-
Natural rubber (Hevea brasiliensis L.) is a globally important industrial commodity and a primary livelihood source in many tropical regions[1]. Accelerating demand, especially across Asia, has driven large-scale conversion of land to rubber monocultures[2]. Although natural rubber is native to the Amazon basin, commercial production shifted to Asia in the late 19th century; currently, Asia accounts for ~89% of global natural rubber supply, with Thailand and Indonesia contributing nearly 60% of production[3,4]. Growing climatic pressures, higher mean temperatures, altered rainfall regimes, and increased frequency of extreme events pose significant risks to tree physiology, latex yields, and the long-term viability of plantation systems.
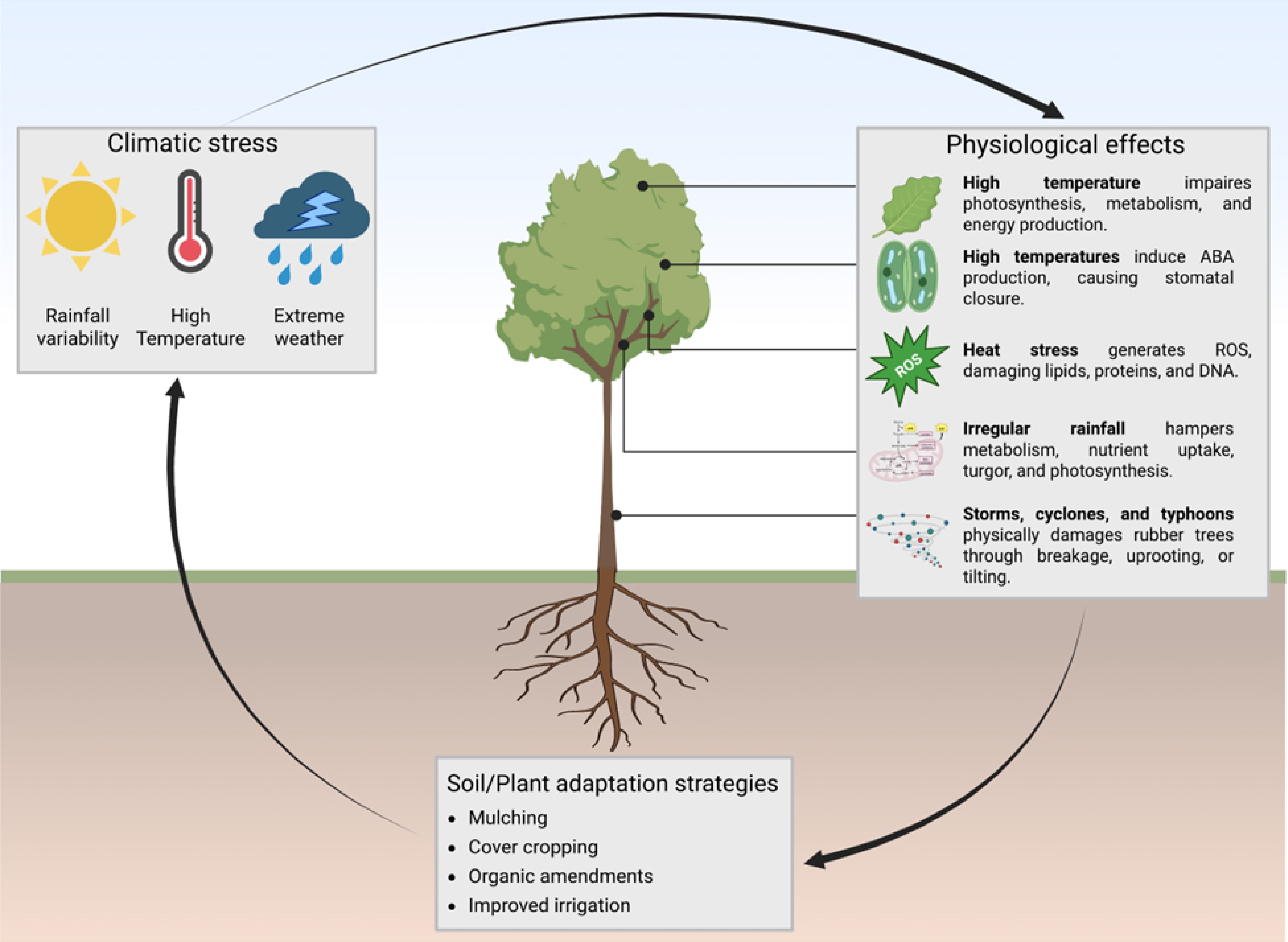
The impacts of climate change on rubber cultivation are multifaceted. Higher temperatures result in increased evapotranspiration, leading to water stress, diminished photosynthesis, disruption of cis-1,4-polyisoprene enzyme activity critical for latex biosynthesis, and a higher respiration rate that depletes carbohydrate reserves necessary for latex synthesis[5]. Moreover, climate change has altered precipitation patterns and reduced rubber productivity. Prolonged drought increases abscisic acid (ABA) hormone concentrations, resulting in stomatal closure and diminished gas exchange, whereas erratic and excessive precipitation leads to land degradation, nutrient depletion, and a lack of essential nutrients for plants[6]. These limitations require adaptive approaches that prioritize soil health and management to improve the resilience of rubber trees to climate change.
Soil attributes, in contrast to climatic or genetic factors, can be rapidly altered by management, as their physical, chemical, and biological functions respond dynamically to field-level interventions. Soil functions as a dynamic system in which processes such as organic matter decomposition, aggregate formation, and microbial regulation play a central role in maintaining ecosystem resilience[7]. Targeted management practices, including conservation tillage, mulching, and organic amendments, promote microbially mediated carbon stabilization and soil aggregation, thereby enhancing water retention, aeration, and nutrient cycling[8]. These mechanisms buffer rubber plantations against climatic stresses by increasing soil water storage, moderating temperature fluctuations, and strengthening root microbe symbioses. In contrast, climatic variability operates beyond management control, and genetic improvements in rubber plantations require long selection cycles with limited adaptability to short-term environmental shifts[9]. Therefore, soil provides a highly manageable biophysical interface through which targeted practices can promptly optimize carbon and nutrient cycling as well as water retention, enhancing the resilience of plantations under fluctuating climate regimes.
Soil management strategies are crucial for enhancing the resilience of rubber plantations against climate variability. Soil characteristics like pH, soil organic matter, microbial activity, such as microbial biomass, enzyme activities (e.g., β-glucosidase, urease, phosphatase), and microbial community functional processes can elucidate nutrient imbalances and provide requisite management interventions[10]. Practices such as mulching, cover cropping, and organic amendments enhance soil organic matter levels, optimize nutrient cycling, and strengthen soil resilience against erosion[11]. Agroforestry and intercropping are viable strategies to mitigate water stress, adjust microclimate, and significantly enhance soil biodiversity. Furthermore, water management techniques, including enhanced irrigation practices and mulching methods customized to water retention capacity, might mitigate drought stress[12]. These measures will only be effective with intensive monitoring and evaluation of soil health. Advanced technologies such as geographic information systems (GIS), remote sensing, and soil sensor instruments provide real-time data, facilitating prompt management decisions to sustain rubber plantation productivity under climate change.
