-
Acute lung injury (ALI) is a clinical syndrome caused by various insults, including infection or trauma[1,2]. ALI has a characteristic pathophysiology with diffuse lung injury involving both alveolar epithelium and pulmonary capillary endothelium[3,4]. ALI can progress to acute respiratory distress syndrome (ARDS), a severe illness with a high case-fatality rate[5,6]. At present, treatment measures of ALI directed to its core pathogenesis are still limited. Therefore, the patient's prognosis is still difficult to control clinically.
Pathogenesis of ALI is associated with interactions among several signaling pathways[4,7]. The mitogen-activated protein kinase (MAPK) pathway, especially ERK1/2, is an important pathway for regulation[8,9]. Pathogenic stimuli, such as lipopolysaccharide (LPS), activate the MAPK/ERK1/2 signaling and induce nuclear translocation and transcriptional activation of nuclear factor-κB (NF-κB)[10−12]. Activated NF-κB subsequently induces the transcription of pro-inflammatory cytokines (e.g., IL-6 and TNF-α)[13−15]. NF-κB not only regulates inflammation responses but also provides a transcriptional foundation for NLRP3 inflammasome induction[8,16,17]. Meanwhile, MAPK/ERK1/2 signaling can directly promote NLRP3 inflammasome formation and maturation, and thus also amplify the inflammatory cascade[18]. Therefore, in ALI, the MAPK/ERK1/2 signaling pathway forms a positive feedback loop, which can enhance the activation of NF-κB and the expression of NLRP3, thereby working together to trigger pulmonary inflammation and damage[16].
Excessive inflammation not only directly causes tissue damage but also induces ferroptosis, which causes a self-amplifying vicious cycle and aggravates ALI even further[19,20]. As ALI evolves, impairment of host defence results in iron deregulation and overload, ferroptosis drives a vicious cycle of oxidative stress, inflammation, and impairment of mitochondrial function[21]. Ferroptosis is associated with multiple molecular disturbances, encompassing intracellular iron overload via hyperactivation of the transferrin receptor solute carrier family 11 member 2 (Slc11a2)-mediated uptake, as well as the downregulation of Slc7a11, and impaired glutathione synthesis due to ferritin deficiency, which collectively weakens cellular antioxidant defense[22,23]. Glutathione peroxidase 4 (GPX4) is the dominant regulatory enzyme that has a pivotal role in the regulation of ferroptosis by reducing lipid peroxides[24]. Therefore, targeting ferroptosis represents a potential therapeutic strategy for ALI.
Xuebijing injection (XBJ) is a formulation of traditional Chinese medicine composed of Carthami Flos, Paeoniae Radix Rubra, Chuanxiong Rhizoma, Salviae Miltiorrhizae Radix Et Rhizoma, and Angelicae Sinensis Radix[25]. Clinical studies have shown that XBJ, when combined with conventional therapy, improved symptoms and reduce inflammatory cytokine levels in patients with severe pneumonia[26,27]. Preclinical research also showed anti-inflammatory, immunomodulatory, and microcirculation-enhancing effects of XBJ[27−30]. Moreover, XBJ has been demonstrated to reverse pulmonary edema and inflammatory cell infiltration in a murine model of ALI[31]. Although its protective effect was well demonstrated, the precise molecular mechanisms of XBJ in ALI is not completely elucidated. This study aims to illustrate the molecular mechanisms of XBJ in ALI, and systematically identify its direct cellular targets in an LPS-induced model. These findings in this study also presented important experimental data for the pharmacological mechanism and clinical effects of XBJ in ALI.
-
XBJ (Lot No. 2501212) was kindly provided by the Tianjin Hongri Pharmaceutical Co., Ltd. (Tianjin, China). Primary antibodies against claudin-1, occludin, Foxp3, RORγt, p38, p-JNK, JNK, p-ERK, p-p65, p65, pro IL-1β, caspase-1, GSDMD, ASC, NLRP3, Slc7a11, DMT1, TFRC, FPN1, GPX4, and GAPDH were obtained from Proteintech (Wuhan, China). Additional antibodies against p-p38, ERK, and ASC were purchased from ABMART (Shanghai, China).
Cell culture
-
MH-S cells were grown at 37 °C in a 5% CO2 humidified incubator. Before a 24 h challenge with LPS (1 μg/mL), cells were pretreated with XBJ at graded concentrations (0.4, 0.8, and 1.6 mg/mL) for 1 h to evaluate its protective effects. Following this, cells were collected for subsequent analyses.
Western blotting
-
Cellular proteins were harvested with a lysis buffer. Following separation via 10% SDS-PAGE, the proteins were transferred onto PVDF membranes. Post-blocking with 5% milk, membranes were first incubated with primary antibodies and then with secondary antibodies. Following the ECL visualization, band intensity was quantified with ImageJ.
Quantitative real-time PCR (qPCR)
-
Total RNA, obtained via a Trizol-based method, was reverse transcripted for cDNA preparation. qPCR amplification was performed using SYBR Green qPCR.
Immunofluorescence
-
After PBS washing and methanol fixation, MH-S cells were blocked with 5% BSA, followed by overnight incubation with primary antibodies. After counterstaining with fluorescent secondary antibodies and DAPI, imaging and quantitative analysis were performed using a Leica DM6B Thunder Microscope (Leica, Germany).
Flow cytometry
-
Lung neutrophils and macrophages were analyzed through fluorescence-labelled CD11B and LY6G antibodies using a CytoFlex flow cytometer (Beckman Coulter, USA). MH-S cells were fixed with methanol and blocked with 5% BSA. Flow cytometric analysis was performed on a CytoFlex instrument following incubation with corresponding primary and secondary antibodies.
Animals and treatment
-
C57BL/6J mice (22–25 g) were sourced from the Experimental Animal Center of Tianjin University of Traditional Chinese Medicine. Mice were housed under SPF conditions and provided a standard diet and water. Mice were randomized into experimental groups (n = 10), including the control, XBJ (10 mL/kg), LPS (5 mg/kg), and LPS+XBJ (2.5, 5, or 10 mL/kg) groups. Mice in the XBJ and LPS + XBJ groups received twice-daily tail vein injections of XBJ for three successive days, and then were administered a single intratracheal dose of LPS. Lung tissues were obtained after euthanasia for further analysis.
Measurement of IL-6, TNF-α, MPO, and LDH
-
IL-6, TNF-α, myeloperoxidase (MPO), and lactate dehydrogenase (LDH) levels in lung tissues were quantified according to the corresponding kit protocols.
Lung pathological assessments
-
Paraffin-embedded lung tissues were sectioned sequentially and processed for hematoxylin and eosin (H&E) staining and immunohistochemical (IHC) examination.
Cellular thermal shift assay (CETSA)
-
MH-S cell lysates were incubated with XBJ or DMSO for 1 h. The samples were then heated at temperatures ranging from 42 to 58 °C for 6 min, followed by centrifugation. The resulting supernatants were collected for further analysis.
Drug affinity responsive target stability (DARTS)
-
Protein extracts from MH-S cells were incubated with XBJ or DMSO after lysis. Proteolytic digestion was carried out using pronase (2 μg/mL, 37 °C, 3 min). The samples were then centrifuged, and the supernatants were harvested for subsequent assays.
Target protein fishing assay
-
Following the previously established method[32], XBJ was conjugated to epoxy-activated 4FF beads. After coupling and blocking, the beads were incubated with protein samples. The captured proteins were eluted and analyzed by Western blot.
Statistical analysis
-
Data are presented as mean ± standard error of the mean (SEM). Statistical comparisons were performed using one-way or two-way ANOVA, through GraphPad Prism software. A p-value < 0.05 was considered statistically significant.
