-
Microbial metabolites hold significant potential across various fields, including agriculture, industry, and medicine[1,2]. From a medical perspective, these compounds have long been essential in disease treatment, with many microbial-derived products forming the basis of clinical drugs. Fungi, in particular, represent an exceptionally diverse group of organisms—estimated to comprise approximately ten million species, of which only a small fraction has been formally described[3]. This vast biodiversity correlates with a wide range of metabolic pathways, making fungi an important and underexplored resource for the discovery of novel bioactive compounds. Since the discovery of penicillin in 1928, fungal secondary metabolites have played a vital role in the development of new drugs due to their diverse and potent biological activities[4−6]. The urgent need for new antimicrobial agents has become increasingly apparent due to the rising incidence of multi-drug-resistant pathogens[7]. Saprobic fungi are particularly promising in this regard, as they naturally produce a wide array of secondary metabolites while decomposing organic matter. These compounds may function as defense mechanisms against competing microorganisms and are potentially valuable for pharmaceutical development[6,8]. Because of the vast diversity of fungal species and their metabolic pathways, fungi are thus expected to represent an important reservoir for the discovery of novel bioactive compounds.
Effective extraction of these metabolites typically involves solvent-based methods, where polarity plays a critical role in isolating specific classes of compounds[9]. In general, sequential extraction using different solvents such as petroleum ether, ethanol, and water can be designed to allow for the separation of metabolites based on their solubility profiles. These extracts can then be screened for biological activity, including antimicrobial properties. Thus, the exploration of fungal metabolite profiles through targeted extraction and activity screening is a vital strategy in the search for new therapeutic agents. In the present study, crude extracts from fourteen saprobic fungal isolates were prepared and subjected to sequential solvent partitioning using petroleum ether, ethanol, and water. The resulting fractions were then analyzed by spectroscopic techniques (FTIR and NMR) and screened for antimicrobial activity to assess their potential as sources of bioactive secondary metabolites.
-
Ten saprobic fungal strains were isolated from fallen leaves of Magnolia liliifera and Cinnamomum iners, as previously described by Monkai et al.[10]. An additional four fungal strains were obtained from the Collection of Microorganisms (DBM), Department of Biochemistry and Microbiology, University of Chemistry and Technology, Prague, Czech Republic. All fungal strains used in this study are listed in Table 1. Throughout the text, fungal isolates are referred to by both their genus name and strain number (e.g., Trichoderma sp. [no. 1]) on first mention in each section, and by strain number alone thereafter for brevity. Seven microbial strains from the DBM collection were used for antimicrobial assays. These included four Gram-positive bacteria (Bacillus cereus DBM 3035, Enterococcus faecalis DBM 3075, Micrococcus luteus DBM 3053, Staphylococcus aureus DBM 3002, and S. epidermidis DBM 3072), two Gram-negative bacteria (Enterobacter cloacae DBM 3126 and Pseudomonas aeruginosa DBM 3081), and one yeast strain (Saccharomyces cerevisiae DBM 2101).
