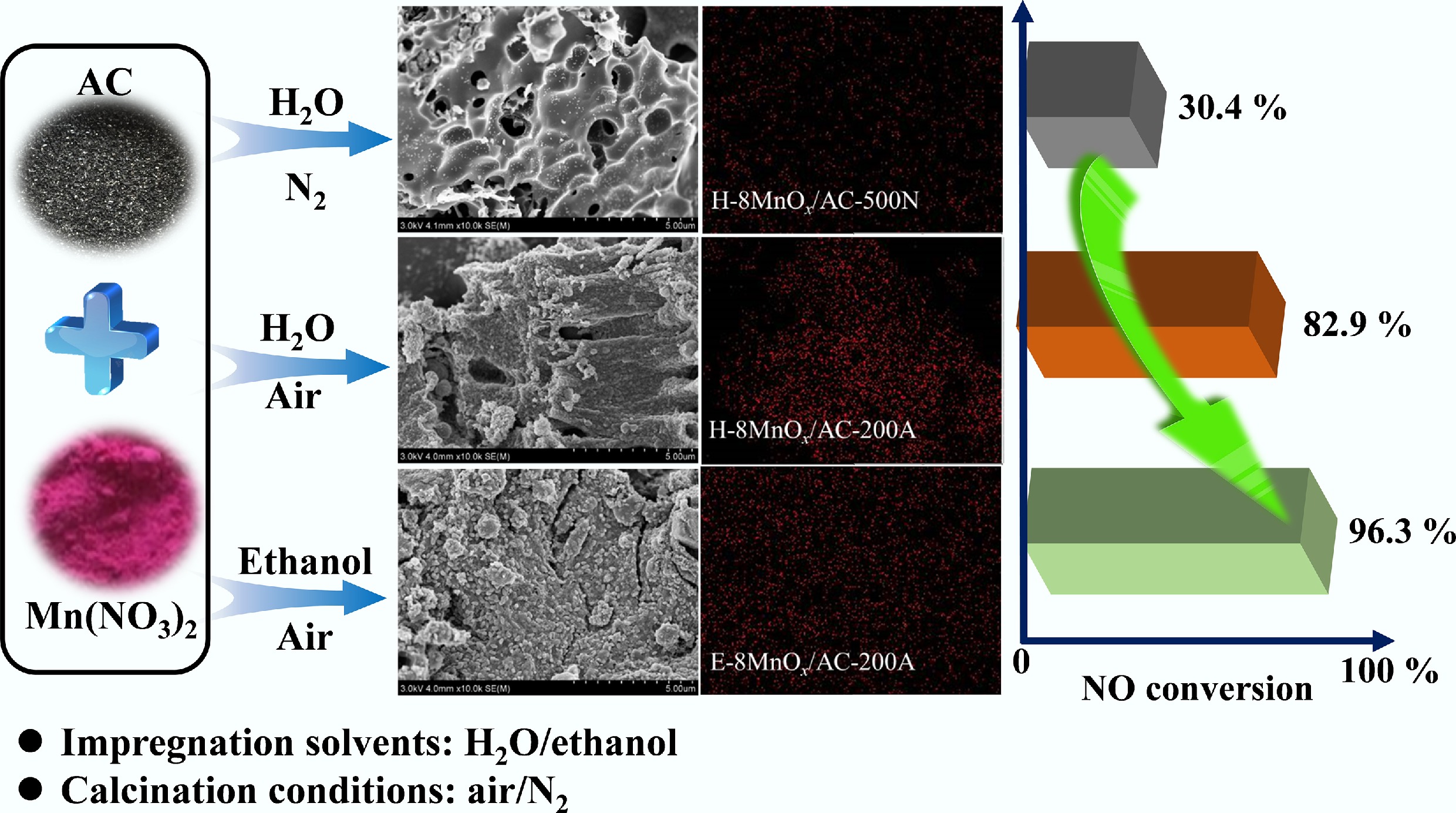
-
The excessive release of nitrogen oxides (NOx) contributes to photochemical smog, acid rain, and other forms of pollution, posing substantial threats to ecosystems and public health. NH3-selective catalytic reduction (NH3-SCR) technology is the most widely used approach for controlling NOx emissions from coal-fired power plants. This technology relies on the use of high-efficiency deNOx catalysts, with vanadium–titanium catalysts currently being the most prevalent in industrial NH3–SCR applications[1]. However, vanadium–titanium catalysts present several challenges and limitations. One primary concern is the high biological toxicity of V2O5, which can sublimate and detach during operation, potentially diffusing into the environment through flue gases. Furthermore, the effective temperature window of vanadium–titanium catalysts is narrow, with high catalytic activity typically achieved only between 300 and 400 °C[2]. To meet the effective temperature requirements, vanadium–titanium catalysts are typically placed upstream of the dust removal and desulfurization units. This positioning makes them susceptible to deactivation and blockage through exposure to particulates and sulfur compounds. Alternatively, if placed downstream of these units, the flue gas temperature is often too low, necessitating reheating to achieve effective catalytic performance, a process that significantly increases energy consumption. In industries such as glass manufacturing, coke production, cement manufacturing, and steel production, flue gas temperatures are typically below 300 °C. As environmental regulations become increasingly stringent, there is a growing demand for low-temperature deNOx catalysts in these sectors. Considering the limitations of vanadium–titanium catalysts in treating low-temperature flue gases, there is an urgent need to develop novel catalysts specifically designed for low-temperature deNOx applications.
