-
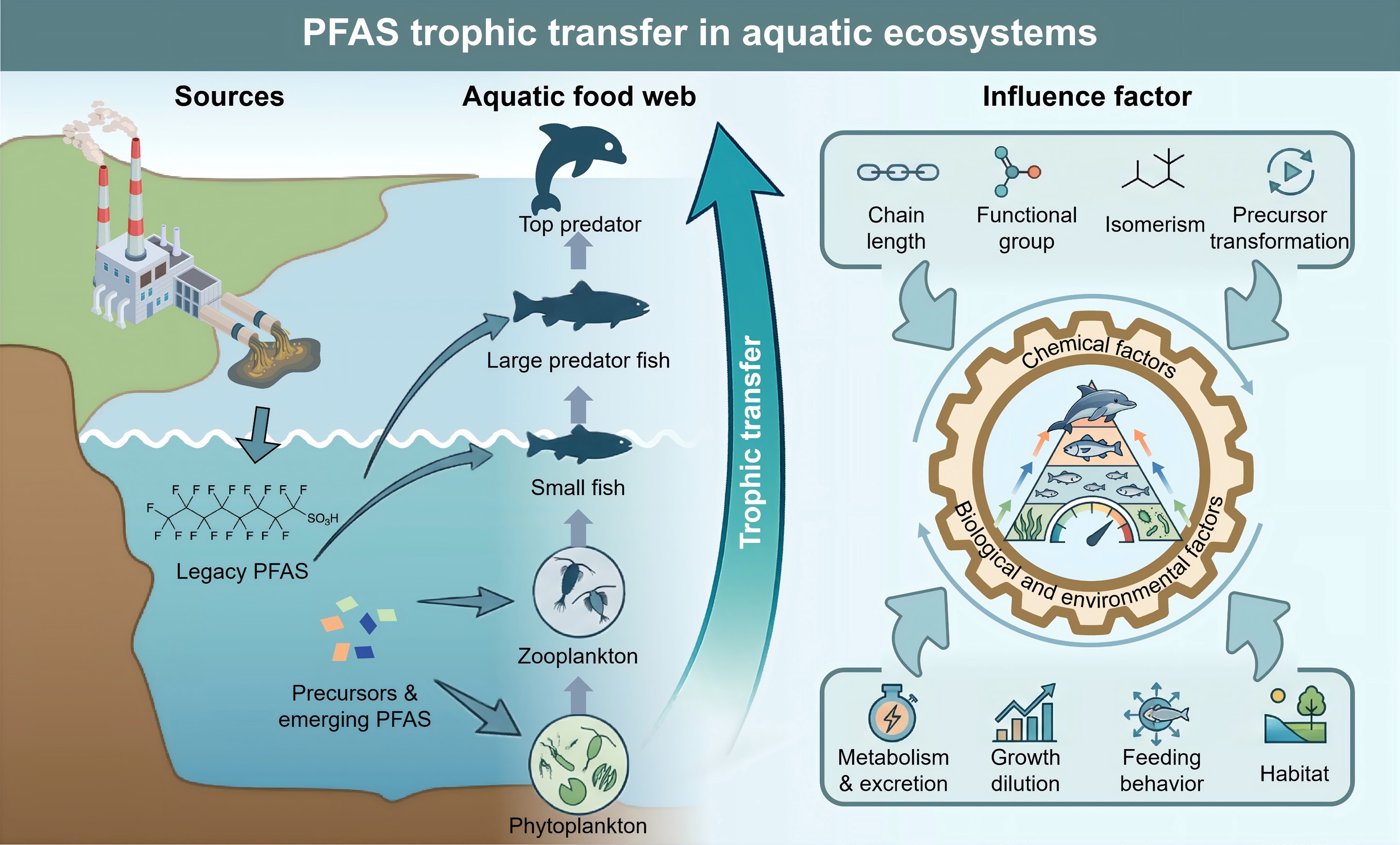
Per- and polyfluoroalkyl substances (PFAS) are a class of fluorinated synthetic compounds. Since the 1950s, PFAS have been widely used in textiles, firefighting foams, non-stick cookware, and food packaging due to their unique properties, such as hydrophobicity, lipophobicity, and thermal stability[1,2]. During the manufacturing, transportation, use, and disposal of products, PFAS are continuously released into the environment. They have been widely detected in air, water bodies, sediments, and organisms[3,4]. Additionally, they are commonly found in human tissues[5]. Due to their widespread presence, environmental persistence, long-range transport, and potential health risks, PFAS have attracted global attention. Many countries and international organizations have taken measures to restrict their production and use[6]. For instance, perfluorooctane sulfonic acid (PFOS) and its related compounds were included in Annex B of the Stockholm Convention in 2009, while perfluorooctanoic acid (PFOA) was listed in Annex A in 2019[7]. As a major convergence and migration channel for PFAS, the aquatic environment continuously receives PFAS through atmospheric deposition, surface runoff, and wastewater discharge, making aquatic ecosystems a key area of concern for PFAS exposure and toxic effects[8−10]. PFAS, including PFOS, PFOA, perfluorononanoic acid (PFNA), and perfluorodecanoic acid (PFDA), can cause significant immunotoxicity in aquatic organisms at concentrations of approximately 10 μg/L. Among them, PFDA has been shown to interfere with immune regulatory mechanisms through the Toll-like receptor signaling pathway[11,12]. Simultaneously, exposure to PFOS, perfluorobutane sulfonic acid (PFBS), and 6:2 chlorinated polyfluorinated ether sulfonic acid (6:2 Cl-PFESA) can induce neuroinflammation, cognitive impairment, and neuronal apoptosis in zebrafish (Danio rerio), among other neurotoxic effects[13]. Short-chain PFAS, such as perfluorohexane sulfonate (PFHxS), which are regarded as substitutes, have also been reported to cause hepatotoxicity in zebrafish at environmentally relevant concentrations[14].
In aquatic food webs, PFAS can accumulate continuously in organisms across trophic levels (TLs) due to: (1) their high stability and resistance to degradation; (2) bioconcentration from water and sediment; and (3) trophic transfer along feeding relationships in the food chain. Biomagnification results in much higher concentrations in higher TL predators than in their prey, sometimes by several orders of magnitude[15,16]. While the initial concentration of PFAS in the environment may be low, organisms at high TLs in the food chain (such as large fish, marine mammals, and fish-eating birds) may accumulate relatively high levels in their bodies[17,18]. Given the persistence, bioaccumulation potential, and toxicity of PFAS, the biomagnification of PFAS in aquatic food webs not only threatens biodiversity and ecosystem stability but also indirectly endangers human health via the consumption of aquatic food sources, resulting in dietary exposure and subsequent ecological health risks[19,20]. Exposure to PFAS has been associated with an increased risk of cancers, such as those of the liver, kidney, and breast. Furthermore, epidemiological and animal studies have demonstrated that PFAS exposure is closely linked to immune dysfunction and hematological damage, inducing immunosuppression, lymphocyte abnormalities, and alterations in blood parameters. These effects can ultimately promote the development of cancer and immune-related diseases through mechanisms involving inflammatory responses, cell cycle dysregulation, and the JAK-STAT signaling pathway[21,22]. Therefore, accurately assessing the biomagnification potential of PFAS in food webs is a critical component of ecological risk assessment. The insights gained from this assessment are essential for understanding exposure levels in high-trophic-level organisms, revealing potential ecological and health risks, and ultimately informing science-based pollution control and environmental management strategies[23]. Currently, the biomagnification factor (BMF) and trophic magnification factor (TMF) are widely used to assess the biomagnification capacity of pollutants in food webs[24]. BMF reflects the ratio of pollutant concentration in a predator to that in its prey; for instance, it has been used to confirm the biomagnification effect of PFAS[25]. TMF, on the other hand, is calculated based on the slope of the relationship between pollutant concentration and TL, representing the amplification trend of pollutants throughout the food web, and has been adopted in the assessment of the biomagnification of polybrominated diphenyl ethers and hexabromocyclododecane in plankton and benthic organisms[26]. However, BMF does not encompass the diversity of different accumulation pathways, whereas TMF, considering the food web as a whole, can be regarded as the weighted average of BMF values for each part and is often considered the 'gold standard' for evaluating the biomagnification potential of chemicals[27,28]. The TMF provides a more robust assessment of biomagnification than the BMF from a single predator-prey pair[29]. This is exemplified by the work of Tomy et al., who reported that PFOA showed biomagnification in specific feeding relationships within an eastern Arctic marine food web, whereas it did not magnify across the whole food web, presenting a contrasting outcome[30].
The biomagnification potential of PFAS in aquatic food webs is the result of complex interactions among chemical properties, biological and ecological characteristics, and the ecosystem's structure[31]. The hydrophobicity of chemicals is often used as a key factor for predicting the bioaccumulation potential of persistent organic pollutants[32]. However, the mechanism by which PFAS bioaccumulate exhibits a distinct complexity as they do not follow the traditional lipid distribution pattern but rather rely more on protein binding, active transport, and specific excretion pathways[33−35]. PFAS exhibit a high affinity for biological macromolecules, such as liver fatty acid-binding protein and serum albumin. This protein-directed accumulation mechanism suggests that even minor differences in their molecular structure, such as carbon chain length and terminal functional groups, can impact binding strength and affinity, tissue distribution, and in vivo clearance kinetics[36,37]. Within aquatic ecosystems, the structure of food webs (such as TL length and benthic-pelagic coupling relationships), species-specific physiological characteristics (including metabolic capacity, growth rate, and feeding strategies), as well as the distribution behavior of PFAS in multiple media, such as water and sediment, will jointly regulate their migration and biomagnification patterns among TLs[38−40]. In addition, while the biomagnification potential of legacy PFAS (such as PFOS and PFOA) is well understood, a systematic understanding of the environmental behavior of emerging PFAS (including short-chain alternatives) and PFAS precursors, which may be degraded into terminal PFAS in organisms, is lacking[41,42]. Current evidence suggests that there are variations in the biomagnification potential of PFAS across different aquatic systems; the underlying causes remain poorly understood, as these differences may arise from complex interactions among water physicochemical properties, pollution inputs, sediment characteristics, and primary productivity, highlighting an urgent need for further investigation[43]. The structural characteristics of PFAS, such as carbon chain length, functional group type, differences in branched and linear structures, and the biotransformation process of precursor substances, can affect their biomagnification behavior in food webs. However, there is still a lack of a unified assessment that can systematically integrate these structural factors that influence the degree of biomagnification[44−46]. Therefore, it is essential to incorporate field monitoring efforts, experimental physiological toxicology assessments, and molecular-level mechanistic studies to elucidate how PFAS migrate across tissues and individuals, as well as upward through TLs within food webs[47,48].