Table 1. Specification of the saprobic fungal strains.
No. Fungal species Strains Sources 1 Trichoderma sp. DBM4197 UCT Prague, Czech Republic 2 Aspergillus oryzae DBM4336 UCT Prague, Czech Republic 3 Botrytis cinerea DBM4208 UCT Prague, Czech Republic 4 Stachybotrys chartarum DBM4297 UCT Prague, Czech Republic 5 Fusicoccum aesculi MFLU10-0260 MFLU Chiang Mai, Thailand 6 Fusicoccum aesculi MFLU10-0266 MFLU Chiang Mai, Thailand 7 Chaetomium sp. MFLU10-0761 MFLU Chiang Mai, Thailand 8 Ascomycete sp. 1 MFLU10-0282 MFLU Chiang Mai, Thailand 9 Clonostachys rosea MFLU10-0261 MFLU Chiang Mai, Thailand 10 Beltrania rhombica MFLU10-0293 MFLU Chiang Mai, Thailand 11 Stachybotrys parvispora MFLU10-0292 MFLU Chiang Mai, Thailand 12 Hyphomycetes sp. 1 MFLU10-0750 MFLU Chiang Mai, Thailand 13 Hyphomycetes sp. 2 MFLU10-0755 MFLU Chiang Mai, Thailand 14 Cladosporium sp. MFLU10-0280 MFLU Chiang Mai, Thailand Extraction and fractionation of fungal metabolites
-
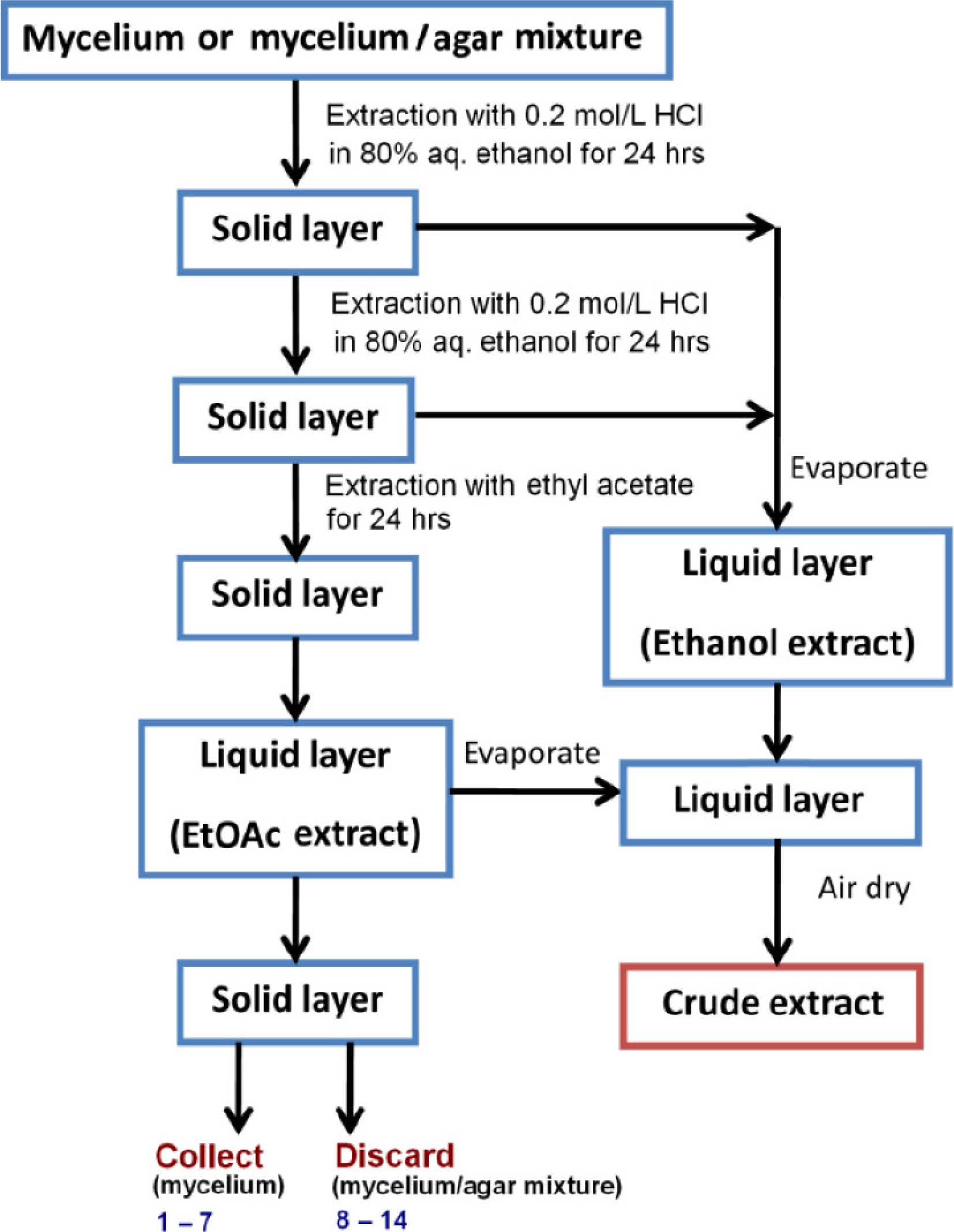
Fungal isolates were cultivated on Sabouraud dextrose agar (SDA) for 30 d at 28 °C. After incubation, the fungal biomass (either scraped from the agar or collected together with agar when necessary) was subjected to a three-step extraction process (Fig. 1). Crude mycelia were first mixed with 20 mL/g of 0.2 M hydrochloric acid in 80% aqueous ethanol. This step was designed to extract polar and moderately polar secondary metabolites efficiently[11]. The solid residues were pelleted by centrifugation, and the extraction was repeated with the same solvent to maximize recovery. A third extraction was performed using ethyl acetate at the same ratio to recover moderately non-polar compounds that might not be efficiently solubilized by acidic ethanol. All extraction steps were conducted by shaking at 150 rpm for 24 h at 28 °C.
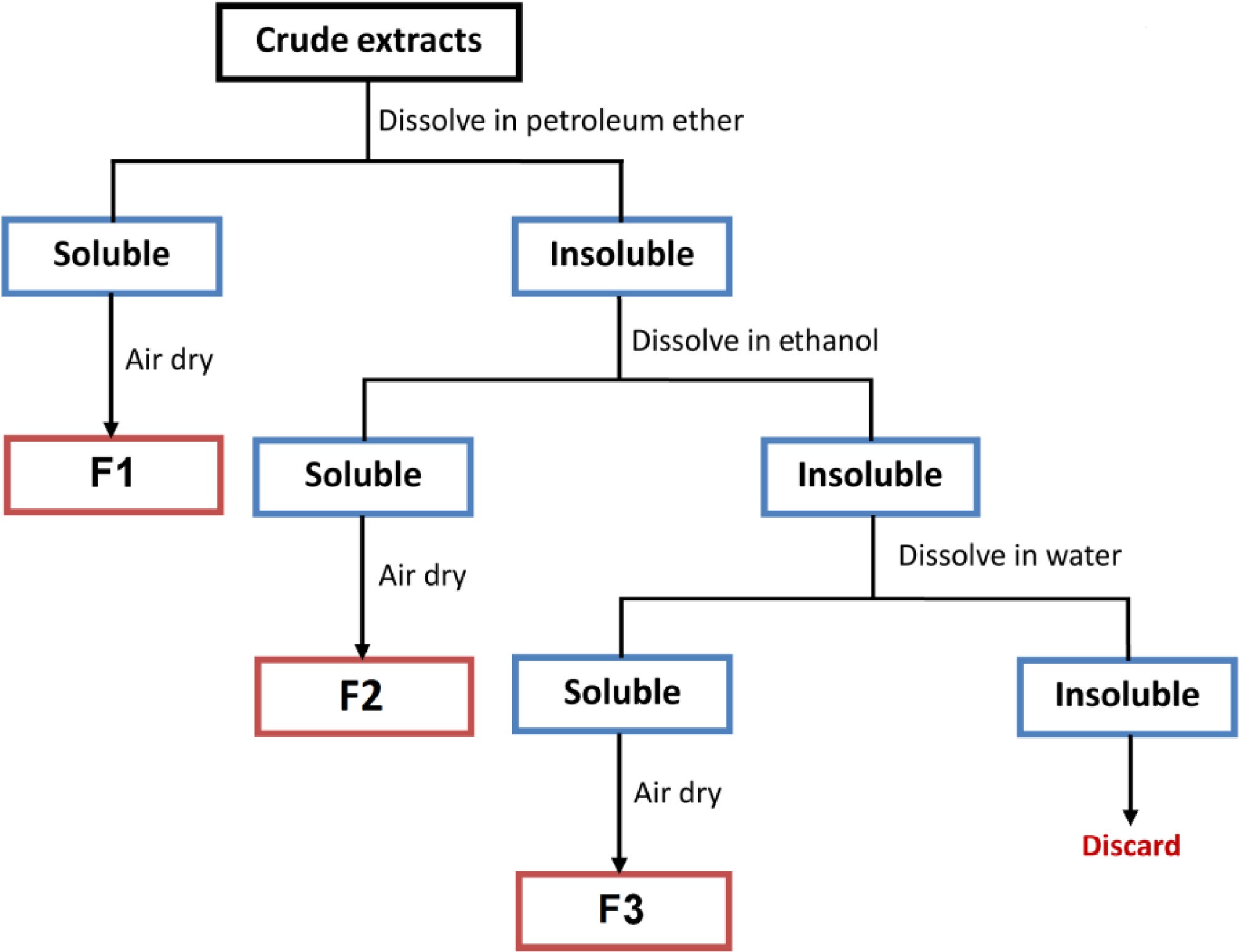
The combined extracts were partially evaporated using a rotary evaporator and then air-dried. The resulting crude extract was subsequently subjected to sequential solvent fractionation by washing with petroleum ether, ethanol, and water (Fig. 2) to separate metabolites based on polarity. This process yielded three fractions: F1 (petroleum ether-soluble), F2 (ethanol-soluble), and F3 (water-soluble). All fractions were air-dried, weighed, and stored at 15 °C until further analysis.
Antimicrobial activity assay
-
The antimicrobial activity of the fungal fractions was evaluated using a modified paper disk diffusion assay. Bacterial strains were cultivated in nutrient broth at 37 °C for 18 h, while S. cerevisiae was grown in potato dextrose broth at 30 °C for the same duration. Cell suspensions were adjusted to approximately 108 CFU/mL and uniformly spread on nutrient agar (NA) for bacteria and potato dextrose agar (PDA) for yeast.
