-
Sweet olive (Osmanthus fragrans Lour.) is a woody, evergreen shrub or small tree in the Oleaceae family[1]. It can reach heights of 7.62 m but more commonly observed at 3.05−3.66 m high and can be grown in United States Department of Agriculture (USDA) hardiness zones 7b to 9b[2]. Osmanthus fragrans has been extensively cultivated for over 2,500 years in China, where it is regarded as one of the most eminent flowers and is deeply associated with traditional Chinese culture[3]. Besides its aesthetic attributes, O. fragrans is highly valued for its strong fragrance. This characteristic has been widely used in tea, food, spices, wine, skincare products, and cosmetics[4]. Additionally, Chinese medicine uses the organs of O. fragrans, including the roots, branches, flowers, and fruits, as a prescription to relieve dampness and cold[5].
More than 170 registered cultivars of O. fragrans have been classified into four groups on the basis of their flower color and blooming season: (1) The Yingui group (albus group) has white to pale yellow flowers that bloom only in autumn once, (2) the Jingui group (luteus group) has yellow flowers that bloom once in autumn, (3) the Dangui group (aurantiacus group) has orange flowers that bloom once in autumn, and (4) the Sijigui group (asiaticus group) has pale yellow to yellow flowers that bloom several times in different seasons except summer[1,4,6,7]. Osmanthus fragrans 'Beni Kin Mokusei' is a cultivar in the aurantiacus group, and the name means "red flowering osmanthus" in Japanese[8]. Over the decades, it has been regarded as the holy grail among Osmanthus collectors because of its red flowers. Its flower color is much darker orange-red than the old forms of O. fragrans var. aurantiacus. Additionally, O. fragrans 'Beni Kin Mokusei' has high ornamental value with its compact and dense growth habit[8].
Stem cutting is the most commonly used method for propagating woody ornamental plants because it overcomes long propagation periods and the low success rate of seed propagation[9,10]. However, the success of cutting propagation heavily depends on the cutting types and when the cutting is taken[11], as cuttings' phenological stage and the climatic conditions significantly affect the rooting ability of woody cuttings[12]. For instance, softwood cuttings of farkleberry (Vaccinium arboreum) had a significantly higher rooting percentage (38.60%) and the most roots per cutting (2.20) compared with semi-hardwood (9.20% and 1.60, respectively) and hardwood cuttings (10.60% and 1.90, respectively) under the same treatment conditions[13]. Additionally, hardwood cuttings of fig (Ficus carica) had a significantly higher rooting percentage (65.02%), more roots per cutting (8.80), and longer roots (7.16 cm) compared with the semi-hardwood cuttings (50.67%, 6.57, and 5.66 cm, respectively) under the same treatment conditions[14].
The success of cutting propagation is also influenced by plant growth regulators (PGRs). PGRs are also known as synthetic plant hormones, which affect rooting percentage and length in cutting propagation[11]. Commercial nursery growers have been using different PGRs to initiate root formation for many years[11,15]. Indole-3-butyric acid (IBA) is one of the most important PGRs for promoting root formation. However, IBA is insoluble in water, so growers have tested different formulations of IBA to promote propagation success[16].
IBA powder, a dry talc product in which IBA has been chemically affixed to talc particles, is a commonly used formulation in commercial propagation and has been shown to significantly enhance root initiation[16]. For example, IBA powder significantly improved the rooting percentage of Chinese native holly (Ilex angulata) cuttings from 25.00% (water dipping) to 66.70% (1,000 mg/L) and 50.00% (3,000 mg/L), as well as increasing the root ball volume from 8.00 cm3 (water dipping) to 107.20 cm3 (1,000 mg/L) and 33.80 cm3 (3,000 mg/L)[17].
Indole-3-butyric Acid potassium salt (K-IBA) is a water-soluble potassium (K) salt of IBA commonly used in liquid applications to ensure absorption and control the precise concentration, improving propagation rates[16]. For instance, in a Chinese native holly (Ilex angulata) propagation study, cuttings treated with 1,000 mg/L K-IBA had a 75.00% rooting percentage and a root ball volume of 118.20 cm3, which was significantly higher than those of water dipping (25.00% and 8.00 cm3, respectively)[17]. In addition, Japanese zelkova (Zelkova serrata) softwood cuttings treated with 1,000 mg/L K-IBA had a higher rooting percentage (31.30%) and longer roots (8.40 cm) compared with water dipping (6.30% and 0.30 cm, respectively)[18].
