-
The rapid development of industry and the economy has significantly improved our quality of life. However, this progress has been accompanied by increasingly severe environmental and resource challenges. Large quantities of pollutants are being released into the environment, with water and soil contamination posing serious threats to ecosystems and human health. Major pollutants include heavy metals (HMs)[1], and organic contaminants such as antibiotics[2,3], dyes[4], and pesticide residues[5]. Heavy metals are persistent, non-biodegradable, and tend to accumulate in organisms through the food chain[6,7]. Organic pollutants are highly toxic, mobile, resistant to degradation, and difficult to remove, presenting substantial risks to public health[8−10].
Given the severity of environmental pollution, there is an urgent need to develop low-cost, high-efficiency contaminant removal technologies. Among the various available methods—such as adsorption[11], catalytic degradation and filtration[10]—adsorption stands out as one of the most successful separation techniques due to its simple design, cost-effectiveness, broad applicability, and high efficiency[12,13]. Recently, biochar (BC) has emerged as a promising adsorbent, demonstrating considerable potential for pollutant removal owing to its wide range of feedstocks, large specific surface area (SSA), abundant functional groups, low cost, and high porosity[14,15]. Biochar produced from natural raw materials represents a stable form of carbon that can persist for centuries or even millennia[16,17], suggesting its potential role in climate change mitigation[18]. Nevertheless, pristine biochar has limitations in selectively adsorbing high concentrations of pollutants, especially under increasingly stringent environmental standards[19,20]. To address these shortcomings, BC-based composites have been developed through various modification or activation methods—such as thermal/gas activation, acid/alkali treatment, organic/inorganic material decoration, and nanomaterial coating—to enhance SSA, pore structure, and surface functional groups, thereby improving adsorption performance[21,22]. Through magnetization modification[23], the addition of nanoparticles[24], or functional materials[25], BC-based composites can be tailored for specific target contaminants, acquiring desirable properties such as unique structures and abundant functional groups. A variety of biomass materials, including straw, rice husks, corncobs, leaves, animal manure, and wastewater sludge, are commonly used to produce biochar. With the rapid development of the traditional Chinese medicine industry, the output of traditional Chinese medicinal herb residues (TCMHR) (mainly including drug residues and non-medicinal waste) has increased dramatically[26]. Currently, TCMHR is still mainly disposed of through stacking, incineration, or landfilling. These extensive treatment methods not only harm the ecological environment but also lead to resource waste[26−28]. TCMHR is rich in resources, containing abundant organic components such as cellulose, hemicellulose, and lignin, which are ideal feedstocks for biochar[29]. Previous studies by our research group have shown that biochar derived from TCMHR, as well as its modified composites, exhibit strong potential for adsorbing heavy metal pollutants[30−32].
This review systematically examines the state-of-the-art advancements in biochar (BC) and BC-based composites derived from traditional Chinese medicine herb residues (TCMHR) for wastewater remediation. The physicochemical properties of TCMHR-BC are first elucidated, emphasizing its unique structural advantages (e.g., high porosity, functional group diversity) governed by lignocellulosic feedstock and pyrolysis conditions. Subsequently, we synthesis strategies for BC and its composites are critically analyzed (e.g., magnetic modification, nanomaterial hybridization), along with their mechanisms for removing inorganic (e.g., Cr[VI], Pb[II]), and organic (e.g., antibiotics, dyes) contaminants. A dedicated discussion unravels pollutant-biochar interactions, including adsorption-reduction-precipitation pathways for metals, and π–π/electrostatic interactions for organics. Finally, key challenges are identified—such as competitive adsorption in multi-pollutant systems and long-term stability of modified BC—and propose future research priorities, including eco-toxicity assessments, and scalable applications of compound TCMHR. This work not only deepens the mechanistic understanding of TCMHR-BC but also provides guidance for its rational design in sustainable resource recovery, and environmental remediation. Furthermore, as a by-product of decoction, TCMHR is reported to retain approximately 30%–50% of the medicinal active constituents[33]. TCMHR biochar demonstrates exceptional adsorption properties, exhibiting outstanding environmental remediation potential and multifunctional development value (antibacterial and antioxidant characteristics). A detailed discussion follows in the subsequent sections.
-
Biochar (BC) is a carbon-rich, microporous material produced through the pyrolysis and carbonization of biomass under high-temperature, oxygen-limited, or anaerobic conditions[34]. Its surface is characterized by a variety of functional groups, such as phenolic hydroxyl, hydroxyl, and carboxyl groups. In recent years, BC has been widely applied in numerous fields, including water pollution treatment[35], energy production[36], soil remediation[37], reducing greenhouse gas (GHG) emissions[38], synthesis of pharmaceutical compounds, and other fields[39,40]. Biochar derived from TCMHR can form porous structures that enhance pollutant removal through mechanisms such as pore filling[41]. In this article, the raw materials for biochar derived from TCMHR have been summarized (see Table 1 for details).
Table 1. Biochar preparation method from TCMHR
Biomass Pyrolysis temperature (°C) Residence time (h) Heating rate (°C/min) Pyrolysis mode Target pollutant Related TCM* Ref. Saussurea involucrata 200–600 2 5 Mild pyrolysis and
high pyrolysisMethyl red (MR) and methyl orange (MO) SAUSSUREAE INVOLUCRATAE HERBA [60] TCM raw material (Forsythia suspensa, Honeysuckle, combination) 700 2 h 5 High pyrolysis Tetracycline (TC) LONICERAE JAPONICAE FLOS, FORSYTHIA FRUCTUS [61] Chrysanthemum residues
(Chuju, Chrysanthemum morifolium)300–700 2 5 Mild pyrolysis and
high pyrolysisQuercetin (QR) CHRYSANTHEMI FLOS [62] Atropa belladonna L. (ABL) 899.85 1–5 5 High pyrolysis Norfloxacin (NOR) BELLADONNAE HERBA [63] Ligusticum chuanxiong Hort. 500 2 10 Mild pyrolysis Cr(VI), As(III),
and Pb(II)CHUANXIONG RHIZOMA [30] Codonopsis pilosula (DGS), Astragalus membranaceus (HQ), Angelica sinensis (DG),
Ligusticum striatum (CX),
Salvia miltiorrhiza (DNS).500 2 − Mild pyrolysis Pb(II) SALVIAE MILTIORRHIZAE RADIX ET RHIZOMA, CHUANXIONG RHIZOMA, ANGELICAE SINENSIS RADIX, CODONOPSIS RADIX, ASTRAGALI RADIX [31] Danshen residue
(dried root of Salvia miltiorrhiza Bunge.)250–800 3 − Mild pyrolysis and
high pyrolysisSulfamethoxazole (SMX) SALVIAE MILTIORRHIZAE RADIX ET RHIZOMA [3] The root of Clematis chinensis Osbeck 500 2 5 Mild pyrolysis Tetracycline (TC) CLEMATIDIS RADIX ET RHIZOMA [2] Codonopsis pilosula (Dangshen) and Angelica sinensis (Danggui) 300–750 2 3–6 Mild pyrolysis and
high pyrolysisMetolachlor CODONOPSIS RADIX, ANGELICAE SINENSIS RADIX [5] Astragalus mongholicus residues 400 3 − Mild pyrolysis Cr(VI) ASTRAGALI RADIX [53] Astragalus membranaceus residue 700 3 − High pyrolysis Ciprofloxacin (CIP) ASTRAGALI RADIX [23] Waste Chinese traditional medicine dregs containing Acanthopanax senticosus, ginseng and Astragalus 450 1 10 Mild pyrolysis As(III) ACANTHOPANACIS SENTICOSI RADIX ET RHIZOMA SEU CAULIS, GINSENG RADIX ET RHIZOMA, ASTRAGALI RADIX [29] Forsythia and honeysuckle 400–600 1 10 Mild pyrolysis and
high pyrolysisChlortetracycline (CTC) FORSYTHIAE FRUCTUS, LONICERAE JAPONICAE FLOS [48] Chinese medicine residues (CMR) contained Polygonum multiflorum, Uncaria, etc. 450–650 2 − Mild pyrolysis and
high pyrolysisAmmonium (NH4+−N) POLYGONI MULTIFLORI RADIX, UNCARIAE RAMULUS CUM UNCIS [64] Raw medicinal residues contained folium eriobotryae, liquorice, platycodon grandiflorum, rhizome typhonii flagelliformis, etc. 600 1 20 High pyrolysis Element mercury (Hg) ERIOBOTRYAE FOLIUM, GLYCYRRHIZAE RADIX ET RHIZOMA, etc. [65] The mixture of polygonatum sibiricum and other herb residues 300–600 2 − Mild pyrolysis and
high pyrolysisPb(II) POLYGONATI RHIZOMA [66] Honeysuckle residue (HR) 400 3 − Mild pyrolysis Cr(VI) LONICERAE JAPONICAE FLOS [67] Chinese medicine residues (CMRs) mainly contain Astragalus, Panax ginseng, licorice, Chen Pi, dry ginger, Ginkgo biloba, etc. 300–700 2 10 Mild pyrolysis and
high pyrolysisHeavy metals
(Cu, Cd and Pb)GINSENG RADIX ET RHIZOMA, ASTRAGALI RADIX, GLYCYRRHIZAE RADIX ET RHIZOMA, ZINGIBERIS RHIZOMA, GINKGO FOLIUM, etc. [68] 100–300 °C: Low pyrolysis; 300–500 °C: Mild pyrolysis; 500–900 °C: High pyrolysis. * Pharmacopoeia of the People's Republic of China (Edition 2020) related Traditional Chinese Medicine (TCM). Various feedstock types correspond to specific pyrolysis conditions owing to variations in inorganic mineral contents, hemicellulose, lignin, and cellulose[42,43]. Consequently, BC produced from different feedstocks under identical temperatures exhibits distinct physicochemical properties[5,44−46]. Current understanding indicates that both raw materials and pyrolysis conditions critically influence the physicochemical characteristics of biochar. Pyrolysis conditions, in particular, affect yield, ash content, specific surface area (SSA), pore size, pH, and surface functional groups[3,5,45]. Pyrolysis temperatures between 200 and 800 °C are commonly selected to achieve the desired properties of biochar[7]. The choice of feedstock and pyrolysis parameters—including temperature, residence time, and heating rate—governs the formation of pores and cracks, SSA, and surface functional groups of biochar[47], which in turn determine its adsorption capacity, as outlined in Table 2. For instance, a study showed that honeysuckle residue biochar pyrolyzed at 500 °C exhibited the highest chlortetracycline (CTC) removal efficiency compared to those produced at 400 and 600 °C[48]. Generally, increasing pyrolysis temperature leads to higher ash content, SSA, total pore volume, and pH, while reducing yield, O/C ratio (indicating lower polarity), and H/C ratio (reflecting higher aromaticity)[49].
