of genome-editing tools, such as CRISPR-Cas systems tailored for regulatory RNA editing, hinders efforts to fine-tune these networks for enhanced sink stability.
In RTCs, achieving a high-efficiency source–sink balance is constrained by regulatory feedback on assimilation, transport, and storage. For example, overloading the source without matching sink capacity may lead to sugar accumulation, which inhibits key metabolic enzymes. One mechanism that mitigates this is the redox regulation of ADP-glucose pyrophosphorylase (AGPase): in tubers, AGPase exists in an inactive dimeric form when sink supply is disrupted, but reducing conditions can restore its activity and drive starch synthesis, linking sucrose import with storage flux[71]. At the transport level, the tuberization signal SP6A physically interacts with SWEET11, restricting sucrose efflux into the apoplast, and thereby promoting symplastic unloading into the developing tuber[72,73]. Computational modeling of this interaction further supports that SP6A-mediated suppression of SWEET11 enhances sucrose flux to stolon tips, increasing sink delivery efficiency[74]. During tuber development, there is also a coordinated metabolic and structural switch; callose metabolism is reprogrammed, and plasmodesmata conductance is increased to strengthen symplastic connectivity and reinforce sink strength[75]. Meanwhile, microRNA156 (miR156), shown to be mobile in grafts and to regulate SPL transcription factors, modulates tuber crop architecture and tuberization under photoperiod control; its overexpression alters miR172 levels through the SPL module, implicating a miR156–SPL–miR172 network in source–sink signaling[76]. Furthermore, hormonal signaling via StBIN2 kinase can influence sink strength by regulating sucrose transporter genes including SWEET11 and SUT3, integrating sugar sensing with developmental signaling[77]. Based on the above, tuber and root crops regulate source-sink-flow energy efficiency via four core mechanisms: (1) the miR156/miR172 hierarchical network acts as an upstream developmental module, orchestrating tuber initiation, sink establishment, and carbon allocation under photoperiodic signals[70,76]; (2) StBIN2 kinase functions as a hormonal hub, phosphorylating key factors to bidirectionally regulate sugar transporters (e.g., SWEET11, SUT3) and metabolism at transcriptional/post-translational levels, converting developmental signals into carbon transport/utilization instructions[77]; (3) SP6A interacts with SWEET11 post-translationally to inhibit apoplastic sucrose efflux and promote symplastic unloading into tubers, directing carbon transport[72−74]; (4) AGPase redox activation couples sucrose import with starch biosynthesis, enhancing sink responsiveness and avoiding photosynthetic feedback inhibition[71]. Additionally, callose metabolism reprogramming boosts plasmodesmatal permeability to reinforce carbon flux channels[75].
These components together form a well-stratified, multi-pathway synergistic molecular regulatory network. With miRNA-mediated developmental regulation as the core upstream switch, and SP6A-SWEET11-mediated directional unloading and AGPase-mediated starch synthesis as the dual engines for yield formation, this network integrates hormonal/redox signaling, transporter-mediated carbon transport, and plasmodesmatal regulation. It stabilizes carbon flow allocation, alleviates photosynthate feedback inhibition, and ultimately achieves the overall optimization of source-sink-flow energy efficiency. However, this regulatory network remains under-optimized in most modern RTC cultivars, and its synergistic function is further constrained by inherent physiological bottlenecks.
Collectively, the combination of intrinsically inefficient carbon fixation, stress-sensitive Rubisco regulation, insufficient sink strength, weak carbon-transport capacity, and under-optimized developmental regulatory modules explain why RTCs maintain relatively low carbon-sequestration efficiency, and why its low-carbon agricultural performance remains below theoretical potential. Strategic, multi-target genetic engineering is therefore required to simultaneously elevate fixation efficiency, strengthen carbon sinks, and stabilize source–sink dynamics under field stress conditions.