Metal oxide catalysts have attracted significant attention because of their excellent low-temperature deNOx performance, low cost, and ease of fabrication[3,4]. The widely used metal oxides include MnOx, CuOx, FeOx, CeOx, and VOx. Among them, MnOx stands out for its versatile valence states, excellent lattice oxygen mobility, and abundance of surface acidic sites, demonstrating remarkable performance in low-temperature deNOx applications[5]. When comparing equal amounts of metal oxides, their deNOx capabilities follow this descending order: MnOx > CuOx ≈ CeOx > VOx > FeOx[6]. MnOx-based catalysts can be categorized into supported and unsupported types[7]. Supported MnOx-based catalysts have gained widespread attention in academia and industry because of their reduced agglomeration of active species, efficient use of active sites, and potential for significant cost savings. Common catalyst supports include activated carbon (AC), polymers, and molecular sieves. Among them, AC offers a high specific surface area and abundant functional groups[8−10], which enhance the dispersion of active components and confer significant advantages as a catalyst support for low-temperature deNOx. Therefore, the preparation of MnOx/AC catalysts using AC as the support shows great potential for practical applications[11,12].
The dispersion of MnOx on the AC support and the valence state of Mn are critical factors influencing the catalytic efficiency of MnOx/AC catalysts. The large specific surface area of AC provides ample space for loading and dispersing active components, as well as for adsorbing gas molecules and facilitating catalytic reactions. The uniform dispersion of the active components ensures thorough contact with NH3 and NOx, minimizing the formation of undesirable byproducts and enhancing low-temperature deNOx efficiency. The catalytic performance varies significantly among Mn species with different valence states, with Mn4+ in MnOx exhibiting the highest deNOx activity, followed by Mn5O8, Mn2O3, Mn3O4, and MnO. Therefore, achieving evenly dispersed active components and a high Mn4+ content is crucial for fabricating high-performance low-temperature deNOx catalysts[13]. Conventional MnOx/AC catalysts are typically prepared by impregnating AC with Mn-containing aqueous solutions, followed by calcination in a nitrogen atmosphere at temperatures ranging from 400 to 600 °C. However, this method often results in uneven MnOx dispersion and a lower ratio of active Mn4+, which compromises the catalytic efficiency. The poor dispersibility of MnOx can be attributed to the relatively high surface tension of water. Since the wetting effect of solvents reduces the surface energy of the target substance and facilitates effective diffusion, lower surface tension generally improves the wettability. Previous studies have demonstrated that ethanol, when used as an impregnation solvent, offers significant advantages in enhancing the uniformity of metal oxides' loading on support surfaces[14−16]. Ling et al.[17] prepared CuO–ZnO–ZrO/Al2O3 catalysts and compared the effects of water and ethanol as impregnation solvents. The results showed that ethanol markedly improved the dispersion of the active components, thereby enhancing the catalytic efficiency of the catalyst. Additionally, during the high-temperature calcination process used to prepare MnOx/AC catalysts, the reduction reaction between high-valence Mn and carbon reduces Mn4+ to lower valence states, potentially diminishing the SCR activity. To address this issue, Liu et al.[18] prepared MnOx/AC catalysts via low-temperature air calcination, which increased the proportion of Mn4+ and generated abundant chemisorbed oxygen, resulting in improved deNOx performance. These findings suggest that optimizing the Mn impregnation conditions and utilizing low-temperature air calcination can effectively improve the dispersion of the catalyst's active component(s), increase the Mn4+ content, and enhance the catalytic performance.
Herein, a novel method was proposed for preparing deNOx catalysts by enhancing Mn loading using ethanol (a solvent with low surface tension) combined with low-temperature air calcination. Characterization of the catalysts and laboratory-scale deNOx tests were systematically conducted to investigate the effects of the impregnation solvents and calcination conditions on the physicochemical properties and deNOx performance of the catalysts. Ultimately, the optimal catalyst preparation conditions were identified.
-
AC derived from coconut shells was purchased from Green Forest Activated Carbon Factory in Pingdingshan, Henan. After being crushed and sieved to a particle size range of 0.42–0.85 mm, the AC was dried at 105 °C for 8 h. Mn(NO3)2 was obtained from China National Pharmaceutical Group Co., Ltd., and ethanol was procured from Shanghai Macklin Biochemical Technology Co., Ltd.
Preparation of the catalysts
-
Initially, an aqueous or ethanolic Mn(NO3)2 solution with a specific concentration was prepared. Subsequently, 4 g of AC was immersed in the solution and subjected to ultrasonic treatment (effective ultrasonic power, 300 W; ultrasonic frequency, 40 kHz) at room temperature for 2 h to produce the catalysts' precursor. The precursors were then calcined at specific temperatures under controlled atmospheres (air or N2) for 2 h to obtain Mn-modified carbon-based deNOx catalysts, labeled as a-bMnOx/AC-cd. Here, a denotes the solvent type used for the Mn(NO3)2 solution, with H and E representing water and ethanol, respectively; b indicates the Mn loading amount (4 wt%, 6 wt%, 8 wt%, 10 wt%, or 12 wt%); c corresponds to the calcination temperature of the precursor (180, 200, 220, 240, 260, and 500 °C); and d specifies the calcination atmosphere, either air (A) or nitrogen (N). Details of the preparation conditions are summarized in Table 1.