Table 2. Effects of pyrolysis temperature on the physicochemical properties of biochar derived from TCMHR
Raw material T (°C) Yield (%) Ash (%) BET (m2/g) Vtotal (cm3/g) pH H/C O/C Ref. Saussurea involucrata 200 95.77 ± 0.77 − 1.6525 0.009217 − − − [60] 300 94.18 ± 0.92 − 1.8538 0.005995 − − − 400 93.23 ± 1.12 − 2.1093 0.005432 − − − 500 92.34 ± 0.88 − 3.0227 0.011160 − − − 600 91.48 ± 0.79 − 1.9364 0.005603 − − − Chrysanthemum residues (Chuju, Chrysanthemum morifolium) 300 52.13 14.85 0.427 0.001943 − 0.074 0.795 [62] 500 38.65 28.52 1.397 0.007105 − 0.063 0.690 700 27.55 31.02 2.390 0.007992 − 0.051 0.516 Danshen residues (dried root of Salvia miltiorrhiza Bunge.) 250 91.90 14.70 3.80 − 6.7 0.240 0.641 [3] 400 37.90 28.30 49.20 0.0420 10.2 0.108 0.165 600 31.20 31.10 51.30 0.0510 10.1 0.039 0.052 800 29.10 37.10 70.30 0.0680 10.6 0.007 0.064 Angelica sinensis (Danggui) and Codonopsis pilosula (Dangshen) 300 − 49.46 3.24 0.0116 7.4 0.870 0.310 [5] 500 − 54.70 54.28 0.0534 9.7 0.480 0.180 750 − 57.65 85.30 0.0803 9.8 0.240 0.170 Radix Isatidis residues 300 − 18.37 4.45 0.0075 7.5 0.079 0.240 [44] 500 − 19.66 8.50 0.0125 7.8 0.033 0.116 700 − 23.42 11.80 0.0178 8.1 0.023 0.055 Chinese medicine residues (CMR) contained Polygonum multiflorum, Uncaria, etc. 450 − 25.64 13.46 0.0072 − 0.060 0.280 [64] 550 − 17.59 12.07 0.0112 − 0.040 0.250 650 − 22.17 14.67 0.0132 − 0.030 0.200 The H/C atomic ratio is a key indicator of biochar carbonization degree or aromaticity, while the O/C atomic ratio reflects hydrophilicity, and the (O + N)/C ratio characterizes polar functional groups[50], with elevated H/C and O/C ratios indicating reduced aromatic structures, and increased polarity in biochar[47]. As the temperature increases, the thermogravimetric loss, hydrogen-to-carbon atom ratio, and carbon oxidation loss rate (K2Cr2O7 method) of biochar gradually decrease, indicating enhanced thermal stability, degree of aromatisation, and chemical oxidation stability. The reduction in O/C and H/C atom ratios may be attributed to the gradual increase in carbon content, and decrease in oxygen and hydrogen content[51]. Biochar adsorption capacity is closely linked to its physicochemical properties, such as functional groups, stable carbon framework, and well-developed pore structure. The aromatic matrix is particularly important for adsorbing hydrophobic organic compounds (HOCs), with studies showing a strong positive correlation between aromaticity and adsorption affinity[52]. For example, magnetic biochar (MBC) prepared from astragalus residue pyrolyzed at 400 °C for 3 h, and modified via Fe3+/Fe2+ coprecipitation achieved a maximum Cr(VI) adsorption capacity of 23.85 ± 0.23 mg/g at pH 2[53]. However, the maximum adsorption capacity for ciprofloxacin at pH 6 was 68.9 ± 3.23 mg/g for the adsorbent obtained by the same method at 700 °C[23], showing good adsorption performance for antibiotics. H/C and O/C ratios are also associated with biochar longevity and stability. The Van Krevelen diagram, which tracks changes in H/C and O/C ratios with pyrolysis temperature, is commonly used to describe the pyrolysis process (Fig. 1). Spokas[54] described BCs with O/C ratios: (a) < 0.2, half-life > 1,000 years (highly stable); (b) between 0.2 and 0.6, half-life between 100 and 1,000 years (moderately stable); and (c) > 0.6, half-life < 100 years (relatively unstable). Additionally, BC with an H/C ratio below 0.7 possesses a more condensed aromatic structure compared to those with higher H/C ratios (> 0.7).

Figure 1.
The relationship between the molar ratio of H/C and O/C and pyrolysis temperature (Van Krevelen Diagram)[49].
Increased ash content and SSA at higher temperatures are attributed to reduced carbon components, and greater micropore volume[55]. As the pyrolysis temperature increased from 250 to 800 °C, the SSA and ash content of biochar respectively increased from 3.8 m2/g, 14.7% to 70.3 m2/g, 37.1%[3]. Ash content correlates positively with pH and negatively with yield; higher ash content elevates pH while reducing BC yield. Moreover, high-temperature pyrolysis promotes volatile loss and organic matter decomposition, further expanding total pore volume through porous structure formation[46,56]. Higher lignin-containing lignocellulosic biomass is reported in the literature to produce higher biochar yields[57], as confirmed by the positive relation between lignin content and biochar yield reported by Yuan et al.[31] (p < 0.05, r = 0.90) (Fig. 2). Recent research has found that biochar yield decreases significantly with increasing residence time[58]. However, Sun et al.[59] suggested that different residence time at high temperatures has little effect on the yields. In summary, extensive research confirms that the physicochemical properties of biochar are influenced by raw materials, pyrolysis temperature, residence time, heating rate, and pyrolysis type. However, these relationships are not absolute; optimal biochar production conditions should be selected based on specific application requirements and contextual constraints.
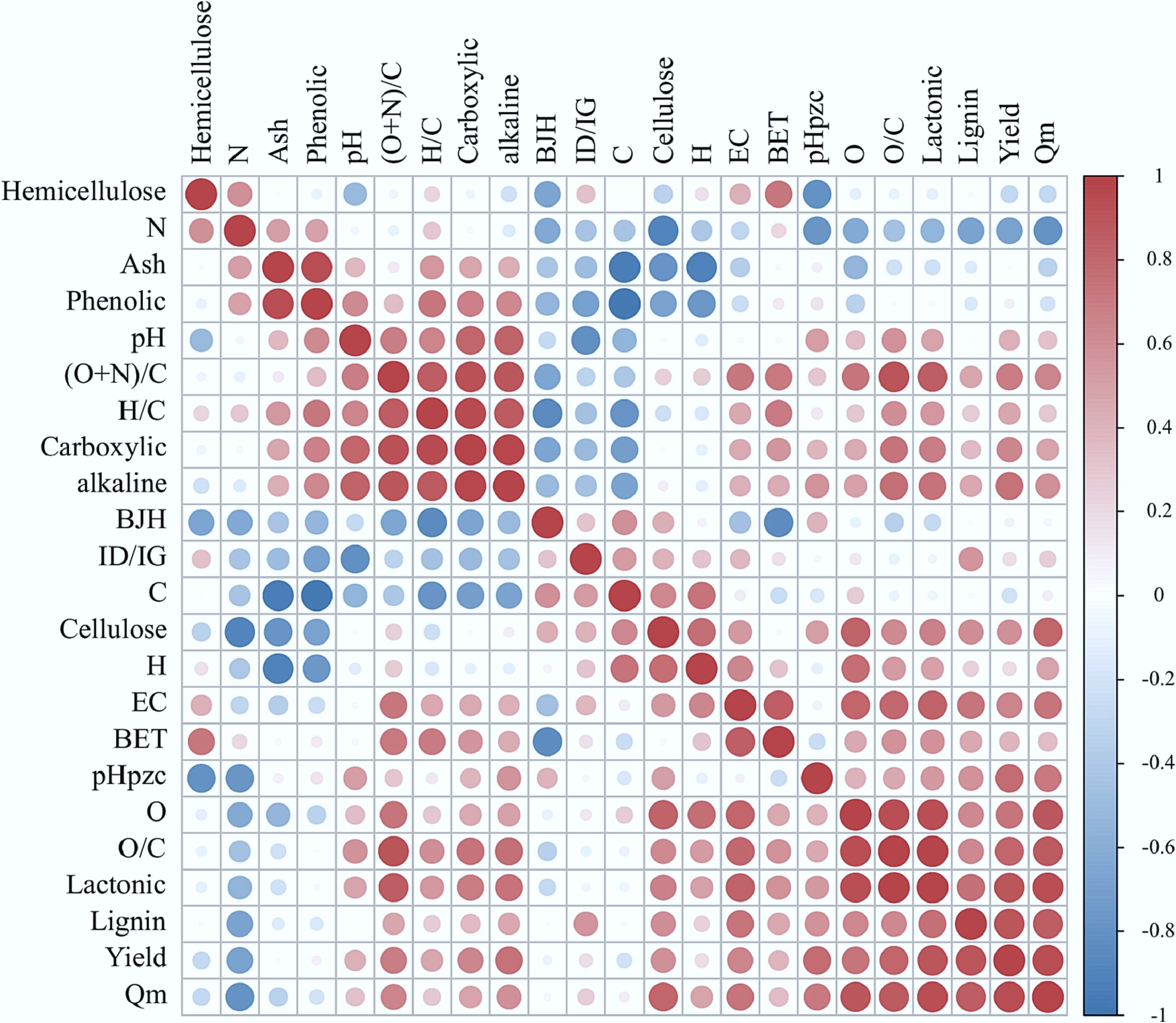
Figure 2.
