-
Glaucoma encompasses a group of complex ocular diseases characterized by progressive optic nerve damage, often accompanied by elevated intraocular pressure (IOP) and irreversible vision loss[1]. Primary open-angle glaucoma (POAG) and chronic primary angle-closure glaucoma (CPACG) represent two common glaucoma subtypes. POAG is the most prevalent subtype globally. Although the anterior chamber angle remains open, POAG results from impaired drainage function of the trabecular meshwork (TM) and aqueous humor. In contrast, CPACG primarily arises from anatomical obstruction of aqueous humor outflow[2]. PAC/PACS exhibits anatomical structures similar to those of CPACG, but optic nerve injury has not yet occurred[3]. Patients with age-related cataract (ARC) frequently display anatomical similarities to POAG patients but lack evidence of optic neuropathy[4]. These distinctions enhance the comparability of aqueous humor metabolomic analyses when investigating hormonal influences on glaucoma pathophysiology.
Steroid hormones, including mineralocorticoids, glucocorticoids, and sex hormones[5], are cholesterol-derived physiological regulators of homeostasis with both systemic and local effects[6−9]. Dysregulated systemic steroid hormones have been linked to retinal and glaucomatous pathologies[10−13]. For instance, elevated androstenedione levels correlate with visual field progression in POAG patients[13]. Sex hormones also protect the optic nerve in glaucoma models and maintain ocular surface integrity[14−16].
Exogenous glucocorticoid (GC) administration is a well-established risk factor for secondary open-angle glaucoma, highlighting cortisol's role in IOP dysregulation[17]. Topical or systemic GC exposure elevates IOP in a significant proportion of individuals by increasing TM outflow resistance through cytoskeletal remodeling, extracellular matrix (ECM) deposition, and matrix metalloproteinase dysregulation[18−20].
Aqueous humor is a dynamic intraocular fluid that nourishes avascular tissues and reflects TM function, making it a crucial medium for investigating glaucoma pathogenesis[21]. Advances in aqueous humor proteomics and metabolomics have facilitated the identification of biomarkers for disease stratification and provided mechanistic insights[21−24]. Additionally, plasma and aqueous humor liquid biopsies offer innovative approaches to investigating tissue-specific metabolic disturbances[13,25,26]. Although early studies using radioimmunoassay detected elevated cortisol levels in aqueous humor from patients with POAG[27,28], these results were limited by the narrow steroid detection range of the method. Furthermore, they did not account for other glaucoma subtypes such as CPACG and PAC/PACS. Thus, the role of endogenous steroid hormones in glaucoma pathogenesis remains incompletely understood.
Liquid chromatography-tandem mass spectrometry (LC-MS/MS) is currently the most accurate method for detecting steroid metabolites in aqueous humor[29]. This approach was optimized in previous studies[30]. Furthermore, given that ocular biometric parameters, such as anterior chamber depth (ACD), are fundamental determinants of the anterior chamber's structure and volume, which constitute the local microenvironment for the aqueous humor, this study sought to explore whether these parameters correlate with the levels of key steroid hormones within the eye. Therefore, the present study employed LC-MS/MS to measure steroid hormone concentrations in aqueous humor and plasma samples from patients with POAG, CPACG, PAC/PACS, and ARC. Clinical data were analyzed to achieve four objectives: (1) to map the distribution of endogenous steroid hormones in aqueous humor and plasma; (2) to evaluate the correlations between aqueous humor and plasma steroid hormone levels; (3) to explore metabolic patterns of GCs and mineralocorticoids in aqueous humor and plasma; and (4) to assess changes in cortisol and aldosterone among glaucoma patients. By clarifying the roles of GCs and mineralocorticoids in glaucoma pathophysiology, this study provides new insights into the mechanisms underlying glaucoma.
-
This study adhered to the principles outlined in the Declaration of Helsinki. The study protocol received approval from the Ethics Committee of the Third People's Hospital of Huizhou (Approval No. 2023-KY-102-01). Furthermore, the study was prospectively registered with the Chinese Clinical Trial Registry (ChiCTR2400081103) and the National Medical Research Registration and Filing Information System (Approval No. MR-44-25-033665). Before inclusion in the study, all participants provided written informed consent.
Clinical samples
-
This study included patients who underwent glaucoma or cataract surgery at the Third People's Hospital of Huizhou, China, from March to October 2024. A total of 121 aqueous humor specimens and 92 plasma samples were collected and categorized. Patients were divided into four groups based on established ophthalmic diagnostic criteria[3,4,31]: a cataract group (ARC, 63 aqueous humor/42 plasma samples), two glaucoma groups (POAG, 19 aqueous humor/12 plasma samples; CPACG, 20 aqueous humor/20 plasma samples) and a pre-glaucomatous group (PAC/PACS, 19 aqueous humor/18 plasma samples). The ARC group served primarily as an age- and surgery-matched control group to differentiate glaucoma-specific changes from effects related to aging or the surgical procedure itself. Each patient's plasma sample was matched with the corresponding aqueous humor sample. A total of 29 plasma samples were excluded for pre-analytical reasons: hemolyzed plasma samples (n = 16), missed blood samples (n = 12), and sample mislabeling (n = 1). All exclusions were made before biochemical analysis to avoid technical artifacts. Although selection bias cannot be entirely ruled out, comparative analysis revealed no significant differences in demographic or clinical characteristics between the included and excluded cohorts.
Inclusion criteria: (1) Patients aged 50–85 years undergoing glaucoma or cataract surgery for POAG, CPACG, or ARC. (2) No history of other ocular surgeries, including vitrectomy, cataract surgery, intravitreal drug injection, or glaucoma surgery in the affected eye. (3) Refractive error more than –6D. (4) No use of corticosteroids, radiotherapy, or chemotherapy within the previous six months. (5) All patients with glaucoma used β-blocker IOP-lowering eye drops, but did not use prostaglandin analog eye drops within one month before surgery.
Exclusion criteria: (1) Monocular patients. (2) Secondary glaucoma, such as neovascular glaucoma or glaucoma due to uveitis or trauma. (3) Other ocular diseases, such as uveitis or trauma. (4) Hereditary eye diseases, such as retinitis pigmentosa. (5) Metabolic disorders, such as hyperuricemia and diabetes (patients with classic symptoms of hyperglycemia or hyperglycemic crisis, a random plasma glucose ≥ 200 mg/dL (≥ 11.1 mmol/L) following American Diabetes Association guidelines[32]. (6) Severe systemic diseases, such as autoimmune diseases, rheumatic diseases, lung cancer, adrenal tumors, pituitary tumors, or breast cancer.
