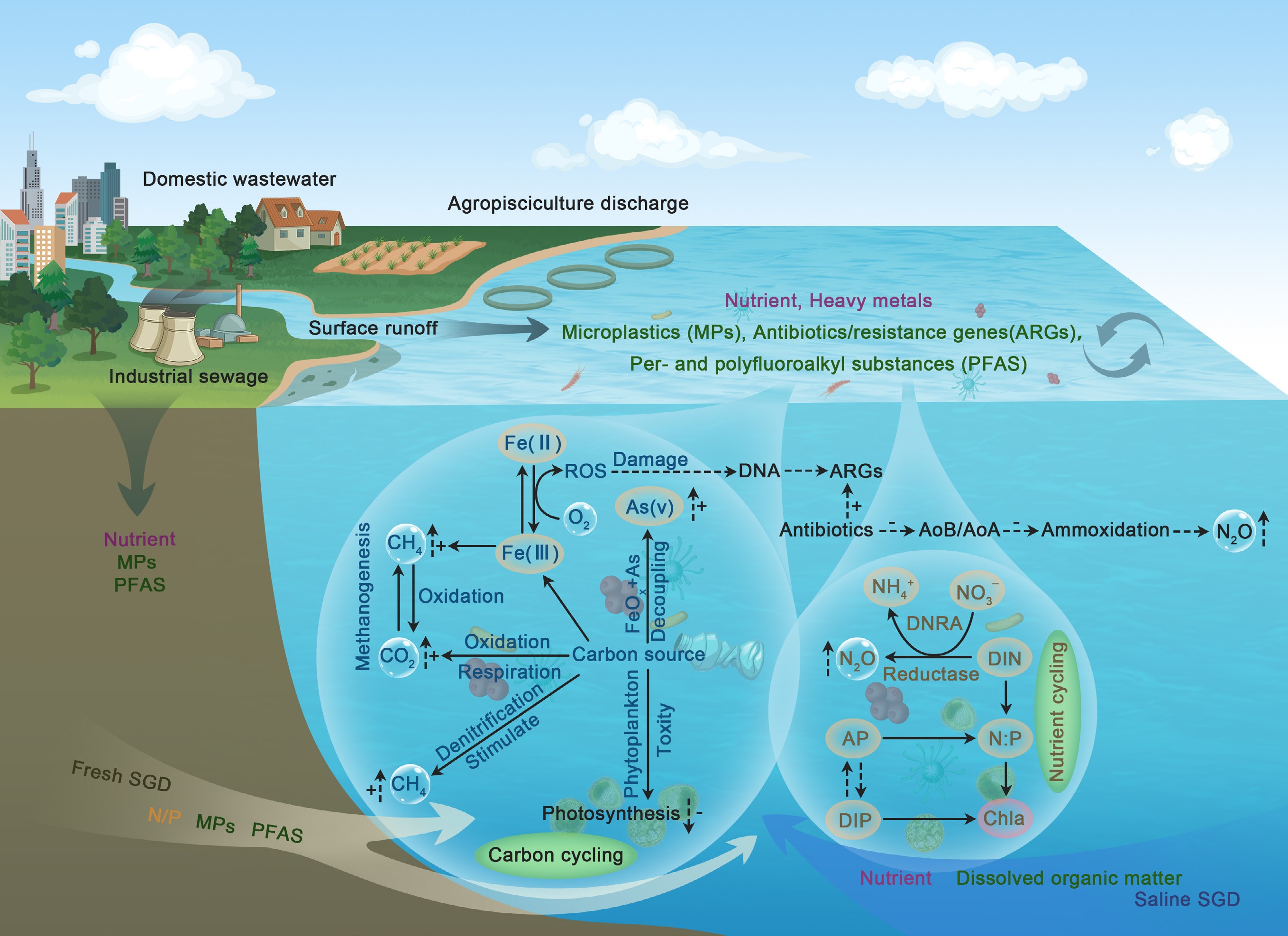
-
Coastal-marine interfaces (CMIs) represent dynamic transition zones where terrestrial and oceanic systems converge. These regions sustain exceptional biodiversity, support productive fisheries, and contribute to global climate regulation through carbon sequestration and elemental cycling[1]. They also facilitate complex biogeochemical interactions crucial for regulating key elemental cycles such as carbon, nitrogen, and phosphorus, heavily influenced by both terrestrial inputs and marine processes[2]. However, anthropogenic pressures have increasingly compromised the integrity of CMIs, primarily through the discharge of both legacy contaminants (LCs; e.g., nutrients, heavy metals, and persistent organic compounds [POPs]) and emerging contaminants (ECs; e.g., pharmaceuticals, antibiotic resistance genes [ARGs], per- and poly-fluoroalkyl substances [PFAS], and microplastics [MPs]) via agricultural runoff, industrial effluents, and urban wastewater[3,4]. Legacy contaminants have long been recognized for their environmental persistence, bioaccumulative potential, and ecotoxicological impacts[5,6]. Mercury, for instance, induces neurotoxic effects in aquatic organisms[7], while petroleum hydrocarbons disrupt metabolic and reproductive functions in marine fauna, with cascading consequences for fishery resources[8]. In coastal settings, these pollutants enter the offshore environment through riverine transport or direct discharge, infiltrating coastal aquifers, and threatening groundwater quality and drinking water safety[9]. Critically, LCs rarely act in isolation, as their co-occurrence with other pollutants can amplify mobility and ecological risk through synergistic physicochemical interactions[10].
Emerging contaminants present a distinct set of challenges due to their chemical diversity and variable environmental behavior[11−13]. Specifically, PFAS exhibit extreme environmental persistence and high aqueous mobility owing to strong C−F bonds and surfactant-like properties[14]. In contrast, certain plastic additives such as phthalates degrade more rapidly but yield toxic transformation products[15]. Microplastics, though limited in solute-like transport, function as reactive vectors by adsorbing trace metals and hosting biofilms that alter local biogeochemistry[16−18]. Their porous surfaces provide abundant binding sites for heavy metals (e.g., Pb2+, Cd2+), potentially prolonging contaminant residence in sediments and enhancing toxicity to primary producers such as microalgae and model organisms like zebrafish[19]. Antibiotics, while not directly altering metal speciation, can indirectly reshape contaminant fate by suppressing functional microbial guilds, particularly denitrifiers, thereby perturbing nitrogen (N) cycling and cascading into imbalances in carbon (C), phosphorous (P), and silicon (Si) dynamics[20,21]. Furthermore, these interactions potentially disrupt carbon storage and biogenic silica fluxes, which are regarded as factors with far-reaching implications for ocean acidification and marine biodiversity[22,23]. Therefore, the heterogeneity of different contaminants in persistence, reactivity, and biological impact complicates environmental risk assessment and necessitates contaminant-specific evaluation within the context of multi-stressor environments[24−26].
The fate and behavior of pollutants in CMIs are further modulated by a suite of environmental variables, including salinity, pH, redox potential, temperature, organic matter content, and microbial activity[27,28]. Heavy metal lability, for instance, is strongly controlled by speciation, with exchangeable fractions being highly mobile, whereas residual forms remain relatively inert in marine sediments[29]. Metal sorption onto particle surfaces, whether mineral or synthetic (e.g., MPs), is enhanced in the presence of organic matter, particularly in high-energy coastal settings such as beaches. Conversely, desorption is favored under acidic conditions, which may promote remobilization in the digestive tracts of organisms and facilitate trophic transfer[30]. Redox transitions exert additional control, particularly through the reductive dissolution of Fe/Mn (hydr)oxides, which governs the mobility of redox-sensitive elements such as arsenic and mercury[31,32]. Hydrodynamic disturbances (e.g., storm events or tidal resuspension) can further mobilize As(III)-rich sediments by eroding reduced layers and accelerating oxide dissolution, thereby elevating environmental and public health risks[33]. When multiple contaminants coexist, their interactions may yield synergistic or antagonistic effects that alter not only toxicity but also environmental persistence and transformation pathways[34,35].
Submarine groundwater discharge (SGD) has emerged as a dominant yet historically underappreciated pathway for land-to-ocean contaminant transfer[36]. Although SGD often constitutes a minor volumetric flux compared to rivers, its solute concentrations can be orders of magnitude higher due to prolonged subsurface residence times and biogeochemical enrichment. Consequently, SGD can deliver nutrient and contaminant loads comparable to, or even exceeding, those from riverine and atmospheric sources[37]. In coastal aquifers, the mixing of oxic freshwater with anoxic saline porewater generates sharp redox gradients that drive intense biogeochemical reactivity. These conditions promote heterotrophic respiration, modify dissolved organic carbon composition, and select for microbial communities capable of degrading complex organic pollutants[9]. Therefore, SGD not only transports contaminants but also actively transforms them en route to the coastal ocean, positioning subterranean estuaries as critical reaction zones for coupled contaminant-element cycling[38].
Despite growing recognition of multi-contaminant pressures in CMIs, significant knowledge gaps persist regarding the long-term ecological consequences and human health implications of co-exposure scenarios. Moreover, the influence of dynamic environmental gradients, particularly salinity and redox state, on contaminant speciation, solubility, and reaction kinetics remains inadequately quantified[9,39]. To advance understanding of multi-contaminant dynamics in CMIs, this review synthesizes peer-reviewed literature through an integrated bibliometric and thematic analysis, with emphasis on spatial patterns, co-occurrence hotspots, and ecological impacts of LCs and ECs. Three integrative hypotheses grounded in current evidence are proposed: (1) a persistent disconnect exists between laboratory-scale mechanistic studies and field observations, resulting in the systematic underrepresentation of SGD-mediated co-transport processes in biogeochemical models and environmental risk frameworks; (2) the co-distribution of LCs and ECs in CMIs is spatially structured by anthropogenic source gradients and hydrological connectivity, with identified hotspots exhibiting disproportionately amplified disruption to C, N, and Si cycling; and (3) SGD-induced redox oscillations concurrently enhance the bioavailability of reduced metals and modify microplastic surface reactivity, thereby reshaping microbial communities responsible for nutrient regeneration. By clarifying where critical uncertainties lie, this review aims to refocus future research on multi-stressor environments where land-sea contaminant exchange exerts its most profound influence on ecosystem function and societal well-being.