This review investigates the facilitative function of soil-based strategies in the adaptation of rubber plantations to climate change, emphasizing the necessity for suitable and effectively managed soil management practices, water management techniques, and soil health monitoring systems. It also consolidates existing knowledge and highlights gaps that must be addressed to establish guidelines for the sustainable growth of rubber plantations in response to escalating climate concerns. Ultimately, it aims to implement this information into actionable recommendations for researchers, policymakers, and practitioners.
-
Climate change exerts profound impacts on agricultural production systems worldwide, particularly on rubber plantations in tropical regions. Alterations in climate variables such as temperature, precipitation, and extreme weather events have both direct and indirect influences on rubber tree growth and latex production[13]. Rubber trees are highly sensitive to changes in temperature, which can have both immediate and long-term effects on their growth, physiological processes, and latex production (Fig. 1). As temperature rises, enzyme activity speeds up, boosting photosynthesis and latex synthesis, which support rubber tree growth[5]. However, temperatures exceeding a certain point (above 35 °C) cause enzyme denaturation, disturbing metabolic processes and adversely affects tree health, growth, and latex production[4,14]. High temperature decreases Rubisco (ribulose-1,5-bisphosphate carboxylase/oxygenase) efficiency, which is a key enzyme responsible for carbon fixation. Lower Rubisco activity decreased the conversion of carbon dioxide into sugar, thereby reducing the energy and sugar production required for growth and latex biosynthesis[15]. In addition, high temperature also causes degradation in chlorophyll and inhibits enzyme synthesis related to chlorophyll, further diminishing the tree's ability to absorb light for photosynthesis[16].
Additionally, high temperatures stimulate abscisic acid (ABA) production, which facilitates guard cells to close the stomata and reduces water loss[7,17]. As a result, this restricts gas exchange, which lowers stomatal conductance and impairs the plant's ability to take in CO2 and release O2 for photosynthesis. Prioritizing water conservation over photosynthesis activity diminishes overall photosynthetic efficiency[18]. Very high temperatures also disrupt energy production by destabilizing respiration enzymes, hindering essential processes in the respiratory pathway, and reducing ATP (Adenosine Triphosphate) production, which is vital for growth, reproduction, and latex synthesis[19]. Furthermore, heat stress leads to the formation of harmful reactive oxygen species (ROS), like superoxide radicals, hydrogen peroxide, and hydroxyl radicals. ROS causes damage to key cellular components like lipids, proteins, and DNA, while also overwhelming antioxidant enzymes like superoxide dismutase (SOD), causing oxidative stress and reduced enzyme function[20].
Rainfall patterns play a key role in the growth and productivity of rubber plantations by affecting cellular function, photosynthesis, and nutrient uptake. Moreover, sufficient rainfall is necessary to maintain the water balance in rubber plantations, cellular processes, and functions like enzyme activity and protein synthesis[21]. In contrast, irregular rainfall disrupts cellular processes, causing wilting, stunted growth, and disrupted energy metabolism, while also triggering stomata closure to conserve water and limit gas exchange[22]. Rainfall patterns significantly influence plant cell turgor pressure and play a crucial role in chloroplast integrity. Sufficient water supply improves light-capturing efficiency and ensures hydration, whereas drought conditions or improper precipitation patterns damage chloroplasts and photosynthesis, mainly Photosystem II, reducing the plant's ability to capture light energy[23]. Photosynthesis efficiency was influenced indirectly by rainfall and low light due to the light available for chlorophyll absorption, particularly in the red and blue wavelengths, which play a vital role in the existence of electrons in the photosystem and regulate the photosynthesis rate[24]. In Photosystem II (PSII), the absorbed energy splits water molecules to release oxygen and protons, transferring electrons to Photosystem I (PSI), where they reduce NADP (Nicotinamide Adenine Dinucleotide Phosphate) to NADPH (Nicotinamide Adenine Dinucleotide Phosphate Hydrogen), providing the energy necessary for carbon fixation in the Calvin cycle[25]. The critical climatic thresholds affecting rubber tree physiology, soil processes, and latex productivity are summarized in Table 1.
Table 1. Critical climatic thresholds affecting rubber tree physiology, soil processes, and latex production.
Stress factor Critical threshold Physiological/soil response Impact on growth and yield Ref. High temperature > 35 °C Rubisco and latex-biosynthesis enzyme denaturation; chlorophyll degradation; ABA-induced stomatal closure ↓Photosynthesis; ↓carbon assimilation; ↓latex yield [26] > 40 °C Rapid ROS accumulation; destabilized respiration enzymes; impaired ATP production Severe growth reduction; canopy dieback [27] Low temperature < 15 °C Impaired enzymatic activation; reduced phloem flow and latex fluidity Reduced tapping efficiency; slowed growth [20] Annual rainfall deficit < 1,100 mm yr−1 Soil moisture deficit; reduced turgor and PSII activity 10%–15% latex yield decline [20, 28] Dry-season length > 20–25 consecutive
dry daysABA-mediated stomatal closure; microbial suppression Growth stagnation; yield loss [18] Soil moisture threshold < 20% VWC Reduced nutrient diffusion; lower microbial mineralization ↓Nutrient uptake; ↓latex flow [5, 29] Waterlogging > 48–72 h saturated soil Root hypoxia; risk of Phytophthora infection Sharp yield decline; wilting [30] Heat–drought interaction > 33–35 °C with soil moisture deficit Synergistic ROS increase; PSII repair collapse Severe canopy stress; strong yield depression [31] Soil pH constraint pH < 5.5 Al toxicity; P fixation Nutrient deficiency; impaired root growth [32] Low SOC SOC < 1.5% Low microbial activity; poor aggregate stability Low climatic resilience [33] Extreme weather events such as storms, cyclones, and typhoons can cause significant physical damage to rubber trees, affecting both their structural integrity and internal physiology. Firstly, strong wind damages rubber trees by breaking branches, tilting or uprooting the trees, and weakening their over-stability. Such damage makes the tree more susceptible to disease and pests[34]. The uprooted lose their ability to absorb nutrients and water, resulting in stunted growth. Even a slight tilt can impair root function, while damage to the trunk and leaves decreases photosynthesis efficiency, further limiting growth[35]. Secondly, strong wind also damages the plant tissues and rupture the bark, cambium layer, and vascular tissues (xylem and phloem). This damage disrupts the transport of water, essential nutrients, and sugar[36], leading to diminished secondary growth and interrupting photosynthesis. Additionally, the breakage of branches and trunks further damages the vascular system, leading to dehydration, wilting, reduced growth, and potential death of the plant[37]. Thirdly, the mechanical damage in rubber plants triggers mechanosensitive receptors, initiating calcium ion-mediated signaling cascades that activate stress-responsive pathways, including ethylene production. However, the overactivation or misregulation of these pathways can lead to negative effects on the plant[37].
