-
Gastric cancer remains a significant global health challenge. According to GLOBOCAN 2022 data, there were over 968,000 new cases of gastric cancer worldwide (accounting for 4.9% of all cancers), with approximately 660,000 deaths (6.8% of total cancer-related mortality). Both the incidence and mortality rates of gastric cancer rank fifth among all malignancies globally[1]. Gastric cancer typically metastasizes to the liver (48%), peritoneum (32%), lungs (15%), and bones (12%)[2]. However, cervical metastasis of gastric cancer is rare in clinical practice. Here, we present a case of cervical metastasis occurring six years after radical gastrectomy for gastric cancer, which achieved PR following chemotherapy combined with immunotherapy.
-
A 70-year-old Chinese female underwent radical distal subtotal gastrectomy with Roux-en-Y gastrojejunostomy on March 20, 2018. Pathological staging according to the 8th edition of the American Joint Committee on Cancer (AJCC) staging system confirmed stage IIIA (pT2, pN3a, cM0) moderately to poorly differentiated adenocarcinoma. The Lauren classification was mixed type. Immunohistochemistry (IHC) report: Her2(1+) C-MET(++), E-cadherin(+), PD-L1 (SP142 assay: tumor cells negative; tumor-infiltrating immune cells = 5% positive), MLH1(+), PMS2(+), AFP(−), P53(+++), NY-ESO1(−), Ki67(60%+), MUC2(focal+), MUC5AC(+), MUC6(+), CD10(−), CK7(+), CK20(focal+), Villin(+++) (Fig. 1). Histopathological findings revealed tumor invasion into the muscularis propria, with negative surgical margins at both resection ends. Lymphovascular invasion and perineural invasion (PNI) were identified. Metastatic carcinoma was found in six out of 16 lymph nodes along the lesser curvature, and five out of seven lymph nodes along the greater curvature. Postoperative the patient received five cycles of adjuvant chemotherapy with the following protocol: Paclitaxel liposome 90 mg on day 1 and day 8 + S-1 50 mg orally twice daily on days 1−14. The patient tolerated the treatment well, and regular follow-up showed no evidence of recurrence.
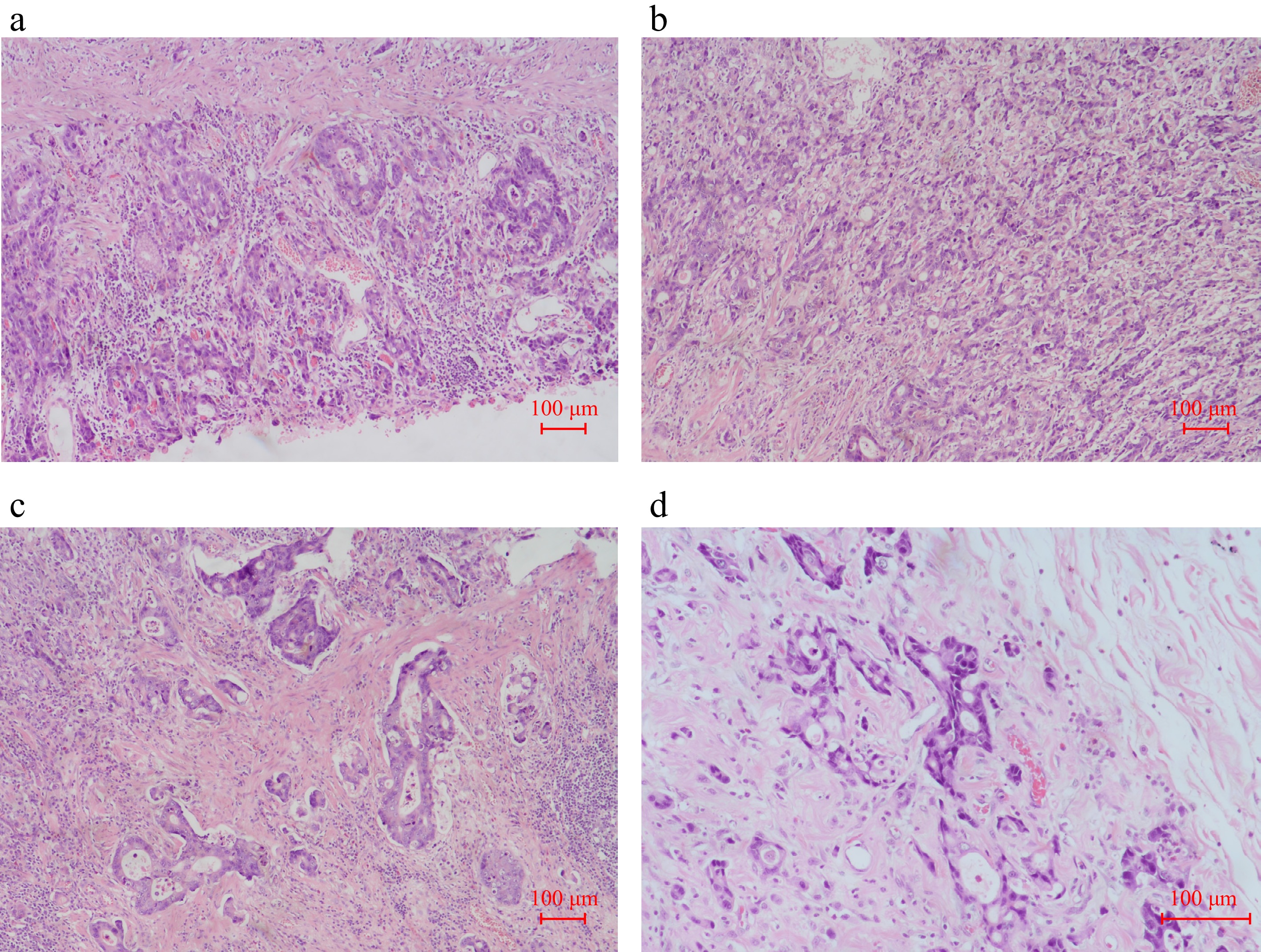
