-
The slowdown in agricultural productivity, driven by changing weather patterns and the depletion of fertile land, poses a significant challenge for vegetable crops. Current statistics predict the need to quadruple food yields by 2050 to sustain a global population of 10 billion[1]. Achieving sustainable food production is increasingly constrained by climate change, which intensifies abiotic and biotic stresses in vegetable crops, resulting in substantial yield losses. Recent studies indicate that heat, drought, and pest pressures could collectively reduce global vegetable yields by 10%–20% by 2050, with the most severe impacts expected in tropical and subtropical regions[2]. Abiotic stresses, including heat, drought, cold, salinity, and ultraviolet radiation, along with pests and diseases, reduce vegetable crop yield and quality. For example, cold stress affects broccoli inflorescence development and vernalization in cabbage and kale[3], while heat stress reduces tomato, chili, and pepper yields by up to 50%[4]. To ensure sustainable vegetable crop development, breeders focus on creating cultivars with enhanced disease resistance, stress tolerance, high yield, and superior nutritional value. Traditional breeding methods have contributed to the evolution of crop varieties with unique traits. They are dependent on phenotypic characteristics, and as a consequence of the breeder's knowledge, many superior crop varieties were produced. Genetic features and selection methods are essential for the development of desirable plant cultivars[5]. However, these conventional breeding methods require several generations to produce stable lines and remove undesirable traits. Thus, directed mutagenesis, rapid variant detection, and efficient mutant screening have significantly enhanced modern breeding programs. Consequently, plant breeding has evolved to incorporate advanced biotechnological approaches such as cisgenesis, genome editing, and speed breeding. The integration of genotypic and phenotypic information through these tools facilitates more precise and efficient crop improvement.
Moreover, the application of molecular technologies, including molecular markers, marker-assisted selection, quantitative trait loci (QTL) mapping, and CRISPR/Cas-based genome editing, has fostered a unified framework for genetic enhancement, providing powerful strategies to improve the resilience and productivity of vegetable crops. This review paper aims to provide a thorough analysis of traditional, molecular, and speed breeding methods. It examines how genomic technologies can be integrated with speed breeding to increase the efficiency and accuracy of cultivar development, and emphasizes the need for contemporary breeding methods to maximize agricultural resources for urban food production. A comparative study has been carried out to clarify the potential roles of conventional, molecular, and speed breeding techniques.
-
Conventional breeding has been the foundation for vegetable crop improvement, relying on natural inheritance, hybridization, and phenotypic selection. This type of breeding is confined to sexually compatible species, hence restricting the introduction of novel traits. Some of the key factors involved in this process are interspecific crossover, male sterility, and the selection of appropriate hybrids or stable lines. Male sterility is affected by both hereditary and environmental influences, illustrated in Fig. 1.
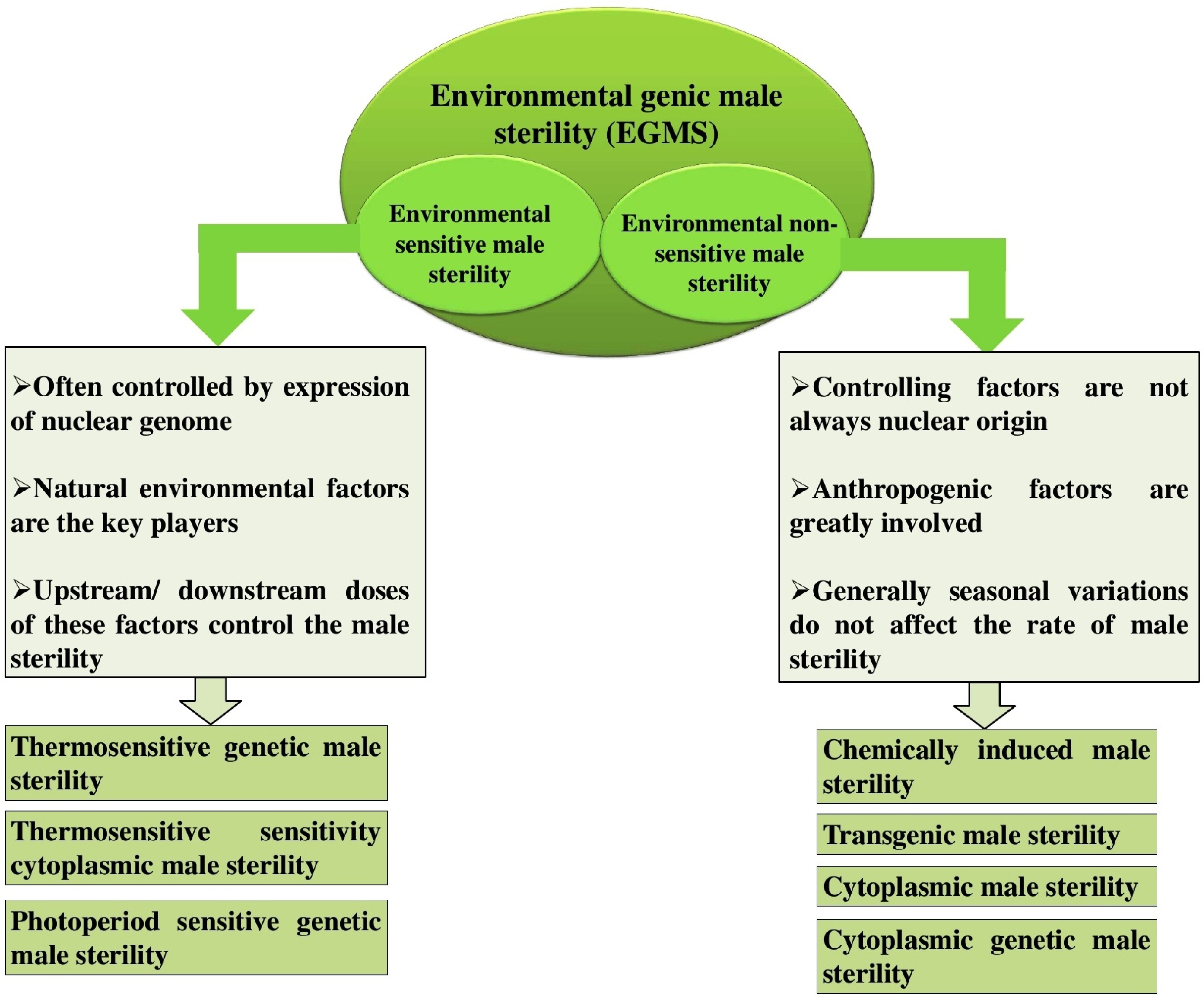
Figure 1.
Types of environmental genic male sterility (EGMS). Environmental sensitive male sterility (ESMS): Male sterility triggered by specific environmental conditions. Environmental non-sensitive male sterility (ENSMS): Male sterility that remains unaffected by environmental variations.
The transmission of characteristics can frequently result in unexpected features, some of which may adversely affect plant development and yield. Table 1 illustrates various ways utilized to improve stress tolerance via plant breeding. Despite these challenges, conventional breeding has been instrumental in developing widely improved varieties of vegetable crops. Figure 2 illustrates several reproductive activities among horticulture plants, including cross-pollination, self-pollination, vegetative pollination, and other techniques.
Table 1. Different breeding approaches for various kinds of stress tolerance.
Types of stress tolerance Breeding approaches Ref. Heat stress tolerance Selection and evaluation of breeding lines under high temperature conditions to identify heat-tolerant genotypes.
Use of physiological trait-based selection, such as canopy temperature and membrane stability.
Implementation of molecular and genomic-assisted breeding to accelerate identification of stress-resilient lines.[6,7] Cold tolerance Conventional breeding to select genotypes adapted to low-temperature environments.
Development of genetically modified plants carrying cold-tolerance genes.
Genome editing technologies are applied to introduce or modify genes regulating cold stress response.
[8]Salt stress tolerance Breeding programs aimed at developing cultivars with stable yield under saline conditions.
Introgression of salt-tolerance alleles into elite, high-yielding backgrounds.
Application of quantitative trait loci (QTL) mapping and marker-assisted selection (MAS) to identify and transfer salt-tolerant traits.
Marker-assisted backcrossing and genome-editing techniques for precise genetic improvement.[9,10] Drought stress tolerance Selection for superior yield stability under both drought and non-stress conditions.
Incorporation of drought adaptation traits, such as root architecture and water use efficiency.
Use of molecular markers, genomic selection, and genome-editing tools like CRISPR/Cas9 to enhance drought resilience.[11] Waterlogging tolerance Developing hybrids through crossing tolerant and susceptible genotypes.
Collection and evaluation of germplasm for waterlogging-tolerant traits.
Investigation of genetic mechanisms involved in anaerobic stress adaptation.