-
To systematically evaluate the evolution of scientific understanding regarding contaminant dynamics in CMIs over the past two decades, a comprehensive literature search was conducted using the Web of Science Core Collection (WOSCC). The search strategy was explicitly designed to capture studies focused on hydrological and biogeochemical coupling at land-sea transition zones, rather than on general coastal or open-ocean systems, and to specifically capture studies that address interactions between LCs and ECs in the context of elemental cycling. An initial broad query (i.e., coastal AND pollutant) retrieved 8,032 records. However, this yielded substantial noise from studies lacking explicit focus on CMI-specific processes or multi-contaminant interactions affecting C, N, P, and Si cycles. To enhance thematic precision, the search string was refined to: ('coastal aquifer*' OR 'subterranean estuar*' OR 'submarine groundwater discharge*' OR 'estuarine groundwater' OR 'freshwater-seawater interface' OR 'coastal groundwater' OR 'intertidal aquifer*' OR 'beach aquifer*' OR 'coastal mixing zone') AND ('emerging contaminant*' OR 'legacy pollutant*' OR 'heavy metal*' OR 'nutrient*' OR 'nitrate*' OR 'persistent organic pollutant*' OR 'pesticide*' OR 'pharmaceutical*' OR 'antibiotic*' OR 'microplastic*' OR 'PFAS*' OR 'perfluoroalkyl' OR 'antibiotic resistance gene*' OR 'ARG*') AND ('biogeochemical cycle*' OR 'redox' OR 'sorption' OR 'desorption' OR 'microbial community' OR 'element cycling' OR 'carbon' OR 'nitrogen' OR 'phosphorus' OR 'silicon'). This targeted approach yielded a final corpus of 5,067 peer-reviewed publications. Each record underwent manual screening to extract structured metadata, including: (1) contaminant categories (e.g., nutrients, heavy metals, persistent organic pollutants, pharmaceuticals, microplastics, antibiotics); (2) environmental matrices (groundwater, SGD-influenced surface water, sediments, and biota); and (3) key biogeochemical or hydrological processes (e.g., redox transitions, sorption/desorption, microbial transformation, SGD-driven transport). Particular emphasis was placed on studies documenting co-occurrence or interaction effects between LCs and ECs and their implications for C, N, P, and Si cycling, which served as a criterion used to prioritize inclusion in subsequent thematic and spatial analyses.
Bibliometric and thematic analysis
-
To map the intellectual structure, collaborative networks, and conceptual trajectories of the field, bibliometric network analysis was integrated with thematic trend detection using CiteSpace (v6.20.R3) and VOSviewer (v1.6.18). Full-record data (including titles, abstracts, keywords, references, authors, and affiliations) were exported from WOSCC in plain text format and preprocessed in OriginPro to resolve inconsistencies among author names, institutional spellings, and keyword synonyms (e.g., 'microplastic' vs 'microplastics'). In CiteSpace, co-citation analysis of cited references was performed to identify foundational works and intellectual turning points. Additionally, burst detection algorithms highlighted rapidly emerging topics. Timeline views visualized the temporal evolution of research clusters. Concurrently, VOSviewer generated keyword co-occurrence networks, where edge weights reflected the frequency of joint appearance across publications. From an initial pool of 4,694 unique keyword-plus-derived terms, only those occurring ≥ 10 times (n = 166) were retained to ensure statistical robustness and semantic clarity. These keywords were grouped into coherent thematic domains through iterative interpretation based on contextual meaning and domain knowledge, enabling identification of dominant research fronts and nascent directions. To assess geographic patterns of contaminant co-occurrence, we compiled a georeferenced dataset comprising > 400 sampling sites across China's coastal aquifers, selected from studies reporting quantitative concentrations of LCs and/or ECs in groundwater or SGD-impacted porewaters. Site coordinates, contaminant classes, and concentration values were organized in Microsoft Excel, and imported into ArcGIS 10.2 for spatial interpolation.
Conceptual synthesis framework
-
To integrate empirical findings into a mechanistic understanding of multi-contaminant impacts on biogeochemical cycling in CMIs, a process-oriented conceptual framework structured around four interdependent components was developed: (1) hydrogeochemical controls including redox stratification, pH shifts, salinity gradients, and mineral transformations (e.g., reductive dissolution of Fe/Mn oxides) that govern contaminant speciation, solubility, and mobility, particularly for redox-sensitive elements like As, Hg, and Cr; (2) matrix-mediated interactions consisting of the roles of sediments, natural organic matter, clay minerals, and biofilms acting as reactive interfaces that adsorb, sequester, or catalyze the transformation of co-occurring contaminants. Microbial communities embedded within these matrices further modulate redox conditions and enzymatic degradation pathways; (3) interactions between contaminants including competitive sorption, complexation, carrier effects (e.g., microplastics as vectors for hydrophobic organic compounds), and altered bioavailability due to LC-EC co-exposure that can amplify or mitigate ecological risks beyond additive expectations; and (4) transport dynamics highlighting SGD as a primary pathway for terrestrial contaminant export to coastal waters. Its magnitude and chemical signature are modulated by sea-level rise, groundwater extraction, seasonal recharge variability, and tidal forcing, which are factors shaping spatiotemporal patterns of contaminant delivery and in situ reactivity in subterranean estuaries.
-
Coastal-marine interfaces constitute dynamic transition zones where terrestrial and marine systems interact through tightly coupled hydrological, biogeochemical, and ecological processes[40]. These zones serve as critical filters or amplifiers of contaminant fluxes from land to sea, governed by redox gradients, salinity transitions, microbial activity, and sediment–water interactions[41]. Over the past two decades, scientific interest in contaminant dynamics within CMIs has expanded markedly, driven by growing awareness of their vulnerability to anthropogenic pressures, including land-use change, wetland loss, reservoir construction, and compounding climatic stressors such as sea-level rise, extreme precipitation events, and ocean warming[42,43]. The present analysis of 5,067 peer-reviewed publications reveals a pronounced upward trajectory in research output, with annual publication counts rising from fewer than 40 prior to 2005, to over 300 after 2018, peaking at 420 in 2022 (Fig. 1). Concurrently, mean annual citations per publication have followed a non-linear but overall increasing trend since 2000, reaching ~40 in 2021, indicating both expanding scholarly engagement and rising scientific impact within environmental science, hydrology, and Earth system disciplines. Notably, 35% of all studies explicitly address coastal groundwater pollution (CGP), underscoring the pivotal role of subsurface pathways, particularly submarine groundwater discharge (SGD), in delivering contaminants to nearshore waters. This emphasis reflects a paradigm shift from viewing CMIs merely as passive recipients of pollution to recognizing them as reactive aquifers where biogeochemical transformations actively modulate contaminant fate[42,44].
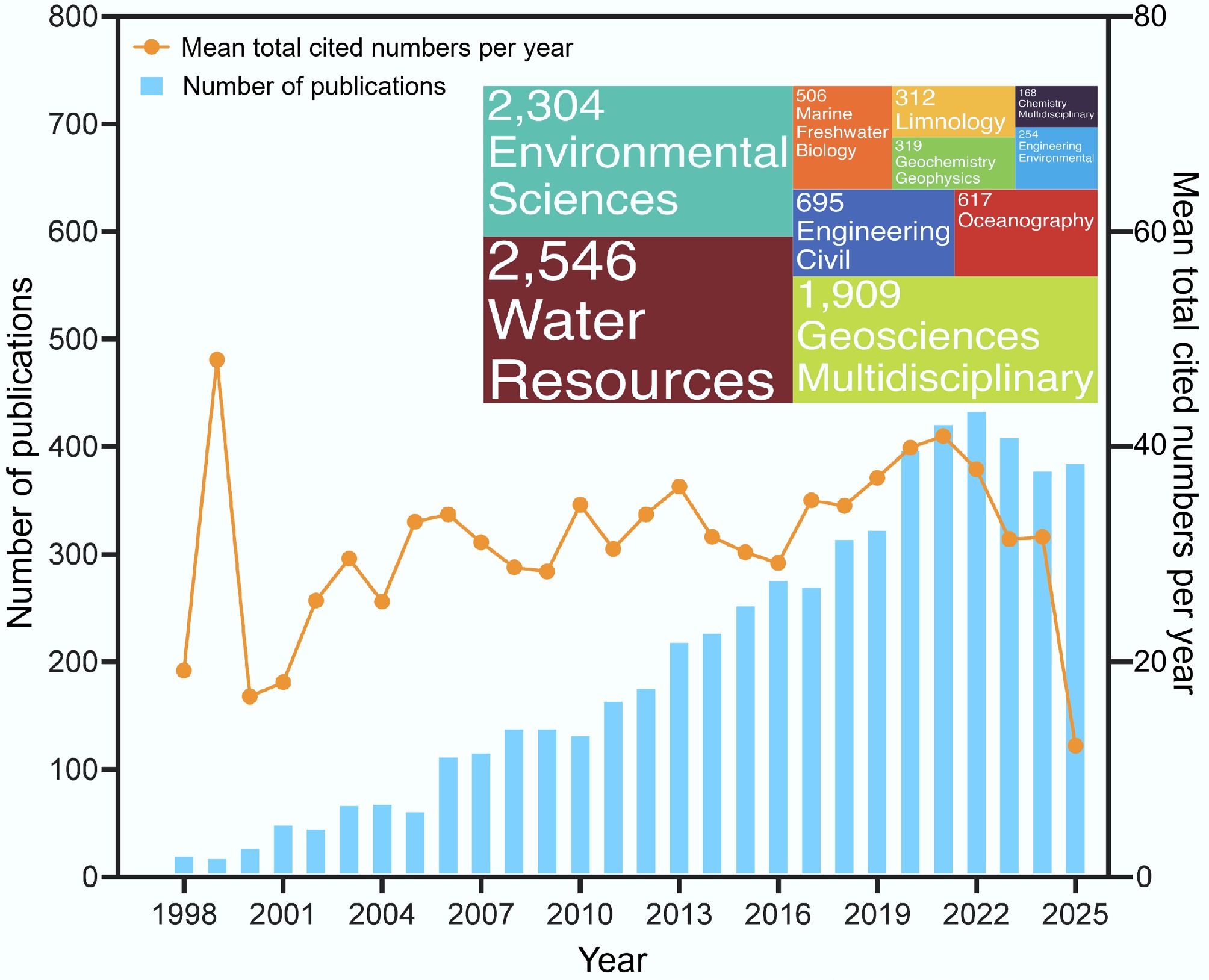
Figure 1.
Trends in the number of published and mean total annually cited articles on CMI pollution identified by WOSCC, and the number and proportion of related disciplines (top 10) for CMI pollution research.
Disciplinary mapping further illustrates the inherently interdisciplinary nature of CMI research. Core contributions are from water resources (~26%), environmental science (~24%), and multidisciplinary geosciences (~20%) (Fig. 1), with significant input from marine/freshwater biology, oceanography, and civil/environmental engineering. This convergence signals a move beyond compartmentalized approaches toward integrated frameworks that link groundwater contamination with land-use dynamics, sediment biogeochemistry, and anthropogenic stressors. Critically, the integration facilitates system-scale modeling and process coupling, which is essential for predicting contaminant behavior under non-stationary hydroclimatic conditions.
Keyword co-occurrence and thematic clustering
-
To delineate the intellectual architecture of the field, keyword co-occurrence networks were constructed using CiteSpace and VOSviewer (Fig. 2a & b). Analysis of 5,067 publications yielded 15 distinct modules. Among these, terms such as submarine groundwater discharge, groundwater quality, dissolved organic matter, seawater intrusion, and denitrification exhibited high centrality and frequency, confirming their status as foundational concepts in CMI contaminant research. Focusing specifically on the refined query 'land-ocean* OR land-sea* AND groundwater* AND pollutant*', 4,694 unique keywords were identified, of which 166 occurred ≥ 10 times. These were clustered into three dominant thematic domains based on linkage strength and semantic coherence (Fig. 2b)[45]. The red cluster represents biogeochemistry and nutrient dynamics, centered on 'nitrogen', 'nitrate', 'heavy metals', 'groundwater', and 'submarine groundwater discharge'. This domain reflects sustained research on nutrient loading, metal mobilization under redox transitions (e.g., Fe/Mn reduction), and SGD-mediated export of terrestrial solutes to coastal waters. The green cluster highlights hydrogeological and salinity dynamics, dominated by 'aquifer', 'seawater intrusion', 'salinity', and 'geochemistry'. This cluster emphasizes the hydrological fragility of coastal aquifers under increasing groundwater extraction and sea-level rise[46,47]. Salinity-driven shifts in ionic strength and pH can trigger desorption or reductive dissolution of mineral-bound contaminants, thereby altering porewater chemistry and contaminant mobility. The blue cluster covers pollution assessment and management, encompassing 'pollution', 'drinking-water', 'river-basin', and 'groundwater quality'. This domain underscores the applied dimension of CMI research, particularly concerns over human exposure via groundwater use and the need for integrated monitoring and risk governance.
