-
Tea, one of the three most consumed non-alcoholic beverages worldwide, is extensively cultivated across the globe. Sexual reproduction in tea plants often leads to genetic variation, failing to preserve their desirable traits; thus, clonal propagation is predominantly employed in practice. Among these methods, cutting is the most prominent and cost-effective technique due to its simplicity, high efficiency, and the ability to maintain parental superiority[1]. The core of cutting propagation lies in adventitious root (AR) formation, which refers to roots developing from non-root tissues such as stems or leaves, in response to wounding or external stimuli like hormones or pathogenic microorganisms[2]. ARs enhance plant adaptability to environmental changes, improve resource acquisition and support growth, playing a vital role in development and environmental responses. Investigating the effects of different plant growth regulators on AR initiation in cuttings can guide their rational application, significantly improving survival rates, root quality, and shortening the breeding cycle, which holds substantial theoretical and practical significance.
The type and concentration of plant growth regulators significantly influence AR formation in tea cuttings. Certain cultivars that are difficult to root often exhibit insufficient endogenous auxin levels, necessitating the application of exogenous regulators[3]. Commonly used auxins for promoting rooting in tea include indole-3-butyric acid (IBA), naphthaleneacetic acid (NAA), indole-3-acetic acid (IAA), and compound hormones such as ABT rooting powder[4]. Other effective regulators include malic acid (MA), citric acid (CA), γ-polyglutamic acid (γ-PGA), and 2,4-epibrassinolide (EBR). Studies indicate that treating cuttings with optimal concentrations of IBA significantly enhances cell division activity at the base, promotes root primordium differentiation, and facilitates the development of new roots and vascular tissues[5,6]. Soaking tea cuttings in IBA solution markedly shortens rooting time, increases root number, improves root robustness and enhances root vitality compared to controls[7]. Organic acids, such as MA and CA, act as metabolic hubs, signaling molecules, and environmental buffers, improving nutrient uptake, transport, and stress resistance[8]. γ-PGA has been shown to increase root biomass in maize[9]. Brassinosteroids (BRs), such as EBR, play critical roles in root development through synergistic interactions with auxin, likely via overlapping activities, shared target genes, or enhanced auxin transport[10].
The formation of AR in tea plants typically occurs in three stages: callus induction, root primordium initiation, and AR elongation[11]. AR formation is thus a complex, multi-stage physiological process, intricately regulated by hormonal signals throughout each phase[3]. Endogenous hormones involved in tea cutting propagation primarily include IAA, cytokinins (CTK), abscisic acid (ABA), and gibberellins (GA). These hormones function by mobilizing nutrients toward root primordia, enhancing enzyme activity, and promoting AR formation[12]. The relationship between AR formation and endogenous metabolism in tea plants is highly complex. During rooting, cuttings consume substantial nutrients, leading to significant changes in carbohydrate and nitrogen compound levels. Carbohydrates serve as energy reserves, providing not only essential energy, but also generating organic substances and ATP through oxidative metabolism, thereby influencing rooting. Multiple studies indicate that higher carbohydrate content, particularly soluble sugars, facilitates callus and AR formation[13]. Soluble sugars accumulate to a peak during root initiation and decline rapidly as roots elongate, showing an initial increase followed by a decrease[14].
During cutting propagation, oxidases significantly influence plant growth, development, root formation, and other metabolic processes. Studies have shown that indole-3-acetic acid oxidase (IAAO), polyphenol oxidase (PPO), and peroxidase (POD), are closely associated with rooting[15]. IAAO degrades IAA, thereby regulating its endogenous level. High IAAO activity reduces IAA concentration and inhibits root primordium formation; lower IAAO activity generally facilitates easier rooting, dynamically affecting AR development[16]. PPO, a copper-containing oxidase, not only contributes to tea quality, but also plays a key role in AR formation by catalyzing the synthesis of the 'IAA-phenolic acid complex,' a recognized co-factor for rooting[17]. POD, involved in auxin metabolism, respiration, photosynthesis, and cell wall synthesis, also oxidizes IAA and influences AR initiation and formation[18].
Existing research has primarily focused on individual regulators, leaving the dynamic changes in key physiological indicators under different treatments, and their contributions to the variation in AR formation poorly understood. Based on preliminary screening of common plant growth regulators and concentrations, this study employed IBA and EBR as experimental treatments, with water-treated cuttings serving as the control. Key nutrient levels (soluble sugars, starch, and proteins), oxidase activities (POD, PPO, IAAO), and endogenous hormone levels (IAA, CTK, ABA), were monitored during AR formation. By analyzing the relationship between these factors and rooting progression, we aim to elucidate the physiological mechanisms underlying IBA, EBR, and their combined effects on AR development in tea cuttings, thereby providing a scientific basis for establishing an efficient propagation system.
-
Plant materials were collected from the Tea Demonstration Station of Northwest A&F University (Xixiang County, Hanzhong, Shaanxi Province, China; 107°46′ E, 33°05′ N), located in a temperate monsoonal climate with an annual average temperature of 14.2 °C, precipitation of 800–1,000 mm, and elevation of 500–800 m. Healthy, pest-free, current-year semi-lignified shoots of 'Zhongcha 108' were harvested in summer and autumn. Cuttings were 3–4 cm long with stem diameters of 2–3 mm, each retaining one fully expanded apical leaf, and one plump axillary bud at the base. The top cut was made perpendicular to the stem axis to avoid cambium damage, while the basal cut was angled at 45° to increase absorption area, with all cuts smooth and clean to ensure uniformity and to minimize experimental error.
The experiment was conducted in a research greenhouse at the Northwest A&F University, with temperature maintained at 14–45 °C (± 1.0 °C), and relative humidity at 60%–90% (± 7% RH). Natural light was used, supplemented with shading, forced ventilation during summer months to keep temperatures below 35 °C to avoid stress, and heating in winter to maintain temperatures above 10 °C for overwintering. A 3:8 (v/v) mixture of substrate and vermiculite served as the growing medium, providing high porosity, aeration, and drainage (V9 substrate: organic matter ≥ 30%; pH 4.5–7.0; moisture ≥ 20%). The medium was disinfected by soaking in 0.1% KMnO4 solution for 30 min, air-dried, and filled into 50-cell trays. Trays were irrigated to full capacity 24 h before insertion, and used when the surface was dry, but the interior remained moist and soft.
During the initial stage, cuttings were watered twice daily to maintain substrate moisture content between 75% and 100%. After rooting, watering was reduced to once every 1–2 d to sustain moist conditions. Throughout the propagation period, cuttings were regularly monitored, and any moldy or decayed specimens were promptly removed to ensure environmental stability.
Treatments
-
The experimental factors included different plant growth regulators, concentration gradients, and soaking durations (set according to regulator properties), with treatments applied via either low-concentration long-soaking, or high-concentration quick-dip methods; control (CK) cuttings were soaked in water. Each treatment comprised three biological replicates, with 150 cuttings per replicate to ensure data reliability. The basal ends (2–3 cm) of the cuttings were immersed vertically in the corresponding regulator solutions. After absorption, cuttings were inserted vertically into the substrate to a depth of two-thirds their length (approximately 2–3 cm), using a moist substrate with moderate water content. All leaves were oriented uniformly to avoid mutual shading, with petioles and axillary buds exposed and maintained 0.5–1 cm above the substrate to prevent leaf-soil contact and decay. The substrate was gently compacted around each cutting to ensure close contact and facilitate water and nutrient uptake. Specific treatments are listed in the supplementary tables (Supplementary Tables S1 and S2).
After insertion, callus and root development were monitored daily. Rooting indices were recorded at 45 d. For biochemical analysis, basal tissues from 35 randomly selected cuttings per treatment were collected at 0, 7, 15, 21, 30, and 45 d after insertion, flash-frozen in liquid nitrogen, and stored at −80 °C.