The Spearman correlation matrix of physicochemical properties of the TCMHR biochar[31].
-
Given the limitations of pristine biochar (BC) that hinder its effectiveness in environmental remediation, an increasing number of studies have focused on modifying the physicochemical properties of BC through activation or functionalization, as well as preparing BC-based composites by combining BC with other materials. These composites integrate the advantages of BC and the incorporated materials, thereby enhancing pollutant removal performance.
Modification of biochar can effectively alter its properties, such as increasing the specific surface area (SSA), improving adsorption capacity, and modifying surface functional groups[69]. Different treatment methods and modifiers directly influence the adsorption capacity and mechanisms of biochar toward pollutants. Therefore, selecting an appropriate modification strategy is crucial for efficient contaminant removal. This section discusses several major types of BC-based composites, including magnetic biochar, biochar-based nanocomposites, and other functionalized biochars, to illustrate recent advances in the application of herbal residue-derived biochars. The modification techniques for composite biochar derived from traditional Chinese medicinal herb residues (TCMHRs) are summarized in Table 3.
Table 3. Diverse techniques for modifying composite biochar derived from TCMHRs
Type Material Synthesis method Composite particle Contaminant Best fit
kinetic modelBest fit isotherm model Ref. Loading magnetic compositions Astragalus membranaceus residue Biochar was added to a solution containing ferric chloride hexahydrate and ferrous sulphate, then NaOH solution (10 mol/L) was added to raise the pH to 10.5, finally stirred at 25 °C, 1 h under nitrogen atmosphere Fe3O4 Cr(VI) Pseudo-second-order Langmuir [53] Astragalus membranaceus residue Added biochar into Ferric and ferrous (Fe3+/Fe2+) mixed solutions, then stirred for 60 min at 25 °C, under a N2 atmosphere at 10.5 pH adjusting by NaOH solution Fe2O3, Fe3O4 Ciprofloxacin (CIP) Pseudo-second-order Langmuir [23] The root of Clematis chinensis Osbeck Chemical co-precipitation method: added BC into a solution containing Mn2+ and MnO4−, then exposed to outdoor conditions for a period of 24 h MnO2 Tetracycline (TC) Pseudo-second-order Langmuir and Freundlich [2] Herb residue Biomass powder was immersed in diluted waste liquor from the pickling of steel, then slow stirring for 12 h, finally pyrolyzed at 600 °C in an N2 atmosphere for
2 h at a timeFe3O4 and
γ-Fe2O3Hexavalent chromium
(Cr(VI))Pseudo-second-order Langmuir [70] Chinese medicine residue (CMR) from a pharmacy in Zhengzhou, China The magnetic biochar was obtained from Fe2+/Fe3+ co-precipitation method, then added the magnetic biochar into Al(NO3)3 solution (0.5 mol/L), next, added NaOH solution (3 mol/L) to the mixture until the pH = 5, finally stirred for 30 min for complete reaction Al/Fe3O4 Aqueous fluoride Pseudo-second-order Freundlich [72] Astragalus membranaceus residues Herb biochar was dissolved in Fe2+ solution and stirred 2 h under N2, then added dropwise the NaBH4 solution, finally stirred another 60 min under N2 n-ZVI particles Cr(VI) Pseudo-second-order − [24] Nanoscale-metal assistant Honeysuckle residue (HR) Added biochar into a solution containing ferric chloride with the solid–liquid ratio 1:20 (W/V), then stirred for 8 h at 150 rpm Fe3C particles Cr(VI) Pseudo-second-order Langmuir [67] Waste Chinese traditional medicine dregs containing Acanthopanax senticosus, ginseng and Astragalus TiO2−loaded biochar (TBC) was synthesized by a modified solgel method: BC was added into a solution containing concentrated hydrochloric acid, butyl titanate and anhydrous ethanol, anhydrous ethanol was added dropwise into the mixture and the solution was slowly stirred until it had formed a gel. Finally, aging at 25 °C for 24 h without light and dried at 60 °C for 24 h TiO2 Arsenic (III) Pseudo-
second-orderSips [29] The residue of Flueggea suffruticosa Biomass was impregnated with ZnCl2
(2.75 mol/L), then stirred for 1 h and dried, finally pyrolyzed at 500 °C for 90 minZinc chloride (ZnCl2) Tetracycline (TC), chlortetracycline (CTC) and oxytetracycline (OTC) Pseudo-
second-orderLangmuir
(TC, CTC, OTC)[73] Surface modification A membranaceus residue Biomass was impregnated with Na2CO3
(10 wt%) for 24 h and dried at 105 °C, finally pyrolyzed at 800 °C for
1.5 h under N2 atmosphereNa2CO3 Cefradine (CF) in wastewater Pseudo-
second-orderFreundlich [74] Glutinous Rehmannia, Codonopsis pilosula,
Pseudo-ginseng,
Rhizoma GastrodiaeHerbal dregs biochar and potassium hydroxide solids were blended at various ratios, added deionized water, stirred and placed in an oven to dry, then heated in a tube furnace, 150 mL of 30% hydrochloric acid solution was slowly mixed with 10 g of the above product, stirred for 3 h at
70 °C and washed to neutralityKOH Pb(II) Pseudo-second-order Langmuir [75] Saussurea involucrata The drug residue was impregnated in a 10% Na2CO3 solution (1:8) for 24 h. The drug residue was accurately weighed, and the modified carbon was washed with
0.1 mol of HClNa2CO3 Methyl red and methyl orange Pseudo-second-order − [60] The most widely applied preparation methods for MBC involve liquid impregnation pyrolysis[70], co-precipitation[2], and liquid-phase reduction[24]. In the liquid impregnation pyrolysis method, biomass is first impregnated in an Fe2+/Fe3+ salt solution or other magnetic ion solution under stirring, then dried, and finally pyrolyzed to obtain MBC. Yi et al.[70] first used steel pickling waste liquor to replace iron salt to synthesize MBC. The biomass powder, consisting of peanut shells, rice straw, and herb residue, was slowly stirred for 12 h in diluted steel pickling waste liquor, before being pyrolyzed at 600 °C for 2 h in a N2 atmosphere. The study indicated that the biomass with lower ash content and higher cellulose content was an ideal feedstock for the preparation of MBC, which demonstrated outstanding removal performance for Cr (VI). The co-precipitation method first requires the preparation of BC, after which BC is added to a solution containing magnetic iron salt to form a suspension. Next, NaOH solution is added dropwise to raise the pH with mixing under a N2 atmosphere at room temperature or in open-air conditions, and then precipitation occurrs. Shang et al.[53] prepared MBC with Astragalus membranaceus residue using the Fe2+/Fe3+ co-precipitation method. MBC from low-cost herb residues had the potential for recycling under an external magnetic field, and can be applied as an environmentally friendly and effective adsorbent for treating Cr(VI) contaminated wastewater. Apart from the above, the liquid phase reduction method is to add dropwise KBH4/NaBH4 solution into a suspension containing BC and Fe2+/Fe3+ under N2 to reduce Fe2+/Fe3+ into Fe0 (e.g., nZVI). Shang et al.[24] applied this approach to prepare nanoscale particles of zerovalent iron supported on biochar made from herbal residue (nZVI/BC), and nZVI/BC was found to be an effective reagent for the treatment of acidic wastewater contaminated with Cr(VI). MBC's major composite particles are Fe3O4 and/or Fe2O3, which aid in the separation of pristine BCs from aqueous solutions. Furthermore, the MBC made by the reduction method with nano zerovalent iron (nZVI) had high adsorption performance for contaminants due to the strong reducing power of nZVI. However, this procedure required the use of a reduction reagent (e.g., KBH4/NaBH4) as well as the introduction of a protective gas (e.g., N2) throughout the preparation process. Furthermore, nZVI is easily oxidized and difficult to maintain, which limits its application.