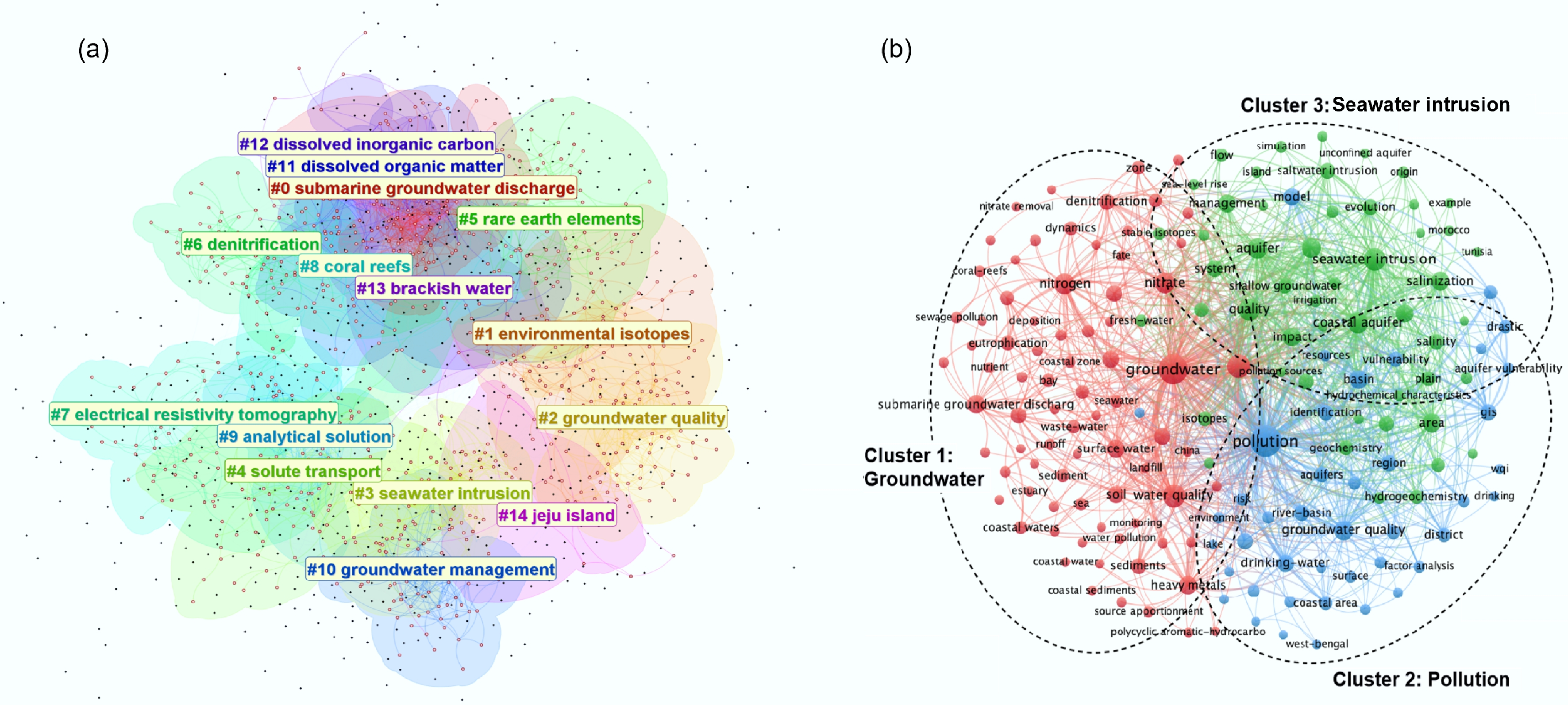
Figure 2.
Network visualization map generated (a) by CiteSpace based on the search terms of ('coastal aquifer*' OR 'subterranean estuar*' OR 'submarine groundwater discharge*' OR 'estuarine groundwater' OR 'freshwater-seawater interface' OR 'coastal groundwater' OR 'intertidal aquifer*' OR 'beach aquifer*' OR 'coastal mixing zone') AND ('emerging contaminant*' OR 'legacy pollutant*' OR 'heavy metal*' OR nutrient* OR nitrate* OR 'persistent organic pollutant*' OR pesticide* OR 'pharmaceutical*' OR antibiotic* OR 'microplastic*' OR 'PFAS*' OR 'perfluoroalkyl' OR 'antibiotic resistance gene*' OR ARG*) AND ('biogeochemical cycle*' OR redox OR 'sorption' OR 'desorption' OR 'microbial community' OR 'element cycling' OR carbon OR nitrogen OR phosphorus OR silicon), and (b) by VOSviewer based on the search terms of 'Land-ocean* or Land-sea* AND groundwater* AND pollutant*' from Web of Science Core Collection database. Different colors represent the specific clusters of extracted terms that were grouped by the software based on the items relations. Size and label represent the weight of keywords, and lines represent links between labels.
Crucially, these clusters are interconnected through bridging concepts. Nitrate links nutrient and salinity dynamics (red-green), pollution sources connect biogeochemical processes to management imperatives (red-blue), and aquifers serve as a shared locus across hydrogeological and water quality domains (green-blue). The term 'pollution' itself functions as a unifying node, integrating natural process understanding with societal relevance.
Temporal evolution of research fronts
-
A burst detection analysis of keyword emergence reveals four distinct phases in the conceptual evolution of CMI contaminant research (Fig. 3). Prior to 2005, research focused primarily on nitrogen, phosphorus, eutrophication, and nitrate, reflecting global concern over nutrient-driven algal blooms and hypoxia, particularly in river-influenced coastal systems, with an emphasis on stoichiometric imbalances (N : P ratios) and their ecological ramifications[48]. Between 2006 and 2011, a shift toward seawater intrusion, salinity, and aquifer dynamics coincided with rapid coastal urbanization and unsustainable groundwater abstraction[49]. Climate-induced sea-level rise intensified scrutiny of saltwater-freshwater interactions and their feedback on contaminant release, e.g., mobilization of adsorbed metals due to ionic competition or pH shifts[50]. From 2012 to 2017, research was characterized by a heightened focus on heavy metals (e.g., As, Cd, Pb) and fluoride, driven by industrial discharges, mining legacies, and geogenic contamination in vulnerable aquifers[51,52]. Fluoride gained prominence due to its dual origin (anthropogenic vs natural) and complex geochemical behavior, including pH-dependent desorption and evaporite dissolution[53]. Since 2018, research has been defined by the ascendancy of ECs, including pharmaceuticals, personal care products, MPs, and ARGs, alongside increased interest in dissolved organic matter (DOM) and organic carbon[54,55]. DOM is now understood not only as a carbon substrate but as a multifunctional agent that influences redox buffering, metal complexation, and pollutant transport[56].
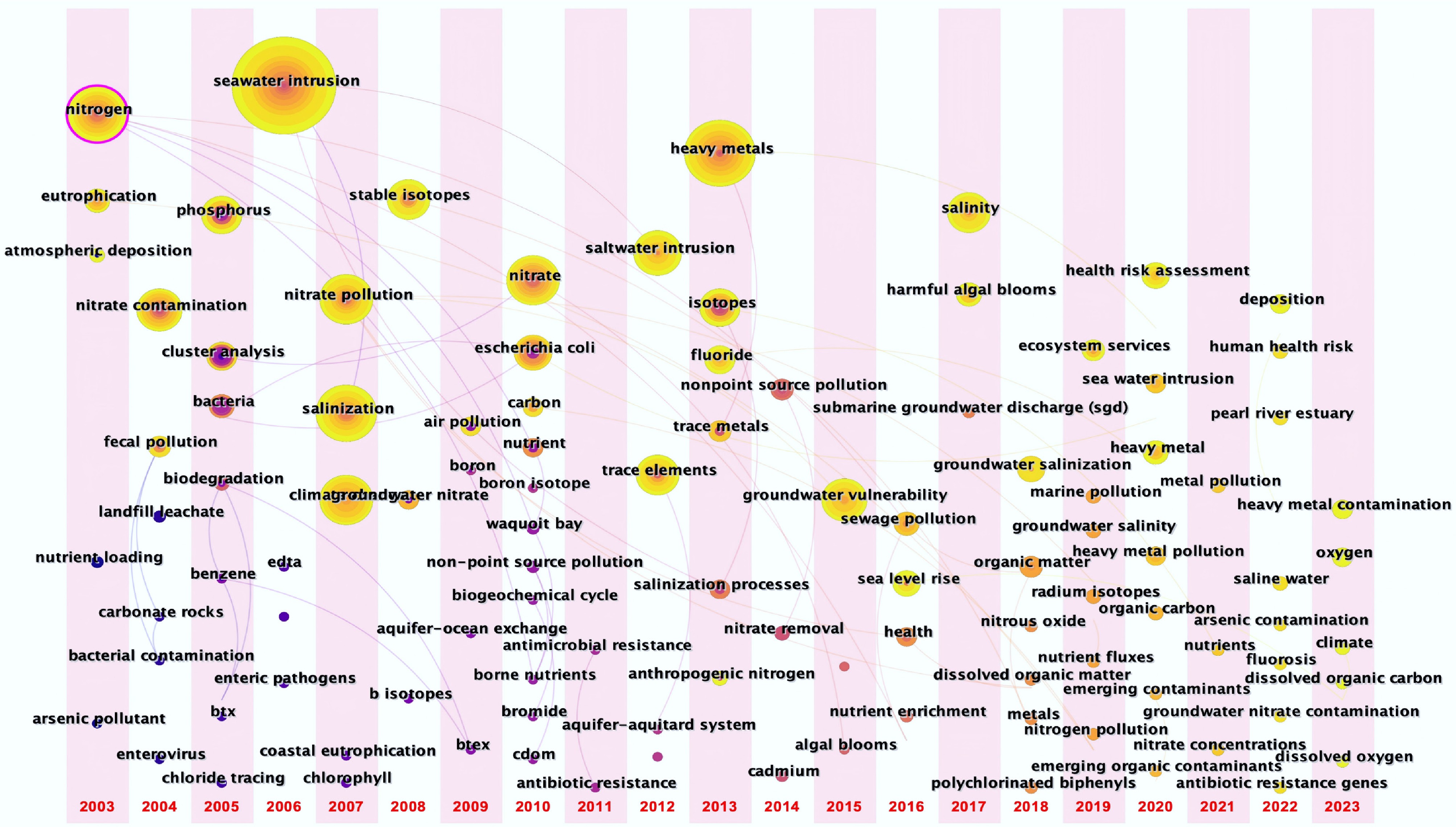
Figure 3.
