-
Fungal endophytes can live within plants for part, or most, of their life cycle without showing any symptoms on the plant[1]. Many fungal endophytes have been shown to confer many benefits to plants[2,3]. Increasing its nutrient availability, its photosynthetic capacity, and the production of plant hormones are among them, as well as enhancing its capacity to resist abiotic stresses[4−7]. In addition, fungal endophytes can reduce plant herbivory[7−9] and/or fungal attack[4,6,7,10], either directly through the production of toxins or indirectly through inducing the plant salicylic and jasmonic acid pathways, increasing the production of volatile organic compounds, antioxidants, and secondary metabolites[11]. Consequently, multifunctional endophytes represent a new paradigm for eco-friendly agricultural practices, ranging from the biological control of pests and pathogens[2,4] to biological promoters of plant growth[5,12]. Therefore, the use of strains with multiple functionalities may represent a promising cost-effective strategy in agriculture.
Purpureocillium lilacinum (Thom) Luangsa-ard, Houbraken, Hywel-Jones & Samson has initially been used for controlling plant nematodes[10,13]. Subsequently, its entomopathogenic capacity was demonstrated[14−16], as was its mycopathogenic role[16−18]. Purpureocillium lilacinum has occasionally been shown to enhance plant growth when applied to the soil as a biofertilizer[10,19,20]. Moreover, this fungal species has been shown to degrade plastic wastes as well[21]. Based on current knowledge, it has been naturally isolated as an endophyte only from cotton plants, monk fruits, and chickpea plants[22−24]. Additionally, its potential as an endophyte has been evaluated experimentally in tomato plants[25,26]. Moreover, the strain isolated from cotton was found to protect this plant against the cotton aphid[27] and bollworm[28]. This cotton strain, as well as one isolated from whiteflies attacking tomato, has been shown to promote plant growth as endophytes[25,26].
Strawberries (Fragaria × ananassa Duc; Eosales: Rosaceae) are widely distributed and represent an important commodity worldwide, with China and the USA the main world producers[29]. Strawberry is a very demanding crop in terms of needed inputs to maintain healthy plants and high fruit yields[30]. In particular, strawberry production requires high levels of fertilization[30,31]. However, chemical fertilizers could damage soil fertility, increase soil salinity, and damage aquatic ecosystems through eutrophication[32,33]. Therefore, given the increasing pressures from organic producers' certification requirements[34], and consumer concerns[35], alternatives to agrochemicals should be found for strawberry cultivars[31]. The use of endophytes represents a promising approach to address these demands. Although some endophytes have occasionally been tested in strawberries with promising results[8,12,36−38], P. lilacinum has not been among them. Therefore, considering both extant pressures for sustainable solutions in strawberry cultivars and the known benefits of P. lilacinum, assessing its potential benefits as an endophyte in strawberry for the first time may provide valuable information to determine whether this fungus could be considered a suitable candidate for the agroecological and organic production of this valuable fruit.
The overall aim of the present research was to evaluate whether a strain of P. lilacinum, previously characterized as entomopathogenic and mycopathogenic, can also function as an endophyte in strawberry plants. It was hypothesized that P. lilacinum, acting as an endophyte, can promote strawberry plant growth. In particular, this study evaluated its ability to colonize internal tissues following root or foliar inoculation, its capacity to spread from the inoculation site to other parts of the plant, the persistence of the endophyte over time, and its effect as a plant growth promoter. For that, strawberry plants were grown in a greenhouse and inoculated using root or foliar inoculation. The percentage of colonization, as well as plant growth parameters, was recorded over time.
-
Strawberry plants (cultivar 'Aroma'; neutral day variety) were obtained from an organic crop when each plant had three to four leaves. After receiving them, the roots of all plants were cut to a total length of 7 cm, as recommended by the seller. Plants were kept in seedling trays with their roots submerged in water until use. According to the total weight and general appearance, the plants were divided into groups containing similar plants. Later, all the plants were transplanted individually into 3 L pots with a non-sterilized substrate composed of 50% fertile soil, 30% peat, and 20% perlite. Plants were kept in a greenhouse at the National University of Quilmes (Bernal, Argentina) under the same conditions, as follows: 106 d (from 8 September to 23 December 2022) at 25 ± 9 °C and 51% ± 22% RH (means and SD) for the root experiment, and 87 d (from 1 November 2022 to 27 January 2023) at 29 ± 9 °C, and 48% ± 21% HR for the foliar one. They were maintained under the same natural light conditions (approximately 16 h light: 8 h dark, corresponding to the spring–summer period in Argentina), and were watered daily using an automatic irrigation system and fertilized weekly with Hoagland solution. A total of 20 plants were assigned to each inoculation treatment (root or leaf inoculation), and 20 additional plants served as controls. Plants were positioned haphazardly within the greenhouse. Some plants died during the experiment. The final number of surviving plants per treatment was used in the analyses.
Plant growth parameters, as well as flowers and fruits produced, were quantified twice per week until the end of each experiment. Leaves were classified as 'healthy' if they were vigorous and completely green; otherwise, they were considered 'unhealthy'. This last category included leaves with brown borders, yellow or red spots, or withered or misshapen (Fig. 1). New leaves and dead leaves were registered throughout the experiment. New flowering buds were also recorded, and at each sampling session, flowers and fruits were counted and identified to prevent double-counting in subsequent measurements. In addition, the number of runners produced over time was quantified.

Figure 1.
Leaves classification. (a) Healthy leaves. Unhealthy leaves: (b) Red border. (c) Senescent leaf. (d) Brown border. (e) Chlorosis. (f) Withered. (g) Yellow and red spots. (h) Misshapen. Dark arrows indicate each symptom in unhealthy leaves.
