-
Gerbera hybrida, as an exemplary perennial herbaceous species within the Asteraceae family, has gained global popularity for its large inflorescences, vibrant color spectrum, and extended blooming period, earning its status among the world's top five commercial cut flowers. Beyond its economic significance, the species' unique transfloral morphology renders it an invaluable model for studying complex inflorescence development and evolution[1,2]. However, the expanding cultivation scale has exacerbated disease challenges, particularly root rot, which has emerged as a critical constraint to its commercial development[3].
G. hybrida root rot arises from single or combined infections by diverse soil-borne pathogens, with documented causal agents including Fusarium[4−6], Rhizoctonia[7], and Phytophthora[8]. Pathogen isolation studies in Yunnan and Fujian provinces (China), identified P. cryptogea as the primary pathogen, while other isolates, such as Fusarium solani, primarily accelerate disease progression rather than initiate independent infections[9,10].
Chemical fungicide drenching remains the predominant control strategy, though no highly effective agents have been identified[11]. Prolonged application has yielded limited efficacy while raising concerns regarding pesticide residues and pathogen resistance. Compared to chemical approaches, biofungicides demonstrate superior environmental compatibility. Hydroponic and pot experiments revealed inhibitory effects of Bacillus subtilis, Trichoderma spp., and Actinomycetes against root rot pathogens[12]. Nevertheless, field application outcomes remain inconsistent due to environmental sensitivity. Extensive evidence has been reported on the biocontrol potential of Piriformospora indica against a wide range of pathogens. Though P. indica does not exhibit direct antagonistic activity against P. cryptogea, its inoculation significantly mitigates disease symptoms in host plants, suggesting the involvement of indirect defense mechanisms that warrant further investigation[13].
Given these challenges, breeding resistant cultivars represents the most direct and economical control strategy. Comprehensive evaluation of G. hybrida germplasm for root rot resistance, coupled with mechanistic investigations into disease tolerance, holds critical importance. This study conducted systematic resistance assessments across 100 G. hybrida accessions, classifying them into six resistance tiers as used in other studies investigating resistance to P. cryptogea: highly resistant, resistant, moderately resistant, moderately susceptible, susceptible, and highly susceptible[14,15]. Validation experiments comparing phenotypic responses of highly resistant vs highly susceptible accessions at sequential post-inoculation timepoints with P. cryptogea were subsequently performed[16].
-
A total of 100 G. hybrida accessions, introduced as commercial cultivars by the Flower Research Institute of the Yunnan Academy of Agricultural Sciences, were evaluated (Table 1, Supplementary Table S1). Accessions 1–77 originated from the Netherlands, while accessions 78–100 were sourced from Fujian Province, China (Fig. 1). For each accession, 20 plants constituted one biological replicate with three replicates, and all accessions were grown in the greenhouse at 25 °C under a 16 h light/8 h dark photoperiod using a randomized block design.
Table 1. Evaluation of the natural incidence and resistance of 100 G. hybrida germplasm.
No. Cultivars Origin Incidence rate (%) Disease index Resistance level 47 Mini Orange BC the Netherlands 91.7 85 HS 63 Micro Pink BC the Netherlands 90.3 81.98 HS 51 Mini Red BC the Netherlands 83.3 81.67 HS 92 JQ-24 Fujian, China 83.3 79.58 S 9 MAXI hot pink the Netherlands 71.7 60.42 S 79 JQ-25 Fujian, China 65 60.42 S 85 JQ-21 Fujian, China 61.7 58.75 MS 20 Midi Red the Netherlands 61.7 50.83 MS 12 Giant Orange the Netherlands 50 48.75 MR 27 Midi Eyecatcher Purple BC the Netherlands 46.7 40.42 MR 36 Midi Light Pink BC the Netherlands 50 40 R 53 Mini Yellow BC the Netherlands 41.7 40 R 21 Midi Yellow the Netherlands 21.7 20.42 R 32 Midi Pink BC the Netherlands 21.7 20.42 R 24 Midi Red BC the Netherlands 13.3 13.33 HR 72 Maxi Yellow BC the Netherlands 13.3 11.67 HR 30 Midi Eyecatcher Twilight BC the Netherlands 8.3 8.33 HR 
Figure 1.
Morphological characteristics of 100 G. hybrida germplasm in full bloom. The top left corner label corresponds to the variety number; scale bar is 1 cm.