Analytical methods
-
Nutrient content was determined as follows: soluble sugars and starch were measured using the sulfuric acid-anthrone colorimetry method. Fresh tissue (1 g) was homogenized in 5 mL of 80% ethanol using an ice bath, extracted at 80 °C for 30 min with intermittent shaking, and centrifuged at 3,000 r/min for 10 min. The supernatant was collected, and the residue was re-extracted twice. The combined supernatants were diluted to 25 mL with 80% ethanol as the soluble sugar extract. A standard curve was prepared using glucose solutions (0–1.0 mL of 0.1 mg/mL), and anthrone reagent, with absorbance measured at 620 nm. For starch, the residue after sugar extraction was hydrolyzed with 5 mL of 6 mol/L HCl at 100 °C for 30 min, neutralized with NaOH, and diluted to 25 mL. The glucose released was quantified similarly using the anthrone method. Soluble protein was determined by the Coomassie Brilliant Blue G-250 method. Tissue (1 g) was homogenized in 5 mL of 50 mmol/L Tris-HCl buffer (pH not specified), centrifuged at 10,000 r/min for 20 min at 4 °C, and the supernatant was collected and diluted to 25 mL. A standard curve was constructed with BSA (0–1.0 mL of 0.1 mg/mL), and absorbance was read at 595 nm after adding Coomassie blue reagent and incubating for 5 min. Sample absorbances were measured accordingly under the same conditions.
Enzyme activities were determined using the following protocols: crude enzyme extracts were prepared by homogenizing 1 g of plant material in 5 mL of phosphate buffer, followed by centrifugation at 4,000 r/min for 15 min. The supernatant was collected as the enzyme extract. PPO activity was measured according to Li[19] using catechol as substrate and monitoring absorbance at 410 nm. POD activity was assayed based on Gao[20]: the extract was diluted to 100 mL with phosphate buffer, and the reaction mixture containing 3 mL of substrate solution, and 1 mL of enzyme extract was monitored at 470 nm every 30 s. IAAO activity was determined using the dichlorophenol colorimetric method as described by Li[19].
Hormone levels were determined according to the method described by Li[19]. Frozen samples collected after rooting treatments were ground into powder using a mortar. Approximately 0.1 g of the powder was weighed into a centrifuge tube, mixed with 1 mL of ethyl acetate extraction solvent, and vortexed at 2,000 rpm for 10 min. After centrifugation at 12,000 rpm for 10 min at room temperature, the supernatant was carefully transferred to a new 2 mL tube. The solvent was evaporated under a nitrogen stream for approximately 45 min. The dried residue was redissolved in 200 μL of 70% methanol, followed by vortexing and centrifugation as above. The resulting supernatant was filtered through an organic solvent-compatible membrane, and stored in a brown injection vial for subsequent analysis. The contents of IAA, ABA, GA3, and tZTR were quantified using liquid chromatography-tandem mass spectrometry (LC-MS/MS). Three biological replicates were performed for each hormone.
MDA content was determined according to the thiobarbituric acid colorimetric method[19]. Briefly, 1 g of plant tissue was homogenized in 5 mL of 5% TCA, followed by centrifugation at 3,000 × g for 10 min. Then, 2 mL of the supernatant was mixed with 2 mL of 0.67% TBA, heated in a boiling water bath for 15 min, and centrifuged again after cooling. The absorbance of the resulting supernatant was measured at 450, 532, and 600 nm (denoted as A450, A532, and A600, respectively), using a reference control consisting of 2 mL distilled water mixed with 2 mL of 0.67% TBA.
Root activity was assessed 45 d after cutting using a root activity assay kit (Solarbio, TTC method) applied to treated cuttings.
Rooting observation and data collection
-
At 0, 7, 15, 21, 30, and 45 d after cutting, 35 cuttings per treatment were randomly selected for measurement. After each observation, plants were carefully returned to their original seedbeds to maintain consistent growth conditions. At 45 d, 60 cuttings per group were collected to assess survival rate, rooting rate, root number, root length, root spread, root diameter, number of root tips, and root activity. Survival rate was calculated as (number of surviving cuttings/total cuttings sampled) × 100%, and rooting rate as (number of rooted cuttings/total cuttings sampled) × 100%.
Data processing and analysis
-
Data are presented as mean ± SD. Preliminary data organization was performed using Microsoft Excel 2019. Statistical analysis was carried out with SPSS software (SPSS Inc., Chicago, IL, USA) using one-way ANOVA, followed by Duncan's multiple range test to assess significant differences between treatments (p < 0.05, p < 0.01). Graphs were plotted, and correlation analyses were conducted using GraphPad Prism version 9.0.
-
To investigate the effects of different auxins on the rooting efficiency of tea cuttings, this study employed IBA, ABT rooting powder, IAA, and NAA, in a comparative experiment following the methods outlined in Supplementary Table S1. Most treatment groups exhibited significantly higher survival and rooting rates compared to the control, indicating that appropriate concentrations of plant growth regulators can enhance cutting survival and root formation. The A14 treatment achieved the highest survival rate (81.73%) among all groups. Significant differences in rooting rates were observed across treatments, with the A10 group showing the highest AR formation rate at 83.33%. At a concentration of 300 mg/L, the rooting rates of ABT, IAA, and NAA treatments were significantly higher than the control; however, when the concentration increased to 500 mg/L, the rooting rates declined across all auxin treatments. The number of ARs in A2 and A10 groups was significantly greater than in other treatments, reaching approximately 18 roots per cutting. Additionally, root length in the A10 group was significantly longer than in A2, representing the maximum value observed among all treatments. The application of 300 mg/L IBA (A10 group) resulted in the highest rooting rate, greatest root length, and significantly superior performance in both survival rate and root number compared to all other treatments (Fig. 1).
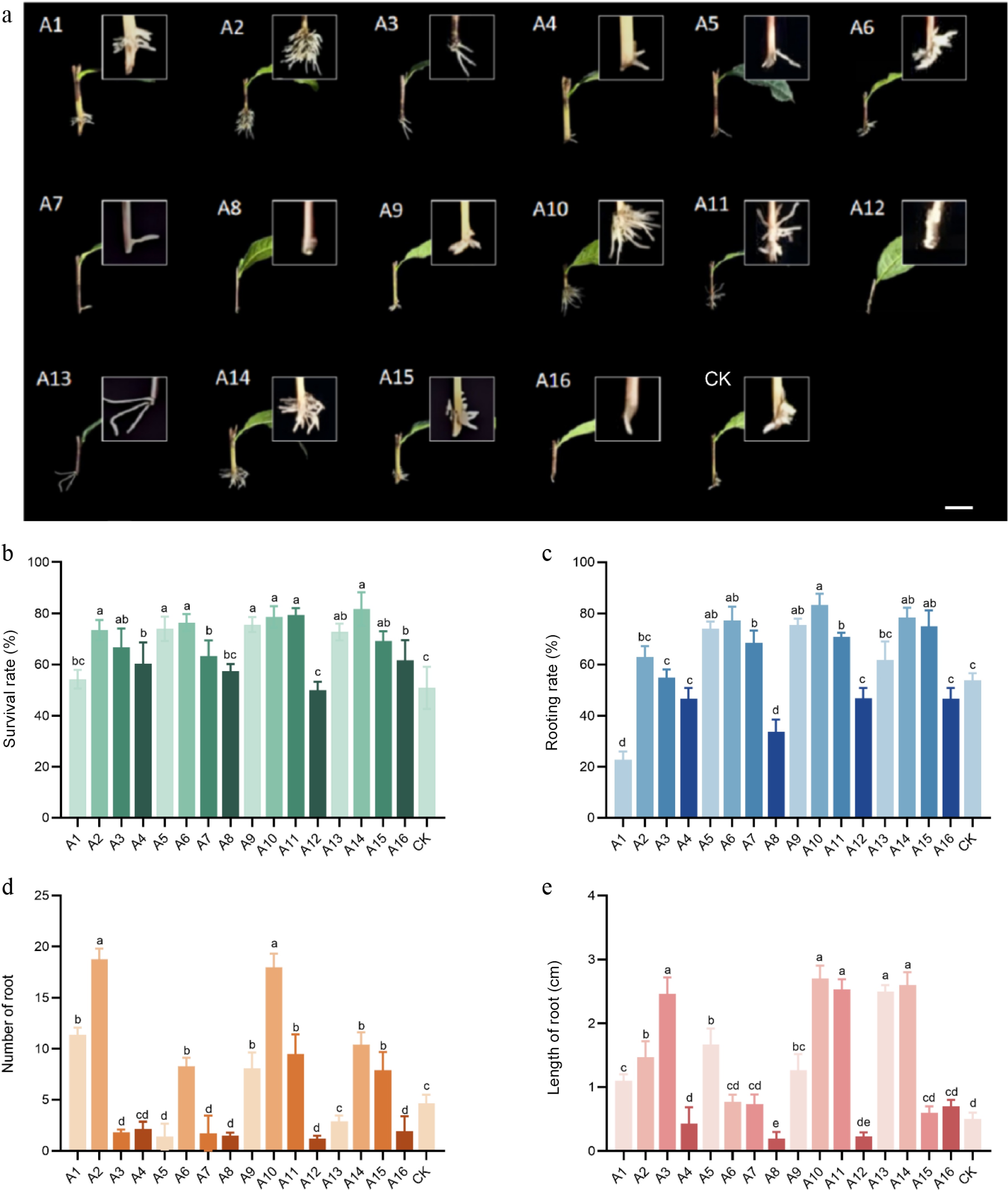
Figure 1.