Plants: inoculation with P. lilacinum
-
Purpureocillium lilacinum strain YLOB, used in this study, was isolated from the fungus garden of a leaf-cutter ant colony (Acromyrmex lobicornis Emery) collected in Corrientes, Argentina. This strain was properly identified morphologically and molecularly, using the nuc elongation factor-1α (EF-1α) gene region[15], and was stored in glycerol 20% at –80 °C in the Laboratory collection until its use. Then, it was plated onto Petri dishes (90 mm × 15 mm) containing potato dextrose agar (PDA) supplemented with antibiotic (ampicillin 100 µg/ml), and incubated at 25 °C in the dark for 7 d. Two successive passages onto culture media were done before using the fungus. Afterwards, conidia were harvested by scraping the surface of the culture with a sterilized spatula with the addition of 10 ml of sterile 0.01% Tween 80. Conidial concentration was determined using a Neubauer hemocytometer and adjusted to 1 × 108 conidia/ml by performing the necessary dilutions with the same Tween 80 solution. This concentration was used in both inoculation treatments. It represents a commonly used concentration[39], and was chosen for producing the greatest entomopathogenic effect[15]. Conidial viability was assessed prior to use by incubating an aliquot of the conidial suspension on PDA at 25 °C for 18 h, after which at least 100 conidia were examined under a light microscope and considered viable when the germ tube was equal to or longer than the conidial diameter. Viability was consistently higher than 95%.
Root treatment
-
Plants were subjected to root inoculation by submerging their roots, altogether, in a container (30 cm × 15 cm × 5 cm) with 200 ml of fungal suspension; roots were left under these conditions for 24 h. After that, these plants and another group of plants without any treatment (control) were transplanted to 3 L pots, as explained above. The inoculated and control groups had 14 surviving plants each. The experiment spanned 106 d, which corresponds to approximately 15 weeks.
After 17 d post-inoculation (hereafter referred to as dpi), samples were harvested from 12 plants, including a young leaf that was not present at the time of inoculation and the oldest leaf, which was present at the time of inoculation. This was also done with ten control plants. Leaves were plated on the same day of harvesting after their sterilization (see protocol below). At 106 dpi, the youngest and oldest leaves from each plant of the inoculated treatment were harvested.
Leaf treatment
-
Leaf inoculation was carried out in 20 plants two months after being planted, since several leaves were needed per plant for harvesting. The same control plants from the previous experiment (N = 14) were used here, which remained non-inoculated throughout the experiment. The inoculation was made on two leaves/plant, a young one and an old one. The conidia suspension was applied by spraying the leaves on both sides. Each leaf received one or more sprays, depending on its size, to completely cover the leaf with the suspension. All the other leaves, plant organs, and the soil were covered with aluminum foil to avoid unwanted inoculations. Afterwards, the inoculated leaves were enclosed with a transparent plastic bag for 48 h. At 7 and 14 dpi, one inoculated leaf and one non-inoculated leaf were harvested from ten inoculated plants, while two non-inoculated leaves were collected from each of the ten control plants. At 21 and 43 dpi, only non-inoculated leaves were harvested because the inoculated leaves had already been collected in previous samplings. The aim was to determine whether the conidia initially applied to the inoculated leaves had translocated within the plant and persisted in other leaves after inoculation. Leaf-inoculated plants and their controls were uprooted at 87 dpi. Each plant was washed with tap water and divided into above-ground and below-ground parts. Subsequently, each of these parts was placed individually inside a paper bag and labeled accordingly before being placed in an oven at 60 °C for 3 d, until a constant dry weight was obtained. Afterwards, the content of each bag was weighed using a precision lab balance (readability 0.01 g), Acculab model VI 350 (Acculab: San Diego, CA) to obtain the dry biomass value. The experiment lasted 87 d (approximately 13 weeks).
Endophytism: plant tissue sterilization and presence of P. lilacinum
-
As recommended by McKinnon[40], published protocols were modified to obtain a suitable one for the plants. Each leaf was surface sterilized by sequential immersion in 0.01% Tween 80 for 1 min, 85% ethanol for 1 min, and 20% sodium hypochlorite solution for 5 min; afterwards, each leaf was rinsed three consecutive times in sterile distilled water. In addition, it was confirmed that the removal of epiphytes and the inoculated fungus by finding no fungi after plating the last water rinse from each leaf on PDA. Each sterilized leaf was cut into sections, which were counted, placed in Petri dishes with PDA and antibiotic (100 µg/ml of ampicillin), and stored in the dark at 22–23 ºC for 10 d. After that time, the number of segments per Petri dish that exhibited P. lilacinum growth was quantified. The same procedure was carried out with equivalent leaves from control plants, for roots from the root experiment, as well as for fruits from all treatments.
The percentage of endophytism was calculated in two ways: by plant and by leaf. Plants that had, at least, one leaf section with the inoculated strain present in any harvested leaf were considered positive for endophytism. The percentage of endophytism/plant was calculated based on the number of all plants/treatment. To obtain the percentage of endophytism by leaf, the number of leaf sections showing the inoculated strain out of the total sections plated was first calculated for each leaf. This data was categorized by leaf age (young and old) and harvest date for all treatments. This calculation was done both for inoculated leaves and non-inoculated leaves harvested from inoculated plants as well as from leaves harvested from non-inoculated plants (i.e., the control group). The median and quartiles of the percentage of endophytism/leaf were calculated considering all the plants/treatments.
Statistical analysis
-
Plants from the control group were compared with the root-inoculated plants using the data gathered from the beginning of the experiment until the end of it (106 dpi). Similarly, plants from the control group were compared with the leaf-inoculated plants, considering only the data collected from the day of leaf inoculation (starting at 56 dpi of the root inoculated treatment) until the end of the experiment (87 dpi of the leaf treatment).
