-
With the rapid growth of the population, the demand for vegetables has been continuously rising. Solanaceous vegetables, due to their nutritional value and multi-functionality, have become important crops worldwide. Their planting area reaches 9 million ha with an annual output of approximately 280 million t, among which, tomatoes, peppers, potatoes, and eggplants account for a prominent proportion. Solanaceous vegetables are rich in essential nutrients for the human body, and their demand at home and abroad is constantly increasing[1,2]. However, the implementation of intensive agricultural production to meet the continuously growing demand has led to the widespread application of long-term continuous cropping systems, triggering a series of biotic and abiotic stress-related challenges. Within solanaceous cropping systems, continuous cropping significantly exacerbates infestations of root-knot nematodes, resulting in economic losses of up to 21%–25%. Taking pepper as an example, long-term monoculture has been demonstrated to reduce yield by as much as 70% and substantially compromise fruit quality[2]. Continuous cropping obstacles (CCOs) refer to the degradation of the crop-growing environment and deviations in plant growth and development caused by repeatedly cultivating the same crop in the same soil[3]. For instance, soil-borne diseases become more prevalent, weakening plant vigor and making crops more susceptible to diseases and developmental abnormalities. This suppression ultimately results in a significant decline in crop yields[4]. To date, most reviews on continuous cropping challenges have focused on medicinal plants, corn, and melons[5−9]. In contrast, there is a notable lack of comprehensive reviews addressing CCOs in solanaceous vegetables. Furthermore, as the demand for solanaceous vegetables continues to grow, their cultivation area is expanding, which exacerbates the occurrence of CCOs in crops including tomatoes, peppers, potatoes, and eggplants[10−13]. For example, one study revealed that 5 years of continuous pepper cultivation led to a decrease in soil enzyme activity and deterioration of the microbial environment, resulting in a significant decline in pepper yields[14]. Similarly, 5 to 7 years of continuous potato cultivation leads to the accumulation of phenolic acid allelochemicals, causing biotic and abiotic environmental stress and thus contributing to CCOs[15]. Jokela et al. also highlighted that continuous cropping disrupts the soil nutrient balance, negatively affecting the growth and nutritional quality of peppers, and ultimately reducing fruit quality[16,17].
Biochar has recently emerged as a promising amendment for mitigating CCOs, though its efficacy is highly crop-specific. Solanaceous vegetables show particularly strong responses, owing to a unique synergy: their main constraints—sensitivity to poor soil structure, pH shifts, and soil-borne diseases—are directly addressed by biochar's ability to improve rhizosphere properties, modulate nutrients, and suppress pathogens. This leads to rapid gains in yield and quality. In contrast, biochar's effects on cereals are soil-dependent; it may inhibit legume nitrogen fixation, and can immobilize nitrogen in leafy vegetables. Biochar often provides only long-term benefits in perennial systems, underscoring its distinct value for solanaceous crops. This paper presents the first bibliometric analysis of research on solanaceous vegetables and soil biochar. We systematically identify the causes of CCOs in these crops, review mitigation strategies, and elucidate the mechanisms by which biochar alleviates these obstacles, thereby providing a foundation for future research and practical application.
-
The literature visualization software VOSviewer was used to conduct data mining and analysis on the Web of Science (WOS) database. First, the main solanaceous vegetables were identified, and then the search formula was applied to retrieve relevant literature. The search date was as follows: January 6, 2026, on which the main solanaceous vegetables (tomatoes, eggplants, peppers, and potatoes) yielded a total of 5,351 relevant documents; for the solanaceous vegetables (including Solanum lycopersicum, Capsicum annuum, Solanum melongena, Solanum tuberosum, Lycium, Physalis spp, Solanum betaceum (Cyphomandra betacea), Solanum tuberosum 'Andigenum' group, Solanum juzepczukii, Solanum curtilobum, Solanum melanocerasum, Solanum nigrum, tomatoes, eggplants, peppers, and potatoes, a total of 5,606 articles were retrieved, among which the literature on tomatoes, eggplants, peppers, and potatoes accounted for 95.4% of the total. The search data was exported in the form of full records, and used as the primary datasets for bibliometric analysis. VOSviewer was used to conduct keyword co-occurrence analysis (Fig. 1), and the annual publication trends for solanaceous vegetables and the main solanaceous vegetables from 2015 to 2025 were determined. The total amounts of fruits and vegetables were compared in Fig. 2. The top 11 high-frequency keywords included tomatoes, potatoes, in vitro, physical and chemical properties, antioxidant activity, growth, resistance, bioactive compounds, oxidative stress, pseudo Arabidopsis, antioxidants, and quality. These keywords reveal that in vitro experiments on tomatoes have become a major research focus in recent years. Long-term continuous cultivation of tomatoes, potatoes, and other solanaceous vegetables has been proven to alter the physical and chemical properties of the soil, and lead to excessive accumulation of bioactive compounds, which seriously affects the normal growth, stress resistance, gene expression, and quality of the crops. To address the challenges posed by continuous cropping, this study reviews the causes of continuous cropping of the main solanaceous vegetables and discusses corresponding mitigation measures. Through in-depth research and discussion, this study aims to provide practical guidance for the healthy cultivation and sustainable production of solanaceous vegetables.
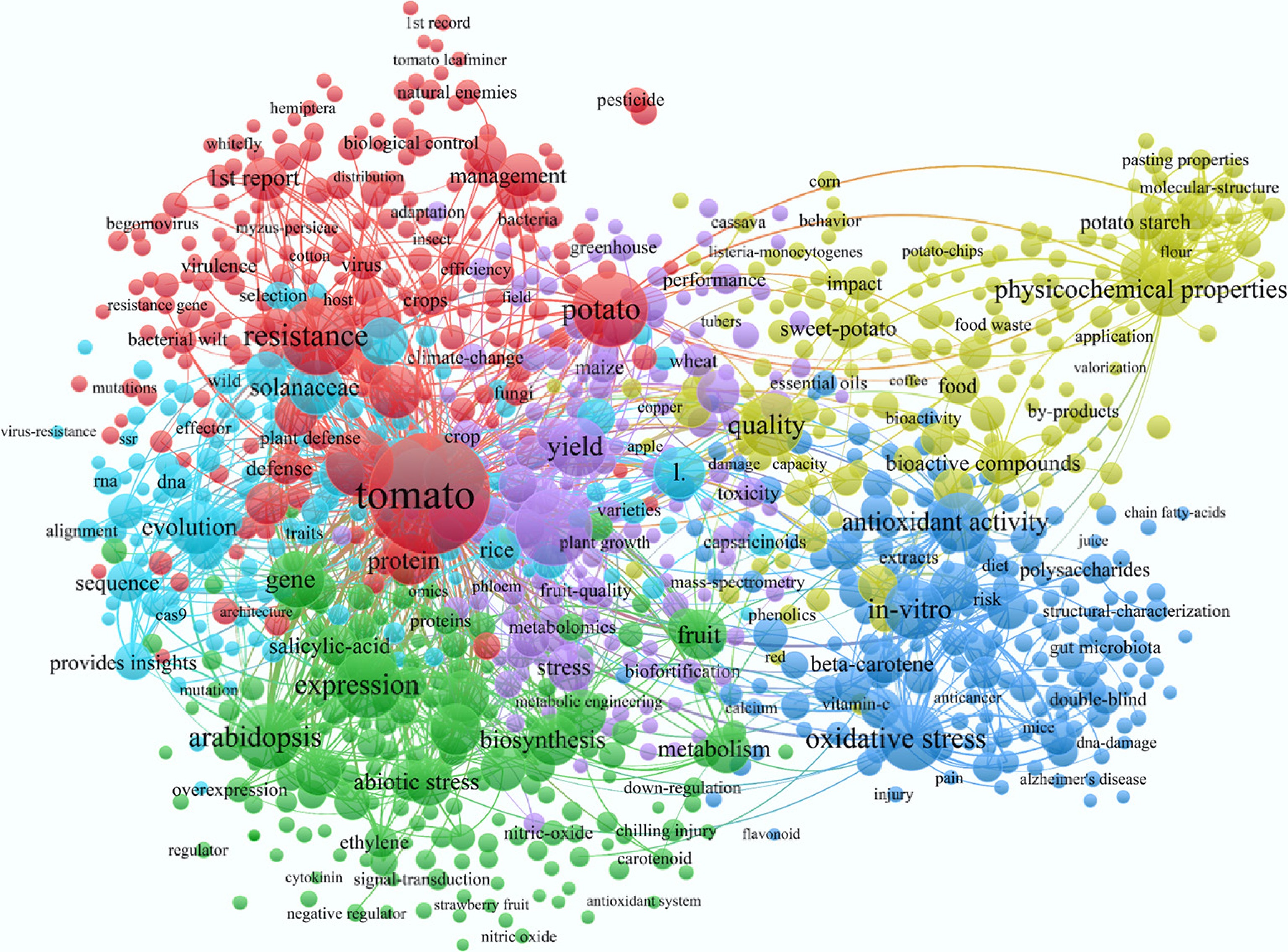
Figure 1.
Visual analysis diagram of the main solanaceous vegetables. The search period was up to January 6, 2026. Based on the Web of Science (WOS) database, the main solanaceous vegetables and nightshade vegetables were searched according to the search formula TS = (Solanum lycopersicum) OR TS = (Capsicum annuum) OR TS = (Solanum melongena) OR TS = (Solanum tuberosum) OR TS = (Lycium) OR TS = (Physalis spp) OR TS = (Solanum betaceum) OR TS = (Cyphomandra betacea) OR TS = (Solanum tuberosum 'Andigenum' Group) OR TS = (Solanum juzepczukii) OR TS = (Solanum curtilobum) OR TS = (Solanum melanocerasum) OR TS = (Solanum nigrum) OR TS = (tomatoes) OR TS = (eggplants) OR TS = (peppers) OR TS = (Potatoes). The data was visualized using VOSviewer (Version 1.6.20).