Patients underwent measurements for axial length (AL) and central ACD using the ZEISS IOL Master 700. Additionally, patients with POAG and CPACG underwent assessments of retinal nerve fiber layer (RNFL) thickness via optical coherence tomography (ZEISS) and visual field testing. Blood samples were collected between 8:00 and 10:00 AM to avoid potential influences of circadian rhythms on steroid hormone levels. Surgeries were performed between 12:00 AM and 2:00 PM to consider the biological rhythm of corticosteroid secretion.
Approximately 2 mL of venous blood was collected during routine preoperative assessment, centrifuged, and stored as plasma at −80 °C. Aqueous humor samples (0.05–0.1 mL) were collected intraoperatively via limbal corneal puncture during cataract or glaucoma procedures, including trabeculectomy or combined cataract-glaucoma surgery. Aqueous humor specimens were immediately stored in liquid nitrogen at −196 °C. Collected clinical data included patient age, gender, diagnosis, AL, ACD, visual field, and RNFL thickness.
Cataract surgery: Phacoemulsification cataract removal combined with intraocular lens implantation was performed. Before the operation, a 1 mL syringe with a 30 G needle punctured the upper limbus (right eye) or lower limbus (left eye) to collect 0.05–0.1 mL aqueous humor. Afterward, Ringer's solution was injected through the puncture site to deepen the anterior chamber, and the puncture site was incorporated into the limbal incision. A main incision was then made at the temporal limbus, and standard phacoemulsification combined with intraocular lens implantation was completed.
Trabeculectomy: After dissecting the upper scleral flap and before trabecular tissue removal, a 1 mL syringe with a 30 G needle was used to puncture the anterior chamber at the nasal or temporal corneal limbus to collect 0.05–0.1 mL aqueous humor. After re-forming the anterior chamber with Ringer's solution, the remaining trabeculectomy procedure was performed.
Steroid hormone extraction
-
Steroid hormones were extracted from 100 µL of aqueous humor or plasma using liquid-liquid extraction. Samples with an initial volume less than 100 µL were brought to this volume by the addition of phosphate-buffered saline (PBS), and the final measured concentrations were corrected for this dilution factor. Briefly, each sample was mixed with 1,500 µL of a 1:9 (v/v) acetonitrile/methyl tert-butyl ether solution, vigorously vortexed, and centrifuged at 10,000 × g. The supernatant was collected, evaporated to dryness under a gentle stream of nitrogen, and the residue was reconstituted in 60% methanol for storage at −20 °C until LC-MS/MS analysis.
LC–MS/MS analysis
-
Chromatographic separation was performed using an ACQUITY UPLC CSH C18 column (Waters) with a gradient of 0.1 mM ammonium fluoride (A) and MeOH (B): 20%–40% B (0–0.5 min), 40%–55% (0.5–3.3 min), and 55%–95% (3.3–6 min). Steroid hormones were quantified using a SCIEX 6500+ mass spectrometer in multiple reaction monitoring (MRM) mode with ion spray voltages of ± 4.5/5.5 kV and a source temperature of 550 °C. Data acquisition and processing were conducted using Analyst 1.7 and MultiQuant 3.0.2 software.
Statistical analysis
-
All data analyses and graphical representations were performed using GraphPad Prism (version 10.1.2). The normality assumption for all continuous variables (steroid hormone concentrations, age, AL, ACD) was verified via quantile-quantile (Q-Q) plots, which confirmed that the data conformed to an approximate normal distribution and thus validated the application of parametric tests. The comparison of three groups of quantitative data was conducted using one-way ANOVA, and the post-hoc test was corrected using the Bonferroni method. The comparison of two groups of quantitative data was conducted using an independent sample t-test. For all subgroup analyses (e.g., comparisons between glaucoma subtypes), p-values were adjusted for multiple comparisons using the Bonferroni correction where appropriate. These subgroup analyses should be interpreted with caution due to the modest sample sizes, as they are exploratory in nature. Linear regression analyses were performed using SPSS. Steroid hormone concentrations, age, AL, ACD, visual field defect values, and RNFL thickness were all expressed as mean ± standard deviation (SD). Statistical significance was defined as p < 0.05 and denoted as follows:* p < 0.05; ** p < 0.01; *** p < 0.001. For one-way ANOVA, effect size was reported as eta-squared (η2) with 95% confidence intervals (CI). For Student's t-tests, the effect size was reported as Cohen's d with 95% CI. Correlation analyses were performed using the Pearson correlation coefficient. All key comparisons and correlations are reported with their respective effect sizes and 95% CIs to quantify the magnitude and precision of the observed effects.
-
In aqueous humor, aldosterone, corticosterone, cortisone, and cortisol were detected at high abundance, while dehydroepiandrosterone and 17-hydroxyprogesterone were not detected. Steroid hormone concentrations were higher in plasma compared to aqueous humor, with plasma-to-aqueous humor ratios ranging from 5.44 to 62.04 times (Table 1). The distribution pattern of steroid hormone concentrations in aqueous humor closely matched that observed in plasma. Specifically, the hierarchy of mean concentrations (aldosterone > cortisol > cortisone > corticosterone) was identical in both compartments (Fig. 1).
Table 1. Concentrations (ng/mL) and ratios of steroid hormones in total PL and AH samples.