Time-line view of keywords and noun-phases. The node size represents the co-occurrence frequency of keywords, the red characters are the clustering labels, the length of the horizontal line represents the duration of each clusters, and the top number is the end year of each time slice.
Despite this diversification, nutrient contamination remains persistent, sustained by diffuse agricultural runoff, aquaculture effluents, and inadequately treated wastewater in densely populated coastal regions[57,58]. More importantly, the temporal trajectory reveals a conceptual shift from studying individual pollutants in isolation to investigating contaminant co-occurrence, interaction mechanisms, and synergistic effects on biogeochemical cycles, which is a frontier still underexplored. For instance, MPs may act as vectors for hydrophobic organic compounds while simultaneously providing substrates for biofilm development that locally alters nitrogen and phosphorus cycling. Similarly, DOM can compete with phosphate for sorption sites on Fe/Mn oxides, potentially enhancing P mobility and bioavailability in estuarine sediments. Emerging evidence also implicates processes such as photo-oxidation of organic carbon, microbial respiration across redox gradients, and mineral-contaminant interactions as critical regulators of C, N, P, and Si cycling, which are elements that collectively govern primary productivity, nutrient limitation regimes, and C sequestration potential in coastal ecosystems[46,47].
Spatial distribution of legacy and emerging contaminants at CMIs, focusing on China
-
China's coastal zone, which supports over 40% of the national population within a 10-km band along its 32,000 km shoreline, experiences intense anthropogenic pressure, resulting in widespread co-contamination by LCs and ECs[59]. Historically, research has emphasized LCs such as nutrients (N, P), heavy metals, and POPs, whose accumulation in coastal environments is linked to eutrophication, ecotoxicity, and long-term ecosystem degradation[7,56]. More recently, CMIs have emerged as critical zones where LCs intersect with a growing array of ECs, including pharmaceuticals, MPs, ARGs, and PFAS, posing complex challenges for biogeochemical cycling, groundwater quality, and water security[30,57,58]. Drawing on a synthesis of data from over 400 peer-reviewed study sites across China's coastal aquifers, this section evaluates the spatial co-distribution of LCs and ECs in CMI groundwater systems (Fig. 4), with emphasis on source attribution, interaction potential, and implications for environmental risk.
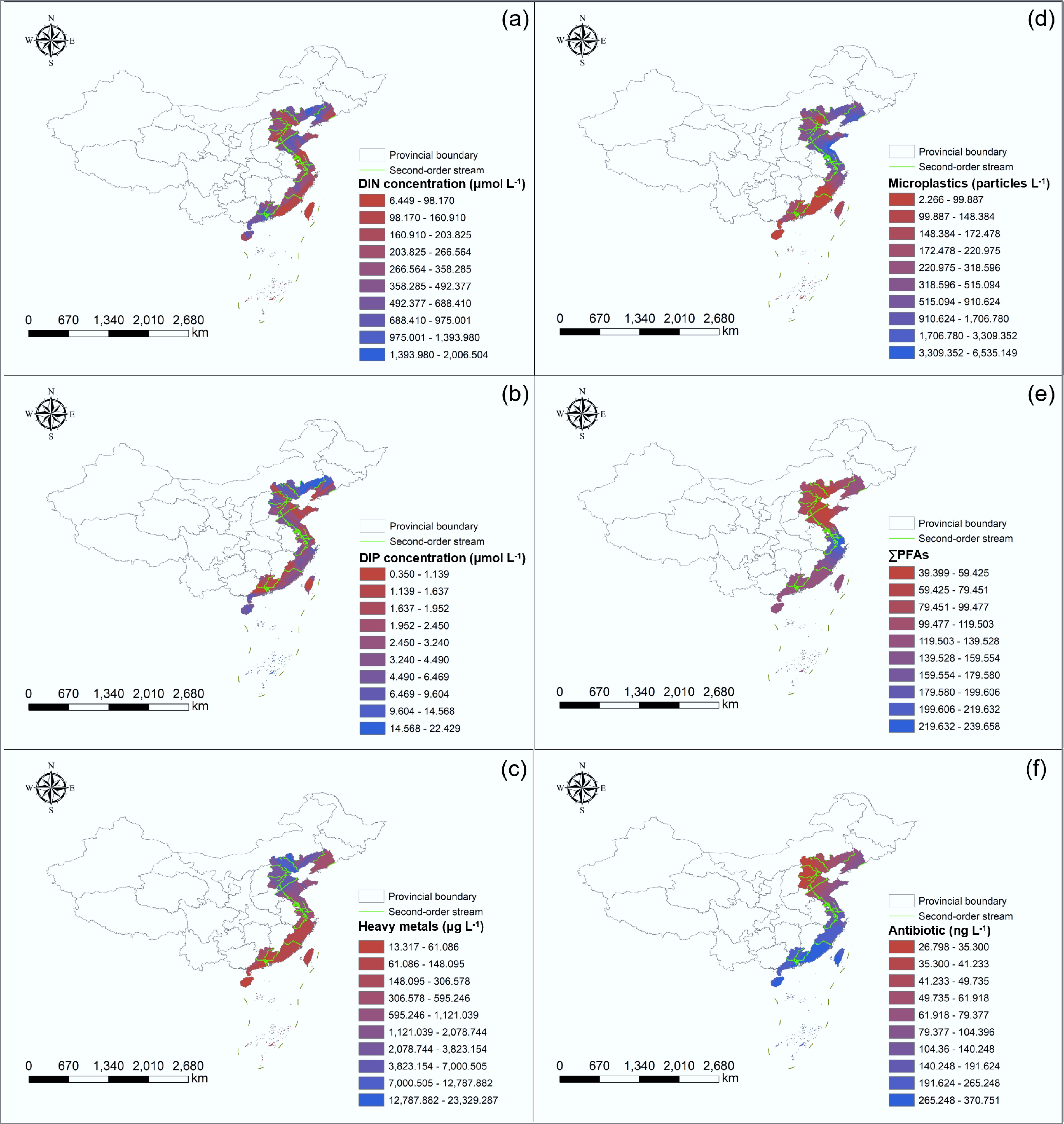
Figure 4.
Spatial distribution of contaminants in groundwater in coastal areas of China: (a) dissolved inorganic nitrogen (DIN), (b) dissolved inorganic phosphorus (DIP), (c) heavy metals, (d) microplastics, (e) per- and poly-fluoroalkyl substances (PFAS), and (f) antibiotics.
In the marine environment, nitrogen and phosphorus serve as primary limiting nutrients for phytoplankton growth, underpinning basal productivity in aquatic food webs when present at balanced concentrations[59]. However, excessive inputs, particularly from agricultural runoff and inadequate wastewater treatment, trigger algal blooms, oxygen depletion, and ecosystem collapse via eutrophication (Supplementary Table S1)[60]. Analysis of 315 groundwater samples from China's CIMs reveals elevated concentrations of dissolved inorganic nitrogen (DIN) and dissolved inorganic phosphorus (DIP), with pronounced hotspots in the Bohai Bay, Liaodong Bay, southern Yellow Sea, and both flanks of the Qiongzhou Strait (including Hainan Island), where concentrations most frequently exceed national groundwater quality standards (Supplementary Table S2; Fig. 4a & b). After excluding non-detects and extreme outliers (> 1,500 μmol L−1), correlation analysis of the remaining 175 samples shows a statistically significant positive relationship between DIN and DIP concentrations (p < 0.05, Fig. 5a), consistent with shared anthropogenic sources and coupled hydrogeochemical transport pathways. This co-enrichment reflects decades of intensive agriculture characterized by excessive nitrogenous fertilizer use and insufficient sanitation infrastructure[61,62]. Nitrates (NO3−) and nitrites (NO2−) exhibit marked spatial heterogeneity across coastal China, governed by gradients in nutrient loading, recharge dynamics, and subsurface redox conditions[61,63]. Critically, N/P-rich groundwater plumes discharge into coastal waters via submarine groundwater discharge (SGD), acting as a sustained, diffuse source of eutrophication with N/P-rich plumes discharging into coastal waters via SGD, contributing significantly to coastal eutrophication[36].
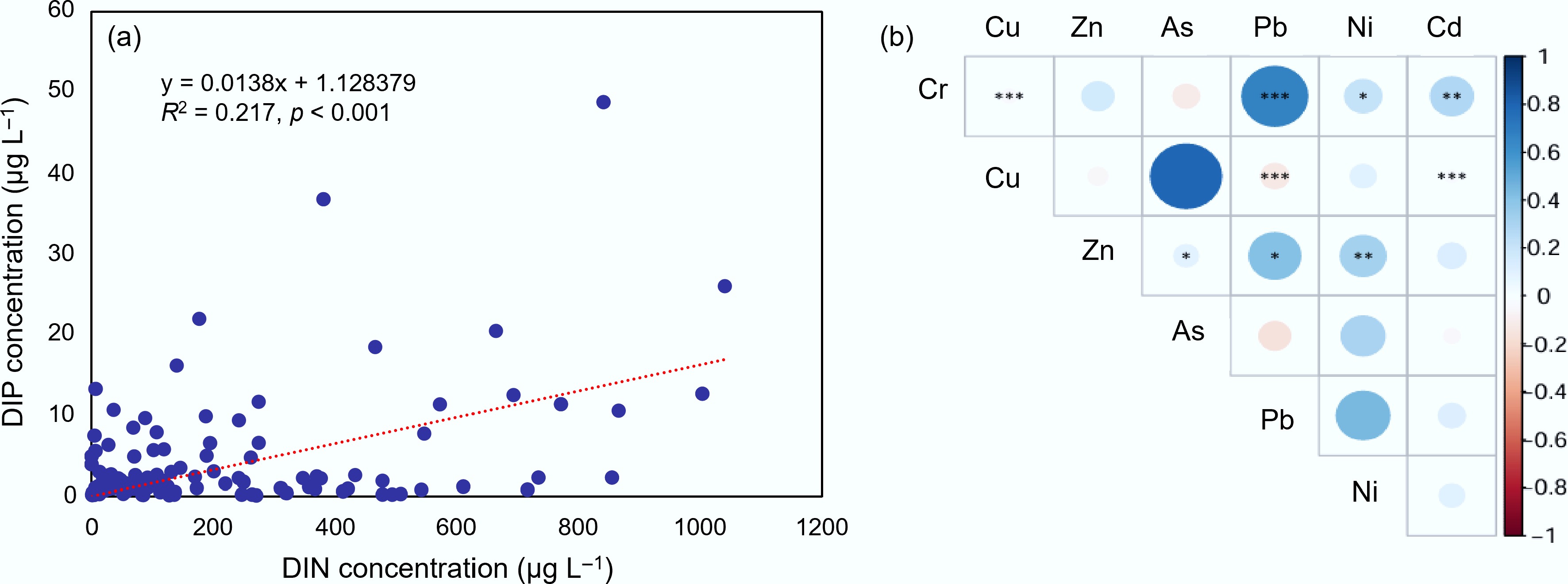
Figure 5.