Utilization of QTL mapping to locate genes associated with tolerance.[12,13] 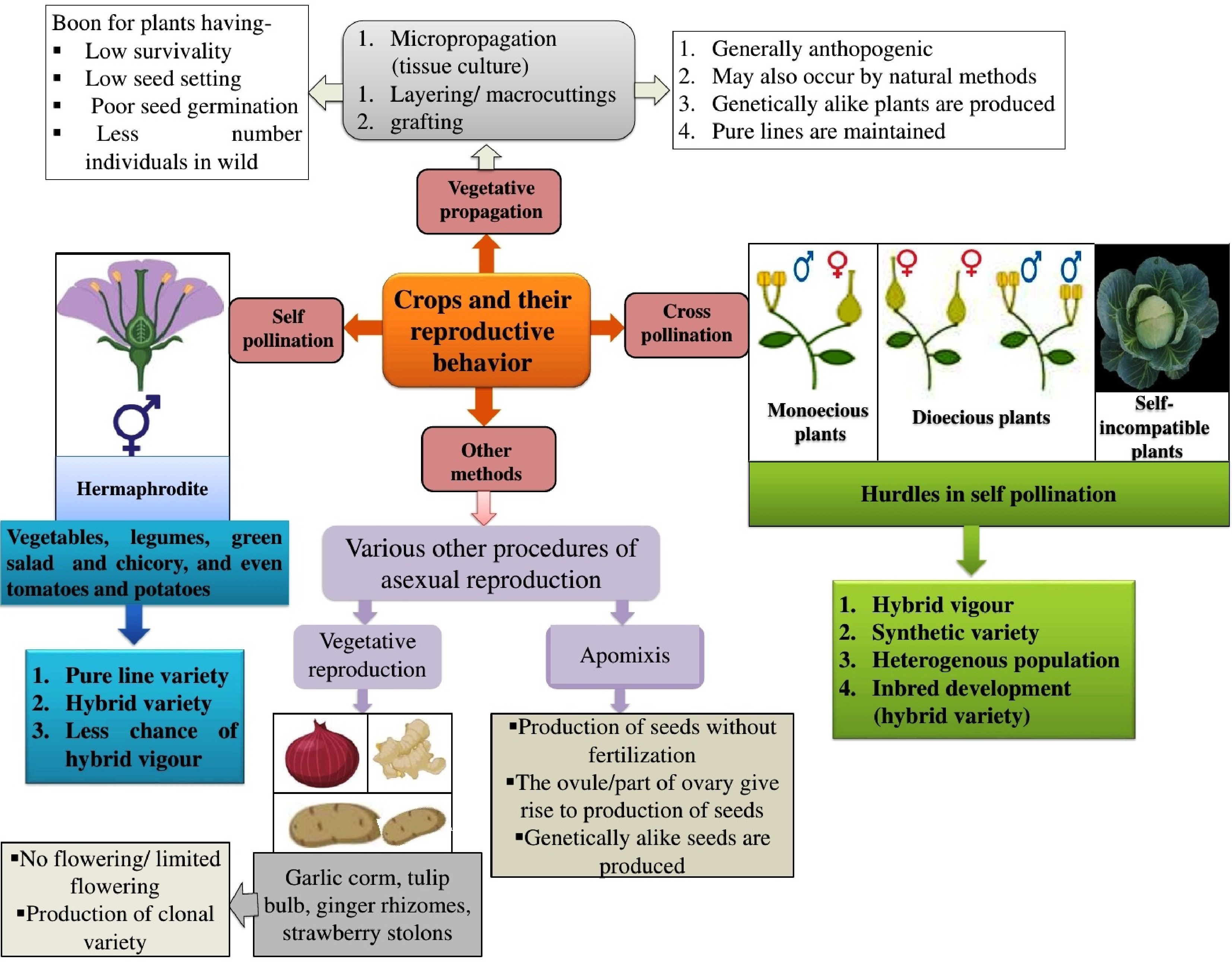
Figure 2.
Different reproductive strategies in plants showcasing the varied reproductive behaviours observed in vegetable crops.
Conventional approaches use several breeding strategies, as shown in Fig. 3, and their suitable examples are discussed in Table 2.
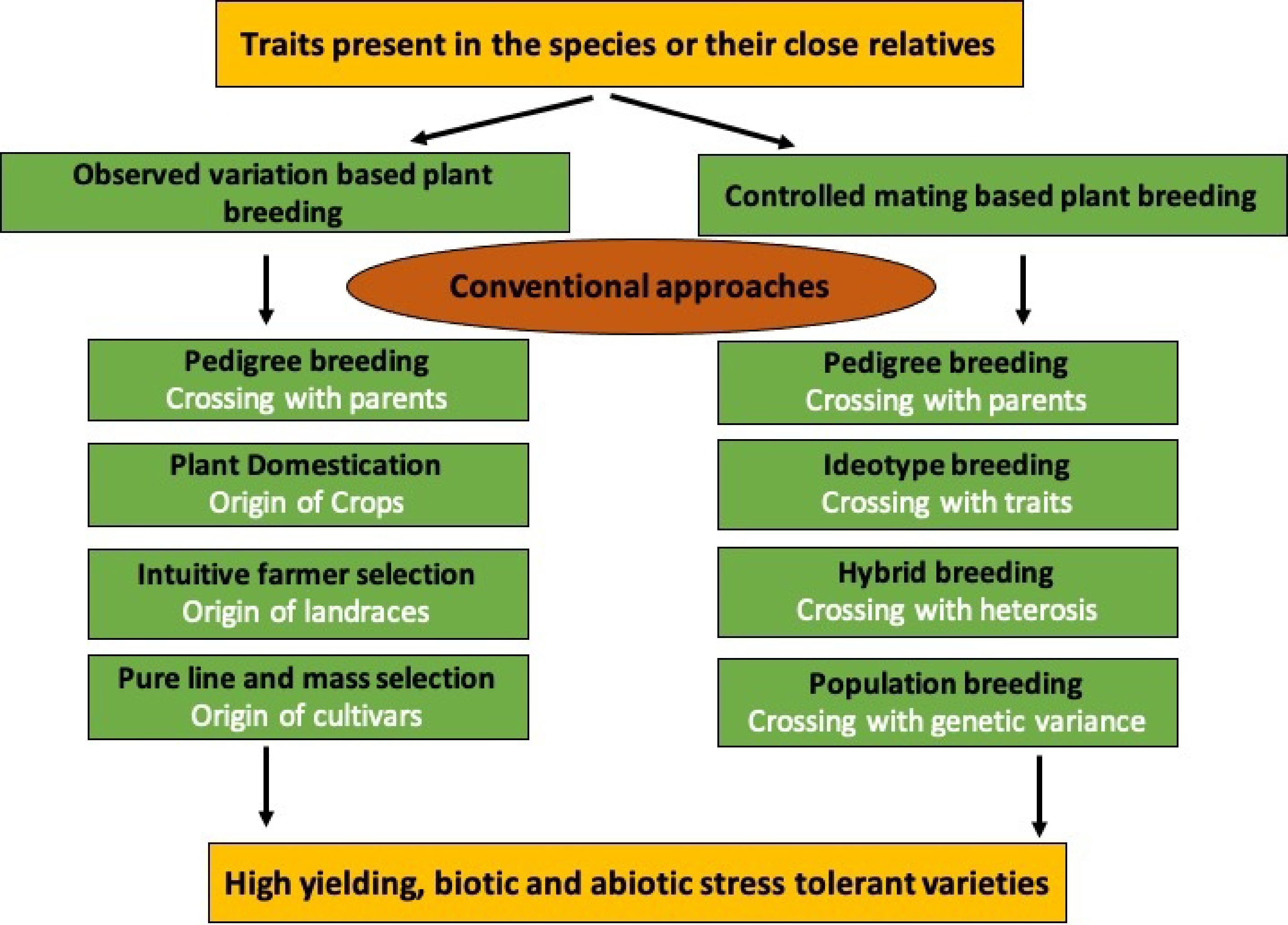
Figure 3.
Graphical overview of conventional breeding methods illustrating the traditional approaches used in vegetable crops.
Table 2. Overview of conventional breeding approaches in vegetable crops and their associated trait improvements.
Vegetable crops Breeding method Trait improved Ref. Tomato (Solanum lycopersicum) Backcross Enhanced tolerance to late blight, improved fruit texture [15] Brinjal/eggplant (Solanum melongena) Backcross Resistance to shoot and fruit borer, and yield stability [16] Chilli/pepper (Capsicum annuum) Pedigree Better fruit quality, improved disease tolerance, consistent pungency [17] Okra (Abelmoschus esculentus) Pure-line Increased yield, early maturity, uniform fruit traits [18] French bean (Phaseolus vulgaris) Pure-line High pod uniformity, disease, and pest resistance [18] Cowpea (Vigna unguiculata) Mass selection Larger fruit size, stress tolerance, and vine vigor [19] Cucumber (Cucumis sativus) Mass selection Bulb size uniformity, improved shelf life, and early maturity [20] Bottle gourd (Lagenaria siceraria) Recurrent selection Compact head formation, tolerance to pests [20] Onion (Allium cepa) Pedigree High curd compactness, adaptability to temperature stress [21] Cabbage (Brassica oleracea var. capitata) Mass selection Improved root color, sugar content, and shape regularity [14] Cauliflower (Brassica oleracea var. botrytis) Recurrent selection Resistance to powdery mildew, enhanced cold tolerance [22] Carrot (Daucus carota subsp. sativus) Mass selection Increased leaf yield, resistance to downy mildew [23] Pea (Pisum sativum) Backcross Higher fruit size, drought tolerance, and yield stability [24] Spinach (Spinacia oleracea) Pedigree Enhanced leaf yield, heat, and drought tolerance [25] Bitter gourd (Momordica charantia Mass selection High fruit set, color intensity, and disease tolerance [20] Mass selection has similarly been employed to increase the frequency of favourable traits within a population, where both male and female gametes can be managed to maximize desirable inheritance (Fig. 4).