Group ALD CORT COR F DHEA 17OHP PL (n = 92) 418.73 ± 229.38 0.99 + 1.02 8.63 ± 6.07 58.72 ± 26.76 0.51 ± 0.73 0.22 ± 0.34 AH (n = 121) 6.73 ± 8.32 0.18 ± 0.20 0.42 ± 0.37 1.95 ± 1.25 PL/AH 62.04 5.44 20.43 29.96 AH, aqueous humor; PL, plasma; ALD, aldosterone; CORT, corticosterone; COR, cortisone; F, cortisol; DHEA, dehydroepiandrosterone; 17OHP, 17-hydroxyprogesterone. 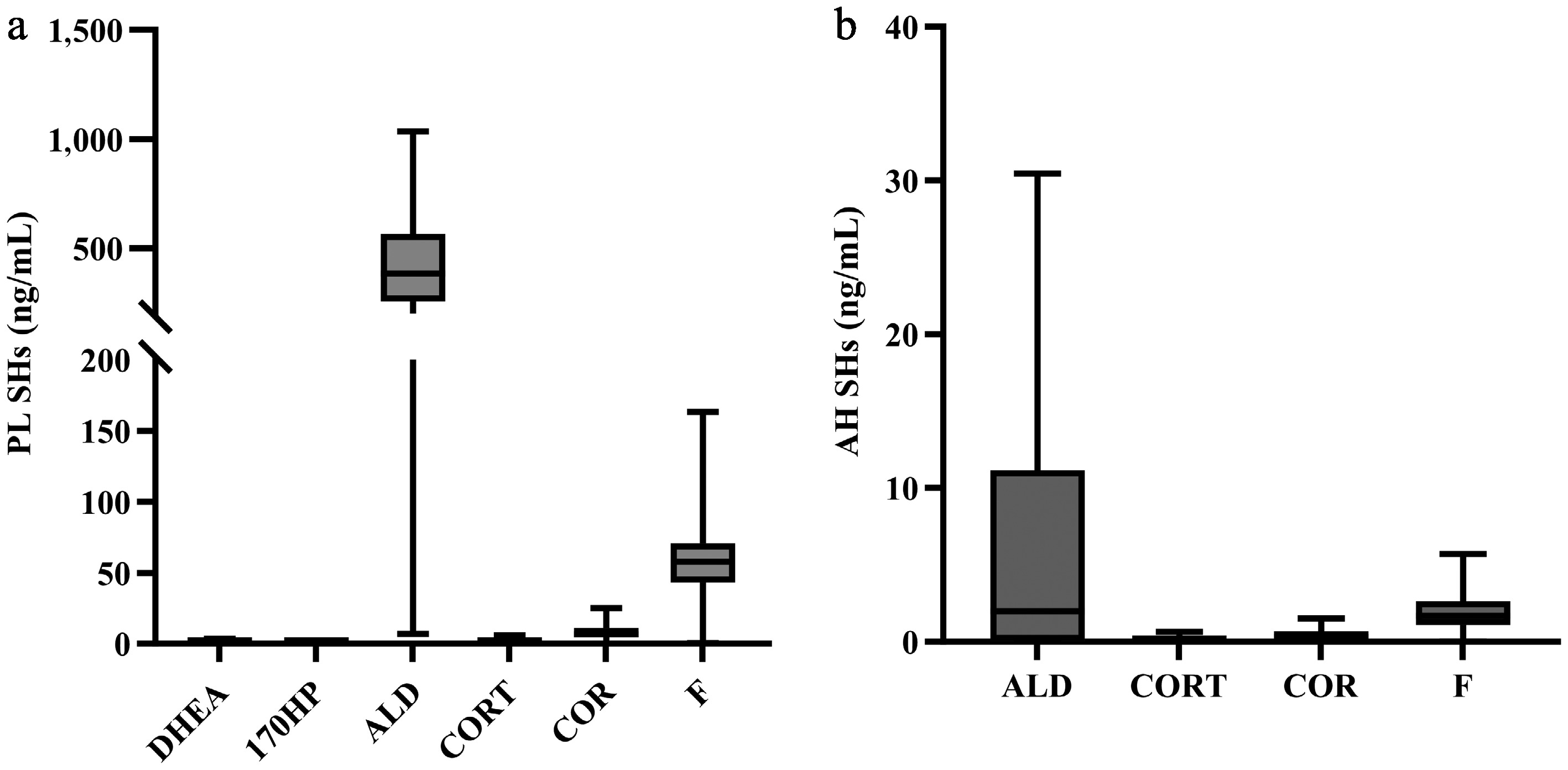
Figure 1.
Distribution of steroid hormones in plasma and aqueous humor. (a) In plasma, six steroid hormones (ALD, CORT, COR, F, DHEA, and 17OHP) were detected at high abundance. (b) In aqueous humor, four steroid hormones (ALD, CORT, COR, and F) were detected at high abundance. Notes: AH, aqueous humor; SHs, steroid hormones; PL, plasma; ALD, aldosterone; CORT, corticosterone; COR, cortisone; F, cortisol; DHEA, dehydroepiandrosterone; 17OHP, 17-hydroxyprogesterone.
Impact of age and gender on steroid hormones
-
Correlation analyses between aldosterone, corticosterone, cortisone, and cortisol and age in the ARC group revealed no significant relationships in either plasma (all |r| ≤ 0.184, p > 0.05) or aqueous humor samples (all |r| ≤ 0.154, p > 0.05) (Supplementary Fig. S1). Similarly, analysis using unpaired t-tests (as presented in Supplementary Table S1) showed no significant correlations with gender (all p > 0.05). These results suggest that age and gender do not significantly affect steroid hormone levels in the control group. It should be noted that, although the overall age distribution did not differ significantly among diagnostic groups (p = 0.064), the gender distribution varied (p = 0.003, Supplementary Table S2).
Metabolic pathway diagram of steroid hormones
-
Steroid hormone metabolic pathways include GC metabolism, mineralocorticoid metabolism, and sex hormone metabolism. The metabolic pathways for steroid hormones are presented in Supplementary Fig. S2, with asterisks indicating the steroid hormones detected in this study.
Next, metabolic correlation patterns in aqueous humor and plasma were analyzed. In the 121 aqueous humor samples, a significant positive correlation was observed between cortisol and aldosterone (r = 0.535, p < 0.01). Corticosterone and aldosterone were negatively correlated (r = –0.556, p < 0.01). There is also a positive correlation between cortisone and cortisol (r = 0.308, p < 0.01), or between cortisone and corticosterone (r = 0.739, p < 0.01) (Fig. 2).
