-
Soybean (Glycine max L.) is a major grain legume cultivated worldwide for its high protein and oil content, and for its capacity to contribute biologically fixed Nitrogen to agricultural production. Through symbiosis with nitrogen-fixing bacteria, soybean reduces reliance on synthetic N inputs and support sustainable cropping systems[1]. However, soybean yield in the Philippines remains consistently low, ranging from 0.9–1.1 t·ha−1, which is substantially below the global average yield (2.8 t·ha−1)[2,3]. This yield gap reflects persistent constraints in tropical soybean production, where suboptimal cultivar adaptation, acidic soils with low organic matter, nutrient limitations, and episodic water stress frequently interact to limit crop performance[4]. Among these constraints, N availability plays a critical role in regulating early vegetative growth, reproductive development, and final yield formation. In tropical soils, efficiency of mineral N fertilization is often restricted by losses through volatilization and leaching, reducing fertilizer use efficiency, and increasing production costs. As a result, biological N fixation (BNF) remains a critical process for meeting crop N demand in soybean-based systems. Consequently, BNF is mediated primarily by Bradyrhizobium species that form nodules on roots and supply N during the later stages of vegetative growth and throughout reproductive development[5,6]. In the Philippines, Bradyrhizobium japonicum, B. elkanii, and B. diazoefficiens have been identified as dominant microsymbionts, with reported N fixation potentials ranging from 200 to 400 kg N ha−1 under favorable conditions[5,7,8]. Despite this substantial potential, effective nodulation and high rates of nitrogen fixation under tropical field conditions usually occur only after early crop establishment[9]. This creates a transient period of N limitation during seedling growth, which can restrict root development, delay canopy expansion, and have lasting effects on reproductive sink formation[3,5]. Early-season N constraints may therefore influence not only vegetative vigor but also subsequent flowering, pod set, and yield component relationships.
In contrast to rhizobia, plant growth–promoting rhizobacteria (PGPR), such as the Azospirillum species, can colonize the rhizosphere and root surfaces and influence early plant growth through modifications of root architecture, nutrient uptake, and plant vigor[10,11]. While the individual contributions of rhizobial symbionts and PGPR to legumes have been widely documented, their combined application has emerged as a promising strategy for improving soybean performance under low-input and stress-prone environments. Several studies have reported positive yield responses to co-inoculation with rhizobia and PGPR across a range of environments; however, the magnitude and consistency of these effects remain highly variable and strongly dependent on environmental conditions[12,13]. Notably, most previous co-inoculation studies have focused primarily on final grain yield, nodulation characteristics, or total N accumulation, with limited consideration of how microbial symbionts influence the internal structure of yield formation[14]. Few studies have examined whether co-inoculation alters phenological development, redistributes yield among its components, or affects trade-offs between seed number and seed size under field conditions[15]. In soybean, grain yield results from complex interactions among flowering time, pod initiation, seed filling, and biomass partitioning during the reproductive phase[16,17]. Well-documented yield compensation mechanisms, such as increased seed size offsetting reductions in seed number, changed yield components, and occurred without proportional changes in total biomass. However, these compensatory responses remain poorly understood in the context of microbial co-inoculation, particularly in tropical production systems.
In the Philippines and other Southeast Asian countries, soybean is commonly grown on marginal soils under rainfed or partially irrigated conditions, where early growth constraints can have persistent effects on subsequent reproductive development. Understanding whether microbial co-inoculation modifies phenological timing, yield, and assimilate allocation between vegetative and reproductive organs is therefore essential for evaluating its agronomic relevance. Such functional responses may provide insight on how microbial inoculants influence yield formation under field conditions, even in the absence of direct measurements of nitrogen fixation or other physiological processes. Therefore, the present study evaluates the effects of single and combined inoculation with rhizobial and associative diazotrophic bacteria on soybean grown under field conditions in the Philippines, with particular emphasis on phenological development, grain yield components, and biomass allocation patterns. We hypothesize that co-inoculation enhances early vegetative development, resulting in increased grain yield and yield components.
-
The field experiment was conducted during the dry season at La Paz, Magalang, Pampanga, Philippines (15°12'54" N, 120°39'43" E; 15 m above sea level). The site is characterized by a Type I tropical climate, with a distinct dry season from November to April. Prior to land preparation, soil sampling was carried out from 0–30 cm depth to check the characteristics of the surface soil (Table 1). Ten soil cores were randomly collected across the experimental area, composited, air-dried, and sieved (2 mm). Soil texture was determined using the hydrometer method, soil pH was measured in a 1:1 soil–water suspension, total N was analyzed using the Kjeldahl method, available P was determined using the Olsen extraction, exchangeable K was extracted with ammonium acetate, and organic matter content was estimated by the Walkley–Black method.