IBA gel, formulated with viscous auxin solutions and a thickening agent typically derived from cellulose, provides better adhesion to the surface of a cutting[16]. The enhanced adhesion improves hormone absorption at the wound area, reduces leaching, and promotes root initiation, resulting in better rooting. In a study of hybrid tea rose (Rosa × hybrida) semi-hardwood cutting propagation, IBA gel significantly increased the number of roots from 9.70 (water dipping) to 16.30 (IBA gel) and increased root dry weight from 0.12 g (water dipping) to 0.19 g (IBA gel)[19]. Similarly, softwood cuttings of dogwood [(Cornus florida × kousa) × florida] treated with 3,100 mg/L IBA gel had a higher rooting percentage (88.10%) compared with water dipping (40.50%)[20].
For Osmanthus species, the IBA formulation used in previous studies was an ethanol-dissolved IBA solution, which can significantly improve the success of cutting propagation of O. fragrans. Zou et al.[21] reported that softwood cuttings of O. fragrans (collected in May) treated with a 100 mg/L IBA solution significantly increased the rooting percentage to 58.43% compared with the water dipped control (11.10%). Additionally, semi-hardwood cuttings (collected in September) and treated with a 1,500 mg/L IBA solution achieved an 86.40% rooting percentage[22].
Similarly, thecutting type also affects the propagation success of Osmanthus species under IBA treatment. Untreated semi-hardwood and hardwood cuttings of O. heterophyllus 'Ilicifolius' had no significant difference in rooting percentages (91.70% and 80.60%, respectively)[23]. However, when treated with a 10,000 mg/L IBA solution (dissolving reagent-grade IBA in 50% isopropyl alcohol), semi-hardwood cuttings had a much higher rooting percentage (77.80%) than hardwood cuttings (52.80%)[23].
However, there are few relevant studies that have evaluated the effects of different formulations and rates of IBA on propagating O. fragrans by cuttings, not to mention the effects of different types of cuttings (semi-hardwood and hardwood). Additionally, the commonly used ethanol-dissolved IBA solution concentration can be changed by the evaporation of ethanol, and commercial products in the United States contain 1-Naphthaleneacetic acid (NAA)[16]. Therefore, this study aimed to identify the most effective and stable formulation and rates of IBA, as well as the optimal cutting type for propagating O. fragrans by stem cuttings, thereby providing growers with production efficiency for the future propagation of O. fragrans.
-
Osmanthus fragrans 'Beni Kin Mokusei' semi-hardwood cuttings were collected from a mature stock plant on 17 August 2024, and hardwood cuttings were collected on 12 October 2024 from the University of Georgia Horticulture Farm (Watkinsville, GA, USA). Semi-hardwood cuttings were partly lignified, easy to cut, and had a lighter gray stem color. In contrast, hardwood cuttings were fully lignified, hard to cut or bend, and the wood was firm. The cuttings remained with three nodes and the top five leaves, measuring around 7–10 cm long. Half of the leaf area was removed to decrease the transpiration rate. The basal end was wounded by hand pruners at an angle to increase the surface area.
IBA materials and treatments
-
Stems were treated with a K-IBA solution prepared by using K-IBA powder (K-IBA, AFG Bioscience, College Station, TX, USA) dissolved in water to 1,000, 3,000, and 8,000 mg/L); IBA powder [Hormodin #1 (1,000 mg/L), Hormodin #2 (3,000 mg/L), and Hormodin #3 (8,000 mg/L), OHP, Inc., Mainland, PA, USA]; or IBA rooting gel (Clonex® Rooting Gel, HydroDynamics International, Lansing, MI, USA) at 3,100 mg/L. Cuttings dipped in tap water for 10 s were used as the control.
For the liquid treatments, the basal ends of the cuttings were immersed in the liquid for 10 s, and each treated cutting was air-dried for at least 10 min before being inserted into the growing medium. For IBA powder and gel treatments, cuttings were dipped into water for 1 s, then into the IBA powder or gel, and lightly tapped to remove excess powder or gel.
After the IBA treatment, each cutting was inserted into a square pot (6.35 × 6.35 × 7.62 cm) filled with a mix of perlite (Garden Horticulture Perlite; Whittemore Company, Inc., Lawrence, MA, USA) and growing mix (PRO-MIX® BK45·V; Premier Tech Horticulture, Rivière-du-Loup, QC, Canada) thoroughly blended at a ratio of 1:1 (by volume). Cuttings were inserted to a depth of 3 cm. Each pot was placed into a single cell of a 32-cell carry tray (Fig. 1).

Figure 1.
