-
Rapid urbanization, industrialization, and economic growth have increased energy demands. The widespread use of fossil fuels has accelerated resource shortages[1,2], and considerably polluted the natural environment, such as water, air, and soil, with highly toxic inorganic, organic, and biological pollutants[3,4]. Therefore, developing new renewable energy conversion and storage technologies, along with sustainable water and air treatment techniques, is necessary to overcome these energy and environmental challenges[5]. Various materials, including unconventional materials, metals/metal oxides, organic–inorganic hybrids/composites, polymers, and dendritic polymers, have been investigated extensively[6]. However, the rapid exhaustion of fossil energy sources and environmental pollution have shifted the research focus toward utilizing sustainable energy sources[7,8]. Carbon-based materials have attracted widespread attention and stand out for energy and environmental applications owing to their availability, simple preparation schemes, engineered properties post-synthesis, low cost, resistance to acidic and alkaline conditions, and excellent recyclability[9,10].
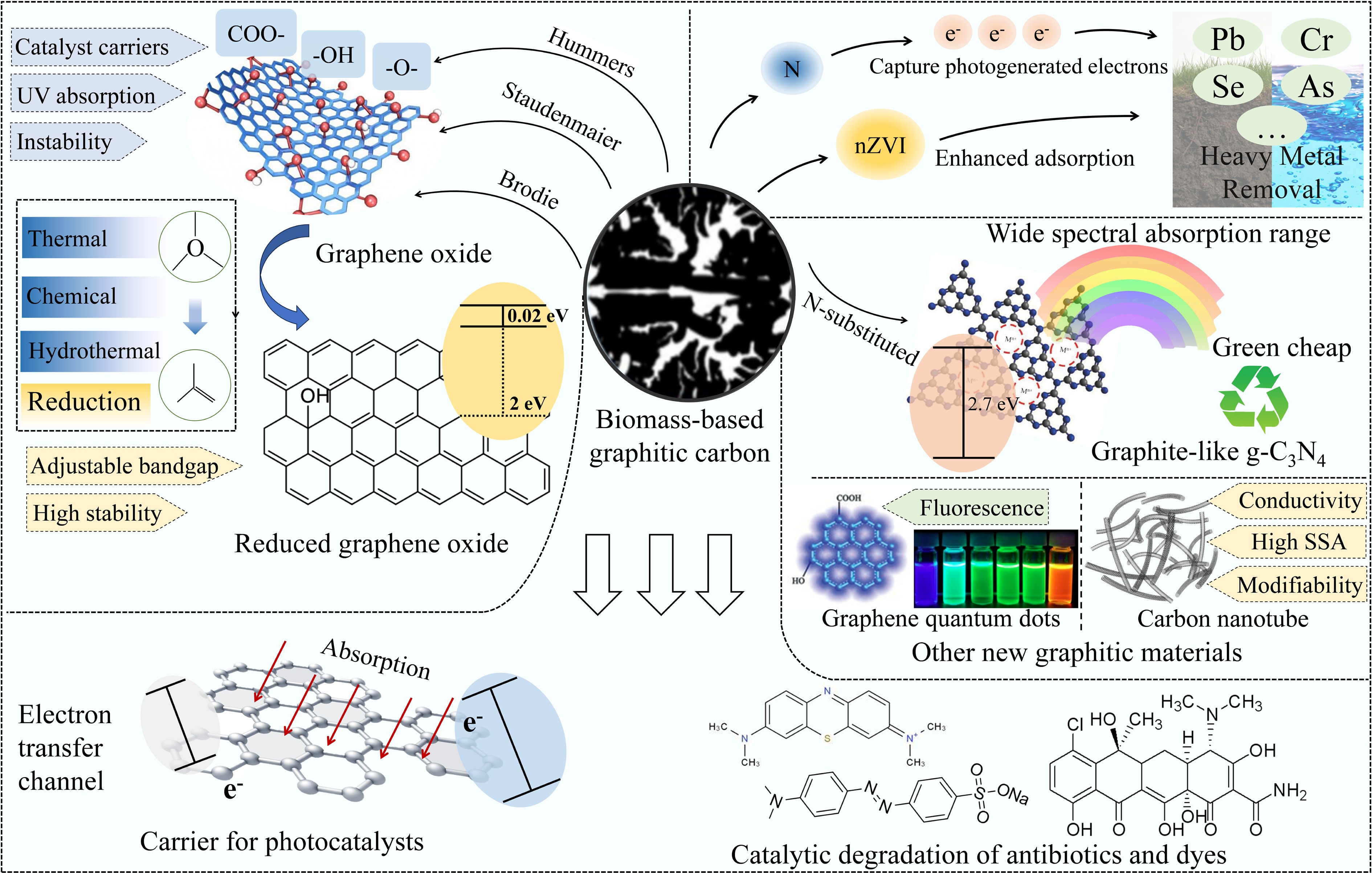
Graphitization is a process whereby the carbon structure of organic materials is reorganized into a graphite-like structure at high temperatures (> 2,500 °C)[11]. In contrast, other pyrolysis processes, such as carbonization, hydrothermal synthesis, chemical activation, chemical blowing, and template-assisted confinement, often produce amorphous carbon, which includes soft and hard carbons, rather than graphitic crystalline structures. Soft carbon, owing to its relatively ordered local structure, can undergo partial graphitization at elevated temperatures. Hard carbon, however, possesses a highly disordered cross-linked structure that exhibits strong resistance to graphitization even under high-temperature conditions, thereby directly limiting the overall graphitization level of biomass-derived carbon[12]. Biomass is a sustainable carbon precursor for the production of graphitic carbon. Therefore, its use as feedstock for graphitic carbon production has remarkable potential[13−15]. Because of the complexity of producing graphitic carbon from biomass, biomass feedstock variations considerably impact the production and application of graphitic carbon[16,17]. For example, biomass naturally exhibits various microstructures that can be perpetuated and augmented by carbon materials generated during graphitization. The three typical components of biomass, namely cellulose, hemicellulose, and lignin, exhibit distinctly different behaviors during carbonization. Meanwhile, the contents of these three components remarkably influence the properties of graphitic carbon products. In addition, biomass is rich in heteroatoms, including non-metallic elements, such as N, S, and P, and alkali and alkaline earth metals (AAEMs) such as K, Na, and Ca[18]. Therefore, a better understanding of the interrelationships among feedstocks, graphitization processes and application technologies can enhance the utilization of the intrinsic properties of biomass.
Unlike conventional biochar, graphitic carbon contains more or less graphitic microcrystalline structures, an important property for energy and environmental applications. Among graphitic carbon materials, graphene stands out in the energy sector due to its unique two-dimensional layered structure, exceptionally high specific surface area, outstanding electronic conductivity, and mechanical stability, demonstrating irreplaceable application potential and becoming a research hotspot in energy storage. It not only enhances ion transport rates and charge storage capacity but also improves the electronic conductivity and structural stability of electrodes[19]. Currently, the two types of graphite are used in industrial applications: mineral and synthetic graphite. Graphite anodes used in lithium-ion batteries (LIBs) typically contain both mineral and synthetic graphite[20]. The processing of mineral graphite for lithium-ion anode applications typically requires the use of hydrofluoric acid for purification. Hydrofluoric acid is highly corrosive and considerably threatens human health and the environment[21]. The production of synthetic graphite typically employs high-aromatic, low-sulfur fossil carbon resources, such as fluid catalytic cracking slurry oil. First, needle coke is formed through delayed coking at 475–530 °C. This is followed by calcination at 1,100–1,500 °C, and then graphitization at ≥ 2,500 °C for more than 7 d[22]. The production of synthetic graphite is associated with considerable carbon emissions, with an estimated emission of CO2 of 20 kg/kg graphite[23]. Therefore, developing novel technologies that utilize abundant, cost-effective, and renewable carbon materials are essential to achieve the sustainable production of graphite anodes under relatively mild processing conditions. The green production and large-scale application of graphene-based materials also offer new solutions for achieving this sustainable development goal.
In environmental science, graphitic carbon is frequently used as a photocatalyst support because of its exceptional physicochemical properties[24,25]. Compared to other materials, such as ZrO2, graphite exhibits higher photoelectron-catalytic activity, and can serve as a co-catalyst to enhance catalytic activity. Two-dimensional (2D) graphite materials exhibit outstanding electrical conductivity, acting as electronic transmitters to improve the separation of photogenerated charge carriers, thereby enhancing photoelectron-catalytic activity[26]. Graphitic materials used in photocatalysis include transition metal-doped graphitic carbon, 2D graphene oxide (GO), g-C3N4, and other novel graphite materials.
Preparatory steps and applications of biomass graphitization have been studied[27]. Biomass-based graphitic carbon (BBGC), derived from biomass, is a product of high temperature or catalytic graphitization, which is environmentally friendly. The urgency for the global community to transition towards sustainable energy solutions, particularly given the increasing threat of climate change, underlines the key role of material science innovation in green technologies[26]. In this context, BBGC can be applied to achieve synergies between the sustainable use of renewable resources and their use in energy storage. Therefore, a life cycle assessment (LCA) of BBGC enables comprehensive assessments of the environmental impacts of biomass materials throughout their life cycle, from production to disposal. This is important for the development of effective waste management and recycling strategies that can help achieve resource recovery, reduce the overall environmental footprint, and support BBGC applications in sustainable energy systems.
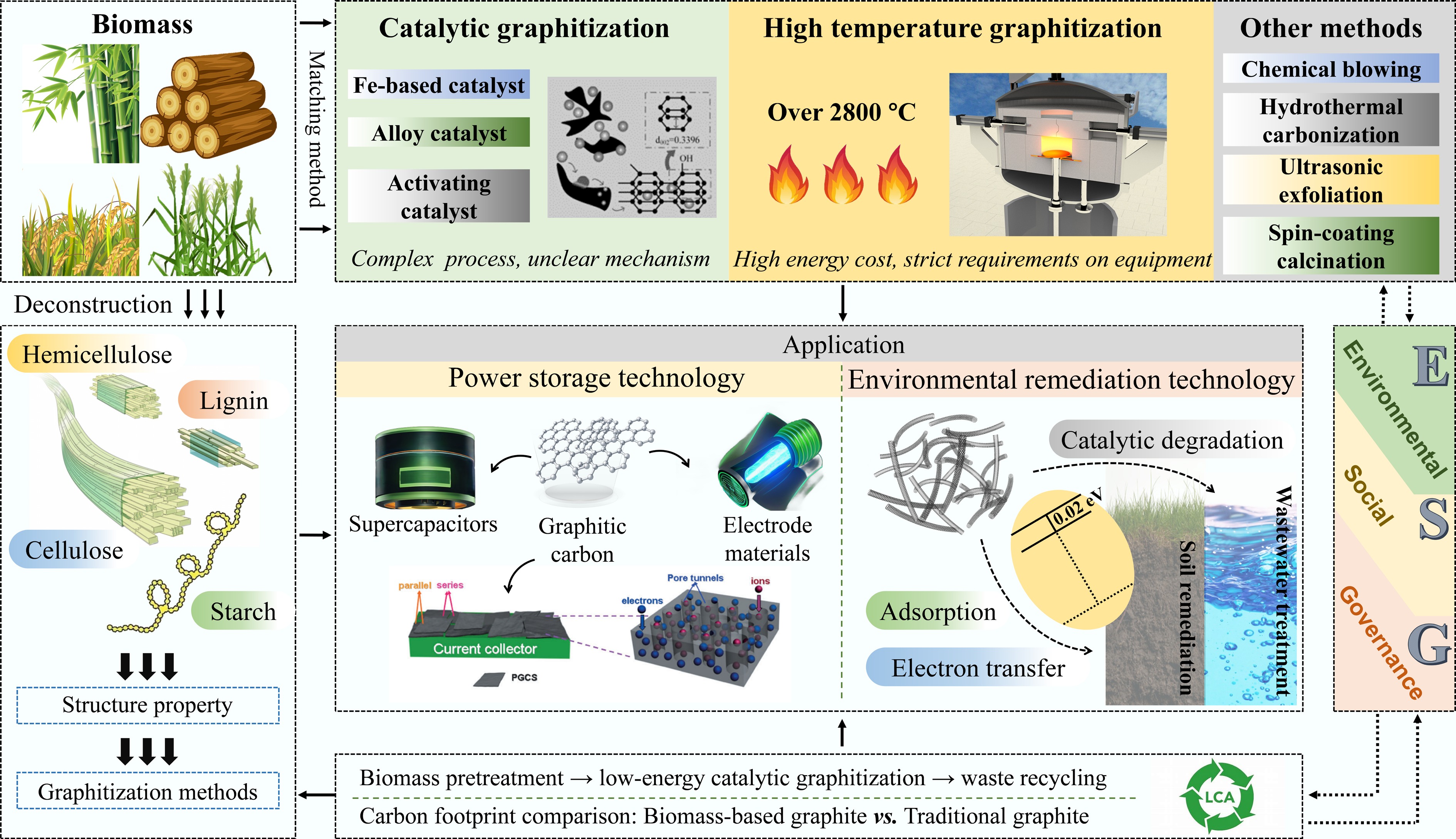
In addition to its economic and environmental benefits, biochar is an environmentally friendly material that offers considerable environmental, social, and governance (ESG) advantages[28]. In agriculture, it enhances crop yields and fosters ecosystem services by enhancing soil properties. Furthermore, the Intergovernmental Panel on Climate Change (IPCC) has acknowledged biochar as an emission-negative technology, emphasizing its potential to reduce atmospheric carbon dioxide levels[29]. However, economic and technological challenges are associated with its widespread use. Government support through financial and non-financial incentives can facilitate the growth of the biochar industry, increase employment, enhance rural livelihoods, and promote biochar use[30]. Promoting biochar can help realize the United Nations Sustainable Development Goals (SDGs). This review provides a systematic investigation into the intrinsic graphitization potential of biomass, as shown in Fig. 1. It covers the regulation of the graphitization process by its main components (cellulose, hemicellulose, lignin), starch, and AAEMs and non-metallic elements. Graphitization technology pathways matching biomass characteristics were screened, and the applications of BBGC and its derivatives in two core fields (energy storage and catalytic degradation of pollutants) were elaborated in detail. Furthermore, this review conducts a comprehensive LCA of BBGC, clarifying its environmental footprint across the entire process from raw material acquisition and graphitization preparation to end-use applications and waste disposal. Integrating an ESG perspective, it further validates the sustainability advantages of BBGC in replacing traditional graphite, including reduced resource dependency, lower carbon emissions, and mitigation of environmental risks.
-
Over the past years, with the increasing global concern for sustainable development and environmental issues, research on biochar has shown a remarkable growth trend, in which BBGC has received extensive attention in the energy and environmental fields. These changes not only reflect the latest trends in scientific research but also mark the intersection of technological advances and market demand. As shown in Fig. 2, there was a significant increase in the number of research publications on biochar from 2015 to 2025. The number of publications in 2015 was 1,195, and it has increased to 9,888 in 2025, suggesting an increasing interest in biochar[31]. From 2015 to 2025, the number of research publications on graphitic carbon increased steadily from 1,344 to 3,919. At the intersection of graphitic carbon and energy, graphitic carbon plays a key role in energy technologies such as batteries and supercapacitors. A pattern of steady growth was observed in the number of articles published on this topic, from 428 in 2015 to 1,720 in 2025. This growth reflects the demand for efficient energy conversion and storage in the energy sector. The utilization of graphitic carbon in the energy sector is expected to continue to expand, in line with the global pursuit of renewable energy and efficient energy storage solutions[32]. Finally, the integration of graphitic carbon into environmental applications is a growing area of research, with the number of publications increasing from 90 in 2015 to 931 in 2025. Research has focused on the utilization of graphitic carbon in environmental protection and pollution control, including water treatment, air purification, and the development of sensors for environmental monitoring. This growth indicates that the need to solve environmental problems drives the expansion of such research.
