-
Rapeseed oil currently occupies a pivotal position in the global edible oil market, widely valued by consumers and manufacturers alike for its distinctive sensory profile and favorable nutritional properties[1,2]. However, crude rapeseed oil is a chemically complex matrix containing free fatty acids, phospholipids, pigments, and trace metal ions, which could compromise its sensory quality and oxidative stability[3]. Consequently, the crude oil must undergo a standardized, multi-stage refining process to ensure consumption safety and meet requisite industrial versatility standards. This refining sequence is essential for optimizing key physicochemical attributes, most notably by elevating the smoke point and ensuring the superior oxidative stability of the final product[4]. Currently, refined rapeseed oil has become indispensable across a wide spectrum of applications, ranging from domestic cooking to large-scale industrial food manufacturing[5].
Traditionally, the chemical refining sequence of rapeseed oil includes hydration degumming, alkali refining, adsorption decolorization, and high-temperature deodorization[6]. Among these unit operations, alkali refining represents a critical intervention point. In this step, free fatty acids are neutralized by aqueous alkaline solutions to form soapstock, a process central to reducing acidity and improving stability[7]. While the primary function of neutralization is the efficient removal of free fatty acids and residual phospholipids, exposure to high pH and elevated temperatures may trigger complex, non-enzymatic secondary reactions[8]. Although standard refining protocols generally yield high-quality neutral oils, sensory deviations manifested as soapy or metallic off-flavors have been frequently observed in substandard batches. Distinct from the well-characterized oxidative rancidity associated with long-term storage, these process-induced off-flavors occur immediately following neutralization[9]. This immediate degradation significantly diminishes consumer acceptance and necessitates aggressive downstream deodorization; conversely, such severe processing can strip the oil of its natural tocopherols and desirable aroma compounds[10]. Although the flavor profile of cold-pressed rapeseed oil has been comprehensively characterized, the molecular mechanisms driving the formation of soapy and metallic off-flavors during alkali refining remain poorly understood.
For the accurate elucidation of off-flavor compounds in complex vegetable oil matrices, modern flavor chemistry has established a systematic research paradigm centered on molecular sensory science[11,12]. In this approach, gas chromatography-olfactometry (GC-O) is first employed to localize key regions within the aroma-active fraction, thereby focusing on odorants that are directly relevant to human perception and avoiding misinterpretation based solely on chemical abundance[13]. Subsequently, AEDA is applied to rank odor potency, followed by quantitative determination of selected compounds and calculation of OAV, which translate analytical concentrations into indices of sensory relevance[14]. Finally, aroma recombination and omission experiments are conducted to verify the causal contribution of candidate key odorants, ensuring that the proposed molecular set is capable of reproducing the characteristic odor of the original sample[15]. Using this analytical strategy, we have previously investigated the volatile off-flavor formation in small mill sesame oil and peanut oil, successfully identifying the key off-flavors responsible for their characteristic sensory defects[16,17].
Thus, this study aims to elucidate the impact of the alkali refining process on the flavor quality of rapeseed oil and its molecular mechanisms. Specific objectives include: (1) using QDA to characterize the evolution of sensory attributes in rapeseed oil from crude to alkali-refined stages; (2) applying GC-O combined with AEDA to screen high-potency odor compounds, and identifying key odorants responsible for soapy and metallic flavors through gas chromatography-mass spectrometer (GC-MS) analysis and OAV calculations; (3) verifying the contributions of key odorants to overall off-flavor characteristics via flavor recombination and omission tests. This research seeks to provide a scientific explanation for alkali-induced off-flavors in rapeseed oil and offer theoretical guidance for targeted regulation and quality optimization in the refining process.
-
Rapeseeds (Brassica napus L., cv. Zhongyouza 501) were obtained from Hubei Province, China. Crude oil was extracted using a low-temperature screw press (CA59G, Komet, Germany) with the oil outlet temperature controlled below 50 °C. The thermally treated control oil was prepared by heating the crude oil in a reactor at 65 °C for 40 min under constant stirring (60 rpm) without water addition. For the preparation of the water-degummed oil, the crude oil was similarly heated to 65 °C and mixed with distilled water (2.5% w/w). The mixture was stirred rapidly (400 rpm) for 2 min to disperse water, followed by a slow agitation period (30 min, 60 rpm) to induce phospholipid hydration and flocculation. The aqueous gum phase was separated by centrifugation (5,000 × g, 20 min, 20 °C). Then, the alkali-refined oil was produced by further processing the water-degummed oil. The oil was heated to 85 °C for deacidification, treated with a sodium hydroxide solution (18° Bé) added dropwise over 10 min under stirring (350 rpm), and maintained for 25 min. After soapstock removal via centrifugation (4,500 × g, 15 min), the oil phase was washed twice with hot deionized water (90 °C, 10% w/w) and vacuum-dried at 50 °C for 30 min. All samples were flushed with high-purity nitrogen and stored in amber glass bottles at −20 °C prior to analysis.
Chemicals
-
All chemicals were of analytical grade, and solvents were of HPLC grade. Authentic flavor standards (purity ≥ 90%) used for identification and quantification, as well as the internal standard 2-methyl-3-heptanone, were obtained from Sigma-Aldrich (St. Louis, MO, USA) and are listed in Supplementary Table S1. Dichloromethane and anhydrous sodium sulfate were supplied by Merck (Darmstadt, Germany). Sodium hydroxide and citric acid used for the refining process were of food grade. Both the recovery rate study and the construction of the aroma recombination model used deodorized sunflower seed oil as a matrix.
Sensory evaluation
-
The sensory profile of the rapeseed oil samples was quantitatively characterized by a panel of 12 trained assessors (six males, six females; aged 20–35 years) recruited from China Agricultural University. Sensory assessors were selected and calibrated according to ISO 8586 protocols to ensure the accurate and consistent application of specific sensory descriptors and intensity scales. The study protocol was approved by the university's human research ethics committee (CAUHR-20240703). Based on preliminary trials and the standard lexicon for rapeseed oil, seven distinct sensory attributes were defined: pungent, cabbage-like, green, fatty, soapy, metallic, and rancid. Authentic chemical reference standards, presented in deodorized sunflower oil, served to anchor the panel's intensity ratings and minimize inter-assessor variation. The definitions and reference concentrations were established as follows (Supplementary Table S2). Sensory sessions were conducted in a climate-controlled sensory laboratory at 22 °C. Ten mL oil samples were presented monadically in coded, capped amber glass vials. Panelists were instructed to assess the intensity of each attribute on a 10-point linear scale (0 = not detected; 10 = very strong). Each sample was evaluated in triplicate by every assessor across three independent sessions.