Cuttings were treated with different auxins. (a) Phenotype of cuttings treated with different auxins. (b) Survival rate of cuttings treated with different auxins. (c) Rooting rate of cuttings treated with different auxins. (d) Number of roots of cuttings treated with different auxins. (e) Root length of cuttings treated with different auxins. Note: bar = 1 cm. Significant differences were found between different alphabets for different treatment groups, p ≤ 0.05.
To evaluate the effects of novel plant growth regulators on the rooting efficiency of tea cuttings, this study applied MA, CA, γ-PGA, and EBR in concentration gradient experiments. The results showed that all treatments except B12 and B16 significantly improved both survival and rooting rates compared to the control. The B2 treatment resulted in the highest rooting rate of 80.17%, which was significantly greater than that of the control. Although increasing concentrations gradually reduced the rooting rate, all values remained higher than the control. The optimal concentration for CA was 500 mg/L, yielding a rooting rate of 75.33%, with no inhibitory effect observed across the tested range (100–1,000 mg/L). The B14 treatment performed best in terms of AR number and root length, with an average of 15.63 roots per plant and a survival rate second only to B2, with no significant difference between the two. Furthermore, it was noted that root number decreased with increasing EBR concentration, indicating that low concentrations of EBR effectively promoted root development, whereas higher concentrations were inhibitory. Thus, the selection of appropriate types and concentrations of growth regulators is crucial for optimizing the rooting of tea cuttings (Fig. 2).
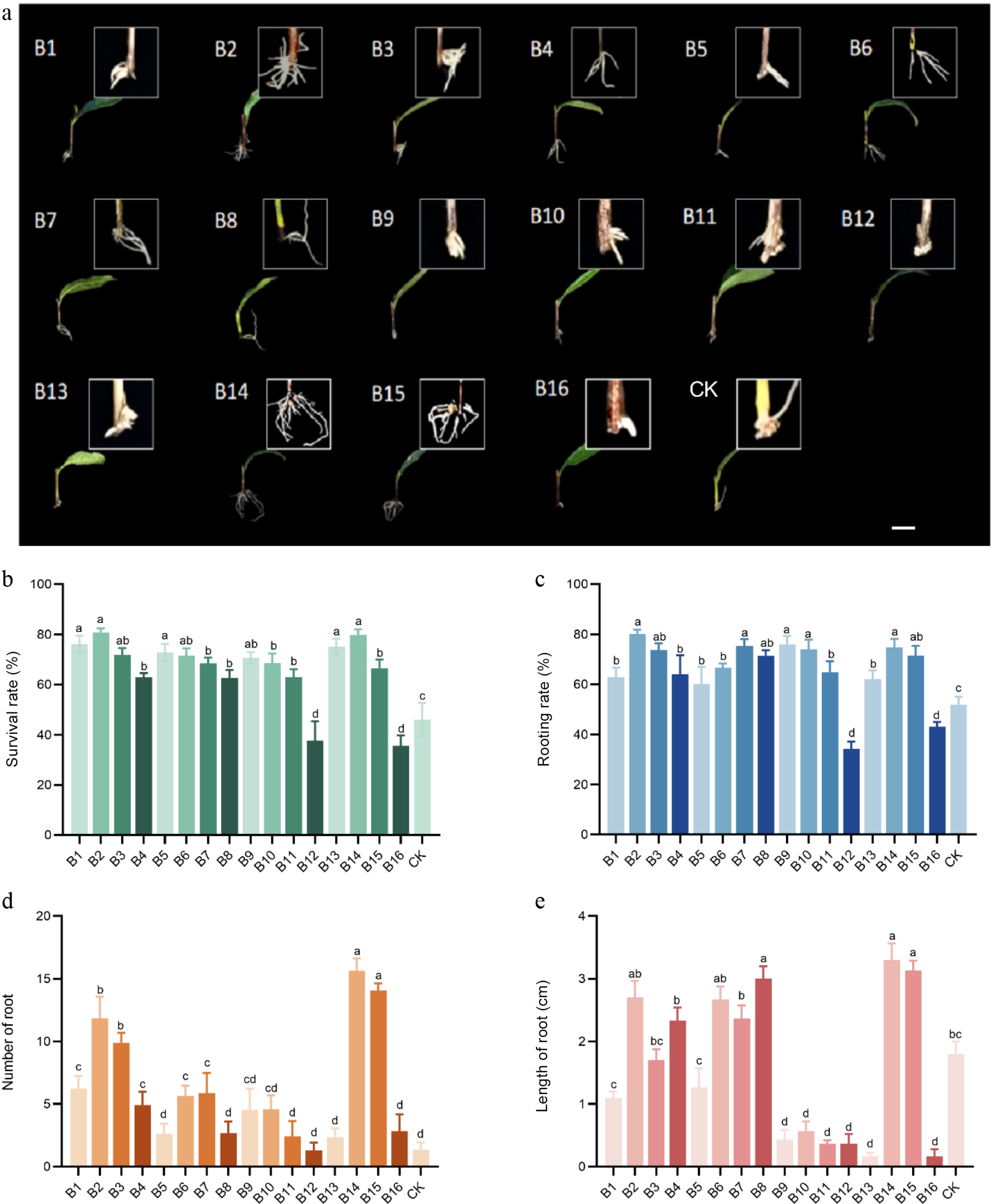
Figure 2.
Cuttings were treated with other plant growth regulators. (a) Phenotype of cuttings treated with other plant growth regulators. (b) Survival rate of cuttings treated with other plant growth regulators. (c) Rooting rate of cuttings treated with other plant growth regulators. (d) Number of roots of cuttings treated with other plant growth regulators. (e) Root length of cuttings treated with other plant growth regulators. Note: bar = 1 cm. Significant differences were found between different alphabets for different treatment groups, p ≤ 0.05.
Changes in external morphology during the AR formation process
-
To investigate the differential effects of two hormone treatments, a dynamic observation experiment was conducted using water as the CK on the previously screened root-promoting treatment groups A10 and B14. The results demonstrated significant differences in the AR formation process among the treatments (A10, B14, and CK), which could be divided into three distinct stages: callus induction, root primordium initiation, and AR elongation, with the onset and duration of each stage varying by treatment. The IBA-treated group (A10) exhibited the shortest callus formation time, with 18.72% of cuttings developing light-yellow callus at the base by day 15, whereas neither the EBR group (B14), nor the control showed visible changes at this timepoint. ARs emerged in A10 after 15 d. In contrast, callus formation in B14 occurred around day 21, by which time the callus in A10 had expanded throughout the basal periphery. Notably, ARs in B14 appeared within 9 d after callus formation, a shorter interval than in other groups. The control cuttings required approximately 30 d to form callus, followed by AR emergence after another 15 d. These results indicate that both A10 and B14 treatments significantly accelerated the adventitious rooting process: A10 markedly shortened the callus induction phase, while B14 reduced the duration of root primordium initiation, leading to a significant decrease in total rooting time (Fig. 3).
