-
As important ornamental plants, wetland irises, including Iris ensata, I. pseudacorus, and I. laevigata, etc., possess a broad range of applications in landscaping, water body restoration, and wetland ecosystems management, owing to their vibrant flowers and strong adaptability[1−3]. Among them, I. ensata, I. ensata var. variegata, and I. sanguinea are particularly notable for their outstanding ornamental qualities and proven performance in practical applications. These species are often favored for use in aquatic landscaping and other wetland-associated plantings due to their resilience to environmental stresses, such as heavy metals and salinity[4−6]. Nevertheless, despite their excellent ornamental and ecological value, several challenges remain in their cultivation and management, including low bud production, inefficient reproduction, slow growth rates, and uneven flowering[7−9]. These issues negatively impact both their landscape appeal and commercial productivity. Plant growth regulators (PGRs) have been extensively used in horticultural crops to enhance growth, development, and overall productivity[10]. Although PGRs are commonly used to improve the growth performance of various plant species, their application in wetland iris cultivation remains limited. Given the potential of PGRs to address the above challenges in wetland irises, investigating their effects on bud sprouting, growth, and flowering is of significant research value and practical importance for enhancing both the aesthetic and economic value of wetland iris cultivation.
PGRs have become increasingly significant in horticultural practices due to their ability to regulate various physiological processes, including cell division, differentiation, and senescence[11,12]. Among these, 6-Benzylaminopurine (6-BA), a synthetic cytokinin, is widely used to promote lateral shoot growth, stimulate bud germination, and delay leaf senescence[13,14]. 6-BA has been shown to enhance lateral shoot production in ornamental plants such as Chrysanthemum[15], and delay leaf senescence in horticultural crops like broccoli (Brassica oleracea var. italica cv. Green King; GK)[16] and Chinese flowering cabbage (Brassica rapa ssp. parachinensis)[17]. Additionally, its application in maize (Zea mays) has demonstrated to increase the rate of carbohydrate transfer from leaves to flower organs, resulting in higher yields[18]. Furthermore, 6-BA stimulates adventitious shoot elongation in mango (Mangifera indica L.)[19], illustrating its broad utility in enhancing plant growth and productivity.
On the other hand, Prohexadione-Ca (PC), a PGR that inhibits the final stages of gibberellin (GA) biosynthesis, has also been widely studied for its impact on PGR[20−22]. PC has been known to reduce plant height and leaf area in species like potato (Solanum tuberosum)[23], and Chinese cabbage (Brassica Rapa)[24]. In fruit trees, such as apple (Malus domestica), PC promotes fruit bud formation and increases cytokinin levels during fruit seed development[25,26]. Furthermore, studies on ornamental plants, including I. germanica, have suggested that cytokinin and PC may regulate rhizome growth and lateral bud production, potentially affecting plant morphology and flowering performance[27].
The effects of PGRs on plant vegetative and reproductive growth may vary considerably with application concentration. As evidenced by studies on I. germanica, treatment with 6-BA at 1,000 mg/L promoted lateral bud emergence more rapidly than at 3,000 mg/L[28]. This differential effect of concentration is also apparent in Camellia oleifera, where 6-BA at 500 mg/L favored vegetative growth, enhancing shoot development and leaf bud formation, whereas a concentration of 1,500 mg/L more effectively promoted flower bud differentiation[29]. Furthermore, research on PC in I. germanica has demonstrated that application at 700 mg/L promoted rhizome growth and lateral shoot production more effectively than at 1,500 mg/L[27]. However, research on the application of 6-BA and PC in irises remains largely unexplored, particularly for wetland irises. This highlights the need for further research to explore the application of these PGRs in Iris cultivation and to assess their potential in enhancing ornamental traits in the Iris genus.
In this study, three preferred wetland Iris species, including I. ensata, I. ensata var. variegata, and I. sanguinea, were selected as research subjects, which was based on their suitability for horticultural cultivation and landscape applications in Hangzhou, China. The research aims to investigate the effects of varying concentrations of 6-BA and PC on key growth characteristics of these species, such as bud number, plant height, increase in rhizome diameter, rhizome weight, and flowering performance. By comparing these parameters under different treatments, the study seeks to evaluate how 6-BA and PC influence the growth and development of wetland irises. The findings will provide valuable insights for optimizing the cultivation practices of wetland irises in the Hangzhou region. Furthermore, this research will contribute to theoretical knowledge on the effective use of PGRs for improving the ornamental and functional traits of wetland irises, offering practical guidance for their application in landscape design.
-
Three superior Iris germplasms, commonly cultivated in the south of the Yangtze River Delta, were used as experimental materials: I. ensata, I. ensata var. variegata, and I. sanguinea. After two years of clonal propagation, healthy and morphologically uniform plants were carefully selected for the experimental study. To ensure accurate data collection, the newly emerged lateral buds on the rhizomes were removed prior to treatment, preventing interference with subsequent sprouting statistics.
The experiment was conducted at the Qingshan Lake Base (120 °E, 30 °N) of Hangzhou Landscaping Incorporated, Hangzhou, Zhejiang Province, China. The prepared plants were transplanted into two-gallon pots filled with a mixed pine scale, peat, and perlite in a volume ratio of 5:4:1, supplemented with controlled-release fertilizer at 4 kg/m3. The experimental pots were maintained under rain-sheltered conditions.
Experimental design
-
The study aimed to compare the effects of exogenous PGRs on their germination, growth, and flowering characteristics, with the goal of establishing optimized propagation protocols for Iris cultivation in Zhejiang Province, China. The experimental design employed a completely randomized design with five distinct treatments, each replicated three times. Each replication comprised 20 individual pots, resulting in a total of 300 pots for the entire experiment (Table 1).
Table 1. Experimental treatments and application concentrations of PGRs.
Treatment Plant growth regulator Application concentration A0 − − A1 6-BA 1,000 mg/L A2 6-BA 2,000 mg/L A3 PC 500 mg/L A4 PC 1,000 mg/L 6-BA, 6-Benzylaminopurine; PC, Prohexadione-Ca. Treatment A0 served as the control group, where plants were treated with distilled water. Each treatment received 1,000 mL per application on Oct. 30, Nov. 30, and Dec. 30, 2019, and on Jan. 30 and Feb. 29, 2020. Plant establishment commenced on Oct. 8, 2019, with the transplantation of prepared rhizomes into two-gallon pots, maintaining one plant per pot to ensure uniform growth conditions. The application of PGR was administered through irrigation at five critical time points: Oct. 30, Nov. 30, Dec. 30 of 2019, and Jan. 30, Feb. 29 of 2020, respectively. The treatment solutions were formulated as follows: A1: 1,000 mg/L 6-BA, A2: 2,000 mg/L 6-BA, A3: 500 mg/L PC, A4: 1,000 mg/L PC, and A0: control (distilled water).