Biochar is a versatile material with a unique structure, ideal for combining with nanomaterials to create biochar-based nanocomposites. The deposition of nanoparticles on BC improved the affinity of BC for heavy metals, increased the adsorption sites and SSA of BC, improved the thermal stability and reaction mechanism of BC, and also improved the oxidation resistance of BC[71]. For instance, composites prepared from BC and nanomaterials (e.g., TiO2) can significantly improve adsorption capacity, surface functional groups, and microporous structure[29]. Zheng & Duan[67] impregnated biochar (HDB) into a solution containing ferric chloride for post-treatment to obtain Iron-Modified Biochar (MHDB). Comparing the physicochemical properties (average pore size and SSA) of HDB and MHDB, it is evident that iron modification significantly enhances the structure, decreases the pore size, and simultaneously increases the SSA. Additionally, on the surface, Fe–O–C structures have been created, which provided adsorption active sites to facilitate wider distributions of Cr (VI) on BC. In summary, biochar-based nanocomposites can significantly increase the SSA, improve surface functional groups or active sites, optimize the pore structure, and increase the adsorption capacity for contaminants effectively.
In addition to the above methods, mineral salt (KH2PO4, Na2CO3, etc.) modification or acid/alkali (HCl, H2SO4, NaOH, etc.) treatment can also have an impact on the physico-chemical characteristics of BCs.
-
Public attention is increasingly focused on the severe issue of heavy metal pollution. As a low-cost adsorbent, biochar (BC) derived from traditional Chinese medicine herb residues (TCMHR) has shown broad potential in wastewater treatment applications. The removal of inorganic contaminants by BCs is summarized in Table 4, with the maximum adsorption capacity (Qmax) for inorganic pollutants ranging from 7.45 to 599.40 mg/g.
Table 4. Removal of inorganic pollutions by biochar derived from TCMHRs
Material Contaminants Modification method pH Best fit kinetic model Best fit isotherm model Adsorption capacity
(mg/g)Sorption mechanism Ref. The TCMHR were collected from a pharmacy Cr(VI) Zinc chloride (ZnCl2) and Iron based waterworks sludge (IBWS) pH = 2 Pseudo-second-order Langmuir 27.04 Physisorption, chemisorption reduction [79] Chinese medicine residue Pb(II) HCl/KOH pH = 5 Pseudo-second-order Langmuir 74.38 Coordination, co-precipitation, surface complexation, electrostatic attraction, ion exchange [75] Astragalus membranaceus (HQ), Codonopsis pilosula (DGS), Angelica sinensis (DG), Ligusticum striatum (CX), Salvia miltiorrhiza (DNS) Pb(II) − − Pseudo-second-order Langmuir 36.42 Complexation with oxygen-containing functional groups, ion exchange,
precipitation[31] Ligusticum chuanxiong Hort. residues Cr(VI)
As (III)
Pb(II)Fe3O4/
Chitosan (CTS)pH = 4 Pseudo-second-order Langmuir 65.74
49.32
69.45Redox interactions, complexation adsorption. Ion exchange, electrostatic attraction. [30] Astragalus membranaceus residue Cr(VI) Fe3O4 pH = 2 Pseudo-second-order Langmuir 45.45 − [53] Astragalus membranaceus residue Cr(VI) nZVI pH = 2 Pseudo-second-order − − Reduction and coprecipitation [24] The TCMHR mainly included acanthopanax senticosus, ginseng and Astragalus As(III) Nano-TiO2 − Pseudo-second-order Langmuir 58.46 O2 acted as an electron accepter and O2•− dominated the oxidation of As(III). [29] Chicken manure and Chinese medicine residue Pb(II) KH2PO4 pH = 6 Pseudo-second-order Langmuir 599.40 Surface complexation,
ions exchange, coprecipitation,[76] Turkish gall (TG) residue Pb(II) − − Pseudo-second-order − − − [56] Honeysuckle residue Cr(VI) FeCl3 pH = 2 Pseudo-second-order Langmuir 13.70 Physical adsorption,
chemical adsorption[67] Herb residue Cr(VI) Fe3O4 and
γ-Fe2O3Pseudo-second-order Langmuir 11.73 Reduction, complexed [70] Chinese medicine residue was collected in a hospital Cu(II) Nano-hydroxyapatite (nHAP) − − Freundlich 61.96 − [80] Zhou et al.[56] produced BCs from Turkish gall residue across a range of pyrolysis temperatures (200 to 600 °C), and investigated their physicochemical properties and adsorption performance for Pb(II). The results indicated that the biochar structure was altered as the pyrolysis temperature increased. Specifically, ash content, fixed carbon, and pH gradually rose with temperature, while yield and the abundance of surface functional groups declined. The adsorption kinetics of Pb(II) followed the pseudo-second-order (PSO) model, and the expected equilibrium adsorption capacity (Qe,exp) of the optimal BC reached 168.34 mg/g. In our laboratory, biochars were also prepared via slow pyrolysis from five common Chinese medicine residues: Astragalus membranaceus (HQ), Codonopsis pilosula (DGS), Angelica sinensis (DG), Ligusticum striatum (CX), and Salvia miltiorrhiza (DNS). The adsorption characteristics and mechanisms of these biochars for Pb(II) were examined, revealing a clear correlation between the physicochemical properties of the biochar and the type of herb residue. Biochar derived from biomass with higher lignin content exhibited superior Pb(II) adsorption, with BDNS showing the highest capacity (36.42 mg/g), followed by BHQ (7.45 mg/g), BCX (7.84 mg/g), BDG (9.00 mg/g), and BDGS (18.67 mg/g)[31].
Previous studies have demonstrated that BC can be modified with magnetic particles to enable easy separation from water using an external magnetic field, and that other modifications (e.g., with KH2PO4 or TiO2) can enhance its adsorption performance and physicochemical properties. Chen et al.[76] developed an efficient adsorbent by preparing KH2PO4-modified biochar (PBC) from Chinese medicine residue (CMR), and chicken manure (CM) for Pb(II) removal. PBC exhibited excellent applicability across a wide pH range (1–6), with zeta potentials varying from –3.2 to –43.1 mV. The results indicated that initial pH significantly influenced PBC performance, and chemical adsorption was identified as the primary mechanism. Moreover, PBC displayed an extraordinary Pb(II) adsorption capacity, with a Qmax of 599.40 mg/g, largely attributed to its abundant functional groups. Our team also developed a novel chitosan-modified magnetic biochar (CMBC), which at pH 4.0 showed maximum adsorption capacities of 49.32 mg/g for As(III), 69.45 mg/g for Pb(II), and 65.74 mg/g for Cr(VI). The removal process followed the Langmuir isotherm and pseudo-second-order kinetics. Based on batch adsorption experiments, density functional theory (DFT) calculations, and characterization analyses (Fig. 3a), the adsorption mechanisms of CMBC for Pb(II), As(III), and Cr(VI) included electrostatic attraction, ion exchange, complexation, and redox interactions[30]. Ren et al.[75] used KOH activation of herbal dregs to prepare activated biochar as an efficient biosorbent for Pb(II), achieving an adsorption capacity of 74.38 mg/g under optimal conditions (adsorption mechanism illustrated in Fig. 3b). Modified biochars consistently demonstrated better heavy metal adsorption than pristine biochar and possessed unique functional properties. However, research on heavy metal adsorption by herb-residue-based biochar remains limited, and further investigations are needed to address practical application challenges.
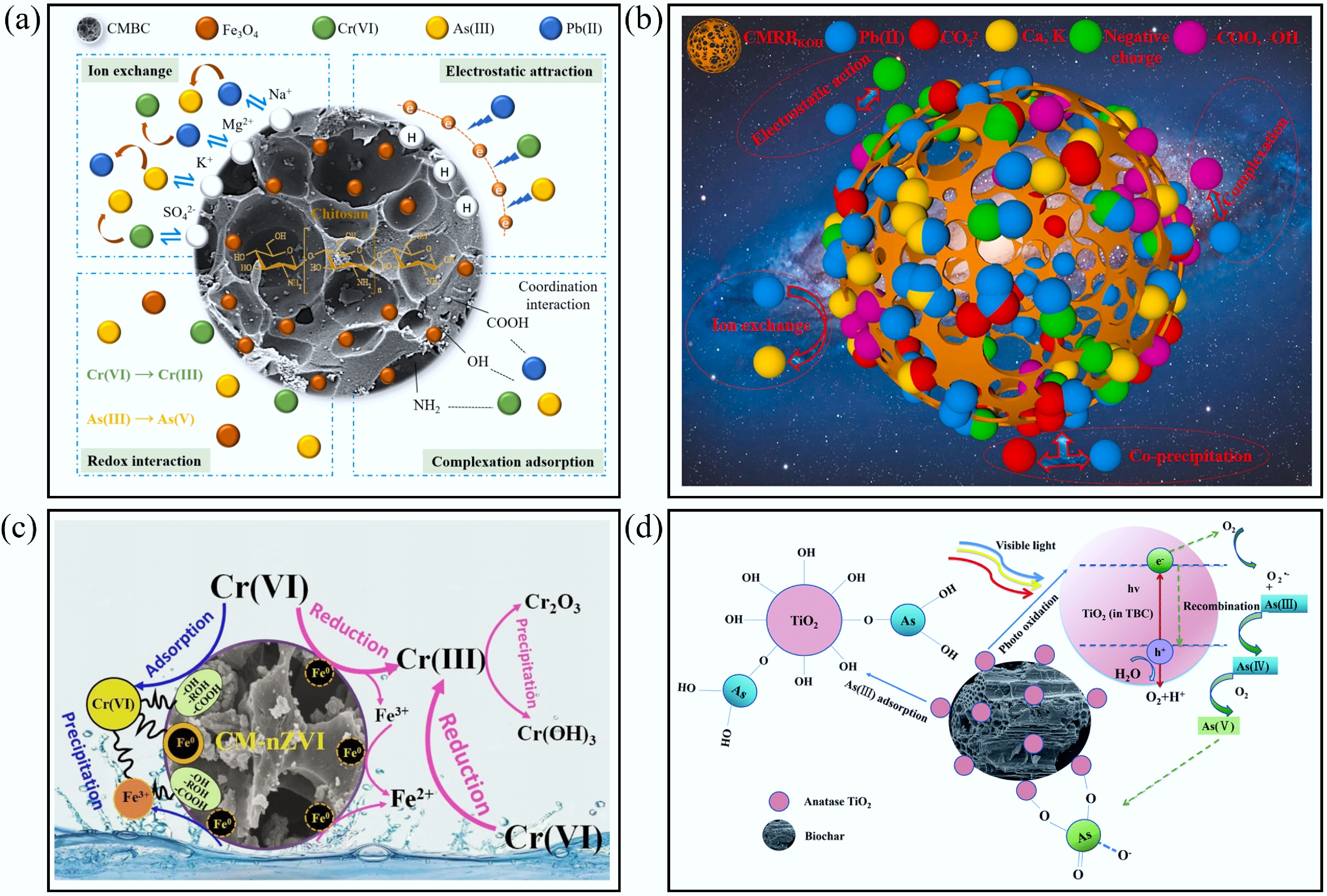
Figure 3.