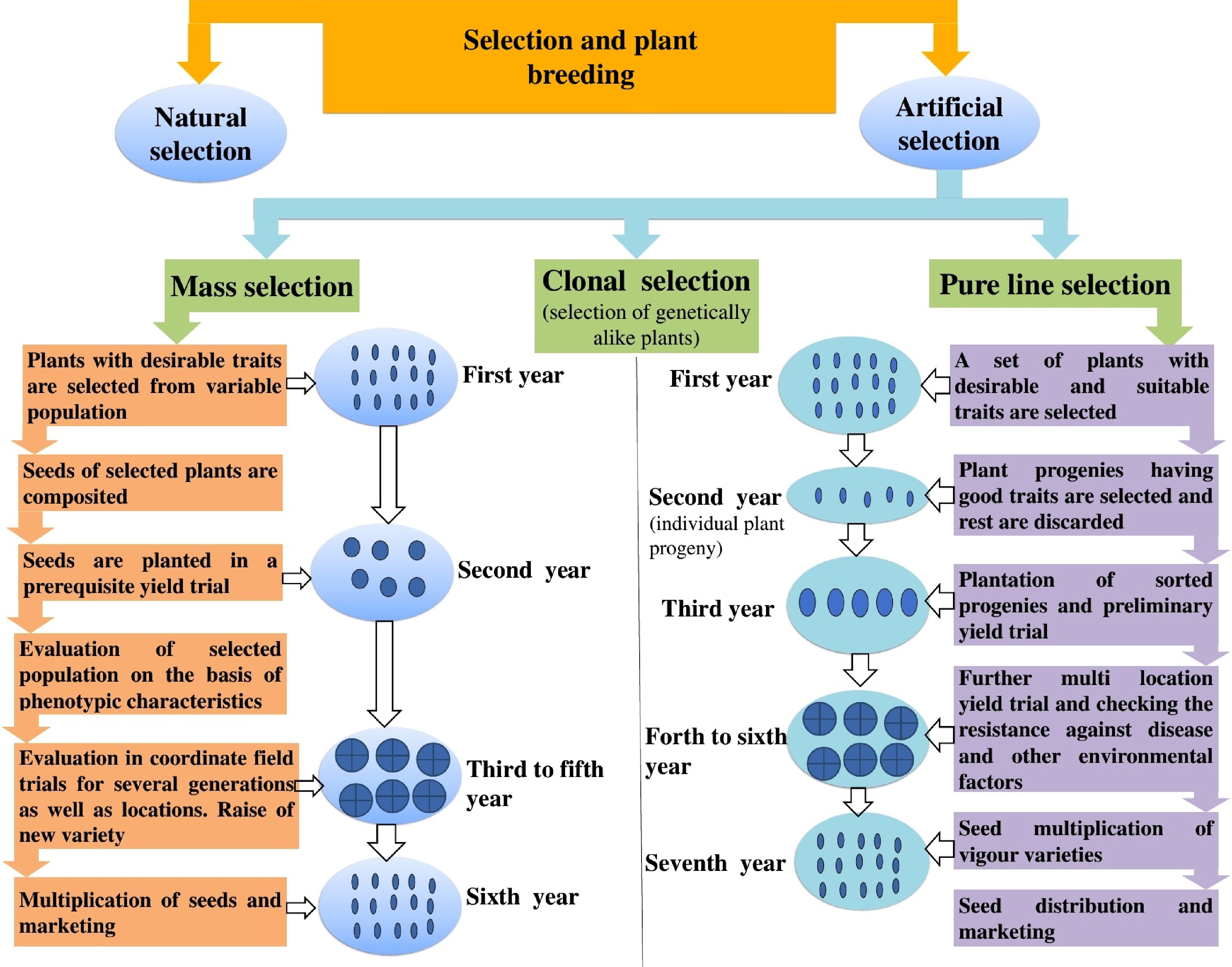
Figure 4.
Role of various selection strategies in plant breeding. This figure demonstrates how different selection methods contribute to the improvement of vegetable crop plants.
Recurrent selection has also been widely applied, using repeated cycles of phenotypic selection within segregating populations to progressively enrich desirable alleles, particularly for complex quantitative traits such as yield and stress tolerance[14]. Pedigree breeding, which traces the ancestry of cultivars through controlled hybridizations, has been successful in enhancing qualitative traits such as vegetable flavour, colour, and disease resistance (Fig. 5). Gene pyramiding through sequential backcrossing is a powerful breeding strategy that allows the integration of multiple resistance genes into a single genotype. This approach has been widely used in long-duration vegetable crops to achieve durable resistance against various pathogens (Fig. 6). Although the process can be slow and labor-intensive, it continues to play a central role in modern vegetable crop improvement programs.
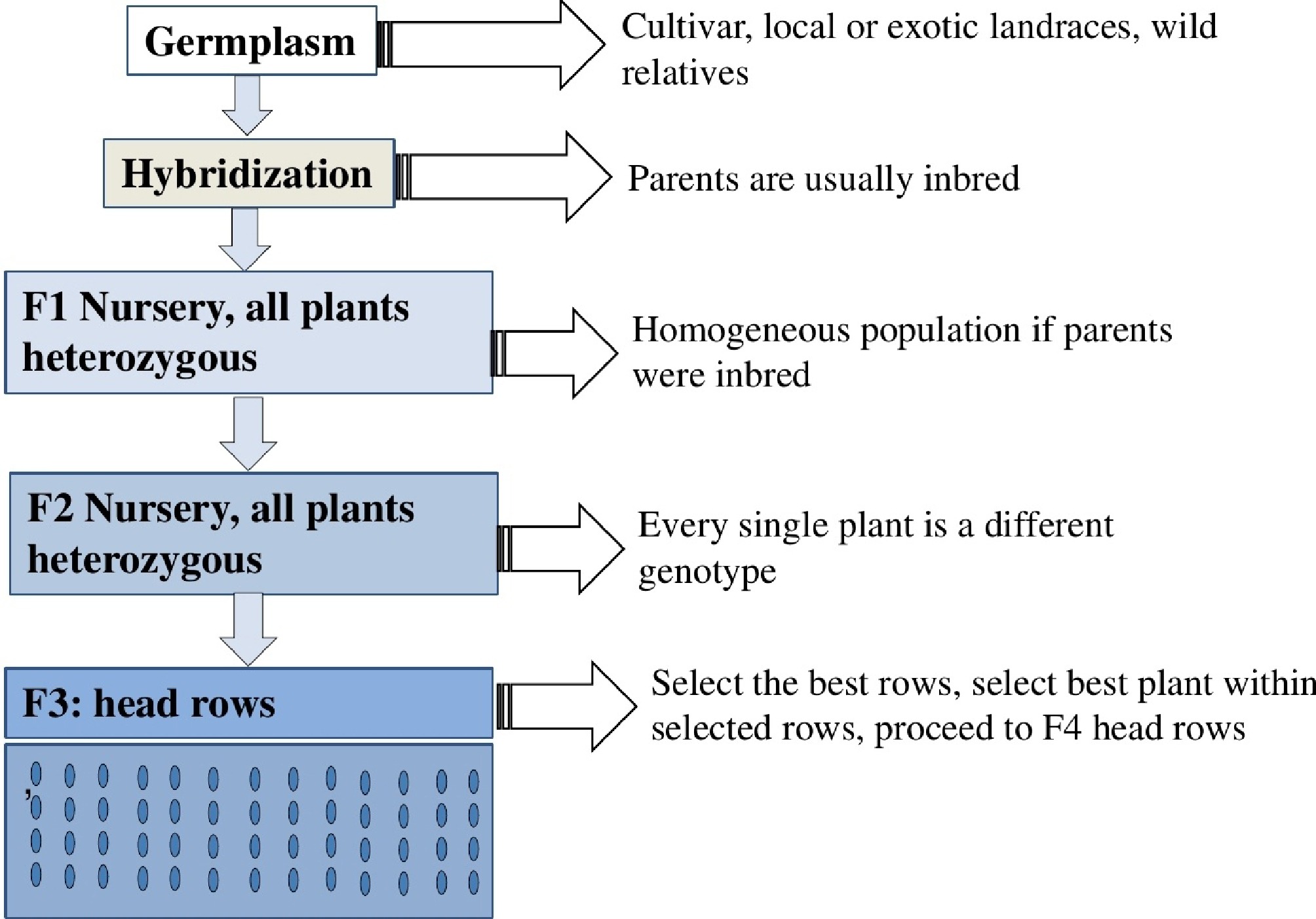
Figure 5.
Graphical presentation of cultivar development in self-pollinated species using the pedigree method, illustrating the typical steps involved in developing new cultivars for self-pollinated species.
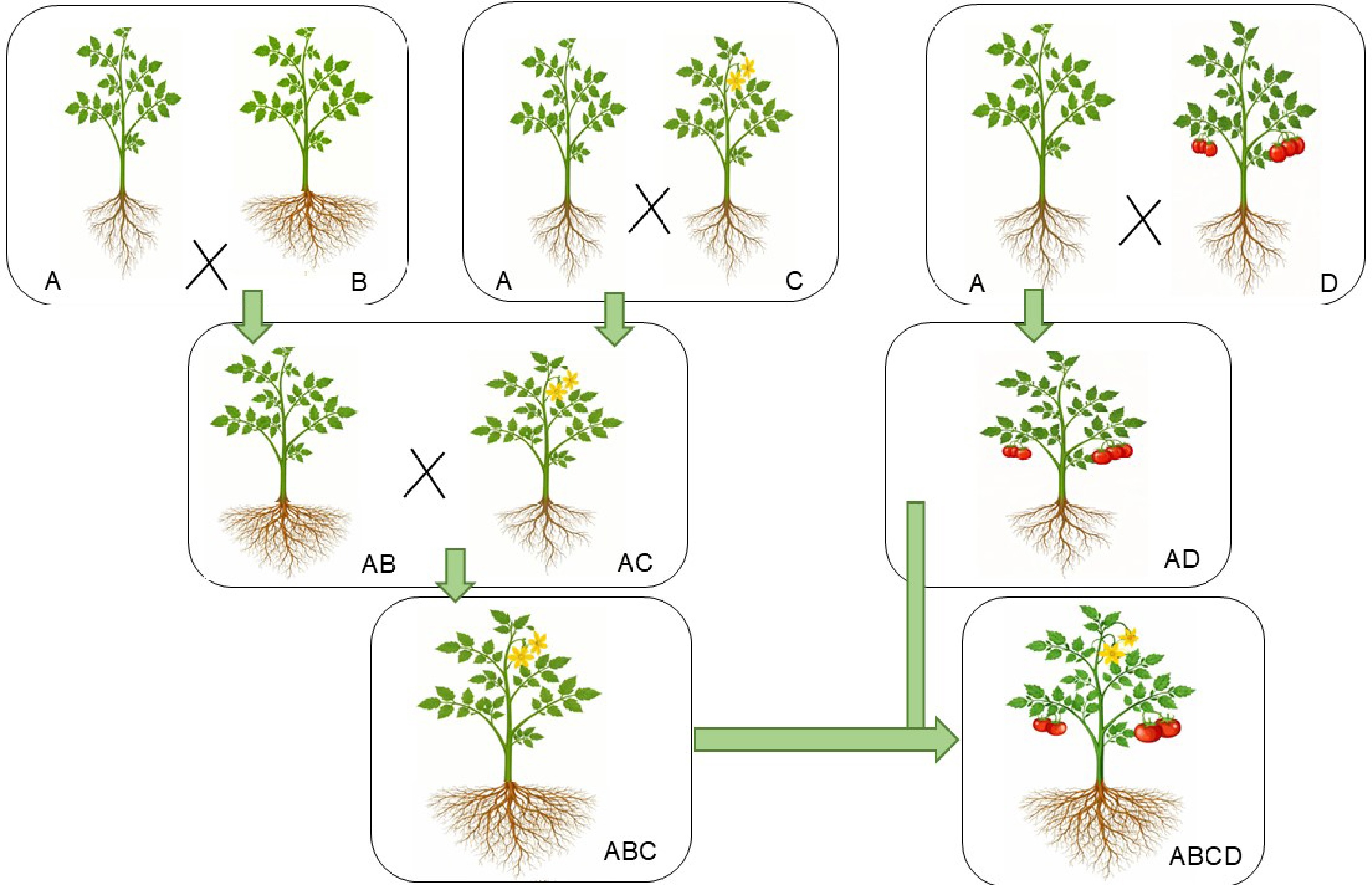
Figure 6.