Isolation, identification, and quantification of volatile flavor compounds
-
An accurately weighed sample of rapeseed oil (50.00 ± 0.01 g) was dispersed in 150 mL of HPLC-grade dichloromethane within a sealed amber flask. Then 200 μL of an internal standard solution containing 2-methyl-3-heptanone (0.816 mg/mL in dichloromethane) was added. The mixture was agitated on a thermostatic shaker at 180 rpm for 8 h at 4 °C. The solvent-assisted flavor evaporation (SAFE) unit was operated under a high vacuum of 10−5 mbar, generated by a molecular turbine vacuum pump. The flask containing the dichloromethane extract was immersed in a circulating water bath maintained at 40 °C. The distilled volatiles were efficiently trapped in a cryogenic collection flask cooled with liquid nitrogen. Upon completion of the distillation, the recovered distillate was dried over anhydrous sodium sulfate. The volume of the dried extract was then reduced to approximately 1 mL using a Vigreux column at 40 °C. A final concentration step to a precise volume of 100.0 µL was performed under a gentle stream of high-purity nitrogen. The resulting concentrated extract was immediately used for all subsequent analyses to minimize the loss of volatile components.
Instrumental analysis was conducted on an Agilent 7890B gas chromatograph interfaced with an Agilent 5977B mass selective detector and a Gerstel ODP 3 olfactory detection port. A 1.0 µL aliquot of the final extract was introduced into the GC system via a splitless injection, with the inlet temperature held at 250 °C. Chromatographic separation was performed on an HP-INNOWax fused-silica capillary column (30 m × 0.25 mm × 0.25 µm). The carrier gas was high-purity helium, maintained at a constant flow rate of 1.0 mL/min. The oven temperature program consisted of an initial hold at 40 °C for 5 min, a 4 °C/min ramp to 160 °C held for 5 min, followed by a 5 °C/min increase to 240 °C held for 5 min. The MSD was configured to operate in electron ionization mode at 70 eV, with the ion source and quadrupole temperatures set at 230 °C and 150 °C, respectively. A solvent delay of 5.0 min was employed. Mass spectra were acquired over an m/z range of 30–500. For olfactometry, the column effluent was split 1:1 (v/v) between the MSD and the ODP. The ODP transfer line was heated to 250 °C, and humidified air was supplied at 50 mL/min.
Flavor compound identification and aroma extract dilution analysis
-
The identification of volatile compounds was established using mass spectral data, retention index, and odor characteristics. Initially, the acquired mass spectra were screened against the NIST 2020 library, requiring a similarity match factor exceeding 80%. To substantiate these spectral matches, the retention indices were experimentally determined relative to a homologous series of n-alkanes on the HP-INNOWax column. These values were aligned with the retention indices of authentic standards analyzed under identical chromatographic conditions. Finally, conclusive identification was secured through specific olfactory confirmation at the olfactory detection port, ensuring that the sensory descriptors perceived by the trained assessors were consistent with the aromas of the corresponding reference standards.
The sensory potency of each odorant was assessed by AEDA. This required the preparation of a serial 1:2 (v/v) dilution series of the original extract using dichloromethane. Each dilution was then analyzed by the GC-O panel, which consisted of three trained assessors (two female, one male; aged 20–30 years). The flavor dilution (FD) factor was established as the maximum dilution at which a characteristic aroma was still discernible and correctly identified by a minimum of two of the three assessors.
Quantitative analysis and odor activity value calculation
-
The quantitative analysis of targeted volatile flavor compounds was conducted utilizing an external calibration method that incorporated an internal standard, 2-methyl-3-heptanone. A series of mixed calibration standard solutions was formulated. The preparation involved dissolving precisely weighed amounts of authentic analyte standards and a consistent mass of the internal standard in dichloromethane to achieve seven distinct concentration levels. Each calibration standard was analyzed in triplicate via GC-MS with a 1.0 µL injection volume. Calibration curves were subsequently established by plotting the peak area ratio of the analyte to the IS against the mass ratio of the analyte standard to the internal standard. The resulting data points were fitted using linear regression analysis to derive the calibration equation, represented as:
$Y=\alpha X + \beta $ In this equation, the variable Y denoted the analyte-to-IS peak area ratio, while X corresponded to the mass ratio of the analyte standard to the IS within the calibration solutions.
To evaluate the method's efficacy, a recovery study was performed. Blank deodorized oil samples were fortified with the target analytes at three separate concentrations, alongside a constant quantity of the internal standard, before being subjected to the entire SAFE extraction and GC-MS analysis procedure. The recovery rate was determined using the expression:
$ R=(A_1-A_0)/A \times 100{\text{%}} $ Here, A1 was the concentration of the analyte determined in the spiked blank oil sample after undergoing the complete SAFE analytical procedure, A0 represented the concentration of the analyte determined in the unspiked blank oil sample after undergoing the complete SAFE analytical procedure, and A was the theoretical concentration of the analyte that was added into the blank oil sample. Furthermore, the limits of detection and quantification for the method were established by analyzing the chromatograms of standard solutions, defining them at signal-to-noise ratios of 3:1 and 10:1, respectively.
To estimate the sensory contribution of each odor-active compound to the overall aroma, OAV were calculated. The OAV for each compound was determined by dividing its quantified concentration by its odor threshold. The odor thresholds were selected from peer-reviewed literature, prioritizing values that were determined in an oil matrix[18,19].
Aroma recombination and omission experiments
-
To validate the accuracy of the identification and quantification results, an aroma recombination model was prepared to simulate the flavor profile of the alkali-refined rapeseed oil[20]. A deodorized oil matrix was used as the base. Stock solutions of the identified odorants were prepared in odorless medium-chain triglyceride oil and added to the blank matrix at the exact concentrations determined in the alkali-refined samples. The mixture was stirred for 30 min and equilibrated at room temperature for 1 h to ensure homogeneity. The sensory profile of the recombination model was then evaluated by the trained panel using the same protocol and attributes described in the sensory evaluation section. The similarity between the recombination model and the original oil sample was assessed by comparing the mean intensity scores of the key attributes.
To elucidate the contribution of specific flavor classes or individual odorants to the soapy and metallic off-flavors, omission experiments were conducted. A series of omission models was prepared by excluding a specific group of compounds, such as nitriles, aldehydes, or ketones, from the complete recombination model described above. The models were presented to the panelists in a randomized order. Assessors were instructed to rate the intensities of the soapy and metallic attributes for each omission model using the 10-point scale. The significance of the sensory differences between the omission models and the full recombination model was determined by statistical analysis.
Statistical analysis
-
All experiments were conducted in triplicate, and data are presented as mean ± standard deviation. Statistical analysis was performed using SPSS software (v. 22.0, IBM, Armonk, NY, USA). Significant differences between means were determined by one-way analysis of variance (ANOVA) followed by Tukey's post-hoc test.