To systematically identify and characterize trophic transfer patterns, this review focuses on TMF as the core evaluation index to comprehensively integrate and assess existing research. Specifically, this review focuses on whether different structural types of PFAS, including traditional long-chain and emerging short-chain homologues, and different functional group categories and substitutes exhibit biomagnification potential in aquatic food webs and their spatial variation. This review systematically analyzes how TMFs are affected by compound structural features such as carbon chain length, functional group type, branched and linear isomerism, and the transformation potential of precursor substances. Additionally, the key driving factors at the biological and ecosystem levels, including species' metabolic and excretion capabilities, growth dilution effects, feeding behaviors, habitat characteristics, and food web structure, are evaluated to understand their influence on biomagnification potential.
This review systematically integrates the key drivers determining the biomagnification behavior of PFAS in aquatic food webs, encompassing multidimensional factors such as compound-specific properties, biological metabolic characteristics, and ecosystem environments. Based on mechanistic analysis, it elucidates how these factors drive the differential biomagnification outcomes and provides an in-depth examination of the transformation pathways of precursor compounds as well as the biomagnification of emerging PFAS. This review offers a novel contribution by conducting the first quantitative comparative analysis of PFAS TMFs across diverse aquatic ecosystems, thereby revealing spatial heterogeneity. Furthermore, it emphasizes that PFAS biomagnification in aquatic systems is not an inevitable process but rather a conditional outcome governed by the interplay of specific physicochemical, biological, and ecological factors. This refined understanding serves as an important corrective to current oversimplifications in ecological risk assessment. Through the systematic review and mechanism analysis of TMF-related evidence, this paper aims to provide a unified conceptual framework for understanding the biomagnification of PFAS and lay a scientific foundation for the formulation of future ecological risk assessment and management strategies.
-
The trophic magnification of PFAS in aquatic ecosystems is a result of the interplay between their unique physicochemical properties and the physiological and ecological processes of organisms. This process begins when PFAS enter primary producers and benthic invertebrates from water and sediment, initiating their transfer through the food web. Via predator-prey relationships, PFAS are transferred to higher TLs, with their magnification potential primarily determined by the dynamic balance between uptake efficiency and elimination rates in organisms[49] (Fig. 1).
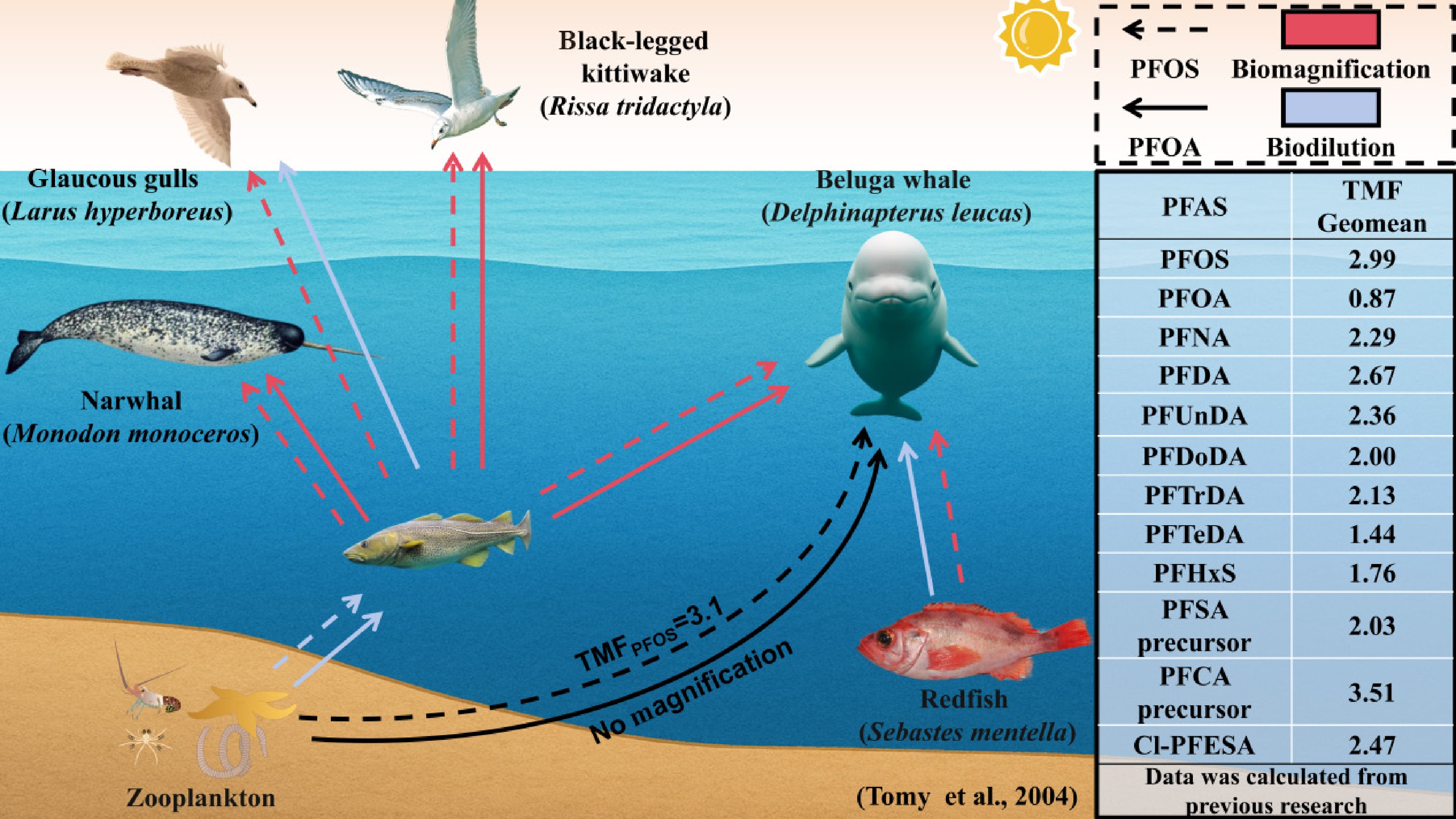
Figure 1.
Biomagnification of PFAS in aquatic ecosystems. The detailed PFAS trophic magnification diagram, adapted from Tomy et al.[30], demonstrates that both PFOS and PFOA exhibit biomagnification in predator-prey relationships. However, only PFOS shows significant biomagnification across the entire food web. The geometric mean (geomean) of the trophic magnification factors (TMFs) for various PFAS is noted, as statistically compiled from previous studies (Supplementary Table S1).
Benthic invertebrates primarily accumulate PFAS from sediment and serve as an essential food source for many fish species, establishing a critical trophic pathway for PFAS transfer from sediments to fish[50]. Dietary intake is the predominant pathway for PFAS accumulation and transfer up TLs in aquatic animals, such as fish. The dietary route of exposure can contribute to 44.9%–76.4% of total PFAS uptake in most fish species[51]. The degree of uptake is influenced by the TL and body composition of the organism. In addition, fish with a higher protein content tend to exhibit more active foraging behavior, leading to a greater dietary PFAS intake[52].
After entering the organism, PFAS are predominantly distributed in protein-rich tissues, such as blood, liver, and kidneys, due to their high affinity for proteins, including serum albumin and fatty acid-binding protein[53]. This strong affinity to proteins is a key factor influencing their capacity to be compartmentalized with an organism or eliminated. In fish, PFAS are eliminated or reduced via gill respiration, fecal excretion, growth dilution, and biotransformation. Among these, gill respiration and fecal excretion represent the two major pathways, whereas growth dilution and biotransformation generally play smaller roles[51]. The elimination pathways and efficiencies of perfluoroalkyl acids (PFAAs) in fish are strongly governed by their chain length and hydrophobicity (LogKow)[51]. Short-chain PFAAs (C < 8), characterized by low molecular weight and weak hydrophobicity, are primarily absorbed and eliminated via gill respiration. In contrast, long-chain PFAAs (typically C9–C12) have a greater binding affinity to proteins, reducing the effectiveness of gill elimination and shifting the dominant elimination route to fecal excretion[33]. The contribution of fecal excretion initially increases with LogKow, peaking at 87.6% for LogKow around 6.5, indicating a higher importance for PFAAs within this mid-hydrophobicity range[51]. There is a significant positive correlation between fecal excretion contribution and fish protein content[49], as high-protein fish consume more food and produce more feces. However, as LogKow increases beyond approximately 6.5, a strong hydrophobicity further reduces the ability for elimination, making growth dilution relatively important in reducing PFAAs. This mechanism is particularly prominent in smaller fish, as there is a marked negative correlation between the contribution of growth dilution and body weight[54].
The combination of high uptake rates and low elimination rates ultimately drives trophic magnification of PFAS. Taking PFOS as an example, it is present at high concentrations in fish diets, leading to substantial dietary intake (405 ng/[kg·d]), in addition to gill uptake (633 ng/[kg·d]). Meanwhile, its elimination rate constant (ke = 0.037 d−1) is remarkably low[51]. This kinetic profile results in a net accumulation rate of PFAS that increases with TL, clearly demonstrating the biomagnification potential of PFOS in aquatic ecosystems.
-
TMFs are calculated using the slope of the relationship between contaminant concentration and TLs from field measurements, and they represent the magnification of the entire food web, as inferred through the following process[29]: (1) Measure the concentrations of pollutant X in various organisms across different TLs within a food chain; (2) Determine the TL of each organism by analyzing the ratio of nitrogen stable isotopes; (3) Assum that the concentration of [X] increases exponentially with TL, so plotting the logarithm of [X] against TL for the relevant segment of the food chain will yield a linear relationship:
$ \log_{\text{10}}[\text{X}]=\mathit{\mathrm{\mathit{a}}}\times\mathrm{TL}+\mathrm{\mathit{b}} $ (1) The TMF is then derived from the slope of the plot:
$ \text{TMF}={\text{10}}^{\text{a}} $ (2) where, [X] represents the concentration of the target PFAS, a represents the intercept, and b represents the slope (both are constants). When TMF is greater than 1, it represents biomagnification, and when TMF is less than 1, it represents biodilution (Fig. 2a).