Sterile paper disks (6 mm diameter) were loaded with 20 μl of F1 fraction (0.1 g/mL), air-dried, and placed on the agar surface. Equivalent volumes of F2 and F3 fractions were directly spotted onto the NA or PDA surface. The inoculated plates were incubated at 37 °C for bacteria and 30 °C for yeast for 24 h. Each test was performed in five replicates. The diameters of the inhibition zones were measured in millimeters.
Spectroscopic analysis
-
For FTIR analysis, approximately 5 mg of each dried fraction was mixed with 50 mg of potassium bromide (MERCK), ground in a mortar and pestle, and compressed into transparent tablets using a hand press. FTIR spectra were recorded using a Nicolet 6700 FTIR spectrometer (Thermo Scientific, USA) and processed with Omnic 8.0 software. Spectra were acquired over the 4,000–400 cm−1 range, with a resolution of 2 cm−1 and 64 scans per sample. Baseline correction and smoothing were applied.
Proton NMR (1H) spectra of selected F2 fractions with antimicrobial activity were recorded on a Bruker Avance III™ 600 MHz spectrometer (Bruker, Billerica, MA, USA) in CD3OD. Two-dimensional NMR experiments, including 1H–1H PFG-COSY and 1H–13C HMQC, were conducted to support signal assignment and compound identification.
-
Fungal mycelia developed on agar surfaces either as loose growth or as compact layers tightly embedded within the agar. In cases where the mycelium was inseparable from the agar, the entire mycelium-agar mixture was used for extraction. After the extraction process, intact mycelia were recovered, while the mycelium-agar mixtures were discarded (Fig. 1).
To improve the extraction of pigmented metabolites, which was observed to be more efficient under acidic conditions, 0.2 M hydrochloric acid in 80% aqueous ethanol was used instead of pure ethanol. Following solvent evaporation and air drying, the extracts yielded intensely colored crude products, which were then subjected to fractionation. Due to the variable water content and composition of the starting material (i.e., wet mycelia or mycelium-agar mixtures), it was difficult or impossible to reliably quantify the yield of crude extracts. Therefore, yield analysis focused on the subsequent fractions. Dried crude extracts were fractionated by sequential solubilization using solvents of increasing polarity: petroleum ether, ethanol, and water. The resulting fractions were labeled F1 (petroleum ether–soluble), F2 (ethanol–soluble), and F3 (water–soluble). The mass ratios of the obtained fractions are presented in Table 2, revealing considerable variation in yield across different fungal strains.
FTIR analysis
-
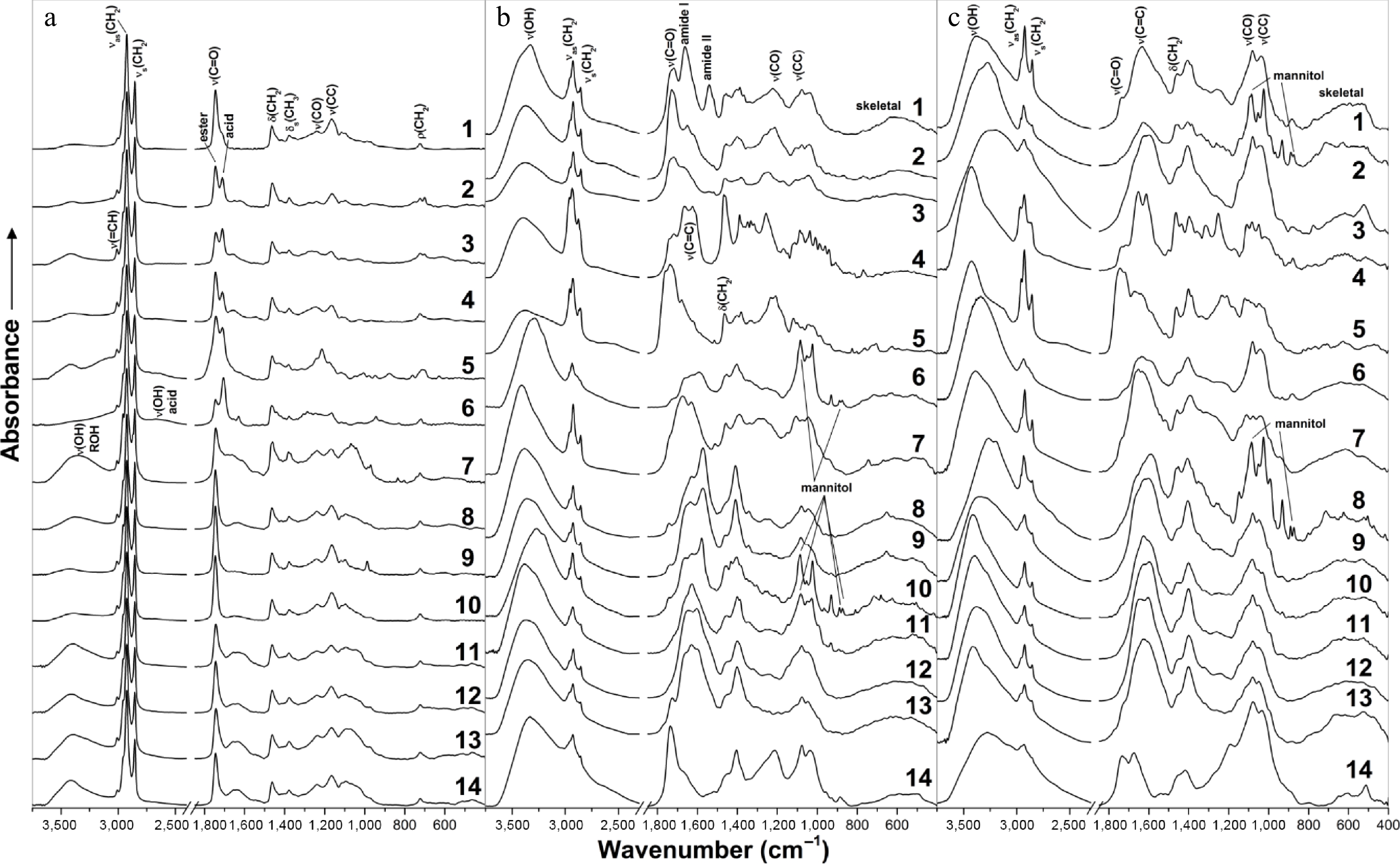
All crude extract fractions (F1, F2, and F3) were analyzed using Fourier-transform infrared (FTIR) spectroscopy to characterize their chemical composition.