Gene pyramiding for vegetable crop enhancement and high-yield varieties. This figure highlights the role of gene pyramiding in developing vegetable crops with multiple desirable traits. The key traits include: Trait A, plants with simple shoot and root systems; Trait B, plants with high-yielding roots and simple shoots; Trait C, plants with potent flowering alongside simple root and shoot systems; Trait D, plants with a high rate of fruit setting and simple shoot and root systems, respectively.
Although these methods are often slow and labour-intensive, they remain integral to vegetable crop improvement programs.
Conventional breeding remains highly effective for developing specific resistance traits; however, it often falls short when addressing complex traits such as multi-stress tolerance. Hence, plant breeders increasingly combine conventional methods with advanced molecular tools to accelerate the development of resilient vegetable varieties capable of withstanding diverse biotic and abiotic stresses.
-
Molecular breeding has significantly advanced the nutritional quality, adaptability, and resilience of vegetable crops by improving traits such as protein digestibility, essential amino acid composition, taste, aroma, and tolerance to abiotic and biotic stress. High-accuracy telomere-to-telomere sequencing has further expanded opportunities for vegetable crop improvement. For instance, genome data in cucumber (Cucumis sativus), has enabled the identification of genes linked to bitterness and fruit length. Mutation breeding has generated high-yielding okra (Abelmoschus esculentus) and disease-resistant onion (Allium cepa) lines, while genome editing through CRISPR/Cas9 has been successfully applied in tomato, lettuce, cabbage, chilli, etc., to enhance tolerance against abiotic stresses. These examples highlight how molecular breeding integrates genomic tools with classical approaches to create cultivars with improved nutritional profiles, stress resilience, and consumer-preferred qualities. Collectively, these molecular advances bridge the gap between genotype and phenotype, enabling the precise improvement of nutritional and agronomic traits.
Marker-assisted selection (MAS): bridging tradition and technology
-
MAS is a powerful tool that helps breeders increase vegetable crop yields by precisely selecting and transferring beneficial genetic traits. It tracks DNA variations within, and between species, creating new genetic diversity. MAS also accelerates traditional breeding steps such as germplasm characterization, genetic mapping, gene tagging, and introducing genes from wild or exotic species into cultivated vegetable crops. A key strength of MAS is its ability to detect and select quantitative trait loci (QTLs) in breeding populations, regardless of environmental conditions, often at early developmental stages[26]. MAS approaches include genome-wide association studies (GWAS), marker-assisted recurrent selection (MARS), genomic selection (GS), and marker-assisted backcrossing (MABC)[16]. MARS and MABC are widely used to develop climate-resilient vegetable crops, especially maize, demonstrating MAS's practical value in molecular breeding. MAS allows early selection of desirable traits, often at juvenile plant stages, and DNA markers help identify genomic regions linked to unwanted traits, which can be eliminated, while retaining beneficial traits[27]. This integration of molecular techniques into conventional breeding enhances vegetable crop productivity and resilience.
Despite its advantages, MAS has some limitations. The development and validation of reliable markers can be time-consuming and costly. MAS is most effective for traits controlled by a few major genes, while complex traits governed by many small-effect genes may be harder to improve. Its application in many vegetable crops remains limited, and field validation across different environments is often lacking. Additionally, integrating MAS with high-throughput phenotyping and genomic selection is still under development, highlighting opportunities for further research. The following section will explore deeper into the individual DNA markers employed in MAS and their significance for plant breeding.
Molecular marker: unravelling the genetic blueprint
-
With the advancement of genetic testing technologies such as DNA chips and genetic bit analysis, molecular markers are emerging as promising tools for the future genetic improvement of vegetable crops[28] (Table 3), such as tomato, cabbage, lettuce, and cucurbits, etc.,[29], bringing programs closer to allele composition screening and accelerating the pace of selection and improvement.
Table 3. List of molecular markers used in vegetable crops and their specific advantages.
Marker type Use in vegetable crops Advantages Ref. RFLP (Restriction Fragment Length Polymorphism) Tomato, cabbage Reliable, co-dominant marker; useful for gene tagging and varietal fingerprinting; not affected by environmental conditions [28,30] AFLP (Amplified Fragment Length Polymorphism) Broccoli, pepper, carrot High polymorphism detection; suitable for genome-wide screening; useful for linkage map construction [30] SSR (Simple Sequence Repeats/microsatellites) Cucumber, tomato, brinjal Highly polymorphic, reproducible, co-dominant; ideal for genetic diversity and parentage studies [28] SNP (Single Nucleotide Polymorphism) Pepper, cucumber, tomato Abundant across the genome; co-dominant; suitable for high-throughput and genomic selection studies [30] SCAR (Sequence Characterized Amplified Region) Tomato, chili Highly specific and reproducible; derived from RAPD markers; efficient for detecting specific genes or traits [31] InDel (Insertion–Deletion) Tomato, pepper Stable marker type; valuable for fine mapping and polymorphism studies in breeding populations [31] CAPS (Cleaved Amplified Polymorphic Sequence) Chili, tomato High specificity and co-dominant; useful for mapping and marker-assisted selection of quality traits [31] SRAP (Sequence-Related Amplified Polymorphism) Cabbage, lettuce, cucurbits Targets open reading frames; detects functional polymorphisms related to agronomic traits [29] CRISPR/Cas9 (Genome Editing System) Tomato, cucumber, lettuce Enables targeted gene modification for stress tolerance, disease resistance, and yield improvement [29,32] Molecular breeding enables precise introgression of stress-resistance genes[33], shortening breeding cycles by 30%–50% in vegetables. Genomic selection further accelerates genetic gains by combining molecular data with predictive models[34]. Despite these advantages, over-reliance on markers may narrow genetic diversity, increasing vulnerability to stress. It also requires advanced bioinformatics, infrastructure, and significant investment. Moreover, regulatory and ethical concerns, particularly around gene-editing technologies remain important challenges. Thus, balancing precision with diversity, cost, and regulation is essential for sustainable application.
-
The rapid increase in the global population necessitates an enhancement in agricultural productivity to ensure sustainability. Projected shifts in climate, marked by rising temperatures and extended periods of droughts and floods are predicted to give rise to a higher number of ailments and infestations[34,35]. To develop plant varieties with improved productivity and adaptability to changing climate conditions, it is essential to accelerate genetic advancement in agricultural breeding programs. To achieve this goal, it is imperative to integrate technologies that can expedite the breeding process. In most traditional vegetable crop breeding programs, developing a new crop variety typically entails selecting and crossing parent plants with desired traits, and then systematically selecting the most promising offspring. However, this approach generally requires more than a decade to produce cultivars that meet market demands, substantially delaying commercialization[36]. Plant breeders have adopted methods such as doubled haploid and shuttle breeding to expedite this process. A modern approach increasingly utilized in breeding programs is speed breeding, in which plant populations are cultivated under controlled conditions that significantly enhance generation turnover[37]. Approximately three to nine generations are obtained per year via speed breeding approaches, a significant improvement over conventional breeding techniques, which usually result in only one to two generations per year. As a result, speed breeding enables the rapid development of stable and homozygous genotypes, conserving both time and resources[38]. Furthermore, for the simultaneous selection of several desired traits, speed breeding methods work well with high-throughput phenotyping and marker-assisted selection (MAS). Speed breeding involves manipulating environmental conditions such as temperature, photoperiod, light quality, and intensity (Table 4). By exploiting various biological processes, it aims to accelerate the rate at which plants advance through generations. Hence, speed breeding modifies multiple growth parameters, as outlined below.
Table 4. Various speed breeding factors and their influence on vegetable crops.
