-
Taxonomically, the genus Paeonia belongs to the family Paeoniaceae, a monogeneric family with approximately 33 accepted species according to the latest molecular phylogenetics research and the World Flora Online (WFO) database. Unlike the traditional three-section classification system, the updated classification based on nuclear and chloroplast genome data divides Paeonia into five monophyletic sections: Section moutan (tree peonies, endemic to East Asia), section paeonia (herbaceous peonies, distributed across Eurasia), section onaepia (North American peonies, two species), section delavayana (three species endemic to the Himalayan Hengduan Mountains), and section sterniana (monotypic, restricted to southwestern China)[1]. This revision reflects the evolutionary relationships among Paeonia species more accurately, providing a robust taxonomic framework for subsequent resource development and pharmacological research.
Since the Shennong Bencao Jing (Shennong's Classic Materia Medica), numerous medical classics in successive dynasties have detailed the medicinal effects of Paeonia plants, which has inspired the research and development of modern drugs[2]. In the original system of Chinese medicinal materials, the main medicinal part of Paeonia plants is the root, commonly known as "moutan cortex" (the root bark of tree peonies), "white peony root" (the processed root of herbaceous peonies), and "red peony root" (the unprocessed root of herbaceous peonies). In 2002, documents issued by the National Health Commission of the People's Republic of China stated that Paeonia roots could be used as a raw material added to health food products[3]. In 2015, Paeonia roots were included in the Pharmacopoeia of the People's Republic of China (PPRC)[4].
Beyond their medicinal relevance, Paeonia plants in China have long exhibited a dual identity as both a prominent ornamental species and an important medicinal resource[5]. Prolonged domestication and cultivation have established tree peonies and herbaceous peonies as iconic elements of traditional garden landscapes, flower appreciation culture, and folk customs[6]. Notably, ornamental and medicinal traditions are historically interconnected rather than independent. Classical medical texts and accumulated clinical experience indicate that traditional processing techniques applied to Paeonia roots—including peeling, boiling, drying, and various stir-frying or wine/vinegar processing methods—were closely associated with different medicinal properties and therapeutic orientations, particularly in blood-related applications[2]. Consistent with these traditional concepts, modern phytochemical studies haev demonstrated that multiple constituents of white peony root undergo significant quantitative changes during different processing procedures, providing a measurable material basis for the traditional notion that processing modulates both the medicinal properties and efficacy[7−9].
Moutan cortex contains a variety of chemical components, mainly acetophenones, terpenoids and their glycosides; organic acids; and other compounds. Among these, acetophenones, terpenoids, and their glycosides have relatively high contents and are the main bioactive components of moutan cortex[10].
Here, we systematically summarize the recent research advances in plant resources' distribution, the exploration of bioactive components, analyses of pharmacological effects, and development and utilization of the genus Paeonia. Furthermore, the core scientific issues of existing studies and future innovative directions are discussed, aiming to provide theoretical references and technical support for the in-depth and sustainable development of this group of medicinal plants.
-
The genus Paeonia comprises several important species, among which Paeonia lactiflora (herbaceous peony) and Paeonia suffruticosa (tree peony) are the most representative members. Paeonia lactiflora is widely distributed in China, ranging from Heilongjiang in the north to Yunnan in the south, Shandong in the east, and Gansu in the west. Paeonia suffruticosa is native to the mountainous areas of the Qinling and Daba Mountains in western China, and its large-scale cultivation has now been established in Luoyang (Henan Province), Heze (Shandong Province), and other regions. Plants of this genus exhibit strong adaptability, distributed from plains and hills at an altitude of several hundred meters to mountainous areas at an altitude of approximately 2,000 m, and they usually grow in hillside grasslands, forests, and thickets[11].
Characteristics of Paeonia
-
The herbaceous and woody types of Paeonia plants have unique morphological characteristics. Herbaceous plants have soft stems, whereas woody plants such as Paeonia suffruticosa have highly lignified stems. Their leaves are morphologically diverse, mostly bipinnate ternate compound leaves, with serrated or lobed leaf margins. The flowers are large and showy, solitary at the top of branches, and exhibit rich colors, including red, pink, white, purple, and other hues. The flowering period mainly occurs from April to May, and the fruiting period is from August to September[12]. The morphological characteristics of Paeonia plants, including buds, flower buds, and flowers, are illustrated in Fig. 1, which also illustrates the key morphological traits of typical species (e.g., Paeonia ostii).

Figure 1.
Morphological characteristics of Paeonia. (a) Shoot of Paeonia ostii. (b) Bud of Paeonia ostii. (c) Flower of Paeonia ostii.
Chemical composition of Paeonia
-
Paeonia plants are rich in chemical constituents, mainly including paeoniflorin, paeonol, flavonoids, terpenoids, and other compounds[13]. Among these, paeoniflorin and paeonol are the bioactive components with the highest medicinal value in this genus. Paeoniflorin exhibits significant effects such as anti-inflammation, analgesia, and immune regulation; paeonol, on the other hand, has pharmacological activities such as anti-inflammatory[14−16], antioxidant[17], antidiabetic[18], and anticancer[19] effects. Similar to Panax notoginseng, for which established systematic research has shown the phytochemical properties, pharmacological effects, and biosynthetic regulation of ginsenosides[20], the core bioactive components of Paeonia also have clear biosynthetic pathways and regulatory mechanisms worthy of in-depth exploration. Compared with other plants containing similar components, Paeonia plants have become an important resource for obtaining these bioactive components in traditional Chinese medicine (TCM) because of their abundant resources, long cultivation history, and high safety. They occupy a crucial position in modern pharmaceutical research and development, as well as in the application of traditional prescriptions. Medicinal Paeonia plants not only have abundant resources and wide distribution but also have proven ability to regulate the soil environment[21]. This has led to the extensive utilization of Paeonia in TCM and has exerted a profound impact on its pharmacology, phytochemistry, and clinical application.
-
Paeonia has a long history of medicinal use, with moutan cortex (the root bark of Paeonia suffruticosa) being first documented in Shennong's Classic Materia Medica. Traditionally, moutan cortex is used for "clearing heat, cooling blood, and promoting blood circulation to resolve stasis". White peony root (the processed root of herbaceous peonies) is used for "nourishing blood and relieving spasms", and red peony root (the unprocessed root of herbaceous peonies) is used for "activating blood circulation and removing stasis". All these traditional uses are supported by modern pharmacology. Specifically, moutan cortex, which is rich in paeonol, exerts antioxidant and anti-inflammatory effects that underpin its traditional "heat-clearing and blood-cooling" efficacy, whereas white peony root, rich in paeoniflorin, achieves its "blood-nourishing and spasm-relieving" effects through immunomodulatory and analgesic activities.
Geographical origin of Paeonia
-
The core distribution areas of the global wild distribution and cultivation patterns of Paeoniaceae plants are centered in East Asia and the Mediterranean region, spreading across the temperate zones of Eurasia. Their distribution extends northward to near the Arctic Circle and southward to Sichuan, China, spanning temperate and subarctic zones and high-altitude subtropical regions. As typical temperate plants, Paeoniaceae are intolerant of humid and hot conditions and require sufficiently low winter temperatures to complete vernalization. Its cultivation areas are extensive, with industrialized cultivation carried out in parts of Eurasia, North America, and Oceania, and the high-altitude regions in southern areas serve as restricted cultivation zones[11]. The ancient medical classic Mingyi Bielu (Supplementary Records of Famous Physicians) contains records of moutan cortex-producing areas, stating that it "grows in Ba Commandery and Hanzhong". Currently, moutan cortex, as a medicinal material, has a relatively wide distribution, mainly concentrated in regions such as Tongling and Bozhou (Anhui Province), Heze (Shandong Province), Dianjiang (Chongqing), and Zhongjiang (Sichuan Province). Notably, the moutan cortex produced in the Fenghuang Mountains in Tongling, Anhui Province, is of the highest quality and is referred to as "Paeonia ostii cortex" in the industry. This geographical specificity of the active component's content is consistent with research on Atractylodes lancea, where genome resequencing revealed that local adaptation drives the remodeling of the rhizome's metabolome, resulting in geographical differences in the content of active components[22].