Table 1. Initial soil properties of the experimental field (0–30 cm depth).
Soil parameter Value and interpretation pH (H2O, 1:1) 5.57 (moderately acidic) Total nitrogen (%) 0.12 (low) Available phosphorus (ppm) 20.1 (moderate) Exchangeable potassium (cmol·kg−1) 0.18 (low) Organic matter (%) 1.43 (low) Texture Sandy loam Experimental layout
-
The field experiment was arranged as a randomized complete block design (RCBD) with four replications and five treatments: control (no inoculation), inoculation with Azospirillum spp., inoculation with Bradyrhizobium spp., co-inoculation with Azospirillum spp. and Bradyrhizobium spp., and a mineral N application representing conventional fertilization practice. Block was used to account for gradual variation in soil conditions across the experimental field. Each plot was 3.5 m × 4.0 m (14 m2) in size and was separated from adjacent plots by 0.5 m alleys. Soybean was planted at a spacing of 60 cm between rows and 40 cm between hills. Seedlings were thinned to one plant per hill, to maintain a population density of 41,700 plants·ha−1. To minimize border effects, a central sampling area of 2.8 m × 2.5 m (7 m2) was established within each plot.
Microbial inoculation
-
A commercially formulated carrier-based biofertilizer (SuprIMO®) was used as the rhizobial inoculant. The product contains a multi-strain consortium of diazotrophic rhizobacteria with a minimum population density of 1 × 108 cfu·g−1, comprising taxa assigned to the genera Rhizobium, Bradyrhizobium, and Sinorhizobium. The taxonomic composition and species identity of these rhizobial strains are based on prior isolation and characterization reported by Mason et al.[8], who documented soybean-nodulating diazotrophic bacteria belonging to these genera. Based on manufacturer analysis, the formulation contains 0.93% N, 0.69% P2O5, 0.96% K2O, 25.01% organic matter, and has a pH of 9.03. Prior to application, the product was stored under ambient temperature (25–28 °C) in a shaded area to maintain microbial viability. A total of 250 g of biofertilizer was dissolved in 16 L of unchlorinated water, which corresponds to the recommended rate for 2,000 m2. Based on the experimental plot size of 14 m2, the equivalent application rate was 1.75 g biofertilizer per plot, diluted proportionally in water to ensure uniform soil application. The inoculant suspension was applied evenly to the soil surface 1 d before planting and lightly incorporated into the topsoil.
For associative N fixation, Bio-N® was used as the Azospirillum inoculant, which contains Azospirillum brasilense and Azospirillum lipoferum isolated from Saccharum spontaneum. The formulation and agronomic effectiveness of Bio-N® have been well documented under Philippine conditions, particularly for enhancing root development and early plant vigor[18,19]. Application followed the standard soil inoculation method recommended by BIOTECH-UPLB, using the same dilution and plot-level volume as the rhizobial inoculant. In the co-inoculation treatment, SuprIMO® and Bio-N® were applied sequentially to ensure adequate establishment of both microbial groups.
Nitrogen fertilization
-
Mineral N was applied at a total rate of 87.4 kg N ha−1 in two equal splits. Based on the plot area (14 m2), this rate is equivalent to 122.4 g N plot−1. Nitrogen in the form of urea (46-0-0) was applied in two equal splits to improve uptake efficiency and reduce losses: 50% (61.2 g N plot−1) was applied at planting, and the remaining 50% was applied at early vegetative stage (approximately 25–30 d after planting). Although mineral N availability is known to influence nodulation in soybean, nodulation traits were not measured in this study, and therefore no inferences regarding nodulation suppression or symbiotic activity under mineral N fertilization are made. Comparisons between mineral N and microbial treatments are thus interpreted strictly in terms of observed phenological responses, yield components, and final yield outcomes.
Agronomic management
-
Crop management practices were applied uniformly across all treatments throughout the study. Weeding was carried out manually at regular intervals to maintain weed-free conditions. Irrigation was applied as needed, based on crop demand and prevailing weather conditions, to avoid moisture stress. Replanting was conducted within the first week after emergence to maintain uniform plant populations across plots. An integrated pest management approach was implemented, with regular monitoring of insect pests and diseases. Control measures were applied only when necessary and uniformly across treatments following recommended practices for soybean. Harvesting was performed at physiological maturity, and plants were harvested manually to minimize yield loss prior to sample processing.