Figure 1.
Pathological images of the gastric mass from the radical distal subtotal gastrectomy with Roux-en-Y gastrojejunostomy on March 20, 2018. (a)–(c) H & E staining (× 10). (d) H & E staining (× 20).
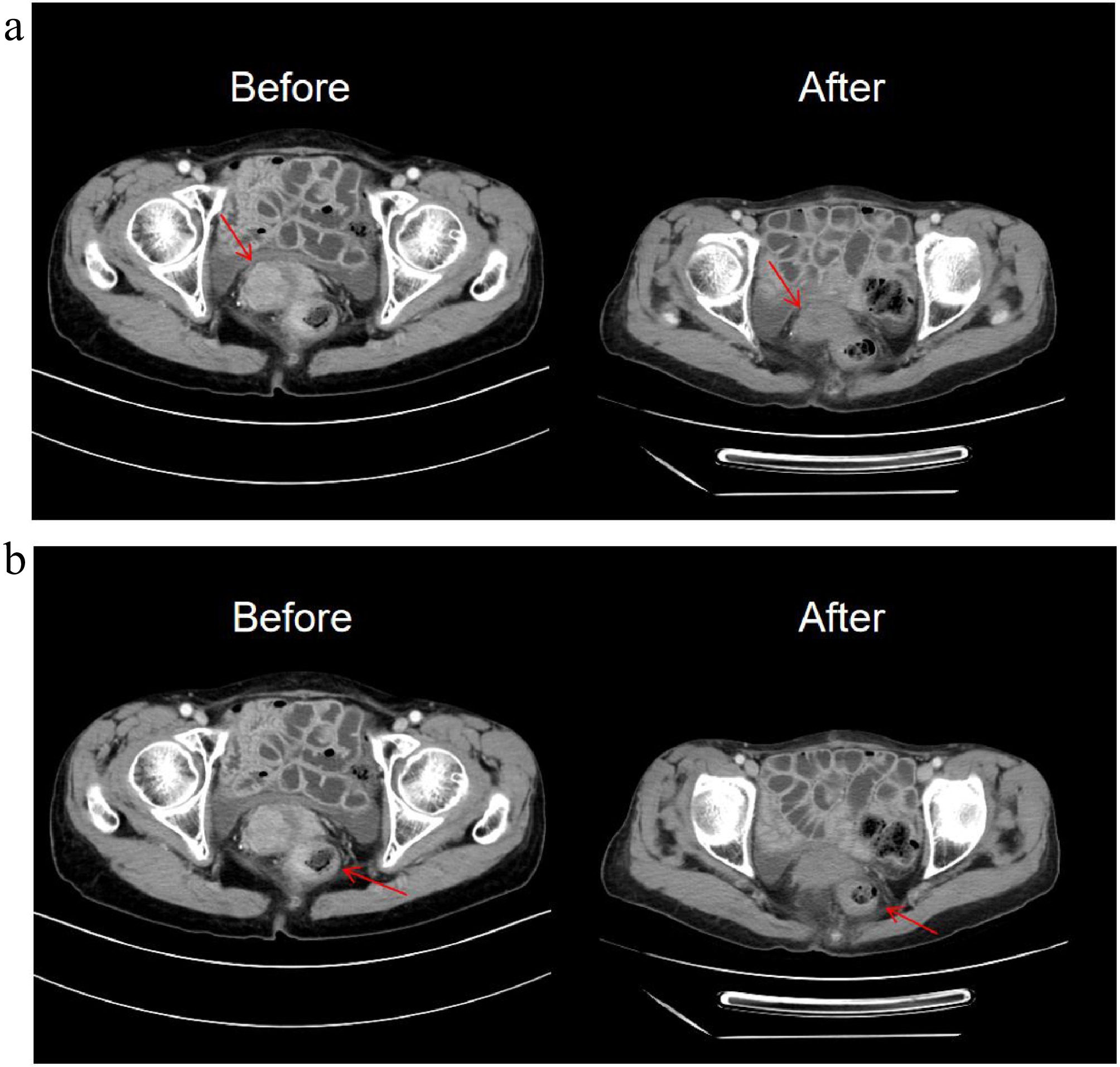
In September 2024, the patient presented with anal tenesmus and dull left lower abdominal pain persisting for six months. Contrast-enhanced CT of the chest and abdomen (2024-09-28) revealed focal rectal wall thickening with enlarged left-sided regional lymph nodes, newly identified compared to prior imaging (2022-09-29), (Fig. 2a, b). Colonoscopy (2024-09-30) indicated a suspicious rectal mass with an extrinsic compression pattern. Histopathology (rectal biopsy): poorly differentiated adenocarcinoma infiltrating the lamina propria and muscularis mucosae. Immunohistochemistry (IHC): CK-P(+), CAM5. 2(+), Syn(−), CgA(−), CD56(−), INSM1(−), SSTR2(0), p40(−), P63(−), CK5/6(−), p53(90% strong+), Ki-67(90%+), Desmin (highlighted muscularis mucosae).
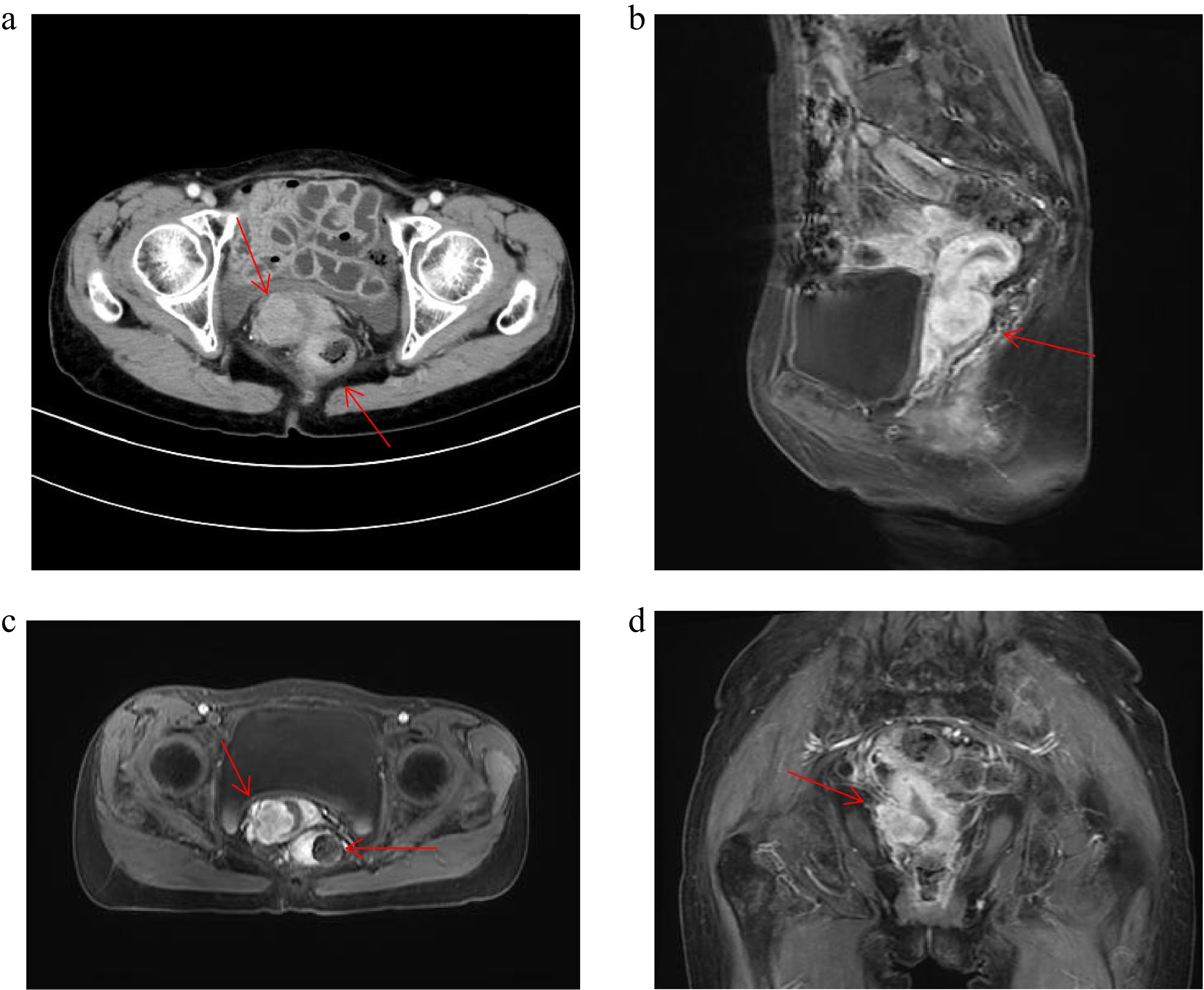
Figure 2.