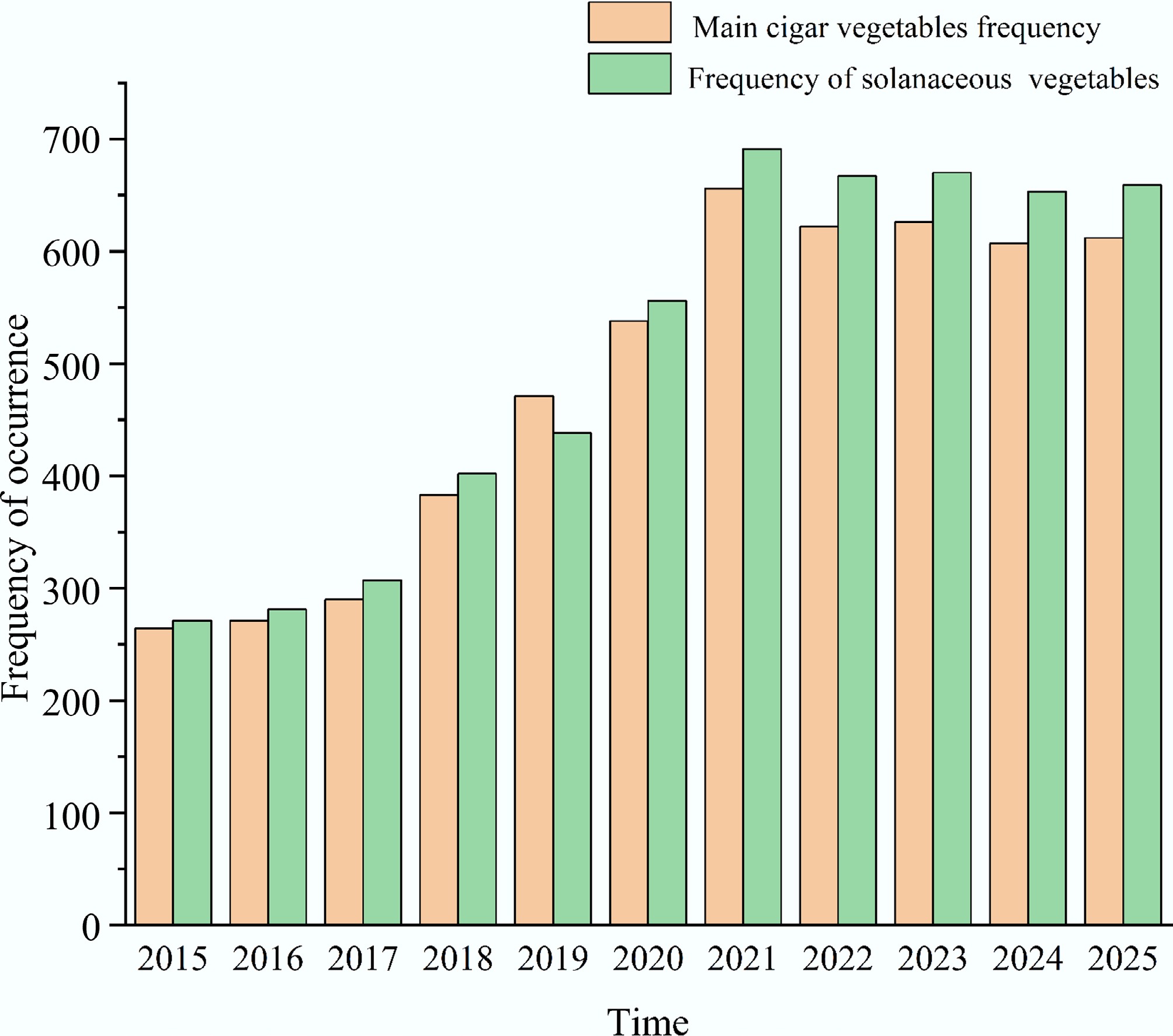
Figure 2.
Comparison of the frequency between solanaceous vegetables and the main solanaceous vegetables.
Visual analysis of research hotspots of biochar application in soil
-
To delve further into the focal points of research and the development trends in biochar application in soil, the literature visualization software VOSviewer was used for data mining and quantitative analysis of the Web of Science (WOS) database. In the WOS core collection, the search language was limited to English, the document type was an article, and the search was performed according to the search formula TS = (biochar). The search ended on January 6, 2026, and a total of 3,163 relevant documents were retrieved. VOSviewer was used to conduct keyword co-occurrence analysis.
Figure 3 presents the co-occurrence network diagram of keywords used in biochar research across multiple fields. The keywords are divided into three major clusters. The top 15 high-frequency keywords include biochar, activated carbon, adsorption, pyrolysis temperature, pyrolysis, aqueous solution, heavy metals, biomass, wastewater, hydrothermal carbonization, sewage sludge, slow pyrolysis, polycyclic aromatic hydrocarbons, black carbon, and soil. These findings indicate that the main areas of biochar application include aqueous solutions, heavy metals, wastewater, sewage sludge, polycyclic aromatic hydrocarbons, and soil. Moreover, the main application areas of biochar include adsorption. In addition, biochar, such as hydrothermal carbon and pyrolytic carbon, applies to soil treatment and other fields.
In cluster 1, biochar is most closely associated with soil, with keywords covering biochar, soil, carbon sequestration, greenhouse gases, microbial communities, plant growth, and related agronomic properties. Its primary applications focus on soil enhancement, carbon sequestration, and microbial community regulation, which facilitate plant growth and mitigate greenhouse gas emissions. Cluster 2 highlights production methods such as hydrothermal carbonization and pyrolysis of biomass, sewage sludge, and lignocellulosic waste, emphasizing biochar's role as an adsorbent, particularly in waste treatment. In cluster 3, frequent terms include activated carbon, adsorption, degradation, removal, aqueous solutions, wastewater treatment, magnetic biochar, and methylene blue. This underscores biochar's ability to adsorb and degrade pollutants, with modified magnetic biochar showing enhanced performance in wastewater remediation. As a strategy to address CCOs, biochar's soil improvement mechanism must consider potential polycyclic aromatic hydrocarbon (PAH) formation during production[18]. Thus, optimizing preparation processes and testing PAH content before use are essential.
-
Key factors influencing CCOs in solanaceous vegetables primarily arise from soil physicochemical degradation, allelopathic autotoxicity, soil-borne pathogens/pests, and microbial community shifts[19−23]. Moreover, long-term continuous cultivation of solanaceous vegetables can produce a large amount of allelopathic substances, which can have a profound negative impact on crops (Fig. 4). This significantly reduces rhizosphere microbial populations, inhibits growth hormone synthesis, disrupts metabolic pathways, and may trigger soil-borne diseases. Long-term continuous potato cultivation triggers soil salinization and physicochemical degradation, coupled with the accumulation of root-secreted secondary metabolites like vanillin. These compounds inhibit the expression of IAA synthesis genes in seedling roots and disrupt the rhizosphere microbial community, reducing beneficial bacteria and increasing pathogenic fungi. Together, these processes synergistically exacerbate crop growth suppression and the incidence of soil-borne diseases[24]. Eggplant monoculture systems demonstrate this phenomenon, where concentrated allelopathic compounds promote soil-borne pathogen transmission and cause yield losses[25]. Multiomics analyses reveal that root-accumulated allelochemicals modify rhizosphere metabolic pathways, steering the microbiota assembly toward plant-suppressive configurations.
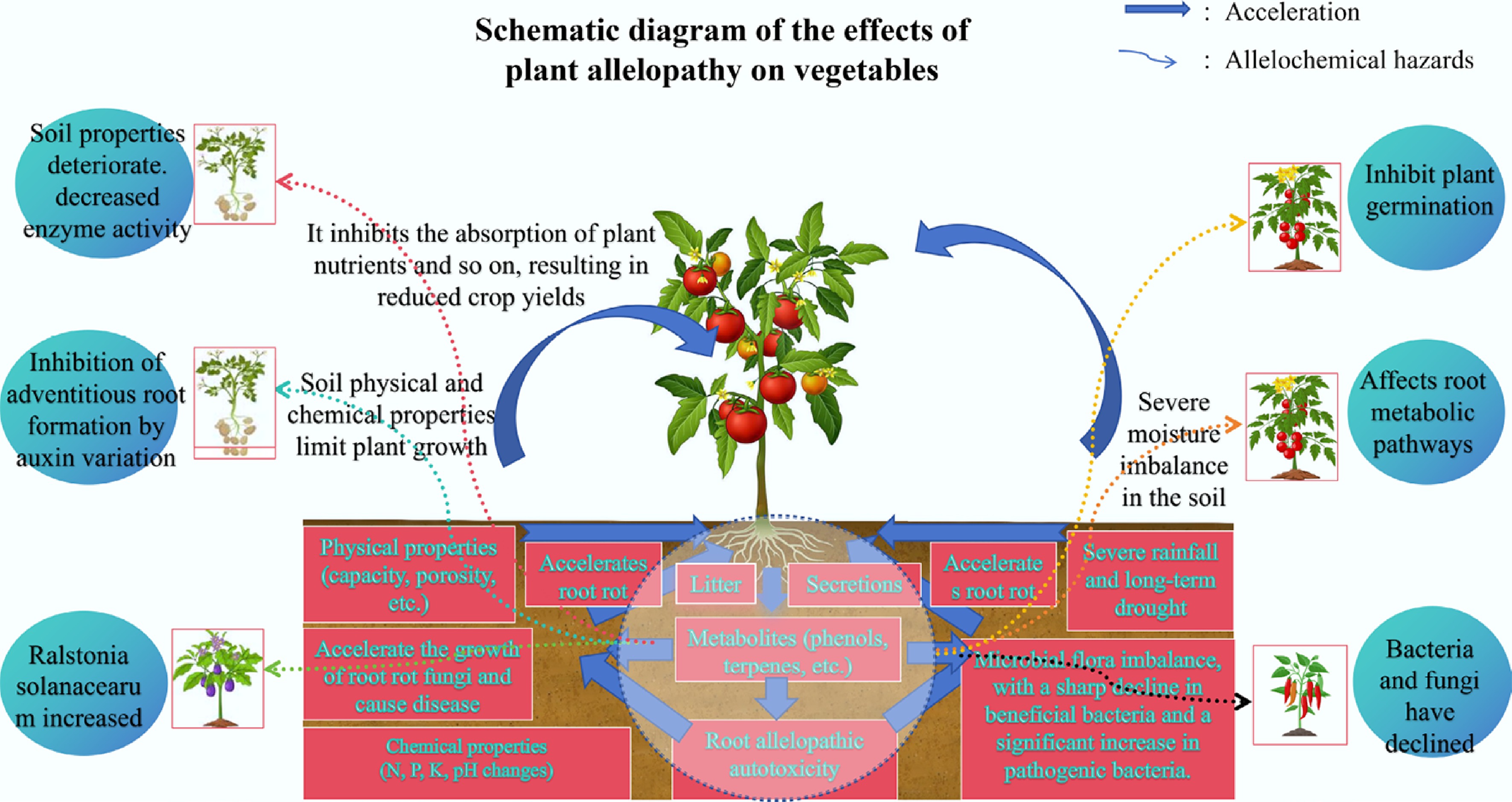
Figure 4.