S. No. Vegetable crop Key factor Influence/observation Ref. 1 Pea (Pisum sativum) Photoperiod length
(22 h light/17 °C day-night cycle)Accelerated flowering and generation turnover; up to six generations per year achieved under controlled light regimes [39] 2 Hot pepper (Capsicum spp.) Light spectrum and far-red proportion Far-red supplementation (30–90 µmol·m−2 s−1) enhanced early bud initiation and shortened the reproductive phase to achieve four generations annually. [40] 3 Cabbage (Brassica oleracea var. capitata) Supplemental lighting
(blue/red ratio 2:1)An extended photoperiod increased budding rates up to threefold compared to field conditions. [24,41,42] 4 Chinese kale (Brassica alboglabra) Far-red (3–6 W·m−2) illumination Budding and flowering rates improved by 20%–35% relative to control treatments. [41,42] 5 Tomato (Solanum lycopersicum 'Micro Tom') Blue light exposure (~100 µmol·m−2 s−1) Stimulated earlier flowering by 3–4 d and faster fruit ripening under indoor photoperiod systems [43] 6 Tomato (Solanum lycopersicum 'Mini Chal') Supplementary UV-A radiation
(0.4 W·m−2)Mild UV-A stress induced floral initiation and reduced the vegetative period [11] 7 Cowpea (Vigna unguiculata) Planting density modulation Closer spacing (60 × 20 cm) advanced flowering time and improved seed set efficiency [44] 8 Spotted bean (Phaseolus vulgaris) Temperature control (20/18 °C) Moderate thermal regimes enhanced flowering and reduced cycle duration to under 60 d [26] 9 Soybean (Glycine max) Light intensity (480 µmol·m−2 s−1) Optimal light intensity reduced the generation cycle to roughly 75 d [45] 10 Faba bean (Vicia faba) Photoperiod variation (14–22 h) Long-day exposure (18 h) produced the most effective balance between rapid cycling and yield consistency. [46] 11 Amaranth (Amaranthus spp.) Temperature (30–35 °C, 16 h light) Induced early floral transition and enabled up to six generations per year under controlled temperature. [47] 12 Chinese cabbage (Brassica rapa) CO2 enrichment (380 vs 100 ppm) Elevated CO2 promoted photosynthetic efficiency and faster seed maturation [41,42] 13 Pea (Pisum sativum) Temperature + light combination Warmer regimes with 22-h photoperiod shortened the flowering phase by 2.5 × compared to field setups. [48] 14 Bean
(Phaseolus vulgaris L.)Planting at different densities
(10 × 30 cm, 20 × 30 cm, 30 × 30 cm,
40 × 30 cm, 50 × 30 cm, 60 × 30 cm)Planting density of 20 × 30 cm resulted in the highest yield [26] Principles and practices of speed breeding
-
This is an idea for enhancing plant growth and reproduction by carefully controlling environmental conditions such as light, temperature, and photoperiod. In vegetable crops, these practices enable multiple generations to be grown within a single year, thereby reducing breeding time. By optimizing factors like LED light spectra, day length, and nutrient supply, breeders can fast-track the development of improved varieties with desirable traits such as higher yield, stress tolerance, and disease resistance. This approach bridges traditional and modern breeding methods, offering a sustainable path for rapid advancement in vegetable crop improvement.
Photoperiod manipulation
-
Photoperiod, defined as the duration of light within a 24-h cycle, serves as one of the most important environmental cues regulating plant growth and development. It enables plants to synchronize key physiological processes such as flowering, fruiting, and seed set according to seasonal changes. In vegetable crops, photoperiod sensitivity varies widely among species, and even among genotypes within a species[49]. For instance, long-day crops like spinach and lettuce respond to extended daylight hours by accelerating flowering, whereas short-day crops such as onion or tomato require shorter days to initiate reproductive development.
Several traits essential for plant growth, yield performance, and adaptations such as leaf expansion, stem elongation, and metabolite accumulation, are tightly influenced by photoperiodic signals. Because these signals play a fundamental role in aligning plant development with environmental rhythms, understanding photoperiod response mechanisms has become a major focus of research in vegetable breeding. Advances in photoperiod manipulation have also revolutionized modern breeding approaches. In particular, controlled photoperiod regimes are now integral to 'speed breeding' protocols, enabling multiple generations of vegetable crops to be grown within a single year. Figure 7 illustrates how adjusting the light duration and intensity can alter developmental timelines, accelerating selection cycles and facilitating the rapid development of improved vegetable varieties adapted to diverse climatic conditions.
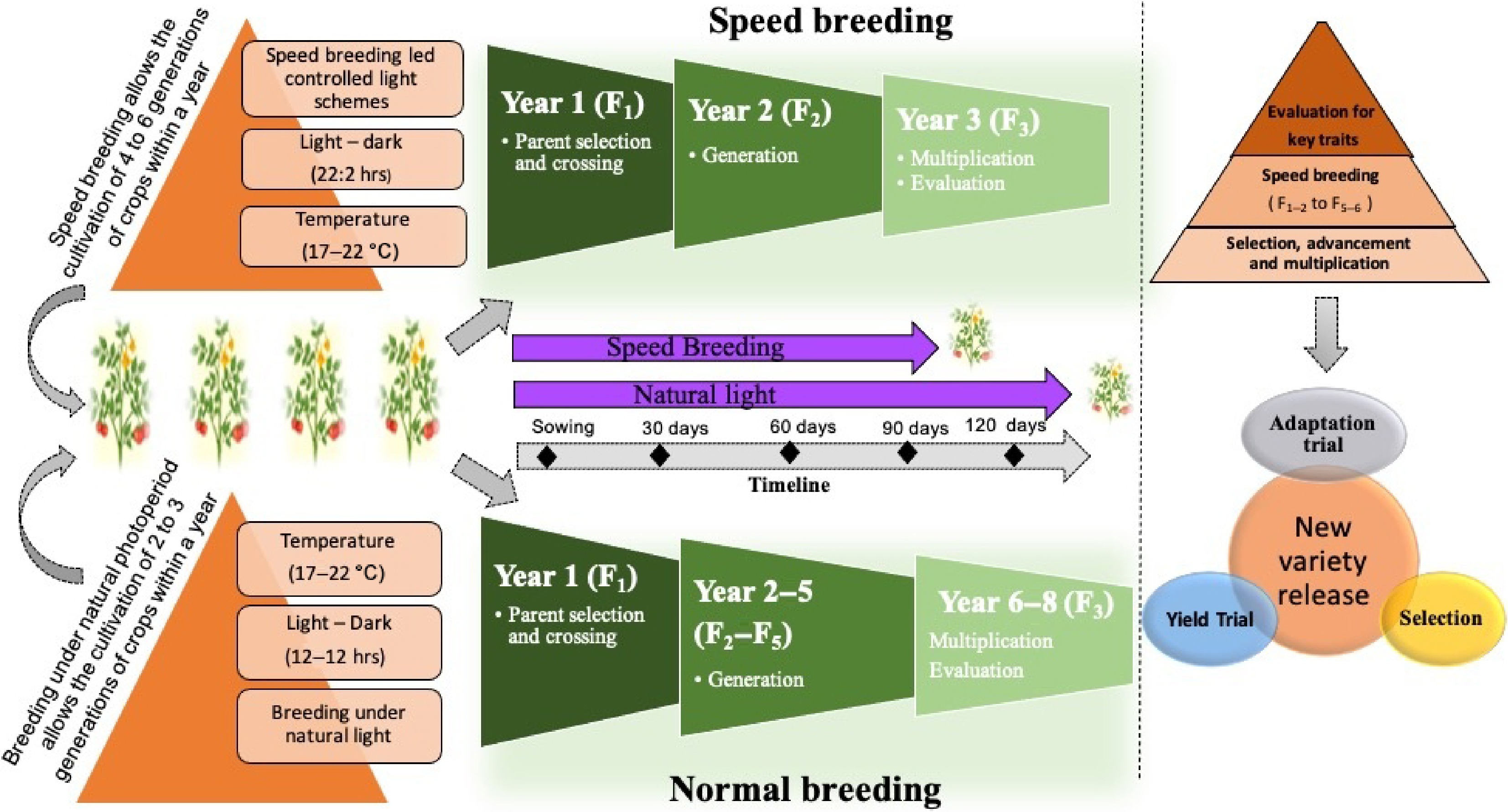
Figure 7.
Impact of photoperiod on standard and speed breeding methods in plant breeding, depicting the differences in photoperiod effects between conventional breeding and speed breeding methods.
The quality of light, including both the light plants receive at any moment, and the total light received during the day, directly affects plant growth, water loss through transpiration, stomatal opening, internal carbon dioxide levels, and overall photosynthesis rate[35,39]. Therefore, these studies underline the importance of photoperiod in regulating the flowering time. For potential applications in optimizing vegetable crop production and breeding strategies, alongside photoperiod, changes in soil moisture and temperatures also have the potential to impact plant germination and growth patterns.
Managing temperature regime
-
Photoperiod interacts with temperature to regulate flowering. In various vegetable crops, e.g., tomato and okra, however, high temperatures over 33 °C can have detrimental effects on pollen viability and male fertility, as such temperatures hinder proper pollen grain development[50]. Additionally, vernalization and dormancy, two plant traits once considered essential for hop flower proliferation, have been reported not to affect hop flower yield or quality[51]. Therefore, only temperatures within the optimal critical range promote the onset of flowering.
Altering soil moisture levels
-
Soil moisture levels can be altered to induce changes in plant height, maturation, flowering time, seed production, and other aspects of plant growth and development. Both drought and flooding are advantageous in speed breeding as they can induce early maturation and flowering in plants like tomato and lettuce. By combining the adjustments in irrigation regimes with temperature and humidity regulation, researchers can enable the annual production of multiple generations[52]. Conversely, drought stress in sorghum and beans has been reported to prolong flowering by up to nine days as opposed to well-watered conditions, underlining the significance of optimization of water management in speed breeding facilities[53,54]. Post-flowering, accelerating grain filling and maturation involves gradually decreasing soil moisture levels. In speed breeding, this technique typically entails reducing watering frequency from daily to twice per week, four to six weeks post-flowering, and then ceasing irrigation entirely in the final week before harvest, benefiting vegetable crops such as tomato and pea (Pisum sativum).
Utilization of plant nutrients and growth regulators
-
Plant nutrition and hormones are known to accelerate plant growth, flowering, seed development, and the germination of immature seeds[51]. For instance, the combined application of auxin and cytokinin in faba bean cultures induced 100% flowering and achieved a 90% seed set[54]. Such responses to plant growth regulators (PGRs) can vary, but controlled environments like greenhouses and growth chambers allow precise regulation of photoperiods and temperatures to optimize these effects.