-
To elucidate the therapeutic potential of XBJ in ALI, an LPS-induced mouse model by intratracheal instillation was established (Fig. 1a). XBJ treatment significantly ameliorated ALI, as evidenced by decreased levels of key pro-inflammatory cytokines (IL-6 and TNF-α) and decreased activities of MPO and LDH in lung tissue (Fig. 1b). Histopathological examination by H&E staining showed that XBJ alleviated the pulmonary pathological damage, with reduced wall thickness and inflammatory cell infiltration (Fig. 1c). Given the pivotal role of inflammatory cell infiltration in ALI progression[33], this process was further analyzed by immunohistochemical and flow cytometric analyses (Fig. 1d, f). These results demonstrated that XBJ significantly suppressed the infiltration of CD68+ macrophages and CD11B+LY6G+ neutrophils in lung tissue after LPS challenge. Collectively, these observations suggested that XBJ attenuated the LPS-induced ALI, as well as improved the related inflammatory and pathological factors.
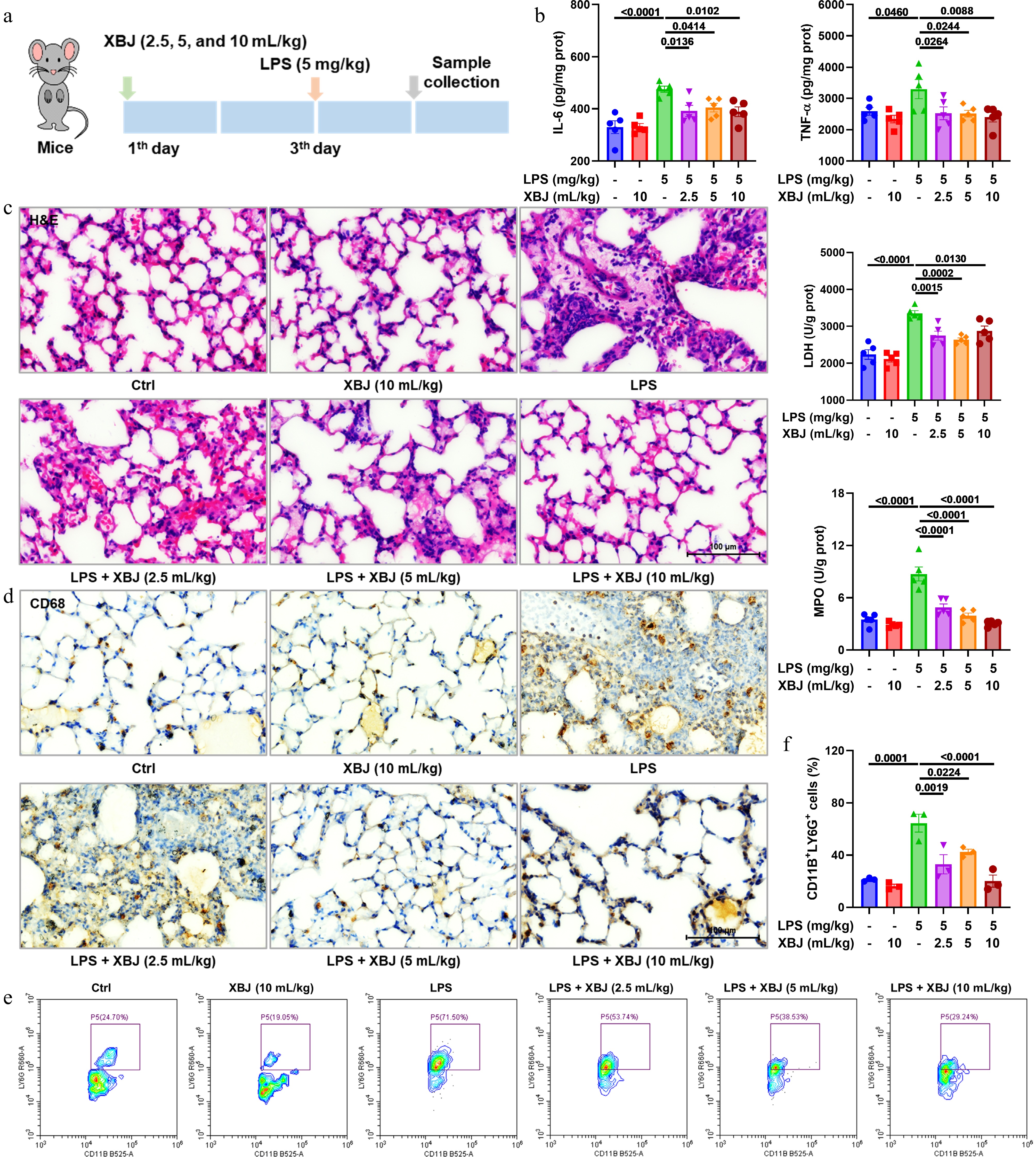
Figure 1.
XBJ suppressed LPS-induced pulmonary inflammation and tissue injury. (a) The experimental protocol; (b) effects of XBJ toward IL-6, TNF-α, LDH, and MPO in ALI (n = 5); (c) H&E staining plots; (d) representative CD68 staining plots; (e), (f) flow cytometric profiling of CD11B+LY6G+ cells (n = 3).
XBJ ameliorated LPS-induced ALI by restoring barrier integrity, modulating Treg/Th17 balance, and inhibiting MAPK/NF-κB pathways
-
Integrity of the pulmonary epithelial barrier maintains lung microenvironmental homeostasis and prevents escalation of inflammation cascades[34]. Therefore, we measured tight junction protein expression in lung tissues of LPS-induced ALI. Immunohistochemistry and western blot analyses showed that XBJ effectively reversed the decrease in LPS-mediated expressions of occludin and claudin-1 (Fig. 2a, b & d). RORγt and Foxp3, transcription factors specific to Th17 and Treg cells influence cell differentiation and function by regulating IL-17a and IL-10 secretion[35,36]. Western blotting showed that XBJ modulated these two cell types (Fig. 2c). Similar to this observation, XBJ treatment also significantly altered the mRNA levels of key pro-inflammatory cytokines (e.g., IL-17a, IL-6, TNF-α, and IL-1α) (Fig. 2e). To understand the underlying mechanisms, relevant critical signaling pathways were analyzed. These results indicated that XBJ treatment markedly affected the expression of proteins in the MAPK and NF-κB pathways in lung tissues (Fig. 2f & g). Finally, immunofluorescence staining indicated that XBJ modulated the expression and cell localization of Foxp3, RORγt, COX-2, and IL-6. In conclusion, these findings suggested that XBJ attenuated pulmonary inflammation and tissue injury by improving the integrity of the epithelial barrier, regulating the balance of Treg/Th17 cells, and inhibiting the MAPK and NF-κB pathways.