Typical adsorption mechanisms of heavy metal ions by biochar composites. (a) The mechanism of adsorption of As(III), Pb(II), and Cr(VI) by CMBC[30]. (b) Possible mechanisms of Pb(II) sorption onto CMRBKOH[75]. (c) The reaction mechanism for the removal of Cr(VI) by CM-nZVI[78]. (d) The removal mechanisms of As(III) from aqueous solution by TBC[29].
Sorption–reduction–precipitation of high-valent metal ions
-
Adsorption is a common process for removing metal ions from aqueous solutions. However, if environmental conditions change, adsorbed metal ions may be released back into solution, necessitating consideration of desorption behavior. High-valence metal ions such as Cr(VI), Se(IV), As(V), and U(VI) can be reduced to lower-valence states upon adsorption, often forming stable precipitates on solid particles[77]. A novel composite of nano zero-valent iron and Chinese medicine residual carbon (CM-nZVI) has been effectively applied for Cr(VI) removal. Mechanistic studies revealed that Cr(VI) is partially adsorbed by CM-nZVI, while a larger fraction is reduced to Cr(III), which subsequently precipitates as Cr2O3 and Cr(OH)3. Additionally, some Cr(VI) species form precipitates through complexation reactions on CM-nZVI, collectively reducing the mobility of Cr(VI). Overall, adsorption, complexation, precipitation, and reduction processes all contribute to Cr(VI) removal by CM-nZVI[78] (Fig. 3c). Zheng et al.[79] successfully synthesized magnetic Fe-Zn@BC for Cr(VI) adsorption using a one-pot method. At pH 2 and 35 °C, the maximum adsorption capacity reached 27.04 mg/g. The primary mechanisms included both physical and chemical adsorption, as well as the reduction of Cr(VI) to Cr(III). Recently, Yang et al.[29] prepared biochar from mixed traditional Chinese medicine dregs (TBC) and modified it with TiO2 for As(III) removal. TiO2 modification reduced the average pore size of TBC but increased its total pore volume and specific surface area compared to unmodified biochar. The maximum As(III) adsorption capacity was 58.46 mg/g. Given that As(III) is more toxic than As(V), converting As(III) to As(V) is an effective strategy to enhance arsenic removal. Nitrogen-scavenging and free-radical-scavenging experiments indicated that oxygen (O2) acted as an electron acceptor and played a dominant role in oxidizing As(III), which significantly influenced the adsorption performance. XPS analysis confirmed the presence of both As(III) and As(V) on the TBC surface, indicating that oxidation of As(III) had occurred. TBC thus removed As(III) through a combination of photocatalytic oxidation and direct adsorption (Fig. 3d). For metal-ion-contaminated lakes, rivers, and soils, reduction-precipitation often represents a more effective treatment than adsorption alone. Since removing contaminants from sediments or soils is particularly challenging, future research should expand the exploration of reduction-precipitation strategies to develop broader and more practical remediation options.
-
Recently, discharging urban effluent containing high levels of organic pollutants from the paper industry, the pharmaceutical industry, and the food industry has caused organic pollution of water. Antibiotics, phenols, dyes, and pesticides are common organic contaminants in wastewater. Biochar (BC) exhibits excellent adsorption performance toward these pollutants owing to its rich surface functional groups and unique pore structure. The removal of organic contaminants by BC is summarized in Table 5, with reported maximum adsorption capacities (Qmax) ranging from 21.36 to 930.3 mg/g.
Table 5. Removal of organic pollutants by biochar derived from TCMHRs
Biomass Contaminant Modification method pH Qm (mg/g) Sorption mechanism Ref. Chinese herbal medicine Tetracycline (TC) MnO2 pH = 3 131.49 Physical adsorption and chemical interactions [2] Salvia miltiorrhiza Bge residue Methylene blue (MB) Citric acid and Na2CO3 pH = 7 161.29;
178.57− [4] Astragalus membranaceus residue Ciprofloxacin (CIP) Fe3O4 pH = 6 68.93 − [23] Astragalus mongholicus residue Ciprofloxacin (CIP) − pH = 7 43.67 − [81] Astragalus membranaceus residue Cefradine (CF) Na2CO3 pH = 3 64.76 Ion exchange, electrostatic adsorption, π-π EDA interaction and hydrogen bonding [74] Astragali radix Tetracycline (TC) ZnCl2 pH = 6 930.3 π-π interaction, electrostatic attraction, hydrogen bonding and pore diffusion [82] Flueggea suffruticosa residue Oxytetracycline (OTC) ZnCl2 pH = 7 129.90 Hydrogen bonding and electrostatic interaction [73] Chinese medicine residue was collected in a hospital Tylosin (TYL) Nano-hydroxyapatite (nHAP) pH = 6 50.02 − [80] Honeysuckle residue Chlortetracycline (CTC) Biochar-microbial (Bacillus subtilis) complexes pH = 7 − Biochar adsorption and microbial degradation [48] Chinese medicine residue was collected in a hospital Tylosin (TYL), sulfamethoxazole (SMX) Nano-hydroxyapatite (nHAP) − TYL: 52.02;
SMX: 51.22Surface adsorption [96] Forsythia suspensa, Honeysuckle, combination Tetracycline (TC) − − 36.92 Electrostatic interaction, π–π interaction, pore filling and hydrogen bonding [61] Atropa belladonna L. Norfloxacin (NOR) − pH = 7.4 − Electrostatic interaction, hydrogen bonding, pore diffusion and π–π interaction [63] Mulberry waste
(Chinese medicine residue)Tetracycline hydrochloride (TC) Ball milling pH = 4 103.7 − [97] Adsorption of organic pollutants by BCs
-
Antibiotics are extensively used in agriculture, animal husbandry, and human healthcare. However, their overproduction, irrational application, and improper disposal have resulted in severe environmental contamination. Most antibiotics are non-biodegradable, and their widespread abuse poses significant ecological risks[74]. Hence, there is a growing need to develop environmentally friendly, low-cost, and functional materials for antibiotic removal from water. Shang et al.[81] investigated the adsorption of ciprofloxacin (CIP) using BC derived from Astragalus mongholicus residue. Biochar produced at 800 °C exhibited the highest Qmax (43.67 mg/g) at pH 7, attributed to its larger surface area. Similarly, magnetic biochar (MBC) from Astragalus membranaceus residue has been applied for CIP removal[23], achieving a maximum adsorption capacity of 68.9 ± 3.23 mg/g at pH 6. Modification with Fe3O4 further enhanced the adsorption capacity of BC. The use of Astragalus-derived biochar for antibiotic removal has gained considerable attention. Recently, Zhao et al.[82] employed hydrothermal carbonization combined with ZnCl2 activation and pyrolysis to produce biochar from astragalus radix (AR) residues, which demonstrated an exceptionally high tetracycline (TC) adsorption capacity of 930.3 mg/g.
Phenolic contaminants primarily originate from industrial wastewater (e.g., oil refineries, plastics, disinfectants, pharmaceuticals, and steel production), as well as domestic and agricultural effluents. Representing a major class of organic pollutants, phenols are highly toxic, resistant to degradation, and prone to bioaccumulation, making their removal critically important[83]. Zhang et al.[84] prepared biochar from waste herbal residues at 400, 500, and 600 °C for phenol adsorption. Experimental results indicated that adsorption time, temperature, biochar preparation temperature, and initial phenol concentration all influenced removal efficiency. Among the three biochars tested, the adsorption capacity followed the order: BC600 > BC500 > BC400. BC600 achieved 97% phenol removal at an initial concentration of 50 mg/L, and a temperature of 45 °C.