Moutan cortex has the effects of clearing heat and cooling the blood, as well as promoting blood circulation and resolving blood stasis. It is widely used in clinical practice for treating various conditions, including symptoms caused by heat invading the nutrient-blood phase and warm-toxin-induced macular eruptions; bleeding disorders such as hematemesis and epistaxis; persistent manifestations of fever (e.g., evening fever with morning remission and "bone steaming" without sweating); pain associated with amenorrhea, dysmenorrhea, and traumatic injuries; and even skin diseases like carbuncles, swellings, and sores.
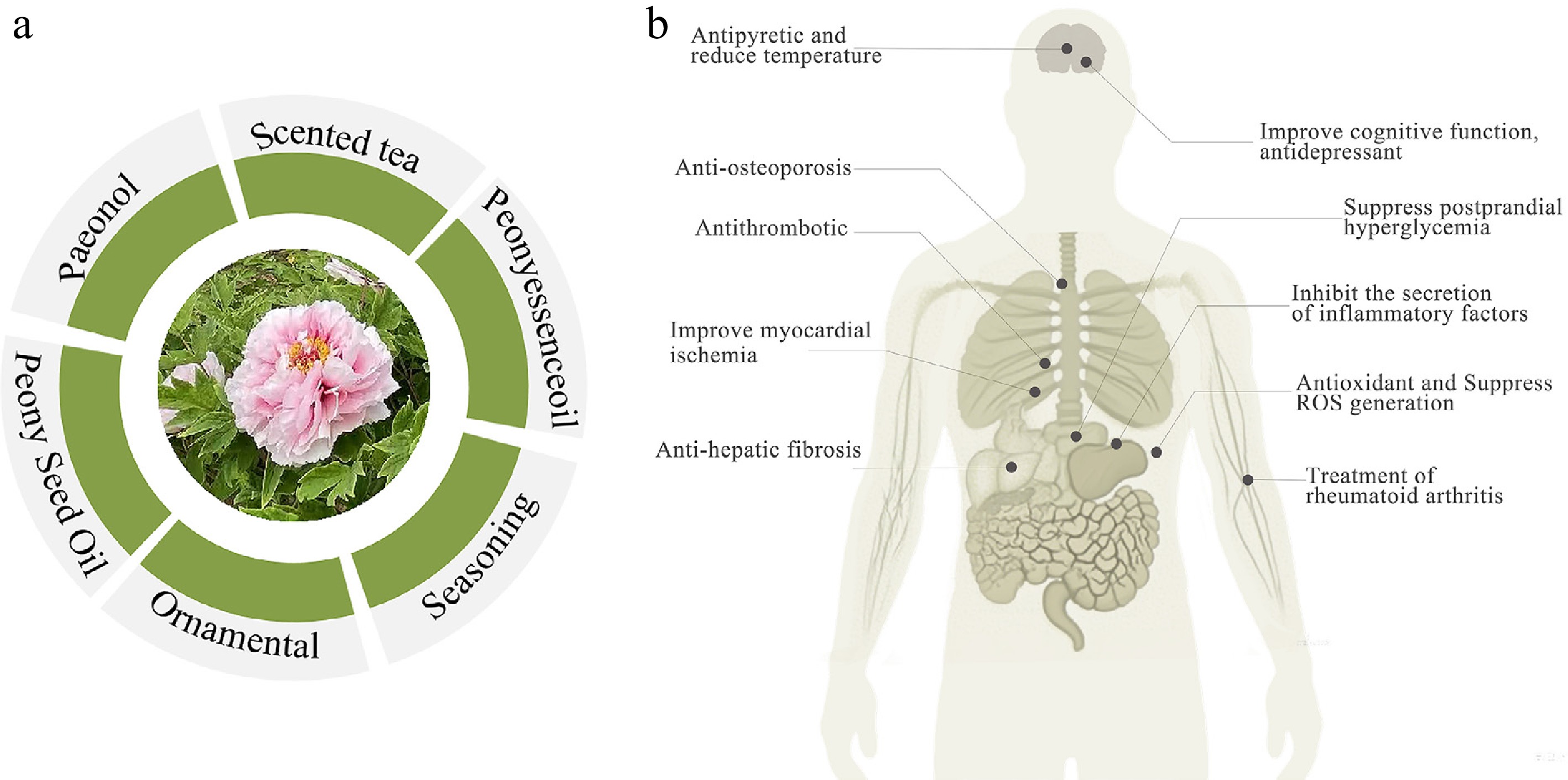
Current applications of Paeonia
-
At present, several registered drugs derived from Paeonia plants have been developed in China. For instance, Paeonol ointment, with moutan cortex as the main component, is used for treating various skin disorders such as eczema, dermatitis, pruritus, and redness/swelling caused by mosquito or bedbug bites; it also exerts a certain effect on allergic rhinitis and in the prevention and treatment of colds. Recently, Paeonia roots have also been utilized in tea production; for example, when combined with lotus leaves and Panax notoginseng, they are processed into "Yanyan tea" which has the effect of clearing heat and removing toxins[23]. Additionally, Paeonia roots can serve as a cooking ingredient, particularly in porridges and barbecues[24]. Figure 2 illustrates various applications of Paeonia plants.
-
Previous research has primarily examined the chemical composition of the Paeonia lactiflora genus, identifying and characterizing numerous compounds. The literature reports the presence of various compound components, notably monoterpene glycosides, tannins, flavonoids, phenolic acids, alkaloids, terpenoids, and glycopeptides. These components play a significant role in influencing the pharmacological activity of Paeonia lactiflora. Specifically, studies have concentrated on acetophenones like paeonol and monoterpenoid glycosides such as paeoniflorin in chemical investigations of Paeonia lactiflora.
Monoterpenoid glycosides and their glycosides
-
Terpenoids constitute a crucial component of the chemical constituents in Paeonia plants, exhibiting rich structural diversity. Monoterpenoids are primarily of the pinane type with a cage-like structure, whereas triterpenoids cover types such as oleanane, hopane, cycloartane, and nor-triterpenoids. Among these, monoterpenoids and their glycosides are the major components with a relatively high content in the root bark of Paeonia suffruticosa (moutan cortex), mostly formed by the condensation of paeonolide aglycone or its analogs with sugars. To date, 56 monoterpenoids have been isolated from Paeonia plants (Compounds 1–56). Their parent nucleus structures (A−C) and other related compounds (Compounds 57–66) are shown in Fig. 3, with detailed information provided in Supplementary Table S1[25−44].