-
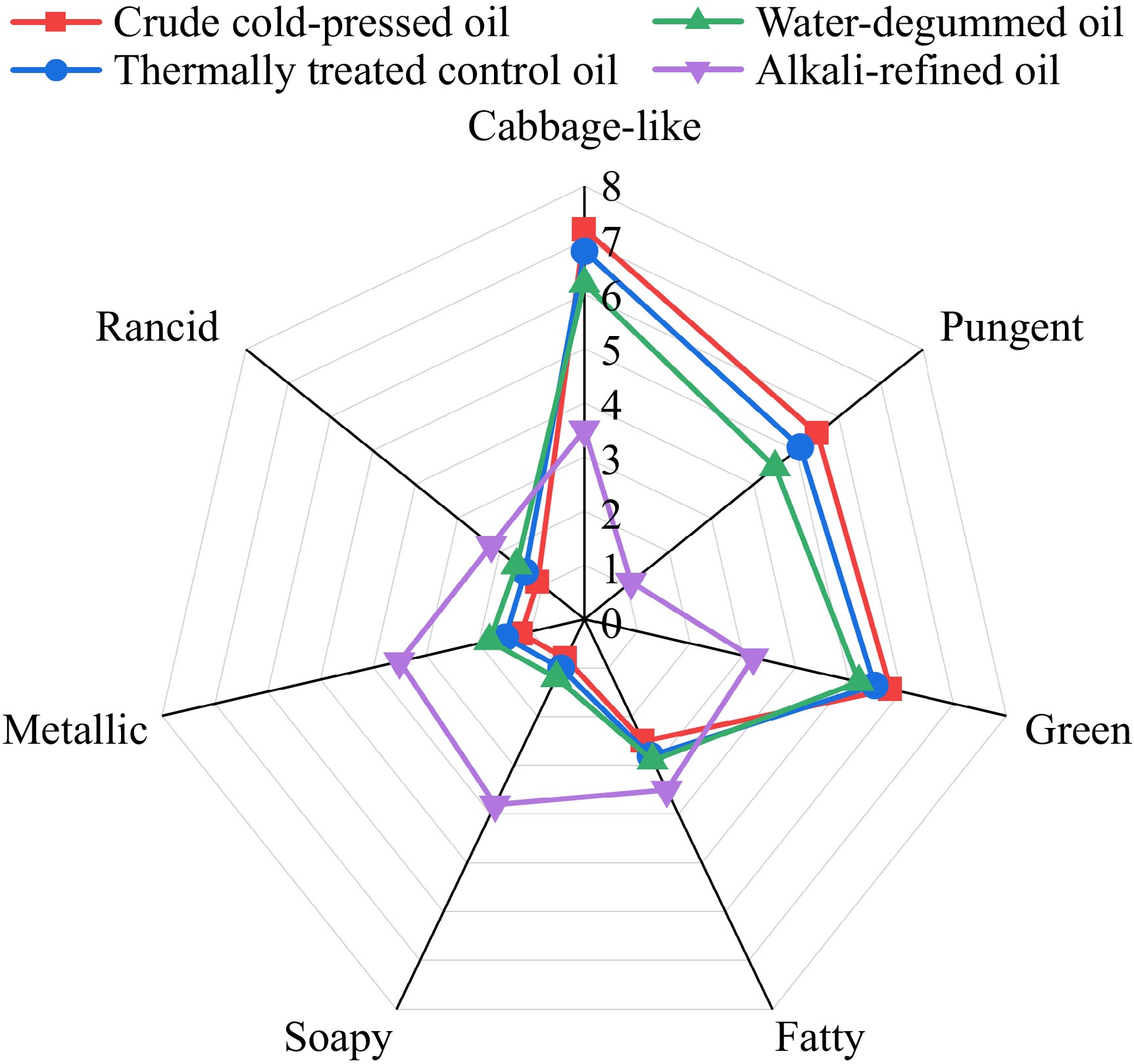
To systematically evaluate the impact of processing stages on the aroma profile of rapeseed oil, a comparative QDA was conducted on the crude cold-pressed oil, the thermally treated control oil, the water-degummed oil, and the alkali-refined oil. As shown in Fig. 1, the refining process significantly changed the sensory profile of the rapeseed oil (p < 0.05).
The crude cold-pressed oil exhibited the highest intensities for cabbage-like (7.2 ± 0.6) and green (5.8 ± 0.5) odors, alongside a notable pungency (5.5 ± 0.7). These attributes are typical of high-quality cold-pressed oils, deriving largely from the enzymatic hydrolysis of glucosinolates and the lipoxygenase pathway activity inherent to the raw seeds[21,22].
In contrast, the alkali-refined oil exhibited a change in its sensory profile. The characteristic pungent and green attributes decreased to levels of 1.1 and 3.2, respectively, indicating a loss of the original characteristics. Concurrently, the alkali treatment was associated with the formation of undesirable off-flavors. The soapy and metallic attributes reached intensities of 3.8 ± 0.7 and 3.5 ± 0.6 in the alkali-refined oil. These values exceeded those observed in all other samples (p < 0.05), including the thermally treated control oil. The thermally treated control oil and water-degummed oil exhibited sensory profiles that were transitional between those of the crude and refined oils. Thermal stress alone in the thermally treated control oil was associated with a slight increase in metallic nuances (1.5) relative to the crude cold-pressed oil (1.2), and a decrease in pungency (5.1). However, these values remained lower than those observed in the alkali-refined oil, reinforcing the hypothesis that the high pH environment is a catalyst for specific off-flavor formation. Interestingly, the cabbage-like attribute, while highest in the crude oil, was reduced in the alkali-refined oil (3.5). These sensory data indicated that while thermal treatment initiates general flavor degradation, the alkali environment represents the primary condition associated with the dominant soapy and metallic defects. These sensory results provided the necessary foundation for the subsequent flavor identification.
Identification of odor-active compounds in rapeseed oil across processing stages via aroma extract dilution analysis
-
To elucidate the chemical basis of the sensory changes described above, AEDA was applied to the volatile flavor compounds isolated from the crude cold-pressed oil, thermally treated control oil, water-degummed oil, and alkali-refined oil. This stepwise screening resulted in the detection of 46 odor-active compounds in the FD factor range of 1 to 512. Comparative evaluation of FD factors across the four processing stages (Table 1) revealed a shift that aligned with the sensory observed by QDA.
Table 1. Flavor dilution factors of volatile flavor compounds in rapeseed oils subjected to different treatments via aroma extract dilution analysis.