Each treatment application consisted of a 1,000 ml solution, standardized across all groups. To enhance solution adsorption and ensure uniform distribution, three to four drops of Tween-20 (a surfactant) were added to each treatment solution, including the control. Following potting and throughout the experimental period, all plants received standardized horticultural management following conventional cultivation practices for Iris species, maintaining consistent environmental conditions across all treatments.
Determination of growth indices
-
To evaluate the treatment effects on plant development, 12 replicate pots from each treatment were randomly selected for morphological analysis. The largest diameter of the rhizome was measured using digital vernier calipers before potting. In the spring of 2020, vegetative growth was quantitatively assessed by recoding shoot emergence rates, specifically measuring the number of shoots that successfully penetrated the soil surface and developed fully extended leaves per individual plant. The plant height measurements were conducted monthly from May to Oct., with recoding taken on the final day of each month. Concurrently, floral development was determined through quantitative analysis of flower production and measurement of maximal floral stem height.
Following natural senescence, indicated by complete leaf yellowing on Nov. 30, the rhizome and root systems were carefully excavated, thoroughly washed, and air-dried for subsequent analysis. Rhizome development was evaluated by measuring the maximum diameter using digital vernier calipers and calculating rhizome diameter variations. The collected data from two years were subjected to a comprehensive comparative analysis to assess the influence of different treatments on the reproductive capacity, vegetative growth parameters, and flowering characteristics of Iris development.
Statistical analysis
-
All experimental data were subjected to statistical analysis using SPSS 18.0 (IBM Corporation, Armonk, NY, USA), and Microsoft Excel 2021 (Microsoft Corporation, Redmond, WA, USA) software packages. To explore the linear relationships between different indices, Pearson's correlation coefficient was used for bivariate correlation analysis. Treatments effects were analyzed through one-way analysis of variance (ANOVA) and Tukey's HSD test for post-hoc comparisons (* p < 0.05, ** p < 0.01, *** p < 0.001). Values with a probability value p < 0.05 were considered significantly different between treatment groups.
-
Four treatments were tested for their effects on the bud production in I. ensata, I. ensata var. variegate, and I. sanguinea (Fig. 1a). However, distinct responses to the types and concentrations of PGRs were observed among the different Iris species. In I. ensata, all four treatments increased the bud number. Specifically, the A1 and A2 treatments with 6-BA resulted in increases of 0.17 and 0.67 buds, respectively. Similarly, the A3 and A4 treatments with PC led to increases of 0.75 and 0.34 buds, respectively. Notably, the highest bud production in I. ensata was observed with the low concentration of PC (A3 treatment), yielding the largest increase in bud number in spring.
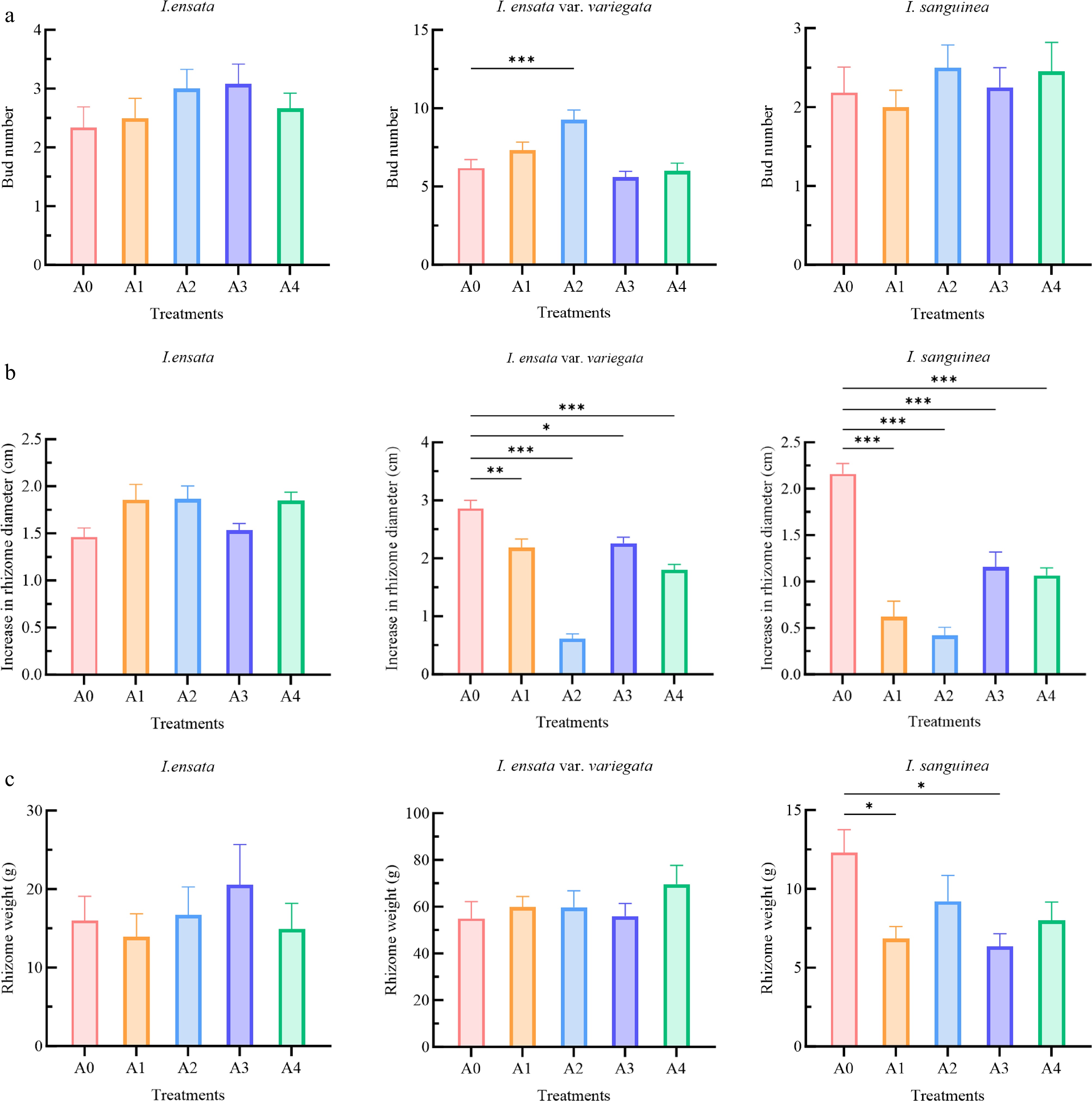
Figure 1.
Effects of plant growth regulator treatments on (a) bud number, (b) increase in rhizome diameter, and (c) rhizome weight in three Iris species (I. ensata, I. ensata var. variegata, and I. sanguinea). The treatments included: A0 (control group), A1 (low concentration of 6-BA, 1,000 mg/L), A2 (high concentration of 6-BA, 2,000 mg/L), A3 (low concentration of PC, 500 mg/L), and A4 (high concentration of PC, 1,000 mg/L). Each bar represents the mean (n = 12) ± standard error. The significant differences within each species compared to the A0 (control group) were analyzed using Tukey's HSD test following one-way ANOVA (* p < 0.05, ** p < 0.01, *** p < 0.001).