Regulating CO2 levels
-
Elevated CO2 levels can enhance plant growth and accelerate the transition to reproductive stages, including early flowering, although responses vary among species and genotypes[39,52]. Speed breeding that manipulates CO2 concentrations requires suitable infrastructure, such as CO2 cylinders, regulators, and growth chambers, and entails associated operational costs. Safety precautions are also essential when handling CO2 cylinders and operating valves. Nagatoshi et al. reported that immature seeds from soybean plants grown under CO2 supplementation exhibited high germination rates comparable to those under control conditions[23].
It can be effectively extended to vegetable crops by incorporating CO2 regulation along with precise control of other environmental factors such as light and temperature. Fine-tuning CO2 levels within controlled environments not only enhances photosynthetic efficiency and biomass accumulation, but it also promotes timely flowering and seed development. When combined with optimized lighting and temperature regimes, this approach enables faster generational turnover and supports the rapid creation of high-yielding, stress-tolerant vegetable varieties. Thus, integrating CO2 enrichment into speed breeding systems offers a species-responsive strategy for accelerating genetic improvement and increasing productivity in vegetable crops.
Strategies for speed breeding implementation
-
Speed breeding in vegetable crops relies on optimizing light regimes, temperature, and controlled environments to hasten growth and reduce generation time. For example, extended photoperiods and regulated temperatures in growth chambers or greenhouses can enable multiple generations per year in crops such as tomato, lettuce, and pepper, far beyond what is possible under conventional field conditions. Some of the speed breeding strategies are discussed below.
Controlled environment agriculture (CEA)
-
The implementation of vertical farms, plant factories, and other controlled environment agriculture (CEA) technologies can bring food production closer to urban areas, addressing the demands of dense populations while supporting global climate goals. These systems utilize greenhouses, growth chambers, or other regulated environments to precisely control factors such as photoperiod, humidity, CO2 levels, and light intensity, enabling consistent year-round cultivation and rapid vegetable crop cycle turnover.
Hydroponics and aeroponics systems
-
Soilless cultivation systems, wherein roots are exposed to nutrient-rich mist or water, can be utilized for growing plants for efficient nutrient absorption and faster growth as opposed to traditional methods using soil. Various substrates like gravel, sand, or water can be used to immerse the roots completely or supplied with nutrients via the nutrient film technique, which utilizes drip irrigation. Hydroponic systems are known for their efficiency and are widely implemented by small farmers and commercial enterprises for growing vegetable crops such as cucumbers and tomatoes. These vegetables can be grown quite efficiently using the deep-water culture method, wherein the roots of the plants are immersed in nutrient-rich water, and air is supplied to the roots via an air stone. To ensure the successful cultivation of vegetable crops in hydroponic systems, pH, electrical conductivity, and temperature must be maintained in the ranges of 5.8–6.4, 1.2–3.5 Mho, and 65−78 °F, respectively[55]. Furthermore, aeroponics is a method wherein plants are suspended in air with their roots exposed. Water is supplied in the systems through a misting system. Fogging systems disperse tiny water droplets suspended longer, enabling increased evaporation and a drier plant canopy.
Enhanced lighting systems
-
Artificial light sources such as light-emitting diodes (LEDs) and high-pressure sodium (HPS) lamps can provide the optimal light spectrum desired for photosynthesis and plant growth. Photoperiods can be extended by using continuous lighting to trigger accelerated growth, thereby enabling the cultivation of multiple vegetable crop cycles within a reduced timeframe. A speed breeding approach allowing the customization of light quality has been reported to be efficient for short-day vegetable crops like amaranth, soybean, and okra. This protocol assists the development of several generations annually by utilizing LED light regimes specific to each crop, thereby removing the need for embryo rescue and other tissue culture techniques. Additionally, this approach makes it feasible to incorporate a multi-tiered 96-cell tray system, integrating speed breeding with genomics for enhanced breeding efficiency[36].
Speed breeding capsules
-
Speed breeding capsules, constructed from repurposed refrigerated shipping containers, equipped with temperature and light controls and irrigation systems, offer a cost-effective solution for accelerated plant growth. These containers, insulated, and featuring slotted floors with drainage ports, eliminate the need for greenhouse benches, allowing tall plants to be placed directly on the floor. Adjustable LED lights enhance flexibility, accommodating plants of varying heights, while regulating light intensity at the canopy level. In warm climates, heat from the lights can be vented out directly, whereas in cooler temperatures, it can be retained for heating. Solar power systems ideally supply most electricity, making these capsules affordable and adaptable for use worldwide in local breeding programs[34].
However, the speed breeding capsule concept and its integration into smallholder and resource-limited farming systems remains challenging. The technique relies on controlled environments, a stable power supply, and advanced infrastructure. These conditions are often lacking in developing or rural areas. Many smallholder farmers operate under minimal technological support, with limited access to continuous electricity supply or climate-controlled facilities. Consequently, the high initial costs and maintenance requirements of such setups can act as major barriers to adoption. To make speed breeding genuinely impactful at the grassroots level, more attention needs to be directed toward developing simplified and low-cost versions of these systems. Innovations such as solar-powered lighting, modular growth chambers, and locally fabricated growth units could make the technology more accessible. Additionally, establishing community-based speed breeding hubs or integrating such facilities with regional research centres may help bridge the gap between cutting-edge breeding techniques and smallholder farming realities. Focusing on adaptive, resource-efficient designs will ensure that the benefits of rapid crop improvement extend beyond well-funded institutions to reach farmers who need them most. Recent studies suggest that simplified growth protocols and low-input lighting solutions may help adapt speed breeding approaches for use in developing regions[52].
Genomic approaches
-
Advanced molecular tools such as genomic selection, gene editing, marker-assisted selection, etc., can be incorporated into speed breeding approaches to enable researchers to identify the desired traits efficiently, thereby expediting the development of enhanced vegetable crop varieties. Moreover, in-vitro germination of immature embryo cultures of vegetables such as pepper, potato, and tomato can facilitate breeders to obtain additional generations as compared to traditional approaches[16,56]. Therefore, integrating these techniques with speed breeding offers a promising approach to address the urgent need for developing climate-resistant and high-yielding vegetable crop varieties within a reduced timeframe.
Speed breeding methods have proven to be a valuable route to speed up traditional breeding programs. By developing vegetable crops with higher yields that can withstand changes and utilize water more efficiently, speed breeding can help provide people with healthier and more nutritious foods, while also reducing the environmental impact of agriculture. Speed breeding complements molecular techniques, reducing breeding cycles from 10–12 years to 5–6 years for key vegetables.
-
Integrating diverse breeding approaches offers a powerful strategy to speed up vegetable crop improvement. By combining conventional, molecular, and genomic tools, breeders can harness the strengths of each method to achieve higher precision and sustainability in plant development.
Conventional and molecular breeding approaches: integration and acceleration through speed breeding
-
Conventional breeding in vegetable crops relies mainly on phenotypic selection, where traits such as yield, disease resistance, fruit size, and colour are evaluated over several growing seasons. While effective, this approach is slow, as long generation cycles and environmental variability often delay progress. In contrast, molecular breeding uses genotype-based tools, including marker-assisted and genomic selection, to identify and transfer desirable alleles with higher precision and speed. Integrating speed breeding with these methods has transformed vegetable crop improvement. Though not a separate breeding approach, speed breeding acts as a generation acceleration tool, enabling the rapid advancement of breeding populations under controlled environmental conditions. By optimizing light, temperature, and photoperiod, facilities can generate up to four to six crop cycles per year in tomato, pepper, lettuce, and cabbage, compared to one or two under field conditions. This accelerated framework supports faster gene pyramiding, quicker trait introgression, and earlier validation of molecular markers. Coupled with genomic tools, it enhances selection for complex traits such as nutrient efficiency, stress resilience, and fruit quality. Tables 5 and 6, together with Fig. 8, illustrate that the amalgamation of traditional, molecular, and rapid breeding techniques markedly enhances the efficacy and accuracy of vegetable crop enhancement.
Table 5. Comparative overview of conventional and molecular breeding approaches, and the complementary role of speed breeding as a generation advancement tool.
S.No. Conventional breeding Molecular breeding Speed breeding 1. Uses conventional techniques for plant crossbreeding to merge desired characteristics from two parent plants. Breeding is focused on phenotype, making it difficult to determine a pure line of genes. Breeding using a genotype-based approach is more accurate in identifying a pure gene line. It brings novel characteristics from allied species or cultivars. Uses methods for controlled environments to speed up the breeding cycle. Entails adjusting growth factors (temperature, light, etc.,) to encourage quicker plant development and more generations in a shorter time. 2. Dominant genes are typically the only ones chosen. It depends on recombination and natural genetic variation. Recessive alleles can be chosen using markers and the identification of certain gene locations, which is a quicker process than the traditional method.
Identifies plants with desired characteristics using molecular markers/DNA sequences.Speed breeding is a viable and effective solution for vegetable crops with huge populations. It can be used on a variety of vegetable crops and requires less work than conventional breeding techniques. 3. Less technological expertise and genetic understanding is required. It may be done using local methods and equipment, thus being less expensive. Molecular plant breeding is more reliable and effective. Due to the need for sophisticated equipment and techniques, it is costly. Needs specific equipment and facilities. Requires a large upfront setup cost for facilities in controlled environments. 4. Usually, takes 10–15 years of generation time to develop a new variety. A new variety can be released in a relatively shorter amount of time. By eliminating some of the trial-and-error procedures, breeding time can be significantly reduced. Significantly reduces the time needed to develop new varieties from years to months. 5. This technique is less accurate as unwanted characteristics could be added. Frequently used in a variety of vegetable crops. More accurate than traditional breeding, focusing on particular features or genes. Utilized for vegetable crops that have particular qualities that are desired, like increased nutritional value, drought tolerance, or disease resistance. With four to six generations every year, it has been successfully used on a variety of vegetable crops and is helpful in research settings seeking quick genetic improvement, or for vegetable crops with lengthy breeding cycles. Table 6. List of conventional, molecular, and integrated speed breeding approaches in vegetable crops.