To reduce data noise and improve model fitting, weekly measurements of growth parameters were averaged for the foliar treatment, and bi-weekly averages (from two consecutive weeks) were used for the root treatment. Data were analyzed using linear mixed-effects models (LMMs) and generalized linear mixed-effects models (GLMMs), incorporating a nonlinear temporal trend when appropriate, fitted using the glmm TMB function in R. Modeling fitting and assumption diagnostics were performed using the DHARMA package (Diagnostics for Hierarchical Regression Models). When normality assumptions were violated, data transformations were applied, or alternative distributions for count data were used. In cases of variance heterogeneity, heteroscedastic models with appropriate dispersion structures were applied.
TREATMENTS, PERIOD of time, and the INTERACTION treatment × period were included as fixed effects in the analysis of each variable. Plant identity (ID) was included as a random effect to account for repeated measures (all details are provided in Supplementary File 1 and Supplementary File 2). For non-linear trends over time (e.g., in healthy leaves, flowers, or fruits), natural splines (ns) were used to identify knots (points where trends shift) and to fit smooth curves in order to capture gradual changes, avoiding overfitting. Model selection was based on model fit evaluation and the Akaike Information Criterion (AIC). Significant interactions between treatment and periods pointed to treatment-specific differences over time, which were further explored via post hoc comparison, using estimated marginal means (emmeans), followed by Tukey's test adjusted with Bonferroni correction for multiple comparisons. The post hoc comparisons were done every two weeks in the root inoculation experiment or every week in the foliar inoculation treatment. In the latter, week zero represented the measurement done prior to the inoculation to examine if there were differences between treatments.
Biomass data from the leaf-inoculated and control treatments were compared using t-tests after testing for normality.
All statistical analyses were done using R version 4.1.0 (Development Core Team 2021).
-
The data were log-transformed to satisfy the model's assumptions. A model with two degrees of freedom, with a knot placed at week eight, was selected. Overall, the total number of leaves increased over time (Fig. 2a). A significant interaction (p = 1.26e−05) between treatment and the second period (period ≥ 8) was detected (Table 1S in Supplementary File 1), allowing further post hoc contrasts. These contrasts revealed that, from week eight onward, plants in the inoculated treatment produced significantly more leaves compared to the control (Fig. 2a). At the end of the experiment, the mean ± standard deviation of the registered leaf number was 11.0 ± 1.9 for inoculated plants and 8.1 ± 1.1 for control plants. The variance associated with the random effect was minimal (Table 1S in Supplementary File 1), indicating low variability attributable to the plants.
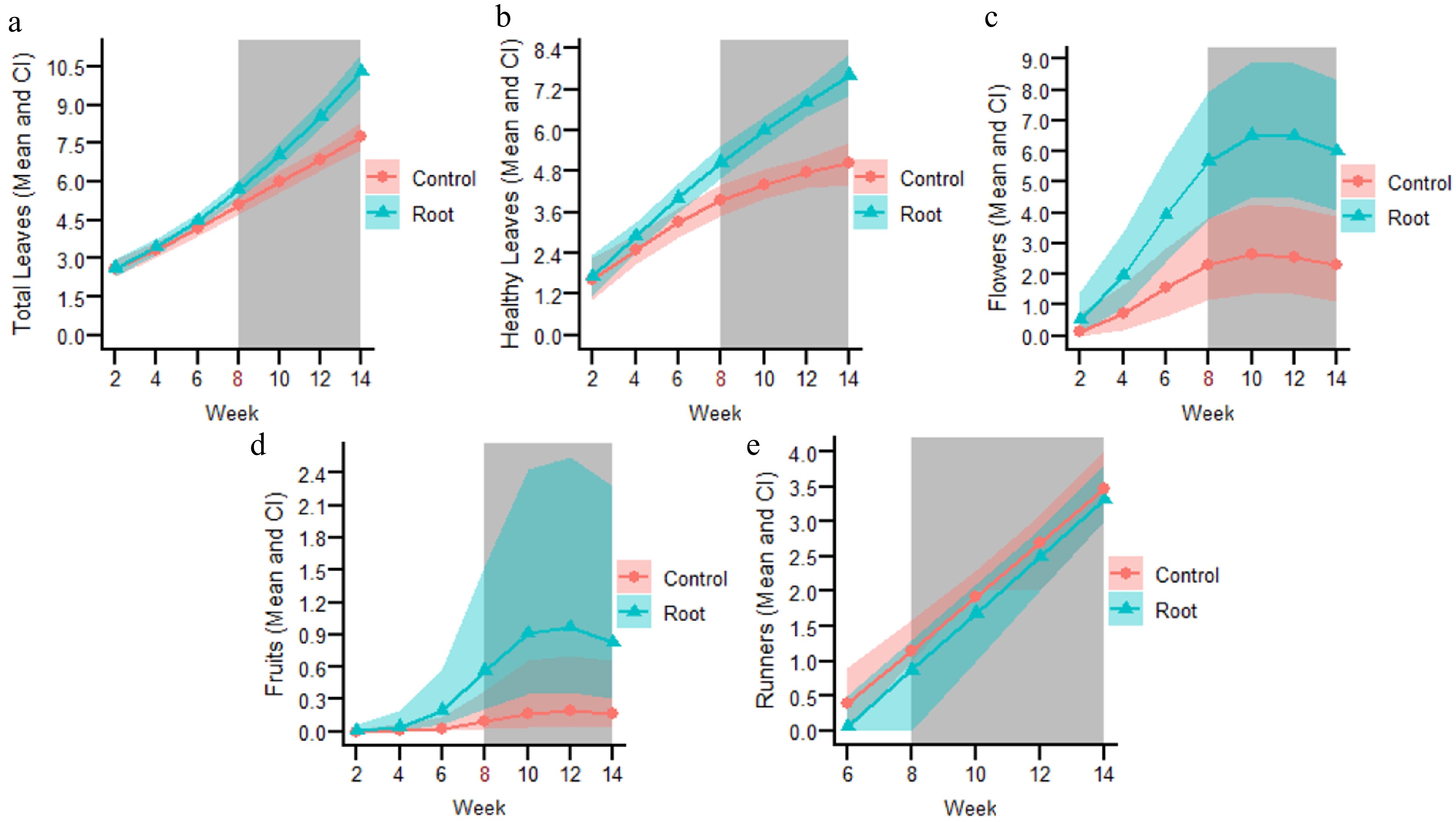
Figure 2.