For I. ensata var. variegata, both 6-BA treatments promoted bud formation. In particular, the A1 treatment increased the number of buds by 1.16, while the A2 treatment resulted in a more pronounced increase of 3.08, which was statistically significant (p < 0.001). In contrast, PC treatment led to a reduction in bud number compared to the control, though the difference was not significant.
In I. sanguinea, the highest bud count was observed with the application of 6-BA in the A2 treatment, which resulted in an increase of 0.32 buds. The A1 treatment led to a decrease in the number of buds by 0.18. Compared to the control group, the number of buds increased by 0.07 and 0.27 under the two PC treatments. However, the effects of the four treatments on I. sanguinea were not significant overall.
Effects of PGR treatments on rhizome growth
-
Iris rhizome diameter and weight are key indicators for evaluating rhizome growth. The influence of four PGR treatments on rhizome diameter and weight was assessed across three Iris species, with varying responses observed for each species (Fig. 1b, c).
In I. ensata, the rhizome diameter increased by 0.40 and 0.41 cm under the A1 and A2 treatments with 6-BA, respectively. Treatments with PC at A3 and A4 resulted in increases of 0.07 and 0.39 cm, respectively. However, none of these treatments showed a significant difference compared to the control group. For rhizome weight, I. ensata rhizome showed increases of 0.72 and 4.54 g under the A2 treatment with the high concentration of 6-BA, and the A3 treatment with the low concentration of PC, respectively. Two further treatments led to lower rhizome weights than the control, but these differences were not significant.
In I. ensata var. variegata, the effects on rhizome diameter were pronounced. The control group showed a 2.90 cm increase in rhizome diameter, whereas the A1 and A2 treatments with 6-BA significantly resulted in reductions of 0.72 cm (p < 0.01), and 2.29 cm (p < 0.001), respectively. Similarly, the average rhizome diameter in the PC-treated groups (A3 and A4) significantly decreased by 0.64 cm (p < 0.05) and 1.10 cm (p < 0.001), compared with the control. For rhizome weight, all treatments resulted in an increase compared to the control, with 6-BA treatments at low (A1) and high (A2) concentrations increasing weight by 4.98 and 4.86 g, respectively. The PC treatments at low (A3) and high (A4) concentrations resulted in average increases of 1.10 and 14.74 g, respectively. However, none of these differences were significant.
For I. sanguinea, rhizome diameter was significantly reduced by both 6-BA and PC treatments (p < 0.001). The 6-BA treatments (A1 and A2) decreased the rhizome diameter by 1.54 and 1.73 cm, respectively, while the PC treatments (A3 and A4) led to reductions of 1.00 and 1.11 cm. Concerning rhizome weight, all treatments resulted in lower values than the control, with reductions of 5.45 (A1), 3.08 (A2), 5.96 (A3), and 4.30 g (A4). It should be noted that the weight losses in treatments A1 and A3 were statistically significant (p < 0.05).
Effects of PGR treatments on plant height during the growing season
-
The impact of PGR treatments on the plant height of Iris species during the growing season varied in both magnitude and response, depending on the concentrations and species (Fig. 2). For I. ensata, the A3 treatment with PC yielded the most pronounced numerical effect on plant height, although this increase was not significant compared to the control. At the end of June, the A1 treatment with 6-BA resulted in a discernible numerical reduction in plant height than the control, while the other treatments showed similar effects to the control. In contrast, for I. ensata var. variegata, all PGR treatments significantly reduced plant height, with measurements lower than the control. The reductions were most pronounced in July, with the A2 and A3 treatments showing high significance (p < 0.01) and the A4 treatment being extremely significant (p < 0.001). This strong inhibitory effect persisted for the A4 treatment in Aug. (p < 0.001), while the effects of A2 and A3 remained significant (p < 0.05). By Sep., the A2 treatment exhibited an extremely significant reduction (p < 0.001), with A3 and A4 also being highly significant (p < 0.01). Finally, in Oct., a significant effect was maintained for the A3 treatment (p < 0.05). But for I. sanguinea, no significant differences in plant height were observed between the four treatments and the control.
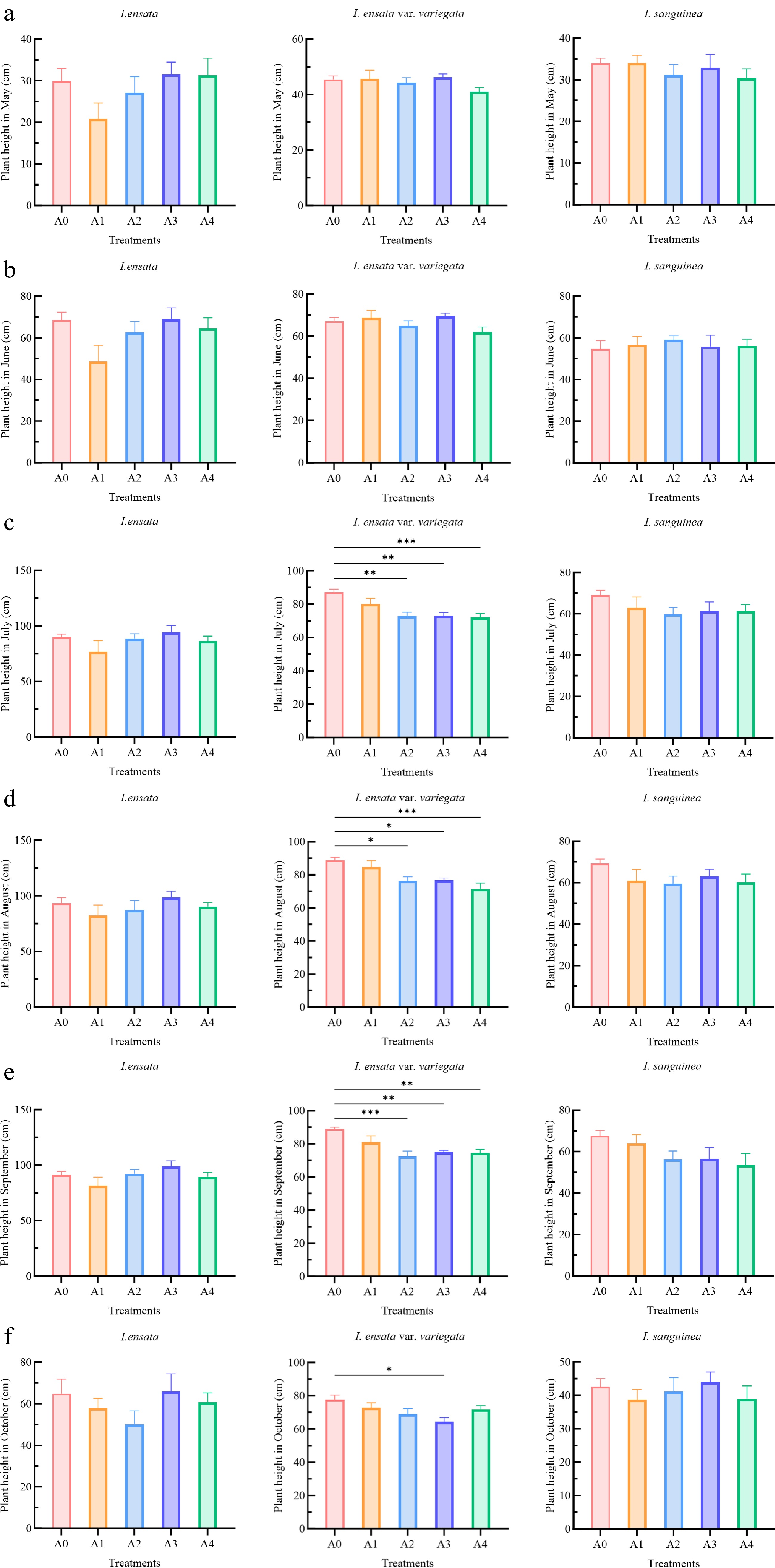
Figure 2.