Mechanism of CCOs in solanaceous vegetables[9].
Changes in soil physicochemical properties
-
The same plant requires different nutrients at different times, and continuous cropping reduces the contents of certain nutrient elements in the soil, resulting in soil nutrient deficiencies[26]. Compared with continuous potato cropping, broad bean-potato rotation significantly improved yield by improving soil nutrient conditions and microorganisms[13]. Continuous cultivation can consume soil nutrients, reduce the soil organic matter content, and alter the soil cation exchange capacity, leading to soil degradation, and the cultivated layer gradually becomes shallower[27,28]. The absence of mechanical deep tillage and organic fertilizer supplementation leads to the progressive shallowing of the plow layer and continuous deterioration of soil physical properties. This inhibits root growth, reduces water infiltration and nutrient cycling efficiency, thereby affecting soil quality and exacerbating compaction. Furthermore, soil aggregates serve as a key physical indicator of soil structure and health[29,30]. Research has shown that continuous cropping may have a negative effect on the composition of soil aggregates, their stability, and size categories, resulting in a loss of soil structure and nutrient deficiencies[31].
Continuous cropping leads to allelopathic autotoxicity
-
Allelopathic autotoxicity refers to the natural phenomenon in which plants exert direct or indirect adverse effects by releasing certain chemicals into the environment in normal life activities[32]. Many studies have shown that allelopathic autotoxicity is widespread in solanaceous vegetables, especially in large-scale crops such as potatoes, tomatoes, eggplants, and peppers[11,33−35]. Allelopathic substances usually refer to secondary metabolites of plants that are widely found in various parts of plants, such as roots, stems, leaves, and fruits[36]. There are many types of allelopathic substances, including phenols, terpenes, alkaloids, and fatty acids[37] (Fig. 5).
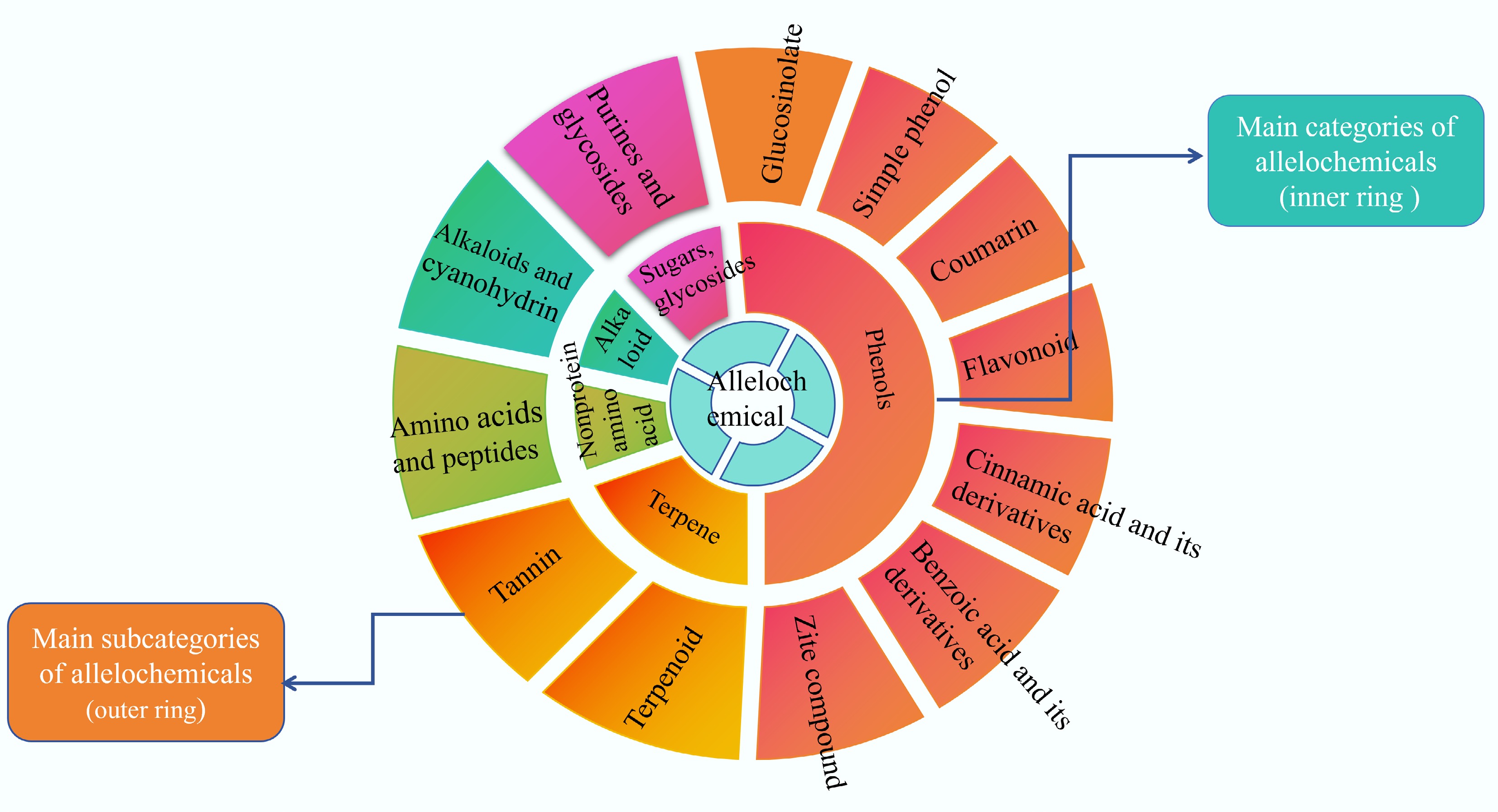
Figure 5.
Classification diagram of allelopathic substances[37].
Allelopathic substances disrupt the balance of soil microorganisms and promote the proliferation of pathogenic microorganisms. This alteration severely hinders the normal growth of solanaceous vegetables, leading to reduced crop yield and quality, and in extreme cases, may even result in total crop failure[38]. Therefore, controlling soil allelopathic substances is crucial for solanaceous vegetable production. There are certain differences in the types of allelopathic substances in the root exudates of different solanaceous vegetables (Table 1). The main allelopathic substances include cinnamic acid, ferulic acid, p-hydroxybenzoic acid, vanillic acid, and their derivatives. These allelopathic substances have adverse effects on plant roots, soil physicochemical properties, key enzyme activities, and microbial community structure and function, thereby indirectly affecting the yield of vegetables.
Table 1. Effects of allelopathy on solanaceous vegetables
Solanaceous vegetable species Allelopathic substances Impact on crops and soil Ref. Potato Vanillin, ferulic acid, and p-hydroxybenzoic acid Reduced root IAA content inhibits adventitious root formation, ultimately
reducing yield.[13,34,39] Eggplant Esters, alcohols, hydrocarbons, fatty acids, ethers, ketones, and benzene Continuous cropping promotes Ralstonia solanacearum proliferation, disrupting microbial community stability and function while lowering diversity. [10,40,41] Pepper Olefins, phenols, alkanes, aromatics, alcohols, ketones, etc. Decreased bacterial and fungal genera abundance; reduced soil microaggregates; nonlinear β-glucosidase decline; altered microbial composition; impaired plant growth and pathogen resistance. [11,42] Tomato Alcohols, terpenes, phenolic acids, etc. Mainly including benzoic acid, ferulic acid, and cinnamic acid Root exudates hinder the germination process of tomato seeds and impede
the spontaneous growth of tomato seedlings.[12] Allelopathic substances originate from plant metabolism, environmental stimuli, and genetic inheritance. When damaged by diseases and insect pests, eggplants produce solanine that inhibits its growth and negatively affects their physiology, reproduction, quality, and stress resistance[43]. To enhance solanaceous vegetable yield and quality, it is essential to investigate their allelopathic/autotoxic mechanisms and implement cultivation and breeding strategies to mitigate these adverse effects.