Growth and phenological development
-
Crop development was monitored throughout the growing season. Flowering time was recorded as days to 50% flowering, defined as the stage at which approximately half of the plants within the sampling area had reached the R1 developmental stage, following the soybean growth scale[20]. Plant height was measured at 90 d after sowing, from the soil surface to the uppermost growing point of the main stem. Root length was assessed at the same stage by carefully excavating selected plants and gently washing the root systems to remove adhering soil. The length of the longest root was then measured. These measurements were taken from the same 10 plants per plot to maintain consistency across growth assessments, using procedures commonly applied in root–shoot studies.
Crop yield assessment
-
For yield component analysis, pods from the sampled plants were harvested at maturity. Pod and seed number per plant, seeds per pods, seed weight per plant, and 100-seed weight were determined through direct counts and dry-weight measurements. Final grain yield was obtained from the total seed weight harvested from the sampling area of each plot. Yield values were adjusted to a standard moisture content of 13% and converted to a per-ha basis using conventional agronomic conversion procedures. Dry matter efficiency and harvest index were calculated to assess assimilate partitioning and yield efficiency. Harvest index (HI) was expressed as the ratio of seed yield to total above-ground biomass to evaluate crop yield efficiency and partitioning[1].
$ \rm{D}\mathrm{ME}\; (\mathrm{g\cdot m}^{-2}\cdot\mathrm{day})=\dfrac{\text{Seed yield}}{\text{Total dry matter produced}}\times\dfrac{\text{100}}{\text{Crop duration}} $ $ \rm{HI\; (\text{%})}=\dfrac{\text{Economic yield (kg)}}{\text{Biological yield (kg)}} $ Statistical analysis
-
Data analyses were conducted using STAR (Statistical Tool for Agricultural Research), version 2.0.1. Prior to analysis of variance (ANOVA), assumptions of normality and homogeneity of variances were evaluated for each parameter using residual diagnostics. Normal probability plots and residual vs fitted value plots were examined to verify compliance with ANOVA assumptions. Analysis of variance was then performed, and treatment means were compared using the Least Significant Difference (LSD) test at p ≤ 0.05 when significant effects were detected. Relationships among phenological traits, growth parameters, and yield components were assessed using Pearson correlation analysis based on plot-level data, and correlation coefficients were visualized using a correlation matrix. All statistical tests were conducted at a 5% level of significance.
-
Days to flowering varied markedly among treatments (Fig. 1a). The uninoculated control reached flowering at 41 d after planting, whereas all treated plants flowered earlier. Among single microbial inoculations, the shortest time to flowering was recorded under Azospirillum spp. (32 d), followed by Bradyrhizobium spp. (37 d). Co-inoculation with Azospirillum spp. and Bradyrhizobium spp. resulted in flowering at 36 d, which was comparable to the mineral N treatment (36 d). Plant height was not significantly affected by treatment (Fig. 1b). Uninoculated plants attained a mean height of 55.38 cm, followed by Azospirillum spp. (53.33 cm), rhizobial inoculation (55.45 cm), and co-inoculation (56.00 cm), with the tallest plants recorded under mineral N fertilization (58.23 cm). Root length showed a strong response to microbial inoculation (Fig. 1c). The shortest roots were observed in uninoculated plants (4.76 cm). Single inoculation with Azospirillum spp. (8.24 cm), and Bradyrhizobium spp. (8.53 cm) significantly increased root length, while the greatest increase was recorded under co-inoculation (13.01 cm). N fertilization also enhanced root length relative to the control (7.94 cm).