F1 fractions: lipid-dominated profiles
-
FTIR spectra of F1 fractions (petroleum ether-soluble) revealed characteristic bands of lipids, primarily triacylglycerols and free fatty acids (Fig. 3a)[12,13]. Prominent methylene stretching vibrations were observed at 2,925 and ~2,854 cm−1, with additional CH2 bands at 1,462–1,465 cm−1 (scissoring) and ~722 cm−1 (rocking). Methyl group vibrations were less intense, with bands at ~2,955 (antisymmetric stretch), ~1,440 , and ~1,378 cm−1 (bending). Carbonyl stretching bands appeared at 1,742–1,746 cm−1 and 1,706–1,711 cm−1, corresponding to esters and free fatty acids, respectively. The ratio of these two bands reflected the relative abundance of fats vs free fatty acids and varied between fungal strains. For instance, esters predominated in F1 from fungi no. 8–13, while F1 from Fusicoccum aesculi MFLU10-0266 (fungus no. 6) contained more free fatty acids. Ester C–O stretching was also evident at 1,235–1,246 cm−1 and 1,163–1,169 cm−1.
Unsaturated fatty acid residues contributed a sharp C=H stretching band at ~3,009 cm−1 and a broad shoulder at 1,650–1,660 cm−1 (C=C stretching). Weaker bands at ~1,418 cm−1 and ~1,400 cm−1 were assigned to =CH bending and rocking vibrations. A broad OH stretching band between 3,396–3,441 cm−1 was associated with water or hydroxyl-containing compounds. Additionally, a broad band envelope from 1,130 to 1,000 cm−1, characteristic of sugar-related vibrations (C–O, C–C, and C–OH), confirmed the presence of glycolipids.
F2 and F3 fractions: complex mixtures
-
FTIR spectra of F2 (ethanol-soluble) and F3 (water-soluble) fractions were notably complex, reflecting their chemically diverse content (Fig. 3b, c). Bands between 2,800–3,100 cm−1 were indicative of aliphatic CH stretching, while strong absorption in the 1,400–1,800 cm−1 region suggested carbonyl (C=O) and unsaturated (C=C) functional groups.
Amide I and II bands (1,663 and 1,541 cm−1, respectively) were detected only in F2 from Trichoderma sp. DBM4197 (fungus no. 1), indicating the presence of peptides or proteins. Strong to moderate bands between 800–1,200 cm−1, associated with C–O and C–C skeletal vibrations, likely arose from sugars. However, this region also overlaps with aromatic and unsaturated =CH in-plane and out-of-plane bending vibrations.
Signatures of mannitol were identified in F2 (fungi no. 6, 10, and 11) and F3 (fungi no. 2 and 8) fractions, based on bands at 418, 476, 931, 1,025, and 1,086 cm−1. Additional mannitol-related vibrations were detected at 623–626, 712–716, 873–875, 890, 967–969, 1,052–1,054, and ~3,290 cm−1, although shifts were observed when compared with commercial β-mannitol[14]. These spectral variations likely reflect different crystalline polymorphs (α, β, δ) or interference from other fungal metabolites. Mannitol, a common fungal metabolite, plays multiple biological roles—as a storage carbohydrate, antioxidant, osmoregulator, and stress protectant[15,16]—and may comprise up to 10%–50% of fungal dry weight[17,18]. Its therapeutic applications include treatment of cerebral edema, renal protection during surgery, and management of rhabdomyolysis[19,20].
FTIR of residual mycelia: cell wall components
-
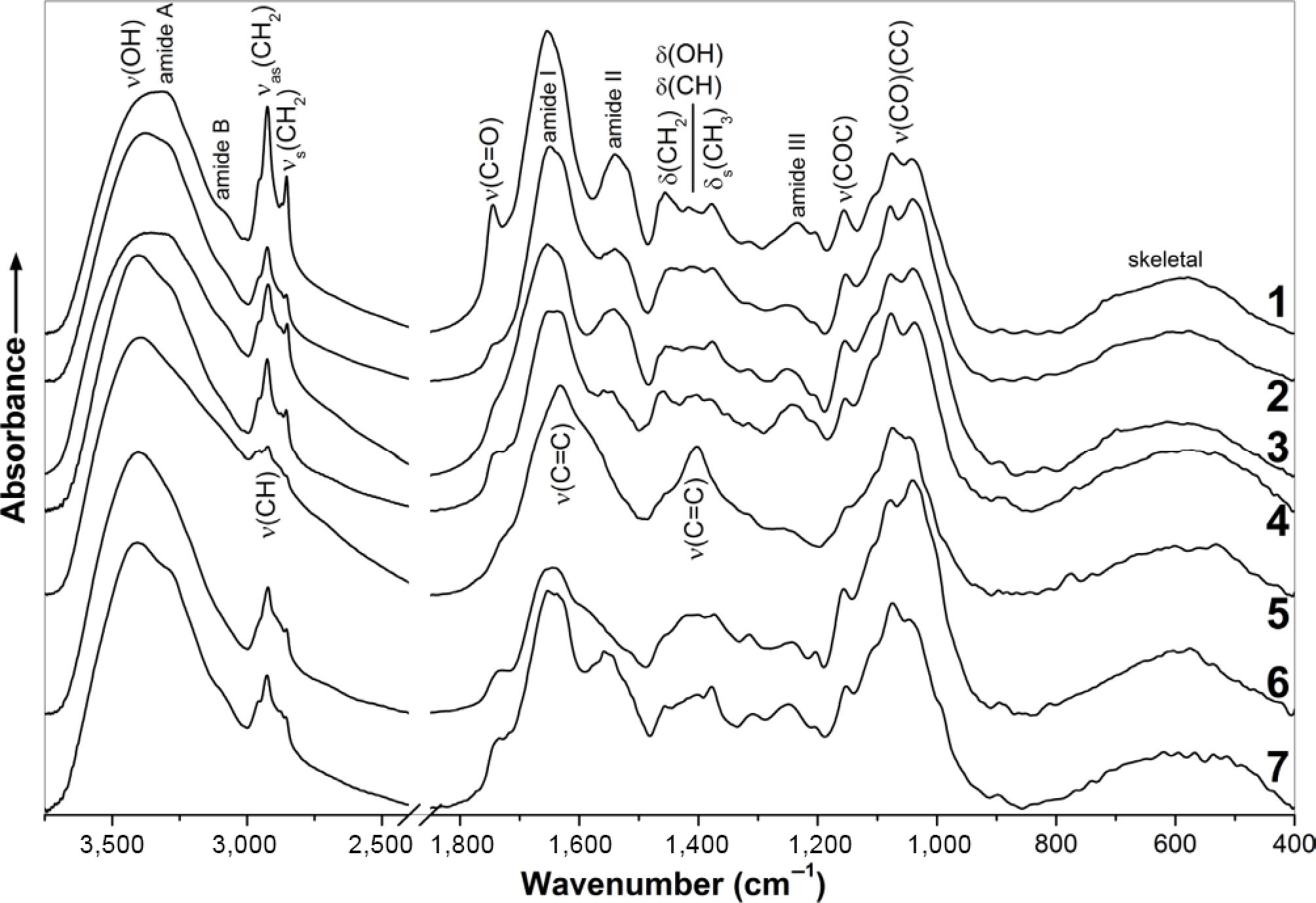
Pure mycelia from fungi no. 1–7 were collected post-extraction for FTIR analysis (Fig. 4). The spectra confirmed the presence of structural biopolymers, predominantly proteins and polysaccharides[21−23]. In Trichoderma sp. DBM4197 (fungus no. 1), strong protein-associated bands were observed at 3,316 cm−1 (amide A), 3,100 cm−1 (amide B, shoulder), 1,655 cm−1 (amide I), 1,541 cm−1 (amide II), and 1,234 cm−1 (amide III), along with lipid signals at 2,925 cm−1, 2,854 cm−1, 1,746 cm−1, and 1,456 cm−1. Polysaccharide-rich spectra (1,200–950 cm−1) were most prominent in F. aesculi MFLU10-0266 (fungus no. 6), in Stachybotrys chartarum DBM4297 (fungus no. 4), and Chaetomium sp. MFLU10-0761 (fungus no. 7), the amide I band appeared split (~1,656 and 1,623–1,626 cm−1), and the amide II band was shifted to 1,558–1,560 cm−1—features characteristic of α-chitin, a major component of fungal cell walls[24,25]. Additional features of β-polysaccharides (e.g., chitin, β-glucans) were identified at 891–899, 1,377–1,380, 1,316–1,320, ~1,110, and ~1,040 cm−1. Notably, the mycelium of F. aesculi MFLU10-0260 (fungus no. 5) showed prominent bands at 1,631 and 1,402 cm−1, indicative of aromatic C=C stretching. These findings suggest that residual mycelia primarily consist of cell wall polysaccharides, with varying contributions from proteins, lipids, and aromatic compounds across fungal species.