Approach Vegetable crop Advantage Limitation Conventional Tomato, potato, chilli, brinjal, broccoli, bottle gourd, etc. Established resistance traits Slow, labor-intensive Molecular Tomato, cucumber, lettuce, etc. Precise trait introgression Costly, infrastructure-heavy Speed Lettuce, tomato, pepper, cabbage, Chinese kale Reduced cycle time Controlled environment required Integrated Tomato, pepper, soyabean, pea Rapid and precise breeding Requires expertise and funding 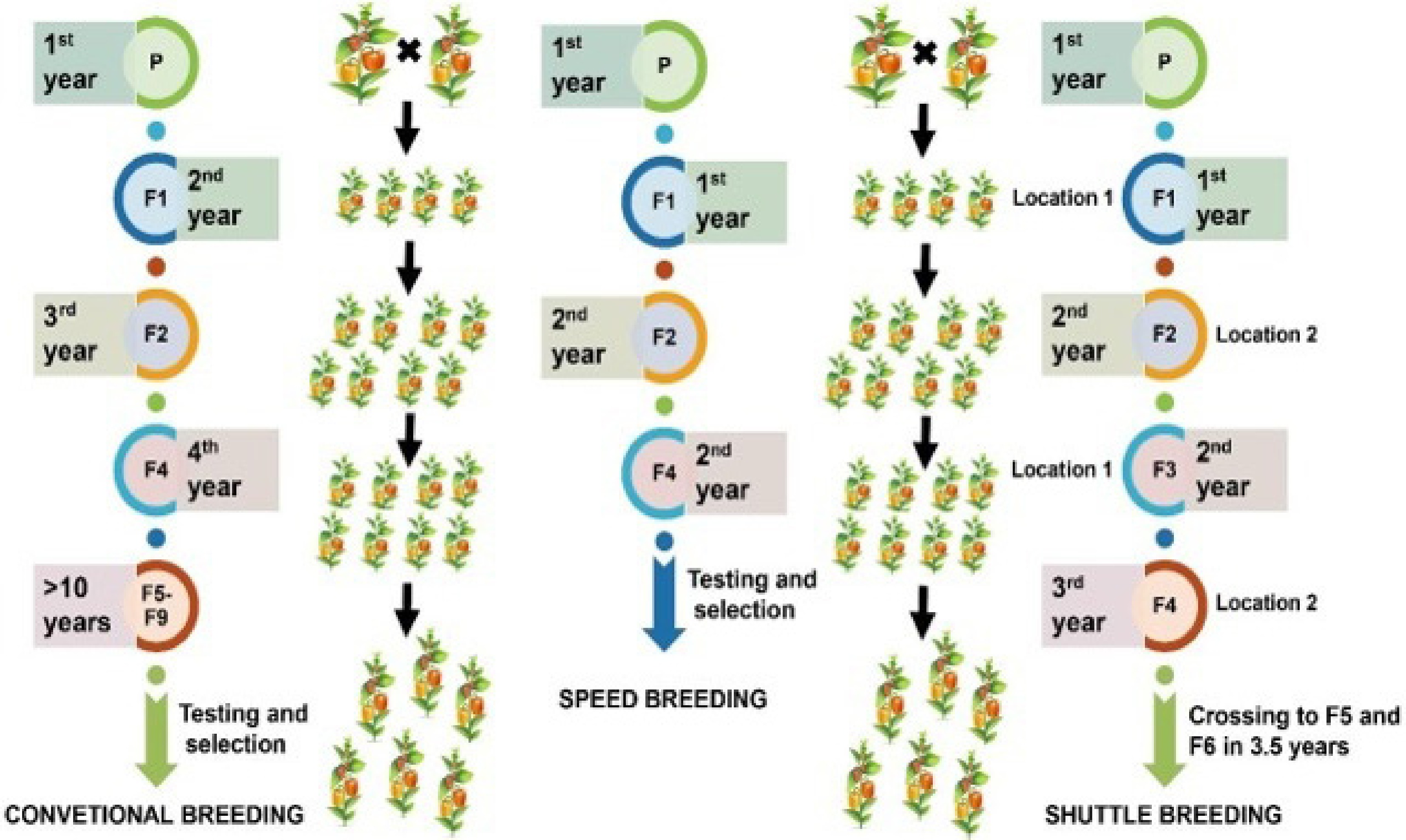
Figure 8.
Comparative representation of conventional, speed, and shuttle breeding techniques in vegetable crop improvement, showcasing the efficiency and timeframes.
Combining approaches such as molecular breeding, speed breeding, and conventional practices can speed up the development of resilient vegetable varieties with enhanced stress tolerance, disease resistance, and yield potential. Integrated breeding strategies offer significant opportunities to increase efficiency, precision, and innovation in vegetable crop improvement[21]. Some of the examples of integrated methods have been discussed below.
Genomic data integration in conventional breeding
-
Conventional breeding based on phenotype and pedigree often requires over a decade to deliver improved varieties without pre-breeding programs. Strategies like rigorous parent evaluation, targeted crosses, shuttle breeding, and elite germplasm testing across diverse locations, supported by effective databases, have helped shorten cycles. The advent of DNA sequencing provided breeders with extensive genomic information, while DNA marker-based genotyping enabled selection based on genetic composition rather than phenotype, enhancing efficiency[57]. Genomic-assisted breeding has evolved over the past four decades, beginning with QTL mapping that linked DNA markers with traits for marker-assisted selection[21]. Early methods, however, suffered from limited allelic variation, low resolution, and dependence on segregating populations[30]. Recent advances now deliver high-resolution genomic data, accelerating breeding efficiency and precision. By integrating conventional and modern genomic tools, plant breeding has become more targeted, reducing breeding cycles, and ensuring the development of superior cultivars adapted to agricultural and market demands.
Speed breeding and precision breeding with molecular tools
-
Speed and precision breeding are innovative approaches that integrate molecular tools to speed up the development of vegetable crop varieties with desirable traits. While molecular breeding introduces new traits into diverse genetic pools, speed breeding enables their rapid development and testing, enhancing genetic diversity and adaptability in vegetable crops. Numerous studies have demonstrated the effectiveness of combining speed breeding with genome editing. For instance, in tomato, CRISPR/Cas9-based precision breeding improved disease resistance and fruit quality while reducing ethylene biosynthesis, significantly extending shelf life[58], with speed breeding shortening the generation time for faster cultivar release. In potatoes, precision breeding targets late blight resistance[59], while rapid-cycle breeding speeds up evaluation. Similarly, in cucumber, speed breeding has facilitated the development of lines with enhanced fruit quality and stress tolerance[34]. These advancements underscore the potential of integrating rapid breeding methods with cutting-edge genomic technologies to improve vegetable crop resilience and quality.
-
With an escalating demand for new varieties of vegetable crops and continuous evolution in science and technologies, breeding programs are also advancing. Currently, breeding is focused on vegetable crops that thrive in controlled environments and have shorter growth cycles to maximize space and resources. Some of the emerging technologies and trends are discussed below.
Emerging technologies and trends
CRISPR-Cas9
-
CRISPR/Cas9 has transformed vegetable breeding by enabling precise, targeted genome modifications that enhance resilience to abiotic and biotic stresses. It can add, remove, or deactivate genes to help plants withstand heat, salinity, and disease. For example, cucumber exhibits improved salt tolerance through editing of ion transport genes[38]. Further, CRISPR has been employed to produce seedless tomatoes[60], delay flowering in broccoli (BoFLC2 gene)[22,61], improve heat tolerance in lettuce[41], modify garlic genes for better storage and shelf life, enhance pepper fruit quality traits, develop disease resistance, and create GABA-enriched tomatoes[62]. When combined with speed breeding, CRISPR enables the production of stable plants in approximately one year, far faster than traditional breeding methods[63]. Genome editing tools such as CRISPR are being used to precisely modify the DNA of vegetable crops, enabling the development of improved varieties with desirable traits. These innovations include disease-resistant cucumbers that can withstand mildew, tomatoes enriched with nutrients like lycopene or vitamin D, and slow-ripening tomatoes with a longer shelf life. Researchers have also created salt-tolerant pumpkins to endure stress conditions and non-browning potatoes or carrots with altered colours for enhanced quality and appeal. Unlike traditional breeding, this approach allows scientists to introduce beneficial traits accurately. As a result, genome-edited crops are emerging as promising tools for sustainable agriculture and food security, with several varieties already approaching or entering the commercialization stage. Overall, integrating CRISPR/Cas9 with speed breeding provides a powerful platform for developing climate-resilient, high-yielding, and nutritionally improved vegetable cultivars[22,64]. Table 7 outlines how CRISPR/Cas9 is being used in vegetable crop improvement. Recent evidence shows that its benefits are not confined to growth chambers, as several edited vegetable lines perform consistently under more realistic cultivation conditions. In tomato, for example, CRISPR/Cas9 has been used to modify genes that regulate fruit texture, resulting in lines with firmer fruits, slower softening after harvest, and better shelf life, while largely preserving yield and eating quality. In lettuce, targeted editing of pathways involved in vitamin and antioxidant biosynthesis has produced plants with markedly higher levels of β-carotene, zeaxanthin, and ascorbic acid, yet with normal growth and morphology, demonstrating that nutritional enhancement can be achieved without obvious penalties to plant performance.
Table 7. Applications and challenges of CRISPR/Cas9 in vegetable crop improvement.