Changes in soil microbial communities
-
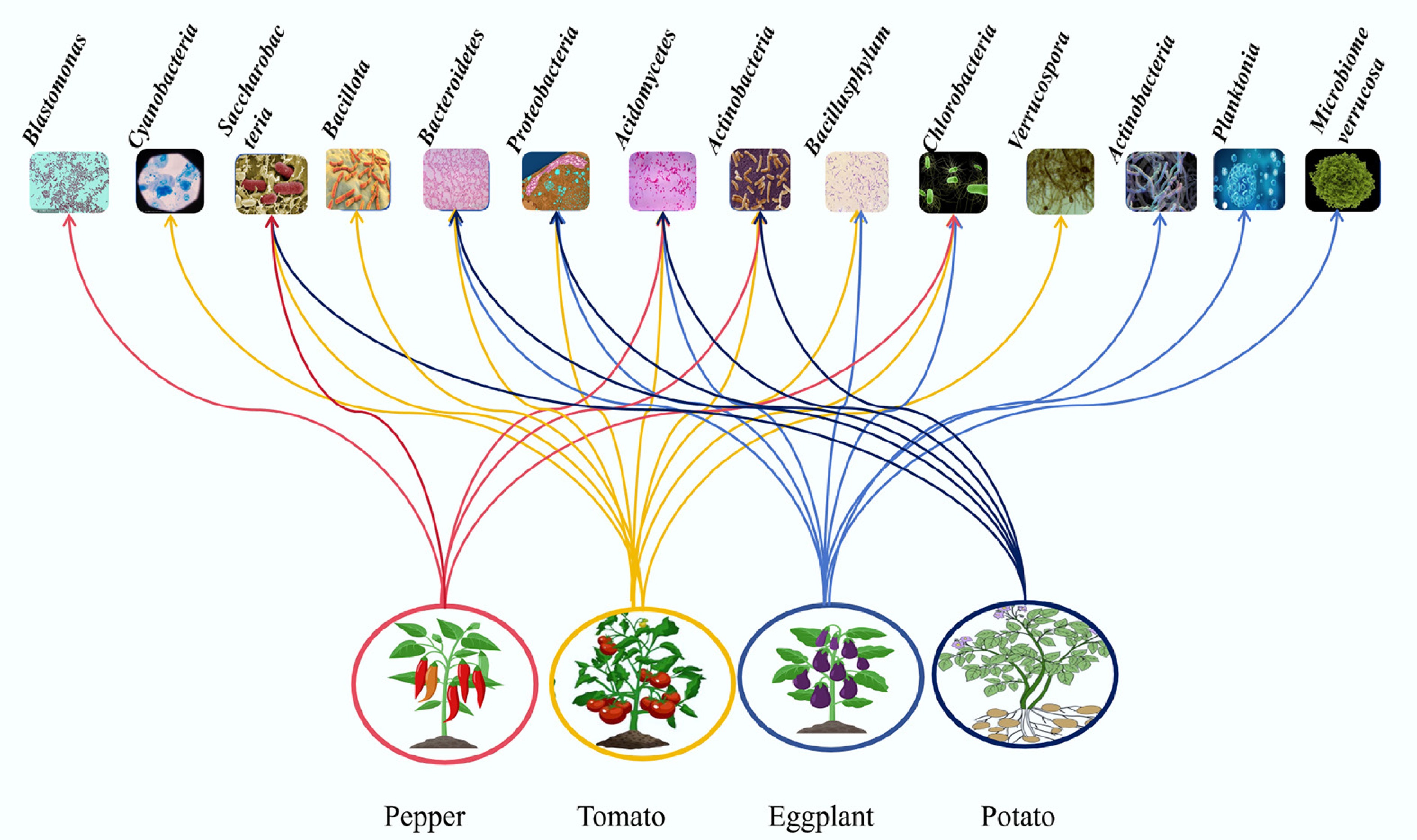
Soil microorganisms maintain soil structure and ecological balance via nutrient cycling. Continuous cropping diminishes rhizosphere microbial diversity, reduces bacterial/fungal ratios, and promotes the dominance of fungal pathogens[44−46]. Specifically, a > 4-year old potato monoculture depletes beneficial bacteria (Rhodospirillales, Rhizobiales), while enriching pathogenic fungi like Fusarium oxysporum, directly driving cropping barriers and pathogen accumulation[24,47−50].
The soil bacteria/fungi ratio reflects ecosystem health, with higher ratios indicating stronger disease resistance[51,52]. Figure 6 reveals distinct dominant phyla in rhizosphere soils across solanaceous vegetables[11,41,53−55], primarily including Acidobacteria, Actinobacteria, Proteobacteria, and Chloroflexi. Continuous cropping induces dynamic microbial shifts: potato rhizospheres exhibit species release, while tomato systems progressively enrich beneficial bacteria during long-term monoculture. Pepper cultivation over 5 years reduces Acidobacteria and Fusarium abundance[11], whereas 7-year potato rotations show annual succession: Mortierella and Rhizobium (continuous cropping for 1 year), Zygosaccharomyces (continuous cropping for 4 years), and Paecilomyces and Botrytis (continuous cropping for 7 years), with the proliferation of pathogenic Fusarium and Pseudocercospora by year 7[24]. These patterns demonstrate that continuous cropping depletes beneficial taxa, amplifies pathogens, and destabilizes root microbiota, ultimately compromising plant health.
Methods to alleviate CCOs
-
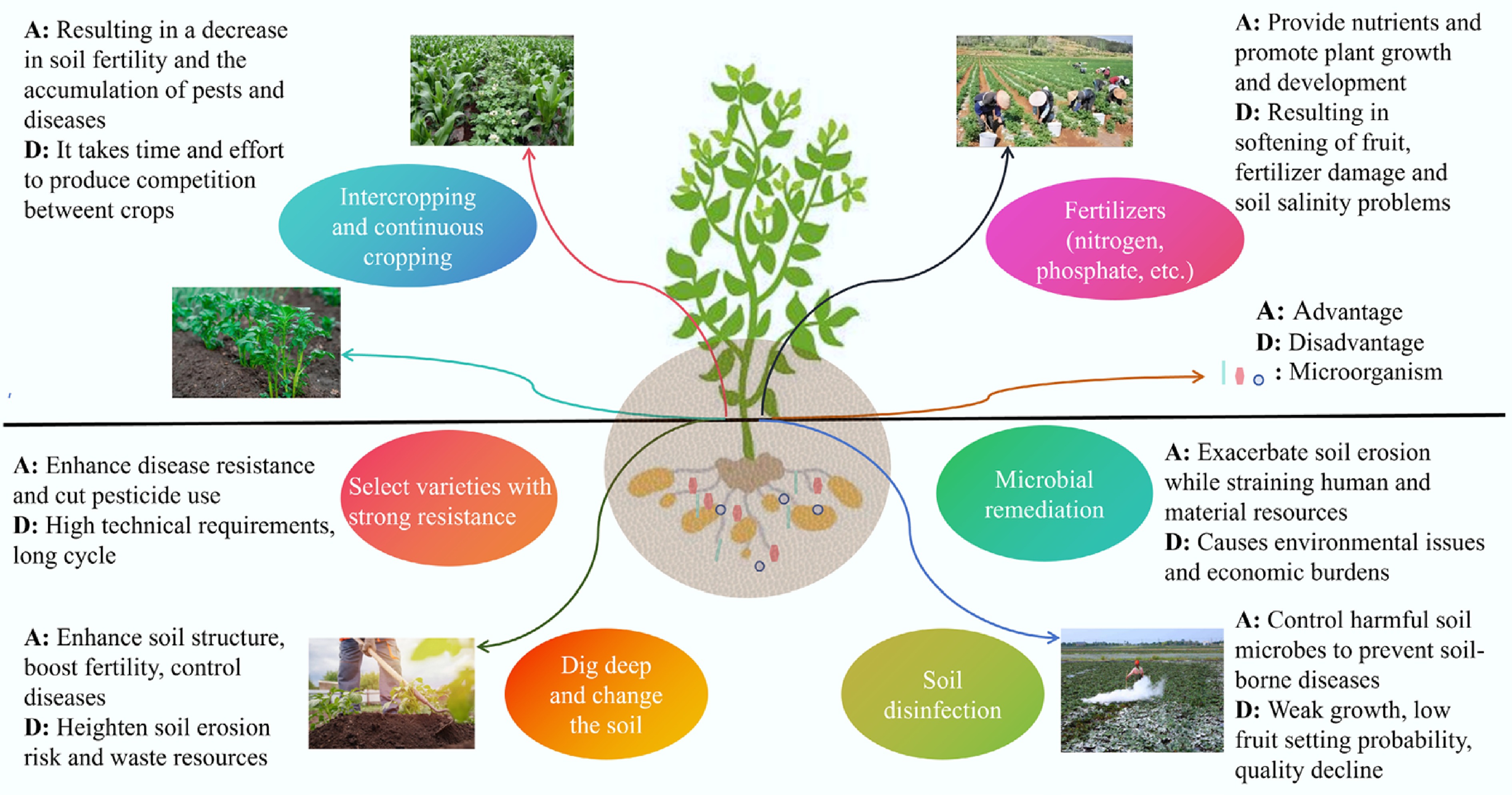
Continuous cropping has become essential for intensive agriculture due to limited arable land[3,31]. While multiple strategies address CCOs (Fig. 7; Table 2)[33,53,56−62], microbial remediation shows particular promise. Beneficial microorganisms enhance crop growth through nitrogen fixation and phytohormone synthesis[63], with corn inoculation improving both yield and nitrogen use efficiency[64]. However, microbial interventions may alter protist communities and microbiome functions[65]. Traditional soil disinfection (physical and chemical) demonstrates dual efficacy: chemical methods regulate carbon utilization and suppress pathogens[66], while quicklime remediates acidic soils. Excessive applications, however, reduce nitrogen availability and organic matter, inhibiting plant growth[67].
Fertilizers (inorganic and organic) remain predominant in agricultural management, with biochar-enhanced formulations shown to boost crop nutrient uptake and soil microbial diversity[68]. However, excessive long-term application induces environmental degradation and soil nutrient imbalances[69]. While crop rotation/intercropping effectively addresses CCOs, their implementation requires technical precision and extended timelines, along with cross-infection risks. Subsurface nutrient reserves can be mobilized through deep ploughing to replenish topsoil fertility, yet this method incurs relatively high labor and resource costs[70]. These limitations highlight the critical need for integrated strategies targeting solanaceous vegetable CCOs.