No. Odorant CAS number Odor quality RIa FD factorsb CCOc TTCd WDOe AROf Nitriles 1 3-Butenenitrile 109-75-1 Pungent, mustard-like 1176 64 64 32 8 2 4-Pentenenitrile 592-51-8 Pungent,metallic 1280 128 128 128 64 3 4-(Methylsulfanyl)butylnitrile 59121-24-3 Broth-like, garlic-like, sulfuric 1721 64 64 32 16 4 5-(Methylsulfanyl)pentanenitrile 59121-25-4 Broccoli-like, cabbage-like 1845 16 16 8 4 5 Phenylacetonitrile 140-29-4 Mushroom-like 1922 8 8 8 2 6 3-Phenylpropanenitrile 645-59-0 Sour, fatty 2051 4 4 4 4 Aldehydes 7 Hexanal 66-25-1 Fatty, grassy, green 1078 32 32 64 64 8 Heptanal 111-71-7 Green, rancid 1192 8 8 16 16 9 Octanal 124-13-0 Citrus-like, fatty, soapy 1280 8 16 16 32 10 Nonanal 124-19-6 Citrus-like, soapy, waxy 1381 16 32 32 128 11 Decanal 112-31-2 Citrus-like, soapy, coriander-like 1492 4 8 16 64 12 Undecanal 112-44-7 Citrus-like, metallic 1592 < 1 2 4 16 13 Dodecanal 112-54-9 Floral, soapy 1690 < 1 < 1 < 1 4 14 (Z)-3-Hexenal 69112-21-6 Fresh green 1129 128 64 32 < 1 15 (E)-2-Heptenal 505-57-7 Fatty, almond 1220 8 16 16 32 16 (E)-2-Octenal 2548-87-0 Fatty, nutty, green 1420 8 16 16 32 17 (E)-2-Nonenal 18829-56-6 Cucumber-like, fatty 1527 4 8 16 64 18 (E,Z)-2,6-Nonadienal 557-48-2 Cucumber-like, fatty 1577 16 16 8 4 19 (E,E)-2,4-Nonadienal 5910-87-2 Deep-fried, fatty, green 1693 8 16 32 64 20 (E)-2-Decenal 3913-81-3 Tallowy, metallic 1629 4 8 16 32 21 (E,E)-2,4-Decadienal 25152-84-5 Deep-fried, fatty, flowery 1801 8 16 32 64 22 Trans-4,5-Epoxy-(E)-2-decenal 134454-31-2 Metallic 2003 < 1 4 8 256 23 3-Methylbutanal 590-86-3 Malty, dark chocolate 927 16 16 16 32 24 2-Methylbutanal 96-17-3 Malty, almond 931 16 16 16 32 25 Phenylacetaldehyde 122-78-1 Flowery, honey-like 1650 32 32 16 8 26 Benzaldehyde 100-52-7 Almond-like, burnt sugar 1512 4 4 4 8 Ketones 27 1-Penten-3-one 1629-58-9 Pungent, fishy 1017 8 8 16 64 28 6-Methyl-5-hepten-2-one 110-93-0 Green, metallic 1340 4 4 2 2 29 1-Octen-3-one 4312-99-6 Mushroom-like, metallic 1294 8 16 32 128 30 (Z)-1,5-Octadien-3-one 65767-22-8 Geranium-like, metallic 1364 < 1 2 4 16 Alcohols & Phenols 31 1-Octen-3-ol 3391-86-4 Mushroom-like 1435 16 16 8 2 32 (Z)-3-Hexen-1-ol 928-96-1 Cut grass, cucumber-like 1381 32 16 8 < 1 33 2-Methoxyphenol 90-05-1 Smoky, phenolic, sweet 1860 64 32 16 < 1 34 4-Methylphenol 106-44-5 Fecal, stable-like 2081 32 16 8 < 1 35 4-Vinylguaiacol 7786-61-0 Spicy, clove-like, woody 2194 64 32 16 < 1 36 2-Phenylethanol 60-12-8 Flowery, honey-like 1909 8 8 4 < 1 Pyrazines 37 2-Isopropyl-3-methoxypyrazine 25773-40-4 Earthy, potato-like 1426 4 4 4 2 Volatile sulfur-containing compounds 38 Allyl isothiocyanate 57-06-7 Green, sulfury, pungent 1360 128 64 32 < 1 39 Dimethyl disulfide 624-92-0 Moldy, onion-like 1066 8 16 32 64 40 4-Isothiocyanato-1-butene 3386-97-8 Pickled, pungent, spicy 1453 32 16 8 < 1 41 Dimethyl trisulfide 3658-80-8 Cabbage-like, sulfury 1365 256 128 64 16 42 3-methylthiopropanal 3268-49-3 Cooked potato-like 1448 32 32 32 8 Others 43 2-Pentylfuran 3777-69-3 Vegetable-like 1232 8 16 32 128 44 Acetic acid 64-19-7 Pungent, sour, vinegar-like 1444 64 64 32 < 1 45 Butanoic acid 107-92-6 Sweaty, cheese-like 1620 32 32 16 < 1 46 Octanoic acid 124-07-2 Soapy, spicy 2054 16 16 16 < 1 a RI meant retention index calculated in this experiment. b FD meant flavor dilution factor. c CCO meant crude cold-pressed oil. d TTC meant thermally treated control oil. e WDO meant water-degummed oil. f ARO meant alkali-refined oil. The characteristic pungency of the raw material, which received the highest intensity score in the crude cold-pressed oil, was clearly attributable to a specific group of nitriles and isothiocyanates. In the crude cold-pressed oil, allyl isothiocyanate (pungent and sulfury odor) exhibited a high FD factor of 128. This compound is a well-known product of sinigrin and is a key marker for the biting taste of Brassica vegetables[19,23]. However, this flavor compound was progressively depleted during the processing simulation (FD = 128 vs 64 vs 32 vs < 1). A similar downward trend was observed for other nitrile compounds, such as 3-butenenitrile (pungent, mustard-like, FD = 64 vs 64 vs 32 vs 8) and 4-pentenenitrile, which dropped from the raw material to the alkali-refined oil. This drastic reduction in the FD factors of nitrogen- and sulfur-containing compounds aligned with the statistical decrease of the pungent sensory attribute in the alkali-refined oil, confirming that the refining process effectively strips the oil of its natural pungency.
Furthermore, the fresh green attribute of the crude cold-pressed oil was dominated by (Z)-3-hexenal. This lipoxygenase-derived aldehyde showed the highest activity in the raw oil (FD = 128) but was labile during refining (fresh green, FD = 128 vs 64 vs 32 vs <1). Its degradation, likely via isomerization to (E)-2-hexenal or oxidation to acid, correlated with the loss of the freshly crushed seed character. In contrast, hexanal, representing a more generic fatty, grassy, and green note, showed an increase in the processed samples (FD = 32 vs 32 vs 64 vs 64)[24]. This increase likely reflected mild oxidative changes occurring during the heating and mixing steps.
The emergence of the unpleasant soapy attribute in the alkali-refined oil was associated with the accumulation of medium-chain saturated aldehydes. Specifically, nonanal showed a notable increase in the alkali-treated sample. While present in the crude oil, its potency increased substantially in the final stage (citrus-like, soapy, FD = 16 vs 32 vs 32 vs 128). This was accompanied by increases in decanal, which is described as having soapy and coriander-like notes (FD = 4 vs 8 vs 16 vs 64). The elevated FD factors of these specific aldehydes in the final stage contrast with the loss of shorter-chain green aldehydes.