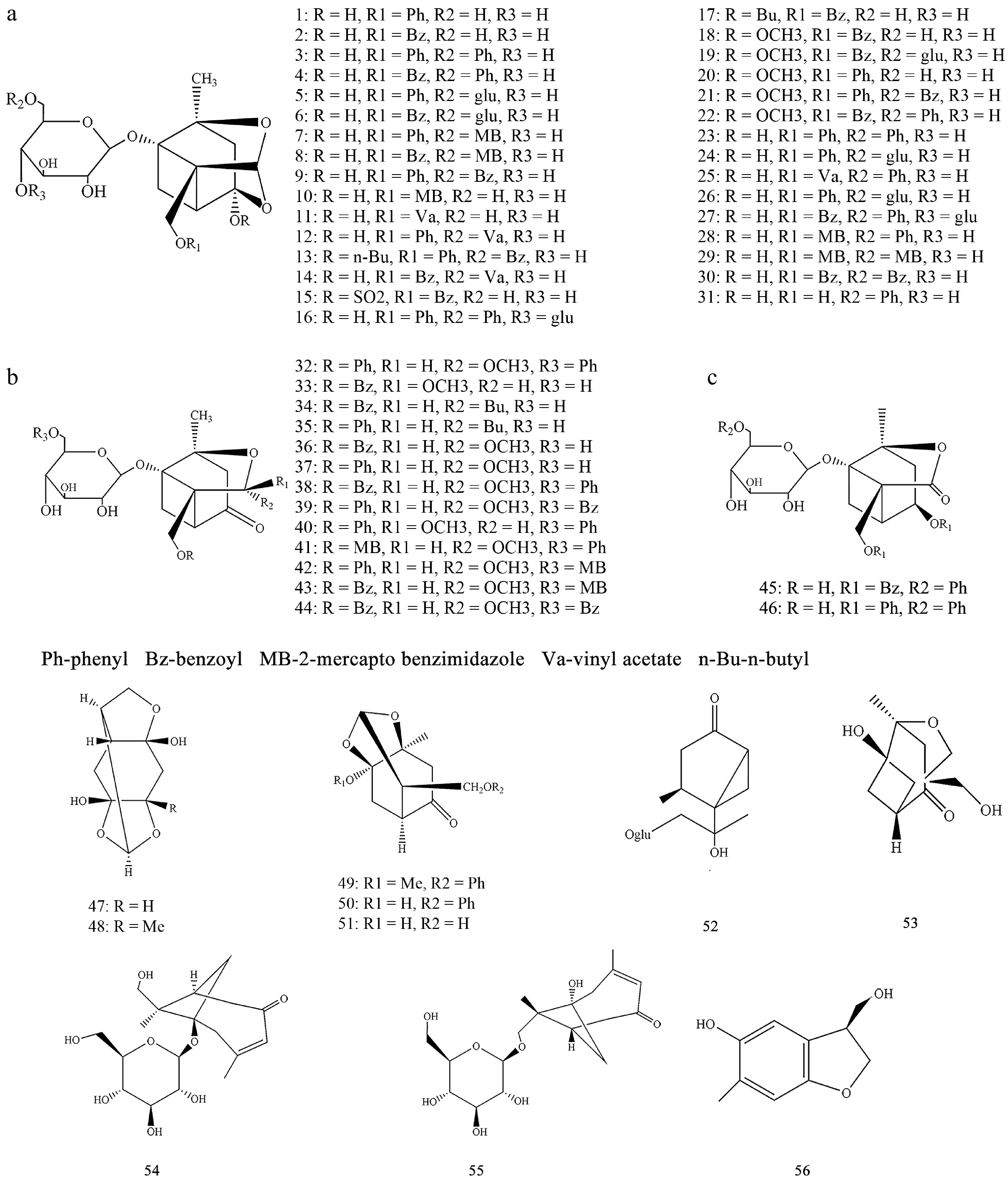
Figure 3.
Core structures of monoterpenes and their glycosides, and the chemical structures of Compounds 1−56 from Paeonia.
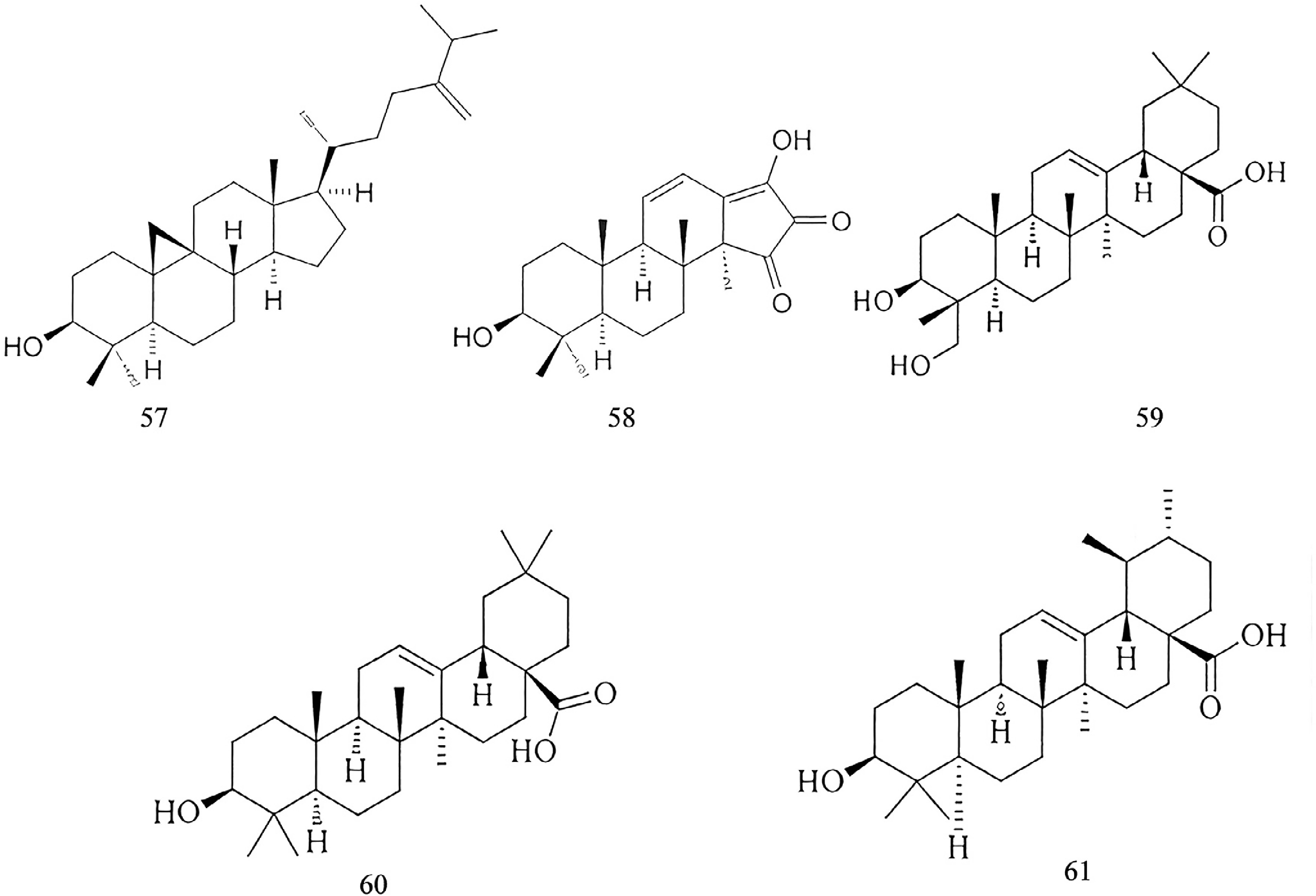
Triterpenoid glycosides
-
Researchers have isolated 33 triterpenoid compounds from various parts of this plant, including roots, root barks, rhizomes, leaves, flowers, and callus tissues. Among these, only Compounds 57 and 58 are tetracyclic triterpenoids, whereas all other compounds belong to the pentacyclic triterpenoids[39]. The structures of some triterpenoid glycosides are illustrated in Fig. 4.