Table 2. Methods and effects of alleviating CCOs in solanaceous vegetables
Solanaceous vegetables Treatment methods Effects Ref. Potato Ridge and furrow mulching planting method Increases crop yield and promotes the proliferation of soil basidiomycete fungi. [60] Tomato Organic fertilizers Promotes the increase of organic carbon, total nitrogen, total phosphorus, and total potassium contents. [56] Tomato Microbial restoration substrate (MRS) Increases the abundances of Proteobacteria, Actinobacteria, and Bacteroidetes, while decreasing the abundances of Acidobacteria, Firmicutes, and Actinomycetes. [72] Tomato New bio-organic fertilizer Improves growth conditions and inhibits the spread of soil pathogen Fusarium wilt. [61] Eggplant Garlic stems or biological fertilizers Enhances photosynthesis and antioxidant systems, and promotes the synthesis of plant hormones and the absorption of mineral nutrients. [58] Eggplant Grafting technology Effectively controls the occurrence of bacterial wilt and increases eggplant yield. [33] Pepper Cannabis and chili pepper crop rotation Significantly reduces the incidence of chili pepper diseases. [59] Pepper Interplanting deep-rooted and shallow-rooted plants Enhances nutrient absorption and nitrogen utilization in the deep soil layer, and reduces nitrate loss. [57] -
Biochar, a carbon-rich material produced by thermochemical conversion (pyrolysis/gasification) of biomass under oxygen-limited conditions, contains abundant nutrients (K, Ca, P) and trace elements (Li, Be, V, Cr)[73]. This porous material demonstrates multifunctional environmental benefits: reducing greenhouse gas emissions, immobilizing soil contaminants (heavy metals and organic pollutants), enhancing cation exchange capacity, and alleviating CCOs[42]. During pyrolysis, biomass components (cellulose, proteins, lipids) generate surface functional groups (carboxyl, carbonyl, ketone), which enhance contaminant adsorption[74]. Furthermore, biochar-soil interactions improve nutrient availability and modify the soil microbial habitat, which plays a crucial role in suppressing soil-borne pathogens and alleviating CCOs.
Biochar has emerged as a globally recognized soil amendment for addressing continuous cropping challenges. Recent studies demonstrate its multifunctional benefits: adding 5% biochar can not only significantly increase the effective water content of sandy arid agricultural soil, but also serve as a safe soil amendment agent for adjusting the pH values of saline-alkali soil and acidic soil[75,76]. Israeli agricultural trials revealed that greenhouse waste-derived biochar enhances tomato plant growth by 63% and reduces the incidence of Fusarium crown rot[77]. Chinese investigations demonstrated biochar's effectiveness in suppressing soil-borne diseases in continuous cropping systems[78]. These beneficial effects are partly attributed to biochar-mediated improvements in soil microbial communities.
The rising demand for solanaceous vegetables has accelerated intercropping adoption. Studies demonstrate biochar's efficacy in suppressing soil-borne diseases, with soybean stalk and cassava biochar reducing the severity of tomato bacterial wilt by 30% in sandy soils[79]. Biochar amendments elevate critical soil parameters, including pH (by 0.58 units), electrical conductivity (by 101 μS/cm), and organic matter decomposition rate (by 9.48%), while also enhancing pepper fruit yield and plant height[80]. Root-knot nematode (RKN) infestations, responsible for up to 30% of vegetable yield losses, can be mitigated by a 3% rice husk biochar combined with biocontrol agents, which also stimulate tomato plant biomass and defense-related gene expression[81]. Additionally, biochar alleviates allelopathic stress by reducing benzoic acid accumulation in soybean root systems[82]. Table 3 summarizes biochar-mediated impacts on carbon cycling organisms in solanaceous cropping systems.
Table 3. Different biochars used to mitigate obstacles to the continuous cropping of solanaceous vegetables
Solanaceous vegetables Preparation of biochar raw materials Effects Ref. Tomato Olive branch Biochar concentration of 10%–15% significantly promotes the development of tomato seedlings, while enhancing its defense against systemic pathogens, including potato spindle tuber viroid and tomato spotted wilt virus. [83] Tomato Vine pruning residue (grapes) Through the improved soil treatment, plant height, leaf count, and collar diameter were over 50% greater than those in other methods, accompanied by increased soil conductivity, pH, and soluble nutrients. Furthermore, the application of biochar reduced soil bulk density by approximately 50%, enhancing root growth and subsequent water and nutrient absorption. [84] Tomato Two biochar nanoparticles were prepared from rice and corn stover Through the application of biochar, the biomass of aboveground parts and roots was enhanced, while their fresh weights increased. Additionally, it suppressed the upward transfer of sodium, thereby enhancing crops' salt tolerance. [85] Tomato Rice husk biochar Through biochar amendment, the microbial biomass C : N : P of soil increased, resulting in enhanced tomato height, stem circumference, and leaf area. [86] Eggplant Date palm, pistachio biochar Through the application of biochar, vegetative growth, yield, and water use efficiency of eggplant were enhanced. [87] Eggplant Leaf waste biochar Through the addition of leaf waste biochar (LWB) with the biocontrol agent trichoderma harzianum (BCA), plants showed higher levels of phenolics, flavonoids, and peroxidase, NPK content in eggplants significantly rose. [88] Potato Barley straw Through biochar application, cation exchange in soil, organic matter, soil pH, and crop yield were enhanced. [89] Potato Corn straw biochar Through the use of 7% biochar, plant growth was optimized, significantly reducing the metal accumulation in potato plants. Biochar enhanced the leaf growth, stem growth, and tuber growth, while mitigating the accumulation of As, Cd, Cu, and Pb. [90] Potato Pig and cow feces Biochar combined with recommended nutrients from mineral fertilizers (RNPK) significantly increased potato yield and water use efficiency. [91] Pepper Biochar Through the combined treatment of chemical fertilizer and biochar, the pepper yield significantly increased. [92] Despite significant progress in biochar application for alleviating intercropping disorders, critical limitations persist. Key challenges include: (1) material specificity—high-ash livestock manure-derived biochar (> 40% ash) risks soil salinization; (2) application thresholds—excessive dosage (> 50 t/ha) induces abnormal soil porosity; (3) temporal effects—prolonged application (> 3 years) may trigger secondary disorders like phosphorus fixation. Current understanding remains incomplete regarding the biochar-soil-microbe interaction network and the molecular mechanisms suppressing soil-borne pathogens[81]. Notably, pyrolysis temperature variations (300–700 °C) induce distinct rhizosphere microbiome modifications, yet their specific regulatory pathways remain undefined.
-
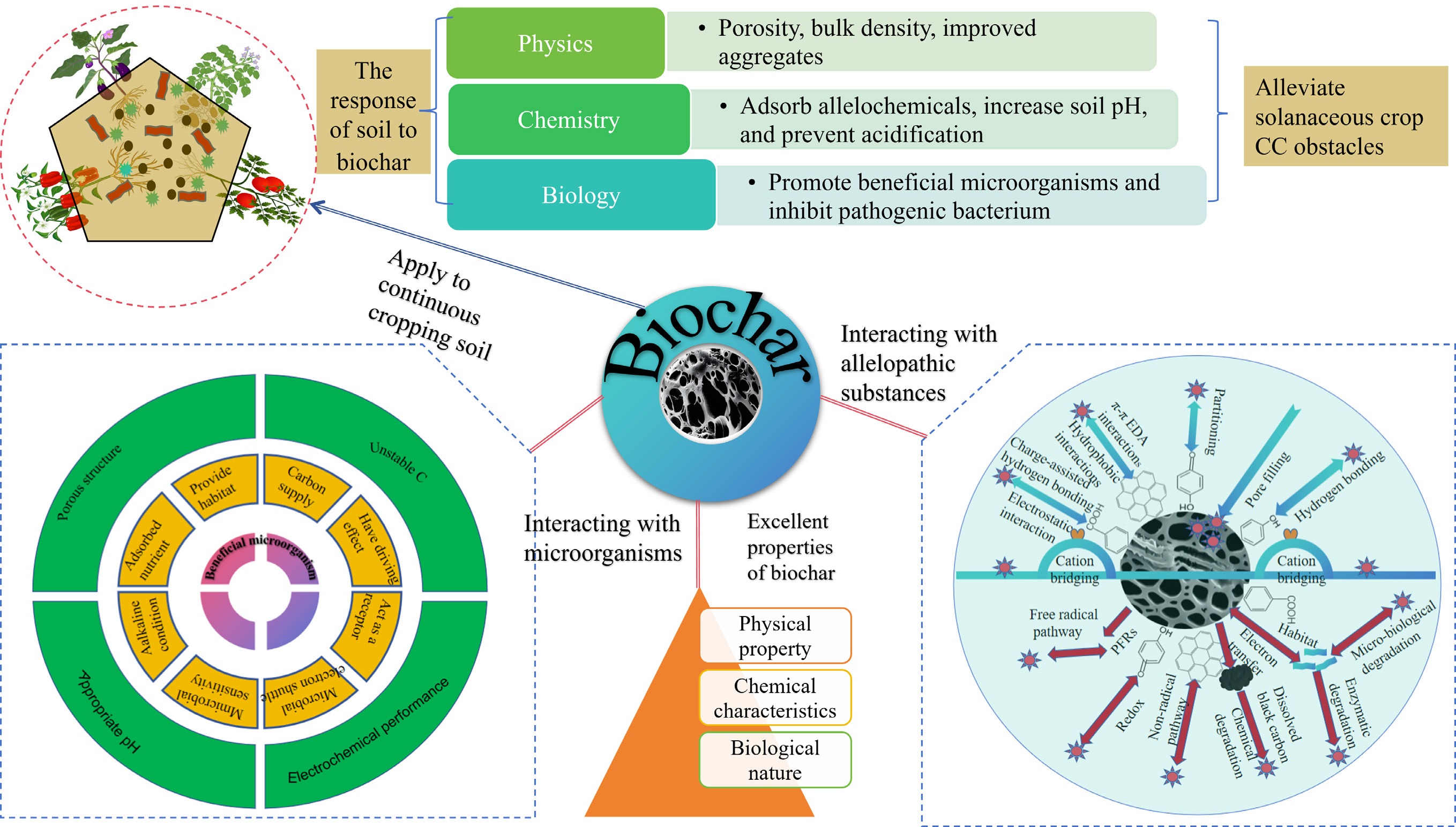
Biochar has been used to treat CCOs in solanaceous vegetables because of its unique advantages (such as its porosity, high adsorption capacity, and abundance of minerals and organic matter) (Fig. 8). Biochar alleviates CCOs in solanaceous vegetables through multiple interconnected mechanisms, primarily via improving soil physicochemical properties, adsorbing and degrading allelopathic substances, and modulating the soil microbial community (Fig. 8). Its porous structure, high adsorption capacity, and richness in minerals and organic matter provide the foundation for these functions[9]. Research has shown that adding biochar to soil can reduce the production and release of benzoic acid from soybean roots and has a positive effect on alleviating allelopathic effects[93]. The excellent characteristics of biochar lay the foundation for mitigating CCOs in solanaceous vegetables. Therefore, the application of biochar is a new pathway for alleviating CCOs in solanaceous vegetables (Table 3). The specific mechanisms are shown in Fig. 8.