Gaps in management of RTCs: from photosynthetic source to soil carbon sink
-
The application of low-carbon agricultural technologies in RTCs production remains limited, which directly restricts the realization of its carbon sequestration potential[78]. At the field management level, enhancing and stabilizing the soil carbon pool is essential for synergistically improving soil fertility and carbon sequestration capacity. Environmental stress, particularly water scarcity, also significantly limits carbon fixation. Studies have shown that under soil-water stress, carbon assimilation in potatoes follows a two-phase response: first, stomatal closure and reduced mesophyll conductance jointly limit the diffusion of CO2 to the chloroplasts; as drought intensifies, the activity of the photosynthetic apparatus itself declines[79]. Detailed limitation analysis indicates that mesophyll conductance is the most critical factor restricting carbon and water exchange in potatoes, with its importance exceeding that of stomatal factors across various soil moisture conditions[79]. Under severe drought, decreased mesophyll conductance accounts for approximately 40% of the decline in photosynthetic rate, while reduced Rubisco carboxylation capacity explains about 30%[80]. In the dryland regions of Northwest China, a major potato-producing area, continuous monocropping is common, while low-carbon practices such as crop rotation and green manure planting are underutilized. This not only leads to soil degradation, but also maintains high greenhouse gas emissions.
In contrast, the adoption of low-carbon rotation systems such as the Durum Wheat–Green Manure–Potato, has been shown to significantly increase soil carbon sequestration and reduce greenhouse gas emissions[81]. Although the application of organic fertilizer alone can increase soil organic carbon content, it may also accelerate the mineralization of organic matter, which is detrimental to the long-term stability of the carbon pool[82]. Between 2011 and 2021, potato production in China was characterized by excessive nitrogen and phosphorus inputs, insufficient potassium application, and limited adoption of low-carbon nutrient management technologies. As a result, greenhouse gas emissions from fields increased by 83.4%, while carbon sequestration efficiency remained low[83]. In summary, the combined application of organic and inorganic fertilizers, particularly balanced fertilization, proves more effective in increasing total organic carbon, easily oxidizable organic carbon, and mineral-associated organic carbon in the soil. This approach also significantly improves the carbon pool management index, thereby enhancing long-term soil carbon sequestration[82]. Additionally, the combination of plastic film mulching with biochar application has been demonstrated as an effective synergistic strategy. Film mulching helps conserve soil moisture and increase yield, while biochar, with its high specific surface area and stability, improves soil water and nutrient retention, and serves as a persistent carbon storage material. Research shows that this integrated measure significantly enhances water use efficiency and tuber yield in potato cultivation[84].
-
As mentioned above, RTCs, as staple crops feeding over one billion people, predominantly rely on the inefficient C3 photosynthetic pathway, resulting in substantial carbon loss through photorespiration, and limited CO2 fixation capacity under elevated temperatures and CO2 predicted in the future. Enhancing their carbon sequestration potential through genetic engineering and biotechnology represents a promising avenue to increase yield and contribute to atmospheric CO2 drawdown, as shown in Fig. 1. The overall ability of tuber crops to store carbon depends on the 'source–sink dynamic balance'. Leaves, as 'source' organs, conduct carbon fixation through photosynthesis; tubers/taproots, as 'sink' organs, convert sucrose produced by the 'source' organs into starch granules for storage. If genetic engineering boosts photosynthesis in leaves but does not improve the tuber's capacity to store carbon, sugars begin to accumulate in the plant. This buildup can trigger feedback inhibition, causing problems such as photoinhibition and sugar–signaling stress[85]. These stresses slow down the Calvin cycle and cancel out the benefits gained from stronger photosynthesis. This tight interdependence creates what scientists call a 'source–sink–energy' optimization challenge.