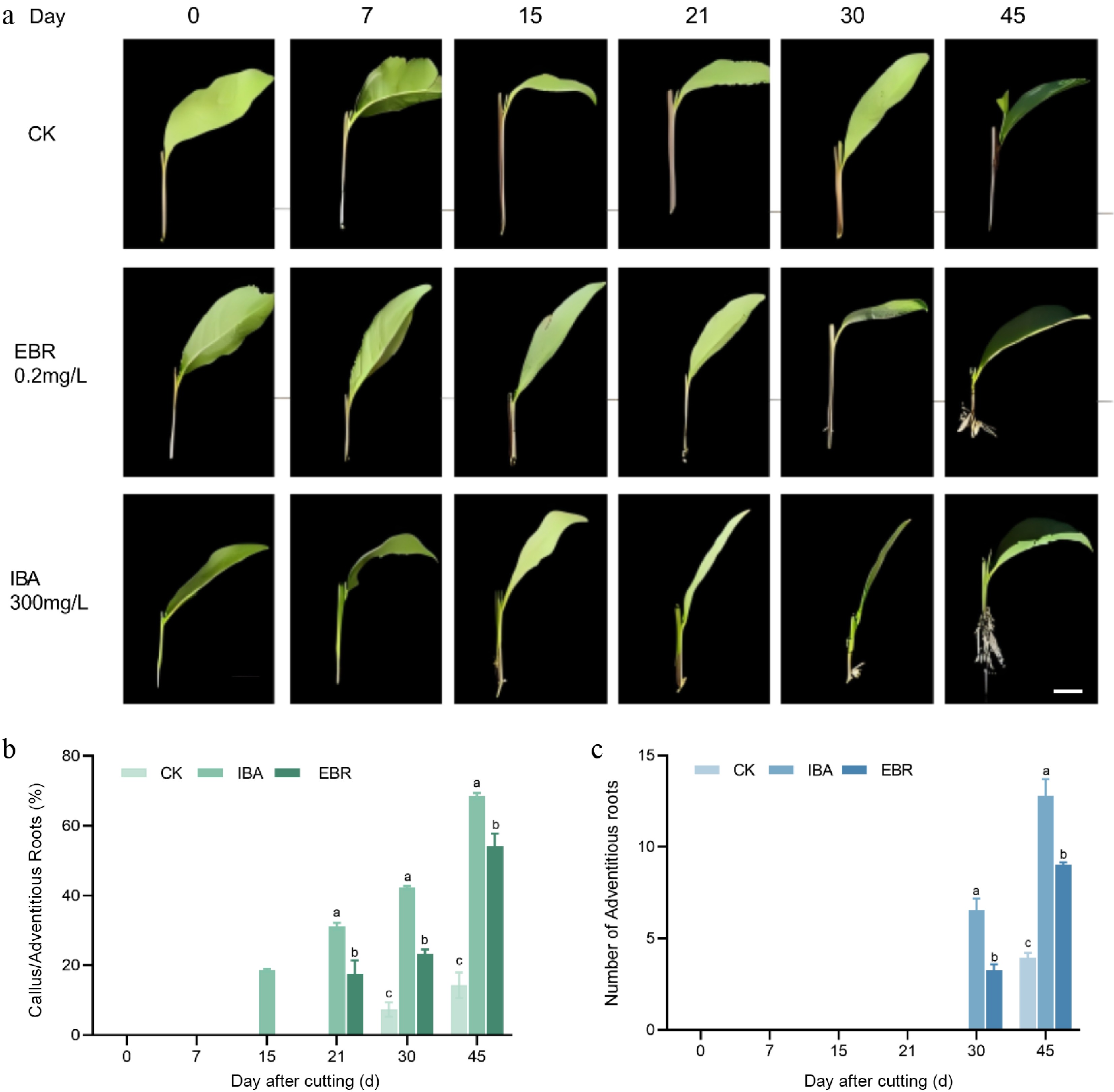
Figure 3.
Comparative analysis of adventitious rooting processes in cuttings treated with IBA and EBR. (a) Phenotypic diagram of IBA and EBR treatment for rhizome rooting process. (b) Incidence of callus and adventitious root formation during the rooting process under IBA or EBR treatment. (c) Number of adventitious roots induced by IBA and EBR treatments. Note: bar = 1 cm. Significant differences were found between different alphabets for different treatment groups, p ≤ 0.05.
Modulation of AR morphology and endogenous substances in Camellia sinensis cuttings by IBA, EBR, and their combination
-
Plant growth regulators play a key role during the short-cutting propagation of tea plants by regulating root primordium differentiation, and vascular bundle development. Based on preliminary screening results, this study selected IBA and EBR—both showing significant rooting-promoting effects—as the target regulators, with a 30-min water soak serving as the control. Single and combined treatments of IBA and EBR were designed. The effects of these individual and combined plant growth regulator treatments on AR formation were evaluated by assessing cutting morphology at 45 d after insertion, as well as by monitoring dynamic changes in nutrients, oxidative enzymes, and key endogenous hormones during the rooting process.
Comparison of rooting efficiency under different hormone treatments
-
Regarding the effects of treatments on the underground parts of the cuttings, the IBA-EBR combined treatment showed the best performance with a survival rate of 82.65%. The rooting rate was 78.56% in the IBA-alone group, and 74.27% in the combined treatment, both being significantly higher than the control. All three treatment groups exhibited significantly greater root length compared to the control, with the IBA-treated group achieving the longest average root length (4.71 cm), followed by the EBR (4.32 cm) and combined treatment groups (4.26 cm). Notably, the IBA-EBR combination resulted in significantly higher root number (15.02 per plant) and root tip count (36.14 per plant) than all other treatments. An increase in root number was accompanied by a corresponding decrease in root diameter. The EBR and control groups developed 10.08 and 7.24 roots per plant, respectively, which were significantly fewer than those in the IBA and IBA-EBR groups, but their root diameters (1.62 and 1.36 mm, respectively) were larger. Although all treatments significantly improved rooting rate, survival rate, and root length compared to the control, no significant differences in these parameters were observed among the three treatment groups. Root activity is a key indicator of the absorptive capacity of plant root systems, with higher values reflecting more developed roots and enhanced nutrient uptake. Compared to the control, both exogenous IBA and EBR treatments improved the root activity of ARs in tea cuttings. However, the combined IBA-EBR treatment exerted a stronger promoting effect than either individual application (Fig. 4).