Patient CT and MRI image. (a) Patient 2024.9.28 CT image. (b)–(d) Patient 2024.10.24 MRI image.
Subsequent pelvic MRI revealed a cervical mass and a rectal mass, which presented with an extrinsic compression pattern (Fig. 2c–e). The pathology report of cervical biopsy (October 25, 2024) suggested poorly differentiated adenocarcinoma. Immunohistochemistry (IHC) showed the tumor cells exhibited the following staining pattern: CK7(−), CK20(+), Villin(+++), P40(−), P16(focal+), PAX8(+), SATB2(rare+), (Fig. 3). Primary cervical adenocarcinoma typically shows diffuse strong positivity for p16 (≥ 70% of tumour cells). In cervical lesions, P16 immunohistochemical staining is often used to assist in determining the HPV infection status, whereas the patient's focal positivity is inconsistent with primary disease. PAX8 is expressed in gynecological tumours, but can also be positive in gastrointestinal metastases, combined with CK7(−) negativity, it does not support a primary cervical origin. These results, together with the patient's gastric cancer history and gastrointestinal IHC profile (CK20+, Villin+++), taking all factors into consideration, the possibility of primary cervical cancer has been ruled out.
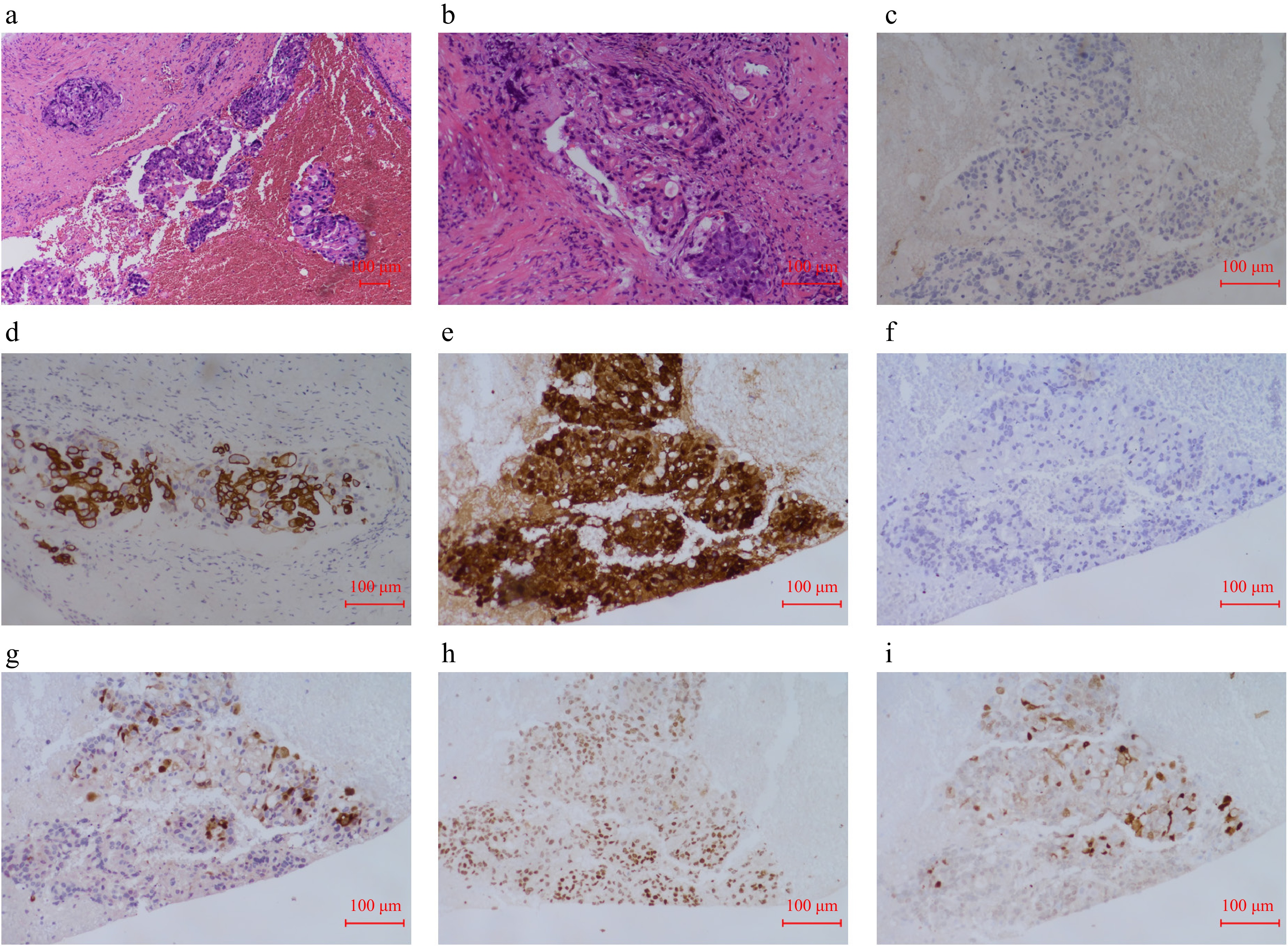
Figure 3.
Cervical pathological images. (a) H & E staining of sections from cervical mass (× 10). (b) H & E staining of sections from cervical mass (× 20). (c) Immunohistochemical CK7 pathological images of the sections from cervical mass. (d) Immunohistochemical CK20 pathological images of the sections from cervical mass. (e) Immunohistochemical Villin pathological images of the sections from cervical mass. (f) Immunohistochemical P40 pathological images of the sections from cervical mass. (g) Immunohistochemical P16 pathological images of the sections from cervical mass. (h) Immunohistochemical PAX8 pathological images of the sections from cervical mass. (i) Immunohistochemical SATB2 pathological images of the sections from cervical mass.