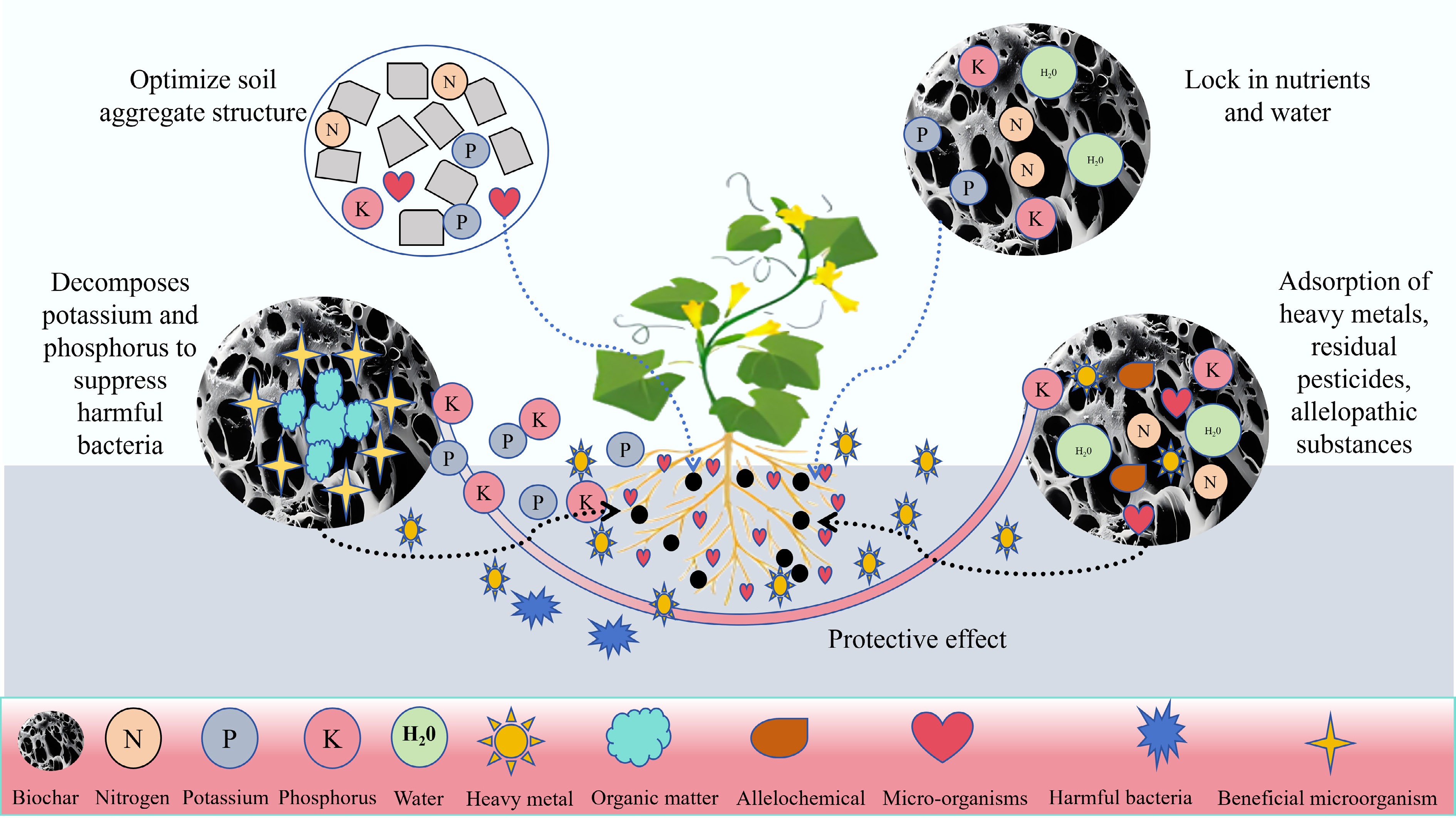
Figure 8.
Mechanistic diagram of the recovery of solanaceous vegetables from CCOs by biochar treatment[9].
Effects of biochar on soil physicochemical properties
-
Previous studies have shown that biochar positively affects soil organic carbon, electrical conductivity, and water content[94,95] (Fig. 8). It can effectively adjust the structural distribution of soil aggregates and optimize soil structure. Due to its high porosity, biochar slows water evaporation from farmland soil. This extends the time for soil moisture to decline to field capacity and enhances the upward flow of deep soil water at night[96]. Research indicates that biochar can significantly improve soil carbon pools and promote the formation of favorable soil aggregates in dryland cropping systems[97]. It is particularly effective for sandy soil improvement. Compared to unamended soil, biochar application increases the available water capacity of sandy soil by an average of 28.5%. Hydraulic conductivity and saturated water content are also significantly enhanced[98]. Studies further show that biochar-soil interaction benefits acidic sandy soils and soil biota[99], and can significantly increase soil nutrient levels. In water-scarce regions, biochar addition can effectively raise soil temperature and improve its insulation properties, thereby enhancing crop stress resistance[100]. Biochar also enriches soil organic matter, retains nutrients, and provides a material basis for organism growth and reproduction[101]. Its hydrophilic nature helps retain soil moisture, significantly reducing evaporation and water loss[102].
Biochar has good adsorption properties due to its high charge density on its surface and easily forms a magnetic field, which can effectively absorb soil nutrients and reduce nutrient loss[103]. In general, biochar has a high adsorption potential for organic matter[104]. Another study further demonstrated that biochar can effectively address key challenges in solar greenhouse agriculture by ameliorating degraded soil. It significantly improves soil structure by reducing bulk density and increasing water-holding capacity, which in turn enhances crop photosynthesis. Additionally, biochar application promotes tomato growth and development by creating a more favorable rhizosphere environment, leading to improved fruit appearance and mitigation of CCOs. Therefore, biochar can improve the physicochemical properties of soil and alleviate CCOs in solanaceous vegetables.
Biochar adsorption and degradation mechanism of allelopathic substances
-
Allelopathic substances are secreted by the rhizosphere of plants. After decomposition, plant residues penetrate the soil growth system through rain and dew, causing adverse effects on crops. Continuous cropping leads to a large accumulation of allelopathic substances in the soil of solanaceous vegetables[14]. Cinnamic acid and benzoic acid are the main allelopathic autotoxic substances in tomatoes. Long-term continuous cropping causes harm to plant cell lipids, proteins, and nucleic acids. It can affect the activity of plant photosynthesis-inhibitory enzymes and reduce the length and biomass of roots and stems[105]. After ten years of field experiments, vanillin is the main allelopathic substance causing potato CCOs, and its inhibitory effect is more obvious than that of ferulic acid and p-hydroxybenzoic acid. Vanillin reduces the concentration of indole-3-acetic acid in potato roots and inhibits the formation of adventitious roots, ultimately leading to a decrease in crop yield[34]. Biochar effectively adsorbs and exchanges ions, lowering allelopathic substance levels in continuously cropped soil, increasing the abundance of beneficial bacterial groups (Actinobacteria) and changing the fungal community. Reductions were observed in Ascomycota and Aspergillus, accompanied by changes in mould abundance[17]. In the presence of the allelochemical dimethyl phthalate, the addition of biochar has a positive effect on the germination of tomato plants[106]. Biochar adsorbs soil-free allelochemicals (such as hydroxy acids, benzoic acids, n-alkanoic acids, acetoxy acids, diols, triols, and phenols) released from the rhizosphere of peppers and tomatoes in cultivation media, thereby promoting the growth of beneficial microorganisms and increasing plant disease resistance, which ultimately improves crop yield[107]. The efficacy and mechanisms of such adsorption can be quantitatively elucidated through specific adsorption models and kinetics. A representative case study on alleviating celery (Apium graveolens) CCOs demonstrated that phosphoric acid (H3PO4)-modified wood biochar (WP400) exhibited superior adsorption of phenolic allelopathic substances such as gallic and ferulic acids. The adsorption isotherm followed the Langmuir model (R2 > 0.97), indicating monolayer chemical adsorption with a maximum capacity (Qmax) of 106–120 mg/g at pH 3–4. Kinetics adhered to the pseudo-second-order model (R2 ≈ 1.0), confirming chemisorption-controlled rates. The high performance was attributed to an exceptionally high specific surface area (1,273 m2/g) and oxygen-containing functional groups introduced by modification, which facilitated adsorption via hydrogen bonding and π-π interactions. Notably, adsorption was highly pH-dependent, with optimal capacity under acidic conditions due to reduced electrostatic repulsion and enhanced π-π donor-acceptor interactions. Pot experiments validated that 0.3% (w/w) WP400 reduced water-soluble phenolic compounds by > 40% in soil and significantly promoted celery growth, directly linking high adsorption capacity to alleviated allelopathy in practice[82].