Vegetable crop Target trait/gene Reported outcome/success Approx. success rate Limitations/knowledge gaps Ref. Tomato Seedlessness (SlAGAMOUS-LIKE) Stable seedless fruit 60%–70% Moderate editing efficiency; few multi-trait studies; limited field trials [65] Tomato Ripening (SlEXP1) Delayed ripening, extended shelf life 65% Mostly greenhouse-tested; off-target effects possible [62] Tomato GABA enrichment Nutritionally enhanced fruit 50%–60% Lab-scale validation; field performance not fully evaluated [65] Cucumber Salt tolerance (ion transport genes) Improved salinity tolerance 55%–60% Limited field validation; genotype-dependent efficiency [66] Broccoli Flowering time (BoFLC2) Delayed flowering observed 50%–55% Transformation efficiency varies; limited multi-trait editing [61] Lettuce Heat tolerance Increased tolerance to heat stress 50%–55% Greenhouse data only; off-target mutations need assessment [67] Garlic Storage and shelf life Improved post-harvest traits 30%–40% Polyploid genome; low transformation efficiency [68] Pepper Fruit quality and disease resistance Enhanced fruit traits, disease resistance 50%–55% Mostly greenhouse trials; multi-trait editing is rarely explored [69] Spinach Downy mildew resistance Resistant lines developed 45%–50% Limited field data; editing efficiency varies by cultivar [10] Eggplant Anthocyanin content Increased antioxidant levels 50% Few studies; field validation lacking [32] Carrot Root shape and carotenoid content Improved root quality and nutritional traits 40%–50% Transformation efficiency is low; few multi-trait edits [70] Cauliflower Flowering time and curd development Delayed flowering and uniform curd size 45%–50% Genotype-dependent; field-level validation limited [71] Potato Late blight resistance Enhanced disease tolerance 40%–50% Tetraploid genome complicates editing; off-targets possible [9,16] Rapeseed Abiotic and biotic stress tolerance Positive results in field trials 60% Protocol optimization is needed for consistent efficiency [4] High-throughput phenotyping
-
High-throughput technologies such as proximal and remote sensing have revolutionized vegetable breeding by enabling rapid, non-invasive, and data-driven phenotyping. Automation and advanced analytics in these platforms streamline the screening of germplasm and breeding populations, reducing labour while increasing precision[58]. UAV-based remote sensing has proven particularly effective in detecting drought and heat stress across diverse genetic backgrounds. In vegetable crops, high-throughput phenotyping in tomato and pepper allows rapid evaluation of drought and heat tolerance, enabling targeted selection of superior lines. Yet, studies by Sanaeifar et al. and Jin et al. reveal persistent gaps between phenotyping and data integration[19,72]. Coupling these systems with omics technologies, genomics, transcriptomics, proteomics, and metabolomics can establish comprehensive frameworks for trait evaluation, driving the development of climate-resilient and high-performing vegetable cultivars[7].
Haplotype breeding
-
Haplotype breeding is a promising genomics-assisted approach for developing specialized vegetable crop varieties. This technique uses resequencing to identify superior haplotypes, specific combinations of alleles at closely linked chromosomal sites that can be incorporated into breeding programs. By targeting these beneficial genetic patterns, breeders can enhance traits such as yield, disease resistance, and stress tolerance[73]. Originally applied in human genetics to reduce the number of markers required in genomic regions via haplotype-tagged SNPs[27], the approach has been extended to vegetable crops. Genome-wide association studies (GWAS) are a key tool for uncovering genetic variation in diverse populations[74], allowing breeders to link specific genetic markers with desirable traits and select optimal haplotypes. Some challenges, such as chromosome doubling and haploid production defects can occur with double-haploid techniques in vegetable crops like tomato[15] and legumes[75]. Haplotype breeding has been successfully applied to identify allele combinations for yield and stress resilience in vegetables such as tomato and lettuce.
Ethical and regulatory considerations in vegetable crop improvement
-
Ethical and regulatory safeguards are fundamental in the improvement of vegetable crops, helping to ensure that new technologies are safe, fair, and sustainable for the environment. Before release, candidate varieties are expected to undergo thorough assessment for potential allergenicity, toxic effects, and unintended changes in nutritional profile. Further, the gene editing tools can support traits such as stronger disease resistance, lower pesticide requirements, better drought tolerance, along with increased productivity offering clear agronomic and environmental advantages when carefully implemented, while maintaining sustainability aspects. When crops are generated without introducing foreign DNA, the resulting plants may resemble those produced by conventional mutagenesis, and some regulators treat them under lighter biosafety regimes than transgenic GMOs. Further, the regulatory systems differ markedly across regions, with the European Union tending to apply stricter GMO rules to many gene-edited crops, while several other jurisdictions have adopted more flexible, trait-based, or case-by-case approaches. Alignment with international reference frameworks, including standards developed under the Codex Alimentarius, and plant variety protection systems, such as UPOV can promote cross-border consistency in safety evaluation and variety protection. However, consumer transparency through clear labelling, where required, is important to support informed choice and maintain trust, particularly for foods eaten fresh[36]. At the same time, intellectual property arrangements for CRISPR tools and edited varieties need to balance innovation with mechanisms that preserve access for public breeding programs, humanitarian uses, and smallholder-oriented seed systems.
Vegetables are dietary staples, yet often remain relatively expensive, highly perishable, and susceptible to pests, the methods/plant varieties that extend shelf life, improve micronutrient content, or reduce losses could improve access to healthy food for low-income populations if embedded in inclusive value chains. Ethically, structuring seed pricing, licensing, and distribution so that benefits do not accrue only to affluent markets or large-scale producers, but also reach smallholders and marginalized consumers. Because vegetables are widely consumed raw or minimally processed, early engagement with farmers, retailers, nutritional professionals, and consumer groups, alongside clear and non-stigmatizing communication about how traits are developed, is essential for maintaining trust. In the longer term, greater convergence or mutual recognition of regulatory approaches, combined with participatory governance and fair benefit-sharing, can enable gene-edited vegetable crops to contribute meaningfully to nutrition security while respecting consumer autonomy and protecting vulnerable farming communities.[76]. Furthermore, greater alignment of regulatory frameworks across countries may support global deployment and trade, while transparent communication and stakeholder involvement are key to improving public confidence in gene-editing technologies.
Sustainability and climate resilience in breeding programs
-
Sustainability and climate resilience are becoming increasingly important in breeding programs. Incorporating these factors ensures vegetable crops are better equipped to cope with environmental changes and resource constraints. Breeding for drought tolerance reduces water requirements, while varieties needing fewer fertilizers minimize environmental impacts and input costs. Improved root growth can enhance soil organic matter, supporting long-term fertility, and natural resistance to pests and diseases reduces reliance on chemical controls. Vegetable crops with inherent herbicide tolerance enable more effective weed management with lower chemical use. Developing varieties with adaptable growth cycles helps plants withstand changing seasonal patterns, and selecting traits that enhance soil carbon sequestration can contribute to climate change mitigation.
-
The convergence of conventional, molecular, and speed breeding approaches has transformed modern vegetable improvement programs. While traditional breeding remains vital for maintaining genetic diversity and trait stability, molecular tools such as marker-assisted selection and genome editing provide precision and efficiency in targeting specific genes for yield, quality, and stress tolerance. The integration of speed breeding enables faster generation turnover, significantly reducing the time required for cultivar development. Together, these complementary strategies speed-up the creation of high-yielding, climate-resilient, and resource-efficient vegetable varieties. The incorporation of artificial intelligence and machine learning into genomic selection and phenotyping further enhances decision-making, allowing rapid identification of superior genotypes. This holistic, technology-driven breeding framework not only supports food and nutritional security, but also promotes sustainable, adaptive, and environmentally responsible horticultural systems.
-
The authors confirm their contributions to the paper as follows: writing − original draft preparation, visualization: Singh J; visualisation, writing − review and editing: Apoorva; writing − review and editing: Singh S; visualisation: Sharma L; writing − review and editing: Rai SK; conceptualization, writing − review and editing, resources, and supervision: Pandey-Rai S. All authors reviewed the results and approved the final version of the manuscript.
-
Data sharing does not apply to this article, as no datasets were generated or analysed during the current study
-
Jagriti Singh acknowledges the financial support provided through the Raja Jwala Prasad Postdoctoral Fellowship. The authors also express their sincere gratitude to IOE-BHU, ISLS-BHU, and CAS -Department of Botany, BHU.
-
The authors declare that they have no conflict of interest.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Singh J, Apoorva, Singh S, Sharma L, Rai SK, et al. 2026. Bridging traditional breeding and molecular approaches: routes to sustainable vegetable crop advancement. Vegetable Research 6: e010 doi: 10.48130/vegres-0026-0004
Bridging traditional breeding and molecular approaches: routes to sustainable vegetable crop advancement
- Received: 29 August 2025
- Revised: 02 January 2026
- Accepted: 28 January 2026
- Published online: 26 March 2026
Abstract: Ensuring global food security increasingly relies on the rapid advancement of nutrient-rich and climate-adaptive vegetable varieties. Conventional plant breeding, though foundational, is limited by slow selection processes and the gradual narrowing of genetic diversity. The advent of genomic-assisted speed breeding offers an innovative avenue to address these challenges by integrating molecular approaches with traditional breeding frameworks. This review highlights progressive techniques, including haplotype-based selection, high-throughput phenotyping, marker-assisted selection (MAS), and quantitative trait loci (QTL) mapping, all aimed at enhancing yield potential, nutritional value, and tolerance to environmental and biological stressors. Moreover, genome-editing platforms such as CRISPR/Cas9 facilitate precise modification of target genes, expediting the development of improved cultivars. This paper also underscores the interplay between classical breeding practices and modern genomic tools, while considering ethical and regulatory dimensions. Collectively, this synergistic approach promotes faster genetic improvement, sustainable vegetable crop productivity, and resilience within horticultural systems, thereby contributing to long-term food and climate security.
-
Key words:
- Abiotic stress /
- Traditional breeding /
- Speed breeding /
- Molecular Marker /
- CRISPR/Cas9.