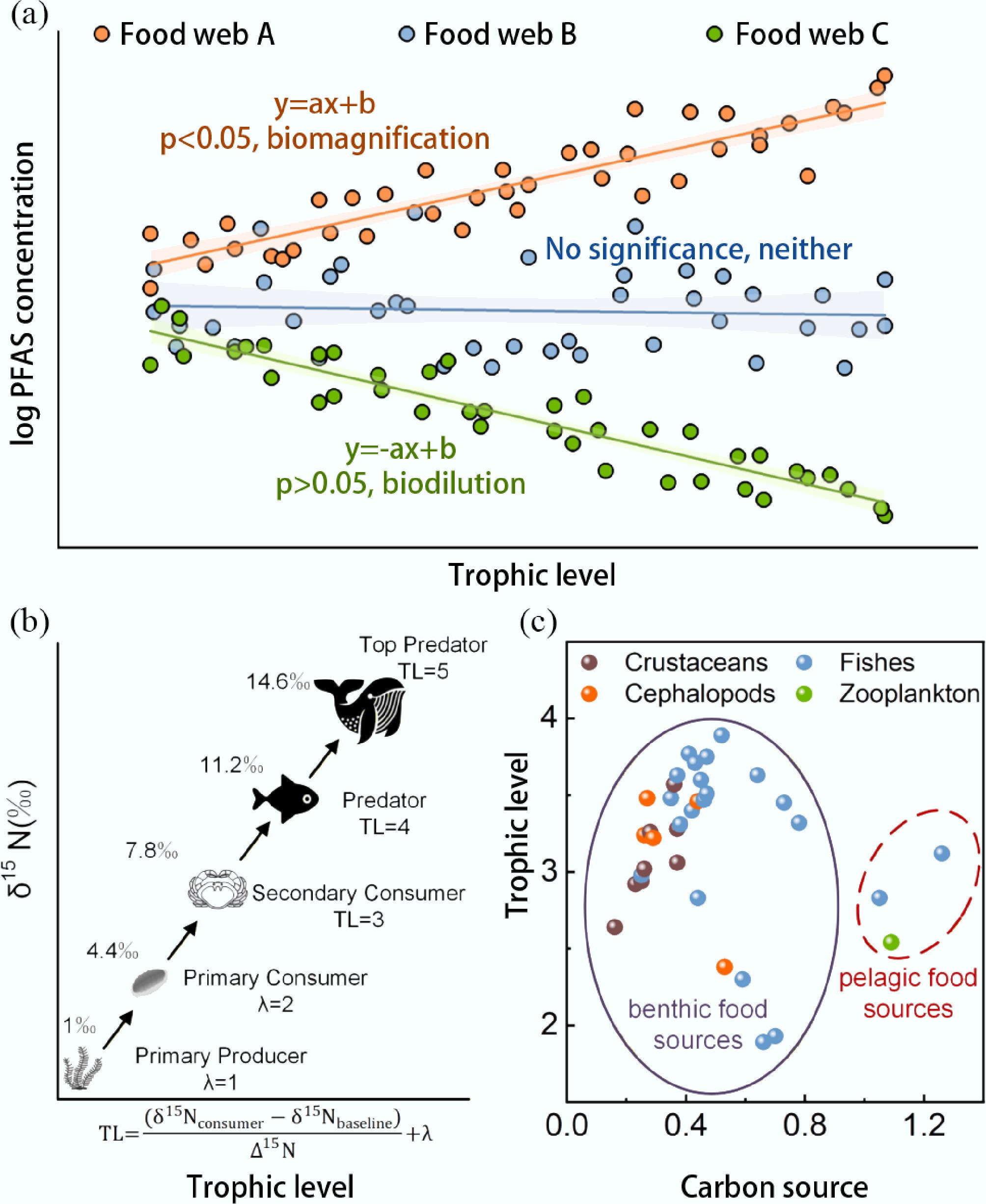
Figure 2.
Integrated assessment of contaminant biomagnification and trophic dynamics. (a) The regression model illustrating PFAS biomagnification, with the slope (a) representing the biomagnification rate and (b) the intercept, according to the equation: Y = aX + b. The shaded areas represent the confidence interval of the fitted line. (b) General trophic positions and bulk delta nitrogen isotope ratios. (c) Carbon sources and trophic levels characterize food web relationships.
The 15N/14N (δ15N) generally reflects a clear and systematic enrichment between predators and prey; the difference between predator and prey is always a constant value (3‰–4‰)[55] and is therefore often used to estimate the TL of consumers[56]. In environmental science, this constant value is conventionally defined as 3.4‰[39]. The calculation formula is as follows:
$ \text{TL}=\dfrac{({\delta}^{\text{15}}{\text{N}}_{\text{consumer}}-{\delta}^{\text{15}}{\text{N}}_{\text{baseline}})}{{\Delta}^{\text{15}}\text{N}}+\lambda $ (3) where, TL is the trophic level; δ15Nconsumer is the nitrogen isotope ratio of consumers; δ15Nbaseline is the nitrogen isotope ratio of baseline species. When the baseline species is the primary producer, λ = 1. When the baseline species is the primary consumer, λ = 2. Δ15N is the fractionation factor, which is the enrichment value of the δ15N in each TL during the material transfer process (Fig. 2b). Nevertheless, it is important to recognize that identifying a definitive baseline organism in complex food webs involves inherent judgment. Variability in baseline selection can propagate into TL estimates and ultimately influence calculated TMFs. To mitigate this, the ecological context and feeding ecology must be considered. Some studies suggest that employing multiple baselines may improve accuracy[57]. Therefore, while commonly standardized, δ15N baseline selection remains a methodological assumption. Future work should explicitly quantify how this choice affects TMFs across systems.
Accurate and reliable identification of the food web is the basis for understanding the biomagnification of contaminants[58]. The 13C/12C (δ13C) shows a slight variation with increasing TL. When the isotopic signatures of carbon sources differ, it can be used to assess the proportion of different nutrient sources for food tracing[55]. Xie et al. distinguished two food webs in subtropical estuaries based on differences in basic food sources, as reflected in δ13C numerical clustering, which indicated variations in basal food sources. They then conducted TMF analysis accordingly[59]. In addition, δ13C can be used to determine the carbon sources of aquatic species and assess whether different species belong to the same food web. This approach considers the contributions of both benthic and pelagic organisms and eliminates the trophic influence using the following formula[38]:
$ \begin{split}& \text{Carbon source}=1-\\ &\dfrac{{\delta}^{\text{13}}{\text{C}}_{\text{zooplankton}}-{\delta}^{\text{13}}{\text{C}}_{\text{consumer}}+\Delta{\delta}^{\text{13}}\text{C}({\text{TL}}_{\text{consumer}}-{\text{TL}}_{\text{zooplankton}})}{{\delta}^{\text{13}}{\text{C}}_{\text{zooplankton}}-{\delta}^{\text{13}}{\text{C}}_{\text{benthic}}} \end{split}$ (4) where, δ13Czooplankton is the carbon isotope ratio of zooplankton and represents the pelagic source. δ13Cconsumer is the carbon isotope ratio of the consumer. δ13Cbenthic is the carbon isotope ratio of benthic species and represents the benthic source. The Δδ13C(TLconsumer−TLzooplankton) term accounts for species occupying varying TLs higher than the sources and thus for δ13C-enrichment between source and consumer depending on the consumer TL relative to the source. Carbon source values close to 0 indicate a high dependence on benthic food sources, while values close to 1 indicate a high dependence on pelagic food sources (Fig. 2c)[39]. A more fundamental yet often implicit limitation stems from the prevalence of mixed feeding strategies in aquatic ecosystems. For organisms with such strategies, their nutritional sources are not singular, which violates the basic linear-model assumption underlying TMF calculations. Although C analysis aids in identifying carbon sources, complex feeding behaviors can still weaken the linear relationship between contaminant concentration and N-derived TL, causing the calculated TMF to deviate from the true biomagnification potential[29,60].
Pan et al. studied the biomagnification of PFAS in a subtropical marine food web, emphasizing the importance of establishing a well-defined food web structure for TMF calculations. They initially found that all species had similar δ13C values, indicating a shared carbon source base among these organisms. To further verify whether these species belonged to the same continuous food chain, they calculated the relative carbon source proportions and ultimately constructed a clear food web structure, except for phytoplankton and Grammoplites scaber. This food web structure served as the basis for the subsequent TMF calculations[61]. In contrast, Fang et al. identified several species with similar carbon sources but did not specify the structure of the food web. In their subsequent analyses, the subjective choice of whether to include invertebrates yielded two distinct TMF results[62]. The comparison above demonstrates that a clear and reasonable food web structure is a prerequisite for ensuring the reliability of TMF assessment results[29,60].
It is also important to note that TMF calculations are based on linear regression analyses, and biomagnification or biodilution effects are only meaningful if the log concentration values of the pollutant and TLs are significantly correlated (p < 0.05)[62,63]. However, this statistical threshold is a necessary but insufficient criterion for robust TMF interpretation. Sole reliance on p-values can be misleading: limited sampling may fail to detect true biomagnification (Type II error), whereas extensive data can render even ecologically negligible trends statistically significant. Therefore, p-values must be evaluated in conjunction with the sample point, its confidence interval, and the coefficient of determination (R2) to distinguish statistical artifact from biologically meaningful amplification. The reliability of TMF regression analysis critically depends on the detection frequency of the target compound. For instance, while Miranda et al. reported biomagnification of L-N-ethyl perfluorooctane sulfonamide (L-EtFOSA) (TMF = 1.8) in an estuarine food web, its detection frequency was only 30%[64]. With over two-thirds of data points imputed, the regression becomes dominated by uniform, non-measured values, distorting the true variance-concentration relationship and severely undermining statistical validity. Therefore, applying a detection frequency threshold such as above 40% to 50% for TMF calculation, as practiced by Gao et al., is essential to ensure robust and comparable conclusions[65].
Furthermore, the biomagnification assessment also has a key limitation regarding tissue-specific differences. Studies have shown that using different tissues as the baseline for concentration measurements can systematically alter the estimated TMFs of PFAS in food webs[62]. For example, a comparison of the ln (PFOS) concentration-TL relationships at two different locations showed that if the PFAS concentrations in top marine mammals are measured in plasma rather than homogenized animal tissues, the TMF derived from the linear regression slope yields higher. In dolphins, the TMF calculated from plasma concentrations was approximately twice that when calculated from whole-body concentrations[66]. In polar food webs, the TMF of PFOS calculated from the homogenized concentrations of beluga and narwhal is approximately threefold lower than that derived from liver concentrations. Moreover, at the compound level, when measured in the liver, the TMF of PFOA is greater than 1, but when calculated using homogenized concentrations, the TMF is less than 1[66]. This evidence consistently suggests that TMFs calculated using plasma or liver concentrations of predators tend to overestimate the biomagnification of PFAS. The methodological root of this bias lies in tissue distribution differences. Compared to lipids, PFOS is more prone to accumulate in the blood and liver. Therefore, the standard lipid normalization methods used for hydrophobic/lipophilic organic pollutants are not applicable to PFOS, and cannot account for the differences in concentration between tissues[30]. When comparing across TLs, if top trophic groups are represented by plasma or liver concentrations, while lower TLs are represented by whole-body or muscle concentrations, the mismatch in concentration baselines based on sample type will systematically raise the TMF estimate. For lower trophic organisms, using whole-body concentrations is more realistic, as they are typically consumed whole by higher TL predators. To reduce systematic bias from tissue mismatches, it is recommended to prioritize whole-body concentrations or body burdens as the unified baseline when estimating TMFs when possible and comparing across studies. If liver or plasma concentrations are used, a comparison with homogenized concentrations should be provided to avoid overestimating the degree of biomagnification.