The various traits of allelopathic substances (such as hydrophobicity, polarity, and molecular size) influence biochar adsorption. Biochar has a greater tendency to adsorb hydrophilic allelopathic substances, whereas hydrophobic substances are attracted via weak hydrophobic effects or van der Waals forces, resulting in different sizes of allelochemical molecules remaining at different positions after being adsorbed[108−110]. In addition, the phenolic hydroxyl groups of biochar and allelopathic substances are prone to redox reactions, which promote the decomposition of allelopathic substances and are beneficial to crop growth, as shown in Fig. 9[111]. The application of biochar to adsorb and degrade allelopathic substances has become an effective method for alleviating CCOs in solanaceous vegetables. As shown in Fig. 9, the mechanism by which biochar decreases allelopathic substances can be roughly divided into two processes: (1) Adsorption process: oxygen-containing functional groups (OFGs) on biochar can form an electron donor‒acceptor complex (EDA) through a binary or ternary adsorption system to adsorb organic substances[112]. Allelochemical and biochar structures feature carboxyl, hydroxyl, aromatic, and quinone groups, some of which can form binary complexes. Hydrogen bonding, including charge-assisted hydrogen bonding, as well as hydrophobic effects and partitioning, potentially aid in allelochemical adsorption on biochar[113,114]. Research has indicated that cation bridges between negative allelochemicals and biochar can increase adsorption[115]. (2) Degradation process: the biochar-mediated degradation pathways of organic pollutants can be divided into free radical pathways and nonradical pathways[111,116]. The primary contributors to the free radical pathway are environmentally persistent free radicals (EPFRs), hydroxyl radicals (•OH), and superoxide radicals (•O2−)[117]. EPFRs on biochar can degrade organic pollutants after direct contact or induce water or oxygen to produce •OH or •O2− to degrade organic pollutants[118,119]. Many allelopathic substances contain abundant phenolic hydroxyl groups, which readily undergo redox reactions with biochar[120]. This process facilitates the transformation and biological conversion of chemical substances[111].
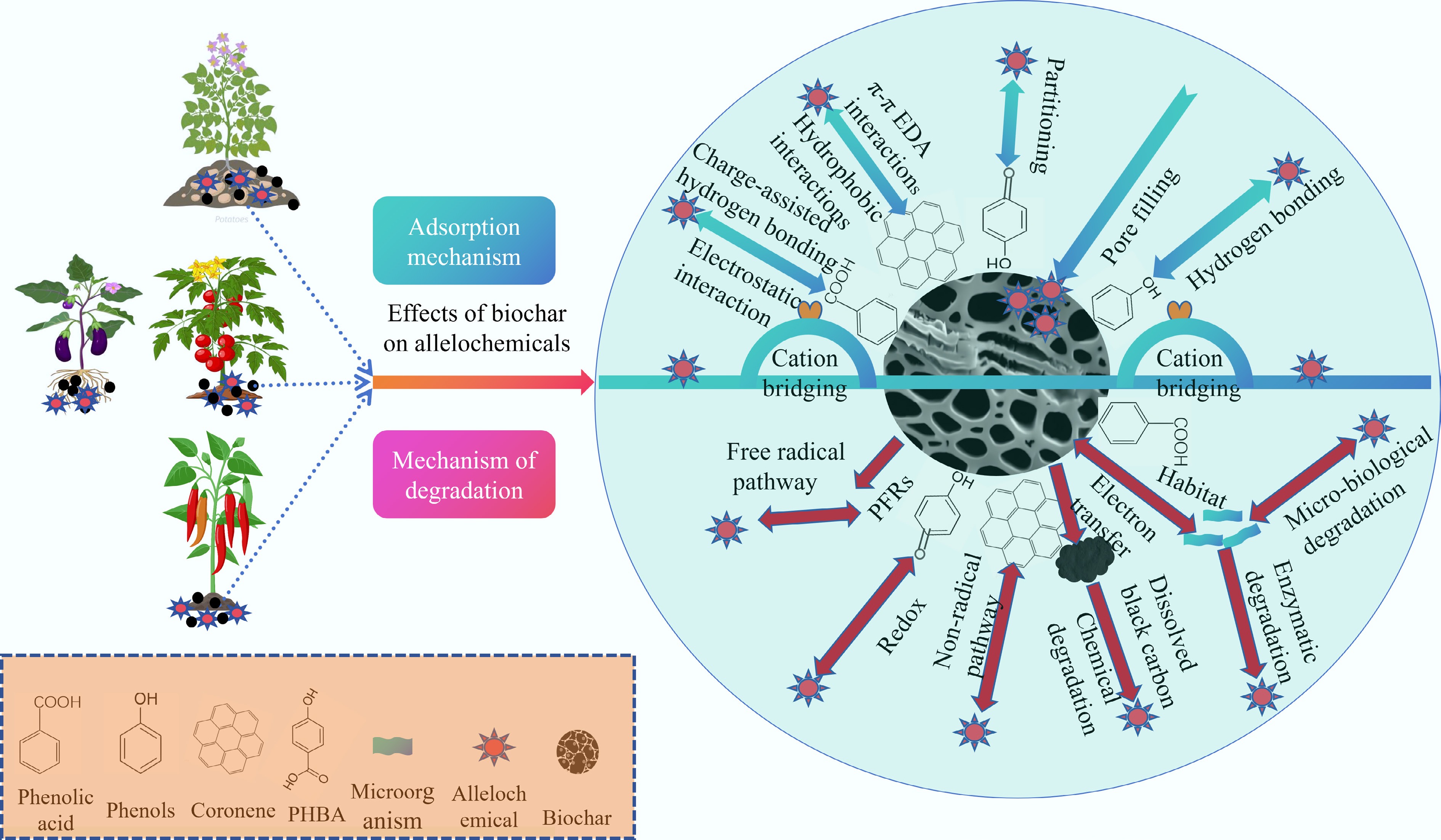
Figure 9.
Adsorption and degradation mechanisms of allelopathy by biochar[111].
Impact mechanism of biochar on the soil microbial flora
-
Enhancing the soil microenvironment is key to overcoming CCOs and improving the yield and quality of solanaceous vegetables. Biochar can improve soil microbial ecology and increase soil microbial abundance and diversity[65, 121] (Fig. 10). Jaiswal et al. reported that biochar promoted the growth of Pseudomonas fluorescens, Actinobacteria, and Trichoderma[122]. For instance, Jaiswal et al. reported that greenhouse waste-derived biochar conferred systemic resistance against Fusarium crown rot in tomato by modulating the rhizosphere microbiome[77]. Similarly, Ge et al. employed ITS rRNA sequencing to demonstrate that biochar application significantly reduced the abundance of pathogenic fungi (e.g., Trichoderma and Monosporangium spp.) in continuous tobacco cropping systems[78]. These effects are facilitated by biochar's creation of microhabitats that selectively stimulate beneficial microbiota and suppress phytopathogens. Moreover, Pseudomonas putida, Pseudomonas koreensis, Pseudomonas moraviensis and Pseudomonas monteilii have been shown to exhibit up to 72% antagonistic activity against root rot pathogens while inhibiting mycelial growth, thus suppressing the occurrence of soil-borne diseases in tomato. After the application of biochar, the dominant fungi in the eggplant rhizosphere (Humicola and Chaetomium, Mortierella, and Tausonia) increased in number and suppressed the occurrence of diseases. Biochar application enriches the rhizosphere with the bacterial genus Bacillus and promotes dominant fungal taxa, including Humicola, Chaetomium, Mortierella, and the yeast Tausonia, which collectively contribute to disease suppression[123]. Biochar increases the number of actinomycetes in the rhizosphere (such as those belonging to Micrococcaceae, Nocardioides, Janibacter, Corynebacteriaceae, and Terrabacter) and Alphaproteobacteria (such as Caulobacter and Qipengyuania), thereby increasing the release of refractory organic carbon and improving soil fertility[124−126]. Biochar can increase the number of beneficial functional microorganisms and metabolic pathways related to energy utilization efficiency.
Biochar alters plant rhizosphere microbe populations, generating beneficial bacteria while inhibiting pathogens. This enhances plant growth, increases disease resistance, and increases crop yields in consecutive planting cycles[127]. The application of biochar to the rhizosphere soil of continuously cropped cucumbers can increase the abundance of beneficial bacterial groups in the soil, such as Firmicutes (gram-positive bacteria), Actinobacteria (actinomycetes), and Bacillus (bacillus), and by altering the pathogenic fungal community (with reduced abundance of Aspergillus and Penicillium), the crop yield can be significantly improved[128]. Graber et al. reported that the addition of biochar promoted the development of microbial populations (for example, Pseudomonas, Mesorhizobium, Microbacterium, Brevibacillus, Bacillus, and Streptomyces) that are beneficial for the growth of tomato and pepper crops[107]. In addition, biochar can increase the number of potentially beneficial organisms (Pseudeurotium and Solirubrobacter) and reduce the number of potential pathogenic organisms (Kribbella and Ilyonectria) while increasing the transformation capacity of microorganisms and improving the ability to convert root nutrients (N, P, K, etc.) to increase crop yields[129,130]. Beyond altering the microbial community composition, biochar fundamentally reshapes the functional potential of the soil microbiome by modulating the abundance of key functional genes and metabolic pathways. Specifically, biochar amendment significantly enriches functional genes critical to soil nutrient cycling. For phosphorus cycling, biochar (especially produced at 350 °C) markedly increases the abundance of the phoD gene, which encodes alkaline phosphatase, thereby enhancing the transformation and availability of soil phosphorus[48]. For nitrogen cycling, biochar application elevates the abundance of genes involved in nitrification (AOA-amoA, AOB-amoA), denitrification (narG, nirS, nirK), and N2O reduction (nosZ), collectively accelerating nitrogen mineralization and nitrification processes, while potentially mitigating N2O emissions[92]. Metabolomic analyses further reveal that biochar addition reprograms soil metabolic pathways. Long-term field studies indicate that biochar triggers the upregulation of differential metabolites and enriches metabolic pathways such as oxidative phosphorylation and benzoxazinoid biosynthesis, which are closely linked to energy metabolism and plant defense[20]. Concurrently, biochar fosters the proliferation of key functional microbial taxa, such as Nitrospira, involved in nitrification, enhancing the soil nitrification capacity[20]. These synergistic shifts—in functional gene abundance, microbial taxa, and metabolic pathway activity—equip the soil microbiome with enhanced capabilities for nutrient transformation, disease suppression, and stress alleviation. This provides a mechanistic bridge between biochar-induced microbial community changes and the observed alleviation of CCOs. Biochar can significantly improve the diversity and structure of the soil microbial communities of solanaceous vegetables, adjust the proportion of beneficial and harmful bacteria, and thereby promote the growth and yield of vegetables.