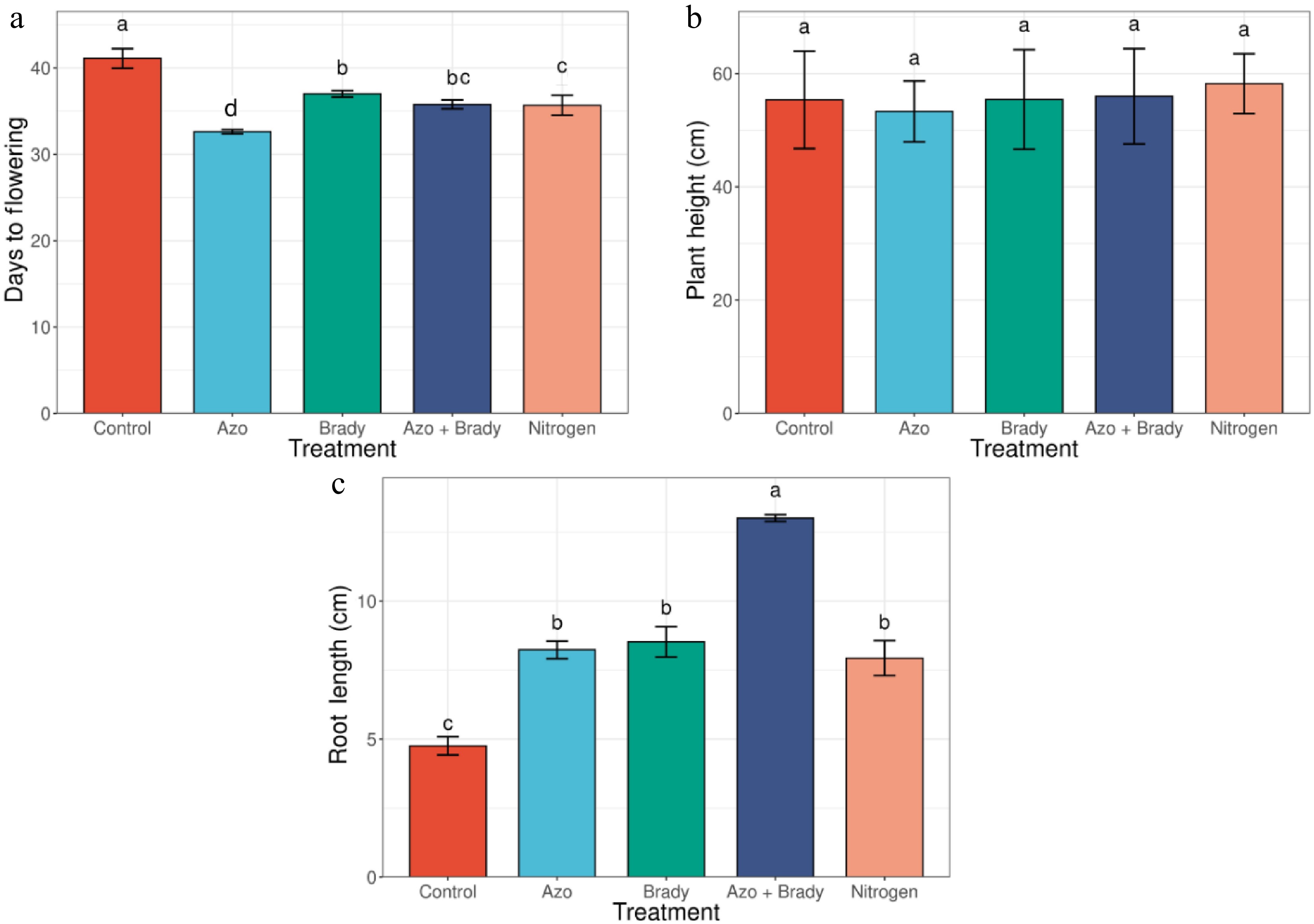
Figure 1.
Phenology and vegetative growth traits of soybean control (no inoculation). Azo, Azospirillium spp.; Brady, Bradyrhizobium spp.; Azo + Brady, Azospirillium spp. + Bradyrhizobium spp.
Reproductive sink formation of soybean
-
Pods per plant was strongly influenced by treatment. Plants without inoculation produced 144.38 pods·plant−1, whereas plants receiving co-inoculation with Azospirillum spp. and Bradyrhizobium spp. produced 315.62 pods·plant−1. Application of mineral N also increased pods·plant−1, while single inoculation with Bradyrhizobium spp. and Azospirillum spp. increased pod number by 56.81% and 30.69%, respectively, relative to the control (Table 2). In contrast, seed number per plant decreased compared with the uninoculated control. Reductions of 12.77% and 14.13% were observed under individual inoculation with Azospirillum spp. and Bradyrhizobium spp., respectively, whereas co-inoculated plants exhibited a more pronounced reduction of 16.32%. A similar pattern was observed for seeds per pod, which declined from 1.64 in uninoculated plants to 1.09 under Azospirillum spp., 0.90 under Bradyrhizobium spp., 0.86 under N fertilization, and 0.62 under co-inoculation (Table 2). Dry matter efficiency did not differ significantly among treatments, with values ranging from 0.80 to 0.88, indicating no consistent increase or decrease relative to the control (0.88). In contrast, harvest index was significantly affected by treatment. Relative to the uninoculated control (26.73%), harvest index increased by 17.08% under single inoculation with Azospirillum spp., and by 11.20% under N fertilization, but declined slightly under Bradyrhizobium spp. inoculation (−0.75%) and co-inoculation (−1.23%).