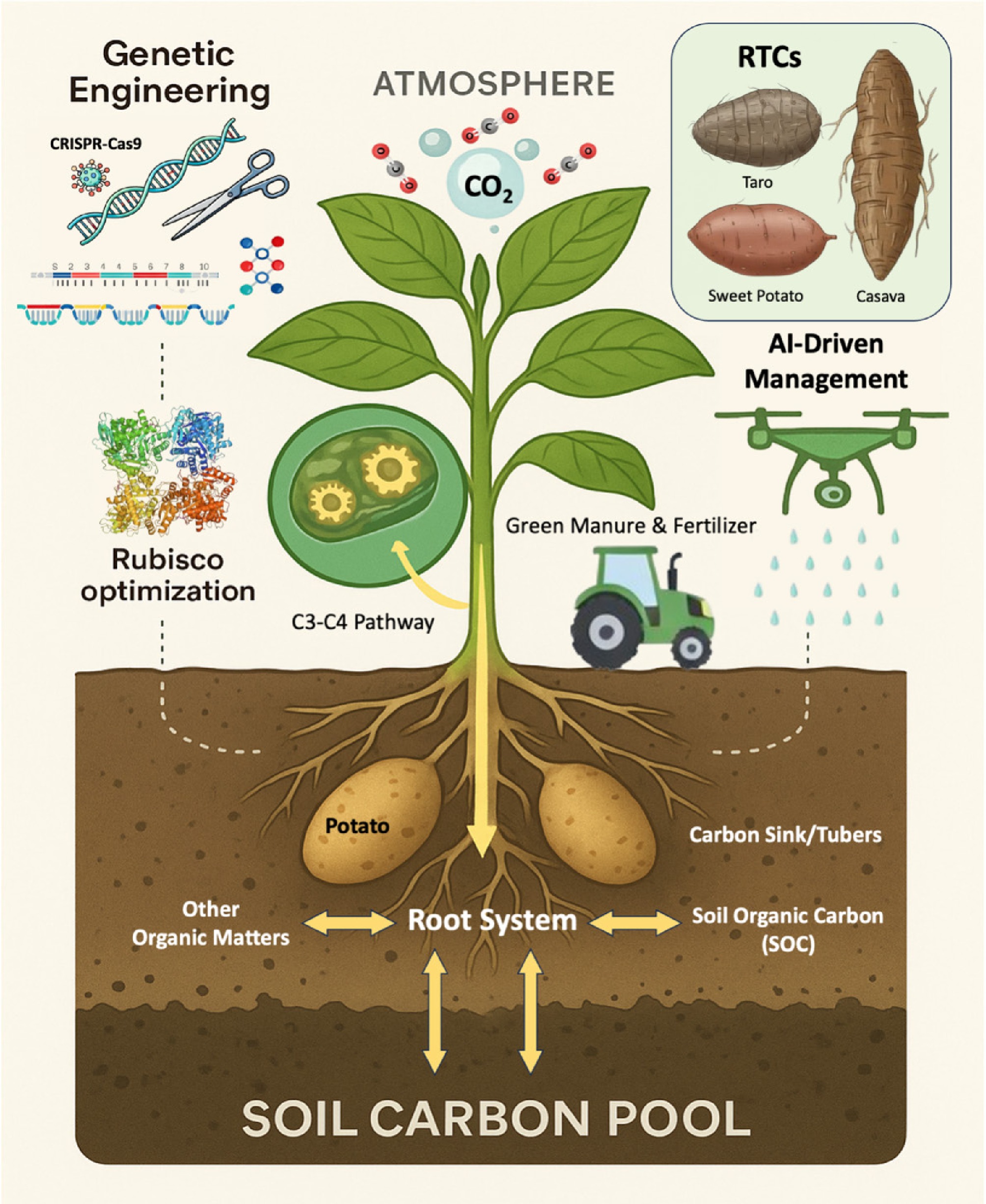
Figure 1.
Photosynthetic pathway of RTCs and their carbon sink potential. Genetic engineering includes a series of targeted molecular modification techniques, such as transgenesis, gene editing (e.g., CRISPR-Cas systems), RNA interference, gene overexpression, knockout, and knockdown, as well as transcriptional regulation via promoter engineering and regulatory element modification.