Based on the clinical history and immunohistochemical profile, the findings suggest a gastric primary origin for both the cervical and rectal masses. Subsequently, gastric cancer treatment-related marker testing was performed on the rectal metastasis. The findings were shown as follows: HER2 (4B5)(−), Claudin18.2(++), c-MET (SP44)(++), MLH1(+), PMS2(+), MSH2(+), MSH6(+). In Situ Hybridization: EBER(−). PD-L1 Companion Diagnostic (22C3): Tumor Proportion Score (TPS) (%): 0. Combined Positive Score (CPS): 1.
This patient the underwent chemotherapy with oxaliplatin + capecitabine (CAPOX) on October 26, 2024. CSCO Gastric Cancer Guidelines (2024): Chemotherapy-immunotherapy (chemotherapy + anti-PD-1/PD-L1) is recommended as the first-line treatment for unresectable advanced gastric cancer, regardless of PD-L1 expression status (including CPS < 5), and the patient lacks alternative targeted therapy (HER2-negative). Based on the CPS, sintilimab (anti-PD-1) was added from November 25, 2024, to April 15, 2025, completing six cycles of CAPOX plus sintilimab. During therapy, the patient developed grade 3 thrombocytopenia (occurred after cycle 6, 2025-04), and grade 2 leukopenia (occurred after cycle 2, 2024-12). Recombinant human thrombopoietin (rhTPO) and granulocyte colony-stimulating factor (G-CSF) were retrospectively administered for symptomatic treatment. The patient completed all six cycles as planned, and no serious or persistent adverse events occurred. Tumor marker CA72-4 monitoring demonstrated a declining trend, eventually normalizing (Supplementary Fig. S1). Follow-up CT imaging revealed a reduction in the cervical and rectal masses, and this patient showed alleviation of intestinal obstruction symptoms. The therapeutic response was evaluated as partial response (PR) per RECIST 1.1 criteria (Fig. 4).
