-
Pharmaceuticals and personal care products (PPCPs) are a large group of chemical substances that are used in medicine, healthcare, and personal hygiene and have been recognized as contaminants of emerging concern[1]. They have been detected in both tap water and several environmental matrices, such as soil, surface waters, groundwater, sediments, and the atmosphere[1], as a result of inappropriate disposal of industrial and domestic effluents in the environment and irregular disposal of garbage, which leads to leaching into the soil and groundwater. In coastal zones, PPCPs in environmental matrices are primarily associated with the disposal of both treated and untreated sewage through submarine outfalls[2,3] and the direct discharge of stormwater. These chemicals have been reported in coastal waters worldwide, such as Costa Rica[4], Taiwan[5], Singapore[6], Malaysia[7], Sri Lanka[8], and Brazil[9,10]. Moreover, because of their continuous usage and release into the environment, most PPCPs tend to occur at high concentrations, despite their half-lives, degradation rates, or dilution. Therefore, PPCPs have been considered to be pseudo-persistent contaminants[3]. Thus, in recent years, there has been increasing concern about the potential environmental risks that PPCPs may pose on aquatic biota and people living in coastal areas, especially in metropolitan zones and densely occupied areas[3].
In December 2019, the COVID-19 pandemic, caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), allegedly started in China and rapidly spread around the world. Because of the lack of vaccines against COVID-19, the rapid dissemination of the virus, and its severe impacts on human health, many drugs were used by the public in attempts to prevent or treat COVID-19[11,12]. These drugs include pharmaceuticals recommended by health authorities such as the World Health Organization (WHO) and the US Center for Disease Control and Prevention (CDC)[13], as was the case of loratadine and betamethasone, and those that became popular through misinformation or political narratives, such as chloroquine, hydroxychloroquine, and ivermectin[14], which were widely used, particularly in Brazil, India, and the USA[14−17]. For example, in Brazil, the sales of these drugs increased by more than 50% compared with 2019[14]. Various authors also stated that because of the inefficiency of municipal sanitation systems, particularly sewage collection and treatment, the release of these pharmaceuticals increased, along with their concentrations in the environment.
Loratadine is a second-generation antihistaminic drug used to treat allergies and inflammation[18]; it is not fully eliminated by conventional wastewater treatments and is found in aquatic environments at concentrations up to 81 ng/L[19]. Betamethasone is a synthetic glucocorticosteroid that mimics the action of cortisol, which is prescribed as an anti-inflammatory drug[20], similar to dexamethasone. Glucocorticosteroids have been associated with adverse effects in aquatic organisms[21−25] at environmental concentrations[26], including endocrine disruption in fish[20]. Hydroxychloroquine is a by-product derived from chloroquine (CQ)[27], and both are used to treat malaria and rheumatic and dermatologic disorders of autoimmune origin[28,29]. Information on the toxicity of hydroxychloroquine in aquatic organisms is limited, but there is evidence that it is toxic to freshwater[30] and marine[31] organisms. Nitazoxanide is a broad-spectrum drug used to treat a wide range of parasites[32]. It is an inhibitor of pyruvate/ferrodoxin oxidoreductases, preventing the binding of pyruvate with thiamine pyrophosphate, which is a critical step in the anaerobic metabolism of protozoa[32]. This compound inhibits the growth of the microalgae Euglena gracilis[33] and causes adverse effects in zebrafish (Dania rerio) embryos[34]. However, there is no information regarding its toxicity to marine organisms. Ivermectin is an antihelminthic drug that is widely used and is considered very toxic to a range of aquatic organisms[35−43]; however, its effects on marine organisms are still poorly understood. The existing literature regarding the environmental distribution of these substances and their toxicity to aquatic organisms is limited[18−43], despite the widespread use of these drugs. Thus, studies on their effects on aquatic life have become crucial for understanding their environmental impact and should be prioritized[14]. In this study, we analyzed the effects of five drugs widely used during the COVID-19 pandemic on the brine shrimp Artemia salina, providing information to understand how the pandemic could have affected coastal ecosystems.