Photo of (a) semi-hardwood and (b) hardwood cutting blocks taken 14 weeks after the cuttings were struck.
Experimental environment
-
The cuttings were placed on a bench under 60% shading cloth and intermittent mist in a greenhouse at the University of Georgia Horticulture Farm (33.886971° N, 83.420468° W; USDA Zone 8a) under conditions of 21.11/32.22 °C night/day temperature and an 11.15-h daylength. The intermittent mist operated for 6 s every 6 min in Week 0, 6 s every 10 m in Week 1, 6 s every 20 min in Week 2, and 6 s every 30 min from Week 3 to Week 14.
Measurements
-
On 26 November 2024 and 21 January 2025 (Week 14 for the semi-hardwood and hardwood cuttings, respectively), the survival rate (the percentage of live cuttings), callus rate (the percentage of cuttings with callus tissue), rooting percentage (the percentage of cuttings with roots), number of roots per cutting, total root length (cm), average root length (cm), longest root length (cm), and root index were recorded. All root lengths were measured using a ruler. A cutting with a root tip greater than 1 mm was counted as having a root, and the root lengths of unrooted cuttings were considered to be 0 cm (Fig. 2). Additionally, eight cuttings from each treatment per block were collected, and cutting biomass (g) was recorded after the cuttings were dried in an oven at 80 °C for 3 d.
$ \mathrm{Root\; index=Root\; number\times Total\; root\; length} $ 
Figure 2.
Photos of semi-hardwood (a) and hardwood (b) cuttings taken 14 weeks after the cuttings were struck.
Experimental design and data analysis
-
For each type of cutting, the experimental design was a randomized complete block design (RCBD) with four blocks. Each block consisted of two 32-cell trays placed on a bench. Each tray included four treatments with eight replicates. Cuttings were randomly collected from plant materials and treated with treatments in a randomized process. The treated cuttings were randomly assigned to one of four blocks. Data were analyzed by generalized linear mixed models (GLMMs) using R software (version 4.3.1; RStudio, Boston, MA, USA). Binomial GLMM was used for percentage data, Poisson GLMM was used for root number, and log-Gaussian linear mixed models were used for root length, index, and biomass. Block was included as a random effect in the models. For root length data, zero values were log-transformed as log(x + 0.001) before analysis. Likelihood ratio tests were used to test the significance of model terms by comparing nested models among treatments. Post hoc pairwise comparisons were performed using the estimated marginal means with Tukey adjustment (emmeans package).
-
For both semi-hardwood and hardwood cuttings, IBA treatment significantly affected the rooting percentage, callus rate, root number per cutting, total root length, average root length, longest root length, root index, and biomass. In addition, IBA treatment significantly affected the semi-hardwood cuttings' survival rate (p = 0.036) but not those of hardwood cuttings (Table 1).
Table 1. GLMM results for survival rate (%), rooting percentage (%), callus rate (%), root number per cutting, total root length (cm), average root length (cm), longest root (cm), root index, and biomass (g) of semi-hardwood (Semi) and hardwood (Hard) cuttings as influenced by the IBA treatments.
Cutting type Parameter Unit $\chi^2 $ df p-Value Semi-hardwood Survival rate % 14.986 7 0.036* Rooting percentage % 18.976 7 0.008** Callus rate % 43.913 7 <0.001** Number of roots 117.95 7 <0.001*** Total root length cm 22.952 7 0.002** Average root length cm 22.625 7 0.002** Longest root cm 21.787 7 0.003** Root index 23.824 7 0.001** Biomass g 25.468 7 <0.001*** Hardwood Survival rate % N/A N/A N/A Rooting percentage % 23.537 7 0.002** Callus rate % 15.472 7 0.03* Number of roots 179.05 7 <0.001*** Total root length cm 37.401 7 <0.001*** Average root length cm 31.757 7 <0.001*** Longest root cm 33.216 7 <0.001*** Root index 41.76 7 <0.001*** Biomass g 34.993 7 <0.001*** * p ≤ 0.05; **, p ≤ 0.01; ***, p ≤ 0.001; NS, no significance; N/A, not applicable. Statistical tests were not performed for the survival rate of the hardwood batch as this had 100% survival across all treatments. The semi-hardwood cuttings treated with the 8,000 mg/L K-IBA solution had the lowest survival rate (87.50%), and all hardwood cuttings resulted in 100.00% survival rates (Fig. 3a).