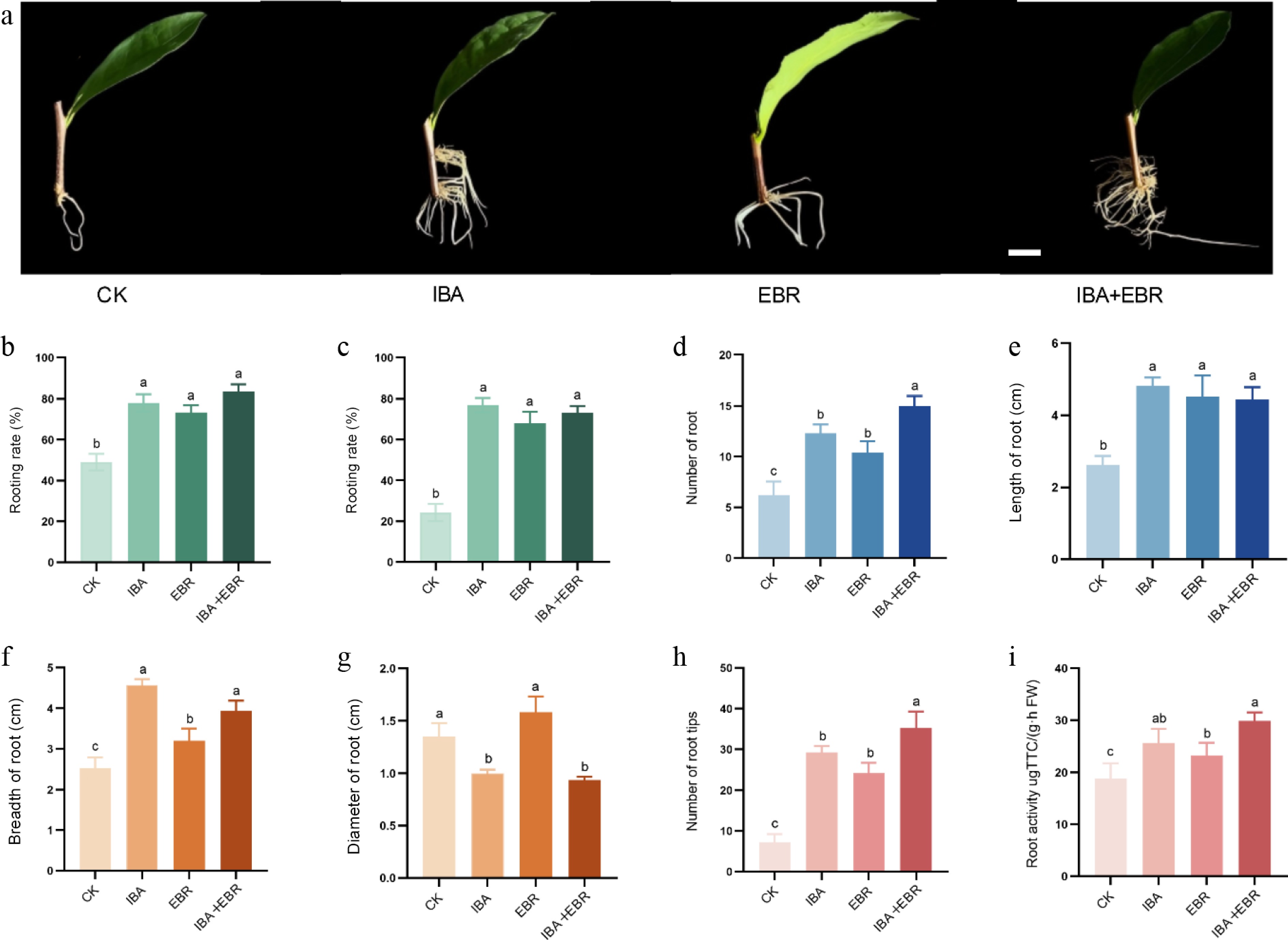
Figure 4.
Effects of IBA, EBR, and their combination on belowground development of Camellia sinensis cuttings. (a) Phenotype of cuttings treated with IBA, EBR, and their combination after 45 d, showing from left to right: CK, IBA, EBR, and IBA–EBR combined treatment. (b) Survival rate. (c) Rooting rate. (d) Root number. (e) Root length. (f) Root spread. (g) Root diameter. (h) Number of root tips. (i) Root activity. Note: bar = 1 cm. Significant differences were found between different alphabets for different treatment groups, p ≤ 0.05.
Overall, the IBA-EBR combination significantly increased the number of ARs and root tips. Although no significant differences were observed in survival rate or root activity between the combined treatment and other groups, these two metrics reached their highest values under the IBA-EBR treatment.
Changes in nutrient content during the AR formation process
-
The results indicated that soluble sugar content initially decreased briefly during the early cutting stage, followed by a recovery after callus formation. A significant reduction occurred during the rooting phase as the cuttings began to establish autonomous nutrient uptake capacity. In the later stage, as the root system became fully functional, soluble sugar levels gradually returned to normal metabolic levels. Throughout the rooting process, the soluble sugar content in cuttings treated with IBA, EBR, and IBA-EBR was generally higher than that in the control group. Variation trends differed among treatments: the IBA-treated group reached a peak of 69.23 mg/g at 21 d after insertion, declining to the lowest value of 41.82 mg/g by day 30; the EBR-treated group showed the lowest level (43.67 mg/g) at day 15; whereas the IBA-EBR co-treatment significantly increased soluble sugar content at both 21 and 30 d, reaching 79.42 and 52.37 mg/g, respectively (Fig. 5a).
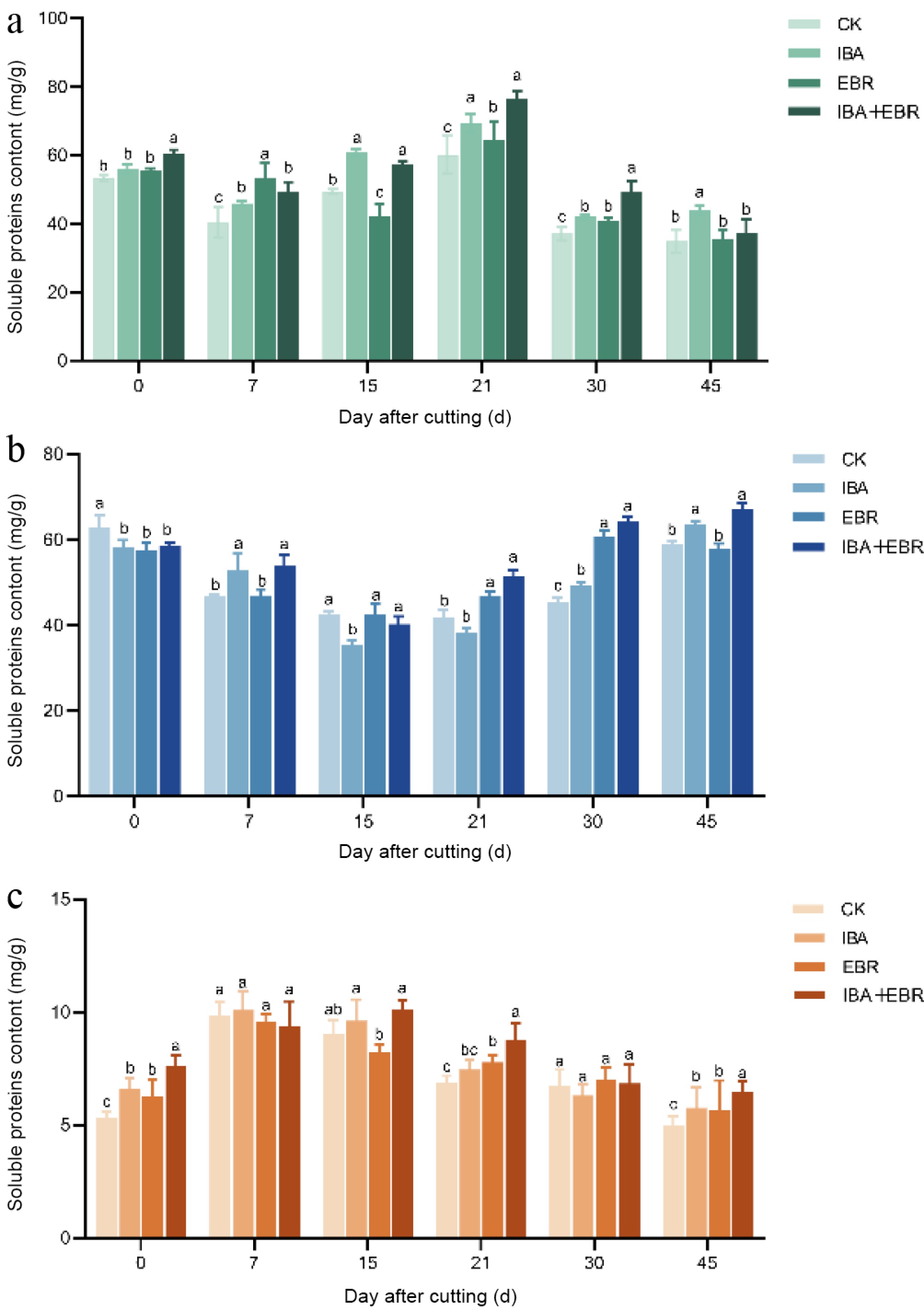
Figure 5.
Effects of IBA, EBR, and their combination on nutrient contents in Camellia sinensis cuttings. (a) Influence on soluble sugar content. (b) Influence on soluble starch content. (c) Influence on soluble protein content. Note: Significant differences were found between different alphabets for different treatment groups, p ≤ 0.05.