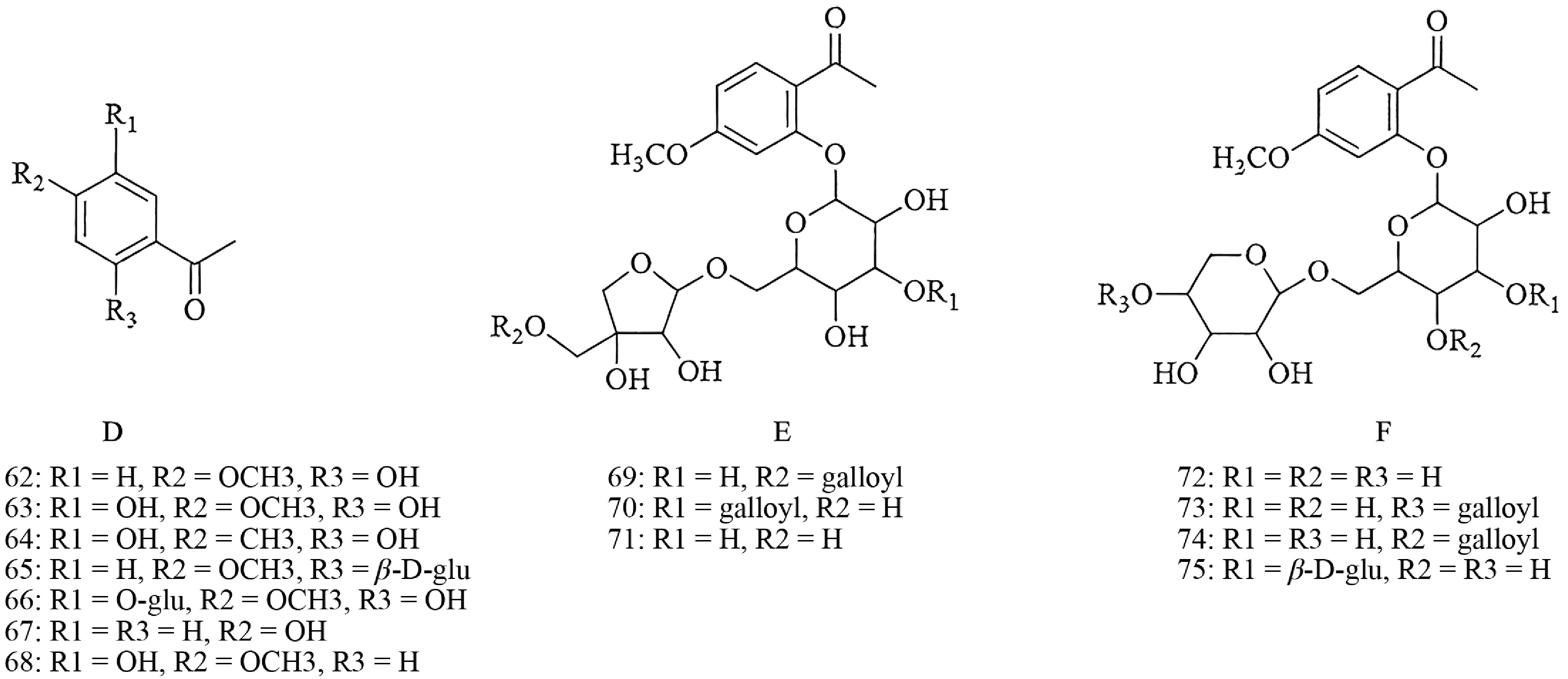
Acetophenone
-
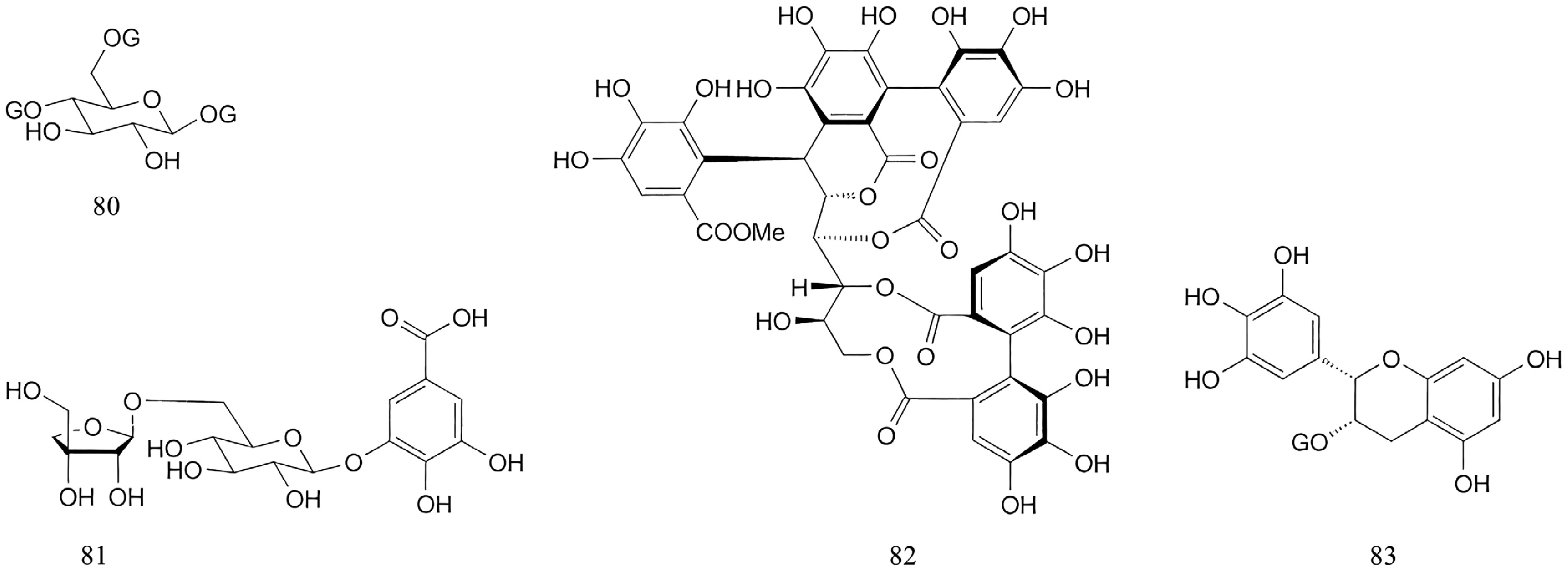
Acetophenone compounds, represented by paeonol, are primarily distributed in the roots or root barks of Paeonia plants. Besides paeonol, other members of this class include paeonolide, apiopaeonoside, and suffruticoside A. On the basis of differences in the attached glycosyl groups, acetophenone compounds can be classified into three categories (D, E, and F). Their parent nucleus structures are shown in Fig. 5, with detailed information provided in Supplementary Table S1 (Compounds 62−75).
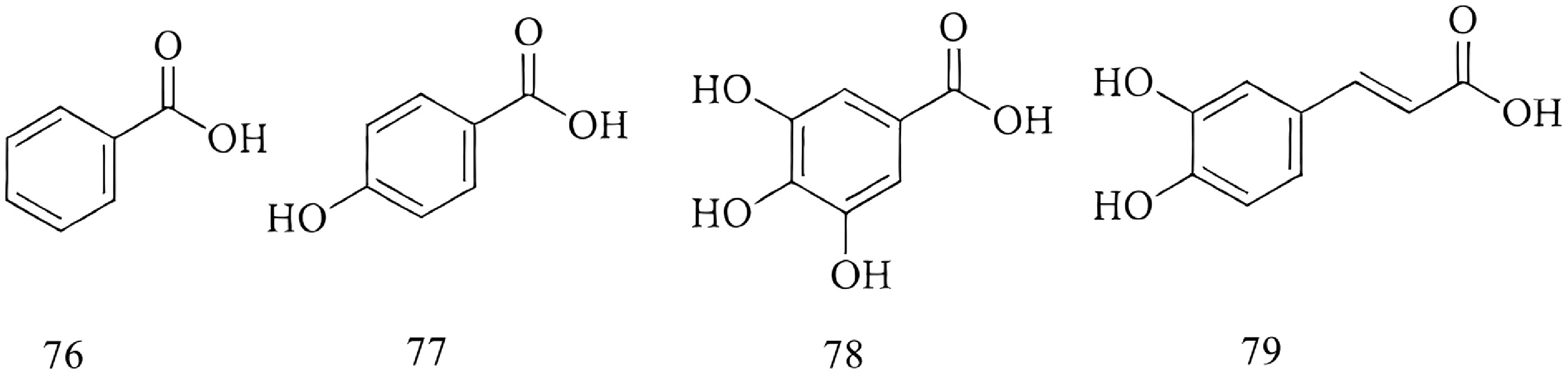
Organic acids
-
Additional compounds identified in Paeonia include benzoic acid32 (Compound 76), p-hydroxybenzoic acid[39] (Compound 77), gallic acid 32 (Compound 78), and caffeic acid[39] (Compound 79). The specific chemical structures of these compounds are presented in Fig. 6.