Semi-hardwood cuttings treated with 3,100 mg/L IBA rooting gel had the highest rooting percentages (87.50%), and 1,000 mg/L IBA powder produced the lowest (53.13%). In hardwood cuttings, 8,000 mg/L IBA powder and 8,000 mg/L K-IBA solution had the highest rooting percentages (84.38% each) (Fig. 3b).
Semi-hardwood cuttings showed the highest callus rate (96.88%) was recorded under 1,000 mg/L IBA powder and the water dip, while the lowest (46.88%) was under 8,000 mg/L IBA powder. Hardwood cuttings had a generally higher callus rate with no significant difference among treatments (Fig. 3c).
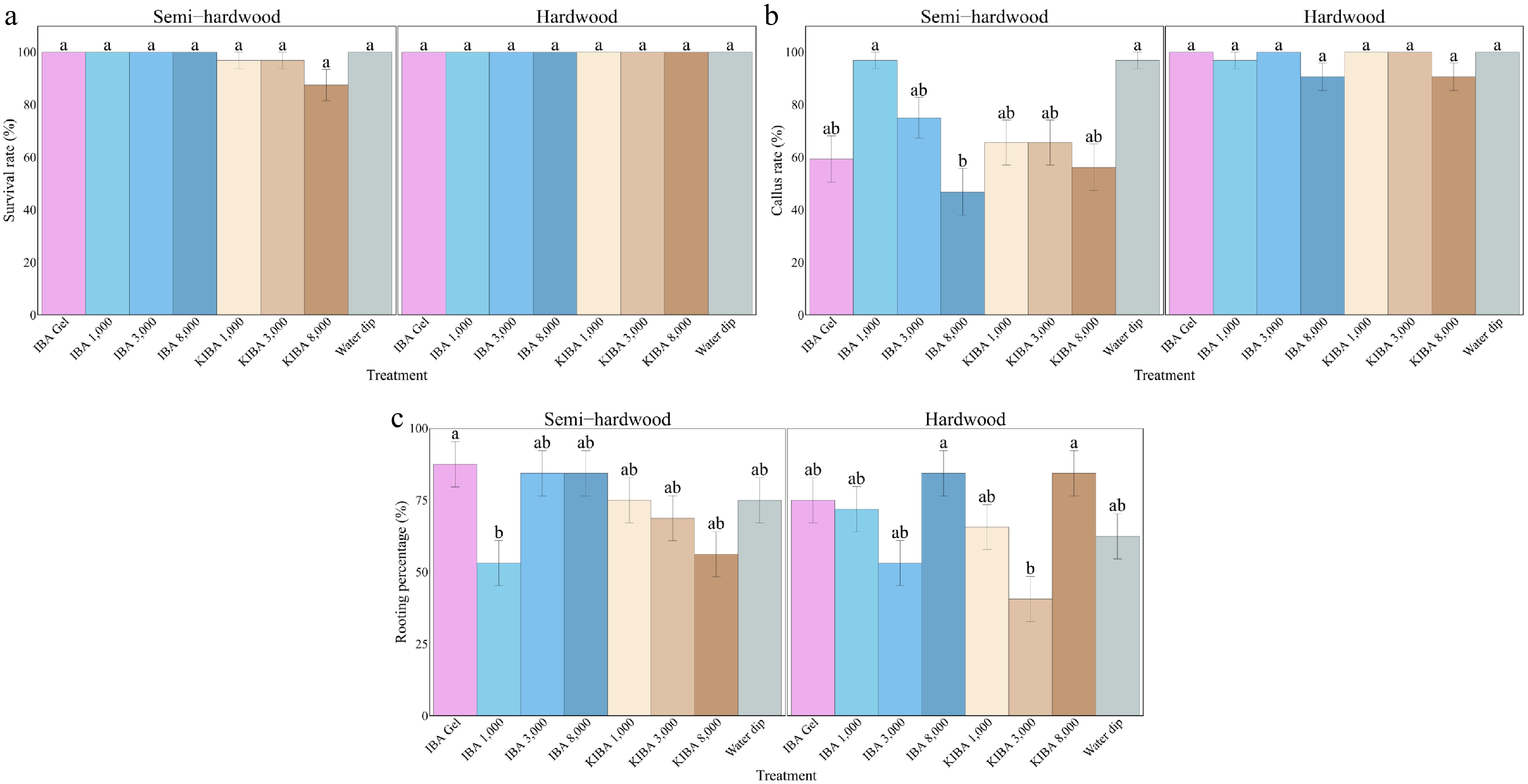
Figure 3.