-
Five compounds were selected for assessment in this investigation, which used high-purity substances purchased from their respective manufacturers: loratadine (CAS #79794-75-5; molecular formula, C22H23ClN2O2; Sigma-Aldrich), betamethasone (CAS #378-44-9; molecular formula, C22H29FO5; Chemical Synthesis), hydroxychloroquine (CAS #118-42-3; molecular formula, C18H26CIN3O; Sigma-Aldrich), ivermectin (CAS #70288-86-7; molecular formula, C48H74O14; Chemical Synthesis), and nitazoxanide (CAS #55981-09-4; molecular formula, C12H9N3O5S). Loratadine and betamethasone have been recommended by health authorities[13] and are used to treat patients infected with COVID-19. In turn, hydroxychloroquine, ivermectin, and nitazoxanide were shown to be ineffective against COVID-19 but were still largely used in some countries because of widespread disinformation campaigns spread through social networks[14−17].
The test concentrations for each substance were selected according to their reported environmental concentrations and the toxicities observed in freshwater organisms[17−43]. They were prepared by diluting the drugs, in powder form, in 1.0 mL of acetone and 99 mL of clean filtered seawater, followed by stirring until all the salts dissolved entirely and no precipitates could be observed in the solutions. The following nominal test solutions were prepared: 0.02, 0.2, 2, and 20 mg/L loratadine; 4, 40, 400, and 4,000 μg/L betamethasone; 0.1, 1, 10, and 100 mg/L hydroxychloroquine; 1, 10, 100, and 1,000 μg/L ivermectin; and 0.05, 0.5, 5, and 50 mg/L nitazoxanide. The control consisted of a 1% acetone solution prepared with clean filtered seawater. Previous data from our laboratory (unpublished data) showed that this concentration of acetone is not toxic to nauplii of Artemia salina, whereas the literature indicates that other marine organisms are not affected by acetone at this and lower concentrations[2,3,7,10]. Quality assurance/quality control (QA/QC) procedures during the toxicity tests consisted of using clean glassware, checking the physicochemical parameters of the test solutions, and using adequate controls. Moreover, the laboratory monitors the sensitivity of the test organism by conducting a toxicity test with a reference toxicant every month and comparing the results with a control chart.
Nauplii of Artemia salina were hatched from dehydrated cysts obtained commercially. Two days before the experiment, the cysts were introduced into a glass beaker containing 0.5 L of clean filtered seawater (salinity: 35‰) and kept under aeration at 25 °C. The nauplii hatched the following day, and after another 24 h, they were used in toxicity tests[44,45]. The acute toxicity test with A. salina nauplii was performed following the protocol described by ABNT NBR 16530[44]. The test was carried out using 15-mL glass test tubes as replicates, each containing 10 mL of the test solution at the abovementioned test concentrations. Four replicates were prepared for each concentration. Ten nauplii were selected and introduced into each replicate using a dropper. At the beginning of the toxicity tests, pH, dissolved oxygen (DO), temperature, and salinity were measured for each test solution as well as for the control; temperature was also monitored every hour. The experiment consisted of exposing the organisms to the solutions for 48 h under controlled laboratory conditions (temperature 25 ± 2 °C; photoperiod, 16:8 light : dark; lack of additional food). After 48 h, the organisms in each test tube were analyzed under a microscope and the survivors were counted.
The test results were initially checked for normality and variance homogeneity using Shapiro–Wilk and Levene's tests, respectively. Next, they were analyzed by one-way analysis of variance (ANOVA), followed by Dunnett's t-test, using a 95% significance level (p = 0.05), in order to determine the lowest observed effect concentration (LOEC) and the no observed effect concentration (NOEC)[44,45]. The lethal concentration for 50% of organisms after 48 h of exposure (LC50-48 h) was calculated by the probit method, using Statistica 6.0 software.
-
The results of the toxicity tests are shown in Fig. 1 and Table 1. Figure 1 shows the survival rates of A. salina nauplii exposed to the tested pharmaceuticals, and the physicochemical parameters of the tested solutions are presented in Table 1. The physicochemical parameters were within the recommended ranges for all treatments, according to the test protocol for A. salina[44,45]. Thus, these factors did not interfere with the survival of the organisms.