-
As a representative legacy long-chain PFAS, PFOS has been restricted or banned globally. However, due to its high environmental persistence and potential for long-range transport, the degradation of PFOS precursors and the continuous use of PFOS-containing products result in PFOS remaining a persistent contaminant in aquatic environments[67,68]. Numerous studies have demonstrated that PFOS can exhibit significant bioaccumulation potential in marine organisms, furthering biomagnification through food webs. The mechanism underlying PFOS biomagnification is closely related to its unique molecular structure, as its perfluoroalkyl chain (C8F17−) confers high chemical stability and resistance to biodegradation[69], while its hydrophilic sulfonic acid group (−SO3H) has a strong negative charge and stable dissociation state, enabling PFOS to form high-affinity complexes with carrier proteins such as serum albumin, fatty acid-binding proteins, and liver fatty acid-binding proteins[70,71]. Consequently, PFOS can accumulate in protein-rich tissues, such as the blood and liver, and result in poor elimination through metabolic pathways. However, the TMF of PFOS varies among aquatic ecosystems, and the factors governing its biomagnification potential require further investigation.
Overall, literature data indicate that the geometric mean of the TMF of PFOS in aquatic food webs is 2.99 (range: 0.93–14.26), suggesting a biomagnification potential. The highest TMF value (14.26) was reported by Chen et al. in the offshore South China Sea food web[63], whereas Kelly et al. observed a TMF of 11 in the Arctic marine food web[72], and Li et al. reported a TMF of 10.9 in an oligotrophic river on the Qinghai-Tibet Plateau[73], all of which highlight the biomagnification potential of PFOS. Notably, Penland et al. (2015) reported an overall TMF of 0.93 for PFOS in the Yadkin-Pee Dee River food web in the temperate Atlantic region. This value was derived from a comprehensive food web spanning from primary consumers (the Asiatic clam, serving as the food web baseline) to top predatory fish at TLs from 0.4 to 4.3, encompassing four mollusk families, 12 aquatic insect families and suborders, two crayfish species, and 10 fish species[74]. A TMF < 1.0 indicates biodilution of PFOS within this system, which contrasts with the biomagnification response typically observed across most ecosystems.
The TMF of PFOS tends to differ across ecosystem types and climatic zones. By ecosystem type, marine food webs have a geometric mean TMF of 3.47 (1.57–14.26), which is overall higher than that of freshwater ecosystems, with a geometric mean of 2.77 (0.93–10.90) (Fig. 3a). TMFs are most widely reported in temperate waters, with a geometric mean of 2.97 (0.93–10.90), while polar waters have the highest biomagnification potential with a geometric mean TMF of 4.49 (2.9–11), and subtropical waters exhibiting a geometric mean TMF of 2.85 (1.3–14.26). In contrast, in tropical waters, the geometric mean was 1.53, suggesting a latitudinal gradient in which TMFs progressively decline from high to low latitudes (Fig. 3b). This spatial pattern indicates that temperature, food web structure, and ecosystem productivity may jointly influence the efficiency of PFOS trophic transfer.
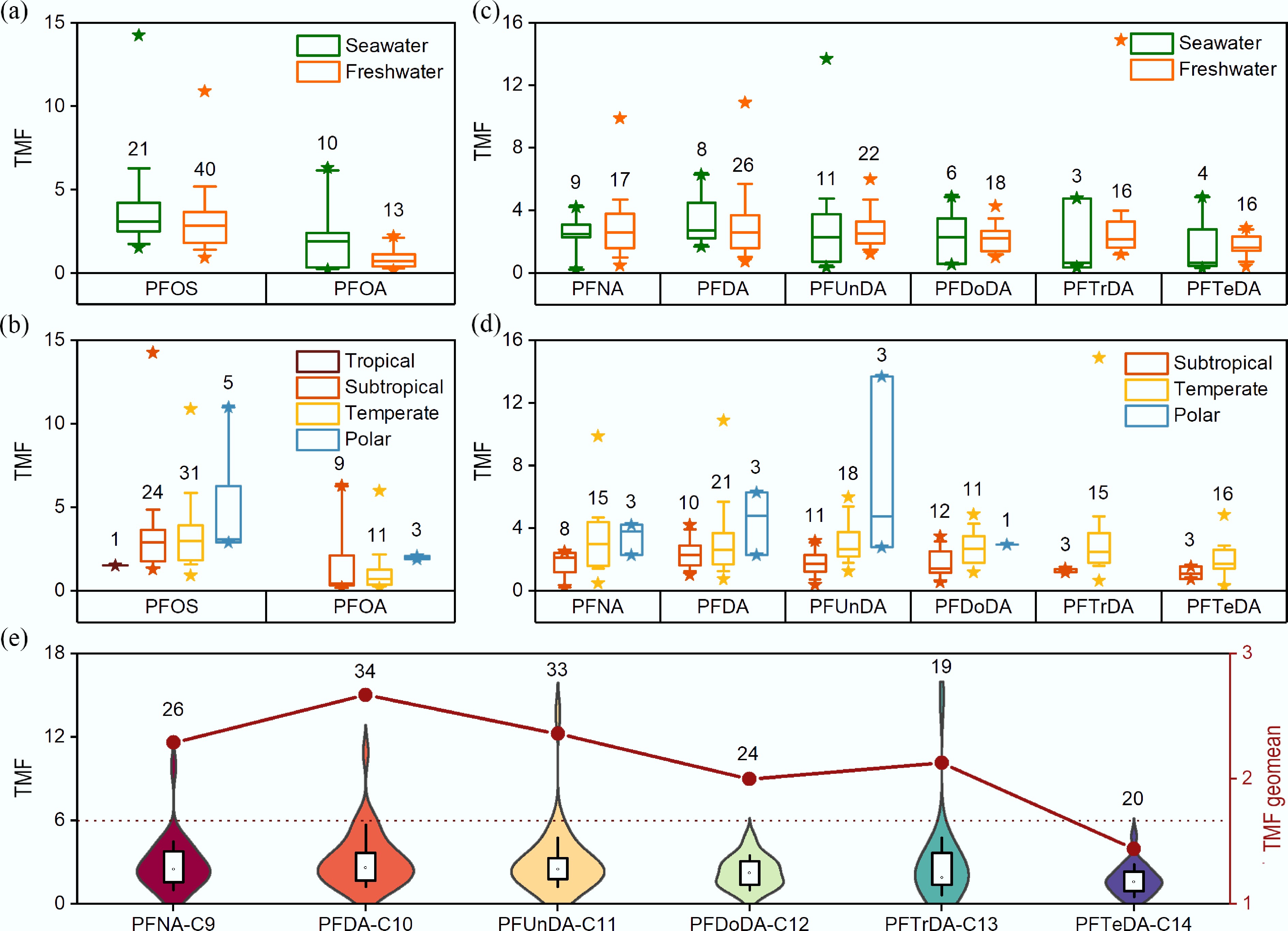
Figure 3.
Trophic magnification factors (TMFs) of per- and polyfluoroalkyl substances (PFAS) in aquatic food webs. (a) Comparison of TMFs for perfluorooctanesulfonic acid (PFOS) and perfluorooctanoic acid (PFOA) in marine versus freshwater ecosystems. (b) Differences in TMFs for PFOS and PFOA across different climate zones. (c) Comparison of TMFs for long-chain perfluoroalkyl carboxylic acids (PFCAs) in marine versus freshwater ecosystems. (d) Differences in TMFs for long-chain PFCAs across different climate zones. The boxplots show the median (horizontal line), 25th–75th percentile (box), and 10th–90th percentile (whisker chart) of TMF, and the asterisk indicates the maximum or minimum values. (e) Variation in TMFs for long-chain PFCAs with increasing carbon chain length. The boxplots show the median (horizontal line), 25th–75th percentile (box), and 10th–90th percentile (whisker chart) of TMFs (left Y-axis). The violin plots represent the data distribution (left Y-axis). The red dotted line represents the geometric mean of TMF, with coordinates on the right Y-axis. The numbers above the bars indicate the sample size (n). Data for this figure were pooled from 33 published studies. Only results with a statistical significance of p < 0.05 were included in the analysis.
Several studies have investigated the differences in TMF among PFOS isomers. Wang et al. calculated TMFs in the northern South China Sea food web as follows: n-PFOS (4.88) > iso-PFOS (4.23) > 5m-PFOS (4.19) > 4m-PFOS (3.49) > 3m-PFOS (3.03)[75]. Similarly, Fang et al. reported that in the Meiliang Bay region of Taihu Lake, the TMF sequence was n-PFOS (3.86) > 3+5m-PFOS (3.35) > 4m-PFOS (3.32) > 1m-PFOS (2.92) > 2m-PFOS (2.67) > iso-PFOS (2.59). This order corresponds well with their elution sequence on a Hydrophobic Interaction Chromatography column, suggesting that hydrophobicity may be an essential factor influencing the TMFs of PFOS isomers[62].
Houde et al. reported that branched isomers (Br-PFOS) are eliminated more rapidly than linear isomers (L-PFOS), with the latter exhibiting a greater biomagnification potential. In the Lake Ontario food web, the TMF of L-PFOS (3.9) was higher than that of Br-PFOS (2.5), indicating a higher biomagnification efficiency for the linear isomer[76]. However, contrasting findings have also been reported. For example, Simmonet-Laprade et al. noted that the relative biomagnification of Br-PFOS and L-PFOS was inconsistent in four river systems of southeastern France (Rhône, Bourbre, Furan, and Luynes). The geometric mean TMF across five sampling sites was higher for Br-PFOS (2.84) than for L-PFOS (2.37)[67]. Similar trends were observed in the Orge River near Paris with Br-PFOS (1.8) > L-PFOS (1.6)[77], and the Gironde River in southwestern France with Br-PFOS (3.9) > L-PFOS (2.5)[78]. Overall, the biomagnification of PFOS isomers does not follow a fixed pattern and is likely influenced by multiple factors, including hydrological conditions, food web composition, and species-specific metabolic traits. Consequently, comparisons of isomer-specific biomagnification and ecological risk should be interpreted with caution and in the context of specific environmental conditions.