The survival rate (a), rooting percentage (b), and callus rate (c) of O. fragrans semi-hardwood and hardwood cuttings under various treatments after 14 weeks. Error bar = standard error (SE). Likelihood ratio tests were used to test the treatment effects, and different letters indicate significant differences at p ≤ 0.05 according to Tukey-adjusted estimated marginal means.
Cutting types and IBA effects on root development and cutting biomass
-
In semi-hardwood cuttings, 3,100 mg/L IBA rooting gel produced the highest value for number of roots per cutting, total root length, longest root length, root index, and cutting biomass (6.50, 18.36 cm, 4.74 cm, 149.12, and 10.32 g, respectively), and 8,000 mg/L IBA powder had the longest average root length (3.22 cm). Conversely, 1,000 mg/L IBA powder had the smallest root system (2.41, 6.15 cm, 2.17 cm, 36.43, and 1.34 cm, respectively), and the 8,000 mg/L K-IBA solution produced the lowest biomass (6.73 g) (Figs 4 and 5).
Hardwood cuttings treated with the 8,000 mg/L K-IBA solution had the strongest root system, with the most roots per cutting, the longest roots (total, average, and longest root length), and the highest root index (5.28, 17.26 cm, 2.61 cm, 4.01 cm, and 164.70, respectively). However, the 1,000 mg/L K-IBA solution had the highest biomass (11.42 g), whereas 1,000 mg/L IBA powder had the lowest biomass (9.61 g) (Figs. 4 and 5).
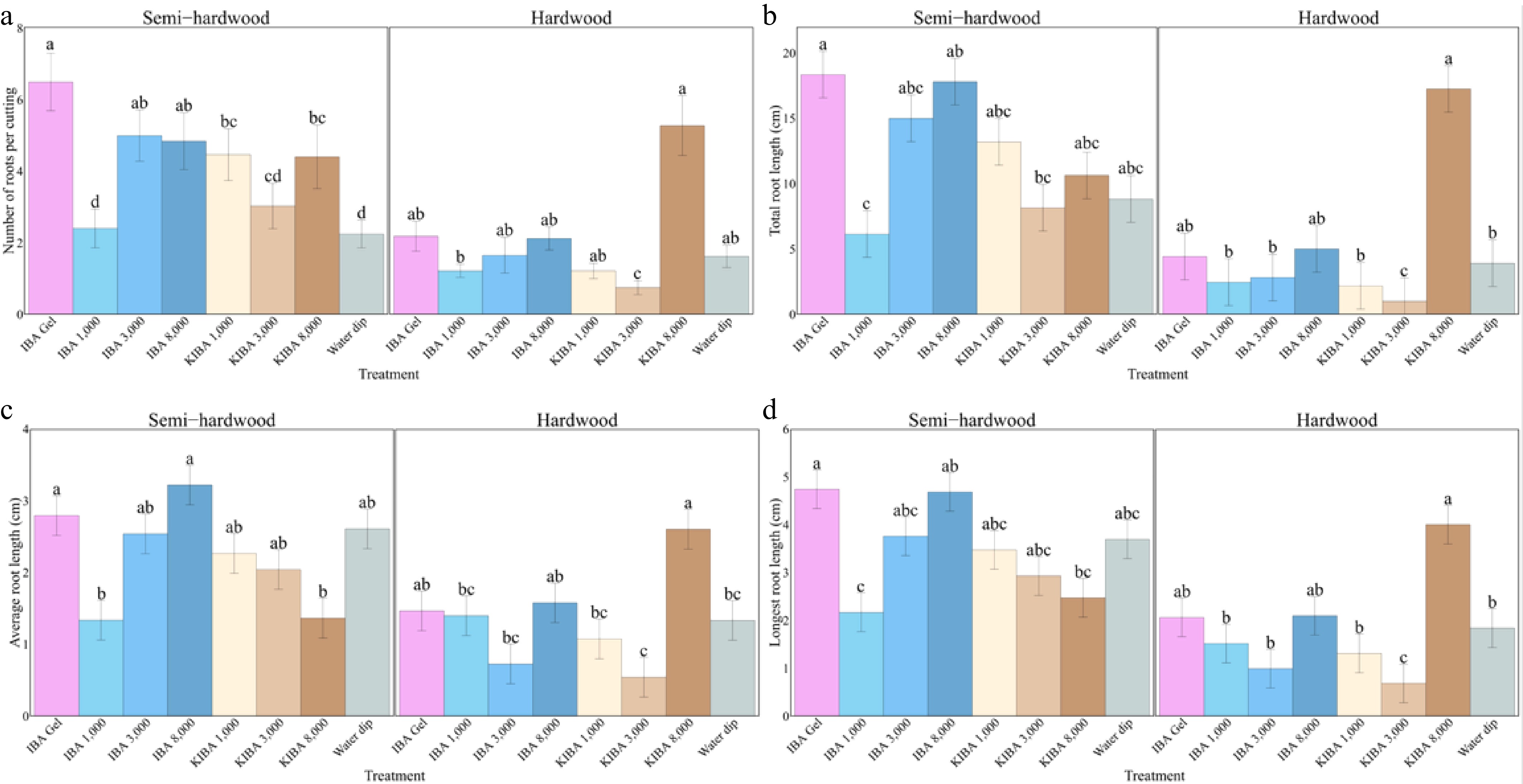
Figure 4.