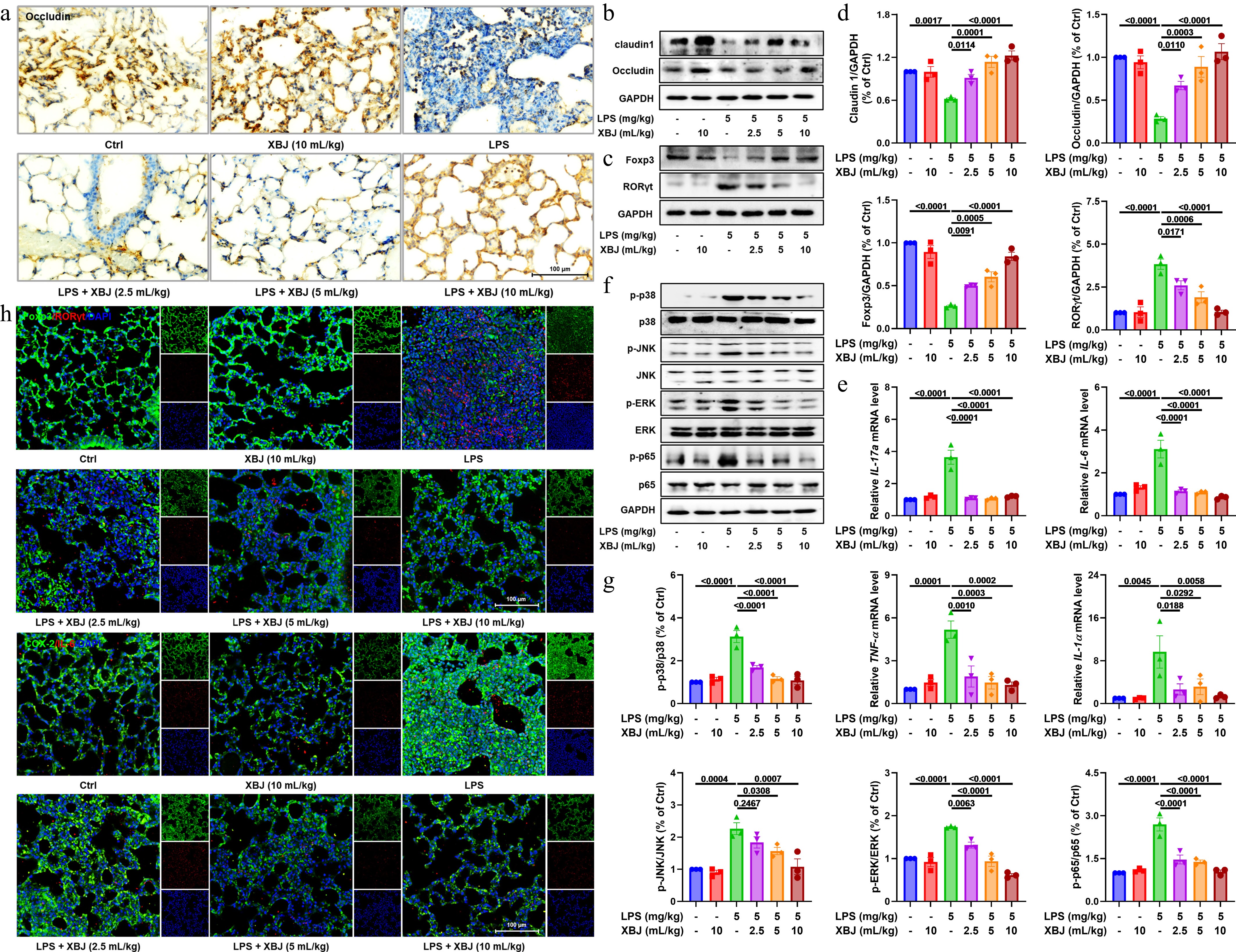
Figure 2.
Protective effects of XBJ against ALI via barrier repair, immune regulation, and MAPK and NF-κB pathway inhibition. (a) Representative occludin staining plots; (b)–(d) expressions of claudin1, occludin, Foxp3, and RORγt proteins (n = 3); (e) mRNA expressions of IL-17a, IL-6, TNF-α, and IL-1α (n = 3); (f), (g) expressions of key proteins in the MAPK and NF-κB pathways (n = 3); (h) immunofluorescence staining of Foxp3, RORγt, COX-2, and IL-6.
XBJ ameliorated LPS-induced ALI by inhibiting NLRP3 inflammasome activation and ferroptosis
-
Emerging evidence shows that ferroptosis and NLRP3 inflammasome activation play an important role in the progression of ALI[37]. To understand the regulatory effect of XBJ on ferroptosis and NLRP3 inflammasome activation, we detected the level of related molecules at the mRNA and protein levels. In Fig. 3a, XBJ treatment modulated mRNA expressions of key ferroptosis-related markers (e.g., GPX4, transferrin receptor [TFRC], acyl-CoA synthetase long chain family member 4 [ACSL4], and Slc7a11), and NLRP3 inflammasome components (e.g., NLRP3 and IL-1β). Further, both mRNA and protein levels of NLRP3 pathway components were substantially modulated by XBJ treatment, indicating that it indeed exerts an inhibitory effect on this inflammatory pathway (Fig. 3b & c). Western blot further verified that XBJ markedly affected protein expression of ferroptosis regulator proteins, such as Slc7a11, divalent metal transporter 1 (DMT1), TFRC, ferroportin1 (FPN1), and GPX4. This indicates that XBJ played a role in regulating cellular iron metabolism and antioxidant defense (Fig. 3d, e; Supplementary Fig. S1). Finally, immunofluorescence staining confirmed that XBJ suppressed the key markers associated with ferroptosis (TFRC) and inflammasome activation (ASC and NLRP3), and upregulated the anti-ferroptotic protein GPX4 (Fig. 3f & g). Collectively, these findings indicated that XBJ attenuated ALI by simultaneously suppressing the NLRP3 inflammasome pathway and the ferroptosis pathway.
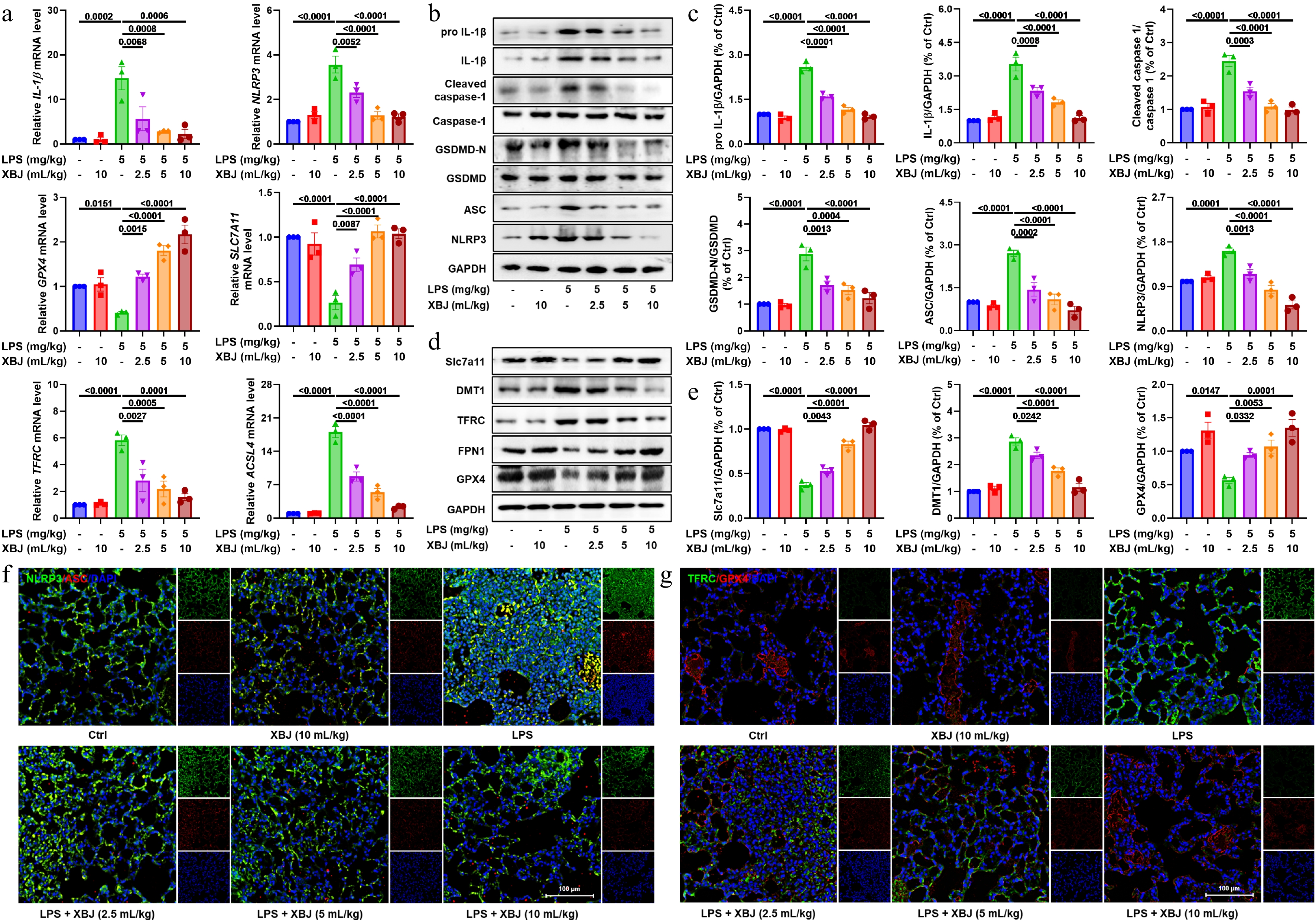
Figure 3.