Another legacy long-chain PFAS, PFOA, is also widely detected in aquatic environments, posing potential ecological and health risks[42]. Unlike PFOS, most studies report that PFOA tends to exhibit biodilution in aquatic food webs, with TMFs typically below 1. The molecular basis for this difference lies predominantly in functional groups, as the carboxylic acid group (−COOH) of PFOA is less negatively charged and shows pKa-dependent dissociation compared with the sulfonate group (−SO3−) of PFOS. This results in weaker binding affinity to carrier proteins, such as serum albumin, which promotes renal elimination and reduces tissue retention[79]. Additionally, the shorter biological half-life and higher water solubility of PFOA facilitate its entry into organisms primarily via direct aqueous exposure rather than trophic transfer.
The geometric mean TMF of PFOA in aquatic food webs is 0.87 (0.18–6.30). The lowest value (0.18) was reported by Ren et al. in the temperate freshwater lake, Lake Erie, food web[80]; Du et al. reported a TMF of 0.2 in the South China Sea marine food web[28], and Pan et al. found a TMF of 0.354 in the Beibu Gulf marine food web[61], all indicating biodilution trends. Nevertheless, PFOA trophic transfer shows ecosystem-specific heterogeneity. Zhu et al. reported TMFs of 2.4 and 1.9 in the South China Sea and Southern Ocean marine food webs, respectively[81], while Munoz et al. found a TMF of 6.0 in the estuarine food web of the Gironde River in southwestern France[78]. These findings suggest that under certain ecological conditions, PFOA may also exhibit a potential for biomagnification.
Across aquatic ecosystem types, PFOA TMFs in marine food webs, with a geometric mean of 1.22 (0.2–6.3), are slightly higher than those in freshwater systems, which range from 0.18 to 2.2, with a geometric mean of 0.68 (Fig. 3a). Consistent with the spatial distribution pattern of PFOS, PFOA TMFs show a negative correlation with temperature across climatic zones. The highest values occur in polar waters between 1.9 and 2.1, with a geometric mean of 1.97, representing the only climatic region with a relatively stable biomagnification. Temperate and subtropical waters have geometric means of TMFs of 0.75 (0.18–2.2) and 0.81 (0.2–6.3), respectively, while tropical waters exhibit the lowest TMF of 0.2, indicating an overall biodilution effect (Fig. 3b).
Long-chain perfluoroalkyl carboxylic acids
-
Long-chain perfluoroalkyl carboxylic acids (PFCAs) typically refer to carbon chain lengths of nine or more carbons. The long-chain PFCAs that are frequently detected include PFNA (C9), PFDA (C10), perfluoroundecanoic acid (PFUnDA, C11), perfluorododecanoic acid (PFDoDA, C12), perfluorotridecanoic acid (PFTrDA, C13), and perfluorotetradecanoic acid (PFTeDA, C14). As representative legacy long-chain PFAS, these compounds have been widely used in industrial and commercial applications since the 1950s. However, compared with PFOA and PFOS, global regulatory actions to restrict long-chain PFCAs were implemented later. This gap between use and regulation has led to the historical accumulation of long-chain PFCAs in aquatic environments worldwide[82]. Ren et al. reported that the concentrations of long-chain PFCAs in organisms from the Lake Ontario food web were considerably higher than those of short-chain PFAS[83], suggesting the necessity of treating long-chain PFCAs as a distinct research category.
A multitude of studies have demonstrated that long-chain PFCAs generally exhibit a biomagnification potential. In the Lake Ontario food web, Ren et al. calculated TMFs of 2.6, 3.7, 3.1, 2.7, 2.7, and 2.7 for PFNA, PFDA, PFUnDA, PFDoDA, PFTrDA, and PFTeDA, respectively, all of which exceeded 1[83]. Similarly, Tomy et al. reported TMFs of 3.8, 6.3, and 13.7 for PFNA, PFDA, and PFUnDA, respectively, in the western Canadian Arctic marine food web based on marine mammal and mesopelagic fish liver samples[84]. Conversely, some studies have observed biodilution of long-chain PFCAs. For example, Pan et al. reported TMFs of 0.2455, 0.7499, 0.5741, and 0.7586 for PFNA, PFUnDA, PFDoDA, and PFTeDA, respectively, in the subtropical Beibu Gulf marine food web of southern China[61]. The TMF of long-chain PFCAs did not exhibit a pattern similar to that of PFOS and PFOA in seawater or freshwater (Fig. 3c).
Current trends from existing research reveal a distinct latitudinal gradient in the biomagnification of long-chain PFCAs, as TMF values tend to decrease with increasing temperature (Fig. 3d). Taking PFUnDA as an example, geometric mean TMFs were highest in polar food webs (5.69), followed by temperate (2.89), subtropical (1.57), and tropical (0.41) systems. Other long-chain PFCAs exhibit similar patterns, suggesting that temperature may be a crucial environmental factor influencing trophic transfer efficiency.
Several studies have reported that the biomagnification potential of long-chain PFCAs does not increase monotonically relative to carbon chain length, but instead follows an 'inverted U-shaped' trend, in which the bioaccumulation potential first increases and then decreases. For instance, in the Lake Erie food web, TMFs for PFNA, PFDA, PFUnDA, PFDoDA, PFTrDA, and PFTeDA were 0.89, 1.10, 2.04, 1.47, 1.65, and 1.55, respectively[80]. Similarly, Ren et al. reported corresponding TMFs of 4.7, 5.3, 5.4, 3.5, 3.7, and 2.6 in the Port of Austin food web in the United States[85]. Across the studies compiled in this review, the TMFs' geometric mean (range) for PFNA was 2.29 (0.25–9.9), PFDA was 2.67 (0.75–10.9), PFUnDA was 2.36 (0.40–13.7), PFDoDA was 2.00 (0.57–4.88), PFTrDA was 2.13 (0.39–14.9), and PFTeDA was 1.44 (0.33–4.87) (Fig. 3e). Overall, this trend is consistent with the studies referred to above.
Emerging PFAS
-
With the increasingly stringent regulation of legacy PFAS, the production and detection of emerging PFAS, including perfluoroalkyl ether sulfonic acids (PFESAs), perfluoroalkyl ether carboxylic acids (PFECAs), cyclic PFAS, and ultrashort-chain PFAS, have markedly increased. This section will systematically review these emerging PFAS by chemical structural class and delve into the underlying mechanisms of their biomagnification potential.
As a major industrial alternative to PFOS, 6:2 Cl-PFESA (commercially known as F-53B) has been widely detected in aquatic ecosystems and exhibits significant biomagnification potential. Its TMF generally exceeds 1, ranging from 1.82 to 3.37 in temperate marine food webs and from 0.47 to 4.76 in subtropical marine environments[39,61,63,75,81,86,87]. Studies suggest that 6:2 Cl-PFESA may possess an even greater biomagnification potential than PFOS, which could be attributed to the chlorine atom in its hydrophobic tail facilitating stronger electrostatic interactions with proteins, enhancing the binding affinity, and reducing renal clearance[88]. The relatively lower biomagnification factor observed for 6:2 H-PFESA in the same studies further supports this mechanism[75]. Notably, the TMF for its homologue, 8:2 Cl-PFESA, has reached as high as 6.44[75], likely due to the extended carbon chain conferring a stronger protein-binding capacity[89]. Current evidence indicates that the introduction of an ether linkage into the perfluoroalkyl chain does not appear to substantially alter the fundamental toxicological mode of action[90].
The biomagnification potential of PFECAs varies dramatically among different compounds, highlighting a high degree of structural specificity. This is particularly evident in the comparison between hexafluoropropylene oxide tetrameric acid (HFPO-TeA) and 4,8-dioxa-3H-perfluorononanoic acid (ADONA). HFPO-TeA exhibits extremely high TMFs, ranging from 5.95 to 11.7, even surpassing those of legacy PFOS[91,92]. The underlying mechanism may involve its additional ether bond, which increases molecular size and potentially hinders elimination. Concurrently, greater hydrophobicity may enhance its binding affinity to specific proteins, ultimately elevating its bioaccumulative potential[93]. In stark contrast, although computational models predict that ADONA has a high binding affinity to liver fatty acid-binding protein[94], experimental observations reveal a pattern of biodilution (TMF = 0.58)[59]. The structural drivers behind this discrepancy remain unresolved. In summary, the biomagnification behavior of PFECAs is not a class-wide characteristic. Their specific environmental fate, bioaccumulative potential, and associated health risks require further in-depth investigation.
Perfluoroethylcyclohexane sulfonate (PFECHS), an emerging cyclic PFAS and a substitute for PFOS, has demonstrated a biomagnification potential in subtropical freshwater food webs[95], though it is generally considered lower than that of PFOS. This may be attributed to its relatively lower hydrophobicity and more efficient elimination pathways[96].
Ultrashort-chain PFAS are attracting increasing attention due to their high environmental mobility and potential toxicity, yet they exhibit minimal biomagnification potential. For instance, in temperate marine food webs, perfluoro-2-methoxyacetic acid (PFMOAA) exhibits TMFs in the low range of 0.03 to 0.26[91,92], indicating rapid metabolic clearance and very low bioaccumulation potential. This aligns with its physicochemical properties, including short chain length and high water solubility, which facilitate rapid renal or metabolic elimination, thereby effectively preventing tissue retention and trophic transfer[97].
Overall, a variety of emerging PFAS and PFAS alternatives are continuously being identified. Differences in molecular structure, environmental matrices, and metabolic stability contribute to increasingly complex behaviors within aquatic ecosystems. Current evidence indicates that typical alternatives, such as Cl-PFESAs, PFECAs, and cyclic PFAS (e.g., PFECHS), generally exhibit significant biomagnification effects.
Precursors
-
In addition to emerging PFAS, PFAS precursors, which can continuously release terminal perfluorinated carboxylic acids (PFCAs) and perfluorinated sulfonic acids (PFSAs) through biotic and abiotic processes, can also exhibit magnification in aquatic ecosystems. As a known precursor of PFOS, perfluorooctane sulfonamide (FOSA) has shown significant biomagnification potential (TMF > 1) in numerous studies. In temperate river food webs, TMF values range from 1.3 to 5.9[67, 77]. However, an exception was reported in Lake Ontario, Canada, where a TMF as low as 0.51 was observed[98], suggesting that trophic magnification of FOSA may be influenced by biotransformation and environmental conditions. Substantial interspecies differences have also been observed, with TMFs of 5.0 and 4.46 reported in the food webs of bottlenose dolphins (Tursiops truncatus) and beluga whales (Delphinapterus leucas), respectively[66,72], indicating pronounced biomagnification in mammal-dominated systems where uptake and storage rates exceed elimination. Similarly, TMF > 1 has been observed in liver-based food webs of beluga[84]. Beyond FOSA, other PFOS precursors exhibit distinct magnification behaviors. For example, N-methyl perfluorooctane sulfonamidoacetic acid (MeFOSAA) exhibited TMFs ranging from 1.5 to 2.3 in temperate freshwater food webs, indicating its potential for biomagnification[67,77]. Furthermore, the TMF of N-ethyl perfluorooctane sulfonamide (N-EtFOSAA) varies across aquatic environments, tending to be higher in seawater (2.93) than in freshwater (1.5) systems[75,95].