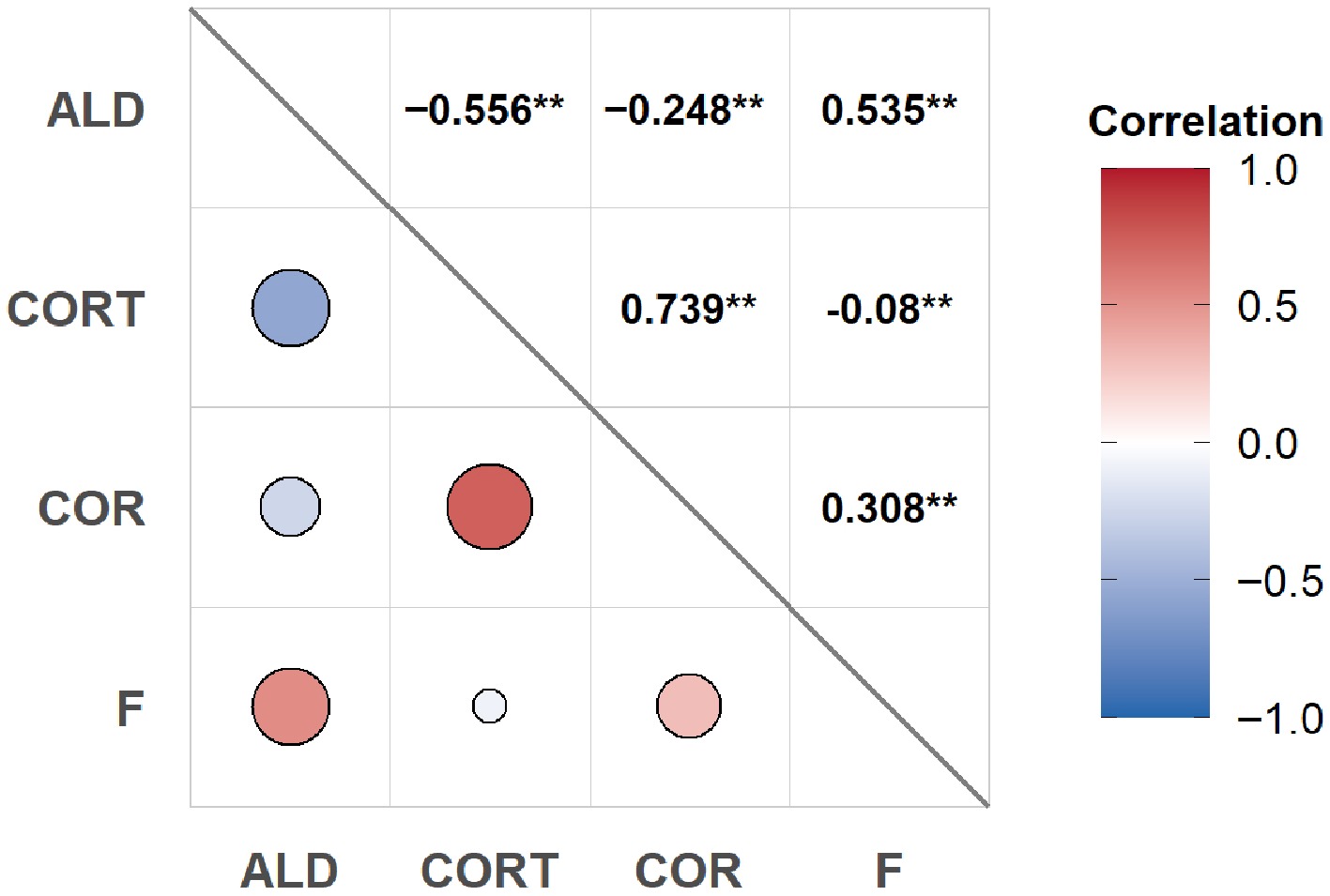
Figure 2.
The correlation of steroid hormones within aqueous humor. Correlation analysis was performed on 121 aqueous humor samples. Notes: * p < 0.05, ** p < 0.01. ALD, aldosterone; CORT, corticosterone; COR, cortisone; F, cortisol.
In the 92 plasma samples, aldosterone was positively correlated with corticosterone (r = 0.703, p < 0.01), and cortisol was positively correlated with cortisone (r = 0.699, p < 0.01). Aldosterone was also positively correlated with cortisol (r = 0.748, p < 0.01). and corticosterone positively correlated with cortisone (r = 0.760, p < 0.01). There was no significant correlation between plasma and aqueous humor levels of cortisol, but a weak negative correlation existed between plasma and aqueous humor levels of aldosterone (r = –0.226, p < 0.05) (Fig. 3).
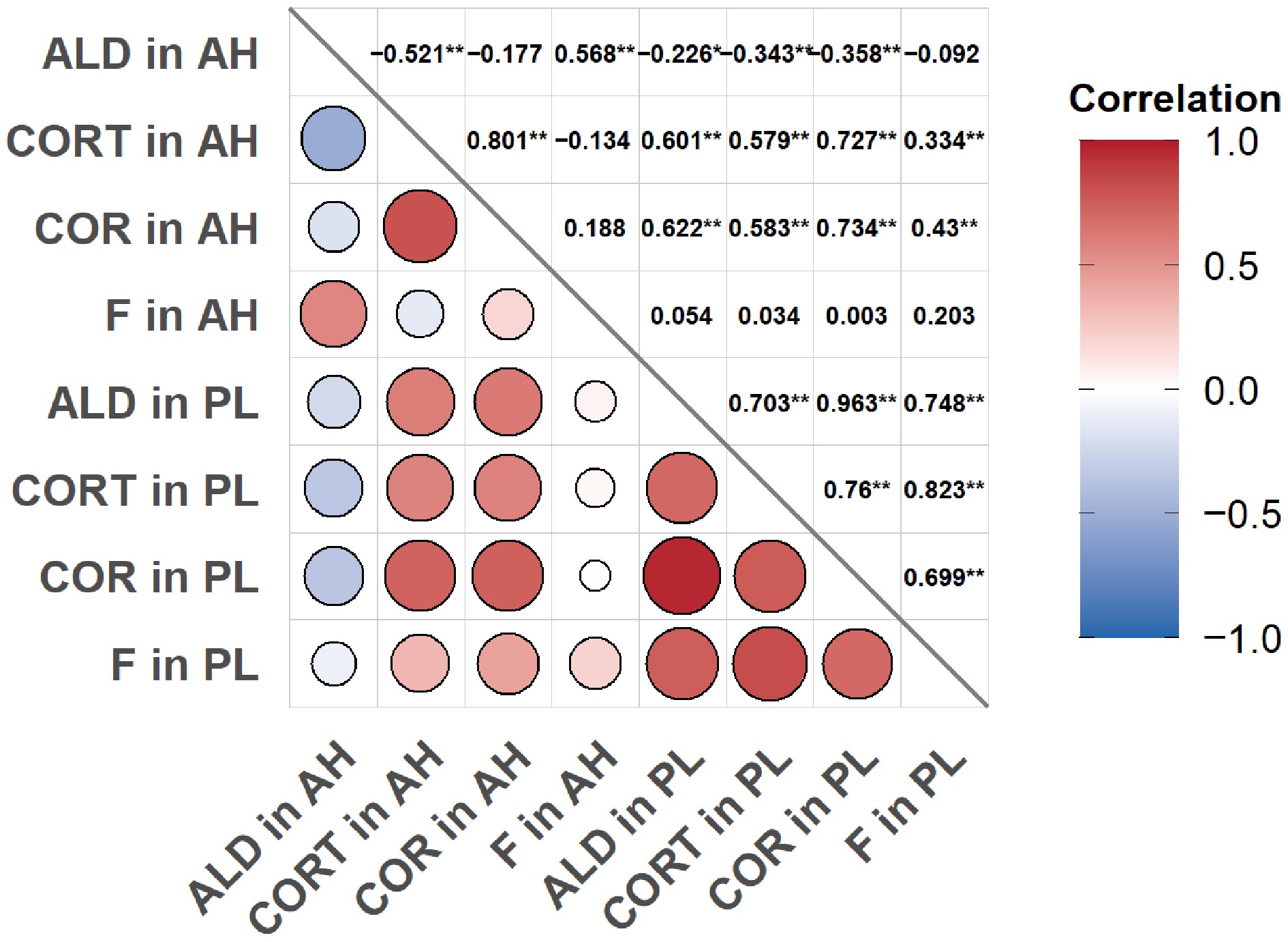
Figure 3.