Antimicrobial activity
-
Fractions F1, F2, and F3 were derived from dried crude fungal extracts through sequential extraction with petroleum ether, ethanol, and water, respectively. This stepwise fractionation aimed to maximize recovery of both hydrophobic and hydrophilic compounds. The antimicrobial activity of the resulting fractions was evaluated in vitro using a modified disc diffusion method against seven test microorganisms. The results are summarized in Table 2.
Table 2. Antimicrobial activities of the fractions of crude fungal extracts against selected microorganisms.
No Fraction Ratioa Antimicrobial activityb Gram-positive Gram-negative BC EF ML SA SE EC PA SC 1 F1 0.21 5.8 ± 1.48 − 3.6 ± 1.14 − 1.2 ± 0.45 − − − F2 0.33 5.4 ± 1.14 − 12.6 ± 1.95 − 1.6 ± 0.55 7.6 ± 1.95 5.6 ± 1.52 − F3 0.46 − − + − − − − − 2 F1 0.12 21.0 ± 2.65 2.8 ± 0.84 3.6 ± 0.89 13.4 ± 2.14 1.8 ± 0.84 − − − F2 0.44 11.4 ± 1.95 1.2 ± 0.45 1.2 ± 0.45 11.6 ± 1.67 7.6 ± 1.67 1.4 ± 0.55 1.0 ± 0.00 − F3 0.44 − − − − − − − − 3 F1 0.08 − − 2.2 ± 0.45 − − − − − F2 0.14 9.6 ± 1.67 5.6 ± 0.55 4.8 ± 0.45 5.0 ± 0.71 6.2 ± 1.10 5.4 ± 1.52 5.0 ± 0.00 − F3 0.78 − − − − − − − − 4 F1 0.18 − 3.6 ± 0.89 − 5.4 ± 0.89 − − − − F2 0.80 6.2 ± 1.10 5.4 ± 1.52 2.8 ± 0.45 6.4 ± 0.89 2.0 ± 0.00 6.4 ± 1.34 6.2 ± 0.45 − F3 0.02 10.0 ± 0.00 − − − − − − − 5 F1 0.07 − − 1.0 ± 0.00 − 1.0 ± 0.00 − − − F2 0.73 5.6 ± 0.55 − 5.0 ± 0.00 − − 6.0 ± 0.71 9.0 ± 1.00 − F3 0.21 − − − − − − − − 6 F1 0.11 − − − − − − − − F2 0.75 7.0 ± 0.00 − − − − 5.0 ± 0.00 − − F3 0.14 4.6 ± 0.55 − − 2.0 ± 0.00 − − − − 7 F1 0.09 7.6 ± 1.34 − − − − − − − F2 0.66 5.6 ± 0.55 − − − − − − − F3 0.25 − − − − 9.4 ± 0.89 1.0 ± 0.00 1.8 ± 0.45 − 8 F1 0.34 − − − 6.8 ± 1.10 − − − − F2 0.11 − − − − − − − − F3 0.54 − − − − − − − − 9 F1 0.21 5.0 ± 0.00 − − − 2.0 ± 0.00 − − − F2 0.46 10.8 ± 1.10 5.0 ± 0.00 2.0 ± 0.00 4.6 ± 0.55 9.6 ± 0.55 6.0 ± 1.22 − 4.6 ± 0.55 F3 0.32 2.8 ± 0.45 − − 4.4 ± 0.55 5.0 ± 1.00 − − − 10 F1 0.39 − − − − − − − − F2 0.24 7.0 ± 1.22 − − 5.8 ± 0.45 5.0 ± 0.00 − − − F3 0.37 − − − − − − − − 11 F1 0.24 − 5.0 ± 0.00 1.0 ± 0.00 6.0 ± 0.71 2.0 ± 0.00 − − − F2 0.50 12.2 ± 1.79 3.0 ± 0.00 4.8 ± 0.84 5.0 ± 0.00 6.2 ± 0.45 − 3.0 ± 0.00 − F3 0.26 − − − − − − − − 12 F1 0.24 − − − − − − − − F2 0.57 − − 5.0 ± 0.00 − − − 1.0 ± 0.00 − F3 0.18 − − 2.8 ± 0.45 3.0 ± 0.00 − − − − 13 F1 0.19 − − − − − − − − F2 0.53 5.8 ± 0.45 − 2.0 ± 0.00 5.2 ± 0.45 − − − − F3 0.29 − − − 5.0 ± 0.00 − − − − 14 F1 0.11 − 5.0 ± 0.71 − − − − − − F2 0.41 7.4 ± 0.89 5.0 ± 0.00 2.0 ± 0.00 5.4 ± 0.55 5.0 ± 0.00 − 2.0 ± 0.00 − F3 0.48 − − − − − − − − a Ratio of the mass of this fraction to the total mass of all isolated factions; + = presence of clear zone, − = absence of clear zone. b BC = B. cereus, EF = E. faecalis; ML = M. luteus; SA = S. aureus, SE = S. epidermidis, EC = E. cloacae, PA = P. aeruginosa, SC = S. cerevisiae. To enable direct comparison of potency among fractions, representative quantitative inhibition values are highlighted here. The ethanol-soluble F2 fractions consistently produced the largest inhibition zones, often exceeding 10 mm. For example, Trichoderma sp. DBM4197 (fungus no. 1) showed a 12.6 mm zone against Micrococcus luteus, while Clonostachys rosea MFLU10-0261 (fungus no. 9) exhibited 10.8 mm inhibition against Bacillus cereus and 9.6 mm against Staphylococcus epidermidis. Similarly, Stachybotrys parvispora MFLU10-0292 (fungus no. 11) produced a substantial 12.2 mm zone against B. cereus. In contrast, fractions showing only weak inhibition typically generated zones of 1–4 mm—for example, the F2 fraction of Beltrania rhombica MFLU10-0293 (fungus no. 10) produced 5.0 mm zones for S. epidermidis but showed no activity against Gram-negatives. These representative values demonstrate clear differences in antimicrobial strength, supporting the conclusion that F2 fractions generally possess the highest potency.