Precursors of PFCAs also exhibited biomagnification in aquatic ecosystems, with 7:3 fluorotelomer carboxylic acid (FHpPA) exhibiting a TMF of 5.82 in subtropical marine food webs[75]. In addition, fluorotelomer sulfonic acids (n:2 FTSAs, n = 6, 8, 10) displayed variable magnification behaviors. Previous studies reported that 10:2 FTSA (4.15) showed a higher TMF than 8:2 FTSA (2.95)[75], while the TMF of 6:2 FTSA reached as high as 6.76 in Tibetan Plateau food web[73], reflecting differential biomagnification potential.
-
Despite trophic magnification being a central concept in assessing the ecological risk of persistent organic pollutants, it must be emphasized that biomagnification is not an inevitable outcome for PFAS transfer within food webs. Even for typical PFAS such as PFOS, which have been widely demonstrated to possess a strong biomagnification potential, they may still exhibit biodilution phenomena in specific food webs. For instance, research by Penland et al. in the Yadkin-Pee Dee River food web found a TMF value of 0.93 for PFOS, indicating a trend of biodilution[74]. This key case demonstrates that the trophic transfer fate of PFAS (magnification or dilution) is not solely an intrinsic property determined by their physicochemical characteristics, but rather the outcome of the interaction between these properties and the structural features of specific ecosystems, environmental conditions, and physiological processes of organisms. Therefore, understanding the behavior of PFAS in food webs must move beyond simplistic attribution based solely on compound-specific properties, and instead systematically examine the multiple interacting factors that drive the direction of their trophic transfer. The following sections will elaborate on these key influencing factors one by one.
Chemical factors
Substance properties
-
The length of the perfluorinated alkyl chain can influence the biomagnification of PFAS in aquatic ecosystems[91]. This impact exhibits a clear 'inverted U-shaped' relationship, which arises from a fundamental shift in the dominance of two core mechanisms governing biomagnification potential: protein-binding affinity and molecular steric hindrance. Specifically, for PFCAs with chain lengths shorter than approximately ten carbon atoms (e.g., C8–C10), biomagnification potential increases with chain length. This ascending phase is primarily driven by enhanced protein-binding affinity. Longer perfluoroalkyl chains form stronger hydrophobic interactions and specific bonds with key transport and storage proteins in organisms, such as liver fatty acid-binding proteins and serum albumin[89]. For example, in the food webs of Chaohu Lake, long-chain PFAAs demonstrated significant trophic magnification, whereas short-chain homologs mostly exhibited biodilution or no significant magnification[69]. This enhancement in binding affinity reaches saturation at approximately C10/C11. Beyond this critical chain length (e.g., C11–C14 PFCAs), despite the continued increase in the compound's hydrophobicity, its biomagnification potential declines instead of rising. Research on the Lake Huron food web supports this: the TMFs of C8–C10 PFCAs increased with carbon chain length. In contrast, those of C11–C14 PFCAs decreased[69]. The determining factor in this descending phase shifts from binding affinity to molecular steric hindrance[99]. Excessively long perfluoroalkyl chains result in a larger molecular volume, which can hinder passive diffusion across cellular membranes[85,100]. Further supporting this phenomenon is the observation that when the perfluoroalkyl chains exceed 11 carbon atoms, the binding affinity of PFCAs to human serum albumin also decreases[101]. This nonlinear relationship between protein binding affinity and perfluoroalkyl chain length may help explain the observed trophic dynamics. Therefore, the entire 'inverted U-shaped' curve essentially represents a trade-off between bioaccumulation potential driven by protein binding and uptake and transport limitations imposed by molecular size. This also supports the view that hydrophobicity alone does not govern the trophic transfer of PFAS from lower to higher TLs[85,100]. This property difference is progressively amplified through predation and trophic transfer along the food chain, ultimately resulting in the significant distinction in bioaccumulation between short- and long-chain PFAS in aquatic organisms, thereby providing a mechanistic explanation for their markedly different biomagnification behaviors.
Distinct isomers of PFAS, namely linear and branched forms, exhibit different biomagnification potentials due to variations in molecular structure, hydrophobicity, and persistence. Linear PFAS isomers generally demonstrate a stronger biomagnification capacity than their branched counterparts[102], likely due to the preferential interactions of linear isomers with cell membranes[62,75,76]. In a study of the Lake Ontario food chain, Houde et al. observed a selective retention of linear PFOS during trophic transfer, with its TMF being higher than that of branched isomers. For instance, during transfer from benthic organisms, such as Mysis, to fish like sculpin, the biomagnification factor of linear PFOS isomers reached 3.9, whereas that of a monobranched isomer was only 2.8[76]. Research by Gui et al. on Indo-Pacific humpback dolphins in the Pearl River Estuary revealed differential bioaccumulation capacities among PFSA isomers, with linear PFOS exhibiting a greater bioaccumulation potential compared to branched isomers[103]. However, a key counterexample was provided by Simmonet-Laprade et al. in a riverine food web in southeastern France, where branched PFOS demonstrated a higher biomagnification potential than its linear isomer at specific sampling sites, indicating that the biomagnification potential of linear PFAS is not perpetually superior to that of branched isomers under all environmental conditions[67,104].
The functional group type of PFAS influences their biomagnification behavior in aquatic ecosystems by modulating molecular polarity, charge distribution, and interaction modes with biological macromolecules. Typically, −SO3H confers the most substantial biomagnification potential, followed by carboxylic acid groups, while specific emerging alternatives with distinct functional groups exhibit minimal effects. For instance, Zeng et al. demonstrated in a zebrafish bioaccumulation study that sulfonate-based PFOS was accumulated at relatively high levels in fish organs during exposure, whereas the carboxylate-based PFOA generally displayed a lower biomagnification potential than PFOS[105]. Similarly, Fang et al. reported that the type of PFAS functional group regulates biomagnification effects in aquatic ecosystems by altering the binding affinity to biomacromolecules. Specifically, −SO3H imparts a stronger protein-binding capability to PFAS molecules than −COOH, as evidenced in the Taihu Lake food web, where PFOS exhibited a significant biomagnification (TMF = 3.74). At the same time, PFOA with an identical carbon chain length showed biomagnification (TMF = 2.13)[62,106]. Furthermore, Farrell et al. observed that the functional group affects the hydrophobicity of PFAS, protein-binding ability, and metabolic resistance, thereby directly determining the bioaccumulation potential and trophic magnification trends in food chains. For example, in their aquatic food chain investigation, PFOS with −SO3H demonstrated remarkably high accumulation patterns in mayflies, with enrichment levels substantially surpassing those of other PFAS, indicating that the sulfonic acid group may potentiate PFAS-protein binding in organisms, resulting in selective retention and amplification during trophic transfer[71].
Precursor transformation
-
Furthermore, the biomagnification of PFAS is partially modulated by the biotransformation of precursor compounds. Taking FOSA as an example, an important precursor to PFOS, its environmental transport and transformation can indirectly enhance the biomagnification potential of PFOS. Research by Brandsma et al. indicated that the in vivo transformation of PFAS precursors amplifies the biomagnification capacity of their terminal forms in aquatic ecosystems, as precursor compounds are subsequently metabolized into persistent terminal PFAS that exhibit reduced elimination efficiency. For instance, rainbow trout (Oncorhynchus mykiss) fed 8:2 fluorotelomer alcohol (8:2 FTOH) metabolize it into PFOA and other PFCA[107]. Similarly, FOSA is biotransformed into PFOS in rainbow trout. Comparable metabolic pathways have been corroborated by Yeung et al. These in vivo conversion processes lead to the continuous accumulation of terminal PFAS, such as PFOA and PFOS, in fish tissues. Due to their limited elimination capacity, they exhibit an enhanced potential for trophic transfer and magnification along the food chain[108]. Consequently, precursors like FOSA can function as cryptic sources of PFOS in food webs, thereby elevating the TMF of PFOS[64,77]. This mechanism is substantiated by the frequent observation of biodilution for FOSA itself in food webs; its decreasing concentration with increasing TL signifies ongoing conversion to PFOS. An analogous mechanism is observed in Arctic marine food webs, where precursors such as FOSA and EtFOSA undergo biotransformation to yield terminal PFOS, markedly strengthening the biomagnification potential of PFOS from fish to top predators like seals[30]. Studies by Simonnet-Laprade et al. further demonstrate that in aquatic environments, (alkyl-) FASAs and (alkyl-) FASAAs can be transformed into PFOS, while FTSAs and polyfluoroalkyl phosphoric acid diesters are converted into PFCAs with similar or shorter carbon chain lengths. These transformation processes collectively intensify the biomagnification of PFOS and PFCAs in food webs[77]. Notably, even if direct emissions of PFOS are controlled, the continual transformation of precursors may sustain PFOS as a long-term source. Retrospective analysis by Gui et al. on Indo-Pacific humpback dolphins in the Pearl River Estuary revealed that, although concentrations of certain PFCA declined between 2004 and 2016, PFOS levels exhibited an increasing trend, likely attributable to ongoing environmental degradation or the metabolism of other PFAS precursors into PFOS. This phenomenon suggests that precursor transformation not only maintains the ecological reservoir of PFOS but may also contribute to its persistent accumulation in top predators, such as Indo-Pacific humpback dolphins, thereby further amplifying its biomagnification potential within the food chain[103].
PFAS mixture interactions
-
In real-world environments, aquatic organisms are continuously exposed to mixtures of PFAS with varying chain lengths and functional groups. Recent studies have revealed that PFAS components do not act independently but rather exhibit competitive interactions, which are crucial for understanding their actual bioaccumulation and trophic magnification behaviors.
Experimental research has shown that long-chain PFAS can effectively inhibit the bioaccumulation of short-chain PFAS in fish tissues due to the former's stronger hydrophobicity and higher affinity for protein-binding sites. The underlying mechanisms primarily involve two aspects. First, competition for transport proteins. In the presence of long-chain PFAS, they compete with short-chain PFAAs for transporters, thereby inhibiting the uptake and elimination of short-chain PFAAs. This leads to a decrease in the uptake and elimination rate constants of short-chain PFAAs, with the reduction in uptake rate being far greater than that in elimination rate[109]. Second, competition for protein-binding sites. For example, Wen et al. demonstrated that the binding affinity of PFAS to fish liver fatty acid-binding proteins increases with fluorocarbon chain length. Under mixed exposure, long-chain PFAS preferentially occupy limited protein-binding sites, displacing short-chain PFAS from these sites. This competitive effect leads to the fractionation of short-chain PFAS during bioaccumulation, meaning that the proportion of long-chain PFAS in organisms increases with their environmental concentration, while that of short-chain PFAS decreases accordingly[89]. More importantly, this fractionation effect is progressively amplified along the food chain. As trophic levels increase, predators assimilate an ever-greater proportion of long-chain PFAS from their prey, while short-chain PFAS are further excluded due to competition, resulting in a decrease in their relative proportion in organisms along the food chain[89].