Natural disease incidence was assessed during peak root rot outbreaks (the time point during the investigation period when the disease incidence or disease index shows the most rapid increase). Due to the delayed appearance of external symptoms in G. hybrida, the hidden progression of root rot, and its strong dependence on environmental conditions, disease assessments were conducted at 0, 2, 4, and 6 d after inoculation based on experimental observations and statistical analyses. Disease severity was quantified using an integrated evaluation of incidence rate and disease index. The disease grading system was established based on the degree of leaf wilting observed in infected G. hybrida plants, thereby reflecting the severity of disease symptoms in individual plants. Disease incidence represents the proportion of infected leaves, indicating whether or not the plants are affected by the disease. The disease grading criteria were as follows:
Grade 0: No symptoms
Grade 1: ≤ 50% leaves wilted
Grade 2: > 50% leaves wilted but not fully desiccated
Grade 3: All leaves wilted, partial leaf desiccation observed
Grade 4: Complete plant death.
Incidence rate (%) was calculated as:
$ \text{Incidence rate}\;({\text{%}})=\dfrac{\text{Number of infected leaves}}{\text{Total leaves surveyed}}\times 100 $ The disease index (DI) was determined by:
$ \text{DI}=\dfrac{\sum (\text{Number of plants per grade}\times \text{Grade value})}{\text{Total plants surveyed}\times \text{Highest disease grade observed}}\times 100 $ Accessions were classified into six resistance tiers based on DI values:
Highly Resistant (HR): DI ≤ 20
Resistant (R): 20 < DI ≤ 40
Moderately Resistant (MR): 40 < DI ≤ 50
Moderately Susceptible (MS): 50 < DI ≤ 60
Susceptible (S): 60 < DI ≤ 80
Highly Susceptible (HS): DI > 80.
Pathogen isolation and identification
-
Gerbera plants exhibiting root rot symptoms were collected from the Jiuxi Gerbera Germplasm Resource Nursery in Yuxi City, Yunnan Province, China. The roots were washed thoroughly with clean water, and tissue samples were excised from the junction between diseased and healthy areas for pathogen isolation. These samples were disinfected in 75% ethanol for 90 s, and rinsed three times with sterile distilled water. Segments were transferred to tomato juice agar (TJA) supplemented with rifampicin (50 μg/mL) and streptomycin (100 μg/mL), then incubated at 28 °C. Emerging hyphae were subcultured from colony margins onto fresh media for purification. Morphological identification via microscopy was corroborated by PCR amplification using P. cryptogea specific primers (Cryp1: 5'-CCTGGTGGCCGACAAGGTCC-3'. Cryp2: 5'-GTTGAGTTCGCGCCGTACC-3'). PCR protocol consisted of two phases: phase 1 included an initial step at 94 °C for 4 min, followed by 10 cycles of denaturation at 94 °C for 15 s, annealing at variable temperature for 30 s, and extension at 72 °C for 20 s. In the first cycle, the annealing temperature was set to 63 °C, and at each of the subsequent cycles, the annealing temperature was decreased by 0.5 °C. Phase 2 consisted of 25 cycles of 94 °C for 15 s, 58 °C for 30 s, and 72 °C for 20 s[17].
Resistance validation
-
The resistance validation experiment was conducted under natural outdoor conditions at the Jiuxi Gerbera Germplasm Resource Nursery, with an ambient temperature of 25 °C. The inoculated strain was P. cryptogea, which has been previously isolated and identified as described in the above section. It was inoculated into PDA medium and cultured it in the dark at 28 °C for 5–7 d. Mycelial plugs (0.5 cm diameter) from colony margins were transferred to potato dextrose broth (PDB) and shaken (120 rpm) for 48 h. Hyphae were removed via filtration to obtain spore suspensions (10,000 spores/mL).
Two cultivars exhibiting the highest and lowest disease index and incidence in the resistance evaluation were selected as experimental materials, and all were uniform 5-month-old seedlings. The spore suspension was poured along the root until the soil was sufficiently wet. The control group was irrigated with the same amount of water as the healthy control group. Each treatment had 30 pots of seedlings and was repeated three times. Three plants were randomly selected from each replicate at 0, 2, 4, and 6 d after inoculation to determine the growth-related indicators.
Statistical analyses
-
GraphPad Prism (version v9.0.0) (GraphPad Software, San Diego, CA, USA) was used for the analysis of variance (ANOVA) of experimental data. Significant differences between the treatments were evaluated using two-way ANOVA followed by Tukey's Honestly Significant Difference (HSD) test at the 5% probability level, and the two factors for the two-way ANOVA were the inoculation time with P. cryptogea and the G. hybrida cultivar.