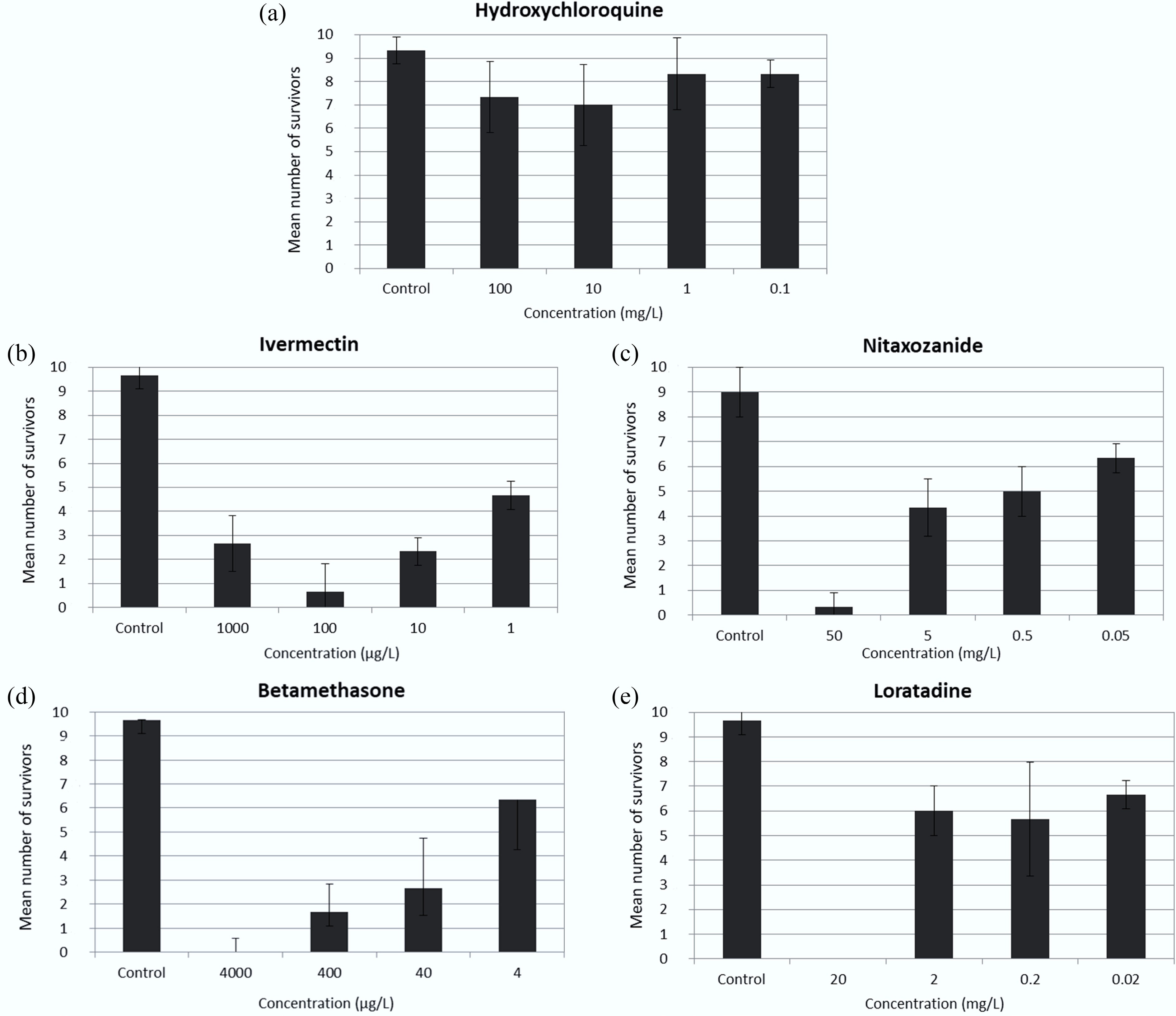
Figure 1.
Mean survival rates of nauplii of Artemia salina exposed to COVID-19 drugs: (a) Hydroxychloroquine, (b) ivermectin, (c) nitazoxanide, (d) betamethasone, and (e) loratadine) for 48 h. The control treatment consists of a 1.0% acetone solution. Error bars indicate standard deviations.