Comparative analysis of genetic improvement strategies for core enzymes
-
To overcome this, genetic improvement must target several systems at the same time. Important enzymes include Rubisco to improve carbon fixation, AGPase to increase starch-synthesis capacity, and PEPC to enhance CO2 use and energy efficiency. Low carbon fixation efficiency of Rubisco is a core issue, and its inherent oxygenase activity leads to substantial consumption of fixed carbon by photorespiration[86]. For this bottleneck, genetic engineering offers a feasible modification pathway. For example, the introduction of the GOC photorespiratory bypass into potato chloroplasts has successfully enabled the direct metabolism of part of the glycolate produced by photorespiration within chloroplasts with CO2 release, creating a localized high-CO2 environment around Rubisco[87]. This technology significantly reduces the photorespiration rate of transgenic plants by 6.59%–12.01%, increases the net photosynthetic rate by 7.02%–11.68%, and ultimately boosts the fresh weight of tubers by up to 10.34%–46.7%[88]. Additionally, progress has been made in attempting to replace potato's endogenous Rubisco with Type II Rubisco from the bacterium Rhodospirillum rubrum. Studies have confirmed that under a high CO2 concentration environment of 0.5%–1.5%, this modification allows the growth and tuber yield of potatoes to reach the level of wild-type plants, providing another technical route for the targeted optimization of Rubisco[89].
Beyond these specific modification cases, the genetic improvement of key enzymes (Rubisco, AGPase) in RTCs mainly relies on three mainstream genetic improvement strategies—site-directed mutagenesis, heterologous expression, and CRISPR-Cas-based gene editing—which exhibit distinct characteristics and application scenarios[89−91]. Site-directed mutagenesis enables precise fine-tuning of enzyme functional sites (e.g., Rubisco's CO2 binding specificity, AGPase's allosteric regulation) without introducing exogenous genes, showing high biosafety and suitability for mild improvement of existing cultivars, though it is limited by low positive mutation rates[90,91]. Heterologous expression of high-activity enzymes from cyanobacteria, C4 plants, or other species (e.g., Type II Rubisco from Rhodospirillum rubrum) can break the inherent enzymatic constraints of RTCs, achieving significant improvement in carbon fixation or starch synthesis efficiency, but faces challenges such as poor metabolic compatibility and complex approval processes[89]. Gene editing allows simultaneous modification of multiple homologous genes in polyploid RTCs (e.g., potato, sweet potato), realizing precise regulation of enzyme expression spatio-temporally (e.g., tuber-specific AGPase overexpression) with high efficiency and stability, yet it requires comprehensive genome information and strict off-target detection[91]. In practical breeding, site-directed mutagenesis is preferred for rapid variety improvement with high biosafety requirements, heterologous expression for breakthrough trait innovation, and gene editing for large-scale precision breeding, while combined strategies (e.g., mutation site screening + gene editing) can balance improvement effect and operational efficiency.
Personalized C4/CAM pathway engineering for major RTCs
-
Another primary strategy involves converting or supplementing the C3 pathway with elements of C4 or crassulacean acid metabolism (CAM)[92]. Overexpression of key C4 enzymes—phosphoenolpyruvate carboxylase (PEPC), pyruvate orthophosphate dikinase (PPDK), and NADP-malic enzyme (NADP-ME)—in mesophyll and bundle-sheath-like cells has shown promise in model species[93]. Concurrently, introducing cyanobacterial or red algal Rubisco with higher catalytic turnover rates, combined with carboxysome-like nanostructures can significantly reduce oxygenation side-reactions. Bypassing photorespiration via synthetic glycolate catabolic pathways further improves net CO2 assimilation in silico predictions[94]. Establishing functional carbon-concentrating mechanisms (CCMs) constitutes another pivotal approach. Coexpression of bicarbonate transporters (e.g., BicA/SbtA) and carbonic anhydrase at the chloroplast envelope elevate intra-chloroplastic CO2 concentrations, mimicking natural CCMs in algae and C4 plants[95]. Simultaneously strengthening sink strength in storage organs—through deregulation of ADP-glucose pyrophosphorylase (AGPase) via site-directed mutagenesis of allosteric sites or tuber-specific promoters—ensures fixed carbon is efficiently partitioned into starch, preventing feedback inhibition of photosynthesis.