-
This report describes a metachronous cervical metastasis identified six years following curative resection of gastric adenocarcinoma. While disease relapse most frequently involves locoregional sites, peritoneum, or liver, cervical involvement represents an extraordinary rarity, conferring significant complexities in both diagnosis and clinical management. The incidence of cervical metastases is only 0.3%, and the average age at diagnosis of metastatic cancer is 44 years (23–75 years)[3]. The underlying mechanism of this rare metastasis remains incompletely understood, but recent studies on the tumor microenvironment (TME) provide new insights. Peng et al. found that inflammatory cancer-associated fibroblasts (iCAFs) in the gastric cancer TME can promote epithelial-mesenchymal transition (EMT) and lipid metabolism crosstalk, enhancing the invasive and metastatic capacity of tumor cells[4]. Additionally, the crosstalk between iCAFs and myeloid cells (e.g., tumor-associated macrophages, TAMs) via ligands such as MIF and CSF1 creates a pro-metastatic niche[4]. In the present case, the patient developed cervical and rectal metastasis six years after curative gastrectomy; although CAFs were not directly detected in the metastatic foci, the rare metastatic pattern may be attributed to the pro-metastatic TME remodeling mediated by CAFs and myeloid cells, as proposed by Peng et al.[4]. This also explains why gastric cancer metastasis usually occurs in visceral organs (e.g., liver) but rarely in the cervix—specific TME components may facilitate tumor cell colonization in unusual sites[4]. For patients with a history of gastric cancer who present with gynecologic symptoms such as genital bleeding, ascites, or lower abdominal pain, the possibility of metastatic recurrence should be carefully considered.
The cervix may be involved by direct tumor extension from adjacent organs, but is rarely affected by metastatic disease. This frequently occurs in endometrial carcinoma, where the endocervical mucosa and/or stroma are infiltrated due to direct tumor spread. The cervix can also be involved by gynecological tumors originating from the ovaries, fallopian tubes, or peritoneum via transtubal and intrauterine dissemination[5]. Metastasis to the female genital tract from extragenital primary sites is exceedingly rare, and the cervix is less commonly involved compared to the uterine corpus[6]. Kumar & Hart reported that the most frequent extragenital malignancies metastasizing to the cervix or uterine body are breast (47.3%), gastric (29%), cutaneous melanoma (5.4%), lung (4.3%), colon (3.2%), pancreas (3.2%), and kidney (3.2%)[7]. Although gastric cancer metastasis to the female genital tract is exceptionally rare, affected patients have an extremely poor prognosis, which is worse compared to other primary sites (e.g., breast and colorectal cancers)[5].
To date, radical resection has been regarded as the only curative treatment for gastric cancer. However, even after radical surgical resection, postoperative recurrence or metastasis occurs in 60.8% of patients, with a median time to recurrence of 29.0 months[8]. In a study by Jiao et al., 68 GC patients had postoperative recurrence; among them, 53 (77.9%) had recurrences within the first two years, and 66 (97.1%) patients had recurrence within five years of surgery[9]. These high recurrence rates are attributed to the rich lymphatic network within the gastric wall, which facilitates mucosal skip lesions, as well as the diverse potential lymphatic drainage pathways extending beyond the stomach. In a study by Imachi et al., the mean interval between primary diagnosis and cervical metastasis in 16 gastric cancer patients was 35.8 months[10], significantly longer than recurrence intervals at other sites. Similarly, in the present case, the patient developed cervical metastasis six years post-curative gastrectomy for gastric cancer. This delayed recurrence pattern may be associated with the unique anatomical position of the cervix and its distinct metastatic route, though the underlying mechanisms remain incompletely understood.