Soluble starch content exhibited a decline-rise pattern, though the timing of peak and trough values varied among treatments. At 15 d after insertion, the IBA-treated group showed significantly lower starch content than other groups. By day 21, the IBA-EBR combined treatment reached the highest soluble starch level of 54.73 mg/g, which further increased to a peak of 70.16 mg/g at day 45. In contrast, the EBR-treated group displayed the lowest soluble starch content among all groups at both 15 and 21 d after insertion (Fig. 5b).
The soluble protein content in all groups exhibited a consistent upward-downward trend, but the peak occured at different times. No significant differences were observed among the groups at 7 and 30 d after insertion. However, at day 15, the EBR-treated group showed the lowest soluble protein content (7.43 mg/g), which was significantly lower than that in the IBA and IBA-EBR combined treatment groups (Fig. 5c).
Changes in oxidase activity during AR formation
-
The activities of antioxidant enzymes, including POD, PPO, and IAAO, significantly influence AR formation and seedling growth. POD activity increased significantly across all treatments at day 7, with the IBA-EBR combined treatment showing the highest activity, which differed significantly from the other groups. By day 15, the IBA-EBR group maintained the highest POD activity, followed by the EBR treatment, with no significant difference between these two, while the IBA group exhibited the lowest activity. After 21 d, POD activity began to decline in all groups except the IBA treatment. By day 30, during the root primordium elongation phase, the metabolic emphasis shifted toward auxin-regulated cell expansion, and POD activity continued to decrease. Once root system establishment was complete, POD activities across groups measured 23.27, 20.70, 24.53, and 25.00 U/g, returning to normal levels. Throughout the rooting process, both the EBR and IBA-EBR combined treatments significantly enhanced POD activity in the cuttings (Fig. 6a).
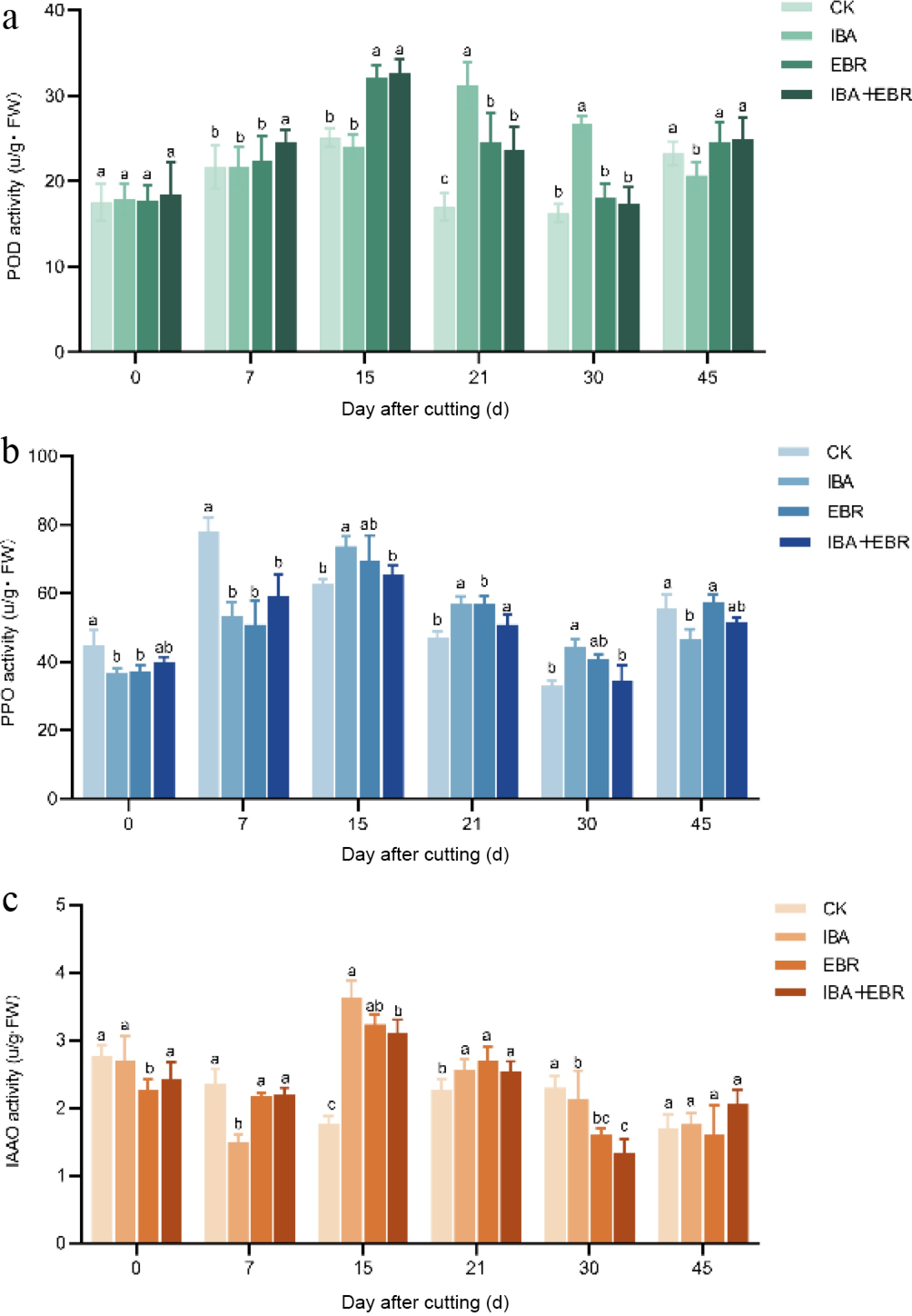
Figure 6.
Effects of IBA, EBR, and their combination on enzyme activities in Camellia sinensis cuttings. (a) Influence on peroxidase (POD) activity. (b) Influence on polyphenol oxidase (PPO) activity. (c) Influence on indole-3-acetic acid oxidase (IAAO) activity. Note: Significant differences were found between different alphabets for different treatment groups, p ≤ 0.05.
PPO activity increased rapidly on day 0 and day 7 after cutting, with values of 62.60, 73.80, 69.40, and 65.47 U/g, respectively. By the root differentiation and elongation stage (day 30), PPO activity decreased to 32.93, 44.30, 40.80, and 34.57 U/g, respectively, yet remained higher in treated groups than in controls, gradually declining as metabolism shifted toward auxin response and cell elongation. After root system establishment, PPO activity returned to normal levels (Fig. 6b).
Early after excision, IAAO activity was suppressed to maintain high IAA concentrations, facilitating root primordium formation. During mid-rooting, IAAO activity reached 1.76, 3.63, 3.23, and 3.10 U/g, respectively, indicating its upregulation to optimize IAA levels, and prevent excessive cell division or oxidative damage in root tips. In the later stage, as new roots elongated and photosynthetic capacity was established, IAAO activity declined again to 2.30, 2.13, 1.60, and 1.33 U/g, respectively, to maintain basal IAA levels required for essential metabolism. Treated groups exhibited significantly lower IAAO activity than controls on days 0, 7, and 30, suggesting that exogenous regulators moderately inhibit IAAO to delay IAA degradation and improve rooting. By day 45, no significant differences in IAAO activity were observed among groups (Fig. 6c).
Changes in endogenous hormone levels and ratios
-
As shown in the figure, IAA content exhibited a pattern of rapid increase, followed by a brief decline and a secondary surge, before eventually stabilizing. During the early cutting stage (0–7 d), IAA levels in IBA-treated cuttings rose rapidly from an initial value of 3.62 to 5.62 ng/g. Throughout callus formation and root primordium development (15–30 d), IAA increased again to regulate cell elongation in the root apical meristem and promote root primordium differentiation, reaching a peak of 5.86 ng/g by day 30. Both the IBA-alone, and the IBA-EBR combined treatments resulted in significantly higher IAA levels than the control at days 7, 15, and 21, which were positively correlated with the rooting rate. In contrast, tZTR content showed a downward-upward-downward trend, inversely related to IAA dynamics. With the exception of days 7 and 21, tZTR levels in all treatment groups were significantly lower than those in the control. ABA content demonstrated an initial increase, followed by a decrease. From day 7 to 21, ABA levels in all treatment groups were significantly higher than in the control, indicating enhanced stress resistance in response to exogenous plant growth regulators. As the cuttings adapted to the new environment, ABA concentrations gradually decreased and remained at low levels during later rooting stages. GA3 content exhibited a rise-decline-rise pattern. With the exception of day 21, GA3 levels in both the IBA and IBA-EBR groups were significantly higher than in the control (Fig. 7).