Table 2. Effects of microbial inoculation and N application on soybean seed set parameters and biomass allocation.
Treatment Pod number·plant−1 Seeds·plant−1 Seeds·pod−1 Harvest index (%) Dry matter efficiency No inoculation 144.38 e 236.20 a 1.64 a 26.73 c 0.86 Azospirillum spp. 188.72 d 206.06 b 1.09 b 31.30 a 0.88 Bradyrhizobium spp. 226.38 c 202.85 bc 0.90 c 26.93 c 0.81 Azospirillum spp. + Bradyrhizobium spp. 315.62 a 197.59 c 0.62 d 26.40 c 0.80 Mineral nitrogen 269.12 b 230.36 a 0.86 c 29.73 b 0.83 Means within a column followed by the same letter are not significantly different at p ≤ 0.05 (LSD). Agronomic yield formation of soybean
-
Hundred-seed weight increased from 8.03 g in uninoculated plants to 12.05 g under Azospirillum spp. inoculation and to 12.21 g under Bradyrhizobium spp. inoculation (Table 3). The highest seed weight was produced under co-inoculation (16.04 g). Mineral N fertilization resulted in a moderate increase in 100-seed weight (11.85 g). Seed weight per plant differed significantly among treatments. Uninoculated plants recorded a seed weight of 18.96 g·plant−1. Relative to the control, seed weight per plant increased to 24.82 g in plants inoculated with Azospirillum spp. and to 24.75 g under rhizobial inoculation. The highest seed weight·plant−1 was observed under co-inoculation (31.70 g), followed by the mineral N (27.29 g), and lowest seed weight was recorded in plants with no inoculation (18.96 g).
Seed mass per pod declined significantly with increasing pod number. Seed mass per pod decreased by 16.85% under rhizobial inoculation, by 21.92% under mineral N fertilization, and by 23.46% under co-inoculation of Azospirillum spp. + Bradyrhizobium spp., with the lowest value observed in the co-inoculated treatment (0.101 g·pod−1). Uninoculated plants produced a yield of 1.57 t·ha−1, whereas yield increased by 31.21% under Azospirillum spp. inoculation and by 31.21% under Bradyrhizobium spp. inoculation. N fertilization increased yield by 42.04%, while the highest yield (2.52 t·ha−1) was obtained under co-inoculation (Table 3).
Table 3. Effects of microbial inoculation and N application on soybean yield components and seed yield.
Treatment 100-seed weight (g) Seed weight·plant−1 (g) Seed mass·pod−1 (g) Seed yield (t·ha−1) No inoculation 8.03 d 18.96 d 0.131 a 1.57 c Azospirillum spp. 12.05 bc 24.82 c 0.132 a 2.06 b Bradyrhizobium spp. 12.21 b 24.75 c 0.109 b 2.06 b Azospirillum spp. + Bradyrhizobium spp. 16.04 a 31.70 a 0.101 c 2.52 a Mineral nitrogen 11.85 c 27.29 b 0.103 bc 2.23 b Means within a column followed by the same letter are not significantly different at p ≤ 0.05 (LSD). Correlation analysis of phenology, growth, and yield components
-
Seed yield (SY) was strongly associated with seed weight per plant (SWP; r = 0.94), 100-seed weight (HSW; r = 0.91), and pods per plant (PP; r = 0.88). Days to flowering (DTF) was negatively associated with seed yield (r = −0.59), and with key yield components (SWP and HSW; r = −0.58 to −0.59). In contrast, seeds per plant (SP) and seeds per pod (SPP) showed strong negative associations with HSW (−0.80 and −0.93, respectively). Pods per plant (PP) was positively associated with SY (r = 0.88), and seed mass–related traits, but negatively associated with SPP (r = −0.93). Dry matter efficiency (DME) showed weak to moderate negative associations with yield and seed mass traits, and a moderate positive association with harvest index (HI; r = 0.35). Vegetative traits showed weaker associations overall; plant height (PH) was weakly correlated with yield, whereas root length (RL) was positively associated with PP, seed mass traits, and SY (r = 0.87–0.98). In general, seed yield was most closely associated with seed mass-related traits and reproductive sink size, with earlier flowering and greater root development consistently accompanying higher yield, while seed number traits exhibited compensatory relationships with seed size (Fig. 2).