Biological factors
Food web structure
-
Habitat constraints can lead to diverse dietary structures among organisms, enabling the same species to occupy different TLs across distinct environments and food webs. Differences in food web structure, particularly food chain length and TL variations, can result in substantially divergent biomagnification behaviors[58]. Generally, longer food chains provide more tiers for the trophic transfer of PFAS, potentially leading to higher degrees of contaminant accumulation in top predators. As demonstrated in a study of the Arctic marine food web, with a long chain extending from macroalgae (TL: 1.0) to polar bears (TL: 5.5), PFOS exhibited a notably high TMF of 17.4, illustrating the enhanced biomagnification capacity associated with elongated food chains for PFAS[72]. Fang et al. reported significantly higher concentrations of long-chain PFAS, such as PFOS, in high TL organisms, specifically top piscivorous fish like mandarin fish, compared to lower TL organisms, indicating an apparent biomagnification effect[62]. However, in the same study, results for PFTrDA and PFTeDA contrasted with those of EtFOSA, suggesting that biomagnification potential does not invariably increase with TL. The factors governing these patterns remain unclear, and differential metabolism of PFAS among species may contribute to explaining this phenomenon[64]. Similarly, when Miranda et al. calculated the TMF of EtFOSA after grouping organisms by TL, biomagnification was observed in the lower TL group (TLs: 2.00–3.22) composed mainly of filter-feeding organisms, whereas biodilution occurred in the second group (TLs: 3.56–4.49) dominated by omnivorous organisms, indicating dilution of the chemical in top predators, potentially due to more efficient metabolism and excretion in higher TL species[64]. This aligns with findings by Diao et al., who reported that PFAS biomagnification was not pronounced in higher-TL species, except in the case of the catfish (Clarias macrocephalus) that preys on the bearded goby (Trypauchen vagina). The underlying reason may be that biomagnification in high TL organisms is a complex process influenced by interspecific variability and intricate dietary patterns[39].
The inclusion of benthic organisms in the food web and the presence of mammalian top predators also modulate the degree of PFAS biomagnification in aquatic ecosystems. Specifically, benthic organisms serve as a critical pathway in which PFAS are taken up from sediments and incorporated into marine food chains. For instance, in the Xiaoqing River Estuary, benthic invertebrates such as snails and mussels directly absorb and accumulate PFAS from sediments, with PFOA and PFMOAA occurring at relatively high concentrations[91]. When fish consume these benthic organisms, contaminants are transferred upward via the benthic-pelagic coupling food web, significantly enhancing the biomagnification potential of PFAS in high TL organisms[91]. Moreover, mammals, which lack efficient gill-based excretion pathways, exhibit strong retention and accumulation capacities for long-chain PFAS, resulting in exceptionally high trophic magnification factors observed in these longer food chains. As such, multiple studies in Chinese coastal and estuarine systems have reported that PFOS concentrations increase from lower to higher trophic levels, with notably elevated levels in mammalian species, such as the Indo-Pacific humpback dolphin, compared to organisms from other lower levels[110].
Bioabsorption and metabolic capacity
-
The metabolic capacity of aquatic organisms, particularly the differences in the efficiency of respiratory excretion pathways, plays a regulatory role in the biomagnification potential of PFAS in marine ecosystems. The fundamental distinction between aquatic mammals and fish in terms of respiratory excretion efficiency is particularly critical. Research by Liang et al.[51] demonstrated that the gill structure of fish plays an important role in eliminating PFAS, although its efficiency is strongly dependent on the physicochemical properties of the compounds. Specifically, the contribution of gill respiration to PFAS elimination decreases with increasing Kow[51]. When PFAS chain lengths exceed eight carbon atoms, the elimination contribution rate via gill respiration drops sharply to merely 0.82%[51]. This indicates that gill respiration primarily functions in the elimination of short-chain PFCAs, while its contribution to long-chain homologues is minimal. The biomagnification potential of PFAS largely hinges on metabolic and elimination efficiency within organisms. Short-chain PFCAs are typically rapidly cleared via efficient biotransformation, such as conjugation with glutathione, and active excretion pathways, resulting in limited accumulation in fish. In contrast, long-chain PFAS, such as PFOS and PFCAs with ≥ 8 carbons, resist biotransformation due to their molecular structure and rely on less efficient pathways, including fecal excretion, leading to significantly prolonged half-lives in organisms, influencing the necessary conditions for bioaccumulation to occur[48]. However, in stark contrast to fish, aquatic mammals such as cetaceans, being air-breathing top predators, lack similarly efficient aquatic excretion pathways, resulting in the propensity for long-chain PFAS to accumulate extensively in their bodies. This largely explains the extreme phenomenon observed in long food chains containing high TL organisms, such as that which occurs in the Arctic marine food web, which extends from algae to polar bears, where PFOS exhibits a trophic magnification factor as high as 17.4[78]. Furthermore, metabolic transformation capacity can profoundly influence the ultimate fate of PFAS; for instance, the precursor FOSA can be metabolically converted to PFOS in fish. Although FOSA itself may exhibit a dilution trend during trophic transfer, its conversion process indirectly elevates the apparent TMF of PFOS. Similarly, Gui et al.'s study on Indo-Pacific humpback dolphins in the Pearl River Estuary found that PFOS concentrations in dolphins far exceeded those in their fish prey, attributed to the distinct physiological structures and metabolic clearance efficiencies between the two groups. Fish possess relatively effective passive diffusion and ion exchange across the gill epithelium, providing a relatively rapid excretion route for PFOS. In contrast, humpback dolphins, as mammals, rely entirely on the hepatorenal system for excretion. This lack of excretion pathway, coupled with insufficient metabolic transformation capacity, results in a substantially extended half-life of PFOS in dolphins, thereby generating a strong biomagnification effect during trophic transfer from fish to dolphins, where dolphins are subsequently bioaccumulating more PFOS[103].
Environmental factors
-
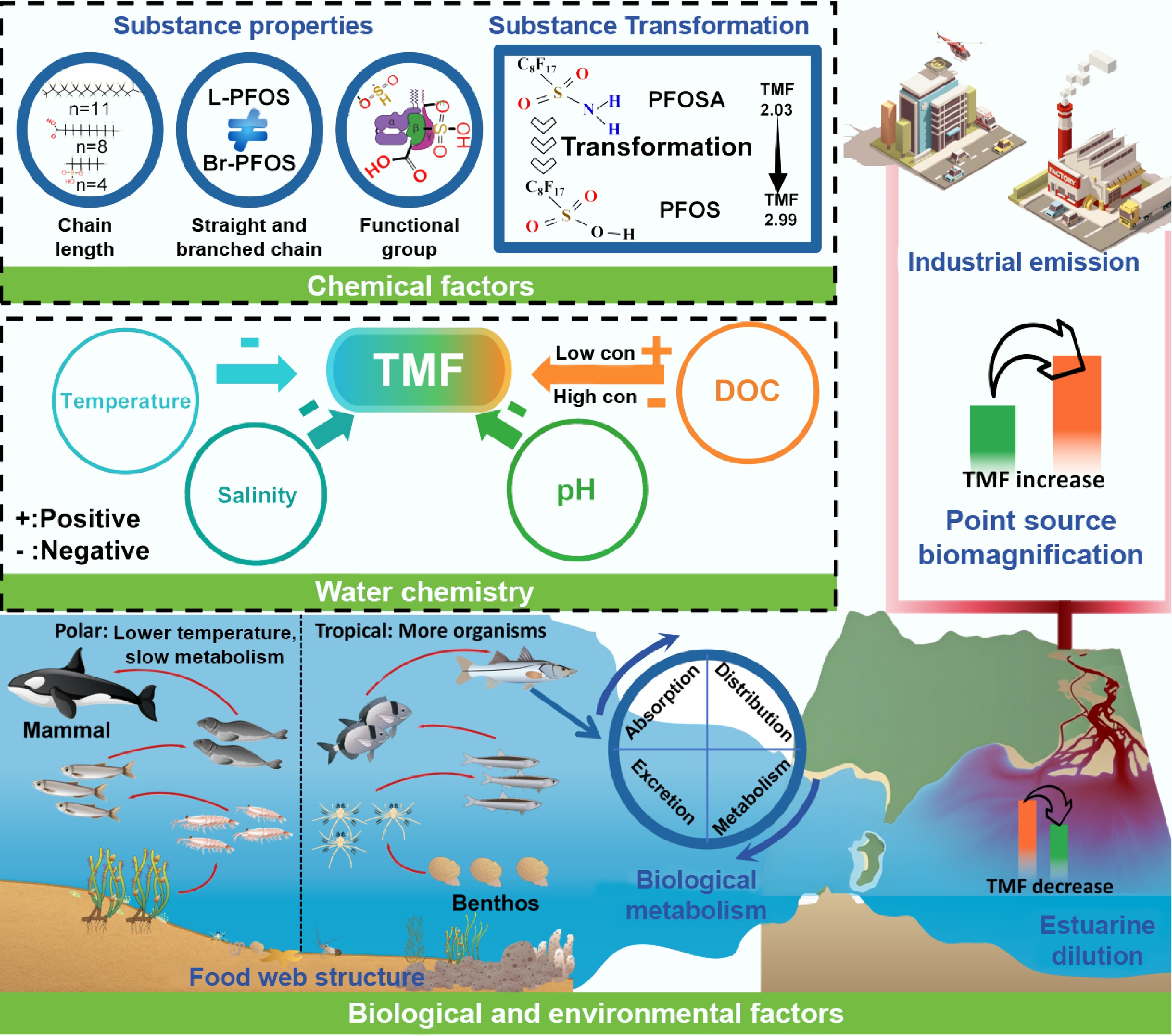
In ecosystems influenced by diverse environmental factors, identical PFAS can exhibit different behavior. When comparing different aquatic ecosystems, it is essential to consider geographic factors. The first of these is temperature. Temperature is one of the primary drivers affecting PFAS biomagnification. Research indicates that higher water temperatures typically enhance the metabolic rate and excretion abilities of aquatic organisms, thus accelerating the absorption and elimination of PFAS[50]. This can lead to lower TMF values in tropical or subtropical regions. In colder areas, however, organisms have slower metabolic and growth rates, as well as higher energy demands, resulting in increased accumulation of PFAS in their bodies. Consequently, higher TMF values are typically observed in polar or high latitude food webs[61]. For example, the TMF of PFOS in the northern polar food web can reach 2.92, whereas in subtropical marine food webs, it is only 1.75[61,65]. According to Wang et al., the concentrations of 13 PFAS in zooplankton and zebrafish increased with rising temperature in laboratory conditions[111]. Additionally, Vidal et al. demonstrated that reduced water temperature led to an increased PFAS burden in rainbow trout[112]. In addition to the physiological effects of temperature, differences in temperature zones may lead to a varying composition of the food web, contributing to TMF differences. The food webs of tropical aquatic ecosystems typically contain more organisms, which may dilute the energy flows between them and therefore dilute pollutants[29]. Spatial differences in habitat also contribute to TMF variability. Xie et al. showed that trophic transfer and bioaccumulation of PFAS in estuarine organisms were masked by a gradient dilution of PFAS in their habitats, and the effect of biotic dilution was more pronounced in species with broader swimming ranges[59]. Additionally, the spatial distribution of pollution sources and ecosystem structure are significant external factors influencing the temporal and spatial fluctuations of TMFs. Regions near PFAS industrial discharge sources, such as Bohai Bay and Taihu Lake, typically exhibit higher environmental concentrations and TMF values in organisms[86,87]. Similarly, in the Northern Hemisphere, populations near point sources generally exhibit higher PFAS concentrations[113].