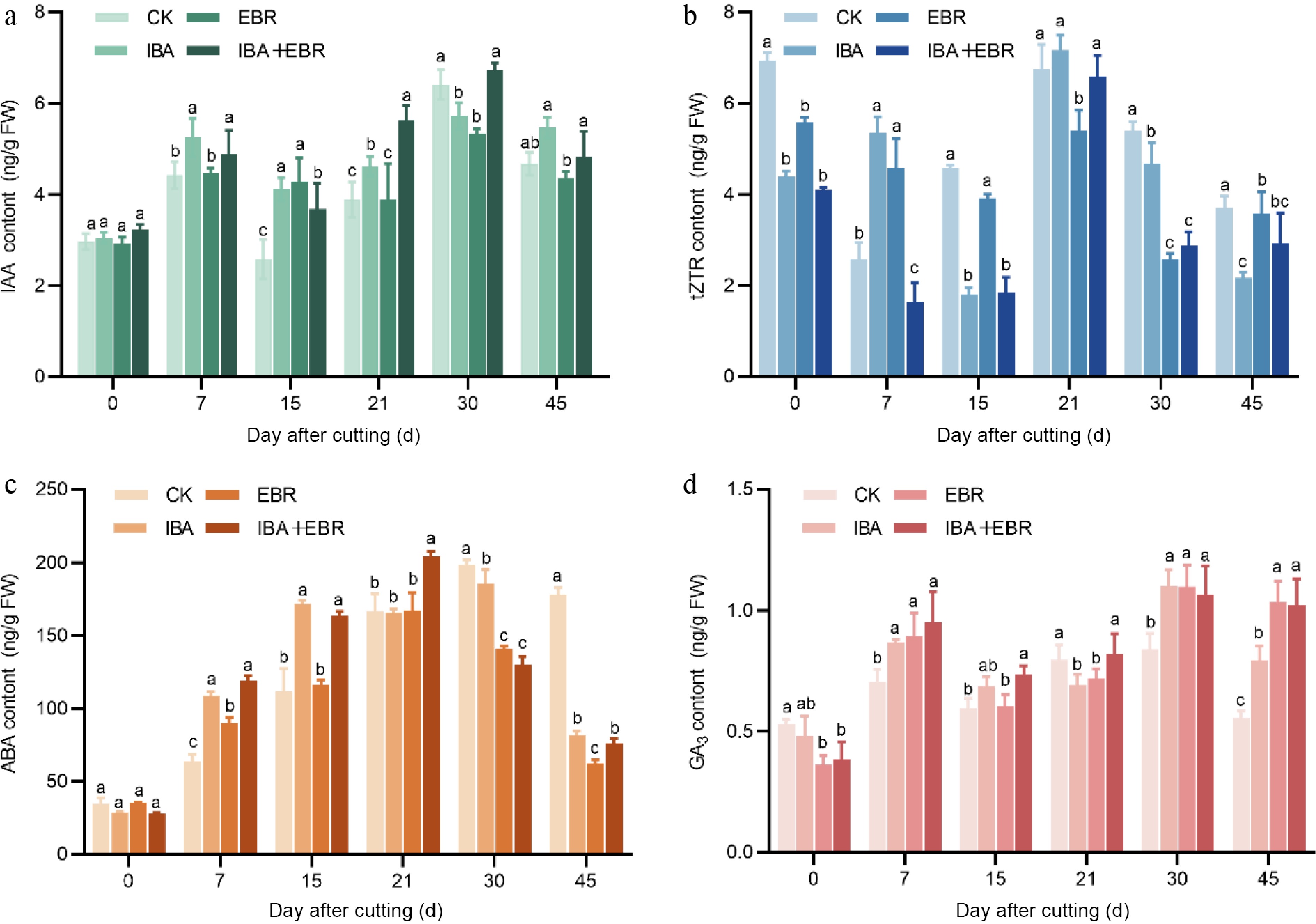
Figure 7.
Effects of IBA, EBR, and their combination on endogenous hormone contents in Camellia sinensis cuttings. (a) Influence on indole-3-acetic acid (IAA) content. (b) Influence on trans-zeatin riboside (tZTR) content. (c) Influence on abscisic acid (ABA) content. (d) Influence on abscisic acid (ABA) content. Note: Significant differences were found between different alphabets for different treatment groups, p ≤ 0.05.
These results indicate that exogenous plant growth regulators significantly improved rooting efficiency by optimizing hormonal balance. AR formation in cuttings is the result of synergistic actions of multiple hormones, with auxin likely playing the most critical role in the rooting process of tea plants.
-
The application of exogenous growth regulators primarily alters plant developmental processes by influencing nutrient content, related oxidase activity, and the balance of endogenous hormones[21]. During cutting propagation, these effects are mainly manifested through the induction of AR formation, ultimately improving the overall rooting rate. The optimal type, concentration, and duration of growth regulator treatment vary significantly among plant species. This study demonstrated that both the A10 and B14 treatments significantly shortened the AR formation period at different stages, and markedly improved parameters such as rooting rate, average root length, and root activity. Moreover, the combined treatment partially integrated the advantages of both individual applications. Further investigation is warranted to refine the optimal treatment protocols for tea cuttings to maximize rooting efficiency.
Nutrients are critical for the cutting propagation of tea plants. In this study, the control group maintained consistently low levels of soluble sugars and starch, and excessive depletion directly delayed AR initiation[22]. In contrast, the A10 and B14 treatments reversed this rapid depletion. Soluble sugar content peaked during AR formation and subsequently declined in the elongation phase, overall showing an initial increase followed by a decrease. Soluble starch content initially decreased due to breakdown for early energy supply, then rebounded after rooting, as photosynthesis resumed, and nutrient uptake capacity improved. The B14 treatment exhibited higher starch content during AR growth, which may explain its advantage in producing thicker roots[23]. Moreover, the combined treatment maintained higher nutrient levels across nearly all stages, and alleviated the low-content trends observed in individual treatments during key rooting periods. These results also explain why semi-lignified cuttings showed the highest survival and rooting rates in preliminary experiments (Supplementary Fig. S1). Overly mature cuttings exhibit reduced cell division and vascular cambium activity due to excessive lignification, significantly inhibiting adventitious rooting. Although young cuttings possess high metabolic activity, their low soluble sugar content and weak mechanical support make them susceptible to water loss and physical damage, thereby reducing survival rates. In contrast, semi-lignified cuttings not only store higher carbohydrate levels but also possess a well-developed vascular system that facilitates efficient long-distance transport of water, nutrients, and hormones[24]. Soluble proteins participate in enzymatic reactions and signal transduction during AR formation. In this experiment, all treatment groups showed a similar trend of initial increase followed by a decrease in soluble protein content, likely related to the rapid synthesis of stress-responsive proteins after excision, and a continuous supply of photosynthetic products from remaining leaves[22]. During the callus induction phase, the A10 treatment exhibited higher protein content than the control and B14, possibly due to IBA upregulating auxin-responsive genes, and activating protein synthesis pathways[1,25]. As root formation progressed, partial protein degradation led to a gradual decline in content. Protein metabolism stabilized once the root system became fully functional.
This study demonstrates that treatment with growth regulators promotes AR formation in tea cuttings by modulating the dynamic activities of three key enzymes closely associated with rooting: POD, PPO, and IAAO. Compared to the control, the treatment groups exhibited higher POD activity during most rooting stages. Its early increase helped mitigate oxidative stress, the mid-phase activity supported root development and lignification, while later activity declined, likely due to consumption for lignin synthesis, consistent with previous reports in IBA-treated plants[26,27]. PPO activity was also significantly higher in treated cuttings during key stages of AR formation. Its appropriate elevation catalyzes the synthesis of the root-promoting 'IAA-phenolic acid complex' to induce root primordia, while a subsequent decrease prevents excessive accumulation of this substance[28,29]. IAAO activity was significantly reduced during the callus induction phase (particularly in the A10 treatment), facilitating the accumulation of endogenous IAA to promote rooting, a result consistent with findings in Paeonia lactiflora, Olea europaea, and other species[15,30,31].