Dyes are widely used in the textile, leather, paint, cosmetics, and food-processing industries. They are characterized by high chemical oxygen demand (COD), acidity, and resistance to degradation, and many are known to be carcinogenic, teratogenic, and hazardous to both ecosystems and human health. Even trace amounts of dyes such as methylene blue (MB), rhodamine B (RhB), methyl orange (MO), and Congo red (CR) can pose serious health risks[85]. Zhao & Zhou[4] used the extracted residue of Salvia mitiorrziza Bge (SM), a traditional Chinese medicine, as an effective biosorbent for methylene blue in contaminated water, and the experimental data showed that the maximum monolayer biosorption capacity of the raw SM was 100.0 mg/g.
The adsorption of organic pollutants onto biochar involves multiple mechanisms, with hydrogen bonding, electrostatic interactions, hydrophobicity, and pore filling being the primary contributing factors[86]. For instance, studies on tetracycline (TC) adsorption by honeysuckle and Forsythia suspensa-derived biochar indicated that electrostatic interactions, pore filling, hydrogen bonding, and π–π interactions played key roles[61]. Similarly, biochar produced from Atropa belladonna L. (ABLB4) was used to adsorb norfloxacin (NOR). At pH below the point of zero charge (pHpzc = 2.9), the negatively charged ABLB4 exhibited strong electrostatic attraction toward both NOR+ and neutral NOR species. In contrast, at pH > 8.51, electrostatic repulsion with NOR– reduced removal efficiency[63]. A generalized adsorption process is illustrated in Fig. 4a. Biochar from Flueggea suffruticosa residue was also employed to remove oxytetracycline (OTC), chlortetracycline (CTC), and TC, with hydrogen bonding, and electrostatic interactions dominating the adsorption (Fig. 4b). Thermodynamic and kinetic analyses revealed that the antibiotic adsorption on Zn-modified BC involved both physical and chemical processes. Hydroxyl and carboxyl groups on the biochar surface formed hydrogen bonds with hydroxyl and amine groups of tetracycline antibiotics, an interaction that plays a crucial role in TC adsorption[73]. By thoroughly elucidating the mechanisms of antibiotic adsorption on biochar, we can better understand the underlying interactions and behavior. Such insights are valuable for optimizing biochar preparation conditions and enhancing adsorption efficiency, ultimately contributing to the development of more effective and sustainable solutions for antibiotic removal and the protection of environmental and human health.
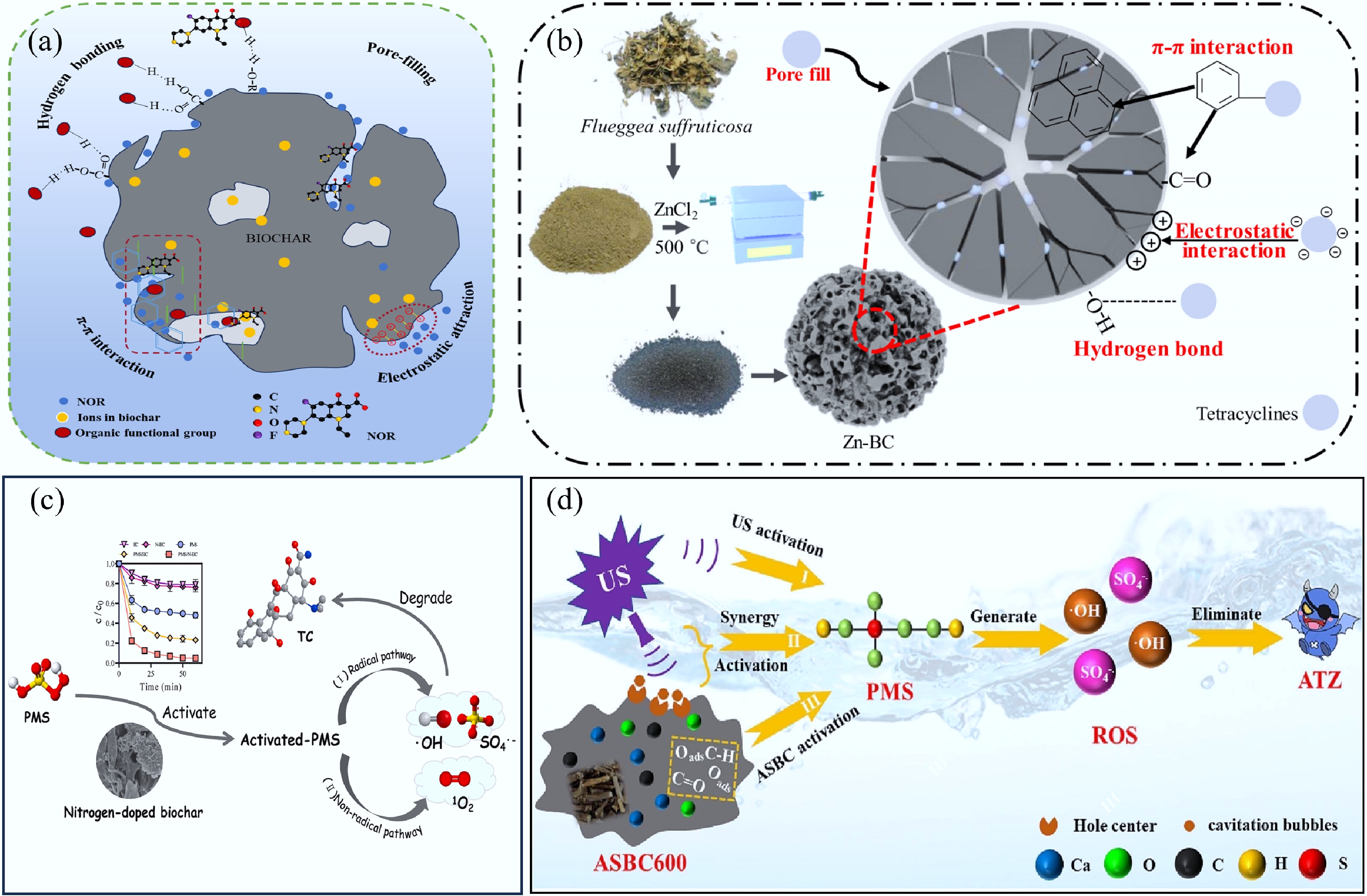
Figure 4.
Typical adsorption mechanisms of organic pollutants by biochar composites. (a) The NOR adsorption mechanisms of ABLB4[63]. (b) Diagram of the Zn-BC preparation and adsorption mechanism[73]. (c) The mechanism of tetracycline removal by N-BC[87]. (d) The mechanism of atrazine removal by ASBC[88].
Degradation of organic pollutants by BC
-
Persistent organic pollutants—such as antibiotics, per- and poly-fluoroalkyl substances (PFAS), and herbicides—are difficult to degrade, and pose long-term threats to drinking water safety and aquatic ecosystems, thereby presenting significant challenges to environmental sustainability. Biochar has shown effectiveness in both adsorbing and degrading such pollutants. While adsorption is a widely used, low-cost, and efficient method for removing antibiotics, it merely transfers contaminants from the aqueous to the solid phase without breaking them down[89]. In contrast, oxidative degradation—which can mineralize persistent pollutants, antibiotics, and other macromolecules into smaller, less harmful compounds—has attracted considerable academic interest for antibiotic removal. Advanced oxidation processes (AOPs) are regarded as highly competitive water treatment technologies in this regard[90]. In AOP, highly reactive oxygen species (ROS), including individual oxygen species (1O2), hydroxyl radicals (•OH), sulfate radicals (SO4•−), and superoxide radicals (O2•−) are produced by catalyzing or activating hydrogen peroxide, oxidants like ozone, peroxymonosulfate (PMS), or persulfate (PS) through carbon materials, heat, electricity, UV light, and transition metals[91−94]. In recent years, improved oxidation strategies, particularly Fenton-like catalytic oxidation and sulfuric acid-based oxidation, have been hot topics for antibiotic degradation[95].
This section specifically examines the reaction mechanism of biochar catalysts in the degradation of organic pollutants using activated persulfate. Li et al.[87] prepared herbal residual nitrogen-doped biochar (N-BC) for activated peroxymonosulfate (PMS) degradation of tetracycline, and the optimum conditions for the degradation efficiency of tetracycline (TC) in 60 min (> 99%) were achieved at a dosage of 1 g/L of N-BC, and a concentration of 5 mM of PMS, and the effect of nitrogen doping was investigated by using density functional theory (DFT) and characterization. Non-radical singlet oxygen (1O2) was found to be the dominant active oxidizing species by electron paramagnetic resonance (EPR) spectroscopy, and free radical scavenging activity. Pyridinic-N played a crucial role in the 1O2-dominated pathway (Fig. 4c). Atrazine is a broad-spectrum herbicide that is extensively used in dryland cultivation and is photosynthetically inhibited. For atrazine (ATZ), the ultrasonic (US)/Acanthopanax senticosus biochar (ASBC)/persulfate oxidation system was established. The results indicated that the optimal degradation performance was obtained at a US frequency of 45 KHz, and 0.01 g/L ASBC600, which resulted in the removal of approximately 70% of ATZ in 50 min. SO4•− and •OH were the dominant active species of the system[88] (Fig. 4d). Biochar and biochar-based catalysts can activate PMS to generate a variety of ROS with strong oxidizing power, which attack refractory organic compounds and ultimately mineralize them into CO2 and H2O. However, the catalytic activity of biochar may diminish over time due to surface deactivation. Therefore, investigating the long-term stability and reusability of biochar catalysts is crucial. In practical applications, further attention should be given to issues such as competitive adsorption in the presence of heavy metals, the reproducibility of biochar catalysts, and their stability during oxidative-catalytic degradation processes.
