-
Plants optimize fitness by dynamically integrating developmental programs with environmental cues. This balance is increasingly challenged by climate change. Rising global temperatures and altered precipitation patterns have amplified the frequency, duration, and severity of drought events, threatening agricultural productivity and ecosystem stability[1]. Drought limits water availability, reduces turgor pressure, inhibits photosynthesis, and ultimately compromises cellular integrity and fertility.
To maintain viability, plants deploy multilayered responses involving stomatal regulation, osmotic adjustment, and hormonal crosstalk between abscisic acid (ABA) and gibberellin (GA)[2]. These mechanisms reallocate resources from growth to stress protection. While transcriptional reprogramming contributes to long-term acclimation, it is often too slow to buffer rapid environmental fluctuations. Therefore, plants also rely heavily on post-translational regulatory mechanisms, particularly the ubiquitin–proteasome system and selective autophagy, to achieve fast adjustments in the growth–stress trade-off[3].
Selective autophagy is mediated by cargo receptors, such as NBR1 and other ATG8-interacting proteins, which recognize specific substrates and deliver them to autophagosomes for degradation[4]. Once viewed primarily as a stress-response pathway, autophagy is now recognized to regulate key developmental processes, including seed development, germination, and flowering. Recent studies reveal essential roles of ATG5 and ATG7 in male gametophyte development, pollen tube growth, and fertility[5]. Additionally, autophagy influences integumentary tapetum-associated programmed cell death and lipid metabolism during embryo development, underscoring its importance beyond vegetative stress responses[6].
The ubiquitin receptor DOMINANT SUPPRESSOR OF KAR 2 (DSK2) functions as a selective autophagy receptor through its interaction with ATG8 via a conserved AIM motif, thereby linking specific targets to autophagic degradation. Initially identified in brassinosteroid (BR) signaling through its role in BES1 turnover[7], DSK2 may regulate a broader network of growth-promoting regulators. Here, we argue that DSK2 functions not as a pathway-specific component, but as a multi-client regulatory hub that coordinates growth suppression during stress, ensuring developmental stability and reproductive success. This perspective explores the expanding role of DSK2 in stress adaptation and proposes a unifying model with implications for both fundamental biology and crop improvement.
-
DSK2 was initially implicated in growth regulation through its role in mediating the degradation of BES1, a central transcription factor in the BR signaling pathway, under abiotic stress[7]. Under favorable conditions, BES1 accumulates in the nucleus, activating genes involved in cell elongation and organ growth[8]. However, stresses such as drought and fixed-carbon starvation trigger BES1 ubiquitination, enabling its recognition by DSK2, which delivers it to both the 26S proteasome and autophagic machinery for degradation[7]. This dual degradation route allows plants to transiently suppress BR-dependent growth and redirect energy toward stress defense mechanisms. The discovery of BES1 as a DSK2 client established a paradigm in which the selective degradation of growth-promoting transcription factors represents a fast-acting strategy for stress adaptation. Nonetheless, whether BES1 is the primary, or even sole, target of DSK2 during drought remains unclear.
-
Our recent findings suggest that DSK2 mediates the turnover of additional regulators, including the F-box protein LAO1 and several class I TEOSINTE BRANCHED1, CYCLOIDEA, PROLIFERATING CELL FACTOR 1 and 2 (TCP) transcription factors (TCP7, TCP14, TCP15, and TCP21) during nitrogen starvation[9]. Although these TCPs function genetically downstream of LAO1, they are not direct degradation targets of LAO1, suggesting that DSK2 operates independently in their regulation. This layered control implies a conserved logic across stress conditions.
Class I TCPs regulate diverse developmental processes, including germination, hypocotyl elongation, flowering, circadian rhythms, and nitrate signaling[9,10]. Emerging evidence links TCP transcription factors to abiotic stress responses. Their expression is modulated by drought, salt, heat, and submergence[11−13]. Notably, TCP9 has been linked to plant drought tolerance[14], raising the possibility that DSK2-mediated regulation of TCP7, TCP14, TCP15, and TCP21 may contribute to drought adaptation.