The dynamic balance of endogenous hormones is crucial for AR formation in tea cuttings. In this study, the control group maintained consistently low IAA levels throughout the rooting process, which may be a key factor limiting its rooting capacity[32]. In contrast, the treatment groups exhibited significantly higher IAA content during the callus and AR formation stages, which decreased in the elongation phase, indicating that exogenous IBA and EBR promote rooting by modulating endogenous IAA levels at critical stages. Meanwhile, CTK inhibits rooting at higher concentrations[13]. The control group maintained high CTK levels during callus formation, whereas the treatment groups significantly reduced CTK content. Specifically, the A10 treatment showed a high IAA/CTK ratio at day 15, characterized by elevated IAA accompanied by a marked decrease in CTK, which favors cell elongation and root primordium formation, and may explain why it produced more roots than B14. At day 21, B14 had significantly lower CTK than the other groups, resulting in a lower IAA/CTK ratio that may contribute to its shortened rooting time. GA play a role in breaking dormancy. The treated cuttings had significantly higher GA3 levels than the control soon after insertion, suggesting that the treatments enhanced endogenous GA3, and helped release dormancy in lignified cuttings. The control group reached its peak GA3 level later, coinciding with the callus formation stage, implying that high GA3 content may promote callus induction[33]. In summary, the growth regulator treatments optimized the hormonal environment for AR formation by coordinating the levels and balance of IAA, CTK, and GA3.
In this experiment, ABA content increased during the initial cutting stage, which can be attributed to mechanical injury at the incision site stimulating ABA production. This promotes starch hydrolysis into sugars, and facilitates the formation of IAA receptors, thereby enhancing the stress resistance of the cuttings[33]. The higher ABA levels in the treatment groups, particularly A10 during the early stage, were consistent with their elevated soluble sugar content. Meanwhile, MDA content—a product of membrane lipid peroxidation—was measured as an indicator of cell membrane damage and oxidative stress levels (Supplementary Fig. S2). Throughout the rooting process, the control group exhibited higher MDA content than most treatment groups, especially compared to A10. This suggests that exogenous plant growth regulators alleviated oxidative damage and reduced oxidative stress in the cuttings, thereby promoting growth. Furthermore, the stronger rooting ability observed in the A10 group may be attributed to the enhanced stress resistance induced by IBA treatment.
Although significant progress has been made in related research globally, several limitations remain. Most studies have focused on the effects of single plant growth regulators on AR formation, while systematic research on the combined application of multiple regulators is relatively scarce, despite the potential superior efficacy of formulated mixtures in practical production. Furthermore, the compatibility between different ecological environments, tea plant varieties, and types of plant growth regulators requires further investigation to meet diverse cultivation needs. Future research should explore the interactions among these substances and optimize application strategies to enhance agricultural efficiency. With improved understanding of their mechanisms of action, more efficient and environmentally friendly plant propagation technologies are expected to be developed. It is also important to note that although plant growth regulators show remarkable advantages in promoting adventitious rooting, their long-term effects and potential environmental impacts still need further evaluation. Future studies should emphasize reducing chemical usage while maintaining cutting success rates, and explore greener and more sustainable methods for plant propagation.
-
In summary, IBA, EBR, and their combined treatment all promoted AR formation in cuttings. IBA exhibited superior efficacy in accelerating callus formation, while EBR shortened the AR emergence time, and resulted in thicker root diameters. The combined treatment demonstrated a synergistic effect to some extent. The earlier callus induction under IBA may be attributed to higher soluble sugar accumulation, and IAAO-mediated modulation of IAA levels at critical stages. EBR established a more favorable IAA/CTK ratio, enhancing rooting competence and reducing the time to AR emergence. Additionally, increased starch content under EBR contributed to thicker roots. All three treatments balanced oxidase activities and hormone levels during key rooting phases. Notably, elevated PPO and POD activities under the treatments accelerated IAA degradation at critical stages, thereby optimizing the rooting process. Higher ABA and lower MDA levels in IBA-treated cuttings indicated stronger stress resistance. Furthermore, the combined treatment showed synergistic improvements in survival rate, root activity, endogenous IAA content, and soluble sugar accumulation, suggesting complementary regulation in hormonal and metabolic pathways. This study confirms that IBA and EBR treatments promote AR development in tea cuttings through coordinated regulation of nutrient utilization, hormonal balance, and enzyme activities.
This work was supported by the National Natural Science Foundation of China (32573087, 32202551), and the Key Project of the Shaanxi Provincial Natural Science Basic Research Program (2025JC-QYCX-025).
-
The authors confirm their contributions to the paper as follows: experiment implementation: Tian S, Liang W; data collection and organization: Tian S, Liang W, Li W, Ma Y; manuscript writing: Tian S; manuscript revision: Ge Z, Wang W; manuscript review: Yu Y, Wang W, Bao L; data analysis: Huang X; material support. Zhou J; hypothesis establishment and experiment design: Bao L. All authors reviewed the results and approved the final version of the manuscript.
-
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
-
The authors declare that they have no known competing finan-cial interests or personal relationships that could have appeared toinfluence the work reported in this paper.
-
# Authors contributed equally: Shenyang Tian, Wenjuan Liang
- Supplementary Table S1 Experimental factor-level design for Camellia sinensis cuttings treated with various auxins.
- Supplementary Table S2 Experimental factor-level design for Camellia sinensis cuttings treated with other plant growth regulators.
- Supplementary Fig. S1 (a) Effects of cutting maturity on the propagation of Camellia sinensis cuttings. (b) Survival rate of cuttings with different maturity levels. (c) Rooting rate of cuttings with differentmaturity levels.
- Supplementary Fig. S2 Effects of IBA, EBR, and their combination on malondialdehyde (MDA) content in Camellia sinensis cuttings.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Tian S, Liang W, Li Y, Ge Z, Ma Y, et al. 2026. Effects of different plant growth regulators on adventitious root formation in Camellia sinensis cuttings. Beverage Plant Research 6: e017 doi: 10.48130/bpr-0026-0002
Effects of different plant growth regulators on adventitious root formation in Camellia sinensis cuttings
- Received: 05 November 2025
- Revised: 22 December 2025
- Accepted: 08 January 2026
- Published online: 06 May 2026
Abstract: This study investigated the mechanisms by which plant growth regulators enhance adventitious root (AR) formation in short cuttings of the tea cultivar 'Zhongcha 108'. Based on preliminary screening using concentration-time gradient treatments of various regulators, indole-3-butyric acid (IBA), and 24-epibrassinolide (EBR) were selected for further observation of their effects on the rooting process. A systematic analysis was conducted to compare morphological and endogenous physiological changes under individual and combined hormone treatments, and to evaluate their correlations with AR development. The results demonstrated that IBA, EBR, and their combination significantly accelerated AR formation, and increased the AR rate. IBA shortened callus formation time, while EBR reduced the time required for AR emergence; IBA also produced a greater number of ARs. The combined treatment resulted in more balanced promotion, increasing the AR number by 140% compared to the control, while achieving root diameter comparable to that of the IBA-treated group. During AR formation, all three treatments enhanced nutrient levels and peroxidase (POD) activity at critical stages, while reducing indole-3-acetic acid oxidase (IAAO) activity to facilitate rooting. Additionally, the treatments provided appropriate indole-3-acetic acid (IAA) levels, and amplified the IAA/cytokinin (CTK) ratio. Both individual regulators promoted AR formation by modulating physiological balance: IBA excelled in accelerating callus formation, whereas EBR shortened the AR emergence period. The combined treatment further exhibited a synergistic effect to some extent.
-
Key words:
- Camellia sinensis /
- Cutting propagation /
- Nutrients /
- Enzyme activity /
- Endogenous hormones