Effects of plant growth regulator treatments on plant height of three Iris species (I. ensata, I. ensata var. variegata, and I. sanguinea) during the growing season. The treatments included: A0 (control group), A1 (low concentration of 6-BA, 1,000 mg/L), A2 (high concentration of 6-BA, 2,000 mg/L), A3 (low concentration of PC, 500 mg/L), and A4 (high concentration of PC, 1,000 mg/L). (a) May; (b) June; (c) July; (d) August; (e) September; and (f) October. Each bar represents the mean (n = 12) ± standard error. The significant differences within each species compared to the A0 (control group) were analyzed using Tukey's HSD test following one-way ANOVA (* p < 0.05, ** p < 0.01, *** p < 0.001).
Effects of PGR treatments on the number of flowers per plant and stalk height
-
The effects of PGR treatments on the number of flowers per plant and flower stalk height were examined in three Iris species (Fig. 3). In I. ensata, the highest number of flowers per plant was observed in plants treated with a low concentration (A3) of PC (Fig. 3a). Interestingly, the same treatment significantly reduced the flower stalk height (p < 0.05) in both I. ensata and I. ensata var. variegata, producing the shortest stalks among all groups (Fig. 3b). The number of flowers per plant of I. ensata var. variegata treated with a low concentration (A3) of PC was the lowest, though the difference was not significant compared to the control and other treatments. For I. sanguinea, both A1 and A3 treatments promoted flowering compared to the control, with the latter (A3) producing the highest average of 10 flowers per pot. In contrast, A2 treatment significantly suppressed flowering (p < 0.001). However, no significant differences in flower stalk height were observed between the treatments and control.
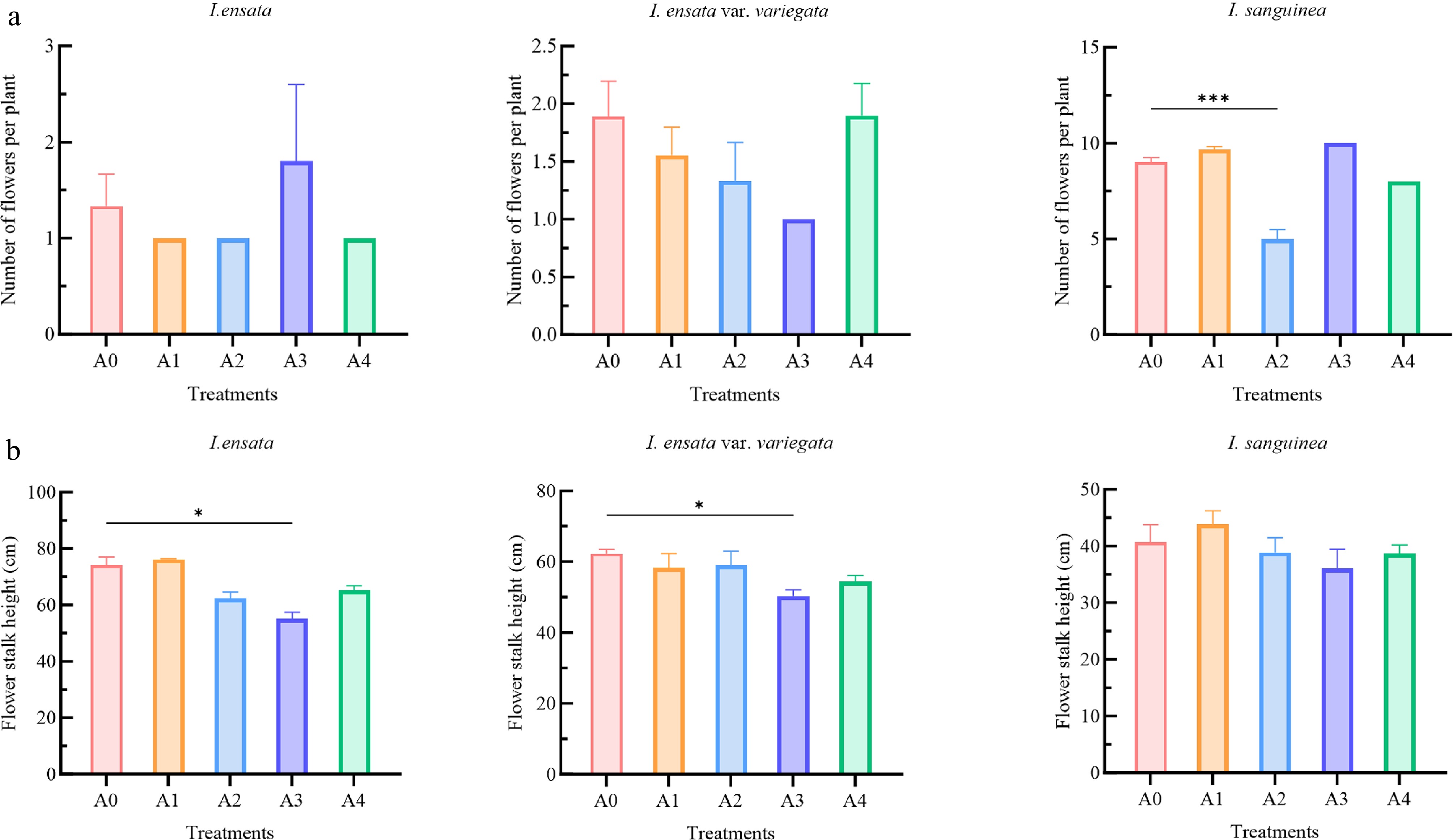
Figure 3.
Effects of plant growth regulator treatments on (a) number of flowers per plant, and (b) flower stalk height in three Iris species (I. ensata, I. ensata var. variegata, and I. sanguinea). The treatments included: A0 (control group), A1 (low concentration of 6-BA, 1,000 mg/L), A2 (high concentration of 6-BA, 2,000 mg/L), A3 (low concentration of PC, 500 mg/L), and A4 (high concentration of PC, 1,000 mg/L). Each bar represents an average (n = 12) ± standard error. The significant differences within each species compared to the A0 (control group) were analyzed using Tukey's HSD test following one-way ANOVA (* p < 0.05, ** p < 0.01, *** p < 0.001).