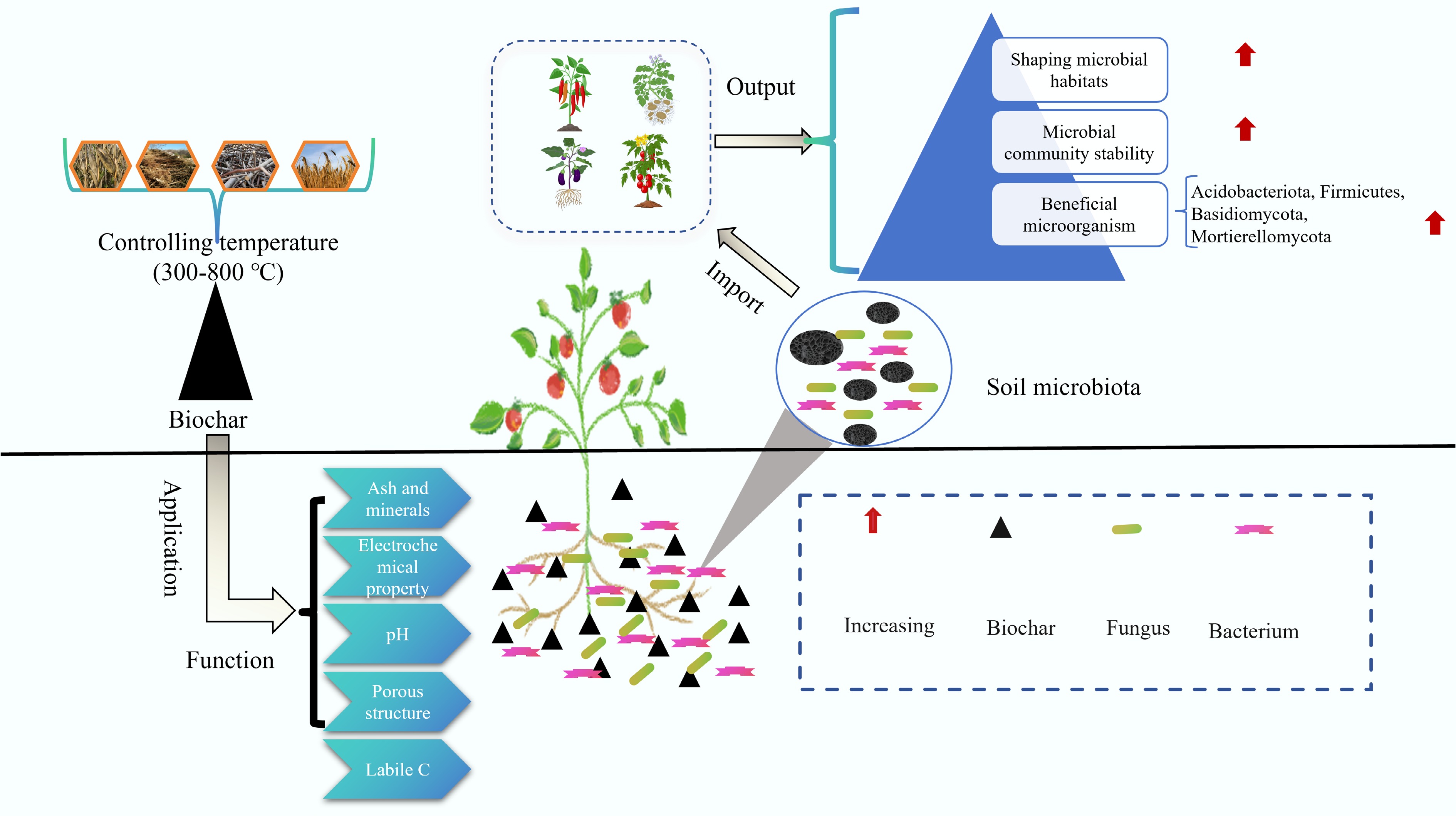
Figure 10.
Effects of biochar on soil microbial communities[131].
-
In this review, the latest research progress of biochar in alleviating CCOs in solanaceous vegetables is discussed in terms of the changes in the way of alleviating CCOs, the improvement of soil quality, the transformation of microbial community structure, and the adsorption of allelopathic toxic substances. Additionally, the adsorption mechanism of biochar as an allelopathic substance adsorbent is explored. This includes two application processes: adsorption and degradation, and emphasizes the specific mechanism of biochar in degrading allelopathic substances. Biochar provides excellent prospects for future development in alleviating CCOs in crops. However, its application is constrained by some inherent limitations, such as low nutrient supply efficiency, limited adsorption capacity, and potential risks, including polycyclic aromatic hydrocarbon pollution, soil alkalization, and micro-ecological imbalance. To address these shortcomings and guide future research, the following key directions are proposed.
Biochar-microbe-fertilizer synergy: interfacial mechanisms
-
Future work must elucidate the physicochemical and biological interactions at biochar-mineral-microbe-organic matter interfaces. Key foci include the co-assembly of biochar pores with organic amendments for controlled nutrient release, the colonization and signaling of functional microbes on biochar surfaces, and the use of isotope tracing and multi-omics to map element fluxes and metabolic networks. This will inform the design of integrated 'carrier-nutrient-consortium' soil amendments.
Aboveground VOC-mediated allelopathy
-
Research should expand from soil-based allelopathy to aerial interactions mediated by plant volatile organic compounds (VOCs). Employing volatilomics, isotopic labeling, and canopy diffusion models can quantify how biochar alters VOC profiles in solanaceous crops and their ecological effects. Mechanistic studies should reveal the phytohormonal pathways involved, advancing the theory of coordinated above-belowground interference for novel ecological management strategies.
Long-term risk and resilience assessment
-
A robust framework is needed to assess the sustainability of long-term use of biochar. This requires modeling pollutant fate (e.g., free radicals, PAHs) across soil-plant-water interfaces, diagnosing microbial network stability to identify vulnerability thresholds, and evaluating trade-offs in soil carbon sequestration, nitrogen cycling, and greenhouse gas emissions. Such an approach would define safe application thresholds and support science-based management.
-
The authors confirm their contributions to the paper as follows: all authors contributed to the conception and design of this study. Zengquan Luo and Wenxuan Quan: material preparation, analysis, and original draft writing. Anping Wang: revising, writing − review. Chaochan Li and Bing Wang: conceptualization, writing − original draft, supervision, manuscript revision and review. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
-
This work was supported by the Project of Guizhou Provincial Department of Science and Technology [Grant Nos. QKHPTRC-GCC(2023)067, QKHCG(2023)YB541, QKHZC(2023)YB078, and QKH-ZK(2024)YB645], the Science and Technology Program of Guizhou Provincial Branch of the CNTC [Grant No. 2023XM16], the Project of Guizhou Education Bureau [Grant No. QJJ(2024)324], and the Project of Guizhou Forestry Bureau [Grant No. GUI(2022)TSLY07].
-
The authors declare that they have no conflict of interest.
-
Full list of author information is available at the end of the article.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Luo Z, Wang A, Quan W, Li C, Wang B. 2026. Application of biochar for the prevention and control of soil continuous cropping obstacles in solanaceous vegetables: a review. Biochar X 2: e013 doi: 10.48130/bchax-0026-0012
Application of biochar for the prevention and control of soil continuous cropping obstacles in solanaceous vegetables: a review
- Received: 18 January 2026
- Revised: 02 March 2026
- Accepted: 20 March 2026
- Published online: 03 April 2026
Abstract: Continuous cropping obstacles (CCOs) pose severe threats to the quality and yield of solanaceous vegetables. Biochar has emerged as an effective soil amendment to mitigate CCOs due to its nutrient-rich composition and unique structural properties. However, its performance can be affected by feedstock types, pyrolysis temperature, soil conditions, and crop species, resulting in inconsistent outcomes in the field. Thus, a review on the effect and mechanism of biochar in the prevention and control of CCOs of solanaceous vegetables is highly needed. This is the first systematic review focusing on the mechanism of biochar in alleviating CCOs in solanaceous vegetables. This review systematically delineates the causes and detrimental impacts of CCOs across major solanaceous crops (tomatoes, eggplants, peppers, and potatoes), and comprehensively evaluates biochar-based soil management strategies in comparison with conventional agronomic, physical, and chemical control measures. The underlying specific mechanisms of biochar in alleviating CCOs are explored, including improving soil properties and soil microbial community structure, as well as alleviating allelopathic autotoxicity in solanaceous plants. Based on the current research status and the nature of biochar, future research should focus on exploring the microscopic reaction mechanism and long-term effects of biochar and its composites on alleviating CCOs. In addition, further research on the allelopathic effects of biochar on the aboveground parts of crops should also be considered. This review provides a foundational reference for the use of biochar to combat CCOs in solanaceous vegetables.