The metallic attribute, which was perceived in the alkali-refined oil, can be primarily traced back to specific lipid peroxidation products. The most potent contributor was identified as trans-4,5-epoxy-(E)-2-decenal. This compound had a negligible FD factor in the crude cold-pressed oil, but increased to the highest FD factor after alkali-refined (intense metallic, FD = < 1 vs 4 vs 8 vs 256). The detection of this epoxy-aldehyde has an extremely low odor threshold, meaning even trace amounts can dominate the aroma profile[9]. Its contribution was likely synergistically enhanced by vinyl ketones, such as 1-octen-3-one (mushroom-like, metallic, FD = 8 vs 16 vs 32 vs 128), which also reached its maximum intensity in the final oil. Finally, the cabbage-like odor was consistent with the presence of volatile sulfur-containing compounds, particularly dimethyl trisulfide, which showed high FD factors in the crude oil (FD = 256) but decreased significantly in the alkali-refined oil (FD = 16), mirroring the sensory reduction of the cabbage-like odor[25].
Quantitative analysis and odor activity value calculation of key aroma compounds in rapeseed oil across processing stages
-
To identify the key aroma compounds responsible for the soapy and metallic off-flavors, the concentrations of the 46 identified volatiles were quantified using external standard calibration curves constructed using peak area ratios relative to the internal standard (Supplementary Table S3). The concentrations (Fig. 2; Supplementary Table S4) were then divided by their respective odor thresholds to calculate OAV (Table 2).
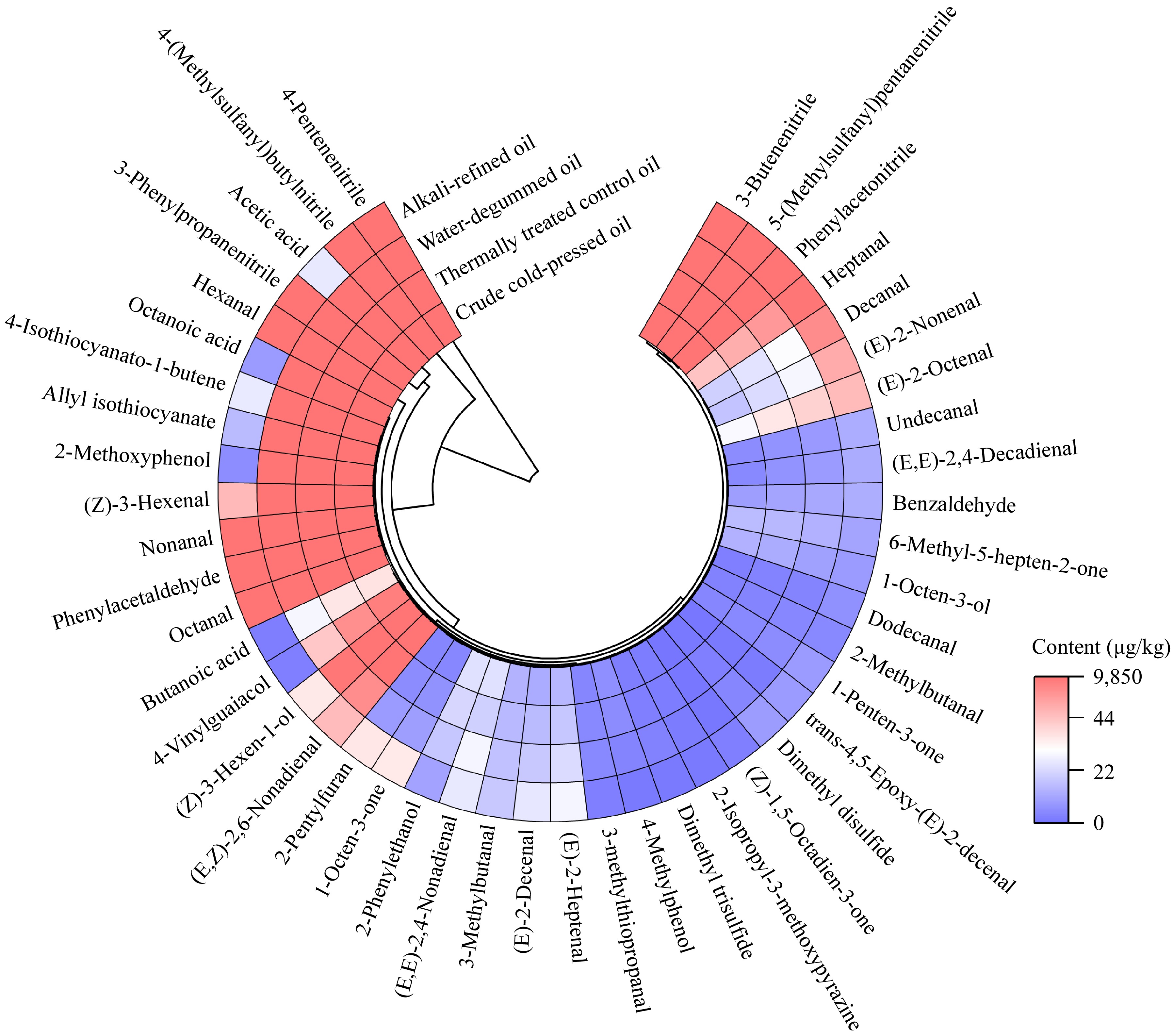
Figure 2.
Concentrations of volatile flavor compounds in crude cold-pressed, thermally treated, water-degummed, and alkali-refined rapeseed oils.
Table 2. Odor activity values of key odorants in crude cold-pressed, thermally treated, water-degummed, and alkali-refined rapeseed oils.