Correlation analysis of growth indices and Iris species under PGR treatments
-
The correlations among various growth variables across Iris species were examined (Table 2). The correlation between Iris species and bud number was weak and negative (r = −0.062), indicating a slight inverse relationship. Similarly, the correlations between Iris species and rhizome growth indices (diameter and weight) were negligible (r = −0.045 and r = −0.104, respectively), suggesting minimal species influence on rhizome growth. Regarding flowering traits, Iris species showed a strong positive correlation with the number of flowers per plant (r = 0.835, p < 0.01), highlighting the species differences in flower production. However, a negative correlation with flower stalk height (r = −0.761, p < 0.01) suggesting that species with a higher number of flowers per plant tend to have shorter flower stalks. These findings underscore the variability in flower production and stalk elongation across species. In terms of plant height, significant correlations were observed across all months, indicating the distinct growth patterns among species. Some species exhibited early growth followed by a slowdown in later months, while others maintained more consistent growth throughout the season.
Table 2. Correlation analysis of growth indices and plant growth regulator treatments in wetland irises.
Iris species Bud number Plant
height
in MayPlant
height
in JunePlant
height
in JulyPlant
height
in Aug.Plant
height
in Sep.Plant
height
in Oct.Number of
flowers per
plantFlower
stalk
heightIncrease in
rhizome
diameterRhizome
weightIris species 1 −0.062 0.152* −0.168* −0.538** −0.559** −0.649** −0.369** 0.835** −0.761** −0.045 −0.104 Bud number −0.062 1 0.484** 0.271** 0.111 0.122 0.104 0.409** −0.568** 0.417** 0.370** 0.727** Plant height in May 0.152* 0.484** 1 0.612** 0.372** 0.297** 0.271** 0.317** −0.286** 0.252* 0.283** 0.501** Plant height in June −0.168* 0.271** 0.612** 1 0.675** 0.571** 0.548** 0.259** −0.315** 0.129 0.137 0.281** Plant height in July −0.538** 0.111 0.372** 0.675** 1 0.865** 0.899** 0.367** −0.455** 0.293** 0.137 0.163* Plant height in Aug. −0.559** 0.122 0.297** 0.571** 0.865** 1 0.872** 0.351** −0.499** 0.360** 0.128 0.174* Plant height in Sep. −0.649** 0.104 0.271** 0.548** 0.899** 0.872** 1 0.453** −0.528** 0.483** 0.149 0.198* Plant height in Oct. −0.369** 0.409** 0.317** 0.259** 0.367** 0.351** 0.453** 1 −0.564** 0.553** 0.192* 0.458** Number of flowers per plant 0.835** −0.568** −0.286** −0.315** −0.455** −0.499** −0.528** −0.564** 1 −0.688** −0.232* −0.544** Flower stalk height −0.761** 0.417** 0.252* 0.129 0.293** 0.360** 0.483** 0.553** −0.688** 1 0.229 0.357** Increase in rhizome diameter −0.045 0.370** 0.283** 0.137 0.137 0.128 0.149 0.192* −0.232* 0.229 1 0.520** Rhizome weight −0.104 0.727** 0.501** 0.281** 0.163* 0.174* 0.198* 0.458** −0.544** 0.357** 0.520** 1 * Significant (p < 0.05), ** Highly significant (p < 0.01). The correlations between other growth indicators reveal some interesting trends. The bud number was negatively correlated with the number of flowers per plant (r = −0.568, p < 0.01), but positively correlated with rhizome weight (r = 0.727, p < 0.01). The plant height across different months also showed significant positive correlations, suggesting a consistent growth trajectory. The number of flowers per plant was negatively correlated with flower stalk height (r = −0.688, p < 0.01), indicating that a higher number of flowers per plant is associated with shorter stalks. The rhizome diameter was weakly positively correlated with plant height in May (r = 0.283, p < 0.01), and moderately positively correlated with rhizome weight (r = 0.520, p < 0.01).
Correlation analysis of growth indices and PGR treatments in irises
-
The relationship between different growth regulator treatments and growth indices in I. ensata showed treatments had positive correlations with bud number (r = 0.159), rhizome diameter (r = 0.020), and weight (r = 0.051, Table 3). However, treatments negatively correlated with the number of flowers per plant (r = −0.027), and flower stalk height (r = −0.660, p < 0.05). Notably, the bud number was positively correlated with rhizome weight (r = 0.354, p< 0.01) and the plant height across months, indicating consistent growth. Rhizome diameter also positively correlated with plant height in May (r = 0.358, p < 0.01), and there was a moderate positive correlation with rhizome weight (r = 0.540, p < 0.01). These findings suggest that rhizome diameter is an important indicator of early-season growth and rhizome development.
Table 3. Correlation analysis of growth indices and plant growth regulator treatments in I. ensata.
Treatments Bud
numberPlant
height
in MayPlant
height
in JunePlant
height
in JulyPlant
height
in Aug.Plant
height
in Sep.Plant
height
in Oct.Number of
flowers per
plantFlower
stalk
heightIncrease in
rhizome
diameterRhizome
weightTreatments 1 0.159 0.152 0.085 0.075 0.064 0.116 0.066 −0.027 −0.660* 0.020 0.051 Bud number 0.159 1 0.421** 0.379** 0.310* 0.298* 0.314* −0.058 0.095 −0.481 0.099 0.354** Plant height in May 0.152 0.421** 1 0.849** 0.671** 0.660** 0.713** 0.060 0.103 −0.582 0.358** 0.383** Plant height in June 0.085 0.379** 0.849** 1 0.836** 0.753** 0.805** 0.109 0.152 −0.451 0.201 0.361** Plant height in July 0.075 0.310* 0.671** 0.836** 1 0.853** 0.937** 0.129 0.212 −0.538 0.166 0.306* Plant height in Aug. 0.064 0.298* 0.660** 0.753** 0.853** 1 0.891** 0.086 0.258 −0.512 0.172 0.296* Plant height in Sep. 0.116 0.314* 0.713** 0.805** 0.937** 0.891** 1 0.138 0.298 −0.588 0.118 0.282* Plant height in Oct. 0.066 −0.058 0.060 0.109 0.129 0.086 0.138 1 0.185 −0.122 −0.078 −0.097 Number of flowers per plant −0.027 0.095 0.103 0.152 0.212 0.258 0.298 0.185 1 NA −0.012 0.088 Flower stalk height −0.660* −0.481 −0.582 −0.451 −0.538 −0.512 −0.588 −0.122 NA 1 −0.436 −0.511 Increase in rhizome diameter 0.020 0.099 0.358** 0.201 0.166 0.172 0.118 −0.078 −0.012 −0.436 1 0.540** Rhizome weight 0.051 0.354** 0.383** 0.361** 0.306* 0.296* 0.282* −0.097 0.088 −0.511 0.540** 1 * Significant (p < 0.05), ** Highly significant (p < 0.01).