-
Soil management is fundamental to agricultural sustainability, particularly for rubber plantations. Sustainable soil management measures are crucial to bolster resistance against climate-induced stresses. The quality and health of the soil profoundly influence the development, productivity, and sustainability of rubber trees. Farmers can alleviate the detrimental impacts of temperature variations, altered precipitation patterns, and severe weather occurrences by preserving soil integrity. Sustainable soil management encompasses strategies aimed at preserving soil resources, maintaining soil fertility, and promoting ecosystem equilibrium. These practices help to improve overall soil structure and water holding capacity as well as organic matter content, which contribute to the resilience of rubber cultivation through better rooting, greater nutrient uptake, and healthier plants, allowing rubber trees to better cope with adverse climate conditions.
Conservation tillage and soil surface protection
-
Soil conservation is integral to sustaining and enhancing agricultural productivity by addressing soil degradation and improving soil health[38]. In order to improve soil fertility, manage water resources, and stop erosion, effective soil conservation techniques such as contour farming, cover crops, conservation tillage, terracing, and agroforestry are essential[39]. Long-term agricultural sustainability, improved soil structure, and higher crop yields are the results of these approaches. For example, conservation tillage and cover crops increase soil organic matter and moisture retention; contour farming and terracing lessen soil erosion and control the discharge of water[40]. By combining trees and crops, agroforestry offers extra advantages like stabilized soil and enhanced biodiversity. Adoption of these methods is hindered by factors like high initial costs, specialist expertise requirements, and insufficient legislative backing, notwithstanding their benefits[41]. To get the most out of soil conservation, these obstacles must be removed. Policies that assist farmers' financial and technical needs must be put in place in order to ensure that these methods are widely adopted and that agricultural production and environmental health continue. Soil conservation is pivotal for sustaining agricultural productivity, and soil conservation practices are essential for enhancing agricultural productivity and ensuring long-term sustainability[42].
Cover cropping and mulching
-
In rubber plantations, cover cropping and mulching represent key soil surface management practices that enhance soil health, conserve water, and mitigate the adverse effects of climate variability. Cover crops, typically herbaceous legumes or grasses, are cultivated between rubber tree rows to provide continuous vegetative ground cover that protects the soil from direct raindrop impact, reducing splash erosion, surface sealing, and runoff[43]. Common leguminous species include Stylosanthes guianensis, Centrosema pubescens, Calopogonium mucunoides, and Pueraria phaseoloides, while grasses such as Paspalum notatum and Brachiaria decumbens are widely adopted[44]. The extensive root systems of these plants stabilize soil aggregates, enhance water infiltration, and minimize erosion, particularly on sloped or degraded lands[45]. Mulching with plant residues further protects the soil surface, reduces evaporation, and contributes organic matter that improves soil structure, nutrient availability, and microbial activity[46]. These practices also suppress weed growth and support biodiversity by providing habitat for beneficial soil organisms and insects. Collectively, cover cropping and mulching improve soil physical and chemical properties and strengthen plantation resilience at the plot level, providing a foundation for sustainable rubber production[47].
Agroforestry and intercropping systems
-
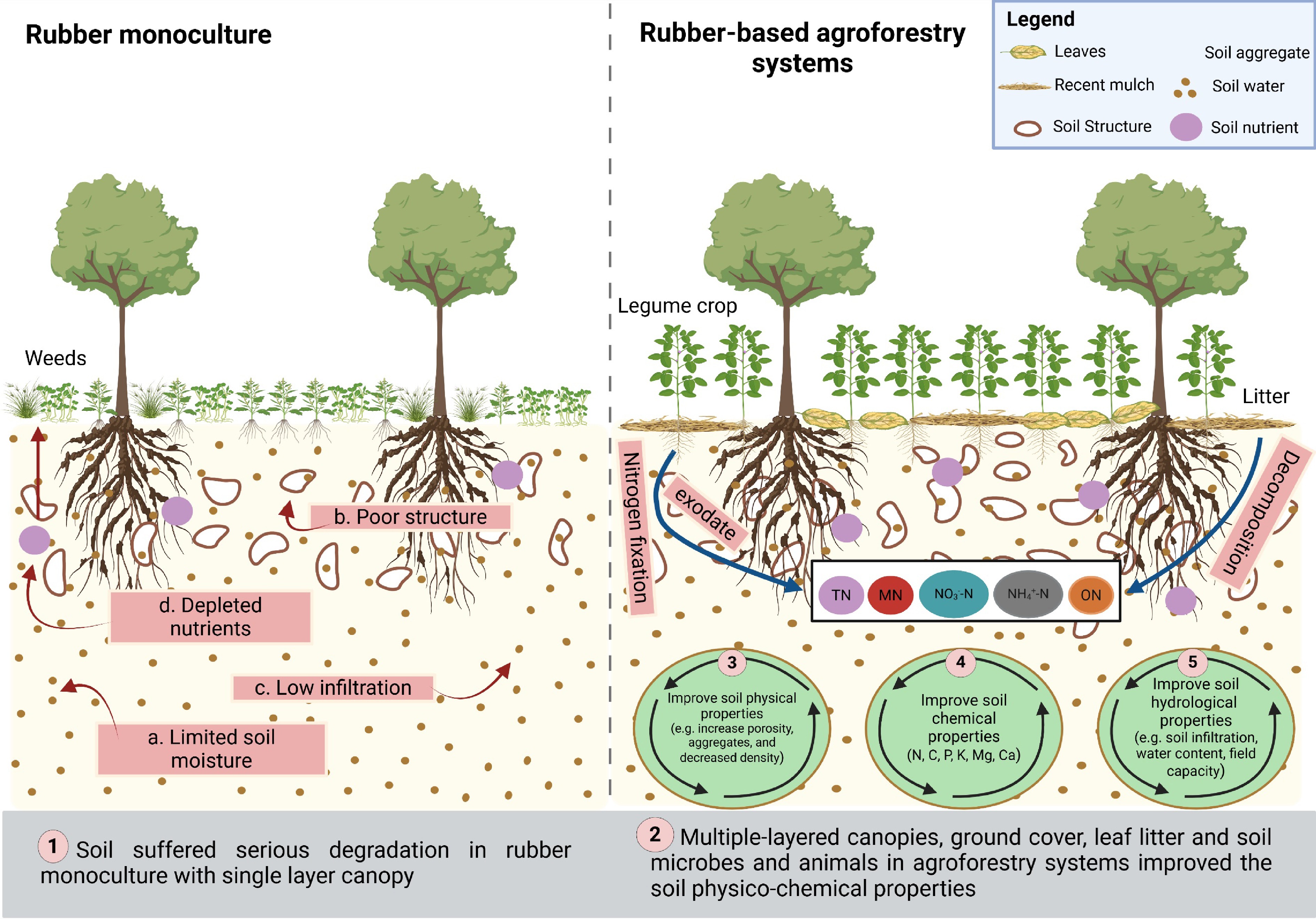
Beyond ground-cover management, agroforestry and intercropping represent system-level diversification strategies that enhance soil fertility, structural stability, and ecological resilience in rubber plantations. Integrating leguminous and other complementary tree species improves soil structure by stimulating root biomass growth and organic matter decomposition, leading to higher soil aggregation and porosity[48] (Fig. 2). The additional canopy cover from diverse tree species protects the soil from direct rainfall, reducing erosion, while leaf litter inputs contribute to soil organic matter and nutrient cycling[49]. Symbiotic relationships, particularly nitrogen fixation by leguminous trees, maintain soil nitrogen availability and reduce the reliance on synthetic fertilizers[50]. Intercropping between rubber trees also supports soil microbial diversity, enhances water infiltration, stabilizes soil aggregates, and promotes beneficial insects for integrated pest management[51]. Together, agroforestry and intercropping provide long-term soil and ecosystem services, reinforcing plantation sustainability and resilience under variable climatic conditions[52].