Odorant OT (μg/kg)a OAVb Crude cold-pressed oil Thermally treated
control oilWater-degummed oil Alkali-refined oil Nitriles 3-Butenenitrile 700 1 1 1 < 1 4-Pentenenitrile 300 33 32 30 20 4-(Methylsulfanyl) butylnitrile 2,000 2 2 2 1 5-(Methylsulfanyl) pentanenitrile 50 6 6 5 2 Phenylacetonitrile 60 6 6 5 2 3-Phenylpropanenitrile 500 2 2 2 2 Aldehydes Hexanal 300 2 2 2 2 Heptanal 250 < 1 < 1 < 1 < 1 Octanal 56 2 2 2 4 Nonanal 150 1 1 1 2 Decanal 650 < 1 < 1 < 1 < 1 Undecanal 5,000 < 1 < 1 < 1 < 1 Dodecanal 3,000 < 1 < 1 < 1 < 1 (Z)-3-Hexenal 3 54 47 37 15 (E)-2-Heptenal 850 < 1 < 1 < 1 < 1 (E)-2-Octenal 4 7 9 10 11 (E)-2-Nonenal 150 < 1 < 1 < 1 < 1 (E,Z)-2,6-Nonadienal 3.8 18 16 15 12 (E,E)-2,4-Nonadienal 1.5 16 13 19 17 (E)-2-Decenal 10 1 1 2 2 (E,E)-2,4-Decadienal 180 1 1 2 3 Trans-4,5-Epoxy-(E)-2-decenal 1.3 < 1 1 1 7 3-Methylbutanal 5.4 3 3 3 3 2-Methylbutanal 2.2 1 2 2 2 Phenylacetaldehyde 83 2 2 2 2 Benzaldehyde 60 < 1 < 1 < 1 < 1 Ketones 1-Penten-3-one 0.73 1 5 6 7 6-Methyl-5-hepten-2-one 1,000 < 1 < 1 < 1 < 1 1-Octen-3-one 10 < 1 1 1 4 (Z)-1,5-Octadien-3-one 0.13 2 4 6 19 Alcohols and Phenols 1-Octen-3-ol 1 13 12 10 9 (Z)-3-Hexen-1-ol 1,100 < 1 < 1 < 1 < 1 2-Methoxyphenol 16 8 8 5 < 1 4-Methylphenol 25 < 1 < 1 < 1 < 1 4-Vinylguaiacol 50 1 1 1 < 1 2-Phenylethanol 211 < 1 < 1 < 1 < 1 Pyrazines 2-Isopropyl-3-methoxypyrazine 0.01 192 184 173 120 Volatile sulfur-containing compounds Allyl isothiocyanate 30 5 4 3 1 Dimethyl disulfide 12 < 1 < 1 < 1 1 4-Isothiocyanato-1-butene 70 2 2 1 < 1 Dimethyl trisulfide 0.03 70 83 93 40 3-methylthiopropanal 0.2 19 21 23 13 Others 2-Pentylfuran 100 5 4 4 1 Acetic acid 750 2 2 1 < 1 Butanoic acid 7.9 5 5 4 < 1 Octanoic acid 3,000 < 1 < 1 < 1 < 1 a Thresholds were derived from the book 'Compilations of odour threshold values in air, water and other media (Edition 2011)'[39]. b OAVs were calculated by dividing the concentrations by the respective odor thresholds. The sensory profile of the crude cold-pressed oil, characterized by pungent and cabbage-like attributes, was rationalized by high concentrations of sulfur-containing compounds. Allyl isothiocyanate (no. 38) was quantified at 139.2 μg/kg in the crude cold-pressed oil. A depletion was observed during the refining simulation, decreasing to 15.4 μg/kg in the alkali-refined oil. This loss aligned with the decline in the perception of pungent attributes[26]. The OAV calculation reflected this trend, showing a decrease from 5 in the crude oil to 1 in the alkali-refined oil. This demonstrated that alkali refining could remove some volatile hydrolysis products of glucosinolates, likely due to their volatility and susceptibility to nucleophilic attack by hydroxide ions. Similarly, (Z)-3-hexenal (no. 14), responsible for the fresh green note, decreased from 162.3 μg/kg in the crude cold-pressed oil to 45.6 μg/kg in the alkali-refined oil. Consequently, its odor activity value decreased across the processing stages (OAV = 54 vs 47 vs 37 vs 15), confirming its heat-lability and the corresponding loss of green odor. The cabbage-like odor driver, dimethyl trisulfide, also decreased from 2.1 μg/kg (OAV = 70) in the crude oil to 1.2 μg/kg (OAV = 40) in the alkali-refined oil, consistent with the sensory reduction.
The metallic sensory defect may be elucidated through the formation of specific lipid peroxidation products. The quantitative data identified trans-4,5-epoxy-(E)-2-decenal (no. 22) as a potent odorant associated with this defect[27]. In the crude cold-pressed oil, this compound was present at trace levels (0.50 μg/kg), resulting in an OAV less than 1. However, the alkali refining process was associated with a significant accumulation, elevating its concentration to 8.50 μg/kg in the alkali-refined oil. Due to its extremely low odor threshold, this increase resulted in a rise in its odor activity value to 7. This metallic profile was further intensified by a group of vinyl ketones. (Z)-1,5-Octadien-3-one (no. 30), characterized by its geranium-like and metallic odor[28], showed a substantial increase from 0.20 μg/kg in the crude oil to 2.50 μg/kg in the alkali-refined oil, raising its OAV from 2 to 19. 1-Octen-3-one (no. 29) also increased from 4.0 μg/kg to 35.2 μg/kg. Although its calculated OAV remained moderate (OAV = 0 to 4) based on the threshold used, its high FD factor (128) and significant concentration surge suggested it played a critical synergistic role. The concomitant rise of these potent compounds provides a molecular explanation for the metallic off-flavor perceived by the sensory panel.
Meanwhile, the quantitative analysis revealed that the soapy attribute was likely to increase in medium-chain saturated aldehydes. The concentration of nonanal (no. 10) exhibited a significant increase, reaching 256.8 μg/kg in the alkali-refined oil compared to 160.5 μg/kg in the crude cold-pressed oil. Similarly, decanal (no. 11) increased from 20.1 μg/kg in the crude oil to 55.4 μg/kg in the refined sample. While the calculated OAVs for decanal remained below 1, decanal reached an FD factor of 64, and nonanal reached 128 in the alkali-refined oil. This discrepancy between OAV and FD data suggested that the soapy perception was likely driven by the additive or synergistic effect of these homologous aldehydes, such as octanal, nonanal, decanal, and undecanal, rather than a single compound exceeding its threshold.
Importantly, volatile acids such as acetic acid and butanoic acid were removed during alkali refining. For example, acetic acid dropped from 1542.4 μg/kg in the crude oil to 25.5 μg/kg in the alkali-refined oil, reducing its OAV from 2 to less than 1. This confirmed the efficacy of the deacidification process in removing free fatty acids and acidic volatiles. Furthermore, the earthy-odor 2-isopropyl-3-methoxypyrazine (no. 37) maintained a relatively constant trace concentration throughout the process (1.92 to 1.20 μg/kg), maintaining high OAVs (192 vs 120). Since pyrazines are stable, they act as a consistent background aroma and do not drive the differential off-flavors generated by the refining steps.
In summary, the quantitative analysis showed that the metallic off-flavors were attributed to lipid peroxidation products such as trans-4,5-epoxy-(E)-2-decenal and (Z)-1,5-octadien-3-one, which were formed during alkali treatment and exhibited significant OAV increases. Meanwhile, the soapy off-flavors were associated with the accumulation of medium-chain saturated aldehydes. Although their individual OAVs were low, their high FD factors and collective concentration implicate them as the contributors of the soapy off-flavor defect. These findings establish the targets for subsequent validation through aroma recombination experiments.