XBJ alleviated ALI by inhibiting the NLRP3 pathway and ferroptosis. (a) mRNA expressions of GPX4, TFRC, ACSL4, Slc7a11, IL-1β, and NLRP3 (n = 3); (b), (c) effects of XBJ toward NLRP3 pathways in ALI (n = 3); (d), (e) Western blotting analysis of Slc7a11, DMT1, TFRC, FPN1, and GPX4 in ALI (n = 3); (f), (g) representative image of immunofluorescence of TFRC, GPX4, ASC, and NLRP3.
XBJ suppressed LPS-induced inflammation in vitro
-
To find out the XBJ cellular mechanisms against ALI, we analyzed its anti-inflammatory effect using an LPS-stimulated MH-S alveolar macrophage model. The CCK-8 assay verified that XBJ had no cytotoxicity to the MH-S cells at the working concentrations (Fig. 4a). XBJ could reduce the production of nitric oxide (NO) induced by LPS (Fig. 4b). Similar to the in vivo results, XBJ dramatically reduced the release of major inflammatory factors such as IL-6 and TNF-α (Fig. 4c). XBJ also down-regulated the LPS-induced mRNA expression of IL-6, TNF-α, COX-2, IL-1α, IL-1β, CCL5, CXCL10, and NLRP3 (Fig. 4d). Flow cytometric analysis further confirmed the inhibitory effect of XBJ on the expression of IL-6 and IL-1β (Fig. 4e, f, g & h). Overall, these results demonstrated that XBJ inhibited LPS-induced MH-S cell effects with potent anti-inflammatory activity by inhibiting the expression and secretion of a wide range of inflammatory cytokines and chemokines.
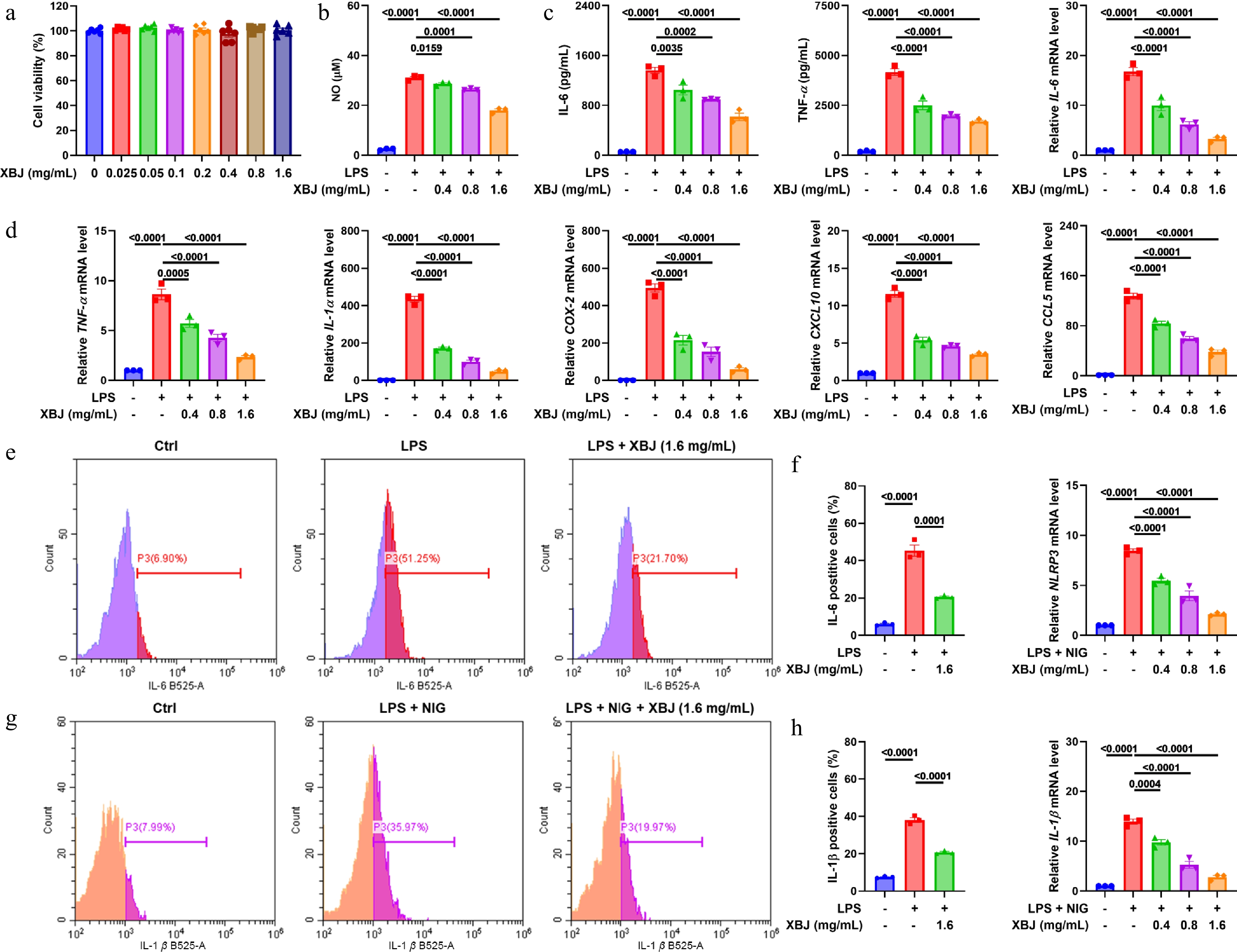
Figure 4.
XBJ suppressed LPS-induced inflammation in MH-S cells. (a) CCK-8 assay of MH-S cells treated with XBJ (n = 5); (b) XBJ inhibited NO production in MH-S cells (n = 3); (c) effects of XBJ toward IL-6 and TNF-α (n = 3); (d) effects of XBJ on mRNA expression levels of IL-6, TNF-α, COX-2, IL-1α, IL-1β, CCL5, CXCL10, and NLRP3 in LPS-mediated MH-S cells (n = 3); (e)–(h) flow cytometry of IL-6 and IL-1β (n = 3).
XBJ ameliorated LPS-induced inflammation by MAPK and NLRP3 pathways in vitro
-
In order to study the in vitro anti-inflammatory mechanism of XBJ, we evaluated the effect of XBJ on the key signaling pathways of LPS-stimulated MH-S cells. Figure 5b and c showed that XBJ could decrease the levels of critical MAPK pathway proteins in LPS-induced MH-S cells. At the same time, XBJ also reduced the expression of protein components of NLRP3 inflammasome, as well as its downstream product proteins, such as NLRP3, pro IL-1β, and IL-1β (Fig. 5d & e). Immunofluorescence staining also showed that XBJ treatment remarkably suppressed the LPS-induced expression of COX-2 and NLRP3 (Fig. 5a & f). These results demonstrated that XBJ alleviated LPS-induced inflammation in MH-S cells, perhaps by inhibiting the MAPK and NLRP3 signaling pathways.