Table 1. Catalysts' preparation conditions
Catalyst Preparation conditions Solvent Loading (wt%) Calcination temperature (°C) Calcination atmosphere E-8MnOx/AC-200A Ethanol 8 200 Air H-8MnOx/AC-200A Water 8 200 Air H-8MnOx/AC-500N Water 8 500 N2 E-8MnOx/AC-180A Ethanol 8 180 Air E-8MnOx/AC-220A Ethanol 8 220 Air E-8MnOx/AC-240A Ethanol 8 240 Air E-8MnOx/AC-260A Ethanol 1 260 Air E-4MnOx/AC-200A Ethanol 4 200 Air E-6MnOx/AC-200A Ethanol 6 200 Air E-10MnOx/AC-200A Ethanol 10 200 Air E-12MnOx/AC-200A Ethanol 12 200 Air NH3-SCR performance
-
The schematic diagram of the deNOx testing apparatus is shown in Supplementary Fig. S1. The setup consisted of gas cylinders, mass flow meters, a gas mixing tank, an SCR deNOx reactor, an infrared flue gas analyzer (MRU MGA5), and an exhaust treatment unit. For each test, 2 g of the catalyst was placed in a quartz tube reactor, and the deNOx reaction was conducted for over 30 min. The simulated flue gas composition was set as follows: an NO concentration of 0.03 vol%, NH3 at 0.035 vol%, and O2 at 3 vol%, with N2 as the balance gas. The deNOx reaction was conducted at 150 °C with a gas hourly space velocity (GHSV) of 20,000 h–1. The gas composition at both the inlet and outlet of reactor was analyzed using the MGA5. Data were recorded after steady-state conditions were reached, with each sample tested in triplicate or more to ensure reproducibility.
The deNOx performance was assessed by using the NO conversion (η) equation[18]:
$ \mathit{\eta}\mathrm{=(1-[NO}_{\mathrm{x}}\mathrm{]}\mathrm{_{\mathit{\mathrm{out}}}\mathrm{/[NO}}_{\mathrm{x}}\mathrm{]}_{\mathit{\mathrm{in}}}\mathrm{)\times100\text{%}} $ (1) where, [NOx]in and [NOx]out represent the NOx concentrations at the inlet and outlet of the SCR reactor, respectively.
Characterization of the catalysts
-
The pore structure of the catalysts was determined using a physical adsorption analyzer (ASAP 2020). Prior to testing, the samples were degassed at 250 °C for 6 h. The Brunauer–Emmett–Teller (BET) method was used to calculate the specific surface area (SBET), whereas the total pore volume (Vtot) was derived from the N2 adsorption at a relative pressure of 0.98. The micropore volume (Vmic) was calculated using the Horvath–Kawazoe method. The surface morphology and elemental distribution were analyzed using scanning electron microscopy combined with energy-dispersive spectroscopy (SEM-EDS, FEI Tecnai G2 F30). X-ray photoelectron spectroscopy (XPS, Thermo Escalab250X) was used to determine the surface chemical composition of the catalysts.
-
The pore structure of the catalyst significantly influences the flow of flue gas and the interaction with active sites, which is a key factor for deNOx efficiency. The pore structure characteristics of H-8MnOx/AC-500N, H-8MnOx/AC-200A, and E-8MnOx/AC-200A catalysts were studied using a physical adsorption analyzer (Fig. 1). As shown in Fig. 1a, all catalysts exhibited combined Type I and Type IV isotherms according to the International Union of Pure and Applied Chemistry (IUPAC) classification. The rapid increase in nitrogen adsorption at P/P0 < 0.1 indicated the presence of microporous structures, whereas the gradual increase at the relative pressure (P/P0) > 0.4 with the emergence of hysteresis loops suggested the existence of mesoporous structures[19,20]. Table 2 summarizes the pore structure parameters of the catalysts. In terms of microporosity, the H-8MnOx/AC-500N, H-8MnOx/AC-200A, and E-8MnOx/AC-200A catalysts displayed high percentages of 89.2%, 91.1%, and 92.1%, respectively, indicating that all catalysts are microporous–mesoporous composite materials dominated by micropores. The low microporosity of H-8MnOx/AC-500N was attributed to the high-temperature treatment inducing pore formation and expansion. Regarding specific surface areas, H-8MnOx/AC-500N exhibited the highest value (743.6 m2/g), whereas H-8MnOx/AC-200A and E-8MnOx/AC-200A showed lower values of 686.2 and 689.3 m2/g, respectively. Compared with H-8MnOx/AC-200A, E-8MnOx/AC-200A showed a marginally larger specific surface area, along with notable enhancements in total pore volume and micropore volume. This improvement was attributed to the more homogeneous distribution of Mn in the ethanol-based solvent system, which effectively prevented pore blockages caused by Mn agglomeration and facilitated the transformation of mesopores into micropores. These changes in pore structure were further corroborated by the pore size distribution: H-8MnOx/AC-500N displayed pore sizes mainly at ≤ 3 and 20–25 nm; H-8MnOx/AC-200A at ≤ 3 and 10–15 nm; and E-8MnOx/AC-200A primarily at ≤ 3 nm.
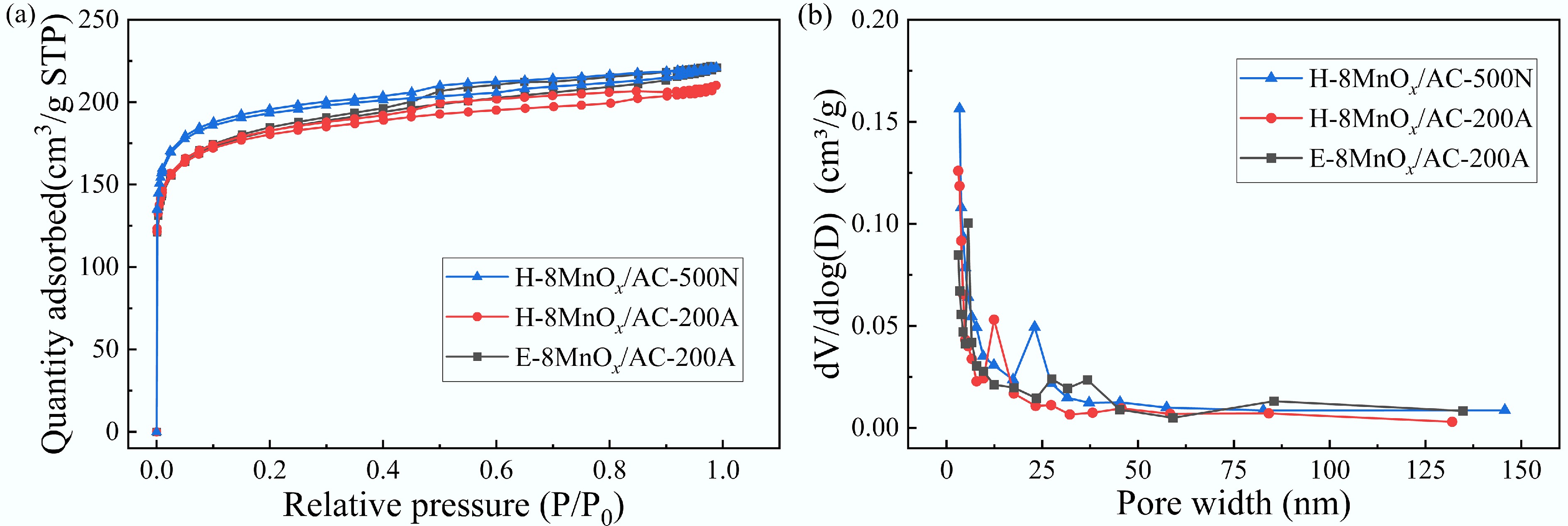
Figure 1.