-
The 100 G. hybrida accessions showed typical symptoms of root rot disease, including leaf chlorosis, wilting, and root necrosis, while the incidence rates (IR) and disease index (DI) were different among cultivars (Table 1). Midi Eyecatcher Twilight BC exhibited the lowest IR of 8.3% and DI of 8.33, and was classified into a highly resistant (HR) cultivar. Mini Orange BC showed the highest IR (91.7%), and DI (85) was classified into a highly susceptible (HS) cultivar (Table 1).
Among the 100 Gerbera germplasm resources, 39 cultivars were classified as resistant (R) with disease indices of 20.42 to 40, 13 cultivars were classified as moderately resistant (MR) with disease indices of 40.42 to 48.75, and three cultivars were classified as highly resistant (HR), with disease indices of 8.33 to 13.33. Ten cultivars were classified as moderately susceptible (MS), with disease indices of 50.83 to 58.75, 32 cultivars were classified as susceptible (S), with disease indices of 60.42 to 79.58, and three cultivars were classified as highly susceptible (HS), with disease indices of 81.67 to 85 (Table 1, Supplementary Table S1). With the increase in DI, the degree of wilting in Gerbera plants intensified, the number of wilted leaves increased, while total roots decreased. Except for four MR cultivars from Fujian (JQ-9, JQ-10, JQ-17, and JQ-18), all resistant Gerbera germplasm originated from the Netherlands. Overall, the proportion of resistant germplasm introduced from the Netherlands (66%, 51/77), was much higher than that from Fujian (17%, 4/23).
Growth performance of highly resistant and susceptible G. hybrida accessions to P. cryptogea inoculation
-
Previous experiments showed that P. cryptogea was the main pathogen of root rot of G. hybrida. Therefore, highly-resistant and highly susceptible G. hybrida varieties were selected for inoculation experiments. The highly resistant variety Midi Eyecatcher Twilight BC showed no phenotypic differences compared to the water-treated control group during the first four days after inoculation (DAI). On 6 DAI, 74% of inoculated plants maintained healthy growth, while 26% plants exhibited mild symptoms (≤ 50% leaf yellowing), consistent with the HR classification (Fig. 2). In contrast, the