Apart from temperature, the chemical properties of water (such as salinity, pH, dissolved organic carbon (DOC), and total phosphorus concentrations) also influence the bioavailability and biomagnification potential of PFAS by affecting their chemical forms and binding states[58]. Among these, salinity, as a key factor distinguishing different aquatic ecosystem types, plays an essential role in regulating the environmental behavior of PFAS[114]. In high-salinity waters, the salting-out effect can reduce the solubility of PFAS in the water phase, promoting their accumulation in particles or biological tissues and thereby altering their bioaccumulation and magnification characteristics. Other physicochemical properties also have specific effects[115]. For example, DOC has a complex and concentration-dependent impact on PFAS biomagnification: at lower concentrations, certain types of DOC (especially humic acids) may promote PFAS bioaccumulation, while at higher concentrations, DOC tends to reduce bioavailability and magnification. This is likely due to the combined effects of DOC type, concentration, and its influence on biological membrane permeability and the nutrient environment[115−117]. Changes in pH also partially affect biomagnification. Under lower pH conditions, changes in the functional groups on adsorbent surfaces and PFAS migration rates and degradation may lead to higher concentrations of PFAS in fish[31,118,119]. Finally, nutrient concentrations can alter TMFs by influencing species growth rates and food web structure. For example, elevated nutrient levels often promote faster growth in organisms. This accelerated growth can lead to a growth dilution effect, where the increase in body mass outpaces the accumulation of PFAS, thereby reducing contaminant concentrations in fish tissues[109].
Overall, these findings indicate that PFAS TMFs in food webs exhibit variation across regions, driven by the combined influence of environmental temperature, water chemical characteristics, salinity, ecosystem structure, and pollution source distribution. Environmental conditions not only regulate the physicochemical behavior of PFAS but also affect the physiological and ecological responses of aquatic organisms, thereby jointly shaping the migration and biomagnification processes of PFAS in food webs. Therefore, in assessing the environmental risks of PFAS and formulating regional pollution management or mitigation strategies, it is crucial to fully consider the comprehensive impact of these environmental variables on PFAS biomagnification mechanisms (Fig. 4).
Methodology
-
Various methods can systematically impact the estimation of TMFs. For instance, matrix effects between samples may lead to inaccuracies in quantification, resulting in biased TMF measurements. In reported cases, adding an adequate amount of isotopically labelled internal standard prior to extraction and using isotope dilution methods, along with smaller sample sizes (approximately 250 mg) and efficient purification, can help mitigate variable recovery rates and matrix effects, ensuring consistent results within the test range[78].
Additionally, the arbitrary replacement of not detected (ND) values (e.g., using 0.5 × LOD or DF × LOD) is common practice but lacks a theoretical basis and can distort correlations and regression slopes, subsequently inflating TMF estimates[78,120]. For instance, for PFUnDA, in the case of a benthic food web, arbitrary substitution resulted in a TMF range of 3.2–3.7 (p < 0.0001, R2= 0.23–0.24), whereas using a robust deletion method (Akritas-Theil-Sen/cenken) yielded non-significant results (p = 0.08, Kendall's τ = 0.13, slope = 0.15, TMF = 1.4). Even with a low deletion rate (less than 12% for PFUnDA non-detections), simple substitution still led to distorted TMF values and conflicting conclusions[78]. Therefore, it is essential to clearly report non-detection treatments and internal standard strategies in the report and conduct sensitivity analyses to quantify the methodological dependency of TMFs. Furthermore, the detection frequency and concentration of pollutants should be carefully determined, as an inaccurate frequency can lead to unexpected TMF results, as highlighted by both Gao et al. and Miranda et al[64,65].
-
The biomagnification of PFAS in aquatic food webs is widely regarded as a potential threat to ecosystem stability and human health. However, in dietary exposure and health risk assessments, it is essential to recognize that biomagnification is not an inevitable outcome, but rather a conditional consequence of integrated processes, including absorption, distribution, metabolism, and excretion. Multiple factors, including the properties of the compounds, the physiological characteristics of the species, the structure of the food web, and the environmental context, influence this process. Therefore, when assessing the biomagnification potential of PFAS in food webs, caution should be exercised, and interpretation should be evidence-based.
This review systematically summarizes the research progress in understanding the transfer and biomagnification of PFAS in aquatic ecosystems. Beginning with the toxicokinetic mechanisms, it elucidates the intrinsic drivers of biomagnification. The review establishes the TMF as a central component of standardized assessment frameworks. It examines the biomagnification behavior of legacy long-chain PFAS, emerging alternatives, and their precursors across diverse aquatic food webs. This work further systematically analyses the influence of multiple dimensions, such as chemical properties, biological characteristics, and environmental conditions, on trophic transfer. Overall, the strong protein-binding affinity and slow elimination kinetics of PFAS underlie their potential for biomagnification. However, this process is intensely regulated by the complex structure of the food web, showing apparent compound dependence, species specificity, and ecosystem differences.
Future research should transition from assessing single substances to studying complex pollution systems, paying attention to the interactions between short-chain and long-chain PFAS, as well as the transformation processes of precursor substances in organisms and their contributions to the accumulation of terminal products. Additionally, it is necessary to thoroughly analyze the physiological and ecological characteristics of different species (especially top predators) in relation to the accumulation and clearance behaviors of PFAS, thereby promoting the deep integration of ecology and toxicokinetics.
To enhance the comparability and regional integration of research, it is recommended to establish a standardized framework for assessing TMFs, including standardized procedures for sample processing, food web structure characterization, and data calculation. Furthermore, the application of omics technologies (such as genomics, proteomics, and metabolomics) should be actively promoted to provide new insights into the toxicokinetics of PFAS. Integrating omics data with traditional toxicokinetic models will enable a more comprehensive understanding of PFAS behavior across different species, particularly in top predators. Moreover, omics tools can be utilized to investigate the transformation of PFAS precursors into persistent terminal products, offering valuable data for assessing long-term bioaccumulation and biomagnification potential.
Ultimately, future efforts should integrate toxicokinetic mechanisms, food web structure, and field observations to develop predictive models that can quantify key uncertainties, such as precursor transformation, interspecies metabolic differences, and environmental influences. Such models will be critical for accurately predicting PFAS biomagnification and formulating targeted ecological and health risk management strategies.
In summary, PFAS biomagnification in aquatic food webs presents a multifaceted scientific challenge that involves numerous processes across various scales. Future studies should extend beyond phenomenological descriptions to achieve a mechanistic understanding and predict the potential for biomagnification. This shift will not only deepen the understanding of contaminant behavior but also address the urgent need to develop effective management strategies that safeguard the integrity of aquatic ecosystems and human dietary health.
-
It accompanies this paper at: https://doi.org/10.48130/newcontam-0026-0004.
-
The authors confirm their contributions as follows: Ge Yang: data collection and analysis, visualization, writing − original draft and review; Yibo Wang: data collection and analysis, visualization, writing − original draft; Di Fang: data collection and analysis, writing − original draft; Jiayi Lin: data collection and analysis, writing − original draft; Xinyu Ma: data collection and analysis, writing − original draft; Minhao Wang: writing − original draft and review; Jason T. Magnuson: writing-review & editing; Wenhui Qiu: writing-review & editing. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets used or analyzed during the current study are available from the corresponding author upon reasonable request.
-
The study was supported by National Natural Science Foundation of China (Grant No. 42322707); Shenzhen Science and Technology Innovation Committee (Grant Nos KCXFZ20240903093700002, JCYJ20241202130703005); Guangdong-Hong Kong Joint Laboratory for Soil and Groundwater Pollution Control (Grant No. 2023B1212120001); Ningbo key research and development plan and 'reveal the list' project (Grant No. 2023Z176); and High-level University Special Fund (Grant No. G03050K001).
-
The authors declare that they have no conflict of interest.
-
Full list of author information is available at the end of the article.
- The supplementary files can be downloaded from here.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Yang G, Wang Y, Fang D, Lin J, Ma X, et al. 2026. Trophic transfer of per- and polyfluoroalkyl substances in aquatic ecosystems. New Contaminants 2: e009 doi: 10.48130/newcontam-0026-0004
Trophic transfer of per- and polyfluoroalkyl substances in aquatic ecosystems
- Received: 10 November 2025
- Revised: 25 December 2025
- Accepted: 11 January 2026
- Published online: 13 March 2026
Abstract: Per- and polyfluoroalkyl substances (PFAS) pose a widespread environmental concern due to their persistence, bioaccumulation potential, and toxicity. Their capacity to biomagnify poses a potential threat to ecosystem stability and human health. However, biomagnification is not an inevitable outcome but rather a conditional consequence of complex toxicokinetic processes interacting with environmental conditions. This review systematically summarizes the current understanding of the trophic transfer of PFAS in aquatic ecosystems, employing the trophic magnification factor as a central tool for standardized assessment. It reveals that biomagnification is related to the specific compound and organism, and is influenced by food web structure and environmental conditions. While legacy long-chain PFAS often demonstrate the capacity to biomagnify, emerging alternatives and precursors exhibit a more variable and uncertain relationship. Future research should shift towards assessing complex mixtures, accounting for precursor transformation, and integrating species-specific physiological ecology with toxicokinetics. The development of predictive models that can integrate mechanistic and food web structures is crucial for accurate risk assessment and for formulating effective, targeted management strategies to protect both aquatic ecosystems and human health.
-
Key words:
- PFAS /
- Trophic magnification /
- Methodology /
- Trophic level /
- Food web /
- Aquatic ecosystems