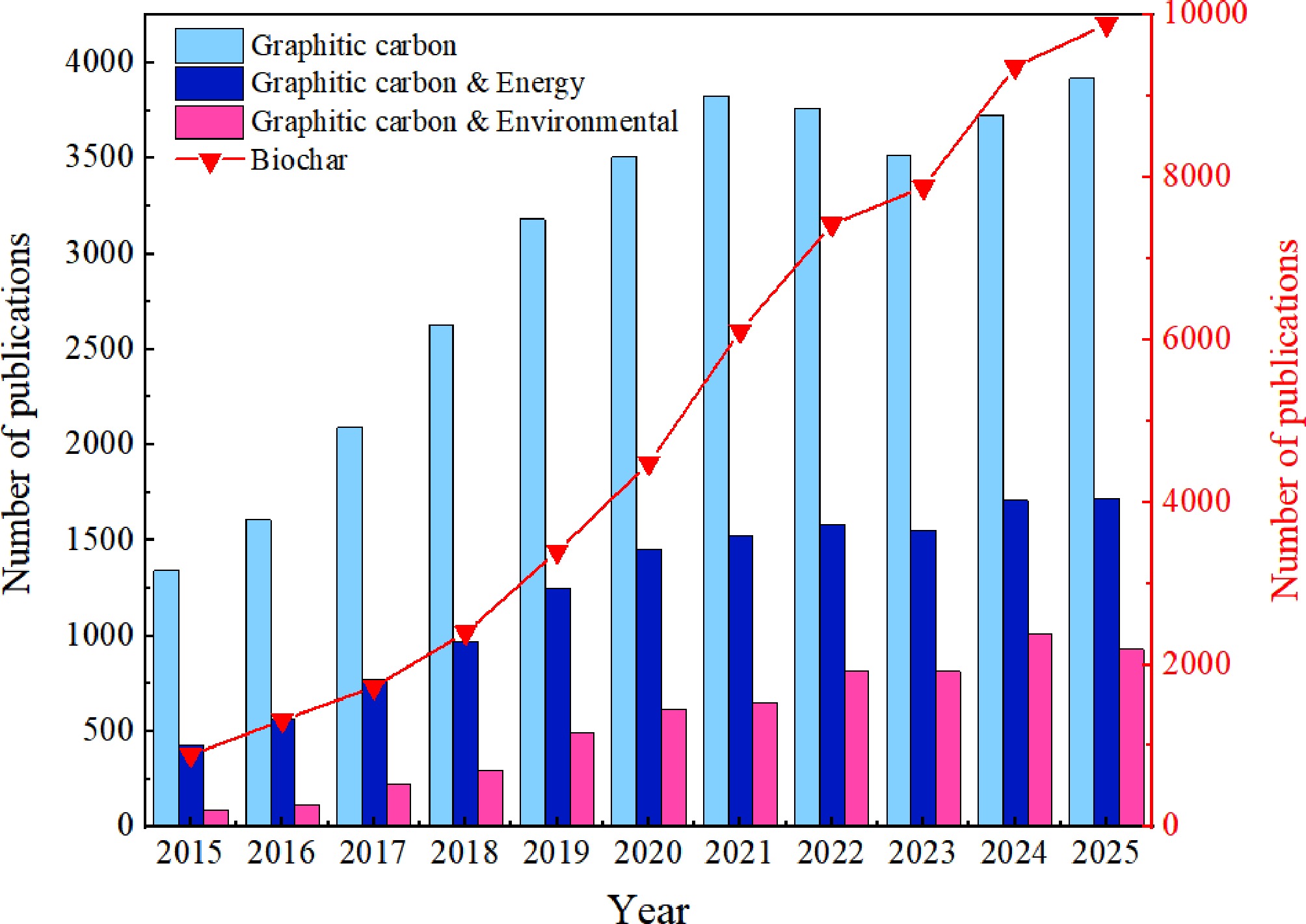
Figure 2.
The publication status of articles related to the application of graphitic carbon in the energy and environmental fields from 2015 to 2025, as of March 02, 2026 (from the Web of Science).
This review conducted a literature analysis of research related to BBGC, aiming to provide reliable recommendations for future studies. Research data on BBGC were retrieved from the Web of Science Core Collection database (as of November 25, 2025). Search terms included 'Graphitic carbon' or 'Graphitic carbon & Energy' or 'Graphitic carbon & Environmental', with the time range set from 2015 to 2025. After primary screening and duplicate removal, 6,000 papers were selected for bibliometric analysis based on relevance (highest to lowest). Visualization was performed using VOSviewer and bibliometric software, ultimately identifying 251 keywords (minimum occurrence threshold: 40 times). The visualization results are shown in Fig. 3. Keyword analysis indicates that over the past decade, graphitic carbon nitride has served as the primary carrier in BBGC-related research, while graphene is commonly employed as a modifying component to construct composite structures with graphitic carbon nitride. In Fig. 2, it is evident that this material finds extensive applications in both photocatalysis and energy storage fields, such as supercapacitors. Such composite systems can be utilized for clean energy production, like photocatalytic hydrogen generation, environmental remediation scenarios, including the degradation of pollutants such as Rhodamine B and tetracycline, as well as CO2 conversion, and simultaneously extend into energy storage domains like supercapacitors. Structural designs incorporating nanosheets and active sites enhance charge separation efficiency and energy storage performance. Research on preparation techniques like in-situ synthesis and heterostructure construction also balances catalytic activity, stability, and energy storage performance. This research focus is no coincidence. It responds to demands for renewable energy development, environmental remediation, and energy storage technology upgrades. Through composite modifications, it overcomes the limitations of single materials, thereby advancing BBGC applications across energy, environmental, and energy storage sectors.
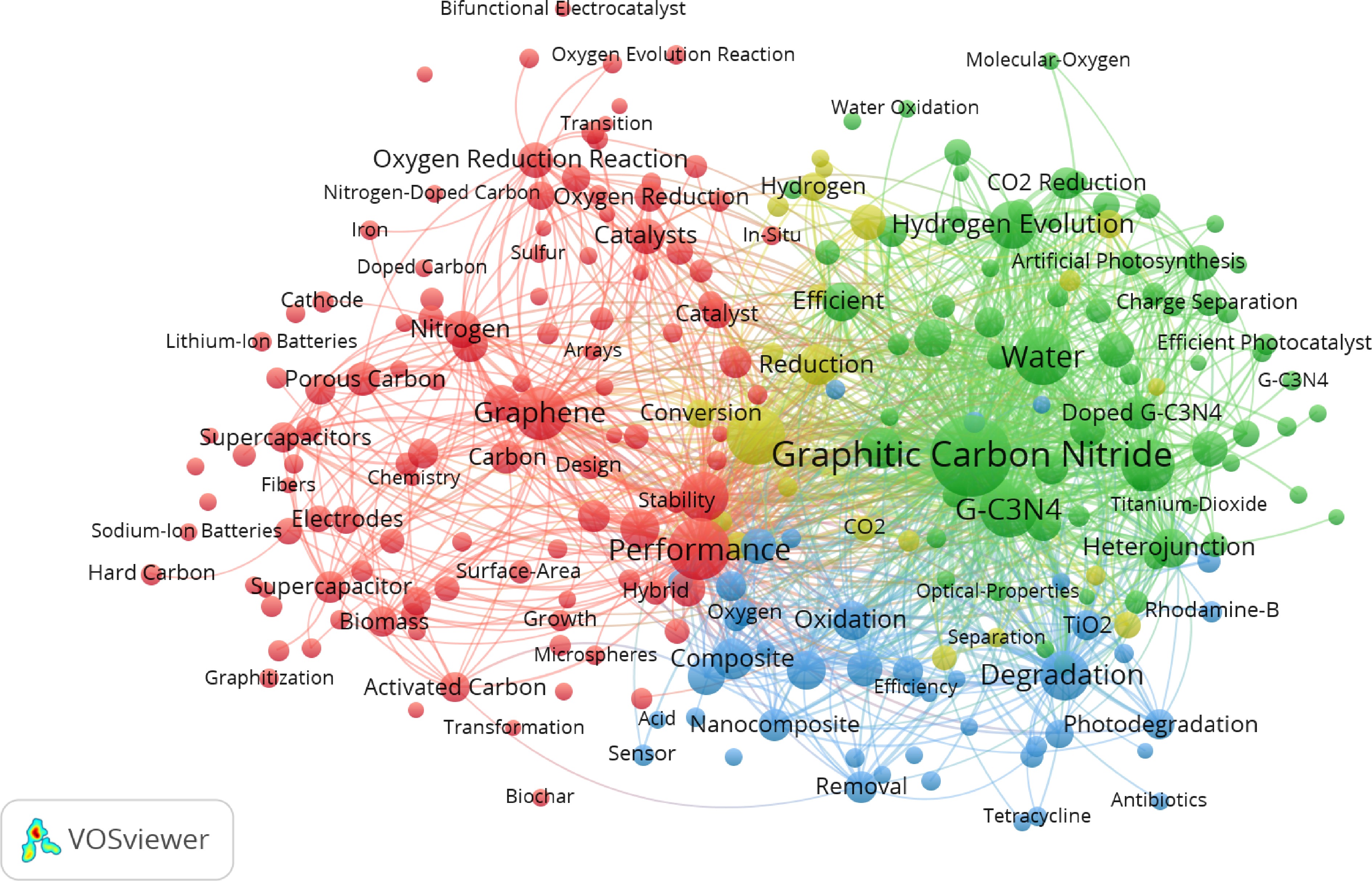
Figure 3.
Scientometric visualization of the top 251 keywords of all peer-reviewed publications related to BBGC released from 2015 to 2025. A total of 6,000 publications were retrieved from the Web of Science Core Collection (as of March 02, 2026) with 'Graphitic carbon', 'Graphitic carbon & Energy', or 'Graphitic carbon & Environmental' as the search keywords. Collected data were analyzed using the built-in function of co-occurrence of all keywords and plotted in 'overlay visualization' using VOSviewer. Each circle stands for a keyword, while its size represents the number of times a pair of keywords co-occurred in the publications.
Reviews in the field of BBGC tend to focus on several key areas. These include the graphitization of biomass, methods for the synthesis of graphitic carbon and its derivatives, and the application of these materials in energy storage and environmental management (illustrated in Table 1). In particular, studies have focused on the development of new synthesis techniques to improve the efficiency and properties of materials, such as electrical conductivity, mechanical strength, and chemical stability. Research on the use of adsorbents for pollution control, including in water treatment and air purification, is also of interest. Although existing reviews provide a comprehensive overview of BBGC, there are some limitations. First, the graphitization potential of biomass, and the associated graphitization methods, which impact the industrial production of graphitic carbon, have not been discussed in detail. This is because it is not easy to identify a method that both matches the raw material and reduces the quality of graphitic carbon. Second, in the discussion of energy and environmental applications, researchers tend to focus on the performance of composites, ignoring the critical role of graphitic carbon and its derived materials. Furthermore, reviews often fail to fully discuss cost-effectiveness, which is a considerable constraint for the wider application of biomass-based carbon materials.
Table 1. Review papers on recent research on BBGC
Review title Main content Citation Ref. Bilayer nanographenes: structure, properties, and synthetic challenges This review explores the synthesis, structural characteristics, and functional significance of bilayer and multilayer nanographene. It investigates how the degree of π-π overlap governs key properties—including the highest occupied molecular orbital (HOMO)-lowest unoccupied molecular orbital (LUMO) energy gap, redox behavior, photoluminescence shift, and quantum yield, as well as chiral optical response—and reveals synthetic approaches for obtaining enantiomerically pure bilayer graphene. − [33] Probing the evolution in catalytic graphitization of biomass-based materials for enduring energetic applications This review aims to bridge the gap between the diverse feedstocks and processing conditions employed in different studies by exploring the potential of biomass materials as raw materials for catalytic graphitization, with a commitment to achieving sustainable and efficient energy applications. 32 [34] Biomass-derived carbon applications in the field of supercapacitors: progress and prospects This paper analyzes recent advances in biomass-derived carbon electrodes for supercapacitors, introduces carbon electrodes from various biomass resources, and comprehensively reviews carbonization, activation, and heteroatom doping techniques for biomass carbon. 98 [35] Biochar: from by-products of agro-industrial lignocellulosic waste to tailored carbon-based catalysts for biomass thermochemical conversions This review explores research on preparing biochar from agricultural and forestry residues as carbon-based catalysts and catalyst supports, along with their applications. By systematically examining biochar formation mechanisms, catalytic mechanisms, synthesis conditions, catalyst or support modification techniques, and application scenarios, it provides guidance for preparing application-oriented biochar catalysts and supports for use in biorefinery thermochemical reactions. 129 [36] Recent advances of 3D graphene-based adsorbents for sample preparation of water pollutants: a review The review emphasizes the significance of employing biomass waste and various techniques to synthesize graphitic carbon. The discussion encompasses recent advancements in the conversion of biomass wastes into carbon-rich precursors, as well as methods for the synthesis of graphitic carbon. Additionally, the article addresses the importance of utilizing agricultural biomass waste, types of potential biomass waste carbon precursors, and their pretreatment methods. 143 [37] Porous and graphitic structure optimization of biomass-based carbon materials from 0D to 3D for supercapacitors: a review The review presents the latest advancements in the preparation methods of BPGCs, with a particular focus on the mechanisms of structural evolution during activation and graphitization processes. Furthermore, it discusses the progress made in optimizing and reconstructing the microstructure from 0D to 3D. 238 [38] From biomass to energy storage: a review on lignocellulosic biomass-derived hard carbon anodes for sodium-ion batteries This review explores the precursors employed and their impact on the final properties of carbon materials—including structure, texture, and surface chemistry—as well as their influence on the electrochemical performance of sodium-ion batteries. It introduces research on heteroatomic doping for hard carbon and elucidates strategies for overcoming performance bottlenecks in sodium-ion batteries. − [39] Iron-catalyzed graphitization for the synthesis of nanostructured graphitic carbons The review presents an overview of the current state of research on iron-catalyzed graphitization, with a particular focus on molecular organic or biomass precursors. Biomass-derived precursors are identified as particularly promising options for the sustainable production of graphitic carbon. It discusses the challenges currently faced by iron-catalyzed graphitization, with a particular focus on the limitations in understanding the mechanisms of graphitization. 188 [27] Upcycling of plastic wastes and biomass for sustainable graphitic carbon production: a critical review The review examines methods for transforming plastic waste and biomass into high-value graphitic carbon materials through co-pyrolysis techniques. It examines the factors influencing the quality of graphitic carbon and discusses its potential applications in electronics, energy storage, and other fields. 44 [40] A route towards graphene from lignocellulosic biomass: technicality, challenges, and their prospective applications The review considers methods for the production of graphene using lignocellulosic biomass as a sustainable and cost-effective feedstock. It examines a range of synthesis techniques, including carbonization, graphitization, and hydrothermal carbonization, and the challenges associated with these methods. It also outlines the extraordinary properties of graphene and its wide range of potential applications in electronics, energy storage, environmental technology, and healthcare. It also considers the environmental impact, economic benefits, and sustainability issues in the production process. 50 [41] Graphene-like carbon structure synthesis from biomass pyrolysis: a critical review on feedstock-process-properties relationship The review presents a summary of the most recent developments in the synthesis of graphene-like carbon (GLC) structures through microwave-assisted pyrolysis of biomass. It includes discussions on biomass selection, the effect of pyrolysis process parameters on product characteristics, and a comparison between microwave pyrolysis and traditional pyrolysis methods. It places particular emphasis on the potential of microwave pyrolysis as a cost-effective and renewable method for the production of graphene. 72 [42] Carbon-based materials derived from green and sustainable chemistry: current perspectives for electrocatalysis and energy applications This review focuses on the application prospects of emerging eco-friendly carbon-based materials derived from renewable or waste biomass resources in the fields of electrocatalysis and energy storage. This paper explores green synthesis routes aligned with sustainable practices (such as hydrothermal carbonization, pyrolysis, and low-energy chemical processing), while also delving into recent advances in heteroatom doping (e.g., N, S, P), and hierarchical structure engineering. It demonstrates that porous carbon materials, graphene, carbon nanotubes (CNTs), and carbon dots can significantly enhance the efficiency of fuel cells, supercapacitors, and rechargeable batteries. 5 [43] -
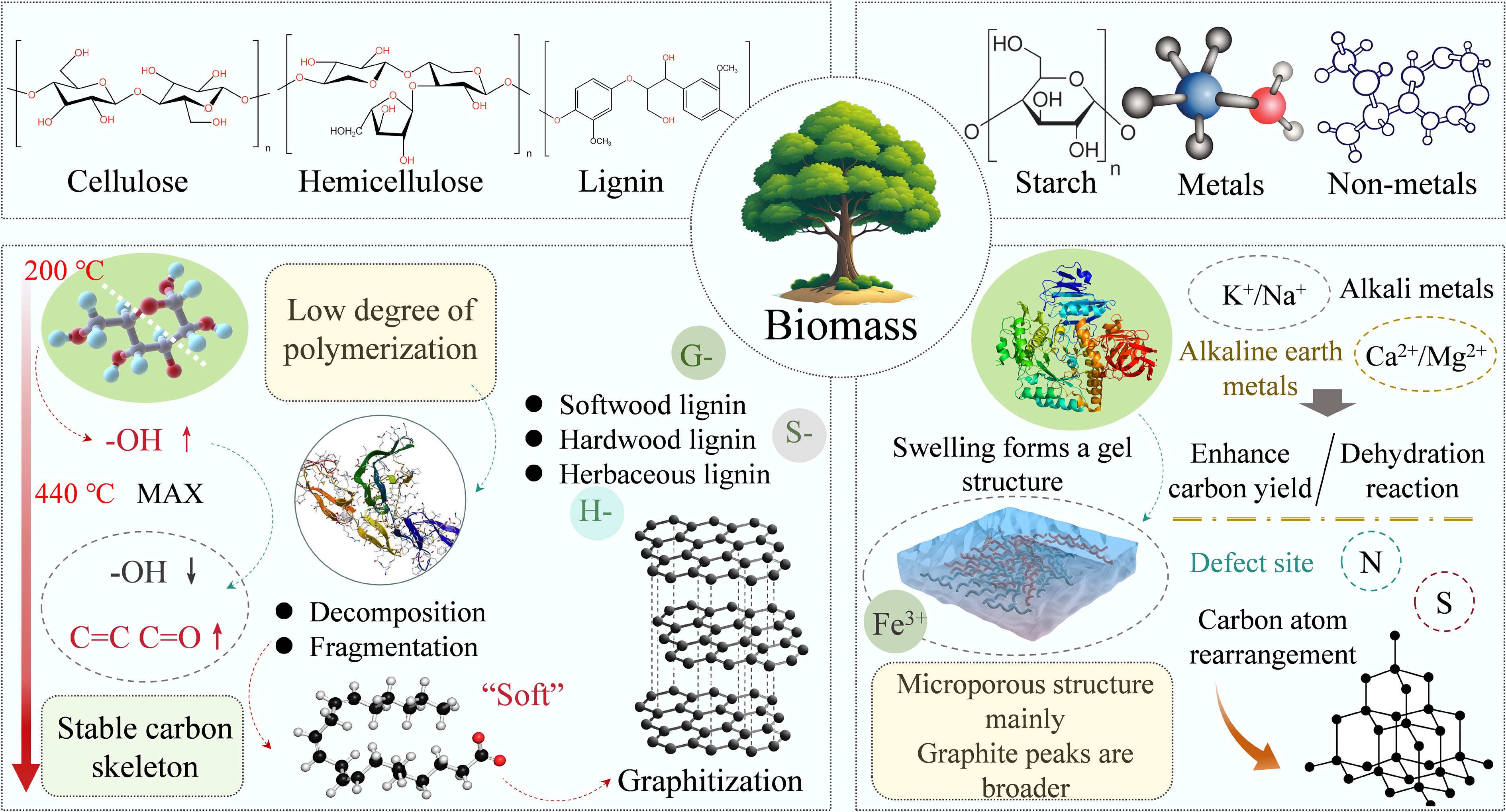
Biomass was defined as biological material produced directly or indirectly by photosynthesis according to the International Energy Agency[44]. It includes organic products, byproducts, and residues derived from forestry, agriculture, and households, as well as livestock production and waste[45]. The main components in biomass (cellulose, hemicellulose, and lignin) exert a certain influence on graphitization, while starch, metallic elements, and non-metallic elements significantly affect the graphitization process[46] (Fig. 4).