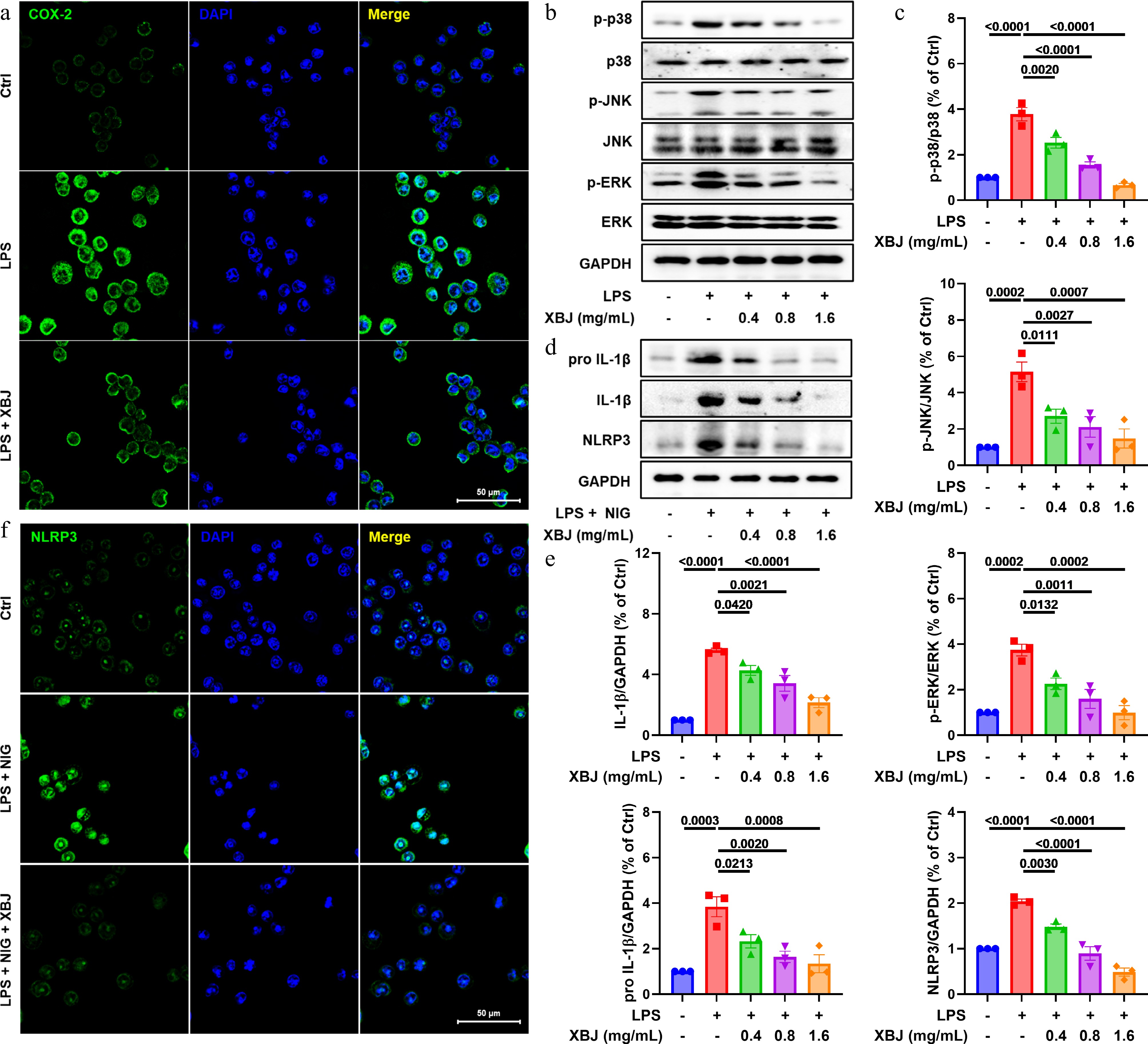
Figure 5.
XBJ suppressed LPS-induced inflammation via MAPK/NLRP3 pathways in vitro. (a), (f) Representative image of COX-2 and NLRP3 staining; (b), (d) XBJ attenuated expression of p-p38, p38, p-JNK, JNK, p-ERK, ERK, pro IL-1β, IL-1β, and NLRP3 (n = 3); (c), (e) quantification of p-p38/p38, p-JNK/JNK, p-ERK/ERK, pro IL-1β, IL-1β, and NLRP3 (n = 3).
XBJ inhibited ferroptosis in LPS-induced MH-S cells
-
As ferroptosis has been increasingly implicated in the pathogenesis of ALI, we further evaluated whether XBJ could modulate this form of cell death[38]. As shown in Fig. 6a, LPS stimulation dramatically increased oxidized lipid accumulation, whereas XBJ treatment reversed this effect. In a concentration-dependent manner, XBJ also attenuated the LPS-induced accumulation of a special probe for ferric ion detection, RhoNOX-1 (Fig. 6b). Furthermore, we analyzed the levels of key mRNAs related to ferroptosis and found that XBJ treatment downregulated the mRNA expressions of ACSL4 and TFRC in MH-S cells after LPS exposure, while upregulating mRNA expressions of Slc7a11, and GPX4 (Fig. 6c). Consistent with these shifts in transcription, XBJ suppressed the expression of DMT1 and TFRC, and enhanced the expression of Slc7a11, FPN1, and GPX4 in LPS-challenged MH-S cells as well (Fig. 6d & e). In summary, these results indicate that XBJ alleviated lipid peroxidation induced by LPS and regulated the ferroptosis-related molecules in macrophages.
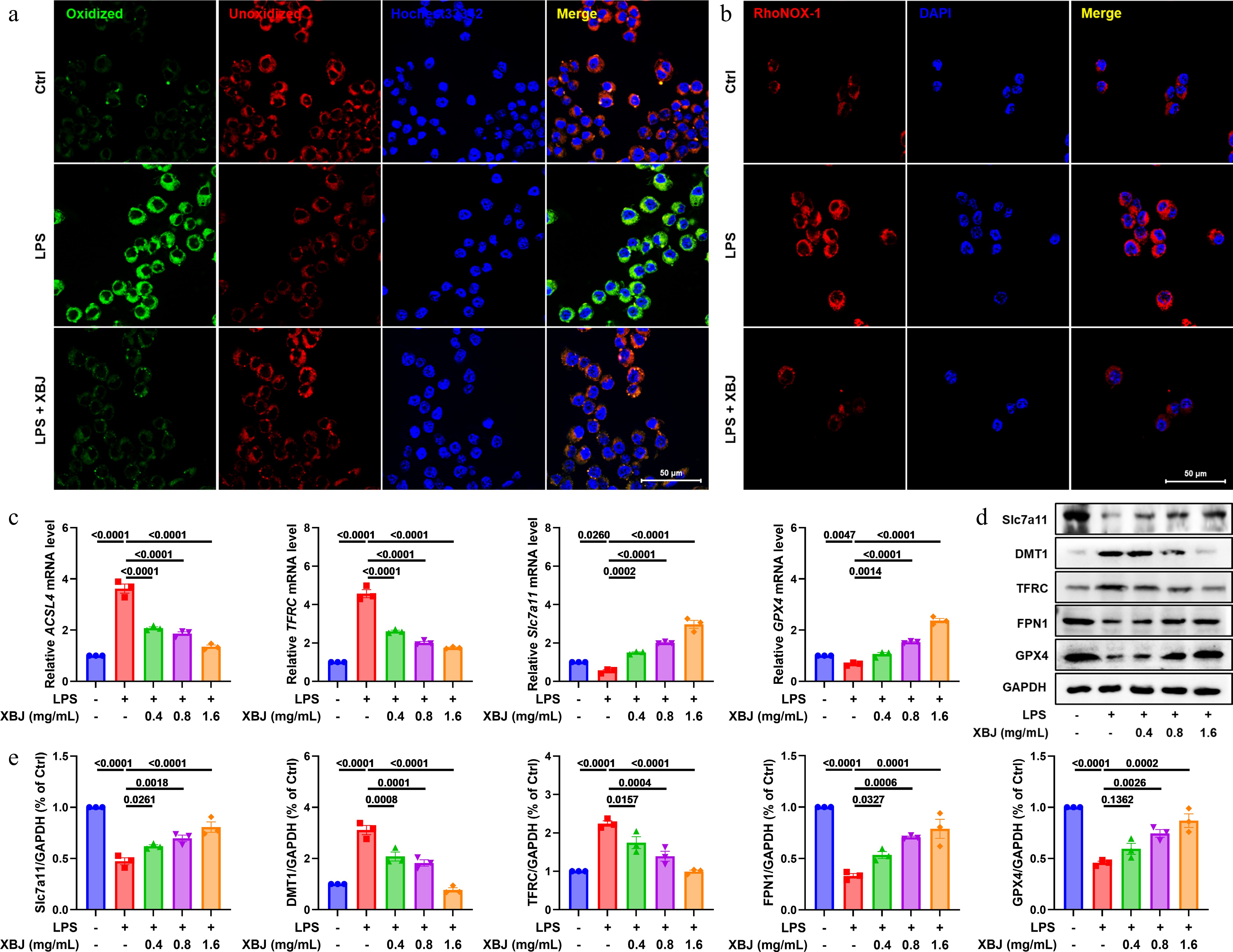
Figure 6.