Correlation analysis of steroid hormones between 92 matched plasma and aqueous humor samples. Notes: * p < 0.05, ** p < 0.01, AH, aqueous humor; PL, plasma; ALD, aldosterone; CORT, corticosterone; COR, cortisone; F, cortisol. Correlation analysis was performed using 92 pairs of matched plasma and aqueous humor samples.
Correlation analysis of steroid hormones with AL and ACD
-
Correlation analyses were performed between AL, ACD, and the levels of aldosterone, cortisol, corticosterone, and cortisone in plasma and aqueous humor. In plasma samples (n = 92), no significant correlations were found between AL or ACD and the levels of any of the four steroid hormones (Supplementary Fig. S3). In aqueous humor samples (n = 121), aldosterone and cortisol levels showed positive correlations with ACD (aldosterone: r = 0.248, p < 0.01; cortisol: r = 0.234, p < 0.05) but not with AL (Fig. 4). A stratified analysis based on angle status was subsequently conducted. In the open-angle subgroup (POAG + ARC), aqueous humor aldosterone remained positively correlated with ACD (r = 0.284, p < 0.01; Supplementary Fig. S4). No other significant correlations between steroid hormones and ACD were observed in the open-angle or angle-closure (CPACG + PAC/PACS) subgroups Supplementary Figs. S4 and S5).
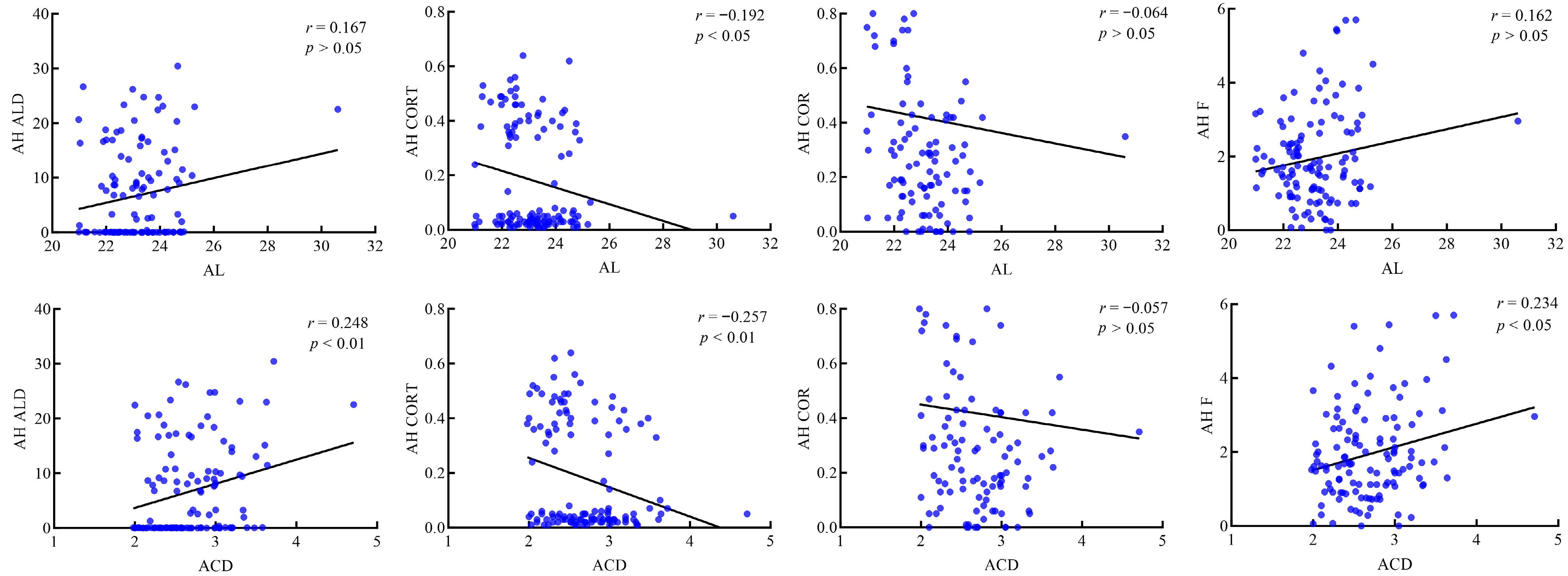
Figure 4.
Correlation analysis of 121 aqueous humor samples with AL and ACD. Notes: AH, aqueous humor; ALD, aldosterone; CORT, corticosterone; COR, cortisone; F, cortisol.
Comparison of cortisol concentrations among the ARC, CPACG, and POAG groups
-
No significant differences were observed in plasma cortisol concentrations among the ARC, CPACG, and POAG groups (η2 = 0.01, 95% CI: 0.00–0.06, F = 0.24, p = 0.79; Fig. 5a). In contrast, aqueous humor cortisol concentrations were significantly higher in the POAG group compared to both the ARC and CPACG groups (η2 = 0.09, 95% CI: 0.01–0.19, F = 4.65, p = 0.01; Fig. 5b). The plasma-to-aqueous humor cortisol ratio was consequently lower in the POAG group (Table 2).