-
Traditional Chinese Medicine Herb Residues (TCMHR) are rich in cellulose, hemicellulose, lignin, and other organic components[82,98]. By comparing the yield, ash content, lignocellulose content, and C/O and C/H ratios of TCMHR biochar with those of agricultural waste biochar[99,100], and fruit waste biochar[101], it was found that the differences in their composition are likely attributable primarily to pyrolysis temperature, with the inherent properties of the feedstock exerting only a minor influence. Yuan et al.[31] prepared biochar from five common types of Chinese medicinal residue, with yields ranging from 29.30% to 38.65%, which are comparable to those of straw-based biochar[102], in terms of cellulose content, the medicinal residue biochar was also similar to straw-derived biochar[103]. Among 40 plant-based Chinese medicinal residues collected before and after decoction, the average total lignocellulosic fraction reached 68.2%, a level comparable to that of common agricultural residues such as maize stalks, wheat straw, sugarcane bagasse, rice straw, and switchgrass. These findings suggest that traditional Chinese medicinal solid waste holds promise as a viable feedstock for biomass energy applications[104]. This composition favors the formation of a well-developed pore structure and high specific surface area during high-temperature pyrolysis. TCMHR-derived biochar typically exhibits a high specific surface area, a developed pore network (rich in micropores and mesopores), and high porosity, all of which contribute to excellent adsorption performance for various pollutants. Additionally, the surface of TCMHR biochar is abundant in functional groups that can form stable complexes with heavy metal ions or undergo chemical reactions with organic pollutants, further improving removal efficiency[82,97]. Moreover, TCMHR biochar can serve as a catalyst support; by loading metal oxides or other catalytic materials, it can catalytically degrade organic pollutants[87]. Furthermore, certain TCMHR may contain natural plant extracts such as alkaloids and polyphenols, which can be converted into specific surface functional groups (e.g., hydroxyl, carboxyl) during carbonization, thereby enhancing interactions with pollutants. Literature reports indicate that organic acids and flavonoids present in Chinese herbs also contribute to chemisorption potential[48].
Excellent environmental restoration potential
-
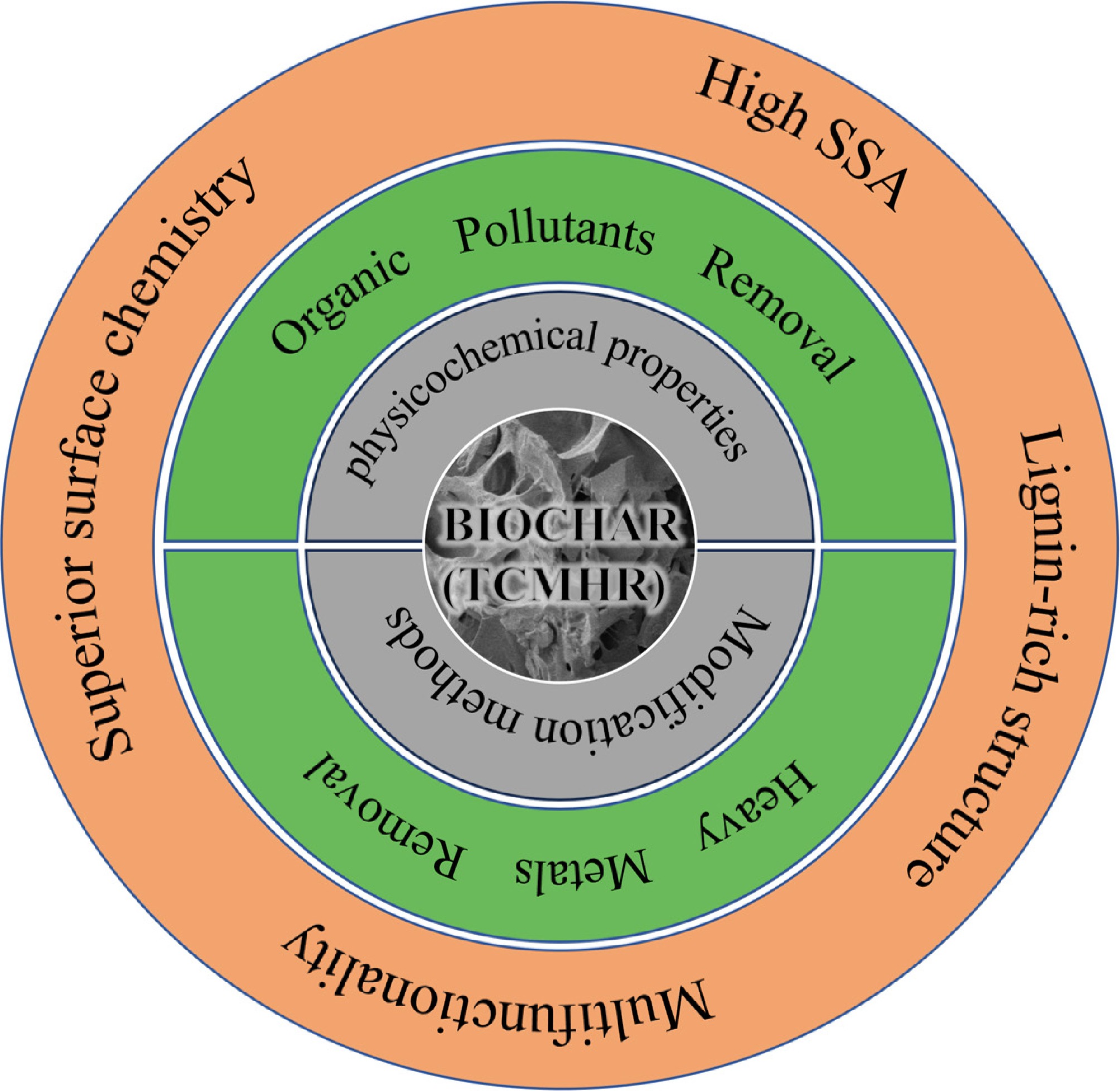
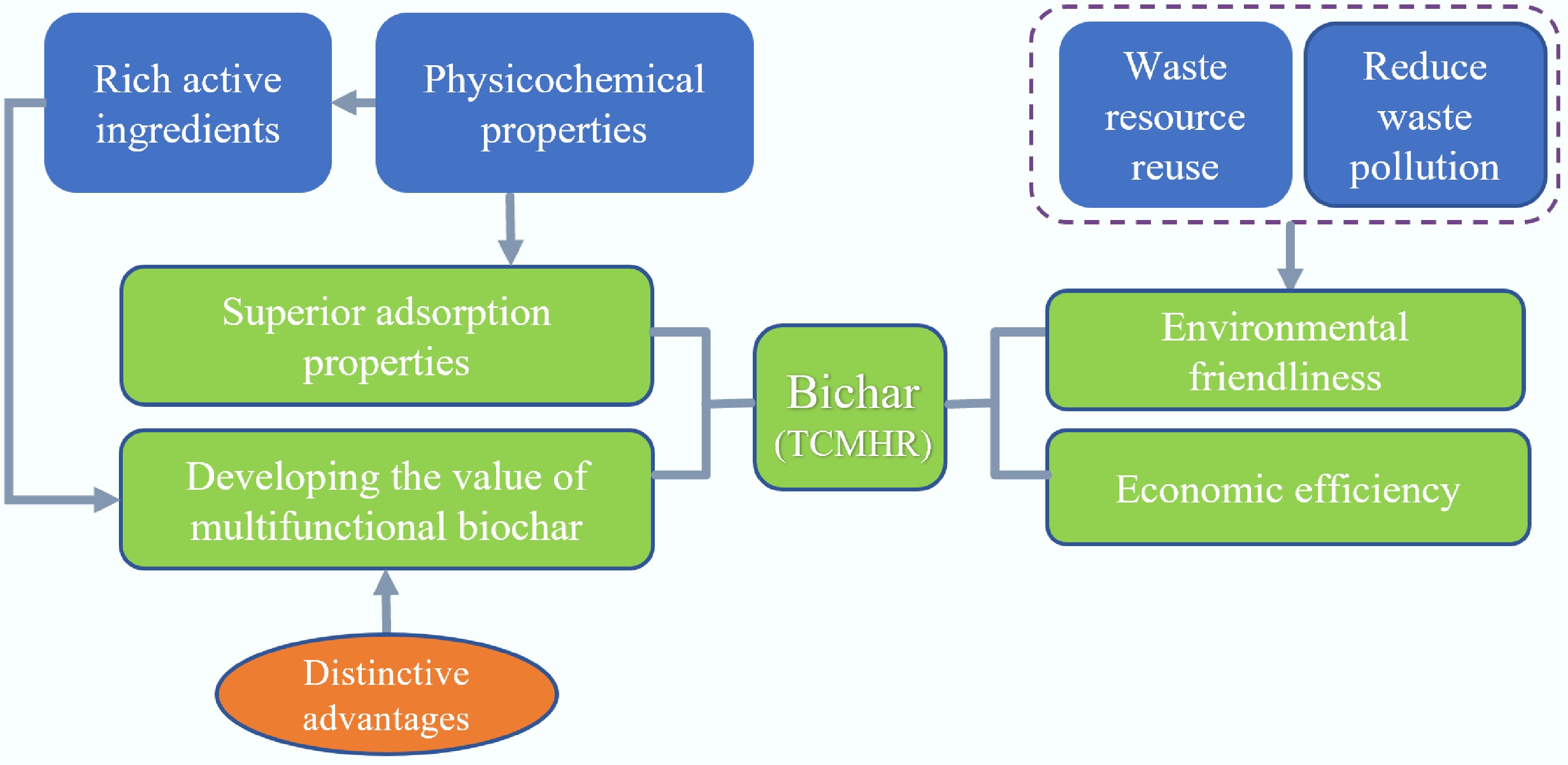
From a low-carbon economy perspective, converting TCMHR into biochar enables resource recovery from waste and reduces environmental impacts. For example, Deng et al.[105] reported that low-temperature pyrolysis of herbal residues to produce biochar can serve as a cost-effective, carbon-negative soil conditioner. As a soil amendment, biochar not only aids carbon sequestration and climate change mitigation[106] but also enhances overall soil health and promotes plant growth[107]. It can also effectively adsorb heavy metals and organic pollutants, and remediate polluted soil and water bodies[108]. As a widely available waste material, TCMHR has very low raw-material costs, requiring only transportation and processing expenses, which makes it suitable for large-scale production. Through rational design and modification, high-performance adsorbent materials with considerable economic value can be prepared. TCMHR-derived biochar offers multifunctional adsorption performance, catalytic degradation capability, environmental friendliness, and cost-effectiveness in pollutant removal. Its unique physicochemical properties and environmental benefits position it as a promising material for addressing pollution challenges and advancing sustainable development. Figure 5 summarizes the key benefits and characteristics of TCMHR biochar in pollutant removal. By utilizing TCMHR biochar rationally, we can effectively reduce environmental pollution, promote resource recycling, and generate significant environmental, economic, and social benefits.