highly susceptible cultivar Mini Orange BC displayed rapid symptom progression: 40% of inoculated plants showed disease signs on 2 DAI, escalating to 55% incidence on 4 DAI, including 11% with complete plant death. On 6 DAI, 47% of inoculated Mini Orange BC plants exhibited severe symptoms (Grades 3–4), characterized by full-leaf yellowing and desiccation, and 24% plants were dead (Fig. 2).
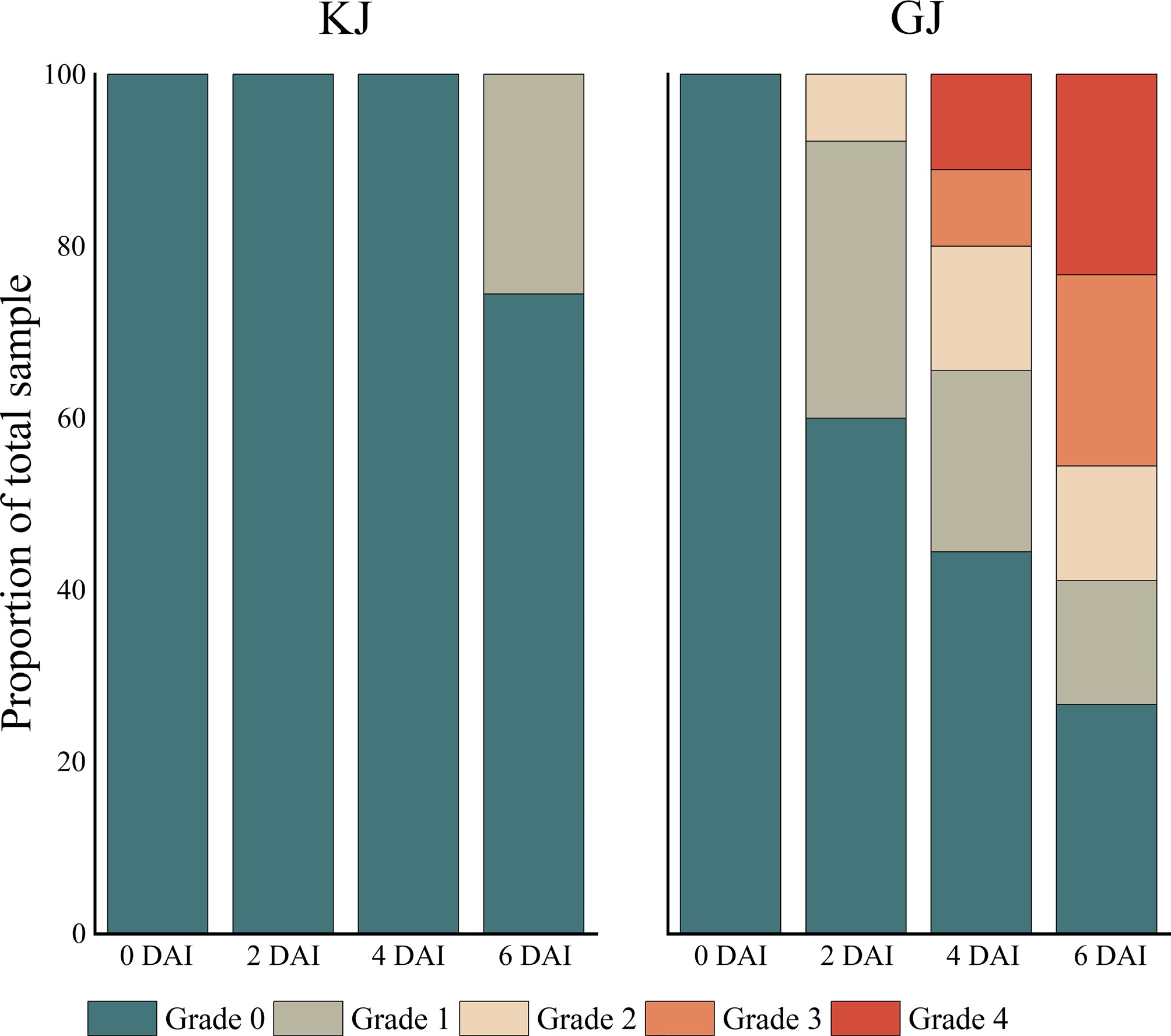
Figure 2.
Disease grading of highly resistant and susceptible G. hybrida inoculated by P. cryptogea. KJ indicates Midi Eyecatcher Twilight BC pots inoculated with P. cryptogea spore suspension liquid, and GJ indicates Mini Orange BC pots inoculated with P. cryptogea spore suspension liquid. Grade 0 indicates no symptoms; Grade 1 indicates ≤ 50% leaves wilted; Grade 2 indicates > 50% leaves wilted but not fully desiccated; Grade 3 indicates all leaves wilted, partial leaf desiccation observed; Grade 4 indicates complete plant death.
Aboveground biomass in different cultivars after P. cryptogea inoculation
-
After P. cryptogea inoculation, the highly susceptible variety Mini Orange BC exhibited progressive deterioration of aboveground growth (Fig. 3). On 2 DAI, partial leaf yellowing had already appeared. the leaf length, number of leaves, and fresh weight of above-ground parts showed no significant differences compared with the water-treated for the highly resistant variety Midi Eyecatcher Twilight BC. A slight reduction in shoot fresh weight was observed on 2 DAI (Fig. 4c). On 4 DAI and 6 DAI, new leaves grew rarely, and the existing leaves wilted gradually, resulting in a reduction in leaves number and shoot fresh weight (Fig. 4).

Figure 3.
Phenotypes of above-ground parts of highly resistant and highly susceptible G. hybrida after P. cryptogea inoculation. KJ indicates Midi Eyecatcher Twilight BC pots inoculated with P. cryptogea spore suspension liquid; CK1 indicates Midi Eyecatcher Twilight BC pots inoculated with equal amount of water; GJ indicates Mini Orange BC pots inoculated with P. cryptogea spore suspension liquid; CK2 indicates Mini Orange BC pots inoculated with equal amounts of water. 0d, 2d, 4d, 6d denote day 0, 2, 4, and 6 after inoculation with P. cryptogea. Scale bar is 1 dm.