Fitted lines from selected models for the following variables of the root-inoculated experiment: (a) Total number of leaves. (b) Total number of healthy leaves. (c) Total number of flowers. (d) Total number of fruits. (e) Total number of runners. The number in red on the X axis represents the knot, where the tendencies of each variable changed over time. The grey-shaded background indicates the weeks at which significant differences (p < 0.05) were observed between the root-inoculated and control treatments.
Table 1. Percentage of endophytism with P. lilacinum calculated by plants and by leaf/fruit from the leaf inoculated experiment. The last column shows data as median with quartiles, plus minimum and maximum values recorded.
Days post inoculation (dpi) Organ harvested Number of plants/fruits Plants with the endophytes (%) Leaf/fruit with endophyte (%) Median (q1–q3) Min–max 7 Old inoculated leaves 10 100 9 (5–14) 4–16 7 Old non-inoculated leaves 10 20 9 (5–12) 5–12 14 Young inoculated leaves 10 90 12 (8–15) 3–29 14 Young non-inoculated leaves 10 40 1 (1–2) 1–3 21 Old non-inoculated leaves 10 20 2 (2–2) 2–2 43 Old non-inoculated leaves 10 0 0 0 7–24 Fruit 13 0 0 0 Number of healthy leaves
-
Data satisfied the model's assumptions using a Gaussian distribution. A model with two degrees of freedom, also with a knot placed at week eight, was selected. The total number of healthy leaves increased over time (Fig. 2b). There were significant interactions between treatment and week one (p = 0.0054) and with week two (p = 4.44e−05) as well (Table 2S in Supplementary File 1). Post hoc contrasts showed a significantly greater number of healthy leaves, from week six until the end of the experiment, in the root inoculated treatment in comparison to the control (Fig. 2b), ending with 7.7 ± 2.2 and 4.7 ± 1.9 healthy leaves, respectively. The variance associated with the random effect was low (Table 2S in Supplementary File 1).
Number of flowers
-
Data satisfied the model's assumptions using a Gaussian distribution, after being square root transformed. The selected model had two degrees of freedom, with a knot placed at week eight. The number of flowers increased from the beginning of the experiment until week ten, when it remained constant or decreased very little until the end of the experiment (Fig. 2c). The interaction between treatment and week was significant for each period (p = 3.18e−05 and 0.00214, respectively; Table 3S in Supplementary File 1). Post hoc adjusted comparisons showed significant differences from week six onward (Fig. 2c), ending with 6.6 ± 3.5 number of flowers for the root inoculated plants and 3.3 ± 3.5 for the control plants. The variance for the random effect for the number of flowers was similar to the ones from the previous models (Table 3S in Supplementary File 1).
Number of fruits
-
This variable was fitted with a Poisson distribution to satisfy the model's assumptions. This selected model also had two degrees of freedom, with a knot placed at week eight. Fruit production started after week four increasing until week ten without producing new fruits afterwards (Fig. 2d). There was no significant interaction (p > 0.05) between treatment and both periods of the selected model, however, there was a significant treatment effect (p = 0.042, Table 4S in Supplementary File 1), which showed greater fruit production in the root inoculated group in comparison to the control from week six onward (Fig. 2d). The final number of fruits produced was 1.6 ± 1.3 for the inoculated group and 0.6 ± 1.1 for the control. The variance associated with the random effect was greater in comparison to other plant parameters (Table 4S in Supplementary File 1).
Number of runners
-
A Gaussian distribution was used, and the model selected analyzed all the time series as one period. Production of runners started at week six and continued increasing until the end of the experiment, with a similar trend for both treatments (Fig. 2e). There were no significant differences for the interaction term nor for the treatment (p > 0.05; Table 5S in Supplementary File 1). In fact, 3.5 ± 1.5 runners were registered in the root-inoculated plants and 3.6 ± 1.3 in the control group. The variance of the random term was intermediate (Table 5S in Supplementary File 1).
Leaf inoculation
Total number of leaves
-
The data were log-transformed, and a dispersion structure was incorporated to satisfy the model's assumptions. The selected model had only one period. Before inoculation, the average total number of leaves, every week, was very similar between the control plants and those later assigned to the leaf-inoculation group (Fig. 1S in Supplementary File 2). Overall, the total number of leaves increased over time (Fig. 3a). A significant interaction (p = 0.0022) between treatment and period was detected (Table 1S in Supplementary File 2). Contrasts revealed that, from week three onward, plants in the inoculated treatment produced significantly more leaves compared to the control (Fig. 3a). At the end of the experiment, the mean ± standard deviation of the registered leaf number was 11.6 ± 1.9 for inoculated plants and 9.2 ± 1.2 for control plants. The variance associated with the random effect was very low (Table 1S in Supplementary File 2).