Table 1. Physicochemical parameter of the tested solutions in the toxicity tests with pharmaceuticals used to treat COVID-19
Hydroxychloroquine (mg/L) Nitazoxanide (mg/L) Ivermectin (μg/L) Loratadine (mg/L) Betamethasone (μg/L) Control 100 10 1 0.1 Control 50 5 0.5 0.05 Control 1,000 100 10 1 Control 20 2 0.2 0.02 Control 4,000 400 40 4 Salinity 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 35 pH 8.1 7.9 7.9 8 8 8.2 7.7 7.7 7.9 7.9 8.2 7.7 7.9 8 8 8.1 8 8 8.1 8 8.1 8 8.1 8 8.1 Temperature (°C) 25 25 25 25 25 25 25 25 25 25 25 25 25 25 25 25 25 25 25 25 25 25 25 25 25 D.O.
(% saturation)92 88 91 92 92 95 80 85 89 90 93 86 89 91 91 91 90 93 87 88 90 82 91 92 90 Hydroxychloroquine showed no toxicity to A. salina nauplii at the tested concentrations. On the other hand, the other four compounds were toxic at all concentrations tested (Fig. 1, Table 2). The LC50-48 h of loratadine was 0.53 mg/L, whereas the LOEC was estimated at 0.02 mg/L. For betamethasone, the LOEC was 4 μg/L and the LC50-48 h was 33.64 μg/L (range: 22.25–50.86); for this compound, mortality was 100% at the highest tested concentration (4,000 μg/L). Regarding nitazoxanide, the LOEC was estimated as 0.05 mg/L, whereas the LC50-48 h was calculated as 0.64 mg/L (range: 0.21–1.93). Finally, the LOEC for ivermectin was estimated at 1 μg/L. The LC50-48 h could not be calculated, as mortality exceeded 50% at all tested concentrations.
Table 2. Lethal concentrations to 50% of organisms (LC50-48 h) and the lowest observed effect concentrations (LOECs) obtained for nauplii of Artemia salina exposed to COVID-19 pharmaceuticals
Test compound LC50-48 h LOEC Hydroxychloroquine Not toxic Not toxic Ivermectin NC 1.0 μg/L Nitazoxanide 0.64 (0.21–1.93) mg/L 0.05 mg/L Betamethasone 33.64 (22.25–50.86) μg/L 4.0 μg/L Loratadine 0.53 mg/L 0.02 mg/L The COVID-19 pandemic has affected populations worldwide, with more than 700 million people infected and > 7 million deaths[46]. Several pharmaceuticals have been proposed or used for treating COVID-19, initially reaching up to 700 to 1,000 potential drugs[47]. From this list, a smaller group remains, including drugs approved by health authorities, such as antibiotics, antiretroviral drugs, anti-inflammatory drugs, and analgesics. Additionally, drugs not officially recommended but widely used in some countries as a result of disinformation campaigns and generalized self-medication during lockdowns include veterinary medicines (e.g., ivermectin) and antimalarial drugs (e.g., hydroxychloroquine)[47,48]. For example, the increase in prescriptions and refills for hydroxychloroquine and chloroquine reported in the US in 2020 reached 1977%[49]. The consequence of this massive use of COVID-19-related drugs was an increase in the release of these substances or their by-products into the environment through wastewater treatment plants or direct discharge into water bodies[47,50,51], similar to those observed for the environmental levels of Tamiflu during the H1N1 outbreak[52]. Therefore, assessing the toxicity of COVID-19 drugs to aquatic organisms is crucial for understanding their environmental impact and the broader consequences of the COVID-19 pandemic. This study provides new insights into the toxicity of five pharmaceuticals used to treat COVID-19 in marine organisms during the SARS-CoV-2 pandemic.