Tannins
-
Tannins are predominantly derived from the fruits of Paeonia, with a fraction obtained from the root bark. The majority of these tannins are classified as gallotannins[45] (e.g., Compounds 80 and 81), with some being ellagitannins (e.g., Compound 82)[46]; a minority, like Compound 83, are condensed tannins. The chemical structures of select tannins are depicted in Fig. 7.
Others
-
This plant family has yielded around 29 additional compounds through isolation, with the list encompassing 2 alkyl hydrocarbons, 12 fatty acids, 2 fatty alcohols, 1 L-amino acid, 1 nucleotide, 1 coumarin, anthraquinone, sugars, amygdalin, benzamide, and (Z)-hex-3-enal[47].
-
On the basis of the three core criteria of Q-Markers, the chemical components of Paeoniaceae plants can be screened into three categories: (1) Monoterpene glycosides represented by paeoniflorin, which have a unique cage-like pinane structure (specificity), exhibit significant anti-inflammatory and immune regulatory activities (effectiveness), and can be accurately quantified by high-performance liquid chromatography (HPLC) and ultrahigh-performance liquid chromatography–tandem mass spectrometry (UPLC-MS/MS) (detectability); (2) the acetophenones represented by paeonol, which are highly enriched in the root bark of Paeonia suffruticosa (specificity), with antioxidant and cardiovascular protective effects (effectiveness), with a detection limit of up to reach 0.01 μg/mL determined by UPLC-MS/MS (detectability); and (3) flavonoids such as gallic acid, which are widely distributed in Paeonia leaves and flowers (specificity), have strong free radical scavenging activity (effectiveness), and can be detected by thin-layer chromatography (TLC) and HPLC (detectability). These components form the core chemical basis for evaluating the quality of Paeoniaceae medicinal plants. The core Q-marker-mediated pathways, mechanisms, and associated diseases of Paeonia are illustrated in Table 1. Detailed comparisons of the components, their corresponding activities, and applications of different parts of Paeonia plants are provided in Supplementary Table S2.
Table 1. Core Q-marker-mediated pathways, mechanisms, and associated diseases of Paeonia.
Core regulatory pathways Associated Q-markers Targets/mechanisms of action Corresponding diseases NF-κB pathway Paeoniflorin, paeonol, and flavonoids Inhibiting NF-κB activation and reducing the release of inflammatory factors (TNF-α, IL-6) Rheumatoid arthritis, skin inflammation PI3K/Akt pathway Paeoniflorin, triterpenoids Regulating PI3K/Akt phosphorylation, inhibiting tumor cell proliferation, and promoting osteoblast differentiation Hepatocellular carcinoma, osteoporosis Mitogen-activated protein kinase (MAPK) pathway Paeoniflorin, paeonol Regulating the activity of p38/JNK/ERK1/2, enhancing macrophage phagocytic function, and inhibiting myocardial fibrosis Immunodeficiency, cardiovascular diseases Brief overview of the core pharmacological effects
-
As a component of TCM, Paeonia exhibits core pharmacological activities such as anti-inflammatory/immunomodulatory effects, cardiovascular protection, and antiosteoporosis, with the key bioactive components including paeoniflorin, paeonol, and total glucosides of paeony (TGP).
Anti-inflammatory and immunomodulatory effects are its core: Paeoniflorin inhibits tumor necrosis factor (TNF)-α/nuclear factor kappa B (NF-κB) pathways for broad-spectrum anti-inflammatory effects, echoing the traditional analgesic effect. Paeonol protects the vascular endothelium and suppresses oxidative stress, corresponding to its use for "promoting blood circulation and smoothing collaterals". TGP exerts definite immunomodulatory effects, validating the traditional function of "nourishing the blood". For cardiovascular protection, paeoniflorin and paeonol act via vasodilation, antioxidative effects, and inhibition of platelet aggregation, further supporting their traditional medicinal value[2,3]. Paeonol acts by scavenging free radical and inhibiting inflammatory mediators. Together, these mechanisms underpin the treatment of rheumatoid arthritis and autoimmune hepatitis, whereas TGP capsules are clinically used for their marked immunomodulation.
For cardiovascular protection, paeoniflorin and paeonol exert multitarget effects (hypotension, antithrombosis, cardiomyocyte protection) via vasodilation, improved endothelial function, and inhibition of oxidative stress/platelet aggregation[4], supporting adjuvant treatment of hypertension and atherosclerosis. Paeoniflorin regulates bone metabolism (promoting osteoblast differentiation and inhibiting osteoclasts) with potential for treating postmenopausal and glucocorticoid-induced osteoporosis[5].
Paeonia's pharmacological effects form a network centered on anti-inflammatory/immunomodulatory effects, synergistic anti-inflammation, immunomodulation, and bone protection in treating rheumatoid arthritis, and supporting other drugs in traditional compounds. Core components serve as quality markers (Q-Markers) and potential compounds for developing novel anti-inflammatory/organ-protective drugs.
Modern pharmacology aligns with traditional uses: Paeoniflorin's anti-inflammatory and blood circulation-promoting effects validate its traditional application in amenorrhea, dysmenorrhea, and traumatic injuries; paeonol's free radical scavenging and hepatoprotective activities scientifically explain its traditional efficacy of "clearing heat, cooling the blood, and detoxifying", validating traditional experience and facilitating the development of modern preparations.
Antioxidant effects
-
Paeoniaceae plants possess significant antioxidant activity, primarily attributed to the flavonoids and paeonol (key bioactive components). These constituents exert their effects by scavenging free radicals and inhibiting lipid peroxidation, Multiple lines of scientific evidence directly support their traditional efficacy in TCM of "clearing heat, cooling the blood, and detoxifying." Such antioxidant properties mitigate the oxidative damage associated with heat-toxin-related disorders, with in vivo and in vitro studies validating these protective effects[48−50].
In vivo and in vitro experiments have shown that Paeonia root extracts can effectively scavenge free radicals and mitigate lipid peroxidation[51], and exert protective effects against atherosclerosis and inflammatory diseases. Through in vitro experiments, phenolic compounds (e.g., flavonoids, tannins) in Paeonia exhibit notable antioxidant activity, which renders them suitable for the prevention of chronic diseases. The content of phenolic acids (e.g., gallic acid) in extracts derived from wild peony petals has been determined to be 12.38 mg/g, and these extracts demonstrate strong free radical scavenging capacity[52].
Additionally, interspecific differences exist in the expression of compounds with antioxidant activity. Compared with Paeonia veitchii, Paeonia lactiflora shows greater antioxidant activity, attributed to its higher levels of flavonoids and hydrolyzable tannins[53]. From the perspective of resource utilization, during the germination stage, Paeonia leaves reach the highest paeoniflorin content (2.8 mg/g), supporting their viability as novel antioxidant resources for cultivation[54].