XBJ inhibited ferroptosis in vitro. (a) Representative images of oxidized and unoxidized staining; (b) representative images of RhoNOX-1 staining; (c) mRNA levels of ACSL4, Slc7a11, TFRC, and GPX4 (n = 3); (d), (e) XBJ decreased expression levels of Slc7a11, DMT1, TFRC, FPN1, and GPX4 in LPS-mediated MH-S cells (n = 3).
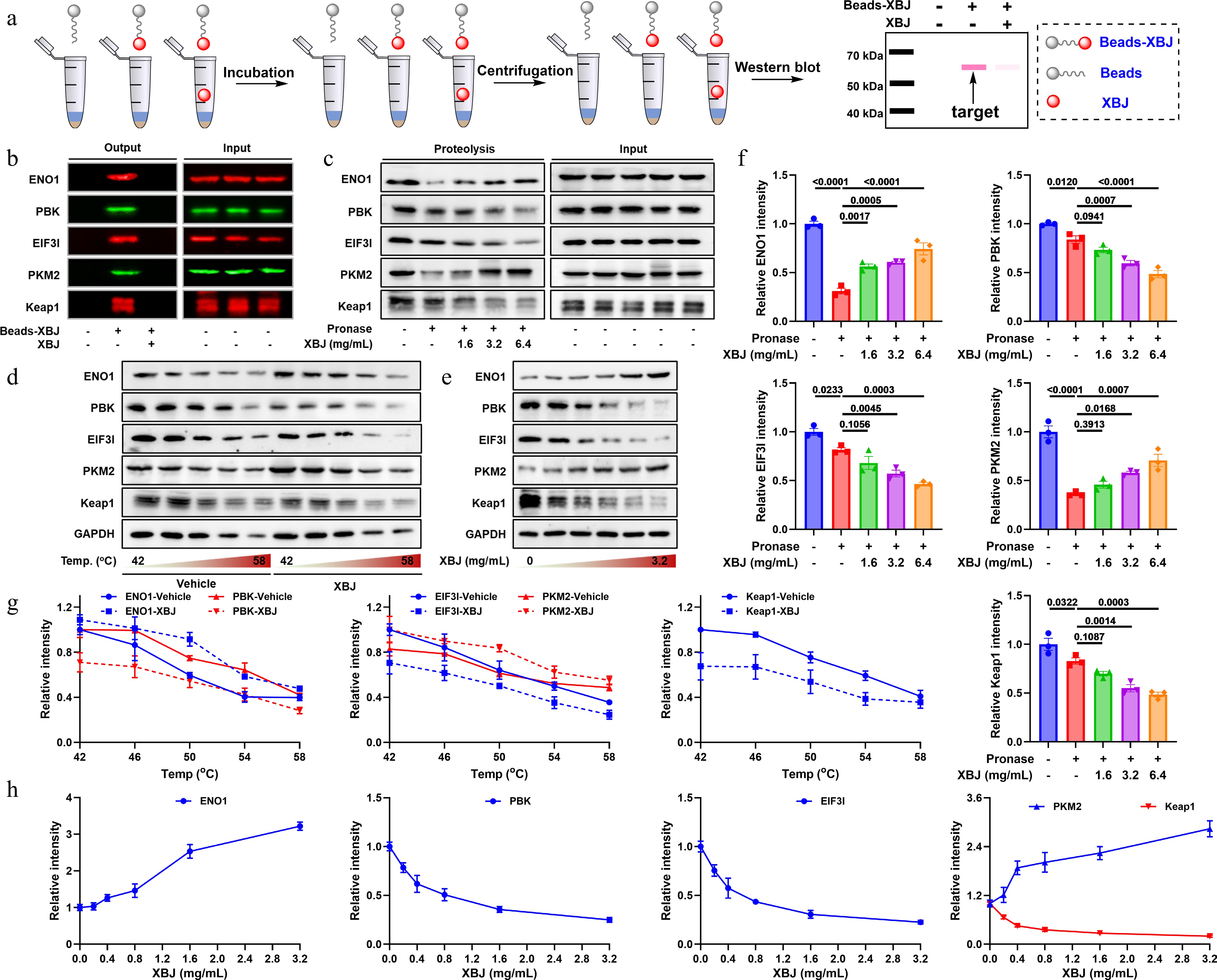
ENO1, PBK, EIF3I, PKM2, and Keap1 were identified as potential targets of XBJ
-
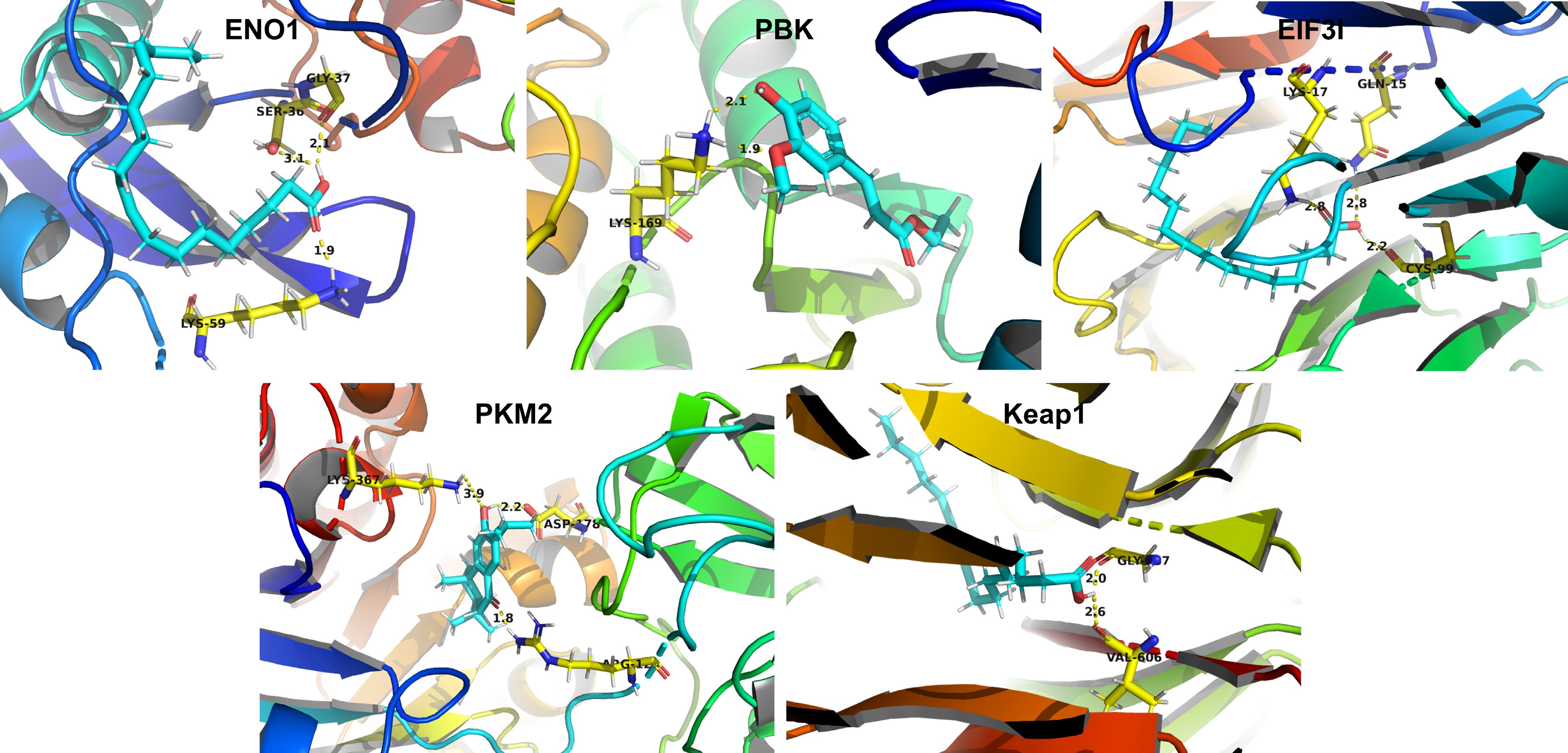
To systematically identify potential molecular targets of XBJ, we developed a bead-conjugated XBJ complex (Beads-XBJ), (Fig. 7a) to capture potential binding proteins as in previous methods[32,39]. Following the affinity pull-down experiment, five proteins were selectively enriched in the Beads-XBJ group, and ENO1, PBK, EIF3I, PKM2, and Keap1 were identified as direct binding targets of XBJ (Fig. 7b), based on the corresponding antibodies. Subsequent validation was performed using CETSA and DARTS assays. The DARTS assay indicated that XBJ altered the susceptibility of five proteins to degradation, which suggested the occurrence of specific binding between XBJ and these proteins (Fig. 7c & f). Consistent with this, CETSA results demonstrated that XBJ treatment altered the thermal stability of five proteins, further confirming their direct interaction with XBJ (Fig. 7d, e, g & h). In order to confirm these results, we analyzed potential active pharmaceutical ingredients of XBJ, introduced into the blood and lungs using LC-MS/MS (Supplementary Figs S2 & S3). We found 13 compounds, and identified them as hydroxysafflor yellow A, ligustilide, isomers of ligustilide, ethyl 4-hydroxy-3-methoxycinnamate, paeoniflorin, neocnidilide, senkyunolide K, 10,12-octadecanedioic acid, oleic