Notably, the strategy of introducing C4/CAM pathway elements should be tailored to the genomic and photosynthetic characteristics of major RTCs[28,35,46]. As a C3−C4 intermediate crop, cassava (diploid genome, low photorespiratory CO2 compensation point (≈25–50 μmol mol−1) requires targeted enhancement of its inherent CO2 recycling mechanism (e.g., overexpressing native PEPC and NADP-ME genes) rather than de novo construction of C4 pathways[28,46]. For pure C3 crops, the tetraploid potato with complex genome and high photorespiration (25%–30% losses) benefits more from establishing simplified carbon-concentrating mechanisms (e.g., coexpressing C4 enzymes with tuber-specific promoters and carboxysome-like structures)[12,95]. Hexaploid sweet potato, characterized by strong sink strength for storage roots, should integrate C4/CAM elements with optimized sugar transporters (e.g., SUT1, StTST2) to avoid feedback inhibition of enhanced photosynthesis[37,69]. Such species-specific customization ensures the efficiency and feasibility of C4/CAM pathway engineering in RTCs.
Rhizosphere and carbon transport engineering for carbon sequestration
-
Beyond leaf-level interventions, rhizosphere engineering offers additional carbon sequestration opportunities. Enhanced root exudation of carboxylates and sugars, achieved by overexpressing malate dehydrogenase or sucrose transporters, stimulates soil microbial carbon fixation and aggregation[65]. Synthetic biology approaches introducing PHA synthesis pathways into roots may immobilize substantial carbon underground as biodegradable bioplastics. For instance, Rubisco subunits (rbcL and rbcS) form the core CO2-fixing enzyme, where engineering for improved kinetics can boost fixation rates[96]. Activators like ribulose bisphosphate carboxylase/oxygenase activase (RCA) and rubisco accumulation factor 1 (RAF1) ensures Rubisco's optimal assembly and activation, reducing photorespiration losses[90]. Calvin cycle regulators (phosphoribulokinase [PRK], glyceraldehyde-3-phosphate dehydrogenase [GAPDH], fructose-1,6-bisphosphatase [FBPase], sedoheptulose-1,7-bisphosphatase [SBPase]) optimize downstream reactions for efficient carbon flow[97]. C4 pathway genes (PEPC, PPDK, NADP-ME/NAD-ME, and carbonic anhydrase [CA]) enable spatial CO2 concentration, minimizing O2 interference[98]. Transporters like TPT facilitate photosynthate export, while sink-related genes (AGPase, starch synthase [SS], sucrose phosphate synthase [SPS]) enhance starch and sucrose accumulation in tubers[91]. In RTCs, these genes can be modified via transgenic strategies such as overexpression, or CRISPR technology, to increase biomass, yield, and soil carbon input, supporting sustainable agriculture under climate stress. By integrating multiplex CRISPR-Cas, GoldenGate-based multigene assembly, and rigorous field testing under elevated CO2, it is feasible to develop 'carbon-negative' potato, sweet potato, and casava varieties that not only exhibit higher productivity but also actively contribute to long-term soil carbon storage and climate change mitigation. Related genes mentioned above were listed in Table 2.
Table 2. Functional genes that may affect the carbon fixation efficiency.