NA indicates that it cannot be calculated because at least one variable is a constant.For I. ensata var. variegata, treatments showed a minor negative correlation with bud number (r = −0.136) and rhizome diameter (r = −0.027), but a significant positive correlation with rhizome weight (r = 0.291, p < 0.05, Table 4). Regarding flowering traits, the number of flowers per plant was negatively correlated (r = −0.038), and flower stalk height strongly negatively correlated (r = −0.613, p < 0.01) with the treatments, suggesting that fewer flowers is associated with taller flower stalks. The plant height exhibited more or less significant negative correlation with treatments across all month, suggesting that growth regulators might inhibit plant growth.
Table 4. Correlation analysis of growth indices and plant growth regulator treatments in I. ensata var. variegata.
Treatments Bud
numberPlant
height
in MayPlant
height
in JunePlant
height
in JulyPlant
height
in Aug.Plant
height
in Sep.Plant
height
in Oct.Number of
flowers per
plantFlower
stalk
heightIncrease in
rhizome
diameterRhizome
weightTreatments 1 −0.136 −0.171 −0.161 −0.512** −0.535** −0.477** −0.275* −0.038 −0.613** −0.027 0.291* Bud number −0.136 1 −0.186 −0.047 −0.126 −0.105 −0.159 0.063 −0.277 0.264 0.231 0.158 Plant height in May −0.171 −0.186 1 0.733** 0.633** 0.630** 0.597** 0.350** −0.127 0.350 −0.414** −0.100 Plant height in June −0.161 −0.047 0.733** 1 0.678** 0.615** 0.599** 0.423** −0.270 0.467* −0.282 −0.029 Plant height in July −0.512** −0.126 0.633** 0.678** 1 0.745** 0.796** 0.667** −0.011 0.655** 0.005 0.035 Plant height in Aug. −0.535** −0.105 0.630** 0.615** 0.745** 1 0.747** 0.416** −0.103 0.603** −0.142 −0.079 Plant height in Sep. −0.477** −0.159 0.597** 0.599** 0.796** 0.747** 1 0.643** 0.196 0.624** 0 0.027 Plant height in Oct. −0.275* 0.063 0.350** 0.423** 0.667** 0.416** 0.643** 1 0.167 0.639** 0.150 0.139 Number of flowers per plant −0.038 −0.277 −0.127 −0.270 −0.011 −0.103 0.196 0.167 1 0.118 0.031 −0.148 Flower stalk height −0.613** 0.264 0.350 0.467* 0.655** 0.603** 0.624** 0.639** 0.118 1 0.087 −0.274 Increase in rhizome diameter −0.027 0.231 −.414** −0.282 0.005 −0.142 0 0.150 0.031 0.087 1 0.407** Rhizome weight 0.291* 0.158 −0.100 −0.029 0.035 −0.079 0.027 0.139 −0.148 −0.274 0.407** 1 * Significant (p < 0.05), ** Highly significant (p < 0.01). The relationships between other growth indicators also revealed some intriguing patterns. Plant height exhibited significant positive correlations across different months, indicating a consistent growth trend over time. Flower stalk height was positively correlated with plant height throughout the season, suggesting that both flower stalk height and plant height increased in tandem during the plant's growth period. Additionally, rhizome diameter showed a moderate positive correlation with rhizome weight (r = 0.407, p < 0.01).
In I. sanguinea, the treatments showed a slight positive correlation (r = 0.116) with bud number, while rhizome growth was negatively affected (rhizome diameter: r = −0.521, p < 0.01; rhizome weight: r = −0.281, p < 0.05, Table 5). This suggests that the treatments had an adverse impact on rhizome growth in I. sanguinea. Regarding flowering traits, the flowering number and flower stalk height also showed negative correlations with treatments. The plant height had a transient positive correlation in June (r = 0.019) but was otherwise negatively correlated. The rhizome diameter positively correlated with both rhizome weight (r = 0.661, p < 0.01) and plant height in July (r = 0.395, p < 0.05), Aug. (r = 0.436, p < 0.05), and Sep. (r = 0.479, p < 0.01). These results indicate that rhizome diameter is a key indicator of overall plant vigor and growth during the mid-season months.
Table 5. Correlation analysis of growth indices and plant growth regulator treatments in I. sanguinea.
Treatments Bud
numberPlant
height
in MayPlant
height
in JunePlant
height
in JulyPlant
height
in Aug.Plant
height
in Sep.Plant
height
in Oct.Number of
flowers per
plantFlower
stalk
heightIncrease in
rhizome
diameterRhizome
weightTreatments 1 0.116 −0.150 0.019 −0.181 −0.178 −0.343** −0.032 −0.119 −0.189 −0.521** −0.281* Bud number 0.116 1 −0.068 0.200 0.035 0.064 −0.104 −0.270* −0.142 0.059 −0.080 0.227 Plant height in May −0.150 −0.068 1 0.190 0.513** 0.109 0.312* 0.270* 0.073 0.191 0.283 0.072 Plant height in June 0.019 0.200 0.190 1 0.410** 0.103 0.203 0.014 −0.047 −0.071 0.044 0.239 Plant height in July −0.181 0.035 0.513** 0.410** 1 0.720** 0.824** 0.248 0.164 −0.056 0.395* 0.489** Plant height in Aug. −0.178 0.064 0.109 0.103 0.720** 1 0.704** 0.231 0.209 −0.063 0.436* 0.466** Plant height in Sep. −0.343** −0.104 0.312* 0.203 0.824** 0.704** 1 0.429** 0.210 0.196 0.479** 0.545** Plant height in Oct. −0.032 −0.270* 0.270* 0.014 0.248 0.231 0.429** 1 0.078 −0.006 0.056 0.002 Number of flowers per plant −0.119 −0.142 0.073 −0.047 0.164 0.209 0.210 0.078 1 0.007 0.196 −0.022 Flower stalk height −0.189 0.059 0.191 −0.071 −0.056 −0.063 0.196 −0.006 0.007 1 0.054 −0.087 Increase in rhizome diameter −0.521** −0.080 0.283 0.044 0.395* 0.436* 0.479** 0.056 0.196 0.054 1 0.661** Rhizome weight −0.281* 0.227 0.072 0.239 0.489** 0.466** 0.545** 0.002 −0.022 −0.087 0.661** 1 * Significant (p < 0.05), ** Highly significant (p < 0.01). -
In this study, two PGRs, including 6-BA and PC, were applied to investigate their effects on bud germination, vegetative growth, and flowering in three species of wetland iris. 6-BA, a synthetic cytokinin, is known to promote bud differentiation and cell division, thereby inducing bud formation and enhancing lateral bud growth[30,31]. In the present experiment, 6-BA significantly increased bud number in I. ensata var. variegata, which aligns with its documented role in stimulating lateral or tiller bud development in other plants, such as Jatropha curcas and rice[32,33]. However, this promotive effect was not consistently observed across all three Iris species. Notably, no clear dose–response relationship was detected within the concentration range applied, suggesting that the selected concentrations may have been insufficient to elicit such a gradient effect. These results collectively underscore that cytokinin activity is highly context-dependent, varying not only with developmental context but also potentially with the tested dosage[30]. In contrast, PC, a triazole-type gibberellin biosynthesis inhibitor[34], is known for suppressing apical dominance and promoting lateral bud formation[27]. However, in wetland iris, PC exhibited limited direct effects on bud germination. The response appeared to be species-specific, with indirect or delayed effects possibly mediated through altered hormonal balances. These differences suggest that the regulatory roles of PC in bud development may be more complex and context-dependent in Iris than in other plant systems.