Aroma reconstitution and omission experiments
-
To validate the accuracy of the above results, an aroma recombination model was prepared using the authentic odorants at their concentrations determined in the alkali-refined oil[29,30]. As presented in Fig. 3, the sensory profile of the recombination model was in good agreement with the original alkali-refined oil. The trained panel rated the soapy intensity of the model at 3.7 and the metallic intensity at 3.6, values which were statistically indistinguishable from the scores of 3.8 and 3.5 observed in the original alkali-refined oil (p > 0.05). The absence of significant differences across all attributes, including the background fatty and green odors, confirmed that the identified and quantified compounds represent the aroma profile of the alkali-refined oil.
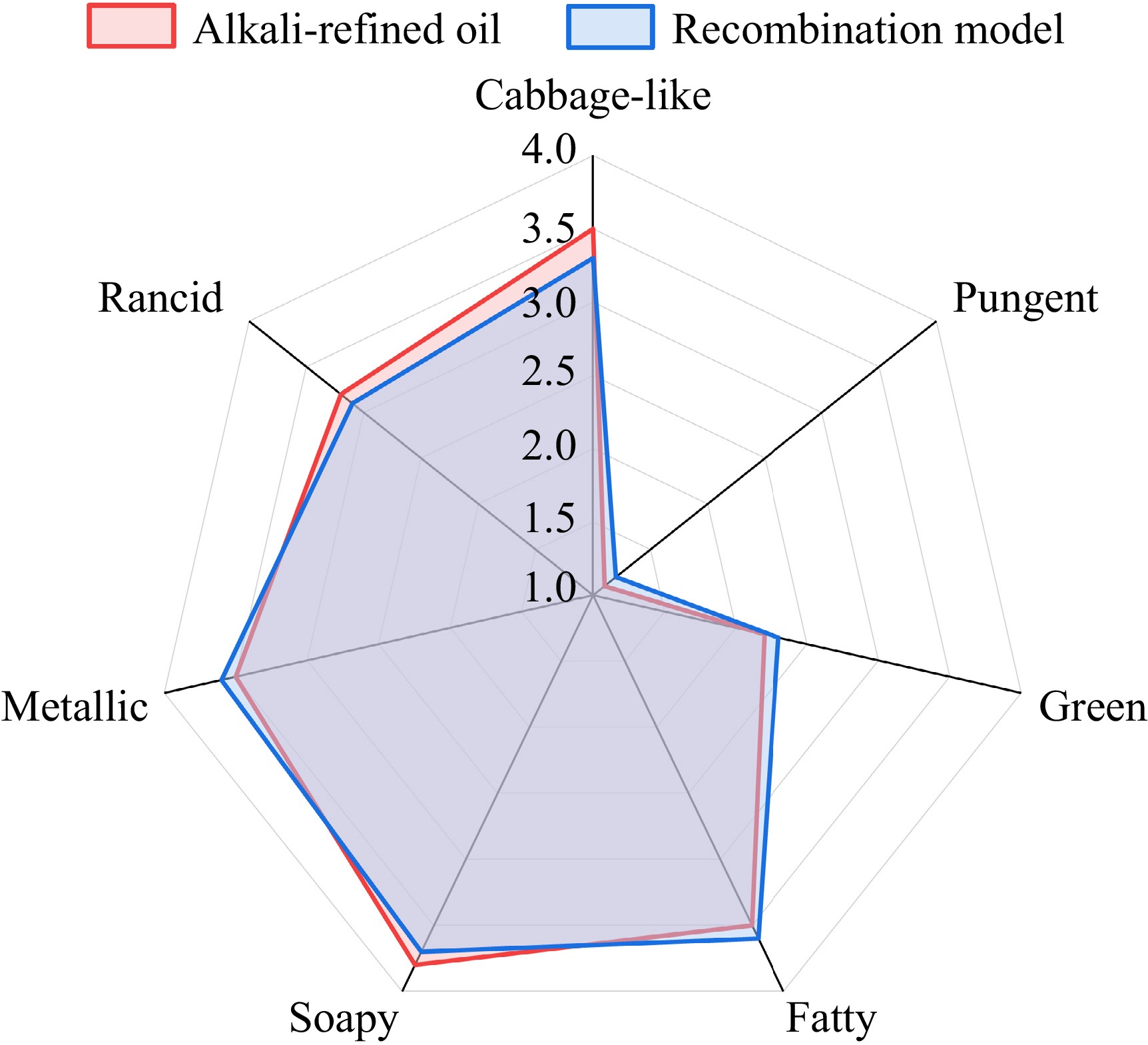
Figure 3.
Comparison of sensory profiles between alkali-refined rapeseed oil and its aroma recombination model.
To further clarify the contribution of specific flavor classes to the key defects, omission experiments were conducted (Table 3). A series of models was prepared by omitting one specific chemical class at a time from the full recombination model. Excluding the aldehyde fraction caused a highly significant decrease (p < 0.001) in both soapy and metallic intensities. These results confirmed the dominance of aldehydes in the off-flavor profile, identifying nonanal and decanal as the drivers of the soapy off-flavor and trans-4,5-epoxy-(E)-2-decenal as the source of the metallic off-flavor.
Table 3. Significance of odorant groups on soapy and metallic off-flavors determined by aroma omission experiments.
No. Omitted group Soapy Metallic 1 Aldehydes ***a ***a 2 Ketones nsc **b 3 Volatile sulfur-containing compounds ns ns 4 Nitriles ns ns 5 Pyrazines ns ns 6 Alcohols ns ns 7 Phenols ns ns 8 Acids ns ns 9 Isothiocyanates ns ns a p < 0.001. b p < 0.01. c Not significant. Furthermore, the omission of ketones significantly lowered the metallic intensity (p < 0.01) but had no observed effect on the soapy note. This specific reduction aligned with the quantitative analysis, which identified vinyl ketones such as (Z)-1,5-octadien-3-one and 1-octen-3-one as potent metallic odorants that do not possess soapy characteristics. This result experimentally verified that the metallic off-flavor is a composite perception driven by them. In contrast, removing volatile sulfur compounds, nitriles, or alcohols resulted in no significant changes to the defect intensities. Although compounds such as dimethyl trisulfide were identified in the oil with high OAVs, their omission suggested they are responsible for the general background aroma derived from the raw seeds rather than the process-induced soapy and metallic off-flavors.
-
Based on established theories of vegetable oil flavor chemistry and the analytical data from this study, the metallic off-flavor characterized by trans-4,5-epoxy-(E)-2-decenal and vinyl ketones was primarily associated with the decomposition of linoleic and linolenic acid hydroperoxides[31,32]. Specifically, the alkaline environment promotes the rearrangement of the linoleic acid 13-hydroperoxide isomer into the epoxy-aldehyde[33]. Furthermore, the significant increase in 1-octen-3-one suggests the concurrent breakdown of the 9-hydroperoxide derived from linolenic acid, which is known to be unstable under basic conditions[34,35].