The potential routes of extragenital metastasis to the cervix include hematogenous spread, retrograde lymphatic dissemination, and transperitoneal spread. The abundant fibromuscular tissue of the cervix may contribute to its low susceptibility to metastatic involvement[11]. Additionally, the cervix has a relatively small surface area compared to common metastatic target organs (e.g., liver, lung, bone, or ovary), and limited blood supply, which may further hinder metastatic implantation[11]. According to the literature, 20%–40% of metastatic cervical cancer cases are misdiagnosed as primary cervical cancer.[12] When cervical metastasis occurs, the most common presenting symptoms include genital bleeding, ascites, and lower abdominal pain[10], which can mimic primary cervical carcinoma and lead to misdiagnosis. Immunohistochemical (IHC) staining may aid in distinguishing primary cervical adenocarcinoma from metastatic lesions. In the present case, the cervical lesion showed IHC features of CK7(−), CK20(+), and Villin(+++), which are typical of gastrointestinal origin, confirming metastatic gastric cancer rather than primary cervical cancer. Notably, the primary tumor was moderately to poorly differentiated adenocarcinoma with a mixed Lauren classification and high Ki67 expression (60%+), indicating high proliferative activity. Suyila et al. demonstrated that poorly differentiated gastric cancer cells (e.g., MGC-803) are more sensitive to interventions targeting the apoptosis pathway, as they exhibit stronger responses to anti-cancer bioactive peptides (ACBP) that activate the TP53 signaling cascade[13]. The present patient's tumor also showed strong P53 expression (+++), which is consistent with the TP53-mediated apoptosis mechanism reported by Suyila et al.[13]; this may partially explain why the tumor responded well to CAPOX chemotherapy (which induces apoptosis) combined with sintilimab (which enhances anti-tumor immunity). Typically, CK7, CEA, and p16 are expressed in primary cervical adenocarcinomas[14]. Therefore, in patients with a history of malignant tumors presenting with masses outside the primary site, tissue biopsy should be obtained whenever feasible to determine the origin of the lesion. Immunohistochemistry guides the identification, classification, and assessment of tumor behavior and prognosis in cancers of unknown primary, thereby informing therapeutic decision-making.
Current identification of primary sites in metastatic tumors relies primarily on imaging (CT, MRI, PET-CT), histopathology, and immunohistochemistry. However, these modalities exhibit inherent limitations, particularly for poorly differentiated and undifferentiated carcinomas where immunohistochemical staining has restricted diagnostic utility. In recent years, the widespread application of molecular profiling-encompassing gene expression profiling, genomic and epigenomic analyses-has enhanced diagnostic accuracy for tumor origin. Emerging technologies such as liquid biopsy and AI-powered image analysis hold promise for further improving primary site detection rates in the future.
For patients with recurrent gastric cancer, the choice of chemotherapy regimen should consider factors such as the Lauren classification and prior treatment history. The present patient had a mixed Lauren classification (non-intestinal type), and we selected the CAPOX regimen (oxaliplatin + capecitabine) combined with sintilimab. The final partial response (PR) confirmed that this combination strategy was effective, which aligns with the principle of 'individualized adjustment based on Lauren classification' proposed by Wang et al.[15]. The present patient had a low PD-L1 expression (TPS = 0, CPS = 1) but still achieved PR with sintilimab, suggesting that PD-L1 may not be the only predictor of immunotherapy response. Li et al. reported that FAT4 mutation is an independent favorable prognostic factor in gastric cancer, as it is associated with high tumor mutation burden (TMB) and increased infiltration of effector immune cells (e.g., CD4 memory-activated T cells)[16]. Although a FAT4 mutation was not detected in the present patient, the good response to immunotherapy may be related to high TMB or other immune-related biomarkers—consistent with Li et al.'s finding that TMB and FAT4 mutation can complement PD-L1 to predict immunotherapy efficacy[16]. Additionally, Li et al. noted that FAT4-mutant patients have fewer regulatory T cells (Tregs) in the TME, which reduces immune suppression[16]; this may also explain why the present patient benefited from immunotherapy, even with low PD-L1 expression. Future studies should include TMB and FAT4 detection to optimize immunotherapy selection for patients with metastatic gastric cancer[16].
Even if the patient achieves PR after treatment, due to the high recurrence risk of pelvic peritoneal metastasis of gastric cancer, we have customized a personalized monitoring plan that conforms to clinical practice for them: Monitoring frequency: once every three months in the first two years (high-risk period of recurrence), once every six months in the next three years, and once a year thereafter. Evaluation method: Imaging: Contrast-enhanced CT of the abdomen/pelvis (for monitoring peritoneal/cervical/rectal lesions) + annual upper gastrointestinal endoscopy (for re-evaluating the remnant stomach); Laboratory: Tumor marker group (CA72-4, CEA, CA19-9) (series changes are associated with recurrence risk); Clinical/gynecological assessment: Routine pelvic examination + record of gynecological symptoms (such as genital bleeding, pelvic pain) (crucial for monitoring cervical metastasis); Management of suspected recurrence: If imaging or tumor markers suggest recurrence, further assessment (such as MRI, biopsy) should be conducted promptly to guide salvage treatment (such as targeted therapy, immunotherapy, and monotherapy). This protocol strikes a balance between the sensitivity of early recurrence detection and minimizing unnecessary intervention, and is in line with the current clinical guidelines for metastatic gastric cancer.