Antitumor effects
-
Paeoniflorin and its derivatives have exhibited significant potential for antitumor research. Studies have shown that these bioactive components exert antitumor effects by inhibiting tumor cells' proliferation and metastasis, inducing apoptosis, and regulating signaling pathways (e.g., PI3K/AKT, mitogen-activated protein kinase [MAPK])[55]. For instance, Paeonia extracts inhibit the proliferation of cancer cells (e.g., breast cancer, lung cancer) and induce their apoptosis, which is closely associated with the ability to interfere with tumor cell signal transduction. The paeoniflorin derivative suffruticosol B can promote osteoblast differentiation and inhibit osteoporosis-related tumors[56].
Additionally, terpenoids (e.g., paeoniflorin) in Paeonia plants also show antitumor activity. Triterpenoids (e.g., paeonisuffral) induce apoptosis in tumor cells by activating caspase-3, and this regulatory effect may be mediated through the alleviation of intracellular calcium overload and the improvement of mitochondrial function in tumor cells[57]. Paeoniflorin downregulates the NF-κB and PI3K/Akt signaling pathways to inhibit the proliferation of HepG2 hepatoma cells, with a half-maximal inhibitory concentration (IC50) of 45 μM. Meanwhile, the combination of paeoniflorin with chemotherapeutic drugs reduces cisplatin resistance and enhances cytotoxicity against A549 lung cancer cells[58].
Antibacterial effects
-
In vitro studies have shown that flavonoids, phenols, and terpenoids from Paeonia exhibit significant antiviral, antibacterial, and antifungal activities, primarily manifested as inhibitory effects against a variety of bacteria (e.g., Staphylococcus aureus, Escherichia coli) and fungi (e.g., Candida albicans). Specifically, the root extract of Paeonia emodi inhibits Candida albicans (with an inhibition zone diameter of 15.3 mm), and an extract of Paeonia lactiflora is effective against Gram-positive and Gram-negative bacteria such as Staphylococcus aureus (mean inhibitory concentration = 0.5 mg/mL) and Escherichia coli[53]. Paeoniflorin exerts antiviral effects through inhibiting viral replication and regulating immune responses. Meanwhile, monoterpenoid glycosides are capable of blocking the neuraminidase activity of the H1N1 influenza virus[59].
Immune regulation
-
Paeoniflorin is a major bioactive monomer of Paeonia. In vitro studies have shown that it can reduce the concentration of intracellular calcium ions by inhibiting the release of pro-inflammatory mediators (e.g., prostaglandin E2, leukotriene B4, nitric oxide, etc.), thereby reducing the inflammatory response. It also bidirectionally regulates lymphocyte function by inhibiting overactivated T cells (e.g., decreasing the proportion of Th17 cells) while enhancing macrophages' phagocytic capacity[60]. Paeonia extracts activate the MAPK pathway (p38/JNK/ERK1/2) through a TLR4-dependent mechanism, which promotes macrophages' immune activity, increases the production of cytokines (e.g., TNF-α, IL-6) and nitric oxide (NO), and enhances phagocytic function[61].
In vitro studies have continuously elucidated the role of paeoniflorin extracts in immune regulation. CP-25, a paeoniflorin derivative prepared through structural modification, improves the pathological progression of autoimmune diseases (e.g., rheumatoid arthritis) in animal models by regulating G protein-coupled receptor kinase 2 (GRK2); the underlying mechanism includes modulating T cell subset balance and cytokine networks[62]. For specific diseases, PSORI-CM02 is a clinically applied TCM for psoriasis, whose mechanism has remained unclear until recently. Subsequent studies have clarified that it inhibits Human adult low Calcium high Temperature keratinocyte (HaCaT) cells' proliferation and alleviates symptoms of psoriasis by regulating the PI3K/Akt/mammalian target of rapamycin (mTOR)-autophagy axis[63]. Rheumatoid arthritis is a chronic inflammatory and systemic autoimmune disease with an unknown etiology. Its pathogenesis involves excessive activation of the synoviocytes and immune cells, increased secretion of inflammatory mediators and cytokines, and dysfunction of the EP-G-protein-cAMP and MAPK signaling pathways in the synoviocytes. TGP and its main active component, paeoniflorin, exert anti-inflammatory and immunoregulatory effects by inhibiting the activation of immune cells and synoviocytes, reducing the production of inflammatory substances, and restoring abnormal signaling in the synoviocytes[64].
Cardiovascular protection
-
In vitro studies have shown that paeoniflorin exhibits significant cardioprotective effects, which are exerted through the inhibition of oxidative stress, the regulation of mitophagy, the suppression of inflammatory responses, and the prevention of cardiomyocyte apoptosis and hypertrophy[65]. Bioactive components derived from paeoniflorin, such as paeonol, mitigate the progression of atherosclerosis by suppressing oxidative stress and inflammatory responses in vascular endothelial cells, as well as inhibiting the proliferation and migration of vascular smooth muscle cells. Additionally, these components exert cardioprotective effects through regulating lipid metabolism and improving cardiac function[66]. Additionally, paeonol exerts such effects by modulating miRNA expression, inhibiting inflammatory signaling pathways (e.g., PI3K/Akt/NF-κB pathway), and preventing vascular endothelial cell damage[67]. The protective role of paeoniflorin in myocardial ischemia–reperfusion injury has also attracted attention, as it can upregulate SIRT1 expression, inhibit myocardial fibrosis, and alleviate myocardial damage[68].
In terms of its pharmacokinetic properties, paeoniflorin exhibits low bioavailability. In vivo studies have shown that it is widely distributed in multiple tissues after absorption, including the heart, liver, spleen, lung, kidney, stomach and intestine, and it is capable of crossing the blood–brain barrier through passive diffusion. This compound has a short half-life (approximately 1.8 hours); however, its absorption rate can be accelerated and the mean residence time prolonged when co-administered with other TCM remedies[69].
Antipyretic and temperature-reducing effects
-
Research on the antipyretic effects of medicinal Paeoniaceae plants has focused on components such as peony roots, paeoniflorin, and Shaoqi polysaccharide glycosides (a compound preparation of Paeonia and Astragalus).
In vivo studies have found that radix paeoniae (peony root) extract exerts a significant hypothermic effect on both normal body temperature and yeast-induced hyperthermia. The efficacy of this extract is similar to that of paracetamol—a standard antipyretic drug—but demonstrating a longer duration of action, which indicates its potential value in antipyretic therapy[70].
For paeoniflorin, in vitro studies have shown that it significantly inhibits carrageenan-induced thermal hyperalgesia, suppresses the production of inflammatory mediators (e.g., nitric oxide, prostaglandin E2), and reduces neutrophil infiltration as well as the expression of inducible nitric oxide synthase and cyclooxygenase-2. This suggests it may exert anti-inflammatory and analgesic effects by inhibiting the production of inflammatory mediators[16].
Cosmetics and health products
-
Owing to their rich natural bioactive components, Paeonia plants are widely used in cosmetics and health products. A large number of in vitro studies have shown that compounds such as polyphenols, flavonoids, and saponins in Paeonia plants exhibit antioxidant, anti-inflammatory, and antiaging effects, suggesting promising application prospects in cosmetics[71]. Paeoniflorin, a monoterpene glucoside isolated from the root of Paeonia lactiflora, possesses multiple pharmacological activities, including anti-inflammatory, antioxidative, analgesic, anticancer, and immunoregulatory effects. Clinically, it is utilized for the treatment of diseases such as rheumatoid arthritis and systemic lupus erythematosus. Additionally, paeoniflorin (PF) has been demonstrated to exert vasculoprotective effects, safeguard against vascular endothelial cell dysfunction, and inhibit hypoxia-induced epithelial–mesenchymal transition (EMT) in human breast cancer cells[72]. The natural components of Paeonia plants are regarded as important sources of natural cosmetics because of their low toxicity and good biocompatibility. Specifically, polyphenols in these plants exert antioxidant effects, helping reduce the skin damage caused by free radicals to achieve antiaging outcomes; natural oils and extracts can enhance the skin's moisturization and elasticity[73,74].