Gene/ref. Function Potential effects in potato rbcL[96] Encodes the large subunit of Rubisco, catalyzing CO2
fixation in the Calvin cycle.Improved catalytic efficiency could increase net photosynthesis by 10%–20%, enhancing tuber yield and carbon sequestration. rbcS[99] Encodes the small subunit of Rubisco, influencing enzyme stability and specificity. Optimized variants reduce photorespiration, potentially boosting biomass accumulation in tubers under high temperatures. RCA[90] Ribulose bisphosphate carboxylase/oxygenase activase; removes inhibitors from Rubisco active sites. Overexpression maintains Rubisco activity during heat stress, leading to sustained CO2 fixation and larger tubers. RAF1[100] Rubisco accumulation factor 1; aids in Rubisco assembly
and folding.Enhances Rubisco biogenesis, improving overall photosynthetic rate and carbon partitioning to storage organs. PRK, GAPDH,
FBPase, SBPase[97]Catalyze and regulate steps in the Calvin cycle (e.g., PRK regenerates ribulose-5-phosphate; GAPDH reduces 3-PGA; FBPase/SBPase hydrolyze sugar phosphates). Coordinated upregulation accelerates carbon assimilation, increasing starch synthesis and tuber size by up to 15%–25%. PEPC[98] Phosphoenolpyruvate carboxylase; initial CO2-fixing enzyme in C4 pathway, producing oxaloacetate. Introduction enables C4-like metabolism, concentrating CO2 and reducing photorespiration losses for higher yields in arid conditions. PPDK[101] Pyruvate orthophosphate dikinase; regenerates PEP to
sustain C4 cycle.Maintains efficient C4 flux, potentially elevating CO2 fixation rates and biomass in potato leaves. NADP-ME/
NAD-ME[102]Malic enzymes; decarboxylate malate in bundle sheath cells, releasing CO2 for Rubisco. Facilitates CO2 delivery, improving photosynthetic efficiency and carbon allocation to tubers. CA[103] Carbonic anhydrase; hydrates CO2 to HCO3−, providing substrate for PEPC. Enhances C4 initiation, leading to reduced water loss and increased carbon gain in drought-prone potato cultivation. TPT[104] Triose phosphate translocator; exports photosynthates
from chloroplasts to cytosol.Optimizes carbon export, preventing feedback inhibition and promoting starch accumulation in tubers. AGPase, SS[91] ADP-glucose pyrophosphorylase and starch synthase;
key in plastidial starch biosynthesis.Deregulation increases tuber starch content, enhancing sink strength and overall plant carbon storage. SPS[105] Sucrose phosphate synthase; catalyzes sucrose synthesis
in cytosol.Upregulation improves photosynthate partitioning, supporting tuber growth and long-term carbon sequestration in soil. Carbon transport is equally important. Strengthening the activity of sugar transport proteins—such as the SUT and SWEET families—or key genes like cell wall invertase can improve phloem unloading and allow carbon to move more efficiently into the developing tubers. In addition, regulatory networks provide precise targets for improvement. Those miRNAs, such as miR172 and miR156 play important roles in tuber development and carbon allocation. Editing these pathways with advanced methods like CRISPR-Cas13 may offer a highly targeted way to boost carbon storage and improve sink development.
Optimizing Rubisco function and photorespiratory metabolic pathways through genetic engineering approaches, combining precision field water management to alleviate mesophyll conductance limitations, and complementing with soil carbon sequestration technologies centered on biochar and balanced fertilization constitutes a multi-level technical system from 'photosynthetic source', to 'soil carbon sink'. This system provides clear modification strategies and implementation pathways for systematically enhancing the carbon fixation and sequestration efficiency of potatoes. Redirecting metabolic resources toward carbon sequestration may alter the plant's source-sink dynamics, potentially creating competition for finite assimilates. Consequently, it is crucial to consider whether this reallocation might inadvertently compromise the potato's resilience to biotic and abiotic stresses, or diminish the nutritional quality of the tubers.
Enhancing carbon sequestration through improved management in RTCs
-
Beyond genetic improvement, multiple optimized technical practices must be used since they have been proven effective in enhancing the carbon sequestration capacity of tuber crops. In terms of soil management, the application of organic fertilizers and green manure can directly increase the soil organic carbon pool, stimulate root growth, and enhance soil carbon sequestration. For instance, manure application in potato cultivation has been shown to improve disease resistance and yield[106]. Further research indicates that the combined application of organic and inorganic fertilizers optimizes the allocation of photosynthetic carbon within the crop-soil system, significantly promoting carbon accumulation in sweet potato tubers[107]. Regarding cropping systems, the adoption of grain-tuber rotations (e.g., switching from wheat-maize to sweet potato) has been demonstrated to significantly reduce total carbon emissions and the carbon footprint per unit yield, thereby improving carbon use efficiency at the system level[108]. In protected cultivation environments, applying CO2 fertilization is a direct and effective method to enhance photosynthetic efficiency. For example, in a technology application by the Jiangmen Dual Carbon Laboratory, this practice increased sweet potato yield by up to 40% and shortened the growth cycle[109]. Simultaneously, the use of IoT technology for integrated intelligent management of light, temperature, water, and fertilizers in greenhouses can create optimal photosynthetic conditions for crops[110]. These exogenous management approaches provide feasible pathways for achieving low-carbon and high-output production in RTCs by synergistically optimizing the source–sink relationship.