Organic amendments, composting, and biochar application
-
Organic amendments and composting are crucial components of soil management strategies aimed at enhancing climate resilience in rubber plantations. Plant residues, animal manures, compost, and biochar collectively improve soil physicochemical and biological properties by increasing soil organic matter, enhancing nutrient availability, and strengthening soil structural integrity. These amendments significantly improve water retention, promote nutrient cycling, and support favourable conditions for rubber tree growth[51]. Recent studies show that composted materials effectively enhance soil moisture storage and structural stability, thereby reducing erosion risks and improving drought tolerance in rubber plantations[53]. Composting also stimulates soil biological activity by supplying decomposed organic substrates that support microbial growth, nutrient mineralization, and aggregate formation, which collectively strengthen soil resilience to climatic fluctuations[54]. In addition, composting contributes to climate mitigation through increased carbon storage and reduced methane emissions relative to unmanaged organic waste[55]. Organic amendments further enhance carbon sequestration potential and sustain nutrient supply while suppressing allelochemicals that interfere with herbicide performance. Importantly, the interaction between biochar properties and soil texture exerts a critical influence on amendment effectiveness. Biochar, as a stable carbon-rich material, modifies soil physicochemical attributes through its high porosity, surface adsorption capacity, and cation exchange properties[56]. However, its benefits vary substantially across soil textures. In coarse-textured soils (sandy or loamy), biochar significantly increases water retention, aggregate stability, and nutrient-holding capacity by filling interparticle voids and reducing percolation losses, resulting in enhanced plant-available water and improved rubber tree performance[57]. Meta-analyses indicate that biochar and cover crop application in soils can increase soil organic carbon (SOC) by 0.5−1.2 g·kg−1·yr−1, whereas organic amendment and biochar can improve soil water holding capacity by 10%−15%[58]. In clay-rich soils, where inherent cation exchange capacity and moisture retention are already high, biochar primarily contributes to improving microbial habitat stability, pH buffering, and gradual carbon stabilization rather than substantial gains in water-use efficiency[56]. These contrasting responses highlight the need for texture-specific biochar management strategies in rubber agroecosystems, including optimized application rates, particle size selection, and feedstock types to maximize soil structural improvement and climate resilience.
-
Rubber cultivation has improved the economic status of farmers, but it has also altered the environment and ecosystem of the habitat. It is important to note that the productivity and sustainability of rubber plantations are closely linked to environmental and climatic factors. Changes in precipitation patterns, soil texture and structure, evapotranspiration rates, groundwater recharge, and organic matter decomposition are essential for rubber tree health and yield. Climate change has increasingly been seen as a threat to this plantation in recent years. In previous studies, it was found that the northwestern and southeastern regions of China are experiencing a climate change phenomenon characterized by increased droughts, floods, and erratic precipitation[59].
Soil health, particularly soil moisture, has decreased as climate change raises air temperature and increases precipitation variability. Under these conditions, plants face drought stress, and agricultural commodities production is reduced. Rubber plantations suffer from inadequate root formation due to drought, resulting in a reduction in latex production. Soil compaction and infiltration decrease caused by prolonged drought can cause water availability issues[60]. Moreover, climate change and land use shifts strongly impact the evapotranspiration process, a key component of ecosystem water cycles[61]. The increasing temperature promotes the demand for plant water in rubber monocultures, where the canopy cover may alleviate some direct solar radiation to the soil[62]. In many regions, this increased water loss surpasses precipitation inputs, which can result in negative water balance scenarios. This puts stress not just on the rubber trees but also on the microbial communities and nutrient cycling that are key to soil health. Despite the high social interest in this topic, only a few studies evaluate the effect of climate change and commercial agricultural activities (e.g., deforestation) on soil moisture and water availability[63].
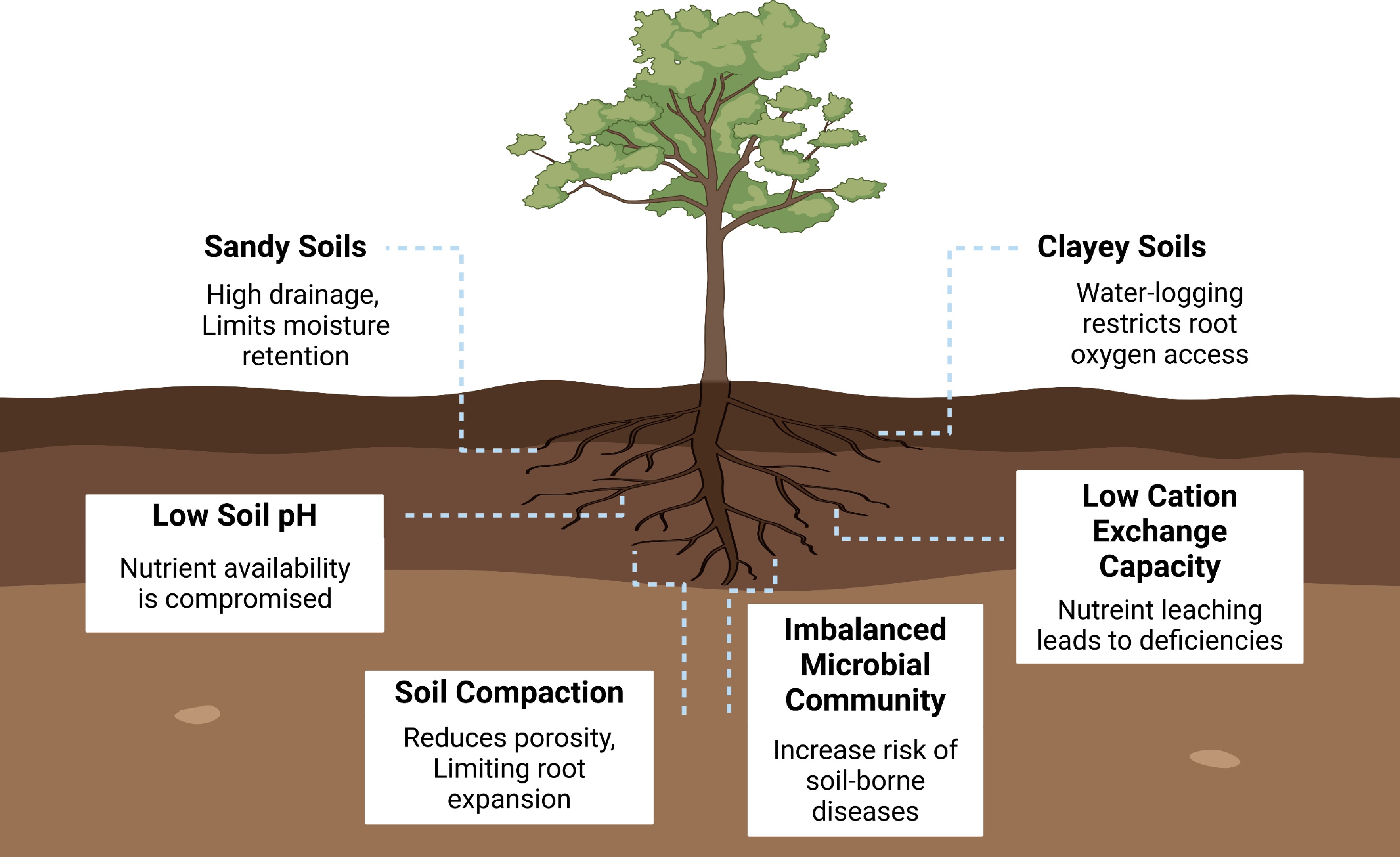