Hydroxychloroquine did not cause toxicity to nauplii of A. salina at concentrations ranging between 0.1 and 100 mg/L, which are significantly higher than the environmental levels reported in the surface waters of Spain (32 ng/L)[48] and the predicted environmental concentration (78.3 ng/L)[48]. However, this range is similar to the concentrations reported in a wastewater treatment plant (0.11 mg/L) and river water (0.013–0.42 mg/L)[53]. Thus, according to our results and these environmental concentrations, acute toxicity to marine organisms would not be expected. However, Ramesh et al.[30] reported adverse biochemical effects in the fish Cyprinus carpio exposed to concentrations similar to those used in the present study. Information on hydroxychloroquine's toxicity to aquatic organisms is limited, and the existing results show varying responses. Zurita et al.[31] observed a lack of effects on the bacterium Vibrio fischeri and relevant toxicity to the cladoceran Daphnia magna, the fish Poeciliopsis lucida, and the microalgae Chrorella vulgaris. Moreover, the predicted no effect concentrations (PNECs) for this compound were 170 ng/L in river water[48], with values ranging from 7.5 ng/L to 85 μg/L[54−56]. In another study on meiobenthic nematodes, hydroxychloroquine caused effects at low concentrations[57]. However, more information is needed on the toxicity of hydroxychloroquine in marine organisms. The lack of toxic effects on A. salina, compared with the toxicity reported in the literature, suggests that A. salina is more resistant to hydroxychloroquine. Hydroxychloroquine is expected to be relatively persistent, with a half-life of 138 days[58], and its degradation products can be toxic[53]. However, more studies are necessary to determine the impact of hydroxychloroquine on marine organisms; in particular, studies focusing on chronic and subchronic effects are required.
This study is the first to investigate the toxicity of nitazoxanide to saltwater organisms. This antiparasite drug is widely used[32], and previous studies have indicated its toxicity to microalgae[33] and zebrafish embryos[34], and its effects involve the inhibition of the pyruvate : ferredoxin oxidoreductase (PFOR) enzyme-dependent electron transfer reactions necessary for anaerobic energy metabolism, inhibition of glutathione-S-transferase, and modulation of the glutamate-gated chloride ion channels[58,59]. In this study, nitazoxanide exhibited toxicity to A. salina at a concentration of 0.05 mg/L, with an LC50-48 h of 0.64 (0.21–1.93) mg/L. In a prospective study using ecological models[58], this compound was considered very toxic to freshwater organisms, with an expected half-life of 12 days. According to a quantitative structure–activity relationship (QSAR) model, the predicted ecotoxicity values (pEC50) for Pseudokirchneriella subcapitata, Daphnia magna, Oncorhynchus mykiss, and Pimephales promelas were 8.61, 5.10, 4.64, and 5.60 mol/L, respectively[59]. This compound was capable of causing oxidative stress and immunotoxicity in zebrafish embryos at concentrations from 0.75 mg/L[34]. In A. salina, lethal toxicity occurred at lower concentrations, but the effects of nitazoxanide on marine organisms remain understudied, similar to hydroxychloroquine.
Among the drugs evaluated in this study, ivermectin has been the most extensively studied. It is a macrocyclic lactone used in both veterinary and human medicine as an antiparasitic drug[50]. Up to 90% of the drug is excreted by the body through the feces and urine[50]; thus, ivermectin can be released into water bodies through sewage, agricultural leachates, and urban drainage, especially in developing countries. Ivermectin is persistent in the environment[60], with a half-life of over 100 days in marine sediments[61]. A previous study reported that approximately 31% of the initially applied ivermectin was transformed[62]. As a result, ivermectin concentrations were high in wastewater treatment plants[63], and this compound was detected in rivers at levels ranging from 5 to 20 ng/L and in irrigation waters at 0.093 μg/L before the COVID-19 pandemic[50,52].