acid, linoleic acid, palmitic acid, danshenxinkun D, and sugiol, respectively (Supplementary Table S1). Furthermore, we screened them for the binding to ENO1, PBK, EIF3I, PKM2, and Keap1 by molecular docking. Based on the binding score, oleic acid displayed the best binding to ENO1 through hydrogen bond interactions with Ser36, Gly37, and Lys59, with distances of 1.9, 2.1, and 3.1 Å (Fig. 8), respectively, among them. In addition, ethyl 4-hydroxy-3-methoxycinnamate, sugiol, palmitic acid, and 10,12-octadecadiynoic acid could form a hydrogen bond with amino acid residues of PBK, EIF3I, PKM2, and Keap1 (Fig. 8), respectively. These findings illustrated XBJ directly bound to, and stabilizes five target proteins, which are ENO1, PBK, EIF3I, PKM2, and Keap1 respectively, thereby elucidating its multi-target mechanism underlying anti-inflammatory activity.
-
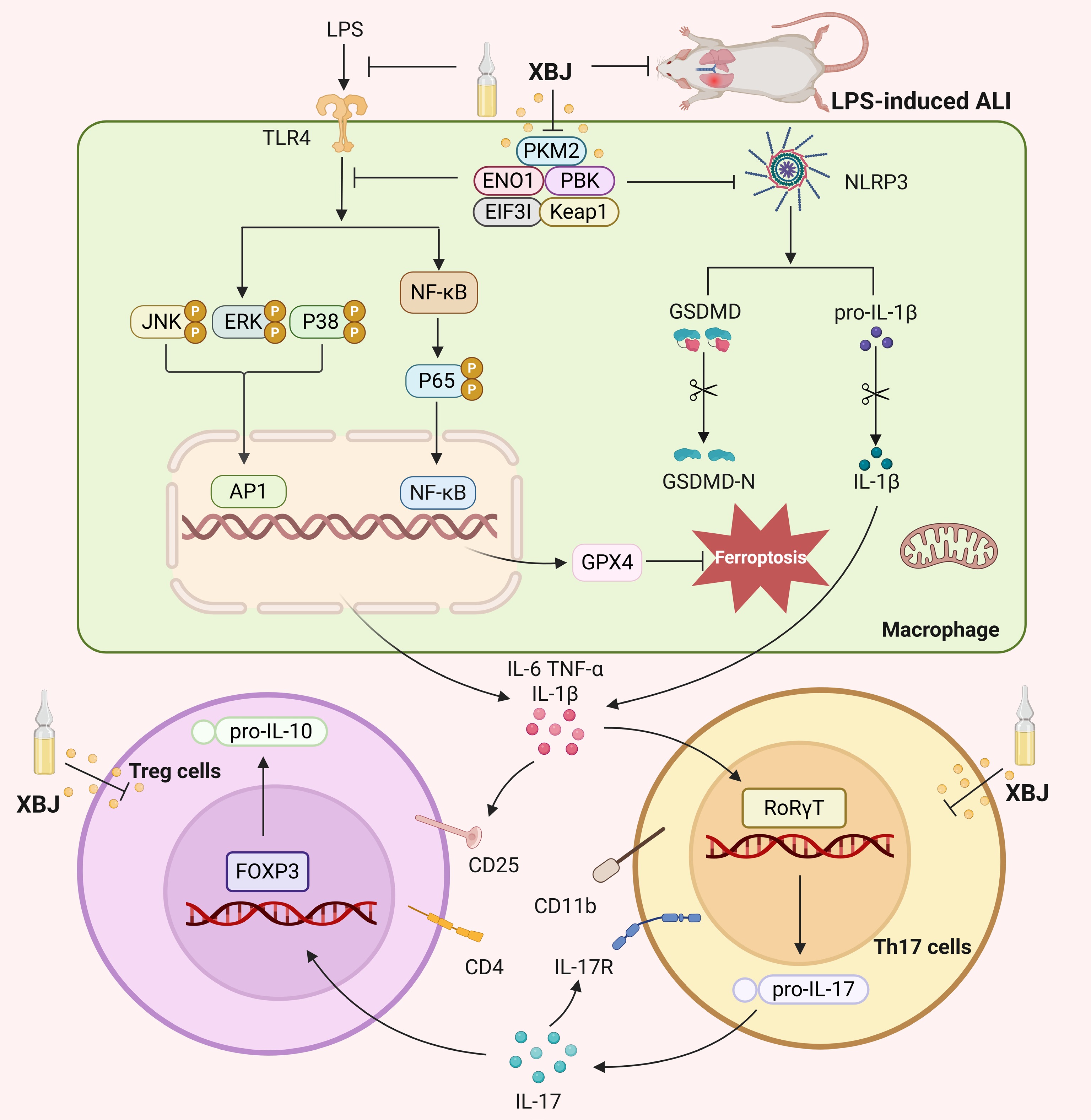
ALI causes direct or indirect damage to lung tissues and leads to severe pathological changes and poor clinical outcomes due to the lack of effective targeted therapies[40−42]. Therefore, the development of therapeutic agents with clear molecular mechanisms holds great significance. This study demonstrated that XBJ improved LPS-induced ALI by restoring the Treg/Th17 balance and inhibiting the release of pro-inflammatory cytokines (including TNF-α, IL-6, and IL-1β). By mechanistic investigations, we identified that XBJ directly combined with five target proteins (PKM2, ENO1, PBK, EIF3I, and Keap1), thereby inhibiting the MAPK/NF-κB pathway, inactivating the NLRP3 inflammasome, and blocking ferroptosis. From these results, we recognized the therapeutic potential of these targets in ALI, positioning XBJ as a promising multi-target modulator for this complicated syndrome.