Consistent with this, the dsk2 mutants exhibit severe drought hypersensitivity, wilting rapidly and failing to recover upon rehydration[7] (Fig. 1a−c). Conversely, multiple tcp mutants, including tcp7/14/21, tcp14/15/21, tcp7/14/15/21 (tcpQ), and tcp septuple (tcpS), show enhanced drought tolerance (Fig. 1a−c). Genetic analysis reveals that dsk2 tcpQ higher-order mutants phenocopy tcpQ, not dsk2 (Fig. 1d, e), indicating that these TCPs act downstream of DSK2 in the drought response pathway. This epistatic relationship suggests that DSK2-mediated TCP degradation accounts for a substantial component of the drought-related phenotype observed in the dsk2 mutants. Given that BES1 is also a downstream target of DSK2 during drought[7], and that TCPs regulate many BR-responsive genes[15], it is likely that BES1 and class I TCPs converge on shared transcriptional programs. They may represent functionally interdependent, though not necessarily redundant, targets of DSK2. Whether they physically interact or co-occupy promoters of growth-related genes remains an open question. Together, these findings support a model in which DSK2 integrates stress signals by simultaneously modulating the stability of multiple transcriptional regulators to fine-tune drought adaptation.
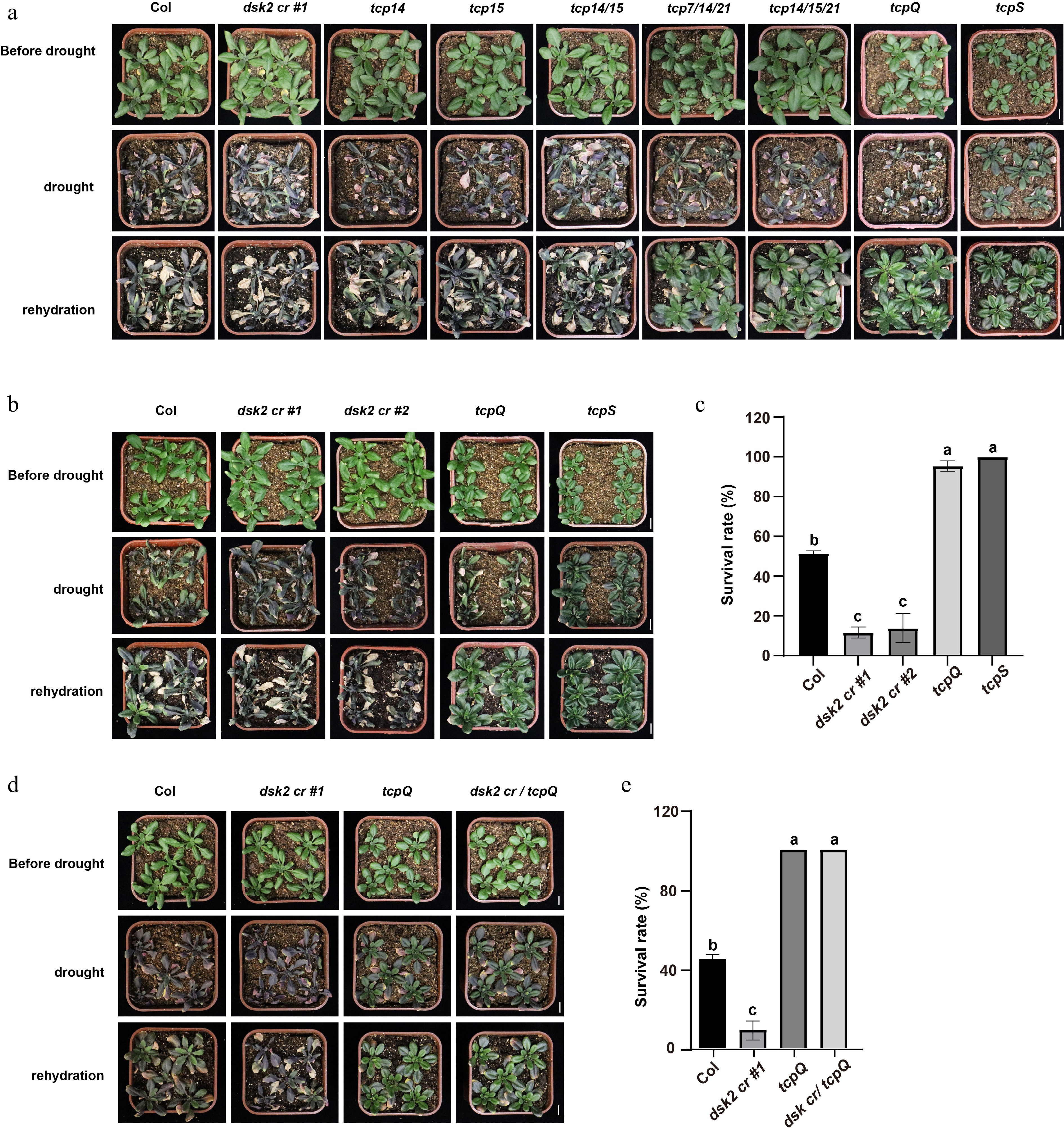
Figure 1.