In rubber-producing regions, the physical, chemical, and biological properties of soil play a pivotal role in determining rubber tree performance, directly influencing both vegetative growth and latex productivity (Fig. 3). For instance, sandy soils exhibit high permeability, which promotes rapid water drainage and reduces the moisture available for plant uptake, particularly during dry periods[64]. The sandy soils with their low percentage of fine particles and low cation exchange capacity (CEC) retain fewer nutrients and are prone to leaching and deficiency of key ions, including nitrogen, potassium, and calcium[65]. Likewise, clayey soil has a very important role in dry climatic regions as it helps hold a considerable amount of water, but becomes a problem in regions with humid climates due to the potential waterlogging. The waterlogging conditions prevent rubber trees from getting oxygen, resulting in root damage, stunted growth, and decreased latex production[8]. Clayey soil has a high CEC, so it retains more nutrients efficiently in the soil. The chemical characteristics of soil influence the nutrition and health of rubber trees, with soil pH being a critical component that significantly impacts soil quality and conditions. An optimal pH range for rubber cultivation is slightly acidic to neutral. Soil pH substantially influences nutrient availability and the possible incidence of shortages. The macronutrients, including nitrogen, phosphorus, and potassium, are essential for the growth and latex production of rubber trees. Phosphorus is tightly linked to iron and aluminum in acidic soil, forming insoluble compounds that restrict root development[10]. Soil pH influences the accessibility of micronutrients, including Fe, Mn, Cu, and Zn, with Fe availability rising in acidic soils and diminishing in alkaline soils. Furthermore, aluminum ions (Al3+) exhibit substantial solubility in soil pH levels below 5.5, resulting in toxicity that impairs root cell division and elongation[66].
Soil biological properties, including microorganisms, soil fauna, and roots, play an important role in the health and productivity of rubber plantations (Fig. 3). The bacteria, fungi, and actinomycetes are important microorganisms for the nutrient cycle, decomposition of organic matter, and soil aggregate formation[52]. Soil microorganisms contribute greatly to the decomposition of organic material, along with releasing microelements necessary for rubber tree growth, such as nitrogen, phosphorus, and potassium, in rubber plantations[66]. However, an imbalanced microbial community, dominated by harmful pathogens like Fusarium or Phytophthora, can lead to soil-borne diseases, adversely affecting rubber tree health and latex yield[67]. Soil microorganisms also produce soil enzymes that catalyze essential processes in nutrient cycling. These enzymes facilitate organic matter decomposition, nitrogen mineralization, and the breakdown of complex compounds such as cellulose and lignin. High enzyme activity, especially in nitrogen mineralization (e.g., urease, protease) and organic matter breakdown (e.g., cellulase, amylase), enhances nutrient availability, therefore promoting robust growth and increased latex output[68]. In addition, SOM serves as an energy source for soil microbes. The decomposition, mostly facilitated by fungi and bacteria, results in the creation of humus, which improves soil structure, water retention, and nutrient availability[69]. These enhancements promote rubber plantation development in tropical areas, where improved soil structure promotes root penetration, aeration, and moisture retention.
Strategies for improving soil water retention and drainage in rubber plantation
-
Rubber trees exhibit distinct root-level physiological adjustments under fluctuating soil moisture, particularly during alternate wetting and drying (AWD) cycles commonly experienced in monsoonal regions. AWD conditions trigger fine-root proliferation during drying phases to enhance soil moisture foraging, while periodic rewetting stimulates deeper root extension to access subsoil water[70,71]. Temporary saturation induces localized hypoxia, prompting aerenchyma formation that improves oxygen diffusion and root survival. At the same time, AWD modulates aquaporin activity and root hydraulic conductivity, which are downregulated during drying to conserve water and rapidly restored upon rewetting to maintain transpiration and latex flow. These coordinated responses enable the rubber tree to stabilize water uptake and maintain metabolic function under variable moisture regimes, underscoring the importance of implementing water-management strategies that support such physiological resilience[72].
In rubber-growing regions, prolonged periods of water deficit represent a major challenge for plantation sustainability, as they reduce soil water availability, disrupt water distribution, and affect the overall water balance[73]. In sandy soils, which predominate in some rubber-growing areas of northern China, water retention is particularly low, leading to increased reliance on groundwater and surface water reserves to sustain latex production during dry seasons[74]. Between 1979 and 2015, drought affected approximately 17% of rubber plantation acreage in China, reducing latex yields by 7.8%–11.6%[75]. Therefore, improving soil water retention and optimizing drainage are crucial for sustaining rubber productivity under variable climatic conditions.