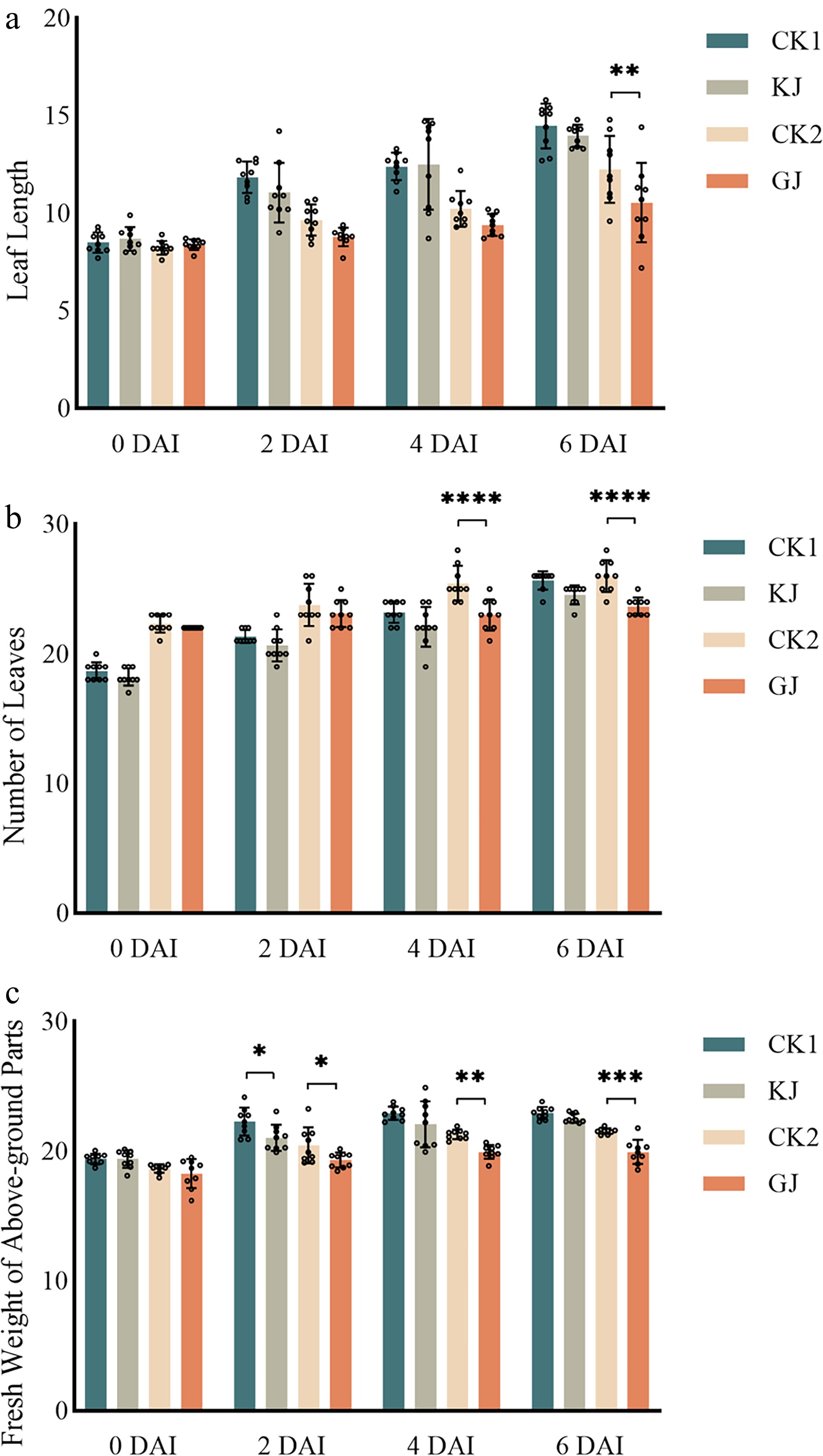
Figure 4.
Aboveground growth indexes of highly resistant and highly susceptible G. hybrida after P. cryptogea inoculation. (a) Leaf length of highly resistant and highly susceptible G. hybrida after P. cryptogea inoculation. (b) Numbers of leaves of highly resistant and highly susceptible G. hybrida after P. cryptogea inoculation. (c) Fresh weight of above-ground parts of highly resistant and highly susceptible G. hybrida after P. cryptogea inoculation. KJ indicates Midi Eyecatcher Twilight BC pots inoculated with P. cryptogea spore suspension liquid; CK1 indicates Midi Eyecatcher Twilight BC pots inoculated with equal amount of water; GJ indicates Mini Orange BC pots inoculated with P. cryptogea spore suspension liquid; CK2 indicates Mini Orange BC pots inoculated with equal amounts of water. * p < 0.05, ** p < 0.01, *** p <0.001, **** p < 0.0001. Treatments that were not significantly different from the control are not labelled on the graph.
Root performance of different cultivars after P. cryptogea inoculation
-
Difference of root response to P. cryptogea were observed among cultivars. In HS cultivar Mini Orange BC, dark lesions appeared at the stem base on 2 DAI, and then spread to root tips progressively (Fig. 5). However, no obvious symptoms were found in the roots of HR cultivar Midi Eyecatcher Twilight BC.

Figure 5.