Role of DSK2-TCP module in plant response to drought stress. (a) Images showing the phenotypes of each genotype before drought stress, after stress treatment, and following rehydration. Scale bars: 1 cm. (b) and (c) Phenotypes of Col, dsk2 cr #1, dsk2 cr #2, tcpQ, and tcpS after drought stress treatment. Plants producing normal leaves after the 2 d recovery period were considered survivors. (b) Representative images. Scale bars: 1 cm. (c) Quantification of survival rates. Data represent mean ± SEM (n = 3 biological replicates). Each replicate contained 10–12 independent plants. (d) and (e) Genetic analysis of DSK2 and TCPs. The dsk2 cr /tcpQ mutants were generated via CRISPR-Cas9 mutagenesis technology in the tcpQ background. (d) Representative images of phenotypes of Col, dsk2 cr #1, tcpQ, and dsk2 cr /tcpQ. Scale bars: 1 cm. (e) Survival rate of each genotype after drought stress. Data was shown as mean ± SEM of three biological replicates. Each replicate contained 6–7 independent plants of Col, dsk2 cr #1, and tcpQ, and 10 independent plants of dsk2 cr /tcpQ. In (c) and (e), the statistical significance was determined using one-way ANOVA (p < 0.05, ANOVA followed by Tukey's post hoc comparison test) analysis. Different letters denoted significant differences. Detailed information on the drought treatment protocol is provided in the Supplementary Text 1.
-
We propose that DSK2 functions as a central regulatory hub that coordinates the degradation of BES1, class I TCPs, and likely additional growth-promoting factors, enabling plants to switch between growth and survival (Fig. 2). Under favorable conditions, BES1, class I TCPs and other regulators accumulate in the nucleus to drive cell expansion, division, and organ development. Under stress conditions, these proteins are ubiquitinated and recognized by DSK2, which channels them into either the ubiquitin–proteasome system or selective autophagy, with autophagy becoming dominant under prolonged stress. This dual-pathway targeting ensures robustness and flexibility in degradation capacity. The coordinated clearance of multiple transcription factors suppresses energy-intensive growth processes, stabilizes cellular homeostasis, and optimizes resource allocation. By targeting regulators across hormonal (e.g., BR and GA) and developmental networks, DSK2 acts as an integrative node that rapidly reconfigures plant