(a) Relationships between the distributed concentrations of dissolved inorganic nitrogen (DIN) and dissolved inorganic phosphorus (DIP) within the reported 175 research sites, and (b) correlations among different heavy metals. *, **, and *** represent significant differences between different pollution conditions at p < 0.05, p < 0.01, and p < 0.001 level, respectively.
Heavy metals and MPs exhibit overlapping spatial patterns, with elevated concentrations observed in Bohai Bay and Jiaozhou Bay (Fig. 4c & d). This convergence likely stems from common emission sources, particularly industrial effluents, and potentially from the role of MPs as carriers facilitating metal transport and retention[64]. Research on heavy metals has focused on As, Cr, Pb, Cd, Zn, Cu, and Ni, which exhibit highly localized but persistent enrichment, especially near industrial and mining districts[65,66]. The Bohai Bay region hosts extensive metallurgical, electroplating, and chemical industries that historically discharged untreated or poorly treated wastewater containing Hg, Cd, and Pb[67]. These inputs, combined with the bay's semi-enclosed geometry, weak hydrodynamics, and limited seawater exchange, promote contaminant accumulation in sediments and facilitate lateral transport via groundwater circulation. Notably, elevated Cr and Zn levels in the Tangshan area are linked to legacy metallurgical activities and improper solid waste disposal. Similarly, the Pearl River Delta represents another major contamination hotspot, shaped by decades of unregulated discharges from electronics manufacturing, battery production, leather tanning, and informal e-waste recycling[68]. Strong inter-element correlations among heavy metals (Fig. 5b) further support common origins or co-migration mechanisms, aligning with global patterns of metal enrichment corridors[69]. MP pollution in the Tianjin and Jiaodong Peninsula areas correlates spatially with nitrogen hotspots, implicating shared sources, such as domestic sewage and agricultural plastic use (e.g., mulching films, packaging). The concurrent input of agricultural plastics, fertilizers, and aquacultural antibiotics, compounded by limited hydrodynamic flushing, fosters synergistic effects. Nutrient enrichment promotes microbial activity that may enhance ARG proliferation, while MPs provide surfaces for biofilm formation and horizontal gene transfer. These interactions threaten both coastal ecosystem integrity and aquaculture product safety.
In contrast to the localized hotspots of LCs, pharmaceuticals, and PFAS exhibit broader, more diffuse distributions, with the highest concentrations observed in the Yellow Sea and southern coastal regions (Fig. 4e & f). Antibiotics, including tetracyclines (TCs), sulfonamides (SAs), macrolides (MLs), quinolones (QNs), and cephalosporins (CPs), are particularly elevated in the Yangtze River Delta and Pearl River Delta. This pattern is driven by intensive aquaculture, livestock operations, and dense urban populations that generate substantial pharmaceutical loads[70,71]. These compounds pose dual threats: direct ecotoxicological effects and selection pressure for antimicrobial resistance. ARGs and associated mobile genetic elements (e.g., integrons, plasmids) are increasingly detected in estuarine sediments and SGD-influenced zones[72,73], positioning CMIs as reservoirs and conduits for antimicrobial resistance dissemination. The frequent co-occurrence of antibiotics and ARGs in groundwater underscore the need to treat CMIs as integrated One Health interfaces. Despite growing concern, the occurrence and fate of MPs and PFAS in groundwater remain poorly constrained due to significant analytical challenges[74,75]. Their detection in complex subsurface matrices is hindered by low concentrations, matrix interference, and the lack of standardized reference materials, limiting cross-study comparability[76,77]. Moreover, the hydrophobic nature of many PFAS and the nano-scale dimensions of MPs challenge conventional filtration and quantification methods. To advance understanding, the application of high-resolution mass spectrometry (HRMS), pyrolysis-gas chromatography/mass spectrometry (Py-GC/MS) for MPs, and compound-specific isotope analysis (CSIA) is urgently needed to improve detection sensitivity, resolve transformation pathways, and elucidate subsurface transport mechanisms.
Sources of legacy contaminants and controls on their migration and transformation
-
Intensive socioeconomic activity along China's coast has led to pervasive contamination of soils, sediments, and groundwater systems through the discharge of untreated industrial effluents, agricultural runoff, and municipal wastewater[38]. Within CMIs, the fate of LCs, including POPs, nutrients, and heavy metals, is governed by a tightly coupled network of hydrological, geochemical, and microbial processes. These mechanisms operate across multiple spatiotemporal scales, from molecular-level redox transformations to regional-scale groundwater-surface water exchange, and collectively shape contaminant mobility, speciation, and ecological impact in dynamic subsurface environments (Supplementary Table S3). This section synthesizes the dominant sources and key biogeochemical controls on LC migration and transformation in CMIs, with emphasis on sediment geochemistry, microbial mediation, redox zonation, salinity gradients, and groundwater-surface water interactions (Fig. 6). Together, these factors define the biogeochemical architecture that regulates contaminant behavior at the land–sea boundary.
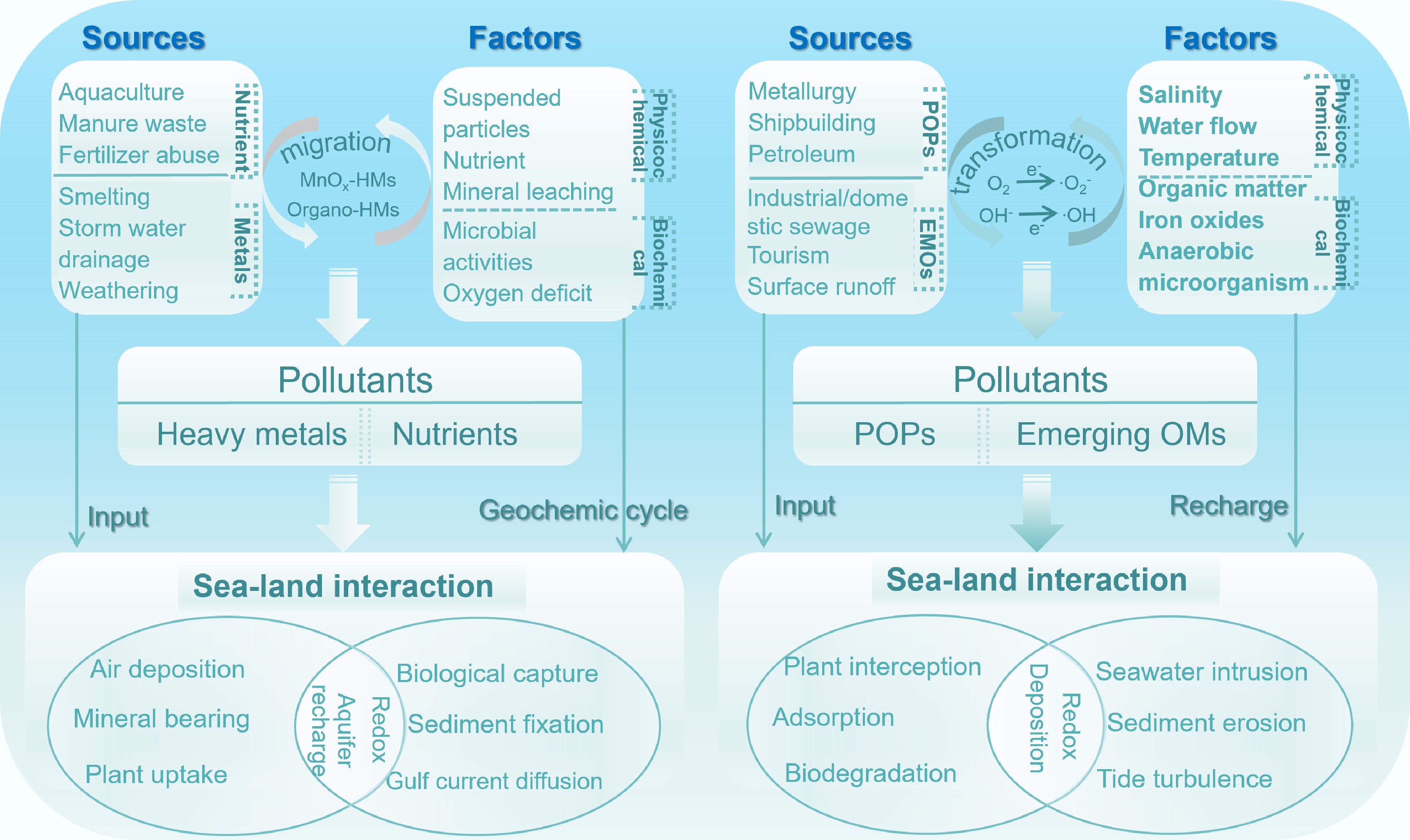
Figure 6.
Sources, drivers, and land–sea interactions governing legacy/emerging pollutant cycling in coastal margine interfaces (CMIs). Left (heavy metals, nutrients): HMs migrate as Mn oxide/organic-ligand complexes; nutrient overloading (aquaculture/manure) induces HM-sediment sorption competition (modifying bioavailability). CMI redox dynamics mediate HM transformation; biotic capture (plants, benthos) links biological uptake to geochemical cycling. Right (organic pollutants): ROS (superoxide, OH) drive POP/emerging OM oxidative transformation (regulating toxicity/persistence). Salinity/microbiota control OM sorption/anaerobic biodegradation. Seawater intrusion/tidal turbulence resuspend sediment-bound organics, while plant interception enables rhizosphere co-metabolic OM degradation.
Persistent organic pollutants (POPs): persistence, remobilization, and biogeochemical regulation
-
Despite global regulatory frameworks such as the Stockholm Convention, many POPs, including polychlorinated biphenyls (PCBs), polycyclic aromatic hydrocarbons (PAHs), and organochlorine pesticides (OCPs), remain widespread in CMIs due to their high environmental persistence, strong hydrophobicity, and capacity for long-range atmospheric and oceanic transport[78]. Primary inputs originate from historical and ongoing industrial discharges, agricultural applications, and petrochemical operations, resulting in significant accumulation in estuarine sediments and coastal aquifers (Fig. 6)[78,79]. Of growing concern is the remobilization of historically sequestered POPs under changing environmental conditions, such as sea-level rise, groundwater table fluctuations, and intensified storm events, which can destabilize sediment-bound reservoirs and reintroduce legacy pollutants into active biogeochemical cycles.
Recent studies underscore the critical role of coastal vegetated ecosystems, including mangroves, salt marshes, and seagrass meadows, as natural biogeochemical filters. These habitats trap particle-associated POPs via root-induced sedimentation and foster enhanced microbial degradation through vertically stratified redox conditions in porewaters[80]. For example, anaerobic consortia in mangrove soils can reductively dechlorinate PCBs under sulfate-reducing or methanogenic regimes, while PAHs undergo partial mineralization by facultative degraders in redox-fluctuating zones. The mobility of POPs is further modulated by soil organic matter (SOM) content and pH. Low-SOM, alkaline conditions reduce sorption capacity, increasing PCB leachability, whereas acidic environments promote protonation and surface complexation, limiting aqueous-phase transport[81]. Seasonal dynamics also exert strong control: elevated rainfall enhances PAH desorption from suspended particulates, boosting solubility and facilitating export to coastal waters[82]. In polar CMIs, freeze-thaw cycles and ice-melt pulses mobilize POPs previously locked in permafrost or glacial matrices, leading to episodic release and potential re-deposition in nearshore sediments[83]. However, the long-term trajectory of these remobilized compounds under accelerating climate change remains poorly constrained, representing a critical knowledge gap for predictive contaminant modeling.