Overall, many fungal extracts exhibited antimicrobial activity, with F2 fractions (ethanol-soluble) exhibiting the most pronounced effects across the majority of strains tested. This indicates that the active compounds are generally polar in nature. One notable exception was Aspergillus oryzae DBM4336 (fungus no. 2), whose F1 fraction (petroleum ether extract) inhibited the growth of all tested Gram-positive bacteria. Additionally, its F2 fraction displayed strong antimicrobial activity against Bacillus cereus, Staphylococcus aureus, and Staphylococcus epidermidis.
Significant antimicrobial potential was also observed in the F2 fractions of Trichoderma sp. DBM4197 (fungus no. 1), Botrytis cinerea DBM4208 (no. 3), and Stachybotrys chartarum DBM4297 (no. 4), which were active against both Gram-positive and Gram-negative bacteria. Similarly, broad-spectrum activity was noted in the F2 fractions of Clonostachys rosea MFLU10-0261 (fungus no. 9), Stachybotrys parvispora MFLU10-0292 (no. 11), and Cladosporium sp. MFLU10-0280 (no. 14). Additionally, F2 fractions from Fusicoccum aesculi MFLU10-0260 (fungus no. 5) and Beltrania rhombica MFLU10-0293 (no. 10) also demonstrated antimicrobial effects, particularly against Gram-positive bacteria. In contrast, their F1 and F3 fractions exhibited little or no inhibitory activity.
NMR analysis of chosen bioactive fractions
-
Proton NMR spectra of the bioactive F2 fractions—derived from Trichoderma sp. DBM4197 (no. 1), Clonostachys rosea MFLU10-0261 (no. 9), and Stachybotrys parvispora MFLU10-0292 (no. 11)—showed intense signals corresponding to residual solvents, including water, methanol, ethanol, and acetic acid, likely originating from the extraction process and NMR solvents. Consistent with FTIR data, mannitol was a major component of F2 fractions from strains 9 and 11 (Fig. 5a), but absent in strain 1. Trehalose, a common fungal disaccharide, was detected in all samples, while free glucose was observed in strains 1 and 11; strain 11 additionally contained β-D-glucoside (Fig. 5b). All three F2 fractions exhibited signals corresponding to aromatic, unsaturated, and saturated protons. The intensity of aromatic proton signals increased progressively from strain 9 to 11 to 1 (Fig. 5c), suggesting a higher abundance of aromatic structures in Trichoderma sp. DBM4197. Numerous singlet aromatic proton signals across all spectra indicate the presence of highly substituted carbo- and heterocyclic systems, which are characteristic motifs in metabolites of Trichoderma[26] and related Hypocreales genera[27].
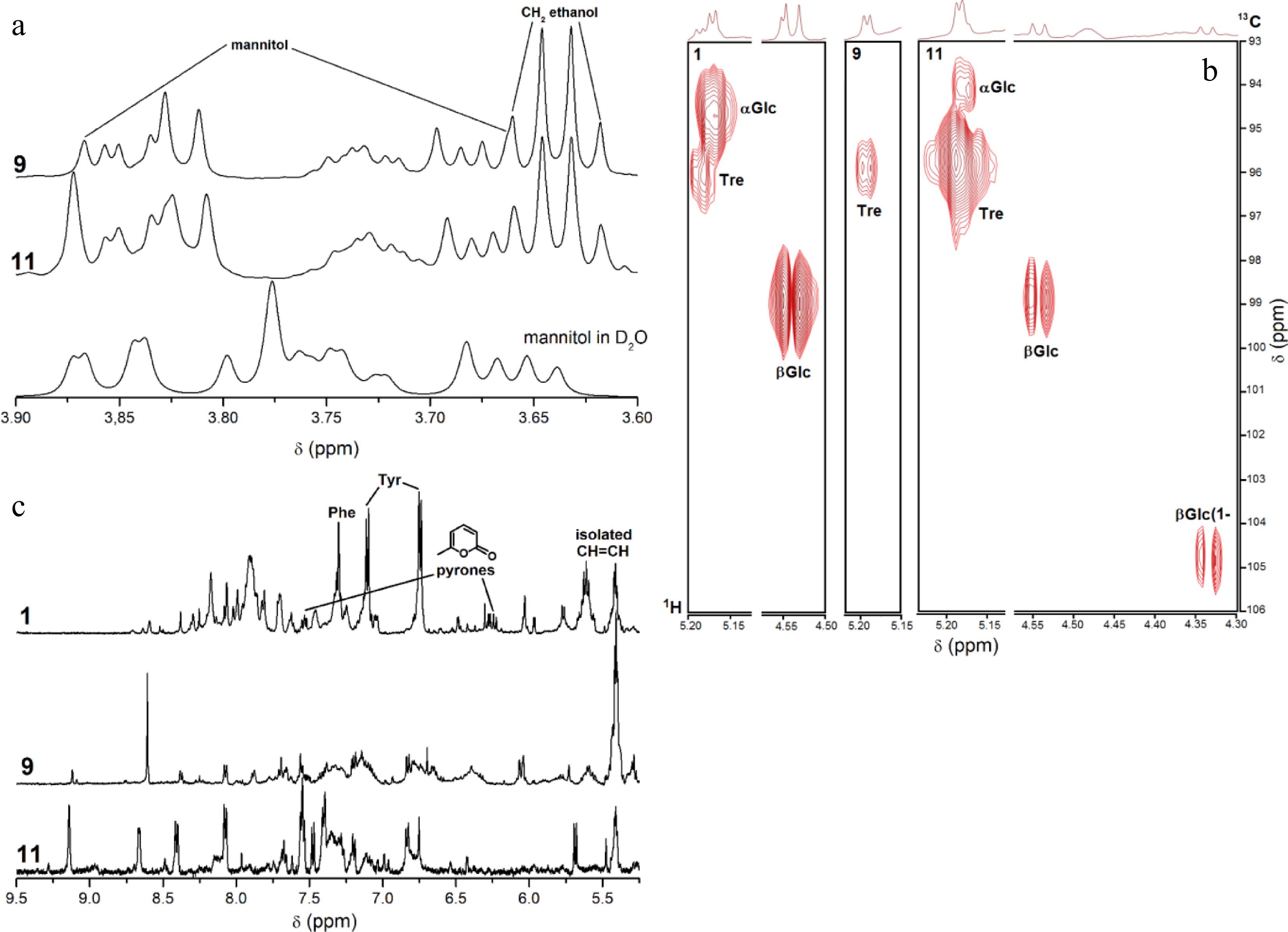
Figure 5.