(a) N2 adsorption–desorption isotherms and (b) corresponding pore size distribution of the catalysts.
Table 2. Pore structure parameters of the catalysts
Catalyst SBET
(m2/g)Vmic
(cm3/g)Vtot
(cm3/g)Vmic/Vtot
(%)Dp
(nm)H-8MnOx/AC-500N 743.6 0.306 0.343 89.2 1.845 H-8MnOx/AC-200A 686.2 0.297 0.326 91.1 1.900 E-8MnOx/AC-200A 689.3 0.315 0.342 92.1 1.988 The micromorphology and dispersion of active components significantly influenced the deNOx efficiency of the catalysts. SEM-EDS analysis (Fig. 2) revealed the microstructure and elemental distribution of the catalysts. The H-8MnOx/AC-500N catalyst exhibited abundant pore structures and a relatively smooth surface, potentially caused by the decomposition of Mn(NO3)2 at high temperatures, where the resulting NO2 and NO gases acted as activators to etch the AC support[21,22]. In contrast, both the H-8MnOx/AC-200A and E-8MnOx/AC-200A catalysts exhibited a more conspicuous reduction in pore structures and rougher surfaces. This was attributed to the loading of MnO2 at lower calcination temperatures, consistent with the N2 adsorption–desorption analysis[23]. Notably, although MnOx aggregated prominently on the surface of the H-8MnOx/AC-200A catalyst, it was dispersed more uniformly on the E-8MnOx/AC-200A catalyst. The dispersibility of MnOx was closely related to the interaction between precursors and carriers, which was largely governed by both the polarity of the carrier surface and the density and polarity of the solvents used during impregnation[24,25]. Compared with water, ethanol's lower polarity, reduced surface tension, and superior wetting properties enhanced the interactions between Mn(NO3)2 and AC[26−28], ensuring better contact and infiltration, thereby promoting MnOx dispersion[29,30].

Figure 2.
SEM-EDS images of the catalysts: (a) H-8MnOx/AC-500N, (b) H-8MnOx/AC-200A, and (c) E-8MnOx/AC-200A.
The chemical states of the catalysts were determined by XPS (Fig. 3). The O 1s peak was deconvoluted into two fitting peaks located at 532.2 and 530.1 eV, corresponding to chemisorbed oxygen (Oα, like O2−, O−) and lattice oxygen (Oβ, like O2−), respectively[31]. Oα, characterized by high activity and mobility, was a pivotal indicator of catalytic performance, primarily existing as OH− and CO32−[32−34]. The Mn 2p3/2 spectra were deconvoluted into three fitted peaks at binding energies of 640.8, 641.9, and 643.5 eV, corresponding to Mn2+, Mn3+, and Mn4+, respectively. The MnOx speciation directly governs the deNOx capability of the catalyst, with Mn4+ demonstrating a superior redox capacity and high catalytic activity during the deNOx process[18,35]. The relative contents of Mn4+ and Oα in the H-8MnOx/AC-500N catalyst were only 18.73% and 56.36%, respectively, whereas the H-8MnOx/AC-200A catalyst exhibited significantly higher Mn4+ and Oα values of 30.52% and 84.54%. This improvement could be attributed to two main factors. First, the lower calcination temperature (200 °C) facilitated the formation of amorphous MnO2 during the thermal decomposition of Mn(NO3)2, in which Mn2+ was oxidized to Mn4+ under the combined action of nitrate and molecular oxygen[36]; second, calcination in an air atmosphere introduced abundant oxygen-containing functionalities on the catalyst's surface, which facilitated the generation of higher-valent Mn species through electron transfer[18]. Notably, under identical calcination conditions (200 °C in air), the relative content of Mn4+ in the E-8MnOx/AC-200A catalyst reached 43.63%, representing a 42.96% increase over its H-8MnOx/AC-200A counterpart. This was attributed to the ethanol-assisted Mn loading, which enhanced the dispersion of MnOx, reduced aggregation, and facilitated intimate contact between MnOx and the catalyst support. As a result, MnOx reacted thoroughly with the oxygen-containing functional groups in the support, leading to a higher production of Mn4+.
