-
Ethylene, a pivotal phytohormone, controls a variety of essential physiological processes, including seedling growth, floral development, fruit ripening, and stress responses[1−9]. Ethylene exerts a concentration-dependent, dual role in plant physiology, where optimal levels promote growth while sub-optimal levels can inhibit processes or induce senescence[10−12]. Its biosynthesis involves a relatively simple two-step enzymatic conversion: ACC synthase (ACS) mediates the conversion of S-adenosylmethionine (SAM) to 1-aminocyclopropane-1-carboxylic (ACC), and ACC oxidase (ACO) then catalyzes the oxidation of ACC to ethylene[13]. Notably, ACS is the rate-limiting enzyme in ethylene synthesis. Among them, ACS2 has emerged as a fascinating locus of evolution and development in cucumber. Recent studies have linked it to the determination of flower sex and the control of fruit development in cucumber. A non-synonymous SNP G87T in CsACS2 caused the transformation of cucumbers from unisexual (gynoecious and monoecious) to bisexual flowers[14]. In cucumber, CsACS2 can be ubiquitinated and degraded by the E3 ligase SF1, thereby precisely regulating ethylene dosage and fruit length[15]. Most strikingly, a groundbreaking study published in Cell further revealed that even synonymous mutations within its coding sequence that do not change the protein sequence can exert profound phenotypic effects[16]. Therefore, the manipulation of ACC synthase at the transcription level or post-translational level provides a means to regulate these traits determined by ethylene in plants. These findings also prompt a critical scientific question: how do different types of mutations within the same gene, ACS2, generate such starkly divergent phenotypes—a binary switch in flower sex vs a continuous variation in fruit length? This question prompts us to re-evaluate how individual genes adeptly manage their functional diversity across different developmental stages and tissues.
Pleiotropy is classically defined as the phenomenon where one gene independently affects two or more distinct traits[17,18]. The prevailing view of pleiotropy often attributes it to non-coding regulatory regions, which dictate a gene's spatiotemporal expression patterns[19−22]. According to the central dogma, the coding region is traditionally seen as the blueprint for the amino acid sequence, determining protein structure and interaction networks[23,24]. While synonymous mutations have been shown in vitro and in cellular models to influence gene expression, translation efficiency, and protein folding[22,25−27], robust in vivo genetic evidence in plants has been scarce. The recent study, published in Cell, demonstrates that synonymous mutations can control gene function in planta by influencing mRNA m6A modification, mRNA structure, and translation efficiency—uncovering a hidden layer of genetic regulation embedded within the coding sequence itself[16]. This discovery provides a crucial new insight for re-evaluating the molecular basis of pleiotropy and for identifying cryptic regulatory sites that orchestrate complex traits.
-
The biosynthesis of ethylene is subject to precise control in horticultural plants, as achieving and maintaining optimal concentrations is essential for numerous physiological processes[28−32]. In cucurbits, the orchestration of both flower sex determination and fruit development hinges on this precise regulation. The development process of flowers and fruit in cucurbits can be divided into five major phases: initiation of floral organs, sex determination, anthesis, fruit growth, and final maturation[33,34]. The pivotal role of ethylene in sex determination is well-established in cucumber, melon, and squash. The genetic basis for flower sex determination in these crops is closely linked to ethylene, as the major regulatory genes are, in fact, those encoding its core biosynthetic enzymes. The M gene (Monoecious) controls unisexual flower development, and encodes the ethylene biosynthetic rate-limiting enzyme ACS2, which is expressed specifically in the carpel of the female flower. The loss-of-function 'm' allele disrupts this pathway, thereby resulting in bisexual flowers[14,35]. The G (gynoecious) gene, which encodes the transcription factor CmWIP1, plays a critical role in cucurbit sex determination[36]. Within carpel primordia, CmWIP1 acts cell-autonomously to repress the carpel-promoting gene CRC, a mechanism that actively promotes the development of male flowers[37]. The female (F) phenotype is determined by CsACS1G, which is expressed during the early developmental stage of the flower bud and acts together with CsACO2 to generate a high dose of ethylene. Ethylene then inhibits CsWIP1 and activates CsACS2 to initiate the development of gynoecy[38]. Female flower development in melon and cucumber is regulated by the A (androecious) gene, which exerts its function by encoding ACS11—a pivotal enzyme that catalyzes the rate-limiting step in ethylene production. Its phloem-specific expression, connected to flowers, inhibits the expression of CmWIP1, thereby orchestrating the co-development of male and female flowers. The loss-of-function mutants of ACS11 result in male plants (androecy)[39]. Furthermore, the role of ethylene synthesis in sex determination is underscored by studies on ACC oxidase (ACO), which catalyzes the final step in ethylene production from ACC. For instance, inactivation of CsACO2 in cucumber disrupts carpel development, causing a female-to-male transition[40], while reduced CpACO1A activity in squash converts female flowers into hermaphrodites[41]. Collectively, these findings establish ethylene as the pivotal hormonal regulator of flower sex in cucurbits.