Three components of biomass
-
The composition of biomass influences the yield and quality of graphitized products. Biomass characteristics, particularly its cellulose, lignin, and hemicellulose content, play a crucial role in the formation of carbon-based materials. Therefore, a detailed analysis of the graphitization potential of each component was conducted.
The molecular structure of cellulose is tightly bound by hydrogen bonds and van der Waals forces, forming microfibrils at the microscopic level[47]. This structure endows cellulose with stable structural properties and excellent mechanical performance. Cellulose, as the core precursor for biomass graphitization, provides crucial support for the formation of graphitized ordered carbon structures through its unique molecular structure and pyrolysis reaction characteristics. During the initial stages of pyrolysis, the number of hydroxyl (−OH) groups first increases as the glucose units decompose, reaching a maximum at 440 °C[48], and dehydration at high temperatures leads to a decrease in the number of −OH groups, which leads to the formation of more double-bonded structures such as olefins (=C−H) or ketones (C=O). This process establishes the carbon framework foundation for graphitization, while the enrichment of double bond structures provides the essential structural units for subsequent aromatization and ordered stacking of carbon domains. As pyrolysis enters the high-temperature carbonization stage, the formation effect of C=C bonds is significantly enhanced[49]. As C=C bonds continuously link to form aromatic rings, these rings further orient themselves through π-π stacking interactions, gradually constructing the layered ordered structure characteristic of graphitization[50]. Meanwhile, the changes in chemical bonds during pyrolysis also exert precise control over the graphitization process. The −OH, C−C, and C=O bonds undergo continuous cleavage through dehydration, dehydrogenation, and bond-breaking reactions[51]. This effectively removes heteroatoms and disordered structures from the carbon skeleton. However, the relatively stable C−O−C structure, by moderately retaining oxygen atoms, regulates the defect density and surface chemistry of the carbon material, thereby preventing excessive disorder[49]. In summary, cellulose provides a controllable and efficient conversion pathway for biomass graphitization by regulating carbon framework restructuring, double bond formation, and heteroatom removal during pyrolysis. Furthermore, the structural advantages of cellulose itself ensure the stability of the carbon framework during pyrolysis, preventing high-temperature collapse, while also guiding the evolution of carbon structures along specific directions. This significantly enhances the order and crystalline quality of the final graphitized products.
As one of the core components of woody lignocellulosic biomass (accounting for 25%–35%)[52], hemicellulose, along with cellulose and pectin, constitutes the plant cell wall[53]. Different from the high crystallinity of cellulose, hemicellulose is composed of pentoses (arabinose, xylose), hexoses (galactose, glucose, mannose), and adipic acid (glucuronic acid) as diverse polysaccharide units, naturally exhibiting an amorphous branched structure[54]. Although this structural feature results in lower self-order, it creates favorable conditions for biomass graphitization during pyrolysis by regulating the evolution of the carbon skeleton. Additionally, compared to the highly polymerized long-chain structure of cellulose, the low polymerization degree of hemicellulose (typically < 200) makes it more prone to depolymerization and fragmentation under heating conditions, rapidly generating small-molecule carbon species[52]. At the same time, the alternating distribution of functional groups such as aldehyde and acetyl groups on its side chains will gradually be removed during pyrolysis, inducing a directed restructuring of the carbon skeleton and driving the carbon structure from disordered to aromatic[55]. Additionally, the 'soft properties' resulting from the low degree of polymerization of hemicellulose (making it easier to process compared to cellulose) can further optimize the performance of graphitized products.
Lignin is a naturally occurring aromatic polymer in nature, whose amorphous three-dimensional (3D) macromolecular structure is inherently rich in phenylpropane units composed of benzene rings and propyl side chains[56]. This provides an irreplaceable aromatic carbon source foundation for graphitization, representing its core advantage over cellulose and hemicellulose. The monomeric composition and chemical bond characteristics of lignin also influence the quality of biomass graphitization products. It consists of three phenylpropane units, categorized into H-, G-, and S-type, with significant variations in the proportion of lignin units across different biomass sources[57]. For example, softwood lignin is predominantly composed of G-type units[58], hardwood lignin contains a large number of S- and G-type units[59], and grass lignin consists of a mixture of three phenylpropane units[58]. This difference leads to variations in the ratio of chemical bonds, thereby influencing graphitization. The S-type unit features two methoxy groups attached to the aromatic ring, making it prone to elimination reactions during pyrolysis. In contrast, the G-type unit contains a higher proportion of C–C bonds, thereby exhibiting greater carbon skeleton stability[60]. This facilitates the maintenance of structural integrity at high temperatures. Meanwhile, the fundamental units of lignin are linked by covalent bonds, including ether, C–C, and ester bonds. Among these, the β–O–4 bond is the most prevalent, occurring in 43%–65% of major bonds. Other notable bonds are α–O–4, 4–O–5, 5–5, and β–β[61]. The ether bond readily breaks at moderate to low temperatures, rapidly releasing aromatic small molecules, while the C–C bond prevents carbon domain collapse, supporting the carbon skeleton's gradual evolution into an ordered graphite structure at high temperatures[62]. Additionally, the aromatic skeleton of lignin can overcome the limitations of its 3D amorphous structure at high temperatures. When the pyrolysis temperature exceeds 2,500 °C, the graphitization degree can reach over 94%[63]. At temperatures below 1,000 °C, the 3D structure is difficult to dissociate, making it challenging to form high-quality graphitic carbon[64]. Besides temperature, the pyrolysis atmosphere also influences the degree of lignin graphitization. For example, Ar serves as an inert atmosphere to prevent carbon loss and provide a stable environment for graphitization; CH4 under Fe catalysis supplies an additional carbon source for lignin pyrolysis, accelerating the formation of multilayer graphene; while H2 and CO2 remove disordered carbon impurities generated during pyrolysis through etching action[65]. Meanwhile, functional groups such as hydroxyl and carboxyl groups in the lignin molecules are gradually removed during pyrolysis, reducing the damage to the graphite structure caused by heteroatoms like oxygen and hydrogen[62].
In summary, cellulose, hemicellulose, and lignin in plant biomass each play crucial roles in graphitization. Cellulose regulates carbon skeleton restructuring and double bond formation through its stable structure and pyrolytic properties, effectively enhancing product order. Hemicellulose, with its low polymerization degree and soft properties, optimizes the product structure. Lignin provides an aromatic carbon source, facilitating the formation of high-quality graphitized carbon. Together, these three components underpin the efficient graphitization of biomass.
Starch content in biomass
-
Starch, as an energy storage substance in biomass, also serves as a unique precursor for graphitization. Its structural characteristics provide differentiated regulatory pathways for the graphitization process. Similar to cellulose, starch is a polymer composed of glucose monomers. However, unlike cellulose, which is insoluble in water, starch can swell to form a gel structure[66]. This property enables it to generate carbon materials with predominantly microporous structures and broader graphite peaks during graphitization (whereas cellulose-derived carbons typically exhibit mesoporous structures)[66]. The porous nature of the gel structure provides space for the growth of graphite microcrystals. In iron salt-catalyzed systems, the complex gel structure of starch particles allows Fe3+ precursors to intercalate between the starch and amylose molecules, achieving uniform dispersion. Concurrently, the excellent thermal stability of starch maintains the organic carbon framework surrounding iron precursors over extended periods, facilitating the gradual aggregation of iron species into iron oxide nanoparticles[67]. During pyrolysis, these small particles further transform into smaller iron carbide nanoparticles, which facilitates the formation of graphitic structures. Starch also exhibits excellent graphitization regulation capabilities, with its particle size, amylopectin content, and crystallinity closely related[68]. The content of branched starch is a key regulatory factor. Experiments demonstrate that taro starch (particle size of approximately ~5 μm)[69], possessing higher amylopectin content, yields derived carbon with significantly higher graphitization levels than sweet potato (approximately 15 μm)[70] or potato (approximately 25 μm)[71] starch carbon after KOH activation, exhibiting optimal electrochemical performance. This result confirms that high-branching starch can optimize the properties of derived carbon, enhance graphitization levels, and simultaneously circumvent the time-consuming and energy-intensive limitations of the template method[46], offering a potential pathway for low-cost preparation of porous carbon with varying degrees of graphitization.
In summary, starch possesses considerable graphitization potential, capable of expanding into the gel structure, exhibiting excellent thermal stability, and demonstrating superior graphitization regulation capability, thereby providing space, environment, and optimization for graphite growth and formation.
AAEMs in biomass
-
In addition to the three main biopolymers, biomass also contains AAEMs such as potassium (K), sodium (Na), calcium (Ca), and magnesium (Mg). These elements, although found in relatively small quantities, have a significant effect on the further graphitization process of biomass[72].
AAEMs can disrupt the stable structure of biomass polymers. In practical studies, biomass samples are generally impregnated in aqueous solutions of AAEMs for pyrolysis experiments to allow ionized metal cations to form ligand bonds with biopolymer molecules[72]. Because of the affinity of the impregnating salts for the organic groups of the polymers, they tend to disrupt existing chemical bonds. The prevailing hypothesis is that the interaction of metals with oxygen atoms results in the weakening of hydrogen bonds, which, in turn, causes the cleavage of the pyranose ring in cellulose[51]. During impregnation and pyrolysis, AAEMs have been demonstrated to reduce the stability of glycosidic bonds and hydroxyl groups, thereby facilitating the cleavage of glycosidic bonds, dehydration reactions, and ring scission. This process promotes the formation of smaller molecules while inhibiting the generation of undesirable byproducts such as levorotatory dextran[73]. These small molecules can reduce the crystallinity of cellulose and enhance its pyrolysis reactivity[74]. Additionally, AAEMs can influence interactions between biopolymers, optimizing the morphology and structure of biomass[75], thereby providing stable support for graphite microcrystal growth.
Two types of metals exert differing effects on graphitization. Alkali metals (K+, Na+) do not directly alter the decomposition temperatures of cellulose, xylan, and lignin[76]. Instead, they enhance pyrolysis efficiency and product quality by promoting the cleavage of tar molecules and guiding the carbon skeleton toward an ordered structure[77]. Alkaline earth metals (AEMs) (Mg2+, Ca2+), as stronger Lewis acids, exhibit higher affinity for oxygen-containing rings and more readily promote solid-state hydrolysis at elevated temperatures[72]. They can significantly reduce the activation energy for graphitization[78], typically demonstrating superior catalytic efficiency compared to alkali metals[79], thereby more effectively facilitating the formation of graphitic microcrystals. This difference is also reflected in reaction selectivity. Alkali metals tend to enhance carbon yield (this effect correlates with the electropositivity of metal cations; stronger electropositivity correlates with higher catalytic activity of cations in carbon formation)[80], while AEMs are more effective at lowering graphitization temperatures (decomposition temperature decrease order: Mg2+ < Ca2+ < K+ < Na+[78] or K+ > Na+ > Ca2+ > Mg2+[72]), and exhibit more pronounced suppression of nitrogen-containing impurities[81]. The mechanisms of action for both in cellulose pyrolysis also vary. Alkali metals primarily weaken the hydrogen bonding network, whereas AEMs facilitate dehydration reactions[72]. This difference is also evident in the primary pyrolysis products of cellulose, where the addition of potassium and sodium has resulted in lower L-glucose yields, whereas magnesium and calcium exhibited the opposite trend[82]. However, the two also have something in common. The presence of these metals can facilitate the formation of crosslinked cellulose, which results in char production[83]. Further studies revealed that both alkali metal and alkaline earth metal additives exhibited a reduction in aromatic and olefin yields, while simultaneously promoting cracking and dehydration reactions and increasing thermally derived COx and char yields[84].
In general, AAEMs significantly influence biomass graphitization, though their effects vary by type. Alkali metals enhance carbon yield, pyrolysis efficiency, and product quality, while AEMs reduce the activation energy of graphitization and lower the graphitization temperature. In cellulose pyrolysis, both types of metals promote the formation of cross-linked cellulose, thereby facilitating char production. The distinction lies in the fact that alkali metals primarily weaken the hydrogen bonding network, whereas AEMs promote dehydration reactions.
Non-metal elements in biomass
-
Nitrogen (N) and sulfur (S), as the primary non-metallic elements in biomass, exert influence on the graphitization process despite differences in their content and existence forms.
The core role of N in biomass graphitization manifests in four aspects: promoting structural ordering, regulating electronic properties, influencing chemical stability, and modifying surface functional groups[85]. First, the incorporation of N facilitates the structural ordering of carbon materials. This phenomenon is due to the embedding of N atoms, which act as defective points, prompting the rearrangement of adjacent carbon atoms and the formation of a graphite structure with a more ordered arrangement[86]. This process not only enhances its thermal stability but also significantly improves its electrical conductivity[87]. The introduction of N can also change the electronic properties of graphitic carbon materials. N atoms are more electronegative than carbon atoms, and when N atoms are doped into a carbon skeleton, additional electrons are introduced, affecting the Fermi energy level of the material and increasing the carrier density, significantly optimizing electrochemical activity[88]. Moreover, this effect is regulated by pyrolysis temperature, with approximately 900 °C being the optimal preparation temperature for N-doped graphitized carbon materials[89]. In addition, the doping forms of N, including pyridinic N, pyrrolic N, and graphitic N, exhibit distinct functionalities due to differences in electron cloud distribution. Pyridinic N, in particular, has been observed to enhance the electron affinity and electron transfer ability, as well as the activity of the oxygen reduction reaction (ORR)[90]. The incorporation of pyrrolic N into a material enhances its chemical stability and electron density, thereby improving the catalyst selectivity[91]. Graphitic N has been shown to enhance electrical conductivity and thermal stability[92]. This provides insights into the design and preparation of functionalized graphitized carbon materials.