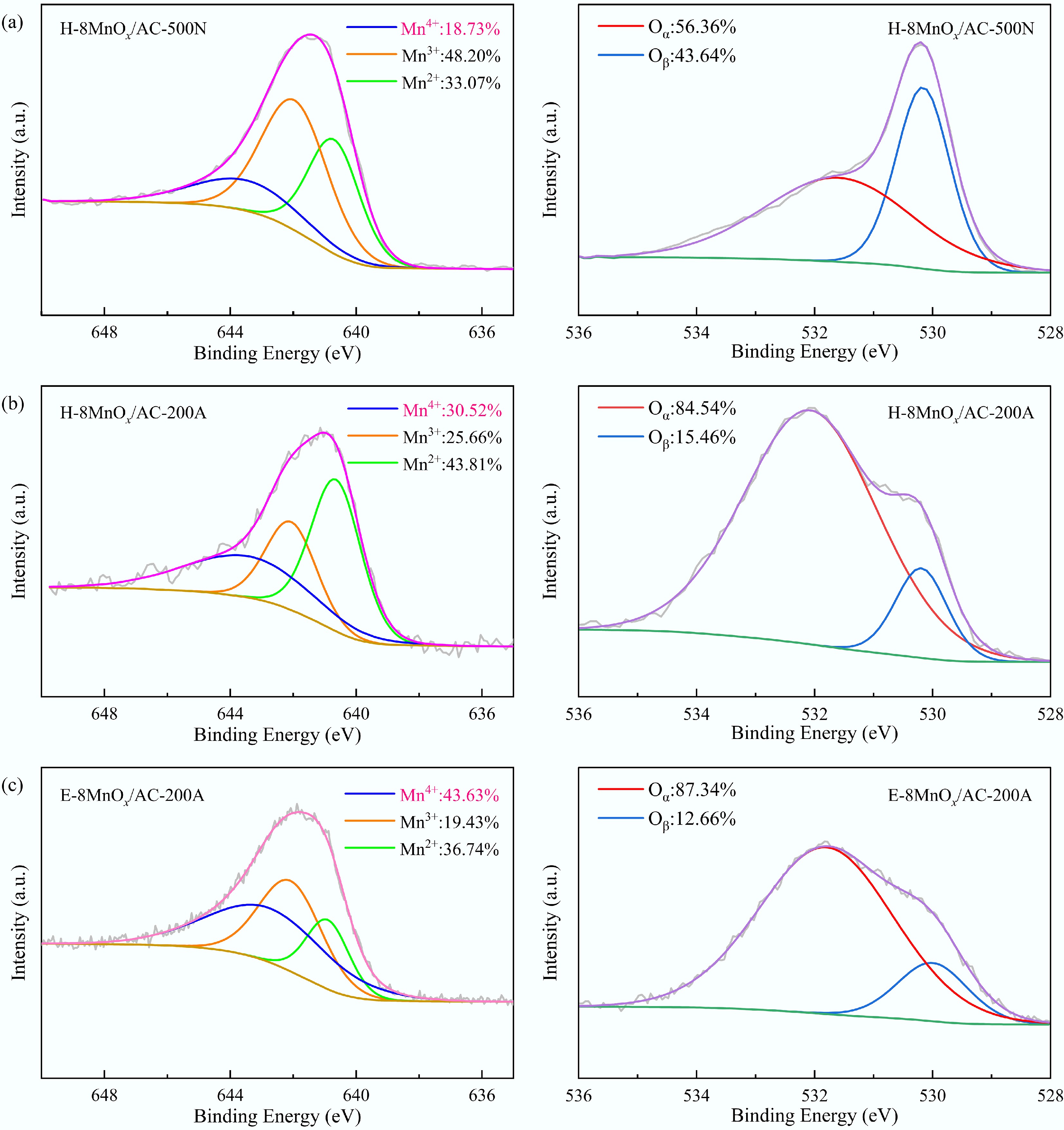
Figure 3.
Mn 2p3/2 and O 1s spectra: (a) H-8MnOx/AC-500N, (b) H-8MnOx/AC-200A, and (c) E-8MnOx/AC-200A.
Catalysts' deNOx performance
Impact of the preparation method
-
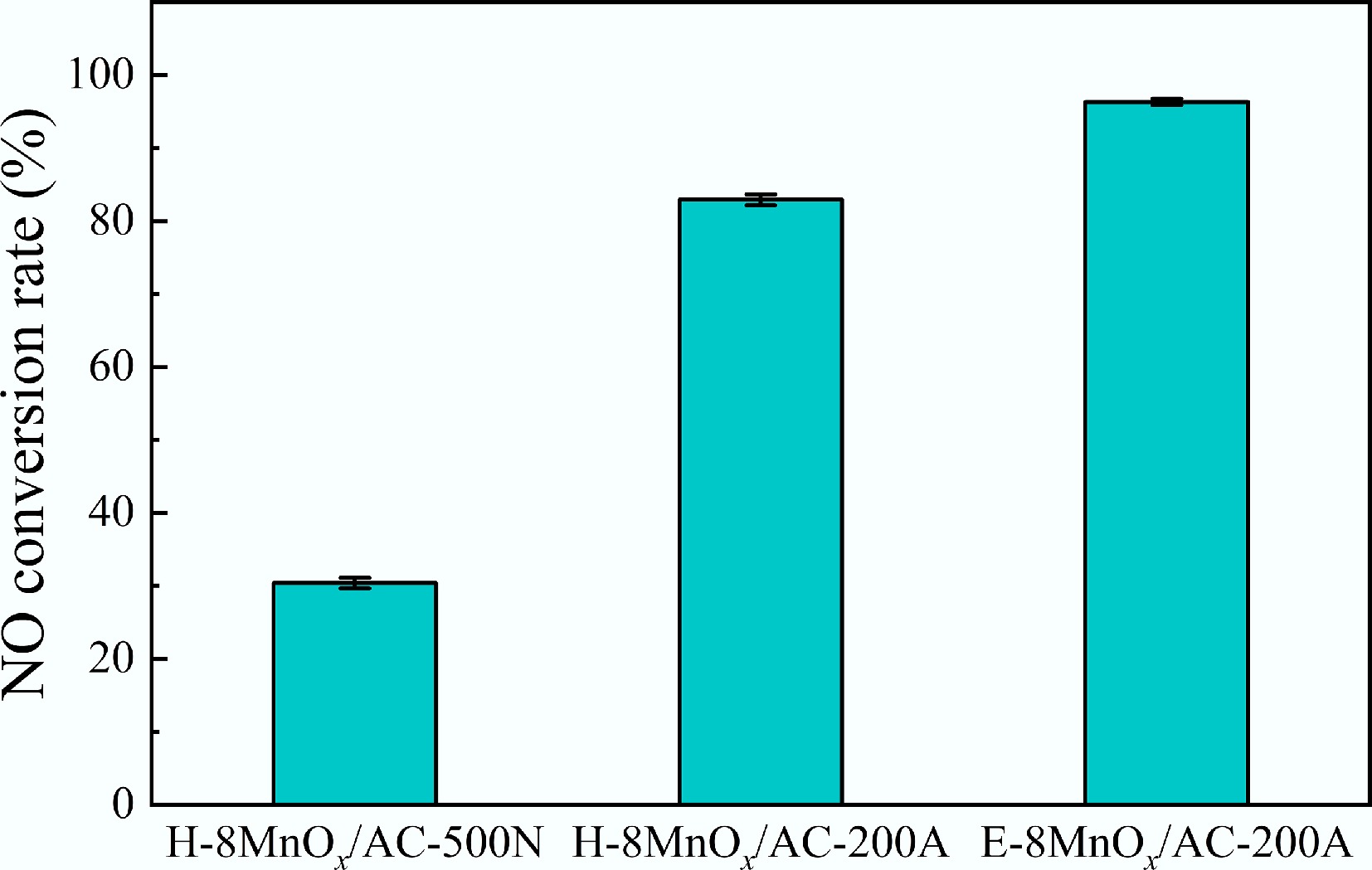
To optimize the preparation method of deNOx catalysts, the NO removal performance of H-8MnOx/AC-500N, H-8MnOx/AC-200A, and E-8MnOx/AC-200A was evaluated at 150 °C and a GHSV of 20,000 h−1, aiming to reveal the influence of the impregnation solvents and calcination conditions on the NO conversion efficiency; the results are depicted in Fig. 4. Although H-8MnOx/AC-500N presented a substantial specific surface area, its deNOx efficiency was limited to 30.4%. Conversely, H-8MnOx/AC-200A displayed a marked improvement, achieving an efficiency of 82.9%. This discrepancy highlighted that deNOx performance is not only determined by physical attributes like surface area but is also intimately tied to the morphology and dispersion of the active components. Although a large surface area provided ample anchoring sites for the active species, the inferior performance of H-8MnOx/AC-500N was attributed to its lower Mn4+ content and poor dispersion. In contrast, H-8MnOx/AC-200A, prepared via low-temperature air calcination, contained a higher amount of active Mn4+ and formed more oxygen-containing functional groups, such