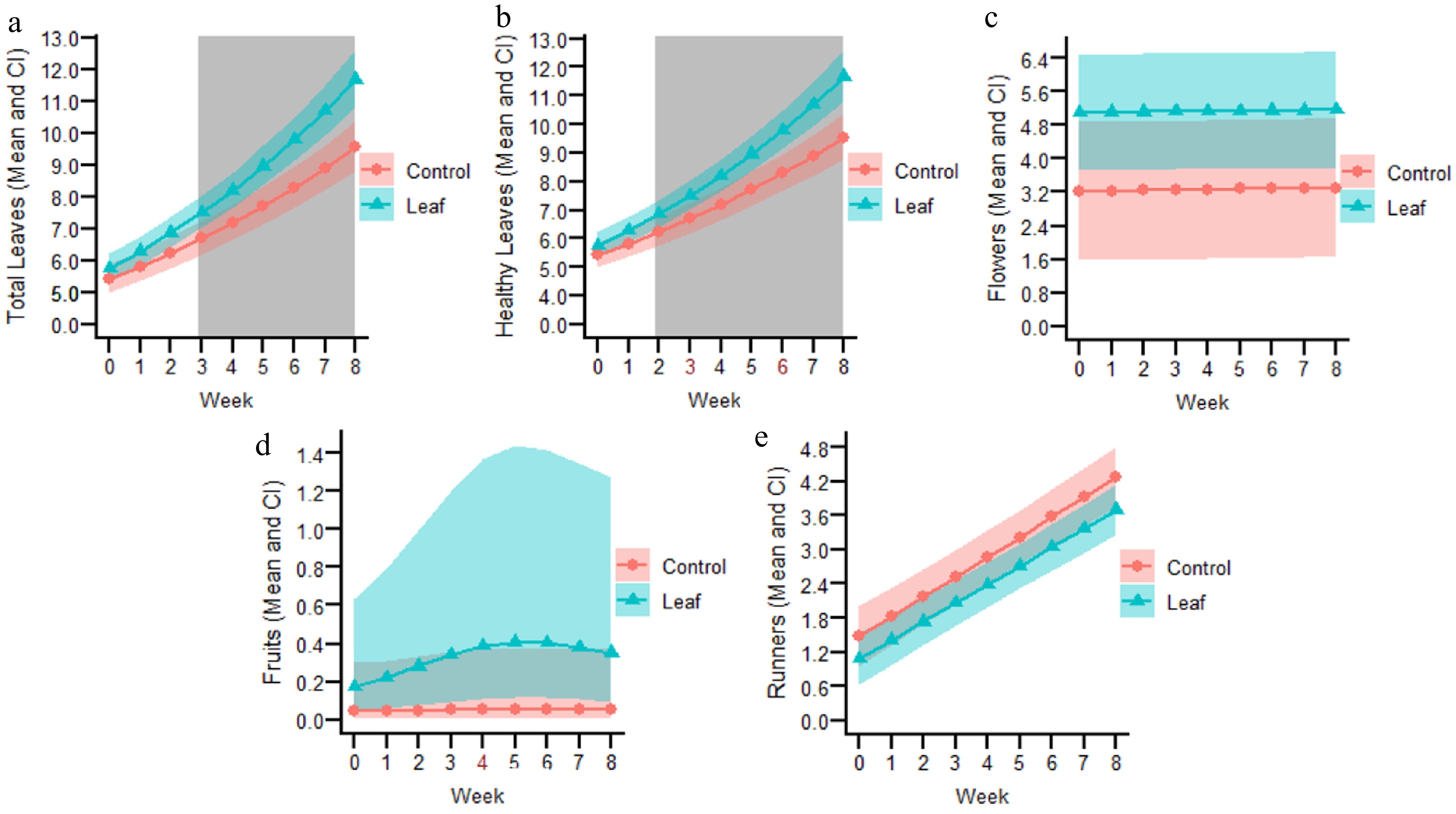
Figure 3.
Fitted lines from selected models for the following variables of the leaf-inoculated experiment. (a) Total number of leaves. (b) Total number of healthy leaves. (c) Total number of flowers. (d) Total number of fruits. (e) Total number of runners. The number in red on the X axis represents the knot, where the tendencies of each variable changed over time. The grey-shaded background indicates the weeks at which significant differences (p < 0.05) were observed between the leaf-inoculated and control treatments.
Number of healthy leaves
-
Data satisfied the model's assumptions after being square root transformed and using a Gaussian distribution. The number of healthy leaves was similar between the plants assigned as control or to be inoculated (Fig. 2S in Supplementary File 2). A model with three degrees of freedom was selected, with knots placed at week three and six (Table 2S in Supplementary File 2). The total number of healthy leaves sharply increased over time for the foliar-inoculated treatment, but the control increased a little at the beginning and later remained almost constant (Fig. 3b). There were significant interactions between treatment and each of the three periods (p = 2.69e−07, 7.9e−06, and 2e−16, respectively) (Table 2S in Supplementary File 2). Post hoc contrasts showed significantly more healthy leaves in the foliar inoculated treatment in comparison to the control from week two onward (Fig. 3b), ending with 10.2 ± 1.7 and 5.5 ± 1.2 healthy leaves, respectively. The fixed variables of the model explained most of the variance in the data, as very little was associated with the random effect or the residuals (Table 2S in Supplementary File 2).
Number of flowers
-
Data fitted with a Gaussian distribution satisfied the model's assumptions, which had only one period. At the time of assignment, plants designated for foliar inoculation already showed a higher number of flowers compared to the control (Fig. 3S in Supplementary File 2), and this difference between treatments was maintained throughout the post inoculation period, although it was not significant (Fig. 3c). The interaction between treatment and period was not significant nor the treatment (both p > 0.05, Table 3S in Supplementary File 2). The observed final number of flowers for the foliar inoculated plants was 5.2 ± 3.1, and for the control plants was 3.3 ± 3.6. The variance for the random effect was high, indicating great differences among plants, but overall most, of the variance was accounted for by the model (low residual) (Table 3S in Supplementary File 2).