S is also a common non-metallic element, and its role in the graphitization should not be overlooked, although it is much less abundant in biomass than N and oxygen in nature[93]. The impact of elemental sulfur on the graphitization of biomass is primarily evident in its capacity to facilitate structural transformation, induce alterations in electronic and chemical properties, and regulate the surface properties of materials[94]. Sulfur has the potential to facilitate the transformation of biomass into graphitic structures. During high-temperature heat treatment, sulfur functions as an intrinsic catalyst that facilitates the cleavage of unstable components and the formation of aromatic structures in biomass. This contributes to the formation of a more ordered graphite structure[95]. Second, it has been demonstrated that doping sulfur can alter both the electronic properties and chemical reactivity of graphitic carbon materials[94]. Sulfur atoms exhibit larger atomic radii and different electronegativities compared to carbon atoms. When sulfur is embedded in the carbon skeleton of graphitic carbon materials, it not only introduces a local charge imbalance and increases the local electron cloud density but also forms sulfur-doped-induced defective sites. These can be used as active sites to enhance the catalytic activity and electrochemical properties of the materials[96]. Furthermore, sulfur-doping can markedly affect the chemical stability and corrosion resistance of these materials, thereby conferring enhanced resistance to external factors. An appropriate amount of sulfur-doping can enhance the oxidation and heat resistance of the materials and improve their stability in harsh environments[97]. Conversely, an excess of sulfur-doping may cause the surface of the materials to become excessively active. This reduces the chemical stability of the materials and affects their service life[98]. Therefore, regulating the quantity and morphology of sulfur doping is of paramount importance to guarantee the efficacy of graphitic carbon materials. Furthermore, sulfur can influence the surface properties of graphitic carbon materials, including their hydrophilicity, adsorption capacity, and types of surface functional groups. The surfaces of sulfur-doped materials often contain polar functional groups, including thiol groups (-SH) and sulfide ether groups (-S-). These polar functional groups enhance the interaction between the materials and polar molecules, thereby improving its adsorption capacity and facilitating subsequent chemical modification[99]. It is noteworthy that in iron-catalyzed biomass graphitization, the promoting effect of sulfur is only manifested above 1,130 °C. At lower temperatures, sulfur completely inhibits Fe-induced graphitization, as Fe is bound to FeS and is therefore incapable of forming iron carbide[93].
In summary, regarding the two main non-metallic elements in biomass, the core role of nitrogen in biomass graphitization is manifested in promoting structural ordering of carbon materials, optimizing electrochemical activity, influencing chemical stability, and modifying surface functional groups. The influence of sulfur on biomass graphitization is primarily reflected in its ability to promote the formation of more ordered graphitic structures, alter the electronic properties and chemical reactivity of graphitic carbon materials, and modulate the surface characteristics of graphitic carbon.
In a word, this section systematically elucidates the influence mechanisms of various biomass components on the graphitization process, as shown in Table 2. In lignocellulosic biomass, the three major components play distinct roles: cellulose regulates the restructuring of the carbon skeleton and enhances ordering through its stable structure, hemicellulose optimizes the microstructural arrangement of the product, while lignin serves as an aromatic carbon source that directly facilitates the formation of graphitized carbon. Starch exhibits significant graphitization potential due to its gelation capability, excellent thermal stability, and regulatory properties, thereby providing a favorable environment for graphite growth. AAEMs exert differentiated effects: alkali metals enhance carbon yield and product quality, whereas AEMs effectively reduce the graphitization activation energy and temperature. Both promote cross-linking of cellulose into char, albeit through distinct mechanisms—alkali metals primarily disrupt the hydrogen bonding network, while AEMs preferentially facilitate dehydration reactions. As for non-metallic elements, nitrogen promotes structural ordering of carbon and optimizes electrochemical performance, while sulfur contributes to the formation of more ordered graphitic structures and modulates the electronic properties of the material.
Table 2. Influence mechanisms of various biomass components on the graphitization process
Component Function and impact Key property Cellulose As the core precursor, regulates carbon skeleton restructuring and double bond formation, significantly enhancing product order and crystallinity. Stable microfibril structure and bond transformations during pyrolysis (e.g., dehydration, C=C formation). Hemicellulose Readily depolymerizes during pyrolysis to generate small carbon species, driving carbon skeleton aromatization. Facilitate the formation of high-surface-area carbon materials, providing space for graphite microcrystal growth. Amorphous, low polymerization degree, soft properties, and branched structure. Lignin Provides an inherent aromatic carbon source, with benzene rings directly converting to graphitic carbon at high temperatures, facilitating the formation of high-quality graphitized carbon. Native aromatic polymer structure;
cleavable ether bonds and stable C–C bonds.Starch Provides microporous space, maintains the carbon framework, and optimizes graphitization levels, enabling low-cost preparation of porous carbon and offering a favorable environment for graphite growth. Gel structure, thermal stability, and high branching content. AAEMs Alkali metals: enhance carbon yield, pyrolysis efficiency and product quality.
AEMs: reduce the activation energy of graphitization and lower the graphitization temperature.Alkali metals: primarily weaken the hydrogen-bonding network.
AEMs: facilitate dehydration reactions.
Both types promote cross-linking of cellulose into char.Non-metal elements N: promotes structural ordering, enhances electrical conductivity and electrochemical activity, modifies surface functional groups.
S: promotes the formation of more ordered graphitic structures, alters the electronic properties and chemical reactivity of graphitic carbon materials, and modulates the surface characteristics of graphitic carbon.N and S: incorporate into the carbon matrix through doping, introducing defective sites and altering the local electron density and charge distribution. -
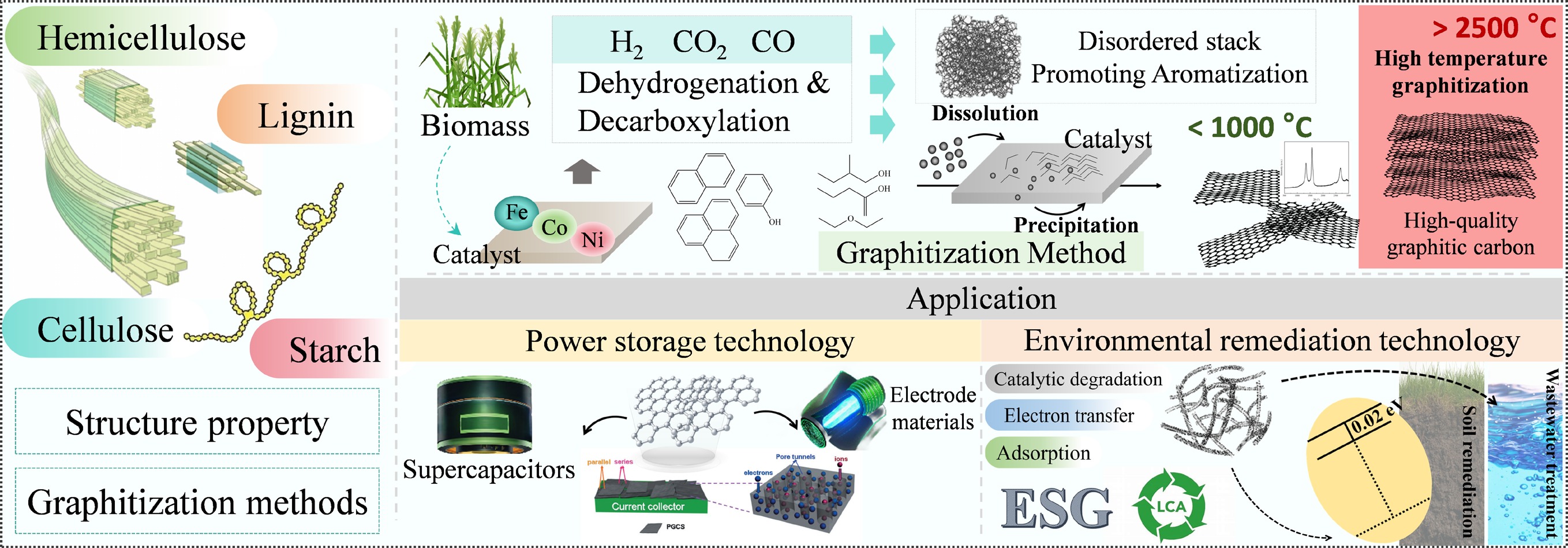
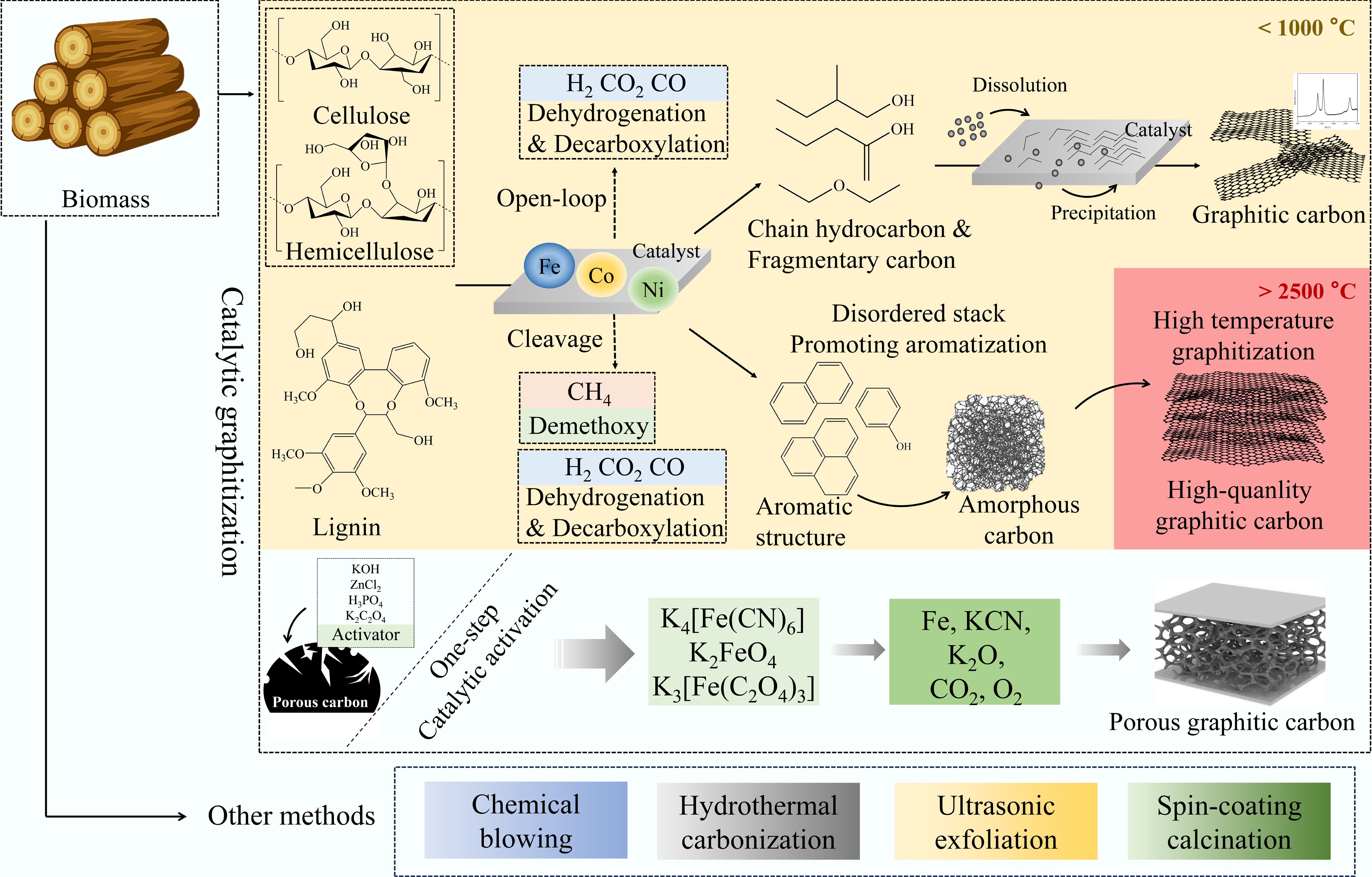
Biomass catalytic pyrolytic graphitization is a technology that achieves the efficient conversion of biomass into graphite-based carbon materials through catalyst-mediated processes. This technology deeply couples pyrolysis with catalysis, not only significantly enhancing biomass carbon conversion rates and optimizing product structures, but also controlling graphitization temperatures within a narrow range below 1,000 °C[27], thereby overcoming the high-energy-consumption bottleneck of conventional graphitization processes. During pyrolysis, biomass is first heated to a specific temperature under anoxic/hypoxic conditions, yielding volatile products and carbon-based residues. Subsequently, catalysts such as metal salts, metal oxides, and acids/bases drive the directed rearrangement of carbon atoms within the carbon-based intermediates and residues, forming a regular graphite structure while efficiently removing other elements and impurities from the biomass[100]. Additionally, metal particles can be used as rigid templates to facilitate the formation of supplementary pores[101], further optimizing the material's microstructure. By meticulous engineering of the catalytic system and optimization of the pyrolysis conditions, the biomass-catalyzed pyrolytic graphitization method can efficiently generate graphitic carbon materials with a highly ordered structure and exceptional electrical conductivity, along with distinctive functional properties.
Catalytic graphitization
-
The exceptional electronic conductivity of carbon materials is the core foundation for achieving low internal resistance, rapid electron transfer, and structural stability, directly determining the material's reaction rate, power density, and cycle life. Enhancing the degree of graphitization is a key method for strengthening the conductivity of carbon materials[102]. Traditional graphitization relies on temperatures exceeding 1,000 °C to induce the rearrangement of carbon atoms, forming sp2 conjugated structures and well-ordered graphite phases[103]. However, biomass-derived carbon typically exhibits amorphous characteristics with complex and diverse structural morphologies[104]. Even when heated to 2,500 °C, it remains challenging to convert it into a perfect graphite structure. Furthermore, high-temperature carbonization ( ≥ 1,000 °C) not only consumes a considerable amount of energy but can also result in the collapse of numerous pores, thereby leading to the underdevelopment of pore structures and a reduction in the specific surface area (SSA)[105]. This severely limits its application scenarios.
Catalytic graphitization, as a key technology for overcoming the aforementioned bottlenecks, enables efficient conversion of amorphous carbon into graphitic carbon at relatively low temperatures (≤ 1,000 °C)[27]. This process is based on a reduction in the activation energy for carbon phase transformation in the presence of transition metal elements such as Ni, Co, and Fe[106,107]. Additionally, metal particles can act as hard templates to create additional pores[101]. This method does not require extreme temperatures, thereby reducing energy consumption while preserving and even optimizing the porous structure of carbon materials.
Additionally, among various catalysts, iron-based compounds (such as FeCl3, FeCl2, K4[Fe(CN)6], and K2FeO4) have emerged as highly efficient catalysts for producing high-quality graphitic carbon materials due to their high catalytic efficiency and controllable costs[108]. Its mechanism of action is primarily divided into two categories: (1) The carburization effect plays a leading role in the synthesis of graphitic carbon[109]; (2) Iron combines with carbon atoms from the precursor to form a dense Fe3C layer. At temperatures exceeding 1,100 °C, carbon precipitation from Fe3C occurs, resulting in the transformation of Fe3C into α-Fe. Subsequently, the carbon atoms that have diffused outward aggregate on the surface of the self-generated iron template, forming a dense 2D carbon layer[110].
Catalytic graphitization of lignin. The core challenge with lignin lies in its complex and heterogeneous chemical structure, which poses significant hurdles for its conversion into high-value materials. However, the polyaromatic ring structure inherent to lignin makes it an ideal precursor for producing aromatic carbon materials such as carbon fibers and electrodes, as well as graphite-based materials[27,82,111].
In recent years, iron-based catalysts have become the mainstream catalysts for the graphitization of lignin. Organic gold lignin, hydrolyzed lignin, and sulfonated lignin, three typical forms of lignin, have all been proven to serve as effective iron-catalyzed graphitization precursors[112]. During heating, lignin melts into a liquid intermediate phase, enabling full contact with the catalyst and resulting in a uniformly distributed graphite structure[59]. However, critical limitations of this technology remain prominent. The resulting graphite products generally exhibit low graphitization levels, with both product quality and crystal size falling short of graphite materials synthesized from lignocellulosic and carbohydrate precursors[113]. It is possible that the high bond dissociation energy was responsible for the poor graphitization of lignin, in addition to catalyst poisoning[57]. Subsequent research also explored using Cu[114] as a catalyst. However, its effectiveness and cost were not as favorable as those of Fe.