as hydroxyl groups, on the AC matrix. These functional groups facilitated the adsorption of NH3 through acidic sites, thus promoting the catalytic conversion of NO. Notably, E-8MnOx/AC-200A, despite having a similar surface area to H-8MnOx/AC-200A, achieved a superior deNOx efficiency of 96.3%, which is a 13.4% increase. This was attributed to the enhanced Mn4+ content and its uniform distribution, which facilitated optimal interaction between the catalyst's active components and the flue gas.
Impact of calcination temperature
-
Figure 5 illustrates the deNOx efficiency of catalysts prepared at calcination temperatures ranging across 180–260 °C. The results indicated that E-8MnOx/AC-180A achieved a deNOx efficiency of 78.1%. As the calcination temperature rose, the deNOx efficiency of the catalysts initially increased and then decreased, peaking at 96.3% for the catalysts calcined at 200 °C, followed by a decrease to 85.9% at 260 °C. This could be attributed to insufficient precursor calcination at lower temperatures, resulting in low Mn4+ content. However, excessive calcination temperatures prompted the conversion of Mn4+ to Mn3+[37]. The catalysts calcined at 200 °C contained a higher amount of Mn4+, whose strong redox capability facilitated the conversion of NO to NO2, thereby enhancing the rapid SCR reaction (NO + NO2 + 2NH3 → 3N2 + 2H2O)[38].