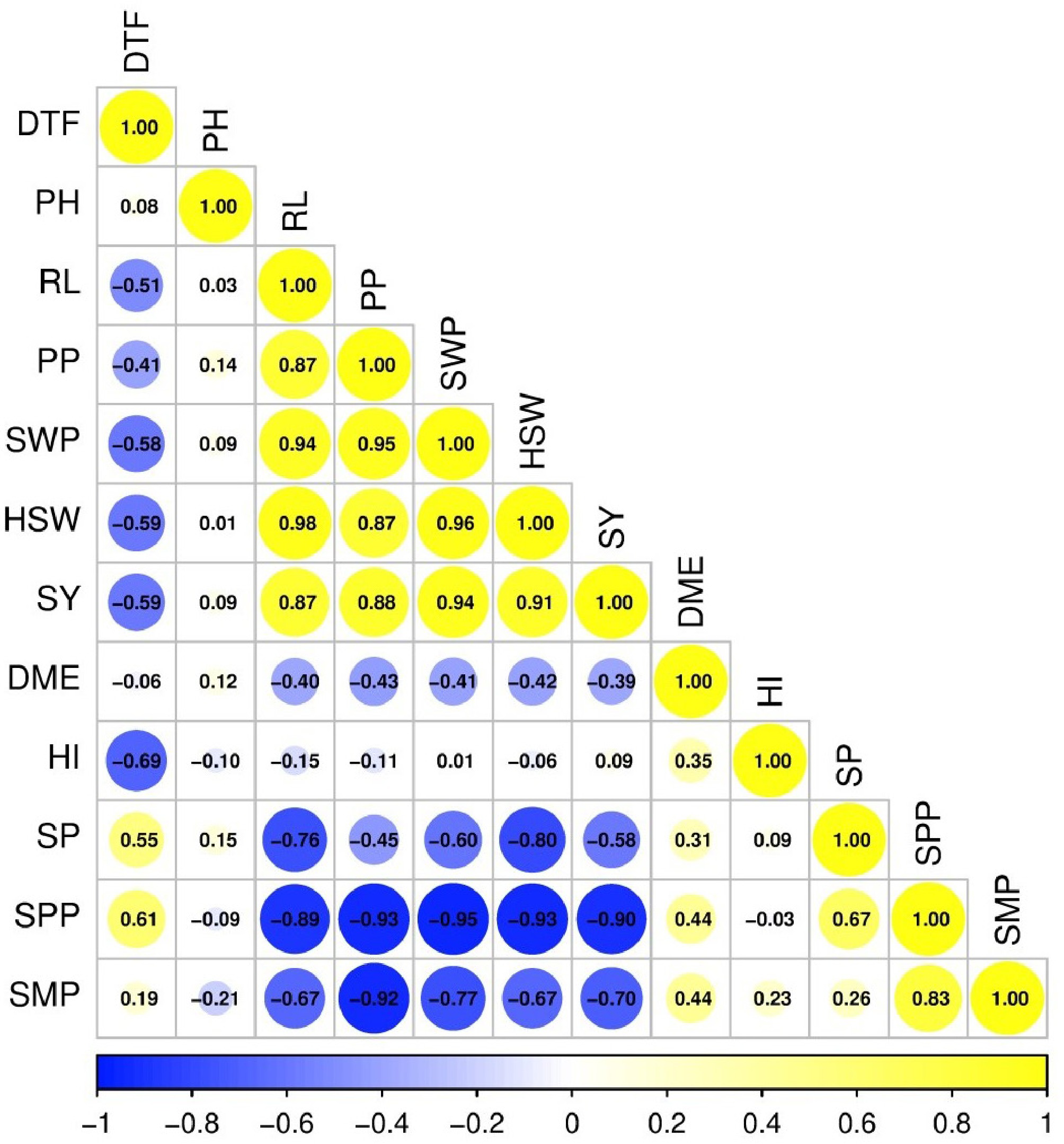
Figure 2.