Similarly, iron-based catalysts can convert lignin-derived carbon materials into graphitic carbon materials at low temperatures[115]. A process combining freeze-drying, carbonization, and graphitization can elucidate the formation mechanism of graphitic carbon: (1) As the temperature rises to 250 °C, lignin rapidly solidifies and partially carbonizes, maintaining a porous microstructure[116]; (2) When the temperature is further increased from 250 to 450 °C, the phenyl rings and side chains in the lignin structural framework decompose[117]; (3) When the temperature is raised to 1,200 °C, the graphitic carbon sheets derived from phenyl rings are chemically bonded by sp3 carbon atoms, forming a graphite structure, and eventually, higher-quality graphitic carbon is formed after the cooling process[118].
Catalytic graphitization of cellulose. Cellulose, as a key component of biomass, possesses the inherent potential to be converted into high-quality graphitic carbon due to its high crystallinity and well-ordered glucose unit framework. Catalysts such as iron can effectively trigger the graphitization process[119]. To further improve the graphitization degree of cellulose and enhance the performance of cellulose-based graphitic carbon, pretreatment techniques and catalysts play an important role.
Pretreatment technology can break down the cellulose structure, laying the groundwork for subsequent graphitization. The acid–base–acid process hydrolyzes cellulose microfibrils into thin-layered 2D crystals, which are then carbonized at high temperatures of 1,200 °C. During this process, glucose units lose oxygen and hydrogen atoms, and the six-carbon units are reorganized along the cellulose crystal structure, forming highly ordered 2D graphite-like carbon crystals[120]. Acetic acid pretreatment promotes cellulose conversion into oligosaccharides through mild hydrolysis, allowing cellulose units to shed in the form of nanocellulose sheets and subsequently transform into graphitic carbon materials at 700 °C[121], significantly reducing energy consumption. Furthermore, adding melamine during the initial pyrolysis stage (1,000 °C) serves as a multifunctional pretreatment strategy. It functions both as a reaction template to guide structural formation and as an N source and pore-forming agent. When mixed with FeCl3, it can form N-rich graphitic structures at 950 or 1,100 °C[122]. In catalytic systems, transition metals such as iron, cobalt, and nickel can all serve as catalysts for cellulose graphitization. They not only accelerate the graphitization reaction by lowering activation energy but also function as structural directing agents, inducing the formation of randomly stacked graphitic carbon nanosheets with wrinkles[100]. During the spark plasma sintering process at 1,250 and 1,350 °C, cellulose nanofibers are transformed into graphitic carbon sheets that connect to sintered engineering ceramics, demonstrating the critical role of pretreatment and catalysts in the cellulose transformation process[123].
In summary, transition metals, such as iron, cobalt, and nickel, effectively promote the graphitization of cellulose and improve the performance of cellulose-based graphitic carbon materials. The application of various pretreatment techniques has further optimized the structure of cellulose-based graphitic carbon.
Catalytic activation methods
-
To achieve outstanding electrochemical performance, carbon materials must possess both a high degree of graphitization and an appropriate porous structure within their carbon matrix. However, obtaining this is a significant challenge because of the trade-off[124]. Porous carbon typically contains abundant disordered amorphous carbon, which can be readily produced during activation but is prone to collapse at ultra-high temperatures[105]. The sp2 conjugated carbon structure required for high graphitization, though dependent on high temperatures for formation, is easily etched and destroyed by activators. To overcome this technical bottleneck, catalytic activation emerged as a solution.
Traditional chemical activation methods predominantly employ either direct activation (directly activating carbon materials) or a two-step carbonization-activation process. Specifically, this involves subjecting a mixture of carbon precursors and activators (or dehydrating agents) to high-temperature carbonization under an inert atmosphere. Common activators include KOH, H3PO4, and ZnCl2[125]. The pore structure (e.g., SSA, pore diameter, pore volume, and pore geometry), and degree of graphitization of activated carbon can be manipulated by utilizing various activation parameters, including the type of activator, ratio of biomass to activator, activation temperature, and activation time. Temperature is a key regulatory variable, and moderate heating can promote pore development by leveraging the high reactivity of the activator[126]. HCOOK and KOH activation can be used to prepare carbon materials with high graphitization, for example, soybean hulls can be converted into N-doped porous graphene by calcination and KOH activation[127,128]. The formed porous graphitic carbon is ultra-thin (thickness of approximately 3.8 nm), has a large SSA (1,816 m2/g), and has a high proportion of mesopores. However, excessively high activation temperatures or excessive activators can significantly increase the pore size and even collapse and destroy the carbon skeleton, resulting in a lower SSA, unreasonable pore size distribution, and poor carbon specific capacity[129].
To further simplify the process and enhance synergistic effects, researchers proposed a simple and economical one-step carbonization process that achieves simultaneous activation and graphitization. This process introduces the activator and graphitization catalyst into the carbon precursor concurrently, accomplishing both pore formation and graphitization in a single reaction. As a specific case, the preparation of bio-oil-derived honeycomb porous graphene carbon (BPGC) involves a combination of chemical activation and Ni(NO3)2 catalyzed graphitization[130]. A similar approach was used to synthesize BPGCs from coconut[131], rice husk[132], and dried kelp[133]. The products generally exhibit outstanding characteristics with SSA exceeding 1,000 m2/g, and IG/ID ratios exceeding 1.0. Controlling the ratio of KOH to FeCl2 is key when optimizing graphitization and amorphous structures[134]. However, KOH combined with transition metal salts tends to form insoluble precipitates in aqueous solutions, leading to difficulties in the uniform distribution of KOH and graphite catalysts in carbon matrices. Therefore, potassium carbonate (K2C2O4) and ZnCl2 are frequently used as activators instead of KOH during the synthesis of BPGC. In particular, ZnCl2 is frequently used in conjunction with FeCl3 to fabricate porous graphene-like nanosheets from coconut shells via a one-step carbonization method[135]. In this process, Fe converts amorphous carbon into graphitic carbon through the formation and decomposition of carbide intermediates. Meanwhile, ZnCl2 activates the carbon skeleton to form numerous pores[136]. Furthermore, the chelation of Fe2+/Zn2+ with functional groups present in biomass facilitates the achievement of a uniform distribution of graphite structures and pores within the resulting material. The conversion of cellulose into porous GLC nanosheets based on a composite of FeCl3 and ZnCl2 demonstrated that the higher the ratio of ZnCl2 to biomass, the greater the degree of graphitization and the larger the total pore volume[137]. This fully demonstrates the synergistic enhancement effect of activation and graphitization.
To enhance productivity and reduce the production cost of graphitic carbon, researchers have been dedicated to investigating a catalyst that can facilitate simultaneous activation and graphitization. Iron-based compounds represent a core category in this endeavor. K4[Fe(CN)6] catalyzed carbonization of corn stover at 1,100 °C, followed by hydrochloric acid deactivation, yields porous graphene nanosheets with a SSA of 540 m2/g and IG/ID = 1.6[138]. This may be attributed to the combined effect of K etching and iron graphitization. Another iron compound, K2FeO4, has also been demonstrated to promote graphitization of carbon[139]. K2FeO4 exhibits a high degree of versatility and can be used as an effective catalytic activator for a wide range of biomasses, including sodium lignosulfonate[140], bamboo[139], peanut shells[141], dandelion flower stems[142], and wood[143]. 3D porous graphitic carbon was prepared from bamboo using K2FeO4. The K can act as an etchant to produce a large number of pores. The prepared carbon exhibits high graphitization and a high SSA (1,732 m2/g). The material exhibits an elevated specific capacitance value of 222.0 F/g at a current density of 0.5 A/g[139]. The pyrolysis temperature is a critical variable in regulating the synthesis and decomposition of its derived intermediate Fe3C. At temperatures up to 700 °C, the Fe3C layer can be retained within the graphitic carbon without the outward diffusion of Fe3C-free carbon[144]. An example of a product that can be obtained by this process is graphene-encapsulated Fe3C embedded in CNTs. This material is produced using a pyrolysis reaction, in which glucose, melamine, and an aqueous solution of FeCl3 are combined. The resulting product has been shown to perform well in hydrogenation reactions, including those involving nitroaromatic hydrocarbons and C–C bonds, at relatively low temperatures, ranging from 40 to 60 °C[145].
This discovery led to the identification and implementation of another green and efficient activator, K3[Fe(C2O4)3][146]. At a high temperature, K3[Fe(C2O4)3] undergoes thermal decomposition, resulting in the formation of K2C2O4 and FeC2O4[147]. K2C2O4 is an effective activator with weak corrosive properties. Therefore, the remarkable activated graphitization effect of K3[Fe(C2O4)3] is mainly due to its decomposition products, K2C2O4 and Fe[148]. The derivatives of K2FeO4 and K4Fe(CN)6, KOH, or KCN, are corrosive or toxic, whereas K3[Fe(C2O4)3], in comparison with K2FeO4 and K4Fe(CN)6, exhibits more environmentally friendly and benign properties and contributes to a mild activated graphitization process.
The above analysis indicates that the graphitization and activation effects of K2FeO4, K4Fe(CN)6, and K3[Fe(C2O4)3] depend on the specific type of K and Fe present. Since the ratio of potassium to iron remains fixed within the compounds described above, variations in the SSA and degree of graphitization of BPGC can be achieved by adjusting either the biomass-to-catalyst ratio or the carbonization temperature.
In conclusion, carbon materials must simultaneously possess high graphitization levels and suitable porous structures to achieve outstanding electrochemical performance. However, the inherent contradiction between these two properties presents a critical technological bottleneck. Traditional chemical activation primarily employs direct activation or a two-step carbonization-activation process, using activators like KOH and H3PO4 to regulate pore structure and graphitization degree. However, excessively high temperatures or excessive activators can easily cause carbon skeleton collapse and performance degradation. The one-step carbonization process simultaneously introduces activators and graphitization catalysts, enabling concurrent activation and graphitization. The resulting products exhibit both high SSA (often exceeding 1,000 m2/g) and high graphitization degree (IG/ID > 1.0). Iron-based compounds serve as core catalysts for simultaneous activation-graphitization. K4[Fe(CN)6] and K2FeO4 exhibit synergistic effects through potassium etching and iron-catalyzed graphitization, though their derivatives may be corrosive or toxic. K3[Fe(C2O4)3], with its thermally decomposed products (K2C2O4, Fe) exhibiting low corrosivity and environmental friendliness, emerges as a more promising green activation-graphitization reagent. The efficacy of these iron-based composite reagents relies on the synergistic interaction between potassium and iron. In practical applications, precise control over the SSA and graphitization degree of carbon materials can be achieved by adjusting the biomass-to-catalyst ratio and carbonization temperature, providing an effective pathway for the low-cost, large-scale production of high-performance carbon materials.
High-temperature graphitization method
-
High-temperature graphitization refers to a process where biomass is subjected to elevated temperatures within an inert atmosphere, using graphite containers as the reaction vessel. A portion of biochar is capable of generating graphitic carbon through high temperatures without catalysts[149]. Applying pressure simultaneously during heating can further enhance graphitization efficiency[150−152]. This has become a key optimization direction for high-temperature graphitization technology.
The precursor structure is a key factor influencing high-temperature graphitization efficiency, with significant differences observed between lignin-derived and cellulose-derived carbon. After high-temperature graphitization, cellulose-derived carbon is composed almost entirely of carbon microcrystals, whereas lignin-derived carbon retains some structural skeleton[153]. This structural divergence directly leads to distinct product characteristics. The results demonstrated that the graphiticity of the graphitized lignin-derived samples at 2,800 °C was 89.53%, with a layer spacing of 0.3363 nm and a conductivity of 104.6 S/cm. In contrast, the graphitization of the cellulose-derived samples was 76.74%, with a layer spacing of 0.3374 nm and a conductivity of only 48.8 S/cm[153]. Lignin possesses structural advantages. Compared to the sp3 hybridized structures found in other biomass precursors, its sp2 hybridized aromatic ring structures are more prone to undergo ring-expansion rearrangements during graphitization, forming large-area continuous graphite layers[154,155]. Even without catalysts, treating lignin at 1,300 °C can produce graphitic carbon sheets with low defect rates and suitable interlayer spacing through its original aromatic monomers and highly cross-linked ether and condensation bonds[156].
However, high-temperature graphitization still faces bottlenecks. The intertwined microcrystals in the carbon-based material, even at such extremely elevated temperatures, strongly limit the expansion of the contiguous lamellar graphite regions, resulting in the formation of a vortex layer structure with a considerable number of structural defects, which ultimately leads to a relatively low electronic conductivity[153]. Although all the components of lignocellulosic biomass decompose during high-temperature pyrolysis, the original macroscopic structure of the biomass still plays an important role in the formation of graphene products[157]. The selection of biomass with flake shapes, such as flower petals, leaves, and grass blades, has the potential to be a valuable source for the synthesis of high-quality graphene products.
Other graphitization methods
-
In addition to the previously mentioned catalytic activation and high-temperature graphitization methods, researchers have developed several novel graphitization techniques suitable for different scenarios. These include the chemical blowing method, hydrothermal carbonization (HTC), ultrasonic exfoliation, and the spin-coating calcination method.
Chemical blow molding is a technique used in the preparation of foams in which a chemical reaction generates gases to form vesicles. In the preparation of graphitic carbon materials, this technique is used to generate graphene foams or graphene sponges with high porosity, low density, and a high SSA[158]. The preparation of graphitic carbon typically involves the use of NH4Cl as a blowing agent, which releases gases (NH3 and HCl) that thin the walls of the biomass precursor. Finally, the biomass is subjected to a process of stripping, whereby it is transformed into a porous graphitic carbon material[159]. Urea[160] and melamine[161] are also effective foaming agents to prepare high pore volume (0.94 cm3/g) and large surface area (~1,998 m2/g) N-doped graphitic carbon nanosheets for the generation of N-doped carbon nanosheets[162]. The chemical blowing method is a simple operation requiring the uniform blending of the activator and carbon source. The activator releases gas during the pyrolysis process, which can thin the walls of the carbon source. The pyrolysis of polymer precursors with a low degree of orientation is an easier process than that of conventional bulk carbon sources. This is because the energy required for the parallel alignment of (002) graphite layers is considerably lower with oriented precursors[159]. The heating rate represents a key variable in the matching reaction of gas release and polymer curing. The extent to which this variable influences the process is contingent upon the kinetics associated with the decomposition of the activator, the growth of bubbles, the initiation of bubble nucleation, and the rate of gas diffusion.
HTC typically occurs in water at temperatures below 200 °C and autogenous pressure. No catalysts or other hazardous substances are necessary for this process to occur[163,164]. Carbon obtained through this process typically takes the shape of a sphere with a relatively low surface area, and abundant oxygen-containing functional groups. However, it exhibits poor electronic conductivity[161,165]. When the HTC process is conducted at 180 °C, glucose-derived carbon primarily consists of cross-linked furan rings and ketone-type fatty chains, requiring high-temperature annealing to transform into graphene-like structures[166,167]. The application of post-activation/charring methods has the potential to significantly enhance the textural properties of a material and to improve its structural ordering. For example, oil palm empty fruit bunches were initially subjected to a hydrothermal environment at 250 °C for a period of 20 min prior to the production of biochar. Following calcination at 800 °C, the biochar was transformed into porous graphene-like nanosheets[168]. Highly curved graphite structures were prepared from coconut shell dust using HTC and subsequent pyrolysis techniques, and samples without hydrothermal pretreatment yielded amorphous carbon during pyrolysis[169], suggesting that the hydrothermal process is important for the formation of graphite-like structures.
Ultrasonic exfoliation is an effective top-down method for separating graphene and graphitic carbon[170]. Graphitic carbon materials can be obtained by stripping biochar prepared by pyrolysis. Meanwhile, exfoliation effectively enhances the porosity of carbon materials[171−173]. For example, graphene can be produced via the pyrolysis of commercial sodium alginate at 900 °C in Ar[174]. The carbon-containing residue was sonicated at 250 W for 30 min. The suspension was then freeze-dried to obtain graphene materials. More interestingly, the graphene material showed good nitro to amino reduction activity in the presence of H2. The active site of the hydrogenation reaction can be attributed to the formation of a frustrated Lewis acid-base pair. Ultrasonic exfoliation of KOH-activated carbon from peanut shells in a 10% H2SO4 aqueous solution yielded a few layers of graphene-like materials[175].