Developing the value of multifunctional biochar
-
TCMHR, which are by-products of decoction, have been reported to retain approximately 30%–50% of medicinal active components[33]. Researchers have innovatively utilized these residues as carbon sources to synthesize traditional Chinese medicine-derived carbon dots (TCM-CDs) via hydrothermal methods. This strategy not only preserves the core pharmacologically active constituents during carbonization but also leverages the nanoscale structural effects to achieve synergistic integration between the bioactive components and the carbon-based matrix. As a result, TCM-CDs exhibit multi-target antibacterial activity and a low propensity to induce drug resistance, along with significantly enhanced biological performance[109]. To date, these carbon dots have shown promising research progress and potential applications in several medical fields, including hemostasis[110], gastrointestinal disorders[111], neuropsychiatric conditions[112], cardiovascular diseases[112], and antimicrobial therapy[113]. The residual medicinal components in traditional Chinese medicine residues can be utilized through well-designed preparation techniques to produce functional biochar. Currently, a variety of single herbs, compound formulations, and processing by-products from traditional Chinese medicine have been successfully used as carbon sources to synthesize antibacterial TCM-CDs. For example, carbon dots derived from white aconite, Cnidium monnieri, and garlic have all demonstrated excellent antibacterial efficacy[109,114]. However, during the aging process, stable heavy metals within biochar may undergo morphological changes and release new pollutants. To ensure its long-term ecological safety, stringent control and assessment of raw materials, processes, and products are required, alongside the implementation of preventive measures such as leaching toxicity testing to mitigate potential risks. TCMHR biochar, produced from waste residues of traditional Chinese medicine, aligns with circular economy principles and holds potential for large-scale production. Simultaneously, its raw material composition exhibits significant variability, affecting product stability; inadequate quality control may lead to secondary pollution. Compared to mainstream adsorbents (such as activated carbon), its economic competitiveness requires further quantification through techno-economic analysis.
In summary, TCMHR biochar demonstrates exceptional adsorption properties, exhibiting outstanding environmental remediation potential and multifunctional development value (antibacterial and antioxidant characteristics). As an emerging and cutting-edge research field, it not only promotes the resource recovery of traditional Chinese medicine residues but also provides an innovative pathway for developing novel biochar.
-
Biochar derived from traditional Chinese medicine herb residues (TCMHR) represents a sustainable and multifunctional material for addressing soil and water pollution. This review highlights the unique advantages of TCMHR-based biochars, including their high lignocellulose content, tailored porosity, and abundant surface functional groups (e.g., hydroxyl, carboxyl), which enable exceptional adsorption capacities for heavy metals (e.g., 599.40 mg/g for Pb[II]) and organic pollutants (e.g., 930.3 mg/g for tetracycline). The removal mechanisms span adsorption-reduction-precipitation for high-valent metals and synergistic interactions (π–π bonding, hydrogen bonding, and non-radical oxidation) for organics. Modified composites, such as magnetic Fe-Zn@BC and TiO2-loaded BC, further enhance recyclability and catalytic efficiency.
Therefore, TCMHR biochar demonstrates exceptional adsorption properties, exhibiting outstanding environmental remediation potential and multifunctional development value, representing an efficient and sustainable approach to resource recycling. However, challenges persist in managing competitive adsorption in multi-pollutant systems, mitigating surface deactivation, and assessing environmental risks of metal oxide-modified variants. Future research should prioritize: (1) selective pollutant removal strategies; (2) long-term stability of biochar in dynamic environments; and (3) comprehensive eco-toxicity evaluations. Additionally, the distinct physicochemical properties of compound TCMHR (vs single-herb biomass) demand systematic exploration to optimize adsorption performance. Addressing these gaps will advance TCMHR biochar from lab-scale innovation to scalable environmental remediation, aligning with circular economy principles, and global carbon neutrality goals. This work not only transforms industrial waste into high-value materials but also paves the way for sustainable pollution control in the TCM sector.
-
The authors confirm their contributions to the paper as follows: Yuzhu Tan, Yanling Liu, Tianzhe Chu, Chengjiu Wang: conceptualization, original draft writing, methodology, writing − review and editing; Yu Gu, Can Chen, Wenlong Fu, Jiandan Yuan: writing − review and editing, formal analysis; Hulan Chen, Cheng Peng: supervision, project management, obtaining funding. All authors reviewed the results and approved the final version of the manuscript.
-
All data generated or analyzed during this study are included in this published article.
-
This research work was financially supported by the National Key R&D Program of China (Grant No. 2025YFC3509100), and the Sichuan Science and Technology Program (Grant No. 2025NSFSC2160).
-
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
-
TCMHR biochar: High SSA, lignin-rich structure, superior surface chemistry, multifunctionality.
Multistep mechanisms: Cr(VI)→Cr(III) reduction; π–π/electrostatic for organics.
Future focus: Scalable real-wastewater stability and AOP-integrated TCMHR biochar composites.
-
Full list of author information is available at the end of the article.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Tan Y, Liu Y, Chu T, Wang C, Gu Y, et al. 2026. From waste to resource: TCM herb residue-derived biochar as a multifunctional material for environmental remediation. Biochar X 2: e010 doi: 10.48130/bchax-0026-0006
From waste to resource: TCM herb residue-derived biochar as a multifunctional material for environmental remediation
- Received: 01 December 2025
- Revised: 07 January 2026
- Accepted: 13 January 2026
- Published online: 27 February 2026
Abstract: The massive generation of traditional Chinese medicine herb residues (TCMHR), a direct consequence of industry expansion, presents a dual challenge: significant waste disposal burdens, and untapped resource potential. Concurrently, the remediation of heavy metal and organic contaminants in aqueous and soil matrices requires innovative, sustainable materials. This review systematically synthesizes recent advancements in the strategic conversion of TCMHR into engineered biochar (BC), and BC-based composites for environmental decontamination. Synthesis protocols (e.g., pyrolysis, hydrothermal carbonization, activation) and their direct influence on critical physicochemical properties are evaluated, such as hierarchical pore architecture, specific surface area (often exceeding 500 m2/g), and tailored surface chemistry. The inherent lignocellulosic and bioactive composition of TCMHR facilitates biochar production with exceptional adsorption capacities, documented here for pollutants including Pb(II) (up to 599.40 mg/g), and tetracycline (up to 930.3 mg/g). The dominant remediation mechanisms are detailed: for heavy metals, a combination of surface complexation, ion exchange, and reduction-precipitation; for organic contaminants, synergistic effects involving π–π stacking, electrostatic attraction, and hydrogen bonding. Furthermore, how engineered modifications—such as magnetic Fe3O4 impregnation for separation or the creation of heteroatom-doped carbon matrices for catalytic processes (e.g., peroxymonosulfate activation)—enhance functionality, reusability, and pollutant degradation efficiency are analyzed. Moreover, well-designed preparation techniques can transform active constituents from TCMHR into functional biochar while retaining core pharmacological components during carbonization, offering potential medical applications. Despite promising laboratory results, key obstacles impede scalable application. These include performance in complex, multi-pollutant systems, long-term environmental stability, and potential secondary leakage of modified BCs, and a need for full life-cycle and ecotoxicity assessments. Addressing these research gaps is essential to transform TCMHR-derived biochar from a promising valorization concept into a viable, circular economy solution that contributes to environmental sustainability and carbon neutrality goals.