Nutrient dynamics: anthropogenic forcing and subsurface export pathways
-
Anthropogenic intensification through agricultural expansion, urbanization, and atmospheric deposition has dramatically increased fluxes of reactive nitrogen (Nr) and bioavailable phosphorus to coastal zones, particularly along China's densely populated margins. Municipal wastewater alone contributes over 6 million metric tons of Nr annually, accounting for nearly 45% of total Nr emissions from agricultural catchments, while atmospheric deposition supplies approximately 30% of nitrate in the Yellow Sea's surface waters[84,85]. Within CMIs, nutrient cycling is mediated by hydrodynamic forcing, redox gradients, and microbial metabolism, including nitrification, denitrification, anaerobic ammonium oxidation (anammox), and organic matter mineralization. Critically, seawater intrusion alters porewater chemistry, shifting redox zonation and thereby redirecting nitrogen transformation pathways (Fig. 6). Oxic to suboxic zones favor nitrification, whereas anoxic microenvironments support denitrification or anammox, processes contingent on organic carbon availability and microbial community composition. Human activities further amplify nutrient mobilization. Spatial co-location of NO3− hotspots with zones of intensive groundwater extraction (e.g., near Tianjin and Qingdao) demonstrates how anthropogenic pumping enhances contaminant transport and submarine groundwater discharge (SGD) fluxes[19,64,86]. In Liaodong Bay, SGD accounts for > 79% of dissolved inorganic nitrogen (DIN), dissolved inorganic phosphate (DIP), and silicate (DSi) inputs, exceeding riverine contributions in both magnitude and ecological significance[86]. Similar patterns are observed globally. In Lake Arend (Germany), SGD delivers up to 50% of total phosphorus loading, acting as a cryptic driver of eutrophication[87], while in the oligotrophic Mediterranean Sea, SGD (~68 km3 yr−1) constitutes the dominant terrestrial source of nutrients and trace metals, sustaining coastal productivity and ecosystem resilience[88].
Heavy metals: redox-mediated mobility and geochemical feedbacks
-
Heavy metal contamination in CMI groundwater arises from a confluence of industrial discharges, agricultural practices, and natural geogenic sources, with elevated risk concentrated in heavily industrialized and port-dominated regions such as Bohai Bay and Jiaozhou Bay. Key contributors include wastewater from petrochemical, metallurgical, and electronics manufacturing sectors, which release Pb, Cd, Hg, and As, as well as agrochemical inputs (e.g., Cd- and As-containing fertilizers) that leach into aquifers via infiltration and surface runoff. The mobility and toxicity of redox-sensitive metals, particularly As, Cr, Pb, and antimony (Sb), are governed by interlinked processes including redox transformations, adsorption/desorption equilibria, and solid-aqueous phase partitioning. These processes are highly responsive to salinity, oxygen availability, and microbial activity (Fig. 6). In tidal flats and estuarine aquifers, diurnal and seasonal fluctuations in hydraulic head generate sharp redox transitions that dictate metal speciation and solubility[89]. A pivotal conceptual advance is the recognition of the 'iron curtain' effect. The discharge of Fe(II)-rich groundwater into oxic seawater triggers rapid oxidation to Fe(III), precipitating amorphous iron (oxy)hydroxides that effectively scavenge oxyanions such as arsenate [As(V)] and chromate [Cr(VI)], thereby attenuating their seaward transport[90]. This natural attenuation mechanism highlights the importance of mixing zone geochemistry in contaminant immobilization. Conversely, under reducing conditions that are common in deep aquifers, reductive dissolution of Fe(III) (oxy)hydroxides liberates adsorbed As(III/V) into groundwater, driving elevated concentrations[91]. Additionally, colloidal transport and complexation with dissolved organic matter (DOM) can enhance metal mobility, facilitating off-site migration and increasing exposure risks[71,92]. The pronounced spatial heterogeneity in metal speciation and bioavailability underscores the necessity of integrating dynamic biogeochemical zonation into contaminant fate assessments at CMIs.
Sources of emerging contaminants and factors governing their transformation
-
Emerging contaminants exhibit distinct spatial distributions in CMIs, often reflecting proximity to point sources and the degree of hydrological connectivity. While LCs, such as heavy metals and nutrients, have been extensively characterized in these zones, ECs remain comparatively underexplored despite their escalating environmental prevalence and potential ecological risks. This section synthesizes recent advances in understanding the sources, transformation pathways, and environmental fate of ECs in CMIs, with particular emphasis on how their interactions with co-occurring pollutants amplify ecological hazards and perturb biogeochemical cycles.
Microplastics (MPs): subsurface transport, plastisphere dynamics, and reactive interfaces
-
Microplastics have become ubiquitous constituents of CMIs, accumulating in sediments, groundwater, and aquatic biota[22,93]. Exceptionally high concentrations, up to ~70,000 items kg−1, have been reported in sediments of the Wenruitang River, among the highest globally documented values, with larger fragments (> 300 μm) dominating in freshwater-influenced reaches[94]. Intriguingly, MP loads at urban sites do not consistently exceed basin-wide averages, suggesting that secondary tributaries may serve as disproportionately significant contributors relative to mainstem channels[94,95]. This non-uniform distribution underscores that MP deposition is governed not solely by emission proximity but also by a suite of environmental controls, including particle density, size, shape, biofouling, hydrodynamics, and biological uptake. Recent evidence identifies seawater intrusion (SWI) as a critical yet previously overlooked driver of MP migration, facilitating the landward transport of marine-derived plastics into coastal aquifers[96]. Moreover, inter-aquifer hydraulic exchanges can mobilize MPs into paleo-saline groundwater reservoirs, implying long-term subsurface retention and potential remobilization under altered hydrological regimes. These findings necessitate an expansion of traditional MP transport models to explicitly incorporate subsurface hydrogeological dynamics, a dimension largely absent from earlier syntheses.
Beyond physical transport, MPs function as reactive substrates and vectors for co-contaminants. The 'plastisphere', a distinct microbial biofilm colonizing plastic surfaces, plays a pivotal role in modulating both MP degradation and the fate of adsorbed pollutants. Organohalogen compounds sorbed onto MPs selectively enrich organohalide-respiring bacteria, reshaping microbial community structure, interaction networks, and functional gene expression within the plastisphere[97]. These consortia exhibit reductive dehalogenation capacity, offering novel insights into the biotransformation of halogenated pollutants in redox-stratified CMIs. Furthermore, synergistic interactions between microbial metabolites and iron-bearing minerals can generate reactive oxygen species (ROS), which accelerate the oxidative breakdown of organic pollutants. In iron-rich coastal wetlands and groundwater systems, iron (oxyhydr)oxide coatings ('iron plaques') form on sediment and particle surfaces under elevated salinity, anaerobiosis, the presence of natural organic matter (NOM), and weakly alkaline pH (8.0–8.3). Experimental studies demonstrate that such plaques enhance MP adhesion by approximately fivefold and promote polymer matrix oxidation, thereby accelerating fragmentation and the release of sequestered contaminants[98]. Consequently, iron plaque formation represents a key biogeochemical control on MP persistence, transformation, and ecotoxicological potential in intertidal environments.
Per- and poly-fluoroalkyl substances (PFAS)
-
Per- and poly-fluoroalkyl substances constitute a class of highly persistent, bioaccumulative, and toxic ECs increasingly detected across CMIs[99]. Wastewater effluents represent a dominant input pathway, with widespread contamination documented in both surface water and groundwater[100]. For instance, Sharma et al. reported PFHxA and PFBS concentrations reaching 4.7 and 10.2 ng L−1, respectively, in the Ganges River basin, with comparable levels in surface and groundwater, highlighting extensive aquifer infiltration driven by anthropogenic discharges[101]. The environmental behavior of PFAS in CMIs is primarily governed by sorption to organic carbon-rich particles and colloidal phases. Longer-chain homologues (e.g., PFOA) exhibit stronger affinity than short-chain analogues (e.g., PFBA), owing to enhanced van der Waals forces, hydrogen bonding, and cation bridging with divalent metals (e.g., Ca2+, Mg2+)[102]. Regulatory phase-outs of long-chain PFAS have prompted industrial shifts toward shorter-chain alternatives, which display greater aqueous mobility and reduced sorption, thereby altering vertical and lateral distribution patterns across salinity gradients and complicating risk assessment and remediation strategies[103]. Vegetated intertidal zones can act as natural 'estuarine filters', attenuating PFAS export through mechanisms including redox-driven precipitation, adsorption onto particulate organic carbon (POC), and colloid retention. However, the efficiency of these processes is highly sensitive to dynamic environmental variables, such as salinity, dissolved oxygen, and DOM composition, all of which vary spatiotemporally across CMIs. A mechanistic understanding of how these factors regulate PFAS speciation, partitioning, and bioavailability remains a critical knowledge gap, particularly in the context of climate-driven hydrological change.
Antibiotics and antibiotic resistance genes (ARGs)
-
The global surge in antibiotic use in human medicine, veterinary practice, and aquaculture has led to pervasive contamination of aquatic systems, with ARGs now recognized as emerging contaminants of dual concern for ecosystem health and public safety[104]. Global human antibiotic consumption rose by 65% between 2000 and 2015, driven largely by rapid development in low- and middle-income countries, while high per capita usage persists in high-income regions due to regulatory and behavioral challenges[105]. China, the world's largest producer and consumer of antibiotics, used approximately 10,125 tons annually by 2020, accounting for nearly half of the global total[106,107], resulting in the widespread detection of antibiotics (ng L−1 to μg L−1)[107]. In the Yellow River Delta, 12 and 18 distinct antibiotics were identified in water and suspended particulate matter (SPM), respectively, at concentrations ranging from 151 to 4.72 × 103 ng L−1, and 118 to 8.84 × 103 ng g−1[108]. The strong correlation between aqueous and particulate-phase trends indicates that SPM acts not only as a temporary sink but also as a potential secondary source during hydrodynamic disturbances such as tides or storms.
Since the onset of mass antibiotic production in the 1940s, ARGs have disseminated widely via horizontal gene transfer (HGT), a process amplified by selective pressures from residual antibiotics and co-occurring pollutants[109]. Rivers serve as primary conduits, transporting ARGs to estuarine and coastal zones where microbial communities face complex multi-stressor environments[110]. Estuarine sediments harbor diverse and abundant ARGs, including those conferring resistance to clinically critical antibiotics (e.g., β-lactams, tetracyclines, and sulfonamides)[111]. Mariculture activities further exacerbate this issue: nutrient enrichment, heavy metal inputs, and shifts in bacterial community composition collectively drive ARG proliferation (Fig. 7)[112]. Notably, co-contaminants such as heavy metals and organic pollutants can induce HGT and co-select for multidrug-resistant bacteria, even in the absence of direct antibiotic exposure[113]. While seawater dilution may reduce ARG abundance in some settings, persistent trace-level contaminants can sustain selective pressures, counteracting dilution effects and maintaining or even amplifying resistance gene loads in coastal receiving waters[114]. This multi-stressor-mediated evolution of microbial communities represents a fundamental mechanism underlying the emergence and spread of antimicrobial resistance in dynamic coastal systems. Future research should therefore prioritize understanding the hydro-biogeochemical processes that govern ARG persistence, mobility, and ecological risk in CMIs.