In summary, a rare case of cervical metastasis occurring six years after radical gastrectomy for gastric cancer is presented. This case holds significant clinical relevance due to its exceptional nature. When gastric cancer infiltrates beyond the serosa, exfoliated tumor cells implant onto the peritoneum and visceral serosal surfaces, forming metastatic nodules. Such seeding metastases predominantly occur in the mesentery, rectum, and ovaries (in females). In this case, both cervical and rectal metastases represent manifestations of peritoneal dissemination. While cervical metastasis is anatomically exceptional, its pathophysiological plausibility is supported by contiguous peritoneal spread. Critically, the significant response to gastric cancer-directed chemoimmunotherapy provides retrospective validation of the metastatic tumor origin. There is no standard for the treatment of metastatic cervical cancer. Studies have shown that overall survival with comprehensive treatment is longer than for radiotherapy or surgery alone (17.7 vs 10.7 or 13.2 months, respectively)[17]. While surgical resection may be an option for isolated cervical metastasis, the present patient had concomitant peritoneal dissemination, prompting the initiation of combination chemotherapy and immunotherapy.
-
This study was approved by the Ethics Committee of Nanjing Drum Tower Hospital and conducted in accordance with the principles of the Declaration of Helsinki (2013 revision). Written informed consent was obtained from the patient for the publication of this case report and any accompanying clinical/imaging materials. All patient-specific information (e.g., name, medical record number) have been fully de-identified to ensure privacy protection.
-
The authors confirm their contributions to the paper as follows: collected clinical data, organized pathological and imaging materials, drafted the initial manuscript: Zhang Z; designed the study framework, interpreted pathological and radiological results, revised the manuscript critically for important intellectual content: Wei J; supervised the study, confirmed the clinical diagnosis and treatment strategy, finalized the manuscript: Yang J. All authors have read and approved the final version of the manuscript and agree to be accountable for all aspects of the work.
-
All clinical data (including patient demographics, treatment records, and follow-up results), pathological images, and imaging materials (CT, MRI scans) used in this study are available from the corresponding authors upon reasonable request. Data sharing is subject to compliance with the Ethics Committee of Nanjing Drum Tower Hospital's regulations and patient privacy protection guidelines (all identifying information has been de-identified to protect patient confidentiality).
-
We would like to thank the Oncology Department and Pathology Department of Nanjing Drum Tower Hospital for providing clinical data access and pathological sample support during the study. We also express gratitude to the patient and her family for providing informed consent for the publication of this case report and accompanying images. Additionally, we appreciate the constructive feedback from the journal's reviewers, which significantly improved the quality of the manuscript.
-
The authors declare that they have no conflict of interest.
- Supplementary Fig. S1 Changes in tumor markers(CA72-4).
- Copyright: © 2025 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Zhang Z, Wei J, Yang J. 2025. Cervical metastasis from gastric carcinoma: a rare case report. Gastrointestinal Tumors 12: e019 doi: 10.48130/git-0025-0019
Cervical metastasis from gastric carcinoma: a rare case report
- Received: 02 September 2025
- Revised: 03 December 2025
- Accepted: 12 December 2025
- Published online: 31 December 2025
Abstract: Secondary cervical malignancies commonly arise from adjacent reproductive tract tumors, whereas metastatic spread from gastric carcinoma to the cervix is exceedingly rare. A rare case of cervical metastasis in a 70-year-old Chinese female with a history of gastric cancer treated by curative gastrectomy six years prior is presented; she presented with anal tenesmus, and dull left lower abdominal pain. Pelvic MRI revealed space-occupying lesions involving the cervix, with pathological evaluation suggesting a gastrointestinal origin, and a multidisciplinary team (including oncologists, pathologists, and radiologists) confirmed the rectal lesion resulted from extrinsic compression due to peritoneal dissemination of gastric cancer rather than a primary rectal tumor. The patient subsequently underwent chemotherapy (oxaliplatin + capecitabine) plus immunotherapy (sintilimab), achieving a notable treatment response: normalization of tumor markers (CA72-4), significant regression of cervical and rectal lesions, resolution of intestinal obstruction symptoms, and partial remission (PR) per RECIST 1.1 criteria. Treatment for cervical cancer differs significantly from that of gastric cancer; thus, in patients with a history of gastric cancer presenting with a cervical mass, the possibility of gastric cancer recurrence should be carefully considered, and a tissue biopsy is essential to confirm the diagnosis and guide appropriate treatment strategies.
-
Key words:
- Stomach neoplasms /
- Cervical metastasis /
- Chemotherapy /
- Immunotherapy /
- Partial response