Vegetative growth in wetland iris, primarily represented by rhizome diameter and weight, and plant height as well, also responded differentially to PGR treatments. Previous studies have demonstrated that 6-BA can enhance plant vegetative growth[35,36]. In this study, 6-BA significantly inhibited rhizome enlargement in both I. ensata var. variegata and I. sanguinea, and significantly reduced biomass accumulation only in I. sanguinea (Fig. 1), suggesting a genotype-specific response. Plant height, conversely, was generally suppressed by 6-BA application (Fig. 2), which may be attributed to resource reallocation toward underground organs. PC primarily inhibited shoot elongation in a concentration-dependent manner[37], though the effect varied by species. No significant promotive effect on final plant height was observed in I. ensata at either concentration (Fig. 2). In I. ensata var. variegata and I. sanguinea, PC application resulted in clear growth suppression (Fig. 2c–e). The inhibitory effect in I. ensata var. variegata appeared after July, whereas it persisted throughout the growth period in I. sanguinea. These findings, together with the results for 6-BA, suggest that the regulatory effects of these PGRs on vegetative growth are species-dependent and are also influenced by growth stage and dosage.
Flowering responses to PGRs also varied significantly among species. The 6-BA is known to promote cell division and differentiation by regulating hormonal balance in plants, thereby increasing the number of flowers[38,39]. In this study, the 2,000 mg/L 6-BA treatment significantly suppressed the number of flowers per plant in I. sanguinea, while a lower concentration showed a non-significant promotive effect. A similar trend was observed for PC, with high and low concentrations exerting inhibitory and promotive effects, respectively, although neither was statistically significant. These responses likely reflect species-specific hormonal sensitivities and endogenous GA levels. As an inhibitor of GA biosynthesis, PC may lead plants allocating more resources to reproductive growth, such as flowering[40]. However, excessively high concentrations of PC may overly inhibit GA synthesis, thereby negatively affecting flowering. The similar concentration-specific effects have been observed in Impatiens walleriana, where 100 mg/L PC promoted flowering, but 200–300 mg/L significantly suppressed floral development due to excessive growth retardation[41].
Taken together, the effects of PGRs on bud germination, vegetative growth, and flowering were highly dependent on species, concentration, and developmental stage (Fig. 4). For I. ensata, while all treatments contributed to an increase in bud number, a significant inhibitory effect in flower stalk height was observed with the 500 mg/L PC treatment. It is also noteworthy that the 500 mg/L PC treatment promoted an increase in flower number, although this effect was not significant. In I. ensata var. variegata, 2,000 mg/L 6-BA significantly promoted bud formation. In contrast, both 6-BA (1,000 and 2,000 mg/L), and PC treatments (500 and 1,000 mg/L) significantly inhibited the rhizome enlargement. Regarding plant height, a significant inhibitory effect from both 6-BA (2,000 mg/L) and PC treatments (500 and 1,000 mg/L) was observed from July onward and continued throughout the observation period in Sep. In I. sanguinea, a significant inhibitory effect on rhizome enlargement was observed for all concentrations of 6-BA (1,000 and 2,000 mg/L), and PC (500 and 1,000 mg/L), with 1,000 mg/L 6-BA and 500 mg/L PC being particularly effective in reducing biomass accumulation. Additionally, 2,000 mg/L 6-BA significantly reduced the number of flowers. These findings highlight the importance of species-specific PGR responses and the need for tailored application strategies to maximize desirable traits.
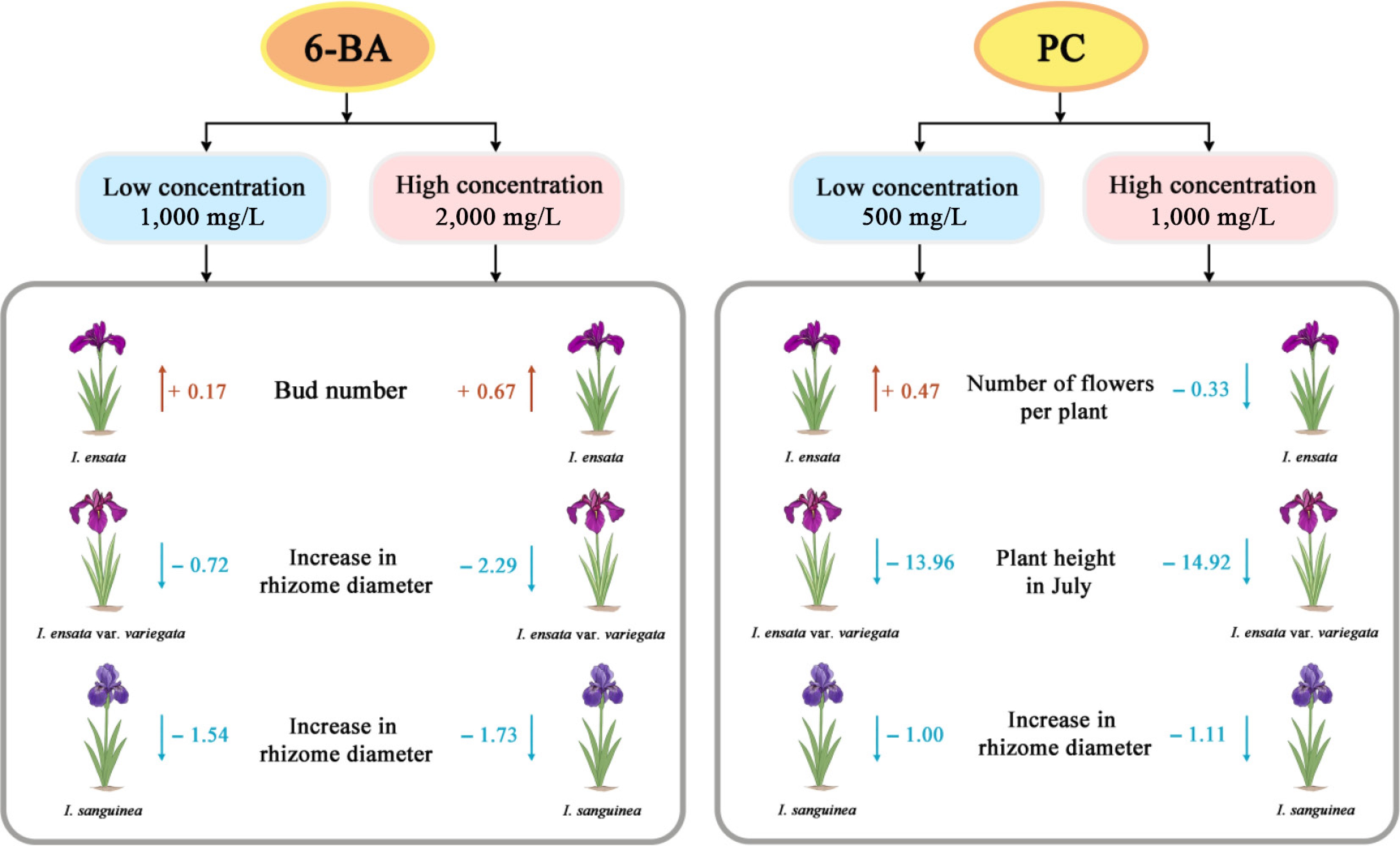
Figure 4.