1H NMR and 1H, 13C HMQC spectra (600 MHz, CD3OD) of fractions F2 from the selected strains: the regions of (a) OCH, OCH2 protons in comparison with pure mannitol measured in D2O, (b) anomeric protons/carbons and aromatic protons. (c) Key signals of NMR spectra in the F2 fractions from Trichoderma sp. DBM4197 (no. 1) were annotated: aromatic protons (δ 7.15–7.35 ppm, Tyr/Phe), pyrone ring protons (δ 6.21 and 6.45 ppm, 6-substituted pyrones), olefinic protons (δ 5.80–6.00 ppm).
For strain 1, key structural assignments are supported by distinctive chemical shifts: singlets at δ 6.21 and δ 6.45 ppm correspond to protons on 6-substituted pyrone rings, consistent with previously reported data for 6-pentyl-2H-pyran-2-one (6PP) derivatives[28]. Aromatic proton signals in the δ 7.15–7.35 ppm range, combined with strong COSY correlations, correspond to para-substituted and unsubstituted phenyl rings and are attributable to tyrosine (Tyr) and phenylalanine (Phe) residues characteristic of viridiofungins[29]. Collectively, these observed δ values and spin systems provide direct spectroscopic evidence supporting the presence of viridiofungin-like compounds and 6-substituted pyrones in the fraction from strain 1.
F2 fractions from strains 9 and 11 displayed similar aromatic patterns and spin systems; however, aromatic amino acids were not detected in these fractions. This suggests the presence of other structurally diverse aromatic or sugar-linked secondary metabolites. Overall, the NMR data confirm that F2 fractions from these saprobic fungi are chemically complex mixtures enriched in bioactive components, including known antifungal and antibacterial metabolites, while also indicating potential novel analogues or previously undescribed compounds.
The NMR spectra of F2 fractions revealed prominent resonances corresponding to aromatic protons (δ 6.2–7.3 ppm) and sugar-like anomeric signals (δ 3.2–4.8 ppm), indicating the presence of both aromatic and carbohydrate-derived components. These observations are consistent with the FTIR spectra, which showed characteristic absorptions for C=C stretching of aromatic rings (~1,600 cm−1) and C–O–C/C–OH vibrations (1,050–1,250 cm−1), confirming that F2 contains complex mixtures of aromatic and polysaccharide-like structures. In contrast, the FTIR spectrum of F1 exhibited strong C–H stretching bands (2,850–2,930 cm−1), supporting the predominance of aliphatic lipid constituents and aligning with the absence of aromatic signals in its NMR spectrum. Together, these complementary analyses demonstrate that the fungal fractions are chemically distinct: F1 is lipid-rich, while F2 (and F3) are enriched in aromatic and polar metabolites.
-
The use of acidic ethanol improved the extraction of pigmented metabolites, likely favoring solubilization of phenolic, aromatic, or conjugated compounds that might not dissolve well in neutral ethanol. Sequential extraction (from nonpolar to intermediate to polar) is a classic approach to fractionate complex mixtures[9]; here it succeeded in partitioning lipids (F1) from more polar metabolites (F2, F3). The variable yields of fractions among fungal strains reflect inherent metabolic diversity and differential partitioning of secondary metabolites.
FTIR data confirmed that F1 was largely composed of lipids, fatty acids, and glycolipids, while F2/F3 were more chemically heterogeneous (sugars, sugar alcohols, proteins/peptides, aromatic compounds). The identification of mannitol, trehalose, and glucose is consistent with well-known fungal osmolytes and storage compounds[15,16]. Mannitol can especially account for substantial fungal dry weight (10%–50%)[17,18]. In many fungi, mannitol functions not only as a carbon reserve but also as an osmoprotectant and antioxidant[15]. Its prevalence here is not surprising. Residual mycelial FTIR also confirmed that structural polymers—chitin, β-glucans, structural proteins—persist post-extraction. Observed α-chitin features (amide I splitting, shifted amide II) align with prior studies[24,25]. The detection of aromatic signals in residual mycelium (e.g., in F. aesculi) suggests possible phenolic or aromatic cross-linking in cell walls.
Linking chemical profiles to antimicrobial activity
-
The ethanol-soluble F2 fractions showed superior antimicrobial activities in most strains, suggesting that active compounds generally have intermediate polarity (neither fully hydrophobic nor purely water-soluble). In contrast, F1 and F3 fractions were less active, indicating that extreme polarity (water-soluble) or nonpolarity (lipid fraction) is less favorable for antimicrobial potency in these fungi, at least under the tested conditions.
The notable exception (A. oryzae F1 activity against Gram-positive bacteria) highlights that some potent lipophilic metabolites (e.g., nonpolar toxins, membrane-active compounds) may reside in F1 for certain species. A. oryzae is known to produce a range of bioactive compounds, including animal-toxic mycotoxins[30], extracellular antimicrobial proteins[31], and indoloditerpenes with antimicrobial and insecticidal activity[32].
The strong activity of the fungal F2 extracts is consistent with existing literature: Trichoderma species are prolific producers of biologically active secondary metabolites[26]; B. cinerea synthesizes antibiotics such as botrydial and dihydrobotrydial with activity against fungi and Gram-positive bacteria[33,34]; and S. chartarum, often found in damp indoor environments, produces a variety of bioactive compounds including trichothecenes, triprenylated phenolics, and atranones[35]. C. rosea is an entomopathogenic saprobe known to produce volatile organic compounds[36,37] and peptaibols, which are effective antagonists of soil-borne pathogens like Sclerotinia sclerotiorum[23,38]. Compounds such as stachybotrin C and parvisporin, isolated from S. parvispora, have shown cell survival activity[39]. Cladosporium sp. is recognized for producing cladospolides, a group of cyclic antibiotics[40].