Number of fruits
-
Data was log-transformed and fitted with a Poisson distribution. The selected model had two degrees of freedom, with a knot placed at week four. During the pre-inoculation time, including one week before inoculation, week zero, the number of fruits were similar between treatments (Fig. 4S in Supplementary File 2), afterwards the number of fruits started to increase only for the foliar inoculated treatment until week four, later no new fruits were produced (Fig. 3d). The interaction between treatment and each period was not significant nor any of the other factors (all p > 0.05, Table 4S in Supplementary File 2). The final number of fruits for the foliar inoculated plants was 1.6 ± 2.1, and for the control plants it was 0.6 ± 1.2.

Figure 4.
Lateral view (above) and top view (below) of strawberry plants from the leaf inoculation (left), root inoculation (middle), and control (right) groups.
Number of runners
-
Data was fitted with a Gaussian distribution. Once runner production began, a slightly greater number was found in control plants compared to those assigned to the inoculated treatment (Fig. 5S in Supplementary File 2), and this difference remained throughout the post-inoculation period (Fig. 3e). The interaction between treatment and period was not significant nor the treatment factor (both p > 0.05) (Table 5S in Supplementary File 2). The final number of runners for the foliar inoculated plants was 3.8 ± 1.2, and for the control plants it was 4.2 ± 1.3. The variance associated with the random effect was low (Table 5S in Supplementary File 2).
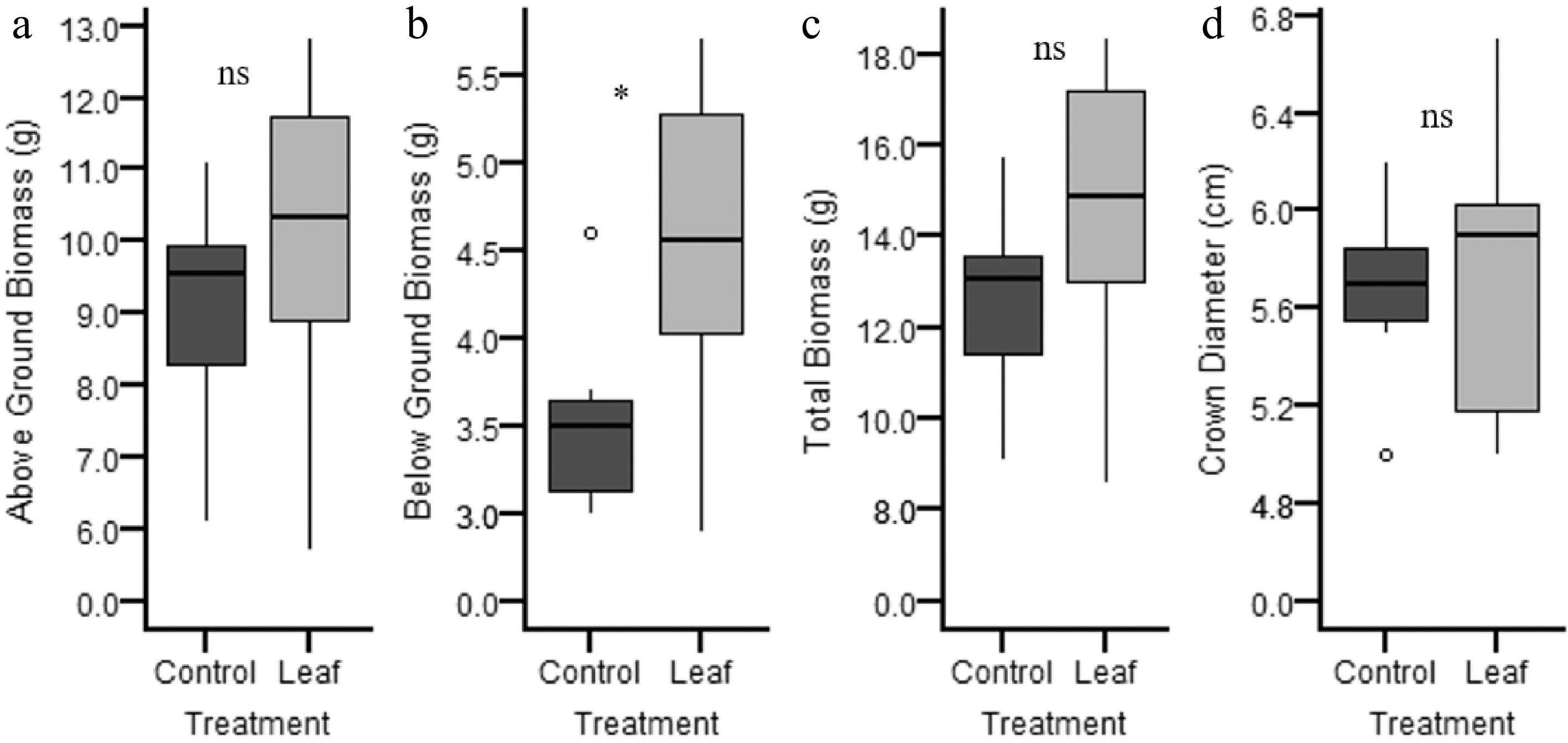
Figure 5.
Boxplot for (a) above-ground, (b) below-ground, and (c) total biomass, plus (d) crown diameter (Y axes are different for each variable) discriminated by treatment: control (light grey) and leaf inoculated (dark grey).
Figure 4 shows a representative plant from each treatment, where the number of total leaves and healthy leaves was evidently greater for the leaf and root inoculated plants in comparison to the control.