According to Juarez et al.[64], ivermectin acts as an agonist of several types of ligand-gated chloride ion channel receptors. It increases the activity of γ-aminobutyric acid (GABA) receptors or glutamate-gated chloride ion channels (Glu-Cl), which signal between the neurons and muscles in parasites, leading to both ionic impairment in cells and increased paralysis in invertebrates[63,64], including crustaceans[65]. These findings are consistent with our results, in which ivermectin was highly toxic to A. salina nauplii, with an LOEC of 1 μg/L; moreover, mortalities were greater than 50% in all tested concentrations. In fact, ivermectin is considered to be highly toxic to freshwater organisms, such as microbes[36,41], microalgae[36,39], fish[36,38], cladocerans[36,37,39,42], and annelids[40]. Less information is available for marine organisms, but the existing data indicate high toxicity. A previous study reported acute toxicity in several marine organisms, with LC50 values ranging between 0.026 (0.013–0.051) μg/L for the mysid Neomysis integer (48 h) and > 10,000 μg/L for Hydrobia ulvae (96 h) and nematodes[35]. Another study reported an LC50-96 = 70 (44–96) ng/L for the mysid Neomysis integer[43]. The toxicities of ivermectin and other similar compounds of the avermectin family were studied by Blizzard et al.[66], who obtained an IC50 of 870 μg/L for A. salina, which was much higher than the values reported in our study and those reported in the literature for other organisms[35,43]. In addition, the ecological models used to predict environmental risks of ivermectin suggest very high risks[63], with toxic threshold levels ranging from 25 ng/L[37] to 1.5 μg/L[63]. Given its environmental concentrations and toxic thresholds, the environmental risks for ivermectin may occur along the coast of São Paulo.
There is limited information on the toxicity of loratadine and betamethasone to aquatic organisms, and data on their toxicity to marine organisms are lacking. In our study, loratadine caused adverse effects in A. salina at the lowest concentration tested (0.02 mg/L), with an LC50-48 h of 0.53 mg/L. If we compare our results with those obtained for freshwater organisms, A. salina was more sensitive than the water flea Ceriodaphnia dubia (LC50 = 0.60 mg/L), the rotifer Brachionus calcyflorus (LC50 = 17 mg/L), and the anostracan crustacean Thamnocephalus platyurus (LC50 = 50.70 mg/L)[18]. Actually, the LC50 obtained for A. salina was higher than the EC50 values obtained for C. dubia (28.14 μg/L) and B. calcyflorus (51.32 μg/L), and lower than that obtained for the microalgae P. subcapitata (2,155 μg/L)[18]. Moreover, the toxicity of loratadine to A. salina was similar to that of the anti-histaminic compound promethazine hydrochloride[67]. The LOEC for A. salina is lower than the maximum concentration reported in surface waters[19], suggesting potential environmental risks for loratadine. These risks may be aggravated by its degradation products, which can also be toxic[19] and are not fully removed by conventional wastewater treatments[19].
Betamethasone was more toxic than loratadine, with an estimated LOEC of 4 μg/L and an LC50 = 33.64 (22.25–50.86) μg/L. These toxic concentrations were higher than the environmental concentrations reported in rivers from the Czech Republic and Switzerland, which were in the range of 8–31 ng/L[68]. These were also higher than the PNEC proposed for endocrine disruption in freshwater fish[20,25,68]. Because little is known about the environmental levels and toxicity of betamethasone, we compared our results with those of dexamethasone, a parent compound that exhibits similar behavior. In this case, dexamethasone's environmental concentrations increased during and after the COVID-19 pandemic[26,50], but its acute toxicity to freshwater organisms tended to be lower than that of betamethasone[21,22,59]. Estimations of the probable effect concentrations of dexamethasone were variable, with values ranging between 0.29 ng/L[48] and 0.003 mg/L[63], which are also comparable with the LOEC estimated for betamethasone in the present study, yet the environmental risks were considered to be low by Tarazona et al.[63].
-
This study provides new information regarding the toxicity of nitazoxanide, hydroxychloroquine, ivermectin, loratadine, and betamethasone to an important aquatic euryhaline species, the brine shrimp A. salina. This species is globally distributed in lakes and coastal lagoons, and is abundant in hypersaline environments[69]. Most of these compounds caused toxicity in A. salina nauplii, except for hydroxychloroquine. The LC50 values ranked the drugs' toxicity as ivermectin > betamethasone > nitazoxanide = loratadine > hydroxychloroquine, but this concentration could not be calculated for ivermectin and hydroxychloroquine. On the basis of their respective LOECs, which represent the toxic threshold, the toxicity of the tested drugs can be ranked as follows: Ivermectin > betamethasone > loratadine > nitazoxanide > hydroxychloroquine. In this context, the increased use of these pharmaceuticals to treat or prevent COVID-19 likely contributed to higher levels of pharmaceuticals and personal care products in the environment.