physiology in response to environmental perturbation. This model implies that stress resilience can be achieved by hijacking conserved proteolysis hubs (e.g., DSK2) to broadly modulate growth-regulatory networks. Such post-translational control enables rapid phenotypic plasticity, adjusting transcriptional outputs through post-translational control of key transcription factors.
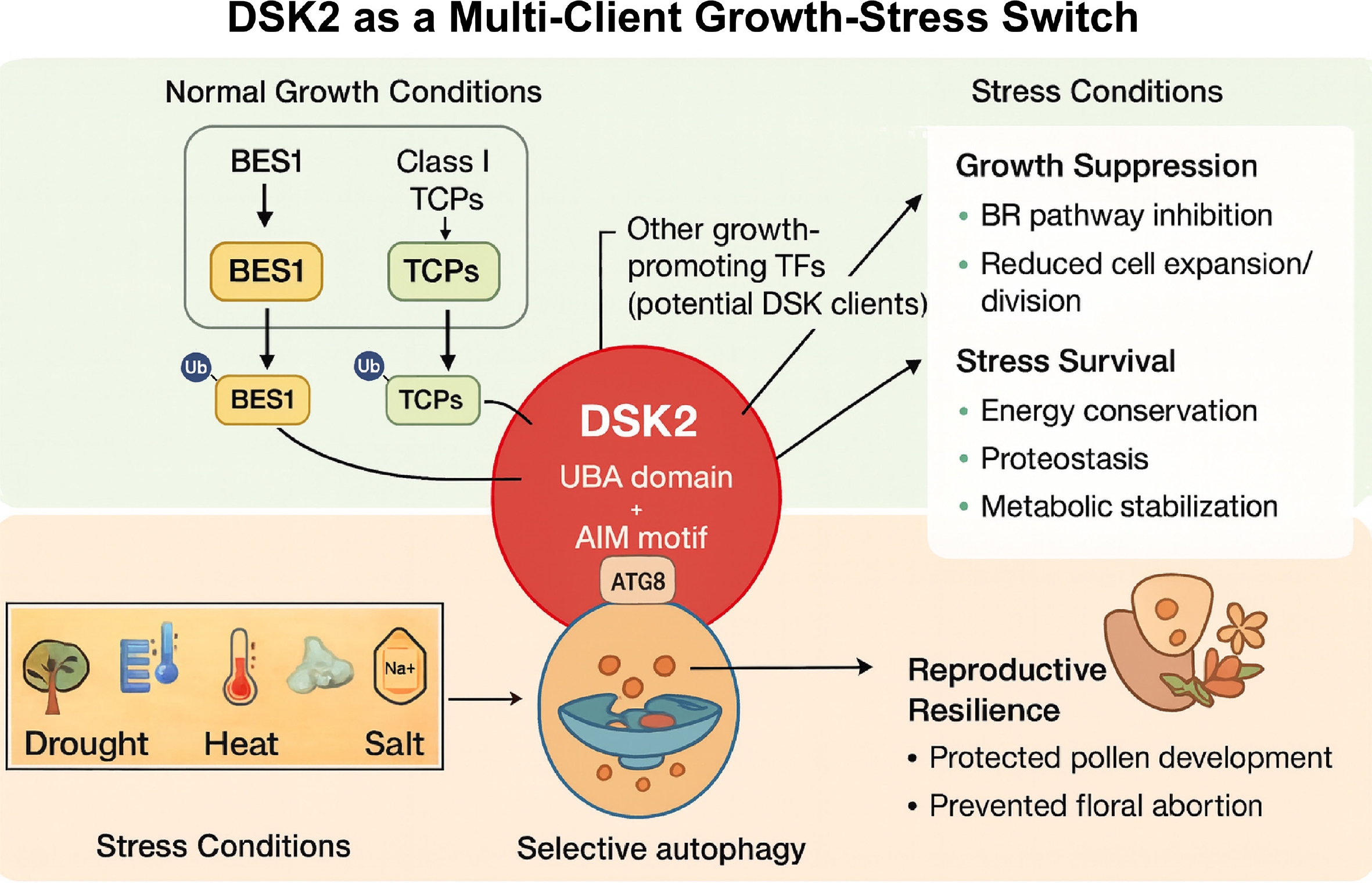
Figure 2.
A unified model of the DSK2 hub coordinating growth-to-stress transitions through multi-client degradation.
Under normal growth conditions, BES1 and class I TCPs accumulate in the nucleus to promote growth. Upon perception of environmental stress, they are ubiquitinated and recognized by the selective autophagy receptor DSK2. Similar to other selective autophagy receptors such as NBR1 and ATG8-interacting adaptor proteins, DSK2 then facilitates the turnover of these proteins primarily via selective autophagy. This coordinated degradation suppresses energy-intensive growth processes, maintains cellular homeostasis, and reallocates metabolic resources to other critical processes such as reproductive development, thereby enhancing stress resilience and yield.
-
The effectiveness of DSK2 as a hub relies not only on its ability to degrade multiple transcription factors but also on the pleiotropic roles of its targets. BES1, for instance, integrates drought and carbon starvation responses[7]. Similarly, Class I TCPs regulate both nitrogen starvation and drought. Notably, our recent genome-wide association study in rapeseed identified the BAK7/BKK1-TCP21 module as essential for submergence tolerance[11]. Paradoxically, the tcpQ mutant is sensitive to submergence treatment, the opposite of its drought-resistant phenotype[11]. This functional reversal highlights the context-dependent roles of TCPs in opposing water-stress conditions. Furthermore, such dualism implies that TCP protein abundance must be dynamically and precisely controlled. In this context, DSK2-mediated selective degradation provides a plausible mechanism for context-specific modulation of TCP stability. Depending on the stress type, DSK2 activity could fine-tune TCP levels to either suppress growth (drought) or maintain metabolic flexibility (flooding). This reinforces the concept of DSK2 as a hub, enabling adaptive growth tuning under contrasting precipitation extremes.