Root phenotypes of highly resistant and highly susceptible G. hybrida after P. cryptogea inoculation. KJ indicates Midi Eyecatcher Twilight BC pots inoculated with P. cryptogea spore suspension liquid; CK1 indicates Midi Eyecatcher Twilight BC pots inoculated with equal amount of water; GJ indicates Mini Orange BC pots inoculated with P. cryptogea spore suspension liquid; CK2 indicates Mini Orange BC pots inoculated with equal amounts of water. 0d, 2d, 4d, 6d denote day 0, 2, 4, and 6 after inoculation with P. cryptogea. Scale bars are 1 cm.
The root length of all treatments increased over time (Fig. 6a). The root length of Mini Orange BC (HS) was significantly lower than the control, whereas no significant difference was observed between the Midi Eyecatcher Twilight BC (HR) and the water-treated for it. The root number of Mini Orange BC (HR) decreased by half compared with the control on 4 DAI, and was reduced by two-thirds on 6 DAI (Fig. 6b). The root fresh weight showed a pattern similar to that of root length and root number. Root growth stagnated from 4 DAI, and roots began to wilt gradually, resulting in a decrease in root fresh weight (Fig. 6c).
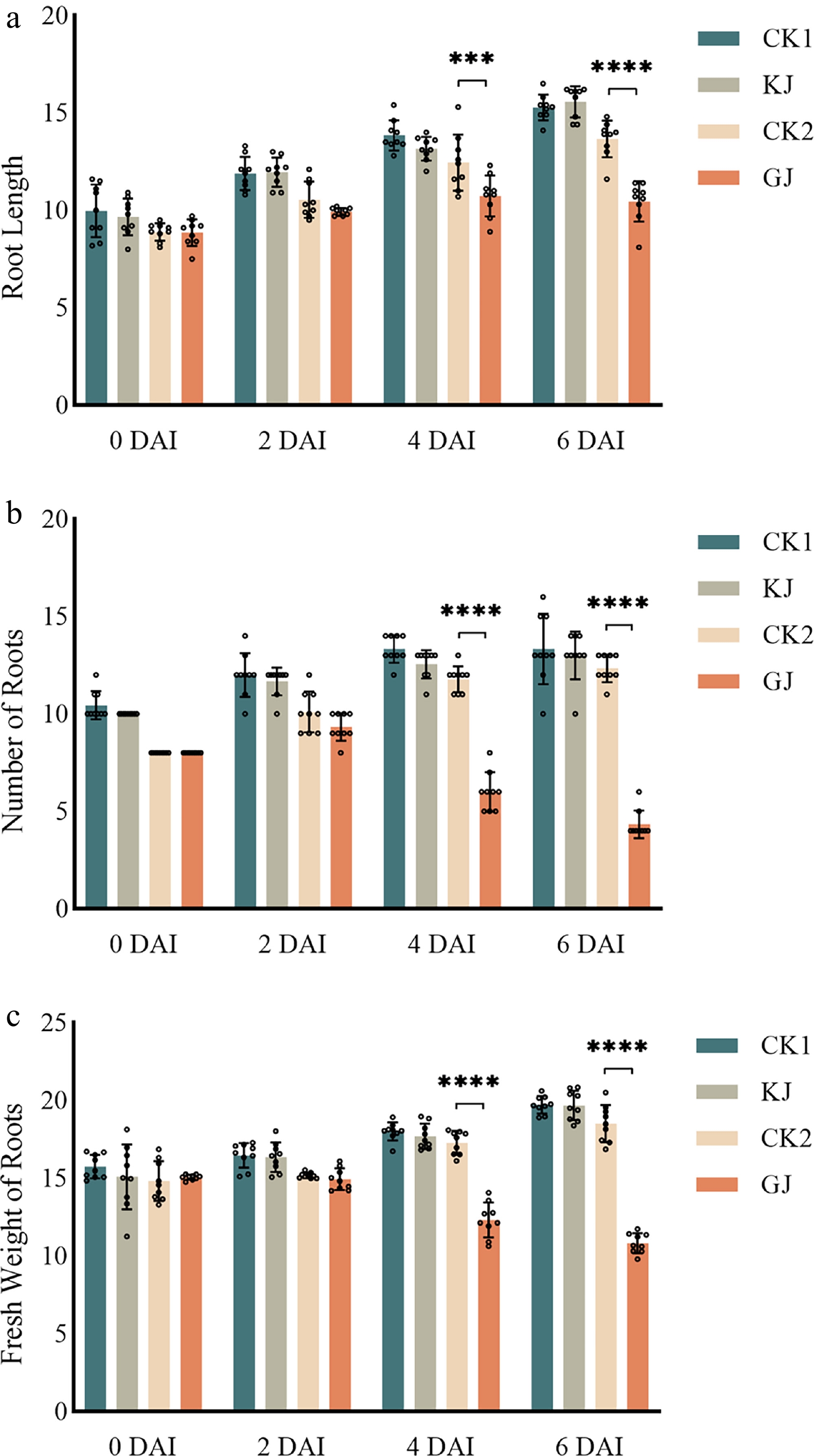
Figure 6.