XBJ is a modernized compound formulation derived from traditional Chinese medicine and widely reported to have anti-infection, anti-inflammation, anti-oxidative stress, and immunomodulatory effects[43,44]. Previous reports also demonstrated that XBJ alleviated ALI by reducing oxidative damage and mitochondrial dysfunction, upregulating miR-181d-5p to inhibit SPP1, and cooperating with miR-223 to suppress the NLRP3 expression and inhibit inflammation and pyroptosis[45]. Moreover, hydroxysafflor yellow A, a major active component of XBJ has been found to protect against ALI by inhibiting the MAPK/p38 and NF-κB/p65 pathways[46]. Quercetin, as one of the main components of XBJ has recently been confirmed by research to inhibit ferroptosis by activating the Sirt1/Nrf2/GPX4 signaling pathway, thereby alleviating ALI induced by LPS[47]. Consistent with previous findings, this work demonstrated that XBJ exerted protection by other mechanisms, such as suppression of ferroptosis, modulating the MAPK/NF-κB/NLRP3 signaling pathway, and the balance between TH17 and Treg cells.
Recent technological advances have greatly accelerated the depth of drug target discovery[48−52]. Heat-protoemic and chemical proteomics are new and good tools that help us systematically learn the interactions of small molecules and proteins and find out how drugs work. Analysis of thermal proteome profiling (TPP) can comprehensively control the thermal stability of proteins and press the chemical proteomics to accurately understand the target protein[14,53]. In comparison, the integrated strategy employed here is especially suitable for the label-free direct screening of multi-component systems such as XBJ, providing a practicable method for target identification in such complicated natural mixtures. In this study, we employed an integrated chemical proteomics strategy to identify the direct target proteins of XBJ, ENO1, PBK, EIF3I, PKM2, and Keap1. These results are supported by affinity pull-down, CETSA and DARTS analyses are a reliable multidimensional approach. It should be noted that, as a multi-component formula, the overall therapeutic effect of XBJ is difficult to attribute precisely to a single constituent-target pair, which is a ubiquitous limitation in pharmacological research on complex formulations. Future analysis of key single constituents will give us a better insight into their respective contributions.
Functionally, these targets are crucial for inflammatory control and metabolic control. PKM2 polarizes macrophages to M1 and enters the nucleus to activate the transcription activity of NF-κB, thus linking metabolic reprogramming and the amplification of inflammatory signaling[54,55]. ENO1 acts as a multifunctional protein and plays an important role in cancer progression, tissue repair, and immune regulation[56,57]. PBK is a serine-threonine kinase in the MAPKK family that promotes inflammatory signaling through the MAPK and RSK pathways, which may regulate the activation of the NLRP3 inflammasome in turn[58,59]. EIF3I participates in cell proliferation, cell cycle progression, and differentiation, mainly through its interaction with PD-L1 IRS4[60,61]. Keap1 is the central hub for the oxidative stress response, regulating the expression of antioxidant genes through the Nrf2 pathway, and has an essential role in suppressing ferroptosis[62−64]. Thus, PKM2, ENO1, PBK, EIF3I, and Keap1 are key regulators of inflammatory cascades and metabolic homeostasis, and they form a regulatory network spanning metabolic reprogramming, signal transduction, translational control, and redox balance. Essentially, this network collectively accounts for the inhibitory effects of XBJ on the MAPK/NF-κB/NLRP3 inflammatory axis and ferroptosis, providing an initial explanation for its multi-target synergistic regulatory effect on treating ALI. In addition, molecular docking analysis showed that key constituents of XBJ, including oleic acid, ethyl 4-hydroxy-3-methoxycinnamate, sugiol, palmitic acid, and 10,12-octadecadiynoic acid, exhibited binding affinities to the aforementioned targets. These results provided a chemical structural basis underpinning the multi-target mechanism of XBJ in ALI (Fig. 9).
-
In summary, this study demonstrated that XBJ alleviated ALI by targeting multiple molecules, including PKM2, ENO1, PBK, EIF3I, and Keap1. This multi-target network coordinately inhibited the MAPK/NF-κB/NLRP3 inflammatory axis and ferroptosis, thereby improving alveolar barrier function and restoring immune homeostasis. These findings provided novel insights into the pathogenesis of ALI, and highlighted potential avenues for developing multi-target therapeutic strategies. It should be noted that this study was mainly conducted in an LPS-induced ALI model, and its broader applicability requires further validation in models of other etiologies or in clinical settings.
-
All animal experiments received formal approval from the Institutional Animal Ethics Committee of Tianjin University of Traditional Chinese Medicine (Approval No. TCM-LAEC2025002s0959).
-
The authors confirm their contributions to the paper as follows: study conception and design: Qiu F, Zhang J, Sun CP; performing experiment: Hui SW, Li XY, Feng XC, Xu XR, Zhang HL, Feng ZQ, Xia Q, Jia YX; manuscript writing: Hui SW, Li XY; manuscript revision: Zhang J, Sun CP. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
-
This work was supported by the Non-communicable Chronic Diseases-National Science and Technology Major Project (No. 2024ZD0528804), Support Program for Young Faculty's Research and Innovation Capability Development in Tianjin Higher Education Institutions Initiated by the Ministry of Education, Outstanding Youth Foundation of Tianjin (No. 25JCJQJC00180), Science Foundation of Tianjin (No. 25JCYBJC00620), Young Scientific and Technological Talents (Level Two) in Tianjin (No. QN20230212).
-
#Authors contributed equally: Siwen Hui, Xinyuan Li, Xinchi Feng
- Supplementary Table S1 Chemical constituents of XBJ that entered into blood and lung.
- Supplementary Fig. S1 Quantification of FPN1 and TFRC expression.
- Supplementary Fig. S2 Total Ion Chromatogram of XBJ in Blood.
- Supplementary Fig. S3 Total Ion Chromatogram of XBJ in Lung Tissue.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press on behalf of China Pharmaceutical University. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Hui S, Li X, Feng X, Xu X, Zhang H, et al. 2026. Xuebijing injection ameliorates LPS-mediated acute lung injury via multi-target synergy by alleviating inflammation and ferroptosis. Targetome 2(1): e005 doi: 10.48130/targetome-0026-0005
Xuebijing injection ameliorates LPS-mediated acute lung injury via multi-target synergy by alleviating inflammation and ferroptosis
- Received: 29 December 2025
- Revised: 22 January 2026
- Accepted: 26 January 2026
- Published online: 09 February 2026
Abstract: Acute lung injury (ALI) is a diffuse alveolar injury caused by infections and other predisposing factors, with associated alveolar dysfunction, pulmonary oedema, and acute respiratory failure. Currently, no specific therapeutic agents are clinically available for ALI. Xuebijing injection (XBJ) is a widely used clinical traditional Chinese medicine formulation, primarily used for respiratory infections. However, the effects and mechanisms of XBJ in ALI are not fully understood. This study demonstrated that XBJ treatment ameliorated ALI progression by heightening alveolar barrier integrity and reducing histopathological lung damage. Mechanistically, XBJ inhibited the activation of the mitogen-activated protein kinase (MAPK), nuclear factor-κB (NF-κB), and NOD-like receptor thermal protein domain associated protein 3 (NLRP3) inflammasome pathways, resulting in reduced production of key pro-inflammatory cytokines (e.g., IL-6, IL-1β, and TNF-α). It also restored immune balance by regulating Treg/Th17 cells and inhibited ferroptosis. Using integrated chemical biology approaches, pyruvate kinase M2 (PKM2), enolase 1 (ENO1), PDZ binding kinase (PBK), eukaryotic translation initiation factor 3i (EIF3I), and kelch-like ECH-associated protein 1 (Keap1) were identified as direct intracellular targets of XBJ, which was further confirmed through various chemical and biological methods. Moreover, compounds from XBJ, which is entered into the blood and lungs, such as palmitic acid, ethyl 4-hydroxy-3-methoxycinnamate, sugiol, oleic acid, and 10,12-octadecadiynoic acid, could bind to these targets, respectively. In summary, XBJ protected against lipopolysaccharide (LPS)-induced ALI by multi-targets, thereby modulasting inflammatory and immune responses, while inhibiting the MAPK/NF-κB/NLRP3 pathways and ferroptosis. These findings offered mechanistic evidence of the application value of XBJ in the treatment of ALI.
-
Key words:
- Acute lung injury /
- Xuebijing injection /
- ALI /
- Multi-target synergy /
- Ferroptosis /
- MAPK/NF-κB/NLRP3