-
Reproductive development is one of the most stress-sensitive phases, with drought commonly causing floral abortion, male sterility, or reduced seed set[16]. Suppressing excessive vegetative growth during these stages is crucial to preserve resources for reproduction[17,18]. While autophagy supports reproductive processes[6,19], and DSK2 may prevent vegetative growth from competing with reproductive demands, the direct role of DSK2-mediated protein degradation in reproductive processes (e.g., tapetal function, pollen viability, or ovule development) remains largely unexplored. Determining whether DSK2 activity in reproductive organs influences yield under stress is a critical next step.
It is very likely that DSK2 targets additional substrates for degradation under different abiotic stresses. Unbiased proteomic and interactome approaches could be utilized to identify new DSK2 targets. Expanding its client spectrum will clarify its position within the cellular signaling network and reveal potential crosstalk with other degradation pathways. Finally, translating these insights into crop improvement is essential. Modulating DSK2 expression, engineering substrate specificity via gene editing, or designing synthetic DSK2 variants with tunable activity could enable the development of 'climate-smart' crops. Such crops might maintain high yield under favorable conditions while rapidly activating protective responses during climatic extremes, enhancing resilience without sacrificing performance.
-
DSK2 emerges as a versatile regulatory node that couples environmental cues with developmental programs, enabling rapid shifts between growth and survival. By directing the turnover of key transcription factors such as BES1 and class I TCPs, DSK2 may function as a critical switch that maintains cellular stability and safeguards reproductive success under abiotic stress. Through selective, context-dependent degradation, DSK2 allows plants to achieve precise growth adjustments across fluctuating environments, optimizing survival in complex stress landscapes. Understanding and harnessing such post-translational regulatory hubs becomes not only a frontier in plant biology but a necessity for sustainable agriculture. DSK2 represents a promising target for engineering crops that are both high-yielding and resilient, a step toward securing global food systems in a changing world.
We apologize to researchers whose work has not been cited in the review due to space limits. This project was funded by National Natural Science Foundation of China (32470286) and Zhejiang A&F University Research Development Fund (2025LFR070).
-
During the preparation of this work, the authors used ChatGPT-4 for language refinement and enhancement of Fig. 2. The authors have reviewed and edited all content produced with the assistance of this tool, verified its accuracy, and take full responsibility for the integrity and originality of the final manuscript. This work represents the authors' own intellectual contribution, and no AI tool is credited as an author.
-
The authors confirm their contributions to the paper as follows: manuscript organization and writing: Li Y, Dong J; manuscript editing: Cui F, Zheng B, Pan R. All authors reviewed and approved the final version.
-
The datasets generated during the current study are available from the corresponding author on reasonable request.
-
The authors declare that they have no conflict of interest.
- Supplementary Text 1 Drought tolerance assay.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press on behalf of Hainan Yazhou Bay Seed Laboratory. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Li Y, Cui F, Zheng B, Pan R, Dong J. 2026. DSK2 as a multi-client regulatory hub in plant stress adaptation. Seed Biology 5: e015 doi: 10.48130/seedbio-0026-0009
DSK2 as a multi-client regulatory hub in plant stress adaptation
- Received: 18 December 2025
- Revised: 31 January 2026
- Accepted: 09 February 2026
- Published online: 06 May 2026
Abstract: Plants must continuously balance growth and stress resilience to survive in fluctuating environments. Selective autophagy and the ubiquitin–proteasome system serve as key post-translational regulatory mechanisms that enable this dynamic equilibrium. The ubiquitin receptor DOMINANT SUPPRESSOR OF KAR 2 (DSK2) is known to mediate stress-induced degradation of the brassinosteroid-activated transcription factor BES1, thereby suppressing growth under adverse conditions. In this perspective, we propose that DSK2 functions more broadly as a multi-client 'growth-switch' hub, coordinating the targeted turnover of additional growth-promoting regulators, including class I TEOSINTE BRANCHED1, CYCLOIDEA, PROLIFERATING CELL FACTOR 1, and 2 (TCP) transcription factors. Phenotypic evidence supports a role for DSK2-mediated TCP degradation in drought tolerance. We present a conceptual model in which DSK2 integrates diverse stress signals to reprioritize metabolic resources, maintain cellular homeostasis, and safeguard reproductive development. Elucidating the full target spectrum of DSK2 will deepen our understanding of post-translational regulation in plant stress adaptation and open new avenues for engineering climate-resilient crops, an urgent imperative for global food security in an era of accelerating climate change.
-
Key words:
- Drought /
- DSK2 /
- Transcription factor /
- TCP