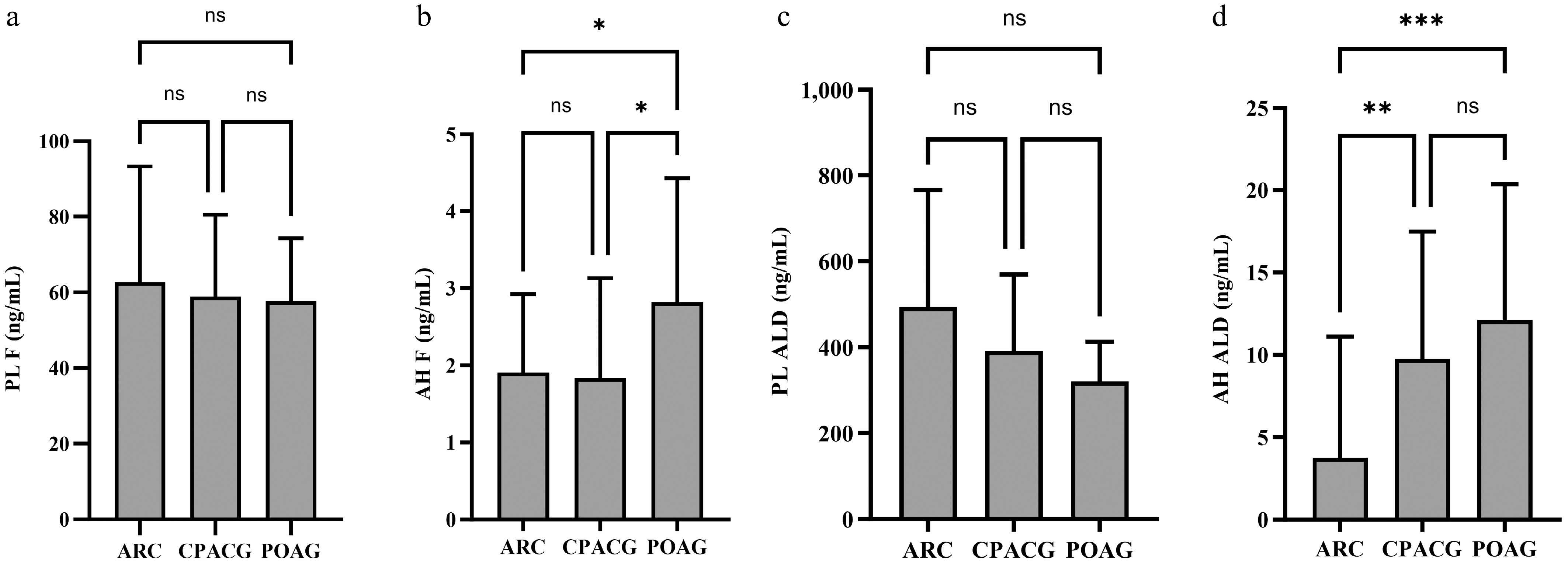
Figure 5.
Comparison of cortisol and aldosterone concentrations among ARC, CPACG, and POAG groups. (a) No significant differences in plasma cortisol concentrations among groups. (b) Aqueous humor cortisol concentrations were significantly higher in POAG compared to ARC and CPACG. (c) No significant differences in plasma aldosterone concentrations. (d) Aqueous humor aldosterone concentrations were elevated in both POAG and CPACG compared to ARC (p < 0.01). Notes: Data are presented as mean ± SD. *p < 0.05, **p < 0.01, ***p < 0.001. AH, aqueous humor; PL, plasma; ALD, aldosterone; CORT, corticosterone; COR, cortisone; F, cortisol. ARC group: 63 aqueous humor, 42 plasma. POAG group: 19 aqueous humor, 12 plasma. CPACG group: 20 aqueous humor, 20 plasma.
Table 2. Comparison of cortisol concentrations (ng/mL) and corresponding ratios in PL and AH among ARC, CPACG, and POAG groups.
Group ARC CPACG POAG η2 (95% CI) F P PL 62.73 ± 30.58 58.89 ± 21.64 57.71 ± 16.55 0.01 (0.00~0.06) 0.24 0.785 AH 1.91 ± 1.02 1.84 ± 1.29 2.82 ± 1.61*# 0.09 (0.01~0.19) 4.65 0.012 PL/AH 32.9 32.03 20.49 AH, aqueous humor; PL, plasma. * Compared with the ARC group, p < 0.05; # Compared with the CPACG group, p < 0.05. Group comparisons: one-way ANOVA and post hoc pairwise comparisons were conducted using the Bonferroni method. Similarly, plasma aldosterone concentrations did not differ significantly among the three groups (Fig. 5c). Aqueous humor aldosterone concentrations were substantially higher in both the CPACG and POAG groups compared to the ARC group (p < 0.01 for both; Fig. 5d), with no significant difference between the CPACG and POAG groups.
Comparisons of structural and functional glaucomatous damage between CPACG and POAG patients revealed no significant differences in global RNFL thickness (62.70 ± 17.88 vs 55.68 ± 13.45 μm; Cohen's d = 0.44, 95% CI: –0.20–1.08, t = 1.38, p = 0.18) or visual field mean deviation (–26.41 ± 7.39 vs –25.42 ± 5.61 dB; Cohen's d = –0.15, 95% CI: –0.78–0.48, t = –0.47, p = 0.64) (Supplementary Table S3).
No significant differences in plasma or aqueous humor cortisol or aldosterone concentrations were found between the CPACG and PAC/PACS groups (all p > 0.05; Table 3).
Table 3. Comparison of ALD and F concentrations (ng/mL) in PL and AH, AL and ACD values (mm) between the CPACG and PAC/PACS groups.
Variables SHs CPACG PAC/PACS d (95% CI) t P PL ALD 391.4 ± 177.8 334.3 ± 178.5 0.32 (−0.32~0.96) 0.99 0.330 F 58.89 ± 21.64 43.30 ± 30.10 0.60 (−0.06~1.25) 1.82 0.079 AH ALD 9.77 ± 7.23 7.94 ± 8.45 0.23 (−0.41~0.85) 0.70 0.486 F 1.84 ± 1.29 1.39 ± 1.13 0.37 (−0.28~1.01) 1.13 0.266 AL 22.38 ± 0.89 22.53 ± 0.84 0.17 (−0.46~0.80) 0.52 0.605 ACD 2.37 ± 0.24 2.34 ± 0.19 0.10 (−0.53~0.73) 0.33 0.746 AH, aqueous humor; PL, plasma; SHs, steroid hormones; ALD, aldosterone; F, cortisol. Group comparisons: unpaired t-test. -
This study investigated the distribution and relationship of steroid hormones in the plasma and aqueous humor of patients with glaucoma. It also examined metabolic patterns in the two steroid pathways. The findings demonstrated that the total steroid hormone concentrations were notably lower in aqueous humor than in plasma. Moreover, steroid hormone concentrations were not significantly associated with age, gender, AL, or ACD in the control group. Furthermore, elevated aldosterone, the active metabolite in the mineralocorticoid pathway, was observed in both patients with POAG and those with CPACG. In addition, cortisol, the active GC metabolite, was increased explicitly in POAG patients, consistent with previous literature[27]. A positive correlation was found between cortisol and cortisone, and a negative correlation was found between corticosterone and aldosterone in aqueous humor. In contrast, positive correlations (corticosterone–aldosterone, cortisol–cortisone) were observed in plasma. These findings suggest that steroid hormones in aqueous humor show tissue-specific metabolic patterns. These results suggest endogenous GC and mineralocorticoid hormones are biologically active, potentially coordinating their effects in the pathogenesis of glaucoma, and may play a role in the pathogenesis of primary glaucoma.