Changes in key growth parameters of three Iris species (I. ensata, I. ensata var. variegata, and I. sanguinea) in response to plant growth regulator treatments. The most significant indicators identified from the dataset are presented. Treatments included two concentrations of 6-BA (1,000 and 2,000 mg/L), and PC (500 and 1,000 mg/L). Values represent the changes relative to the control.
The extensive use of 6-BA and PC in other horticultural crops further supports their potential in Iris cultivation. For instance, 6-BA has been used to promote tiller bud sprouting in rice[32], lateral bud outgrowth in jatropha (Jatropha curcas)[33], and flowering in orchids[13]. Regarding PC, the previous findings in other Iris species indicated that 700 mg/L PC was the most effective in promoting lateral bud formation. This informed the present experimental selection of a similar or broader concentration range to ensure that potential effects could be captured[27]. PC is widely applied in rice, vegetables, and legumes due to its low toxicity and short persistence[42−46], effectively reducing vegetative vigor in pear trees[47,48], and in peppers, PC treatments at 25–50 mg/L are used to control seedling elongation, whereas 100 mg/L PC treatments can significantly reduce yield[49]. These examples underscore the importance of precise PGR regulation for optimizing plant development.
Application prospects of PGRs in the cultivation and breeding of wetland iris
-
The differential responses of I. ensata, I. ensata var. variegata, and I. sanguinea to 6-BA and PC observed in this study provide a valuable basis for optimizing the cultivation and breeding strategies for wetland irises. Rather than a one-size-fits-all approach, the results suggest that species-specific PGR application protocols are necessary to achieve targeted outcomes such as enhanced flowering, compact plant architecture, or robust vegetative propagation.
From a horticultural perspective, low concentrations of PC (e.g., 500 mg/L) appear particularly promising for promoting reproductive development in I. ensata and I. sanguinea, while higher concentrations of 6-BA may be more effective for stimulating the bud formation in I. ensata var. variegata. These findings can inform nursery and landscape practices, where goals such as synchronized flowering, space-efficient growth, or clonal propagation via rhizomes may differ. For example, in managed aquatic landscapes or constructed wetlands, the use of PC could help in controlling plant height, while simultaneously enhancing floral display. Conversely, in breeding programs aiming to rapidly expand vegetative stock, 6-BA treatments may be prioritized to accelerate rhizome proliferation.
Beyond current findings, future work should explore combined PGR strategies, including co-application of 6-BA with auxins (e.g., IAA, NAA), or low concentrations of GA3 to balance vegetative and reproductive growth. Stage-specific treatments, such as applying PC during early vegetative growth to restrict height, followed by 6-BA at floral transition stages may yield synergistic effects. Additionally, the interaction between PGRs and nutrient inputs, especially nitrogen and phosphorus, should be systematically examined to develop the integrated management regimes that enhance Iris performance in both production and landscape settings.
Finally, identifying and testing alternative PGRs beyond 6-BA and PC could further optimize Iris cultivation. Brassinosteroids, strigolactones, or newer-generation growth regulators may offer distinct advantages for traits like stress tolerance or post-harvest longevity. Overall, the strategic application of PGRs represents a promising and practical tool for elevating the ornamental value and propagation efficiency of wetland irises in diverse cultivation scenarios.
This research was funded by the general project of the Zhejiang Sci-Tech University Start-up Fund (No. 24052162-Y), the Zhejiang Provincial Department of Education (Y202456763), and the National Natural Science Foundation of China (NSFC, No. 31901352).
-
The authors confirm contribution to the paper as follows: study conception and design: Li D, Zhou H; data collection, analysis and interpretation of results: Cui Y, Shen Y, Shao L, Wei J, Qiu S, Qian Y; draft manuscript preparation: Cui Y, Li D; review of the manuscript: Li D, Xia Y, Zhang J. All authors reviewed the results and approved the final version of the manuscript.
-
All data generated or analyzed during this study are included in this published article.
-
The authors declare that they have no conflict of interest.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Cui Y, Shen Y, Shao L, Wei J, Qiu S, et al. 2026. Effects of different plant growth regulators on the cultivation of wetland irises. Technology in Horticulture 6: e005 doi: 10.48130/tihort-0025-0042
Effects of different plant growth regulators on the cultivation of wetland irises
- Received: 30 June 2025
- Revised: 23 November 2025
- Accepted: 02 December 2025
- Published online: 04 March 2026
Abstract: Wetland irises are highly valued for their vibrant flowers and strong adaptability, making them important ornamental plants with broad applications in landscaping. However, their cultivation is often constrained by inefficient propagation. The application of plant growth regulators (PGRs) in wetland irises remains limited. This study systematically examined the effects of different concentrations of 6-benzylaminopurine (6-BA; 1,000 and 2,000 mg/L), and prohexadione-Ca (PC; 500 and 1,000 mg/L) on key growth parameters of three wetland iris species: Iris ensata, Iris ensata var. variegata, and Iris sanguinea. Specifically, the study focuses on evaluating the impacts of these PGRs on bud number, plant height, rhizome diameter increment, rhizome weight, and flowering performance. Quantitative analyses of the physiological and morphological responses of these species to PGR treatments revealed distinct, species-specific responses to PGR treatments. Low concentrations of PC increased bud number by up to 0.75 and flower number by 0.47 per plant in I. ensata, while high concentrations of both regulators significantly reduced plant height and rhizome enlargement, particularly in Iris ensata var. variegata and I. sanguinea. These findings provide critical insights for optimizing cultivation practices of wetland irises and also contribute to both theoretical knowledge and practical guidelines for leveraging PGRs to improve the ornamental and functional traits of these plants in landscape applications.
-
Key words:
- Wetland iris /
- 6-Benzylaminopurine /
- Prohexadione-Ca /
- Bud number /
- Flowering performance