Beyond sex determination, ethylene is also crucial for fruit development[42], especially in the elongation of the inferior ovary, which develops from accelerated growth of the receptacle due to the continuous activity of meristematic stem cells in the basal part of the cucumber flower organs[43]. Almost all the sex-determining genes, such as ACO2, ACS, WIP, and CRC, are expressed in the receptacle or receptacle-derived tissues, which suggests that the development of the receptacle promotes the evolution of unisexual flowers in Cucumis species[43]. In melon, CmACS7 plays a dual role in sex determination and fruit shape. A speculated mechanism is that the expression of CmACS7 in the carpel primordium at the early stage suppresses the stamen development in a non-cellular autonomous manner. Following sex determination, the ethylene synthesis mediated by CmACS7 in the primordia then governs cellular processes essential for normal ovary expansion and fruit formation[44]. Ethylene can also regulate the cell division and subsequent fruit elongation in a dose-dependent manner in cucumber (Fig. 1)[15]. All these studies highlight the multiple regulatory functions of ethylene in cucurbits, coordinating the suppression of stamen development with the promotion of fruit elongation.
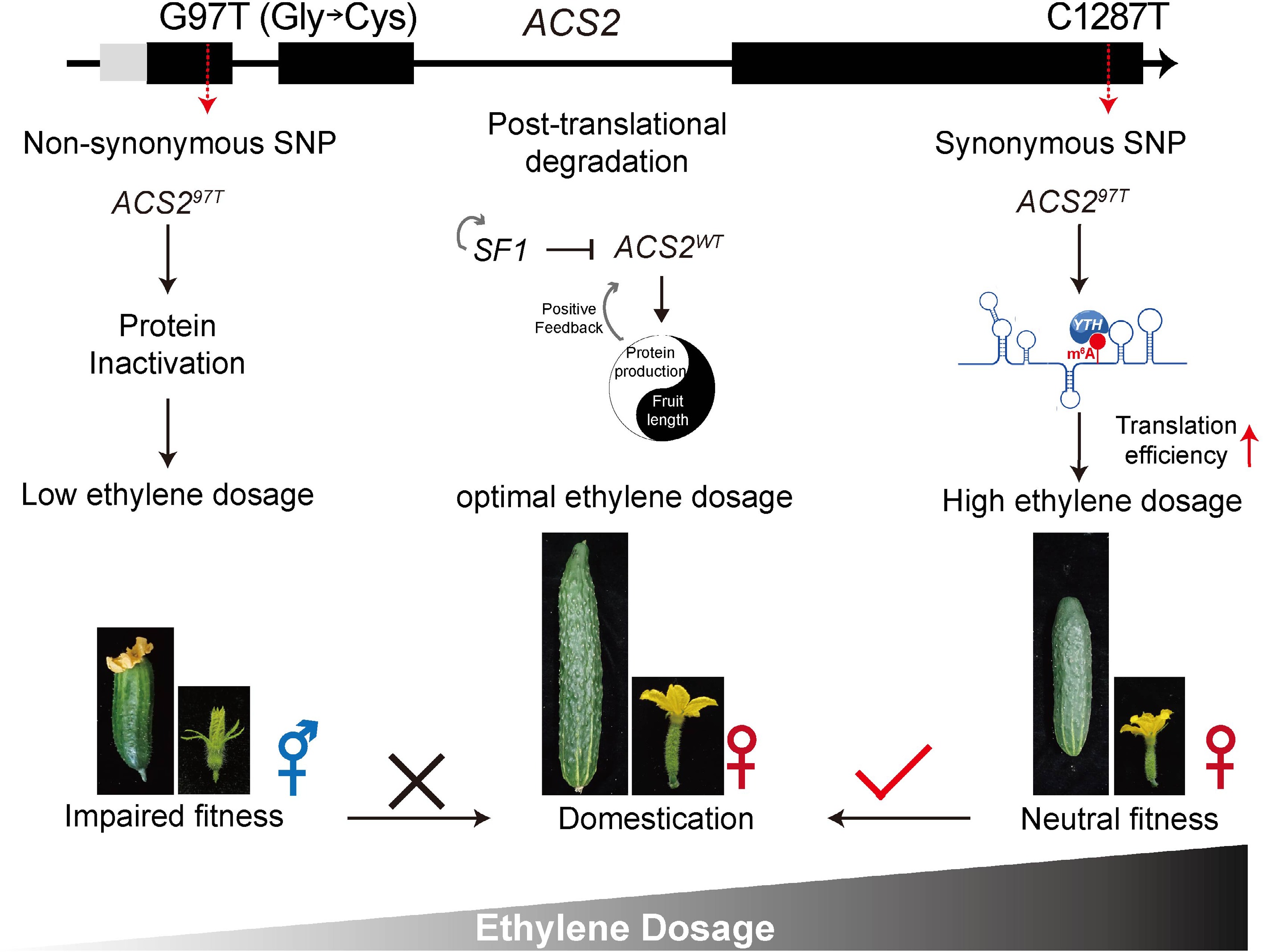
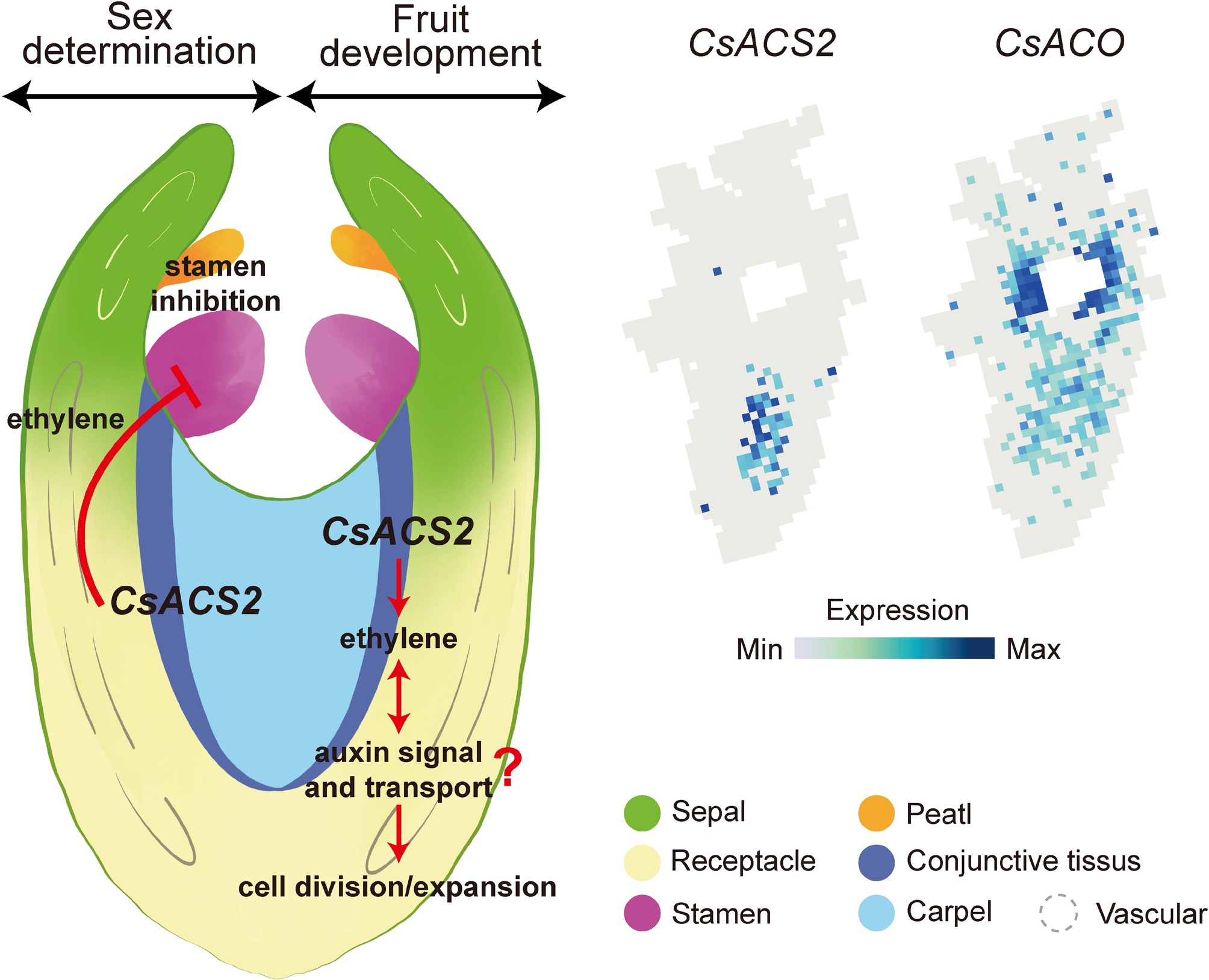
Figure 1.