Distribution and relationship of steroid hormones between aqueous humor and plasma
-
Regarding the distribution and relationship of steroid hormones between aqueous humor and plasma, an intriguing paradox emerges from the data: while the overall distribution pattern of steroids in aqueous humor mirrors that in plasma, no significant correlation was found between matched plasma and aqueous humor levels of individual key hormones, such as cortisol. This apparent contradiction challenges the notion of simple passive diffusion, instead pointing to a more complex, regulated interplay between systemic availability and local ocular dynamics. The lack of individual-level correlation, despite population-level distribution similarity, can be explained by several mechanisms:
First, selective blood-aqueous barrier (BAB) regulation: The BAB may act as a selective regulator rather than a passive conduit, with active transport mechanisms and tight junction integrity determining the penetration of hormones into the anterior chamber. Small lipophilic molecules, such as cortisol and aldosterone, diffuse freely across cell membranes; however, many essential metabolites depend on active transcellular transport systems[33,34]. In vitro and animal studies confirm that the BAB significantly regulates intraocular GC levels, and barrier disruption can alter plasma-to-ocular transport[35−37]. Thus, both steroid hormone properties and disease-induced changes to the BAB likely contribute to differences in aqueous humor hormone concentrations. This could create similar concentration gradients across populations while allowing individual variability based on BAB function[33].
Second, local ocular metabolism: The absence of a correlation between plasma and aqueous humor cortisol levels, despite similar distribution patterns, suggests significant local metabolism within the eye. The strong correlation between cortisol and cortisone in aqueous humor indicates active 11 β-HSD2-mediated conversion within the eye, potentially buffering against fluctuations in plasma levels[38].
Third, tissue sequestration and binding: Differential protein binding in plasma vs aqueous humor may dissociate total hormone levels from bioavailable fractions, while tissue sequestration in the iris, ciliary body, or TM could create local reservoirs that modulate aqueous humor concentrations independently of immediate plasma levels[39].
Therefore, the data support a refined model in which aqueous humor hormones are primarily plasma-derived but are selectively regulated and dynamically modulated at the ocular level. The overall hierarchy is preserved (hence mirrored distribution), but fine-tuning occurs locally, explaining the lack of individual correlation.
Synergistic roles of GCs and mineralocorticoids in glaucoma pathogenesis
-
This study reveals coordinated dysregulation of GC and mineralocorticoid pathways in glaucoma patients. Critically, the elevated aqueous humor cortisol in POAG appears to be a specific pathophysiological feature rather than a mere consequence of greater overall disease severity. This conclusion is supported by similar structural and functional glaucomatous damage observed in both POAG and CPACG groups. Furthermore, CPACG patients did not show identical hormone elevations compared to PAC/PACS patients, reinforcing the specificity of these changes to POAG. This evidence strengthens the interpretation that the observed hormonal differences are linked to distinct underlying mechanisms specific to POAG, such as BAB dysfunction or TM-specific receptor activation, rather than being a non-specific correlate of advanced neural damage or general increases in IOP.
The pathological mechanisms are likely to involve the signaling of glucocorticoid receptors (GR) and mineralocorticoid receptors (MR) in the TM. Human TM cells express functional GR and MR[40,41]. Activation of these receptors can lead to cytoskeletal changes and the deposition of ECM, thereby increasing outflow resistance. For instance, Raghunathan et al.[42] demonstrated that dexamethasone treatment significantly stiffens the TM and its cellular matrix. Furthermore, Patel et al.[43] provided a comprehensive review of GR signaling as a key pathway in glaucoma pathogenesis.
Based on evidence suggesting BAB dysfunction in glaucoma[33], including clinical studies demonstrating increased aqueous flare and inflammatory markers in POAG patients[44] and reviews of barrier pathology[33], a primary BAB disruption in POAG is hypothesized to lead to elevated cortisol levels in the aqueous humor. This excess cortisol could then impair TM function via GC receptors and subsequently raise IOP, thus creating a vicious cycle[40,42,43]. This potential mechanism resembles the pathogenesis of glaucoma induced by GC. While this hypothesis remains speculative in the present study, lacking direct BAB integrity markers, it is supported by established links between barrier breakdown and aqueous humor composition in other ocular conditions[45].
Similar to cortisol, aldosterone levels in aqueous humor were elevated in both POAG and CPACG patients. This is a novel finding that has not been previously reported in the literature. Earlier studies on central serous chorioretinopathy have suggested that GC and mineralocorticoid imbalances in the aqueous humor are involved in the disease's pathogenesis[29]. Recent research identified the ocular renin-angiotensin-aldosterone system (RAAS) as a contributing factor in glaucoma pathogenesis. Overactivation of the RAAS promotes inflammation, tissue remodeling, and oxidative stress, while RAAS modulators may slow glaucoma progression and reduce disease-related damage[46]. Therefore, increased aldosterone in the aqueous humor, likely acting through the MR as part of the RAAS pathway, may be involved in early glaucoma stages, including PAC/PACS.
The strong positive correlation between cortisol and aldosterone in both aqueous humor and plasma suggests a coordinated dysregulation that may be more pathologically significant than the action of either hormone alone. This synergy could operate through several mechanisms: (1) Receptor cross-talk: GR and MR exhibit significant cross-regulation, with GR activation potentially potentiating MR-mediated transcription[47,48]; (2) Shared upstream regulation: Both cortisol and aldosterone production are under the stimulatory control of adrenocorticotropic hormone and other stress-responsive mediators of the hypothalamic-pituitary-adrenal axis[49]; (3) Complementary pathological effects: While cortisol primarily induces ECM deposition and cytoskeletal changes in TM cells[42,43], aldosterone promotes pro-inflammatory and pro-fibrotic responses in various tissues, including the eye[50,51]. Previous studies have reported that systemic GC and mineralocorticoid metabolic pathways play synergistic roles in adapting to different stressors[47,48]. Findings suggest similar positive synergy between GC and mineralocorticoid metabolic pathways in the aqueous humor of glaucoma patients, potentially regulating ocular stress. The positive correlation between aldosterone and cortisol in aqueous humor with ACD suggests enhanced activation of these metabolic pathways in glaucoma patients with a deeper anterior chamber, such as those with POAG.
Clinical implications and therapeutic perspectives
-
The clinical implications of this coordinated dysregulation are substantial. Rather than targeting a single hormonal pathway, dual GR/MR modulation may provide superior therapeutic efficacy. This approach may be particularly relevant for patients with POAG who exhibit concurrent elevations of both hormones. Recent studies in cardiovascular diseases demonstrate that combined GR/MR inhibition produces synergistic benefits beyond single-pathway blockade[46], suggesting similar strategies might be effective in glaucoma management. From a clinical perspective, the cortisol-aldosterone correlation suggests that hormonal profiling might identify glaucoma subtypes most likely to benefit from targeted endocrine therapies.