Correlation matrix of phenological, growth, and yield-related traits in soybean. DTF, days to flowering; PH, plant height; RL, root length; PP, pods per plant; SWP, seed weight per plant; HSW, 100-seed weight; SY, seed yield; DME, dry matter efficiency; HI, harvest index; SP, seeds per plant; SPD, seeds per pod; SMP, seed mass per pod.
-
The present study demonstrates that microbial inoculation influences soybean growth and yield formation under field conditions, with the strongest response observed under co-inoculation of Bradyrhizobium spp. (including Bradyrhizobium japonicum and Bradyrhizobium diazoefficiens) with Azospirillum spp., notably Azospirillum brasilense and Azospirillum lipoferum. Yield improvement was not associated with increased vegetative biomass. Instead, yield gains coincided with shifts in phonological timing and yield component distribution, signifying that microbial inputs altered yield construction rather than enhancing overall plant size or biomass accumulation[21−23]. Consequently, these findings extend earlier work on rhizobial-PGPR co-inoculation by showing that, under field conditions, microbial effects are expressed through temporal organization of crop development, in addition to their role in N acquisition[24]. Earlier flowering was consistently observed following co-inoculation, particularly under Azospirillum spp., combined with Bradyrhizobium spp.[22,25]. In tropical and subtropical soybean systems, where crop duration is frequently constrained by climatic variability and late-season stress, advancement of reproductive onset can be agronomically advantageous[26,27]. The study shows that earlier flowering concurred with increased seed yield and higher individual seed mass, indicating that microbial inoculation shifted reproductive development toward a period more conducive to effective seed filling[28]. As physiological traits and stress indicators were not quantified, this response reflects phenological adjustment rather than evidence of enhanced stress tolerance or intrinsic physiological efficiency. Root length responded strongly to microbial symbionts, particularly under co-inoculation, and showed positive associations with pod number, seed mass traits, and seed yield. These root responses likely reflect early developmental changes accompanying altered phenological progression and reproductive sink establishment. However, in the absence of direct measurements of nutrient uptake or symbiotic N fixation, root traits are part of coordinated architectural changes, rather than as direct mechanistic drivers of yield enhancement[22,24].
Yield improvement under microbial inoculation was primarily governed by changes in the reproductive sink structure and compensatory adjustment among yield components. Co-inoculated plants produced a greater number of pods per plant, while reductions in seeds per pod, and seeds per plant were evident. Consistent with several reports showing that inoculation with Bradyrhizobium spp. and PGPRs such as A. brasilense preferentially promote reproductive initiation and sink establishment rather than sustained increases in assimilate supply[29,30]. Despite reductions in seed number, final grain yield increased due to a consistent growth in individual seed mass, reflected by higher 100-seed weight. This compensation pattern is well documented in soybean, whereby expansion of sink number is offset by adjustments during seed filling, resulting in fewer but heavier seeds. An increased in seed mass following inoculation with Azospirillum spp. and rhizobia supports the seed filling, which is highly sensitive to altered source–sink relations rather than a simple enhancement of carbon acquisition[31,32]. In the present study, the decline in seed mass per pod with increasing pod number, particularly under co-inoculation, further indicates intensified competition for assimilates among reproductive units. Nevertheless, compensation at the seed level was sufficient to increase total yield, demonstrating that microbial inoculation modified assimilate allocation within reproductive sinks rather than improving pod-level efficiency[33,34]. Meanwhile, harvest index did not increase under co-inoculation despite substantial yield gains. It illustrated that yield enhancement did not arise from improved whole-plant partitioning efficiency, but rather from redistribution among yield components, highlighting the plasticity of soybean yield architecture under microbial influence[35−37].