The different mutations and regulation of ACS2 determine the sex and fruit length of cucumbers. A G97T non-synonymous mutation in the first exon of CsACS2 coding region, impairing the enzyme activity and thus producing differences between the unisexual and the bisexual alleles. RING-type E3 ligase short fruit 1 (SF1) regulates fruit length by modulating ethylene dosage via ubiquitination and degradation of both itself and CsACS2 at the post-translational level. A synonymous SNP at the 1,287 position can determine ethylene production through epitranscriptomic (m6A methylation and RNA structure,) thereby regulating the length of cucumber fruits.
-
The function of ACS2 in sex determination of cucumber has been primarily characterized through non-synonymous mutations that directly alter the protein sequence. The conversion of G97T in the CsACS2 coding region caused a substitution of glycine by cysteine at residue 33 in the protein, impairing the enzyme activity and thus producing differences between the unisexual and the bisexual alleles (Fig. 1)[14]. Similarly, a conserved non-synonymous mutation, A57V results in a loss of ACC synthase activity. Since this enzymatic activity is necessary for female flower development in monoecious plants, its reduction can lead to hermaphroditism in flowers that would otherwise be male[35]. These mutations follow the classical model of 'loss-of-function' effects, resulting in dramatic phenotypic shifts that have profound implications for cucumber breeding.
This classical model effectively explains how amino acid alterations of ACS2 at the M locus influence sex determination. However, its limitation emerges when considering that the same site affecting sex determination might be expected to similarly impact fruit development. In practice, these traits can vary independently, suggesting additional layers of regulation beyond simple protein sequence alterations. This also lays the foundation for the discovery of a more subtle regulatory mechanism involving synonymous mutations that specifically affect fruit elongation without disrupting sex determination.
-
Unlike non-synonymous mutations that affect gene function by altering amino acid encoding, synonymous mutations were historically considered 'silent' or neutral, as they do not change the encoded amino acid sequence. Notably, our recent study, published in Cell, has found that synonymous mutations in ACS2 can regulate cucumber fruit length domestication without altering sex expression, which has changed our understanding of traditional genetic regulation in plant[16].
This discovery is based on backcross introgression lines (ILs) initially constructed with short wild cucumbers and long cultivated cucumbers. Genetic analysis identified two epistatically interacting genes that contributed to cucumber fruit length domestication: one encodes YTH1, an RNA m6A reader, and the other encodes ACS2, an ACC synthase. The causative mutation is a synonymous mutation in ACS2, which changes base C1,287 to T1,287 without altering the amino acid sequence of the enzyme. In wild cucumber, the ACS21,287C leads to m6A modification on the base A at position 1,286, forming a loose RNA structure conformation. The m6A modification is then recognized by YTH1, which shifts the folding equilibrium away from the weakest RNA structural conformation. This increases ACS2 protein levels, thereby elevating ethylene production. The enhanced ethylene signal inhibits cell division in early fruit development, ultimately resulting in shorter fruits. In cultivated cucumbers, the 1,287T synonymous mutation disrupts this pathway: it abolishes m6A methylation, forming a compact RNA structure. This leads to reduced ACS2 protein and ethylene synthesis, promoting longer fruit development (Fig. 1).
The synonymous mutations appear to modulate translation efficiency rather than protein function, by affecting mRNA m6A methylation and structure—mechanisms that would quantitatively fine-tune ethylene output without qualitatively changing enzyme function, thereby regulating the length of cucumber fruits.