Regarding the biomass, no significant differences were observed between treatments for total biomass and above-ground biomass (t = 1.26 and 0.86; p = 0.23 and 0.54, respectively) when the leaf inoculated treatment was compared with the control group. However, the leaf inoculated treatment produced significantly greater below-ground (root) biomass (t = 2.16; p = 0.05) (Fig. 5).
Percentage of endophytism
Root inoculation
-
Three plants with the endophyte in new leaves were found after 7 dpi with the following percentages of endophytism/leaf: 4%, 7%, and 50%; whereas leaves harvested from control plants showed no P. lilacinum growth. After 106 dpi, none of the leaves or roots had any signs of the inoculated strain. Out of 21 fruits, none showed P. lilacinum growth.
Leaf inoculation
-
A greater percentage of plants tested positive for endophytes when comparing inoculated leaves to non-inoculated ones, for harvests done 7 and 14 dpi (Table 1). Twenty percent of the plants were positive after 21 dpi, but none at 43 dpi (Table 1). None of the control plants exhibited P. lilacinum growth at 7 and 14 dpi on old and young leaves, respectively.
The median percentage of endophytism/leaf was the same for inoculated and non-inoculated old leaves at 7 dpi, but it was greater for inoculated young leaves at 14 dpi. A small percentage of endophytism/leaf was found at 21 dpi and none at 43 dpi (Table 1). Of 13 evaluated fruits, none contained P. lilacinum.
-
This work shows, for the first time, the endophytic ability of P. lilacinum in strawberry plants, highlighting its multifunctional potential as a fungal species. This fungus successfully colonized plants after being inoculated via roots or leaves, spreading from the inoculation site to other non-inoculated parts of the plants. Moreover, in the leaf inoculation experiment, the endophyte was detected at least until 21 dpi, although it was no longer detectable at 43 dpi. Nonetheless, this fungus proved to have positive effects beyond that time, lasting up to two to three months after inoculation, depending on the inoculation treatment.
This study is also the first to demonstrate the growth-promoting effects of an endophytic P. lilacinum in strawberry plants. In the root experiment, all growth parameters were significantly greater in the inoculated group in comparison to the control, except for the number of runners. In the leaf experiment, there was also a significant increase in the number of total and healthy leaves in the inoculated plants; additionally, final root biomass was also significantly greater in this group at the end of the experiment. The significantly higher number of healthy leaves and fruits recorded in endophyte-inoculated plants provides strong evidence of a beneficial effect of P. lilacinum YLOB on strawberry development and productivity.
The functional outcome of the effect of an endophyte depends on the specific endophyte-plant combination[28,41]. For instance, in the case of P. lilacinum, a growth-promoting effect of P. lilacinum was observed in cotton plants[28], while no such response was found in beans using the same strain tested in this study[42]. Likewise, in tomato, three P. lilacinum strains were evaluated, and only one promoted plant growth, though it was not the same strain that exhibited endophytic colonization in roots[25]. This variation was also observed when selecting the best endophytic strain based on its biological control capacity[13]. A meta-analysis on the effect of root endophytes on plants showed that the results changed depending on the fungal strain and plant species used[41]. Therefore, it is difficult to generalize the effect of a given endophytic fungal species, or even a strain, across different plant species.
Understanding whether and for how long P. lilacinum persists in planta is essential to assess its potential as a functional endophyte. To be used in crop protection, theoretically an endophyte should persist throughout the plant life cycle or, at least, during its most vulnerable periods[43,44], and be present in all the plants. After 7 dpi, the endophyte was detected in 100% of the strawberry plants in the leaf inoculated group; however, this percentage decreased over time, as reported in other studies[26,45,46], until it was no longer found at 43 dpi. In the root-inoculated experiment, the number of plants showing endophytic colonization in new leaves was much lower, probably because it was measured too early (7 dpi) or too late (at the end of the experiment). Interestingly, positive effects on plant development were observed even after the endophyte was no longer detected. There are two non-exclusive possible explanations for this outcome. First, it is possible that the endophyte may have triggered plant responses through hormones or metabolites, which continued to have an impact even after the initial stimulus ceased. Second, the endophyte may have induced a plant response that became evident only at a specific stage of the plant's life cycle, such as increased fruit production observed at harvest time. The extent of the benefit to the plant may also depend on the level of colonization of the endophyte, especially in providing effective protection against herbivores and pathogens. For example, in this study, the median percentage of endophytism/leaf in the leaf-inoculated group did not exceed 12%, which was similar to, or lower than, values reported in other studies[26,27,44]. Possible avenues to increase the level of endophytism, both per plant and plant tissue, could be done by combining multiple inoculation methods or repeating inoculations through time[47]. Furthermore, most studies using leaf inoculation apply the fungus to only one or two leaves/plant, as in this study[3,4]. However, it is likely that if more leaves per plant were inoculated, higher endophytism/leaf levels could be achieved. It is strongly believed that any of the mentioned avenues could increase the percentage of endophytism, especially when applied simultaneously. Ideally, several methods should be tested first to compare the success of the endophytism colonization and to identify the best inoculation method[43,48−50]. Optimizing endophyte inoculation and ensuring its effects during critical stages of plant development are essential to achieve tangible agricultural benefits and promote sustainable practices. However, from an environmental perspective, a decline in endophyte presence over time may be desirable if it could contribute to maintaining ecosystem balance after application, especially in the case of multifunctional organisms[51].