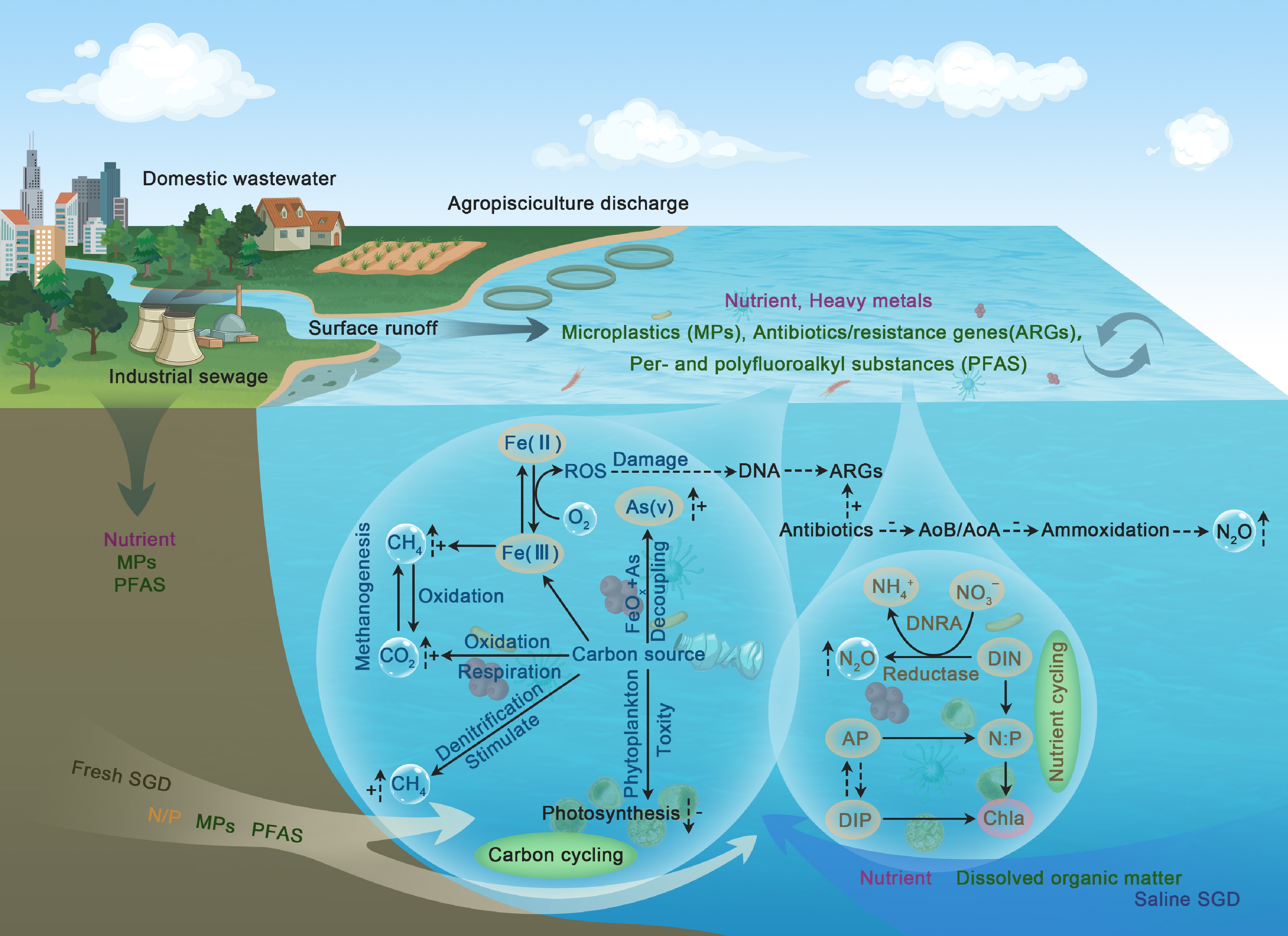
Figure 7.
Pollutant-driven perturbations to biogeochemical cycles in coastal-marine interfaces (CMIs): Conceptual framework linking emerging (ECs) and legacy contaminants (LCs). Terrestrial sources deliver co-contaminants to coastal waters, with fresh/saline submarine groundwater discharge (SGD) being a key land-sea vector for cross-medium transport. Iron (Fe) redox cycling mediates reactive oxygen species (ROS) production: Fe(II)-O2-derived ROS drive ARG proliferation (DNA damage) and As mobilization. Pollutants stimulate microbial methanogenesis/denitrification but inhibit phytoplankton photosynthesis, disrupting carbon cycling via biotic-abiotic stress coupling. Antibiotics suppress ammonia-oxidizing microbes (AOB/AOA), shifting nitrogen metabolism toward N2O-producing ammoxidation and nutrient-retaining DNRA. Alkaline phosphatase (AP)/chlorophyll a (Chla) tracks pollutant-driven N : P imbalance, regulating DIN/DIP cycling via pollutant-nutrient co-limitation.
Synergistic and antagonistic interactions between emerging and legacy contaminants
Cross-coupling interactions: integrating nutrients, metals, and environmental drivers
-
The soil-sediment-groundwater continuum governs the transformation and export of contaminants through microbially driven redox processes, sorption, and degradation[89]. Colloidal organic matter (COM) plays a pivotal role in metal transport. Sañudo-Wilhelmy et al. reported that 22%–96% of dissolved trace metals, and ~40% of organic carbon in coastal bays exists in colloid-associated forms, facilitating translocation across redox boundaries and enhancing bioavailability[115]. Mercury (Hg) cycling exemplifies such cross-coupling. Despite elevated total Hg (up to 96.3 ± 59.3 ng g−1), and methylmercury (MeHg, up to 0.12 ± 0.12 ng g−1) in coastal wetlands, methylation is tightly regulated by soil organic carbon (TOC), pH, and salinity, all of which are factors influencing the microbial community composition (e.g., sulfate-reducing bacteria) and Hg speciation[116]. Conversely, eutrophication-driven hypoxia promotes reductive dissolution of Fe(III) and Mn(IV) oxides, releasing adsorbed arsenic (As) into groundwater[117,118]. Under suboxic conditions, nitrate (NO3−) serves as an electron acceptor for denitrification and simultaneously drives the abiotic oxidation of As(III) to As(V), which exhibits a stronger affinity for iron oxide surfaces[91]. This NO3−-mediated oxidation enhances As immobilization, particularly in aquifers influenced by agricultural or urban recharge. Moreover, NO3− can oxidize As-bearing sulfide minerals (e.g., pyrite, greigite), transiently releasing As before its re-sequestration by newly precipitated Fe oxides, thereby altering long-term As speciation and mobility[119]. These mechanisms extend beyond classical models of As behavior, highlighting the critical role of nutrient-metal-redox coupling in CMIs[120].
Seawater intrusion, intensified by groundwater over-extraction and sea-level rise, further destabilizes metal retention. Elevated ionic strength induces competitive desorption of metals from sediment surfaces, while sulfate reduction fosters the precipitation of iron sulfides (e.g., pyrite), which are less effective at sequestering As than Fe(III) oxides (Fig. 7)[91]. Concurrently, groundwater table fluctuations (GTF) trigger physical and chemical perturbations that amplify contaminant mobilization: GTF enhances colloid detachment from pore surfaces, facilitating the co-transport of nutrients, metals, and organic matter[121]. Associated shifts in reactive oxygen species (ROS) expand redox heterogeneity, increasing the spatial extent and flux of contamination to coastal waters. Nutrient enrichment further complicates metal biogeochemistry. In eutrophic systems, protein-like dissolved organic matter (DOM) increases complexation capacity for metals such as Cd and Zn. Simultaneously, competition between silicate and selenium for diatom uptake can lead to antagonistic assimilation of trace elements, thereby altering trophic transfer[122]. Climate change acts as a meta-driver, modifying precipitation regimes, DOM composition in peatlands, and salinity intrusion patterns, all of which influence contaminant speciation, mobility, and ecological risk[123]. Nevertheless, critical knowledge gaps persist regarding the interactive effects of GTF, salinity, cations, and colloids on coupled contaminant transport in coastal groundwater systems.
Critical pathways regulating the transformation of emerging contaminants
-
Submarine groundwater discharge has emerged as a significant yet underquantified conduit for EC transport from land to sea. Although volumetrically minor compared to riverine inputs, SGD delivers persistent and bioactive pollutants, including pharmaceuticals, endocrine disruptors, MPs, PFAS, and ARGs, continuously and diffusely into sensitive coastal ecosystems[124]. Many ECs exhibit strong sorption under freshwater or brackish conditions[125], leading to their accumulation within subterranean estuaries. These reservoirs can episodically release stored contaminants during SGD events, resulting in delayed but prolonged ecosystem exposure and complicating source attribution and risk assessment. Of particular concern are SGD's roles in ARG dissemination, a pathway historically underestimated in marine antimicrobial resistance dynamics[124]. Given the frequent co-occurrence of antibiotic residues and resistant microbes in impacted coastal aquifers, SGD may act synergistically to amplify pulses of co-contamination, potentially inducing long-term shifts in microbial community structure and threatening aquatic health.
Critically, EC fate is modulated by interactions with LCs. Arsenic species (As[III], As[V]) alter MP surface charge, enhancing their colloidal stability and subsurface mobility[126]. Conversely, negatively charged MPs compete with As(V) for binding sites on iron (hydr)oxides, promoting arsenic desorption[92]. Moreover, MPs induce electrostatic repulsion that destabilizes Fe-rich colloids, facilitating the transport of particle-bound arsenic, which is a previously unrecognized linkage between plastic pollution and metalloid cycling. Similarly, divalent cations (e.g., Cu2+ and Cd2+) enhance PFOS and PFDA adsorption via ternary complexation, illustrating how LCs can act as geochemical modifiers that either immobilize or mobilize ECs depending on local conditions[127]. These findings underscore the inadequacy of single-pollutant frameworks in predicting environmental risk. A holistic approach integrating multi-contaminant interactions, dynamic redox transitions, and matrix-specific sorption is essential to unravel the complexity of contaminant transformations in CMIs, particularly for refining predictive hydrological models and informing management of stressed coastal aquifers.