(a) The number of roots per cutting, (b) total root length, (c) average root length, and (d) longest root of O. fragrans semi-hardwood and hardwood cuttings under various treatments after 14 weeks. Error bar = standard error. Likelihood ratio tests were used to test the treatments' effects, and different letters indicate significant differences at p ≤ 0.05 according to the Tukey-adjusted estimated marginal means.
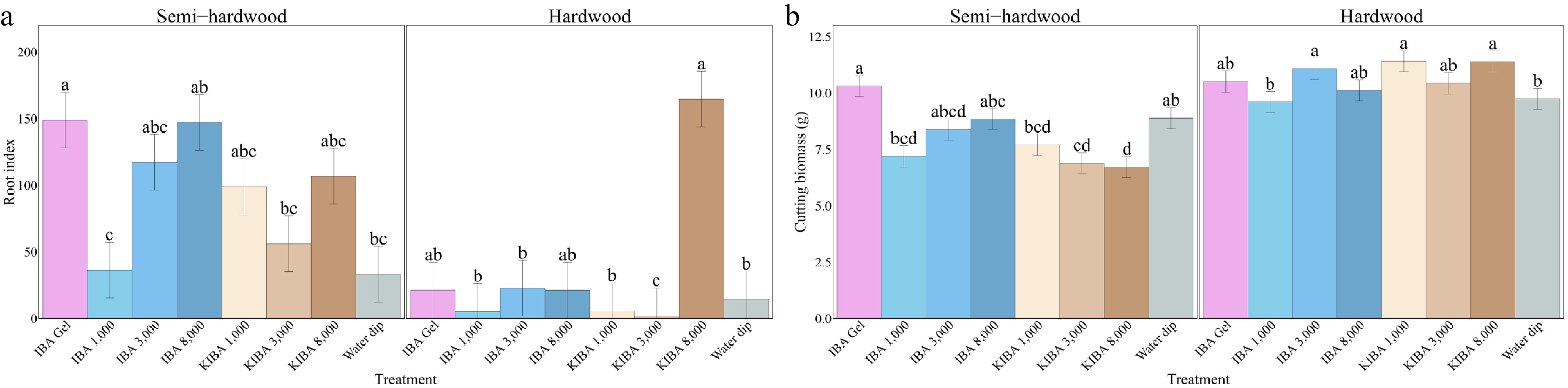
Figure 5.
(a) The root index and (b) cutting biomass of O. fragrans semi-hardwood and hardwood cuttings under various treatments after 14 weeks. Error bar = standard errors. Likelihood ratio tests were used to test the treatments' effects, and different letters indicate significant differences at p ≤ 0.05 according to the Tukey-adjusted estimated marginal means.
-
Different cutting types exhibited different responses in root initiation. For instance, according to the overall means, the semi-hardwood cuttings had more roots per cutting (4.11), longer total root length (12.27 cm), and a lower callus rate (70.31%) compared with hardwood cuttings (2.01, 4.89 cm, and 97.27%, respectively) (Figs 3c and 4a, b). This finding aligned with previous research by Sharma and Sharma[24], who reported significant increases in root number per cutting and root length in wild pomegranate (Punica granatum) semi-hardwood (2.66 and 5.58 cm, respectively) and hardwood cuttings (4.94 and 17.49 cm, respectively). Similarly, Zamir et al.[25] reported that common guava (Psidium guajava) softwood and semi-hardwood cuttings had significant differences in their survival rate (19.80% and 15.00%, respectively). Additionally, Bowerman et al.[13] reported that sparkleberry (Vaccinium arboreum) hardwood cuttings had a significantly higher callus rate (43.80%) than semi-hardwood cuttings (29.60%).