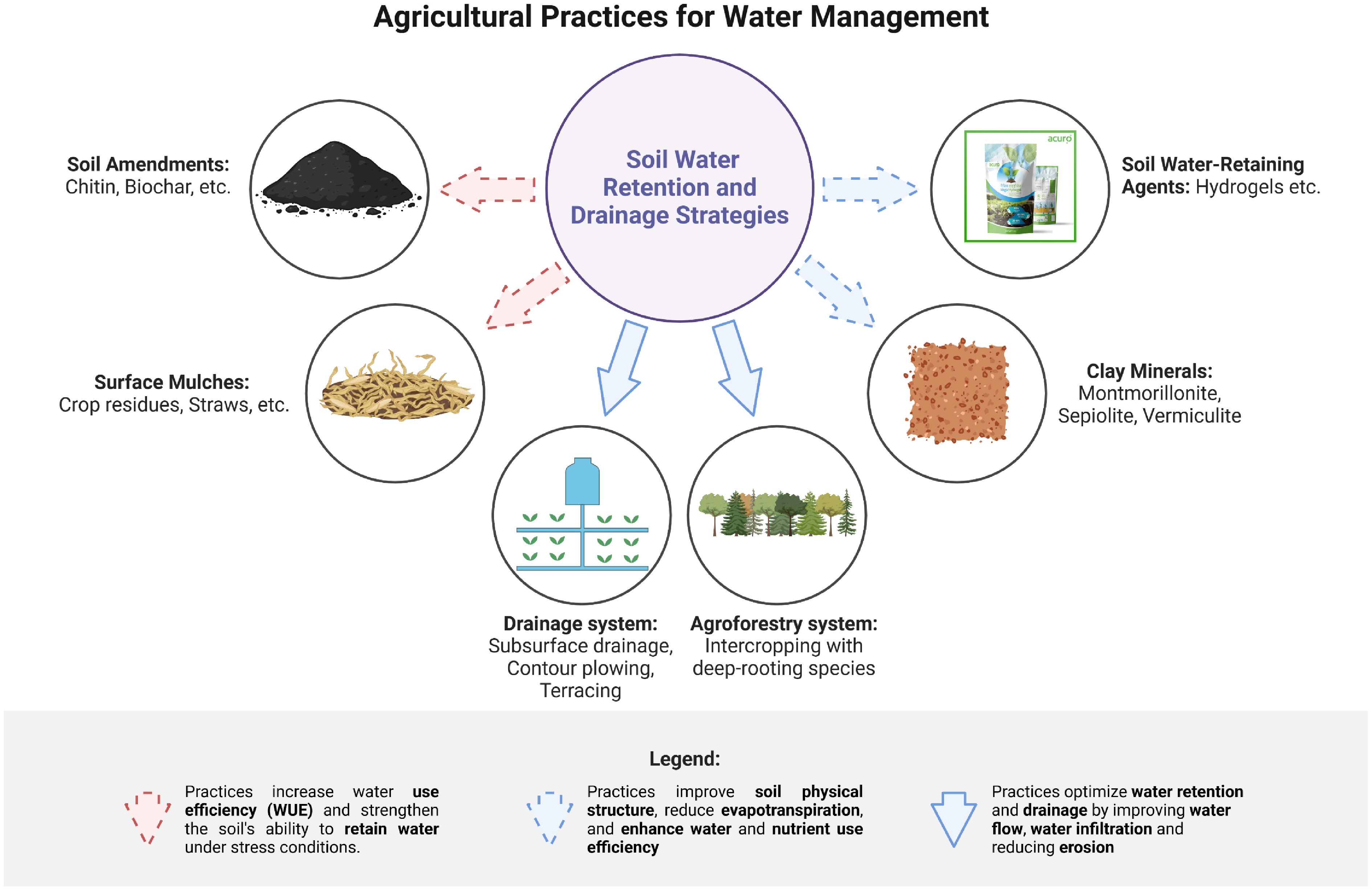
Several strategies have been experimentally applied in rubber plantations to enhance soil moisture availability during dry periods (Fig. 4). Soil water-retaining agents (WRAs), such as hydrogels, have been tested in rubber soils to improve water-holding capacity (WRC). These hydrophilic polymers contain functional groups (e.g., carboxyl, hydroxyl) that increase saturated water content, field capacity, and plant-available water, while reducing deep infiltration losses and evaporation[76]. Field trials in rubber plantations have demonstrated that hydrogel applications can maintain soil moisture for extended periods, alleviating drought stress and stabilizing latex yield. Firstly, soil amendments such as biochar have also been evaluated in rubber plantations. Biochar improves soil physical structure by decreasing bulk density and increasing porosity, leading to enhanced water-holding capacity and water-use efficiency (WUE). For example, studies in tropical rubber soils reported that biochar incorporation increased soil field capacity by 10%–15% and improved root penetration, directly supporting tree growth and latex production[77]. It is important to acknowledge that the effectiveness of soil amendments such as biochar and hydrogels is not universal but strongly mediated by soil type, climatic conditions, and amendment characteristics. For instance, hydrogels show substantial improvements in sandy soils but exhibit limited performance in clay-rich soils where natural water-holding capacity is already high[78]. Similarly, biochar's effects vary with feedstock origin, pyrolysis temperature, and particle size, leading to inconsistent outcomes across tropical rubber-growing regions[79]. Moreover, many published results stem from controlled pot experiments with uniform environmental conditions, which may not fully capture field-scale heterogeneity, seasonal moisture fluctuations, or long-term amendment stability. These methodological limitations underscore the need for site-specific application strategies and more extensive field-based, long-duration trials to validate their efficacy under realistic plantation conditions.
Secondly, drainage management is equally critical in clay-rich rubber soils prone to waterlogging. Field studies have shown that subsurface drainage systems, combined with deep tillage, effectively remove excess water from the root zone, improving aeration and reducing the incidence of root diseases that can limit latex production[80]. Terracing and contour ploughing have been implemented in hilly rubber plantations to minimize surface runoff, prevent erosion, and maintain a balanced water regime[47].
Thirdly, clay minerals such as montmorillonite, sepiolite, and vermiculite naturally enhance soil water retention by increasing absorptive capacity, ion exchange, and swelling potential, thereby improving water and nutrient use efficiency[81]. In addition, surface mulching with crop residues or organic material has been shown to reduce soil evaporation by approximately 6.2% and enhance WUE by 5.0% in rubber plantations, contributing to sustained moisture availability during dry periods[82]. Finally, agroforestry approaches in rubber plantations have been documented as effective strategies for balancing water retention and drainage. Intercropping rubber with deep-rooted trees or shrubs enhances soil structure, improves infiltration, reduces erosion, and promotes biodiversity in both above- and below-ground ecosystems[83]. Deep-rooted species stabilize soil water movement, ensuring that moisture is retained in the root zone while preventing waterlogging, thus supporting both tree growth and latex productivity.
Improved irrigation techniques and water conservation practices
-
Soil water management with proper irrigation methods and applicable water conservation practices plays a vital role in the sustainability and productivity of rubber plantations. These include efficient irrigation systems: drip, micro-sprinkler, and subsurface drip, along with best practices like growing mulch and harvesting rainwater that provide the significant benefits of water use efficiency improvements that lead to increased crop yields[84]. Integrated, multi-strategy approaches can optimize gains and ensure the long-term viability of rubber plantations even in the wake of expanding ecological and financial challenges. It is essential to conduct further research, innovate with technology, and have supportive policies in place the current barriers to the widespread uptake of these next-generation water management techniques are to be overcome.