A spin-coating process using precursor solutions followed by calcination is an effective method for the synthesis of GLC materials, with the potential to produce either monolayer or multilayer architectures[176]. The success of this process depends on the selection and handling of precursors and the precise control of calcination conditions, which together determine the quality and properties of the final product. Chitosan is a natural polysaccharide composed of alternating N-acetylglucosamine and glucosamine units. When a chitosan solution is applied to a quartz surface by spin coating and calcined at the appropriate temperature, the chitosan decomposes to form an N-containing carbon layer, which forms an N-doped graphene-like structure[177]. The N-doping process has the potential to significantly enhance the electrochemical activity and electrical conductivity. The calcination process is a key stage in the production of the final material, as it exerts a profound influence on its quality. The selection of calcination temperature, holding time, and atmosphere (e.g., inert gas, oxygen, or ammonia, etc.) has a significant impact on the microstructure and doping level of carbon materials[178]. A higher calcination temperature is conducive to the graphitization of the material, yet it may also result in a reduction in the quantity of N doping. Conversely, an ammonia atmosphere can facilitate the N doping effect, yet it may also impact the pore structure of the material[161]. Furthermore, the optimization of the spin-coating process is of paramount importance for the enhancement of the quality of the films. The spin-coating velocity, the evaporation rate of the solvent, and the uniformity of coating directly influence the microstructure and thickness of the film, which in turn impact the formation of the carbon layer and the distribution of dopant elements during the calcination process[179]. By accurately regulating these variables, it is possible to fabricate N-doped graphene-like films with exceptional electrochemical and physical characteristics. These methods are summarized in Fig. 5.
Chemical blow molding generates gas foaming through chemical reactions, thinning the walls of biomass precursors before final exfoliation to achieve a porous graphitized structure; HTC produces carbon with poor conductivity but low surface area and abundant oxygen-containing functional groups at temperatures below 200 °C using autogenous pressure—crucial for forming graphite-like structures. Ultrasonic exfoliation enhances the porosity of carbon materials and effectively separates graphene from graphitic carbon. Spin-coating calcination produces N-doped graphene-like films with outstanding electrochemical and physical properties. This depends on the selection and treatment of precursors, precise control of calcination temperature, holding time, and atmosphere, as well as the management of spin-coating velocity, solvent evaporation rate, and coating uniformity.
In summary, biomass graphitization methods include catalytic, high temperature, and other approaches. The catalytic method utilizes metals such as iron, cobalt, and nickel to efficiently convert lignin and cellulose into conductive, structurally ordered graphitic carbon at lower temperatures (≤ 1,000 °C). Through simultaneous activation and graphitization processes, coupled with further catalyst development, this method achieves higher yields, reduced production costs, and controllable pore structures. The high-temperature method directly treats biomass at elevated temperatures under an inert atmosphere, eliminating catalyst requirements but incurring high energy consumption. Lignin, due to its aromatic structure, readily forms highly graphitized carbon materials. Other methods—such as chemical foaming, hydrothermal carbonization, ultrasonic exfoliation, and spin-coating calcination—suit specific production scenarios, enabling the preparation of graphitic carbon materials with porous structures or nitrogen doping.
-
BBGC, as a high-performance carbon material derived from biochar, demonstrates broad application potential in the energy storage field, including supercapacitors and sodium ion batteries (SIBs)[180,181]. Supercapacitors are utilized in a multitude of applications in our daily lives, they can be integrated into advanced devices for the generation and storage of electrical energy. SIBs, on the other hand, are excellent materials promising to replace LIBs, and have good prospects for development in the fields of stationary energy storage, electric vehicles, portable electronic devices, and so on. The electrode material represents the most critical aspect in the preparation of batteries and capacitors[100], one of the most representative materials is BBGC, which is capable of large-scale production from biomass waste through clean and green technologies, thus realizing high-value utilization of biomass.
Supercapacitors
-
Supercapacitors exhibit several advantageous characteristics, including high power density, rapid charging, rapid discharging, and ultra-long lifetime[182]. BBGC, with its large SSA, interconnected pore structure, excellent electrical conductivity, and surface electrochemical stability, perfectly equips it to meet the application requirements of supercapacitors. As an illustrative example, a 3D structure of graphene-like materials can be obtained via chemical blowing with NH4Cl, followed by activation with CO2 and KOH[183]. The high SSA (3,657 m2/g), interconnected hierarchical pore network, and high degree of graphitization exhibited by 3D supported graphene result in a high specific capacitance of 175 F/g in ionic liquid electrolytes. The construction of supercapacitors using this 3D graphene resulted in a maximum energy density of 74 Wh/kg, and a maximum power density of 408 kW/kg[183]. Its advantage lies in the covalent sp2 bond structure between carbon atoms, which enhances the material's conductivity while reducing the overall resistance of the system. For example, the combination of NH4Cl and (NH4)2CO3 can be used as a blowing agent to produce porous GLC with a small number of graphene layers. This particular material exhibits an exceptionally high SSA, and a remarkably high sp2-to-carbon ratio. These characteristics result in an exceptionally high power density and high energy density in organic electrolytes[19]. Moreover, the GLC materials mentioned above have excellent cycle stability due to their electrochemically stable surfaces.
Carbon-based supercapacitors rely mainly on the surface energy storage mechanism, and in general, the specific capacitance increases with the increase of SSA[184]. However, both theoretical and experimental studies have confirmed that simply increasing the SSA is not the optimal solution[146]. A large SSA or pore volume reduces the volumetric density of the carbon material, which in turn limits the energy and power density. In fact, the pore structure that governs ion migration and adsorption is the key factor influencing the formation of double-layer capacitance. Compared to the pursuit of large SSA alone, a reasonable pore size distribution is more crucial for achieving high specific capacitance[185]. Small pores inhibit the diffusion of ions, and too large pores are unfavorable for the attachment of ions, all of which affect the specific capacitance of the supercapacitor[186]. It has been demonstrated that the optimal pore size of carbon materials within organic and aqueous electrolytes is 0.8 and 0.7 nm, respectively[65,186]. However, the dense and slow diffusion of ions in sub-nanometer micropores limits the participation of active sites in energy storage at high current densities. This results in a limitation of rate capability and power density, which in turn affects the overall performance of the micropores[187]. The utilization of mesopores as storage layers enables the creation of rapid ion transfer pathways, accelerates the kinetics of ion diffusion, and exhibits a high multiplicative capacity during the charging and discharging processes, thus improving overall battery performance[188]. However, the presence of excessively large pores can result in a reduction in SSA, and an associated decrease in specific capacitance and energy density[189]. In any case, it is recommended that well-developed pores have an appropriate pore size distribution. Such distribution should provide abundant active sites for ionic interactions, thus ensuring abundant ion storage, while also providing fast-connecting mass transfer channels. Therefore, it is of paramount importance to achieve an optimal ratio of ionizable materials, and a reasonable combination of micropores and mesopores, to ensure optimal rate performance, high energy, and power density[190].
The prevalence of defects in carbon materials ensures a high SSA and a significant number of active sites for ion adsorption and desorption. However, defects also reduce the stability and electrical conductivity of carbon materials. The incorporation of carbon materials with high electrical conductivity can markedly diminish internal resistance, thereby enhancing multiplicity performance and energy density[190]. It has been demonstrated that the graphitization of carbon materials can enhance their conductivity and stability, although this process is accompanied by a reduction in porosity and SSA[105]. Therefore, the challenge of achieving an optimal balance between porosity and electrical conductivity has become a critical concern. Porous graphitic carbon structures combining a reasonable number of disordered and graphitized structures have abundant active sites and excellent electrical conductivity, which are conducive to ion/electron transfer for superior electrochemical performance[190]. Therefore, balancing or even mitigating the trade-off between porosity and conductivity has become a pressing issue. For carbon-based materials in supercapacitor applications, maintaining an approximately equal ratio of graphitized to amorphous carbon (e.g., IG/ID close to 1.0) is an effective strategy for achieving the requirements of good conductivity and high SSA. This balance is critical to achieving a high energy and power density.
SIBs
-
Compared to LIBs, SIBs are considered an alternative energy storage technology with great potential due to the abundance of sodium and its low cost[191]. As an important energy storage system, research and development of SIBs have focused on finding electrode materials with high energy density, long cycle life, and low cost[192]. Graphitic carbon materials are considered ideal electrode materials due to their excellent electrical conductivity, chemical stability, and tunable microstructure[193].
SIBs and LIBs operate on essentially the same principle; both of them realize energy storage and release through the embedding and de-embedding of alkali metal ions in the electrode materials. However, the large radius of sodium ions makes it difficult to achieve efficient embedding and de-embedding in conventional graphite materials. To overcome this limitation, research has turned to the use of graphene nanocomposites and polymer/graphene nanomaterials to achieve high specific capacitance and high current density (~2,000 mAh/g and > 100 mA/g, respectively)[192]. Effective intercalation and reversible de-insertion of sodium ions in graphite through a co-intercalation mechanism, a process that significantly improves the cycling stability and power capability of the anode[194]. In addition, experiments conducted in an ether-based electrolyte showed that the graphite anode was able to achieve a sodium storage capacity of more than 100 mAh/g, and a high Coulombic efficiency of more than 99.87%, which remained stable after 1,000 cycles[195]. To increase the graphite spacing and improve sodium ion intercalation, researchers developed expanded graphite anodes. This material successfully increased the layer spacing to about 0.43 nm through oxidation and partial reduction treatments, resulting in a high sodium storage capacity of about 300 mAh/g[196]. Graphene and its oxides have also been widely investigated as anode materials for SIBs. Through doping strategies, such as phosphorus doping, not only are abundant sodium adsorption sites introduced between graphene layers, but also the mobility of electrons and sodium ions is enhanced by enlarging the layer spacing (d-spacing), which results in high cycling capacity and excellent multiplicity performance[197]. Taking advantage of graphene's high electrical conductivity and mechanical flexibility, the researchers have also developed graphene-based composite anodes, which, by combining them with reactive particles such as Stannic (Sn), Stannic dioxide (SnO2), and titanium dioxide (TiO2), achieve high sodium storage capacity through alloying/conversion reactions, intercalation reactions, or redox reactions[198]. Furthermore, it is possible to modify the electronic structure and surface chemistry of graphitic carbon materials by doping them with heteroatoms, such as N, boron, or sulfur, or by introducing functional groups, such as oxides or hydroxyls[199]. The combination of active phases, including metal oxides, sulfides, and organic materials, in graphene-based composites has been demonstrated to enhance the storage capacity of SIBs, while simultaneously improving the electrical conductivity and structural stability of the electrodes. In practice, graphitic carbon needs to be suitably modified according to the application.
-
BBGC, as a biomass-derived graphitic carbon material, possesses unique advantages, including renewability, low cost, structural tunability, and environmental friendliness. Through targeted functional modification, it demonstrates excellent application performance in the catalytic degradation of pollutants. The following sections detail its degradation performance and mechanisms based on different modification types.
BBGC loaded with iron nanoparticles
-
Biomass-derived graphitic carbon typically forms under iron salt catalysis, naturally loading iron-based nanoparticles such as nano-zero-valent iron (nZVI) and Fe3O4. This makes it an excellent functional material for removing multiple types of pollutants[168,200,201], including metal and non-metal ions[202], dyes[203], and aromatic pollutants[204] are excellent adsorbents. Its core mechanisms include reduction conversion, surface complexation, precipitation fixation, and synergistic adsorption. For example, the addition of chitosan and nZVI-modified biochar to soil can significantly reduce Cr(VI) to Cr(III), which can be adsorbed onto the biochar or immobilized in the soil through strong surface complexation and precipitation reactions[205]. A series of innovative studies demonstrated the effectiveness of magnetic biochar in wastewater treatment. It was demonstrated that the synthesis of magnetic biochar is an effective means of removing Cr(VI) through adsorption with an uptake of up to 55 mg/g[206], in which Fe3O4 helped to immobilize the reduced Cr(III) as Cr(III)-Fe(III) hydroxide. Meanwhile, the removal ability of magnetic biochar prepared from waste coffee grounds for As(V) was mainly attributed to the effects of Fe3C and Fe3O4, not limited by the pore structure or functional groups of the biochar[207]. Furthermore, the presence of organic matter resulted in enhanced oxidation of As(III) and reduction of Cr(VI) in the biochar system, whereas in the absence of organic matter, the reduction of Cr(VI) occurred simultaneously with the oxidation of As(III)[208]. A biochar-based approach utilizing n-ZVI was used to investigate the immobilization of As(V) in soil. The results demonstrated that nZVI was capable of effectively reducing and immobilizing adsorbed As(V) to As(III)[209]. In a separate study, ZVI-based biochar demonstrated efficacy in the removal of Se(IV) and Se(VI) from solution, exhibiting adsorption quantities of 62.5 and 35.4 mg/g, respectively. This was attributed to the synergistic action of ZVI in composite materials and the contribution of a positive surface charge[210].
N-modified BBGC
-
N-doped graphitic carbon plays a key role in photocatalytic systems, and has been extensively demonstrated to be an efficacious component for enhancing photocatalytic performance[211]. Photocatalysts can efficiently adsorb oxygen, capture photo-generated electrons, and convert them into reactive species, thereby significantly influencing charge separation efficiency. In N-doped graphitic carbon-coupled α-Fe2O3 photocatalysts, the regions surrounding graphite nitrogen sites are more prone to hydrogen bond formation, leading to enhanced acidity that further promotes the degradation of pollutants such as acetaldehyde and phenol[212]. In addition, the photocatalytic degradation of pollutants can be further enhanced by the construction of composites, such as those co-modifying nanoparticles with N-doped graphitic carbon quantum dots (N-GQDs) on the surface of the material. Deng et al. constructed graphitic carbon composites co-decorating silver nanoparticles and N-GQDs, which enabled them to capture visible and near-infrared light and convert it into shorter wavelength light[213], a process that will further enhance the localized surface plasmon resonance (LSPR) effect of the silver nanoparticles, increase the light energy utilization across the entire spectrum, and effectively degrade tetracycline. Furthermore, N-GQDs can act as photoelectron acceptors, thus inhibiting the recombination of photoelectrically generated charges. The synergistic effect of the upconversion property of N-GQDs and the LSPR of silver nanoparticles significantly enhances the photocatalytic degradation activity of graphitic carbon[214]. Furthermore, the synergistic effect between different N atoms in NG is also worthy of mention. In a recent study, biochar@CoFe2O4/Ag3PO4 composites were successfully prepared and applied to the photocatalytic degradation of bisphenol A (BPA) under visible light irradiation. The results demonstrated high catalytic activity and stability[215]. A new class of materials was developed for the removal of methyl orange (MO) from aqueous solutions. These FeOOH/Fe3O4/graphitic carbon composites exhibited high magnetic and photocatalytic activities, with a photodegradation efficiency 2.03 times that of traditional biochar. Despite having a significantly smaller surface area than biochar, these composites were found to be effective in the photocatalytic degradation of MO. The presence of •OH radicals, which are responsible for this process, was confirmed through ESR and radical quenching analysis[216]. Thanks to the stable structure of graphitic carbon in the composite, the catalyst demonstrated remarkable stability and reusability. After undergoing five cycles, the photocatalytic activity remained at least 98% of its original level. Additionally, the catalyst could be readily separated from the solution by employing magnetic separation technology[202]. Furthermore, the graphitic carbon catalyst loaded with nZVI demonstrated high catalytic performance in the degradation of sulfamonomethylpyrimidine. This was attributed to its abundant pore and defect structure, as well as a high degree of graphitization. This enabled the catalyst to efficiently degrade sulfamonomethylpyrimidine via both the non-radical and radical pathways within 20 min. Additionally, the catalyst exhibited reusability[217].
The nZVI-loaded graphitic carbon exhibits a distinctive affinity for a diverse array of pollutants, including heavy metals and dyes, in the environment. In contrast, nitrogen-modified graphitic carbon can effectively capture photo-generated electrons, thereby enhancing photocatalytic performance while offering both low cost and environmental compatibility.
Other typical BBGC
-
In addition to the above, S doping, P doping, and structural regulation have also attracted the attention of researchers.
S-containing biochar is doped with elemental sulfur by adding sulfide during biochar preparation or carbonization in a sulfur-containing atmosphere[218]. S atoms can improve the electrochemical activity of BBGC. S doping can also enhance the activation of BBGC towards persulfate by adding more carbonyl groups and defects[219]. The introduction of P into the preparation of biochar improves the thermal and chemical stability of the material and increases its catalytic properties and biological activity[220]. P-doped biochar can improve the P fertilizer efficiency of soil in soil improvement, and also shows good potential for application in electrochemical energy storage and conversion systems[221].