Having healthier plants is one of the main goals of the strawberry production industry[52], as it contributes to a good fruit yield[53]. In this study, healthier leaves were used as a surrogate for overall plant health, since leaves are responsible for producing food and energy needed for plant growth and function. An improved leaf condition could lead to better resistance to abiotic and biotic stresses in plants inoculated with endophytes. Healthier leaves may also result from enhanced immunological status and/or increased phytohormone production; the latter of which has been measured in several plants with endophytes[47,54−56]. In addition, these leaves probably contain higher nutrient levels, possibly due to greater nutrient acquisition, a benefit previously reported in several plant–endophyte systems[12,57−59]. Based on current knowledge, leaf health has not been explicitly quantified in studies that test the beneficial effect of endophytes. In addition, a more developed root system, as observed in the experiment where biomass was evaluated, may facilitate nutrient acquisition from the soil, indirectly contributing to the development of healthier leaves.
The importance of strawberry crops in the market is directly linked to fruit production. It was particularly encouraging that a higher yield was found in root-inoculated plants, with no detection of the endophyte in the harvested fruit. While some studies have also shown the benefits of fungal endophytes on strawberry fruit yields[47,55,60], they have not examined the presence of these fungi in the fruits themselves (with the exception of one study employing a proteomic approach[47]). This lack of testing could raise concerns among consumers. It was noteworthy that no significant increase in fruit production was detected in the leaf-inoculated experiment. A statistical explanation may lie in the higher variability observed in fruit yield among plants in this treatment. Biologically, the lack of effect could be related to the fruiting phenology of strawberry plants and the differing start dates of the two experiments. Fruiting began in mid-October and lasted approximately six weeks. Root-inoculated plants produced fruits steadily throughout the entire period, consistently outperforming control plants from the beginning. In contrast, leaf-inoculated plants only produced more fruits than controls during a three-week window. Importantly, root inoculation occurred five weeks prior to the onset of fruiting, whereas leaf inoculation was performed after fruiting had already begun. As a result, root-inoculated plants were under the influence of the endophyte for a longer period before fruit production began, likely contributing to the significantly higher fruit yield observed in this group compared to the control. In contrast, the shorter exposure time in leaf-inoculated plants may have limited the endophyte's ability to significantly affect fruit production. Further experiments with leaf inoculation prior to the onset of fruit production should be conducted to evaluate this hypothesis.
Purpureocillium lilacinum is emerging as a classic example of a multifunctional fungus with diverse lifestyles[10,48]. The same strain, previously shown to possess entomopathogenic and mycopathogenic activity[15,17], has now been demonstrated to function as an endophyte, enhancing plant health and fruit production in strawberry. These results suggest the strong potential of strain YLOB for application in strawberry cultivation, especially in the current context of rising concerns about synthetic chemical use. Further studies should assess its efficacy across different strawberry varieties and under multiple stress conditions. To implement this strategy under field conditions, inoculation methods such as foliar sprays could be applied in soil-grown strawberry plants without major modifications to current agronomic practices. Subsequent field trials will be valuable for assessing the practicality of endohyte inoculation, its persistence throughout the growing season, and the technical feasibility of inoculum production. Overall, such studies will provide crucial insight into the practical adoption of P. lilacinum as a multifunctional endophyte for use in intensive cultivar systems, like strawberry.
The authors acknowledge the support of the National Council for Scientific and Technological Research (CONICET) and the National University of Quilmes of Argentina. We thank Hernán Farina for kindly allowing us to use his greenhouse, and Josefina Bompadre for helping us with the automatic irrigation system. This study was supported by the National Agency for Scientific and Technological Promotion (Grant No. PICT-2019-3634) and by the National University of Quilmes (Grant No. PUNQ 2259/22), both to PJF.
-
The authors confirm their contributions to the paper as follows: study conception and design: Folgarait PJ, Goffré D; data collection: Folgarait PJ, Goffré D, Rocano Domínguez HS; analysis and interpretation of results: Folgarait PJ, Goffré D, Rocano Domínguez HS; draft manuscript preparation: Folgarait PJ. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
-
The authors declare that they have no conflict of interest.
- Supplementary File 1 Details and selection of the statistical models applied to the results of the root-inoculated experiment.
- Supplementary File 2 Details and selection of the statistical models applied for leaf vs control analysis.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Folgarait PJ, Goffré D, Rocano Domínguez HS. 2026. Beneficial effects of a multifunctional strain of Purpureocillium lilacinum in strawberry plants. Studies in Fungi 11: e003 doi: 10.48130/sif-0026-0001
Beneficial effects of a multifunctional strain of Purpureocillium lilacinum in strawberry plants
- Received: 20 July 2025
- Revised: 27 August 2025
- Accepted: 29 November 2025
- Published online: 31 January 2026
Abstract: The extensive use of agrochemicals poses significant risks to both environmental and human health. The fungus Purpureocillium lilacinum (Ascomycetes: Ophiocordycipitaceae), through its application, represents a promising biotechnological tool to reduce synthetic chemical inputs in crop production. The entomopathogenic and mycopathogenic capabilities of P. lilacinum strain YLOB have already been demonstrated, underscoring the relevance of multifunctional fungal strains in integrated pest and disease management. This study evaluated its potential to establish endophytically and promote growth in strawberry. In separate experiments, roots or leaves were inoculated with the fungus, and endophytic colonization, growth parameters, and final biomass were assessed. Purpureocillium lilacinum successfully colonized both inoculated and distant tissues. Leaf inoculation resulted in higher endophyte recovery from leaves compared to root inoculation at 7 d post-inoculation. Both inoculation treatments led to plants with significantly greater numbers of leaves and improved leaf health compared to controls. Additionally, leaf inoculation increased root biomass, while root inoculation improved flower and fruit production compared to controls. This is the first study demonstrating endophytic P. lilacinum benefits in strawberry plants, expanding its known functional scope and highlighting its potential application in strawberry production systems.