Semi-hardwood cuttings showed a different response, depending on the IBA formulation. Cuttings showed an increasing trend in rooting percentage with higher concentrations of IBA powder and lower concentrations of K-IBA solution (Fig. 3b). In a related study of Blazich and Acedo[23], O. heterophyllus 'Ilicifolius' semi-hardwood cuttings' rates decreased when the concentration of the IBA solution (reagent-grade IBA dissolved in 50% isopropyl alcohol) increased. The different findings from those two studies are likely the results of the different formulations of IBA used, as these formulations may induce changes in the reactive oxygen species (ROS) concentration in root cells, which modulate the hormonal signaling pathways to regulate root initiation and the response to environmental stress[26].
Hardwood cuttings responded differently to the various IBA concentrations and formulations. The 3,000 mg/L IBA powder resulted in reduced root initiation (rooting percentage, average root length, and root index) compared with 1,000 mg/L and 8,000 mg/L IBA powder (Figs. 3b, 4c, and 5a), which aligns with Blazich and Acedo[23]. In their study, hardwood cuttings of O. heterophyllus 'Ilicifolius' treated with a 5,000 mg/L IBA solution had a lower rooting percentage (50.00%) than those treated with 2,500 mg/L (89.10%) and 7,500 mg/L IBA solutions (69.40%)[23]. This nonlinear result may be caused by the increased production of rooting inhibitors and lignified tissues in the more physiologically mature cuttings, leading to delayed or suppressed rooting initiation[27]. In addition, hardwood cuttings treated with 8,000 mg/L K-IBA exhibited the most roots per cutting, the longest roots, and the highest root index (Figs. 4 and 5a). These results are consistent with the findings of Li et al.[18]. In their study, hardwood cuttings of Japanese zelkova (Zelkova serrata) treated with 3,000 mg/L K-IBA developed longer roots (16.20 cm) than those treated with 3,000 mg/L IBA powder (~11.00 cm)[18]. The reason behind those results might be that the hardwood cuttings had a low endogenous auxin content and low uptake capacity, which may result in hardwood cuttings depending on potent water-soluble IBA, since water-soluble salts can produce similar rooting effects with higher concentrations compared with other IBA formulations, even at lower concentrations[16,27].
Effects of IBA formulations on rooting
-
Different formulations of IBA had different effects on the rooting abilities of cuttings. For semi-hardwood cuttings, 8,000 mg/L IBA powder produced the longest average root length, whereas IBA gel yielded the most roots per cutting, as well as the longest total and longest root lengths (Fig. 4a, b, d). These results suggested that the cuttings had different absorption rates for different formulations of IBA, which led to different rooting results[10]. These findings might be caused by the IBA gel adhering more uniformly to the wound surface of the cuttings compared with the K-IBA solution and IBA powder, which provide a greater volume of IBA, resulting in different growth-promoting effects[20].
For semi-hardwood cuttings, their survival rate, rooting percentage, average root length, longest root length, and biomass decreased with increasing K-IBA concentrations. Additionally, the root index of those treated with 3,000 mg/L and 8,000 mg/L K-IBA was lower than that of IBA powder (Figs. 3a, b; 4c, d; and 5a, b). However, higher concentrations of IBA may lead to an increase in ethylene synthesis, which reduces root initiation[28]. These results were not observed with IBA powder in our study, suggesting that higher concentrations of K+ may negatively impact root initiation and plant growth, consistent with the findings of Tian et al.[29]. Their study found that because of the high plasticity of the root system's architecture, high concentrations of K+ can reduce the allocation of photosynthetic products to the root, thereby inhibiting root initiation, reducing total root length, and lateral root density[29]. However, without the pH, electrical conductivity, and tissue K+ content data in our study, it is very hard to determine the negative effect of the K+. This difference between IBA powder and K-IBA may also indicate that different formulations affect the IBA gradients within the cutting tissues, ultimately leading to a significant difference in rooting parameters[30].
Experimental design considerations
-
Several experimental design factors may affect the rooting performance. One factor is that the observation periods were not long enough to determine whether the rooted cuttings ultimately survived. In previous experiments, we found that some rooted cuttings can still die after transplanting, though they have complete root systems, which is aligned with the report of Hartmann et al.[12].
In addition, the same experiment duration was used for different types of cuttings. Our study indicated that semi-hardwood cuttings are easier and faster to root than hardwood cuttings. Additionally, some hardwood cuttings had already initiated root development, as shown by the high rooting percentage and callus rate. However, the root system was still underdeveloped. Therefore, extending the experiment's duration may result in more roots and more complete root systems.