-
What is the underlying molecular mechanism by which ACS2 regulates the female flowers and fruit length during development? Based on spatiotemporal transcriptome data, we found that CsACS2 is specifically expressed in the conjunctive tissue, located between the pericardium and the receptacle. This is different from the previous recognition that it was expressed on the carpel. This also suggests that the conjunctive tissue may play an important role in coordinating ovary development and sex determination (Fig. 2). A plausible model emerges from these studies. During the floral development, ACS2 is specifically expressed in the receptacle-derived connective tissues and catalyzes the synthesis of ACC. This ACC can then be transported via the receptacle vasculature to stamens, where the highly expressed ACO converts it into high doses of ethylene. This ethylene burst is a well-established signal that triggers the developmental arrest or programmed cell death of the stamen primordia, thereby promoting female flower development (Fig. 2)[39,40]. On the other hand, the ethylene specifically produced by ACS2 in the same receptacle-derived connective tissue could interact with auxin signaling and polar transport to regulate fruit development. This crosstalk between ethylene and auxin in the conjunctive tissue could precisely regulate rates of cell division and expansion, thereby promoting the formation of the ovary and the elongation of fruit (Fig. 2).
-
ACS2 plays a pleiotropic role in cucumber, coordinating both floral sex determination and fruit development through the same hormone pathway, yet via distinct mutational mechanisms. The capacity of different mutation types in ACS2 to generate distinct biological effects, with non-synonymous mutations primarily affecting sex determination and synonymous mutations specifically influencing fruit length. Understanding this phenomenon requires consideration of both the molecular genetics of these mutations and the developmental biology processes.
At the molecular level, non-synonymous mutations directly alter the protein sequence of ACS2, potentially affecting enzyme activity, stability, substrate affinity, or protein-protein interactions. These qualitative changes typically have substantial impacts on ethylene biosynthesis capacity, leading to dramatic shifts in sex expression. In contrast, synonymous mutations leave the protein sequence unchanged but may influence mRNA stability, splicing patterns, or translation efficiency, thereby subtly modulating enzyme-specific activity without completely disrupting function. The synonymous mutations essentially act as a 'switch knob' for ethylene production in specific tissues or developmental stages, unlike the 'on/off' switching characteristic of non-synonymous mutations.
At the development level, the spatial and temporal expression of ACS2 and ethylene action contribute to these divergent effects. At the early development stage, functional ACS2 generates ethylene, which suppresses stamen development to promote femaleness. Subsequently, at floral stage 8—after sex determination but before fertilization—final fruit shape is determined. Ethylene in carpel primordia can inhibit stamen development via HB40 and also impact fruit length by regulating cell division and elongation[44,45]. Ultimately, plants achieve precise developmental outcomes by fine-tuning RNA function to modulate ethylene levels, allowing for adjustments in fruit development without compromising the essential inhibition of stamens in female flowers.
-
The finding that synonymous mutations in ACS2 can influence fruit length without altering sex determination has profound implications for our understanding of adaptive evolution in plants, particularly in domesticated species like cucumber. The longstanding view, supported by the higher frequency of synonymous changes in related species, holds that most are neutral variants[46−48]. Recent phylogenomic evidence challenges the traditional neutral view of synonymous mutations. An analysis of the Solanaceae family revealed that among 367,499 deleterious variants, synonymous mutations constitute 15%, a frequency second only to nonsynonymous mutations[49]. This finding implies that a significant proportion of synonymous SNPs are functionally consequential and subject to selection, despite their higher prevalence than nonsynonymous changes in closely related species. While nonsynonymous mutations often face strong selective constraints due to their potential disruptive effects on protein function, synonymous mutations accumulate more freely while still providing a source of phenotypic variation that natural and artificial selection can act upon. ACS2 is crucial for both sex determination and fruit development. Loss-of-function via nonsynonymous mutations leads to andromonoecy or hermaphroditism, traits likely detrimental under natural selection. In contrast, the 1,287C > T synonymous mutation affects only fruit length without altering flower sex, a variation that may be selectively neutral and thus persist evolutionarily. During crop domestication and improvement, synonymous mutations offer a unique evolutionary advantage: they allow for the fine-tuning of specific traits without compromising other essential functions. For a pleiotropic gene like ACS2 that regulates multiple aspects of development, nonsynonymous mutations typically affect all functions simultaneously, creating correlated changes that may not always be advantageous. Synonymous mutations provide a mechanism to break such trade-offs under selection pressure by modifying post-transcriptional processes. These findings challenge the traditional view and highlight the potential significance of synonymous mutations for crop improvement.