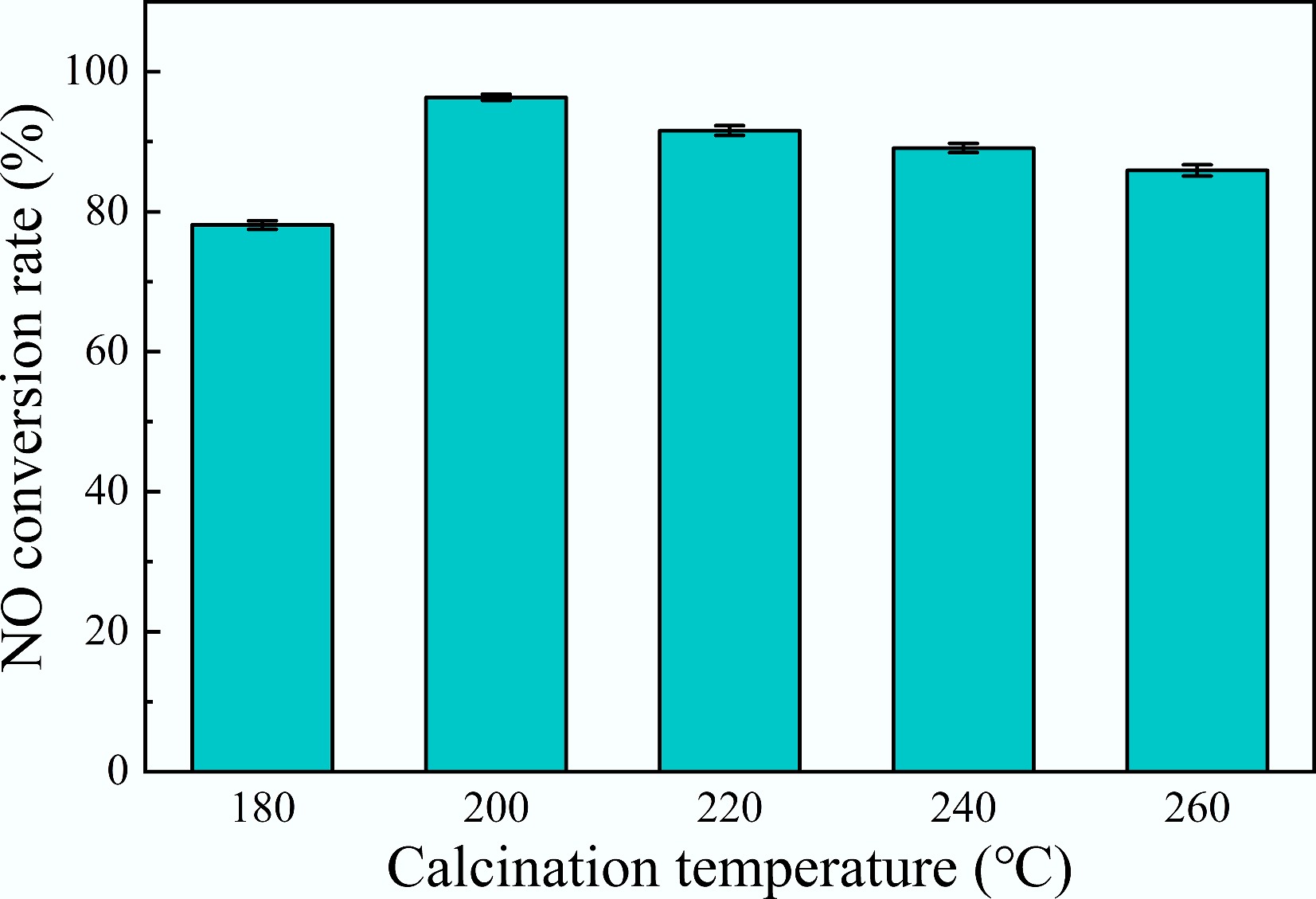
Figure 5.
Influence of calcination temperature on the catalysts' deNOx efficiency (GHSV of 20,000 h−1).
Impact of Mn loading
-
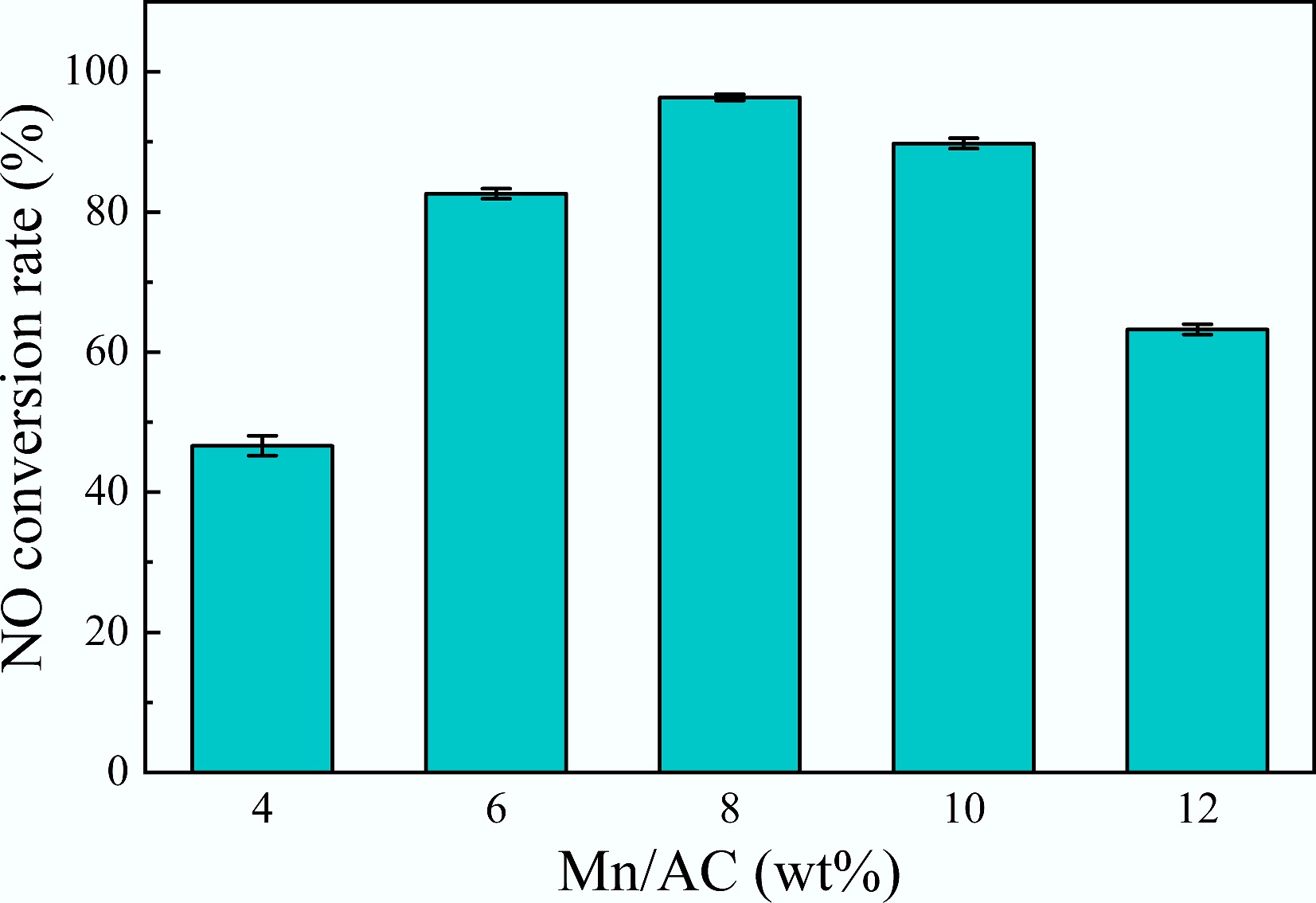
To clarify the impact of Mn loading on the deNOx performance, catalysts with loadings of 4 wt%, 6 wt%, 8 wt%, 10 wt%, and 12 wt% were assessed, with the outcomes presented in Fig. 6. Notably, the E-8MnOx/AC-180A catalyst with the 8 wt% Mn loading showcased the highest catalytic efficiency, reaching 96.3%, whereas both excessive and insufficient Mn loading adversely affected NO conversion. The deNOx efficiency was merely 46.9% at a 4 wt% Mn loading and increased to 63.2% at a 12 wt% Mn loading. This trend occurred because low Mn loading provided insufficient active sites, limiting the catalytic conversion of NO, whereas excessive Mn loading caused the agglomeration of active components on the catalyst's surface, reducing the exposure of active sites and potentially blocking the catalyst's pores, hindering interaction with the flue gas and impeding the deNOx reaction.