More studies on these chemicals are necessary, particularly investigations aimed at detecting and quantifying COVID-19 drugs in marine environments[47]. Additionally, studies should focus on assessing their fate and behavior, bioavailability, bioaccumulation, toxicity, and environmental risks. In addition, effective strategies should be implemented to reduce or eliminate environmental contamination by COVID-19 drugs and their by-products. Such strategies involve improving sanitation systems by collecting domestic, hospital, and veterinary wastewater and adapting wastewater treatment plants to use a combination of advanced technologies for removing these drugs and other contaminants[47,50,52,63]. Some authors also recommend information campaigns aimed at avoiding the use of ineffective drugs by the population[63], as was the case for ivermectin, hydroxychloroquine, and nitazoxanide. O'Flynn et al.[70] highlighted the life cycle assessment of pharmaceuticals from production to release into the aquatic environment, with the purpose of tracking the route of pharmaceuticals into the environment and following the sustainable development targets of SDG 6 (Clean Water and Sanitation), SDG 12 (Responsible Consumption and Production), and SDG 14 (Life Below Water).
We thank Drs. Guacira Pauly (UNESP) and Fernando Perina (University of Aveiro) for technical support. DMSA thanks Instituto Nacional de Identificação, Quantificação, Dispersão, Riscos Ambientais e Mitigação da Poluição por Contaminantes Emergentes em Ambientes Marinhos e Costeiros (INCT-CEMAR) for the financial support. We also dedicate this manuscript to all those people lost during the COVID-19 pandemic and those professionals who dedicated their efforts to save lives all around the world.
-
The authors confirm their contributions to the paper as follows: Denis Moledo de Souza Abessa: conceptualization, methodology, validation, formal analysis, investigation, resources, writing − original draft preparation, supervision, funding acquisition. Maysa Ueda de Carvalho: methodology, investigation, writing − review and editing. Both authors have read and agreed to the published version of the manuscript.
-
All data generated or analyzed during this study are included in this published article.
-
This research was funded by the São Paulo Research Foundation (FAPESP) (Grant Nos. 2020/04779-6, 2022/09070-0, and 2025/27086-0), and the National Council for Scientific and Technological Development (CNPq) (Grant Nos. 308533/2018-6, 313420/2023-8, and 408782/2024-2).
-
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
-
Full list of author information is available at the end of the article.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
de Souza Abessa DM, de Carvalho MU. 2026. Toxic effects of drugs used to treat COVID-19 on Artemia salina. New Contaminants 2: e013 doi: 10.48130/newcontam-0026-0012
Toxic effects of drugs used to treat COVID-19 on Artemia salina
- Received: 06 January 2026
- Revised: 01 April 2026
- Accepted: 10 April 2026
- Published online: 27 April 2026
Abstract: During the COVID-19 pandemic, several drugs were extensively used to treat and prevent infections worldwide, increasing their release into the aquatic environment. However, the effects of COVID-19 drugs on marine organisms are poorly understood; therefore, studies on this topic are required. This study aimed to evaluate the acute toxicity of hydroxychloroquine, ivermectin, nitazoxanide, loratadine, and betamethasone in nauplii of the brine shrimp Artemia salina. Acute toxicity tests were conducted following established protocols for this species (NBR 16530) for 48 h of exposure, using a range of test concentrations for each substance, defined according to reported environmental concentrations or toxicity for other species. Hydroxychloroquine was not toxic at the tested concentrations; thus this drug does not pose a risk to A. salina. The other drugs caused significant mortality in brine shrimps. The lowest observed effect concentrations (LOECs) were as follows: Ivermectin, 1.0 μg/L; nitazoxanide, 0.05 mg/L; betamethasone, 4 μg/L; loratadine, 0.02 mg/L. The toxicity of the tested drugs to A. salina was ranked as ivermectin > betamethasone > loratadine > nitazoxanide > hydroxychloroquine. Further studies with marine species are necessary to determine the environmental risks posed by the drugs tested in this study.
-
Key words:
- Marine ecotoxicology /
- COVID-19 drugs /
- Hydroxychloroquine /
- Ivermectin /
- Betamethasone /
- Loratadine /
- Nitazoxanide