Among irrigation methods, drip and subsurface drip systems have been found to be very effective for water application in rubber plantations as they apply the water directly to the root zone[85]. In comparison to conventional flood irrigation, drip irrigation reduces water consumption by 30%. Another study found that water use efficiency can be increased by 15% through sub-surface irrigation systems[86]. The use of these methods minimizes water losses due to evaporation and runoff, making it a suitable choice for areas with water scarcity, which enhances rubber tree growth and latex production[85]. It should be noted that the reported 30% water-saving efficiency of drip irrigation systems and the 15% improvement in water-use efficiency under sub-surface irrigation are derived from controlled pot-scale experiments; therefore, field-level trials across heterogeneous rubber-growing environments are still required to validate these magnitudes of water savings and agronomic benefits under real plantation conditions. The micro-sprinkler systems are another advanced step toward more uniform watering and improving canopy health. This system can increase soil moisture by up to 20%−25%, improving root development and drought resistance[86]. These irrigation systems can also make fertilizing very easy, which will benefit the tree by improving nutrient uptake.
In addition, rainwater harvesting also serves as a great support to these irrigation technologies, helping to retain soil moisture. They reduce the extent of requirement on external sources for water supply. The rainwater harvesting can reduce external water inputs by 25% providing a backup water supply during drought periods. Adding automated soil moisture monitoring to these practices is a significant step towards precision agriculture[87]. The wireless, solar-powered soil moisture sensors bring rubber plantations closer to precision agriculture when integrated with automated irrigation systems. Those sensors relay information to the irrigation system, so the water is delivered only when needed as a result. It has been found that such systems can increase water use efficiency by 35%, reduce labor costs, and contribute to maintaining soil moisture at an approximate level required for optimal growth of rubber trees[11]. Combining various irrigation techniques and conservation practices in integrated water management strategies can provide combined benefits. The use of drip irrigation and rainwater together results in a powerful system to save water and make the soil healthy. A case study on Brazilian rubber plantations has shown how an agro-systemic approach can boost overall water efficiency by 40%, improving productivity and resilience[3].
-
Healthy soil plays a crucial role in the production of sustainable agriculture, especially in rubber plantations, where long-term viability is an issue. Sustainability of the rubber plantations is positively correlated with soil health, which includes physical, chemical, and biological properties that support plant growth and ecosystem functions[88]. Continuous monitoring of soil health in rubber plantations helps to determine nutrient deficits and degradation of the soil, thereby guiding towards better management practices. Studies on soil health monitoring in rubber plantations indicate the importance of continuous soil assessments to maximize rubber yields, enhance ecosystem services, and ensure sustainability.
Soil health in rubber plantations may be analyzed based on several indicators such as soil organic matter (SOM), pH, available nutrients and texture, microbial biomass, and enzyme activities[10]. SOM is necessary for nutrient holding and soil structure, while pH affects nutrient solubility as well as microbial activity[89]. Macro-nutrients (N, P, K) and micronutrients are generally major plant nutrients for rubber tree growth, but current nutrient assessment focuses on macronutrients to a greater extent[90]. Moreover, soil biological indicators, which include microbes, represent the most valuable component of the soil system for understanding functions such as diversity and enzyme activities that can define a comparable interaction to biological fertility in general[91]. In addition to optimizing fertilization strategies, which reduce nutrient leaching and environmental pollution, regular assessments would improve rubber yield and quality through a balanced supply of a wide range of nutrients[92].
Assessment of soil physico-chemical and biological properties on a routine basis can also encourage the activities of soil organisms, which play a critical role in organic matter decomposition and nutrient cycling. These soil organisms can also contribute to eliminating/suppressing diseases. Soil biodiversity has been linked with both ecosystem resilience and crop productivity[93]. Regular soil health monitoring showed that sustainable land management practices, including crop rotation, agroforestry, and intercropping, play a significant role in latex production[94]. It is important to note that these practices not only improve soil health and fertility but are also economically beneficial.
Furthermore, periodic assessments can identify soil compaction, erosion, and fertility decline well in advance, allowing for necessary management interventions such as cover crops, mulch, or organic amendments. Soil degradation can be minimized with improved sustainable agricultural production for the long term by adopting these interventions[95]. Soil health monitoring technologies have changed the way soil is evaluated. Geographic Information Systems (GIS), remote sensing, and soil sensor technologies have developed active monitoring of soil properties with high precision and effectiveness[96]. Moreover, results from molecular techniques (metagenomics) provide insights into the functioning of soil microbial communities[97]. However, regular soil health assessment across the plantation landscape is hampered by challenges such as cost, technical expert requirements, and variation in soil properties. It is reported that soil health monitoring using indigenous knowledge and technological innovation is more cost-effective and reliable. The use of modern technologies with traditional knowledge is integral for the continuity of rubber pluckers and for sustainable latex production.
Techniques for monitoring soil properties and health in rubber plantation
-
Recent advancements in soil monitoring technologies have substantially enhanced the ability to evaluate soil health in rubber plantations. Three major methodological groups: spectral approaches (remote and proximal sensing), genomic techniques (DNA-based microbial profiling), and soil-sensor systems (IoT-enabled in-situ probes), provide complementary insights into soil physical, chemical, and biological conditions[98,99]. Their comparative operational characteristics, including resolution, cost, and robustness, are summarized in Table 2 to facilitate practical application by researchers and plantation managers. Spectral tools such as multispectral and hyperspectral imaging, especially when integrated with geographic information systems (GIS), offer high-resolution spatial mapping of soil moisture, organic carbon, and nutrient variability, allowing targeted interventions such as precision irrigation and fertilizer optimization[100]. Soil-sensor networks further strengthen monitoring capacity by delivering real-time measurements of moisture, temperature, pH, and electrical conductivity, enabling timely management decisions and preventing both drought stress and waterlogging[101]. In parallel, non-destructive spectroscopic methods combined with chemometric modelling provide rapid and accurate assessments of soil organic matter and nutrient status, supporting precision nutrient management while reducing unnecessary chemical inputs[102]. Collectively, these technologies provide actionable data that improve soil stewardship, mitigate degradation risks, and sustain latex productivity under variable climatic conditions. Recent modelling approaches integrating Sentinel-2 multispectral indices have demonstrated high predictive ability for soil organic carbon (SOC) across plantation landscapes. For instance, a random-forest regression model using Sentinel-2 bands (B3, B4, B8, B11) achieved strong performance (R2 = 0.78; RMSE = 2.1 g kg−1). Such models enable rapid SOC estimation over large areas at 10–20 m resolution, offering a practical, low-cost alternative to intensive soil sampling[103]. This approach is particularly valuable in rubber plantations where spatial heterogeneity in soil fertility and organic matter is pronounced.
Table 2. Comparative overview of spectral, genomic, and sensor-based methods for soil monitoring in rubber plantations.