-
This study introduced an innovative preparation method for carbon-based low-temperature deNOx catalysts using an ethanol-assisted Mn impregnation method, systematically analyzing the effects of the selected impregnation solvent and calcination conditions on the deNOx performance of Mn-modified carbon-based catalysts. The study found that using ethanol as a solvent for enhancing Mn loading significantly improved the uniformity of the active components' dispersion on the carbon support, effectively suppressed metal agglomeration, optimized the MnOx phase, and increased the proportion of Mn4+, thereby greatly enhancing the NO conversion of the catalyst. Moreover, the proper calcination temperature and optimal Mn loading were identified as critical factors for increasing the Mn4+ content and active site density in the catalyst, effectively advancing the deNOx reaction. Specifically, at a calcination temperature of 200 °C and a Mn loading amount of 8 wt%, the catalyst prepared via ethanol-assisted Mn modification exhibited excellent performance, with a relative Mn4+ content of 43.63% in its MnOx phase. At a GHSV of 20,000 h−1 and a deNOx temperature of 150 °C, this catalyst achieved an NO conversion of 96.3%. This method offers the advantages of low cost, compatibility with existing equipment, and minimal additional energy consumption, which makes it highly promising for low-temperature deNOx applications.
-
It accompanies this paper at: https://doi.org/10.48130/scm-0026-0009.
-
The authors confirm their contributions to the paper as follows: Donghong Nan: conceptualization, methodology, data curation, and writing − original draft. Jinheng Xie: investigation, data curation, and validation. Tong Wu: investigation and methodology. Qi Niu: validation and writing − review and editing. Xiangyu Zhang: validation and methodology. Shiguan Yang: writing − original draft. Gangying Huang: methodology and data curation. Kai Li: conceptualization, supervision, writing − review and editing, and funding acquisition. Qiang Lu: conceptualization, supervision, and funding acquisition. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
-
The authors thank the National Key R&D Program of China (2024YFE0111000), the Science and Technology Project of China Huaneng Group (HNKJ22-H150), the Fundamental Research Funds for the Central Universities (2024MS039), and the Postdoctoral Fellowship Program of China Postdoctoral Science Foundation (CPSF) (GZC20230788) for financial support.
-
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
-
The MnOx/AC catalyst was prepared using ethanol as solvent to enhance Mn loading.
Ethanol improved the uniform dispersion of MnOx on activated carbon.
Low-temperature air calcination promoted the formation of Mn4+.
At a deNOx temperature of 150 °C, NO conversion reached 96.3%.
-
Full list of author information is available at the end of the article.
- The supplementary files can be downloaded from here.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Nan D, Xie J, Wu T, Niu Q, Zhang X, et al. 2026. Preparation and performance evaluation of a novel ethanol-enhanced Mn-modified carbon-based deNOx catalyst. Sustainable Carbon Materials 2: e009 doi: 10.48130/scm-0026-0009
Preparation and performance evaluation of a novel ethanol-enhanced Mn-modified carbon-based deNOx catalyst
- Received: 18 December 2025
- Revised: 21 January 2026
- Accepted: 28 January 2026
- Published online: 05 March 2026
Abstract: Mn-modified carbon-based catalysts, with their extensive pore network and excellent redox activity, are gaining prominence for low-temperature deNOx applications. However, conventional preparation methods often suffer from the uneven dispersion of active MnOx species and a low proportion of highly active Mn4+, which limit deNOx's efficiency. To address these limitations, this study developed an efficient strategy for preparing low-temperature carbon-based deNOx catalysts by enhancing Mn modification through the use of ethanol as the impregnation solvent. The results indicated that, compared with deionized water, ethanol, with its lower polarity, surface tension, and better wettability, significantly enhanced the interaction between manganese nitrate and activated carbon during the impregnation process. This improvement facilitated better contact and penetration, promoted the uniform dispersion of MnOx on the activated carbon, and ultimately improved the performance of deNOx. Systematic process optimization identified 200 °C as the optimal calcination temperature and 8 wt% as the ideal Mn loading. Under a gas hourly space velocity of 20,000 h–1 and a deNOx temperature of 150 °C, the NO conversion reached 96.3% using the ethanol-impregnated catalyst, which was 13.4% higher than the 82.9% conversion achieved by the water-impregnated catalyst.
-
Key words:
- Low-temperature SCR /
- Activated carbon /
- MnOx /
- Ethanol solvent /
- Impregnation method