The bioactive components of Paeonia plants have pharmacological effects including anti-inflammation, antitumor, and antidepressive effects, endowing them with potential application value in health products. Polyphenols and saponins derived from Paeonia plants exhibit anti-inflammatory and antioxidant activities, and these bioactive components may contribute to improvedimmune system function as well as the promotion of physical health[75].
Antiosteoporotic effects
-
Paeonia plants exhibit significant antiosteoporotic effects, such as promoting osteoblast differentiation, inhibiting apoptosis, and improving bone metabolism[76,77]. Paeoniflorin plays a crucial role in osteoporosis treatment by exerting dual regulatory effects on bone metabolism. In vitro studies have shown that it promotes osteogenesis in MC3T3-E1 cells and inhibits osteoclast formation in RAW264.7 cells. Notably, this compound exhibits no cytotoxicity, making it a key candidate for osteoporosis therapy[77].
-
This section systematically reviews the safety data and potential risks of peony-derived preparations. First of all, at the level of clinical application, the monitoring of adverse reactions of preparations represented by TGP capsules showed that the spectrum of adverse reactions was mainly mild and transient reactions. The most common reports were gastrointestinal discomfort (such as nausea, abdominal distension, diarrhea) and skin reactions (such as rash and itching), which can be relieved by themselves after reduction or withdrawal[69]. At present, reports of serious adverse reactions (such as liver and kidney dysfunction) are rare, but attention still needs to be paid to their use as long-term, high-dose medications.
At the nonclinical research level, toxicological evaluation of its core active ingredients (such as paeoniflorin) provides basic safety evidence. Acute toxicity experiments have shown that paeoniflorin does not cause obvious toxic reactions or death in experimental animals within the range equivalent to clinical therapeutic doses[69]. Long-term toxicity studies suggest that its toxicity is dose- and time-dependent, and ultrahigh doses or long-term administration may have reversible effects on organs such as the liver and kidney, which provides an important reference for determination of the clinical safety window[63].
This section also analyzes the key variables that affect security. Dose and course of treatment are core factors influencing the safety of medicinal Paeoniaceae plants, as exceeding the recommended dose or long-term administration may significantly increase the risk of adverse reactions. Additionally, processing technology plays a crucial role in safety and efficacy. For instance, sulfur fumigation used for whitening or insect prevention not only results in sulfur dioxide residues but also impairs the stability of active components such as paeoniflorin, reduces the therapeutic effects, and may generate uncertain complex products that introduce potential safety hazards. Furthermore, preliminary studies have indicated that pharmacokinetic or pharmacodynamic interactions may occur when these medicinal plants are combined with certain anticoagulants, immunosuppressants, or drugs metabolized by the same hepatic drug-metabolizing enzymes, requiring vigilance against efficacy-related superposition or antagonism and an elevated risk of adverse reactions.
-
This section focuses on the in vivo processes of the core active ingredients (paeoniflorin, paeonol) of peony-derived preparations. In terms of absorption, the oral bioavailability of paeoniflorin is low. Studies have shown that combining it with specific TCM remedies (such as Pueraria lobata) may improve its absorption efficiency by inhibiting intestinal metabolism or regulating transporters[69]. In terms of distribution, both of them show good tissue permeability and can be widely distributed in target organs such as heart, liver and kidney, and paeoniflorin has the ability to cross the blood–brain barrier, suggesting its potential role in the central nervous system[69]. The metabolic process is mainly carried out in the intestinal flora and liver, and is converted into active metabolites (such as albiflorin) through hydrolysis and other reactions, which is the key link in its pharmacological effects[78]. The excretion pathway is mainly the kidney and biliary tract, and it is finally excreted via urine and feces[69]. These pharmacokinetic characteristics have been fully supported by recent research[78], which provides a scientific basis for understanding its mechanism of action and optimizing the dosing regimen.
-
Quality control methods for medicinal Paeonia plants are documented in the PPRC (2020 edition), including macroscopic characteristics, identification, tests, extracts, and assay. Among these, the most commonly used and relatively specific identification methods include TLC, HPLC, and UPLC-MS/MS[78].
The pharmacological activity and clinical application value of medicinal Paeonia plants are highly dependent on their specific chemical component profiles. Recent studies have confirmed that monoterpenes and their glycosides, triterpenoids, flavonoids, and phenols are core quality markers (Q-Markers) for characterizing their quality; their contents and proportions directly affect the safety and efficacy of the medicinal materials[79]. Relevant information is summarized in Table 2.
Table 2. Comparison of quality control methods.
Analysis and comparison TLC HPLC UPLC-MS/MS Core principle Depending on the differences in the partition coefficients of compounds between the stationary phase and the mobile phase, separation is achieved. Depending on the differences in the interactions of compounds with the stationary phase and the mobile phase in the chromatographic column, high-efficiency separation is achieved. Depending on the ultrahigh separation efficiency of ultrahihg performance liquid chromatography and the high sensitivity, high selectivity, and structural analysis capability of tandem mass spectrometry Main application level Basic qualitative preliminary screening Multicomponent quantitative analysis routine quality control Precise identification and traceability analysis of complex systems Dissociation efficiency Low High Ultrahigh Detection sensitivity Low High Ultrahigh Main detection objectives Several key markers (such as Paeonia lactiflora glycosides, paeonol) Multiple target components Broad-spectrum screening, trace/unknown substance identification, complex metabolite analysis, characteristic metabolic profiling Quantitative capability Limited Strong Extremely strong Advantage Simple, rapid, and economical, suitable for preliminary screening Dissociation good; quantitative accuracy; polydactyly standards can establish fingerprint profiles Powerful identification of Broussonetia papyrifera, with great potential for resolving complex systems and integrating omics approaches. Limitations Low resolution, limited sensitivity, inaccurate quantification, and insufficient information content Limited analytical capability for difficult-to-dissociate substances or trace substances Complex operation, high development requirements, and high operating costs Thin-layer chromatography-based identification
-
As a basic qualitative tool, TLC is mainly used for preliminary screening of medicinal Paeonia plants, enabling authenticity to be identified via target components such as paeoniflorin and paeonol. The PPRC (2020 edition) documents the methods of sample preparation for TLC-based identification of white peony root (processed Paeonia lactiflora roots) and red peony root (unprocessed Paeonia roots), laying a foundation for subsequent research[80].
TLC enables the rapid detection and localization of compounds in complex extracts derived from three Paeonia species. When combined with anatomical analysis, this technology not only verifies the botanical differentiation of the three Paeonia species but also provides a simple, economical, and unambiguous method for their authentication—an outcome that is crucial for guaranteeing the safety and efficacy of their application in TCM[81]. Quantitative analysis showed variations in the proportion of chemical components among different samples, and this reference extract could be effectively used to identify red peony root materials with superior quality.
High-performance liquid chromatography-based identification
-
High-performance liquid chromatography has been applied to determine eight components in peony roots. This analytical approach enables effective differentiation of peony varieties and production areas, laying a foundation for the development of variety-specific quality control strategies for peonies[82]. An HPLC method has been established for the simultaneous determination of four components (e.g., sodium benzoylpaeoniflorin sulfonate) in sulfur-fumigated white peony root. This analytical method revealed the effect of sulfur fumigation on the active components of white peony root and also provided a novel index for the quality evaluation of this medicinal material[83].