Monitoring method Resolution Cost level Robustness Key outputs/applications Spectral methods (e.g., Sentinel-2, UAV hyperspectral, proximal VIS–NIR sensors) Medium to high (10–30 m for Sentinel-2; cm-scale for UAV) Low–Medium High for spectral stability; sensitive to cloud cover SOC estimation, moisture mapping, nutrient status, spatial variability Genomic/Metagenomic techniques (DNA sequencing, qPCR) Very high (microbial taxa & functional genes) High Medium (requires lab conditions; sensitive to contamination) Microbial diversity, nutrient cycling potential, disease-related microbial shifts In-situ soil sensors (moisture, EC, pH, temperature; IoT networks) Point-scale, continuous temporal data Medium Very high (field-ready; long-term monitoring) Real-time moisture tracking, fertigation scheduling, early stress detection High-throughput sequencing and metagenomic analyses have revolutionized the understanding of soil microbial communities[104]. In rubber plantations, these techniques can identify beneficial microbes responsible for nutrient cycling, disease suppression, and organic matter decomposition. Such knowledge informs management strategies, including biofertilizer application, organic residue management, and microbiome-enhancing practices, which directly contribute to soil health and sustainability. Soil enzyme activity measurements, soil respiration assays, and composite indices like the Soil Health Index (SHI) provide quantitative evaluations of soil functionality[105]. In rubber plantations, these assessments allow managers to monitor the effectiveness of interventions such as mulching, biochar amendments, and irrigation scheduling, ensuring that soil biological, chemical, and physical properties are maintained or enhanced. Engaging local stakeholders in soil monitoring allows the practical implementation of site-specific management practices[106]. For example, smallholder rubber farmers can combine sensor data, visual field assessments, and SHI metrics to make adaptive decisions regarding irrigation, fertilization, and soil amendment application.
To enhance adoption among smallholder farmers, low-cost citizen-science soil monitoring kits are increasingly promising. Smartphone-based colorimetric applications (e.g., for SOC or nitrate estimation), simple pH and EC field test strips, and portable handheld readers enable rapid on-site soil assessment without laboratory infrastructure. These tools empower growers to monitor soil nutrient imbalances, moisture deficits, and pH constraints at minimal cost. Integrating such participatory monitoring with broader remote-sensing platforms can substantially strengthen community-level soil stewardship and support more timely, data-driven decision-making in rubber plantations.
-
This review highlights the central role of soil processes in mediating how rubber plantations respond to climate change. Rather than functioning as isolated components, soil properties, climatic variability, and rubber tree physiology operate as an integrated system, jointly determining resilience and productivity. Climatic stressors, such as rainfall deficits, heatwaves, and extended dry seasons, alter soil moisture regimes, nutrient diffusion, microbial activity, and aggregate stability. These soil changes directly influence physiological responses, including stomatal regulation, root system plasticity, photosynthetic efficiency, and carbon allocation to latex biosynthesis. Conversely, physiological adjustments feed back to soil processes by altering root exudation patterns, rhizosphere microbial activity, and nutrient turnover. Thus, soil serves as the primary interface through which climate signals are transmitted to tree function. Moreover, emerging digital technologies, including GIS-based mapping, remote sensing, and automated soil monitoring networks, enable continuous spatial and temporal assessment of soil and environmental variables in rubber plantations. These systems provide real-time data on soil moisture, nutrient dynamics, and degradation risks, allowing managers to implement precision irrigation, fertilizer optimization, and erosion control measures. Integrating such technologies with conventional conservation practices fosters data-driven decision-making that reduces resource loss, improves soil health, and enhances the long-term sustainability and climate resilience of rubber plantations. To advance evidence-based soil-focused climate resilience strategies, future research should evaluate the following testable hypotheses:
H1: Biochar at 2 t·ha−1 combined with straw mulch increases water-use efficiency (WUE) by ≥ 20% in rubber plantations located in sub-1,200 mm annual rainfall regimes, due to improved soil moisture retention and reduced evaporative loss.
H2: Sentinel-2–derived soil organic carbon (SOC) maps achieve an accuracy of R2 ≥ 0.75 at 10-m spatial resolution in rubber-growing zones when calibrated with spectrally optimized soil–vegetation indices and field SOC datasets.
H3: Intercropping rubber with deep-rooted legumes reduces surface runoff by ≥ 30% on slopes exceeding 15°, through enhanced soil aggregation, greater infiltration capacity, and increased belowground biomass.
H4: Automated drip irrigation guided by soil-moisture sensors increases latex yield by ≥ 8% compared with farmer conventional irrigation practices, by maintaining optimal root-zone moisture and reducing subsoil drying cycles.
Future studies should integrate long-term field experiments, remote sensing technologies, and mechanistic tree–soil–climate models to quantify these interactions across diverse agroecological settings. In addition, policies and capacity-building programs should support the adoption of soil-restorative practices and sensor-guided water management, ensuring that both smallholder and industrial plantations can adapt to a rapidly changing climate.
This work was supported by Open Fund Project of Key Laboratory of Forest Plant Ecology, Ministry of Education (Grant No. KLP2024B2), Provincial Education Department Research Project-Innovation Team (Grant No. 2024AH010010), and Start-Up Foundation for Advanced Talents of Anhui Agricultural University (Grant No. rc372210).
-
The authors confirm contribution to the paper as follows: data collection: Shah A; visualization: Ali W, Shehzad K; draft manuscript preparation: Ali W; writing – review & editing: Ali W, Shehzad K, Shah A, Li Z, Zhao C, Xia Z; supervision, project administration: Xia Z. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets generated during and/or analyzed in the current study are available from the corresponding author on reasonable request.
-
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Ali W, Shehzad K, Shah A, Li Z, Zhao C, et al. 2026. Soil-based adaptation strategies of rubber plantations to climate change: a global synthesis. Forestry Research Advances 1: e003 doi: 10.48130/fra-0025-0003
Soil-based adaptation strategies of rubber plantations to climate change: a global synthesis
- Received: 16 September 2025
- Revised: 27 November 2025
- Accepted: 22 December 2025
- Published online: 02 February 2026
Abstract: Globally, rubber (Hevea brasiliensis L.) plantations play a critical role in promoting a sustainable economy and poverty alleviation. However, their productivity is increasingly threatened by rising temperatures, altered precipitation regimes, and the growing frequency of extreme climate events. These climatic pressures adversely affect tree physiology, latex yield, and soil processes, thereby challenging long-term plantation sustainability. This review synthesized soil-based strategies that can improve the adaptive capacity of rubber plants under climate change. Across tropical systems, soil-enhancing practices such as cover cropping have been shown to increase soil organic carbon by 0.5–1.2 g·kg−1·yr−1, while organic amendments and biochar can improve soil water-holding capacity by 10%–15%, contributing directly to drought resilience. Soil management interventions provide substantial physical benefits by improving soil aggregation, enhancing water retention, and increasing drainage efficiency, thereby stabilizing moisture dynamics under variable climates. They also generate important chemical and biological benefits by enhancing nutrient cycling, stimulating microbial activity, and promoting carbon stabilization, which collectively improve soil fertility and ecosystem functioning. Emerging monitoring technologies, including remote sensing, geographic information system (GIS) based soil assessment, and in-situ sensor networks, offer new opportunities to track soil moisture, nutrient availability, and degradation risks with greater spatial and temporal precision. Together, these insights highlight that implementing targeted soil management and advanced monitoring strategies can enhance the resilience of rubber trees and help sustain latex productivity under climate change.
-
Key words:
- Rubber plant /
- Climate change /
- Soil health /
- Agroforestry /
- Drought tolerance /
- Management practices