Root growth indexes of highly resistant and highly susceptible G. hybrida after P. cryptogea inoculation. (a) Root length of highly resistant and highly susceptible G. hybrida after P. cryptogea inoculation. (b) Numbers of roots of highly resistant and highly susceptible G. hybrida after P. cryptogea inoculation. (c) Fresh weight of roots of highly resistant and highly susceptible G. hybrida after P. cryptogea inoculation. KJ indicates Midi Eyecatcher Twilight BC pots inoculated with P. cryptogea spore suspension liquid; CK1 indicates Midi Eyecatcher Twilight BC pots inoculated with equal amount of water; GJ indicates Mini Orange BC pots inoculated with P. cryptogea spore suspension liquid; CK2 indicates Mini Orange BC pots inoculated with equal amounts of water. * p < 0.05, ** p < 0.01, *** p < 0.001, **** p < 0.0001. Treatments that were not significantly different from the control are not labelled on the graphs.
Collectively, these results demonstrate that P. cryptogea infection severely impaired root growth in Mini Orange BC (HS), leading to shorter roots, fewer root numbers, and reduced root fresh weight, whereas Midi Eyecatcher Twilight BC (HR) maintained relatively normal root development throughout the infection period.
-
In this study, a transnational germplasm introduction strategy was employed to systematically evaluate the resistance of 100 G. hybrida accessions originating from different regions, including the Netherlands and Fujian Province, China. A total of 55 accessions exhibiting significant resistance were successfully identified, among which Midi Red BC, Maxi Yellow BC, and Midi Eyecatcher Twilight BC showed particularly strong resistance. These elite resistant accessions can serve as important parental materials for direct use in breeding programs, thereby accelerating the development of disease-resistant cultivars and reducing reliance on chemical control measures in commercial production. The germplasm collection established in this study encompasses a complete resistance–susceptibility continuum, with disease index values ranging from 8.33 to 85 (Table 1), providing an ideal platform for in-depth investigation of the molecular mechanisms underlying G. hybrida resistance to P. cryptogea. Notably, the proportion of resistant accessions among germplasm introduced from the Netherlands (66%) was significantly higher than that observed among accessions originating from Fujian (17%).
Root rot is a general term for soil-borne diseases characterized by progressive decay and necrosis of subterranean plant tissues[18]. The pathogenesis follows a defined sequence: initial pathogen invasion compromises root function, impairing water and nutrient acquisition, which subsequently triggers systemic growth inhibition manifested as leaf wilting, chlorosis, and ultimately plant mortality. The disease etiology involves diverse pathogens, including fungi (notably Fusarium oxysporum and P. cryptogea), bacteria, and nematodes[19,20]. P. cryptogea was by far the most common species of plant pathogen found on annuals and herbaceous perennials[21].
In 2017, a new pathogen was identified on Gerbera in Izmir Province in Turkey. This pathogen caused significant losses of Gerbera cultivars. Based on molecular and morphological data, the pathogen was identified as P. cryptogea. In subsequent pathogenicity tests, blackish-brown discoloration and necrosis were observed in the roots and crowns of all inoculated plants[22]. In California, USA, field-grown lettuce (Lactuca sativa)[23] and shallot (Allium cepa L. var. aggregatum)[24] also developed root rot caused by P. cryptogea. When healthy plants were inoculated with P. cryptogea, they exhibited symptoms within seven or 10 d, including leaf chlorosis, wilting, and root necrosis, which closely matched those observed in the field. In this study, Gerbera also exhibited such symptoms after infection with P. cryptogea (Fig. 7), further demonstrates the broad host range of P. cryptogea among herbaceous plants.

Figure 7.
Phenotypes of different resistance levels of G. hybrida after infection. (a) Growth performance of aerial part of diseased G. hybrida; (b) Root growth performance of G. hybrida; (c) Stem bases growth performance of diseased G. hybrida. Scale bar is 1 cm.