B. rhombica has previously been reported to produce a wide range of antimicrobial compounds with broad-spectrum activity, including against Gram-positive and Gram-negative bacteria, yeast, and phytopathogenic fungi[41]. Rukachaisirikul et al. reported the isolation of two novel eudesmane sesquiterpenes—rhombidiol and rhombitriol—as well as five enantiomeric sesquiterpenes from this species[42]. These findings underscore the potential of saprobic fungi as sources of novel antimicrobial agents, particularly ethanol-soluble secondary metabolites.
Candidate metabolites: viridiofungins, 6PP, and others
-
The NMR evidence (aromatic proton singlets, COSY connectivity, presence of citrate-like spin systems, unsaturated/heterocyclic moieties) strongly suggests that the F2 fractions—especially from Trichoderma—contain viridiofungins. Viridiofungins are aminoacyl alkyl citrate derivatives first isolated from Trichoderma viride[43]. They exhibit potent antifungal activity (MFC in µg/mL range) and inhibit serine palmitoyltransferase (SPT), a key enzyme in sphingolipid biosynthesis, thereby disrupting fungal cell membrane integrity[43]. The presence of citric acid moieties in the NMR is consistent with this class.
Additionally, detection of a 6-substituted pyrone ring suggests 6-pentyl-2H-pyran-2-one (6PP) or analogs. 6PP is a well-characterized volatile secondary metabolite from Trichoderma spp.[26,44]. It has broad antifungal and antibacterial properties, inhibits spore germination, and can act at low concentrations. Some recent studies (e.g., in Trichoderma erinaceum and T. atroviride) show 6PP affects pathogen metabolism (e.g., amino acid pathways, induction of autophagy) and even modulates the physiology of plant hosts[28].
Hence, it is plausible that the observed antimicrobial activity arises from a mixture of viridiofungins, 6PP (or related α-pyrones), and perhaps other aromatic polyketides, peptides, or small terpenes. Given that the aromatic signal intensity was highest in Trichoderma sp. (no. 1), this strain may produce a richer complement of these metabolites.
-
This study demonstrated that saprobic fungi are valuable reservoirs of antimicrobial secondary metabolites, with several strains exhibiting strong and broad-spectrum bioactivities. Among the tested isolates, Trichoderma sp. (no. 1), Clonostachys rosea (no. 9), and Stachybotrys parvispora (no. 11) emerged as the most potent sources of antimicrobial compounds. Their ethanol-soluble (F2) fractions showed the highest activity, quantitatively inhibiting both Gram-positive and Gram-negative bacteria as well as yeast, indicating the presence of bioactive metabolites with intermediate polarity. Spectroscopic analyses (FTIR and NMR) revealed that these active fractions contained multiple classes of secondary metabolites, including aromatic and heterocyclic compounds, citrate derivatives, and 6-substituted pyrone structures. These features are consistent with known antifungal and antibacterial metabolites such as viridiofungins and 6-pentyl-2H-pyran-2-one (6PP), commonly found in Trichoderma and related genera. However, the diversity and complexity of the spectra, together with unique resonance patterns not fully matching known references, suggest that these fungi may also produce novel structural analogues or previously undescribed compounds. The high antimicrobial activity of Trichoderma sp. (no. 1) highlights this strain as a promising candidate for isolating new viridiofungin-like and pyrone-derived metabolites. Similarly, the bioactive fractions of C. rosea (no. 9) and S. parvispora (no. 11) contained mannitol-rich but chemically diverse profiles, indicating potential co-production of sugar-linked or aromatic secondary metabolites. These findings reinforce the potential of saprobic fungi as rich and underexplored sources of structurally diverse antimicrobial agents. Future research should focus on large-scale cultivation and purification of these bioactive fractions, followed by comprehensive structural elucidation using LC-HR-MS/MS and NMR. Such analyses may reveal new analogues of viridiofungins, substituted pyrones, or entirely novel metabolite classes with pharmaceutical potential.
This work was financially supported by Mae Fah Luang University, Thailand (Grant No. 56101020033) and Institute of Chemical Technology, Czech Republic by the 'Operational Program Prague – Competitiveness' (Grant No. CZ.2.16/3.1.00/24503) and the 'National Program of Sustainability I' – NPU I (LO1601 - Grant No. MSMT-43760/2015).
-
The authors confirm their contributions to the paper as follows: study conception and design, review and editing: Chukeatirote E, Synytsya A, Ruml T; data collection, analysis and interpretation of results, and draft manuscript preparation: Monkai J, Synytsya A, Bleha R. All authors reviewed the results and approved the final version of the manuscript.
-
All data generated or analyzed during this study are included in this published article [and its supplementary information files].
-
The authors declare that they have no conflict of interest.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Chukeatirote E, Monkai J, Synytsya A, Bleha R, Ruml T. 2026. Characterization and antimicrobial screening of secondary metabolite fractions from saprobic fungi. Studies in Fungi 11: e004 doi: 10.48130/sif-0026-0002
Characterization and antimicrobial screening of secondary metabolite fractions from saprobic fungi
- Received: 01 August 2025
- Revised: 14 November 2025
- Accepted: 18 December 2025
- Published online: 31 January 2026
Abstract: Fungal secondary metabolites are a rich source of bioactive compounds with significant pharmaceutical and agricultural potential, particularly for their antimicrobial properties. In this study, secondary metabolite fractions (F1, F2, and F3) were isolated from fourteen strains of saprobic fungi through a three-step solvent extraction using petroleum ether, ethanol, and water. The resulting fractions, soluble in their respective solvents, were analyzed by FTIR spectroscopy, with selected samples further characterized by NMR. Antimicrobial assays were conducted against seven pathogenic microorganisms. Among the tested fractions, the ethanol-soluble fractions (F2) generally exhibited the highest antimicrobial activity. Notably, F2 fractions from Trichoderma sp. DBM4197, Clonostachys rosea MFLU10-0261, and Stachybotrys parvispora MFLU10-0292 showed broad-spectrum inhibition against both Gram-positive and Gram-negative bacteria, as well as fungi. NMR analysis confirmed the presence of bioactive compounds, such as 6-substituted pyrenes and viridiofungins, in the F2 fraction of Trichoderma sp., which are known for their antimicrobial properties. These results indicate that the studied saprobic fungi are promising sources of secondary metabolites with potential for development into bactericidal agents.
-
Key words:
- Antimicrobial activity /
- Fungi /
- Saprobe /
- Secondary metabolites