Microbial inoculation vs mineral N
-
A principal contribution of this study lies in the comparison between microbial inoculation and mineral N fertilization with respect to yield. Rhizobial symbionts such as B. japonicum and B. diazoefficiens remain fundamental to soybean N nutrition through biological N fixation, which can supply a substantial proportion of crop N demand under favorable conditions[25,38,39]. However, the role of supplementary mineral N in high-yield soybean systems remains contentious, as excessive N inputs can suppress nodulation and diminish the functional contribution of symbiotic rhizobia[40]. Although similar yield levels were achieved under microbial inoculation and mineral N fertilization, the associated yield formation pathways differed. Mineral N primarily supported yield through maintenance of seed number, whereas microbial inoculation—particularly co-inoculation with Azospirillum spp. and Bradyrhizobium spp.—favored earlier reproductive transition and enhanced seed filling. This divergence simply shows that biological and mineral N inputs are not functionally equal, even when final yield outcomes converge[37]. Evidence from other legumes, including common bean (Phaseolus vulgaris L.), further suggest that mineral N can complement microbial inputs only when applied at rates that do not inhibit symbiotic activity[41,42]. The different responses observed under co-inoculation likely reflects interactions among microbial consortia, soil fertility, and crop phenology rather than an inherently superior inoculation strategy[37,43,44]. Hence, the co-inoculation with Azospirillum spp. and Bradyrhizobium spp. does not simply replace mineral N, but alters the internal balance of yield components and the timing of reproductive development[45,46]. This difference represents a substantive insight into microbial symbionts capable of shaping crop partitioning in soybean cultivation, rather than solely augmenting N supply.
-
Under tropical field conditions, microbial inoculation significantly modified soybean yield, with the strongest response observed under co-inoculation with Azospirillum spp. and Bradyrhizobium spp. Higher yield without marked changes in plant height or biomass efficiency indicates that productivity was not driven by enhanced vegetative growth. Instead, yield improvement was associated with changes in phenology, root development, and yield component balance. Co-inoculated plants flowered earlier and developed longer roots, accompanied by a substantial increase in pod number (316 pods), and a compensatory reduction in seeds per pod. Final yield increases were primarily driven by improved seed filling, as reflected by higher 100-seed weight under inoculation. Generally, these results demonstrated that microbial co-inoculation advances soybean productivity by reshaping yield architecture toward greater sink establishment and seed mass accumulation, rather than through proportional increases in biomass or harvest index. The findings underscore the agronomic relevance of microbial inputs for low-input tropical soybean systems and suggest their potential to complement or reduce reliance on mineral N fertilization. However, the absence of direct measurements of nodulation and plant N dynamics highlight the need for further studies to clarify the physiological mechanisms underlying these functional yield responses.
-
The authors confirm contributions to the paper as follows: study conception and design: Guanzon IM; data collection: Gamboa J; analysis and interpretation of results: Guanzon IM, Guzman WM, Rafael RR; draft manuscript preparation: Guanzon IM, Gamboa J. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
-
The authors received no financial support for the research, authorship, and/or publication of this article.
-
The authors declare that they have no conflict of interest.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Guanzon IM, Gamboa J, Guzman WM, Rafael RR. 2026. Co-inoculation with Azospirillum and Bradyrhizobium modifies yield components and phenological timing in soybean (Glycine max L.). Circular Agricultural Systems 6: e008 doi: 10.48130/cas-0026-0008
Co-inoculation with Azospirillum and Bradyrhizobium modifies yield components and phenological timing in soybean (Glycine max L.)
- Received: 05 January 2026
- Revised: 12 February 2026
- Accepted: 27 February 2026
- Published online: 23 April 2026
Abstract: Soybean productivity in tropical environments is often constrained by nutrient limitation. This study evaluates how microbial inoculation modifies soybean yield formation under field conditions in the Philippines, with emphasis on phenology, root development, and yield components. Treatments included no inoculation (control), inoculation with Azospirillum spp., Bradyrhizobium spp., their combined application, and mineral nitrogen (N) treatment. Grain yield differed significantly among treatments, with co-inoculation producing the highest yield (2.52 t·ha−1) compared with the control (1.57 t·ha−1) and mineral N fertilization (2.23 t·ha−1). Yield increases under co-inoculation were associated with earlier flowering (36 d) and greater root length (13.01 cm), rather than increased vegetative biomass. Reproductive development was strongly altered, with pod number increased to 316 pods·plant−1, while seeds per pod declined from 1.64 to 0.62. Despite reduced seed number, individual seed size increased markedly, with hundred-seed weight rising from 8.03 to 16.04 g. Correlation analysis showed that grain yield was positively associated with seed weight per plant (r = 0.94), hundred-seed weight (r = 0.91), pod number (r = 0.88), and root length (r = 0.87–0.98), while seed number traits were negatively related to seed size. Overall, microbial co-inoculation enhanced soybean productivity in tropical low-input systems by shaping yield components and phenological development, offering a viable alternative to mineral N fertilization.
-
Key words:
- Biological inputs /
- Nitrogen fixation /
- Phenological shift /
- Source-sink