Breeding resistant cultivars remains the most effective control strategy. Previous work by Li et al. established 'Linglong' as a key resistant material screened from 13 cultivars[25]. In the present study, three G. hybrida cultivars with highly resistance (HR), Midi Red BC, Maxi Yellow BC, and Midi Eyecatcher Twilight BC were successfully identified. These resistant cultivars can be directly utilized in breeding programs to accelerate the development of root rot–resistant varieties. This finding provides important insights for domestic gerbera resistance breeding, indicating that targeted introduction of germplasm from regions enriched in resistant resources represents an effective strategy for rapidly acquiring superior resistance sources. In addition, future studies should focus on resistance gene mining, elucidation of defense signaling pathways, analysis of root exudates, and, in particular, investigations into the physiological and biochemical mechanisms underlying root defense responses.
Moreover, in this study, resistance evaluation was primarily conducted under controlled conditions targeting a single pathogen (P. cryptogea), whereas field root rot is usually caused by one or more soil-borne pathogens acting alone or in combination. Subsequent studies must assess the resistance spectra of these elite accessions against other major pathogens such as Fusarium and Rhizoctonia species. The molecular mechanisms underlying resistance in these accessions remain to be elucidated. Furthermore, experimental validation focused on extreme HR/HS contrasts—future work should incorporate materials across the resistance gradient to comprehensively characterize the resistance continuum.
-
This study establishes a framework for developing P. cryptogea-resistant G. hybrida cultivars. We evaluated the resistance of 100 Gerbera cultivars to P. cryptogea. The HR accession Midi Eyecatcher Twilight BC emerged as a prime candidate for both commercial propagation and mechanistic studies, while the susceptibility of Mini Orange BC provides a model for dissecting host-pathogen dynamics. These insights not only address immediate agricultural challenges but also enrich our understanding of Asteraceae-pathogen interactions—a critical step toward sustainable floriculture in the face of escalating soil-borne disease pressures.
-
The authors confirm their contribution to the article as follows: contributed to the conception and design of the experiments, performed the experiments, collected and analyzed the data, and drafted the manuscript: Zhao J, Liu B; provided experimental guidance and technical support throughout the study: Ruan J, Yang C; contributed to the overall research design and supervision, provided critical revision of the manuscript, and secured funding for the project: Li S, Lu Z. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
-
This work was supported by Yunnan Academy of Agricultural Sciences Pre-research Project (2023KYZX-07), and Yunnan Xingdian Talents − Special Selection Project for High-level Scientific and Technological Talents and Innovation Teams-Team Specific Project, Xingdian Talent Support Project (CYRC2020004).
-
The authors declare that they have no conflict of interest.
-
accompanies this paper online at: https://doi.org/10.48130/tihort-0025-0044.
- Supplementary Table S1 Evaluation of the natural incidence and resistance of 100 G. hybrida germplasm.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Zhao J, Liu B, Ruan J, Yang C, Li S, et al. 2026. Geographic disparity in root rot resistance of 100 Gerbera hybrida accessions and their horticultural merit. Technology in Horticulture 6: e006 doi: 10.48130/tihort-0025-0044
Geographic disparity in root rot resistance of 100 Gerbera hybrida accessions and their horticultural merit
- Received: 29 June 2025
- Revised: 07 November 2025
- Accepted: 02 December 2025
- Published online: 19 March 2026
Abstract: Root rot is an extremely severe disease affecting Gerbera hybrida, caused by various soil-borne pathogens through single or co-infections. Among these, Phytophthora cryptogea has been identified as the primary causal agent according to pathogens isolated from reported regions such as Yunnan and Fujian provinces. Breeding resistant varieties to P. cryptogea is considered one of the economic strategies. This study evaluated and classified the root rot resistance of 100 G. hybrida germplasm resources for the first time. The present results indicated that 55 of these 100 germplasm resources exhibited resistance to root rot, with Midi Red BC, Maxi Yellow BC, and Midi Eyecatcher Twilight BC standing out as highly resistant varieties. To validate their resistance, the highly resistant (HR) variety Midi Eyecatcher Twilight BC and the highly susceptible (HS) variety Mini Orange BC were selected for further experiments, and compared their growth characteristics of the aboveground parts and root systems at different growth stages after inoculation with P. cryptogea. The results showed that the root system of the HR variety demonstrated remarkable defensive capabilities when infected by P. cryptogea, effectively preventing the spread of the disease to the aboveground parts, thereby ensuring the healthy growth of the plant.
-
Key words:
- Disease resistance /
- Gerbera hybrida /
- Root rot /
- Phytophthora cryptogea /
- Germplasm screening