-
The pervasive challenge of gene pleiotropy—whereby one gene influences multiple phenotypic traits—has long constrained progress in crop improvement. Traditional approaches to modifying agriculturally important genes frequently encounter trade-offs wherein enhancement of one desirable characteristic comes at the expense of another. The finding that synonymous mutations can exert fine-scale regulatory effects without altering protein sequences offers a new strategy to overcome these limitations.
The molecular basis for this strategy lies in the effects of synonymous mutation on protein expression through mRNA structure, RNA modification, codon usage, or ribosome occupancy rates. Implementing this strategy requires sophisticated screening platforms to identify functional synonymous mutations with desired effects. High-throughput approaches, such as the Prime Editor-based Screening Technology (PRESENT) system and deleterious synonymous mutations finder (DS Finder)[50], allow systematic evaluation of synonymous mutation libraries in human cells. These computational analysis features, including codon usage, mRNA structure, and splice site recognition patterns, prioritize synonymous mutations with the highest likelihood of generating desired phenotypic effects. Such models can also be optimized for use in horticultural crops.
The practical application of synonymous mutation engineering would extend beyond ACS2 to numerous other pleiotropic genes controlling critical agronomic traits. For instance, transcription factors regulating both plant productivity and stress response, or biosynthetic enzymes influencing both yield and quality, represent prime targets for this approach. The implications for crop improvement are profound. If we can decipher the rules governing how specific synonymous codons influence gene expression and protein yield in vivo, we enter a new era of 'rational codon design'. By developing synonymous codon-optimized alleles, breeders can overcome the trade-offs between productivity and resilience, or between yield potential and nutritional quality. Looking forward, the integration of synonymous mutation engineering with advanced prime editing technologies promises to revolutionize precision crop design. More importantly, by operating within the existing coding framework without introducing foreign DNA, synonymous mutation strategies may face fewer regulatory hurdles than transgenic approaches, potentially accelerating their adoption in breeding programs worldwide.
-
The authors confirm their contributions to the paper as follows: study conception and design, draft manuscript preparation: Xin T, Yang X; data collection: Xin T; analysis and interpretation of results: Xin T, Dong Z. All authors reviewed the results and approved the final version of the manuscript.
-
This study is a opinion article and does not contain any original datasets. All referenced data can be found in the cited publications.
-
We are deeply grateful to Prof. Wenyang Li for his invaluable insights and constructive suggestions on this manuscript. This work was supported by the National Natural Science Foundation of China (NSFC) (Grant No. 32525052 to XY), the National Key Research and Development Program of China (2021YFF1000102), and the Science and Technology Innovation Program of the Chinese Academy of Agricultural Sciences (CAAS-ASTIP).
-
The authors declare that they have no conflict of interest.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press on behalf of Chongqing University. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Xin T, Dong Z, Yang X. 2026. The ACC synthase (ACS): a key regulator of flower sex and inferior ovary formation in cucurbits. Plant Hormones 2: e006 doi: 10.48130/ph-0026-0003
The ACC synthase (ACS): a key regulator of flower sex and inferior ovary formation in cucurbits
- Received: 19 December 2025
- Revised: 06 February 2026
- Accepted: 15 February 2026
- Published online: 20 March 2026
Abstract: Ethylene is a crucial phytohormone that modulates flower sex determination and fruit development in cucurbit crops. In cucumbers, ACC synthase 2 (ACS2) controls the key step in the synthesis of ethylene, which regulates these two traits, demonstrating significant genetic pleiotropy. Previous studies have found that a non-synonymous mutation can change the protein sequence of ACS2, leading to the loss of enzyme function and thereby changing the sex of the flower. Recent breakthrough research has found that a synonymous mutation on this gene can influence translation efficiency by affecting the m6A modification and mRNA structure, precisely regulate ethylene production, and thereby 'fine tune' fruit length without affecting sex determination. This finding reveals that the coding sequence itself contains a layer of regulatory mechanism that is independent of the protein sequence. This article discusses how different types of mutations on the same gene coordinate two distinct traits. This provides a new perspective for understanding the molecular basis of gene pleiotropy. Finally, we proposed a strategy of designing synonymous mutations to fine-tune specific agronomic traits, providing a new avenue for crop precise improvement and breeding.
-
Key words:
- Ethylene /
- Cucurbits /
- Fruit development /
- Sex determination /
- ACS2 /
- Pleiotropy