Simultaneously, the soapy defect observed in the alkali-refined oil is likely derived from the autoxidation of oleic acid. Unlike polyunsaturated precursors that degrade readily under thermal stress, oleic acid hydroperoxides exhibit relatively higher thermal stability, which may explain their lower abundance in the thermally treated control. However, the high pH environment significantly reduces the activation energy for their decomposition. This facilitates the breakdown of the 8- and 10-hydroperoxides, leading to the specific scission of the carbon chain that releases medium-chain saturated aldehydes, including nonanal and decanal[36−38]. Regarding the soapy perception, it is worth noting that while the individual OAVs of decanal and nonanal were relatively low in the alkali-refined oil, their FD factors were high. This discrepancy suggests that the soapy off-flavor is not driven by a single compound exceeding its threshold, but rather by the additive or synergistic effects of octanal, nonanal, decanal, and undecanal.
Implications for industrial processing and study limitations
-
Several limitations of this work should be acknowledged. First, the refining process was simulated on a laboratory scale. While care was taken to replicate industrial parameters, the mass transfer rates and surface-to-volume ratios in a laboratory flask differ from those in industrial continuous refining towers, which could influence the exact yield of volatile oxidation products.
Second, this study focused on the identification of volatile odorants, whereas critical non-volatile factors were not quantified. Specifically, the reaction kinetics were likely influenced by the presence of trace metal ions and specific hydroperoxide isomers, neither of which was monitored in this study. Although the alkaline environment is the primary driver for off-flavor formation, transition metals such as iron and copper could act as co-catalysts, synergistically enhancing the decomposition of hydroperoxides even at low concentrations. Future studies should, therefore, incorporate the quantification of trace metals and hydroperoxide precursors and employ chelators to verify their specific contributions to the reaction pathways.
Third, the omission tests confirmed the role of aldehydes and ketones but did not investigate potential perceptual interactions between the off-flavors and the remaining trace amounts of native pungent compounds, which might act as masking agents in the raw material.
Despite these limitations, the findings offer actionable insights for the edible oil industry. The study identifies the alkali neutralization step as a critical control point for flavor quality. Reducing the residence time or lowering the temperature during the alkali contact phase could mitigate the formation of potent off-flavors. Monitoring the peroxide value alone may be insufficient for quality control, as the breakdown of peroxides into secondary odorants represents the actual source of the defect. Targeted monitoring of the identified key markers, specifically the epoxy-aldehyde and nonanal, would provide a more sensitive and relevant metric for assessing the sensory impact of the rapeseed oil refining process.
-
By applying a sensomics strategy that integrates quantitative descriptive analysis and aroma extract dilution analysis, this study identified the key odorants associated with soapy and metallic off-flavors in alkali-refined rapeseed oil. On the basis of the calculation of odor activity values and omission experiments, the metallic off-flavor was attributed to the formation of trans-4,5-epoxy-(E)-2-decenal, (Z)-1,5-octadien-3-one, and 1-octen-3-one. The soapy off-flavors were associated with the accumulation of medium-chain saturated aldehydes, particularly nonanal and decanal. Aroma recombination and omission experiments validated that aldehydes and ketones constitute the primary flavor classes responsible for soapy and metallic off-flavors. The alkali neutralization step was identified as the critical control point for flavor quality. This work provides a theoretical basis for targeted process optimization and improvement of flavor and quality control in rapeseed oil refining.
-
Ethical clearance for the involvement of human subjects in this study was obtained from the Research Ethics Committee of China Agricultural University, under reference number CAUHR-20240703.
-
The authors confirm contributions to the paper as follows: study conception and design: Wu J, Zhou Y, Shi H, Liu C, Xu X; data collection: Yu P, Lao F; analysis and interpretation of results: Yu P, Lao F, Wu J; draft manuscript preparation: Wang J. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
-
Thanks for the financial support from Arawana Charity Foundation. Thanks to all the R&D personnel of Wilmar (Shanghai) Biotechnology Research and Development Center Co., Ltd, for their technical guidance on this project. The author extends special thanks to distinguished engineers Ms. Jing Wang, Ms. Yunfei Zhou, and Mr. Chuan Zhou for their invaluable assistance and significant contributions to the experimental design of the vegetable oil off-flavor identification project over the past four years.
-
The authors declare that they have no conflict of interest.
-
accompanies this paper online at: https://doi.org/10.48130/fia-0026-0010.
- Supplementary Table S1 Purity and suppliers of authentic standards used for identification and quantification.
- Supplementary Table S2 Sensory attributes, definitions, and reference standards used for descriptive analysis of rapeseed oil samples.
- Supplementary Table S3 Method validation parameters for the quantification of volatile flavor compounds in cold-pressed rapeseed oil.
- Supplementary Table S4 Concentrations of volatile compounds in crude cold-pressed, thermally treated, water-degummed, and alkali-refined rapeseed oils.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press on behalf of China Agricultural University, Zhejiang University and Shenyang Agricultural University. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Yu P, Wang J, Zhou Y, Lao F, Shi H, et al. 2026. Identification of the key odorants causing soapy and metallic off-flavors in alkali-refined rapeseed oil via molecular sensory science. Food Innovation and Advances 5(1): 123−132 doi: 10.48130/fia-0026-0010
Identification of the key odorants causing soapy and metallic off-flavors in alkali-refined rapeseed oil via molecular sensory science
- Received: 31 December 2025
- Revised: 27 January 2026
- Accepted: 04 March 2026
- Published online: 27 March 2026
Abstract: Alkali refining may sometimes induce soapy and metallic off-flavors in cold-pressed rapeseed oil, yet the molecular mechanisms remain unclear. A comparative flavor sensomics study was performed on crude cold-pressed, thermally treated control, water-degummed, and alkali-refined rapeseed oils. Quantitative descriptive analysis (QDA) showed that alkali refining increased soapy and metallic sensory attributes. Aroma extract dilution analysis (AEDA) detected 46 odor-active compounds across processing stages. The metallic off-flavors were primarily associated with elevated levels of trans-4,5-epoxy-(E)-2-decenal, (Z)-1,5-octadien-3-one, and 1-octen-3-one. The soapy attribute was linked to the accumulation of medium-chain saturated aldehydes, particularly nonanal and decanal. Omission experiments confirmed that these aldehydes collectively contributed to the soapy off-flavors, despite their low individual odor activity values (OAVs). Collectively, aldehydes and ketones were identified as the primary sources of off-flavor, marking the alkali neutralization step as the critical control point for flavor quality. These findings can offer theoretical guidance for process optimization of cold-pressed rapeseed oil refining.
-
Key words:
- Cold-pressed rapeseed oil /
- Alkali-refined /
- Off-flavor /
- Odor activity value /
- Aroma recombination