Limitations and future directions
-
This study proposes a novel perspective to elucidate glaucoma pathogenesis. Specifically, the endogenous hormone metabolic activity in POAG may represent a promising direction for future treatments. However, this study has limitations. First, the modest sample size, particularly in the glaucoma subgroups, may have reduced statistical power for some comparisons, limited generalizability, and rendered the exploratory subgroup analyses (e.g., RNFL/VF comparisons) underpowered to detect subtle differences. The considerable biological variability in steroid hormone levels, as evidenced by the large standard deviations, further underscores the need for larger cohorts to confirm these findings with greater precision. Second, without direct measurements of GR/MR receptor activity, downstream signaling, or ECM changes in TM tissues, the precise pathological significance of elevated aqueous humor steroids remains speculative. Third, the hypothesized role of BAB dysfunction remains speculative without direct measurement of integrity markers (e.g., aqueous humor protein or cytokines). Fourth, the correlational analyses presented are exploratory and were not adjusted for potential confounders such as age, gender, or medication use. While this approach provides a clear view of direct associations for hypothesis generation, it does not establish causality or independent effects. Fifth, although the systemic effects of topical β-blockers are believed to be minimal based on existing literature, the potential contribution of these agents to the observed hormonal differences cannot be definitely excluded, as the perfect collinearity between medication use and disease status precluded statistical adjustment for this factor. Ultimately, the lack of clinical validation limits the ability to draw definitive associations between hormone levels and specific clinical outcomes.
Future research should: (1) quantify GR/MR expression in TM tissues from glaucoma patients; (2) examine cortisol/aldosterone-induced ECM changes in primary TM cell cultures; (3) test whether MR antagonists can prevent hormone-mediated TM dysfunction; (4) incorporate direct BAB integrity assessment through proteomic analysis of aqueous humor; and (5) employ larger cohorts to conduct multivariate analyses adjusting for confounders and to perform sensitivity analyses, thereby confirming the robustness and independence of the observed associations.
-
The study adhered to the Declaration of Helsinki, and the study protocol received approval from the Ethics Committee of the Third People's Hospital of Huizhou (Approval No. 2023-KY-102-01; dated November 17, 2023). All sample collection procedures followed the approved ethical protocols, and informed consent was obtained from all participants.
-
The authors confirm contribution to the paper as follows: study conception and design: Zhao J, Chou T; data acquisition, analysis and interpretation, critical revision of manuscript: Deng S, Huang X, Dai D, Zhou H, Yin L, Zhang D, Zeng H, Zhu Y, Chou T, Zhao J; statistical analysis: Deng S, Huang X; draft manuscript preparation: Deng S, Huang X; administrative, technical and material support: Zhao J, Chou T; study supervision: Zhao J, Chou T; funding acquisition: Zhao J, Deng S, Huang X, Zhu Y. All authors reviewed the results and approved the final version of the manuscript.
-
All data used in this study are available from the corresponding author; however, the data are unsuitable for posting, as the research data includes sensitive or confidential information, such as patient data.
-
This research was financially supported by the National Natural Science Foundation of China (Grant No. 82471056), Guangdong Basic and Applied Basic Research Foundation (Grant Nos 2024A1515140098 and 2024A1515013058), Shenzhen Science and Technology Program (Grant No. JCYJ20250604184013017).
-
The authors declare that they have no conflict of interest.
-
Authors contributed equally: Shuifeng Deng, Xiaosheng Huang
- Supplementary Table S1 Comparison of steroid hormone concentrations (ng/mL) by gender in the ARC group.
- Supplementary Table S2 Comparison of age and gender distribution among diagnostic groups.
- Supplementary Table S3 Comparison of RNFL (μm) and visual field defects (dB) between CPACG and POAG groups.
- Supplementary Fig. S1 Correlation analysis of aldosterone, corticosterone, cortisone, and cortisol with age in 42 plasma and 63 aqueous humor samples from the ARC group.
- Supplementary Fig. S2 Metabolic pathway diagram of steroid hormones.
- Supplementary Fig. S3 Correlation analysis of steroid hormones in 92 plasma samples with AL and ACD.
- Supplementary Fig. S4 Correlation analysis of steroid hormones and ACD in 82 AH samples from open-angle (POAG + ARC) subgroup.
- Supplementary Fig. S5 Correlation analysis of steroid hormones and ACD in 39 AH samples from angle-closure (CPACG + PAC/PACS) subgroup.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Deng S, Huang X, Dai D, Zhou H, Yin L, et al. 2026. Investigation of steroid hormone imbalance in the aqueous humor and plasma of glaucoma patients. Visual Neuroscience 43: e012 doi: 10.48130/vns-0026-0004
Investigation of steroid hormone imbalance in the aqueous humor and plasma of glaucoma patients
- Received: 08 July 2025
- Revised: 29 December 2025
- Accepted: 05 January 2026
- Published online: 23 March 2026
Abstract: Steroid hormones regulate intraocular pressure and may influence the pathophysiology of glaucoma. However, the role of endogenous steroid hormones in glaucoma patients remains poorly understood. Steroid hormones were analyzed using liquid chromatography-tandem mass spectrometry in paired aqueous humor (n = 121) and plasma (n = 92) samples from patients with primary open-angle glaucoma (POAG), chronic primary angle-closure glaucoma (CPACG), primary angle-closure (PAC)/suspect (PACS), and age-related cataract (ARC). Group differences and correlations were assessed using ANOVA and t-tests, along with linear regression, reporting effect sizes and confidence intervals. Six steroid hormones were quantified in plasma; four (aldosterone, corticosterone, cortisone, cortisol) were detected in aqueous humor at significantly lower concentrations (all p < 0.05; plasma-to-aqueous humor ratios: 5.44–62.04), with identical concentration rankings. Cortisol and aldosterone were positively correlated in both aqueous humor (r = 0.535, p < 0.01), and plasma (r = 0.748, p < 0.01). Aqueous humor cortisol levels were significantly higher in POAG than in CPACG and ARC (η2 = 0.09, 95% CI: 0.01–0.19). Aqueous humor aldosterone levels were elevated in POAG and CPACG compared to ARC (both p < 0.01), with no significant difference between CPACG and POAG. No significant differences in aldosterone or cortisol were observed between CPACG and PAC/PACS. Steroid hormones in aqueous humor originate primarily from plasma but exhibit selective distribution. Endogenous glucocorticoid and mineralocorticoid levels are altered in glaucoma, suggesting a potential coordinated role in its pathophysiology. This study has registered in Chinese Clinical Trial Registry (www.chictr.org.cn/showprojEN.html?proj=220916; ID: ChiCTR2400081103; Date of Registration: 2024-02-22).