In addition, BBGC exists in different forms, and for biomass feedstocks such as cellulose and lignin are able to produce one-dimensional biomass-based carbon materials, i.e., graphitic carbon fibers, through processes such as carbonization, stretching, and surface treatment. These fibers have high strength, high modulus, and good thermal stability, and can be used as a reinforcement for composites or as an electrode material for energy storage devices, exhibiting excellent electrochemical properties[222]. Carbon thin films as 2D biomass-based carbon materials are then prepared from biomass-derived carbon-containing precursors by methods such as chemical vapor deposition (CVD) or biomass carbonization. Such thin film materials have high transparency, excellent electrical conductivity, and mechanical strength and are suitable for applications such as flexible electronics, transparent conductive films, sensors, and energy storage devices[223].
The development of these BBGC utilizes the renewable and environmentally friendly nature of biomass resources, while endowing the materials with unique properties through elemental doping and structural modulation. With the in-depth research and optimization of these materials, they will offer greater flexibility in their applications across the fields of energy, environment, and materials science.
Other graphite materials
-
To highlight the unique advantages of BBGC, the following materials are selected for comparison: GO and its derivatives, graphitic carbon nitride (g-C3N4), and other new graphite materials.
GO and its derivatives
-
The 2D structure of GO, its abundance of surface oxygen-containing functional groups, and its elevated SSA collectively render this material an optimal choice as a carrier for the enhancement of the adsorption capacity of composite photocatalysts[224]. The chemical composition of GO is analogous to that of graphite oxide, which is comprised of aromatic and aliphatic carbon regions. The C/O ratios in GO are typically maintained within the range of 1.5 to 2.5[225,226]. However, the precise structural configuration of GO remains a matter of contention due to its non-stoichiometric composition and strong hygroscopic properties[227]. Currently, the primary techniques for preparing GO include the Hummers method, the Staudenmaier method, and the Brodie method. The traditional Hummers method employs a strong oxidizing system comprising concentrated sulfuric acid, sodium nitrate, and potassium permanganate to intercalate and oxidize natural flake graphite, but carries the risk of generating toxic nitrogen oxide byproducts[228]. The Staudenmaier method employs fuming nitric acid and potassium chlorate as oxidants, achieving higher oxidation levels but featuring longer reaction cycles and greater corrosivity. The Brodie method utilizes nitric acid and potassium permanganate as oxidants, allowing controllable oxidation levels, yet suffers from low reaction efficiency and significant environmental concerns[229]. In recent years, researchers have explored greener, more sustainable preparation pathways. Examples include utilizing ultrasonic-assisted oxidation to enhance reaction uniformity[230] or developing green synthesis strategies based on natural biomass precursors[231]. Different preparation processes directly influence GO's oxidation degree, defect density, and functional group distribution, thereby determining its subsequent reduction behavior and ultimate performance in composite materials. Among the various models proposed, the Lerf-Klinowski model, which was first introduced in 1998, has gained the most widespread acceptance. This model contains epoxy, hydroxyl, and carboxyl groups[232]. Regarding the structures of raw graphite, GO, and reduced graphene oxide (RGO). Research indicates that at a relative humidity of 30%, RGO exhibits conductivity approximately five orders of magnitude higher than GO, yet about ten times lower than raw graphite[233]. Furthermore, thermal reduction, electrochemical reduction, and chemical reduction are frequently used to transform GO into RGO. Thermal reduction techniques are typically more efficacious in restoring the electrical conductivity of GO than chemical reduction methods[234]. However, despite some controversy surrounding the structure and characteristics of GO, its potential for enhancing the efficiency of composite photocatalysts remains significant. In particular, conversion to RGO via a variety of reduction methods has the potential to greatly expand the scope for its application in the fields of environmental remediation and energy conversion, due to its significantly enhanced electrical conductivity.
Table 3 presents a series of GO and its derived materials, which can be utilized in a variety of photocatalytic applications. A combination of ZnO-RGO and ZnO-NanoRGO, which exhibit comparable structures, was created through the use of GO and NanoGO as precursors. The results of the photocatalytic activity tests on ZnO-RGO and ZnO-NanoRGO composites demonstrated that the photosensitization efficiency of NanoRGO was significantly higher than that of RGO[224]. In this system, BiVO4 was selected as the photocatalyst for O2 generation and Ru-SrTiO3:Rh was selected as the photocatalyst for H2 generation[235]. The electrons initially migrate from BiVO4 to RGO, and subsequently, the electrons accumulated on RGO combine with the holes on Ru-SrTiO3:Rh. Furthermore, control experiments and cycling tests have demonstrated that the presence of residual oxygen-containing functional groups is the primary factor influencing the photosensitizing properties of RGO. Theoretical calculations have demonstrated that the residual oxygen-containing functional groups on the surface of RGO exert a considerable influence on its band gap[236]. The increase in residual oxygen-containing functional groups results in a broadening of the band gap of RGO, accompanied by an upward shift of the conduction band (CB). This phenomenon improves the efficiency of electron reduction and enhances the photosensitization capacity of RGO. In comparison to GO, the H2 evolution rates of NiO-GO and Ni-GO hybrids were observed to be enhanced by approximately 4 and 7 times, respectively[224]. Another effective method for improving the performance of GO is doping heteroatoms into the material. For example, it has been demonstrated that the synthesis of phosphorus-doped GO (denoted as P-GO) results in a markedly enhanced hydrogen generation rate in comparison to undoped GO[237]. Similarly, N-doped GO (denoted as N-GO) exhibits a similar performance enhancement phenomenon[89]. Furthermore, GO can be regarded as a p-type semiconductor due to the significant presence of oxygen-containing functional groups. P-type GO and N-type titanium dioxide (TiO2), when assembled in a p-n heterostructure, facilitate the efficient separation of electron-hole pairs under light-induced conditions[238]. The results indicate that doping GO with different heteroatoms can enhance its catalytic performance while also improving the effective separation of charge carriers in photocatalytic processes.
Table 3. GO and its derivative materials for various photocatalytic applications
Composite photocatalyst Light source Photocatalytic applications Ref. Pure GO Visible light xenon lamp Degradation of phenol [239] 2D GO nanosheets Solar irradiation Degradation of methyl blue (MB) [240] TiO2/GO Visible light xenon lamp Degradation of Rhodamine B [241] SiO2-ZrO2@rGO Visible light xenon lamp Degradation of Bisphenol A (BPA) [242] ZnO-GO UV light xenon lamp Degradation of MB [243] ZnO-rGO UV light xenon lamp Degradation of MB [244] TiO2 nanotube array-RGO Visible light xenon lamp Degradation of MB [245] TiO2/GO/Ag Solar irradiation MO [246] Persulfate-rGO UV light xenon lamp Degradation of BPA [247] Ag/AgCl/GO Visible light xenon lamp Degradation of MO [248] BiVO4/TiO2/GO Visible light xenon lamp C.I. Reactive Blue 19 (RB-19) [249] TiO2-Pt/GO UV light xenon lamp & Natural sunlight Sunset yellow and Tartrazine [250] TiO2/Fe3O4/GO Visible light xenon lamp Degradation of MB [251] rGO-Au Visible light xenon lamp Degradation of BPA [252] Graphitic carbon nitride (g-C3N4)
-
In the course of investigating effective environmental purification techniques, g-C3N4, a novel material with a distinctive graphite-like structure and defective sites, has been extensively used for the remediation of deleterious pollutants in aqueous environments[253]. In subsequent studies, g-C3N4 was found to be capable of being composited with traditional biochar, and a variety of other materials. These composites demonstrated the ability to effectively reduce the concentration of heavy metals and organic dyes, while also exhibiting good photocatalytic properties and stability[254], which provided new solutions for wastewater treatment and environmental remediation. In complex aqueous environments where heavy metal ions are present, the composites can still function effectively. For example, g-C3N4@CA/B-PET is a material that not only reduces Cr(VI) to Cr(III) but also removes sodium sulfoquinolines. Furthermore, it exhibits high stability and reusability in applications[255]. In a separate study, the biochar/g-C3N4 composite was also used for the removal of Cr(VI) and Orange G from aqueous solutions. In this case, Orange G could be oxidatively mineralized to organic acids and CO2, while Cr(VI) was photoreduced to Cr(III) under UV light conditions. The photodegradation efficiency of formaldehyde by g-C3N4/biochar under visible light conditions was found to be significantly higher than that of g-C3N4. This enhanced performance can be attributed to two factors: firstly, an increase in the SSA of the biochar allows for enhanced separation of the photogenerated electron and hole pairs, and secondly, the biochar skeleton exhibits high absorption of visible light, which further enhances the photocatalytic activity[256]. Replacement of conventional biochar with magnetic graphitic carbon was able to further enhance the photodegradation performance of the composites against dyes under visible light conditions, which was attributed to the effective photogenerated electron-hole pair separation and inhibition of their recombination, as well as the involvement of reactive oxygen radicals[254,257]. In addition, biochar/g-C3N4 composites are also highly scalable, for example, g-C3N4/FeVO4/Fe@NH2-Biochar composites were used for the removal of methylparaben and 2-cholophenol, which has a high SSA, porous nano-sheet structure, high stability, and good electron-hole pair separation ability, and the presence of FeVO4 and Fe favors the absorption of more light and improves the photocatalytic activity[258]. In the field of environmental remediation and wastewater treatment technologies, g-C3N4 can be combined with biochar in various forms, and exhibits excellent adsorption and photocatalytic properties, high stability, and reusability. These properties enable it to demonstrate remarkable effects in the removal of heavy metals and organic pollutants. These materials are not only highly effective in the treatment of contaminants in complex aqueous environments but also demonstrate remarkable abilities in the remediation of environmental issues. Their optimized structure and material properties allow for effective separation of photogenerated electron-hole pairs, and enhanced photocatalytic activity, making them invaluable tools for the treatment of pollutants in complex aqueous environments.
Other new graphite materials
-
The novel graphitic carbon materials, including graphene quantum dots, and CNTs, have attracted considerable interest due to their distinctive physicochemical properties[259]. These materials have demonstrated excellent potential for application in a number of fields, including energy storage, biomedicine, and electronics. They have also shown great application prospects in environmental science. The potential of novel graphitic carbon materials is particularly attractive for water treatment, air purification, heavy metal removal, organic pollutant degradation, and environmental monitoring.
Graphene quantum dots (GQDs) and their N-doped derivatives (N-doped graphene quantum dots, or N-GQDs) have demonstrated considerable potential for application in the field of environmental sciences due to their distinctive physicochemical properties. These nanocarbon materials, which have a diameter less than 100 nm, possess a number of characteristics similar to those of graphene. These include a high SSA, excellent electrical conductivity, and mechanical strength. However, they also exhibit the quantum effect, edge effect, and the ability to modify the electronic band structure, which renders them highly valuable for a number of applications in the field of environmental monitoring and the removal of pollutants. In the field of environmental monitoring, GQDs and N-GQDs are used to construct highly sensitive and selective chemical and biological sensors. This is achieved by virtue of their stable fluorescence properties and high SSA[260]. These materials demonstrate the capacity for rapid detection and identification of a diverse range of organic pollutants, heavy metal ions, and biomarkers, with particular efficacy in the recognition and detection of certain analytes, including N-GQDs, which exhibit a heightened ability to bind with specific analytes and lower detection limits. This can be attributed to the unique interactions of their surface N functional groups[261]. Furthermore, the fluorescent properties of GQDs and N-GQDs can be used in environmental labeling and tracking, offering novel instruments for the identification of pollutant sources, and the investigation of environmental behavior. With regard to the treatment of pollutants, GQDs and N-GQDs display excellent adsorption performance and photocatalytic activity[262]. They are capable of effectively adsorbing heavy metal ions and organic pollutants in water, and achieve pollutant removal through physical adsorption and complex chemical interactions on the surface. In the field of photocatalysis, GQDs and N-GQDs exhibit the capacity to absorb visible light and generate highly active electron–hole pairs under light irradiation. These charge-separated states can act as catalysts for the decomposition of organic pollutants and facilitate the cleansing of water and air environments. In particular, N-GQDs, with their N doping-induced energy band structure tuning and alteration of surface electronic properties, have demonstrated enhanced efficiency and stability in the photocatalytic process[263].
CNTs have the potential to be a valuable tool in the field of environmental remediation due to their unique physicochemical properties. In particular, CNTs demonstrate superior performance in the adsorption and degradation of heavy metals and organic pollutants. CNTs are capable of effectively removing a wide range of contaminants from water and wastewater, including heavy metals[264], radionuclides[265], and a variety of organic pollutants[266]. This is achieved through adsorption mechanisms, such as pharmaceuticals, dyes, pesticides, and polycyclic aromatic hydrocarbons (PAHs), among others. The adsorption of heavy metals by CNTs is primarily contingent on the surface functionalization modification. For example, oxygen-containing functional groups, such as hydroxyl and carboxyl groups, can be introduced on the surface of CNTs through oxidative modification. These functional groups can enhance the affinity of CNTs for heavy metal ions[267]. Furthermore, the adsorption capacity of CNTs for heavy metals can be enhanced by incorporating metal and metal oxide nanoparticles, polymers, and other materials into CNTs[268]. For example, magnetic iron oxide-decorated CNTs were used to extract heavy metal ions from aqueous solutions, exhibiting remarkable adsorption efficacy[269]. The applications of BBGC and its derivative materials in catalytic degradation of pollutants are summarized in Fig. 6. Graphitic carbon has a good effect on the adsorption and removal of heavy metals, and GO and RGO are the adjustments of functional groups on the surface of graphitic carbon, which make the carbon material more suitable as a carrier for photocatalysts. g-C3N4 is a graphite-like structural material formed by substituting some C atoms with nitrogen. It, along with graphene quantum dots, CNTs, and other novel materials, finds a wide range of applications in the catalytic degradation methods of antibiotics and dyes.
-
LCA enables evaluation and optimization of the entire process from raw material acquisition to end-of-life disposal. It identifies environmental bottlenecks at each stage, thereby establishing optimal application models that balance efficiency and environmental protection. This section will use LCA as the core framework to integrate key stages, elucidate the utility of BBGC across diverse application scenarios, and clarify its ecological footprint throughout the entire lifecycle from production to disposal. It will demonstrate the process sustainability and environmental friendliness, thereby providing support for the large-scale green application of BBGC.
Raw materials form the foundation of BBGC's lifecycle sustainability and directly determine energy consumption and environmental impact in subsequent processes. Biomass has been used as feedstock when preparing graphitic carbon because it is renewable and carbon-rich[270]. The factors that affect the susceptibility of biomass sources to variation and, therefore, the sustainability of the production process include the availability, renewability, and carbon neutrality of biomass sources. Non-food biomass such as agricultural byproducts (straw, fruit shells), forestry residues, and municipal organic waste should be prioritized. These feedstocks are widely distributed, abundantly available, and do not require additional arable land, do not contribute to deforestation[271], and significantly reduce methane emissions, and leachate pollution from landfills[257], demonstrating excellent sustainability, and environmental friendliness. Apart from being a major source of carbon in biotechnological processes, it is equally important for waste utilization projects in managing potential landfill waste. Concerning its relevance to the circular economy, which emphasizes the disposal of waste and the proper utilization of resources, the present strategic approach aligns with the previously mentioned framework. It emphasizes a focus on biomass that allows for cultivation with high yields, causes little disturbance and, therefore, has a negative impact on food security and land use diversification, and is supported by science based on the efficiency of solid biofuels[272], bioenergy gained from agricultural residuals and livestock manure[273], and cooking fuels[274]. Moreover, the features of the biomass feedstock, such as carbon content, moisture content, and heteroatom composition, significantly influence the conversion and characteristics of graphitic carbon[275]. Biomass with high carbon content and rich in heteroatoms such as nitrogen, sulfur, and phosphorus can be used to prepare high-performance graphitic carbon, endowing it with high electrical conductivity and large SSA[276]. In summary, the inherent properties of the raw materials result in lower environmental impact and energy consumption, thereby enhancing the sustainability and eco-friendliness of the entire process from the outset.