High-performance liquid chromatography has also been applied to the simultaneous analysis of 15 monoterpenoid glycosides. This analytical approach confirmed a significant correlation between the components' contents in peony seed cakes and their producing regions, thereby providing a basis for origin tracing and quality tracing for this material[84]. Another HPLC-based method has been developed for the quantification of paeoniflorin, and this analytical approach contributes to the standardization of quality criteria for dried peony extracts[85]. Compared with TLC (using a single compound as the quality marker), HPLC expands the control dimension to a certain extent.
Ultrahigh-performance liquid chromatoragphy–tandem mass spectrometry-based identification
-
Ultra-high performance liquid chromatography–tandem mass spectrometry (UHPLC-MS/MS) has been successfully applied to screen sulfonate derivatives in white peony root. This analytical technique reveals the impact of content variations in these derivatives on efficacy and also highlights its advantages in safety control[8]. Ueno combined UPLC-MS/MS with Proton Nuclear Magnetic Resonance (1H-NMR) metabolomics and found that differences in the chemical composition of peony roots from Japan and China originated from the growth environments rather than the species, providing data support for standardizing cultivation[86]. Quantitative proton nuclear magnetic resonance (qHNMR) has also been applied to quantify metabolite differences, confirming the potential of metabolomics in the quality evaluation of multisource medicinal materials. Ultra-high performance liquid chromatography-tandem mass spectrometry, characterized by ultra-high resolution and sensitivity, addresses challenges in component analysis within complex systems[87].
By integrating the advantages of three techniques, namely using TLC for rapid preliminary screening (front end), HPLC for multiindex quality control (middle), and UPLC-MS/MS for in-depth verification (back end), a three-level quality control network of preliminary screening, quantification, and traceability can be constructed, aiming to further enhance the internationalization and universality of quality standards for medicinal Paeoniaceae materials[88].
Existing questions
-
Mainstream medicinal materials from Paeoniaceae such as baishao, red peony root, and moutan cortex are currently assessed for quality control using paeoniflorin and paeonol as chemical markers[89]. A rudimentary quality control framework has been implemented to verify the authenticity of the materials and to establish a minimum threshold for active components. Nevertheless, several significant challenges persist: Challenges exist in medicinal component extraction technology. For example, processing varies. Baishao requires peeling and boiling, red peony root is sun-dried without peeling, and moutan cortex needs peeling and pith removal. These different processed products show significant component differences, whereas targeted quality control methods are lacking. The contents of nine major active components in Baishao (white peony root) undergo changes during processing procedures including sulfur fumigation, fermentation, cutting, stir-frying, wine-processing, and vinegar-processing. Among these components, five (including paeoniflorin) exhibit significant changes, with statistical significance set at p < 0.01[9].
Quality control standards still rely on low-sensitivity techniques such as TLC, which uses characteristic spots of paeoniflorin (retardation factor [Rf] 0.35 ± 0.05) and paeonol (Rf 0.68 ± 0.05) for rapid species identification. However, UPLC-MS/MS-based multicomponent analysis has not been incorporated into the mainstream pharmacopoeial methods.
-
Paeonia, a core genus within the medicinal plants, encompasses extensive medicinal resources. Its taxa not only occupy a prominent niche in TCM but also exhibit considerable potential for modern pharmaceutical development and application. In recent years, in-depth investigations into medicinal Paeonia have achieved remarkable progress in the profiling of chemical constituents, in vitro/in vivo bioactivity screening, and pharmacodynamic assessment findings that offer critical guidance for new drug discovery, existing drug repositioning/optimization, and the sustainable exploitation of plant resources.
Emerging research directions, such as senescence mechanisms in Paeonia have made initial progress[88], and integrating Q-Marker analysis with specialized research will further expand the depth and breadth of medicinal Paeoniaceae resources and their development. Systematic phytochemical and pharmacological research on medicinal Paeonia provide a scientific rationale for the development of related pharmaceuticals. For instance, studies on its chemical composition and pharmacology have verified a broad spectrum of bioactivities, including anti-inflammatory, anti-tumor, and anti-coagulant effects. In-depth exploration of these bioactive components has established the pharmacological basis for their clinical translation. Additionally, research into the lipid utilization of Paeonia opens up novel pathways for its multipurpose exploitation.
Studies on strategies for the sustainable utilization of Paeonia resources hold significant social, economic, and ecological value in terms of natural resource conservation, environmental pollution abatement, and guaranteeing the sustainable production of pharmaceuticals. Measures such as elite cultivar selection, optimizing cultivation techniques, and rational resource allocation can effectively alleviate the environmental impacts while ensuring sustainable access to medicinal plant resources.
Future research will prioritize the structural modification of bioactive components from medicinal Paeonia, de novo drug design, and evaluations of clinical safety and efficacy (e.g., pharmacokinetics/pharmacodynamics). Concurrently, with the intensification of globalization, international collaboration will assume an increasingly crucial role in the research and development of medicinal Paeonia; this collaboration is expected to accelerate the process of new drug development, facilitate the global utilization of Paeonia resources, and make greater contributions to human health care.
In summary, research on medicinal Paeonia resources is not only key for enhancing the cognition, conservation, and rational utilization of these taxa but also exerts profound impacts on advancing new drug development, ensuring the sustainable supply of pharmaceuticals, and boosting the development of related biopharmaceutical industries. Therefore, future studies should focus on in-depth exploration and cross-disciplinary collaboration in these domains to yield more impactful outcomes.
-
The authors confirm their contributions to the paper as follows: study conception and design: Lu P, Zhang X; data collection: Lu P, Yuan Q; analysis and interpretation of results: Lu P, Ma X, Chen Y, Lin T; draft and revised manuscript preparation: Zhang X, Zhang Y. All authors reviewed the results and approved the final version of the manuscript.
-
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
-
This work was supported by the Natural Science Basic Research Program of Shaanxi (No. 2024JC-YBQN-0182) and the Forestry Science and Technology Innovation Project of Shaanxi Province (No. SXLK2023–02–25).
-
The authors declare that they have no conflict of interest.
- Supplementary Table S1 Key Bioactive compounds in the genus Paeonia.
- Supplementary Table S2 Comparison of components-activities-applications in different parts of Paeonia plants.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Lu P, Yuan Q, Ma X, Chen Y, Lin T, et al. 2026. From traditional materia medica to modern drugs: a review of the chemistry, pharmacology, and exploitation of the genus Paeonia. Medicinal Plant Biology 5: e009 doi: 10.48130/mpb-0026-0004
From traditional materia medica to modern drugs: a review of the chemistry, pharmacology, and exploitation of the genus Paeonia
- Received: 25 October 2025
- Revised: 06 February 2026
- Accepted: 09 February 2026
- Published online: 23 April 2026
Abstract: As a crucial medicinal group in Paeoniaceae, Paeonia plants are rich in bioactive monoterpenoids and phenolic compounds (e.g., paeoniflorin, paeonol, and flavonoids) in their roots, flowers, and other parts. They serve as core medicinal resources in traditional Chinese medicine (TCM) for nourishing blood, activating blood circulation, alleviating inflammation, and relieving pain. In recent years, in-depth studies have clearly elucidated the molecular mechanisms of these bioactive components. With breakthroughs in whole-genome sequencing, transcriptomics, and metabolomics technologies, over 100 secondary metabolites in Paeonia plants has been accurately identified, and multiple pharmacological mechanisms have been analyzed. This review systematically summarizes the current status of Paeonia plants in terms of plant resources, the exploration of bioactive components, analysis of the pharmacological mechanisms, and development/utilization. Combined with the cutting-edge progress in multi-omics technologies and synthetic biology, this review indicates the key directions of Paeonia in innovative drug research and development, aiming to provide a theoretical reference and technical support for the in-depth development of this group of medicinal plants.
-
Key words:
- Paeonia /
- Pharmacology /
- Phytochemistry