The significance of enhancing RTCs carbon sequestration for tropics worldwide
-
In underdeveloped tropical countries, particularly in sub-Saharan Africa and Southeast Asia, RTCs are regarded as 'insurance crops', owing to their drought tolerance, adaptability to poor soils, and low management requirements[4]. They directly underpin food security for vulnerable households and children. As highlighted earlier, the leaf photosynthetic efficiency of RTCs is generally lower than that of typical C4 crops (e.g., sugarcane, maize, and sorghum)[111]. Nevertheless, their substantial underground root biomass and leaf litter can significantly enhance SOC when integrated with conservation agriculture practices, such as crop residue incorporation, organic fertilizer supplementation, and cover crop combinations[112]. Multiple field trials across Africa and Asia have demonstrated that RTC-based conservation agriculture can remarkably increase SOC stocks, improve soil structure, and boost crop yields[112]. However, this 'carbon sequestration effect' does not occur spontaneously: intensive cultivation of crops like cassava or their monoculture for industrial purposes (e.g., fuel ethanol production) may instead reduce SOC, as evidenced by the risk of land conversion to cassava-based biofuel feedstock in Vietnam[113]. While strategies derived from model organisms provide a theoretical framework, their translation to RTCs is significantly impeded by the complex, polyploid, and heterozygous nature of tuber genomes. Consequently, overcoming technical bottlenecks—such as genotype-dependent recalcitrance and phenotypic instability—remains a critical prerequisite for the scalable application of these technologies in agronomic settings.
As the world's largest developing country, China cultivates RTCs as important cash crops in its tropical and subtropical regions (Guangxi, Guangdong, Hainan, and Yunnan provinces)[114]. In northern China, potato and sweet potato serve as crucial staple food crops[6]. Incorporating RTCs into low-carbon agricultural management systems through technologies such as residue return, reduced deep tillage, cover cropping, and increased organic matter input offers a practical pathway for agricultural carbon sequestration under China's 'dual carbon goals'[115]. This approach is particularly suitable for sloping lands, areas with limited water and fertilizer resources, and smallholder farming systems. However, consistent with international experiences, China must establish concurrent SOC monitoring frameworks during the industrialization of cassava production to avoid 'carbon debt' resulting from the conversion of forests or grasslands to cassava plantations[116].
For the United Nations 2030 Sustainable Development Goals (SDGs), RTCs simultaneously contribute to SDG 1 (no poverty), SDG 2 (zero hunger), and SDG 13 (climate action)[117]. Their irreplaceable value in ensuring food and income security in vulnerable regions align with SDG 1 and SDG 2, while RTC-based practices such as soil carbon sequestration, restorative agriculture, and low-emission agricultural technologies correspond to SDG 13 and SDG 15 (life on land)[117]. Achieving long-term climate benefits will depend on policy incentives (e.g., ecological compensation and carbon payments), varietal improvement, and optimized farmland management.
Overall, the carbon sequestration value of RTCs does not rely on transient high photosynthetic rates, but on an integrated system encompassing land management, underground biomass accumulation, residue return, and the prevention of land degradation. Thus, their significance lies in constructing a comprehensive pathway integrating 'food security, soil health, and soil carbon sinks', rather than substituting for crops with high light-use efficiency.
-
RTCs possess unique advantages in carbon sequestration, driven by efficient source–sink–flow coordination and specialized underground carbon storage, complementing their role in global food security. Key bottlenecks, including photosynthetic inefficiencies of the C3 pathway, source–sink mismatches, and suboptimal field management, have constrained their full carbon sequestration potential. Integrating genetic engineering (e.g., modifying Rubisco, enhancing CCMs, and strengthening sink capacity) with targeted agronomic practices (e.g., balanced fertilization, crop rotation, and biochar application) is critical to overcoming these limitations. Future efforts should prioritize multiplex genetic modification and site-specific management optimization. By leveraging RTCs' multifunctional value, we can advance climate-smart agriculture, enhance soil carbon sequestration, and contribute significantly to global carbon.