Pretreatment is a critical step in ensuring stable graphitization and activation outcomes. Effective pretreatment not only reduces energy consumption in subsequent processes but also guarantees product performance stability. Its scientific design and optimization decisively influence the energy efficiency, product performance consistency, and environmental sustainability of the entire production flow. Among these, drying and milling serve as core physical pretreatment steps. They directly impact material handling, storage, and reaction kinetics, while also determining the macro-level energy balance, and economic viability of the entire process chain. The drying process aims to reduce the initial moisture content of raw materials, thereby minimizing the substantial latent heat consumption required for moisture evaporation during subsequent thermal processing. Significant differences exist in energy consumption among various drying technologies. Heat pump drying typically achieves over 30% greater energy efficiency than conventional hot-air drying due to its closed-loop system that recovers latent heat[277]. Additionally, the sequence of drying and grinding significantly impacts overall energy consumption. For pine wood chips, drying before grinding reduces total energy consumption by up to 20% compared to grinding before drying. This is because wet material exhibits high stickiness and poor flow during grinding, increasing mechanical resistance, and power consumption[278]. Therefore, optimizing drying parameters and rationally planning the sequence between drying and grinding processes are primary strategies for achieving energy savings and reduced consumption. To ensure raw material particle size meets requirements for subsequent conversion processes, grinding is typically indispensable. This mechanical processing ensures uniform raw material properties, thereby determining the stability of pyrolysis and graphitization processes. Hong et al.[279] suggested that grinding in some way has an impact on the end product, where a coarse ground particle sample provides high carbon conversion, whereas fine particles provide high energy conversion rates for gases. Therefore, optimizing the milling technology and operating conditions of a mill may help increase the energy efficiency of grinding operations. For instance, research has found that equipping each mill classifier with a uniform particle size meeting Stokes' requirements can minimize mill blockage[280]. Furthermore, pretreatments aimed at softening the structure of biomass and reducing its brittleness, such as torrefaction[82] or self-heat recuperation[281], can further reduce the energy used to grind the biomass and, hence, minimize the environmental impact of this step. Microwave pretreatment selectively heats internal moisture within the material, effectively disrupting cell wall structures and reducing subsequent grinding energy consumption by 15%–30%[282]. Hydrothermal pretreatment, utilizing high-temperature and high-pressure water, can also effectively degrade hemicellulose and improve the grindability of the material[283]. Furthermore, energy consumption and emissions associated with biomass transportation are often overlooked. Promoting the local sourcing of biomass resources can minimize fuel consumption and pollution emissions during transportation, enabling low-energy synergies between pretreatment and subsequent processes, and enhancing the overall sustainability of the process.
Traditional graphitization processes often involve pyrolysis, followed by high-temperature treatment to rearrange carbon atoms into graphitic structures[160]. This results in graphitization being one of the most energy-intensive stages in its life cycle, as raising temperatures to extremely high levels consume significant amounts of energy. In this regard, energy-efficient production techniques, such as catalytic graphitization and HTC, can help minimize the carbon footprint associated with graphite production. Catalyzed graphitization reduces reaction temperatures through catalyst addition[100], significantly lowering energy consumption. However, attention must be paid to the environmental impacts of catalyst preparation, usage, and disposal. Priority should be given to selecting abundant, non-toxic catalysts, and developing recycling technologies to prevent secondary pollution during catalyst preparation, application, and disposal. HTC treats biomass in water at elevated temperatures and pressures, facilitating the breakdown of biomass components into a carbon-based solid known as hydrochar[284]. The following stages have various benefits to the environment, such as a low energy demand, as a result of the relatively lower temperatures involved, and the utilization of wet biomass without requiring extensive drying. In addition, there is an opportunity to recycle or recover valuable chemicals from the aqueous phase created during HTC, thereby rendering the process sustainable in accordance with herbaceous biomass[285], sewage sludge[286], and algae[287]. Coupling HTC with other processes, such as anaerobic digestion, can further enhance energy efficiency[288]. However, hydrochars obtained through HTC require further development through activation and graphitization procedures to obtain the properties required for energy storage applications. In summary, during the graphitization process, the goal of process sustainability can be achieved by selecting low-energy, low-pollution, high-performance coupled graphitization techniques.
Physical[289] and chemical[290] activation play a key role in enhancing the porosity and surface area of carbon-based products. Chemical activation involves impregnating a carbon precursor with activating agents such as KOH, followed by thermal treatment, as demonstrated by Xing et al.[291], who used ultrasonic-assisted immersion pyrolysis. Mbarki et al.[292] obtained inexpensive activated carbon from corn stigmata fibers produced using KOH, H3PO4, and ZnCl2. This formed a highly porous structure and introduced functional groups that could improve the electrochemical performance of a material. The activation conditions[212] (temperature, time, and atmosphere), and the activating agent selected are crucial in regulating the surface chemistry and distribution of pore sizes in activated carbon. Chemical activation is a viable method for producing high-performance carbon materials. However, it also comes with environmental concerns regarding the use and disposal of chemical agents. While physical activation using CO2 or water vapor as a medium is relatively environmentally friendly, its low activation efficiency makes it difficult to construct an ideal hierarchical pore structure, limiting its application in high-performance electrode materials[293]. In contrast, the one-step process offers distinct advantages. Dong et al.[294] reported a one-step process combining ball milling with MgCO3, utilizing the high-energy CO2 jet effect generated during MgCO3 decomposition to in situ form a loose, defect-rich structure within low-rank coal matrices. This structure further evolved into hierarchical porous carbon with outstanding electrochemical performance. The resulting material exhibits not only excellent pore connectivity but also an IG/ID ratio > 1.0, confirming high graphitization[295]. The one-step process eliminates intermediate steps, reducing energy consumption and equipment requirements while demonstrating significant environmental advantages. Notably, thermal management strategies are also critical. Recovering heat released during high-temperature stages for reuse in subsequent stages substantially enhances overall energy efficiency[296]. Furthermore, coupling physical activation with chemical activation preserves the efficient pore formation capability of chemical activation while leveraging mild gas activation to modulate surface chemistry, thereby balancing performance and sustainability.
The sustainability of subsequent processing stages directly determines the environmental benefits of BBGC throughout its entire lifecycle. To minimize their environmental impact, graphitic carbon materials derived from biomass must be recycled and biodegradable upon expiration. By recovering and reusing carbon materials from spent energy storage devices, recycling can remarkably decrease the demand for virgin materials and reduce the life cycle emissions of carbon-based energy saving technologies[297]. Recycling processes that recover high-purity carbon materials are necessary for the continuous use of these resources. Modern separation and purification methods, including solvent-based extraction optimize the recovery process while maintaining the integrity of materials for reuse[298]. Conversely, biodegradability provides a sustainable means of disposing carbon materials that cannot be recycled. The design of carbon-based materials with regulated biodegradability, factor analysis, and consideration of the material composition and microbes are essential for end-of-life management. From the perspective of nano-biomedicine, Mokhtari-Farsani et al.[299] highlighted the need to maintain the biodegradability of carbon-based nanomaterials in the context of biomolecular coronas. It is challenging to design materials that possess the desired electrical and mechanical characteristics for their intended purposes without rendering them indestructible in the environment.
In summary, identifying specific issues in BBGC's LCA throughout raw material selection, preparation, graphitization, and processing enables targeted optimization of these processes. By carefully planning and managing these stages, BBGC can maintain high performance while balancing technical feasibility and environmental sustainability, making them valuable components of sustainable energy storage solutions.
-
With the increasing focus on environmental, social, and economic challenges, many countries aim to achieve the UN SDGs through sustainable practices. In this regard, numerous industries have adopted eco-friendly initiatives. One notable approach involves the use of biomass-derived carbon materials. In 2018, the IPCC officially acknowledged biochar as a negative carbon emission technology, highlighting its capability to remove carbon dioxide from the atmosphere[300]. Biochar is a comprehensive term that connects various environmental management applications. This term extends beyond its role in agriculture and soil improvement, branching into diverse areas in which its properties can be coupled for environmental and ecological benefits[301].
Moreover, the efficient utilization of biomass for green graphite production is a highly effective strategy for satisfying the demand for graphite while minimizing both energy consumption and carbon emissions[302]. Graphite and its derivatives, designated as 'strategic and critical minerals' by the US government and the European Union, play a vital role in various industries such as battery manufacturing and metallurgy[303]. Although bio-based graphite is predominantly used in battery anodes, its potential applications in metallurgy and refractory materials remain largely unknown[112,304]. This transition was further underscored during COP28 in the UAE, where there was a clear call to move away from the use of fossil fuels. This shift has amplified the demand for LIBs, emphasizing the importance of sustainable solutions, such as biomass-derived graphite, for industries such as battery manufacturing and metallurgy. LIBs play a pivotal role in clean energy storage. Recent studies indicated that biomass-derived carbon materials are promising negative electrode materials for LIBs because of their abundant availability, cost-effectiveness, and stable performance in electrochemical applications[270].
Regarding BBGC, several challenges remain, such as low energy density, high costs, considerable self-discharge rates, and inadequate rate performance. Addressing these issues requires the development of cost-effective, high-performance electrode materials. Currently, carbon materials are the most advanced choices for supercapacitor electrodes. BBGC holds promise as a viable material for energy storage applications. Furthermore, extensive research has explored the viability of graphitic carbon as an adsorbent for contaminant removal in pollution control[305]. Carbon nanomaterials such as CNTs, graphene, and nanodiamonds are increasingly recognized for their potential as effective non-metallic catalysts for environmental pollution mitigation. These materials show promise as advanced methods for treating water pollution, particularly through the oxidative degradation of organic pollutants activated by persulfate[306]. Despite the diverse array of benefits associated with biomass-based carbon materials, their widespread large-scale use has not yet been fully realized. Various factors, such as economic considerations, technological challenges, and awareness gaps, may contribute to the limited adoption of these biomass-derived carbon materials[307,308].
With regard to graphite mineral mining, numerous environmental issues have arisen, including habitat destruction due to deforestation and disruption of local ecosystems, water and air pollution through the release of heavy metals and chemicals used in processing and high energy consumption[309]. Additionally, social concerns such as the displacement of communities, labor rights violations, and conflicts over land use often accompany mining operations. For example, Amnesty International revealed that tens of thousands of children in the Democratic Republic of Congo engage in artisanal mining, where they work long hours under hazardous conditions, without essential safety equipment. These children often endure prolonged exposure to toxic dust, which increases their risk of respiratory illnesses and organ damage. Hence, the transition to BBGC offers a sustainable alternative that mitigates the environmental and social challenges associated with traditional graphite mining.
Government support through various financial incentives, such as tax credits, subsidies, and loan guarantees, can play a pivotal role in advancing the growth of the biomass-derived carbon materials industry. These incentives can reduce the economic barriers associated with graphitic biochar production and applications. Commercial financial incentives aim to provide financial assistance to producers or production facilities operating at a commercial scale[310]. The initial stage of the production of biomass-based carbon materials can be positively influenced by programs that specifically target biomass or bio-waste management. Implementing such initiatives focused on the responsible and sustainable management of biomass and bio-waste can not only address environmental concerns but also provide valuable feedstock for the production of biomass-based carbon material. By positioning the production of biomass-based carbon material as a waste management strategy, governments can align environmental goals with economic incentives and encourage the conversion of organic waste into a resource with multiple benefits, including carbon sequestration and energy generation. In addition, research and development funding is directed to grants supporting applied research and pilot/demonstration projects related to biomass-based carbon materials, production technology, or various stages within the value chain[310].
Biomass-based carbon materials have emerged as environmentally sustainable products within the ESG framework, making noteworthy contributions to the UN SDGs (Fig. 7).
-
In summary, the use of BBGC and its applications in the fields of energy and the environment were reviewed. Particular emphasis has been placed on technologies that convert biomass to graphitic carbon materials, as well as the potential applications of these materials. Biomass with a high content of cellulose and hemicellulose exhibits favorable performance under low-temperature graphitization conditions, whereas lignin-rich biomass requires temperatures exceeding 2,500 °C. The graphitization of biomass is evidently influenced not only by its chemical composition but also by the pretreatment techniques and graphitization conditions applied to the material. Catalytic methods are most commonly used for low-temperature graphitization. Combining different catalysts with various physical and chemical treatment methods can enhance the order and electrical conductivity of graphitic carbon materials, thereby regulating the properties and functions of BBGC. Catalysts can achieve efficient graphitization at lower temperatures, reducing production costs and enhancing the performance of carbon materials. Moreover, BBGC materials have a wide range of applications, including supercapacitors and SIBs. It is noteworthy that the pore structure and degree of graphitization of graphitic carbon must be properly designed prior to these applications. In environmental remediation, BBGC and its derivatives can be used for the adsorption and degradation of different pollutants. Regardless of the application field, the transformation of biomass reduces the dependence on fossil fuels, contributes to the reduction of greenhouse gas emissions and the carbon footprint, and demonstrates remarkable advantages in reducing environmental pollution and promoting sustainable development.
On the basis of existing research, future work may investigate more economical and environmentally friendly graphite production technologies, particularly under low-energy consumption conditions in the future. They may also explore ways to retain and utilize non-carbon elements in biomass during the graphite production process. Such investigations may lead to the development of new techniques for the functionalization of graphitic carbon materials. Additionally, BBGC materials exhibit excellent scalability. The surface and structural functionalization of graphitic carbon materials, such as the improvement of their electrochemical performance or catalytic activity through doping, compounding, or surface modification, could be widely applied in emerging fields such as biomedicine and sensor technology. In addition, the introduction of artificial intelligence to create predictive models that can predict the outcome of different graphitization processes, based on input parameters (e.g., temperature, heating rate, and type of biomass), would be a significant boost to the biomass graphitization process. Although the production and application of BBGC materials offer significant environmental and social benefits, their economic viability remains a key factor in promoting their widespread application. Future research must investigate methods for reducing production costs and enhancing market competitiveness through technological innovation and policy support.
Baojun Yi acknowledges the support from the China Scholarship Council (202206765005) and Korea University for hosting his visiting research. This research was supported by OJEong Resilience Institute, Korea University.
-
The authors confirm their contributions to the paper as follows: all authors contributed to the study conception and design; material preparation, data collection, and analysis were performed by Zhengshuai Sun; the first draft of the manuscript was written by Baojun Yi, Zhengshuai Sun, and Jiaqi Deng; all authors commented on previous versions of the manuscript; Baojun Yi, Juin Yau Lim, Sachini Senadheera, Xiangzhou Yuan, Jin Shang, and Yong Sik Ok were involved in the writing − review and editing; project administration and resources were performed by Baojun Yi and Yong Sik Ok; Yong Sik Ok also contributed to the supervision. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
-
Key Research and Development Program of Hubei Province (2023BBB001), National Key Research and Development Program of China (2021YFD1600503), Support Enterprise Technology Innovation and Development Projects of Hubei Province (2021BAB115), the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (RS-2025–00555967) (PI: Yong Sik Ok). This research was supported by the OJEong Resilience Institute of Korea University (PI: Yong Sik Ok).
-
All authors declare that there are no competing interests.
-
Full list of author information is available at the end of the article.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Yi B, Sun Z, Deng J, Lim JY, Senadheera S, et al. 2026. Sustainably graphitizing biomass into advanced carbon materials for energy and environmental applications. Sustainable Carbon Materials 2: e014 doi: 10.48130/scm-0026-0003
Sustainably graphitizing biomass into advanced carbon materials for energy and environmental applications
- Received: 03 December 2025
- Revised: 31 December 2025
- Accepted: 16 January 2026
- Published online: 12 March 2026
Abstract: The demand for graphitic carbon materials is growing steadily, yet traditional production methods heavily rely on fossil fuels or mineral graphite, failing to achieve sustainable development goals. Biomass, as a renewable, low-cost, and environmentally friendly carbon precursor, represents a highly promising alternative for graphitic carbon production. This review explores the graphitization potential of biomass components: cellulose enhances product orderliness, hemicellulose optimizes the microstructure of graphitic carbon, and lignin facilitates the formation of high-quality graphitic structures. Starch-derived carbon mainly forms microporous carbon, and a high branched-chain starch content is required for electrochemical applications. Additionally, the various metallic and non-metallic elements present in biomass exert differing effects on the graphitization process. This review also summarizes methods for producing biomass-based graphitic carbon (BBGC). Catalyzed graphitization can overcome the high-energy-consumption bottleneck of traditional processes, while high-temperature graphitization can leverage the aromatic structure of lignin to produce highly crystalline products. Emerging techniques such as chemical foaming, hydrothermal carbonization, and ultrasonic exfoliation enable the preparation of graphitic carbon tailored for specific applications. This review also explores the application of graphitic carbon in energy storage and environmental remediation, where its sustainability and cost-effectiveness surpass those of traditional materials. Furthermore, this review identifies an environmentally optimized pathway through life cycle assessment (LCA). From an environmental, social, and governance (ESG) perspective, BBGC, as carbon negative technology, can reduce greenhouse gas emissions while avoiding the ecological damage and social issues associated with mineral graphite mining, aligning with the United Nations Sustainable Development Goals (SDGs).
-
Key words:
- Biomass /
- Graphitic carbon /
- Energy storage /
- Sustainability /
- Environmental impacts