The last factor is that each cutting was treated with only a single formulation of IBA, without considering combinations of different IBA formulations. In the study by Nair et al.[10], the application of a 5,000 mg/L K-IBA solution followed by 3,000 mg/L IBA powder resulted in a higher survival rate (78.10%), more roots per cutting (22.70), and greater total root length (71.90 cm) compared with the 8,000 mg/L K-IBA solution (51.50%, 20.00, and 57.50 cm, respectively) or IBA powder alone (43.60%, 8.70, and 26.90 cm, respectively). Therefore, the combined IBA formulation may lead to better results than the method used in our study.
-
In semi-hardwood cuttings, IBA treatments significantly affected all the parameters measured. However, in hardwood cuttings, all parameters were affected except the survival rate. Under August collection conditions, the cuttings rooted faster when treated with IBA gel, higher concentrations of IBA powder, and lower concentrations of the K-IBA solution. Additionally, under October collection conditions, the cuttings required a longer time for rooting initiation, and only 8,000 mg/L K-IBA significantly promoted overall root development (rooting percentage, number of roots per cutting, root length, and root index). Under August collection conditions and a mist greenhouse environment, 3,100 mg/L IBA rooting gel is the most effective method for promoting O. fragrans propagation by cuttings, as those had the highest rooting percentage, the most roots per cutting, the longest total and longest root lengths, the second highest root index value, and the second longest average root length. However, the same experiment durations were used for different cutting types and the short observation periods limited observations of survival in rooted cuttings and an evaluation of rooting ability in hardwood cuttings. In conclusion, under August collection conditions and a mist greenhouse environment, 3,100 mg/L IBA rooting gel cuttings is the most effective method for propagating O. fragrans by cuttings. Further research should be conducted to test a broader range of collection times, experiment durations, and conditions for propagating O. fragrans by stem cuttings.
The authors received no external funding to acknowledge.
-
The authors confirmed that their contributions to the paper are as follows. Study conception and design: Chen Y, Zhang D, Yu P; validation, data collection: Chen Y, Chen L; formal analysis, analysis and interpretation of the results, writing – original draft preparation, visualization: Chen Y; supervision: Yu P; writing – review and editing: Chen Y, Yu P, Zhang D, Chen H. All authors reviewed the results and approved the final version of the manuscript.
-
All data analyzed during this study are included in this published article.
-
The authors declare that they have no conflict of interest.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Chen Y, Chen L, Zhang D, Chen H, Yu P. 2026. Effect of indole-3-butyric acid concentration and cutting type on the propagation of Osmanthus fragrans. Technology in Horticulture 6: e013 doi: 10.48130/tihort-0026-0004
Effect of indole-3-butyric acid concentration and cutting type on the propagation of Osmanthus fragrans
- Received: 25 June 2025
- Revised: 12 February 2026
- Accepted: 06 March 2026
- Published online: 29 April 2026
Abstract: Osmanthus fragrans Lour. (sweet olive) is an evergreen shrub or small tree in the Oleaceae family with small and fragrant flowers. Stem cutting is the most commonly used asexual method for propagating O. fragrans. Different cutting types of O. fragrans respond differently to formulations and concentrations of plant growth regulators (PGRs) in terms of propagation success. This study evaluated the effects of seven formulations/rates of indole-3-butyric acid (IBA) (1,000, 3,000, and 8,000 mg/L of a potassium IBA [K-IBA] solution; 1,000, 3,000, and 8,000 mg/L of IBA powder; and 3,100 mg/L IBA rooting gel) on O. fragrans 'Beni Kin Mokusei' semi-hardwood cuttings collected in August and hardwood cuttings collected in October 2024. Water dipping was used as the control. Propagation parameters, including survival percent (%), rooting percentage (%), number of roots per cutting, total root length (cm), longest root length (cm), average root length (cm), root index, and cutting biomass (g) were measured. The results showed that the semi-hardwood cuttings treated with 3,100 mg/L IBA rooting gel produced the highest rooting percentage (87.50%), the most roots per cutting (6.50), and the longest total and individual root lengths (18.36 and 4.74 cm, respectively). In conclusion, under August collection conditions and a mist greenhouse environment, treating cuttings with 3,100 mg/L IBA rooting gel is the most effective method for propagating O. fragrans by cutting. Further research should be conducted to test different collection times, longer experiment durations, and the conditions of stem-cutting propagation for other O. fragrans cultivars.
-
Key words:
- Indole-3-butyric acid /
- Stem cutting /
- Rooting /
- PGR /
- Ornamental shrubs /
- Sweet olive