To advance RTCs' carbon sequestration potential, future research should prioritize three directions. First, synthetic biology-enabled pathway engineering: constructing modular carbon fixation circuits (e.g., C4 enzyme cassettes, carboxysome-like nanostructures, photorespiratory bypasses) via CRISPR multiplex editing to optimize CO2 assimilation and starch biosynthesis[87,92,95]. Second, AI-assisted breeding: using machine learning to predict multi-gene modification outcomes and high-throughput phenotyping to accelerate screening of high-carbon-sequestration varieties[110]. Third, rhizosphere microbiome engineering: manipulating root exudates to recruit carbon-fixing microbes and mycorrhizal fungi for enhanced soil carbon stabilization[24,65]. These genetic improvements should be integrated with digital agriculture technologies (IoT-based monitoring, precision fertilization) for site-specific management. By synergizing these biotechnologies, RTCs can serve as a 'food security-carbon sequestration dual-purpose' system, contributing to climate-smart agriculture and global carbon neutrality goals.
-
During the preparation of this review paper, the author used Doubao (Version 2.1.8_mac) and Gemini (Gemini 3.0) for English language refinement and figure elements enhancement. The author(s) reviewed and edited all content produced with the assistance of this tool, verified its accuracy, and take full responsibility for the integrity and originality of the final manuscript. This work represents the author(s)' own intellectual contribution, and no AI tool is credited as an author.
-
The authors confirm contributions to the paper as follows: study conception and design: Mu Z, Yang S; literature retrieval and collation: Yang S, Deng K; integration and refinement of review content: Mu Z, Na T, Guo H, Zhou Y, Zhao L, Wang J; draft manuscript preparation: Yang S, Deng K, Na T, Guo H. All authors reviewed the results and approved the final version of the manuscript.
-
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
-
The authors would like to acknowledge Aborncommander Scientist Project of Qinghai Province "Capacity Improvement of Innovation system of potato breeding and seed industry production in plateau" (2023-NK-146) and Qinghai Provincial Department of Science and Technology International Cooperation Project (2025-HZ-812) for supporting this work.
-
The authors declare that they have no conflict of interest.
-
Received 11 December 2025; Accepted 9 March 2026; Published online 27 March 2026
-
# Authors contributed equally: Zhihua Mu, Shuya Yang, Ke Deng
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press on behalf of Hainan University. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Mu Z, Yang S, Deng K, Na T, Guo H, et al. 2026. Engineering tuber crops for carbon sequestration: genetic improvement strategies and technological advances. Tropical Plants 5: e009 doi: 10.48130/tp-0026-0008
Engineering tuber crops for carbon sequestration: genetic improvement strategies and technological advances
- Received: 11 December 2025
- Revised: 28 February 2026
- Accepted: 09 March 2026
- Published online: 27 March 2026
Abstract: Root and tuber crops (RTCs), such as potato, cassava, and sweet potato, are globally critical staple foods and exhibit substantial potential for carbon sequestration. Their unique source-sink-flow synergy, high photosynthetic efficiency, and underground carbon storage capacity make them pivotal for climate change mitigation. However, RTCs face inherent bottlenecks: inefficient C3 photosynthesis with photorespiratory losses, source-sink imbalance, and inadequate low-carbon management practices. To address these limitations, this review synthesizes genetic engineering strategies (e.g., optimizing Rubisco function, introducing C4/CAM pathway elements, enhancing sink strength via AGPase and sugar transporters), and improved field management (e.g., balanced fertilization, crop rotation, biochar application, and IoT-based precision agriculture). These integrated approaches synergistically boost carbon fixation, optimize carbon allocation, and strengthen soil carbon sinks. RTCs thus represent a promising avenue to reconcile food security with carbon neutrality goals, providing actionable pathways for developing climate-resilient and sustainable agricultural systems globally.