Multi-contaminant interactions as emerging controls on C-N-P-S-Si cycling under climate change
-
CMIs serve as biogeochemical reactors where terrestrial and marine processes converge, regulating the cycling of C, N, P, S, and Si. Steep gradients in redox potential, salinity, and microbial activity create hotspots for coupled contaminant transformation and elemental cycling (Fig. 7). Anthropogenic nutrient loading from agricultural runoff, wastewater infiltration, and septic systems has heightened eutrophication risks along 14%–26% of global coastlines, particularly in ecologically sensitive regions such as coral reefs and estuaries[128]. SGD is a major, often overlooked, vector for dissolved inorganic nitrogen (DIN), phosphate, and silicate delivery to coastal oceans, directly influencing primary productivity[129]. Notably, SGD-derived dissolved silicate (DSi) sustains diatom growth through both regeneration of biogenic silica and mineral dissolution, thereby maintaining Si cycling in coastal food webs[42]. Subterranean estuaries host active microbial communities capable of attenuating contaminants prior to oceanic discharge[3,130], yet sustained nutrient inputs can overwhelm these natural buffers, triggering harmful algal blooms, hypoxia, and reduced carbonate buffering capacity[86]. Climate change intensifies these pressures as extreme precipitation and storm events amplify both surface runoff and SGD-driven nutrient pulses[129]. Furthermore, legacy effects from historical land use introduce time-lagged nutrient releases, complicating contemporary management. This review emphasizes SGD's critical role in linking terrestrial and marine nutrient cycles, offering a paradigm shift from surface-centric to integrated subsurface-surface perspectives.
Emerging contaminants further perturb these cycles. MPs in sediments enhance labile carbon availability and foster anaerobic microzones, stimulating denitrification and modulating C-N coupling[25,131]. The effect of MPs on greenhouse gas emissions varies with polymer composition and environmental context[132]. Polymer type dictates greenhouse gas outcomes. Polyethylene (PE) MPs suppress CH4 (> 40%) and CO2 (~26%) emissions, likely via altered sedimentary C chemistry[133]. In contrast, biodegradable polylactic acid (PLA) MPs promote sulfate reduction in mangroves, generating sulfide and precipitating Fe(II), processes that may constrain iron cycling and carbon sequestration[134]. Although MPs contribute modestly (0.001%–1.197%) to sedimentary carbon pools[22], their cumulative influence on carbon budgets warrants attention. PFAS compounds disrupt microbial nitrification and anammox pathways by altering community structure and inhibiting key functional groups[135]. While direct evidence in CMIs remains sparse, marine PFAS exposure correlates with impaired phytoplankton photosynthesis, shifted microbial functionality, and reduced carbon sequestration, suggesting indirect but significant feedbacks to oceanic carbon cycling and climate regulation[136]. Antibiotics primarily interfere with N cycling. Sulfamethazine (SMT) inhibits denitrification in estuarine sediments, elevating N2O emissions and compromising eutrophication control[137]. Paradoxically, high SMT concentrations may stimulate nitrite-dependent anaerobic methane oxidation (n-DAMO) by enhancing methanogen activity[138]. Differential impacts on ammonia oxidizers are also evident. Streptomycin sulfate reduces total ammonia oxidation by 61.2%–84.0%, while penicillin causes 50.0%–74.5% inhibition, attributed to distinct modes of action and, in penicillin's case, possible facilitation of archaeal cooperation[139]. Under co-exposure scenarios (e.g., MPs + antibiotics), elevated ROS, DNA damage, and microbial stress responses accelerate horizontal gene transfer of ARGs. MP presence further drives deterministic assembly of nitrogen-cycling microbiomes and ARG hosts, amplifying ecological risks[140].
-
This review consolidates recent advances in understanding the fate and interactions of contaminant mixtures at coastal-marine interfaces (CMIs), with emphasis on the dynamic interplay between emerging contaminants (ECs) and legacy pollutants (LCs). By synthesizing bibliometric trends, geochemical evidence, and process-level insights, an integrative conceptual framework is proposed that links anthropogenic drivers, such as industrialization, intensive agriculture, and aquaculture, with natural biogeochemical regulators, including redox gradients, salinity transitions, organic matter lability, and mineral surface reactivity. This framework elucidates how these coupled forces govern the co-transport, transformation, and ultimate fate of complex contaminant assemblages in CMIs. The present synthesis demonstrates that competitive and cooperative interactions between ECs and LCs, particularly on reactive surfaces such as Fe (hydr)oxides, can profoundly alter contaminant speciation, mobility, and bioavailability, thereby exerting cascading effects on key elemental cycles (C, N, P, S, Si). Submarine groundwater discharge (SGD) emerges as a critical, yet often underestimated, conduit that bridges terrestrial contaminant sources with coastal oceans, delivering not only nutrients and metals but also persistent organic pollutants, microplastics, antibiotics, per- and poly-fluoroalkyl substances, and antibiotic resistance genes. Concurrently, natural coastal ecosystems including mangroves, salt marshes, and seagrass meadows, serve as vital biogeochemical buffers, attenuating pollution through microbial degradation, physical filtration, and sorptive retention. Given the dual role of CMIs as biodiversity hotspots, blue carbon reservoirs, and frontline filters against land-derived pollution, their integrity is central to both ecological resilience and climate mitigation. To advance predictive understanding and inform effective management, five priority research directions are identified:
(1) Cross-compartmental integration of contaminant dynamics. Most existing studies remain confined to isolated environmental compartments (e.g., water column or sediment). Future work must adopt holistic, multi-media approaches that explicitly track contaminant fluxes and transformations across groundwater, surface water, sediment, and biota. Such efforts should incorporate the functional roles of vegetation, microbial consortia, and reactive mineral phases in mediating adsorption, biodegradation, and ecotoxicological outcomes.
(2) Elucidating multi-pollutant interaction mechanisms under dynamic conditions. Persistent organic pollutants and heavy metals continue to accumulate in CMIs due to their environmental persistence and toxicity. Their interactions with ECs, whether synergistic, antagonistic, or additive, during transport and transformation remain poorly constrained, especially under fluctuating hydrogeochemical regimes (e.g., tidal pumping, redox oscillations, salinity intrusion). Mechanistic studies under environmentally realistic conditions are urgently needed.
(3) Assessing the biogeochemical footprint of microplastics in subterranean estuaries. While seawater intrusion may facilitate MP infiltration into coastal aquifers, the long-term consequences of MP accumulation on microbial community structure, enzymatic activity, and elemental cycling (particularly C, N, P, and Si) in subterranean estuaries are largely unknown. A combination of long-term field monitoring and controlled mesocosm experiments is essential to quantify these impacts and identify potential tipping points.
(4) Expanding the scope of emerging contaminant characterization and impact assessment. ECs, including MPs, antibiotics, and PFAS, are increasingly recognized not merely as passive pollutants but as active participants in biogeochemical processes, either through their chemical backbones, associated additives, or transformation byproducts. Systematic identification of novel contaminants, coupled with the development of high-resolution analytical tools (e.g., non-target screening, molecular characterization), is crucial for mapping transformation pathways and ecological risks. Equally important is evaluating their effects on organismal physiology, microbial metabolism, and feedbacks to global biogeochemical cycles.
(5) Bridging the gap between field observation and mechanistic experimentation. A persistent disconnect exists between field-based surveys often lack mechanistic resolution, and laboratory studies may oversimplify environmental complexity. Future research should integrate high-frequency in situ monitoring with advanced molecular techniques (e.g., metagenomics, metatranscriptomics, stable isotope probing) under variable salinity, organic matter, and redox conditions to resolve the biochemical underpinnings of pollutant-microbe interactions.
Finally, there is an urgent need to develop coupled reactive transport models that simulate multi-pollutant behavior across the groundwater-seawater continuum. Integrating these models with machine learning algorithms and scenario-based projections will enhance our capacity to forecast CMI contamination risks under future climate trajectories, including sea-level rise, intensified storm events, and altered freshwater recharge patterns. Such predictive capabilities are indispensable for designing adaptive management strategies that safeguard the ecological and biogeochemical functions of these vulnerable yet invaluable transition zones.
-
It accompanies this paper at: https://doi.org/10.48130/newcontam-0026-0006.
-
The authors confirm their contributions to the paper as follows: Linan Liu: conceptualization, writing-original draft, funding acquisition. Yizi Hua: data curation, formal analysis, methodology; Gang Wang: conceptualization, writing−review and editing; Honghong Lyu: conceptualization, investigation, methodology; Qiang Li: conceptualization, data curation, formal analysis; Jingmin Sun: conceptualization, investigation, writing−review and editing; Yuguo Zhuo: investigation, writing−review and editing; Xin He: investigation, writing−review and editing; Jingchun Tang: supervision, conceptualization, funding acquisition, writing−review and editing. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets used or analyzed during the current study are available from the corresponding author upon reasonable requests.
-
This work was supported by the National Key Research and Development Program of China (Grant No. 2023YFC3709002), Marine Ecological Restoration and Smart Ocean Engineering Research Center of Hebei Province (Grant No. HBMESO2509), the National Natural Science Foundation of China (Grant Nos 42477524, 42407645, 42377225), the Key R&D Program of Tianjin (Grant No. 23YFXTHZ00170), and the 111 Program, Ministry of Education, China (Grant No. B17025).
-
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
-
# Authors contributed equally: Linan Liu, Yizi Hua
Full list of author information is available at the end of the article. - The supplementary files can be downloaded from here.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Liu L, Hua Y, Wang G, Lyu H, Li Q, et al. 2026. Unveiling the hidden ecological risks of emerging entangled with legacy pollutants within coastal-marine interfaces. New Contaminants 2: e008 doi: 10.48130/newcontam-0026-0006
Unveiling the hidden ecological risks of emerging entangled with legacy pollutants within coastal-marine interfaces
- Received: 03 November 2025
- Revised: 26 December 2025
- Accepted: 14 January 2026
- Published online: 11 March 2026
Abstract: Coastal-marine interfaces (CMIs) serve as critical biogeochemical transition zones, where terrestrial and oceanic systems intersect, and legacy and emerging contaminants frequently interact through sorption/desorption, precipitation/dissolution, and redox-mediated processes. However, the mechanisms by which these interactions perturb elemental cycling remain fragmented. This study conducts a comprehensive bibliometric synthesis of more than 5,000 peer-reviewed studies, integrating co-citation networks, keyword co-occurrence analysis, and spatial hotspot mapping, with a focused assessment of China's heavily contaminated coastal aquifers. The present analysis reveals that the co-transport and transformation of legacy and emerging contaminants in CMIs are jointly governed by hydrodynamic forcing, mineral surface reactivity, and redox gradients. Competitive interactions at Fe/Mn (hydr)oxide surfaces can enhance the mobility of trace metals such as Pb2+ and Cd2+. Microplastics function as reactive vectors that facilitate metal transport, while antibiotics disrupt key microbial guilds, particularly denitrifiers, thereby altering the cycling of carbon, nitrogen, phosphorus, and silicon. Moreover, redox transitions driven by submarine groundwater discharge (SGD), as freshwater mixes with saline porewater, induce reductive dissolution of Fe(III) oxides, mobilizing arsenic and amplifying nitrate fluxes to coastal waters. Three priority knowledge gaps are highlighted: (1) the interfacial processes linking microbes, organic matter, and minerals that control cross-media contaminant transfer; (2) the quantitative contribution of SGD to marine elemental budgets and the associated in situ geochemical transformations within subterranean estuaries; and (3) the feedback between climate change and multi-contaminant stress on coupled biogeochemical cycles. This review establishes a process-oriented framework for future research integrating biogeochemical and ecological dimensions in anthropogenically pressured CMIs.